Published online Jun 15, 2021. doi: 10.4239/wjd.v12.i6.767
Peer-review started: December 2, 2020
First decision: December 24, 2020
Revised: December 29, 2020
Accepted: March 7, 2021
Article in press: March 7, 2021
Published online: June 15, 2021
Processing time: 183 Days and 23.3 Hours
A large amount of evidence has supported a clinical link between diabetes and inflammatory diseases, e.g., cancer, dementia, and hypertension. In addition, it is also suggested that dysregulations related to Ca2+ signaling could link these diseases, in addition to 3'-5'-cyclic adenosine monophosphate (cAMP) signaling pathways. Thus, revealing this interplay between diabetes and inflammatory diseases may provide novel insights into the pathogenesis of these diseases. Publications involving signaling pathways related to Ca2+ and cAMP, inflammation, diabetes, dementia, cancer, and hypertension (alone or combined) were collected by searching PubMed and EMBASE. Both signaling pathways, Ca2+ and cAMP signaling, control the release of neurotransmitters and hormones, in addition to neurodegeneration, and tumor growth. Furthermore, there is a clear relationship between Ca2+ signaling, e.g., increased Ca2+ signals, and inflammatory responses. cAMP also regulates pro- and anti-inflammatory responses. Due to the experience of our group in this field, this article discusses the role of Ca2+ and cAMP signaling in the correlation between diabetes and inflammatory diseases, including its pharmacological implications. As a novelty, this article also includes: (1) A timeline of the major events in Ca2+/cAMP signaling; and (2) As coronavirus disease 2019 (COVID-19) is an emerging and rapidly evolving situation, this article also discusses recent reports on the role of Ca2+ channel blockers for preventing Ca2+ signaling disruption due to COVID-19, including the correlation between COVID-19 and diabetes.
Core Tip: There are several reviews in the literature on diabetes and inflammatory diseases. Nonetheless, to my knowledge, this is the first review which clearly discusses the role of Ca2+/3'-5'-cyclic adenosine monophosphate (cAMP) signaling in the link between diabetes and inflammatory diseases. This article also includes a timeline of the major events in Ca2+/cAMP signaling, and discusses recent reports on the role of Ca2+ channel blockers for preventing Ca2+ signaling disruption due to coronavirus disease 2019 (COVID-19), including the correlation between COVID-19 and diabetes.
- Citation: Bergantin LB. Diabetes and inflammatory diseases: An overview from the perspective of Ca2+/3'-5'-cyclic adenosine monophosphate signaling. World J Diabetes 2021; 12(6): 767-779
- URL: https://www.wjgnet.com/1948-9358/full/v12/i6/767.htm
- DOI: https://dx.doi.org/10.4239/wjd.v12.i6.767
The concept of a complex clinical link between diabetes and inflammatory diseases, e.g., cancer, dementia, and hypertension, has been described in several reports[1-12]. Observational reports provided the first evidence for the link between inflammation and diabetes[11]. An interesting study correlated inflammation and diabetes by showing, in animal models, that tumor necrosis factor-alpha is correlated with both obesity and insulin resistance[13]. In addition, an epidemiologic link between inflammation and diabetes was made when circulating concentrations of markers, and mediators, of inflammation were demonstrated to be increased in these diseases[14-17], e.g., white cells, interleukin (IL)-6, fibrinogen, C-reactive protein, plasminogen activator inhibitor-1, and sialic acid. Besides diabetes, the link between cancer and inflammation was proposed in 1863 by Virchow, which postulated that the etiology of cancer is related to chronic inflammation[18]. It is now clear that persistent cell proliferation at sites that are abundant in inflammatory cells, DNA-damage-promoting agents, growth factors, and activated stroma, actually increases the risk of neoplasia[19]. Inflammation also plays a central role in dementia, e.g., Alzheimer’s disease (AD)[20]. For instance, persistent stimulation of immune cells, and macrophages in the brain (microglia), has been shown to aggravate both amyloid and tau pathology, which then may operate as a link in the etiology of the disease[20]. Finally, recent interest has focused on studying the avenues, and inflammatory mediators, through which immune cells operate to lead to hypertension and end-organ injury[21].
Currently, it is also well-known that dyshomeostasis of Ca2+, through an increase in Ca2+ levels within the cells [Ca2+]c, is correlated with the pathogenesis of diabetes, cancer, hypertension, and dementia[9,10,12,22-26]. In fact, there is a clear relationship between Ca2+ signaling, e.g., increased Ca2+ signals, and inflammatory responses[10,27]. Corroborating this idea, several studies have highlighted that Ca2+ channel blockers (CCBs), classic antihypertensive medicines, can improve cognitive function, in addition to decreasing the symptoms of both cancer and diabetes[12,28-31]. A pharmacological tenet for these exciting findings is linked with reestablishing [Ca2+]c, in addition to modulating 3'-5'-cyclic adenosine monophosphate (cAMP) signaling pathways (Ca2+/cAMP signaling)[9,10,24,25]. Due to the experience of our group in this field[9,10,12,24,25,32-36], this article discusses the contributions of Ca2+ and cAMP signaling in the correlation between cancer, hypertension, diabetes, and dementia. Publications involving dementia, diabetes, hypertension, and cancer were obtained by examining PubMed and EMBASE, using a search strategy with a high sensitivity for studies of etiology, as follows: (1) Searches applied the following strings: Risk (in title or abstract) odds ratio (OR) risk [as a Medical Subject Heading (MeSH) term, not exploded] OR cohort studies (as a MeSH term) OR group (as a text word). Outcomes of these searches were linked with sets conceived with diabetes OR dementia OR cancer OR hypertension OR inflammation; and (2) Bibliographies of the articles obtained were also reviewed for possible data sources.
This article included as a novelty: (1) A timeline of the major events in Ca2+/cAMP signaling; and (2) As coronavirus disease 2019 (COVID-19) is an emerging and rapidly evolving situation, this article also discusses recent reports on the role of CCBs for preventing Ca2+ signaling disruption due to COVID-19, including the correlation between COVID-19 and diabetes.
Dementia: Dementia, like AD, and aging are classically associated with each other[10,24,37,38]. Dementia is often characterized by synaptic dysfunction and death of neurons, leading to a gradual decline in cognitive abilities[24,38]. Among the hallmark features of AD, an accumulation of plaques containing the amyloid beta (Aβ) peptide is established; then in the preclinical phase of AD, Aβ is among the most prevalent pathological markers[10,24,38]. Briefly, the amyloid precursor protein (APP) undergoes proteolysis, thus producing Aβ as a product, which can be quantified from both blood and cerebrospinal fluid, and by imaging[10,24]. In addition, Ca2+ dyshomeostasis has also been associated with dementia, resulting in the death of neurons[10,24,33,37]. Aβ has been linked with both an increased [Ca2+]c and an enhanced susceptibility to neuroexcitotoxicity[10,37]. Ca2+ signaling has been investigated due to modulation of neuronal death[37].
Cancer: Like dementia, cancer dramatically affects the health of individuals, and is considered an uncontrolled division of the body’s cells[9,39]. Ca2+ dysregulations have also been observed in carcinogenesis[9,39-44]. For example, several studies corroborated the involvement of Ca2+ channels overexpression, or hyperactivation, in different types of cancer[40-44]. For instance, a Ca2+-binding protein that exerts a protagonist role in intracellular Ca2+ homeostasis is regucalcin (RGN), a protein which was observed to have reduced expression in cancer of the prostate gland[45,46]. It has also been observed that an elevation of [Ca2+]c stimulates diverse responses to the proliferation of cells from both neoplastic and non-neoplastic cancer of the prostate gland, suggesting a correlation with RGN expression[9,45,46]. Furthermore, increased expression of RGN reduced the migration of NSCLC A549 cells from adenocarcinoma of lung in vitro[45,46]. The protein and mRNA expression of RGN was also decreased in (1) HepG2 cells from human hepatoma; (2) MCF-7 cells from breast cancer; and (3) LNCaP cells from prostate cancer[35,36]. Thus, reduced expression of RGN may be correlated with the stimulation of carcinogenesis[45,46]. Besides RGN, there are other Ca2+-binding proteins that regulate Ca2+ homeostasis, which are implicated in cancer. In addition, expression levels of S100B, TM4SF3 and OLFM4 have been discovered to be highly associated with metastasis of liver cancer[9,47]. These findings confirm the participation of Ca2+ dyshomeostasis in cancer, opening new perspectives for the advancement of therapeutics linked to Ca2+ signaling.
Hypertension: Sympathetic hyperactivity, due to dysregulation of Ca2+ signaling as in dementia and cancer, has been correlated with hypertension. In fact, studies by Miranda-Ferreira et al[48,49] validated this principle by demonstrating changes in the kinetics of the release of catecholamines from spontaneously hypertensive rats (SHRs), when compared with normotensive rats. Ca2+ signaling is argued to be an issue involved in these differences. The authors[48,49] reinforced that Ca2+ dyshomeostasis might explain the increased release of catecholamines seen in SHRs, when compared with normotensive rats.
Diabetes: Like dementia, cancer, and hypertension, diabetes is also a serious medical condition. Diabetes is presently categorized according to its origin. For example, in type 1 diabetes if there is a deficiency of insulin released by the pancreas, then it can be categorized as a juvenile diabetes; while in type 2 diabetes if there is resistance to insulin, then it can be categorized as adult-onset diabetes[50]. From a cellular point of view, whereas a physiological increase in the cytoplasmic concentration of Ca2+ is a significant trigger for releasing insulin, an abnormal elevation of [Ca2+]c could stimulate β-cell apoptosis, then decrease insulin levels, contributing to diabetes[9,12,25]. Besides Ca2+, cAMP modulates the release of various hormones, including insulin released from pancreatic β-cells[51,52]. Although increasing cAMP levels, e.g., via adrenaline, might stimulate the production of hepatic glucose, increasing levels of cAMP in pancreatic β-cells may stimulate insulin release. The start signal for release of insulin is achieved by elevating [Ca2+]c, and this signal is later amplified via cAMP[53]. Additionally, cAMP is also implicated in other cellular phenomena of β-cells, e.g., inhibiting apoptosis[53].
Nowadays, a clinical link between these discussed diseases (hypertension, cancer, diabetes, and dementia) has been described in several reports[1-12,25,26].
Cancer and dementia: A link between dementia and cancer can be established through numerous cellular phenomena that are implicated in the etiology of both diseases, e.g., inflammation, oxidative stress, and angiogenesis[1,3,54]. For instance, inflammatory biomarkers linked with a lower cognitive performance have been shown to be increased, including fibrinogen and IL-6[1-3]. In fact, several proteins may be involved in the etiology of this link, e.g., Aβ peptide[1-3]. It is suggested that Aβ peptide overexpression is correlated with cancer as the overexpression of APP has been found in several tumors, and was then linked with cell proliferation, migration, and invasion[1]. In addition, a recognized tumor suppressor protein, the BRCA1 protein, has been linked to AD[2]. Thus, Aβ pathology can partially result from overactivation of BRCA1, and then promote neurodegeneration[2]. Converging with this concept, plasma levels of Aβ peptide have been found to be increased in patients with different cancer types[3].
In addition, a deficiency in DNA repair mechanisms and/or oxidative stress could lead to DNA damage, an issue which is also important for the etiology of both cancer and dementia[55]. A reduced capacity to repair damaged DNA due to genetic polymorphisms can be correlated to an augmented risk of both cancer and cognitive impairment. Genetic defects in DNA damage repair mechanisms could lead to syndromes such as xeroderma pigmentosum and ataxia telangiectasia, characterized by an augmented risk of cancer and cognitive problems[55]. Thus, understanding the clinical link between cancer and dementia could result in novel therapeutics for both diseases. Therefore, it is essential to determine the etiology of this link, e.g., by analyzing the preclinical phases of both diseases.
Hypertension and cancer: A correlation between hypertension and a higher incidence of cancer has been established by epidemiological and clinical reports[4-8]. However, this correlation is not completely elucidated, and has been highly discussed. For example, the Metabolic Syndrome and Cancer Project assessed this issue, and included cohort studies from Norway, Austria, and Sweden[4]. The goal of the Metabolic Syndrome and Cancer Project was to study the association between metabolic issues and the increased incidence of cancer[4,5]. Patients from cohorts related to the Metabolic Syndrome and Cancer Project were enrolled in health inspections between the 1970’s and 2000’s[4]. The study observed a strong correlation between hypertension and an enhanced incidence of prostate, oropharynx, rectum, pancreas, bladder, lung, and kidney cancer[4]. Additionally, strong correlations between hypertension and an enhanced incidence of pancreas, breast, corpus uteri cancer and malignant melanoma were observed in women. A positive correlation was also observed for esophagus cancer in men and women[4,5]. Indeed, cancer incidence is augmented linearly by increasing blood pressure levels[4-6]. The augmentation of cancer incidence among men was 1% to 2% points higher in hypertensive patients, compared with normotensive men[4-6].
Finally, in observational reports on renal cell carcinoma, hypertension has been documented as a cancer risk factor[4-8]. A meta-analysis of 18 studies observed a 1.6-fold increase in the incidence of renal cell carcinoma in hypertensive patients[7]. Nonetheless, this positive association between hypertension and increased cancer incidence could occur in other disorders, such as obesity[7,8]. Moreover, CCBs, antihypertensive drugs which decrease the influx of Ca2+ into the cells, have shown anti-cancer activity[31].
Diabetes and hypertension: Diabetes is correlated with a higher risk of developing hypertension[56]. Several findings reinforce a clear interaction between diabetes and hypertension[57]. Scientific data suggest that obesity, inflammation, oxidative stress, and insulin resistance could be associated with these diseases[56,57]. Advances in the knowledge of how to prevent these diseases may provide new insights, and perspectives, for the treatment of both diseases.
In fact, hypertension and diabetes are highlighted as the leading risk factors for atherosclerosis, including heart attacks and strokes[58]. For instance, in the Hong Kong Cardiovascular Risk Factor Prevalence report, just 42% of patients with diabetes had regular blood pressure, and just 56% of patients with hypertension had regular glucose homeostasis[58]. In the United States, patients with type 2 diabetes have a prevalence of hypertension ranging from approximately 50% to 80%[59]. In fact, a prospective cohort report from the United States concluded that hypertensive patients had an increased risk of almost 2.5-fold for developing type 2 diabetes[60]. It is clear that diabetes and hypertension are present in the same individual more often than would occur by causality, suggesting both common genetic and environmental factors in their etiology.
Diabetes and dementia: The concept of an association between diabetes and memory dysfunctions has been frequently explored[50,61-63]. Type 2 diabetes has been linked with a decrease in both processing and speed of psychomotor functioning, in addition to a memory deficit associated with speech and fluency[61-63]. Additionally, patients with diabetes have a lengthier walking pace[61-63]. In fact, mild cognitive impairment was observed in approximately 42% of diabetic patients[64]. The association between diabetes and cognitive imbalance was examined in a report[65], and the authors concluded that patients with type 2 diabetes had a lower score in the Mini-Mental State Examination[65]. An interesting study[66] assessed if lesions in the brain, associated with both vascular and degenerative disorders, could be the cause of the association between diabetes and cognitive deficit[66]. The authors concluded that memory performance in diabetic patients was significantly reduced[66].
In addition, these findings were confirmed by reports involving neuroimaging[67]. Brain atrophy was concluded to be highly correlated with type 2 diabetes[67], which usually progresses up to 3 times faster[68,69]. Patients suffering from type 2 diabetes have also demonstrated an enhanced incidence of dementia, e.g., AD[70,71]. In fact, approximately 17.5% of people with type 2 diabetes have shown a modest to a serious deficiency in day-to-day activities[72,73], while 11.3% have shown a loss of cognition, and 14.2% have shown symptoms of depression[74], consequently adversely influencing cognition[75].
A decrease in brain glucose metabolism manifested before the beginning of a quantifiable cognitive decline in cohorts of patients at risk of AD[76-79]. Reports from in vitro and animal experiments propose that a decrease in brain glucose metabolism comes first and, therefore, may stimulate the neuropathologic cascade, finally resulting in cognitive decline in AD[76,77]. Additionally, aging is associated with an augmented risk of worsening glucose homeostasis, which, in turn, may increase the risk of decreasing brain glucose uptake[76,79]. Thus, pharmacotherapy to decrease the risk of AD could provide the following: (1) Improve insulin sensitivity, and then restore glucose homeostasis; or (2) Reduce the decline in brain glucose metabolism by applying strategies that carefully stimulate a maintainable ketonemia[76,79].
Diabetes and cancer: A relationship between diabetes and an increased risk of several types of cancer has also been described[9,18,19]. It is now clear that persistent cell proliferation in conditions with abundant inflammatory cells, growth factors, stimulated stroma, and DNA-damage-promoting molecules, increases neoplastic risk[19]. In addition, an inflammatory process is also involved in the etiology of diabetes[17], thus it could be an issue which could link both diseases.
In fact, in laboratory reports, metformin, the most frequently used drug in patients with type 2 diabetes, has been demonstrated to prevent cell proliferation and decrease colony development, causing partial cell cycle arrest in cancer cell phenotypes[9,19]; thus, it is important not to neglect the disruption of glucose homeostasis as a relevant mediator of this link between diabetes and cancer.
Hypertension and dementia: Midlife hypertension (aged 40-64 years) increases the incidence of AD in later life (≥ 65 years); and hypertension has been linked with increased amyloid deposition and neurofibrillary tangles, both hallmarks of AD[12]. Indeed, the brain of patients with hypertension, when compared with normotensive patients, had higher concentrations of β-amyloid plaques, atrophy, and neurofibrillary tangles[12,20]. Thus, hypertension has been recognized as a risk factor for the deposition of cortical fibrillar β-amyloid[12].
Diabetes and COVID-19: COVID-19, triggered by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is an emerging and rapidly evolving situation[80,81]. It is accepted that angiotensin-converting enzyme 2, which is a component of the renin-angiotensin-aldosterone system, is the key entry receptor for SARS-CoV-2[80,81].
Intriguingly, some reports have observed increased severity of COVID-19 in patients with diabetes. To assess this issue, a meta-analysis[80] was performed by conducting a literature review of Scopus, PubMed, Science Direct, and Web of Science. Observational studies, case-reports, and case-series reports that analyzed diabetes in COVID-19 patients were included in the meta-analysis[80]. It was concluded that diabetes is a risk factor, and plays a role in disease severity and the mortality of individuals with COVID-19. This study also provided suggestions, and guidelines, which could be helpful for the prevention and treatment of diabetic patients suffering from COVID-19[80]. As already discussed in the present article, hyperglycemia may modulate both immune and inflammatory processes, thus prejudicing patients and resulting in severe COVID-19, and possible fatal consequences.
In addition, a retrospective clinical report involving hospitalized patients with COVID-19 and hypertension, concluded that therapy with the CCB amlodipine besylate was correlated with decreased mortality[82]. In fact, CCBs were described to possess antiviral activity against several evolving viruses, including bunyaviruses, arenaviruses, and flaviviruses[82]. Furthermore, CCBs were described to possess anti-inflammatory ability to control patients’ intracellular Ca2+ levels, including decreasing the death rate in septic animal models with severe inflammatory outcomes[83,84]. Severe inflammatory outcomes are described to be linked to a critical COVID-19 result[85]. Thus, it is plausible that CCBs may operate in a synergistic way by combining their antiviral efficacy with alleviation of inflammatory responses[85].
Complementing the present discussion, and in addition to Ca2+ signaling, the participation of cAMP signaling (Ca2+/cAMP signaling) in the correlation between cancer, hypertension, diabetes, and dementia is considered.
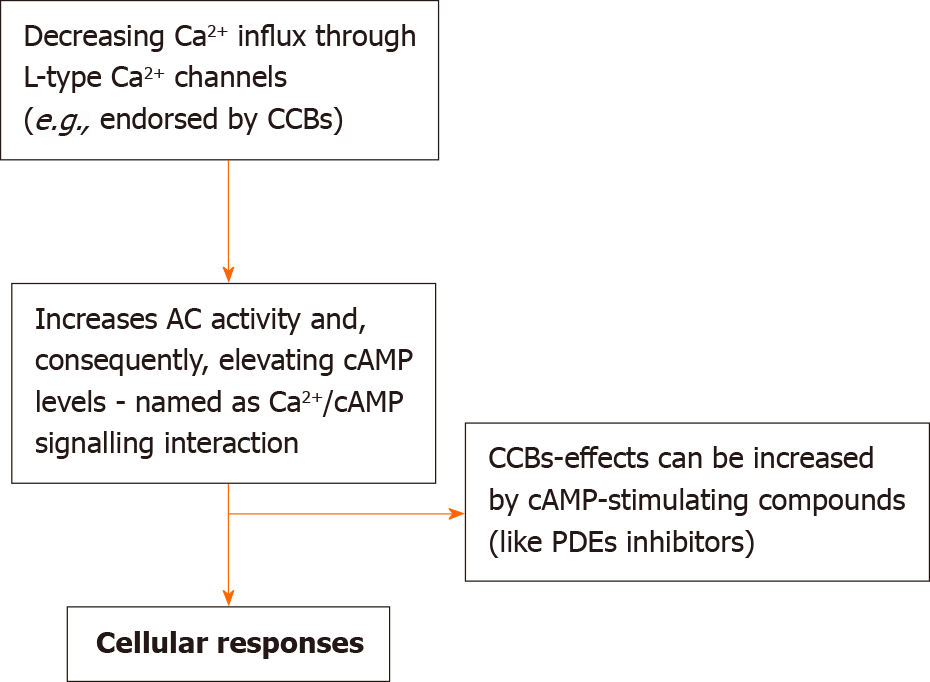
Our reports on Ca2+/cAMP signaling have recognized the participation of these cellular processes in regulating the release of both neurotransmitters and hormones, as well as the death of neurons and tumor growth[9,10,12,24,25,32-36,86,87]. Our studies proved that by decreasing the influx of Ca2+ via voltage activated Ca2+ channels (VACCs), adenylyl cyclases (ACs) are stimulated (thus increasing the levels of cAMP, and the Ca2+/cAMP signaling interaction, Figure 1).
Considering this working model, CCBs-responses can be significantly increased through their pharmaceutical association with cAMP-enhancer agents [such as phosphodiesterase inhibitors]. The working model through which the release of both transmitter and hormone can be significantly augmented by regulating Ca2+/cAMP signaling is related to (1) elevating the concentrations of transmitters and hormones in the secretory apparatus; and (2) enhancing the release of transmitters and hormones[10,12]. Actually, Ca2+ signaling is essential for supporting the release process: Via rising cAMP levels, this can augment the release of Ca2+ from endoplasmic reticulum (ER), thus increasing the release of transmitters and hormones. The timeline of the major events in Ca2+/cAMP signaling can be found below (Table 1).
| 1970s | 1980s and 1990s | 2000s and 2010s | 2019-2020 | |
| Major events | Verapamil paradoxically enhanced the contractions of smooth muscles, e.g., rat vas deferens | Other CCBs (besides verapamil) also paradoxically enhanced the contractions of smooth muscles, e.g., rat vas deferens | 2013. Bergantin et al[32] discovered that the paradoxical increase in the contractions of smooth muscles, produced by CCBs, was due to an interaction of Ca2+/cAMP signaling | Bergantin[12,25,86,87] discussed the involvement of Ca2+/cAMP signaling in the pathogenesis of several diseases, including hypertension, diabetes, neurodegenerative diseases, asthma, and cancer |
| 2015-2016. Bergantin et al[24] proposed that the pharmacological manipulation of Ca2+/cAMP signaling could be a new therapeutic strategy for increasing neurotransmission in psychiatric disorders, and producing neuroprotection in neurodegenerative diseases | ||||
| Articles indexed in PubMed (PMID) | PMID: 1143442 | PMID: 3113986; PMID: 2466518 | PMID: 23849429; PMID: 26516591; PMID: 27349146 | PMID: 30117399; PMID: 30639385; PMID: 30771427; PMID: 30648516; PMID: 31291877; PMID: 31456527; PMID: 31995022; PMID: 32077833; PMID: 32186273; PMID: 32026774; PMID: 32065096; PMID: 32562933; PMID: 33210037; PMID: 33176668 |
Additionally, a higher [Ca2+]c from critical dysregulations of Ca2+ signaling, such as an enhanced Ca2+ influx, has been linked to dementia, diabetes, hypertension, and cancer[9,10,12,24,25]. For instance, it was observed that L-type Ca2+ channels are significantly up-regulated in different types of cancer cells, contributing to abnormal cell proliferation[9,40-44]. The pharmaceutical modulation of these channels could then improve the therapeutics for antitumor purposes.
As well as cancer, dysregulations related to aging have also been detected in Ca2+ signaling pathways, stimulating the death of neurons, e.g., an increase in intracellular Ca2+ levels, an augmented Ca2+ influx via the VACC and abnormalities in Ca2+ regulation in ryanodine and IP3-sensitive Ca2+ stores[10,12,24].
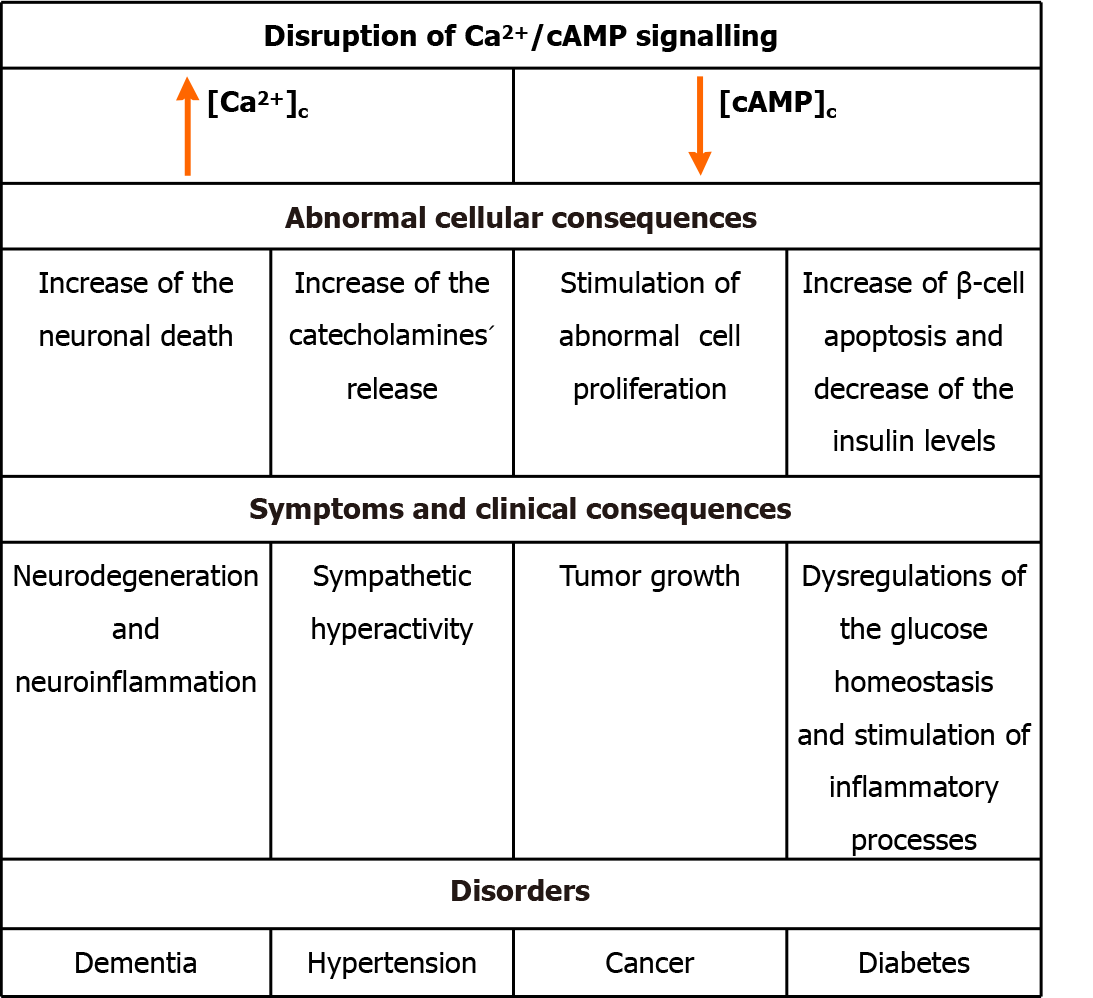
Opposing Ca2+ signaling, stimulation of cAMP/protein kinase/cAMP-response element binding protein pathways can reduce both neuronal death and abnormal cell proliferation, thus resulting in anti-cancer and anti-dementia effects[88-91]. Thus, both the death of neurons and abnormal cell proliferation may also be a consequence of reduced activity of signaling pathways controlled by cAMP, as well as an increase in [Ca2+]c, resulting from disruption of Ca2+/cAMP signaling interactions. Indeed, there is a clear relationship between Ca2+ signaling, e.g., increased Ca2+ signals, and inflammatory responses[92]. cAMP also modulates inflammatory responses: medicines which increase intracellular levels of cAMP can diminish the generation of pro-inflammatory factors, and enhance the generation of anti-inflammatory molecules[93]. Furthermore, whereas a physiological increase in the cytoplasmic concentration of Ca2+ is a significant trigger to release insulin, an abnormal elevation in [Ca2+]c could stimulate β-cell apoptosis, then decrease insulin levels, contributing to diabetes[9,12,25]. Together with Ca2+, cAMP modulates the release of various hormones, as well as insulin from the pancreatic β-cells[51,52]. Although increasing cAMP levels, e.g., via adrenaline, might stimulate the biosynthesis of hepatic glucose, increasing cAMP levels within pancreatic β-cells can stimulate insulin release. A start signal for the release of insulin is achieved through an increase in Ca2+ concentration, and after this signal is amplified via cAMP[53]. Besides the cellular effect of increasing the biosynthesis of insulin, cAMP is implicated in other cellular processes of β-cell, e.g., stimulating both proliferation and differentiation of the cell, and by rescuing the cells from death[53]. These effects are summarized in Figure 2.
Ca2+/cAMP signaling has been highlighted as a protagonist in hypertension, cancer, diabetes, and dementia[9,10,12,24,25,32-36,86,87]. Considering the experience of our group in this field, our reports undoubtedly show that Ca2+ release from the ER can be induced by an increase in [cAMP]c. Therefore, considering the participation of Ca2+/cAMP signaling pathways in modulating the release of both neurotransmitters and hormones, as well as tumor growth and neurodegeneration, dysregulations of these signaling pathways can result in disorders such as hypertension, dementia, diabetes, and cancer[9,10,12,24,25,32-36,86,87].
In addition, several findings have confirmed that CCBs, despite their classical antihypertensive effect, can attenuate the symptoms of dementia, diabetes, and cancer[9,10,12,29,94-96]. Similar effects could be achieved by increasing [cAMP]c[9,10,12,24,88-90]. Please see Table 2.
| CCBs | cAMP signaling (enhancer compounds) |
| Verapamil | Rolipram |
| Nifedipine | 3-isobutyl-1-methylxanthine (IBMX) |
| Diltiazem | Forskolin |
| Isradipine | Aminophylline |
| Amlodipine | Theophylline |
| Nicardipine | Paraxanthine |
Undeniably, an increase in the release of neurotransmitters, and a reduction in the death of neurons in the CNS (e.g., limbic brain sites), could cause as a consequence a decrease in symptoms related to dementia, a working model that can be controlled by Ca2+ and cAMP signaling pathways[10,12,25,33]. Analogous to dementia[10,12,33], discoveries have shown that CCBs can also mitigate the symptoms of both cancer and diabetes[9,12,94-96]. Reestablishing the dyshomeostasis associated with Ca2+ signaling is a working model for these CCBs-mentioned responses, reached due to intervening in the Ca2+/cAMP signaling interactions. In fact, CCBs stimulate the activity of ACs, following an increase in [cAMP]c, promoting Ca2+ release from the ER, ultimately inducing the release of both neurotransmitters and hormones, and decreasing the death of neurons and attenuating tumor growth. Considering that the link between diseases (cancer, diabetes, hypertension, and dementia) could be a consequence of persistent dysregulations of [Ca2+]c, the persistent increase in [Ca2+]c might also disturb Ca2+/cAMP signaling interactions.
Both Ca2+ and cAMP signaling pathways regulate the release of neurotransmitters and hormones, including those involved in neurodegeneration and tumor growth. Furthermore, there is a clear relationship between Ca2+ signaling, e.g., increased Ca2+ signals and inflammatory responses. cAMP also regulates pro- and anti-inflammatory responses. It is concluded that both signaling pathways play an important role in the link between diabetes and inflammatory diseases, thus impacting therapeutics including CCBs and medicines which increase the levels of cAMP. Finally, as COVID-19 is an emerging and rapidly evolving situation, it is also concluded that Ca2+ channel blockers could be useful for preventing Ca2+ signaling disruption due to COVID-19.
Dr. Leandro Bergantin would like to thank the reviewers and editors for their suggestions to improve this manuscript.
| 1. | Lim S, Yoo BK, Kim HS, Gilmore HL, Lee Y, Lee HP, Kim SJ, Letterio J, Lee HG. Amyloid-β precursor protein promotes cell proliferation and motility of advanced breast cancer. BMC Cancer. 2014;14:928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 83] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 2. | Wezyk M, Szybinska A, Wojsiat J, Szczerba M, Day K, Ronnholm H, Kele M, Berdynski M, Peplonska B, Fichna JP, Ilkowski J, Styczynska M, Barczak A, Zboch M, Filipek-Gliszczynska A, Bojakowski K, Skrzypczak M, Ginalski K, Kabza M, Makalowska I, Barcikowska-Kotowicz M, Wojda U, Falk A, Zekanowski C. Overactive BRCA1 Affects Presenilin 1 in Induced Pluripotent Stem Cell-Derived Neurons in Alzheimer's Disease. J Alzheimers Dis. 2018;62:175-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 3. | Jin WS, Bu XL, Liu YH, Shen LL, Zhuang ZQ, Jiao SS, Zhu C, Wang QH, Zhou HD, Zhang T, Wang YJ. Plasma Amyloid-Beta Levels in Patients with Different Types of Cancer. Neurotox Res. 2017;31:283-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 4. | Stocks T, Van Hemelrijck M, Manjer J, Bjørge T, Ulmer H, Hallmans G, Lindkvist B, Selmer R, Nagel G, Tretli S, Concin H, Engeland A, Jonsson H, Stattin P. Blood pressure and risk of cancer incidence and mortality in the Metabolic Syndrome and Cancer Project. Hypertension. 2012;59:802-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 218] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 5. | Stocks T, Borena W, Strohmaier S, Bjørge T, Manjer J, Engeland A, Johansen D, Selmer R, Hallmans G, Rapp K, Concin H, Jonsson H, Ulmer H, Stattin P. Cohort Profile: The Metabolic syndrome and Cancer project (Me-Can). Int J Epidemiol. 2010;39:660-667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 6. | Andreotti G, Boffetta P, Rosenberg PS, Berndt SI, Karami S, Menashe I, Yeager M, Chanock SJ, Zaridze D, Matteev V, Janout V, Kollarova H, Bencko V, Navratilova M, Szeszenia-Dabrowska N, Mates D, Rothman N, Brennan P, Chow WH, Moore LE. Variants in blood pressure genes and the risk of renal cell carcinoma. Carcinogenesis. 2010;31:614-620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 7. | Corrao G, Scotti L, Bagnardi V, Sega R. Hypertension, antihypertensive therapy and renal-cell cancer: a meta-analysis. Curr Drug Saf. 2007;2:125-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 8. | Seretis A, Cividini S, Markozannes G, Tseretopoulou X, Lopez DS, Ntzani EE, Tsilidis KK. Association between blood pressure and risk of cancer development: a systematic review and meta-analysis of observational studies. Sci Rep. 2019;9:8565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 151] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 9. | Bergantin LB. Diabetes and cancer: Debating the link through Ca2+/cAMP signalling. Cancer Lett. 2019;448:128-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 10. | Bergantin LB, Caricati-Neto A. The "Calcium Paradox" and its Impact on Neurological and Psychiatric Diseases. Cambridge Scholars Publishing; 2018. |
| 11. | Tsalamandris S, Antonopoulos AS, Oikonomou E, Papamikroulis GA, Vogiatzi G, Papaioannou S, Deftereos S, Tousoulis D. The Role of Inflammation in Diabetes: Current Concepts and Future Perspectives. Eur Cardiol. 2019;14:50-59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1203] [Cited by in RCA: 975] [Article Influence: 139.3] [Reference Citation Analysis (1)] |
| 12. | Bergantin LB. Hypertension, Diabetes and Neurodegenerative Diseases: Is there a Clinical Link through the Ca2+/cAMP Signalling Interaction? Curr Hypertens Rev. 2019;15:32-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 13. | Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science. 1993;259:87-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6065] [Cited by in RCA: 5408] [Article Influence: 163.9] [Reference Citation Analysis (0)] |
| 14. | Ogston D, Mcandrew GM. Fibrinolysis in obesity. Lancet. 1964;2:1205-1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 67] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Emerging Risk Factors Collaboration, Kaptoge S, Di Angelantonio E, Lowe G, Pepys MB, Thompson SG, Collins R, Danesh J. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet. 2010;375:132-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2004] [Cited by in RCA: 1898] [Article Influence: 118.6] [Reference Citation Analysis (0)] |
| 16. | Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med. 1997;336:973-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4153] [Cited by in RCA: 3708] [Article Influence: 127.9] [Reference Citation Analysis (0)] |
| 17. | Duncan BB, Schmidt MI, Pankow JS, Ballantyne CM, Couper D, Vigo A, Hoogeveen R, Folsom AR, Heiss G; Atherosclerosis Risk in Communities Study. Low-grade systemic inflammation and the development of type 2 diabetes: the atherosclerosis risk in communities study. Diabetes. 2003;52:1799-1805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 853] [Cited by in RCA: 777] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 18. | Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6201] [Cited by in RCA: 5752] [Article Influence: 230.1] [Reference Citation Analysis (0)] |
| 19. | Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860-867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12133] [Cited by in RCA: 11340] [Article Influence: 472.5] [Reference Citation Analysis (3)] |
| 20. | Kinney JW, Bemiller SM, Murtishaw AS, Leisgang AM, Salazar AM, Lamb BT. Inflammation as a central mechanism in Alzheimer's disease. Alzheimers Dement (N Y). 2018;4:575-590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1799] [Cited by in RCA: 1633] [Article Influence: 204.1] [Reference Citation Analysis (1)] |
| 21. | De Miguel C, Rudemiller NP, Abais JM, Mattson DL. Inflammation and hypertension: new understandings and potential therapeutic targets. Curr Hypertens Rep. 2015;17:507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 191] [Cited by in RCA: 177] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 22. | Hemminki K, Hemminki O, Försti A, Sundquist K, Sundquist J, Li X. Surveillance Bias in Cancer Risk After Unrelated Medical Conditions: Example Urolithiasis. Sci Rep. 2017;7:8073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 23. | Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7:280-292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5966] [Cited by in RCA: 5261] [Article Influence: 350.7] [Reference Citation Analysis (0)] |
| 24. | Bergantin LB, Caricati-Neto A. Challenges for the pharmacological treatment of neurological and psychiatric disorders: Implications of the Ca(2+)/cAMP intracellular signalling interaction. Eur J Pharmacol. 2016;788:255-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 25. | Bergantin LB. Debating the "bidirectional link" between diabetes and depression through the Ca2+/cAMP signalling: Off-label effects of Ca2+ channel blockers. Pharmacol Res. 2019;141:298-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Marques R, Peres CG, Vaz CV, Gomes IM, Figueira MI, Cairrão E, Verde I, Maia CJ, Socorro S. 5α-Dihydrotestosterone regulates the expression of L-type calcium channels and calcium-binding protein regucalcin in human breast cancer cells with suppression of cell growth. Med Oncol. 2015;32:228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Dalal PJ, Muller WA, Sullivan DP. Endothelial Cell Calcium Signaling during Barrier Function and Inflammation. Am J Pathol. 2020;190:535-542. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 216] [Cited by in RCA: 193] [Article Influence: 32.2] [Reference Citation Analysis (0)] |
| 28. | Vaz CV, Rodrigues DB, Socorro S, Maia CJ. Effect of extracellular calcium on regucalcin expression and cell viability in neoplastic and non-neoplastic human prostate cells. Biochim Biophys Acta. 2015;1853:2621-2628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 29. | Wu CL, Wen SH. A 10-year follow-up study of the association between calcium channel blocker use and the risk of dementia in elderly hypertensive patients. Medicine (Baltimore). 2016;95:e4593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 30. | Tully PJ, Peters R, Pérès K, Anstey KJ, Tzourio C. Effect of SSRI and calcium channel blockers on depression symptoms and cognitive function in elderly persons treated for hypertension: three city cohort study. Int Psychogeriatr. 2018;30:1345-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Yoshida J, Ishibashi T, Nishio M. G1 cell cycle arrest by amlodipine, a dihydropyridine Ca2+ channel blocker, in human epidermoid carcinoma A431 cells. Biochem Pharmacol. 2007;73:943-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 40] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 32. | Bergantin LB, Souza CF, Ferreira RM, Smaili SS, Jurkiewicz NH, Caricati-Neto A, Jurkiewicz A. Novel model for "calcium paradox" in sympathetic transmission of smooth muscles: role of cyclic AMP pathway. Cell Calcium. 2013;54:202-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 33. | Caricati-Neto A, García AG, Bergantin LB. Pharmacological implications of the Ca(2+)/cAMP signaling interaction: from risk for antihypertensive therapy to potential beneficial for neurological and psychiatric disorders. Pharmacol Res Perspect. 2015;3:e00181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 34. | Caricati-Neto A, Bergantin LB. Pharmacological modulation of neural Ca2+/camp signaling interaction as therapeutic goal for treatment of Alzheimer's disease. J Syst Integr Neurosci. 2017;3. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 35. | Caricati-Neto A, Bergantin LB. The passion of a scientific discovery: the "calcium paradox" due to Ca2+/camp interaction. J Syst Integr Neurosci. 2017;3. [RCA] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 36. | Caricati-Neto A, Bergantin LB. From a "eureka insight" to a novel potential therapeutic target to treat Parkinson's disease: The Ca2+/camp signalling interaction. J Syst Integr Neurosci. 2017;4. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 37. | Bezprozvanny I, Mattson MP. Neuronal calcium mishandling and the pathogenesis of Alzheimer's disease. Trends Neurosci. 2008;31:454-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 781] [Cited by in RCA: 722] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 38. | St George-Hyslop PH, Petit A. Molecular biology and genetics of Alzheimer's disease. C R Biol. 2005;328:119-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 124] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 39. | Kale VP, Amin SG, Pandey MK. Targeting ion channels for cancer therapy by repurposing the approved drugs. Biochim Biophys Acta. 2015;1848:2747-2755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 40. | Gomez-Ospina N, Tsuruta F, Barreto-Chang O, Hu L, Dolmetsch R. The C terminus of the L-type voltage-gated calcium channel Ca(V)1.2 encodes a transcription factor. Cell. 2006;127:591-606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 313] [Cited by in RCA: 280] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 41. | Dziegielewska B, Gray LS, Dziegielewski J. T-type calcium channels blockers as new tools in cancer therapies. Pflugers Arch. 2014;466:801-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 42. | Ohkubo T, Yamazaki J. T-type voltage-activated calcium channel Cav3.1, but not Cav3.2, is involved in the inhibition of proliferation and apoptosis in MCF-7 human breast cancer cells. Int J Oncol. 2012;41:267-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 43. | Gackière F, Bidaux G, Delcourt P, Van Coppenolle F, Katsogiannou M, Dewailly E, Bavencoffe A, Van Chuoï-Mariot MT, Mauroy B, Prevarskaya N, Mariot P. CaV3.2 T-type calcium channels are involved in calcium-dependent secretion of neuroendocrine prostate cancer cells. J Biol Chem. 2008;283:10162-10173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 44. | Latour I, Louw DF, Beedle AM, Hamid J, Sutherland GR, Zamponi GW. Expression of T-type calcium channel splice variants in human glioma. Glia. 2004;48:112-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 67] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 45. | Yamaguchi M, Osuka S, Shoji M, Weitzmann MN, Murata T. Survival of lung cancer patients is prolonged with higher regucalcin gene expression: suppressed proliferation of lung adenocarcinoma A549 cells in vitro. Mol Cell Biochem. 2017;430:37-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 46. | Yamaguchi M. Suppressive role of regucalcin in liver cell proliferation: involvement in carcinogenesis. Cell Prolif. 2013;46:243-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 47. | Huang MY, Wang HM, Chang HJ, Hsiao CP, Wang JY, Lin SR. Overexpression of S100B, TM4SF4, and OLFM4 genes is correlated with liver metastasis in Taiwanese colorectal cancer patients. DNA Cell Biol. 2012;31:43-49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 48. | Miranda-Ferreira R, de Pascual R, de Diego AM, Caricati-Neto A, Gandía L, Jurkiewicz A, García AG. Single-vesicle catecholamine release has greater quantal content and faster kinetics in chromaffin cells from hypertensive, as compared with normotensive, rats. J Pharmacol Exp Ther. 2008;324:685-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 49. | Miranda-Ferreira R, de Pascual R, Caricati-Neto A, Gandía L, Jurkiewicz A, García AG. Role of the endoplasmic reticulum and mitochondria on quantal catecholamine release from chromaffin cells of control and hypertensive rats. J Pharmacol Exp Ther. 2009;329:231-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 50. | McCrimmon RJ, Ryan CM, Frier BM. Diabetes and cognitive dysfunction. Lancet. 2012;379:2291-2299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 766] [Cited by in RCA: 687] [Article Influence: 49.1] [Reference Citation Analysis (0)] |
| 51. | Henquin JC. The interplay between cyclic AMP and ions in the stimulus-secretion coupling in pancreatic B-cells. Arch Int Physiol Biochim. 1985;93:37-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 52. | Bratanova-Tochkova TK, Cheng H, Daniel S, Gunawardana S, Liu YJ, Mulvaney-Musa J, Schermerhorn T, Straub SG, Yajima H, Sharp GW. Triggering and augmentation mechanisms, granule pools, and biphasic insulin secretion. Diabetes. 2002;51 Suppl 1:S83-S90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 196] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 53. | Tengholm A. Cyclic AMP dynamics in the pancreatic β-cell. Ups J Med Sci. 2012;117:355-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 99] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 54. | van der Willik KD, Schagen SB, Ikram MA. Cancer and dementia: Two sides of the same coin? Eur J Clin Invest. 2018;48:e13019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 55. | Ahles TA, Saykin AJ. Candidate mechanisms for chemotherapy-induced cognitive changes. Nat Rev Cancer. 2007;7:192-201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 756] [Cited by in RCA: 666] [Article Influence: 35.1] [Reference Citation Analysis (0)] |
| 56. | Weycker D, Nichols GA, O'Keeffe-Rosetti M, Edelsberg J, Vincze G, Khan ZM, Oster G. Excess risk of diabetes in persons with hypertension. J Diabetes Complications. 2009;23:330-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 57. | Cheung BM, Li C. Diabetes and hypertension: is there a common metabolic pathway? Curr Atheroscler Rep. 2012;14:160-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 423] [Cited by in RCA: 377] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 58. | Cheung BM. The hypertension-diabetes continuum. J Cardiovasc Pharmacol. 2010;55:333-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 56] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 59. | Landsberg L, Molitch M. Diabetes and hypertension: pathogenesis, prevention and treatment. Clin Exp Hypertens. 2004;26:621-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 66] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 60. | Gress TW, Nieto FJ, Shahar E, Wofford MR, Brancati FL. Hypertension and antihypertensive therapy as risk factors for type 2 diabetes mellitus. Atherosclerosis Risk in Communities Study. N Engl J Med. 2000;342:905-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 908] [Cited by in RCA: 737] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 61. | Munshi M, Grande L, Hayes M, Ayres D, Suhl E, Capelson R, Lin S, Milberg W, Weinger K. Cognitive dysfunction is associated with poor diabetes control in older adults. Diabetes Care. 2006;29:1794-1799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 268] [Cited by in RCA: 229] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 62. | Sinclair AJ, Girling AJ, Bayer AJ. Cognitive dysfunction in older subjects with diabetes mellitus: impact on diabetes self-management and use of care services. All Wales Research into Elderly (AWARE) Study. Diabetes Res Clin Pract. 2000;50:203-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 222] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 63. | Alencar RC, Cobas RA, Gomes MB. Assessment of cognitive status in patients with type 2 diabetes through the Mini-Mental Status Examination: a cross-sectional study. Diabetol Metab Syndr. 2010;2:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 64. | Feinkohl I, Keller M, Robertson CM, Morling JR, Williamson RM, Nee LD, McLachlan S, Sattar N, Welsh P, Reynolds RM, Russ TC, Deary IJ, Strachan MW, Price JF; Edinburgh Type 2 Diabetes Study (ET2DS) Investigators. Clinical and subclinical macrovascular disease as predictors of cognitive decline in older patients with type 2 diabetes: the Edinburgh Type 2 Diabetes Study. Diabetes Care. 2013;36:2779-2786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 65. | Zhao Q, Roberts RO, Ding D, Cha R, Guo Q, Meng H, Luo J, Machulda MM, Shane Pankratz V, Wang B, Christianson TJ, Aakre JA, Knopman DS, Boeve BF, Hong Z, Petersen RC; Shanghai Aging Study (SAS); Mayo Clinic Study of Aging (MCSA). Diabetes is Associated with Worse Executive Function in Both Eastern and Western Populations: Shanghai Aging Study and Mayo Clinic Study of Aging. J Alzheimers Dis. 2015;47:167-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 66. | Qiu C, Sigurdsson S, Zhang Q, Jonsdottir MK, Kjartansson O, Eiriksdottir G, Garcia ME, Harris TB, van Buchem MA, Gudnason V, Launer LJ. Diabetes, markers of brain pathology and cognitive function: the Age, Gene/Environment Susceptibility-Reykjavik Study. Ann Neurol. 2014;75:138-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 105] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 67. | Moran C, Phan TG, Chen J, Blizzard L, Beare R, Venn A, Münch G, Wood AG, Forbes J, Greenaway TM, Pearson S, Srikanth V. Brain atrophy in type 2 diabetes: regional distribution and influence on cognition. Diabetes Care. 2013;36:4036-4042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 388] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 68. | Kooistra M, Geerlings MI, van der Graaf Y, Mali WP, Vincken KL, Kappelle LJ, Muller M, Biessels GJ; SMART-MR Study Group. Vascular brain lesions, brain atrophy, and cognitive decline. The Second Manifestations of ARTerial disease--Magnetic Resonance (SMART-MR) study. Neurobiol Aging. 2014;35:35-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 69. | van Elderen SG, de Roos A, de Craen AJ, Westendorp RG, Blauw GJ, Jukema JW, Bollen EL, Middelkoop HA, van Buchem MA, van der Grond J. Progression of brain atrophy and cognitive decline in diabetes mellitus: a 3-year follow-up. Neurology. 2010;75:997-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 153] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 70. | Whitmer RA. Type 2 diabetes and risk of cognitive impairment and dementia. Curr Neurol Neurosci Rep. 2007;7:373-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 134] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 71. | Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract. 2014;103:137-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3448] [Cited by in RCA: 2942] [Article Influence: 245.2] [Reference Citation Analysis (0)] |
| 72. | Bruce DG, Casey GP, Grange V, Clarnette RC, Almeida OP, Foster JK, Ives FJ, Davis TM; Fremantle Cognition in Diabetes Study. Cognitive impairment, physical disability and depressive symptoms in older diabetic patients: the Fremantle Cognition in Diabetes Study. Diabetes Res Clin Pract. 2003;61:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 93] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 73. | Watson GS, Craft S. The role of insulin resistance in the pathogenesis of Alzheimer's disease: implications for treatment. CNS Drugs. 2003;17:27-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 322] [Cited by in RCA: 287] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 74. | Yaffe K, Blackwell T, Whitmer RA, Krueger K, Barrett Connor E. Glycosylated hemoglobin level and development of mild cognitive impairment or dementia in older women. J Nutr Health Aging. 2006;10:293-295. [PubMed] |
| 75. | Kodl CT, Seaquist ER. Cognitive dysfunction and diabetes mellitus. Endocr Rev. 2008;29:494-511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 645] [Cited by in RCA: 576] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 76. | Cunnane S, Nugent S, Roy M, Courchesne-Loyer A, Croteau E, Tremblay S, Castellano A, Pifferi F, Bocti C, Paquet N, Begdouri H, Bentourkia M, Turcotte E, Allard M, Barberger-Gateau P, Fulop T, Rapoport SI. Brain fuel metabolism, aging, and Alzheimer's disease. Nutrition. 2011;27:3-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 515] [Cited by in RCA: 464] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 77. | Cunnane SC, Plourde M, Pifferi F, Bégin M, Féart C, Barberger-Gateau P. Fish, docosahexaenoic acid and Alzheimer's disease. Prog Lipid Res. 2009;48:239-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 207] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 78. | Yassine HN, Croteau E, Rawat V, Hibbeln JR, Rapoport SI, Cunnane SC, Umhau JC. DHA brain uptake and APOE4 status: a PET study with [1-11C]-DHA. Alzheimers Res Ther. 2017;9:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 52] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 79. | Croteau E, Castellano CA, Richard MA, Fortier M, Nugent S, Lepage M, Duchesne S, Whittingstall K, Turcotte ÉE, Bocti C, Fülöp T, Cunnane SC. Ketogenic Medium Chain Triglycerides Increase Brain Energy Metabolism in Alzheimer's Disease. J Alzheimers Dis. 2018;64:551-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 101] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 80. | Abdi A, Jalilian M, Sarbarzeh PA, Vlaisavljevic Z. Diabetes and COVID-19: A systematic review on the current evidences. Diabetes Res Clin Pract. 2020;166:108347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 143] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 81. | Lim S, Bae JH, Kwon HS, Nauck MA. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol. 2021;17:11-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 770] [Cited by in RCA: 608] [Article Influence: 121.6] [Reference Citation Analysis (0)] |
| 82. | Zhang LK, Sun Y, Zeng H, Wang Q, Jiang X, Shang WJ, Wu Y, Li S, Zhang YL, Hao ZN, Chen H, Jin R, Liu W, Li H, Peng K, Xiao G. Calcium channel blocker amlodipine besylate therapy is associated with reduced case fatality rate of COVID-19 patients with hypertension. Cell Discov. 2020;6:96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 83. | Silva IVG, de Figueiredo RC, Rios DRA. Effect of Different Classes of Antihypertensive Drugs on Endothelial Function and Inflammation. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 84. | D'Elia JA, Weinrauch LA. Calcium Ion Channels: Roles in Infection and Sepsis Mechanisms of Calcium Channel Blocker Benefits in Immunocompromised Patients at Risk for Infection. Int J Mol Sci. 2018;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 85. | Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21249] [Cited by in RCA: 18025] [Article Influence: 3004.2] [Reference Citation Analysis (7)] |
| 86. | Bergantin LB. The Interplay Between Asthma and Other Diseases: Role of Ca2+/cAMP Signalling. Endocr Metab Immune Disord Drug Targets. 2020;20:321-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 87. | Bergantin LB. Diabetes and Parkinson's Disease: Debating the Link Through Ca2+/cAMP Signalling. Curr Diabetes Rev. 2020;16:238-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 88. | Fajardo AM, Piazza GA, Tinsley HN. The role of cyclic nucleotide signaling pathways in cancer: targets for prevention and treatment. Cancers (Basel). 2014;6:436-458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 157] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 89. | Drees M, Zimmermann R, Eisenbrand G. 3',5'-Cyclic nucleotide phosphodiesterase in tumor cells as potential target for tumor growth inhibition. Cancer Res. 1993;53:3058-3061. [PubMed] |
| 90. | Xiao L, O'Callaghan JP, O'Donnell JM. Effects of repeated treatment with phosphodiesterase-4 inhibitors on cAMP signaling, hippocampal cell proliferation, and behavior in the forced-swim test. J Pharmacol Exp Ther. 2011;338:641-647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 91. | Uddin MS, Stachowiak A, Mamun AA, Tzvetkov NT, Takeda S, Atanasov AG, Bergantin LB, Abdel-Daim MM, Stankiewicz AM. Autophagy and Alzheimer's Disease: From Molecular Mechanisms to Therapeutic Implications. Front Aging Neurosci. 2018;10:04. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 370] [Cited by in RCA: 330] [Article Influence: 41.3] [Reference Citation Analysis (0)] |
| 92. | Ribeiro CM. The role of intracellular calcium signals in inflammatory responses of polarised cystic fibrosis human airway epithelia. Drugs R D. 2006;7:17-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 93. | Raker VK, Becker C, Steinbrink K. The cAMP Pathway as Therapeutic Target in Autoimmune and Inflammatory Diseases. Front Immunol. 2016;7:123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 255] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 94. | Novotny M, Klimova B, Valis M. Nitrendipine and Dementia: Forgotten Positive Facts? Front Aging Neurosci. 2018;10:418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 95. | Takada K, Kashiwagi S, Asano Y, Goto W, Takahashi K, Fujita H, Takashima T, Tomita S, Hirakawa K, Ohira M. Verification of the effects of calcium channel blockers on the immune microenvironment of breast cancer. BMC Cancer. 2019;19:615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 96. | Taghizadehghalehjoughi A, Sezen S, Hacimuftuoglu A, Güllüce M. Vincristine combination with Ca+2 channel blocker increase antitumor effects. Mol Biol Rep. 2019;46:2523-2528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country/Territory of origin: Brazil
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Aureliano M, Avtanski D S-Editor: Fan JR L-Editor: Webster JR P-Editor: Yuan YY