Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.117635
Revised: January 8, 2026
Accepted: February 24, 2026
Published online: May 15, 2026
Processing time: 150 Days and 20 Hours
Zuo Gui pill (ZGP) is a traditional Chinese herbal formula originating from Jing Yue Quan Shu, a medical text from China’s Ming Dynasty. It is commonly admi
To explore the effects and underlying mechanisms of ZGP in alleviating oxidative damage and inflammatory responses in T2DOP.
Network pharmacology was applied to screen the active components of ZGP, followed by pathway enrichment analysis to identify potential therapeutic targets. A T2DOP rat model was established to evaluate the therapeutic effect of ZGP in T2DOP and observe the levels of oxidative damage, inflammatory response, pro
A total of 76 active compounds were identified in ZGP. Pathway enrichment analysis showed that the treatment of T2DOP is linked to the Nrf2 signaling pathway. Animal studies showed that compared with the model group the ZGP group and ZGP + Fer-1 group showed no significant decrease in blood glucose but significant reductions in total cholesterol, triglycerides, and low-density lipoprotein cholesterol (P < 0.001). Bone microstructure parameters showed sig
ZGP activated the Nrf2/HO-1/XCT/GPX4 signaling pathway to inhibit bone marrow macrophage M1 polarization, effectively preventing and treating T2DOP.
Core Tip: Regulation of M1 polarization of bone marrow macrophages in type 2 diabetic osteoporosis is dependent on the activation of the nuclear factor E2-related factor 2/hemoxygenase-1/XCT/glutathione peroxidase 4 signaling pathway. Zuo Gui pill, metformin, and Fer-1 can work synergistically to restore nuclear factor E2-related factor 2, upregulate hemoxygenase-1, and inhibit M1 polarization of bone marrow macro
- Citation: Zhao Y, Feng XZ, Cao WQ, Chen YR, Yang Y, Liu JC, Xu A, Ren YL, Wang ZM. Zuo Gui pill alleviates M1 macrophage polarization in type 2 diabetic osteoporosis through the Nrf2/HO-1/XCT/GPX4 pathway. World J Diabetes 2026; 17(5): 117635
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/117635.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.117635
Type 2 diabetic osteoporosis (T2DOP) is a common complication of type 2 diabetes mellitus (T2DM). It is characterized by reduced bone mineral density (BMD) and impaired bone microstructure, which increase the risk of fractures and impose a serious economic burden on patients[1]. Currently, the main treatment for T2DOP relies on the combined use of antidiabetic drugs and anti-osteoporosis medications. However, commonly used antidiabetic drugs in clinical practice, such as thiazolidinediones, sulfonylureas, and sodium-glucose co-transporter-2 inhibitors, can actually decrease BMD and increase the risk of fractures[2,3]. Bisphosphonates, widely used anti-resorptive drugs, may cause gastrointestinal discomfort with long-term use and in some cases may lead to atypical femoral fractures and osteonecrosis of the jaw. These drugs all have limitations and potential side effects. Therefore, seeking safer and more effective treatment options is an urgent priority. Traditional Chinese medicine (TCM) is a distinctive therapy that targets multiple pathways and promotes overall balance in the human body. TCM can also alleviate the destruction of bone structure. It mi
Zhang Jingyue, a physician of the Ming Dynasty in China, created the classic formula ZGP, which consists of Radix Rehmanniae Praeparata, Rhizoma Dioscoreae Oppositae, Fructus Lycii Fructus Corni, Radix Cyathulae, Semen Cuscutae, Colla Cornus Cervi, Colla Carapacis et Plastri (Supplementary Table 1). It primarily functions by nourishing the kidney yin essence with the supplementary function of warming the yang. As a commonly used TCM formula, ZGP is frequently used in improving blood microcirculation, treating osteoporosis, and delaying ovarian aging[4]. Recent studies have shown that ZGP works well in preventing and treating T2DOP. However, the exact mechanisms of its effects are still unclear[5].
Chronic inflammation can cause T2DOP. Oxidative stress is the initiating signal of most inflammatory responses. The nuclear factor E2-related factor 2 (Nrf2)/hemoxygenase-1 (HO-1) signaling pathway is crucial for regulating oxidative stress and inflammatory response. Nrf2 controls cellular antioxidant stress. It serves as the main regulator. HO-1 is an enzyme, breaking down heme. It is a downstream effector of Nrf2. Nrf2 activation stimulates HO-1 expression. It promotes the breakdown of heme. This reduces the synthesis of reactive oxygen species (ROS) and increases superoxide dismutase (SOD). It inhibits the activation of proinflammatory pathways[6]. Problems in this pathway in T2DOP cause osteoclasts to become active and stop osteoblasts activity. This leads to bone loss. The Nrf2 signaling pathway is found in many mammalian cells. It helps control inflammation and ferroptosis[7]. Ferroptosis and inflammation induced by oxidative stress can lead to the occurrence of bone loss[8]. This pathway is a crucial link between inflammation, oxidative stress, ferroptosis, and bone metabolism. The exact role of this in the treatment effects of ZGP is incompletely understood.
This research explored how the active components of ZGP regulate the M1 polarization of bone marrow macrophages through the Nrf2/HO-1/XCT/glutathione peroxidase 4 (GPX4) pathway, thereby helping to alleviate T2DOP.
Active components of ZGP were identified using TCMSP and SwissADME, and their corresponding predicted targets were obtained from the Swiss Target Prediction database. Targets associated with T2DOP were screened from databases such as GeneCards and TTD. Next, the intersection of targets between T2DOP and ZGP was analyzed using Venny 2.1. The intersecting targets were investigated in the STRING database. The protein-protein interaction information was shown in Cytoscape 3.10.2. This predicted possible core targets. Enrichment analyses of Gene Ontology, Kyoto En
The experimental animals were SPF-grade male Sprague Dawley rats, weighing 200 ± 20 g (6 weeks old). They were purchased from the Liaoning Academy of TCM. The rats were housed in a comfortable environment with a temperature range of 20-26 °C as well as humidity ranges from 40% to 70%, under a 12-h light and dark period. The experimental animal research procedures were conducted in accordance with the principles of animal care and usage guidelines. This study protocol was approved by the Ethics Committee of the Liaoning Academy of Traditional Chinese Medicine (No. LZYY240404).
Animals were randomly assigned to one of the seven groups. After 1 week of acclimatization, the remaining rats, except for the 10 rats in the control group, were fed a high-fat, high-sugar diet for 6 weeks. Streptozotocin (STZ) was injected intraperitoneally at a dose of 30 mg/kg body weight. Subsequently, blood glucose levels were measured on three consecutive occasions, and rats with glucose levels ≥ 16.7 mmol/L were considered diabetic. The high-fat, high-sugar diet was continued for 8 weeks, and a reduction in bone mass as observed through micro-CT was considered as successful establishment of the model. To minimize bias blinding was implemented throughout the experiment. Furthermore, all data collection, outcome assessments and statistical analyses were performed by researchers who were blinded to the experimental groups. The control group was fed a standard diet and injected with an equal volume of citrate buffer.
To study the preventive and therapeutic effects of ZGP in mediating ferroptosis inhibition, the T2DOP rats were divided into 6 groups with 10 rats in each group: T2DOP group; ZGP group; metformin (MET) group; Fer-1 group; ZGP + Fer-1 group; and MET + Fer-1 group. The ZGP group and ZGP + Fer-1 group were administered by gavage at a dose of 18.9 g/kg of ZGP in awake rats. This dose is twice the clinically equivalent concentration. The dose of ZGP used in this study was determined with reference to the Chinese Pharmacopoeia. The optimal ZGP dose was verified in preliminary animal experiments by our research team. The MET group and MET + Fer-1 group were administered by gavage at a dose of 0.18 g/kg (clinically equivalent concentration). The control group and T2DOP group were gavaged with an equal volume of saline. The gavage was administered for a total of 8 weeks. During the last 2 weeks, the Fer-1 group, ZGP + Fer-1 group, and MET + Fer-1 group were intraperitoneally injected with 2.5 μmol/kg of Fer-1. The remaining groups of rats were intraperitoneally injected with an equal volume of physiological saline. The rats were anesthetized using a 3% pentobarbital sodium solution, and serum and bone samples were collected for subsequent experiments.
Weekly monitoring of blood glucose levels via the rat tail vein and body weight changes were conducted. Serum levels of total cholesterol (TC) (A111-1-1, Jiancheng, Nanjing, China), triglycerides (TG) (A110-1-1, Jiancheng, Nanjing, China), low-density lipoprotein cholesterol (LDL-C) (A113-1-1, Jiancheng, Nanjing, China), insulin (YJ302840, Meilian, Shanghai, China), glycated hemoglobin (HbA1c) (A056-1-1, Jiancheng, Nanjing, China), malondialdehyde (MDA) (BC0025, Solarbio, Beijing, China), SOD (BC5165, Solarbio, Beijing, China), and glutathione/glutathione disulfide (GSH/GSSH) (A061-1-2, Jiancheng, Nanjing, China) were measured according to the manufacturer’s instructions.
Micro-CT (Quantum GX, PerkinElmer) was used to scan the distal femur of rats, and the system’s built-in software was employed to reconstruct and analyze the three-dimensional images. CT scanning parameters were: (1) Voltage 50 kV; (2) Current 160 μA; (3) Field of view 25 mm; and (4) Voxel size 50 μm. The outcomes were analyzed following the guidelines of the American Society for Bone and Mineral Research Bone Histomorphometry Nomenclature Committee.
Histopathological examination of the distal femur was performed to further evaluate bone tissue deterioration. Femoral tissues were fixed in 4% paraformaldehyde, decalcified with EDTA, dehydrated, paraffin embedded, sectioned, and stained with hematoxylin and eosin. Histological changes were examined under a light microscope.
Blood samples from rats were collected and processed in compliance with the instructions provided by the ELISA Kit. The OD values and standard curve were detected using an iMark microplate reader (Bio-Rad, United States). Finally, the concentrations of bone resorption marker TRACP5b (YJ003177, Meilian, Shanghai, China), bone formation marker propeptide of type I procollagen (PINP) (YJ038224, Meilian, Shanghai, China), and inflammatory factors interleukin (IL)-6 (YJ064292, Meilian, Shanghai, China) and tumor necrosis factor-alpha (TNF-α) (YJ002859, Meilian, Shanghai, China) were obtained.
Femur and tibia bones from both hind limbs of rats were collected, and the bone marrow cavities were flushed with PBS to collect bone marrow mononuclear cells. The cells were centrifuged at 1000 rpm for 5 min, and the supernatant was discarded. The cell pellet was resuspended in PBS and filtered through a 70 μm mesh filter. Then 1 mL of red blood lysis buffer was added. After a 5-min incubation on ice, bone marrow monocytes were collected. Finally, the cells were resuspended in 3% BSA and stained with APC-labeled CD11b (No. APC-65229, Proteintech, China), FITC-labeled CD86 (No. 200305, Biolegend, United States), and PE-labeled anti-CD80 (No. 200205, Biolegend, United States). The expression levels of CD11b, CD86 and CD11b double-positive, and CD11b and CD80 double-positive cells were observed by staining in the dark for 30 min 4 °C. Subsequently, all samples were sequentially analyzed using a NovoCyte flow cytometer. The outcomes obtained were analyzed using FlowJo software.
Total RNA was extracted from rat femurs using liquid nitrogen grinding and RNA-easy Separation Reagent (R701-01, Novozan, Nanjing, China). Complementary DNA was synthesized using a reverse transcription kit (RR047A, Takara, Kyoto, Japan). The primer sequences are listed in Table 1. In a 25 μL reaction system, amplification was performed using TB Green Premix Ex Taq II (RR820A, Takara, Kyoto, Japan) on a CFX96 Real-Time PCR Detection System. The expression trends were calculated using the 2-△△CT method (Table 1).
| Primer name | Primer sequence |
| β-actin | Forward: 5’-TGCTATGTTGCCCTAGACTTCG-3’ |
| Reverse:5’-GTTGGCATAGAGGTCTTTACGG-3’ | |
| GPX4 | Forward: 5’-ATTCCCGAGCCTTTCAACCC-3’ |
| Reverse: 5’-TATCGGGCATGCAGATCGAC-3’ | |
| XCT | Forward: 5’-TATGCTGAATTGGGTACGAGC-3’ |
| Reverse: 5’-TATTACCAGCAGTTCCACCCA-3’ | |
| Nrf2 | Forward: 5’-GACATCCTTTGGAGGCAAGACAT-3’ |
| Reverse: 5’-TGGGAATGTGGGCAACCTG-3’ | |
| HO-1 | Forward: 5’-TAAGACCGCCTTCCTGCTCAA-3’ |
| Reverse: 5’-ACGGTCGCCAACAGGAAACT-3’ |
Protein was extracted by grinding rat femurs in liquid nitrogen and homogenizing in Radioimmunoprecipitation Assay (RIPA) lysis buffer with protease inhibitors. Protein concentration was determined using the Bicinchoninic Acid assay (BCA) assay kit. Protein samples were separated by Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis (SDS-PAGE) and then transferred onto a Polyvinylidene fluoride (PVDF) membrane. Blocking was performed with 5% skim milk powder for 2 h. The primary antibodies GPX4 (No. 67763-1-Ig, Proteintech, Wuhan, China), HO-1 (No. 10701-1-AP, Proteintech, Wuhan, China), XCT (No. ANT-111, Thermo, United States), Nrf2 (No. PA5-27882, Thermo, United States), and receptor activator of nuclear factor-kappa B (Rankl) (No. ab239607, Abcam, United Kingdom), RUNX2 (20700-1-AP, Proteintech, Wuhan, China) were incubated overnight on a rocking platform at 4 °C. After membrane washing the secondary antibody was incubated for 1 h at room temperature on a rocking platform. The membrane was then washed with Tris-Buffered Saline with Tween-20 (TBST) (5 min × 6 times) followed by detection.
The tissue sections were incubated with 3% hydrogen peroxide to block endogenous peroxidase activity. Following antigen retrieval, the sections were incubated overnight at 4 °C with the primary antibody. After washing the sections were incubated with the appropriate secondary antibody at 37 °C for 1 h. Immunoreactivity was visualized using chromogenic substrate development, and the sections were subsequently mounted. Staining intensity was examined under a light microscope.
All quantitative outcomes were presented as mean ± SD. Data analysis was performed using GraphPad Prism 9 software with one-way analysis of variance used to evaluate outcomes. P < 0.05 indicates a statistically significant difference, and a P < 0.01 indicates a more pronounced significant difference.
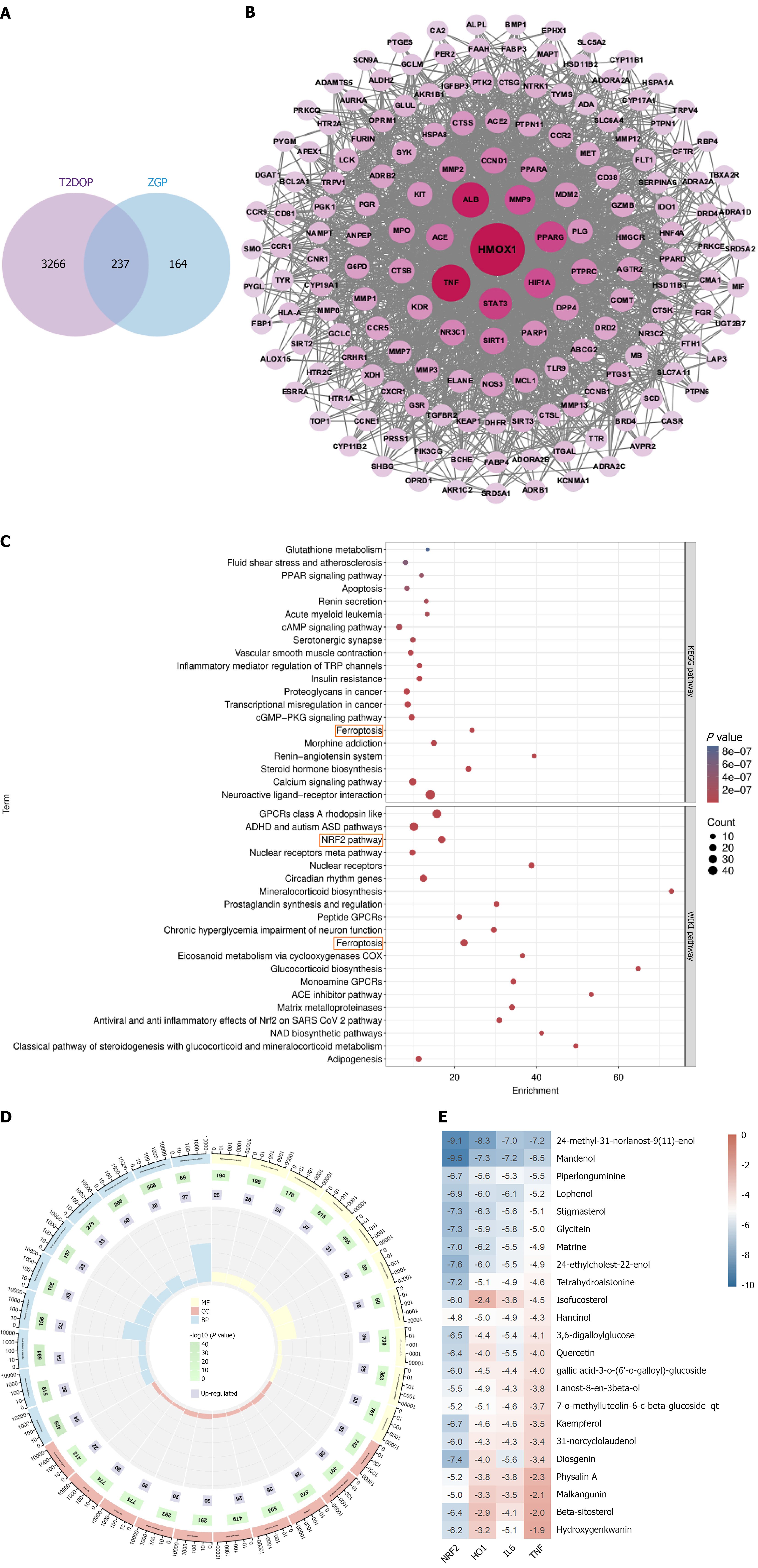
A total of 76 active components were identified from ZGP. It regulates 401 distinct biological targets. Further, 3503 T2DOP-related targets were identified from databases such as GeneCards. Therefore, we identified approximately 237 potential targets of ZGP for treating T2DOP (Figure 1A). We constructed a protein-protein interaction network as shown in the Figure 1B. After removing isolated nodes the main targets obtained included HO-1, TNF, and others. Enrichment analysis was performed using Gene Ontology genes, Kyoto Encyclopedia of Genes and Genomes, and WikiPathways. The analysis revealed that the related pathways include ferroptosis, the Nrf2 signaling pathway, and the TNF signaling pathway (Figure 1C).
The enriched biological process terms included the regulation of homeostasis in blood circulation, which plays an essential role in maintaining physiological stability. At the molecular function level, the focus was on hydrolase activity and nuclear receptor transcriptional regulation. These functions help maintain homeostasis of the body. From the cellular component perspective, the enrichment results primarily focused on the plasma membrane and its specialized structures in polarized cells as well as associated organelles with the core emphasis on membrane regionalization and functional differentiation. This is a crucial location for the realization of cellular functions (Figure 1D).
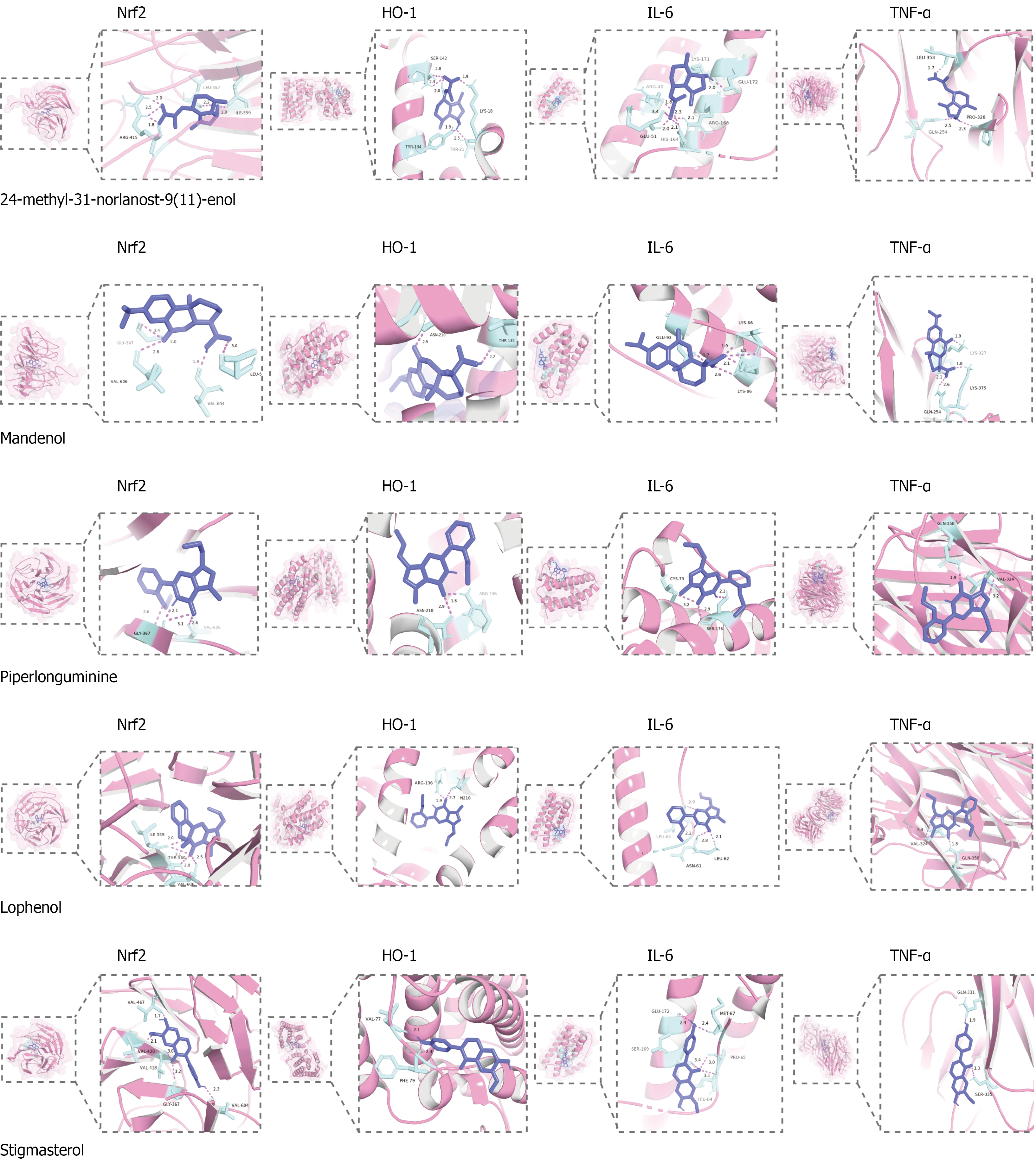
We used molecular docking to validate the interactions between the active compounds in the formula and crucial targets within key targets in the Nrf2 and TNF signaling pathways. An affinity lower than 5.0 kcal/mol indicates a strong binding between the ligand and receptor. These results indicated that the 23 active compounds have the strongest binding affinity to the crucial targets (Figure 1E). The main results of the molecular docking visualization are shown in Figure 2.
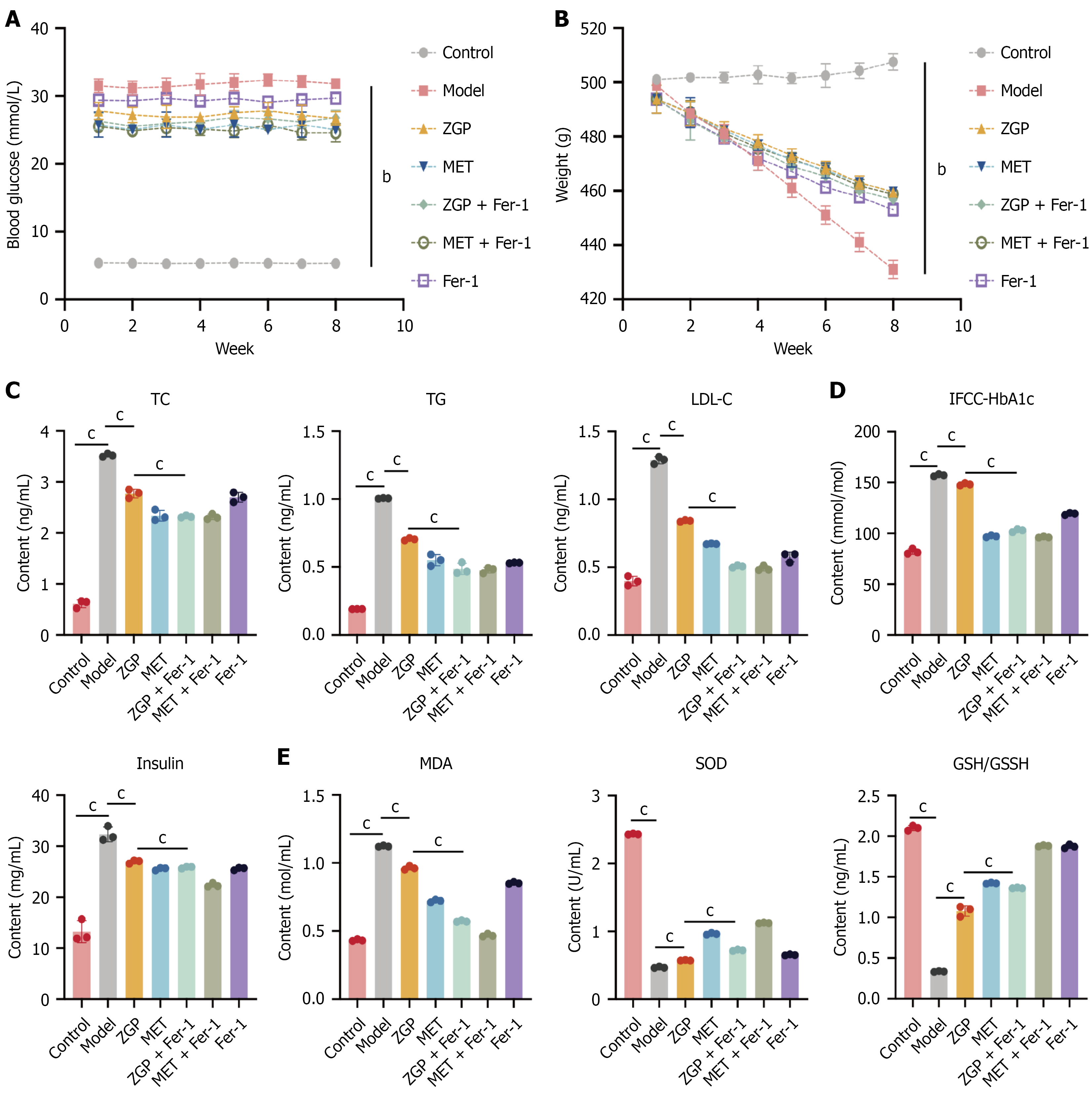
The blood glucose of rats in the T2DOP group was substantially higher than in the control group (P < 0.01). Relative to the model group, the blood glucose of rats in the ZGP group, Fer-1 group, and ZGP + Fer-1 group was reduced (Figure 3A). In comparison with the control group, the body weight of rats in the T2DOP group was significantly lower (P < 0.01). Relative to the model group, the body weight of rats in the ZGP group, Fer-1 group, and ZGP + Fer-1 group was slightly higher (Figure 3B). The serum levels of TC, TG, LDL-C, insulin, and International Federation of Clinical Che
The model group rats had higher serum MDA levels compared with the control group. They also had lower levels of SOD and GSH/GSSH. The differences were significant (P < 0.001). In comparison with the model group, the ZGP group, MET group, Fer-1 group, ZGP + Fer-1 group, and MET + Fer-1 group showed a significant decrease in MDA levels and a significant increase in SOD and GSH/GSSH levels (P < 0.001). Relative to the ZGP group, the ZGP + Fer-1 group exhibited a substantial decrease in MDA levels and a marked increase in SOD and GSH/GSSH levels (P < 0.001). In contrast to the Fer-1 group, the ZGP + Fer-1 group and the MET + Fer-1 group showed a substantial decrease in MDA levels and a significant increase in SOD levels (P < 0.001; Figure 3E).
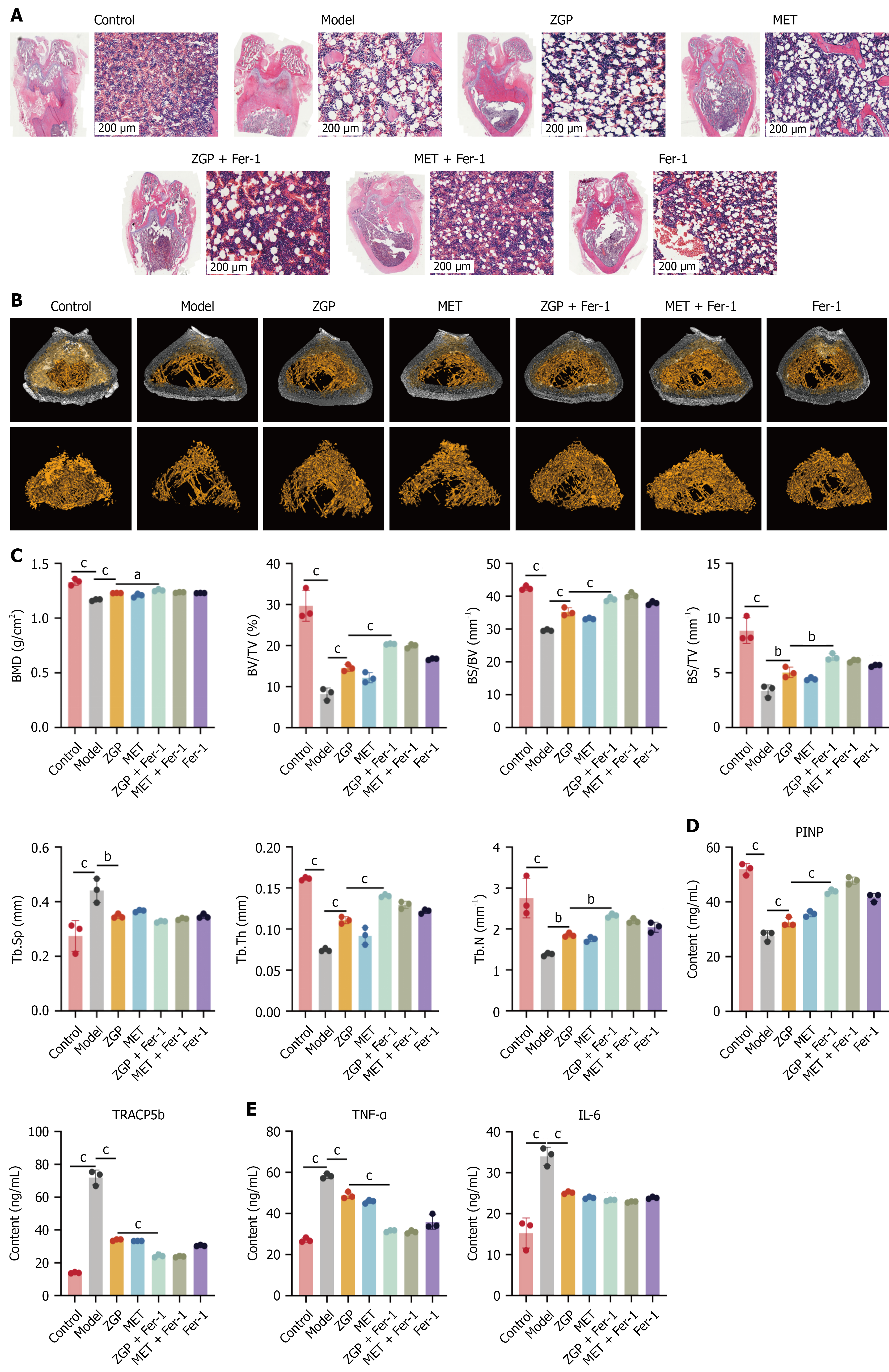
Hematoxylin and eosin staining revealed that in comparison with the control group the T2DOP group exhibited decreased bone density at the distal femur, increased vacuolization, disrupted trabecular structure, disordered trabecular arrangement, widened trabecular gaps, reduced osteocytes, and increased osteoclasts. The bone tissue density at the distal femur in the ZGP group, MET group, Fer-1 group, ZGP + Fer-1 group, and MET + Fer-1 group registered a notable improvement compared with the T2DOP group. They had a more complete trabecular structure. Bone lipid vacuolization was reduced. The trabeculae were more organized (Figure 4A).
Micro-CT imaging showed that the T2DOP group had more bone loss in the distal femur than the normal group. The T2DOP group also had less trabecular bone (Figure 4B). BMD, bone surface (BS)/total volume (TV), BS/bone volume (BV), BV/TV, trabecular number (Tb.N), and trabecular thickness (Tb.Th) were substantially decreased in the ZGP group, and trabecular spacing was markedly increased (P < 0.001). Relative to the T2DOP group, the BMD, BS/TV, BS/BV, BV/TV, Tb.N, and Tb.Th were higher in the ZGP group, MET group, Fer-1 group, ZGP + Fer-1 group, and MET + Fer-1 group (P < 0.01). On the other hand, trabecular spacing was lower in these groups (P < 0.01). Compared with the ZGP group, the ZGP + Fer-1 group exhibited a rise in BMD, BS/TV, BS/BV, BV/TV, Tb.N, and Tb.Th (P < 0.05; Figure 4C).
Relative to the control group, the TRACP5b levels in the model group were significantly increased (P < 0.001). The PINP levels were conversely diminished (P < 0.001). In the ZGP group and the ZGP + Fer-1 group, the TRACP5b levels were decreased more than those in the model group (P < 0.001). The PINP levels were considerably higher than those in the model group (P < 0.001). Compared with the ZGP group, the TRACP5b levels in the ZGP + Fer-1 group decreased more substantially (P < 0.001). The increase in PINP levels was more pronounced (P < 0.001; Figure 4D).
The serum levels of IL-6 and TNF-α in the model group were markedly higher than those in the control group (P < 0.001). In contrast to the model group, the levels of IL-6 and TNF-α were significantly decreased in the ZGP and ZGP + Fer-1 groups (P < 0.001). In the treatment groups, the levels of TNF-α in the ZGP + Fer-1 group were lower than the ZGP and Fer-1 treatment groups (P < 0.001; Figure 4E).
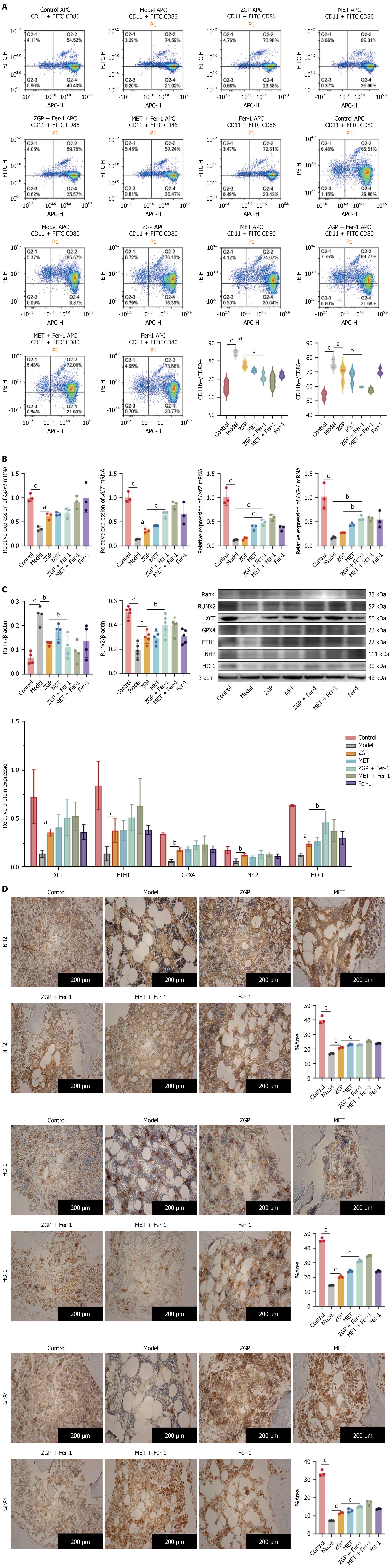
The model group showed a greater increase in CD11b+/CD86+ and CD11b+/CD80+ double positive M1 bone marrow macrophages compared with the control group (P < 0.001). In comparison with the model group, the ZGP and ZGP + Fer-1 groups exhibited a considerable decrease in CD11b+/CD86+ and CD11b+/CD80+ double-positive M1 macrophages (P < 0.05). In contrast to the ZGP and Fer-1 groups, the rats in the ZGP + Fer-1 group showed a relative decrease in CD11b+/CD86+ and CD11b+/CD80+ double-positive M1 macrophages (P < 0.01; Figure 5A).
Compared with the control group, the rats in the model group showed a significant decrease in the expression of Nrf2 mRNA, HO-1 mRNA, GPX4 mRNA, and XCT mRNA (P < 0.001). In contrast to the model group, the expression of Nrf2 mRNA, HO-1 mRNA, GPX4 mRNA, and XCT mRNA was significantly increased in the ZGP group, MET group, and ZGP + Fer-1 group after treatment (P < 0.05). In comparison with the ZGP and Fer-1 groups, the expression of Nrf2 mRNA and GPX4 mRNA was markedly increased in the ZGP + Fer-1 group of rats (Figure 5B).
Compared with the control group, the model group showed a substantial decrease in Runx2 protein expression (P < 0.001) and a significant increase in Rankl protein expression (P < 0.001). After intervention Runx2 protein expression was markedly increased in the ZGP group, MET group, Fer-1 group, ZGP + Fer-1 group, and MET + Fer-1 group. Rankl protein expression was significantly decreased (P < 0.01). Runx2 protein expression in the ZGP + Fer-1 and MET + Fer-1 groups was similar to that in the ZGP, MET, and Fer-1 groups with a substantially statistical difference (P < 0.01; Figure 5C).
The rats in the model group had substantially lower levels of Nrf2, HO-1, GPX4, and XCT proteins than the rats in the control group (P < 0.001). Relative to the model group, the expression of Nrf2, HO-1, GPX4, and XCT proteins was significantly increased in the rats treated with ZGP, MET, Fer-1, ZGP + Fer-1, and MET + Fer-1 groups (P < 0.05). Relative to the ZGP and Fer-1 groups, the ZGP + Fer-1 group exhibited a stronger inhibitory effect on the expression of Nrf2, HO-1, GPX4, and XCT proteins. Immunohistochemical analysis revealed that compared with the model group rats ZGP significantly regulated the expression of Nrf2, HO-1, and GPX4 in bone tissue (P < 0.001). Additionally, compared with the ZGP group, the ZGP + Fer-1 group further increased the expression levels of Nrf2, HO-1, and GPX4 (P < 0.001; Figure 5C and D).
T2DOP is a common complication of T2DM. This condition is marked by bone loss, reduced bone formation, and degeneration of the bone microstructure, causing pain and fractures[9,10]. The global population is aging and the number of people with T2DM is increasing. As a result the number of T2DOP cases is anticipated to grow exponentially[11]. This makes it a widespread public health problem. Studies show that inflammation is a main cause of T2DOP. Hyperglycemia triggers a systemic inflammatory response, disrupting the bone metabolic balance. Currently, treatment focuses on medications for T2DM and osteoporosis. Nonetheless, these therapies often come with adverse effects or are expensive. Therefore, developing safer, more effective, and economical TCM treatment strategies is still a critical challenge.
In the present study, a T2DOP rat model was established using a high-fat diet combined with low-dose STZ administration to investigate the therapeutic mechanisms of ZGP. This study demonstrated that ZGP in combination with ferroptosis inhibitors significantly stimulated bone growth while inhibiting bone breakdown. It significantly enhanced various parameters of bone microstructure, such as BMD, BS/TV, BS/BV, BV/TV, Tb.N, and Tb.Th. Additionally, compared with the model group, ZGP combined with Fer-1 increased the serum level of the bone formation biomarker PINP. At the same time it reduced the levels of the bone resorption marker TRAP5b. The expression level of the osteogenic marker Runx2 was found to be upregulated, and the expression level of the osteoclast marker Rankl was found to be downregulated, further confirming the potential impact of ZGP in combination with Fer-1 on the regulation of bone metabolism.
During the synthesis of active type I collagen, PINP is a precursor protein cleaved by osteoblasts. Its concentration directly reflects the activity of osteoblasts within bone tissue[12]. Runx2 is widely defined as a positive regulator of osteoblast differentiation and bone formation[13,14]. Runx2 is an essential master transcription factor for osteoblast maturation. It is crucial for osteoblast maturation and bone formation[15]. Mice with a knockout of the Runx2 gene have lower bone density. They also exhibit decreased trabecular bone volume. Bone formation and resorption markers are at lower levels[16]. Studies show that high ROS levels cause Runx2 to oxidize. This leads to its breakdown by the proteasome. As a result osteoblast differentiation is blocked[17]. During the process of bone mineral dissolution, TRACP5b is an acidic hydrolase secreted by osteoclasts and involved in the degradation of the bone matrix[18]. Therefore, TRACP5b is considered a biomarker for assessing bone resorption status. Dynamic monitoring is helpful for evaluating changes in bone quality[19-21]. Rankl primarily regulates bone remodeling and is related to the immune system. Rankl is considered one of the key factors at the crossroads of bone metabolism and immunity[22]. The changes in these biomarkers indicate that ZGP together with Fer-1 not only stops osteoclast formation but also increases osteoblast activity. This helps to restore bone balance in T2DOP.
This study used TCMSP to screen 76 active compounds. Pathway enrichment analysis indicated that the Nrf2/HO-1 signaling axis is a crucial pathway. The prediction results are consistent with relevant mechanisms in the existing literature[23]. It plays a key role in anti-inflammatory action, regulation of oxidative stress, and cell death modulation. This pathway is intricately linked to the pathogenesis of T2DOP[24]. Nrf2 serves as a key redox-sensitive transcription factor responsible for regulating the oxidative-reduction balance in mammalian cells[25]. HO-1 and GPX4 are both antioxidant enzymes downstream of Nrf2. GPX4 is a critical suppressor of ferroptosis as it reduces lipid hydroperoxides to non-toxic lipid alcohols using GSH as a cofactor. Ferroptosis is marked by the buildup of lipid peroxides and ROS due to oxidative stress[26]. An increase in ROS can significantly affect the formation and survival of osteoblasts, while inhibiting their differentiation into osteoclasts. Therefore, oxidative stress and ferroptosis have been widely reported as important factors in the pathogenesis of T2DOP[27,28].
Research on molecular docking shows that the active compounds of ZGP strongly interact with the Nrf2/HO-1 pathway. This supports the idea that ZGP affects T2DOP through a multicomponent, multitarget mechanism. Based on this, quantitative PCR and western blot analyses were used. These analyses confirmed that ZGP in conjunction with Fer-1 can intervene on the targets Nrf2, HO-1, Gpx4, and XCT in T2DOP rats. Our findings suggest that ZGP may attenuate ferroptosis-related processes in bone, potentially involving the Nrf2/HO-1/XCT/GPX4 axis. This exerts antioxidative stress effects and inhibits the occurrence of ferroptosis. This, in turn, enhances the formation and survival of osteoblasts while inhibiting osteoclast activation. Macrophages play an important role in bone metabolism. Their function and polarization help regulate this process[29].
In response to inflammation activating the Nrf2 signaling pathway raises the levels of inflammatory factors and cytokines. This causes macrophages to polarize toward the M1 phenotype[30-32]. In this study flow cytometry results showed that ZGP combined with Fer-1 significantly reduced the levels of proinflammatory M1 macrophage markers (CD86, CD80). This indicates that ZGP in conjunction with Fer-1 effectively inhibited M1 polarization of bone marrow macrophages. It also suppressed the inflammatory response. This suggests that ZGP may also regulate the bone immune microenvironment. It could also contribute to increasing osteogenesis and decreasing osteoclastic activity. The beneficial effects of ZGP observed in T2DOP are the result of interactions between multiple elements and various targets. This highlights its promise as a clinical solution for T2DOP.
This study demonstrated that ZGP in combination with Fer-1 had a synergistic effect in inhibiting ferroptosis. ZGP enhanced the expression of HO-1, GPX4, and XCT. This activates Nrf2 and restores the positive regulatory mechanisms. As a result oxidative stress and inflammation were alleviated. M1 macrophage polarization was reduced, ferroptosis was inhibited, and T2DOP was improved. The complex mechanism provides foundation for using ZGP to treat T2DOP and emphasizes the unique benefits of natural products. These findings help us understand how ZGP regulates bone metabolism in T2DOP. Compared with the multicomponent ZGP intervention, single-target agents typically act on a single dominant pathway and may therefore be insufficient to address the multifactorial pathophysiology of diabetes-associated osteoporosis[33].
ZGP has shown its potential as a new treatment for osteoporosis. Nevertheless, we acknowledge several important limitations. Network pharmacology was employed as an exploratory, hypothesis-generating tool to identify pathways potentially involved in the effects of ZGP on T2DOP; however, it does not provide direct causal evidence. Moreover, the absence of comprehensive chemical fingerprinting and quantitative analysis of key constituents represents an additional limitation. Other constraints include species differences between rodents and humans, variations in disease duration and comorbidities, the inability of the high-fat diet/Low-dose STZ model to fully capture the heterogeneity of human T2DOP, and uncertainties regarding human-equivalent dosing, standardization, and long-term safety.
Accordingly, further clinical validation in well-designed human studies will be required before these findings can be applied to patients. Moreover, we acknowledge the need for further research to completely confirm these mechanisms. Future studies should focus on in vivo experiments with monocyte macrophages. These studies can directly investigate their role in the therapeutic effects of ZGP extracts in T2DOP treatment. Additionally, gene knockout techniques could help us understand the molecular mechanisms better. They can provide deeper insights into how these mechanisms work. Based on these findings, an optimized ZGP formula will be developed. It will include more refined ingredients, precise dosing, and clearer mechanisms. This will lay a solid foundation for clinical precision intervention in T2DOP.
The potential synergistic effects of ZGP with other drugs will be explored to possibly mitigate drug resistance. Our study validated the findings of ZGP. It aimed to improve the treatment strategy for T2DOP and patients’ quality of life. Therefore, this study laid the foundation for the future development of ZGP in the treatment of T2DOP.
Our study showed that ZGP can regulate macrophage polarization through the Nrf2/HO-1 pathway. This helped prevent and treat T2DOP as demonstrated by network pharmacology and animal experiments. ZGP exhibited a synergistic effect with Fer-1 in inhibiting ferroptosis. These effects were exerted through different ingredients, aims, and mechanisms of action. Our research showed that ZGP can be used for the prevention and treatment of T2DOP, providing worthwhile evidence for its clinical application.
We sincerely thank the laboratory at Liaoning University of Traditional Chinese Medicine for its support.
| 1. | Sheu A, Greenfield JR, White CP, Center JR. Contributors to impaired bone health in type 2 diabetes. Trends Endocrinol Metab. 2023;34:34-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (1)] |
| 2. | Rajagopalan S, Brook R. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377:2098-2099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 194] [Article Influence: 21.6] [Reference Citation Analysis (3)] |
| 3. | Silverman S, Kupperman E, Bukata S. Bisphosphonate-related atypical femoral fracture: Managing a rare but serious complication. Cleve Clin J Med. 2018;85:885-893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Zeng L, Ye J, Zhang Z, Liang Y, Li J, Zeng L, Cao L, Zhu L, Luo S. Zuogui pills maintain the stemness of oogonial stem cells and alleviate cyclophosphamide-induced ovarian aging through Notch signaling pathway. Phytomedicine. 2022;99:153975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Shi T, Liu T, Kou Y, Rong X, Meng L, Cui Y, Gao R, Hu S, Li M. The Synergistic Effect of Zuogui Pill and Eldecalcitol on Improving Bone Mass and Osteogenesis in Type 2 Diabetic Osteoporosis. Medicina (Kaunas). 2023;59:1414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 6. | Loboda A, Damulewicz M, Pyza E, Jozkowicz A, Dulak J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci. 2016;73:3221-3247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2294] [Cited by in RCA: 2112] [Article Influence: 211.2] [Reference Citation Analysis (6)] |
| 7. | Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol. 2007;47:89-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3126] [Cited by in RCA: 2907] [Article Influence: 153.0] [Reference Citation Analysis (0)] |
| 8. | Arai M, Shibata Y, Pugdee K, Abiko Y, Ogata Y. Effects of reactive oxygen species (ROS) on antioxidant system and osteoblastic differentiation in MC3T3-E1 cells. IUBMB Life. 2007;59:27-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 173] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 9. | Napoli N, Chandran M, Pierroz DD, Abrahamsen B, Schwartz AV, Ferrari SL; IOF Bone and Diabetes Working Group. Mechanisms of diabetes mellitus-induced bone fragility. Nat Rev Endocrinol. 2017;13:208-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 929] [Cited by in RCA: 807] [Article Influence: 89.7] [Reference Citation Analysis (4)] |
| 10. | Papaioannou I, Pantazidou G, Kokkalis Z, Georgopoulos N, Jelastopulu E. Systematic Review: Are the Elderly With Diabetes Mellitus Type 2 Prone to Fragility Fractures? Cureus. 2021;13:e14514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 11. | Ma RX, Lin BH, Feng SX, Bu YT, Chen ZH, Huang YX, Li EL, Weng SJ, Yang L. Evaluation of proanthocyanidins in treating Type 2 diabetic osteoporosis via SIRT6/Nrf2/GPX4 pathways. FASEB J. 2025;39:e70487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Koivula MK, Risteli L, Risteli J. Measurement of aminoterminal propeptide of type I procollagen (PINP) in serum. Clin Biochem. 2012;45:920-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 13. | Drissi H, Luc Q, Shakoori R, Chuva De Sousa Lopes S, Choi JY, Terry A, Hu M, Jones S, Neil JC, Lian JB, Stein JL, Van Wijnen AJ, Stein GS. Transcriptional autoregulation of the bone related CBFA1/RUNX2 gene. J Cell Physiol. 2000;184:341-350. [PubMed] [DOI] [Full Text] |
| 14. | Hu G, Yu Y, Sharma D, Pruett-Miller SM, Ren Y, Zhang GF, Karner CM. Glutathione limits RUNX2 oxidation and degradation to regulate bone formation. JCI Insight. 2023;8:e166888. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 54] [Reference Citation Analysis (0)] |
| 15. | Komori T. Roles of Runx2 in Skeletal Development. Adv Exp Med Biol. 2017;962:83-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 159] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 16. | Komori T. Whole Aspect of Runx2 Functions in Skeletal Development. Int J Mol Sci. 2022;23:5776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 161] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 17. | Qin X, Jiang Q, Komori H, Sakane C, Fukuyama R, Matsuo Y, Ito K, Miyazaki T, Komori T. Runt-related transcription factor-2 (Runx2) is required for bone matrix protein gene expression in committed osteoblasts in mice. J Bone Miner Res. 2021;36:2081-2095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 18. | Lv Y, Wang G, Xu W, Tao P, Lv X, Wang Y. Tartrate-resistant acid phosphatase 5b is a marker of osteoclast number and volume in RAW 264.7 cells treated with receptor-activated nuclear κB ligand. Exp Ther Med. 2015;9:143-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 19. | Sobacchi C, Menale C, Crisafulli L, Ficara F. Role of RANKL Signaling in Bone Homeostasis. Physiology (Bethesda). 2025;40:0. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 20. | Eastell R, Szulc P. Use of bone turnover markers in postmenopausal osteoporosis. Lancet Diabetes Endocrinol. 2017;5:908-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 421] [Article Influence: 46.8] [Reference Citation Analysis (1)] |
| 21. | Ma H, Wang X, Zhang W, Li H, Zhao W, Sun J, Yang M. Melatonin Suppresses Ferroptosis Induced by High Glucose via Activation of the Nrf2/HO-1 Signaling Pathway in Type 2 Diabetic Osteoporosis. Oxid Med Cell Longev. 2020;2020:9067610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 321] [Article Influence: 53.5] [Reference Citation Analysis (4)] |
| 22. | Huang Y, Li W, Su ZY, Kong AN. The complexity of the Nrf2 pathway: beyond the antioxidant response. J Nutr Biochem. 2015;26:1401-1413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 358] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 23. | Wang YB, Li ZP, Wang P, Wang RB, Ruan YH, Shi Z, Li HY, Sun JK, Mi Y, Li CJ, Zheng PY, Zhang CJ. Iron dysregulation, ferroptosis, and oxidative stress in diabetic osteoporosis: Mechanisms, bone metabolism disruption, and therapeutic strategies. World J Diabetes. 2025;16:106720. [PubMed] [DOI] [Full Text] |
| 24. | Zhang Q, Zhao L, Shen Y, He Y, Cheng G, Yin M, Zhang Q, Qin L. Curculigoside Protects against Excess-Iron-Induced Bone Loss by Attenuating Akt-FoxO1-Dependent Oxidative Damage to Mice and Osteoblastic MC3T3-E1 Cells. Oxid Med Cell Longev. 2019;2019:9281481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 25. | Jing X, Du T, Chen K, Guo J, Xiang W, Yao X, Sun K, Ye Y, Guo F. Icariin protects against iron overload-induced bone loss via suppressing oxidative stress. J Cell Physiol. 2019;234:10123-10137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (3)] |
| 26. | Behera J, Ison J, Voor MJ, Tyagi N. Exercise-Linked Skeletal Irisin Ameliorates Diabetes-Associated Osteoporosis by Inhibiting the Oxidative Damage-Dependent miR-150-FNDC5/Pyroptosis Axis. Diabetes. 2022;71:2777-2792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (4)] |
| 27. | Fan H, Tang HB, Shan LQ, Liu SC, Huang DG, Chen X, Chen Z, Yang M, Yin XH, Yang H, Hao DJ. Quercetin prevents necroptosis of oligodendrocytes by inhibiting macrophages/microglia polarization to M1 phenotype after spinal cord injury in rats. J Neuroinflammation. 2019;16:206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 163] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 28. | Li Z, Liang Y, Wang Y, Lin Y, Zeng L, Zhang Y, Zhu L. Zuogui Pills alleviate cyclophosphamide-induced ovarian aging by reducing oxidative stress and restoring the stemness of oogonial stem cells through the Nrf2/HO-1 signaling pathway. J Ethnopharmacol. 2024;333:118505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 29. | Ren Q, Xing W, Jiang B, Feng H, Hu X, Suo J, Wang L, Zou W. Tenascin-C promotes bone regeneration via inflammatory macrophages. Cell Death Differ. 2025;32:763-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 30. | Montoya T, Aparicio-Soto M, Castejón ML, Rosillo MÁ, Sánchez-Hidalgo M, Begines P, Fernández-Bolaños JG, Alarcón-de-la-Lastra C. Peracetylated hydroxytyrosol, a new hydroxytyrosol derivate, attenuates LPS-induced inflammatory response in murine peritoneal macrophages via regulation of non-canonical inflammasome, Nrf2/HO1 and JAK/STAT signaling pathways. J Nutr Biochem. 2018;57:110-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 31. | Liu H, Dai Q, Yang J, Zhang Y, Zhang B, Zhong L. Zuogui Pill Attenuates Neuroinflammation and Improves Cognitive Function in Cerebral Ischemia Reperfusion-Injured Rats. Neuroimmunomodulation. 2022;29:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 32. | Chen M, Lin W, Ye R, Yi J, Zhao Z. PPARβ/δ Agonist Alleviates Diabetic Osteoporosis via Regulating M1/M2 Macrophage Polarization. Front Cell Dev Biol. 2021;9:753194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 33. | Compston JE, McClung MR, Leslie WD. Osteoporosis. Lancet. 2019;393:364-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2087] [Cited by in RCA: 1742] [Article Influence: 248.9] [Reference Citation Analysis (0)] |