Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.116053
Revised: December 12, 2025
Accepted: January 30, 2026
Published online: May 15, 2026
Processing time: 191 Days and 15.2 Hours
In our search to unravel the pathophysiological mechanisms underlying the development of insulin resistance (IR) in the young population, we evaluated the waist-to-height ratio (WtHR) in 39 participants, setting 0.5 as the cut-off value. A high WtHR has been associated with metabolic alterations and increased risk of developing IR.
To explore the relationship between WtHR and IR in young individuals with endoplasmic reticulum (ER) stress molecular mechanisms.
This was a cross-sectional study conducted at the Faculty of Medicine of the Autonomous University of Baja California (Mexico). A total of 39 young parti
Young individuals with high WtHR showed greater body weight, body mass index, and visceral fat area per
High-WtHR in young people is associated with IR and ER stress, driven by JNK activation in platelets, suggesting molecular alterations in early metabolic dysfunction.
Core Tip: Insulin resistance (IR), a health complication commonly associated with advanced age, is increasingly affecting the young population due to poor eating habits and a sedentary lifestyle. In this study, conducted with 39 young adults, participants with a waist-to-height ratio greater than 0.5 showed elevated insulin levels, higher Homeostatic Model Assessment of Insulin Resistance, and altered body composition. Likewise, these participants exhibited platelet molecular disturbances, including IR and activation of proteins associated with endoplasmic reticulum stress. The results suggest that excessive central adiposity in the young population triggers early activation of molecular mechanisms underlying the development of metabolic diseases, such as type 2 diabetes mellitus.
- Citation: Casillas OE, Hernández-Aguirre CA, Leija-Montoya AG, Romero-García T, Vázquez-Jiménez JG. Association between high waist-height ratio and endoplasmic reticulum stress of young adults with insulin resistance. World J Diabetes 2026; 17(5): 116053
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/116053.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.116053
Excessive adiposity leads to dyslipidemia, overweight, and obesity and its comorbidities, which constitute the most common chronic degenerative disease worldwide and is the main condition that favors the development of type 2 diabetes mellitus (T2DM)[1]. There is solid evidence showing an association between alterations in lipid metabolism and the development of insulin resistance (IR), a condition that, if left uncorrected, favors the progression to T2DM[2]. Some studies show a growing prevalence of IR among young adults, with a current rate of around 40%. Interestingly, almost half of those insulin-resistant young are non-obese, which prevents rapid and effective diagnosis and treatment before the onset of T2DM[3]. Given this diagnostic challenge, anthropometric indicators that can identify central adiposity in apparently healthy-weight individuals are essential. In this context, waist-to-height ratio (WtHR) is widely used as a clinical marker, and there is strong evidence that a WtHR > 0.5 means a higher risk of suffering from chronic de
Regarding adiposity, especially visceral fat, and excessive circulating free fatty acids (FFAs), these are the leading causes of IR and T2DM by disrupting several metabolic mechanisms, including chronic inflammation, adipocyte enlargement, increased oxidative stress, and endoplasmic reticulum (ER) stress. In fact, ER stress is linked to lipid accumulation and inflammation, both of which contribute to the progression of IR and T2DM[8]. In this context, high FFA plasma concentrations are associated with the activation of kinases, which leads to the negative regulation of insulin signaling, primarily through IRS-1 phosphorylation in serine residues[9].
At the cellular level, the ER is responsible for maturing newly synthesized proteins; for proper functioning, this organelle requires maintaining high luminal calcium concentrations. In this respect, the SERCA maintains high calcium levels in the ER. Recently, it has been shown that high concentrations of saturated fatty acids decrease SERCA activity and expression. These mechanisms cause an impairment of calcium homeostasis in the ER, allowing activation of the unfolded protein response (UPR), which induces the phosphorylation and activation of the PERK protein[10,11]. PERK phosphorylation serves as a sensor of ER stress, a process that activates the UPR pathway. The UPR pathway induces the activation of JNK, which negatively regulates the insulin signaling pathway by phosphorylating the insulin receptor at serine residues, blocking the PI3K/Akt pathway[12]. Thus, the failure to correct ER stress favors the development of IR, as it has been widely reported in human tissues[12-14].
Interestingly, these pathophysiological mechanisms have only been observed in cell cultures and murine models. However, alterations in SERCA expression and activity for maintaining Ca2+ homeostasis have been reported in platelets from individuals with T2DM. Therefore, to understand the disease's development and progression, one missing link is to determine whether these changes are present in apparently healthy young people with IR.
Since biopsies from tissues with high metabolic activity are difficult to obtain, while blood sampling is comparatively practical, isolating platelets is a feasible method that provides a suitable model for evaluating calcium homeostasis. Considering that platelet dysfunction is a key link between IR and increased cardiometabolic risk, in this context, due to the loss of insulin´s normal antiplatelet effect, platelets exhibit hyperactivity with increased activation and aggregation, favoring a prothrombotic state[15]. Hence, to understand the initial molecular disruptions in the insulin pathway that can lead to the early onset of chronic diseases, this study aimed to evaluate whether young adults with high WtHR exhibit IR and whether this state is associated with ER stress in platelets.
The study was previously approved by the Autonomous University of Baja California, Sports Faculty-Ethics Committee (Approval No. 265/2023-1), in accordance with the principles of the Declaration of Helsinki, as revised in 2000. Informed consent was obtained and documented before initiating the protocol.
Since the purpose of this study was to investigate metabolic alterations and their association with IR in apparently healthy young adults, we conducted a cross-sectional descriptive study, recruiting all newly enrolled students in 2023 at the Faculty of Medicine and Nutrition of Mexicali, the Autonomous University of Baja California.
After applying the exclusion criteria, which included having any of the following conditions—infectious diseases, cancer, liver disease, polycystic ovary syndrome, type 2 diabetes, or pregnancy—and excluding participants with a BMI outside the normal range (< 18.5 kg/m2 or > 40 kg/m2), a total of 39 young adults (men and women) aged 18-5 years were enrolled in the study. The sample size was calculated using G*Power v. 3.1.9.7[16].
Anthropometric parameters were measured after participants had fasted for 8 hours, were barefoot, and wore light clothing. Height was measured using a calibrated SECA 213 stadiometer, and waist circumference was measured using a SECA 201 tape. The WtHR was then calculated by dividing the abdominal circumference by the height, expressed in centimeters. Blood pressure was measured with a digital monitor (OSZ 5, Welch Allyn). Body composition was measured by bioelectrical impedance (Jawon, Ioi 353), and the obtained data were analyzed with Body Pass Plus software. Participants were classified into two groups based on WtHR, using 0.5 as the cutoff: Those with a WtHR < 0.5 were assigned to the low-WtHR group, and those with a WtHR > 0.5 to the high-WtHR group.
Peripheral blood was collected and used to determine glucose using a blood glucose monitoring system (Freestyle Optium neo, Abbott, United Kingdom), as well as total cholesterol (TC), triglycerides (TGs), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C) using a Mission Lipid Panel Test Device (Acon, United States). The TG/HDL-C was then calculated by dividing the blood TG by the HDL-C levels expressed in mg/dL. The remnant peripheral blood was then centrifuged at 350 × g for 15 minutes to obtain plasma, and insulin levels were determined by ELISA assay using a specific kit (Cat. KAQ1251, Thermo Fisher, United States) following the manu
Peripheral blood was collected and centrifuged at 350 × g for 15 minutes to obtain platelet-rich plasma, as described by Burzynski et al[17]. Then, the platelet-rich plasma was centrifuged at 2000 × g for 20 minutes, and the pellet was resuspended in PBS to obtain platelet proteins. Total protein was quantified using a BCA protein assay kit following the manufacturer's instructions (Cat. 23227, Thermo Fisher, United States). Laemmli buffer was added to the platelet protein samples before they were loaded into 7.5% polyacrylamide gels. The SDS-PAGE-resolved proteins were transferred to PVDF membranes and then blocked for 60 minutes in 5% non-fat milk in TBS-T buffer. Specific primary antibodies were incubated overnight at 4 °C, including: SERCA, α-actin, pJNK, JNK, PERK, and pPERK (Cat. sc-53010, sc-1616, sc-6254, sc-7345, sc-377400 Santa Cruz Biotechnology; and MA5-15033 Thermo Fisher, respectively), and then incubated with specific secondary antibodies for 1 hour (anti-mouse, Cat. 170-6516, Bio-Rad, United States; anti-rabbit, Cat. 170-6515, Bio-Rad, United States). Finally, the proteins were visualized using a gel documentation system (Gel Doc XR, Bio-Rad, United States). Quantitative analysis was performed with ImageJ software (v. 1.50i, NIH, United States). Actin served as the loading control for SERCA protein levels normalization, total PERK served as the loading control for pPERK normalization, and total JNK served as the loading control for pJNK normalization.
Data are presented as the mean ± SEM. The number of participants for each experiment is indicated as n. Statistical significance was evaluated using One-Way ANOVA followed by the Dunn post hoc test in GraphPad PrismTM v7.0 (GraphPad Software, Inc., San Diego, CA, United States). A P value < 0.05 was considered statistically significant. The statistical methods of this study were reviewed by Rafael I. Ayala from the National Technological Institute of Mexico, Mexicali Campus.
When analyzing participants' anthropometric characteristics, we found that 21 participants (9 men and 12 women) had a WtHR below 0.5, and 18 participants (11 men and 7 women) had a WtHR above 0.5. The high-WtHR group exhibited a greater body weight, a higher BMI, a larger visceral fat area, and a higher percentage of visceral fat compared to the low-WtHR group (Table 1).
| Low-WtHR (n = 21) | High-WtHR (n = 18) | P value | Mean with 95%CI | |
| Weight (kg) | 56.84 ± 2.02 | 82.11 ± 3.83a | < 0.0001 | -25.26 (-33.66 to -16.8) |
| Height (cm) | 165.87 ± 2.09 | 169.38 ± 2.46 | 0.2811 | -3.5 (-10.03 to 2.99) |
| BMI (kg/m2) | 20.8 ± 0.50 | 28.4 ± 0.88a | < 0.0001 | -7.83 (-9.82 to -5.84) |
| Waist (cm) | 74.73 ± 1.44 | 95 ± 2.37a | < 0.0001 | -20.43 (-25.89 to -14.98) |
| WtHR | 0.450 ± 0.007 | 0.561 ± 0.008a | < 0.0001 | -0.11 (-0.13 to -0.08) |
| Body fat (%) | 21.01 ± 1.377 | 31.63 ± 1.59a | < 0.0001 | -10.53 (-14.77 to -6.29) |
| Visceral fat area (cm2) | 42.71 ± 3.41 | 97 ± 5.33a | < 0.0001 | -54.39 (-66.88 to -41.91) |
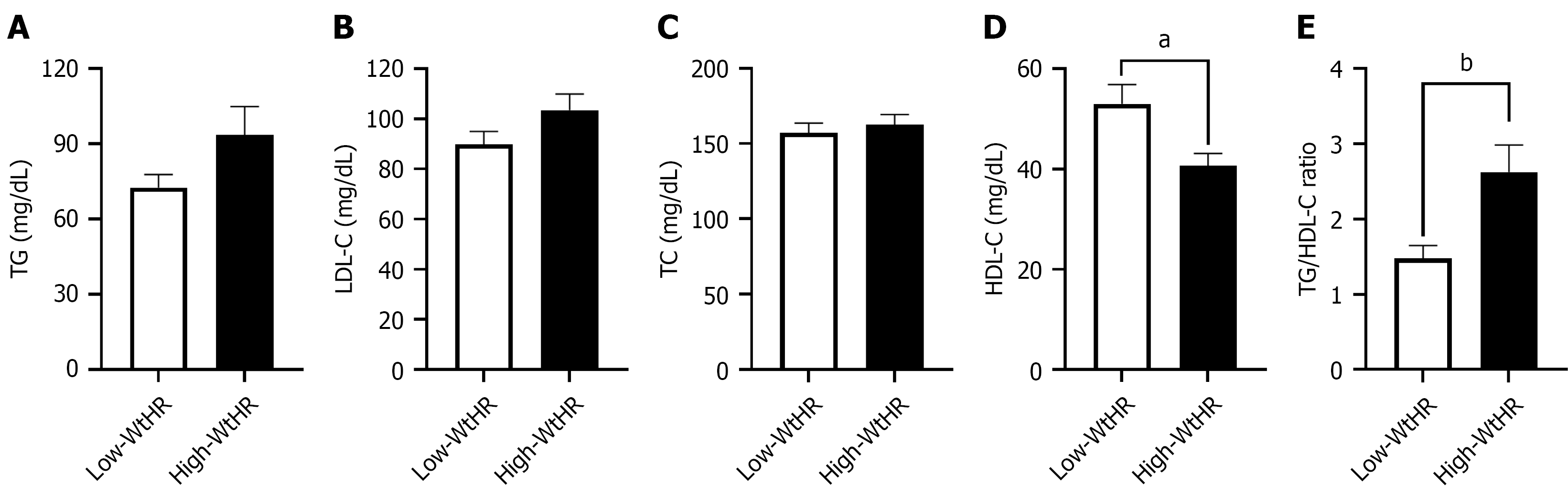
Participants in the high-WtHR group showed no significant differences in TGs, TC, and LDL-C levels compared to those in the low-WtHR group (Figure 1A-C). However, those in the high-WtHR group had significantly lower HDL-C levels (Figure 1D), which contributed to the increased TG/HDL-C ratio observed in this group, an indicator of atherogenic dyslipidemia risk (Figure 1E).
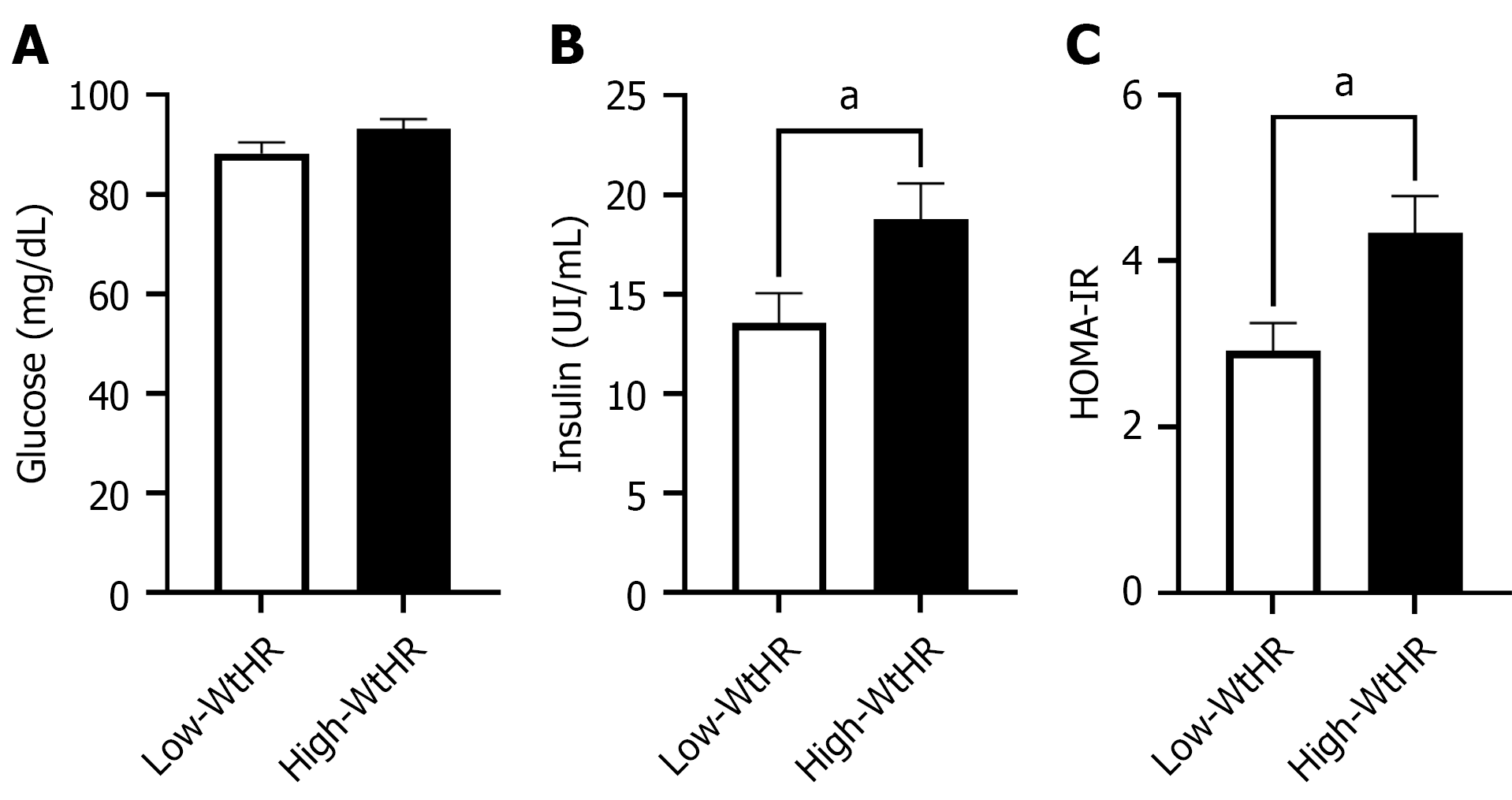
Considering that an increased TG/HDL-C ratio, besides being an indicator of increased cardiometabolic disease predisposition, also serves as an indirect marker of the IR state, we proceed to analyze the glucose and insulin levels. As shown in Figure 2A, there were no statistically significant differences in blood glucose levels between the two groups; however, individuals in the high-WtHR group had greater insulin levels compared to those in the low-WtHR group (18.76 ± 2.354 IU/mL vs 13.54 ± 1.5 IU/mL, P < 0.05; Figure 2B). Using these data, the HOMA-IR index was calculated for the two groups. Interestingly, individuals in the high-WtHR group had an HOMA-IR score associated with IR, in contrast to the low-WtHR group, which had an optimal score (4.34 ± 0.54 vs 2.9 ± 0.3, P < 0.05; Figure 2C).
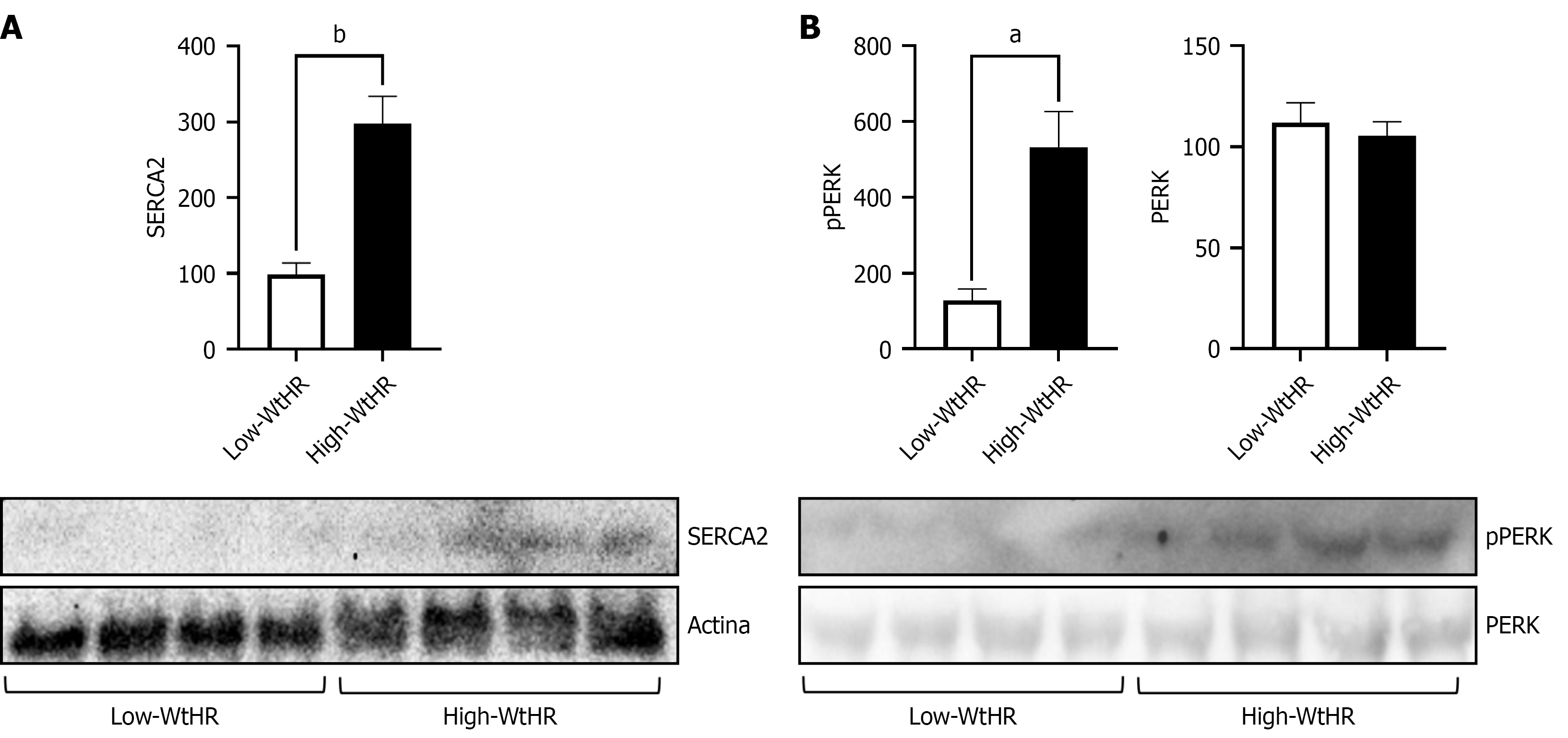
Considering that imbalances in lipid metabolism and the IR are factors associated with ER stress, our goal was to determine whether changes in SERCA expression were linked to the metabolic state of the high WtHR group. To address it, we evaluated SERCA protein levels, since previous reports have shown that platelets from individuals with T2DM display altered expression of the SERCA calcium pump, which disrupts ER calcium homeostasis[18].
As shown in Figure 3A, platelets from individuals in the high-WtHR group expressed higher levels of SERCA, compared to individuals in the low-WtHR group (297.3 ± 39.39 vs 98.75 ± 14.9, P < 0.0002). Although increased SERCA expression is a characteristic response to ER stress, we corroborated that platelets from individuals in the high-WtHR group exhibited ER stress by analyzing pPERK, a post-translational modification that activates PERK and is a widely reported marker of ER stress. Platelets from individuals in the high-WtHR group have significantly greater levels of phosphorylated PERK, compared to participants in the low-WtHR group (532.0 ± 98.87 vs 127.8 ± 29.6, P < 0.001), with no differences in the total expression of this protein, as seen in Figure 3B.
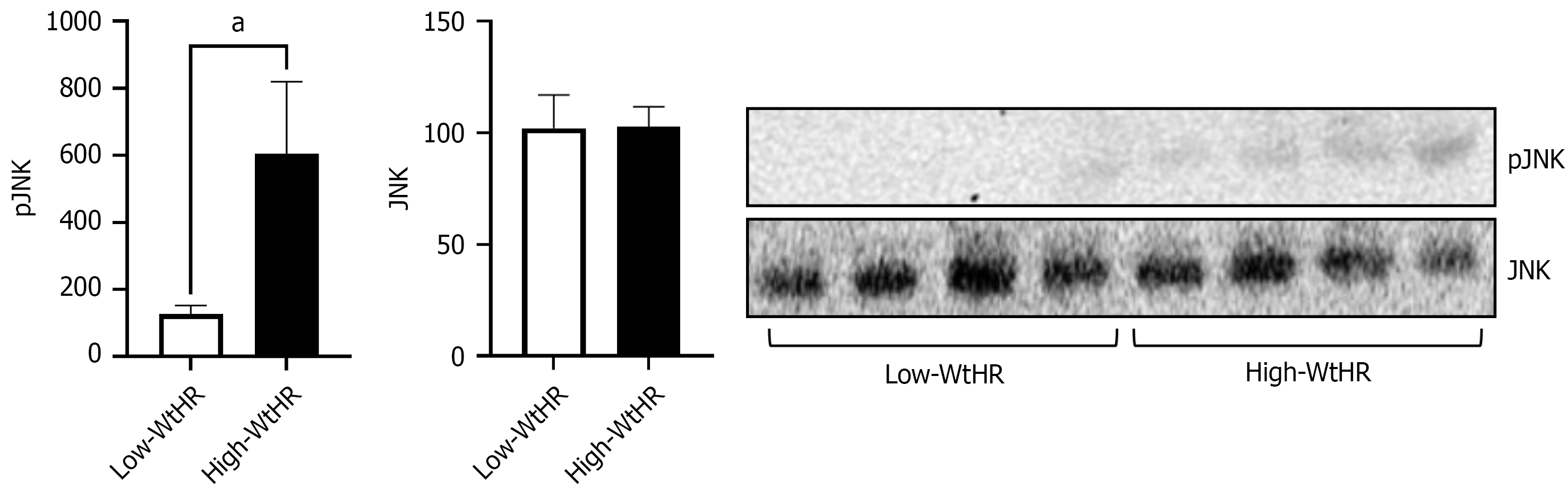
Increased SERCA levels, together with PERK activation, suggested the activation of the UPR pathway in platelets from individuals with high-WtHR. Since the activation of the UPR pathway induces pJNK, the active form of this kinase, which represents a crucial step in the genesis of IR, as pJNK can inhibit insulin signal transduction. As expected, platelets from individuals in the high-WtHR group presented increased levels of pJNK, compared to those from individuals in the low-WtHR group (604.9 ± 216.3 vs 126.7± 25, P < 0.04), confirming the inhibition of the insulin signaling pathway, as shown in Figure 4.
Moreover, downstream mechanisms can block the signaling pathway; for example, the phosphatase and tension homologue (PTEN) can dephosphorylate PI3K. Also, PTEN can negatively modulate insulin signaling by dephosphorylating IRS1/2. Another example is the SHIP-2, which dephosphorylates PIP3[14]. Specifically, these mechanisms interfere with the proper activation of the PI3K/Akt signaling pathway.
Since this research is a cross-sectional descriptive study and the sample size is relatively small, the purpose of this manuscript is to demonstrate that the pathophysiological mechanisms that promote the development of IR, which have been described in cell cultures and murine models[11,19], are also present in apparently healthy young individuals. By examining these mechanisms, we have identified platelets as a safe and reliable model for association[18,20].
It has become evident that there are obese individuals who present a metabolic healthy profile, as well as individuals with IR, metabolic syndrome, and other disorders, despite presenting a normal weight[21]. This has prompted the search for more accurate indicators that include an estimation of adipose tissue and its metabolism, as the mechanistic key linking obesity and cardiometabolic disorders. In this regard, multiple studies consistently show that the prevalence of the metabolically healthy obesity state is higher in children, adolescents, and young adults, which highlights the relevance of early interventions, as the metabolically healthy obesity tends to decline with advancing age[22]. In this context, we opted for the use of WtHR as a tool to identify adipose tissue dysfunction and metabolic risk, since there is solid and recent evidence that associates a high WtHR with prediabetes, IR, diabetes, and cardiovascular disease, justifying its convenience in different populations, including young people[23].
In accordance, our findings demonstrate that all participants with increased WtHR not only exhibit altered anthropometric characteristics (Table 1), but also present a heightened cardiometabolic risk, as evidenced by a TG/HDL-C ratio greater than 2 (Figure 1D) and elevated circulating insulin levels[5]. As previously reported, higher blood insulin levels are indicative of IR, even when glucose concentrations are within normal range[24]. This was corroborated by calculating HOMA-IR (Figure 2C), in which individuals with high WtHR had a mean HOMA-IR greater than 3, a threshold pre
Considering that studies indicate that platelets from people with T2DM have lower expression of SERCA protein[18,27], we therefore examined whether young adults with IR also exhibit alterations in SERCA expression. Interestingly, our data show that individuals with high WtHR had higher SERCA expression levels (Figure 3A). These data contrast with a study we previously published, in which we performed experiments using a human endothelial cell model, demon
In the present study, platelets from young individuals with high WtHR exhibited ER stress, as evidenced by PERK phosphorylation (Figure 3B). Likewise, ER stress induces IR by activating JNK, a serine kinase that can block insulin receptor signaling, thereby favoring IR[10,11,29]. In our research, people with high WtHR presented greater activation of JNK (Figure 4), which constitutes an important fact, by demonstrating beyond in vitro and animal models, that in platelets of young human individuals, without evident clinical manifestations of T2DM, the interconnection of ER stress and IR is present through the molecular activation of JNK. Remarkably, WtHR showed significant independent associations with HDL-C, the TG/HDL-C ratio, fasting insulin, HOMA-IR, pJNK, and SERCA expression (Supplementary Table 1). Overall, these findings indicate that central adiposity is strongly linked to dyslipidemia, IR, and ER stress–related molecular alterations in young adults, even after accounting for sex differences, as demonstrated after linear regression analyses (Supplementary Table 2). At this stage, the observed platelet changes were not associated with early metabolic liver involvement, as indicated by the assessment of C-reactive protein and hepatic enzymes ALT, AST, and γ-GGT, with significant changes observed only in γ-GGT levels (Supplementary Figure 1), supporting the notion that the participants were apparently healthy young individuals in whom metabolic alterations are only beginning to emerge.
Nevertheless, this study has some limitations, including a small sample size and a cross-sectional design, which prevent a causal relationship between high WtHR and metabolic disturbances from being fully established. Furthermore, inflammatory markers and dietary or physical activity parameters—factors that could influence IR and ER stress—were not assessed. Nevertheless, the results provide valuable evidence for the usefulness of platelets as an accessible model for detecting early metabolic alterations in young adults.
Finally, it is worth noting that these pathophysiological alterations are entirely reversible. However, common lifestyle habits among young people, such as consuming high-fat and high-sugar diets, being physically inactive, and experiencing socioeconomic deprivation, all contribute to ER stress and IR through inflammation, oxidative damage, and lipid toxicity. Therefore, it is essential to create conditions that encourage healthier lifestyles and help prevent overweight and obesity. Moreover, our results open up possibilities for intervening through pharmacological ER stress modulators or JNK/PERK inhibitors as potential treatments, in addition to diet and physical activity, as long as effectiveness and health security are assured.
In conclusion, this study demonstrated for the first time that young people with a high WtHR exhibit activation of the ER stress and UPR pathways in platelets, along with IR and an atherogenic lipid profile, despite no medical manifestations of T2DM. These findings highlight the relevance of WtHR as an early marker of metabolic dysfunction and suggest a possible compensatory role for SERCA protein overexpression in response to cellular stress. Altogether, our results provide novel evidence on the molecular mechanisms linking central adiposity to cardiometabolic risk in young populations, demonstrating the importance of preventive strategies based on healthy lifestyles and early detection.
| 1. | Młynarska E, Czarnik W, Dzieża N, Jędraszak W, Majchrowicz G, Prusinowski F, Stabrawa M, Rysz J, Franczyk B. Type 2 Diabetes Mellitus: New Pathogenetic Mechanisms, Treatment and the Most Important Complications. Int J Mol Sci. 2025;26:1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 142] [Article Influence: 142.0] [Reference Citation Analysis (1)] |
| 2. | American Diabetes Association Professional Practice Committee. 2. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes-2025. Diabetes Care. 2025;48:S27-S49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 727] [Cited by in RCA: 654] [Article Influence: 654.0] [Reference Citation Analysis (1)] |
| 3. | Parcha V, Heindl B, Kalra R, Li P, Gower B, Arora G, Arora P. Insulin Resistance and Cardiometabolic Risk Profile Among Nondiabetic American Young Adults: Insights From NHANES. J Clin Endocrinol Metab. 2022;107:e25-e37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 67] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 4. | Ashwell M, Gunn P, Gibson S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: systematic review and meta-analysis. Obes Rev. 2012;13:275-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1439] [Cited by in RCA: 1427] [Article Influence: 101.9] [Reference Citation Analysis (3)] |
| 5. | Feng Q, Bešević J, Conroy M, Omiyale W, Woodward M, Lacey B, Allen N. Waist-to-height ratio and body fat percentage as risk factors for ischemic cardiovascular disease: a prospective cohort study from UK Biobank. Am J Clin Nutr. 2024;119:1386-1396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 6. | Ashwell M, Gibson S. Waist-to-height ratio as an indicator of 'early health risk': simpler and more predictive than using a 'matrix' based on BMI and waist circumference. BMJ Open. 2016;6:e010159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 463] [Cited by in RCA: 371] [Article Influence: 37.1] [Reference Citation Analysis (0)] |
| 7. | Gibson S, Ashwell M. A simple cut-off for waist-to-height ratio (0·5) can act as an indicator for cardiometabolic risk: recent data from adults in the Health Survey for England. Br J Nutr. 2020;123:681-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 62] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 8. | Boden G, Cheung P, Kresge K, Homko C, Powers B, Ferrer L. Insulin resistance is associated with diminished endoplasmic reticulum stress responses in adipose tissue of healthy and diabetic subjects. Diabetes. 2014;63:2977-2983. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 9. |
Vázquez-Jiménez JG, RouraA, Jiménez-Mena LR, Olivares-Reyes JA, Role of free fatty acids on insulin resistance.
|
| 10. | Ozcan U, Yilmaz E, Ozcan L, Furuhashi M, Vaillancourt E, Smith RO, Görgün CZ, Hotamisligil GS. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science. 2006;313:1137-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2054] [Cited by in RCA: 1968] [Article Influence: 98.4] [Reference Citation Analysis (5)] |
| 11. | Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, Tuncman G, Görgün C, Glimcher LH, Hotamisligil GS. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science. 2004;306:457-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3156] [Cited by in RCA: 2874] [Article Influence: 130.6] [Reference Citation Analysis (3)] |
| 12. | Gustavo Vazquez-Jimenez J, Chavez-Reyes J, Romero-Garcia T, Zarain-Herzberg A, Valdes-Flores J, Manuel Galindo-Rosales J, Rueda A, Guerrero-Hernandez A, Olivares-Reyes JA. Palmitic acid but not palmitoleic acid induces insulin resistance in a human endothelial cell line by decreasing SERCA pump expression. Cell Signal. 2016;28:53-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 13. | Boden G, Duan X, Homko C, Molina EJ, Song W, Perez O, Cheung P, Merali S. Increase in endoplasmic reticulum stress-related proteins and genes in adipose tissue of obese, insulin-resistant individuals. Diabetes. 2008;57:2438-2444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 367] [Article Influence: 20.4] [Reference Citation Analysis (1)] |
| 14. | Villalobos-Labra R, Subiabre M, Toledo F, Pardo F, Sobrevia L. Endoplasmic reticulum stress and development of insulin resistance in adipose, skeletal, liver, and foetoplacental tissue in diabesity. Mol Aspects Med. 2019;66:49-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 99] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 15. | Kelem A, Adane T, Shiferaw E. Insulin Resistance-Induced Platelet Hyperactivity and a Potential Biomarker Role of Platelet Parameters: A Narrative Review. Diabetes Metab Syndr Obes. 2023;16:2843-2853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 16. | Faul F, Erdfelder E, Buchner A, Lang AG. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods. 2009;41:1149-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29582] [Cited by in RCA: 18596] [Article Influence: 1093.9] [Reference Citation Analysis (0)] |
| 17. | Burzynski LC, Pugh N, Clarke MCH. Platelet Isolation and Activation Assays. Bio Protoc. 2019;9:e3405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | Galindo-Hernandez O, Machado-Contreras JR, Martinez-Corella R, Romero-Garcia T, Vazquez-Jimenez JG. Inverse correlation between levels of glycated haemoglobin and expression levels of SERCA protein in Mexican patients with type 2 diabetes mellitus. Arch Med Sci. 2020;16:1226-1228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Jakubiak GK, Badicu G, Surma S, Waluga-Kozłowska E, Chwalba A, Pawlas N. The Visceral Adiposity Index and Its Usefulness in the Prediction of Cardiometabolic Disorders. Nutrients. 2025;17:2374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 20. | Park SW, Zhou Y, Lee J, Lee J, Ozcan U. Sarco(endo)plasmic reticulum Ca2+-ATPase 2b is a major regulator of endoplasmic reticulum stress and glucose homeostasis in obesity. Proc Natl Acad Sci U S A. 2010;107:19320-19325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 199] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 21. | Vazquez-Jimenez JG, Corpus-Navarro MS, Rodriguez-Chavez JM, Jaramillo-Ramirez HJ, Hernandez-Aranda J, Galindo-Hernandez O, Machado-Contreras JR, Trejo-Trejo M, Guerrero-Hernandez A, Olivares-Reyes JA. The Increased Expression of Regulator of G-Protein Signaling 2 (RGS2) Inhibits Insulin-Induced Akt Phosphorylation and Is Associated with Uncontrolled Glycemia in Patients with Type 2 Diabetes. Metabolites. 2021;11:91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 22. | Craveiro V, Ramos E, Araújo J. Metabolically healthy overweight in young adulthood: is it a matter of duration and degree of overweight? Nutr Metab Cardiovasc Dis. 2021;31:455-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Bray GA, Jablonski KA, Fujimoto WY, Barrett-Connor E, Haffner S, Hanson RL, Hill JO, Hubbard V, Kriska A, Stamm E, Pi-Sunyer FX; Diabetes Prevention Program Research Group. Relation of central adiposity and body mass index to the development of diabetes in the Diabetes Prevention Program. Am J Clin Nutr. 2008;87:1212-1218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 180] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 24. | Li C, Ford ES, Meng YX, Mokdad AH, Reaven GM. Does the association of the triglyceride to high-density lipoprotein cholesterol ratio with fasting serum insulin differ by race/ethnicity? Cardiovasc Diabetol. 2008;7:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 162] [Article Influence: 9.0] [Reference Citation Analysis (4)] |
| 25. | Sollano Trejo OA, Romero García T, Jaramillo Ramírez HJ, Trejo Trejo M, Leija Montoya AG, Vázquez Jiménez JG. Association between visceral fat and components of metabolic syndrome in young Mexicans: a preliminary study. Nutr Clín Diet Hosp. 2025;45. [DOI] [Full Text] |
| 26. | Murguía-Romero M, Jiménez-Flores JR, Méndez-Cruz AR, Sigrist-Flores SC, Villalobos-Molina R. Insulin and HOMA-IR in Healthy Young Mexicans: A Cut-off Points Proposal. Intern Med. 2014;S6:001. [DOI] [Full Text] |
| 27. | Zhou H, Liu R. ER stress and hepatic lipid metabolism. Front Genet. 2014;5:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 91] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 28. | Randriamboavonjy V, Pistrosch F, Bölck B, Schwinger RH, Dixit M, Badenhoop K, Cohen RA, Busse R, Fleming I. Platelet sarcoplasmic endoplasmic reticulum Ca2+-ATPase and mu-calpain activity are altered in type 2 diabetes mellitus and restored by rosiglitazone. Circulation. 2008;117:52-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 79] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 29. | Romero-García T, Vázquez-Jiménez JG, Sánchez-Hernández R, Olivares-Reyes JA, Rueda A. Insulin resistance, Ca(2+) signaling alterations and vascular dysfunction in prediabetes and metabolic syndrome. Front Physiol. 2025;16:1535153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |