Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.118275
Revised: February 8, 2026
Accepted: March 16, 2026
Published online: May 15, 2026
Processing time: 132 Days and 17.7 Hours
Clinical obesity, a major type 2 diabetes mellitus risk factor linked to insulin resistance and dyslipidemia, responds poorly to single-target therapies. Danggui
To elucidate the synergistic anti-obesity/insulin-sensitizing effects of the DG-HJ (DH) pair and identify epidermal growth factor receptor (EGFR)/phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT)-mediated mechanisms via network pharmacology and experimental validation.
Network pharmacology, molecular docking, 100 nanosecond molecular dynamics simulation, and in vivo experiments in high-fat diet-fed mice (normal control/mo
Network pharmacology analysis identified 15 core components, 73 targets, and PI3K/AKT as the core pathway, with key components showing high affinity to estrogen receptor 1/EGFR/signal transducer and activator of transcription 3. In vivo, DH reduced body weight and fat mass, improved glucose-lipid profiles, alleviated steatosis and inflammation, enhanced insulin sensitivity, normalized leptin/adiponectin balance, and
DH exerts additive or synergistic anti-obesity effects via the EGFR/PI3K/AKT pathway, ameliorating metabolic disorders and insulin resistance, providing a scientific basis for traditional Chinese medicine-Western medicine integration and type 2 diabetes mellitus prevention.
Core Tip: Clinical obesity, a major type 2 diabetes mellitus risk factor with insulin resistance and inflammation, responds poorly to single-target therapies. This study integrated network pharmacology, molecular docking, 100 nanosecond molecular dynamics simulation, and high-fat diet-fed mouse experiments to clarify the synergistic mechanism of Danggui-Huangjing herb pair. Danggui-Huangjing targets epidermal growth factor receptor/estrogen receptor 1/signal transducer and activator of transcription 3 via the phosphatidylinositol 3-kinase/protein kinase B pathway, reducing body weight/fat, improving glucose-lipid metabolism, and alleviating inflammation – efficacy superior to single herbs – providing a scientific basis for clinical application.
- Citation: Li WJ, Xu JJ, Tiang HS, Liu TH, Wu LL. Network pharmacology: Anti-obesity and insulin-sensitizing effects of Danggui-Huangjing herb-pair via epidermal growth factor receptor/phosphatidylinositol 3-kinase/protein kinase B pathway. World J Diabetes 2026; 17(5): 118275
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/118275.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.118275
Obesity has emerged as a critical public health challenge worldwide. According to the World Obesity Federation’s latest World Obesity Atlas, 2025[1], the number of adults with obesity globally is projected to increase from approximately 524 million in 2010 to 1.13 billion by 2030, representing a > 115% surge.
Recently, a landmark consensus statement published in The Lancet redefined obesity, introducing a novel classification system that distinguishes between clinical obesity (CO) and preclinical obesity and explicitly classifies CO as a chronic disease involving abnormal adipose accumulation and metabolic dysfunction. In addition to significantly increasing the risks of type 2 diabetes mellitus (T2DM), cardiovascular disease, and malignancy[2], CO also predisposes to osteoarthritis and metabolic dysfunction-associated steatotic liver disease, which severely impair patients’ quality of life and shorten life expectancy[3,4]. Therefore, the identification of safe and effective intervention strategies for CO – particularly those targeting multiple pathological defects – would be of substantial clinical and public health significance.
The pathogenesis of CO is centered on a vicious cycle involving insulin resistance (IR), disordered lipid metabolism, and low-grade chronic inflammation. The adipose tissue of individuals with obesity secretes large quantities of pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6), which impair insulin signaling. In addition, hyperinsulinemia and IR result in the upregulation of lipogenic transcription factors such as sterol regulatory element-binding protein 1 (SREBP-1), promoting abnormal lipid accumulation in the liver and adipose tissue[5]. The phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) pathway occupies a central position in insulin signaling, regulating glucose uptake and lipid metabolism, and its dysfunction is a hallmark of obesity-associated metabolic abnormalities. Recent studies have shown that the aberrant activation of key proteins in growth factor and inflammatory signaling pathways, such as epidermal growth factor receptor (EGFR) and signal transducer and activator of transcription 3 (STAT3), exacerbates hepatic steatosis and systemic inflammation, further amplifying the metabolic dysregulation[6,7]. These defects form a self-reinforcing pathological loop of lipid deposition, inflammation, and IR[8,9].
The current management strategies for CO include lifestyle interventions and pharmacotherapies, such as glucagon-like peptide-1 receptor agonists[10]. Critically, drugs with single targets are insufficient to treat the multiple metabolic disturbances associated with CO[11]. Accordingly, the development of novel safe therapeutic strategies with multiple targets is imperative.
Given this unmet clinical need, attention has increasingly turned to traditional Chinese medicine (TCM), which emphasizes “multicomponent-multi-target” regulation. Danggui (Angelica sinensis, DG) and Huangjing (Polygonatum sibiricum, HJ) are both medicinal and edible herbs that can be administered long-term. This combination aligns with the TCM principle of the “simultaneous regulation of qi and blood”: DG promotes blood circulation and nourishes the blood, while HJ replenishes qi and nourishes yin. Clinically, the combination has been used to improve the glycolipid metabolism and reduce the body weight of patients with obesity. Experimental studies have shown that DG polysaccharide significantly ameliorates high-fat diet (HFD)-induced fatty liver and dyslipidemia by activating peroxisome proliferator-activated receptor γ and the adiponectin/sirtuin 1/AMP-activated protein kinase and PI3K/AKT signaling pathways[12-14]; and that HJ extract markedly reduces weight gain and fat accumulation and improves IR in HFD-fed mice[15]. However, whether DG and HJ have additive or synergistic effects, and which bioactive components and molecular targets mediate their anti-obesity effects when used in combination remain unclear. To address this gap in knowledge, we adopted an integrated research strategy combining network pharmacology, molecular docking, molecular dynamics (MD) simulation, and in vivo experimental validation to systematically elucidate the potential anti-obesity mechanisms of DH. In addition, we aimed to determine whether DG and HJ have additive or synergistic effects compared with either herb alone. This work should lay a scientific foundation for the clinical use of DH in the management of CO and its further translational development[16,17].
We searched for the chemical constituents of DH using the keywords “Danggui” and “Huangjing” in the Traditional Chinese Medicine System Pharmacology Database and Analysis Platform (TCMSP). Compounds were screened on the basis of their pharmacokinetic parameters, to identify those with oral bioavailability ≥ 30% and drug likeness (DL) ≥ 0.18[16]. Lists of the active components meeting these criteria and their corresponding target proteins were compiled.
Further searches were then conducted of the High-throughput Experiment and Reference Database for Traditional Chinese Medicine and the China National Knowledge Infrastructure for relevant publications, to add potentially active components that were not included in the TCMSP or did not fulfill the initial screening criteria but have effects that are supported by published reports. Subsequently, the molecular structures and chemical substance registry numbers for all the active components were verified in the PubChem database. Components with unclear structures or for which data were missing were excluded. In this way, we established an active component database for DH.
The canonical SMILES sequences of the identified active components were imported into the SwissTargetPrediction database for target prediction (probability > 0)[18]. All predicted target protein names were submitted to the UniProt database for standardization. They were converted to recognized gene symbols, restricted to the species Homo sapiens, and used to construct a predicted target library for DH.
Using “clinical obesity” as the keywords, we systematically searched the GeneCards database, the OMIM database, the Comparative Toxicogenomics Database, and the Therapeutic Target Database and compiled a list of known disease-associated targets. To enhance the reliability of target selection, a relevance score threshold of > 2 was used with the GeneCards database output[19]. All the disease targets were standardized using UniProt IDs, the lists were merged, and duplicates were removed to establish a disease target repository for CO.
The predicted target library for DH was compared with the disease target library for CO to identify potential targets of the combination that might mediate its effects on CO. This target set was submitted to the STRING database to construct a protein-protein interaction (PPI) network. The species was set to “human”, a minimum interaction confidence threshold of > 0.400 was used, and to focus the analysis on functional clusters of proteins, isolated nodes (proteins with no interactions) were hidden, thereby reducing redundancy and improving the visualization of core modules.
The PPI network generated using STRING was imported into Cytoscape software (version 3.10.3)[20] to facilitate visualization and analysis. We used topological importance analysis to precisely identify key targets within the network. Cytoscape’s ‘CytoNCA’ plugin was used to calculate four critical topological parameters for each node in the network: (1) Their degree centrality; (2) Betweenness centrality; (3) Closeness centrality; and (4) Eigenvector centrality[19]. The targets with scores for all metrics exceeding their respective mean values were designated as core targets for subsequent in-depth analysis.
To systematically elucidate the biological functions of core targets, Gene Ontology (GO) enrichment analysis and Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis were performed using the DAVID database. The analysis was configured using the species “Homo sapiens” and the identifier “OFFICIAL_GENE_SYMBOL”. The GO analysis encompassed three categories: (1) Biological process (BP); (2) Cellular component; and (3) Molecular function (MF). The enrichment results were filtered using a threshold of P < 0.05 to identify the key biological processes and signaling pathways containing the core targets.
To investigate the interactions between the active constituents of DH and potential anti-CO targets, molecular docking was performed using AutoDock. The core targets identified using the network pharmacology analysis were analyzed as receptor molecules and the active constituents of DH were analyzed as ligands. The molecular structures of the primary active components of DH were downloaded from the PubChem database. Subsequently, PyMOL 2.5.7 was used to save these structures in Protein Data Bank (PDB) format. PyMOL software was then used to remove the ligands and water molecules from the target macromolecules, which were saved in PDB format, and we used the Getbox Plugin to obtain the binding pocket parameters. We then imported the processed protein and active ingredient files into AutoDock Tools 1.5.6, converting them to PDBQT format. Subsequently, we performed semi-rigid molecular docking using AutoDock 4.2.6 software. The binding potentials were evaluated using affinity scoring to select the optimal docking model. Finally, we visualized the molecular docking results using PyMOL 2.6.0 software.
The three pairs of compounds with the highest binding energies in the molecular docking analysis were selected for MD simulation using GROMACS 2022. Force field parameters for the receptors were generated using the AMBER14SB force field and the pdb2 gmx tool. For ligands, topology files for the GAFF2 force field were constructed using sobtop 1.0 (dev3.1) software, and partial charges were assigned using the RESP method. The complex systems were solvated in a cubic TIP3P water box with a 1 nm edge length, and Na+/Cl- ions were added to achieve charge neutrality (0.15 M NaCl). Long-range electrostatic interactions were handled using the particle mesh Ewald method, with a cutoff distance of 1 nm.
Prior to the MD simulations, the system was subjected to three-stage energy minimization: (1) Water molecule minimization and restraint of solutes (5000 steps of steepest descent); (2) Minimization of counterions (3000 steps of steepest descent); and (3) Unrestrained minimization (3000 steps of steepest descent plus 2000 steps of conjugate gradient). Simulations were conducted under normal pressure and temperature ensemble conditions (310 K, 1 bar) for 100 nanoseconds, with a 2 femtosecond times step. Temperature coupling was performed using the V-rescale method, and pressure coupling using the Parrinello-Rahman method. The post-simulation analyses included root-mean-square deviation, root-mean-square fluctuation, radius of gyration, solvent-accessible surface area, and hydrogen bond analysis (using built-in tools in GROMACS). The molecular mechanics-Poisson Boltzmann surface area binding free energy was calculated using the g_mmpbsa package, with the solvent model set to Poisson Boltzmann surface area and ionic strength set to 0.15 M. Statistical analysis of the simulation results was performed using GraphPad Prism v.9 (GraphPad software, Inc., La Jolla, CA, United States), and the data were presented as mean ± SD.
The DG and HJ granules used in the study were manufactured by Beijing Kangrentang Pharmaceutical Co., Ltd. (Beijing, China) and have been officially registered with the National Medical Products Administration of China (registration No. 1121000016001 for DG and No. 1122000605002 for HJ). These products comply with the Technical Requirements for Quality Control and Standard Development of Traditional Chinese Medicine Formula Granules (National Medical Products Administration announcement, No. 16 of 2021), which requires all TCM formula granules to undergo multicomponent screening and comparative profiling against standard decoctions to ensure consistency in their chemical composition and therapeutic equivalence with the corresponding raw herbs[21].
The DG granules were compliant with the national drug standard (YBZ-PFKL-2021037), which designates ferulic acid as the official quality control marker, with a minimum content requirement of 0.05%[22]. The HJ formula granules were compliant with the Beijing municipal drug standard (BJ-PFKL-2022101), which involves a multicomponent control strategy to guarantee equivalence between the formula granules and the raw material of HJ[23,24]. Furthermore, according to TCMSP, diosgenin-3-O-β-D-fructofuranosyl-α-L-rhamnopyranoside (DFV) (MOL001792) and Zhonghualiaoine 1 (MOL009766) are bioactive components of HJ[25]. The stated compliance with these regulations and the evidence from the databases support the presence of the key active components in the experimental drugs.
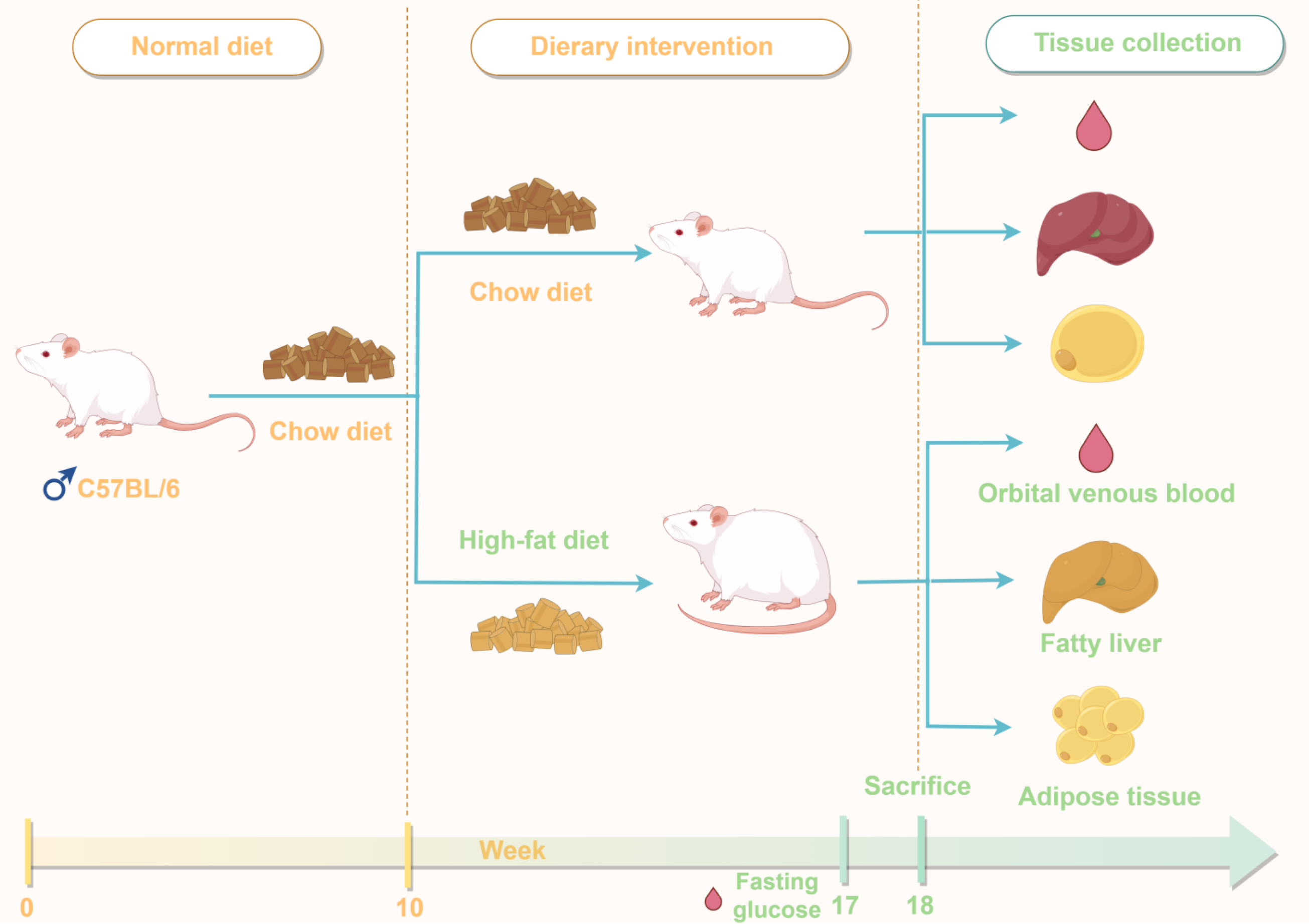
Sixty male C57BL/6 specific pathogen-free mice (8 weeks old, weighing 18 ± 2 g) were supplied by Beijing Sibeifu Biotechnology Co., Ltd. [Beijing, China; license No. SYXK(Jing)2023-0011] and housed in the animal laboratory of the Liangxiang Campus, Beijing University of Chinese Medicine under specific pathogen-free conditions with a 12-hour light/dark cycle, at 24 ± 2 °C, and 50% ± 5% relative humidity. After a 1-week acclimatization period, during which the mice had free access to standard chow and water, they were randomly assigned to five groups (n = 12 per group) using a computerized random-number generator: (1) A normal control (NC) group, fed standard chow (10% kcal from fat, TP23104, Jiangsu Nantong Telofe Feed, Nantong, Jiangsu Province, China) and intragastrically administered purified water; (2) A model control (MC) group, fed a HFD (45% kcal from fat, TP23100, Jiangsu Nantong Telofe Feed, Nantong, Jiangsu Province, China) and intragastrically administered purified water; (3) A DG group, fed the HFD and intragastrically administered DG granules (1.82 g/kg/day, batch No. 24022621, Beijing Kangrentang Pharmaceutical, Beijing, China); (4) An HJ group, fed the HFD and intragastrically administered HJ granules (1.82 g/kg/day, batch No. 24016691, Beijing Kangrentang Pharmaceutical, Beijing, China); and (5) A DH group, fed the HFD and intragastrically administered a 1:1 mixture of DG and HJ granules (total quantity 1.82 g/kg/day). Investigators were single-blinded to group allocation during biochemical and histological analyses.
The administered doses were calculated using a body surface area-based conversion formula for the translation of doses between species: Mouse dose = human clinical dose × mouse body surface area coefficient/human body surface area coefficient (mouse body surface area coefficient: 0.0063 m2/20 g mouse; human body surface area coefficient: 1.73 m2/70 kg adult)[26]. The human clinical daily dose of each herb (DG and HJ) was set to 10 g[27-29], which is the commonly used dose for the treatment of abnormalities in glucose and lipid metabolism. The calculated mouse dose (1.82 g/kg/day) was approximately 12.3 times the human clinical dose.
All of the granules were suspended in purified water (10 mL/kg) and ultrasonicated after warming to 40 °C. The suspensions generated were stored at 4 °C and then administered intragastrically once daily.
The experimental protocol comprised two phases: (1) The induction of obesity during weeks 1-10, during which the MC and treatment groups were fed the HFD; and (2) The treatment phase, during weeks 11-18, during which all the groups continued their respective diets, the treatment groups were administered the herbal suspensions, and the NC and MC groups were administered equal volumes of purified water. Obesity was considered to have been successfully induced when the MC group’s mean body mass exceeded that of the NC group by > 20% (P < 0.05)[30]. The food intakes and body masses of the mice were measured every 3 days, and their body fat percentages were assessed biweekly using an Echo MRI 100H analyzer (Shanghai Niumag Electronic Technology, Shanghai, China). The key timeline of dietary intervention, critical assessments, and sample collection is illustrated in Figure 1.
The study was approved by the Beijing University of Chinese Medicine Experimental Animal Ethics Subcommittee (No. BUCM-2024091301-3253) and conducted in strict accordance with the ARRIVE 2.0 Guidelines for Reporting Animal Research.
At the end of week 18, the mice were fasted for 12 hours and anesthetized with isoflurane, then blood samples were collected by retro-orbital sinus puncture. The blood samples were allowed to coagulate at 24 °C for 3 hours, then centrifuged at 4 °C and 3000 × g for 15 minutes using a 3-30K refrigerated centrifuge (Sigma-Aldrich, St. Louis, MO, United States). The serum samples collected were aliquoted and stored at -80 °C (to preserve cytokine and protein activity) for subsequent biochemical assays and enzyme-linked immunosorbent assay. The liver and epididymal white adipose tissue (eWAT) and brown adipose tissue (BAT) depots were dissected in each mouse within 30 minutes of euthanasia. One portion of each of these was fixed in 4% paraformaldehyde for histopathologic examination, and the other was snap-frozen in liquid nitrogen and stored at -80 °C for subsequent molecular biological assays. The liver and eWAT masses were recorded immediately after the dissection.
Oral glucose tolerance testing was performed at week 17. The mice were fasted for 12 hours, administered glucose (2 g/kg, by oral gavage), and their tail vein blood glucose concentrations were measured 0 minute, 30 minutes, 60 minutes, 90 minutes, and 120 minutes later using a calibrated Contour Next blood glucose meter (Bayer AG, Leverkusen, Germany). The area under the curve (AUC0-120) for each mouse was calculated using the trapezoidal rule.
The serum concentrations of triglyceride (TG), total cholesterol (TC), high-density lipoprotein-cholesterol (HDL-C), and low-density lipoprotein-cholesterol (LDL-C) were measured using a BS-420 automated biochemistry analyzer (Shenzhen Mindray Bio-Medical Electronics, Shenzhen, Guangdong Province, China) and commercial kits (Shenzhen Redu Life Sciences, Shenzhen, Guangdong Province, China; catalog No. S03027, S03042, S03025, and S03029), according to the manufacturer’s instructions. The serum IL-6, TNF-α, IL-1β, leptin, and adiponectin concentrations were measured using enzyme-linked immunosorbent assay kits (Ruixin Biotechnology, Guangdong Province, China; catalog No. RX203049M, RX202412M, RX203063M, RX202420M, and RX20211211M13; specification: 48 tests/kit) and an E9032 microplate reader (Promega Corporation, Madison, WI, United States; model: E9032), with all the assays being performed in duplicate.
Fixed tissue samples were dehydrated, cleared, embedded in paraffin, and sectioned at 5 μm thickness. The sections were stained with hematoxylin and eosin or Oil Red O, then examined using a BX53 optical microscope (Olympus Cor
RNA was extracted from liver samples using an RNA extraction kit (Aikrui Biological Engineering Co., Ltd., Hunan Province, China; catalog No. AG21017) and quantified using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, United States). The purity of the samples was assessed using the 260 nm/280 nm absorbance ratio, and samples with values of 1.8-2.0 were used. Complementary DNA was synthesized using a reverse transcription kit (Aikrui Biological Engineering Co., Ltd., Changsha, Hunan Province, China; catalog No. AG11728). Real-time quantitative polymerase chain reaction was performed using a StepOne Plus real-time PCR system (Thermo Fisher Scientific, Waltham, MA, United States) and a SYBR Green qPCR kit (Aikrui Biological Engineering, Changsha, Hunan Province, China; catalog No. AG11739).
The reaction volume was 10 μL, comprising SYBR Green Mix, complementary DNA template, and specific primers (Table 1). The reaction protocol comprised 30 seconds of denaturation at 95 °C, followed by 40 cycles of 5 seconds at 95 °C and 30 seconds at 60 °C, and melting curve analysis. β-actin was used as the reference gene, and the relative mRNA expression of each target gene was calculated using the 2-ΔΔCt method.
| Primer | Sequence (5’-3’) | Length (bp) |
| actin | Forward primer: TGTACCCAGGCATTGCTGAC | 238 |
| Reverse primer: AACGCAGCTCAGTAACAGTCC | ||
| Egfr | Forward primer: CCTATGCCACGCCAACTGTA | 90 |
| Reverse primer: CACTGCCATTGAACGTACCC | ||
| Pi3k | Forward primer: CGGAGAACCTATTGCGAGGG | 106 |
| Reverse primer: TGCTTGACTTCGCCGTCTAC | ||
| Akt | Forward primer: CCTTTATTGGCTACAAGGAACGG | 170 |
| Reverse primer: CACATGGAAGGTGCGCTCAA | ||
| Fasn | Forward primer: CAAGCAGGCACACACAATGG | 176 |
| Reverse primer: GAAGCGTCTCGGGATCTCTG | ||
| Srebp1 | Forward primer: CGGCTGCATTGAGAGTGAAG | 201 |
| Reverse primer: CTGCTCAGGAAGAAGCGTGTC | ||
| Acc | Forward primer: TCGGCACATGGAGATGTACG | 119 |
| Reverse primer: GATCTACCCGACGCATGGTT | ||
| Stat3 | Forward primer: GCCAAATGCTTGGGCATCAA | 92 |
| Reverse primer: AGGTTCCAATTGGCGGCTTA | ||
| IL6 | Forward primer: CGGCCTTCCCTACTTCACAA | 184 |
| Reverse primer: GCAAGTGCATCATCGTTGTTC | ||
| IL1b | Forward primer: AATGAAAGACGGCACACCCA | 119 |
| Reverse primer: ACTCCACTTTGCTCTTGACTTCT | ||
| Tnfa | Forward primer: ACTCCAGGCGGTGCCTATGT | 160 |
| Reverse primer: GTGAGGGTCTGGGCCATAGAA | ||
| Cd11b | Forward primer: GCACCACACATGCAGAGAGA | 151 |
| Reverse primer: GCTACCCGAGCCATCAATCA | ||
| Nos2 | Forward primer: ACTACTGCTGGTGGTGACAA | 106 |
| Reverse primer: GAAGGTGTGGTTGAGTTCTCTAAG |
Liver lysates were prepared in radioimmunoprecipitation assay lysis buffer containing protease inhibitors (Solarbio, No. BC3710, China). Their protein concentrations were determined using a bicinchoninic acid protein quantification kit (Yamei Biotechnology Co., Ltd., Beijing, China; catalog No. PC201). Lysates each containing 20 μg protein were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis using a MiniPROTEAN Tetra System vertical electrophoresis apparatus (Bio-Rad Laboratories, Inc., Hercules, CA, United States), then the proteins were transferred onto polyvinylidene fluoride membranes. These membranes were blocked in 5% skimmed milk for 1 hour at room temperature, then incubated overnight at 4 °C with specific primary antibodies (1:1000 dilution): (1) Anti-phosphorylated PI3K (p-PI3K, No. 4228); (2) PI3K (No. 4257); (3) Phosphorylated AKT (p-AKT, No. 13038); (4) AKT (No. 4685); (5) Fatty acid synthase (FASN) (No. 3180); (6) Phosphorylated STAT3 (p-STAT3, No. 9145); (7) STAT3 (No. 4904); (8) β-actin (No. 8457T) from Cell Signaling Technology, Inc. (Danvers, MA, United States); (9) Anti-acetyl-CoA carboxylase (ACC) (No. ab45174); (10) EGFR (No. ab72046); and (11) SREBP-1 (No. ab28481) from Abcam plc (Cambridge, United Kingdom). Subsequently, the membranes were incubated with the corresponding horseradish peroxidase-labeled secondary antibody (diluted 1:1000) at room temperature for 1 hour. Specific bands were visualized using enhanced chemiluminescence and captured with a Nikon DS-U3 digital imaging system (exposure time: 45-90 seconds; gain setting: 1.0). Sixteen-bit grayscale images were acquired using Image Lab software (Bio-Rad, v.6.0), ensuring the absence of pixel saturation, and saved in tagged image file format. Grayscale analysis of the target bands was conducted using ImageJ software (NIH, Bethesda, MD, United States), and their intensities were normalized to that of β-actin. The western blot analyses were performed using lysates obtained from three mice per group.
The data were analyzed using SPSS v.26.0 (IBM, Inc., Armonk, NY, United States). The normality and homogeneity of variance of all key continuous outcome variables were verified using the Shapiro-Wilk and Levene’s tests, respectively, with all metrics meeting the two assumptions (P > 0.05 for both tests). All continuous data were therefore presented as mean ± SD. Differences among groups were analyzed by one-way analysis of variance followed by Bonferroni’s post hoc test when the above assumptions were met, or by Kruskal-Wallis test with Dunn’s correction otherwise. The Bonferroni test was used for predefined pairwise comparisons (NC vs MC, MC vs each treatment, and DH vs DG/HJ) due to the confirmed homogeneity of variance. P < 0.05 was considered to indicate statistical significance.
To systematically elucidate the potential mechanisms underlying DH has beneficial effects in patients with CO, we conducted a network pharmacology analysis.
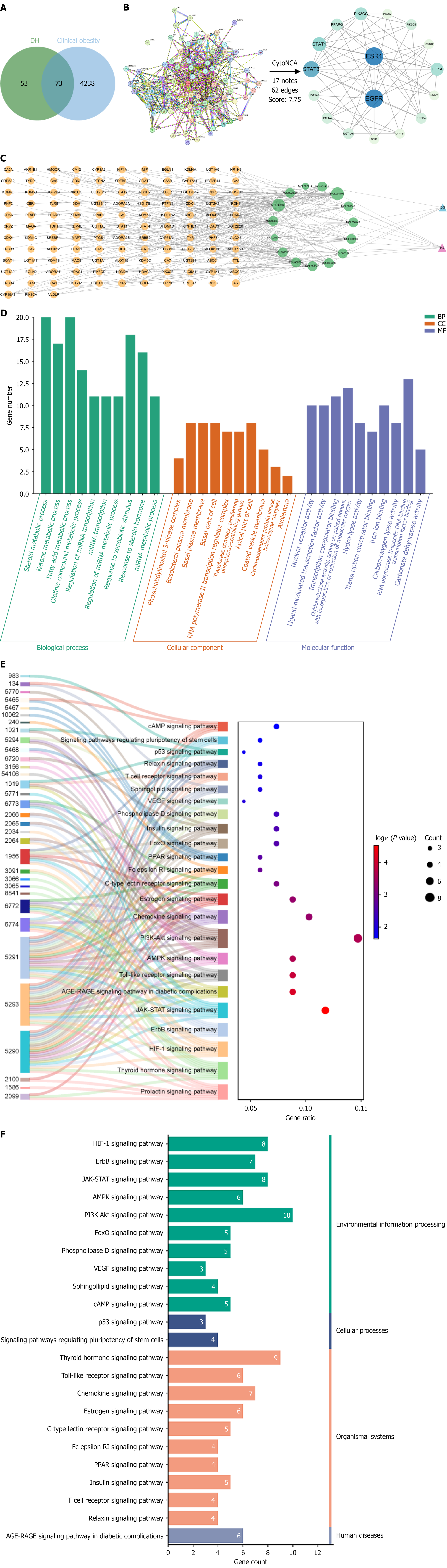
First, through absorption, distribution, metabolism, and excretion parameter screening (oral bioavailability ≥ 30% and drug likeness ≥ 0.18), fifteen active components of DH (five from DG, ten from HJ) (Table 2) and their 127 potential targets were identified. By intersecting these targets with 4238 CO-associated targets, 73 key targets were obtained (Figure 2A). A PPI network was then built (Figure 2B left), and topological analysis using Cytoscape-CytoNCA identified core targets (Figure 2B right). The degree of a node (target) is defined as the number of connections (edges) it has with other nodes, and targets with higher degrees are considered to play more central and biologically critical roles. Through this analysis, we obtained a core PPI subnetwork (Figure 2B right), and the top five targets, ranked in descending value of degree, were: (1) EGFR; (2) ESR1; (3) STAT3; (4) STAT1; and (5) Hypoxia-inducible factor-1α. A “DH-active ingredient-disease target” interaction network was constructed (Figure 2C), with orange nodes for CO targets, green nodes for bioactive compounds [larger node size indicates higher degree; core components include MOL001792 (DFV), MOL009766 (Zhonghualiaoine 1), and MOL000389 (ferulic acid) targeting EGFR/estrogen receptor 1 (ESR1)/STAT3], red nodes for DG/HJ, and grey edges for compound-target interactions.
| Mol ID | Ingredient | Oral bioavailability (%) | Drug likeness | Degree |
| MOL001792 | Diosgenin-3-O-β-D-fructofuranosyl-α-L-rhamnopyranoside | 32.76 | 0.18 | 31 |
| MOL003889 | Methylprotodioscin_qt | 35.11 | 0.85 | 31 |
| MOL009763 | (+)-Syringaresinol-O-beta-D-glucoside | 43.35 | 0.76 | 16 |
| MOL009766 | Zhonghualiaoine 1 | 34.71 | 0.77 | 16 |
| MOL000358 | Beta-sitosterol | 36.91 | 0.75 | 16 |
| MOL000449 | Stigmasterol | 43.82 | 0.75 | 16 |
| MOL002201 | Cis-ligustilide | 51.3 | 0.06 | 16 |
| MOL000359 | Sitosterol | 36.91 | 0.75 | 16 |
| MOL006331 | 4′,5-dihydroxyflavone | 48.55 | 0.18 | 16 |
| MOL000346 | Succinic acid | 29.62 | 0.01 | 16 |
| MOL000389 | FERULIC ACID (CIS) | 54.96 | 0.05 | 16 |
| MOL002959 | 3′-methoxydaidzein | 48.56 | 0.24 | 15 |
| MOL000546 | Diosgenin | 80.87 | 0.80 | 15 |
| MOL009760 | Sibiricoside A_qt | 35.25 | 0.86 | 1 |
| MOL002714 | Baicalein | 33.51 | 0.20 | 1 |
These targets are closely associated with CO pathophysiology: (1) EGFR promotes lipogenesis and inflammation, thereby worsening metabolic disorders[17]; (2) ESR1 regulates lipid homeostasis and insulin sensitivity[31]; (3) STAT3 mediates leptin resistance and chronic inflammation[6]; (4) STAT1 participates in interferon signaling and synergizes with STAT3 to amplify inflammation[32]; and (5) Hypoxia-inducible factor-1α responds to adipose hypoxia, promoting fibrosis and IR[33]. Notably, these core targets are all key nodes of the PI3K/AKT signaling pathway, suggesting that DH may regulate this pathway through multi-target binding to exert anti-obesity effects. These findings suggest that DH may ameliorate CO through effects on these proteins. Furthermore, based on topology analysis of the “DH-active ingredient-disease potential target” interaction network, the top eight active compounds by node degree were[34]: (1) MOL003889 (Methylprotodioscin_qt); (2) MOL001792 (DFV); (3) MOL000389 (ferulic acid); (4) MOL006331 (4′,5-dihydroxyflavone); (5) MOL000359 (sitosterol); (6) (+)-Syringaresinol-O-beta-D-glucoside; (7) MOL000358 (β-sitosterol); and (8) MOL009766 (Zhonghualiaoine 1) (Figure 2C).
GO enrichment analysis revealed 422 significantly enriched entries [271 biological process, 34 cellular component, and 117 MF terms, mainly involving transcriptional regulation (e.g., RNA polymerase II promoter transcription) and MFs such as enzyme and DNA binding (Figure 2D)]. Kyoto Encyclopedia of Genes and Genomes analysis identified 52 enriched pathways (P < 0.05), among which the PI3K/AKT pathway showed the highest enrichment (gene count = 12, P < 0.001; Figure 2E). The “environmental information processing” category contained the most targets (Figure 2F), suggesting that DH modulates cellular responses to extracellular signals.
In summary, network pharmacology analysis identified 15 core bioactive components (with DFV, Zhonghualiaoine 1, and ferulic acid as key) and 73 potential targets of DH, with the PI3K/AKT pathway as the central regulatory mechanism and EGFR/ESR1/STAT3 as key regulatory targets, thus laying a mechanistic foundation for further validation.
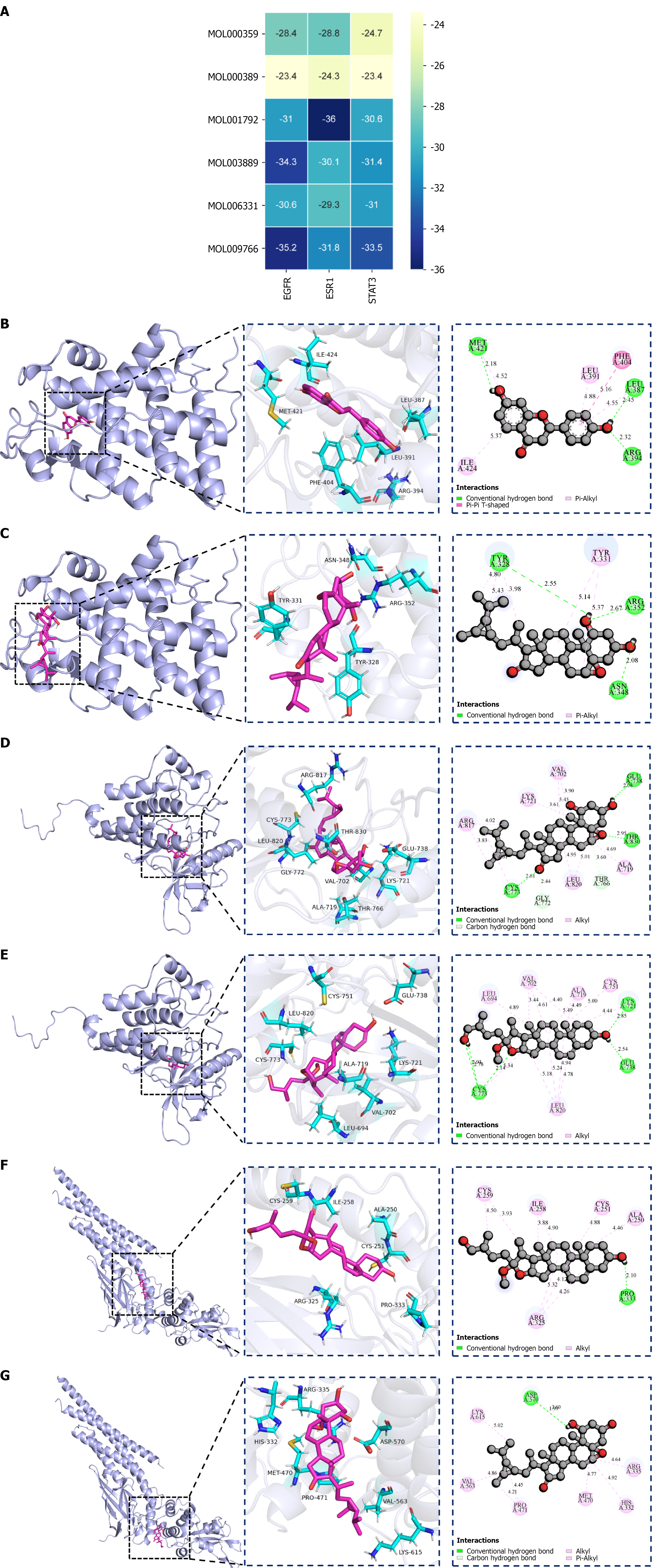
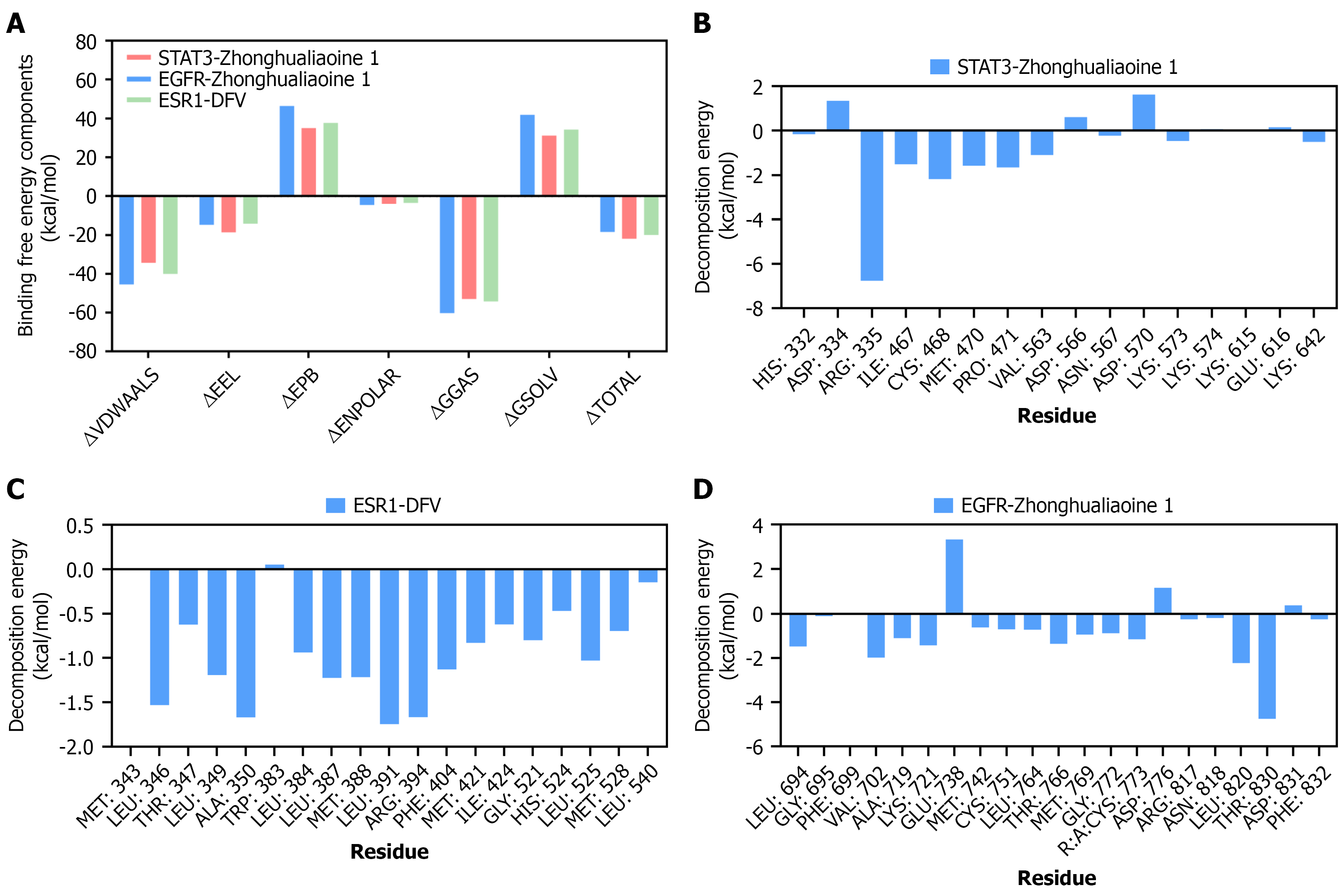
To validate the binding interactions between DH’s active components and core targets, molecular docking analysis was subsequently performed. Previous studies have shown that a minimum binding energy of < -29.3 kJ/mol is indicative of strong binding affinity of a ligand for a target protein and that a minimum binding energy of < -20.9 kJ/mol is indicative of favorable docking of the ligand with the target protein[35]. ESR1 showed optimal binding with HJ-derived DFV (-36.0 kJ/mol), followed by EGFR with HJ-derived Zhonghualiaoine 1 (-35.2 kJ/mol) and Methylprotodioscin_qt (-34.3 kJ/mol). DG-derived ferulic acid exhibited weaker binding but associated with other metabolism/inflammation-related molecules. Both the two-dimensional chemical structures of key ligands and the three-dimensional docking conformations of active ingredient-core target pairs are visualized in Figure 3. Key binding sites included Leu391/Arg394 (ESR1), Thr830/Leu820 (EGFR), and Arg335/Cys468 (STAT3), which may regulate PI3K/AKT signaling.
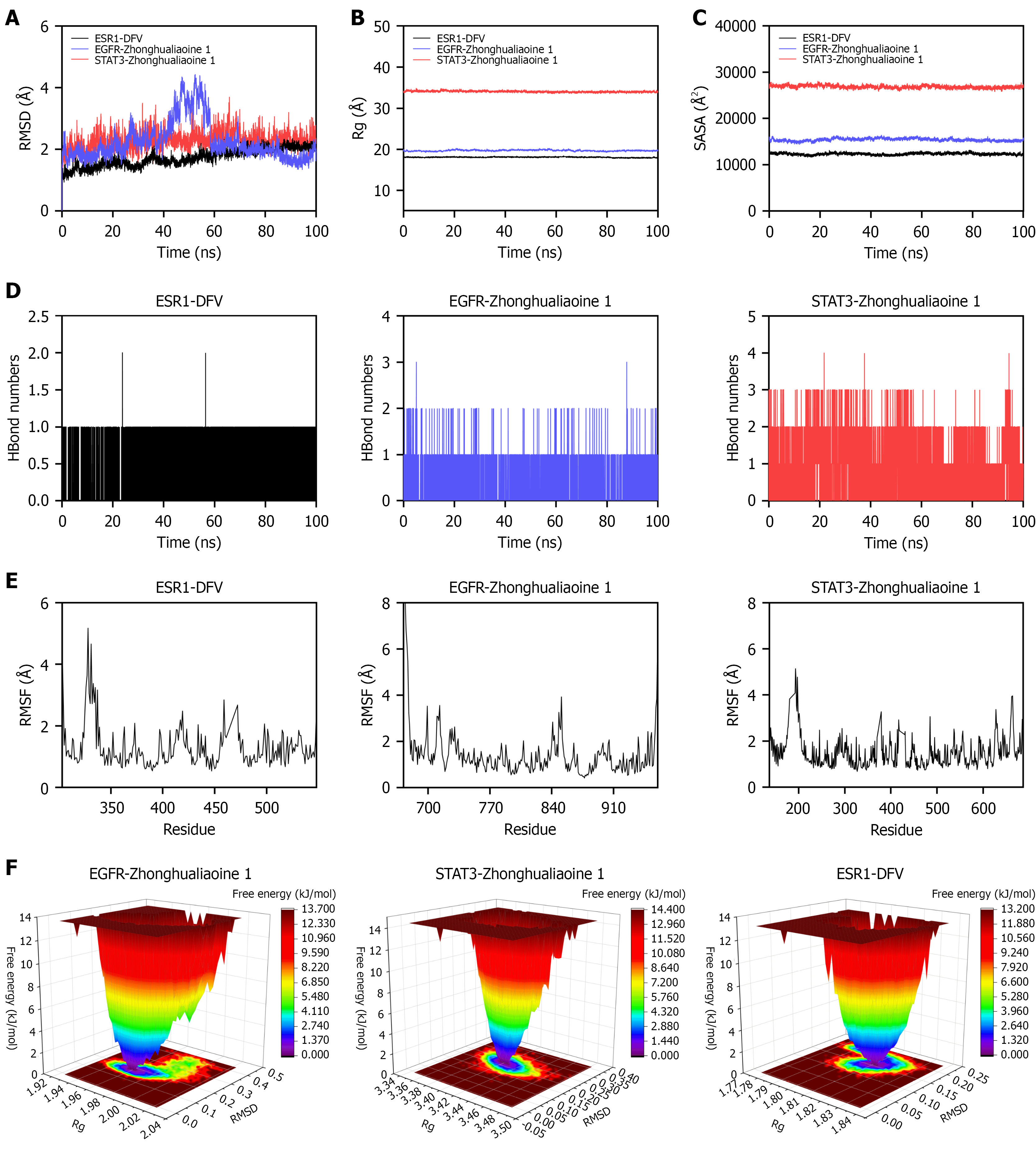
Subsequently, MD simulations were performed to assess the binding stability of DH’s key components to obesity-related core targets (ESR1, EGFR, STAT3) during the simulation period. The root-mean-square deviations for the ESR1/DFV, EGFR/zhonghualiaoine 1, and STAT3/zhonghualiaoine 1 complexes stabilized after 35 nanoseconds (ESR1/DFV), 60 nanoseconds (EGFR/zhonghualiaoine 1), and 15 nanoseconds (STAT3/zhonghualiaoine 1), respectively, and subsequently fluctuated around 2.1 Å, 4.1 Å, and 2.7 Å, indicating conformational stability (Figure 4A). In addition, the radius of gyration (indicative of protein compactness) and solvent-accessible surface area (reflecting protein surface structure) of each complex remained stable throughout the simulation, suggesting no significant expansion, contraction, or surface alteration (Figure 4B and C). Hydrogen bond analysis (Figure 4D) further showed that each complex ma
These results confirm the high binding reliability and stability of DH’s key components to the core targets, providing molecular evidence for the regulation of the PI3K/AKT pathway.
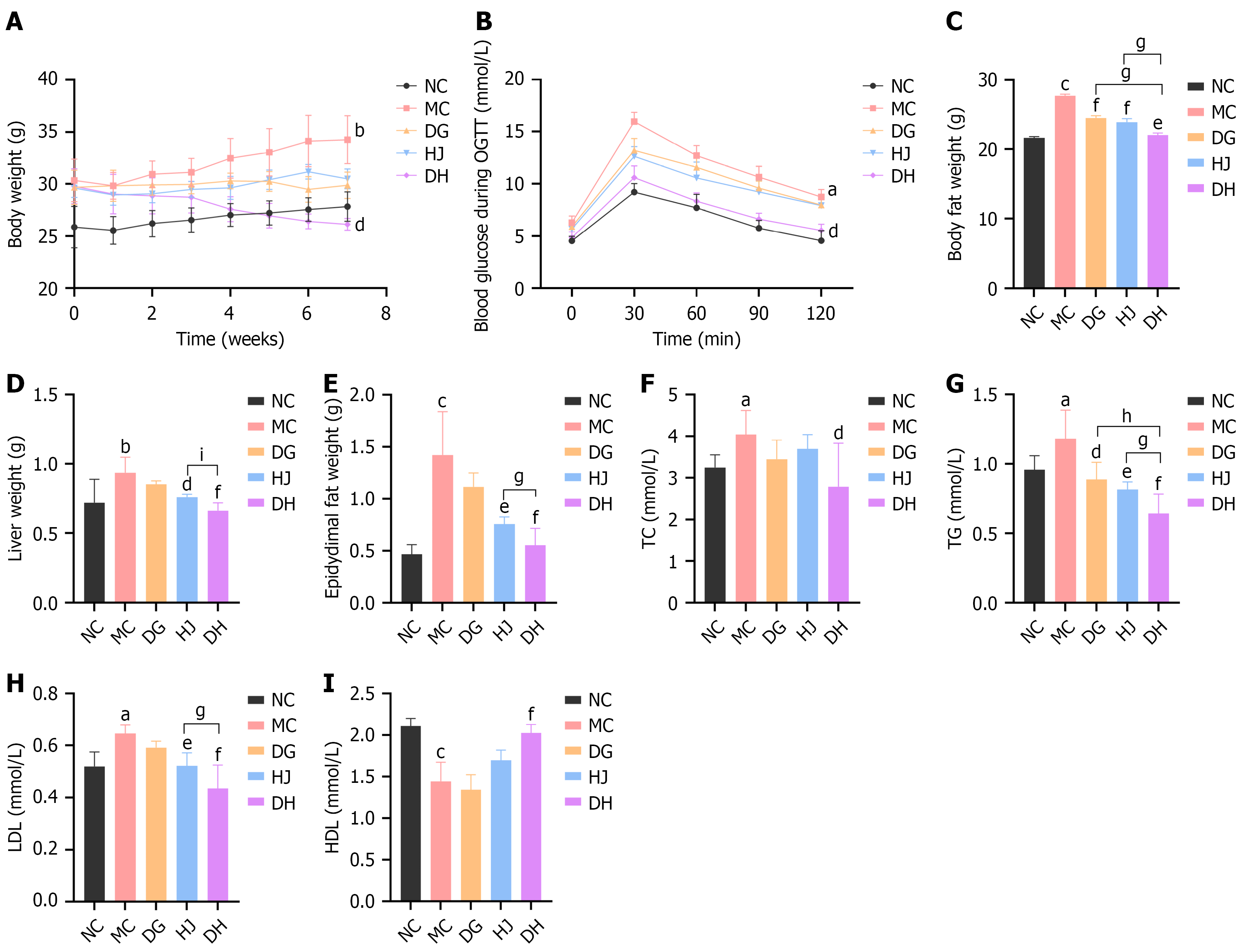
To verify the in silico predictions, the metabolic effects of DH in HFD-fed obese mice were next evaluated (Figure 6). The MC group showed greater body mass gain than NC (P < 0.01); only DH significantly reduced body mass vs MC (P < 0.05). All treatments reduced body fat percentage (DG and HJ: P < 0.001, DH: P < 0.01), with DH’s effect superior to DG/HJ (P < 0.05; Figure 6A and C). DH also significantly reduced liver and epididymal fat masses (both P < 0.001), with more pronounced effects than HJ (P < 0.001 and P < 0.05; Figure 6D and E). The MC group had impaired oral glucose tolerance testing (AUC0-120 increased by 72%, P < 0.01); DH reduced peak blood glucose (from 16.0 ± 1.3 mmol/L to 10.6 ± 0.9 mmol/L) and AUC0-120 (P < 0.05), while DG/HJ had no notable effects (Figure 6B). For dyslipidemia [MC group: (1) High TC/TG/LDL-C; and (2) Low HDL-C; P < 0.05-0.001], DH reduced TC (P < 0.05), TG (P < 0.001), LDL-C (P < 0.001), increased HDL-C (P < 0.01), with superior TG/LDL-C-lowering effects vs HJ (P < 0.05; Figure 6F-I). These findings confirm that DH exerts superior metabolic regulation compared with single herbs, reducing body and fat mass, improving glucose tolerance, and correcting dyslipidemia – consistent with predicted EGFR/PI3K/AKT pathway modulation.
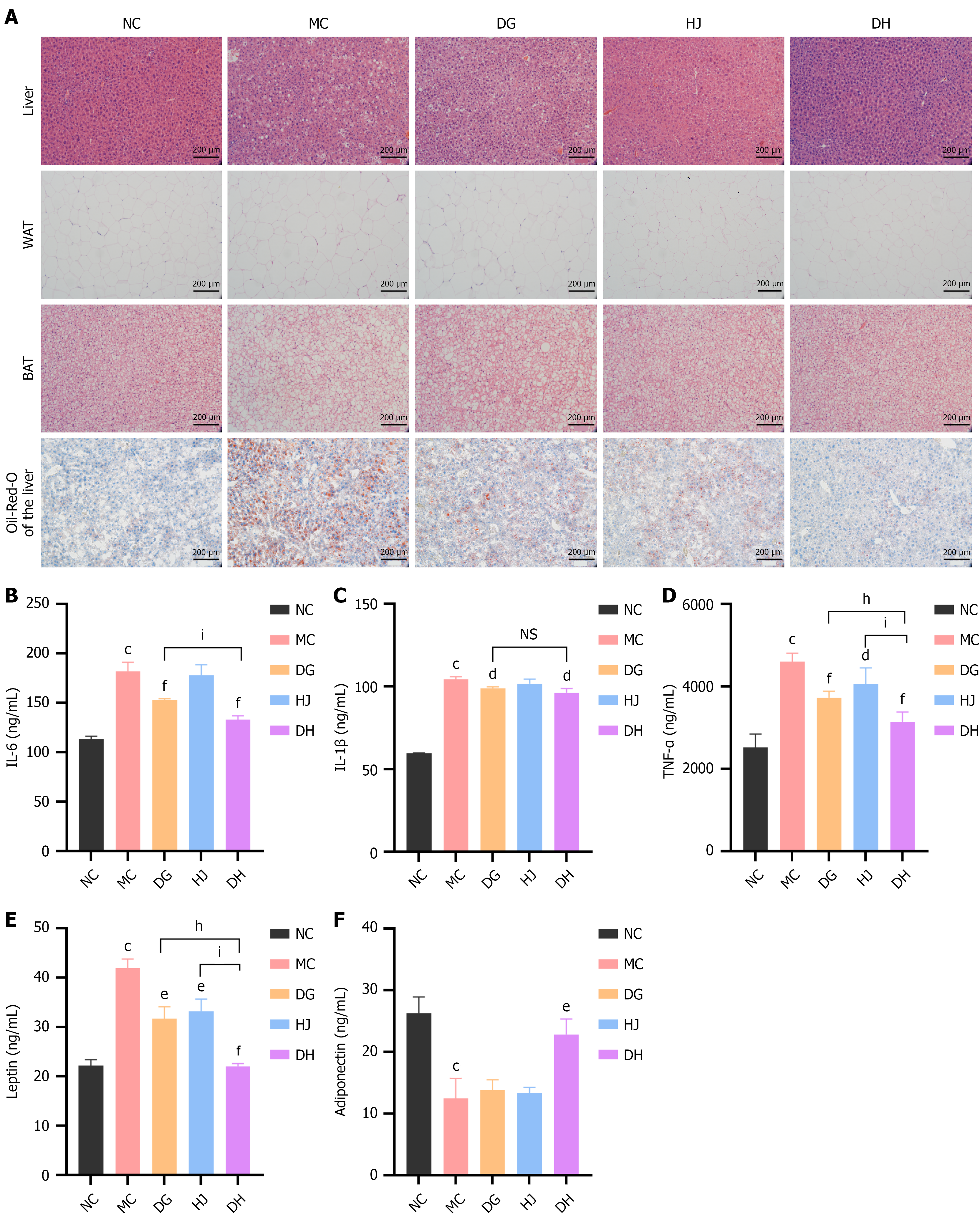
Histological analysis of hematoxylin and eosin/Oil Red O-stained sections showed the MC group had marked hepatic steatosis, eWAT hypertrophy/disorder, and increased BAT lipid deposition vs NC. DG/HJ/DH ameliorated these pathologies, with DH showing the most pronounced effects: (1) Reduced hepatic lipid vacuoles; (2) Regular WAT arrangement; and (3) Less BAT lipid deposition. Oil Red O staining confirmed DH’s superior lipid-lowering effect in the liver (Figure 7A). DH effectively alleviates obesity-induced hepatic steatosis and adipose tissue disorganization via reducing abnormal lipid accumulation, with effects superior to single herbs – consistent with PI3K/AKT-mediated lipogenesis inhibition.
Serum pro-inflammatory cytokines (IL-6, IL-1β, TNF-α) and adipokines (leptin, adiponectin) were measured (Figure 7B-F). The MC group had higher IL-6/IL-1β/TNF-α vs NC (P < 0.001), indicating systemic inflammation. DG/DH reduced IL-6 (both P < 0.01), with DH’s effect superior (P < 0.01 vs DG); DG/DH reduced IL-1β (both P < 0.05). All treatments reduced TNF-α (DG: P < 0.001; HJ: P < 0.05; DH: P < 0.001), with DH outperforming DG (P < 0.01) and HJ (P < 0.001). All treatments reduced leptin (DG/HJ: P < 0.01; DH: P < 0.001), with DH’s effect superior (P < 0.01 vs DG, P < 0.001 vs HJ). Notably, only DH increased adiponectin (P < 0.01). DH has superior anti-inflammatory effects vs single herbs, reducing pro-inflammatory cytokines and restoring leptin/adiponectin balance – related to EGFR/STAT3 inflammatory axis inhibition downstream of PI3K/AKT.
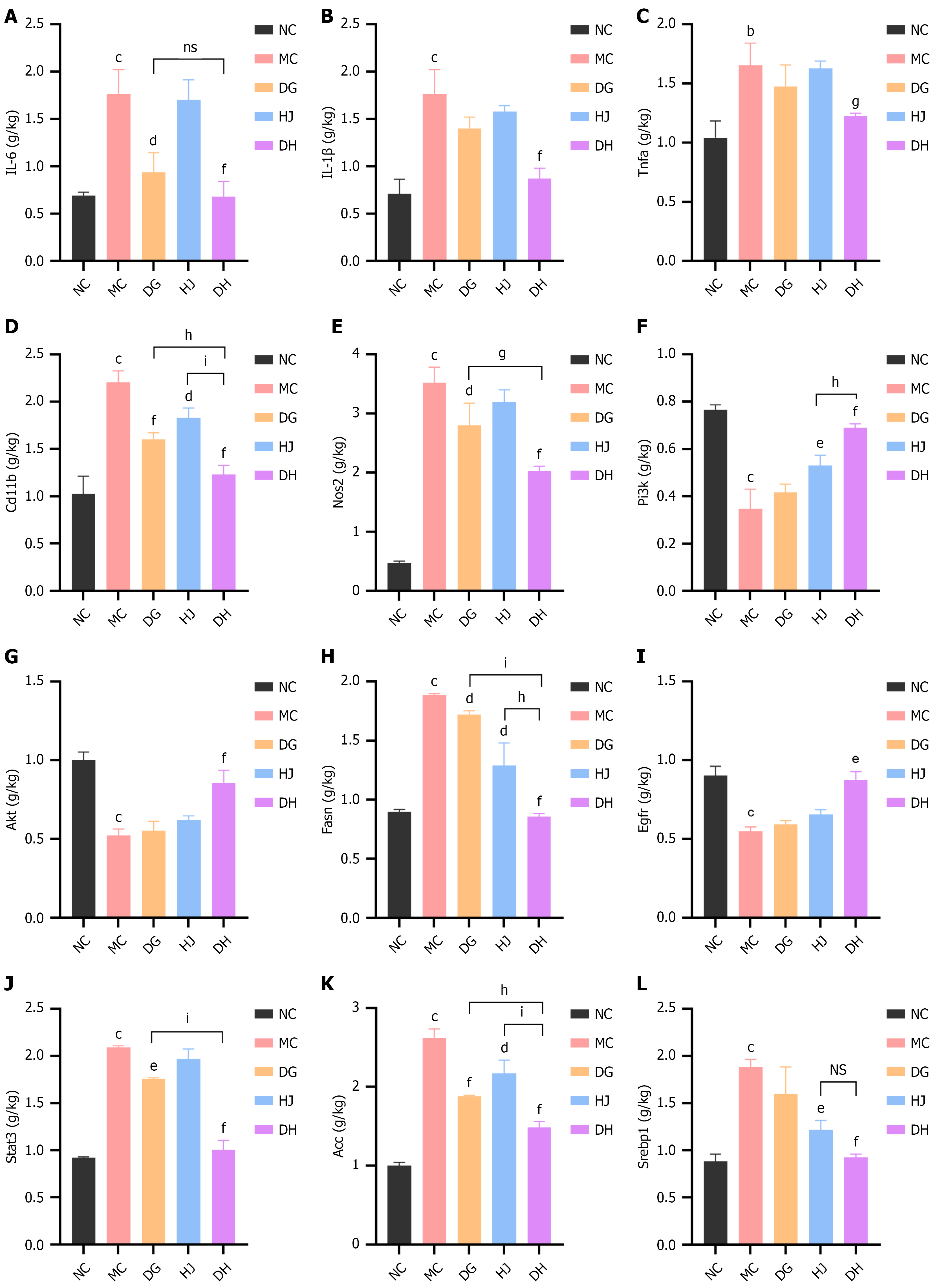
Hepatic expression of key genes was analyzed (Figure 8). The MC group showed aberrant expression of PI3K/AKT pathway genes (low Pi3k/Akt, high Egfr), lipid synthesis genes (Fasn/Stat3/Acc/Srebp-1), and pro-inflammatory genes (IL6/IL1b/Tnfa) (all P < 0.001). DG/HJ/DH reversed these abnormalities, with DH showing the widest and most potent effects: (1) Only DH reduced IL-1b (P < 0.001) and Tnfa (P < 0.05); (2) DH strongly inhibited M1 polarization (Cd11b/Nos2 downregulation, P < 0.001); (3) DH upregulated Pi3k/Akt (P < 0.001) and downregulated Egfr (P < 0.01) (unique vs single herbs); and (4) DH reduced Fasn/Stat3/Acc/Srebp-1 (P < 0.001), with effects superior to DG/HJ (P < 0.01-0.001). DH additively/synergistically regulates the PI3K/AKT/STAT3 pathway and downstream inflammation/lipid synthesis genes, with broader/potenter effects than single herbs – consistent with predicted mechanisms.
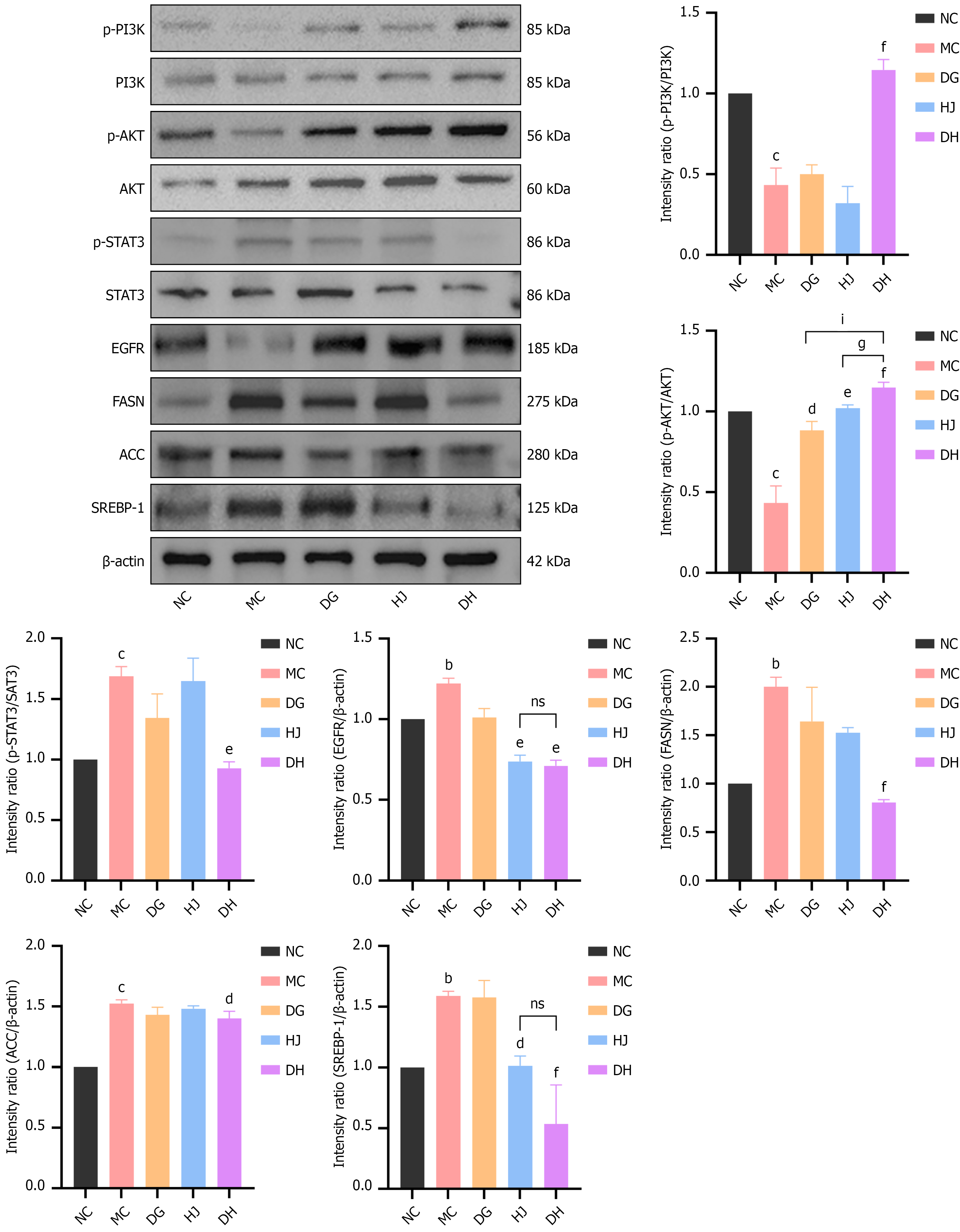
Western blot analysis validated protein expression (Figure 9). The MC group had upregulated lipogenic proteins (Srebp-1/FASN/ACC), EGFR, and p-STAT3 (Tyr705), but downregulated p-PI3K (Tyr458, a phosphorylation site associated with insulin signaling pathway activation) and p-AKT (Ser473, a phosphorylation site specifically reflecting insulin-mediated physiological signals) (P < 0.01/P < 0.001), indicating impaired PI3K/AKT signaling with enhanced lipo
In the present study, we have systematically elucidated the anti-obesity mechanism of the DH combination through an integrated strategy combining network pharmacology, molecular docking, MD simulation, and in vivo validation. The key findings were as follows: (1) The core bioactive components of DH [DFV (MOL001792) and Zhonghualiaoine 1 (MOL009766), both from HJ] act additively or synergistically to target EGFR and STAT3 (predicted by network pharmacology and validated by molecular docking and MD simulation); (2) DH improves metabolic homeostasis, reduces lipid accumulation, and suppresses chronic inflammation through regulation of the PI3K/AKT pathway (validated in vivo); and (3) DH exhibits greater efficacy than DG or HJ alone, supporting the potential for enhanced beneficial effects of the combined herb pair. These findings provide a biological basis for the TCM principle of “simultaneous regulation of qi (vital energy) and blood”, as applied to obesity treatment, and they are consistent with those of previous studies, which showed that HJ extract attenuates weight gain, reduces adipose tissue lipid accumulation, reduces serum TG concentrations, and ameliorates the IR of HFD-fed mice[23,24]. In addition, polysaccharides from DG have been reported to reduce body weight, blood glucose levels, and lipid levels and suppress inflammatory cytokine expression in obesity and diabetes models[13,36]. Since obesity is a major risk factor for T2DM, the metabolic improvements and insulin-sensitizing effects induced by DH may have direct implications for diabetes prevention and early metabolic control[37,38]. Notably, the core pathological features of prediabetic individuals include IR and impaired glucose tolerance, often accompanied by metabolic abnormalities such as obesity and dyslipidemia, for which safe and effective long-term interventions are currently lacking[39]. By regulating the EGFR/PI3K/AKT pathway to improve insulin sensitivity, while correcting glucose-lipid metabolism disorders and inhibiting chronic inflammation, DH precisely addresses the pathological needs of prediabetes. As a natural medicinal and edible combination, it exhibits a favorable safety profile and can be administered long-term, holding promise as a novel intervention strategy for prediabetic individuals to delay disease progression and reduce T2DM onset risk – particularly suitable for those intolerant to chemical drugs or preferring natural therapies.
Importantly, we have shown that the DH combination had superior effects to those of either DG or HJ alone, suggesting additive or synergistic pharmacodynamic interactions between the two herbs. This is consistent with the TCM principle of “monarch-minister-assistant-guide”, wherein auxiliary herbs enhance the efficacy, target specificity, and safety of the principal herb. Beyond reducing adiposity, DH significantly improved glucose tolerance and insulin sensitivity, indicating that it effectively alleviates IR – an essential pathological link between obesity and T2DM. By modulating the EGFR/PI3K/AKT pathway, DH restored downstream insulin signaling and normalized systemic glucose-lipid metabolism. These findings highlight DH as a dual-function intervention with both anti-obesity and anti-diabetic potential.
The additive or synergistic anti-obesity effects of DH probably derive from the complementary targeting of three major pathological processes involved in obesity – metabolic dysregulation, lipid accumulation, and chronic inflammation – by DG and HJ. The principal active compounds of HJ [DFV (MOL001792) and Zhonghualiaoine 1 (MOL009766)] were found to interact with EGFR and STAT3, key regulators of hepatic lipid metabolism and inflammatory signaling. As a receptor upstream of the PI3K/AKT pathway, EGFR promotes the cleavage and nuclear translocation of SREBP-1 via the AKT-mechanistic target of rapamycin complex 1 (mTORC1) axis, thereby enhancing the transcription of genes encoding lipogenic enzymes such as FASN and ACC[40,41]. In obesity, despite impaired insulin signaling, EGFR-induced PI3K/AKT/mTORC1 activity remains partially functional and contributes to hepatic lipid synthesis and steatosis – a core pathological feature of obesity that also predisposes to T2DM[42,43].
AKT activation requires phosphorylation at two distinct regulatory residues, and there is substantial evidence for the identities of the associated upstream signaling pathways and functional outputs. Thr308 phosphorylation is primarily mediated by phosphoinositide-dependent kinase 1, which is recruited to the plasma membrane by PI3K-generated PIP3, downstream of growth factor receptors such as EGFR. This phosphorylation event is linked to pathological processes including aberrant lipogenesis[44,45], because activated AKT activates mTORC1 signaling, which increases the transcriptional activity of SREBP1c and lipogenic gene expression[46]. In contrast, Ser473 is phosphorylated by mTORC2 in response to metabolic signals, including insulin and insulin-like growth factor 1, and plays a pivotal role in mediating the metabolic effects of insulin on hepatic glucose utilization and lipid homeostasis[47]. These two phosphorylation events mediate context-specific signaling: EGFR-induced Thr308 phosphorylation drives obesity-related metabolic dysfunction, while mTORC2-dependent Ser473 phosphorylation maintains metabolic homeostasis[48-50].
Notably, the anti-p-AKT antibody used in the study specifically targets Ser473, and we have shown that DH significantly upregulates p-AKT (Ser473), while downregulating EGFR. This selective modulation may explain why DH inhibits EGFR-induced pathological lipogenesis (via suppression of the EGFR-PI3K-phosphoinositide-dependent kinase 1-Thr308-AKT-mTORC1-SREBP1 axis) while restoring insulin-dependent PI3K-mTORC2-Ser473-AKT activation[51]. Consistent with this, DH improved the glucose tolerance and normalized the leptin/adiponectin ratio of the mice, consistent with a restoration of insulin sensitivity, indirectly confirming the reinstatement of normal AKT pathway signaling. Taking the literature and the present data together, it seems that the mechanism of the effects of DH involves high-specificity binding of HJ-derived DFV to ESR1 (Leu391/Arg394) and Zhonghualiaoine 1 to EGFR (Thr830/Leu820), as demonstrated by the MD simulation, and this selectively inhibits pathological AKT phosphorylation, while restoring insulin-induced PI3K/AKT activation. DH also inhibits EGFR-induced lipogenesis and alleviates the inflammation-induced defect in insulin receptor/insulin receptor substrate signaling. Thus, it has the dual effects of the inhibition of lipogenesis and the promotion of insulin sensitivity, consistent with the “multicomponent-multitarget” effects of TCMs. DG independently modulates inflammatory pathways by reducing the expression of IL-6, IL-1β, and Cd11b, which complements the signaling effects of HJ. Thus, the superior efficacy of DH can be explained by its multiple effects on signaling, inflammation, and abnormal lipid metabolism.
In addition, DH significantly reduced serum concentrations of TNF-α, IL-6, and IL-1β and corrected the high leptin/adiponectin ratio. This rebalancing of adipokine secretion and attenuation of macrophage infiltration may collectively alleviate chronic inflammation and IR – two mutually reinforcing mechanisms central to obesity and T2DM pathogenesis[52]. Given that IL-6 and leptin drive STAT3 activation and SOCS3 upregulation[53-55], the inhibition of the IL-6/Leptin-STAT3 axis by DH may underpin both its anti-inflammatory and insulin-sensitizing effects.
Zhonghualiaoine 1, a bioactive component of HJ, may contribute to the restoration of PI3K/AKT signaling and suppression of inflammation via modulation of EGFR and STAT3. DFV exhibited high binding affinity and stable interaction with ESR1 in MD simulations. ESR1 is a well-characterized regulator of lipid homeostasis and insulin sensitivity[56-58]; thus, the DFV-ESR1 interaction may further enhance insulin responsiveness through crosstalk with the EGFR/PI3K/AKT pathway.
ESR1, a core target identified by network pharmacology, has sex-specific metabolic functions. We used only male C57BL/6 mice due to their more stable and reproducible HFD-induced obesity/steatosis/IR phenotypes (female phe
The present study has some limitations: (1) The use of a single-dose HFD-fed mouse model; (2) Reliance on network pharmacology predictions; (3) Lack of high-performance liquid chromatography/liquid chromatography-mass spectrometry phytochemical characterization and quantitative analysis of the DH granules (while compliance with national/municipal drug standards and indirect evidence confirm the presence of core bioactive components); and (4) Lack of data on other metabolic tissues and the gut microbiota. Future studies should include dose-response verification, bioactive component isolation, high-performance liquid chromatography/liquid chromatography-mass spectrometry fingerprinting and quantitative analysis, pharmacokinetic and toxicity evaluation, and early-phase clinical trials to es
Collectively, DH alleviates IR and restores glucose-lipid metabolic balance through EGFR/PI3K/AKT pathway mo
Future work will address the following: (1) Isolate core components to verify their individual and combined effects, clarifying additive or synergistic molecular mechanisms, and fully validate the “component-target-efficacy” axis; (2) Conduct pharmacokinetic and acute/chronic toxicity studies to support clinical translation; (3) Evaluate DH’s efficacy in human primary hepatocytes and adipocytes to confirm species conservation; and (4) Design phase I clinical trials to verify safety and optimal dose window, and explore its combination with glucagon-like peptide-1 agonists.
DH exerts multi-target additive or synergistic anti-obesity effects by regulating metabolic homeostasis, insulin sensitivity, and chronic inflammation through the coordinated modulation of the EGFR/ESR1/STAT3 axis and PI3K/AKT signaling. This integrated mechanism underlies its superior efficacy over single-herb treatments and highlights its potential as a novel therapeutic candidate for obesity, IR, and related metabolic disorders such as metabolic dysfunction-associated steatotic liver disease and T2DM.
The authors sincerely acknowledge the invaluable support and guidance provided by their mentors and fellow team members.
| 1. | Federation WO. World obesity atlas 2025. Available from: https://data.worldobesity.org/publications/world-obesity-atlas-2025. |
| 2. | World Health Organization. Obesity and overweight 2025. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. |
| 3. | Quek J, Chan KE, Wong ZY, Tan C, Tan B, Lim WH, Tan DJH, Tang ASP, Tay P, Xiao J, Yong JN, Zeng RW, Chew NWS, Nah B, Kulkarni A, Siddiqui MS, Dan YY, Wong VW, Sanyal AJ, Noureddin M, Muthiah M, Ng CH. Global prevalence of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the overweight and obese population: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8:20-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 496] [Article Influence: 165.3] [Reference Citation Analysis (1)] |
| 4. | Zheng H, Chen C. Body mass index and risk of knee osteoarthritis: systematic review and meta-analysis of prospective studies. BMJ Open. 2015;5:e007568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 252] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 5. | NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403:1027-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1583] [Cited by in RCA: 1359] [Article Influence: 679.5] [Reference Citation Analysis (15)] |
| 6. | Hu W, Lv J, Han M, Yang Z, Li T, Jiang S, Yang Y. STAT3: The art of multi-tasking of metabolic and immune functions in obesity. Prog Lipid Res. 2018;70:17-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 7. | Bhushan B, Michalopoulos GK. Role of epidermal growth factor receptor in liver injury and lipid metabolism: Emerging new roles for an old receptor. Chem Biol Interact. 2020;324:109090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 8. | Welsh A, Hammad M, Piña IL, Kulinski J. Obesity and cardiovascular health. Eur J Prev Cardiol. 2024;31:1026-1035. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 127] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 9. | Ahmed SK, Mohammed RA. Obesity: Prevalence, causes, consequences, management, preventive strategies and future research directions. Metabol Open. 2025;27:100375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 105] [Article Influence: 105.0] [Reference Citation Analysis (0)] |
| 10. | Ismaiel A, Scarlata GGM, Boitos I, Leucuta DC, Popa SL, Al Srouji N, Abenavoli L, Dumitrascu DL. Gastrointestinal adverse events associated with GLP-1 RA in non-diabetic patients with overweight or obesity: a systematic review and network meta-analysis. Int J Obes (Lond). 2025;49:1946-1957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 43] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 11. | Roomy MA, Hussain K, Behbehani HM, Abu-Farha J, Al-Harris R, Ambi AM, Abdalla MA, Al-Mulla F, Abu-Farha M, Abubaker J. Therapeutic advances in obesity management: an overview of the therapeutic interventions. Front Endocrinol (Lausanne). 2024;15:1364503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 31] [Reference Citation Analysis (0)] |
| 12. | Wang K, Cao P, Wang H, Tang Z, Wang N, Wang J, Zhang Y. Chronic administration of Angelica sinensis polysaccharide effectively improves fatty liver and glucose homeostasis in high-fat diet-fed mice. Sci Rep. 2016;6:26229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 13. | Ren C, Luo Y, Li X, Ma L, Wang C, Zhi X, Zhao X, Li Y. Pharmacological action of Angelica sinensis polysaccharides: a review. Front Pharmacol. 2024;15:1510976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 14. | Wang K, Cao P, Shui W, Yang Q, Tang Z, Zhang Y. Angelica sinensis polysaccharide regulates glucose and lipid metabolism disorder in prediabetic and streptozotocin-induced diabetic mice through the elevation of glycogen levels and reduction of inflammatory factors. Food Funct. 2015;6:902-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 15. | Hariri N, Thibault L. High-fat diet-induced obesity in animal models. Nutr Res Rev. 2010;23:270-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 552] [Cited by in RCA: 730] [Article Influence: 45.6] [Reference Citation Analysis (0)] |
| 16. | Qin Z, Chen Y, Liu N, Wang Y, Su L, Liang B, Huang C. Mechanisms of Bushenyiqi decoction in the treatment of asthma: an investigation based on network pharmacology with experimental validation. Front Pharmacol. 2024;15:1361379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 17. | Cao S, Pan Y, Tang J, Terker AS, Arroyo Ornelas JP, Jin GN, Wang Y, Niu A, Fan X, Wang S, Harris RC, Zhang MZ. EGFR-mediated activation of adipose tissue macrophages promotes obesity and insulin resistance. Nat Commun. 2022;13:4684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 18. | Zhang T, Wei W, Chang S, Liu N, Li H. Integrated Network Pharmacology and Comprehensive Bioinformatics Identifying the Mechanisms and Molecular Targets of Yizhiqingxin Formula for Treatment of Comorbidity With Alzheimer's Disease and Depression. Front Pharmacol. 2022;13:853375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 19. | Liu T, Wang J, Tong Y, Wu L, Xie Y, He P, Lin S, Hu X. Integrating network pharmacology and animal experimental validation to investigate the action mechanism of oleanolic acid in obesity. J Transl Med. 2024;22:86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (2)] |
| 20. | Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13:2498-2504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43766] [Cited by in RCA: 36496] [Article Influence: 1586.8] [Reference Citation Analysis (4)] |
| 21. | National Medical Products Administration. Technical requirements for quality control and standard development of traditional Chinese medicine formula granules: Circular No. 16 of 2021. Available from: http://www.nmpa.gov.cn/. |
| 22. | Commission CP. Pharmacopoeia of the People’s Republic of China, 2020 version. Beijing: China Medical Science Press, 2020. |
| 23. | Liu D, Tang W, Han C, Nie S. Advances in Polygonatum sibiricum polysaccharides: Extraction, purification, structure, biosynthesis, and bioactivity. Front Nutr. 2022;9:1074671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (0)] |
| 24. | Ou X, Chen J, Li B, Yang Y, Liu X, Xu Z, Xiang X, Wang Q. Multiomics reveals the ameliorating effect and underlying mechanism of aqueous extracts of polygonatum sibiricum rhizome on obesity and liver fat accumulation in high-fat diet-fed mice. Phytomedicine. 2024;132:155843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 25. | Ru J, Li P, Wang J, Zhou W, Li B, Huang C, Li P, Guo Z, Tao W, Yang Y, Xu X, Li Y, Wang Y, Yang L. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform. 2014;6:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4043] [Cited by in RCA: 3505] [Article Influence: 292.1] [Reference Citation Analysis (4)] |
| 26. | Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008;22:659-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3894] [Cited by in RCA: 5038] [Article Influence: 265.2] [Reference Citation Analysis (4)] |
| 27. | Chen Z, Cheng L, Zhang J, Cui X. Exploring the mechanism by which Angelica sinensis improves haematopoietic function in aplastic anaemia. Aging (Albany NY). 2024;16:11535-11552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | Zhou K, Zhang J, Xu L, Lim CED. Chinese herbal medicine for subfertile women with polycystic ovarian syndrome. Cochrane Database Syst Rev. 2021;6:CD007535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 29. | Ren Y, Sun Y, Liao YY, Wang S, Liu Q, Duan CY, Sun L, Li XY, Yuan JL. Mechanisms of action and applications of Polygonatum sibiricum polysaccharide at the intestinal mucosa barrier: a review. Front Pharmacol. 2024;15:1421607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 30. | Xiong Y, Wang X, Gong M, Ji Q, Li Y, Hu A, Lu M, Xu B. Acupoints catgut embedding recovers leptin resistance via improving autophagy progress mediated by AMPK-mTOR signaling in obese mice. Heliyon. 2024;10:e29094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 31. | Ahmed F, Kamble PG, Hetty S, Fanni G, Vranic M, Sarsenbayeva A, Kristófi R, Almby K, Svensson MK, Pereira MJ, Eriksson JW. Role of Estrogen and Its Receptors in Adipose Tissue Glucose Metabolism in Pre- and Postmenopausal Women. J Clin Endocrinol Metab. 2022;107:e1879-e1889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 32. | Cox AR, Chernis N, Bader DA, Saha PK, Masschelin PM, Felix JB, Sharp R, Lian Z, Putluri V, Rajapakshe K, Kim KH, Villareal DT, Armamento-Villareal R, Wu H, Coarfa C, Putluri N, Hartig SM. STAT1 Dissociates Adipose Tissue Inflammation From Insulin Sensitivity in Obesity. Diabetes. 2020;69:2630-2641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Ban JJ, Ruthenborg RJ, Cho KW, Kim JW. Regulation of obesity and insulin resistance by hypoxia-inducible factors. Hypoxia (Auckl). 2014;2:171-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 34. | Li X, Tang Q, Meng F, Du P, Chen W. INPUT: An intelligent network pharmacology platform unique for traditional Chinese medicine. Comput Struct Biotechnol J. 2022;20:1345-1351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 35. | Li X, Wu Y, Wang H, Li Z, Ding X, Dou C, Hu L, Du G, Wei G. Deciphering the Molecular Mechanism of Escin against Neuropathic Pain: A Network Pharmacology Study. Evid Based Complement Alternat Med. 2023;2023:3734861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 36. | Li MM, Zhang Y, Wu J, Wang KP. Polysaccharide from Angelica Sinensis Suppresses Inflammation and Reverses Anemia in Complete Freund's Adjuvant-induced Rats. Curr Med Sci. 2020;40:265-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 37. | Chandrasekaran P, Weiskirchen R. The Role of Obesity in Type 2 Diabetes Mellitus-An Overview. Int J Mol Sci. 2024;25:1882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 351] [Cited by in RCA: 264] [Article Influence: 132.0] [Reference Citation Analysis (0)] |
| 38. | Grams J, Garvey WT. Weight Loss and the Prevention and Treatment of Type 2 Diabetes Using Lifestyle Therapy, Pharmacotherapy, and Bariatric Surgery: Mechanisms of Action. Curr Obes Rep. 2015;4:287-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 39. | Bkaily G, Jazzar A, Abou-Aichi A, Jacques D. Pathophysiology of Prediabetes Hyperinsulinemia and Insulin Resistance in the Cardiovascular System. Biomedicines. 2025;13:1842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 40. | Zhao Q, Lin X, Wang G. Targeting SREBP-1-Mediated Lipogenesis as Potential Strategies for Cancer. Front Oncol. 2022;12:952371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 88] [Reference Citation Analysis (0)] |
| 41. | Cheng C, Geng F, Cheng X, Guo D. Lipid metabolism reprogramming and its potential targets in cancer. Cancer Commun (Lond). 2018;38:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 251] [Cited by in RCA: 630] [Article Influence: 78.8] [Reference Citation Analysis (0)] |
| 42. | Choung S, Kim JM, Joung KH, Lee ES, Kim HJ, Ku BJ. Epidermal growth factor receptor inhibition attenuates non-alcoholic fatty liver disease in diet-induced obese mice. PLoS One. 2019;14:e0210828. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 43. | Bhave S, Ho HK. Exploring the Gamut of Receptor Tyrosine Kinases for Their Promise in the Management of Non-Alcoholic Fatty Liver Disease. Biomedicines. 2021;9:1776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 44. | Toker A, Dibble CC. PI 3-Kinase Signaling: AKTing up inside the Cell. Mol Cell. 2018;71:875-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 45. | Vidal S, Bouzaher YH, El Motiam A, Seoane R, Rivas C. Overview of the regulation of the class IA PI3K/AKT pathway by SUMO. Semin Cell Dev Biol. 2022;132:51-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 46. | Yecies JL, Zhang HH, Menon S, Liu S, Yecies D, Lipovsky AI, Gorgun C, Kwiatkowski DJ, Hotamisligil GS, Lee CH, Manning BD. Akt stimulates hepatic SREBP1c and lipogenesis through parallel mTORC1-dependent and independent pathways. Cell Metab. 2011;14:21-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 574] [Cited by in RCA: 534] [Article Influence: 35.6] [Reference Citation Analysis (4)] |
| 47. | Kim T, Nason S, Antipenko J, Finan B, Shalev A, DiMarchi R, Habegger KM. Hepatic mTORC2 Signaling Facilitates Acute Glucagon Receptor Enhancement of Insulin-Stimulated Glucose Homeostasis in Mice. Diabetes. 2022;71:2123-2135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | McKenna M, Balasuriya N, Zhong S, Li SS, O'Donoghue P. Phospho-Form Specific Substrates of Protein Kinase B (AKT1). Front Bioeng Biotechnol. 2020;8:619252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 49. | Yung HW, Charnock-Jones DS, Burton GJ. Regulation of AKT phosphorylation at Ser473 and Thr308 by endoplasmic reticulum stress modulates substrate specificity in a severity dependent manner. PLoS One. 2011;6:e17894. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 134] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 50. | Balasuriya N, Kunkel MT, Liu X, Biggar KK, Li SS, Newton AC, O'Donoghue P. Genetic code expansion and live cell imaging reveal that Thr-308 phosphorylation is irreplaceable and sufficient for Akt1 activity. J Biol Chem. 2018;293:10744-10756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 51. | Guo D, Prins RM, Dang J, Kuga D, Iwanami A, Soto H, Lin KY, Huang TT, Akhavan D, Hock MB, Zhu S, Kofman AA, Bensinger SJ, Yong WH, Vinters HV, Horvath S, Watson AD, Kuhn JG, Robins HI, Mehta MP, Wen PY, DeAngelis LM, Prados MD, Mellinghoff IK, Cloughesy TF, Mischel PS. EGFR signaling through an Akt-SREBP-1-dependent, rapamycin-resistant pathway sensitizes glioblastomas to antilipogenic therapy. Sci Signal. 2009;2:ra82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 285] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 52. | Song MY, Kim SH, Ryoo GH, Kim MK, Cha HN, Park SY, Hwang HP, Yu HC, Bae EJ, Park BH. Adipose sirtuin 6 drives macrophage polarization toward M2 through IL-4 production and maintains systemic insulin sensitivity in mice and humans. Exp Mol Med. 2019;51:1-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 53. | Wunderlich CM, Hövelmeyer N, Wunderlich FT. Mechanisms of chronic JAK-STAT3-SOCS3 signaling in obesity. JAKSTAT. 2013;2:e23878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 54. | Liu J, Wang T, Yang F, Pubu C. Advancements in the understanding of mechanisms of the IL-6 family in relation to metabolic-associated fatty liver disease. Front Endocrinol (Lausanne). 2025;16:1642436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 55. | Yamaguchi K, Nishimura T, Ishiba H, Seko Y, Okajima A, Fujii H, Tochiki N, Umemura A, Moriguchi M, Sumida Y, Mitsuyoshi H, Yasui K, Minami M, Okanoue T, Itoh Y. Blockade of interleukin 6 signalling ameliorates systemic insulin resistance through upregulation of glucose uptake in skeletal muscle and improves hepatic steatosis in high-fat diet fed mice. Liver Int. 2015;35:550-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 56. | Fabre A, Tramunt B, Montagner A, Mouly C, Riant E, Calmy ML, Adlanmerini M, Fontaine C, Burcelin R, Lenfant F, Arnal JF, Gourdy P. Membrane estrogen receptor-α contributes to female protection against high-fat diet-induced metabolic disorders. Front Endocrinol (Lausanne). 2023;14:1215947. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 57. | Hevener AL, Zhou Z, Moore TM, Drew BG, Ribas V. The impact of ERα action on muscle metabolism and insulin sensitivity - Strong enough for a man, made for a woman. Mol Metab. 2018;15:20-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Bowers LW, Cavazos DA, Maximo IX, Brenner AJ, Hursting SD, deGraffenried LA. Obesity enhances nongenomic estrogen receptor crosstalk with the PI3K/Akt and MAPK pathways to promote in vitro measures of breast cancer progression. Breast Cancer Res. 2013;15:R59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 59. | Zhu BT, Liao QQ, Tian HY, Yu DJ, Xie T, Sun XL, Zhou XM, Han YX, Zhao YJ, El-Kassas M, Liu XX, Sun XD, Zhang YY. Estrogen: the forgotten player in metaflammation. Front Pharmacol. 2024;15:1478819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 60. | Cao LH, Qiao JY, Huang HY, Fang XY, Zhang R, Miao MS, Li XM. PI3K-AKT Signaling Activation and Icariin: The Potential Effects on the Perimenopausal Depression-Like Rat Model. Molecules. 2019;24:3700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 61. | Ribas V, Drew BG, Le JA, Soleymani T, Daraei P, Sitz D, Mohammad L, Henstridge DC, Febbraio MA, Hewitt SC, Korach KS, Bensinger SJ, Hevener AL. Myeloid-specific estrogen receptor alpha deficiency impairs metabolic homeostasis and accelerates atherosclerotic lesion development. Proc Natl Acad Sci U S A. 2011;108:16457-16462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 150] [Article Influence: 10.0] [Reference Citation Analysis (0)] |