INTRODUCTION
Diabetes mellitus (DM) is one of the most pervasive chronic metabolic disorders, characterized by impaired glucose utilization and excessive hepatic glucose output through glycogenolysis and gluconeogenesis, ultimately resulting in sustained hyperglycemia (American Diabetes Association, 2025)[1]. The diagnosis of DM is established through elevated plasma glucose concentrations and increased glycated hemoglobin (HbA1c) levels[2]. Beyond metabolic disruption, DM contributes significantly to the burden of cardiovascular diseases (CVD), particularly coronary artery disease (CAD), underscoring the intricate interplay between metabolic and vascular dysfunction[3].
CAD, also known as ischemic heart disease, arises from progressive narrowing or obstruction of the coronary arteries due to atherosclerosis, thereby impairing oxygen and nutrient delivery to the myocardium[4]. As the disease advances, myocardial ischemia ensues when oxygen demand exceeds supply, which may culminate in total arterial occlusion and infarction. Clinically, patients often present with chest pain, dyspnea, diaphoresis, nausea, and generalized malaise hallmarks of myocardial infarction. The poor management of CAD causes malfunction of heart structure and function deformities and ultimately cause heart failure and arrhythmias[5]. Moreover, in diabetic individuals the consequences of CVD which show the urgent need to explore rigorous association hyperglycemia and CVD specifically molecular mechanism. In the pathophysiology of hyperglycemia-associated coronary heart disease (HACHD) it is suggested that the elevated level of glucose non-enzymatically reacts with collagen molecule and form advanced glycation end products and these advanced glycation end products bind with its receptors of advanced glycation end product and start various pathways which cause CVD. Further, hyperglycemia itself generates oxidative stress and affect various metabolic pathways and cause CVD[6].
To bridging cardiac dysfunction and hyperglycemia, Li and Zhang[7] conduct a retrospective study in which authors keen to observe the association between heart rate dynamics and blood glucose variability in type 2 DM with CAD in elderly patients. Authors measure various fluctuations of glucose in a specific period and calculate mean postprandial glucose excursions (MPPGE) and mean amplitude of glycemic excursions (MAGE) correlate with reduced heart rate variability (HRV). In statistical analysis the authors found inverse association between these glycaemia parameters inversely associated with HRV. Further, combined analysis of MPPGE and MAGE shows higher predictive accuracy for myocardial ischemia compared to traditional HRV indices. These observations emphasize the glycemic control plays major and important role in pathophysiology of CVD and suggest contentious glucose monitoring increases the risk stratification in HACHD.
For further expanding these clinical associations, several studies in molecular biology uncovered and functional and regulatory role of long non-coding RNAs (lncRNAs) in HACHD. The lncRNAs have more than 200 nucleotide long noncoding transcripts and show regulatory role in gene expression at various levels like post transcriptional, transcriptional and epigenetic level[8]. Several studies have identified lncRNAs specifically NEAT1, HOTAIR and MALAT1 are major regulators in HACHD, affecting cellular function of pancreatic beta cells, resistance against insulin, endothelial injury and vascular inflammation[9,10]. Overall, it can be inferred that lncRNAs may act as both potential biomarkers and molecular mediators in HACHD and its progression.
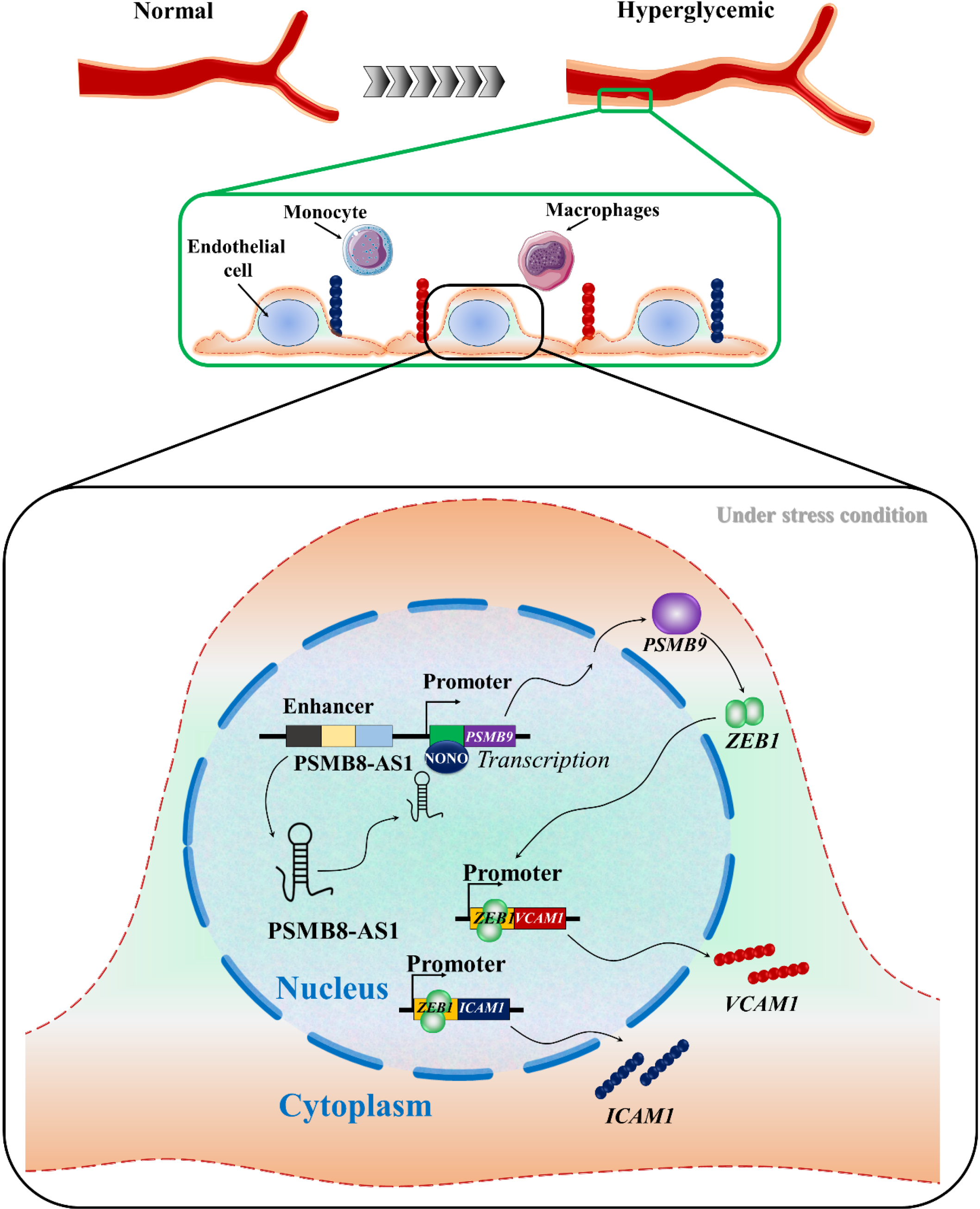
Further recent study by Wang et al[11] identified PSMB8-AS-1 (PSMB8 antisense RNA 1) lncRNA as a major gene regulator in acute coronary syndrome, where it promotes vascular inflammation by targeting and upregulation of vascular cell adhesion molecule 1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1). Further, Li et al[12] revealed that PSMB8-AS-1 (forced expression) increase atherogenesis by upregulating endothelial adhesion molecule expression like VCAM-1 and ICAM-1. Mechanistically, PSMB8-AS-1 recruits the transcription factor NONO to the PSMB9 promoter, thereby increasing VCAM-1 and ICAM-1 expression via ZEB1 activation. This PSMB8-AS-1/NONO/PSMB9/ZEB1 regulatory axis has emerged as a key contributor to vascular inflammation and atherosclerotic progression, with its inhibition offering therapeutic potential[7]. Thus, the molecular crosstalk between lncRNAs and transcriptional networks provides a promising avenue for novel therapeutic interventions.
In this context, PSMB9, an immunoproteasome-specific subunit, plays an indispensable role in protein homeostasis, particularly under cellular stress and mitochondrial dysfunction. Upregulation of PSMB9 enhances proteasomal degradation, preventing toxic protein aggregation and mitigating inflammatory stress. However, in hyperglycemic conditions, dysregulation of PSMB9 impairs proteostasis, exacerbates inflammation, and precipitates endothelial dysfunction[13,14]. EEF1A2, a translation elongation factor, further modulates PSMB9 expression, while molecular chaperones such as HSPB1 safeguard protein folding during oxidative stress[15]. The delicate balance of these molecular interactions is therefore critical to maintaining cardiovascular health under diabetic stress.
Additionally, the transcription factor ZEB1, a zinc finger protein, regulates a wide range of physiological processes. Under normal physiological conditions, ZEB1 supports endothelial cell survival and vascular integrity; however, in disease condition like diabetes it promotes various cell processes like angiogenesis, cell migration, and epithelial-to-mesenchymal transition[16], it also regulates adhesion of the monocyte, macrophage and accumulation of lipid and smooth muscle proliferation and ultimately increases atherosclerosis[17]. Moreover, studies reported that in diabetic condition elevated level of ZEB-1 increases the level of inflammatory cytokines like interleukin-6 and interleukin-8 and promotes myocardial injury and causes CVD[18]. Experimental studies suggested that repression of ZEB-1 by microRNA miR-200b-3p reduce the inflammation and CVD consequences[19]. These findings shed light on ZEB1 may act as a double-edged regulator, promoting angiogenesis and exacerbating vascular injury in HACHD. Furthermore, it is also suggested that regulation of ZEB1 affects the NONO, plays important roles extracellular matrix remodeling, gene transcription and cardiac metabolism. Studies suggested that deficiency of NONO leads to affect collagen synthesis, myocardial fibrosis and impaired diastolic function[20]. Additionally, it also regulates the metabolic pathways glucose and lipids, linking metabolic disturbances in diabetes to cardiac dysfunction. Downregulation of NONO causes cardiomyopathic and remodeling of cardiac cells[21]. Taken together, the NONO centrality in bridging cardiac homeostasis by structural and metabolic regulation.
The combined effect of the molecular triad PSMB9/ZEB1/NONO by PSMB8-AS-1 operates may act as network that mediates cellular adaptation, stress response, and vascular homeostasis. It is also reported that cellular oxidative stress potentially affects the regulation of transcription factors triad PSMB9/ZEB1/NONO and disrupt normal physiology and cause endothelial dysfunction, vascular inflammation, and maladaptive cardiac remodeling[15,21,22]. Figure 1 represents that hyperglycemia associated oxidative stress affects the regulation triad of transcription factors PSMB9/ZEB1/NONO by PSMB-AS-1 lncRNAs and upregulates expression of ICAM and VCAM which promote the adhesion of various cells like monocyte and macrophage and permeate lipid and cholesterol across the smooth muscle and promote atherosclerosis and increase the risk of CVD.
Figure 1 Mechanistic pathway linking hyperglycemia to atherosclerosis and cardiovascular disease via oxidative stress and endothelial activation.
It illustrates how hyperglycemia under stress conditions increases PSMB8-AS-1 expression, which then promotes vascular inflammation by upregulating intercellular adhesion molecule-1 and vascular cell adhesion molecule 1 through interactions with NONO and PSMB9. This process facilitates blood cell adhesion, endothelial dysfunction, and plaque formation, ultimately leading to cardiovascular disease. ICAM-1: Intercellular adhesion molecule-1; VCAM-1: Vascular cell adhesion molecule 1.