Published online Mar 15, 2026. doi: 10.4239/wjd.v17.i3.112177
Revised: September 18, 2025
Accepted: January 12, 2026
Published online: March 15, 2026
Processing time: 226 Days and 1.5 Hours
Diabetic skin ulcers significantly reduce the quality of life of patients with dia
Core Tip: The mechanism of mitochondrial autophagy in skin ulcers and cognitive decline associated with diabetes is the main topic of this review. This study aimed to clarify the crucial role of mitochondrial autophagy in maintaining the metabolic balance of cells, provide potential therapeutic strategies and intervention methods, and offer new concepts for the clinical management of diabetes-related complications. It also exa
- Citation: Chen X, Zhang LL, Zhou Y, Dong DX, Qian XY. Mitochondrial autophagy in diabetes-related cognitive decline and skin ulcers: Mechanistic insights and therapeutic implications. World J Diabetes 2026; 17(3): 112177
- URL: https://www.wjgnet.com/1948-9358/full/v17/i3/112177.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i3.112177
One common consequence of diabetes is cognitive impairment[1-3]. Diabetes greatly raises the incidence of cognitive impairment-related diseases[4-8]. It has been reported that diabetes raises the risk of Alzheimer's disease (AD) by 1.43 times, vascular dementia by 1.91 times, and dementia from all causes by 1.25 times[9]. Patients with diabetes and cognitive impairment, particularly those who are young or elderly, have a lower quality of life and a higher financial burden. This impacts their neurological development, worsens disability, and adds to the strain of family care[10-12]. Likewise, diabetic skin ulcers are another terrible consequence, and their weakened wound-healing systems lead to high morbidity and medical expenses.
Due to impaired mitophagy (mitochondrial-selective autophagy) activity, damaged mitochondria build up in individuals with diabetes, exacerbating tissue damage and cellular dysfunction. Inadequate mitophagy, particularly in skin tissue, can impede keratinocyte and fibroblast activity, interfere with wound healing, and result in the development of persistent skin ulcers. In addition, diabetes-related vasculopathy and neuropathy can potentially worsen the occurrence and progression of skin ulcers by altering the local blood supply and nerve transmission.
Mitochondrial dysfunction has drawn more attention in relation to diabetes-related cognitive impairment[13-16]. The survival and activity of neurons, which are heavily reliant on energy, form the foundation of cognitive function[17]. At the same time, sufficient energy availability and tolerance to oxidative stress are necessary for glial-neuron metabolic interactions, such as neurotransmitter reuptake and energy substrate transfer. In addition to producing energy, mi
Even while the significance of mitophagy in diabetes complications is becoming more widely acknowledged, there are still a number of unanswered questions about the precise pathways relating to mitophagy failure and the development of skin ulcers and cognitive deterioration. The best therapeutic modulation techniques are yet unknown, and the directional changes in mitophagy across various diabetes situations are contradictory. Therefore, this review aims to systematically summarize the regulatory mechanisms of mitophagy in diabetes-related cognitive impairment and skin ulcers, with a particular focus on pathway-specific alterations, tissue heterogeneity, and their implications for therapeutic targeting.
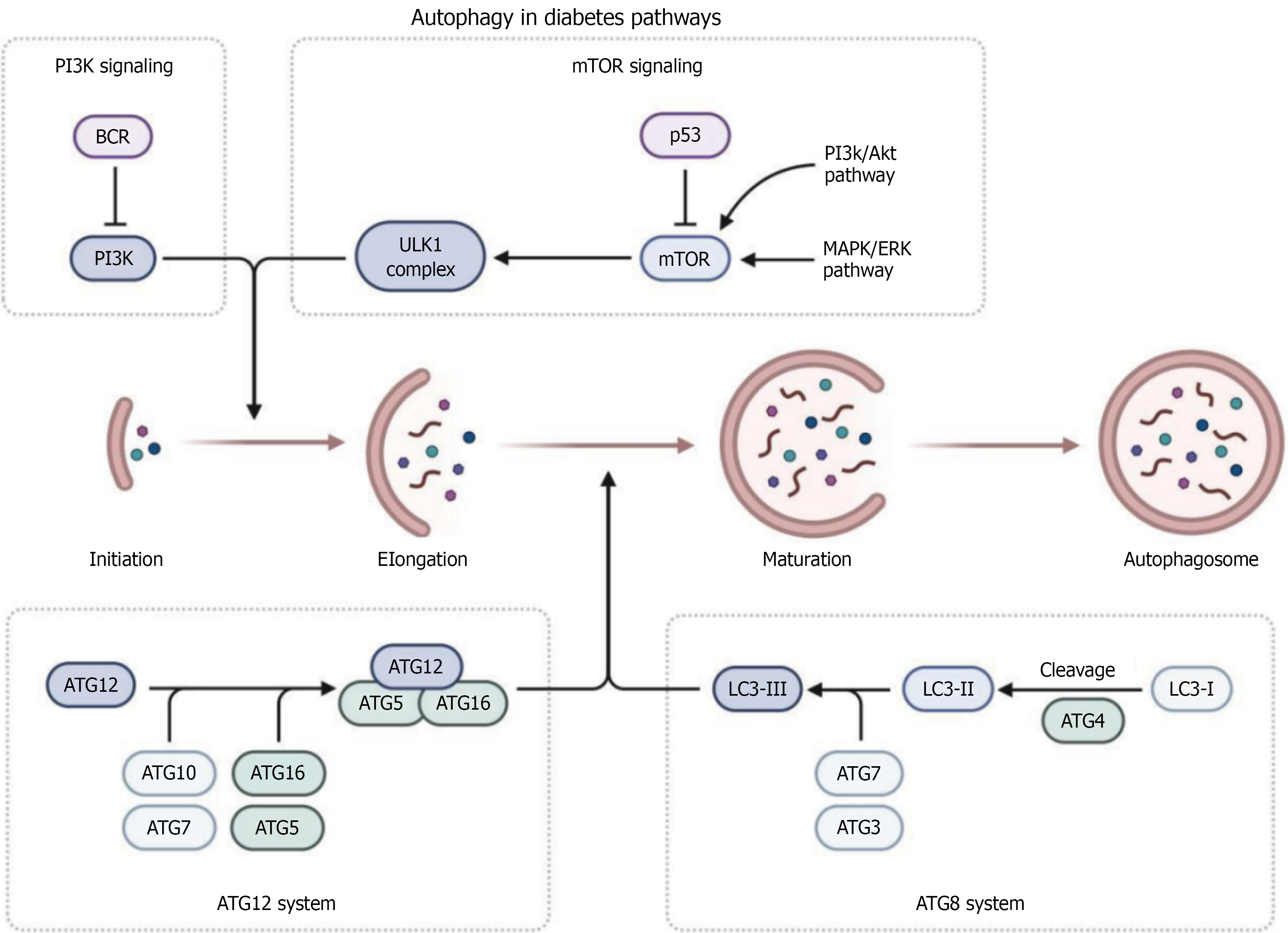
The self-defense mechanism against radiation, oxidative stress, hypoxia, nutritional deficiencies, and foreign microbes is autophagy, a catabolic process that sends the cell's cytoplasmic components-including proteins, organelles, aggregates, and any other intracellular material-to lysosomes for breakdown[31-33]. Macrophage, microautophagy, and chaperone-mediated autophagy are all forms of autophagy[34]. In general, autophagy is synonymous with macroautophagy, a process that involves the formation of a double-layered membrane autophagosome to consume damaged organelles or biochemical molecules. This autophagolysosome subsequently combines with the lysosome to break down the phagocyte[35-38]. Under normal circumstances, cellular autophagy levels stay low, allowing cells to preserve the circulation and regeneration of cell material, get rid of substances that induce cell death, and keep the cellular environment in a state of homeostasis. In order to maintain the energy supply, autophagy becomes highly active during stress, and nonselective autophagy degrades excess 3 proteins in cells (Figure 1).
Following identification and labeling of damaged mitochondria, light chain 3 (LC3), a microtubule-associated protein necessary for autophagy, changes from cytoplasmic soluble LC3I to membrane-type LC3II. This latter is the primary constituent of the bilayer structure of mature autophagosomes and is enlisted to form autophagosomes surrounding damaged mitochondria[39-42]. By binding to proteins tagged on the mitochondrial surface, LC3II phosphorylates ubiquitin (UB) via the phagophore assembly site (PAS). Mitophagosomes are later formed by engulfing damaged mitochondria, and these cells work in tandem with lysosomes to degrade damaged mitochondria[43-45]. Cell damage is the result of insufficient mitophagy, which causes damaged mitochondria to accumulate and ROS levels to rise[46]. Furthermore, lysosomes can release a number of proteases in response to excessive mitophagy, which can result in the improper removal of healthy mitochondria and ultimately cell death[47-50]. Sustaining mitochondrial function and cell viability requires balanced mitophagy (Figure 2). Thus, scientists have been concentrating on the ideal amount of mitophagy and how to balance it.
The most traditional method of initiating mitophagy is PINK1/PARKIN-mediated[51-53]. PINK1, a mitochondrial serine/threonine kinase, is physiologically transported by the translocation complex translocase in the outer membrane of the mitochondria to the intermembrane space and then by the inner mitochondrial membrane (IMM) translocase to the IMM[54-56]. Once in the IMM, PINK1 is released into the cytoplasm for destruction after being cleaved by mitochondrial matrix proteases[57-60]. PARKIN is activated by p-UB, which is created when PINK1 phosphorylates UB at Ser65[61-64]. By altering the UB chains of mitochondria, which PINK1 phosphorylates successively, PARKIN, a crucial UB E3 ligase, enhances the original signal and establishes a positive feedback loop with PINK1[65]. Autophagy-associated proteins are drawn to the vicinity of mitochondria when p-Ser65 chains build up on the outer mitochondrial membrane[66-68]. It has been shown that the multiubiquitination signal in PINK1/PARKIN-mediated mitophagy is recognized by proteins such SQSTM1/p62, the UB-binding protein p62, NBR1, NDP52, OPTN, and TAX1BP1[69-72]. Both the UB binding domain and the LC3 interaction region (LIR) are present in the aforementioned autophagy-associated proteins, and they bind to the UB chain on the target mitochondria[73-75]. The autophagy initiation factor ULK1, FYVE and coiled-coil DFCP1, and WIPI1 are then enlisted to encourage the formation of autophagosomes surrounding mitochondria[76-78]. Among them, SQSTM1/p62 has been frequently employed in conjunction with LC3II as a measure of autophagic flux and has long been linked to selective autophagy, including mitophagy[79-83].
Proteins with LIR motifs on mitochondrial membranes are involved in receptor-mediated mitophagy. In certain circumstances and cell types, these proteins act as mitophagy receptors by binding directly to the PAS and enabling direct interactions between mitochondria and LC3 or other members of the LC3/GABA receptor-associated protein (GABARAP) family to form mitophagosomes[84-90]. BCL2 adenovirus E1B 19 kDa interacting protein 3 (BNIP3) and its analogs BNIP3 L (NIX), BCL2 L13, FUNDC1, anti-mitochondrial antibody-related autophagy and Beclin-1-regulated autophagy protein 1 (AMBRA1), PHB2, and cardiolipin are among the proteins that have been shown to act as mitophagy receptors thus far (Figures 3 and 4).
Importantly, these two mitophagy pathways appear to be regulated differently in diabetes and may therefore contribute to tissue-specific complications. Under chronic hyperglycemia, mitochondrial oxidative stress and impaired mitochondrial quality control have been repeatedly implicated in diabetes-associated neurocognitive impairment. In this context, disruption of the PINK1/Parkin axis-either through insufficient PINK1 accumulation on the outer mitochondrial membrane or inefficient Parkin recruitment-may compromise ubiquitination of damaged mitochondria and weaken mitophagic clearance, thereby favoring persistent mitochondrial dysfunction and ROS burden in the central nervous system[40,75,84]. Experimental evidence in diabetic models further supports that high-glucose conditions can suppress neuronal mitophagy and aggravate cognitive deficits, consistent with a potential role of PINK1/Parkin-related mitophagy impairment in diabetes-related cognitive decline[63,66,76].
By comparison, receptor-mediated mitophagy pathways (e.g., FUNDC1-, BNIP3/BNIP3 L-associated mechanisms) are often discussed in connection with hypoxia/inflammation-driven stress responses and metabolic perturbations, which are also common features across multiple diabetic complications[84,87,89]. Given that ischemia/hypoxia and inflammatory activation are central components of difficult-to-heal diabetic ulcers and related peripheral tissue injury, dysregulated receptor-mediated mitophagy could plausibly limit stress-adaptive mitochondrial turnover and thereby interfere with cellular repair programs under diabetic conditions[56,75]. Although direct mechanistic evidence may differ across tissues, the overall framework that “distinct mitophagy routes respond to distinct pathological cues” provides a useful lens to interpret heterogeneous diabetic complications[87,89].
Therefore, distinguishing between UB-dependent and receptor-mediated mitophagy may help explain why mi
In addition to the brain damage brought on by diabetes, such as insulin resistance, hyperglycemia, and hypoglycemia, diabetes-related cognitive impairment also includes AD and vascular cognitive impairment (VCI), which are directly linked to the onset of diabetes[91-95]. There are parallels and differences in the function of mitophagy in various cognitive diseases. In addition to experimental and animal studies, emerging evidence from human investigations also supports the involvement of mitophagy dysregulation in diabetes-related cognitive impairment. Clinical and observational studies have reported alterations in autophagy- and mitophagy-associated biomarkers in peripheral blood or cerebrospinal fluid of patients with cognitive dysfunction, suggesting compromised mitochondrial quality control in the human central nervous system[20,39]. Moreover, studies in patients with diabetes have demonstrated close associations between metabolic dysregulation, mitochondrial dysfunction, and cognitive decline, indirectly implicating impaired mitophagy in disease progression. Although direct assessment of mitophagy activity in human brain tissue remains technically challenging, these clinical findings provide important translational support for the relevance of mitophagy in diabetes-associated cognitive decline[87,93].
In various stages of the same disease, mitophagy is a dynamic process that can be strengthened or inhibited[96-98]. Although there are currently few thorough studies on the role of mitophagy status in hyperglycemia-induced cognitive impairment, what is known indicates that mitophagy disruption is linked to cognitive impairment in diabetes[99-104]. Proteins whose expression significantly changed in peripheral blood platelets were significantly enriched in the dysregulated mitophagy/autophagy pathway, according to a clinical study[105] of diabetes patients with mild cognitive impairment (MCI) that used proteomics to investigate peripheral blood markers of type 2 diabetes mellitus (T2DM) patients with MCI. OPTN, SQSTM1, and TBC1D15 are some of these proteins. A strong correlation was found between a decrease in the MMSE score and an increase in OPTN, which facilitates the initiation of mitophagy. This suggests that T2DM patients with MCI may activate mitophagy mediated by OPTN[106-110]. These results imply that elevated OPTN may be a distinguishing factor between patients with T2DM-MCI and T2DM-nMCI[111-114]. However, there are still significant unknowns regarding the state of mitophagy in the central nervous system, and mitophagy markers in peripheral blood only offer circumstantial evidence.
Patients with type 1 diabetes who had consistently poor glucose control and reported brain tissue abnormalities and neuronal degeneration were evaluated for brain injury in one postmortem research[115]. Increased LC3 levels and ATG7 expression were found in one investigation, indicating a possible role for autophagy system activation in diabetic brain neuronal injury[116-120]. Although the cellular autophagy system's activation implies that mitophagy may also be engaged, the mitophagy state in the central nervous system of diabetic patients has not been explicitly investigated in humans. However, mitophagy-specific markers are needed to validate this assumption.
Constant exposure to high blood sugar impairs mitochondrial activity and, by generating too many ROS, encourages oxidative stress and neuroinflammation[121-126]. The mechanisms of mitophagy-mediated mitochondrial dysfunction have been uncovered by a number of fundamental investigations[127-130]. When synaptic integrity and autophagy in the cortex and hippocampus of 3xTg-AD and T2DM model mice were investigated, researchers observed that brain damage in the two groups was identical[131-134]. It was suggested that the autophagy-mediated lysosomal system was impeded by the decreased expression of ATG7 and LAMP1[135]. Although this study only looked at macroautophagy markers rather than mitophagy-specific indicators, it did find similarities between AD and diabetic brain damage in terms of mitochondrial dysfunction.
The expression of PINK1/PARKIN, which are essential proteins for mitophagy, was downregulated, and autophagic flux was compromised, according to a later study[136] that examined alterations in mitophagy in PC12 brain cell lines grown in vitro under high glucose conditions. The decrease in PINK1/PARKIN-mediated mitophagy in nerve cells under high glucose conditions was further supported by the observation of decreased colocalization between the mitochondrial marker protein COX IV and LAMP2[137-140]. Furthermore, in PC12 cells grown with high glucose, increasing FUNDC1-mediated mitophagy reduced mitochondrial dysfunction and increased cell resilience to oxidative stress and inflammation caused by ROS[141-147]. These findings imply that FUNDC1-mediated mitophagy plays a protective role in the mitochondrial dysfunction produced by high glucose.
While research on animals and cells have shown that the mitophagy system is inhibited, the mitophagy system is activated in hyperglycemia-related cognitive impairment[148-150]. The complicated and dynamic nature of mitophagy, which is impacted by a number of factors such as age, the duration of the disease, blood glucose regulation patterns, and study participant problems, may be reflected in this seeming contradiction. There are difficulties in simulating hyperglycemia models in their entirety, and different experimental methodologies may differ[151-156]. Nevertheless, these experimental studies have provided vital insights into the impact of hyperglycemia on central nervous system autophagy and particular mechanisms of mitophagy failure, affording important indications for clinical study.
Patients with diabetes who experience hypoglycemia frequently experience complications during glycemic treatment, and the development of hypoglycemia can raise their risk of cognitive impairment[157-160]. The pathogenesis of cognitive impairment in diabetics with hypoglycemia is significantly influenced by mitochondrial dysfunction[161-164]. The majority of research has been on cellular-level autophagy, with relatively few studies examining the connection between hypoglycemia-induced mitochondrial malfunction in central nervous system cells and mitophagy dysfunction[165-168]. Similar to high glucose reperfusion after hypoglycemic episodes, blood sugar levels in diabetic patients can rise sharply above normal levels after hypoglycemia[169]. According to research, elevated levels of LC3II and p62 occur together with autophagy inhibition in the rat hippocampal and cerebral cortex during acute hypoglycemia[170-174]. Activating autophagy can lessen cell damage. Another study showed that in mouse neuroblastoma cell lines (Neuro-2A), autophagy was activated and LC3II levels rose following glucose deprivation, but autophagy was blocked during hyperglycemic reperfusion, which resulted in cell death[175-178]. According to these investigations, autophagy suppression exacerbated damage to nerve cells[179]. Although there aren't many comprehensive research on how mitophagy failure contributes to hypoglycemia cognitive impairment in diabetes, those that do exist offer crucial hints: Impaired cellular autophagic flux could suggest that impaired mitophagy flux plays a role in the pathogenesis of cognitive impairment brought on by mitochondrial malfunction linked to hypoglycemia[180-182]. For a variety of reasons, including irregular medication, an irregular food, and high glucose reperfusion following recovery from hypoglycemia, diabetic patients may have blood sugar variations during glycemic treatment[183-186]. Cognitive disturbance in diabetic individuals has been associated with blood sugar variations[187]. Nevertheless, it is still unknown how blood glucose fluctuations and mitochondrial dysfunction relate to one another in people with cognitive impairment[188]. Research has connected changes in glucose to neural mitochondrial dysfunction, which includes increased formation of ROS in the mitochondria, decreased activity of manganese-dependent superoxide dismutase, and decreased mitochondrial membrane potential[189-192]. Although neuronal studies have not fully examined the role of mitophagy dysfunction in the cognitive impairment linked to blood glucose fluctuations, researchers have found that extreme blood glucose fluctuations can reduce mitophagy, leading to mitochondrial dysfunction in other cell types[193]. Therefore, we speculate that mitochondrial dysfunction and cognitive impairment associated with blood glucose variations may be caused by impaired mitophagy; however, further research is required to clarify the function and mechanism of mitophagy in this setting.
The most prevalent neurodegenerative disease, AD, shares a pathophysiology with type 2 diabetes and is frequently referred to as “type 3 diabetes” due to its central insulin resistance and decreased brain glucose metabolism[194]. Reduced mitochondrial oxidative phosphorylation capability, decreased ATP generation, elevated oxidative stress, and unbalanced mitochondrial quality control systems are all hallmarks of mitochondrial dysfunction, which is common in T2DM and AD[195]. In reaction to neurons' intake of glucose, insulin signaling controls mitochondrial activity[196]. The cognitive impairment linked to type 2 diabetes, oxidative stress, and neuroinflammation in AD, as well as neurodegeneration, can all be exacerbated by inadequate insulin signaling in insulin resistance, which can affect mitochondrial function and quality control[197-200]. One crucial element of the mitochondrial quality control system is mitophagy. Examining how aberrant mitophagy contributes to the pathogenesis of AD offers important new information about the mechanisms underlying type 2 diabetes patients’ cognitive decline.
Much attention has been paid to mitochondrial dysfunction in AD brought on by mitophagy problems and how it contributes to the emergence of cognitive impairment[201]. The postmortem hippocampus tissue of AD patients, AD model mice, and AD-like mouse neuronal cell cultures has been found to exhibit abnormalities in mitophagy[202]. The primary manifestation of aberrant mitophagy in AD is “ineffective autophagy”, which is brought on by a disruption in mitophagy activities and the buildup of damaged mitochondria and mitochondrial debris. Intracellular neurofibrillary tangles, which are mostly made up of extracellular beta-amyloid protein (Aβ) plaques and hyperphosphorylated tau protein (p-tau), are among the primary pathogenic characteristics of AD[203-205]. P-tau and Aβ cause excessive mitochondrial fragmentation and the formation of free radicals through their aberrant interactions with mitochondrial proteins, including VDAC1 and DRP1. Reduced mitophagy hinders the removal of damaged mitochondria and mitochondrial fragments, which leads to the accumulation of damaged mitochondria and a decrease in mitochondrial ATP generation. This impairs physiological functions that depend on cellular energy and ultimately leads to cell death.
It's interesting to note that mitophagy activation might happen early in AD. Research shows higher lysosomal and mitophagy-related gene expression in the CA1 region of the hippocampus, along with raised levels of p62 and LC3 in damaged mitochondria and increased recruitment of PARKIN[206-208]. This could be a neuron's compensating reaction to harmful proteins. Mitophagy progressively becomes dysfunctional and disorganized with aging and disease progression. Cell models, AD patient brains, and animal models have all shown decreased levels of PINK1/PARKIN, BCL2 L13, and BNIP3 L/NIX. Additionally, mitophagy initiating proteins, including phosphorylated ULK1, and p-TBK1 are inhibited. Additionally, through aberrant interactions with PARKIN, Aβ and p-tau can interfere with mitophagy.
Furthermore, in the brains of AD patients, the buildup of mitochondrial debris brought on by damaged mitochondria and inadequate mitophagy impacts not only neurons but also microglia and astrocytes. Damaged and defective mitochondria build up in microglia of APP/PS1 double-transgenic AD model mice, impairing phagocytosis and Aβ plaque removal[209]. Astrocytes and neurons undergo mitochondrial transfer, in which the astrocytes take defective mitochondria from the neurons, complete mitochondrial rejuvenation by mitophagy and mitochondrial biosynthesis, and then transfer healthy mitochondria to the neurons. Because astrocytic mitophagy is compromised in AD, astrocytes’ capacity to sustain neurons is diminished.
Based on these findings, we postulate that although early AD is characterized by a transient compensatory mitophagy activation, as the disease progresses to the decompensation stage, several significant cell types in the central nervous system experience mitophagy abnormalities. Mitophagy activation might be a useful strategy for treating AD patients' cognitive impairment.
Diabetes is one risk factor for cerebrovascular disease, and atherosclerosis is a chronic consequence of diabetes. From moderate cognitive impairment to dementia, VCI encompasses a range of symptoms brought on by cerebral hemorrhage, cerebral infarction, and other cerebrovascular illnesses and associated risk factors[210]. The two primary mechanisms of injury are chronic cerebral hypoperfusion (CCH) and cerebral ischemia-reperfusion injury (I/R). Cognitive dysfunction may result from the inflammatory response, oxidative stress, energy supply disruptions, and synapse loss, among other pathways of cerebral ischemia injury. These many processes entail mitochondrial dysfunction.
Ischemic brain damage is influenced by mitophagy, a vital physiological process that maintains the quantity and quality of mitochondria. However, during I/R damage, increased mitophagy encourages neuronal death. Mitophagy levels progressively rise after early I/R injury, and active mitophagy lowers intracellular oxidative stress levels by stopping defective mitochondria from producing ROS. Twenty-four hours after I/R damage, mitophagy activity peaks and then declines. Later phases of I/R injury may cause mitophagy to deteriorate, however stimulation of mitophagy during this period can have some neuroprotective effects via the PINK1/PARKIN, FUNDC1, BNIP3 L/NIX, and other pathways. Mitophagy appears to decline in CCH damage, following patterns seen in AD. Instead of the traditional UB pathway, BNIP3-mediated mitophagy reduction may be the primary cause of neuronal damage under prolonged hypoxia.
The pathophysiology of diabetes-related cognitive impairment has been shown to involve mitochondrial dysfunction mediated by mitophagy malfunction. Thus, encouraging proper mitophagy activation or avoiding excessive mitophagy will offer new approaches to preserving mitochondrial function, reducing damage to the central nervous system, and preventing and treating cognitive impairment. Nowadays, the main methods for promoting mitophagy are targeted control of genes linked to mitophagy or pharmaceutical intervention[211].
The classic autophagy activator rapamycin (RAPA) has been shown to enhance mitochondrial function and decrease mitochondria-dependent apoptosis in AD model mice (level C evidence). RAPA does this by inhibiting the activity of the mTOR and activating AMPK to mediate autophagy and mitophagy. It dramatically lessens the cognitive impairments and impairment of synaptic plasticity brought on by AD. Other medications that increase mitophagy by generally boosting cellular autophagy include metformin (level B proof for diabetes applications) and NAD+ precursors. Although there is currently little evidence linking urolithin A and substances like spermidine to cognitive impairment caused by diabetes, they can directly increase the expression of key proteins in the UB and receptor pathways, which facilitates the start of mitophagy (level C evidence). Further mitophagy-regulating medications, such as resveratrol, melatonin, and glucagon-like peptide-1 receptor agonists, are being investigated for the treatment of diabetes-related cognitive impair
Some researchers have successfully improved AD-related cognitive impairment by activating mitophagy through focused intervention in the PINK1/PARKIN pathway, which relates to the tailored regulation of important mitophagy proteins. In mice with APP-induced AD, stereotactic injection of PINK1-overexpressing vectors into the hippocampus to stimulate mitophagy alleviated mitochondrial dysfunction and markedly decreased oxidative stress, synaptic dysfunction, amyloid-related pathology, and hippocampus Aβ levels. The cognitive impairments and pathological alterations in the hippocampus brought on by intraventricular STZ injection can be avoided by adenovirus-mediated PINK1 overexpression, while PINK1 deletion considerably worsens damage. In contrast to control mice, APP/PS1 mice that overexpressed PARKIN showed enhanced cognitive function, decreased Aβ-amyloid burden, and downregulated APP protein production. To fully understand the role of additional receptor-mediated mitophagy pathways in diabetes-related cognitive impairment, more study is required, as studies on targeted therapies for these pathways are still lacking[212].
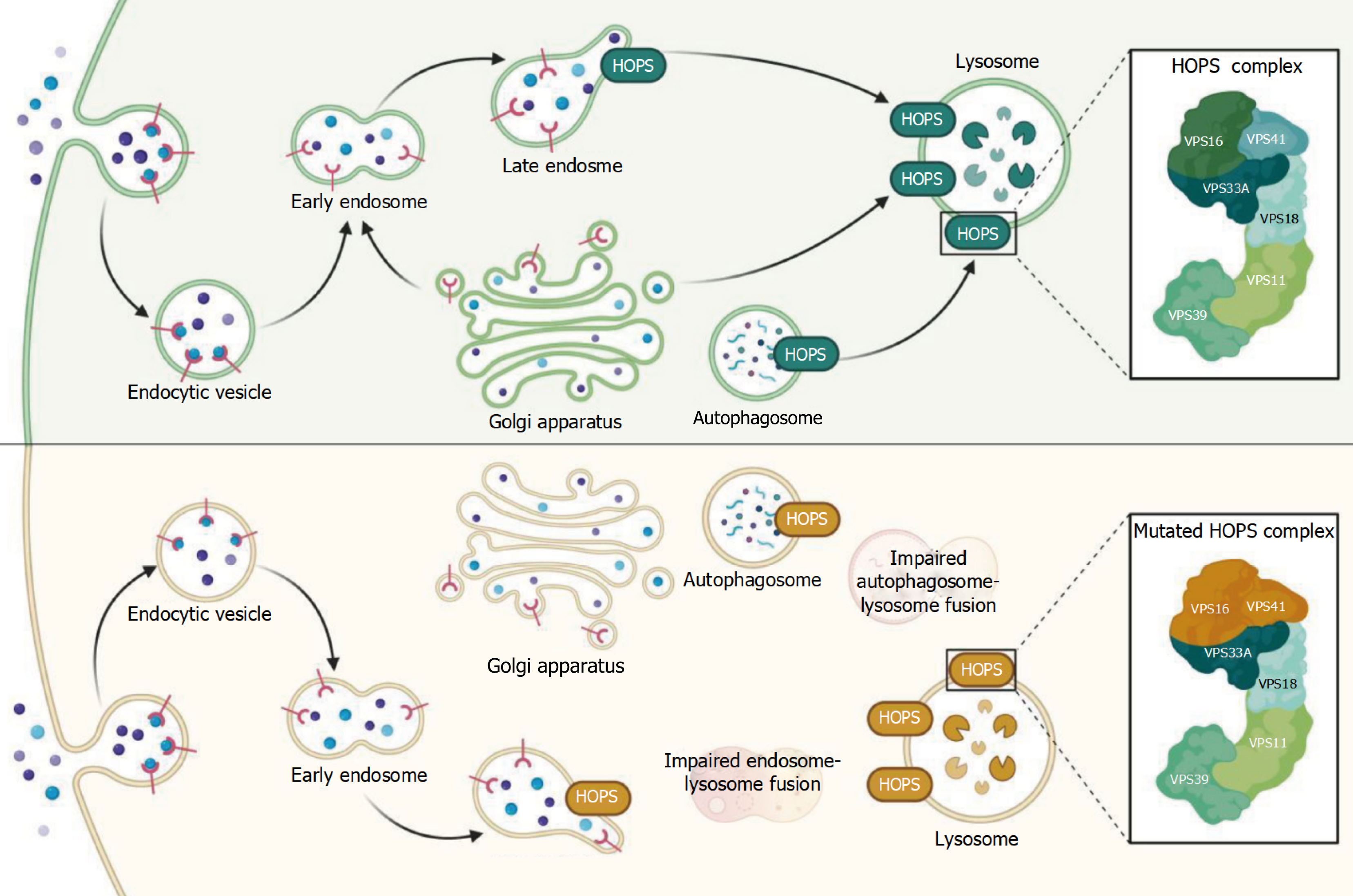
Mutations in the HOPS complex subunit have been linked to diabetes; these mutations may affect insulin production and autophagy, upsetting the glucose metabolic balance. Subunit mutations in the HOPS complex, which is necessary for lysosomal fusion, may result in inadequate insulin secretion or islet beta-cell malfunction, which could cause or worsen diabetes. By altering autophagy processes, HOPS complex anomalies may exacerbate the development of diabetes by increasing stress responses and islet beta-cell destruction. Though more research is needed to determine any direct links between neuronal and skin mitophagy, these findings offer fresh insights into the etiology of diabetes and lay the groundwork for future studies of possible treatment targets (Figure 5).
A particular type of selective autophagy called mitophagy is crucial for studying skin ulcers brought on by diabetes. One of the main causes of diabetic skin ulcers is mitochondrial damage brought on by the oxidative stress and inflammation that hyperglycemia frequently causes in diabetic individuals. By lowering oxidative stress, preserving intracellular energy balance, and selectively eliminating damaged mitochondria, mitophagy mitigates these pathogenic alterations[120-123].
Normal cellular metabolic progression is ensured by mitophagy's effective identification and removal of damaged or defective mitochondria. However, conditions like insulin resistance and chronic hyperglycemia frequently prevent mitophagy in diabetic individuals. This inhibition exacerbates cellular dysfunction and disturbance of energy meta
The health of keratinocytes and fibroblasts in skin tissue is directly impacted by impaired mitophagy. The function of the epidermal barrier and wound healing depend on these cells. Cellular oxidative stress rises when damaged mitochondria cannot be eliminated, consequently impairing cellular structure and function. In addition to delaying wound healing, this could encourage the development of persistent ulcers. Furthermore, diabetes-related neuropathy and vasculopathy worsen the severity of skin ulcers by impairing nerve conduction and lowering the local blood supply[56,94,120].
More and more recent research has concentrated on the specific mechanisms of mitophagy in diabetic skin ulcers. In diabetic mice, studies have shown that controlling important mitophagy proteins including PINK1 and PARKIN greatly enhances skin healing. These results suggest that the integrity and functionality of skin cells are maintained in large part by mitophagy. In addition to encouraging the regeneration and repair of damaged tissue, activation of the mitophagy pathway can lower the risk of complications from diabetes[91,94].
Furthermore, cellular energy consumption and signal transduction are closely related to mitophagy. In addition to being unable to produce energy effectively, damaged mitochondria in diabetics also release an excessive amount of ROS, which damages large areas of tissue. At the most basic level, the pathological state of skin ulcers can be improved by promoting mitophagy, which can efficiently remove these dangerous mitochondria and restore normal cellular function and metabolic balance[123].
However, there is still a significant information gap about mitophagy in human diabetic foot ulcer tissue, as evidenced by the paucity of available data. Future research should examine mitochondrial DNA damage, mitophagy-specific transcriptomics, PINK1/PARKIN colocalization, and electron microscopy in human tissue samples to confirm the mechanistic findings[132].
In conclusion, mitophagy represents a critical regulatory mechanism underlying the pathogenesis of diabetes-associated skin ulcers, particularly through its role in maintaining mitochondrial quality control under conditions of chronic hyperglycemia, hypoxia, and inflammation. Further elucidation of mitophagy regulatory networks and their cell-type-specific functions in skin tissues may provide a theoretical foundation for the development of innovative therapeutic strategies aimed at improving wound healing outcomes in patients with diabetes[87,94].
Beyond skin complications, accumulating evidence also suggests that dysregulated mitophagy contributes to diabetes-related cognitive impairment, highlighting mitophagy as a shared pathological link across distinct diabetic complications. Future mechanistic studies are therefore required to clarify the origins and heterogeneity of mitophagy phenotypes observed in both diabetic skin ulcers and cognitive dysfunction, as well as to determine whether common or tissue-spe
From a translational perspective, priority research directions include the identification of reliable mitophagy-related biomarkers, optimization of therapeutic timing and intervention windows, and the implementation of well-designed clinical studies to validate findings from preclinical models. Integrating mitophagy-targeted interventions with established diabetes management strategies may ultimately offer novel avenues for preventing and treating these de
Beyond classical pharmacological agents such as rapamycin and metformin, emerging therapeutic strategies targeting mitophagy are being actively explored. These include mitochondrial-targeted antioxidants, modulation of mitophagy-related signaling pathways, and gene-based approaches aimed at restoring mitochondrial quality control. Recent advances in gene editing technologies, such as CRISPR-Cas9, have also raised the possibility of selectively manipulating key mitophagy regulators; however, such strategies remain largely experimental and require careful evaluation of safety and feasibility[89,91].
Importantly, several challenges hinder the direct translation of mitophagy-targeted interventions from preclinical models to clinical practice. These include the lack of reliable and noninvasive biomarkers to monitor mitophagy activity in patients, uncertainty regarding optimal timing and dosage of interventions, and potential tissue-specific or context-dependent effects of mitophagy modulation. Moreover, diabetes is a multifactorial systemic disease, suggesting that mitophagy-targeted therapies may be most effective when integrated with established metabolic control strategies rather than applied as standalone treatments. These discrepancies highlight the tissue-specific and stage-dependent nature of mitophagy regulation, underscoring the need to interpret mechanistic findings in light of clinical context rather than extrapolating from single disease models.
Dysregulated mitophagy is a shared pathogenic mechanism contributing to both diabetes-related cognitive impairment and impaired skin-ulcer healing by compromising mitochondrial quality control under chronic metabolic stress. Targeting mitophagy therefore represents a promising therapeutic avenue to restore neuronal resilience and improve wound repair, but effective translation will require defining disease- and stage-specific “optimal” mitophagy rather than indiscriminate activation or inhibition. Next steps toward clinical application include establishing accessible mitophagy-related biomarkers, identifying appropriate therapeutic windows, and validating mitophagy-modulating strategies in well-designed human studies relevant to cognitive outcomes and diabetic wound healing. Future studies should prioritize validation of mitophagy-related mechanisms in human populations, followed by the identification of reliable circulating or tissue-based biomarkers and the definition of optimal therapeutic windows. Such efforts will be essential for translating mitophagy-targeted strategies into clinically feasible interventions for diabetes-related cognitive impairment and skin ulcers.
| 1. | Fang EF, Hou Y, Palikaras K, Adriaanse BA, Kerr JS, Yang B, Lautrup S, Hasan-Olive MM, Caponio D, Dan X, Rocktäschel P, Croteau DL, Akbari M, Greig NH, Fladby T, Nilsen H, Cader MZ, Mattson MP, Tavernarakis N, Bohr VA. Mitophagy inhibits amyloid-β and tau pathology and reverses cognitive deficits in models of Alzheimer's disease. Nat Neurosci. 2019;22:401-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1514] [Cited by in RCA: 1366] [Article Influence: 195.1] [Reference Citation Analysis (1)] |
| 2. | Ding H, Li Y, Chen S, Wen Y, Zhang S, Luo E, Li X, Zhong W, Zeng H. Fisetin ameliorates cognitive impairment by activating mitophagy and suppressing neuroinflammation in rats with sepsis-associated encephalopathy. CNS Neurosci Ther. 2022;28:247-258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 3. | Zheng D, Wang H, Zhou Y, Chen Y, Chen G. Ac-YVAD-cmk ameliorated sevoflurane-induced cognitive dysfunction and revised mitophagy impairment. PLoS One. 2023;18:e0280914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 4. | Yang K, Wu J, Li S, Wang S, Zhang J, Wang YP, Yan YS, Hu HY, Xiong MF, Bai CB, Sun YQ, Chen WQ, Zeng Y, Yuan JL, Yin CH. NTRK1 knockdown induces mouse cognitive impairment and hippocampal neuronal damage through mitophagy suppression via inactivating the AMPK/ULK1/FUNDC1 pathway. Cell Death Discov. 2023;9:404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 5. | Chen C, Yang C, Wang J, Huang X, Yu H, Li S, Li S, Zhang Z, Liu J, Yang X, Liu GP. Melatonin ameliorates cognitive deficits through improving mitophagy in a mouse model of Alzheimer's disease. J Pineal Res. 2021;71:e12774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 144] [Article Influence: 28.8] [Reference Citation Analysis (1)] |
| 6. | Hu F, Nie H, Xu R, Cai X, Shao L, Zhang P. Vinpocetine and coenzyme Q10 combination alleviates cognitive impairment caused by ionizing radiation by improving mitophagy. Brain Res. 2022;1792:148032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 7. | Hou M, Bao W, Gao Y, Chen J, Song G. Honokiol improves cognitive impairment in APP/PS1 mice through activating mitophagy and mitochondrial unfolded protein response. Chem Biol Interact. 2022;351:109741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 8. | Jiang W, Liu F, Li H, Wang K, Cao X, Xu X, Zhou Y, Zou J, Zhang X, Cui X. TREM2 ameliorates anesthesia and surgery-induced cognitive impairment by regulating mitophagy and NLRP3 inflammasome in aged C57/BL6 mice. Neurotoxicology. 2022;90:216-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 32] [Reference Citation Analysis (1)] |
| 9. | Wang W, Gao W, Zhang L, Xia Z, Zhao B. SNAP25 ameliorates postoperative cognitive dysfunction by facilitating PINK1-dependent mitophagy and impeding caspase-3/GSDME-dependent pyroptosis. Exp Neurol. 2023;367:114463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 10. | Zhou Y, Zhao Q, Zhang Y, Di L, Xue F, Xu W, Gao W, Guo Y, He Y, Kou J, Qin Y, Xie X, Du L, Han G, Pang X. A new andrographolide derivative ADA targeting SIRT3-FOXO3a signaling mitigates cognitive impairment by activating mitophagy and inhibiting neuroinflammation in Apoe4 mice. Phytomedicine. 2024;124:155298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 11. | Zhou Y, Luo D, Shi J, Yang X, Xu W, Gao W, Guo Y, Zhao Q, Xie X, He Y, Du G, Pang X. Loganin alleviated cognitive impairment in 3×Tg-AD mice through promoting mitophagy mediated by optineurin. J Ethnopharmacol. 2023;312:116455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 12. | Lautrup S, Lou G, Aman Y, Nilsen H, Tao J, Fang EF. Microglial mitophagy mitigates neuroinflammation in Alzheimer's disease. Neurochem Int. 2019;129:104469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 86] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 13. | Wen J, Wang Y, Wang C, Yuan M, Chen F, Zou Q, Cai Z, Zhao B. Dietary High-Fat Promotes Cognitive Impairment by Suppressing Mitophagy. Oxid Med Cell Longev. 2023;2023:4822767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 14. | Wang J, Zhu S, Lu W, Li A, Zhou Y, Chen Y, Chen M, Qian C, Hu X, Zhang Y, Huang C. Varenicline improved laparotomy-induced cognitive impairment by restoring mitophagy in aged mice. Eur J Pharmacol. 2022;916:174524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 15. | Xu F, Wu Y, Yang Q, Cheng Y, Xu J, Zhang Y, Dai H, Wang B, Ma Q, Chen Y, Lin F, Wang C. Engineered Extracellular Vesicles with SHP2 High Expression Promote Mitophagy for Alzheimer's Disease Treatment. Adv Mater. 2022;34:e2207107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 101] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 16. | Lin X, Wang H, Zou L, Yang B, Chen W, Rong X, Zhang X, He L, Li X, Peng Y. The NRF2 activator RTA-408 ameliorates chronic alcohol exposure-induced cognitive impairment and NLRP3 inflammasome activation by modulating impaired mitophagy initiation. Free Radic Biol Med. 2024;220:15-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 17. | Ahmedy OA, Abdelghany TM, El-Shamarka MEA, Khattab MA, El-Tanbouly DM. Apigenin attenuates LPS-induced neurotoxicity and cognitive impairment in mice via promoting mitochondrial fusion/mitophagy: role of SIRT3/PINK1/Parkin pathway. Psychopharmacology (Berl). 2022;239:3903-3917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 18. | Sun W, Li X, Chu Z, Huang T, Wu W. Euxanthone improves cognitive impairment by attenuating mitochondrial fragmentation and suppressing oxidative stress. Cent Eur J Immunol. 2021;46:446-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 19. | Luan Y, Jiang L, Luan Y, Xie Y, Yang Y, Ren KD. Mitophagy and Traumatic Brain Injury: Regulatory Mechanisms and Therapeutic Potentials. Oxid Med Cell Longev. 2023;2023:1649842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 20. | Castellazzi M, Patergnani S, Donadio M, Giorgi C, Bonora M, Bosi C, Brombo G, Pugliatti M, Seripa D, Zuliani G, Pinton P. Autophagy and mitophagy biomarkers are reduced in sera of patients with Alzheimer's disease and mild cognitive impairment. Sci Rep. 2019;9:20009. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 21. | Liu N, Cui X, Yan W, Guo T, Wang Z, Wei X, Sun Y, Liu J, Xian C, Ma W, Chen L. Baicalein: A potential GLP-1R agonist improves cognitive disorder of diabetes through mitophagy enhancement. J Pharm Anal. 2024;14:100968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 22. | Wang W, Zhao B, Gao W, Song W, Hou J, Zhang L, Xia Z. Inhibition of PINK1-Mediated Mitophagy Contributes to Postoperative Cognitive Dysfunction through Activation of Caspase-3/GSDME-Dependent Pyroptosis. ACS Chem Neurosci. 2023;14:1249-1260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 23. | Chakravorty A, Jetto CT, Manjithaya R. Dysfunctional Mitochondria and Mitophagy as Drivers of Alzheimer's Disease Pathogenesis. Front Aging Neurosci. 2019;11:311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 169] [Article Influence: 24.1] [Reference Citation Analysis (1)] |
| 24. | Zheng G, Wang L, Li X, Niu X, Xu G, Lv P. Rapamycin alleviates cognitive impairment in murine vascular dementia: The enhancement of mitophagy by PI3K/AKT/mTOR axis. Tissue Cell. 2021;69:101481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 25. | Tang W, Yan C, He S, Du M, Cheng B, Deng B, Zhu S, Li Y, Wang Q. Neuron-targeted overexpression of caveolin-1 alleviates diabetes-associated cognitive dysfunction via regulating mitochondrial fission-mitophagy axis. Cell Commun Signal. 2023;21:357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 34] [Reference Citation Analysis (1)] |
| 26. | Wang YM, Qiu MY, Liu Q, Tang H, Gu HF. Critical role of dysfunctional mitochondria and defective mitophagy in autism spectrum disorders. Brain Res Bull. 2021;168:138-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 27. | Zheng W, Li K, Zhong M, Wu K, Zhou L, Huang J, Liu L, Chen Z. Mitophagy activation by rapamycin enhances mitochondrial function and cognition in 5×FAD mice. Behav Brain Res. 2024;463:114889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 28. | Yu W, Chang X, Liao J, Quan J, Liu S, He T, Zhong G, Huang J, Liu Z, Tang Z. Long-term oral tribasic copper chloride exposure impedes cognitive function and disrupts mitochondrial metabolism by inhibiting mitophagy in rats. Environ Pollut. 2023;336:122474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 29. | Picca A, Faitg J, Auwerx J, Ferrucci L, D'Amico D. Mitophagy in human health, ageing and disease. Nat Metab. 2023;5:2047-2061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 474] [Cited by in RCA: 417] [Article Influence: 139.0] [Reference Citation Analysis (1)] |
| 30. | Um JH, Shin DJ, Choi SM, Nathan ABP, Kim YY, Lee DY, Jeong DJ, Kim DH, Kim KH, Kim YH, Nah J, Jeong JH, Yoo E, Shin HK, Park HT, Jo J, Cho JH, Yun J. Selective induction of Rab9-dependent alternative mitophagy using a synthetic derivative of isoquinoline alleviates mitochondrial dysfunction and cognitive deficits in Alzheimer's disease models. Theranostics. 2024;14:56-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 31. | Sharma C, Kim S, Nam Y, Jung UJ, Kim SR. Mitochondrial Dysfunction as a Driver of Cognitive Impairment in Alzheimer's Disease. Int J Mol Sci. 2021;22:4850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (1)] |
| 32. | Wu L, Zheng Y, Liu J, Luo R, Wu D, Xu P, Wu D, Li X. Comprehensive evaluation of the efficacy and safety of LPV/r drugs in the treatment of SARS and MERS to provide potential treatment options for COVID-19. Aging (Albany NY). 2021;13:10833-10852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 33. | Ge X, Wang L, Cui Q, Yan H, Wang Z, Ye S, Zhang Q, Fei A. Electroacupuncture improves cognitive impairment in diabetic cognitive dysfunction rats by regulating the mitochondrial autophagy pathway. J Physiol Sci. 2022;72:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 34. | Zhong X, Chen B, Li Z, Lin R, Ruan S, Wang F, Liang H, Tao J. Electroacupuncture Ameliorates Cognitive Impairment Through the Inhibition of NLRP3 Inflammasome Activation by Regulating Melatonin-Mediated Mitophagy in Stroke Rats. Neurochem Res. 2022;47:1917-1930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 35. | Lee DY, Lee KM, Um JH, Kim YY, Kim DH, Yun J. The Natural Alkaloid Palmatine Selectively Induces Mitophagy and Restores Mitochondrial Function in an Alzheimer's Disease Mouse Model. Int J Mol Sci. 2023;24:16542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 36. | Zou M, Wang D, Chen Y, Yang C, Xu S, Dai Y. Dajianzhong decoction ameliorated D-gal-induced cognitive aging by triggering mitophagy in vivo and in vitro. J Ethnopharmacol. 2024;319:117212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 37. | Song M, Zhao X, Song F. Aging-Dependent Mitophagy Dysfunction in Alzheimer's Disease. Mol Neurobiol. 2021;58:2362-2378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (1)] |
| 38. | Farsi RM. The Role of Mitochondrial Dysfunction in Alzheimer's: Molecular Defects and Mitophagy-Enhancing Approaches. Life (Basel). 2023;13:970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 39. | Veverová K, Laczó J, Katonová A, Horáková H, Matušková V, Angelucci F, Laczó M, Nedelská Z, Hort J, Wang HL, Zhang J, Shi L, Fei Fang E, Vyhnálek M. Alterations of human CSF and serum-based mitophagy biomarkers in the continuum of Alzheimer disease. Autophagy. 2024;20:1868-1878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 40. | Luo JS, Ning JQ, Chen ZY, Li WJ, Zhou RL, Yan RY, Chen MJ, Ding LL. The Role of Mitochondrial Quality Control in Cognitive Dysfunction in Diabetes. Neurochem Res. 2022;47:2158-2172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 41. | Liang J, Wang C, Zhang H, Huang J, Xie J, Chen N. Exercise-Induced Benefits for Alzheimer's Disease by Stimulating Mitophagy and Improving Mitochondrial Function. Front Aging Neurosci. 2021;13:755665. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 42. | Ye JS, Chen L, Lu YY, Lei SQ, Peng M, Xia ZY. Honokiol-Mediated Mitophagy Ameliorates Postoperative Cognitive Impairment Induced by Surgery/Sevoflurane via Inhibiting the Activation of NLRP3 Inflammasome in the Hippocampus. Oxid Med Cell Longev. 2019;2019:8639618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 43. | Mandal N, Das A, Datta R. Unravelling a mechanistic link between mitophagy defect, mitochondrial malfunction, and apoptotic neurodegeneration in Mucopolysaccharidosis VII. Neurobiol Dis. 2025;206:106825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 44. | Yan T, Zhao Y, Jiang Z, Chen J. Acetaldehyde Induces Cytotoxicity via Triggering Mitochondrial Dysfunction and Overactive Mitophagy. Mol Neurobiol. 2022;59:3933-3946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 45. | Sawant N, Morton H, Kshirsagar S, Reddy AP, Reddy PH. Mitochondrial Abnormalities and Synaptic Damage in Huntington's Disease: a Focus on Defective Mitophagy and Mitochondria-Targeted Therapeutics. Mol Neurobiol. 2021;58:6350-6377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 46. | Shi Y, Fang Q, Hu Y, Mi Z, Luo S, Gan Y, Yuan S. Melatonin Ameliorates Post-Stroke Cognitive Impairment in Mice by Inhibiting Excessive Mitophagy. Cells. 2024;13:872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 47. | Reddy AP, Sawant N, Morton H, Kshirsagar S, Bunquin LE, Yin X, Reddy PH. Selective serotonin reuptake inhibitor citalopram ameliorates cognitive decline and protects against amyloid beta-induced mitochondrial dynamics, biogenesis, autophagy, mitophagy and synaptic toxicities in a mouse model of Alzheimer's disease. Hum Mol Genet. 2021;30:789-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 48. | Wang W, Gao W, Gong P, Song W, Bu X, Hou J, Zhang L, Zhao B. Neuronal-specific TNFAIP1 ablation attenuates postoperative cognitive dysfunction via targeting SNAP25 for K48-linked ubiquitination. Cell Commun Signal. 2023;21:356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 29] [Reference Citation Analysis (1)] |
| 49. | Yu W, Lyu J, Jia L, Sheng M, Yu H, Du H. Dexmedetomidine Ameliorates Hippocampus Injury and Cognitive Dysfunction Induced by Hepatic Ischemia/Reperfusion by Activating SIRT3-Mediated Mitophagy and Inhibiting Activation of the NLRP3 Inflammasome in Young Rats. Oxid Med Cell Longev. 2020;2020:7385458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 50. | Li Y, Xia X, Wang Y, Zheng JC. Mitochondrial dysfunction in microglia: a novel perspective for pathogenesis of Alzheimer's disease. J Neuroinflammation. 2022;19:248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 135] [Reference Citation Analysis (1)] |
| 51. | Jiang W, Fan X, Wu H, Song J, Yang C, Zhao Z. TREM2 inhibits LPS-induced pyroptosis and inflammation by promoting mitophagy via SYK in BV2 cells. Neurotoxicol Teratol. 2025;110:107500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 52. | Yu M, Zhang M, Fu P, Wu M, Yin X, Chen Z. Research progress of mitophagy in chronic cerebral ischemia. Front Aging Neurosci. 2023;15:1224633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 53. | He Y, He T, Li H, Chen W, Zhong B, Wu Y, Chen R, Hu Y, Ma H, Wu B, Hu W, Han Z. Deciphering mitochondrial dysfunction: Pathophysiological mechanisms in vascular cognitive impairment. Biomed Pharmacother. 2024;174:116428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 54. | Zhao B, Wei D, Long Q, Chen Q, Wang F, Chen L, Li Z, Li T, Ma T, Liu W, Wang L, Yang C, Zhang X, Wang P, Zhang Z. Altered synaptic currents, mitophagy, mitochondrial dynamics in Alzheimer's disease models and therapeutic potential of Dengzhan Shengmai capsules intervention. J Pharm Anal. 2024;14:348-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 55. | Dai W, Xiao Y, Tu Y, Xiao F, Lu Y, Qin Y, Xie Y. Propofol protects hippocampal neurons in sleep-deprived rats by inhibiting mitophagy and autophagy. Ann Transl Med. 2021;9:1427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 56. | Kshirsagar S, Alvir RV, Pradeepkiran JA, Reddy AP, Reddy PH. Therapeutic potential of DDQ in enhancing mitochondrial health and cognitive function in Late-Onset Alzheimer's disease. Mitochondrion. 2025;83:102036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 57. | Zhang H, Niu Y, Qiu L, Yang J, Sun J, Xia J. Melatonin-mediated mitophagy protects against long-term impairments after repeated neonatal sevoflurane exposures. Int Immunopharmacol. 2023;125:111210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 58. | Li LL, Jin MF, Ni H. Zinc/CaMK II Associated-Mitophagy Signaling Contributed to Hippocampal Mossy Fiber Sprouting and Cognitive Deficits Following Neonatal Seizures and Its Regulation by Chronic Leptin Treatment. Front Neurol. 2018;9:802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 59. | Xie C, Zhuang XX, Niu Z, Ai R, Lautrup S, Zheng S, Jiang Y, Han R, Gupta TS, Cao S, Lagartos-Donate MJ, Cai CZ, Xie LM, Caponio D, Wang WW, Schmauck-Medina T, Zhang J, Wang HL, Lou G, Xiao X, Zheng W, Palikaras K, Yang G, Caldwell KA, Caldwell GA, Shen HM, Nilsen H, Lu JH, Fang EF. Amelioration of Alzheimer's disease pathology by mitophagy inducers identified via machine learning and a cross-species workflow. Nat Biomed Eng. 2022;6:76-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 241] [Article Influence: 60.3] [Reference Citation Analysis (1)] |
| 60. | Demarest TG, Waite EL, Kristian T, Puche AC, Waddell J, McKenna MC, Fiskum G. Sex-dependent mitophagy and neuronal death following rat neonatal hypoxia-ischemia. Neuroscience. 2016;335:103-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 61. | Yang Y, Chen W, Lin Z, Wu Y, Li Y, Xia X. Panax notoginseng saponins prevent dementia and oxidative stress in brains of SAMP8 mice by enhancing mitophagy. BMC Complement Med Ther. 2024;24:144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 62. | Wu L, Zheng Y, Ruan X, Wu D, Xu P, Liu J, Wu D, Li X. Long-chain noncoding ribonucleic acids affect the survival and prognosis of patients with esophageal adenocarcinoma through the autophagy pathway: construction of a prognostic model. Anticancer Drugs. 2022;33:e590-e603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (4)] |
| 63. | Song Y, Du Y, Zou W, Luo Y, Zhang X, Fu J. Involvement of impaired autophagy and mitophagy in Neuro-2a cell damage under hypoxic and/or high-glucose conditions. Sci Rep. 2018;8:3301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 64. | Zhang Y, Liao B, Hu S, Pan SY, Wang GP, Wang YL, Qin ZH, Luo L. High intensity interval training induces dysregulation of mitochondrial respiratory complex and mitophagy in the hippocampus of middle-aged mice. Behav Brain Res. 2021;412:113384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 65. | Sun L, Hong X, Wang D, Li Y. Overexpression of SESN1 improves mitochondrial damage and mitophagy, a potential therapeutic strategy for cognitive dysfunction after anaesthesia. Eur J Neurosci. 2024;59:208-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 66. | Cho JH, Chae CW, Lim JR, Jung YH, Han SJ, Yoon JH, Park JY, Han HJ. Sodium butyrate ameliorates high glucose-suppressed neuronal mitophagy by restoring PRKN expression via inhibiting the RELA-HDAC8 complex. Autophagy. 2024;20:1505-1522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 67. | Wu K, Huang C, Zheng W, Wu Y, Huang Q, Lin M, Gao R, Qi L, He G, Liu X, Liu X, Wang L, Chen Z, Liu L. Activation of mitophagy improves cognitive dysfunction in diabetic mice with recurrent non-severe hypoglycemia. Mol Cell Endocrinol. 2024;580:112109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 68. | Suo L, Wang M. Dexmedetomidine alleviates sevoflurane-induced neurotoxicity via mitophagy signaling. Mol Biol Rep. 2020;47:7893-7901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 69. | Harvey MR, Rodrigues J, McEachan J, Shiu Chung Tam S, Harrison C. Computerized adaptive testing for PRWHE measurements using domains of pain and motor function. J Hand Surg Eur Vol. 2025;50:940-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 70. | Lu J, Zong Y, Tao X, Dai H, Song J, Zhou H. Anesthesia/surgery-induced learning and memory dysfunction by inhibiting mitophagy-mediated NLRP3 inflammasome inactivation in aged mice. Exp Brain Res. 2024;242:417-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 71. | Guo J, Niu K, Ma BF, Sun LN, Fang QW, An JX. Electroacupuncture ameliorates surgery-induced spatial memory deficits by promoting mitophagy in rats. Ann Transl Med. 2023;11:74-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 362] [Article Influence: 362.0] [Reference Citation Analysis (1)] |
| 72. | Chiu CC, Lu CS, Weng YH, Chen YL, Huang YZ, Chen RS, Cheng YC, Huang YC, Liu YC, Lai SC, Lin KJ, Lin YW, Chen YJ, Chen CL, Yeh TH, Wang HL. PARK14 (D331Y) PLA2G6 Causes Early-Onset Degeneration of Substantia Nigra Dopaminergic Neurons by Inducing Mitochondrial Dysfunction, ER Stress, Mitophagy Impairment and Transcriptional Dysregulation in a Knockin Mouse Model. Mol Neurobiol. 2019;56:3835-3853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 73. | Wang C, Hu Z, Zou Y, Xiang M, Jiang Y, Botchway BOA, Huo X, Du X, Fang M. The post-therapeutic effect of rapamycin in mild traumatic brain-injured rats ensuing in the upregulation of autophagy and mitophagy. Cell Biol Int. 2017;41:1039-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 74. | Cen X, Xu X, Xia H. Targeting MCL1 to induce mitophagy is a potential therapeutic strategy for Alzheimer disease. Autophagy. 2021;17:818-819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 75. | Wu X, Yang J, He J. Research Progress of Mitochondrial Dynamics and Autophagy in Diabetic Complications: New Treatment Strategies. Diabetes Metab Syndr Obes. 2025;18:3167-3180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 76. | Wang Z, Xia P, Hu J, Huang Y, Zhang F, Li L, Wang E, Guo Q, Ye Z. LncRNA MEG3 Alleviates Diabetic Cognitive Impairments by Reducing Mitochondrial-Derived Apoptosis through Promotion of FUNDC1-Related Mitophagy via Rac1-ROS Axis. ACS Chem Neurosci. 2021;12:2280-2307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 77. | Lesage S, Drouet V, Majounie E, Deramecourt V, Jacoupy M, Nicolas A, Cormier-Dequaire F, Hassoun SM, Pujol C, Ciura S, Erpapazoglou Z, Usenko T, Maurage CA, Sahbatou M, Liebau S, Ding J, Bilgic B, Emre M, Erginel-Unaltuna N, Guven G, Tison F, Tranchant C, Vidailhet M, Corvol JC, Krack P, Leutenegger AL, Nalls MA, Hernandez DG, Heutink P, Gibbs JR, Hardy J, Wood NW, Gasser T, Durr A, Deleuze JF, Tazir M, Destée A, Lohmann E, Kabashi E, Singleton A, Corti O, Brice A; French Parkinson's Disease Genetics Study (PDG); International Parkinson's Disease Genomics Consortium (IPDGC). Loss of VPS13C Function in Autosomal-Recessive Parkinsonism Causes Mitochondrial Dysfunction and Increases PINK1/Parkin-Dependent Mitophagy. Am J Hum Genet. 2016;98:500-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 350] [Article Influence: 35.0] [Reference Citation Analysis (1)] |
| 78. | Chen Y, Wei G, Feng X, Lei E, Zhang L. Dexmedetomidine enhances Mitophagy via PINK1 to alleviate hippocampal neuronal Pyroptosis and improve postoperative cognitive dysfunction in elderly rat. Exp Neurol. 2024;379:114842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 79. | Chi H, Song M, Zhang J, Zhou J, Liu D. Relationship between acute glucose variability and cognitive decline in type 2 diabetes: A systematic review and meta-analysis. PLoS One. 2023;18:e0289782. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 80. | Zhao Z, Xie L, Shi J, Liu T, Wang S, Huang J, Wu D, Zhang X. Neuroprotective Effect of Zishen Huoxue Decoction treatment on Vascular Dementia by activating PINK1/Parkin mediated Mitophagy in the Hippocampal CA1 Region. J Ethnopharmacol. 2024;319:117172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 81. | Hou Y, Wei Y, Lautrup S, Yang B, Wang Y, Cordonnier S, Mattson MP, Croteau DL, Bohr VA. NAD(+) supplementation reduces neuroinflammation and cell senescence in a transgenic mouse model of Alzheimer's disease via cGAS-STING. Proc Natl Acad Sci U S A. 2021;118:e2011226118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 409] [Article Influence: 81.8] [Reference Citation Analysis (1)] |
| 82. | Zhang S, Wang A, Liu S, Liu H, Zhu W, Zhang Z. Glycemic variability correlates with medial temporal lobe atrophy and decreased cognitive performance in patients with memory deficits. Front Aging Neurosci. 2023;15:1156908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 83. | Wang ZT, Lu MH, Zhang Y, Ji WL, Lei L, Wang W, Fang LP, Wang LW, Yu F, Wang J, Li ZY, Wang JR, Wang TH, Dou F, Wang QW, Wang XL, Li S, Ma QH, Xu RX. Disrupted-in-schizophrenia-1 protects synaptic plasticity in a transgenic mouse model of Alzheimer's disease as a mitophagy receptor. Aging Cell. 2019;18:e12860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 80] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 84. | Shan Z, Fa WH, Tian CR, Yuan CS, Jie N. Mitophagy and mitochondrial dynamics in type 2 diabetes mellitus treatment. Aging (Albany NY). 2022;14:2902-2919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 85. | Blagov AV, Summerhill VI, Sukhorukov VN, Popov MA, Grechko AV, Orekhov AN. Type 1 diabetes mellitus: Inflammation, mitophagy, and mitochondrial function. Mitochondrion. 2023;72:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 86. | Narendra DP, Youle RJ. The role of PINK1-Parkin in mitochondrial quality control. Nat Cell Biol. 2024;26:1639-1651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 267] [Reference Citation Analysis (1)] |
| 87. | Apostolova N, Vezza T, Muntane J, Rocha M, Víctor VM. Mitochondrial Dysfunction and Mitophagy in Type 2 Diabetes: Pathophysiology and Therapeutic Targets. Antioxid Redox Signal. 2023;39:278-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 88. | Han YC, Tang SQ, Liu YT, Li AM, Zhan M, Yang M, Song N, Zhang W, Wu XQ, Peng CH, Zhang H, Yang S. AMPK agonist alleviate renal tubulointerstitial fibrosis via activating mitophagy in high fat and streptozotocin induced diabetic mice. Cell Death Dis. 2021;12:925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 202] [Article Influence: 40.4] [Reference Citation Analysis (0)] |
| 89. | Iorio R, Celenza G, Petricca S. Mitophagy: Molecular Mechanisms, New Concepts on Parkin Activation and the Emerging Role of AMPK/ULK1 Axis. Cells. 2021;11:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 160] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 90. | Cai C, Guo Z, Chang X, Li Z, Wu F, He J, Cao T, Wang K, Shi N, Zhou H, Toan S, Muid D, Tan Y. Empagliflozin attenuates cardiac microvascular ischemia/reperfusion through activating the AMPKα1/ULK1/FUNDC1/mitophagy pathway. Redox Biol. 2022;52:102288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 192] [Article Influence: 48.0] [Reference Citation Analysis (1)] |
| 91. | Tang S, Hao D, Ma W, Liu L, Gao J, Yao P, Yu H, Gan L, Cao Y. Dysfunctional Mitochondria Clearance in Situ: Mitophagy in Obesity and Diabetes-Associated Cardiometabolic Diseases. Diabetes Metab J. 2024;48:503-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 92. | He F, Huang Y, Song Z, Zhou HJ, Zhang H, Perry RJ, Shulman GI, Min W. Mitophagy-mediated adipose inflammation contributes to type 2 diabetes with hepatic insulin resistance. J Exp Med. 2021;218:e20201416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 122] [Article Influence: 24.4] [Reference Citation Analysis (1)] |
| 93. | Paul S, Saha D, Bk B. Mitochondrial Dysfunction and Mitophagy Closely Cooperate in Neurological Deficits Associated with Alzheimer's Disease and Type 2 Diabetes. Mol Neurobiol. 2021;58:3677-3691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 94. | Hu X, Lv J, Zhao Y, Li X, Qi W, Wang X. Important regulatory role of mitophagy in diabetic microvascular complications. J Transl Med. 2025;23:269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (1)] |
| 95. | Xiang J, Zhang C, Di T, Chen L, Zhao W, Wei L, Zhou S, Wu X, Wang G, Zhang Y. Salvianolic acid B alleviates diabetic endothelial and mitochondrial dysfunction by down-regulating apoptosis and mitophagy of endothelial cells. Bioengineered. 2022;13:3486-3502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 96. | Li C, Li L, Yang M, Yang J, Zhao C, Han Y, Zhao H, Jiang N, Wei L, Xiao Y, Liu Y, Xiong X, Xi Y, Luo S, Deng F, Chen W, Yuan S, Zhu X, Xiao L, Sun L. PACS-2 Ameliorates Tubular Injury by Facilitating Endoplasmic Reticulum-Mitochondria Contact and Mitophagy in Diabetic Nephropathy. Diabetes. 2022;71:1034-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 97. | Fan LH, Yang X, Wang ZG. [Role of mitophagy in diabetes mellitus and its complications and traditional Chinese medicine intervention: a review]. Zhongguo Zhong Yao Za Zhi. 2024;49:46-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 98. | Tong M, Saito T, Zhai P, Oka SI, Mizushima W, Nakamura M, Ikeda S, Shirakabe A, Sadoshima J. Mitophagy Is Essential for Maintaining Cardiac Function During High Fat Diet-Induced Diabetic Cardiomyopathy. Circ Res. 2019;124:1360-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 411] [Article Influence: 58.7] [Reference Citation Analysis (6)] |
| 99. | Li JM, Li X, Chan LWC, Hu R, Zheng T, Li H, Yang S. Lipotoxicity-polarised macrophage-derived exosomes regulate mitochondrial fitness through Miro1-mediated mitophagy inhibition and contribute to type 2 diabetes development in mice. Diabetologia. 2023;66:2368-2386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 76] [Reference Citation Analysis (1)] |
| 100. | Liu L, Bai F, Song H, Xiao R, Wang Y, Yang H, Ren X, Li S, Gao L, Ma C, Yang X, Liang X. Upregulation of TIPE1 in tubular epithelial cell aggravates diabetic nephropathy by disrupting PHB2 mediated mitophagy. Redox Biol. 2022;50:102260. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 98] [Article Influence: 24.5] [Reference Citation Analysis (1)] |
| 101. | Huang Q, Liu Z, Yang Y, Yang Y, Huang T, Hong Y, Zhang J, Chen Q, Zhao T, Xiao Z, Gong X, Jiang Y, Peng J, Nan Y, Ai K. Selenium Nanodots (SENDs) as Antioxidants and Antioxidant-Prodrugs to Rescue Islet β Cells in Type 2 Diabetes Mellitus by Restoring Mitophagy and Alleviating Endoplasmic Reticulum Stress. Adv Sci (Weinh). 2023;10:e2300880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (1)] |
| 102. | Prasertsuksri P, Kraokaew P, Pranweerapaiboon K, Sobhon P, Chaithirayanon K. Neuroprotection of Andrographolide against Neurotoxin MPP(+)-Induced Apoptosis in SH-SY5Y Cells via Activating Mitophagy, Autophagy, and Antioxidant Activities. Int J Mol Sci. 2023;24:8528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 33] [Reference Citation Analysis (1)] |
| 103. | Zheng H, Zhu H, Liu X, Huang X, Huang A, Huang Y. Mitophagy in Diabetic Cardiomyopathy: Roles and Mechanisms. Front Cell Dev Biol. 2021;9:750382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (1)] |
| 104. | Xiao L, Xu X, Zhang F, Wang M, Xu Y, Tang D, Wang J, Qin Y, Liu Y, Tang C, He L, Greka A, Zhou Z, Liu F, Dong Z, Sun L. The mitochondria-targeted antioxidant MitoQ ameliorated tubular injury mediated by mitophagy in diabetic kidney disease via Nrf2/PINK1. Redox Biol. 2017;11:297-311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 505] [Cited by in RCA: 476] [Article Influence: 52.9] [Reference Citation Analysis (0)] |
| 105. | Chang X, Li Y, Cai C, Wu F, He J, Zhang Y, Zhong J, Tan Y, Liu R, Zhu H, Zhou H. Mitochondrial quality control mechanisms as molecular targets in diabetic heart. Metabolism. 2022;137:155313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 124] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 106. | Yang YY, Gong DJ, Zhang JJ, Liu XH, Wang L. Diabetes aggravates renal ischemia-reperfusion injury by repressing mitochondrial function and PINK1/Parkin-mediated mitophagy. Am J Physiol Renal Physiol. 2019;317:F852-F864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 107. | Cai C, Wu F, He J, Zhang Y, Shi N, Peng X, Ou Q, Li Z, Jiang X, Zhong J, Tan Y. Mitochondrial quality control in diabetic cardiomyopathy: from molecular mechanisms to therapeutic strategies. Int J Biol Sci. 2022;18:5276-5290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 108. | Horton WB. Elucidating molecular mechanisms that alleviate cardiac microvascular dysfunction in diabetes: The potential benefit of targeting mitophagy and mitochondrial integrity. Acta Physiol (Oxf). 2023;238:e13983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 109. | Lin J, Duan J, Wang Q, Xu S, Zhou S, Yao K. Mitochondrial Dynamics and Mitophagy in Cardiometabolic Disease. Front Cardiovasc Med. 2022;9:917135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 110. | Yao L, Liang X, Liu Y, Li B, Hong M, Wang X, Chen B, Liu Z, Wang P. Non-steroidal mineralocorticoid receptor antagonist finerenone ameliorates mitochondrial dysfunction via PI3K/Akt/eNOS signaling pathway in diabetic tubulopathy. Redox Biol. 2023;68:102946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 86] [Article Influence: 28.7] [Reference Citation Analysis (1)] |
| 111. | Wang T, Wang X, Fu T, Ma Y, Wang Q, Zhang S, Zhang X, Zhou H, Chang X, Tong Y. Roles of mitochondrial dynamics and mitophagy in diabetic myocardial microvascular injury. Cell Stress Chaperones. 2023;28:675-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 112. | Kobayashi S, Liang Q. Autophagy and mitophagy in diabetic cardiomyopathy. Biochim Biophys Acta. 2015;1852:252-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 151] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 113. | Babbar M, Basu S, Yang B, Croteau DL, Bohr VA. Mitophagy and DNA damage signaling in human aging. Mech Ageing Dev. 2020;186:111207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 114. | da Silva Rosa SC, Martens MD, Field JT, Nguyen L, Kereliuk SM, Hai Y, Chapman D, Diehl-Jones W, Aliani M, West AR, Thliveris J, Ghavami S, Rampitsch C, Dolinsky VW, Gordon JW. BNIP3L/Nix-induced mitochondrial fission, mitophagy, and impaired myocyte glucose uptake are abrogated by PRKA/PKA phosphorylation. Autophagy. 2021;17:2257-2272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 85] [Article Influence: 14.2] [Reference Citation Analysis (1)] |
| 115. | Zhang X, Zhou H, Chang X. Involvement of mitochondrial dynamics and mitophagy in diabetic endothelial dysfunction and cardiac microvascular injury. Arch Toxicol. 2023;97:3023-3035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 81] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 116. | Soleimanpour SA, Ferrari AM, Raum JC, Groff DN, Yang J, Kaufman BA, Stoffers DA. Diabetes Susceptibility Genes Pdx1 and Clec16a Function in a Pathway Regulating Mitophagy in β-Cells. Diabetes. 2015;64:3475-3484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 117. | Yang YY, Gao ZX, Mao ZH, Liu DW, Liu ZS, Wu P. Identification of ULK1 as a novel mitophagy-related gene in diabetic nephropathy. Front Endocrinol (Lausanne). 2022;13:1079465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 118. | Wu S, Lu Q, Ding Y, Wu Y, Qiu Y, Wang P, Mao X, Huang K, Xie Z, Zou MH. Hyperglycemia-Driven Inhibition of AMP-Activated Protein Kinase α2 Induces Diabetic Cardiomyopathy by Promoting Mitochondria-Associated Endoplasmic Reticulum Membranes In Vivo. Circulation. 2019;139:1913-1936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 242] [Article Influence: 34.6] [Reference Citation Analysis (1)] |
| 119. | Cao P, Wang Y, Zhang C, Sullivan MA, Chen W, Jing X, Yu H, Li F, Wang Q, Zhou Z, Wang Q, Tian W, Qiu Z, Luo L. Quercetin ameliorates nonalcoholic fatty liver disease (NAFLD) via the promotion of AMPK-mediated hepatic mitophagy. J Nutr Biochem. 2023;120:109414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 80] [Article Influence: 26.7] [Reference Citation Analysis (1)] |
| 120. | Wada J, Nakatsuka A. Mitochondrial Dynamics and Mitochondrial Dysfunction in Diabetes. Acta Med Okayama. 2016;70:151-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 65] [Reference Citation Analysis (1)] |
| 121. | Higgins GC, Coughlan MT. Mitochondrial dysfunction and mitophagy: the beginning and end to diabetic nephropathy? Br J Pharmacol. 2014;171:1917-1942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 217] [Article Influence: 18.1] [Reference Citation Analysis (1)] |
| 122. | Russell JW, Golovoy D, Vincent AM, Mahendru P, Olzmann JA, Mentzer A, Feldman EL. High glucose-induced oxidative stress and mitochondrial dysfunction in neurons. FASEB J. 2002;16:1738-1748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 374] [Cited by in RCA: 399] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 123. | Belosludtsev KN, Belosludtseva NV, Dubinin MV. Diabetes Mellitus, Mitochondrial Dysfunction and Ca(2+)-Dependent Permeability Transition Pore. Int J Mol Sci. 2020;21:6559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 88] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 124. | Zhu Y, Han XQ, Sun XJ, Yang R, Ma WQ, Liu NF. Lactate accelerates vascular calcification through NR4A1-regulated mitochondrial fission and BNIP3-related mitophagy. Apoptosis. 2020;25:321-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 93] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 125. | de Marañón AM, Díaz-Pozo P, Canet F, Díaz-Morales N, Abad-Jiménez Z, López-Domènech S, Vezza T, Apostolova N, Morillas C, Rocha M, Víctor VM. Metformin modulates mitochondrial function and mitophagy in peripheral blood mononuclear cells from type 2 diabetic patients. Redox Biol. 2022;53:102342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 82] [Reference Citation Analysis (1)] |
| 126. | Su CJ, Shen Z, Cui RX, Huang Y, Xu DL, Zhao FL, Pan J, Shi AM, Liu T, Yu YL. Thioredoxin-Interacting Protein (TXNIP) Regulates Parkin/PINK1-mediated Mitophagy in Dopaminergic Neurons Under High-glucose Conditions: Implications for Molecular Links Between Parkinson's Disease and Diabetes. Neurosci Bull. 2020;36:346-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 127. | Alka K, Kumar J, Kowluru RA. Impaired mitochondrial dynamics and removal of the damaged mitochondria in diabetic retinopathy. Front Endocrinol (Lausanne). 2023;14:1160155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 128. | Guo Y, Jiang H, Wang M, Ma Y, Zhang J, Jing L. Metformin alleviates cerebral ischemia/reperfusion injury aggravated by hyperglycemia via regulating AMPK/ULK1/PINK1/Parkin pathway-mediated mitophagy and apoptosis. Chem Biol Interact. 2023;384:110723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 129. | Yang F, Wang X, Qi J, Zhang K, Jiang Y, Feng B, Lv T, Yang L, Yang Q, Zhao M, Liu S, Ma X. Glucagon-like Peptide 1 Receptor Activation Inhibits Microglial Pyroptosis via Promoting Mitophagy to Alleviate Depression-like Behaviors in Diabetic Mice. Nutrients. 2022;15:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (1)] |
| 130. | Zhou P, Xie W, Meng X, Zhai Y, Dong X, Zhang X, Sun G, Sun X. Notoginsenoside R1 Ameliorates Diabetic Retinopathy through PINK1-Dependent Activation of Mitophagy. Cells. 2019;8:213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 138] [Article Influence: 19.7] [Reference Citation Analysis (1)] |
| 131. | Li H, Zheng J, Wu Y, Zhou H, Zeng S, Li Q. Dendrobium officinale polysaccharide decreases podocyte injury in diabetic nephropathy by regulating IRS-1/AKT signal and promoting mitophagy. Aging (Albany NY). 2023;15:10291-10306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 132. | Bhansali S, Bhansali A, Walia R, Saikia UN, Dhawan V. Alterations in Mitochondrial Oxidative Stress and Mitophagy in Subjects with Prediabetes and Type 2 Diabetes Mellitus. Front Endocrinol (Lausanne). 2017;8:347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 101] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 133. | Zhu Z, Luan G, Peng S, Fang Y, Fang Q, Shen S, Wu K, Qian S, Jia W, Ye J, Wei L. Huangkui capsule attenuates diabetic kidney disease through the induction of mitophagy mediated by STING1/PINK1 signaling in tubular cells. Phytomedicine. 2023;119:154975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 32] [Reference Citation Analysis (1)] |
| 134. | Saxena S, Mathur A, Kakkar P. Critical role of mitochondrial dysfunction and impaired mitophagy in diabetic nephropathy. J Cell Physiol. 2019;234:19223-19236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 78] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 135. | Anderson G. Type I Diabetes Pathoetiology and Pathophysiology: Roles of the Gut Microbiome, Pancreatic Cellular Interactions, and the 'Bystander' Activation of Memory CD8(+) T Cells. Int J Mol Sci. 2023;24:3300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 136. | Zhu J, Wang KZ, Chu CT. After the banquet: mitochondrial biogenesis, mitophagy, and cell survival. Autophagy. 2013;9:1663-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 249] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 137. | Hoshino A, Ariyoshi M, Okawa Y, Kaimoto S, Uchihashi M, Fukai K, Iwai-Kanai E, Ikeda K, Ueyama T, Ogata T, Matoba S. Inhibition of p53 preserves Parkin-mediated mitophagy and pancreatic β-cell function in diabetes. Proc Natl Acad Sci U S A. 2014;111:3116-3121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 197] [Article Influence: 16.4] [Reference Citation Analysis (1)] |
| 138. | Lai TC, Chen YC, Cheng HH, Lee TL, Tsai JS, Lee IT, Peng KT, Lee CW, Hsu LF, Chen YL. Combined exposure to fine particulate matter and high glucose aggravates endothelial damage by increasing inflammation and mitophagy: the involvement of vitamin D. Part Fibre Toxicol. 2022;19:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 139. | Sidarala V, Pearson GL, Parekh VS, Thompson B, Christen L, Gingerich MA, Zhu J, Stromer T, Ren J, Reck EC, Chai B, Corbett JA, Mandrup-Poulsen T, Satin LS, Soleimanpour SA. Mitophagy protects β cells from inflammatory damage in diabetes. JCI Insight. 2020;5:e141138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 140. | Zhao W, Zhao B, Meng X, Li B, Wang Y, Yu F, Fu C, Yu X, Li X, Dai C, Wang J, Gao H, Cheng M. The regulation of MFG-E8 on the mitophagy in diabetic sarcopenia via the HSPA1L-Parkin pathway and the effect of D-pinitol. J Cachexia Sarcopenia Muscle. 2024;15:934-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 141. | Serikbaeva A, Li Y, Ganesh B, Zelkha R, Kazlauskas A. Hyperglycemia Promotes Mitophagy and Thereby Mitigates Hyperglycemia-Induced Damage. Am J Pathol. 2022;192:1779-1794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 142. | Du Q, Wu X, Ma K, Liu W, Liu P, Hayashi T, Mizuno K, Hattori S, Fujisaki H, Ikejima T. Silibinin alleviates ferroptosis of rat islet β cell INS-1 induced by the treatment with palmitic acid and high glucose through enhancing PINK1/parkin-mediated mitophagy. Arch Biochem Biophys. 2023;743:109644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 30] [Reference Citation Analysis (1)] |
| 143. | Van Huynh T, Rethi L, Rethi L, Chen CH, Chen YJ, Kao YH. The Complex Interplay between Imbalanced Mitochondrial Dynamics and Metabolic Disorders in Type 2 Diabetes. Cells. 2023;12:1223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 144. | Zhang L, Lin H, Yang X, Shi J, Sheng X, Wang L, Li T, Quan H, Zhai X, Li W. Effects of dapagliflozin monotherapy and combined aerobic exercise on skeletal muscle mitochondrial quality control and insulin resistance in type 2 diabetes mellitus rats. Biomed Pharmacother. 2023;169:115852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 145. | Zhang Y, Zou R, Abudureyimu M, Liu Q, Ma J, Xu H, Yu W, Yang J, Jia J, Qian S, Wang H, Yang Y, Wang X, Fan X, Ren J. Mitochondrial aldehyde dehydrogenase rescues against diabetic cardiomyopathy through GSK3β-mediated preservation of mitochondrial integrity and Parkin-mediated mitophagy. J Mol Cell Biol. 2024;15:mjad056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 146. | Durga Devi T, Babu M, Mäkinen P, Kaikkonen MU, Heinaniemi M, Laakso H, Ylä-Herttuala E, Rieppo L, Liimatainen T, Naumenko N, Tavi P, Ylä-Herttuala S. Aggravated Postinfarct Heart Failure in Type 2 Diabetes Is Associated with Impaired Mitophagy and Exaggerated Inflammasome Activation. Am J Pathol. 2017;187:2659-2673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 65] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 147. | Zhou H, Du W, Li Y, Shi C, Hu N, Ma S, Wang W, Ren J. Effects of melatonin on fatty liver disease: The role of NR4A1/DNA-PKcs/p53 pathway, mitochondrial fission, and mitophagy. J Pineal Res. 2018;64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 247] [Article Influence: 30.9] [Reference Citation Analysis (1)] |
| 148. | Watada H, Fujitani Y. Minireview: Autophagy in pancreatic β-cells and its implication in diabetes. Mol Endocrinol. 2015;29:338-348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 149. | Liu J, Liu L, Han YS, Yi J, Guo C, Zhao HQ, Ling J, Wang YH. The molecular mechanism underlying mitophagy-mediated hippocampal neuron apoptosis in diabetes-related depression. J Cell Mol Med. 2021;25:7342-7353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 150. | Wang Y, He X, Xue M, Sun W, He Q, Jin J. Germacrone protects renal tubular cells against ferroptotic death and ROS release by re-activating mitophagy in diabetic nephropathy. Free Radic Res. 2023;57:413-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 151. | Zhong Y, Jin R, Luo R, Liu J, Ren L, Zhang Y, Shan Z, Peng X. Diosgenin Targets CaMKK2 to Alleviate Type II Diabetic Nephropathy through Improving Autophagy, Mitophagy and Mitochondrial Dynamics. Nutrients. 2023;15:3554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |
| 152. | Audzeyenka I, Rachubik P, Typiak M, Kulesza T, Topolewska A, Rogacka D, Angielski S, Saleem MA, Piwkowska A. Hyperglycemia alters mitochondrial respiration efficiency and mitophagy in human podocytes. Exp Cell Res. 2021;407:112758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 153. | Gupta P, Sharma G, Lahiri A, Barthwal MK. FOXO3a acetylation regulates PINK1, mitophagy, inflammasome activation in murine palmitate-conditioned and diabetic macrophages. J Leukoc Biol. 2022;111:611-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 154. | Tang Y, Liu J, Long J. Phosphatase and tensin homolog-induced putative kinase 1 and Parkin in diabetic heart: Role of mitophagy. J Diabetes Investig. 2015;6:250-255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 155. | Qu M, Zuo L, Zhang M, Cheng P, Guo Z, Yang J, Li C, Wu J. High glucose induces tau hyperphosphorylation in hippocampal neurons via inhibition of ALKBH5-mediated Dgkh m(6)A demethylation: a potential mechanism for diabetic cognitive dysfunction. Cell Death Dis. 2023;14:385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |
| 156. | Hu Y, Zhou Y, Yang Y, Tang H, Si Y, Chen Z, Shi Y, Fang H. Metformin Protects Against Diabetes-Induced Cognitive Dysfunction by Inhibiting Mitochondrial Fission Protein DRP1. Front Pharmacol. 2022;13:832707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 157. | Wei PZ, Szeto CC. Mitochondrial dysfunction in diabetic kidney disease. Clin Chim Acta. 2019;496:108-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 164] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 158. | Mao C, Hu C, Zhou Y, Zou R, Li S, Cui Y, Tian W. Electroacupuncture Pretreatment against Cerebral Ischemia/Reperfusion Injury through Mitophagy. Evid Based Complement Alternat Med. 2020;2020:7486041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 159. | Zhang Y, Tan X, Cao Y, An X, Chen J, Yang L. Punicalagin Protects against Diabetic Liver Injury by Upregulating Mitophagy and Antioxidant Enzyme Activities. Nutrients. 2022;14:2782. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 160. | Soleimanpour SA, Gupta A, Bakay M, Ferrari AM, Groff DN, Fadista J, Spruce LA, Kushner JA, Groop L, Seeholzer SH, Kaufman BA, Hakonarson H, Stoffers DA. The diabetes susceptibility gene Clec16a regulates mitophagy. Cell. 2014;157:1577-1590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 175] [Article Influence: 14.6] [Reference Citation Analysis (1)] |
| 161. | Seabright AP, Fine NHF, Barlow JP, Lord SO, Musa I, Gray A, Bryant JA, Banzhaf M, Lavery GG, Hardie DG, Hodson DJ, Philp A, Lai YC. AMPK activation induces mitophagy and promotes mitochondrial fission while activating TBK1 in a PINK1-Parkin independent manner. FASEB J. 2020;34:6284-6301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 120] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 162. | Bai T, Wang F, Zheng Y, Liang Q, Wang Y, Kong J, Cai L. Myocardial redox status, mitophagy and cardioprotection: a potential way to amend diabetic heart? Clin Sci (Lond). 2016;130:1511-1521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 163. | Guo F, Wang W, Song Y, Wu L, Wang J, Zhao Y, Ma X, Ji H, Liu Y, Li Z, Qin G. LncRNA SNHG17 knockdown promotes Parkin-dependent mitophagy and reduces apoptosis of podocytes through Mst1. Cell Cycle. 2020;19:1997-2006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 164. | Ketenci M, Zablocki D, Sadoshima J. Mitochondrial Quality Control Mechanisms during Diabetic Cardiomyopathy. JMA J. 2022;5:407-415. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 165. | Yan LJ. NADH/NAD(+) Redox Imbalance and Diabetic Kidney Disease. Biomolecules. 2021;11:730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 166. | Yu H, Liu Y, He T, Zhang Y, He J, Li M, Jiang B, Gao Y, Chen C, Ke D, Liu J, He B, Yang X, Wang JZ. Platelet biomarkers identifying mild cognitive impairment in type 2 diabetes patients. Aging Cell. 2021;20:e13469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 167. | Li G, Jian Z, Wang H, Xu L, Zhang T, Song J. Irisin Promotes Osteogenesis by Modulating Oxidative Stress and Mitophagy through SIRT3 Signaling under Diabetic Conditions. Oxid Med Cell Longev. 2022;2022:3319056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 168. | Yang M, Zhang Q, Luo S, Han Y, Zhao H, Jiang N, Liu Y, Li L, Li C, Liu C, He L, Zhu X, Liu Y, Sun L. DsbA-L alleviates tubular injury in diabetic nephropathy by activating mitophagy through maintenance of MAM integrity. Clin Sci (Lond). 2023;137:931-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 169. | Zhou L, Su W, Wang Y, Zhang Y, Xia Z, Lei S. FOXO1 reduces STAT3 activation and causes impaired mitochondrial quality control in diabetic cardiomyopathy. Diabetes Obes Metab. 2024;26:732-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 170. | Rodriguez YA, Kaur S, Nolte E, Zheng Z, Blagg BSJ, Dobrowsky RT. Novologue Therapy Requires Heat Shock Protein 70 and Thioredoxin-Interacting Protein to Improve Mitochondrial Bioenergetics and Decrease Mitophagy in Diabetic Sensory Neurons. ACS Chem Neurosci. 2021;12:3049-3059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 171. | Anderson A, Alfahad N, Wimalachandra D, Bouzinab K, Rudzinska P, Wood H, Fazey I, Xu H, Lyons TJ, Barnes NM, Narendran P, Lord JM, Rauz S, Ganley IG, Curtis TM, Wallace GR, Hombrebueno JR. Relaxation of mitochondrial hyperfusion in the diabetic retina via N6-furfuryladenosine confers neuroprotection regardless of glycaemic status. Nat Commun. 2024;15:1124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 172. | Zhijun K, Xudong Z, Baoqiang W, Chunfu Z, Qiang Y, Yuan G, Xihu Q. Increased oxidative stress caused by impaired mitophagy aggravated liver ischemia and reperfusion injury in diabetic mice. J Diabetes Investig. 2023;14:28-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 173. | Torres-Esquivel C, Montiel T, Flores-Méndez M, Massieu L. Effect of β-Hydroxybutyrate on Autophagy Dynamics During Severe Hypoglycemia and the Hypoglycemic Coma. Front Cell Neurosci. 2020;14:547215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 174. | Chen K, Jin HJ, Wu ZH, Zhang BF, Wu J, Huang ZY, Huang YP, Lu XW, Zheng XT. Glucagon-like peptide-1 receptor agonist exendin 4 ameliorates diabetes-associated vascular calcification by regulating mitophagy through the AMPK signaling pathway. Mol Med. 2024;30:58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 175. | Ji H, Zhao Y, Ma X, Wu L, Guo F, Huang F, Song Y, Wang J, Qin G. Upregulation of UHRF1 Promotes PINK1-mediated Mitophagy to Alleviates Ferroptosis in Diabetic Nephropathy. Inflammation. 2024;47:718-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |
| 176. | Wang X, Song M, Li X, Su C, Yang Y, Wang K, Liu C, Zheng Z, Jia Y, Ren S, Dong W, Chen J, Wang T, Liu L, Guan M, Zhang C, Xue Y. CERS6-derived ceramides aggravate kidney fibrosis by inhibiting PINK1-mediated mitophagy in diabetic kidney disease. Am J Physiol Cell Physiol. 2023;325:C538-C549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 30] [Reference Citation Analysis (1)] |
| 177. | Singh LP, Devi TS, Yumnamcha T. The Role of Txnip in Mitophagy Dysregulation and Inflammasome Activation in Diabetic Retinopathy: A New Perspective. JOJ Ophthalmol. 2017;4:555643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 178. | Guo H, Rogg M, Keller J, Scherzinger AK, Jäckel J, Meyer C, Sammarco A, Helmstädter M, Gorka O, Groß O, Schell C, Bechtel-Walz W. ADP-Ribosylation Factor-Interacting Protein 2 Acts as a Novel Regulator of Mitophagy and Autophagy in Podocytes in Diabetic Nephropathy. Antioxidants (Basel). 2024;13:81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 179. | Sekiya FS, Silva CPND, Oba-Shinjo SM, Santos-Bezerra DP, Ravagnani FG, Pasqualucci CA, Gil S, Gualano B, Baptista MDS, Correa-Giannella ML, Marie SKN. Identification of two patterns of mitochondrial DNA-copy number variation in postcentral gyrus during aging, influenced by body mass index and type 2 diabetes. Exp Gerontol. 2022;168:111932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 180. | Kakoti BB, Alom S, Deka K, Halder RK. AMPK pathway: an emerging target to control diabetes mellitus and its related complications. J Diabetes Metab Disord. 2024;23:441-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 181. | Huang C, Huang L, Huang Q, Lin L, Wang L, Wu Y, Wu K, Gao R, Liu X, Liu X, Qi L, Liu L. Mitophagy disorder mediates cardiac deterioration induced by severe hypoglycemia in diabetic mice. Mol Cell Endocrinol. 2023;575:111994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 182. | Jang BG, Choi BY, Kim JH, Kim MJ, Sohn M, Suh SW. Impairment of autophagic flux promotes glucose reperfusion-induced neuro2A cell death after glucose deprivation. PLoS One. 2013;8:e76466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 183. | Zheng D, Chen L, Li G, Jin L, Wei Q, Liu Z, Yang G, Li Y, Xie X. Fucoxanthin ameliorated myocardial fibrosis in STZ-induced diabetic rats and cell hypertrophy in HG-induced H9c2 cells by alleviating oxidative stress and restoring mitophagy. Food Funct. 2022;13:9559-9575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 184. | Khan I, Preeti K, Kumar R, Kumar Khatri D, Bala Singh S. Piceatannol promotes neuroprotection by inducing mitophagy and mitobiogenesis in the experimental diabetic peripheral neuropathy and hyperglycemia-induced neurotoxicity. Int Immunopharmacol. 2023;116:109793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 185. | Ding J, Shi Q, Tao Q, Su H, Du Y, Pan T, Zhong X. Correlation between long-term glycemic variability and cognitive function in middle-aged and elderly patients with type 2 diabetes mellitus: a retrospective study. PeerJ. 2023;11:e16698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 186. | Zhao Y, Sun M. Metformin rescues Parkin protein expression and mitophagy in high glucose-challenged human renal epithelial cells by inhibiting NF-κB via PP2A activation. Life Sci. 2020;246:117382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 187. | Sherkhane B, Kalvala AK, Arruri VK, Khatri DK, Singh SB. Renoprotective potential of myo-inositol on diabetic kidney disease: Focus on the role of the PINK1/Parkin pathway and mitophagy receptors. J Biochem Mol Toxicol. 2022;36:e23032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 188. | Dai W, Lu H, Chen Y, Yang D, Sun L, He L. The Loss of Mitochondrial Quality Control in Diabetic Kidney Disease. Front Cell Dev Biol. 2021;9:706832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 189. | Liu S, Zhang R, Zhang L, Yang A, Guo Y, Jiang L, Wang H, Xu S, Zhou H. Oxidative stress suppresses PHB2-mediated mitophagy in β-cells via the Nrf2/PHB2 pathway. J Diabetes Investig. 2024;15:559-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 190. | Sun D, Wang J, Toan S, Muid D, Li R, Chang X, Zhou H. Molecular mechanisms of coronary microvascular endothelial dysfunction in diabetes mellitus: focus on mitochondrial quality surveillance. Angiogenesis. 2022;25:307-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 191. | Sharma I, Yadav KS, Mugale MN. Redoxisome and diabetic retinopathy: Pathophysiology and therapeutic interventions. Pharmacol Res. 2022;182:106292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 192. | Wang F, Bai J, Zhang X, Wang D, Zhang X, Xue J, Chen H, Wang S, Chi B, Li J, Ma X. METTL3/YTHDF2 m6A axis mediates the progression of diabetic nephropathy through epigenetically suppressing PINK1 and mitophagy. J Diabetes Investig. 2024;15:288-299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 193. | Chen Y, Liu X, Liu Y, Li Y, Li D, Mei Z, Deng Y. Mitochondrial quality control in diabetes mellitus and complications: molecular mechanisms and therapeutic strategies. Cell Death Dis. 2025;16:652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 194. | Pu K, Yang S, Sheng R, Chen J, Dai Y, Wood IC, Zhong Z, Xu S. Chuanxiong-Danggui herb pair alleviated cognitive deficits of APP/PS1 mice by promoting mitophagy. J Ethnopharmacol. 2025;350:119988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 195. | Yu W, Gao B, Li N, Wang J, Qiu C, Zhang G, Liu M, Zhang R, Li C, Ji G, Zhang Y. Sirt3 deficiency exacerbates diabetic cardiac dysfunction: Role of Foxo3A-Parkin-mediated mitophagy. Biochim Biophys Acta Mol Basis Dis. 2017;1863:1973-1983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 219] [Article Influence: 21.9] [Reference Citation Analysis (1)] |
| 196. | Zheng H, Li W, Huang G, Zhu H, Wen W, Liu X, Sun L, Ma T, Huang X, Hu Y, Huang Y. Secreted frizzled-related protein 2 ameliorates diabetic cardiomyopathy by activating mitophagy. Biochim Biophys Acta Mol Basis Dis. 2024;1870:166989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 197. | Yang J, Yu Z, Jiang Y, Zhang Z, Tian Y, Cai J, Wei M, Lyu Y, Yang D, Shen S, Xing GG, Li M. SIRT3 alleviates painful diabetic neuropathy by mediating the FoxO3a-PINK1-Parkin signaling pathway to activate mitophagy. CNS Neurosci Ther. 2024;30:e14703. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 30] [Reference Citation Analysis (1)] |
| 198. | Luo Y, Zhang L, Su N, Liu L, Zhao T. YME1L-mediated mitophagy protects renal tubular cells against cellular senescence under diabetic conditions. Biol Res. 2024;57:10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 199. | Wu Y, Deng H, Sun J, Tang J, Li X, Xu Y. Poricoic acid A induces mitophagy to ameliorate podocyte injury in diabetic kidney disease via downregulating FUNDC1. J Biochem Mol Toxicol. 2023;37:e23503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 200. | Sheng J, Li H, Dai Q, Lu C, Xu M, Zhang J, Feng J. NR4A1 Promotes Diabetic Nephropathy by Activating Mff-Mediated Mitochondrial Fission and Suppressing Parkin-Mediated Mitophagy. Cell Physiol Biochem. 2018;48:1675-1693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 201. | Wang S, Zhao Z, Fan Y, Zhang M, Feng X, Lin J, Hu J, Cheng Z, Sun C, Liu T, Xiong Z, Yang Z, Wang H, Sun D. Mst1 inhibits Sirt3 expression and contributes to diabetic cardiomyopathy through inhibiting Parkin-dependent mitophagy. Biochim Biophys Acta Mol Basis Dis. 2019;1865:1905-1914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 79] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 202. | Tian W, Zhu M, Zhou Y, Mao C, Zou R, Cui Y, Li S, Zhu J, Hu C. Electroacupuncture Pretreatment Alleviates Cerebral Ischemia-Reperfusion Injury by Regulating Mitophagy via mTOR-ULK1/FUNDC1 Axis in Rats. J Stroke Cerebrovasc Dis. 2022;31:106202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 203. | Song Z, Yan C, Zhan Y, Wang Q, Zhang Y, Jiang T. Melatonin attenuates lung ischemia-reperfusion injury through SIRT3 signaling-dependent mitophagy in type 2 diabetic rats. Exp Lung Res. 2023;49:101-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 204. | Sobrevia L, Valero P, Grismaldo A, Villalobos-Labra R, Pardo F, Subiabre M, Armstrong G, Toledo F, Vega S, Cornejo M, Fuentes G, Marín R. Mitochondrial dysfunction in the fetoplacental unit in gestational diabetes mellitus. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 205. | Ko YS, Jin H, Park SW, Kim HJ. Salvianolic acid B protects against oxLDL-induced endothelial dysfunction under high-glucose conditions by downregulating ROCK1-mediated mitophagy and apoptosis. Biochem Pharmacol. 2020;174:113815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 206. | Feng W, Lv C, Cheng L, Song X, Li X, Xie H, Chen S, Wang X, Xue L, Zhang C, Kou J, Wang L, Zhao H. Targeting ERS-mitophagy in hippocampal neurons to explore the improvement of memory by tea polyphenols in aged type 2 diabetic rats. Free Radic Biol Med. 2024;213:293-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 207. | Sadoshima J. Alternative mitophagy is a major form of mitophagy in the chronically stressed heart. Autophagy. 2022;18:2252-2253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 208. | Wang S, Zhao Z, Feng X, Cheng Z, Xiong Z, Wang T, Lin J, Zhang M, Hu J, Fan Y, Reiter RJ, Wang H, Sun D. Melatonin activates Parkin translocation and rescues the impaired mitophagy activity of diabetic cardiomyopathy through Mst1 inhibition. J Cell Mol Med. 2018;22:5132-5144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 109] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 209. | Shim JY, Chung JO, Jung D, Kang PS, Park SY, Kendi AT, Lowe VJ, Lee S. Parkin-mediated mitophagy is negatively regulated by FOXO3A, which inhibits Plk3-mediated mitochondrial ROS generation in STZ diabetic stress-treated pancreatic β cells. PLoS One. 2023;18:e0281496. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 210. | Tabassum S, Wu S, Lee CH, Yang BSK, Gusdon AM, Choi HA, Ren XS. Mitochondrial-targeted therapies in traumatic brain injury: From bench to bedside. Neurotherapeutics. 2025;22:e00515. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 211. | Li H, Wang Y, Su X, Wang Q, Zhang S, Sun W, Zhang T, Dong M, Zhang Z, Lv S. San-Huang-Yi-Shen Capsule Ameliorates Diabetic Kidney Disease through Inducing PINK1/Parkin-Mediated Mitophagy and Inhibiting the Activation of NLRP3 Signaling Pathway. J Diabetes Res. 2022;2022:2640209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 212. | Rabinovich-Nikitin I, Dhingra R, Kirshenbaum LA. Activation of Mitophagy in High-Fat Diet-Induced Diabetic Cardiomyopathy. Circ Res. 2019;124:1288-1290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |