Published online Dec 15, 2019. doi: 10.4239/wjd.v10.i12.546
Peer-review started: July 26, 2019
First decision: August 19, 2019
Revised: October 20, 2019
Accepted: October 27, 2019
Article in press: October 27, 2019
Published online: December 15, 2019
Processing time: 143 Days and 5.2 Hours
It is known that under conditions of tissue tolerance to insulin, observed during type 2 diabetes mellitus (DM2), there is an increased production of reactive oxygen species. Moreover, the free radicals can initiate lipid peroxidation (LPO) in lipoprotein particles. The concentration of LPO products can influence the state of insulin receptors, repressing their hormone connection activity, which is expressed as a reduction of the glucose consumption by cells. It is possible that reduction in glucose concentration during administration of 10-(6-plastoquinonyl) decyltriphenylphosphonium (SkQ1) to rats with DM2 may be related to the antioxidant properties of this substance.
To establish the influence of SkQ1 on free-radical homeostasis in the heart and blood serum of rats with streptozotocin-induced hyperglycemia.
To induce hyperglycemia, rats were fed a high-fat diet for 1 mo and then administered two intra-abdominal injections of streptozotocin with a 7-d interval at a 30 mg/kg of animal weight dose with citrate buffer equal to pH 4.4. SkQ1 solution was administered intraperitoneally at a 1250 nmol/kg dose per day. Tissue samples were taken from control animals, animals with experimental hyperglycemia, rats with streptozotocin-induced glycemia that were administered SkQ1 solution, animals housed under standard vivarium conditions that were administered SkQ1, rats that were administered intraperitoneally citrate buffer equal to pH 4.4 once a week during 2 wk after 1-mo high-fat diet, and animals that were administered intraperitoneally with appropriate amount of solution without SkQ1 (98% ethanol diluted eight times with normal saline solution). To determine the intensity of free radical oxidation and total antioxidant activity, we used the biochemiluminescence method. Aconitate hydratase (AH), superoxide dismutase, and catalase activities were estimated using the Hitachi U-1900 spectrophotometer supplied with software. The amount of citrate was determined by means of the Natelson method. Real-time polymerase chain reaction was carried out using an amplifier ANK-32.
It was found that the mitochondrial-directed antioxidant elicits decrease of biochemiluminescence parameter values that increase by pathology as well as the levels of primary products of LPO, such as diene conjugates and carbonyl compounds, which indicate intensity of free radical oxidation. At the same time, the activity of AH, considered a crucial target of free radicals, which decreased during experimental hyperglycemia, increased. Apparently, increasing activity of AH influenced the speed of citrate utilization, whose concentration decreased after administering SkQ1 by pathology. Moreover, the previously applied anti-oxidant during hyperglycemia influenced the rate of antioxidant system mobilization. Thus, superoxide dismutase and catalase activity, as well as the level of gene transcript under influence of SkQ1 at pathology, were changing to the direction of control groups values.
According to the results of performed research, SkQ1 can be considered a promising addition to be included in antioxidant therapy of DM2.
Core tip: The results of this research suggest that the mitochondria targeted antioxidant 10-(6-plastoquinonyl) decyltriphenylphosphonium (SkQ1) might be a potential substance for incorporation into the antioxidant therapy of type 2 diabetes mellitus. The ability of this compound to lower the intensity of free-radical processes, acting as the key component of the pathogenesis of the type 2 diabetes mellitus, serves as the basis for this conclusion. Thus, after the introduction of SkQ1 to the animals with streptozotocin induced hyperglycemia, the values of the biochemiluminescence parameters reflecting the free-radical oxidation intensity, the concentration of diene conjugates and carbonyl products of protein oxidation, aconitate hydratase activity, and citrate content approached those of control values. At the same time, the activity level of the antioxidant enzymes superoxide dismutase and catalase approached those of normal values.
- Citation: Agarkov AA, Popova TN, Boltysheva YG. Influence of 10-(6-plastoquinonyl) decyltriphenylphosphonium on free-radical homeostasis in the heart and blood serum of rats with streptozotocin-induced hyperglycemia. World J Diabetes 2019; 10(12): 546-559
- URL: https://www.wjgnet.com/1948-9358/full/v10/i12/546.htm
- DOI: https://dx.doi.org/10.4239/wjd.v10.i12.546
Core tip: The results of this research suggest that the mitochondria targeted antioxidant 10-(6-plastoquinonyl) decyltriphenylphosphonium (SkQ1) might be a potential substance for incorporation into the antioxidant therapy of type 2 diabetes mellitus. The ability of this compound to lower the intensity of free-radical processes, acting as the key component of the pathogenesis of the type 2 diabetes mellitus, serves as the basis for this conclusion. Thus, after the introduction of SkQ1 to the animals with streptozotocin induced hyperglycemia, the values of the biochemiluminescence parameters reflecting the free-radical oxidation intensity, the concentration of diene conjugates and carbonyl products of protein oxidation, aconitate hydratase activity, and citrate content approached those of control values. At the same time, the activity level of the antioxidant enzymes superoxide dismutase and catalase approached those of normal values.
Type 2 diabetes mellitus (DM2) is a pandemic metabolic disease and is becoming a serious problem for health protection due to the global increase of its prevalence. Endocrinopathic complications, such as nephropathy, retinopathy, brain angiopathy, myocardial angiopathy, and lower limbs angiopathy, prove to be the major reason for incapacity, disability, or patient mortality. It is known that diabetic cardiomyopathy develops in patients with insulin resistance and DM regardless of diagnosed coronary heart disease or hypertension[1].
The crucial target is vascular endothelium, which is affected by a number of metabolic, hemodynamic, and immunologic factors that characterize the development of the disease[2]. It has been shown in DM2 that the delivery of fatty acids to the myocardium is intensified and glycolysis is slowed down[3]. At the same time, the concentration of reactive oxygen species (ROS) have a damaging effect on the lipids of cardiomyocyte membranes and contribute to the mitochondrion mechanism malfunction and, as a result, to the inhibition of ATP elaboration. This leads to calcium imbalance in cardiomyocytes and results in muscular relaxation and contraction. Hyperglycemia as well contributes to the development and progression of fibrotic degeneration of cardiomyocytes due to the increased deposition of collagen in the interstitium[4].
Taking into account the most important factors of free radical oxidation activation in the pathogenesis of DM, as well as in the development of its complications, antioxidant therapy should be considered among the modern medical endocrinopathy treatment technologies. In this context, there is an urgent need for effective substances that would protect cellular structures from oxidative stress.
Ten-(6'-plastoquinonyl) decyltriphenylphosphonium (SkQ1) is an aromatic cation (triphenylphosphonium) conjugated with a 10-12 atom aliphatic compound as well as plastoquinone, which is an active molecular component of this substance[5].
Several Sk-compounds with modified lipophilic and antioxidant parts were synthesized and tested by varying the length of the aliphatic linker. All these compounds have abbreviated names derived from the surname of Russian academician Skulachev VP (Sk), the letter to designate ubiquinone (Q), and the letter or numerical symbol to designate the modification.
When used in nanoconcentrations, this substance participates in the ROS balance regulation as it has the ability to neutralize free radicals (FR), including OH radicals in aqueous solutions. This may protect cells from apoptosis and necrosis induced by ROS[6].
The goal of this study was to evaluate the influence of SkQ1 on biochem-iluminescence (BCL) parameters, which reflect the free radical processes reactions rate, the total activity of the antioxidant system (AOS), the level of primary lipid peroxidation (LPO) products such as diene conjugates (DC), the activity of aconitase, which is the sensitive target of FR action and citrate content, the degree of protein oxidative modification, the activity of superoxide dismutase (SOD) and catalase, and the level of their genes’ transcripts in heart and blood serum of the rats with hyperglycemia induced by the administration of streptozotocin (STZ).
To conduct the study, laboratory rats were selected of nursery rat males from Federal State-financed Organization of Health Service “Voronezh Hygiene and Epidemiology Center”. The animals were divided through stratified randomization by their weight and age. The rats used for the study weighed 200-250 g and aged 3-5 mo. The experimental animals were kept for 14 d before the start of the study under the following conditions: 12-h light day, temperature 21-25 °C, and food ad libitum. The experiment was carried out in accordance with European legislation on the protection of animals (Directive 2010/63/EC).
Hyperglycemia was induced by feeding rats a high-fat diet for 1 mo, followed by two intra-abdominal injections of STZ with a 7-d interval at a 30 mg/kg of animal weight dose with citrate buffer equal to pH 4.4[7].
Hyperglycemia in rats was verified by measuring glucose level in the blood serum using a glucose oxidase test. The blood was collected twice with a day-interval from caudal vein after the second administration of STZ and 1-d food deprivation. The reagent kit used for the study was purchased from Vital Diagnosticum, Saint-Petersburg, Russia.
At 2 wk after STZ administration, drugged animals were devitalized for further research. The laboratory rats were divided into four groups: Group 1 (n = 20), animals housed under standard vivarium conditions (control group); group 2 (n = 20), animals with STZ injection-induced hyperglycemia; group 3 (n = 12), animals with STZ-induced glycemia that were administered SkQ1 solution intraperitoneally at a 1250 nmol/kg dose per day, starting from the second week; and group 4 (n = 8), animals housed under standard vivarium conditions that were administered with SkQ1 at a 1250 nmol/kg dose per day, during the second week of conducting the experiment. Group 2 also included rats (n = 8) that were administered intraperitoneally with appropriate aliquot quantity of citrate buffer equal to pH 4.4 once a week during 2 wk after 1-mo high-fat diet. Group 3 included animals (n = 8) that were administered intraperitoneally with appropriate amount of solution without SkQ1 (98% ethanol diluted eight times with normal saline solution).
To obtain tissue homogenate, heart tissue sample was homogenized in triple amount of cooled medium (0.1 mol/L Tris-HCl buffer (pH 7.8) containing 1 mmol/L EDTA, 1% beta-mercaptoethanol) and centrifuged for 10000 g for 15 min. The serum was obtained from venous blood collected in test tubes without anticoagulant. For this purpose, the blood was thermostated at a temperature of 37 °C until phase immiscibility, and supernatant fluid was centrifuged for 4000 g for 10 min. The obtained serum was used for further examination.
The free-radical oxidation and total antioxidant status processes intensity was measured by applying the Fe2+-induced biochemical luminescence method. The principle of the method is based on catalytic degradation of peroxide by transition valence metal ions (Fe2+) in accordance with the Fenton reaction. Catalytic degradation leads to the formation of FR that enter free-radical oxidation initiation process in the examined biotic substrate. Recombination of RO2 radicals results in unstable tetroxide formation that causes liberation of light quantum when it degrades. Biochemicoluminescence kinetic curve was recorded for 30 seconds using BCL-07 with software (Medozons OOO, Nizhny Novgorod, Russia), and the following parameters were measured: Chemicoluminescence light sum (S) and flash intensity (Imax) that characterized free-radical oxidation intensity and slope of curve magnitude (tgα2) depicting total antioxidant status.
The medium for estimating BCL intensity contained 0.4 mL of 0.02 mol/L potassium phosphate buffer (pH 7.5); 0.4 mL of 0.01 mol/L FeSO4, and 0.2 mL of 2% H2O2 solution that had been added immediately prior to measurement. The test material had been added in an amount of 0.1 mL prior to measurement.
The amount of DC was measured by means of spectrophotometric method at 233 nm[32].
The oxidative modification of proteins valuation method is based on the interaction between oxidized amino acid residues and 2.4-dinitrophenylhydrazine (2.4-DNPH) that forms 2.4-DNPHs[10]. Protein load was identified by the biuret test.
The concentration of DC was analyzed spectrophotometrically at 233 nm[8].
For the analysis of protein oxidative modification (POM), we used the method based on the interaction between oxidized amino acid residues and 2.4-DNPH using the Hitachi U-1900 spectrophotometer (Hitachi High-Technologies, Tokyo, Japan)[9].
Aconitate hydratase (AH) activity was estimated using the Hitachi U-1900 spectrophotometer supplied with software at 233 nm in the medium that contained 0.05 mmol/L of Tris-HC1-buffer (pH 7.8) and 4 mmol/L of sodium citrate (PanReac, Barcelona, Spain)[10].
The amount of citrate was determined by means of the Natelson method[11].
SOD activity was determined by the nitroblue tetrazolium recovery rate inhibition in the non-enzymatic system of phenazine methosulfate (PMS) and NADH.
The incubation medium, with a total volume of 3 mL, contained 0.1 M phosphate buffer (pH 7.8), 0.33 mmol/L EDTA, 0.41 mmol/L NBT, 0.01 mmol/L PMS, and 0.8 mmol/L NADH. The activity was measured spectrophotometrically according to the extinction augmentation after 5 min using the Hitachi U-1900 spectrophotometer at 540 nm[12].
Catalase activity was determined at a wavelength of 410 nm using the method based on the ability of hydrogen peroxide to form stable colored complex with ammonium molybdate[13].
The amount of the enzyme that was required for the conversion of 1 mM of substrate per min at 25 °C was defined as the enzyme unit (E). Biuret method was used to determine the protein content.
Total RNA was isolated from heart tissues and blood cells of experimental animals using Extran RNA reagent kit (Syntol Company, Moscow, Russia). The severity of RNA degradation was determined via 1% denaturing agarose gel electrophoresis. The amount of RNA was determined by measuring the absorption at a wavelength of 260 nm using a Hitachi U-1900 spectrophotometer.
M-MuLV Reverse Transcriptase (Fermentas, Vilnius, Lithuania) was used to carry out reverse transcription. DNA, or complementary mRNA, was obtained via Oligo(dT) 18 Primer. The reaction was carried out at 40 °C for 1 h with the following inactivation of reverse transcriptase at 70 °C per 15 min. Ready-to-use сDNA was used for real-time amplification.
The primers selected via a database of a web-based system Universal Probe Library (Universal Probe Library Assay Design Center) were applied to amplify the region of a gene.
PCR amplification was based on reagent kits containing the SYBR Green I (Syntol Company). Real-time PCR was carried out using an amplifier ANK-32 (Syntol Company) according to the following pattern: Per 5 min at 95 °C, then 40 cycles: 95 °C ﹣15 s, 60 °C ﹣15 s, 72 °C ﹣30 s. The next step was to analyze the threshold cycle value obtained by PCR amplification. Among other scientific experiments, there was also a negative control for: (1) An impurity of foreign DNA set components; and (2) A purity level of sample preparation for the amplification. Negative control consisted of a separate test tube at each process of amplification with an equal amount of water instead of DNA test sample.
During the study, the following chemicals were applied: STZ, Tris-HCl, citrate, nitroblue tetrazolium, PMS, NADH (Sigma, St Louis, MO, United States), SkQ1 synthesized according to the following method[5], EDTA (Reanal, Budapest, Hungary), and other chemicals of “chemically pure” or “analytically pure” grade purchased from Russian manufacturers.
Experiments were done in at least 8-20 biological and two analytical replicates. The results were compared with the control. The data were statistically analyzed using a software package STATISTICA 6.0 with numerical variables – arithmetical mean (M), mean error (m), and statistical significance level (P). Normal distribution data were compared by applying Student's t-test for Bonferroni correction in independent samples[14]. Significance level was set at aP ≤ 0.0167 and bP ≤ 0.0167.
The study showed that SkQ1 administration lowered glycemic level by 2.5 times, which was initially upregulated 2.7 times relative to the control (Table 1). The glucose level of the animals of the fourth experimental group was not significantly different from the stated value. Moreover, glucose concentration was within the stated value in rats of the second experimental group having been administered citrate buffer and of the third group having been administered an aliquot of 12% ethanol.
| Glucose concentration in rats’ blood, mmol/L | ||
| The 9th d after STZ administration | The 11th d after STZ administration | |
| Control group | 5.09 ± 0.081 | 5.41 ± 0.088 |
| Animals with experimental hyperglycemia induced by streptozotocin | 10.01 ± 0.166a | 14.6 ± 0.241a |
| SkQ1 is administered to animals with pathology | 6.04 ± 0.099b | 5.87 ± 0.096b |
| SkQ1 is administered to animals at control group | 5.1 ± 0.083 | 5.30 ± 0.086 |
It is also established that S of BCL value increased by 2.2 times in rats’ hearts under pathological conditions and in blood serum by 2.1 times. Imax BCL increased by 2.5 and 2.0, respectively, compared to the control group, demonstrating increased intensity of free radical oxidation. It is known that the processes of glucose autoxidation and its metabolic intermediates, glycosylation of protein and end-product accumulation of its modification, sorbitol exchange mobilization, glucose utilization via hexosamine pathway, and protein kinase C (PKC) activation may be free radical oxidation sources in DM2[15].
The values of BCL tgα2 increased in rats’ hearts with pathology by 2.1 times and in blood serum by 2.3 times (Table 2), indicating compensatory mechanisms implementation and general antioxidant potential organism mobilization during the development of experimental hyperglycemia.
| Parameters | Experiment conditions | ||||
| Control group | Animals with experimental hyperglycemia induced by streptozotocin | SkQ1 is administered to animals with pathology | SkQ1 is administered to animals at control group | ||
| Biochemicoluminescence measurements | |||||
| Light sum, mV × c | Heart | 485.01 ± 7.912 | 1062.76 ± 17.124a | 818.76 ± 13.611b | 478.33 ± 7.971 |
| Blood serum | 307.70 ± 5.141 | 654.14 ± 10.114a | 410.53 ± 6.713b | 302.33 ± 5.012 | |
| Maximum flash intensity, mV | Heart | 51.53 ± 0.851 | 126.93 ± 1.987a | 83.10 ± 1.374b | 47.33 ± 0.779 |
| Blood serum | 25.20 ± 0.412 | 52.23 ± 0.864a | 33.67 ± 0.559b | 22.15 ± 0.362 | |
| Angle tangent of slope of kinetic curve of biochemicoluminescence pathway | Heart | 7.10 ± 0.101 | 14.94 ± 0.241a | 10.10 ± 0.165b | 7.05 ± 0.112 |
| Blood serum | 13.33 ± 0.223 | 30.43 ± 0.498a | 21.71 ± 0.359b | 12.93 ± 0.183 | |
| Content of conjugated dienes, μmol/mL | Heart | 17.70 ± 0.281 | 41.90 ± 0.687a | 29.76 ± 0.492b | 17.22 ± 0.267 |
| Blood serum | 7.55 ± 0.122 | 24.83 ± 0.401a | 15.00 ± 0.247b | 7.33 ± 0.113 | |
| Level of carbonyl compounds, nmol/L/mg of protein | Heart | 0.072 ± 0.001 | 0.199 ± 0.003a | 0.136 ± 0.002b | 0.070 ± 0.001 |
| Blood serum | 0.021 ± 0.0003 | 0.154 ± 0.002a | 0.076 ± 0.001b | 0.019 ± 0.0003 | |
| Citrate content, nmol/L | Heart | 0.201 ± 0.003 | 0.43 ± 0.007a | 0.28 ± 0.004b | 0.204 ± 0.003 |
| Blood serum | 0.57 ± 0.0097 | 1.45 ± 0.021 | 1.07 ± 0.016b | 0.59 ± 0.0093 | |
The administration of SkQ1 led to the S of BCL values decrease in rats’ hearts by 1.3 times and in blood serum by 1.6 times and Imax BCL by 1.5 and 1.6, respectively (Table 2). BCL tgα2 parameter decreased in rats’ hearts by 1.5 times and in blood serum by 1.4 times (Table 2).
No statistically significant BCL parameters changes in rats of the fourth experimental group and the animals belonging to the second experimental group that have been administered citrate buffer and to the third group that have been administered aliquot of 12% ethanol compared with control have been observed (Table 2).
The data received as a result of a research conducted upon the mitochondrial-directed antioxidant and its effect on biochemical luminescence parameters correspond with the results of the assessment of DC level and POM products. It has been shown that the level of DC in the heart and blood serum of animals of the third group has declined 1.4 and 1.7 times, correspondingly, in comparison with the pathology (Table 2). It was found that the concentration of primary products of LPO in the heart and blood serum of rats from the second group increased 2.4 and 3.3 times, correspondingly (Table 2). The progression of hyperglycemia with animals injected with STZ went along with the increase in POM products content: 2.7 times in the heart and 6.3 times in blood serum in comparison with the control group. After the injection of rats with pathology with SkQ1 carbonyl compounds, the level decreased 1.5 times in the heart and 2.0 times in blood serum in contrast with the pathology (Table 2).
DC and carbonyl compounds content in the heart and blood serum of rats under the standard mode of the vivarium and injected with SkQ1 and of animals from the second experimental group injected with citrate buffer and the third group that were administered with aliquot of ethanol with mass fraction of 12% did not change significantly in comparison with the control level (Table 2).
It is known that FR may have aconitase as a target of action. Decline in activity of aconitase is interrelated with the accumulation of citrate that, in turn, is an effective low molecular weight antioxidant due to its chelating properties in relation to ions Fe2+[16].
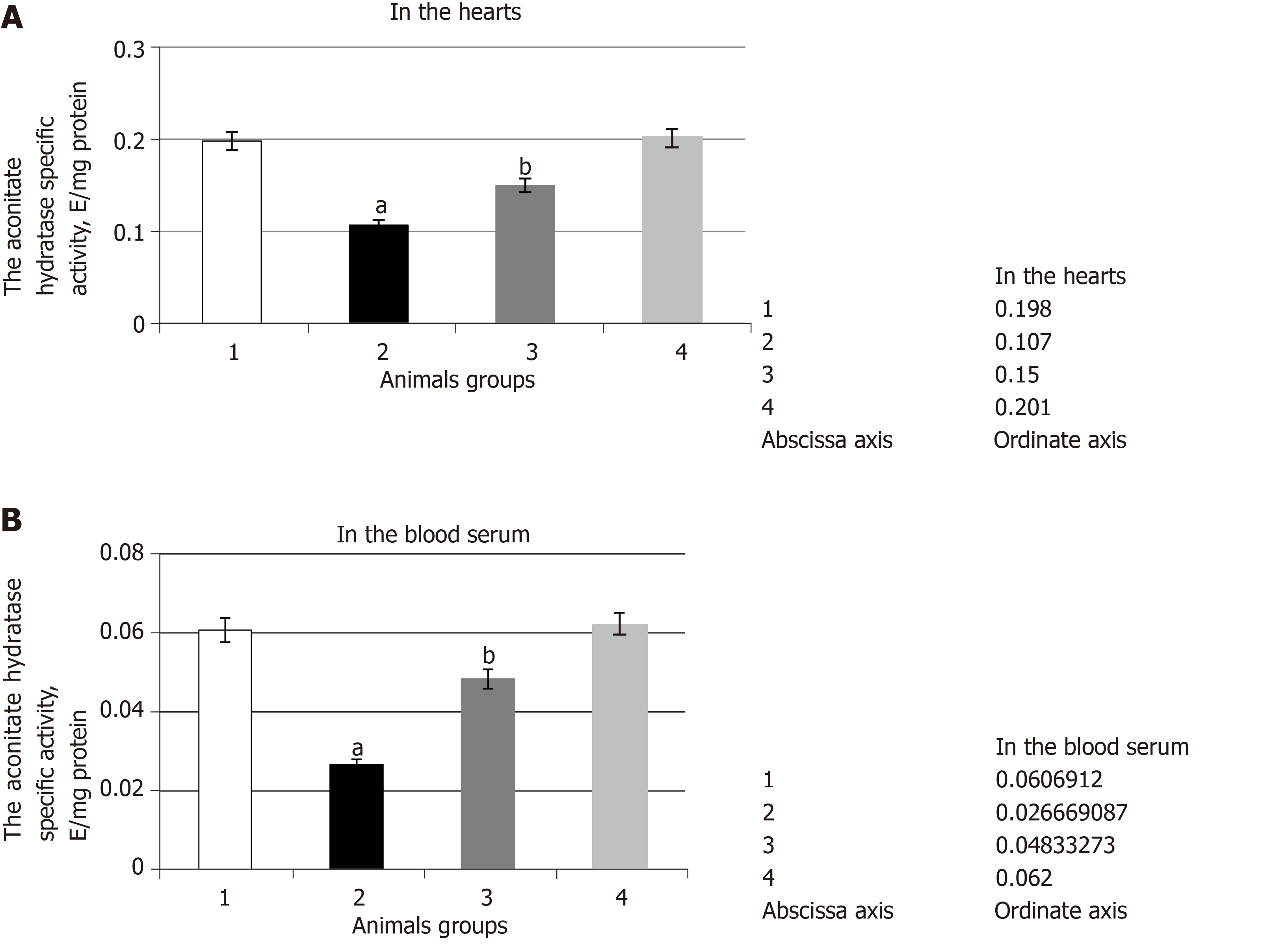
It has been established that in case of hyperglycemia, specific activity of aconitase declines 1.9 times in the heart and 2.3 times in blood serum of rats in comparison with the control level (Figure 1). The slowdown in activity of aconitase within the pathology is clearly connected with intensification of ROS formation and the induction in the development of oxidative stress. It was found that FR participate directly in the oxidation and inactivation of the aconitase iron-sulfur cluster. The long-term exposure of FR leads to the dismantlement of [4Fe-4S] cluster, carbonylation, and degradation of the enzyme.
As a result of SkQ1 exposure, the specific activity of the given enzyme increased 1.4 times in the heart and 1.8 times in blood serum in comparison with the data received from experimental hyperglycemia (Figure 1). It appears that the administration of protector in the bodies of animals with pathology facilitated a decline in the level of ROS and, as a result, a decrease in the damage degree of the molecule of the enzyme and a change in aconitase activity towards control.
Along with the development of the examined pathology, citrate content increased 2.1 times in the heart and 2.5 times in blood serum (Table 2). Presumably, it was mediated by the inhibition of aconitase. In turn, citrate accumulation could be an adaptive process with the development of oxidative stress at pathology, due to the fact that citric acid chelates iron ions and decreases the Fenton’s reaction rate. As a result, the possibility of hydroxyl radical formation declines.
It was found that the injection of SkQ1 leads to the decline of citrate content by 1.5 times in the heart and by 1.3 times in blood serum (Table 2). Presumably, the antioxidant effect provided by SkQ1 was manifested by the decline in the level of oxidative stress and by damage degree of the aconitase molecule. As a result, the activity of the given enzyme increased and led to the increase in citrate utilization rate.
The administration of SkQ1 to the animals of the fourth experimental group did not cause any statistically significant changes in aconitase activity and in heart and blood serum citrate levels in comparison with the control group. The administration of citrate buffer to the animals of the second group and of 12% ethyl solution to the third group did not cause such changes either.
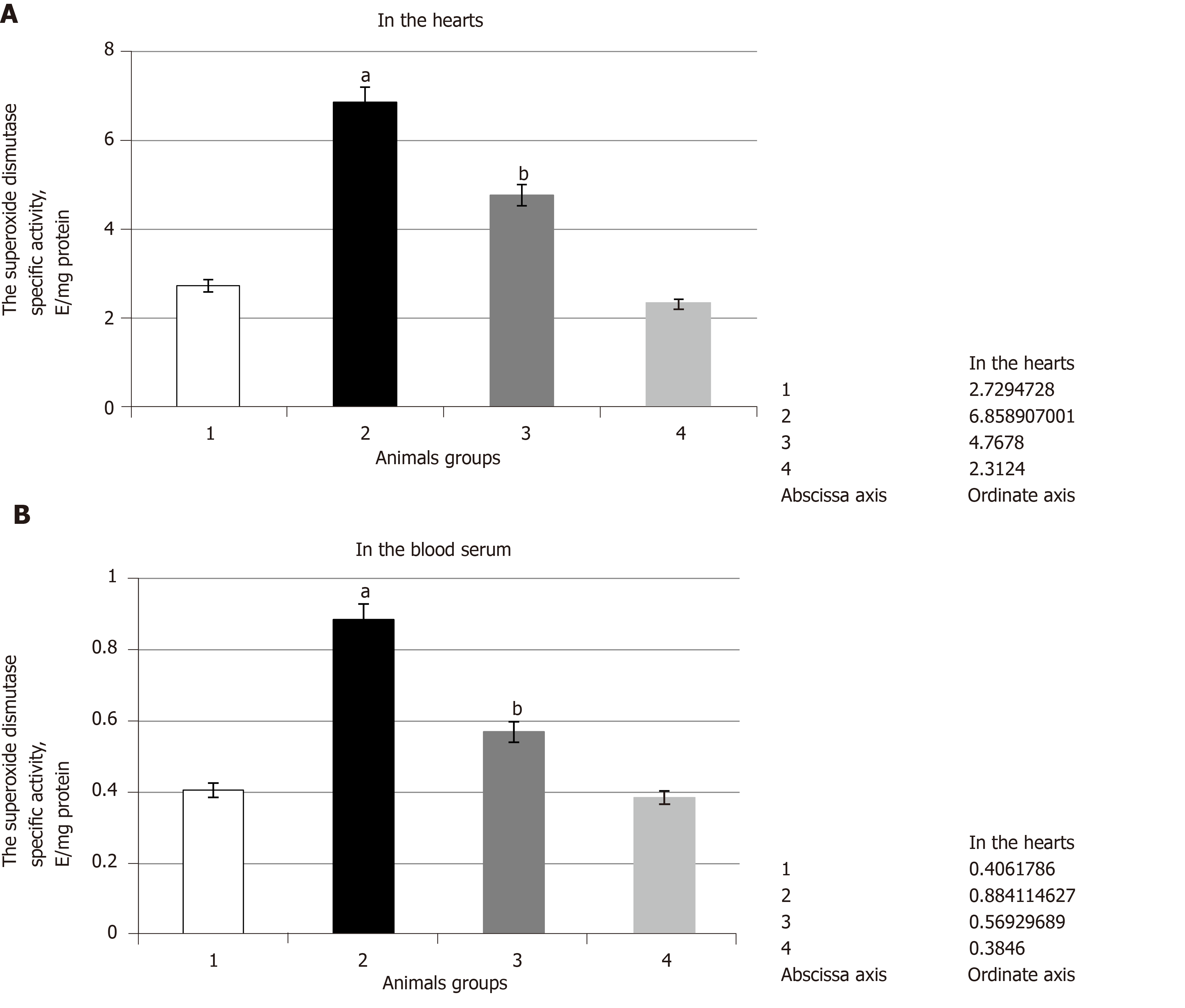
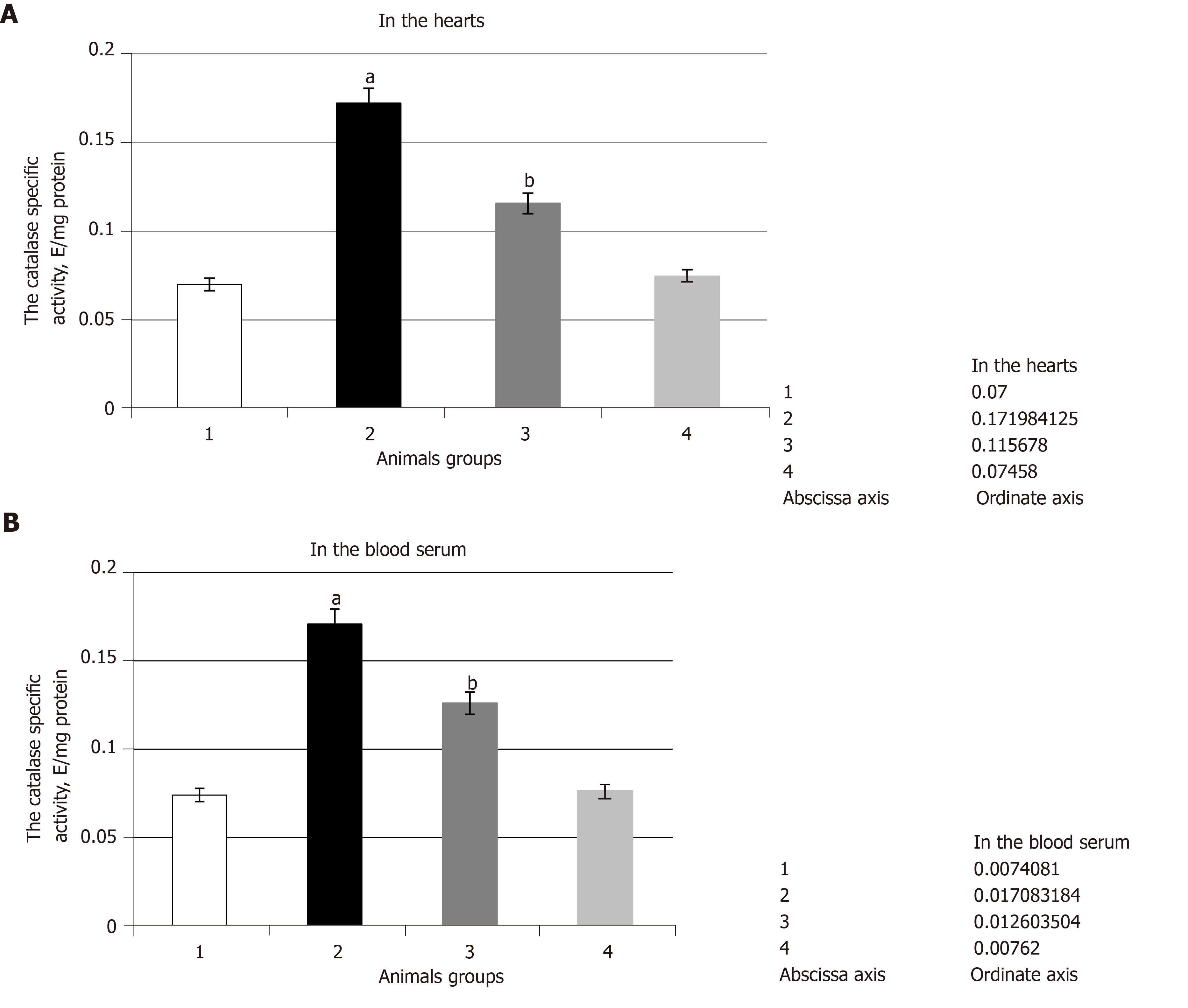
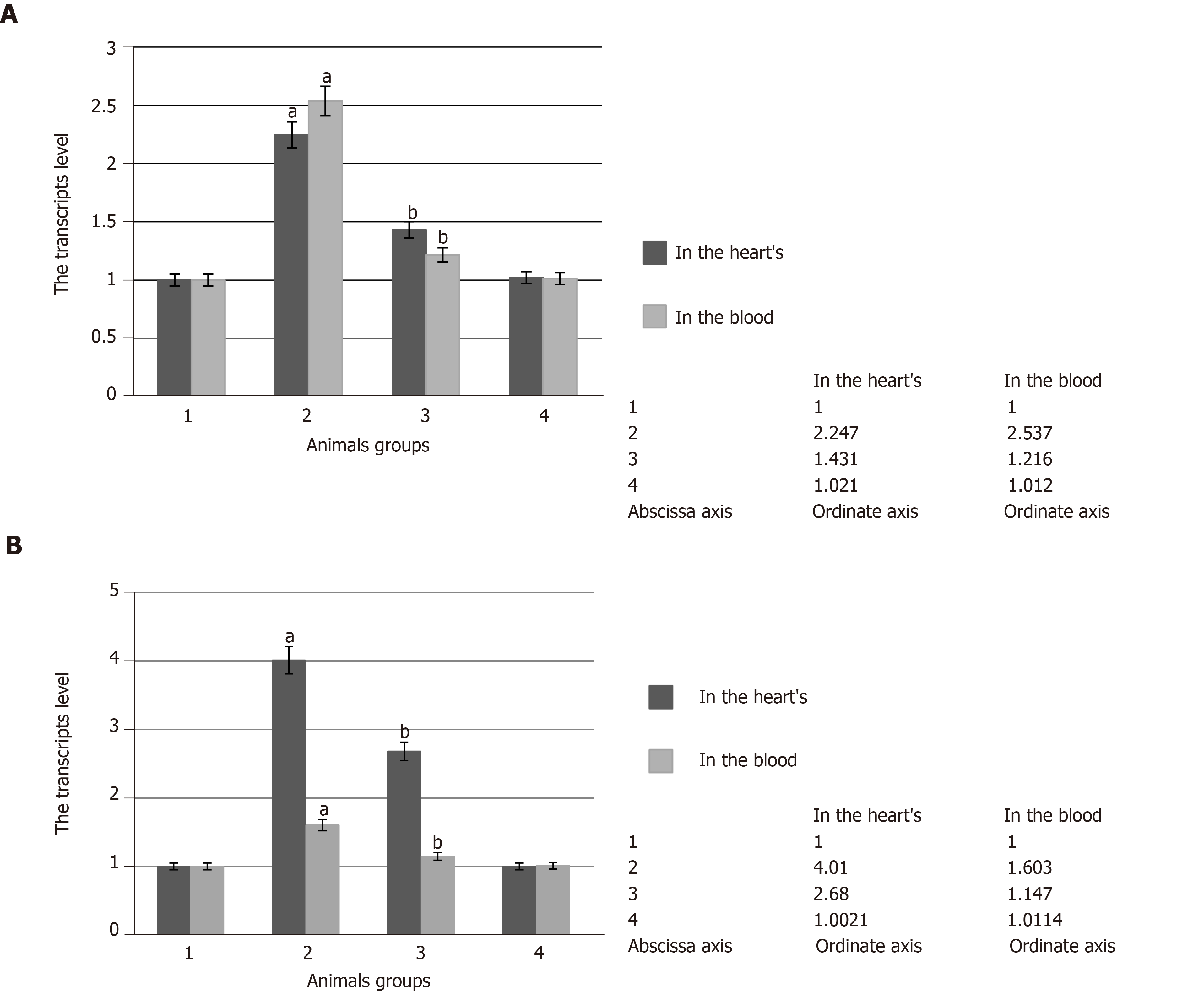
The experiments showed that pathological conditions caused the increase in SOD and catalase specific activities in comparison with the control group of the animals. They were increased by 2.5 times in the heart and by 2.2 and 2.3 times in the blood serum, respectively (Figure 2 and Figure 3 respectively). It was noted that SOD gene transcript and catalase gene transcript blood cell levels increased by 2.5 and by 1.6 times, respectively, and their heart levels increased by 2.3 and by 4.0 times, respectively (Figure 4). These enzyme activity changes in the case of hyperglycemia can probably be considered as a compensatory reaction to intensifying free-radical processes.
The administration of SkQ1 to the animals caused a decrease in SOD and catalase activities in comparison with pathological conditions. It was found that the impact of SkQ1 leads to the decrease in SOD and catalase heart and blood serum specific activities in comparison with the second group of animals. Thus, SOD heart and blood serum specific activities were decreased by 1.4 times and by 1.6 times, respectively (Figure 2), and catalase heart and blood serum activities were decreased by 1.5 and 1.4 times, respectively (Figure 3).
The identified studied enzyme activity is connected with the decrease in the number of their gene transcripts. Thus, the concentration of SOD1 and CAT gene products decreased by 1.5 and 1.3 times in the blood cells and by 1.4 and 1.3 times in the heart (Figure 4). Apparently, the reduction of free radical formation and, consequently, the reduction of oxidative stress, caused indirectly by SkQ1 antioxidant effect, were accompanied by decreased pressure on AOS, which was reflected in the approximation of SOD and catalase activities to the control values.
In comparison with the first group, the rats of the fourth experimental group, the rats of the second experimental group, to which citrate buffer was administered, and the rats of the third group, to which the aliquot of 12% ethyl alcohol solution was administered, did not have any statistically significant changes in SOD and catalase activities as well as in SOD and CAT gene transcript levels.
It is known that the inclusion of high fat content in rat diet contributes to the occurrence of animal tolerance to insulin[17]. Small dose administration of STZ leads to the moderate decline in insulin production, which is similar to a later stage of DM2[18]. Thus, the administration of STZ causes a significant basal increase in the blood glucose level. It also causes glucose tolerance, increase in the glycated hemoglobin level, significant reduction of insulin concentration, and resistance.
It should be noted that under conditions of tissue tolerance to insulin, observed during DM2, there is an increase in superoxide production by mitochondria[19], cytochrome P450, xanthine oxidase, and PKC-dependent NADPH oxidase activation. Moreover, the FR, which are generated during glucose autoxidation or glycosylation end products, can initiate LPO in lipoprotein particles[20]. The concentration of LPO products through an increase in hydrophilic hydrocarbon tails contents, in turn, can lead to formation of membrane pores and membrane stiffening through downregulation of unsaturated fatty acids, and thus it can influence state of insulin receptors, repressing their hormone connection activity, which is expressed as a reduction of the glucose consumption by cells[21]. It is possible that reduction in glucose concentration during administration of SkQ1 to rats with DM2 may be related to the realization of antioxidant properties of this substance[22].
It is well known that during several pathological states, including DM, LPO activation occurs and can lead to a number of defects, structural changes in membranes, and cell metabolic disturbance in particular. At the same time, ROS, including LPO products, act as the main POM inducers[23].
The use of SkQ1 may contribute to an inhibition of accumulation of LPO molecular products and a normalization of the structural condition of lymphocytes' membranes and their apoptosis level during oxidative stress[24].
Reviewing past literature, it is well known that through administration of SkQ1, the rate of development of such diseases as cataract, retinopathy, glaucoma, osteoporosis, hypothermia, and renal ischemia decreases[25]. Furthermore, in animal experiments, SkQ1 efficiency of correction of the several neurodegenerative states was shown[26]. The effect of SkQ1 led to acceleration of the end of the inflammatory phase, the formation of granulation tissue, vascularization, and epithelization of a wounded area[27]. Moreover, it was shown that in OXYS rats of an experimental model of accelerated aging, which is a famous animal model of human age-related macular degeneration, SkQ1 reduces clinical features of retinopathy[28].
According to the results obtained, it was found that administration of the antioxidant compound is accompanied by reduction in concentration of primary LPO products in both blood serum and heart closer to its control values.
During the experiment, it was found that an increase in activity of SOD and catalase in rats of the DM2 group may be related to induction of the synthesis of these enzymes under conditions of the oxidative stress formed in pathology.
Thus, according to the data provided by the literature, the regions reacting with nuclear factor kappa B (NF-kB) were discovered in the coding regions of all three SOD genes[29]. NF-kB is a redox-sensitive transcription factor and acts as a regulator of genes, playing a role of the actual defendant to injurious effects on a cell. It should be noted that H2O2, generated in the reaction that is catalyzed by Cu, Zn-SOD on the endosomal surface, can cause oxidation-reduction NF-κB activation. NF-κB activation leads to an increase in Cu, Zn-SOD expression[30].
It is evident that an increase in the dismutation rate of a superoxide anion radical leads to the accumulation of hydrogen peroxide. It is known that the amount of catalase is controlled by the presence of substrate[31].
It is stated that superoxide anion radicals activate various signaling systems. It includes Keap1 - Nrf2, which is responsible for the induction of a significant number of genes, including antioxidant enzyme genes[32]. Besides, c-jun N-terminal kinase, which is the main inducer activity of forkhead homeobox type O (FOXO) transcription factors, can be activated during hyperglycemia that contributes to the development of oxidative stress. FOXO can also be considered as messengers during the development of oxidative stress, since their activity is regulated by H2O2 and depends on the state of the cell[33].
It is known that protection against oxidative stress of human cardiac fibroblasts after myocardial infarction is mediated by expression of antioxidant enzymes regulated by FOXO, such as SOD, catalase, and peroxidase[34]. In addition, FOXO3a, a member of the FOXO family, induces catalase gene expression in human cells[35]. Moreover, it activates the expression of Mn-SOD by regulating the SOD2 gene after exposure to resting cells with hydrogen peroxide[36].
The increase in the activity and level of transcripts of the studied antioxidant enzymes in pathology probably could be related to the considered molecular genetic mechanisms of the redox regulation of gene expression activity under conditions of excessive generation of ROS. A decrease in the intensity of free radical oxidation under the influence of SkQ1 led to a change in the studied parameters towards control values.
In conclusion, the results of this research suggest that the mitochondria targeted antioxidant SkQ1 might be a perspective substance for incorporation into the antioxidant therapy of DM2. The experimentally revealed ability of this compound to lower the intensity of free-radical processes, acting as the key component of the pathogenesis of the DM2, may serve as the reason for this conclusion. Thus, after the introduction of SkQ1 to the animals with STZ induced hyperglycemia, the values of the BCL parameters reflecting the free radical oxidation intensity, the concentration of DC and carbonyl products of protein oxidation, AH activity, and citrate content changed towards the control values. At the same time, the activity level of the antioxidant enzymes SOD and catalase changed to the direction of normal values.
All treatments of the experiment were consistent with the requirements of the European legislation on the protection of animals (Directive 2010/63/EU).
The laboratory rats were divided into four groups: Group 1 (n = 20), animals housed under standard vivarium conditions (control group); group 2 (n = 20), animals with streptozotocin (STZ) injection-induced hyperglycemia; group 3 (n = 12), animals with STZ-induced glycemia that were administered with 10-(6-plastoquinonyl) decyltriphenylphosphonium (SkQ1) solution intraperitoneally at a 1250 nmol/kg dose per day, starting from the second week; and group 4 (n = 8), animals housed under standard vivarium conditions that were administered with SkQ1 at a 1250 nmol/kg dose per day, during the second week of conducting the experiment. Group 2 also included rats (n = 8) that were administered intraperitoneally with appropriate aliquot quantity of citrate buffer equal to pH 4.4 once a week during 2 wk after 1-mo high-fat diet. Group 3 included animals (n = 8) that were administered intraperitoneally with appropriate amount of solution without SkQ1 (98% ethanol diluted eight times with normal saline solution). The experimental unit was a single animal. Rats were kept in laboratory, and they were divided through stratified randomization by their weight and age.
Hyperglycemia was induced by feeding rats a high-fat diet for 1 mo. STZ was administered intra-abdominally at a 30 mg/kg of animal weight dose with citrate buffer equal to pH 4.4. Hyperglycemia was verified by measuring the glucose level in the blood serum with a glucose oxidase test. SkQ1 solution was administered intraperitoneally at a 1250 nmol/kg dose per day. Appropriate aliquot quantity of citrate buffer equal to pH 4.4 was administered intraperitoneally.
To conduct the study, laboratory rats were selected of nursery rat males from Federal State-financed Organization of Health Service “Voronezh Hygiene and Epidemiology Center”. The rats used for the study weighed 200-250 g and aged 3-5 mo. Rats were kept at 12-h light day, room temperature, and access to water and food ad libitum for 2 wk before the study. Type of housing – plastic. Bedding material – sawdust. Number of cage companions - two rats per cage. The total number of animals used in experiment – 76 rats. The number of animals in each experimental group: Group 1 (n = 20 + 8); group 2 (n = 20 + 8); group 3 (n = 12); and group 4 (n = 8).
The number of animals was necessary for obtaining statistically significant results. The animals were divided through stratified randomization by their weight and age. The rats used for the study weighed 200-250 g and aged 3-5 mo.
The order in which the animals in the different experimental groups were treated and assessed: Group 2 compared with group 1; group 3 compared with group 2; and group 4 compared with group 1. Weight gain, increased water intake, and slowness in rats of the second group were seen. SkQ1 administration lowered glycemic level by 2.5 times, which was initially upregulated 2.7 times relative to the control. The glucose level of the animals of the fourth experimental group was not significantly different from the stated value. Moreover, glucose concentration was within the stated value in rats of the second experimental group, having been administered citrate buffer and of the third group, having been administered aliquot of 12% ethanol. The unit of analysis was group of animals.
Experiments were done at least in 8-20 biological and two analytical replicates. The results were compared with the control. The data were statistically analyzed using software package STATISTICA 6.0 with numerical variables – arithmetical mean (M), mean error (m), and statistical significance level (P). Normal distribution data were compared by applying Student's t-test for Bonferroni correction in independent samples. Significance level was set at aP ≤ 0.0167 and bP ≤ 0.0167.
It was found that influence of the mitochondrial-directed antioxidant elicits decrease of biochemiluminescence (BCL) parameters values that increase by pathology as well as the level of primary products of lipid peroxidation such as diene conjugates and carbonyl compounds, which indicate intensity of free radical oxidation. At the same time, the activity of aconitate hydratase (AH), considered as a crucial target of FR, which was decreasing during experimental hyperglycemia, increased. Apparently, increasing activity of AH influenced the speed of citrate utilization, whose concentration was decreasing after administering SkQ1 by pathology. Moreover, the previously applied anti-oxidant administration during hyperglycemia influenced the rate of antioxidant system mobilization. Thus, superoxide dismutase and catalase activity as well as the level of gene transcript under influence of SkQ1 at pathology were changing towards control groups values.
The results of this research suggest that the mitochondria targeted antioxidant SkQ1 might be a perspective substance for incorporation into the antioxidant therapy of type 2 diabetes mellitus (DM2). The experimentally revealed ability of this compound to lower the intensity of free-radical processes, acting as the key component of the pathogenesis of the DM2, may serve as the reason for this conclusion. Thus, after the introduction of SkQ1 to the animals with STZ induced hyperglycemia, the values of the BCL parameters reflecting the free radical oxidation intensity, the concentration of diene conjugates and carbonyl products of protein oxidation, the AH activity, and citrate content changed towards the control values. At the same time, the activity level of the antioxidant enzyme SOD and catalase changed to the direction of normal values.
The effects of SkQ1 on the stage of compensatory response occurrence in pathology may contribute to decrease of the degree of oxidative stress, normalization of antioxidant system functioning, and blocking of the development of decompensation, characterized by the inhibition of protective systems.
Thus, the mitochondria targeted antioxidant SkQ1 might be considered a perspective substance for incorporation into the antioxidant therapy of DM2.
The authors are very grateful to Academician of RAS, Prof. Vladimir P Skulachev for useful advice and attention to this study.
| 1. | Jia G, DeMarco VG, Sowers JR. Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol. 2016;12:144-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 816] [Cited by in RCA: 748] [Article Influence: 74.8] [Reference Citation Analysis (4)] |
| 2. | Ametov AS. Diabetes mellitus type II. Problems and solutions. Moscow: GEOTAR-Media 2014; 1032. |
| 3. | Peterson LR, Herrero P, Schechtman KB, Racette SB, Waggoner AD, Kisrieva-Ware Z, Dence C, Klein S, Marsala J, Meyer T, Gropler RJ. Effect of obesity and insulin resistance on myocardial substrate metabolism and efficiency in young women. Circulation. 2004;109:2191-2196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 484] [Article Influence: 22.0] [Reference Citation Analysis (3)] |
| 4. | Boudina S, Abel ED. Diabetic cardiomyopathy revisited. Circulation. 2007;115:3213-3223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1256] [Cited by in RCA: 1175] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 5. | Antonenko YN, Avetisyan AV, Bakeeva LE, Chernyak BV, Chertkov VA, Domnina LV, Ivanova OYu, Izyumov DS, Khailova LS, Klishin SS, Korshunova GA, Lyamzaev KG, Muntyan MS, Nepryakhina OK, Pashkovskaya AA, Pletjushkina OYu, Pustovidko AV, Roginsky VA, Rokitskaya TI, Ruuge EK, Saprunova VB, Severina II, Simonyan RA, Skulachev IV, Skulachev MV, Sumbatyan NV, Sviryaeva IV, Tashlitsky VN, Vassiliev JM, Vyssokikh MYu, Yaguzhinsky LS, Zamyatnin Jr. AA, Skulachev VP. Mitochondria-Targeted Plastoquinone Derivatives as Tools to Interrupt Execution of the Aging Program. 1. Cationic Plastoquinone Derivatives: Synthesis and in vitro Studies. Biochemistry (Mosc). 2008;73:1589-1606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 222] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 6. | Ter-Tatevosyan LP, Sarkisyan LV, Yeranosyan LA, Arakelyan LN, Shirinyan EA, Galoyan AA. Enzymes of carbohydrate-phosphorus metabolism in the bone marrow and spleen after sympathectomy. Effects of neuropeptide PRP-1. Neurochem J. 2009;3:301–304. [DOI] [Full Text] |
| 7. | Zhang M, Lv XY, Li J, Xu ZG, Chen L. The characterization of high-fat diet and multiple low-dose streptozotocin induced type 2 diabetes rat model. Exp Diabetes Res. 2008;2008:704045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 421] [Cited by in RCA: 424] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 8. | Recknagel RO, Ghoshal AK. Lipoperoxidation of rat liver microsomal lipids induced by carbon tetrachloride. Nature. 1966;210:1162-1163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 9. | Reznick AZ, Packer L. Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol. 1994;233:357-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1916] [Cited by in RCA: 1703] [Article Influence: 53.2] [Reference Citation Analysis (0)] |
| 10. | Popova TN, Agarkov AA, Verevkin AN. Intensity of Free Radical Processes in Rat Liver under Type 2 Diabetes and Introduction of Epifamin. Acta Naturae. 2013;5:118-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Afanas’ev VG, Zaytsev VS, Vol’fson TI. A micromethod of detecting citric acid in the blood serum by photoelectrocolorimeter. Lab Delo. 1973;2:115-116. [PubMed] |
| 12. | White A, Hendler F, Smith E, Hill R, Lehman I. Fundamentals of Biochemistry: In 3 volumes. V.3. Trans. from English. Moscow: Mir 1981; 7264. |
| 13. | Góth L. A simple method for determination of serum catalase activity and revision of reference range. Clin Chim Acta. 1991;196:143-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1298] [Cited by in RCA: 1309] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 14. | Glantz S. Medico-biological statistics. Moscow: Praktika 1998; 459. |
| 15. | Zanozina OV, Borovkov NN, Shcherbatyuk TG. Free-radical oxidation at a diabetes mellitus of the 2nd type: sources of formation, components, pathogenetic mechanisms of toxicity. Sovremennyye tekhnologii v meditsine. 2010;3:104-112. |
| 16. | Matasova LV, Popova TN. Aconitate Hydratase of Mammals under Oxidative Stress. Biochemistry (Mosc). 2008;73:957-964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Chukanov CN, Dvoracka M, Iskakova SS, Kurmambayev EZh. Modeling of Type 2 Diabetes Mellitus for the Study of Medicines with Antidiabetic Activity. Nauka i zdravookhraneniye. 2004;4:4-8. |
| 18. | Srinivasan K, Viswanad B, Asrat L, Kaul CL, Ramarao P. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacol Res. 2005;52:313-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1453] [Cited by in RCA: 1315] [Article Influence: 62.6] [Reference Citation Analysis (4)] |
| 19. | Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7058] [Cited by in RCA: 6183] [Article Influence: 247.3] [Reference Citation Analysis (3)] |
| 20. | Inoguchi T, Li P, Umeda F, Yu HY, Kakimoto M, Imamura M, Nawata H. High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C-dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes. 2000;49:1939-1945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1121] [Cited by in RCA: 1120] [Article Influence: 43.1] [Reference Citation Analysis (3)] |
| 21. | Mikaelyan NP, Potemkin VV, Frantseva EYu, Kulaeva IO. Functional state of the membrane-receptor apparatus of blood cells with newly diagnosed type 2 diabetes mellitus. Problemy endokrinologii. 2012;4:40-41. |
| 22. | Voronkova YG, Popova TN, Agarkov AA, Skulachev MV. Effects of 10-(6'-plastoquinol)-decyltriphenylphosphonium (SkQ1) on oxidative status in rats with protamine sulfate-induced hyperglycemia. Biochemistry (Mosc). 2015;80:1871-1879. [DOI] [Full Text] |
| 23. | Dean RT, Hunt JV, Grant AJ. Free radical damage to proteins: the influence of the relative localization of radical generation, antioxidants, and target proteins. Free Radic Biol Med. 1991;11:161-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 133] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 24. | Teixeira J, Deus CM, Borges F, Oliveira PJ. Mitochondria: Targeting mitochondrial reactive oxygen species with mitochondriotropic polyphenolic-based antioxidants. Int J Biochem Cell Biol. 2018;97:98-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 25. | Skulachev VP, Bogachev AV, Kasparinskiy FO. Membrane Bioenergetics. Moscow: Publishing House of Moscow University 2010; 365. |
| 26. | Lakomkin VL, Kapel’ko VI. Protective effect of mitochondrial antioxidant SkQ1 at cardiac ischemia and reperfusion. Kardiologiya. 2009;49:55-60. [PubMed] |
| 27. | Demyanenko IA, Popova EN, Zakharova VV, Ilyinskaya OP, Vasilieva TV, Romashchenko VP, Fedorov AV, Manskikh VN, Skulachev MV, Zinovkin RA, Pletjushkina OY, Skulachev VP, Chernyak BV. Mitochondria-targeted antioxidant SkQ1 improves impaired dermal wound healing in old mice. Aging (Albany NY). 2015;7:475-485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 28. | Perepechaeva ML, Grishanova AY, Rudnitskaya EA, Kolosova NG. The Mitochondria-Targeted Antioxidant SkQ1 Downregulates Aryl Hydrocarbon Receptor-Dependent Genes in the Retina of OXYS Rats with AMD-Like Retinopathy. J Ophthalmol. 2014;2014:530943. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Hoffmann A, Natoli G, Ghosh G. Transcriptional regulation via the NF-kappaB signaling module. Oncogene. 2006;25:6706-6716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 448] [Cited by in RCA: 515] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 30. | Rojo AI, Salinas M, Martín D, Perona R, Cuadrado A. Regulation of Cu/Zn-superoxide dismutase expression via the phosphatidylinositol 3 kinase/Akt pathway and nuclear factor-kappaB. J Neurosci. 2004;24:7324-7334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 165] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 31. | Miroshnichenko OS. Biogenesis, physiological role, and properties of catalase. Biopolym Cell. 1992;8:3-25. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 32. | Hayes JD, Dinkova-Kostova AT. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem Sci. 2014;39:199-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1217] [Cited by in RCA: 1736] [Article Influence: 144.7] [Reference Citation Analysis (4)] |
| 33. | Essers MA, Weijzen S, de Vries-Smits AM, Saarloos I, de Ruiter ND, Bos JL, Burgering BM. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 2004;23:4802-4812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 648] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 34. | Storz P. Forkhead homeobox type O transcription factors in the responses to oxidative stress. Antioxid Redox Signal. 2011;14:593-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 271] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 35. | Nemoto S, Finkel T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science. 2002;295:2450-2452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 722] [Cited by in RCA: 682] [Article Influence: 28.4] [Reference Citation Analysis (3)] |
| 36. | Kops GJ, Dansen TB, Polderman PE, Saarloos I, Wirtz KW, Coffer PJ, Huang TT, Bos JL, Medema RH, Burgering BM. Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature. 2002;419:316-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1331] [Cited by in RCA: 1255] [Article Influence: 52.3] [Reference Citation Analysis (3)] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See:
Manuscript source: Unsolicited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: Russia
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Gabriel S, Tangvarasittichai S S-Editor: Tang JZ L-Editor: Filipodia E-Editor: Liu MY