Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.116210
Revised: December 22, 2025
Accepted: January 7, 2026
Published online: May 15, 2026
Processing time: 187 Days and 7.6 Hours
Diabetic foot ulcers (DFUs) are a common complication of diabetes. The injection of mesenchymal stem cells (MSCs) has emerged as a potential therapeutic strategy for DFUs, and several related clinical cohort studies have been conducted. Our previous research found that MSCs derived from umbilical cord and bone mar
Core Tip: Although mesenchymal stem cells (MSCs) can be isolated and expanded from a variety of tissues, their regenerative efficacy for diabetic foot ulcers (DFUs) exhibits significant source-dependent variation, and the specific regulatory mechanisms involved remain incompletely understood. Cao et al demonstrated that subcutaneous injection of 5 × 105 adipose-derived stem cells (ADSCs) into the foot yielded the most effective wound healing outcomes for DFUs. Further angiogenesis experiments revealed that ADSCs promote blood vessel formation by activating the PI3K-AKT pathway to enhance VEGF secretion. Additionally, it was confirmed that ADSCs can suppress the overactivated Notch signaling pathway, thereby reducing local inflammatory responses and improving collagen deposition. This study not only elucidates the underlying molecular mechanisms by which ADSCs accelerate DFU healing but also provides critical evidence to support the clinical application of MSCs.
- Citation: Wan XX, Yan WT, He WH, Xiong K. Synergistic mechanism of adipose-derived stem cells in healing diabetic foot ulcers. World J Diabetes 2026; 17(5): 116210
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/116210.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.116210
This editorial refers to "Optimizing adipose-derived stem cell therapy for diabetic foot ulcers" by Cao et al, 2025; https://doi.org/10.4239/wjd.v16.i11.109859.
Diabetic foot ulcers (DFUs) are a common complication of diabetes with a high incidence rate[1,2]. The development of DFUs involves a complex interplay of multiple pathological conditions, among which lower extremity arterial disease and diabetic peripheral neuropathy are widely recognized as the primary causative factors[3,4]. Approximately 19%-34% of diabetic patients may develop DFUs in their lifetime[5]. Recent meta-analyses estimate that the proportion of diabetic patients at risk of foot ulcers is as high as 53.2%[6]. The prognosis for DFUs is poor, with about half of the cases complicated by infection, an annual amputation rate of up to 17%, and a mortality rate as high as 15%, posing a serious threat to patients' health[7,8]. The key treatment for DFU mainly includes management of infection, debridement of the wound, revascularization. Notably, the healing rate of DFUs within the first four weeks significantly influences their overall prognosis[9]. Therefore, the core objective of treatment is to accelerate wound healing. Various techniques, such as growth factors, nano-silver, and negative pressure wound therapy, have been applied to promote healing in diabetic foot wounds[10-15]. However, these methods often show limited efficacy for ulcers with higher Wagner grades. Stem cell therapy, particularly approaches based on mesenchymal stem cells (MSCs), has been widely adopted in the treatment of DFUs with demonstrated efficacy[16-18]. This therapeutic strategy can effectively accelerate wound healing and significantly reduce the risk of amputation.
MSCs have been reported to be isolated from a variety of sources, including the umbilical cord, placenta, Wharton's jelly, adipose tissue, bone marrow, and dental pulp[19-23]. MSC are characterized by the positive expression of CD105, CD73, and CD90, and the absence of expression for CD45, CD34, CD14 or CD11b, and CD19[24]. Coupled with their wide availability and accessibility, MSCs hold considerable promise for broader clinical translation[25,26]. Recently, a number of clinical cohort studies have investigated the application of MSCs in foot ulcer management, consistently reporting notable therapeutic outcomes with minimal adverse effects[27,28].
MSCs offer multiple advantages in DFU treatment. First, they secrete a variety of growth factors and cytokines that stimulate angiogenesis, improve local microcirculation, and promote granulation tissue formation and epithelial migration, thereby facilitating wound closure[29,30]. Second, MSCs modulate the local immune response by reducing pro-inflammatory cytokine levels and alleviating systemic inflammation, thereby creating a favorable microenvironment for tissue regeneration[31-33]. Moreover, given that diabetic patients often suffer from lower limb vascular complications leading to foot ischemia, MSCs not only possess the potential to differentiate into vascular endothelial cells but also induce neovascularization through paracrine mechanisms, fundamentally enhancing perfusion—a critical factor for successful ulcer healing[34,35]. In our previous studies, we have conducted extensive research on MSCs and demonstrated that those derived from umbilical cord and bone marrow exhibit robust regenerative capabilities. They promote angiogenesis via the c-Jun/VEGF pathway and significantly accelerate wound healing[36-38]. In patients with refractory foot ulcers that do not respond to conventional treatments, the combination of MSCs and endothelial progenitor cells (EPCs) has promoted wound healing in multiple cases and prevented amputations[39].
However, the efficacy of MSCs in promoting wound healing and vascular repair varies depending on their tissue source[40]. For instance, umbilical cord-derived MSCs (UCMSCs) are generally recognized for their strong proliferative and differentiation capacity[41], yet their clinical use may be limited by ethical concerns. In contrast, autologous MSCs (e.g., bone marrow-derived) avoid these ethical issues. However, their application is characterized by a prolonged preparation time, and the cell quality is donor-dependent, being subject to variation based on the patient's age and overall health[42]. Therefore, the selection of an optimal MSC source should be based on a comprehensive evaluation of factors such as cell availability, quantity, quality, regulatory guidelines, and treatment strategy.
Bone marrow-derived MSCs (BMSCs) represent the predominant cell source in autologous transplantation for DFUs, largely due to a harvesting procedure that circumvents prolonged enzymatic digestion, thereby minimizing contamination risks and enhancing theoretical safety[43]. Clinical evidence confirms that BMSC implantation markedly enhances a spectrum of clinical endpoints in DFU, including perfusion metrics (e.g., ankle-brachial index, transcutaneous oxygen pressure), angiogenesis, walking capacity, and pain scores, while accelerating ulcer healing and reducing amputation rates without documented adverse events[44,45]. While intramuscular delivery is a well-established route for addressing critical limb ischemia, alternative strategies such as localized topical application or injection around the wound periphery are emerging as potentially superior methods for facilitating complete wound closure[46,47].
UCMSCs exhibit a markedly enhanced proliferative potential under in vitro culture conditions and have shown considerable promise in promoting the repair of diabetic foot wounds[41]. A demonstrated advantage of UCMSCs is their potent stimulation of keratinocyte proliferation and migration. This enhanced functionality indicates that UCMSCs may be more effective in accelerating the critical process of wound re-epithelialization[48]. Evidence from various transplantation studies and clinical cohorts indicates that the administration of UCMSCs can substantially expedite the healing process of DFUs and contribute to the restoration of vascular function. In comparative clinical assessments, a single injection of UCMSCs led to significantly improved wound closure outcomes relative to placebo controls, with no associated adverse events reported, underscoring their considerable therapeutic promise[49]. Nonetheless, as UCMSCs are sourced from postnatal umbilical cord tissue and are generally applied in an allogeneic setting, their use is accompanied by specific ethical considerations. These concerns present ongoing challenges for the broader integration of UCMSCs into routine clinical practice.
Adipose-derived stem cells (ADSCs) are highly abundant and readily obtainable from adipose tissue, presenting a practical alternative to BMSCs in both therapeutic and research contexts[50,51]. These adult stem cells demonstrate robust self-renewal capability and multipotent differentiation potential[52,53]. Their reduced immunogenic potential further supports broad clinical application. ADSCs are distinguished from BMSCs by their lack of CD106 expression and positivity for CD36. Furthermore, they do not express SSEA-4, a marker characteristic of UCMSCs, further delineating their unique immunophenotype[54,55]. In the context of cell-based therapies for atherosclerosis, studies indicate that ADSCs hold distinct advantages over BMSCs. Specifically, ADSCs display enhanced anti-inflammatory effects, improved phagocytic function, strengthened resistance to apoptosis, and greater cell survival rates[56]. Research by Noël et al[57] utilizing proteomic and transcriptomic approaches revealed differential expression of 18% of proteins and 13.2% of genes between BMSCs and ADSCs under standard culture conditions. Notably, ADSCs were more effective in inducing a more substantial upregulation of IL-10 production in dendritic cells, indicating their potential for more efficient regulation of chronic inflammatory processes in wound healing. Furthermore, comparative investigations[58] have illustrated that ADSCs possess superior angiogenic potential through the activation of EPCs relative to both BMSCs and UCMSCs, implying their enhanced capacity to stimulate new blood vessel formation in chronic ischemic wounds and facilitate tissue repair.
The specific surface markers of MSCs from different sources remain a subject of debate, and their biological functions and immunogenicity exhibit notable variations. Moreover, potential allergens may be introduced during the processes of isolation, extraction, and in vitro culture, all of which contribute to the complexity of MSC clinical application (Table 1). Given the currently limited clinical evidence and research on alternative MSC sources—particularly umbilical cord blood, peripheral blood, and dental pulp—a comprehensive assessment suggests that ADSCs offer a superior cellular therapeutic strategy for the management of DFUs.
| Type | Source | Pros | Cons | Recommended dose |
| BMSCs | Bone marrow | Theoretical safety; high differentiation effects | Low-yield invasive bone marrow aspiration in diabetics | Subcutaneous injection 9 × 107 cells[53] |
| ADSCs | Adipose tissue | Abundant and easily extracted from adipose tissue; multipotent differentiation potential; display enhanced anti-inflammatory effects | Contamination risk from extraction | Subcutaneous injection 5 × 105 cells[67] |
| UCMSCs | Human umbilical cord | Strong proliferative capacity and low immunogenicity | Ethical issues | Intravenous administrations 2 × 105 cells/kg with an upper limit of 1 × 107 cells[59] |
MSCs demonstrate therapeutic potential in both diabetic wound healing and vascular complications through their multifaceted regulatory mechanisms. During wound repair, MSCs exert dual-phase regulation: In the inflammatory phase, they facilitate inflammation resolution by promoting macrophage polarization from the pro-inflammatory M1 phenotype toward the reparative M2 phenotype; in the proliferative phase, they primarily function through paracrine signaling, upregulating key growth factors including VEGF, TGF-β1, and PDGF to enhance epithelial regeneration, granulation tissue formation, and angiogenesis. In diabetic vascular pathologies, MSCs contribute to vascular repair by improving endothelial function and stimulating collateral vessel formation[59-62].
Current research efforts are focused on optimizing MSC-based therapeutic strategies for DFUs. These include combinatorial approaches utilizing MSC-hydrocolloid dressing systems and preconditioning techniques with growth factors or inflammatory cytokines to enhance cellular treatment potency[63-65]. Additionally, the development of engineered MSC-derived exosomes represents a promising cell-free alternative for targeted therapy[66]. Nevertheless, the precise molecular mechanisms underlying MSC-mediated regulation of growth factor secretion and macrophage polarization remain incompletely elucidated. Most existing studies, including our own investigations, have primarily documented phenotypic changes in growth factor profiles, while the intrinsic regulatory networks—including the potential involvement of long non-coding RNAs and metabolic reprogramming—governing MSC secretory behavior in the diabetic microenvironment require further systematic exploration.
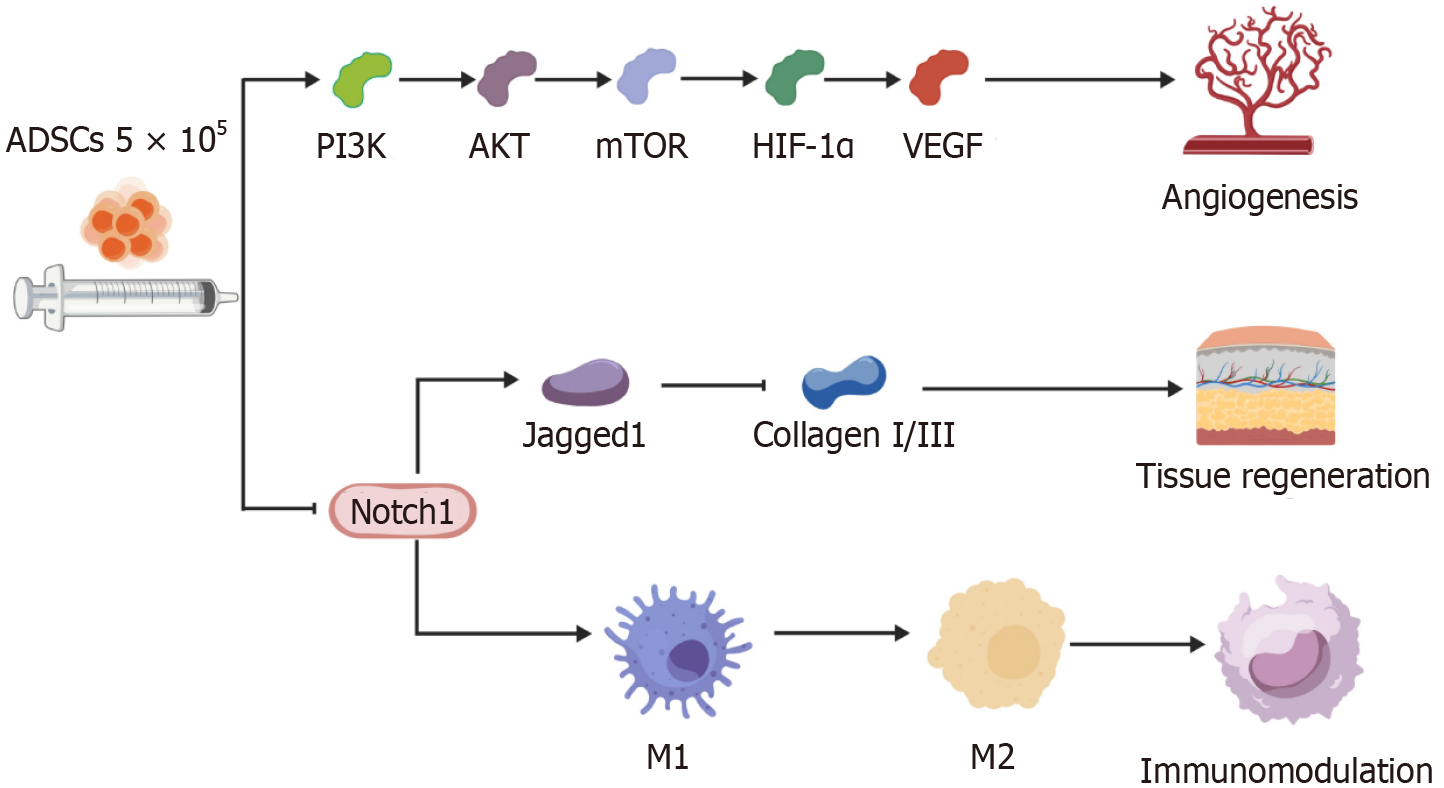
In the article published by Cao et al[67] in the World Journal of Diabetes, the authors systematically elucidated the multifaceted therapeutic mechanisms of ADSCs in a DFU model. The study demonstrated that ADSCs not only accelerate wound closure, promote angiogenesis, modulate inflammatory responses, and facilitate tissue regeneration, but also established a clear dependence of therapeutic efficacy on cell dosage and administration route. Specifically, subcutaneous injection of 5 × 105 ADSCs, particularly when administered in the foot, was identified as the optimal protocol, which significantly prolonged cellular retention and yielded superior treatment outcomes. At the mechanistic level, in vitro experiments revealed that ADSCs promote angiogenesis by activating the PI3K signaling pathway to enhance VEGF secretion, while concurrently modulating the Notch1 pathway to suppress inflammation and support tissue repair. Using DiD labeling, the study visually documented the in vivo distribution and persistence of ADSCs, providing direct evidence for the superiority of local subcutaneous delivery over intramuscular injection by better maintaining MSC viability and function.
As summarized in the schematic diagram illustrating the synergistic mechanism of ADSCs (Figure 1), the in-depth analysis showed that advanced glycation end products (AGEs) suppress the expression of PI3K, AKT, mTOR, and HIF-1α in HUVECs, thereby inhibiting VEGF production—effects that were reversed by ADSC treatment. The application of a PI3K inhibitor abolished the pro-angiogenic capacity of ADSCs under AGE conditions, impairing HUVEC tube formation and migration. Corresponding in vivo experiments in mice verified that ADSCs promote AKT expression, confirming their role in enhancing VEGF production via the PI3K-AKT pathway. Additionally, the results revealed ADSCs significantly enhanced collagen thickness in the wounds by modulating the ratio of collagen I to collagen III through the inhibition of Notch1, which plays a critical role in tissue regeneration[68,69]. On the immunomodulatory front, ADSCs inhibited Notch1, which effectively suppressed M1 macrophage polarization and promoted a shift toward the M2 phenotype. This effect was attributed to the ability of ADSCs to reverse AGE-induced overactivation of the Notch1 signaling pathway, leading to the upregulation of the M2 marker.
In summary, this research not only confirms the pro-angiogenic role of ADSCs but also systematically unveils their dual mechanism in regulating both inflammatory response and tissue regeneration through the PI3K/AKT/VEGF and Notch1 signaling pathways, providing a critical experimental foundation for optimizing ADSC-based therapeutic strategies.
DFUs represent a significant and persistent clinical challenge, driven by complex pathophysiological impairments that conventional therapies often fail to address adequately. The pursuit of innovative treatment strategies capable of targeting multiple facets of the dysfunctional healing process is thus imperative. Among these, MSC-based therapies have garnered considerable attention as a promising regenerative approach, owing to their tripartite capacities for immunomodulation, angiogenesis, and tissue repair. When comparing various MSC sources, ADSCs present distinct practical and therapeutic advantages, including minimally invasive harvesting, abundant tissue availability, robust ex vivo expansion potential, and potent paracrine activity. Looking forward, the successful translation of ADSC-based treatments into routine clinical practice for DFUs will hinge on several critical developments. These include the establishment of standardized protocols for cell isolation, characterization, and delivery; a deeper mechanistic understanding of ADSC behavior within the diabetic wound microenvironment; and the rational design of combinatorial strategies, such as incorporating ADSCs into bioactive scaffolds or pre-conditioning cells to enhance their therapeutic potency. As ongoing research continues to delineate the intricate signaling networks and epigenetic mechanisms that govern ADSC function, the prospect of developing a robust, efficacious, and clinically viable cell-based therapy for DFUs becomes increasingly attainable.
| 1. | Su Y, Fu X, Huang Y; and Wound Repair Professional Committee of Chinese Medical Doctor Association. Consensus statement on the clinical application of extracorporeal shock wave therapy for diabetic foot ulcers (2025 Edition). Int J Surg. 2026;112:71-83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 2. | Cavanagh PR, Lipsky BA, Bradbury AW, Botek G. Treatment for diabetic foot ulcers. Lancet. 2005;366:1725-1735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 407] [Cited by in RCA: 314] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 3. | Wu Y, Dong D, Zhu L, Luo Z, Liu Y, Xie X. Interpretable machine learning models for detecting peripheral neuropathy and lower extremity arterial disease in diabetics: an analysis of critical shared and unique risk factors. BMC Med Inform Decis Mak. 2024;24:200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 4. | Sinacore DR, Jones MA, Kline PW. Major adverse foot events and functional mobility deficits associated with diabetic neuropathy and nephropathy. Metab Target Organ Damage. 2024;4:15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 5. | Armstrong DG, Boulton AJM, Bus SA. Diabetic Foot Ulcers and Their Recurrence. N Engl J Med. 2017;376:2367-2375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3392] [Cited by in RCA: 2719] [Article Influence: 302.1] [Reference Citation Analysis (5)] |
| 6. | Maldonado-Valer T, Pareja-Mujica LF, Corcuera-Ciudad R, Terry-Escalante FA, Chevarría-Arriaga MJ, Vasquez-Hassinger T, Yovera-Aldana M. Prevalence of diabetic foot at risk of ulcer development and its components stratification according to the international working group on the diabetic foot (IWGDF): A systematic review with metanalysis. PLoS One. 2023;18:e0284054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Wang R, Gu S, Kim YH, Lee A, Lin H, Jiang D. Diabetic Wound Repair: From Mechanism to Therapeutic Opportunities. MedComm (2020). 2025;6:e70406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (2)] |
| 8. | Ndosi M, Wright-Hughes A, Brown S, Backhouse M, Lipsky BA, Bhogal M, Reynolds C, Vowden P, Jude EB, Nixon J, Nelson EA. Prognosis of the infected diabetic foot ulcer: a 12-month prospective observational study. Diabet Med. 2018;35:78-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 181] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 9. | Lavery LA, Barnes SA, Keith MS, Seaman JW Jr, Armstrong DG. Prediction of healing for postoperative diabetic foot wounds based on early wound area progression. Diabetes Care. 2008;31:26-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 96] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 10. | Zheng SY, Wan XX, Kambey PA, Luo Y, Hu XM, Liu YF, Shan JQ, Chen YW, Xiong K. Therapeutic role of growth factors in treating diabetic wound. World J Diabetes. 2023;14:364-395. [PubMed] [DOI] [Full Text] |
| 11. | Li X, Yi M, Song Z, Ni T, Tu L, Yu M, Zhang L, Shi J, Gao W, Zhang Q, Yan W. A calcitonin gene-related peptide co-crosslinked hydrogel promotes diabetic wound healing by regulating M2 macrophage polarization and angiogenesis. Acta Biomater. 2025;196:109-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 12. | Piccin A, Di Pierro AM, Canzian L, Primerano M, Corvetta D, Negri G, Mazzoleni G, Gastl G, Steurer M, Gentilini I, Eisendle K, Fontanella F. Platelet gel: a new therapeutic tool with great potential. Blood Transfus. 2017;15:333-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 53] [Reference Citation Analysis (0)] |
| 13. | Zheng Q, Chen C, Liu Y, Gao J, Li L, Yin C, Yuan X. Metal Nanoparticles: Advanced and Promising Technology in Diabetic Wound Therapy. Int J Nanomedicine. 2024;19:965-992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 14. | Blume PA, Walters J, Payne W, Ayala J, Lantis J. Comparison of negative pressure wound therapy using vacuum-assisted closure with advanced moist wound therapy in the treatment of diabetic foot ulcers: a multicenter randomized controlled trial. Diabetes Care. 2008;31:631-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 361] [Article Influence: 20.1] [Reference Citation Analysis (3)] |
| 15. | Schaper NC, van Netten JJ, Apelqvist J, Bus SA, Hinchliffe RJ, Lipsky BA; IWGDF Editorial Board. Practical Guidelines on the prevention and management of diabetic foot disease (IWGDF 2019 update). Diabetes Metab Res Rev. 2020;36 Suppl 1:e3266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 453] [Article Influence: 75.5] [Reference Citation Analysis (0)] |
| 16. | Wang J, Liang YJ, Pan X. Advances in the Role of Stem Cell-Derived Exosomes in Diabetic Foot Wound Healing. Diabetes Metab Syndr Obes. 2025;18:2767-2781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 17. | Raghav A, Tripathi P, Mishra BK, Jeong GB, Banday S, Gautam KA, Mateen QN, Singh P, Singh M, Singla A, Ahmad J. Mesenchymal Stromal Cell-Derived Tailored Exosomes Treat Bacteria-Associated Diabetes Foot Ulcers: A Customized Approach From Bench to Bed. Front Microbiol. 2021;12:712588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 18. | Dias Â, Ferreira G, Vilaça M, Pereira MG. Quality of Life in Patients with Diabetic Foot Ulcers: A Cross-sectional Study. Adv Skin Wound Care. 2022;35:661-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 19. | Yang R, Yang S, Zhao J, Hu X, Chen X, Wang J, Xie J, Xiong K. Progress in studies of epidermal stem cells and their application in skin tissue engineering. Stem Cell Res Ther. 2020;11:303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Alpargu BT, Ozdamar S, Gonen ZB, Kara A, Salkin H. Effect of TGF-β1 transfected dental pulp derived mesenchymal stem cells on ın vivo cartilage regeneration. Iran J Basic Med Sci. 2025;28:1505-1515. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 21. | Akhtar M, Nashwan AJ. Evaluating Wharton's jelly-derived stem cell therapy in autism: Insights from a case study. World J Methodol. 2025;15:100074. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (72)] |
| 22. | Zhong F, Zheng W, Peng M. Ginkgolide B Combined With Umbilical Cord Mesenchymal Stem Cell-Derived Exosomes Alleviate Diabetic Nephropathy by Modulating the Nuclear Factor Erythroid-2-Related Factor 2/Heme Oxygenase 1 Pathway. Chem Biol Drug Des. 2025;106:e70194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 23. | Cheng X, Zhang M, Xie Y, Xu Y, Du R, Wu B, Guan Z, Wang W, Sun W, Xu T, Zhu S, Wu L, Wang X, Shi H, Sun B, Zhang Y. Bone marrow-derived mesenchymal stem cells accelerate angiogenesis in pregnant experimentally induced deep venous thrombosis rat model via up-regulation of pro-angiogenic secretogranin II. Int Immunopharmacol. 2023;118:110025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 24. | Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14328] [Cited by in RCA: 12735] [Article Influence: 636.8] [Reference Citation Analysis (4)] |
| 25. | Jing Y, Cai Y, Li Q, Zheng Z, Yang H, Yu Y, Zhang C. Empagliflozin-pretreated BMSC exosomes attenuate myocardial ischemia-reperfusion injury by enhancing atad3a/pink1-dependent mitophagy. Stem Cell Res Ther. 2025;16:595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 26. | Costa-Ferro ZSM, da Silva KN, Santana Cunha R, de Oliveira GB, Dias Vieira Leite PA, Aranha Rossi E, de Freitas Souza BS, Macedo Rocco PR. Mesenchymal stromal cell-derived extracellular vesicles as a next-generation therapy for ARDS: mechanisms, advances, and future directions. Expert Rev Respir Med. 2025;1-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 27. | Sadyah NAC, Heri-Nugroho, Putra A, Riwanto I. Secretome of Human MSC Gel Improves DFU Healing through NF-?B p50 and CD163 mRNA Expression. Pak J Biol Sci. 2025;28:151-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Kishta MS, Hafez AM, Hydara T, Hamed Z, Bahr MM, Shamaa AA, Abdallah AN. The transforming role of wharton's jelly mesenchymal stem cell-derived exosomes for diabetic foot ulcer healing: a randomized controlled clinical trial. Stem Cell Res Ther. 2025;16:559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 29. | Wei L, Xu Y, Zhang L, Yang L, Zhao RC, Zhao D. Mesenchymal Stem Cells Promote Wound Healing and Effects on Expression of Matrix Metalloproteinases-8 and 9 in the Wound Tissue of Diabetic Rats. Stem Cells Dev. 2023;32:25-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 30. | Wang Z, Xu H, Xue B, Liu L, Tang Y, Wang Z, Yao K. MSC-derived exosomal circMYO9B accelerates diabetic wound healing by promoting angiogenesis through the hnRNPU/CBL/KDM1A/VEGFA axis. Commun Biol. 2024;7:1700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (4)] |
| 31. | Toh WS, Zhang B, Lai RC, Lim SK. Immune regulatory targets of mesenchymal stromal cell exosomes/small extracellular vesicles in tissue regeneration. Cytotherapy. 2018;20:1419-1426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 32. | Li M, Yin H, Yan Z, Li H, Wu J, Wang Y, Wei F, Tian G, Ning C, Li H, Gao C, Fu L, Jiang S, Chen M, Sui X, Liu S, Chen Z, Guo Q. The immune microenvironment in cartilage injury and repair. Acta Biomater. 2022;140:23-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 279] [Article Influence: 69.8] [Reference Citation Analysis (0)] |
| 33. | Aslam S, Khan I, Jameel F, Zaidi MB, Salim A. Umbilical cord-derived mesenchymal stem cells preconditioned with isorhamnetin: potential therapy for burn wounds. World J Stem Cells. 2020;12:1652-1666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 34. | Wu S, Zhou Z, Li Y, Jiang J. Advancements in diabetic foot ulcer research: Focus on mesenchymal stem cells and their exosomes. Heliyon. 2024;10:e37031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 35. | Verdi J, Shirian S, Saleh M, Khadem Haghighian H, Kavianpour M. Mesenchymal Stem Cells Regenerate Diabetic Foot Ulcers: A Review Article. World J Plast Surg. 2022;11:12-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 36. | Zhao L, Guo Z, Chen K, Yang W, Wan X, Zeng P, He H, Luo Y, Xiao Q, Mo Z. Combined Transplantation of Mesenchymal Stem Cells and Endothelial Colony-Forming Cells Accelerates Refractory Diabetic Foot Ulcer Healing. Stem Cells Int. 2020;2020:8863649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 37. | Yue C, Guo Z, Luo Y, Yuan J, Wan X, Mo Z. c-Jun Overexpression Accelerates Wound Healing in Diabetic Rats by Human Umbilical Cord-Derived Mesenchymal Stem Cells. Stem Cells Int. 2020;2020:7430968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 38. | Wang L, Wang F, Zhao L, Yang W, Wan X, Yue C, Mo Z. Mesenchymal Stem Cells Coated by the Extracellular Matrix Promote Wound Healing in Diabetic Rats. Stem Cells Int. 2019;2019:9564869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 39. | Luo Y, Liang F, Wan X, Liu S, Fu L, Mo J, Meng X, Mo Z. Hyaluronic Acid Facilitates Angiogenesis of Endothelial Colony Forming Cell Combining With Mesenchymal Stem Cell via CD44/ MicroRNA-139-5p Pathway. Front Bioeng Biotechnol. 2022;10:794037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 40. | Cao Y, Yan J, Liu H. [Clinical research progress of mesenchymal stem cells in treatment of chronic wounds]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2021;35:496-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 41. | Hashemi SS, Mohammadi AA, Kabiri H, Hashempoor MR, Mahmoodi M, Amini M, Mehrabani D. The healing effect of Wharton's jelly stem cells seeded on biological scaffold in chronic skin ulcers: A randomized clinical trial. J Cosmet Dermatol. 2019;18:1961-1967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 42. | Sethe S, Scutt A, Stolzing A. Aging of mesenchymal stem cells. Ageing Res Rev. 2006;5:91-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 508] [Cited by in RCA: 464] [Article Influence: 23.2] [Reference Citation Analysis (1)] |
| 43. | Harris DT. Banking of Adipose- and Cord Tissue-Derived Stem Cells: Technical and Regulatory Issues. Adv Exp Med Biol. 2016;951:147-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Kirana S, Stratmann B, Prante C, Prohaska W, Koerperich H, Lammers D, Gastens MH, Quast T, Negrean M, Stirban OA, Nandrean SG, Götting C, Minartz P, Kleesiek K, Tschoepe D. Autologous stem cell therapy in the treatment of limb ischaemia induced chronic tissue ulcers of diabetic foot patients. Int J Clin Pract. 2012;66:384-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 112] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 45. | Dash NR, Dash SN, Routray P, Mohapatra S, Mohapatra PC. Targeting nonhealing ulcers of lower extremity in human through autologous bone marrow-derived mesenchymal stem cells. Rejuvenation Res. 2009;12:359-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 232] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 46. | Zhang Q, Wang T, Wu X, Wang Y, Wu X, Zhou M, Li Z, Liu B, Zheng F, Sun X, Liu T. Thrombospondin-4 (TSP4) gene-modified bone marrow stromal cells (BMSCs) promote the effect of therapeutic angiogenesis in critical limb ischemia (CLI) of diabetic rats. Biochem Biophys Res Commun. 2020;532:231-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 47. | Humpert PM, Bärtsch U, Konrade I, Hammes HP, Morcos M, Kasper M, Bierhaus A, Nawroth PP. Locally applied mononuclear bone marrow cells restore angiogenesis and promote wound healing in a type 2 diabetic patient. Exp Clin Endocrinol Diabetes. 2005;113:538-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 48. | Wu M, Zhang R, Zou Q, Chen Y, Zhou M, Li X, Ran R, Chen Q. Comparison of the Biological Characteristics of Mesenchymal Stem Cells Derived from the Human Placenta and Umbilical Cord. Sci Rep. 2018;8:5014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 143] [Article Influence: 17.9] [Reference Citation Analysis (1)] |
| 49. | Hoang DH, Nguyen TD, Nguyen HP, Nguyen XH, Do PTX, Dang VD, Dam PTM, Bui HTH, Trinh MQ, Vu DM, Hoang NTM, Thanh LN, Than UTT. Differential Wound Healing Capacity of Mesenchymal Stem Cell-Derived Exosomes Originated From Bone Marrow, Adipose Tissue and Umbilical Cord Under Serum- and Xeno-Free Condition. Front Mol Biosci. 2020;7:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 133] [Article Influence: 22.2] [Reference Citation Analysis (4)] |
| 50. | Gu Y, Mu Z, Chen Y, Wu C, Shi J, Bai N. Therapeutic potential of ADSCs in diabetic wounds: a proteomics-based approach. Front Cell Dev Biol. 2024;12:1468220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 51. | Shi R, Jin Y, Zhao S, Yuan H, Shi J, Zhao H. Hypoxic ADSC-derived exosomes enhance wound healing in diabetic mice via delivery of circ-Snhg11 and induction of M2-like macrophage polarization. Biomed Pharmacother. 2022;153:113463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 111] [Article Influence: 27.8] [Reference Citation Analysis (2)] |
| 52. | Tseng SL, Kang L, Li ZJ, Wang LQ, Li ZM, Li TH, Xiang JY, Huang JZ, Yu NZ, Long X. Adipose-derived stem cells in diabetic foot care: Bridging clinical trials and practical application. World J Diabetes. 2024;15:1162-1177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 53. | Lu DB, Jiang YZ, Deng WQ, Zhang Y, Liang ZW, Wu Q, Jiang XY, Zhang L, Gao F, Cao Y, Chen B, Xue YM. Long-Term Outcomes of BMMSC Compared with BMMNC for Treatment of Critical Limb Ischemia and Foot Ulcer in Patients with Diabetes. Cell Transplant. 2019;28:645-652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 54. | He H, Nagamura-Inoue T, Tsunoda H, Yuzawa M, Yamamoto Y, Yorozu P, Agata H, Tojo A. Stage-specific embryonic antigen 4 in Wharton's jelly-derived mesenchymal stem cells is not a marker for proliferation and multipotency. Tissue Eng Part A. 2014;20:1314-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 55. | Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy. 2013;15:641-648. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 56. | Li JZ, Cao TH, Han JC, Qu H, Jiang SQ, Xie BD, Yan XL, Wu H, Liu XL, Zhang F, Leng XP, Kang K, Jiang SL. Comparison of adipose and bone marrowderived stem cells in protecting against oxLDLinduced inflammation in M1macrophagederived foam cells. Mol Med Rep. 2019;19:2660-2670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 57. | Noël D, Caton D, Roche S, Bony C, Lehmann S, Casteilla L, Jorgensen C, Cousin B. Cell specific differences between human adipose-derived and mesenchymal-stromal cells despite similar differentiation potentials. Exp Cell Res. 2008;314:1575-1584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 248] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 58. | Wang S, Lu H, Cheng L. [Angiogenic ability of 3 different tissues-derived mesenchymal stem cells on endothelial progenitor cells]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2018;43:184-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 59. | Zhang C, Huang L, Wang X, Zhou X, Zhang X, Li L, Wu J, Kou M, Cai C, Lian Q, Zhou X. Topical and intravenous administration of human umbilical cord mesenchymal stem cells in patients with diabetic foot ulcer and peripheral arterial disease: a phase I pilot study with a 3-year follow-up. Stem Cell Res Ther. 2022;13:451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 60. | Zhang C, Liao W, Li W, Li M, Xu X, Sun H, Xue Y, Liu L, Qiu J, Zhang C, Zhang X, Ye J, Du J, Deng DYB, Deng W, Li T. Human umbilical cord mesenchymal stem cells derived extracellular vesicles alleviate salpingitis by promoting M1-to-M2 transformation. Front Physiol. 2023;14:1131701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 61. | Ong HT, Dilley RJ. Novel non-angiogenic role for mesenchymal stem cell-derived vascular endothelial growth factor on keratinocytes during wound healing. Cytokine Growth Factor Rev. 2018;44:69-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 62. | Li Q, Yao D, Ma J, Zhu J, Xu X, Ren Y, Ding X, Mao X. Transplantation of MSCs in combination with netrin-1 improves neoangiogenesis in a rat model of hind limb ischemia. J Surg Res. 2011;166:162-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 63. | Xu B, Luo Y, Liu Y, Li BY, Wang Y. Platelet-derived growth factor-BB enhances MSC-mediated cardioprotection via suppression of miR-320 expression. Am J Physiol Heart Circ Physiol. 2015;308:H980-H989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 64. | Wong R, Smith CJ, Allan SM, Pinteaux E. Preconditioning with interleukin-1 alpha is required for the neuroprotective properties of mesenchymal stem cells after ischemic stroke in mice. J Cereb Blood Flow Metab. 2023;43:2040-2048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 65. | Siraj A, Haneef K. Role of hydrogen peroxide preconditioning in mesenchymal stem cell-mediated heart regeneration: Molecular insights. World J Cardiol. 2025;17:107437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 66. | Ghavam A, Sheikhnia F, Heidari MM, Valilo M, Mahmoudnejad Z, Gur S. An updated narrative review on revolutionizing erectile dysfunction treatment: the crucial role of trophic factors in Adipose-Derived stem cell therapy. BMC Urol. 2025;25:206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 67. | Cao J, Liu ZC, An WQ, Zhang S, Zhang X, Li LJ, Ji HL, Long X, Yang YM. Optimizing adipose-derived stem cell therapy for diabetic foot ulcers. World J Diabetes. 2025;16:109859. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 68. | Zhang Y, Shi L, Li X, Liu Y, Zhang G, Wang Y. Placental stem cells-derived exosomes stimulate cutaneous wound regeneration via engrailed-1 inhibition. Front Bioeng Biotechnol. 2022;10:1044773. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 69. | Dodig M, Li M, Dasarathy S, Kumarasamy S, Kasumov T, Najjar SM, McCullough AJ. Insulin increases type I collagen synthesis in hepatic stellate cells via α5β1 integrin. Metab Target Organ Damage. 2024;4:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |