Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116882
Revised: January 15, 2026
Accepted: February 10, 2026
Published online: May 15, 2026
Processing time: 172 Days and 7.2 Hours
The prevalence of human epidermal growth factor receptor 2 (HER2)-positive gastric cancer (GC) reflects a distinct subtype of this malignancy, characterized by unique molecular properties and clinical behavior. Revolutionary developments in the therapeutic arena of HER2-positive GC have included advances in targeted therapy, immunotherapy, and antibody-drug conjugates. This review summarizes the knowledge on the molecular basis and current therapeutic advances in HER2-positive GC, with particular focus on the recent advances in overcoming drug resistance and improving therapeutic efficacy such as antibody-drug conjugate therapies (including trastuzumab deruxtecan and disitamab vedotin) and com
Core Tip: The therapeutic profile of human epidermal growth factor receptor 2 (HER2)-positive gastric cancer is shifting to a paradigm where it is no longer dependent on the traditional HER2 inhibition. This change has been achieved by two im
- Citation: Xu JJ, Ni CX, Wang P, Qin LD, Xu JJ. Advancing human epidermal growth factor receptor 2-positive gastric cancer therapy: Toward targeted immunotherapy and antibody-drug conjugates. World J Gastrointest Oncol 2026; 18(5): 116882
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/116882.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.116882
Gastric cancer (GC) is the fifth most prevalent cancer and the fourth leading cause of cancer-related death in the world[1,2]. The modern mode of care in the management of GC is the combination of surgical excision with multidisciplinary modalities such as radiotherapy, chemotherapy, targeted therapy and immunotherapy[3]. But, since there are no specific early symptoms, many of the patients do not receive a diagnosis until the disease has reached an advanced stage, thus losing the chance to undergo a surgical procedure[4,5]. The 5-year survival rate of GC patients with peritoneal metastasis is only 6%[6], which is alarming because it is necessary to find new and effective therapeutic targets.
With the introduction of precision medicine, GC management has been transformed by molecular subtyping and therapeutic approaches based on biomarkers. Overexpression of human epidermal growth factor receptor 2 (HER2) with a reported prevalence of 15%-20% represents a critical therapeutic target and is significantly correlated with increased tumor aggressiveness and unfavorable prognosis, defining a specific molecular subtype with specific clinicopathological features[3,7-11]. The original breakthrough of the ToGA trial was the addition of trastuzumab to first-line standard chemotherapy for the treatment of HER2-positive GC[8]. But the efficacy of this and other traditional HER2-targeted strategies is fundamentally limited by several structural failures, including incomplete blockade of HER2 signaling networks, reliance on antibody-dependent cellular cytotoxicity (ADCC) which can be suppressed within an immunosuppressive tumor microenvironment (TME), failure to eradicate tumor clones with low or heterogeneous HER2 expression, and the absence of a bystander effect to target neighboring HER2-negative cells. These intrinsic constraints lead to a plateau of survival benefit with median overall survival (mOS) at about 13-16 months and the rapid evolution of resi
The challenge to overcome these barriers, therefore, has become of primary concern in the field of therapeutic innovation. The emergence of breakthroughs is redefining the treatment paradigm along two complementary axes: (1) Strategic combination regimens that involve combinations of immune checkpoint inhibitors (ICIs) with targeted therapy and chemotherapy to modulate the tumor immune microenvironment as exemplified by the KEYNOTE-811 trial[17]; and (2) The development of antibody-drug conjugates (ADCs), such as trastuzumab deruxtecan (T-DXd)[18] that are engi
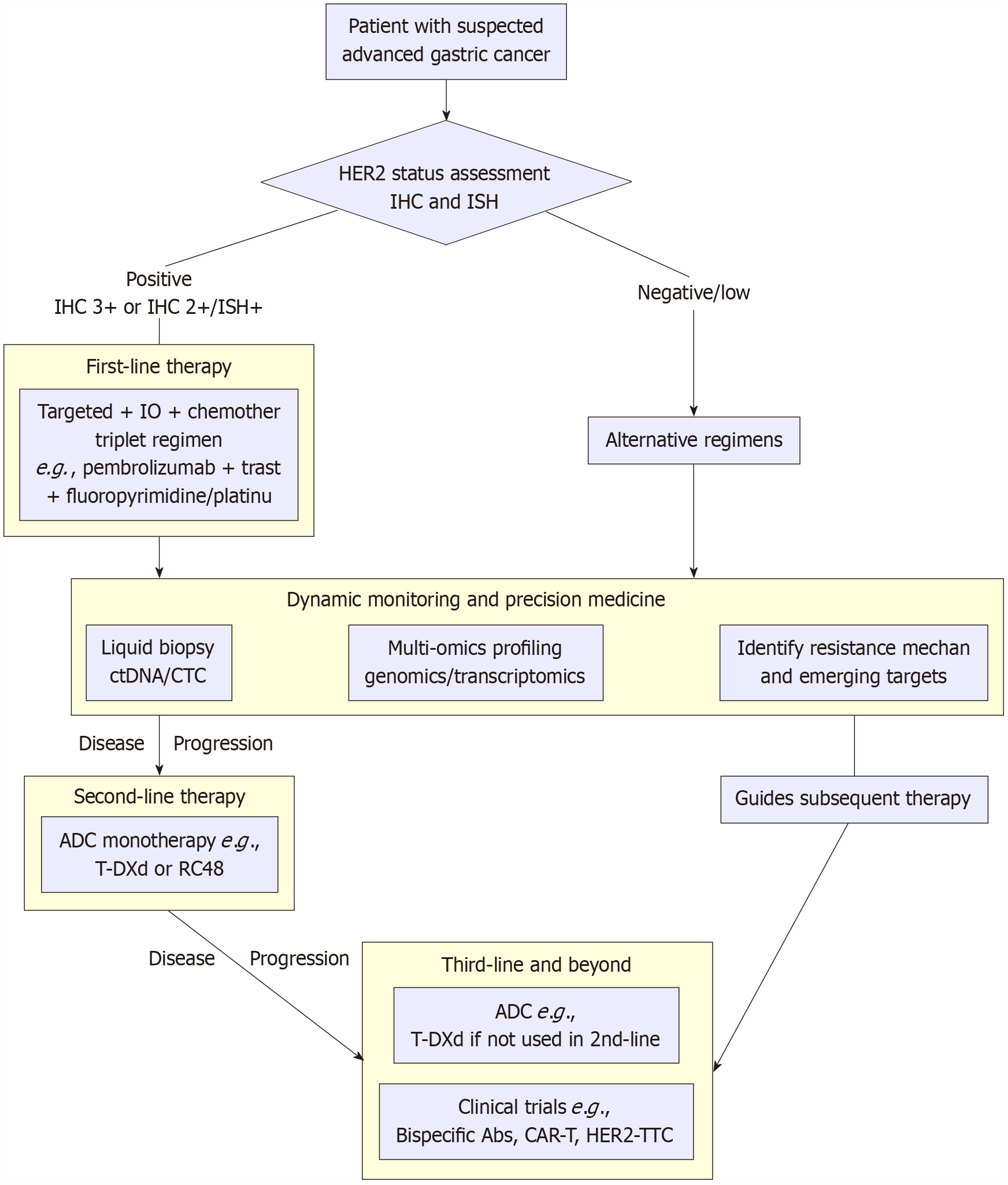
This is a systematic review that discusses the clinicopathology and therapeutic innovations of HER2-positive GC. The underlying constraints of traditional therapies are then critically examined in order to set a problem-based rationale. Next, focusing especially on the combination of targeted and immunotherapy and ADC translational research, we discuss their complementary roles and make a comparative analysis of the most relevant ADC agents. We hope to deliver a conceptual roadmap of how to optimize clinical decision-making and further develop precision oncology by synthesizing existing knowledge about resistance mechanisms in a dynamic, biomarker-informed clinical decision framework and discussing the integrative use of liquid biopsy and multi-omics technologies.
HER2 is a protein that is a key component of the family of ERBB receptor tyrosine kinase. This transmembrane gly
According to international consensus, the presence of HER2 in GC requires immunohistochemistry (IHC) 3+ or IHC 2+ with fluorescence in situ hybridization (FISH)-confirmed amplification of the gene[26]. HER2 expression shows a high level of heterogeneity with a positive correlation to histological subtypes: Intestinal-type carcinomas show high rates of HER2 overexpression of 15%-50% which are significantly higher than those of diffuse-type (2%-25%) or mixed-type tumors[27]. Subsequent research confirms that HER2 overexpression and gene amplification is significantly correlated with an intestinal-type differentiation (P < 0.05) and well-differentiated adenocarcinomas (P < 0.05)[28]. In early-stage GC, heterogeneity of HER2 expression remains, with 18% of cases being classified as HER2-positive, 31.8% as HER2-low, and 50.3% as HER2-negative[7], which also highlights its clinical relevance in regards to stratification.
The HERACLES diagnostic criteria of grading the levels of expression (0/1+/2+/3+)[29] is currently used to determine the level of HER2 expression through the application of IHC and FISH. Research indicates that there is a significant correlation between HER2-low status and clinicopathological factors like patient age, grade of differentiation of tumor, anatomical localization and the Ki-67 index[7]. Nonetheless, HER2 testing has technical issues, such as pre-analytical variability and interpretative inconsistency, which require standardized protocols of gastroesophageal adenocarcinoma to improve reproducibility[30]. The development of new anti-HER2 ADCs emphasizes the increased need to detect HER2-low tumors and reconsider the traditional binary scoring systems to enhance a stratification accuracy[31].
Mechanistically, the HER2 overexpression continues to activate the PI3K/AKT and RAS/MAPK signaling pathways, which promote tumor proliferation, survival, and metastatic potential[7], which biologically underlies the aggressive phenotype of HER2-positive GC.
Clinicopathologically, HER2-positive GC usually affects younger patients and is associated with certain features, such as intestinal differentiation, the location of the gastroesophageal junction, and high indices of Ki-67[7]. Although there are initial reports suggesting that the overexpression of the HER2 is a poor prognostic factor when it comes to disease-free survival or overall survival (OS) in early-stage disease[7], HER2-low status is not an independent poor prognostic factor in disease-free survival or OS in early-stage disease[7]. Rather, the age, differentiation status, tumor location and Ki-67 index are more influential prognostic determinants.
To summarize, HER2-positive GC is a specific molecular subtype with its clinicopathological characteristics. The accurate detection and stratification of HER2 is critical to inform individualized therapeutic approaches and to develop targeted and immunotherapeutic modalities.
The therapeutic pathway of HER2-positive GC has improved significantly; nevertheless, the traditional ones still have some fundamental structural weaknesses that restrain their overall effectiveness. The use of conventional chemotherapy, although historically the backbone of treatment in the advanced disease, is not tumor specific and thus, offers subo
| Therapeutic category | Regimen/agents | Mechanism of action | Limitations and challenges | Key clinical evidence |
| Traditional chemotherapy | Fluorouracil + platinum/taxanes | Interferes with DNA synthesis or cell division | Limited survival benefit; low response rates; short duration of response; lack of specificity; significant adverse effects | Multiple phase III trials |
| Trastuzumab + chemotherapy | Trastuzumab + chemotherapy (various combinations) | Blocks HER2 signaling; mediates ADCC; suppresses angiogenesis | Enhanced benefit in HER2-high; limited efficacy in HER2-low; primary/acquired resistance; HER2 heterogeneity affects efficacy; first-line: Median OS plateaued at 13-16 months; lack of breakthrough improvements; limited survival benefit PFS; second-line: Cross-line trastuzumab failed to improve PFS; lack of HER2 stratification; most patients eventually develop resistance | TOGA[11,14-16,32,33,37,38] |
| Other HER2-targeted agents | Pertuzumab | Forms dual blockade with trastuzumab | Failed primary endpoints in GC phase III; divergent responses between GC and breast cancer | JOSHUA, MARIANNE[40] |
| Small-molecule TKIs | Inhibits HER2 intracellular tyrosine kinase activity | Lapatinib improved ORR but not OS; lapatinib + trastuzumab not superior to monotherapy; different HER2 expression patterns; more complex signaling pathways; unique tumor microenvironment | TYTAN, BO15970[32,38,40,41] |
Being the initial targeted agent to HER2-positive GC, trastuzumab has transformed the treatment scenario. This hu
The subsequent research established a 30-percent OS benefit of a HER2-targeted therapy coupled with chemotherapy over chemotherapy alone[33]. Recent standard regimens emphasize the maximization of chemotherapy: FLOT based combinations (fluorouracil, leucovorin, oxaliplatin and docetaxel) demonstrate significant improvements in PFS and OS[33], whereas cisplatin containing ECF regimens (epirubicin, cisplatin, and fluorouracil) demonstrate relatively modest gains in PFS and OS[34]. Dual HER2 blockade in combination with chemotherapy is also associated with the higher objective response rate (ORR) and the prolonged PFS[35] but with the risk of cardiac toxicity and necessitating cardiac monitoring vigilance[36].
Additional adverse events, such as myelosuppression, require proactive management[34]. Despite the fact that HER2-targeted agents, such as trastuzumab in combination with chemotherapy have dramatically improved the prognosis of HER2-positive GC patients, traditional therapies still present significant limitations, including difficulties in dealing with primary or acquired resistance[32]. The mOS of first-line therapy has not improved with a second-line therapy (mOS = 9.6 months) due to the lack of HER2 stratification[37]. Cross-line trastuzumab therapy did not produce any improvement in ORRs or PFS (3.7 months vs 3.2 months; HR = 0.91, P = 0.33)[14] and most patients eventually develop resistance, resulting in a post-progression median PFS of 6-7 months[11]. Heterogeneity of HER2 further limits its therapeutic efficacy, with 30%-40% of IHC 2+ patients (FISH-negative) showing no clinical benefit[38]. Moreover, the conventional therapy is inadequate to appropriately modulate the tumor immune microenvironment to prevent residual tumor era
The complexity of resistance mechanisms further complicates therapeutic issues, including the dysregulation of HER2 downstream pathways (e.g., PI3K/AKT/mTOR), tumor heterogeneity, compensatory bypass signaling [e.g., mesen
To conclude, chemotherapy using trastuzumab will be a milestone in treating HER2-positive GC. Although success in the TOGA trials transformed the standards, drug resistance and limitations in efficacy of drugs underscore the unmet needs, which have driven innovation in targeted-immunotherapy combinations, ADC development, and multi-pathway inhibition to extend patient survival.
Another HER2-targeted monoclonal antibody (pertuzumab) augments the therapeutic efficacy by forming a dual block of trastuzumab through binding to different HER2 epitopes. The combination approach has shown considerable clinical advantages in breast cancer[39]. The dual blockade effectively suppresses downstream signaling and overcomes the shortcomings of single-agent targeted therapy by simultaneously targeting different regions involved in HER2 dimerization[35]. However, in GC, post-TOGA phase III clinical trials (e.g., JOSHUA and MARIANNE) of pertuzumab-trastuzumab combinations did not meet their primary endpoints, highlighting differences in response to HER2-targeted therapy between gastric and breast cancers[40]. Despite the limited research on the topic of pertuzumab in GC, its mechanistic rationale and effectiveness in other HER2-positive cancers can be valuable insights in the development of therapeutic agents.
Another important group of HER2-targeted agents includes small-molecule tyrosine kinase inhibitors (TKIs), such as lapatinib, neratinib, and pyrotinib. These medications inhibit HER2 intracellular tyrosine kinase activity, therefore, preventing downstream signaling pathways. In HER2-positive GC, phase III TYTAN trial compared lapatinib in combination with capecitabine as the second-line therapy. Although the regimen was able to improve ORRs, it was not able to achieve a statistically significant difference in OS[32]. Likewise, the BO15970 trial that compared lapatinib combined with trastuzumab showed no better result compared to trastuzumab used alone[40].
The poor performance of these targeted agents in GC clinical trials could be the result of a number of factors. The patterns of HER2 expression in GC are different as compared to breast cancer, with lower levels of expression and higher heterogeneity. Moreover, GC displays more complicated signaling pathways with common bypass activation mecha
Nevertheless, despite these difficulties, there is still a possibility of clinical utility of HER2-targeted agents. Dual blockade approach involving pertuzumab and trastuzumab can be useful in certain subgroups specifically those patients whose breast tissue is highly expressed or amplified in expression with the HER2/neu protein[41]. TKIs may be more effective in patients with particular HER2 mutations or pathway addictions[32]. Moreover, the use of these agents in combination with chemotherapy, immunotherapy, or ADCs can help address the limitation of monotherapy and improve the treatment effect[40].
Future studies must seek to determine accurate biomarkers to stratify patients who are likely to respond to targeted therapies, to improve combinations regimens and to devise new approaches to counter resistance. Real-time evaluation of the response to the treatment and the guidance of therapeutic changes could be possible with the help of dynamic monitoring through liquid biopsy[38]. As the understanding of the HER2 signaling pathways and development of novel agents deepens, these HER2-targeted therapies have the potential to be applied more precisely in HER2-positive GC.
The use of ICIs in HER2-positive GC is a considerable breakthrough in cancer immunotherapy. Although single-agent ICIs have a low response rate (20%-40%) in this population[42], emerging data indicates that the combination of ICIs and other modalities could be more effective in this group[43]. Chemotherapy in combination with immunotherapy would potentially enhance the release of tumor antigens as well as remodel the immunosuppressive TME, ultimately improving patient outcomes in HER2-positive GC. The other potentially effective technique is the combination of immunotherapy and targeted therapy. In particular, the targeted-immunotherapy-chemotherapy (pembrolizumab + trastuzumab + fluoropyrimidine/capecitabine + oxaliplatin) triplet regimen, which was proven by the KEYNOTE-811 trial[17] to be a cornerstone of first-line treatment of advanced HER2-positive GC, has become a cornerstone of the first-line treatment of advanced HER2-positive GC.
ADCs have become a game changer in cancer treatment. ADCs facilitate specific tumor targeting and reduce the off-target toxicity by conjugating monoclonal antibodies to cytotoxic payloads via linkers[44]. This strategy of biological missiles provides new treatment choices to HER2-positive GC. Integration of HER2 ADCs with chemotherapy, targeted therapy or immunotherapy can broaden treatment options[45]. Ongoing phase III trials, such as DESTINY-Gastric05 (T-DXd ± pembrolizumab + fluorouracil/capecitabine)[46] and HERIZON-GEA-01 (zanidatamab ± tislelizumab + chemo
The above examples of the established triplet regimen to the promising ADC combinations are a good illustration of how the management of HER2-positive GC is being radically changed by strategic therapeutic integration. The following chapter will proceed to discuss these new breakthroughs in more depth starting with the paradigm-shifting breakthrough of the so-called “targeted + immunotherapy + chemotherapy” triplet regimen and going on to the more advanced use of ADCs. The fundamental reasons and elements of these dominant strategies are comparatively tabulated in Table 2, highlighting the strategic change of sequential monotherapy to multi-pronged attacks aimed at overcoming the resis
| Therapeutic category | Regimen/agents | Mechanism of action | Key clinical trials/evidence | Ref. |
| Targeted + immunotherapy + chemotherapy triplet regimens | Pembrolizumab + trastuzumab + chemotherapy | Pembrolizumab: PD-1 inhibitor, reverses T-cell exhaustion; trastuzumab: Blocks HER2 signaling and induces ADCC; chemotherapy: Induces immunogenic cell death, enhancing tumor antigen presentation | KEYNOTE-811 (phase III): Became the new first-line standard for advanced HER2-positive GC/GEJA, demonstrating superior PFS and OS (median OS: 20.0 months vs 16.8 months) | Ding et al[17]; Wang et al[48]; Cheng et al[49]; Yamashita et al[51]; Li et al[55]; Zhu et al[56]; Yu et al[57]; Yi et al[58] |
| Atezolizumab + trastuzumab + XELOX | Atezolizumab: PD-L1 inhibitor, enhances antitumor immunity; trastuzumab and chemotherapy: Provides direct HER2 blockade and tumor cell killing | A phase II randomized trial: In the perioperative setting for locally advanced resectable GC, significantly improved pathologic complete response rate (38% vs 14%) | Peng et al[1] | |
| ADCs | Trastuzumab deruxtecan | HER2-targeted ADC with a topoisomerase I inhibitor payload and a cleavable linker, enabling a potent “bystander killing effect” against heterogeneous tumors | DESTINY-gastric series: DG-04 (phase III): Redefined 2nd-line standard (median OS: 14.7 months vs 11.4 months). DG-01 (phase II): Robust activity in later-line (ORR = 51.3%). DG-03/05: Evaluating 1st-line combinations | Shitara et al[46]; Oaknin et al[59]; Aoki et al[60]; Chen et al[61]; Janjigian et al[62]; Janjigian et al[63]; Shitara et al[64]; Shitara et al[65]; Yamaguchi et al[66]; Peng et al[67] |
| Disitamab vedotin | HER2-targeted ADC with the microtubule inhibitor MMAE, enabling precise cytotoxicity and efficacy in HER2-low expressions | Phase I/II trials: Showed promising efficacy in heavily pretreated patients with HER2-overexpressing and HER2-low GC | Chen et al[61]; Xu et al[69]; Peng et al[70] | |
| Ado-trastuzumab emtansine | HER2-targeted ADC with the maytansinoid payload DM1, utilizing a stable, non-cleavable linker | Phase III trials (e.g., GATSBY): Failed to demonstrate superior survival benefit over standard chemotherapy in GC, limiting its clinical application | Pegram et al[45]; Barfield et al[71] | |
| Investigational ADC agents | A166 | A site-specifically conjugated ADC with a uniform drug-antibody ratio (approximately 4), delivering the potent microtubule inhibitor duostatin-5 | Early-phase I trials: Showed preliminary antitumor activity in HER2-positive solid tumors (including GC), primarily in breast cancer models to date | Zhang et al[20]; Hu et al[73]; Liu et al[74]; Hu et al[75] |
| LCB-ADC | A novel ADC with an optimized cleavable linker and MMAF payload, designed for enhanced tumor-specific payload release and a wider therapeutic window | Preclinical studies: Demonstrated superior potency and efficacy in HER2-high and ado-trastuzumab emtansine-resistant patient-derived xenograft models | Shin et al[21]; Díaz-Rodríguez et al[68]; You et al[72] |
The use of ICIs and the HER2-targeted agents is an extremely crucial development in the management of the HER2-positive GC. ICIs can overcome immune suppressive TME which may overcome resistance to HER2-targeted therapy[48]. This synergistic strategy takes advantage of complementary activities: The HER2-targeted agents directly suppress tumor growth and cause ADCC, and increase the tumor immunogenicity with antigen release[48]. At the same time, the T-cell antitumor activity is restored by the inhibitors of the programmed cell death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) interaction of the immune checkpoints[49]. This dual modulation of the tumor cells as well as the immune microenvironment create a synergistic effect, increasing overall antitumor responses[48].
The efficacy of the pembrolizumab, trastuzumab and chemotherapy triplet regimen in the treatment of the HER2-positive advanced gastroesophageal adenocarcinoma was conclusively established by the phase III KEYNOTE-811 trial[17]. The ultimate analysis proved the statistically significant increase in OS, which has reduced the risk of dying by 16% [mOS: 20.0 months vs 16.8 months; HR = 0.84, 95% confidence interval: 0.70-1.01]. This benefit was enhanced in the PD-L1 combined positive score (CPS) ≥ 1 subgroup (mOS: 20.1 months vs 15.7 months; HR = 0.81, 95% confidence interval: 0.67-0.98). The study has offered a conclusive new first-line standard which can provide critical direction to clinical practice all over the world.
Recent investigator-initiated phase II randomized trial evaluated atezolizumab, in combination with trastuzumab and XELOX, as perioperative treatment against locally advanced, resectable, HER2-positive gastric/gastroesophageal junction cancer (cT3-4b/N+/M0). The patients underwent three cycles of neoadjuvant therapy then underwent surgery and five cycles of adjuvant therapy. The experimental arm (atezolizumab + trastuzumab + XELOX) had a much higher pathologic complete response (pCR) rate than did the control arm (trastuzumab + XELOX) (38% vs 14%, difference 23.8%). Subgroup analyses indicated an increased pCR rate in patients with the age of < 65 years, males, and with an intestinal-type Lauren classification[1]. The study is the first to use triplet therapy in the perioperative environment in the case of locally advanced disease, and it shows a significant enhancement of pCR and even survival advantages.
It has been confirmed in numerous studies that such combination regimens have a major impact on increasing the rate of response to treatment and also increases survival in clinical practice. These strategies, in comparison to monotherapy, not only enhance ORR, but also demonstrate the potential to prolong PFS and OS[48]. Moreover, the combination therapies can also assist in overcoming the resistance to the HER2-targeted agents, which will allow extending their therapeutic effect[49]. The marked efficacy of the “targeted-immunotherapy-chemotherapy” triplet regimen in HER2-positive GC arises from multidimensional synergistic mechanisms[50,51].
Efficacy of the triplet regimen is as a result of multidimensional synergy. Clinical evidence shows that immune parameters such as high levels of tumor-infiltrating lymphocytes (TILs) predict improved responses to the HER2-targeted therapy and extended survival[52]. Trastuzumab engages the natural killer (NK) cells via ADCC to form an immune-activated microenvironment[53,54], and also to induce tumor PD-L1 expression through the release of interferon-γ[51]. Immunogenic cell death (ICD) is caused by chemotherapy and releases tumor antigens and damage-associated molecular patterns that enhance the presentation of antigens and priming of T-cells[55], and depletes immunosuppressive cell populations to potentiate ICI efficacy[56]. PD-1/PD-L1 inhibitors also reverse a T-cell state of exhaustion[57,58], coo
The toxicity profile of triplet therapy is additive and defined by the individual components, including immune-related adverse events (irAEs) and HER2-targeted toxicities (e.g., cardiotoxicity and gastrointestinal reactions)[49]. It is crucial to have a multidisciplinary management of irAEs with oncologists, cardiologists, and immunologists involved to enable the early recognition and management of irAEs to avoid severe complications[49]. The issue of patient selection continues to be critical in enhancing therapeutic outcomes. Stratification of treatments, based on predictive biomarker expression, such as PD-L1 expression, tumor mutational burden, microsatellite instability status, and TIL levels may be used to guide treatment stratification[49]. With the currently studied PD-1/PD-L1 inhibitors (e.g., pembrolizumab, nivolumab, atezolizumab) there are variations in efficacy, safety and dosing schedules, which require further optimization of combination regimens and treatment sequences[48].
In the future, the paradigm of treatment of HER2-positive GC will focus more on the individual approach. Future studies will focus on the biomarker-directed combinations (e.g., ICIs + HER2-targeted agents + antiangiogenics) and new ADC-based regimens[35]. The future of next-generation HER2-targeted agents, such as BsAbs, optimized ADCs, and novel TKIs, has potential in increased therapeutic efficacy[48,49]. At the same time, improving precision medicine will be achieved by enhancing treatment duration, toxicity management guidelines, and predictive biomarker panels. These strategies are supposed to be confirmed by emerging clinical trials, and eventually enhance survival and quality of life.
Finally, the example of the so-called ICI + HER2-targeted + chemotherapy is the illustration of the potential of changing the perspective on the integration of precision oncology and immunotherapy. This methodology overcomes several vital issues in managing HER2-positive GC with a new sense of hope in the patients. Further research of new combinations and biomarker-based approaches will continue to cement its importance in clinical practice and usher a new era in the struggle against this malignancy.
Although it has a transformative efficacy, the success of the triplet regimen is necessarily limited by the fact that it requires the presence of an already established immune-active TME. Patients with low TILs, low PD-L1 levels, or other immunosuppressive aspects can, potentially, benefit little[52]. This crucial constraint highlights the necessity of additional therapeutic approaches that could effectively target tumors irrespective of underlying immune condition. In that aspect, ADCs will be a synergistic and complementary model. Compared with the triplet regimen that restructures and exploits antitumor immunity, ADCs express high levels of cytotoxic payloads directly into HER2-expressing cells, and have shown antitumor activity that is largely independent of host immune status[59-61]. In addition, some ADCs can cause ICD[55] and therefore potentially cause a conversion of immunologically “cold” tumors to the immunologically “hot” tumors and provide a mechanistic explanation of sequential or combination immunotherapy. This theoretical complementarity does not make the two pillars of contemporary HER2-positive GC-based therapy independent modalities, but rather complementary parts of an integrated, resistance-defying, therapeutic arsenal.
T-DXd: T-DXd, a novel ADC, is a combination of an anti-HER2 antibody, a cleavable tetrapeptide linker, and a payload of a topoisomerase I inhibitor. It is designed in a unique way that involves a “bystander killing effect” that allows it to be used as antitumor activity in not only the HER2-positive cells but also the adjacent HER2-negative tumor cells[59,60]. This process enables T-DXd to specifically target the cancer cells by binding the receptors and releasing the lysosomal payloads, thus destroying the tumor cells selectively. The series of clinical trials called the DESTINY has provided strong evidence on its use in HER2-positive GC and gastroesophageal junction adenocarcinoma (GEJA).
The DESTINY-Gastric03 trial[62,63] tested the efficacy and safety of T-DXd [5.4 mg/kg intravenous (IV) every 3 weeks] combined with fluoropyrimidine (5-fluorouracil 600 mg/m2 continuous IV or capecitabine 750 mg/m2 twice a day) in patients with HER2-positive GC/GEJA resistant to trastuzumab and fluoropyrimidine. Preliminary results demonstrated favorable clinical activity. The current phase III DESTINY-Gastric05 trial[46] will compare T-DXd + fluorouracil ± pembrolizumab vs the standard chemotherapy + trastuzumab ± pembrolizumab as the first line therapy of unresectable, locally advanced, or metastatic HER2-positive GC/GEJA (PD-L1 CPS ≥ 1).
In HER2-positive advanced GC patients who progressed after trastuzumab-based therapy, the phase III DESTINY-Gastric04 trial[64] compared T-DXd (6.4 mg/kg IV every 3 weeks) to ramucirumab plus paclitaxel as second-line therapy. T-DXd significantly improved mOS (14.7 months vs 11.4 months; HR = 0.70, P = 0.0044), PFS (6.7 months vs 5.6 months; HR = 0.74, P = 0.0074), ORR (44.3% vs 29.1%, P = 0.0006), and disease control rate (DCR) (91.9% vs 75.9%). Treatment-emergent adverse events (TEAEs) of grade ≥ 3 were manageable (68.0% vs 73.8%) and serious TEAEs (41.0% vs 43.3%). Nevertheless, 13.9% of T-DXd patients (33 grade 1-2 cases; 1 grade 3 cases) had been diagnosed with drug-related interstitial lung disease, requiring close pulmonary monitoring. Taken together, these findings resulted in T-DXd being the first HER2-targeted therapy to enhance OS in this patient population, setting a new standard on second-line treatment[65].
In later-line settings, the phase II DESTINY-Gastric01 trial[66] enrolled 187 HER2-positive advanced GC patients who had received ≥ 2 prior therapies (including trastuzumab). T-DXd achieved an ORR of 51.3%, a mOS of 12.5 months, and a DCR of 85.7%. Consistent efficacy in Chinese populations was confirmed in the phase IIIb DESTINY-Gastric06 trial[67], which reported a mOS of 12.4 months after excluding four coronavirus disease 2019-related deaths, supporting T-DXd’s use in this population.
Exploratory analyses of DESTINY-Gastric01[66] showed a difference in response to T-DXd in HER2-low [IHC 2+/in situ hybridization (ISH)- and IHC 1+] metastatic GC/GEJA. Current research is seeking to establish the therapeutic potential of T-DXd in HER2-low subsets, especially when used with other strategies.
In short, T-DXd exhibits a great clinical value in HER2-positive GC due to its unique mechanism, high antitumor activity, and manageable safety profile. With the continued development of clinical evidence and the growth of indi
Disitamab vedotin: Disitamab vedotin (RC48-ADC) is a new humanized anti-HER2 ADC that is comprised of an anti-HER2 immunoglobulin G1 antibody, a cleavable linker and an antimicrotubule, monomethyl auristatin E (MMAE). It has a dual mechanism that includes inhibition of HER2-signaling and cytotoxic functions of MMAE that allows precise delivery of the cytotoxic payload to HER2-positive tumor cells[61].
The preclinical research reveals the strong antiproliferative and pro-apoptotic effects of RC48-ADC on HER2-positive GC cells[68]. Dose-dependent antiproliferative activity, especially in the case of HER2-positive cell lines, is demonstrated in vitro[61]. In vivo studies with HER2-expressing GC patient-derived xenograft (PDX) models demonstrate better antitumor activity of RC48-ADC than trastuzumab even in models with moderate-to-low HER2 expression[61]. RC48-ADC, acting at a mechanistic level, inhibits HER2 phosphorylation, induces G2/M phase arrest and activates apoptosis pathways[61].
The phase I clinical trial involved the use of RC48-ADC in the treatment of HER2-overexpressing advanced solid tumors, using an open-label dose-escalation/expansion design. The maximum tolerated dose was found to be 2.5 mg/kg with the recommended phase II dose being 2.0 mg/kg with a DCR of 49.1%[69]. These results confirm the clinical activity of RC48-ADC in the treatment of HER2-positive GC.
The effect of RC48-ADC was further proven by a phase II trial that involved 125 HER2-overexpressing GC patients and demonstrated an ORR of 24.8, a median PFS of 4.1 months, and a mOS of 7.9 months[70]. It is noteworthy that even in highly pretreated population, clinical benefit can be observed with ORRs of 19.4 and 16.9 in patients who have undergone 2 and 3 prior therapies, respectively[70]. HER2-low (IHC 2+/FISH-negative) GC is also promising activity, a population that has traditionally been lacking in effective targeted options[61,70].
Safety studies indicate that there are manageable toxicity profiles that are in line with the mechanism of action of RC48-ADC. Hematologic (leukopenia, neutropenia, anemia) and non-hematologic (alopecia, fatigue, hypoesthesia)[69,70] or adverse events are common. The majority of events are grade 1-2, and dose adjustments and supportive care prove effective to reduce the risks. In the phase II trial, serious adverse events were experienced by 36.0% of the patients with neutropenia (3.2%) being the most frequent RC48-ADC-related serious adverse event[70].
Finally, RC48-ADC shows clinically significant antitumor activity and an acceptable safety profile across HER2-expressing GC populations and treatment-refractory and HER2-low subgroups. Further improvements to the treatment outcomes could be made by future studies that explore the combination of strategies with an immunotherapy or other targeted agents.
Ado-trastuzumab emtansine: The first-in-class HER2-targeted ADC, ado-trastuzumab emtansine (T-DM1) has proven to play a critical role in the HER2-positive malignancies. T-DM1 is a site-specific cytotoxic targeted drug but retains the inherent antitumor activities of trastuzumab including ADCC[71]. The therapeutic conjugate selectively binds to HER2-overexpressing cells, followed by receptor-mediated internalization and lysosomal degradation, which enables intracellular release of DM1 and exert cytotoxic effects[71].
Despite its clinical success in HER2-positive breast cancer (e.g., the phase III KATHERINE trial: HR = 0.50 for invasive disease-free survival; P < 0.001)[45], T-DM1 has demonstrated limited therapeutic efficacy in HER2-positive GC. Compared with conventional chemotherapy, comparative clinical trials have found no statistically significant survival advantage in advanced GC populations[45]. The origin of this discrepancy must have both been biological aspects unique to the GC, as well as inherent constraints in the design of T-DM1. Pharmacokinetic studies reveal that there is poor exposure to drugs because of dose-limiting toxicities, especially those involving hematologic and hepatic adverse effects, that limits the highest possible plasma concentrations[71]. In addition, the heterogeneous drug-to-antibody ratio (mean drug-antibody ratio = 3.5, range = 0-8) of T-DM1 leads to increased off-target toxicity and reduced therapeutic index in GC[71].
The overall clinical use of T-DM1 is limited by several drawbacks. Firstly, its therapeutic efficacy in HER2-positive GC is limited by toxicity-related limitations on maximum serum concentrations, making optimum drug exposure impossible[45]. Compromising further efficacy is the suboptimal pharmacokinetic properties such as delayed internalization kinetics inefficiently delivering intracellular payloads. There are also additional mechanisms of resistance that include loss of lysosomal transporter solute carrier family 46 member 3 and upregulation of drug efflux pumps multidrug resistance protein 1 and multidrug resistance-associated protein 1[45]. Importantly, the absence of bystander effects by T-DM1 constrains its activity in tumors with heterogeneous HER2 expression, a hallmark of gastric malignancies[45]. These interacting issues highlight the necessity of next-generation ADCs designed to overcome pharmacokinetic bottlenecks, resistance pathways, and address TME heterogeneity.
Emerging HER2-targeted ADCs: Developments in ADC technologies have stimulated the development of next-generation HER2-targeted agents to overcome the limitations of current therapies, including narrow therapeutic indices, distinctive toxicities, resistance to therapy, and suboptimal biomarker selection[44]. These new ADCs use optimized tumor specific antibody-linker-payload architectures which reduce systemic toxicity and increase tumor specificity. Site-specific conjugation is among the innovations that enhance homogeneity and stability, leading to positive pharmacokinetic profiles[72].
A166 is a new HER2-targeted ADC containing a trastuzumab-biosimilar antibody site-specifically conjugated to duostatin-5, a microtubule-disrupting derivative of monomethyl xanthine F via protease-cleavable valine-citrulline linkers[73]. This design guarantees consistent drug loading (drug-antibody ratio of about 4), which increases the accuracy with which payloads are delivered. A166 is targeted to HER2-expressing refractory tumors that are resistant to trastuzumab or T-DM1[20].
Its therapeutic potential is proven by the phase I trials in HER2-positive breast cancer. A dose-escalation study (0.3-4.8 mg/kg, n = 35) reported no dose-limiting toxicities, with efficacy signals emerging at ≥ 3.6 mg/kg. Ocular adverse events were reported in 80% and 83% of patients at the doses of 3.6 mg/kg and 4.8 mg/kg, respectively, but were mostly grade 1-2 and reversible. Out of 27 evaluable patients, 7 patients (DCR = 59%) had partial responses, and 9 patients had stable disease[74]. Dose-proportional pharmacokinetics (half-life 1.17-11.04 days) coupled with activity at therapeutic doses (ORR: 59.1% at 4.8 mg/kg; 71.4% at 6.0 mg/kg) was confirmed in a Chinese trial (n = 57; 61.4% with ≥ 5 prior therapies)[73]. Another study showed best ORRs of 73.91% (4.8 mg/kg) and 68.57% (6.0 mg/kg), alongside median PFS of 12.3 months and 9.4 months, respectively. Genomic analyses found fibroblast growth factor receptor (FGFR) 1 amplification as a potential negative predictor of response[75].
HER2-positive gastroesophageal tumors (n = 23, including 7 gastric/esophageal cases) showed antitumor activity at 3.6-4.8 mg/kg, and partial responses in 4/8 evaluable patients (DCR = 75%)[20]. The safety analyses across trials show that manageable toxicities: Grade 1-2 ocular events (blurred vision, dry eye) and peripheral neuropathy were predo
In short, A166 demonstrates a strong antitumor activity (ORR: 59.1%-73.91%; DCR ≥ 59%) in heavily pretreated HER2-positive breast cancer at doses of 4.8-6.0 mg/kg, with a toxicity profile dominated by reversible ocular and neurological events[73-75]. Early warnings in gastroesophageal cancers and biomarker-driven knowledge (e.g., FGFR1 status)[75] should be validated on greater cohorts. The small sample sizes, under-representation of non-breast tumors, and lack of verification of dosing thresholds all indicate the necessity of phase II/III trials to establish its clinical utility.
LCB-ADC is a new HER2-targeted ADC consisting of trastuzumab in combination with monomethyl xanthine F through a cleavable linker that is meticulously engineered[21]. Preclinical trials confirm that it has two mechanisms: Direct cytotoxicity of tumor cells against HER2 and topoisomerase II, which inhibits DNA replication and repair to enhance antitumor activity[68]. LCB-ADC has a better cytotoxic potency and a higher proportion of cells in G2/M-phase cell cycle arrest as compared to T-DM1[21], which is probably due to its optimized linker system, which promotes tumor-specific payload delivery. Its special linker structure allows the efficient release of the payload within the TME and reduces the off-target toxicity consequently increasing the therapeutic window[72].
LCB-ADC demonstrated much greater tumor growth inhibition over trastuzumab or T-DM1 in the HER2-high N87 xenograft models[21]. It is also notable that it was also able to demonstrate strong efficacy in the HER2-positive GC PDX models[21]. Moreover, LCB-ADC maintained potent antitumor effects in models with T-DM1-resistance such as HER2-low JIMT-1 xenografts and PDX tumors[21], and has therapeutic potential in traditionally recalcitrant cancers. The reason is the enhanced biostability and payload retention, which is due to their advanced linker chemistry[21].
Continued phase I clinical trials are based on preclinical demonstrations of the superiority of LCB-ADC over existing ADCs, such as increased cytotoxicity, higher G2/M arrest rates, and greater tumor suppression in HER2-high models[21]. Its effectiveness in both HER2-positive gastric PDX models and resistance to T-DM1-treatment of HER2-low context further supports its translational promise.
Overall, LCB-ADC and A166 represent the groundbreaking steps in the field of HER2-targeted therapy of GC. Through the surmounting of the drawbacks of the conventional ADCs including narrow therapeutic indices and resistance mechanisms these next-generation agents exhibit attractive preclinical and early clinical activity. Massive trials should be conducted to prove their potential in offering new treatment options, especially to patients who are refractory to current therapies.
Comparative analysis of leading ADCs and future therapeutic integration: The very specific profile of T-DXd and RC48 requires the absolute comparative analysis to inform the clinical decision-making process and research priorities with key differences summarized in Table 2.
Mechanistic and efficacy profile: The main difference between such agents is the cytotoxic payloads. T-DXd is a topoisomerase I inhibitor with the ability to cause irreparable DNA double-strand breaks and exert a potent bystander effect. This characteristic allows the removal of HER2-low or HER2-negative cells adjacent to it, making T-DXd especially effective against heterogeneous tumours[59,60]. By comparison, RC48 uses the cell cycle disrupting agent MMAE, which mostly causes cell cycle arrest[61]. T-DXd has been shown to have clinically superior benefits in terms of OS, in global phase III trials (e.g., DESTINY-Gastric04[64]) with a median OS of 14.7 months vs 11.4 months in the control arm (HR = 0.70). RC48 has demonstrated strong and consistent antitumor activity in later-line settings with significant impact of clinical evidence largely based on Chinese populations, which makes it a cornerstone therapy in the region[61,70].
Safety and accessibility: The two agents have significant differences in their safety profiles. A well-characterised risk of interstitial lung disease/pneumonitis is linked to T-DXd, which requires proactive monitoring and early treatment[64,65]. In comparison, RC48 has a more traditional toxicity profile which is mainly marked by the hematological adverse events and peripheral sensory neuropathy[69,70]. Regional disparities in accessibility also affect clinical usage. T-DXd has now gained global regulatory approval, whilst RC48 approvals and wide real-world experience are now concentrated in Asia, therefore, defining region-specific treatment algorithms.
Synthesis and future direction: This comparative discussion brings up a critical question for the field: Should future research focus more on optimization of next-generation ADC monotherapy or the design of rational ADC-based combination strategies. Evidence available supports a non-mutually exclusive, biomarker guided approach. Tumors with high and homogeneous levels of HER2 may be adequately treated with increasingly potent ADC monotherapy. Nonetheless, in order to effectively respond to the multidimensional resistance environment, such as antigen heterogeneity, an immunosuppressive TME, and compensatory pathway activation[76], the use of rational combination strategies is likely to be needed. Of these, incorporation of ADCs with immunotherapy is an especially promising avenue[77]. ADCs have the potential to achieve a significant tumor debulking effect and induce ICD[55] with the potential to sensitize the TME to immune checkpoint blockade. This synergistic idea is actively being clinically tested (e.g., DESTINY-Gastric05[46]). In this regard, the future therapeutic paradigm would be predicted to shift to a dynamic and context-dependent therapeutic approach, using ADC monotherapy in favorable environments and strategically implementing ADC-immunotherapy or ADC-targeted combinations in more complex or resistant disease states, guided by continuous biomarker monitoring.
The ongoing problem of treatment resistance in HER2-positive GC requires treatment approaches that are not limited to traditional paradigm. This section discusses the state of the art in drug development by describing three paradigm-shifting modalities: BsAbs, chimeric antigen receptor (CAR) T-cell immunotherapy (CAR-T) cell therapy, and targeted thorium conjugates (TTC). They all have a unique mechanism of action to get around resistance and achieve more precise and powerful control of tumors, as summarized in Table 3.
| Therapeutic category | Regimen/agents | Mechanism of action | Current limitations/challenges | Ref. |
| Bispecific antibodies | PRS-343 | Targets HER2 and 4-1BB, promoting T-cell proliferation and cytokine production via HER2-dependent 4-1BB clustering | Preclinical stage; clinical validation needed for potential risks like CRS | Hinner et al[78] |
| IBI315 | Concurrently blocks PD-1 and HER2 signaling, establishing a self-reinforcing immunostimulatory cycle via gasdermin B-mediated pyroptosis | Efficacy and safety need confirmation in large-scale clinical trials | Lin et al[79] | |
| KN026 | Recognizes two distinct HER2 domains, achieving potent dual HER2 signal blockade | Optimal combination regimens and long-term benefits need exploration, despite promising ORR (56%) | Ji et al[80] | |
| CD40 × HER2 | Activates CD40 signaling to repolarize macrophages towards the M1 antitumor phenotype, reversing C-C motif chemokine ligand-driven resistance | Preclinical stage; long-term in vivo safety and efficacy require evaluation | Sun et al[82] | |
| IMM2902 | Targets CD47 and HER2, stimulates macrophages to recruit T and NK cells via CXCL9/CXCL10 | Clinical potential awaits validation in human trials | Zhang et al[83] | |
| CAR-T cell therapy | HER2 CAR-T | Genetically engineered T cells express HER2-specific CARs for targeted tumor cell elimination | Immunosuppressive TME; risk of on-target/off-tumor toxicity; cytokine release syndrome and neurotoxicity; antigen heterogeneity | Budi et al[85]; Xu et al[86]; Qi et al[88]; Simon et al[89]; Guzman et al[90] |
| ARC-T cells | Achieves selective tumor cell killing while minimizing off-tissue toxicity through spar X affinity and dose modulation | Early development stage; clinical translation potential needs validation | Mu et al[91] | |
| Targeted thorium conjugates | HER2-TTC | Delivers the alpha-particle emitter thorium-227 to HER2+ cells, inducing potent, localized DNA double-strand breaks | Efficacy depends on sustained HER2 expression; limited clinical data (trial No. NCT04147819 ongoing); long-term safety requires evaluation | Pernas et al[35]; Wickstroem et al[92]; Karlsson[93]; Garg et al[94]; Anderson et al[95] |
BsAbs, which simultaneously bind the HER2 and immune cell surface molecules [e.g., cluster of differentiation (CD) 3], can be used to recruit T cells directly to tumor sites, thereby improving antitumor immune responses[32]. Being a novel treatment method, BsAbs have proven to possess significant promise in the treatment of HER2-positive cancer. In contrast to traditional monoclonal antibodies, BsAbs interact with two different epitopes, usually HER2 on tumor cells and critical immune modulators such as CD3 or the co-stimulatory molecule 4-1BB on T cells. This special design creates an “immune bridge” in the TME that redirects cytotoxic T cells to destroy the HER2-positive tumor cells and enhance antitumor immunity[19,35].
Immune checkpoint-modulating agents are found in mechanistically diversified HER2-targeted BsAbs. Cinrebafusp alfa (PRS 343) (4-1BB/HER2) promotes the growth of T cells and cytokines via HER2-dependent 4-1BB clustering, and inhibits tumor growth in preclinical models[78]. Fidasimtamab (IBI315) (PD-1/HER2) simultaneously inhibits PD-1 and HER2 signaling, which has proved effective in HER2-positive GC, through a gasdermin B-mediated pyroptosis, which creates a self-reinforcing immunostimulatory loop[79]. Dual epitope-binding BsAbs such as KN026, which bind two domains of HER2 that overlap the trastuzumab and pertuzumab binding sites. The results of the clinical trial in phase II showed that HER2-high GC patients had a 56% ORR and good safety[80]. Trasintuzumab is a dual HER2-targeting, potent tumor suppressor that is developed using dual variable domain-immunoglobulin technology[81]. Resistance-based BsAbs like CD40 × HER2 activate CD40 signalling to repolarise macrophages to the M1 antitumour phenotype, reversing C-C motif chemokine ligand-mediated trastuzumab resistance[82]. IMM2902 (CD47/HER2), a drug to target trastuzumab-resistant breast cancer, induces macrophage production of CXCL9 and CXCL10 to recruit T and NK cells, and has been shown to be preclinically safe[83]. Phase II consolidation therapy showed that T cell-engaging HER2 bispecific armed T cells (anti-CD3 × anti-HER2 armed T cells) increased 4-month disease stabilization rates and mOS in the HER2-negative metastatic breast cancer[84].
The HER2-positive GC patients have a limited treatment choice and high treatment resistance. BsAbs provide rejuvenated hope, as they target both HER2 and immune cells[32]. BsAbs are better able to overcome resistance in two ways of action compared to monoclonal antibodies[35]. They can be applied to a larger patient population, such as that with low or heterogeneous HER2 expression[38].
The main benefits of HER2-targeted BsAbs over monoclonal antibodies include: (1) Dual capabilities simultaneously inhibiting HER2 signaling and activating immune responses[35]; (2) Enhanced effectiveness against resistant tumors through intensified inhibition of the HER2 pathway[35]; (3) Induction of sustained antitumor immune memory via activation of immune cells[19]; (4) Expanded utility in the HER2-low/heterogeneous populations[38]; and (5) Off-the-shelf use without complex manufacturing[19]. The following features make BsAbs a groundbreaking development in the treatment of HER2-positive GC.
Although promising, there are challenges in optimizing the pharmacokinetics, reducing cytokine release syndrome, and defining the optimal combination strategies[35]. Nevertheless, the potential of HER2-targeted BsAbs is significant, which is supported by its dual mechanism, resistance efficacy and broad applicability which position it as a key break
The radical immunotherapy carried out through CAR-T cell therapy has shown success in the treatment of hematologic malignancies, but faces significant challenges when treating solid tumors, including HER2-positive GC[85]. The prominent molecular target, HER2, of this malignancy provides a rationale behind the use of CAR-T applications. CAR-T cells are genetically modified T cells derived in patients, which are engineered to express CARs that specifically recognize tumor surface antigens, which allows them to be targeted to eliminate cancer cells[85].
CAR design structural development has continued to increase therapeutic effectiveness. There was limited activity of first-generation CARs because of the lack of co-stimulatory signals. Inclusion of co-stimulatory molecules (CD28 and 4-1BB) in second- and third-generation CARs significantly enhanced cytotoxicity, proliferative capacity, and in vivo persistence. Additional modules of cytokine secretion or of signaling pathway regulation can be found in fourth- and fifth-generation CARs, which fully extend antitumor capabilities and versatility[85].
Preclinical data shows promising antitumor action of HER2-targeted CAR-T cells. In vitro models show that the potent killing of HER2-positive GC cells is accompanied by a high secretion of cytokines[86]. Animal model, such as NCG mouse GC xenografts and PDX models, demonstrate successful tumor growth inhibition and metastatic clearance[86]. Major histocompatibility complex-independent recognition of the HER2 antigens triggers target cell death, leading to superior tumor suppression and cytotoxicity as compared to controls in PDX experiments[87]. An initial phase of CAR-T studies with Claudin18.2 and Claudin6 also demonstrate potential in the treatment of gastrointestinal cancers[88,89].
Although there are encouraging preclinical results, there are several challenges in clinical translation of HER2 CAR-T. The physical barriers of fibroblasts, combined with the presence of inhibitory cytokines and the infiltration and functioning of regulatory T cells, significantly hinder the infiltration and functioning of CAR-T cells in solid tumors[90]. The heterogeneity of antigens can result in the development of therapeutic escape, whereas low level target expression in normal tissues can lead to on-target/off-tumor toxicity[90]. Adverse events related to treatment, such as cytokine release syndrome and neurotoxicity, also need to be carefully handled[85].
In order to overcome these shortcomings, scientists are seeking multipronged optimization solutions. ICI, chemotherapy or radiotherapy based combinatoric approaches aim to remodel the TME and enhance CAR-T efficacy[90]. New structural designs of CARs, including next-generation constructs that secrete cytokines or co-stimulatory ligands are aimed at enhancing persistence and antitumor activity[90]. Rapid targeting systems such as acrobatic recombinase cassettes-T cells achieve selective tumor cell killing with minimal off-tissue toxicity by sparX affinity and dose modulation[91].
Overall, HER2-targeted CAR-T therapy is a new treatment option in HER2-positive GC. Although traditional CAR-T has shown strong preclinical efficacy, new platforms, such as acrobatic recombinase cassettes-T cells, have demonstrated improved ability to target and achieve high levels of safety and efficacy. It will be essential to overcome the problems of clinical translation by optimizing the design of CARs, using combination strategies, and gaining experience. As further developments take place, CAR-T therapy has a future as a foundation of multimodal therapy in treating HER2-positive GC, ultimately leading to improved patient survival and quality of life.
HER2-TTC is an innovative development in a radiopharmaceutical treatment. The novel therapeutic agent is a co
Preclinical studies show that HER2-TTC has a strong antitumor effect in cancer models with varying HER2 expression levels, including breast, gastric, lung, bladder and colorectal cancers (IHC scores 1+ to 3+)[92,93]. It is important to note that it is still effective in trastuzumab- and T-DM1-resistant models and tumors with low HER2 levels (IHC 2+/ISH- or IHC 1+)[92]. Synergistic effects are noted in combination strategies: e.g., HER2-TTC combined with the poly-adenosine diphosphate ribose polymerase inhibitor olaparib improves the therapeutic effects in BRCA-mutant models[92]. The special mechanism of the agent also contributes to rational combinations of therapies. First, ICD induced by HER2-TTC which can reverse immunosuppressive TMEs, potentially enhancing antitumor immunity when used with checkpoint inhibitors[35,92]. Second, biomarker-based interventions [e.g., detecting DNA damage response (DDR) pathway mu
In short, HER2-TTC presents a novel paradigm to deal with HER2-targeted therapy resistance, and to treat HER2-low malignancies. The dual nature of a monotherapy and a combination platform with synergy with DDR inhibitors and immunotherapies make it a transformational tool in precision oncology. The future clinical trials and biomarker research will push HER2-TTC towards the next wave of bringing a precise-integrated radiopharmaceutical therapy.
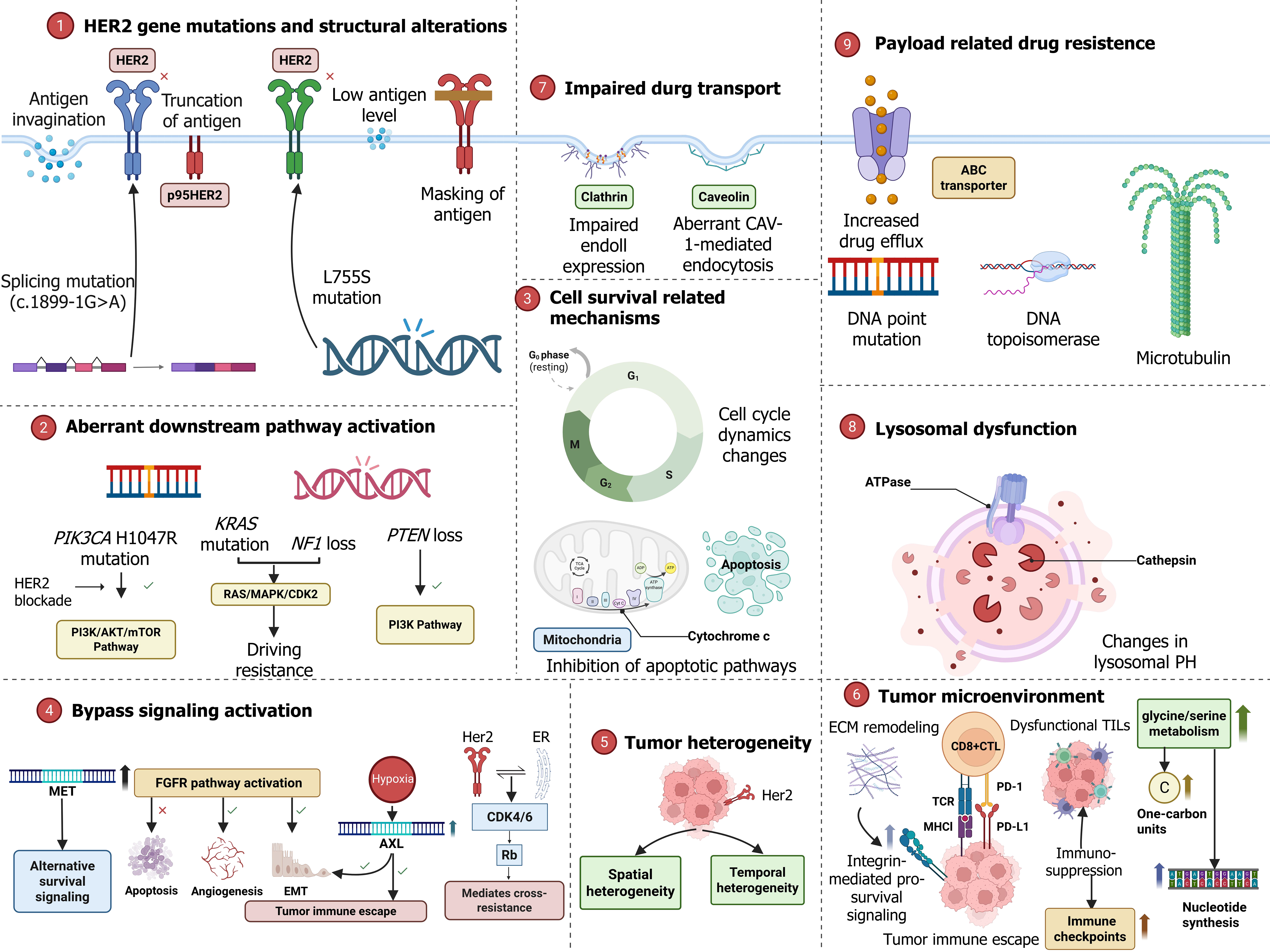
Primary and acquired resistance is an inevitable occurrence in HER2-targeted therapy of patients with HER2-positive GC, which poses a significant clinical challenge. The various mechanisms of resistance are quite heterogeneous and entail numerous molecular and pathway changes (Table 4), which requires a more profound comprehension to design effective counter-mechanisms.
| Resistance mechanism | Key molecular events and evidence | Consequences | Potential overcoming strategies | Ref. |
| HER2 gene mutations and structural alterations | Antibody-mediated drug resistance. L755S mutation: Mediates acquired resistance to TKIs. p95HER2 truncation: Lacks the extracellular domain, evading trastuzumab binding while constitutively activating downstream signaling. Splicing mutation (c.1899-1G>A): Leads to exon skipping, altering the HER2 protein structure | Reduced antigen expression, antigen masking, antigen truncation, target mutations, and antigen internalization. Markedly reduced drug-binding affinity. Sustained activation of downstream oncogenic signaling. Therapeutic escape facilitated by intratumoral heterogeneity | Switch to agents with distinct mechanisms (e.g., ADCs). Implement dual HER2 blockade (e.g., trastuzumab + pertuzumab). Employ NGS to guide therapy selection | Schiff et al[96]; Chen et al[97]; O'keefe et al[98]; Jebbink et al[99]; Jiao et al[100]; Marchiò et al[101]; Sperinde et al[102]; Goh et al[103]; Janiszewska et al[104] |
| Aberrant downstream pathway activation | PIK3CA H1047R mutation: Sustains PI3K/AKT/mTOR signaling despite HER2 blockade. PTEN loss: Leads to constitutive PI3K pathway activation. NF1 loss/KRAS mutation: Activates the RAS/MAPK pathway, driving resistance via the MEK-CDK2 axis | Bypasses upstream HER2 inhibition, maintaining survival and proliferation signals. Alters oncogenic dependency, driving cell cycle progression | Combine PI3K/mTOR inhibitors (e.g., alpelisib). Combine MEK inhibitors (e.g., trametinib). Combine CDK4/6 or explore CDK2 inhibitors | Schiff et al[96]; Janiszewska et al[104]; Smith and Chandarlapaty[105]; Yu et al[106]; Garay et al[107]; Garay et al[108]; Smith et al[109] |
| Cell survival related mechanisms | AKT-mediated phosphorylation: Inhibits pro-apoptotic proteins (e.g., BAD, caspase-9), blocking mitochondrial apoptosis (cytochrome c release). AKT/mTOR signaling: Promotes G1/S transition by regulating cyclin D1/CDKs and downregulating p27Kip1 | Induces an “apoptosis-resistant” phenotype, elevating cell survival threshold. Disrupts cell cycle checkpoints, enabling continuous proliferation (reflected by elevated Ki-67) | Target persistent downstream survival signals (e.g., with AKT inhibitors). Exploit cell cycle vulnerabilities (e.g., with CDK inhibitors) | Smyth et al[3]; Bang et al[8]; Gravalos and Jimeno[9]; Friedlaender et al[22]; Sareyeldin et al[23]; Dumitru et al[24]; Jensen et al[25]; Wang et al[110]; Bassi et al[111] |
| Bypass signaling activation | MET amplification/overexpression: Provides potent alternative survival signaling. FGFR pathway activation: Suppresses apoptosis and induces angiogenesis and EMT. AXL upregulation: Induced by hypoxia, promotes EMT and immune resistance. ER-HER2 crosstalk: Mediates cross-resistance via the CDK4/6-Rb axis | Establishes independent signaling circuits for proliferation and survival. Fuels malignant progression and facilitates immune evasion | Co-administer MET, FGFR, or AXL inhibitors. For HR+ patients, combine CDK4/6 inhibitors with endocrine therapy. Modulate the TME (e.g., alleviate hypoxia) | Pernas and Tolaney[35]; Schiff et al[96]; Szymczyk et al[112]; Wang et al[113]; Recondo et al[114]; Mami-Chouaib et al[115]; Koirala et al[116]; Mahdi et al[117]; Shagisultanova et al[118]; Clark et al[119] |
| Tumor heterogeneity | Spatial heterogeneity: Non-uniform HER2 expression within a tumor, risking sampling error in biopsies. Temporal heterogeneity: Clonal evolution under therapeutic pressure selects for resistant subpopulations | Inherent treatment failure due to untargeted cell populations. Leads to acquired resistance and disease relapse | Perform multi-region biopsy for accurate assessment. Utilize liquid biopsy for dynamic monitoring. Initiate potent combination regimens (e.g., dual HER2 blockade) | Schiff et al[96]; Wang et al[113]; Suenaga et al[120] |
| Tumor microenvironment remodeling | Metabolic reprogramming: Enhanced glycine/serine metabolism supports one-carbon units and nucleotide synthesis. Immunosuppression: Dysfunctional TILs and upregulated immune checkpoints. ECM remodeling: Integrin-mediated pro-survival signaling | Provides biosynthetic precursors and energy for tumor growth. Creates a physical and immunosuppressive barrier against therapy | Target key metabolic enzymes. Combine immune checkpoint inhibitors. Develop novel strategies targeting the ECM | Schiff et al[96]; Abuelreich et al[121] |
HER2 gene mutations and structural alterations: The mutation and structural changes in the HER2 gene are one of the critical mechanisms of resistance to targeted therapy in HER2-positive GC by induction of antibody-mediated drug resistance[96]. This may happen via ways decreased antigen expression, antigen masking, antigen truncation, antigen target mutations, and antigen internalizing[96]. The mutations, including L755S, are acquired in metastatic lesions and mediate resistance to TKIs[96]. These changes cause either impairment of trastuzumab binding or reduction in therapy efficacy, thus promoting acquired resistance.
HER2 mutations can mainly be point mutations, insertions/deletions (indels), and splice site variants[97,98]. The example of the HER2 c.1899-1G>A splice site mutation is the direct consequence of the mutation, which is the skipping of the exon 16 of the HER2 gene, which directly interferes with trastuzumab binding[99,100]. Also, the truncated p95HER2 isoform that lacks the extracellular binding domains but has constitutive tyrosine kinase activity, evades monoclonal antibody targeting[96]. There are three main mechanisms that cause HER2 heterogeneity: Overexpression of HER2 protein, amplification of the gene, and mutation, which together underlie the therapeutic resistance[99].
A truncated form of HER2, p95HER2, which lacks extracellular domains I-IV that are important to trastuzumab binding, continues to activate downstream signaling pathways (e.g., PI3K/AKT) independent of HER2 inhibition[101,102]. This truncated isoform constitutively activates downstream signaling pathways independent of trastuzumab regulation, thereby driving therapeutic resistance[102,103]. The expression of p95HER2 is associated with HER2 gene amplification[103], and can be due to aberrant messenger RNA splicing or alterations in proteolytic processing[101].
HER2 mutations and structural changes impair the efficacy of treatment in a variety of ways: (1) Reduced trastuzumab binding affinity[99]; (2) Sustained activation of alternative signaling pathways such as the PI3K/AKT signal[104]; and (3) Tumor heterogeneity, in which HER2-negative subclones evade therapy a factor strongly predictive of treatment outcomes[104].
Multimodal methods are required to diagnose the HER2 alterations: IHC examines the HER2 protein levels, ISH identifies the amplification of the genes, and next-generation sequencing (NGS) detects mutations[99]. Nevertheless, absence of a standardized definition of HER2 positivity renders clinical practice complex[99]. Transcriptomic profiling can lead to better identification of heterogeneity of HER2 to guide the treatment in a personalized manner[101]. It is important to note that, HER2 mutations facilitate resistance by not only disrupting drug binding but also by activating alternative pathways, remodeling of the TME[104].
Overall, HER2 resistance in HER2-positive GC is centrally associated with mutations in the HER2 gene (e.g., c.1899-1G>A) and structural changes (e.g., p95HER2) that allow tumor cells to avoid therapy due to impaired drug interactions and constant oncogenic signaling. These changes worsen tumor heterogeneity, making it very difficult to diagnose the tumor. Their molecular mechanisms are vital to understand comprehensively to develop new therapeutic interventions and overcome resistance. The focus of future research should be to clarify the clinical impact of the heterogeneity of HER2 and the integration of molecular biomarkers into individualized treatment regimens.
Aberrant activation of downstream signaling pathways: Another important mechanism that contributes to resistance to HER2-targeted therapies in HER2-positive GC[96,105] is the aberrant activation of the PI3K/AKT/mTOR and RAS/MAPK signaling pathways. These down-stream pathways are important in the growth, survival, metabolism and metastasis of tumor cell. They can stimulate tumor cell survival and proliferation in response to targeted therapy inhibition of the HER2 receptor[106].
The PI3K/AKT/mTOR signaling is commonly dysregulated by a variety of molecular events. HER2 positive tumors tend to be co-occurring with mutations in PIK3CA which further increases their resistance to treatment[104]. PIK3CA gene encodes the catalytic subunit of PI3K, and mutations in its catalytic subunit domain (e.g., H1047R) but not its helical domain (e.g., E545K) mutations enhance resistance to the HER2-inhibitor lapatinib in cancer cells expressing the HER2 protein[107]. This resistance is fuelled by continuous AKT signalling with mutations in the kinase domain maintaining PI3K pathway activation despite the presence of HER2 blockade[107,108]. Remarkably, in ”HER2-addicted” tumors with high amplification of the HER2 gene and protein expression, PIK3CA mutations or PTEN loss (a PIP3 phosphatase whose deficiency results in PI3K hyperactivation) can keep the PI3K/AKT/mTOR pathway active, thus avoiding the HER2-targeted inhibition[96,107,108].
RAS/MAPK pathway is the other significant contributor to resistance. The mutations of RAS-MAPK components (e.g., NF1, ERBB2, KRAS) are enriched in patients receiving HER2-targeted treatment[105,109]. NF1-deficient HER2-positive breast cancer models are resistant to HER2 inhibitors (lapatinib, neratinib), but sensitive to MAPK kinase (MEK)/extracellular signal-regulated kinase (ERK) inhibitors, with decreasing sensitivity to AKT inhibitors[105,109]. This resistance is mechanistically associated with MEK-dependent cyclin-dependent kinase (CDK) 2 activation, which promotes progression of the cell cycle and cell survival[109]. Likewise, BRAF, KRAS and ERBB2 mutations induce resistance through the MAPK/CDK2 pathway[105,109]. In tumors with PI3K/AKT/mTOR hyperactivation, cells retain persistent survival signaling; however, when RAS/MAPK is activated, dependency shifts to CDK2-driven proliferation, which cannot be sufficiently countered by monotherapy[105,107].
The combination strategies have potential in curbing resistance. Combining PI3K-dysregulated tumor HER2-targeted therapies with PI3K/AKT/mTOR inhibitors (e.g., alpelisib) is effective[106]. In the case of MEK/ERK inhibitors (e.g., trametinib) or CDK2 inhibitors, which are used in resistance of RAS/MAPK, may be active[105].
Notably, subtypes of PIK3CA mutation (e.g., kinase vs helical domain) are associated with different signaling dynamics and therapeutic responses and thus, must be evaluated based on mutation-specific mechanisms in clinical practice[107]. Also, the expression of HER family ligands (e.g., neuregulin-1) changes the response to treatment, which underlines the necessity of a comprehensive molecular profiling to inform individualized regimens[107].
In short, resistance to HER2-targeted therapies arises not only from HER2-specific mutations (e.g., L755S, p95HER2) but also from PI3K/AKT/mTOR and RAS/MAPK pathway dysregulation. These alterations enable tumor cells to bypass HER2 blockade and sustain oncogenic signaling. Combination therapies using HER2 inhibitors with pathway-specific agents (e.g., PI3K/AKT/mTOR or MEK/ERK inhibitors) represent an effective strategy to combat resistance in HER2-positive GC. Abnormal activation of downstream pathways not only directly drives proliferation but, more importantly, remodels cell survival and cycle regulatory mechanisms, providing tumor cells with powerful intrinsic protection[105,107].
Cell survival related mechanisms: Besides spurring proliferative signaling, HER2 overexpression has a potent effect on tumor cell survival and progression of the cell cycle, by directly activating key downstream pathways, such as PI3K-AKT-mTOR, which directly contribute to resistance to therapy. The essence of this mechanism is the inhibition of apoptotic pathways and the disorder of the dynamics of the cell cycle.
As to the suppression of apoptosis, activated AKT is an active supporter of the mitochondrial apoptotic pathway, inhibiting it by phosphorylating and inhibiting a cascade of pro-apoptotic proteins (e.g., BAD, caspase-9). This inhibits crucial events such as the mitochondrial release of cytochrome c thus vastly lowering the survival threshold of tumor cells and allowing them to endure numerous stresses, including targeted therapy and chemotherapy[3,8,9,22,23]. Bio
With regard to cell cycle behaviour, HER2 signaling directly results in abnormal cell cycle progression in GC cells[24]. Its downstream effectors, including AKT and mTOR, facilitate the G1-S transition by regulating some key cyclins (e.g., cyclin D1) and CDKs[110]. At the same time, the activity of cell cycle inhibitor proteins (e.g., p27 Kip1)[111]) is downregulated by these signaling pathways. It is this loss of cell cycle checkpoints that enable tumor cells to continue proliferating. This biological feature is clinically manifested in HER2-positive GC which usually has an increased index of Ki-67[25], which represents a direct indication of active proliferation of cells.
Subsequently, this inherent resilience mediated by unopposed downstream signals of survival and cycle progression, represents a key pathway through which tumor cells can avoid cytotoxic effects of HER2-targeted therapy, even after success in receptor blockade.
Bypass signaling activation: A key mechanism that contributes to resistance to HER2-targeted therapies in HER2-positive GC is bypass signaling activation[96]. Tumor cells exhibit high plasticity in rewiring signaling networks, allowing them to escape HER2/neu inhibition via alternative pathways. In response to the HER2 signaling blockade, tumor cells activate compensatory mechanisms that maintain tumor survival and growth, thus circumventing the HER2 blockade. Compensatory dimerization of HER receptors further complicates therapy due to functional redundancy of these receptors[96]. Alternatively, activation of receptor tyrosine kinases such as AXL and FGFR, or increased expression of downstream effectors such as SRC and YES1, provides additional proliferative stimuli[96]. For example, tumor cells may overexpress or aberrantly activate MET, FGFR, or AXL to bypass HER2 inhibition[112,113].
Dysregulation of MET pathways, through MET gene amplification, mutation, rearrangement or overexpression, continues to stimulate downstream signaling despite inhibition by HER2, ensuring cell proliferation, survival and migration[114]. MET inhibitors have the ability to reverse resistance caused by MET amplification emphasizing the
In HER2-positive tumors co-expressing ER, unblocked ER signaling provides alternative survival pathways, necessitating dual HER2/ER inhibition[96]. Bidirectional crosstalk between ER and HER2 via the CDK4/6-cyclin D-Rb axis drives resistance to both hormone and HER2-targeted therapies[35,116]. CDK4/6 inhibitors (e.g., palbociclib, ribociclib, abemaciclib) synergize with HER2 inhibitors by blocking Rb phosphorylation and early 2 factor release, inducing G1 cell cycle arrest[116]. Preclinical trials in HER2-positive ovarian and endometrial cancers show increased effectiveness of neratinib (a pan-HER inhibitor) used in combination with abemaciclib, as compared to monotherapy[117]. Indicatively, triple-targeted therapy with tucatinib (a HER2 inhibitor), palbociclib and fulvestrant (an ER degrader) suppress tumor viability, clonogenicity and resistant subclones in HR+/HER2+ cancers and serves to represent a novel approach to dual-resistant patients[118]. Moreover, resistance to CDK4/6 inhibition might be overcome by combining PI3K inhibitors with CDK4/6 inhibitors[119].
Overall, resistance in HER2-positive GC occurs due to overlapping and flexible tumor signaling networks that involve MET, FGFR, AXL and ER pathways. Subsequent therapeutic interventions should focus on multitargeted therapy, including the combination of HER2-based therapy and MET-based therapy, and AXL-based therapy or CDK4/6-based therapy, to defeat resistance and achieve better results in patients.
Tumor heterogeneity: Another decisive factor that has been involved in resistance to HER2-targeted therapies is the tumor heterogeneity. There are high levels of intratumoral heterogeneity in HER2-positive tumors, which are associated with non-homogenous distribution of amplification of the HER2 gene, protein expression, and signaling activity[96]. This heterogeneity leads to the tumor cells being heterogenous, meaning that some cells can survive since they can avoid the therapeutic pressure. Moreover, the network has functional redundancy in the family of receptors in HER and com
Temporal heterogeneity is demonstrated as clonal evolution, which can be observed during time under the influence of therapeutic pressure. The resistance to tumors initially sensitive to the targeted therapy of HER2 may happen via the selective growth of cell subpopulations expressing different signaling pathways[113]. This dynamic change requires the constant modification of treatment plans in order to overcome the evolution of tumors.
Spatial heterogeneity is a genetic and phenotypic differences between cells in various parts of the same tumor. This causes irregular responses to HER2-targeted therapy where some subclones may have a survival advantage due to bypass signaling activation[113]. As an example, in metastatic colorectal cancer, we find polymorphisms in the CCL5 and CCR5 genes which are associated with the location of the primary tumor and significantly affect response of patients to EGFR signaling blockade therapy[120]. The fact that left-sided and right-sided primary tumours have marked differences in their response to treatment further underlines the importance of spatial heterogeneity on treatment outcomes.
Overall, heterogeneity in the tumor is one of the major sources of resistance in the HER2-targeted therapy. It can be spatially reflected in the uneven expression and activity of HER2, which result in therapeutic escape. On the temporal scale it causes clonal evolution, which causes the appearance of resistant subpopulations[113]. This means that often single-agent targeted therapy is not sufficient. It is essential to implement the more powerful and comprehensive strategies of dual HER2 blockade at the beginning of treatment to overcome the heterogeneity and postpone the emer
Remodeling of the TME: Changes in the TME is a central factor in the resistance mediation. One of the adaptive responses of tumor cells to therapeutic pressure is metabolic reprogramming. Research has indicated that cells that have HER2-positive breast cancer display aberrations in the pathways involved with metabolism of non-essential amino acids, especially the active metabolic pathways of glycine and serine. These pathways favour one-carbon metabolism, supplying essential building blocks and energy to support tumor cell growth[121]. At the same time, the capacity of tumor cells to change their metabolism by using aerobic glycolysis, albeit with inefficient energy production, is based on an increase in glutamate consumption which is critical in energy production as well as biosynthesis of nucleotides[121].
The TME also has components that are immunological and that have a significant effect on treatment response. Functional and the density of TILs have a close relationship with therapeutic results[96]. Moreover, components of the extracellular matrix can regulate tumor response and resistance by integrin-mediated signaling[96]. All these microenvironmental elements together form a complex resistance network which allows the tumor cell to survive and progress with therapeutic pressure. Overall, it is through intrinsic metabolic changes, interactions with immune cells, and interactions with the extracellular matrix that tumor cells are able to achieve resistance and survival under the pressure of HER2-targeted therapy.
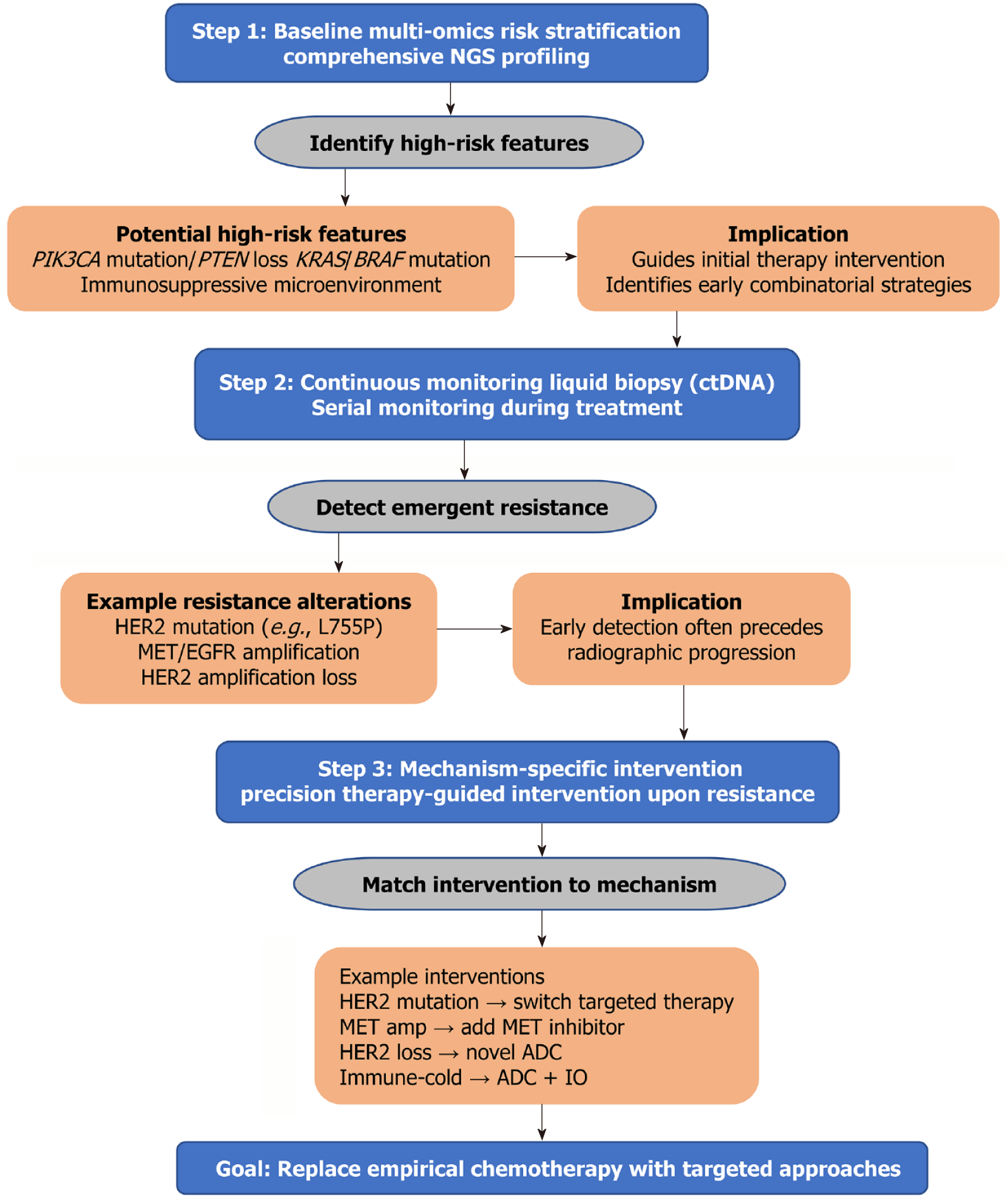
Towards a dynamic, biomarker-driven decision model for overcoming resistance: The resistance in HER2-positive GC is too complex to be treated using the traditional, empirical approaches[94]. This proactive framework that is in line with the modern standards of therapy individualization based on extensive molecular profiling[122], combines continued molecular surveillance with mechanism-specific interventions, with the aim of substituting empirical approaches to chemotherapy at progression with rational, targeted ones.
Initially, risk stratification by multi-omics profiling at the baseline. When diagnosing, extensive molecular characterization, say with NGS, should be carried out to provide a baseline molecular map. This assay goes beyond confirming the HER2 positivity to co-alterations with intrinsic resistance or different therapeutic responses[96]. High-risk molecular features may include concomitant PIK3CA mutations or PTEN loss, suggesting upstream pathway reactivation, KRAS/BRAF mutations indicative of bypass signaling potential, or biomarkers reflecting an immunosuppressive TME. This early stratification guides the severity of first-line therapy, e.g., a more aggressive combination regimen in high-risk molecular settings, and helps to identify early that there are potential combinatorial targets.
This would be followed by constant monitoring through liquid biopsy of early resistance to detect them. Serial analysis of circulating tumor DNA (ctDNA) by liquid biopsy is critically important during active treatment, to identify early emerging resistance clones, and often several months before radiographic progression occurs[94]. The progression of certain resistance-related changes, such as HER2 extracellular domain mutations (e.g., L755S) that confer antibody resistance or MET or FGFR amplification reflecting bypass pathway activation or clonal evolution resulting in loss of HER2 amplification[96]. This surveillance is a liquid-based surveillance which provides a real-time perspective of tumor dynamics in response to therapeutic stress.
Lastly, intervention mechanism-specific intervention on establishment of resistance. Therapeutic choices on standard next-line chemotherapy should be based on underlying mechanism-rather than defaulting back to uniform next-line chemotherapy. This strategy allows a rechallenge strategy in which choice of treatment is determined by the change in resistance detected. As an example, a mutation in a kinase domain typically associated with HER2 can be detected to justify switching to a TKI such as tucatinib or pyrotinib which specifically targets the altered kinase domain[96]. Com
In sum, this dynamic model redefines resistance management as a series of standardized lines of treatment as reactive to various circumstances, and instead, as an emergent and adaptive process. It also highlights the fact that resistance is not a unitary phenomenon but a heterogeneous and dynamic topography[96] and requires a no less flexible and informed responding therapeutic response. This can be achieved by ensuring that there is a wide access to reliable technologies in molecular profiling and liquid biopsy, as well as clinical trials which are specifically designed to validate the use of biomarkers in driving interventions in resistant disease settings. This framework is based on a rational clinical translation of the resistance mechanisms identified above and is critical towards achieving long-lasting precision oncology in HER2-positive GC[94].
Dual HER2 blockade: To overcome resistance resulting due to functional redundancy or heterogeneity of the HER2 receptor, dual HER2 blockade therapy is a powerful investigational approach within the dynamic decision model, especially when baseline profiling or initial longitudinal tracking demonstrates its relevance. This strategy, which involves simultaneous targeting of different epitopes or components within the HER2 signaling axis can be effectively used to suppress tumor growth and to possibly delay the development of resistance[35]. In the framework suggested, patients with a baseline molecular profiling or on-treatment monitoring with particular characteristics (such as HER2 hyper-amplification or an early indication of intratumoral heterogeneity) would be prioritized to receive strategies to use dual blockade therapy. Much higher response rates of up to 62.5% and significantly superior compared to monotherapy are achieved in HER2 hyper-amplified GC xenograft models with dual blockade, which induces complete and sustained tumor regression[123]. Interestingly, it was observed that antitumor activity was not compromised even in models that contained HER2 mutations or KRAS amplification[123], which suggests that the given approach can be considered as a mechanism-specific intervention.
A second example of TKI with ability to overcome resistance is lapatinib, a dual EGFR and HER2 TKI. Molecular context can be used to refine the utility of this strategy. An example is the loss-of-function mutations in the PI3K and MAPK signaling pathways that have been linked to resistance to lapatinib[124]. This environment promotes a combination of mechanism-specific combination approach that is supported by the dynamic decision model. This strategy has been demonstrated in preclinical research as simultaneous suppression of key downstream signaling nodes that have been reactivated[124]. This multi-targeted blockade does not only act at the level of HER2, but also at the level of its critical downstream effectors leading to even greater inhibition of tumor growth.
Molecular justification behind the concept of dual HER2 blockade is strong. This strategy further blocks the activation of downstream pathways and HER2 dimerization by targeting different domains of the signaling axis[122,123]. Nevertheless, the clinical implementation in the future will have to be entrenched in a biomarker-based platform. Future research should aim to define predictive biomarkers (e.g., specific thresholds of HER2 copy numbers and specific dimerization partner expression), to identify patients most likely to benefit and position dual blockade as a targeted option within the dynamic decision model[35,123]. Further studies on new combinations, such as ADCs as an adjunct to more traditional anti-HER2 agents, can also be seen as a logical extension of the concept of dual blockade in the context of precision rechallenge[35].
Sequential therapy and treatment sequencing optimization: The efficient treatment of biological complexity and the subsequent use of the information to guide therapeutic intervention requires sophisticated computational methods. This is the necessary enabling framework that can be supplied by systems pharmacology[125]. It provides a computational basis to simulate which mechanism-specific intervention, such as switching to a TKI, adding a MET inhibitor or initiating an ADC, is most likely to be effective on the identification of specific resistance markers.
It is a model-driven approach that goes beyond the inflexible and linear treatment paradigms and instead encourages adaptive sequencing, where the selection and timing of interventions is continually informed by emerging molecular information. In this regard, the fact that combination strategies involving concurrent combination may be superior to simple sequential administration[126] underlies the limitations of empirical, one-size-fits-all sequencing and supports the need of the model-informed flexibility proposed here. Clinical evidence that supports this paradigm is that therapy focused on specific molecular changes may enhance outcomes following the emergence of resistance[127]. Moreover, complementary strategies, including optimization of the drug delivery systems, can be used to further improve the therapeutic efficacy of the selected agents[128].
Conclusively, it is important to note that systems pharmacology is not an alternative to the dynamic decision model but rather its central driving force to personalization. It makes possible the conversion of continuous monitoring of biomarkers into optimal, patient-specific treatment regimens. The future will be based on the incorporation of these computational strategies and real-life clinical data to further refine the dynamic decision model so that each therapeutic adjustment is proactively guided by the molecular insight rather than reactively by the empirical convention[125].
Even though ADCs are a disruptive therapeutic paradigm in HER2-positive GC, their clinical effectiveness can be undermined by development of resistance. The need to understand the underlying mechanisms of ADC resistance is therefore crucial to the development of effective counterstrategies and enhancement of long-term patient outcomes, as discussed in Table 5.
| Resistance mechanism | Key molecular/cellular processes | Consequences | Potential overcoming strategies | Ref. |
| Alterations in target antigen expression | HER2 protein expression levels. Spatial and temporal tumor heterogeneity | Subpopulations with low/no HER2 evade ADC binding, leading to therapeutic escape. Dynamic downregulation under therapeutic pressure drives acquired resistance | Implement dynamic HER2 status monitoring (e.g., via liquid biopsy). Develop ADCs effective against HER2-low tumors. Explore bispecific antibodies or therapies targeting alternative antigens | Ocaña et al[129] |
| Impaired drug transport | Receptor-mediated endocytosis. ADC intracellular trafficking. Drug efflux pumps (e.g., P-glycoprotein) | Impaired ADC internalization prevents payload delivery. Efflux pumps reduce intracellular payload concentration, diminishing cytotoxicity | Engineer ADCs with improved internalization efficiency. Develop payloads resistant to common efflux pumps. Investigate combination therapies with efflux pump inhibitors | Alrhmoun and Sennikov[32]; Mahalingaiah et al[130]; Chen et al[131] |
| Lysosomal dysfunction | Lysosomal protease activity. Intralysosomal pH. Lysosomal membrane permeability | Inefficient linker cleavage and payload release, even after successful internalization. Altered pH environment inactivates the payload | Design linkers optimized for specific lysosomal proteases. Utilize pH-sensitive linkers that release payload in early endosomes, bypassing lysosomal dependency | Chen et al[131]; Liu-Kreyche et al[132] |
| Payload-specific resistance | DDR pathways. Expression of the payload’s molecular target. Activity of drug-metabolizing enzymes | Enhanced DDR capacity repairs payload-induced DNA damage (e.g., from topoisomerase I inhibitors). Target mutation/downregulation reduces payload binding and efficacy. Enzymatic inactivation of the payload | Develop novel payloads with unique mechanisms of action to bypass pre-existing resistance. Combine ADCs with targeted agents (e.g., PARP inhibitors for DDR). Engineer ADCs with dual, synergistic payloads | Alrhmoun and Sennikov[32]; Chen et al[131]; Ceci et al[133] |
Alterations in target antigen expression: A fundamental aspect of ADC resistance is heterogeneous and dynamic changes in the expression of HER2. Intra-tumoral heterogeneity of HER2 expression permits subpopulations of cells with low or no HER2 to escape ADC binding and resultant killing[129]. Moreover, the dynamic change in the expression of HER2 under treatment pressure may gradually result in the development of resistance in patients who initially respond. It has been shown that the level of changes in HER2 can be affected by the TME, the pressure of clonal selection, and the adaptive responses of tumor cells[32]. The molecular heterogeneity of GC is inherently high and therefore contributes to the difficulty in ADC therapy of this malignancy.
Impaired drug transport: The mechanism of action of ADCs is based on the effective internalization upon antibody binding to the target antigen which releases the cytotoxic payload into the cell. There are a number of ways in which tumor cells can interfere with this process[130]. On the one hand, they can decrease or change the process of the inner cell absorption of the target antigen, making the ADC unable to be efficiently absorbed[131]. Alternatively, the drug efflux pumps (such as the P-glycoprotein) can be increased by the tumor cells, which expel the cytotoxic load of the cell, and this reduces its intracellular concentration[32]. These mechanisms, together, largely reduce the effective intracellular drug concentration, compromising the therapeutic effect of the ADC.
Lysosomal dysfunction: Following internalization, ADCs depend on the lysosomal system for the proteolytic de
Payload-specific resistance mechanisms: The final activity of an ADC depends on the characteristics and mechanism of action of the cytotoxic payload. The payload can be resistant to tumor cells in a variety of ways[131,133]. To begin with, they can improve their DNA repair ability especially against payloads that act by inducing DNA damage, such as topoisomerase I inhibitors[131]. Secondly, tumors can change the expression or structure of the molecular target of the payload, decreasing its binding affinity[131]. Also, the other important mechanism is metabolic inactivation of the payload; tumor cells can enhance the rate of inactivation of the payload by increasing the activity of drug-metabolizing enzymes[32].
Resistance challenges and counterstrategies for novel ADCs: Although the next-generation ADCs (such as T-DXd) are highly potent, long-term administration can ultimately result in resistance, usually with more complex and multifaceted mechanisms[129]. To overcome this, a multi-pronged approach is needed: (1) Optimization of ADC design: This includes designing novel antibodies, optimization of linker technology, and engineering more potent payloads or those with different mechanisms of action[131]; and (2) Innovative clinical methods: These include regular monitoring of HER2 status, rational combinations with immunotherapy or other targeted agents, sequential use of ADCs with different mechanisms after resistance develops, and predictive biomarkers of early intervention[32,131].
To sum up, the problem of resistance to anti-HER2 therapy in HER2-positive GC is a wide-ranging and multi-faceted problem. As shown in Figure 2, it includes both primary resistance to targeted therapies driven by HER2 alterations, persistent PI3K/AKT and RAS/MAPK signaling, bypass pathway activation, and tumor heterogeneity and specific mechanisms for ADCs, including reduced HER2 expression, impaired internalization, lysosomal dysfunction, and payload-specific resistance[96,100,130-132]. To overcome such a complex resistance, especially the entire process of ADC action between “target recognition” and “intracellular killing”, a combination of new and old strategies is necessary. The way forward is in the profound intertwining of the continuous ADC technological innovation with biomarker-directed clinical approaches. Some of the evolving methods are dual HER2 blockade, rational combinations with pathway-specific inhibitors, the development of next-generation ADCs, and sequential therapy guided by dynamic biomarker monitoring[106,124,128,132]. It is this synergy that promises us a systematic way to overcome resistance and to provide more enduring survival benefits to patients.
With the treatment strategies of HER2-positive GC becoming increasingly complex, the role of biomarkers has shifted away as a simple diagnostic tool up to the point of the decision-making process throughout the entire treatment course. This part will cohesively expand on the innovations in the standardization of HER2 testing, the utility of liquid biopsy in dynamic monitoring, and how coordinated multi-omics studies are all coming together to form a next-generation platform to enable precision oncology. These aspects, when combined, are developing a next-generation precision oncology model of HER2-positive GC, as described in Table 6.
| Core domain | Key technologies/strategies | Clinical application and value | Current limitations and challenges | Ref. |
| Standardized HER2 testing | IHC; FISH/CISH; NGS | IHC is the primary screening method. FISH/CISH confirm gene amplification. NGS provides a comprehensive genomic profile (e.g., HER2 amp, mutations, co-alterations like PIK3CA) | Subjectivity in IHC interpretation. Tumor heterogeneity leading to false-negatives. Chromosome 17 polysomy confounding FISH. NGS is not yet a routine primary test | Jebbink et al[99]; McLemore et al[134]; Taylor et al[135]; Klc et al[136]; Ciesielski et al[137]; Vermij et al[138] |
| Liquid biopsy | ctDNA; CTCs; exosomes | Enables dynamic monitoring of resistance and evolution. ctDNA tracks HER2 status and resistance mutations. CTC enumeration and phenotyping reflect tumor burden | Variable sensitivity in early-stage disease. Lack of standardization across platforms. Integration strategy with tissue biopsy is not yet defined | Ho et al[139]; Koessler et al[140]; Rossi and Ignatiadis[141]; Li et al[142]; Massihnia et al[143] |
| Integrated multi-omics analysis | Genomics; transcriptomics; proteomics | Provides a holistic view of tumor biology. Identifies molecular subtypes for targeted therapies (e.g., ADCs). Guides rational combination strategies | Requires bioinformatics expertise. High cost challenges routine use. Needs prospective validation for clinical utility | Yuan et al[144]; Pfeifer and Schimek[145]; Kerr and Yang[146]; Bueno et al[147] |
| Future directions | AI; biomarker-driven individualized framework | AI enhances IHC objectivity and integrates multi-omics for predictive modeling. Establishes refined molecular characterization to guide personalized therapy intensity | AI models require large-scale data for validation. Translating novel biomarkers requires interdisciplinary collaboration and workflow redesign | Jebbink et al[99]; Yuan et al[144]; Bueno et al[147] |
Precision therapy requires accuracy of assessment of the status of HER2. The main HER2 testing methods are currently IHC, FISH, chromogenic ISH (CISH), and NGS with their own specific features and complementary roles[99]. Nevertheless, the challenges still exist, such as interpretive subjectivity, tumor heterogeneity, and technical shortcomings.
The most frequently used first-line screening method is IHC, which determines HER2 status by assessing protein expression levels and offers the advantages of being straightforward to perform and relatively inexpensive. Interpretation of IHC results is subjective, and inter-laboratory consistency should be enhanced[134]. The revised 2018 American Society of Clinical Oncology/College of American Pathologists guidelines narrowed the IHC scoring criteria, allowing more accurate stratification of equivocal (2+) cases, achieving 99.6% concordance with the oncotype DX assay[134]. Research has shown that about 44.7% of all cases of IHC 2+ have ambiguous staining patterns, incomplete and/or weak-to-moderate intense staining in less than 10% of tumor cells. The rate of FISH amplification in such atypical cases is much lower than the rate of FISH amplification in typical IHC 2+ cases (3.3% vs 12.1%)[135]. Hence, these special cases can be given special consideration and may need more stringent interpretative criteria or extra test.
FISH and CISH are the gold standards of identifying gene amplification by determining the copy number of HER2 gene. FISH is highly sensitive and specific yet technically complicated and expensive, and necessitates specialized equipment[99]. CISH can be used to do more intuitive morphological correlation and has high concordance with FISH (98.9%)[136]. A notable challenge is that chromosome 17 polysomy can confound FISH interpretation, as the result is typically based on the HER2/centromeric region of chromosome 17 ratio; an increase in centromeric region of chromosome 17 signals may lead to false-negative results[137]. Thus, the potential impact of chromosome 17 polysomy must be considered when interpreting FISH results.
NGS technology can simultaneously detect HER2 gene amplification, mutations, and alterations in other relevant genes, providing a more comprehensive molecular profile. Studies have shown that the HER2 amplification rate detected by NGS (10.5%) can be lower than that by IHC/CISH, with a 91.6% concordance rate with CISH and a positive predictive value of 60.3%[136]. The strength of NGS lies in its ability to identify co-occurring downstream mutations that may influence response to HER2-targeted therapy, such as those in PIK3CA (33.1%), KRAS (2.5%), and PTEN (1.3%)[136]. Nevertheless, NGS is yet to be implemented as a standard approach to primary HER2 testing, and its usefulness in guiding therapy still needs to be established.
The heterogeneity of HER2 is a crucial determinant of the test results accuracy. A biopsy sample might not be representative of the overall HER2 status of the entire tumor, due to intratumoral heterogeneity[99]. Spatial heterogeneity is defined by the lack of consistency in the HER2 status between tumor sites, and temporal heterogeneity as the possibility of alterations in HER2 status during disease progression[99]. Multi-region sampling and re-biopsy at progression are some of the main optimization strategies that need to be put in place to address these issues.
The optimization of the testing process is important in enhancing the accuracy of the HER2 test. To achieve quality and integrity of tissues, sample collection and processing needs to be standardized[135]. Second, strict quality control system must be laid down, which comprise regular calibration of equipment, and enrollment in external quality assessment programs. Third, a multi-platform verification strategy, e.g., the combined use of IHC with FISH/CISH/NGS can be used to increase reliability[134,136]. Lastly, a case review system especially when it comes to borderline and equivocal findings of numerous pathologists is suggested.
Strategies of HER2 testing might have to be customized in various types of cancers. An example is that in endometrial carcinoma a molecular subtype-directed HER2 test strategy is recommended almost exclusively to the p53-abnormal molecular subtype[138]. In the future, it will be possible to further optimize and innovate HER2 testing methodology. Image analysis with the help of artificial intelligence may help to improve the objectivity and consistency of the interpretation of IHC; the methods of liquid biopsy could help overcome the limitations of tissue biopsy methods by enabling the dynamic monitoring of the status of the HER2; and integrated multi-omics analyses will provide a more comprehensive molecular classification to guide personalized therapy[99]. Such improvements will take another step towards the perfect diagnosis and treatment of HER2-positive tumors, which will eventually lead to improvements in patient outcomes.
To conclude, the rationalization of the testing procedures of HER2 is the basis of precision medicine. The existing paradigm is based on a complementary approach of IHC to initial screening and FISH/CISH to confirmation, but they all have their problems regarding interpretive subjectivity, polysomy of chromosome 17, and heterogeneity of tumors. The use of techniques such as NGS gives more genomic context, but is not yet commonplace. The future directions depend on the promotion of the standardized process of testing, using artificial intelligence to improve the interpretive objectivity, and developing liquid biopsies to monitor dynamically, and, in this way, guide a more effective and personalized treatment.
Liquid biopsy is a non-invasive diagnostic method that uses the circulating tumor-derived components in body fluids to derive tumor information. Liquid biopsy in the management of HER2-positive GC is mainly associated with the detection of the biomarkers, ctDNA, circulating tumor cells (CTCs), and exosomes[139]. These technologies offer a good com
ctDNA is one of the cornerstones of liquid biopsy; ctDNA is fragmented DNA released into the circulation by the tumor cells undergoing apoptosis or necrosis. In HER2-positive GC, ctDNA analysis has the potential to detect the amplification of the HER2 gene, mutations, and other alterations of the HER2 gene in the ctDNA samples[140]. The ctDNA profiling technique, which uses the latest technologies, such as high-throughput sequencing and digital pCR, is critical in providing real-time information on the tumor genomic landscape, which is essential in guiding the HER2-targeted therapy[139].
CTCs are another critical liquid biopsy marker, referring to cancer cells that have detached from the primary or metastatic site and entered the bloodstream. In HER2-positive GC, enumeration of CTCs and the determination of their status as to whether they express HER2 can provide an indication of tumor burden and responsiveness to treatment[141]. Studies have shown that detection of CTC has been of great importance in tracking the progression of the disease as well as predicting the prognosis, particularly in determining the efficacy of HER2-targeted agents[141].
Exosomes are nano-sized vesicles released by cells which contain proteins, nucleic acids and other biomolecules. Exosomes may deliver HER2 protein and associated molecules in yet another non-invasive tumor surveillance method in HER2-positive GC[139]. Exosome analysis does not only indicate the molecular properties of the tumor, it may as well be involved in the regulation of the TME[142], which will provide new insights into the biology of HER2-positive GC.
Liquid biopsy is privileged to have a special edge in the dynamic observing of the disease progression. Liquid biopsies can be conducted more often than conventional tissue biopsy therefore allowing real-time monitoring of tumor progression[143]. Under HER2-positive GC, dynamic variations in the level of ctDNA can reflect variations in tumor burden, whereas changes in the status of the HER2 amplification can herald the changes in the tumor burden or the development of resistance[140]. This ability to constantly monitor enables clinicians to manipulate treatment plans in a timely fashion thus maximizing therapeutic returns.
In determining treatment response, liquid biopsy offers earlier efficacy assessment than conventional radiological imaging. Research indicates that a reduction in ctDNA levels is frequently followed by shrinkage of tumors on imaging, providing the opportunity to make early judgments of the effectiveness of HER2-targeted therapies[143]. Changes in molecular markers of HER2 in ctDNA within patients receiving a treatment based on trastuzumab can reflect treatment response, which can help clinicians decide whether a treatment change is required[140]. Moreover, counts and exosome analysis may be used as additional measures of treatment response, which represent a more comprehensive assessment of efficacy[141].
Liquid biopsy has been of great importance in the prediction of drug resistance. One of the main factors causing failure of therapy in HER2-positive GC is the emergence of resistance. It can be possible to monitor the mutations or changes in amplification of the genes related to the HER2 pathway in ctDNA to detect the development of resistance mechanisms early[139]. As an example, the increase in the level of HER2 gene amplification or downstream signaling gene mutations may serve as a predictor of resistance to trastuzumab[140]. Also, liquid biopsy is able to identify other molecular changes related to resistance, including activation of PI3K/AKT/mTOR pathway, which points to new therapeutic targets to overcome resistance[141].
Although the liquid biopsy technology has a lot of potential, it encounters a number of challenges in the management of the HER2-positive GC. First, the detection sensitivity of ctDNA differs by tumor type and cancer stage, and may be difficult to detect adequate ctDNA in early-stage HER2-positive GC[141]. Second, the liquid biopsy results standardization and interpretation need to be refined further, since the results may vary depending on the detection platform and data analysis method[139]. Moreover, the complementing roles of liquid and tissue biopsies and plans to integrate them require further study to make sure they are optimally applied to clinical practice[140].
Overall, the liquid biopsy technology offers new resources and insights into the management of HER2-positive GC. Liquid biopsy allows dynamic disease monitoring, early evaluation of treatment response, and prediction of resistance through the detection of such markers as ctDNA, CTCs, and exosomes. As technology advances and clinical studies become more in-depth, liquid biopsy is likely to become an even more important tool in the precision medicine of HER2-positive GC, opening the way to more individualized treatment approaches to patients.
This is not because the technical complexity of multi-omics platforms themselves is the key to realizing the real value of biomarker-driven precision medicine, but the ability to solve critical clinical dilemmas that are limiting patient benefit. Beyond a simplistic definition of HER2 status, based alone on protein expression, there is need to combine genomic, transcriptomic, immunophenotypic data to inform therapeutic decision-making in three critical clinical settings.
Predicting response to advanced combination regimens and identifying primary resistance: One of the biggest clinical dilemmas is the inability to reasonably predict which patients have the highest likelihood of benefiting with powerful yet expensive combinations regimens, such as pembrolizumab-trastuzumab-chemotherapy. The use of HER2 IHC or FISH status, as well as PD-L1 CPS alone, is not enough to do so. The combination of multi-omics analysis allows the detection of more informative predictive signatures. For example, combining genomic alterations (e.g., co-mutations in PIK3CA or PTEN), transcriptomic indicators of active immune infiltration (e.g., interferon-γ response and cytotoxic T-cell signatures), and immunophenotypic features reflecting the spatial distribution of CD8+ T cells and PD-L1 expression can stratify patients into subgroups with high or low probabilities of response[144]. This integrative strategy enables one to identify HER2-positive tumors that have intrinsic, or primary, resistance that is driven by a molecular context that is characterized by simultaneous pathway activation and immunosuppression. This will allow saving patients who will not benefit from the ineffective intensive therapy and redirect them to other strategies (e.g., ADCs).
Defining “molecular functional subtypes” to explain heterogeneous outcomes: The extreme inconsistency in the response to treatment of patients categorized as being HER2-positive according to standard criteria underscores the importance of more sophisticated biological taxonomies. Multi-omics clustering can identify such “molecular functional subtypes” within the context of HER2-positive GC. These subtypes may include: (1) Immune-activated, characterized by numerous downstream oncogenic signatures and likely to optimally respond to an immunotherapy-based combination; (2) RAS/PI3K pathway-dominant, driven by downstream oncogenic signatures and likely to derive optimal benefit of immunotherapy-based combinations; and (3) Metabolic or mesenchymal, associated with alternative survival programs and adaptive resistance mechanisms[145]. This kind of functional stratification is what accounts for the fact that two tumors with IHC 3+ status can undergo significantly different clinical courses and is what explains the fact that two tumors with IHC 3+ status can have significantly different clinical courses.
Guiding therapeutic escalation and de-escalation: The application of precision medicine needs to be carefully balanced in terms of both therapeutic efficacy and treatment-related toxicity. In this regard, rational intensification, escalation, simplification, de-escalation of treatment are crucial in this context and cannot be achieved without multi-omics tools. Patients with high-risk biological factors (e.g., complex genomic changes or low immune response) might be good candidates to escalate past the usual first-line therapy, possibly by early incorporation of an ADC or enrolment into clinical trials assessing novel combination regimens. On the other hand, a minority of patients with exceptionally good tumor biology, including a high level of HER2 amplification, strong immune signatures, and no evidence of changes in the bypass pathway may become an ideal candidate in future de-escalation strategies, including chemotherapy-free regimens combining dual HER2 blockade with ICI[146,147]. This biomarker-based framework is aimed at maximizing the therapeutic index by matching the intensity of treatment with inherent biological aggressiveness and vulnerabilities of each tumor.
To conclude, the clinical use of multi-omics analysis is shifting towards a prospective system of decision support, rather than a retrospective research tool. Its systematic incorporation in clinical trials to stratify patients, along with the construction of validated and clinically-available biomarker panels, is one of the next critical steps. Integration of multi-omics will play a key role in the development of truly personalized management of HER2-positive GC.
The paper is a systematic review of the state of HER2-positive GC, including its molecular mechanism, traditional therapeutic options, and innovative developments. Although the introduction of targeted therapy by trastuzumab was welcomed, limited efficacy and emergence of resistance have fueled a paradigm shift. New directions such as ADCs (e.g., T-DXd) and combination strategies that involve the integration of targeted therapy, immunotherapy and chemotherapy now mark a new era. We suggest a precision medicine road map to HER2-positive GC (Figure 3), which combines dia
The authors would like to acknowledge all the authors whose work has been reviewed in the preparation of the manuscript.
| 1. | Peng Z, Zhang X, Liang H, Zheng Z, Wang Z, Liu H, Hu J, Sun Y, Zhang Y, Yan H, Tong L, Xu J, Ji J, Shen L. Atezolizumab and Trastuzumab Plus Chemotherapy for ERBB2-Positive Locally Advanced Resectable Gastric Cancer: A Randomized Clinical Trial. JAMA Oncol. 2025;11:619-624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 2. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21479] [Cited by in RCA: 20392] [Article Influence: 1853.8] [Reference Citation Analysis (3)] |
| 3. | Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4042] [Cited by in RCA: 3475] [Article Influence: 579.2] [Reference Citation Analysis (13)] |
| 4. | Lordick F, Carneiro F, Cascinu S, Fleitas T, Haustermans K, Piessen G, Vogel A, Smyth EC; ESMO Guidelines Committee. Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:1005-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1159] [Cited by in RCA: 1021] [Article Influence: 255.3] [Reference Citation Analysis (13)] |
| 5. | Chen ZD, Zhang PF, Xi HQ, Wei B, Chen L, Tang Y. Recent Advances in the Diagnosis, Staging, Treatment, and Prognosis of Advanced Gastric Cancer: A Literature Review. Front Med (Lausanne). 2021;8:744839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (4)] |
| 6. | Rau B, Brandl A, Piso P, Pelz J, Busch P, Demtröder C, Schüle S, Schlitt HJ, Roitman M, Tepel J, Sulkowski U, Uzunoglu F, Hünerbein M, Hörbelt R, Ströhlein M, Beckert S, Königsrainer I, Königsrainer A; Peritoneum Surface Oncology Group and members of the StuDoQ|Peritoneum Registry of the German Society for General and Visceral Surgery (DGAV). Peritoneal metastasis in gastric cancer: results from the German database. Gastric Cancer. 2020;23:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 7. | Yang T, Xu R, You J, Li F, Yan B, Cheng JN. Prognostic and clinical significance of HER-2 low expression in early-stage gastric cancer. BMC Cancer. 2022;22:1168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T, Aprile G, Kulikov E, Hill J, Lehle M, Rüschoff J, Kang YK; ToGA Trial Investigators. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5933] [Cited by in RCA: 5470] [Article Influence: 341.9] [Reference Citation Analysis (5)] |
| 9. | Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 936] [Cited by in RCA: 886] [Article Influence: 49.2] [Reference Citation Analysis (4)] |
| 10. | Abrahao-Machado LF, Scapulatempo-Neto C. HER2 testing in gastric cancer: An update. World J Gastroenterol. 2016;22:4619-4625. [PubMed] [DOI] [Full Text] |
| 11. | Lote H, Chau I. Emerging HER2-directed therapeutic agents for gastric cancer in early phase clinical trials. Expert Opin Investig Drugs. 2022;31:59-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 12. | Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, Cunningham D, Rougier P, Komatsu Y, Ajani J, Emig M, Carlesi R, Ferry D, Chandrawansa K, Schwartz JD, Ohtsu A; RAINBOW Study Group. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15:1224-1235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1995] [Cited by in RCA: 1848] [Article Influence: 154.0] [Reference Citation Analysis (3)] |
| 13. | Xu RH, Zhang Y, Pan H, Feng J, Zhang T, Liu T, Qin Y, Qin S, Yin X, Liu B, Ba Y, Yang N, Voon PJ, Tanasanvimon S, Zhou C, Zhang WL, Shen L. Efficacy and safety of weekly paclitaxel with or without ramucirumab as second-line therapy for the treatment of advanced gastric or gastroesophageal junction adenocarcinoma (RAINBOW-Asia): a randomised, multicentre, double-blind, phase 3 trial. Lancet Gastroenterol Hepatol. 2021;6:1015-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (2)] |
| 14. | Makiyama A, Sukawa Y, Kashiwada T, Kawada J, Hosokawa A, Horie Y, Tsuji A, Moriwaki T, Tanioka H, Shinozaki K, Uchino K, Yasui H, Tsukuda H, Nishikawa K, Ishida H, Yamanaka T, Yamazaki K, Hironaka S, Esaki T, Boku N, Hyodo I, Muro K. Randomized, Phase II Study of Trastuzumab Beyond Progression in Patients With HER2-Positive Advanced Gastric or Gastroesophageal Junction Cancer: WJOG7112G (T-ACT Study). J Clin Oncol. 2020;38:1919-1927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 156] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 15. | Kim TH, Do Cho H, Choi YW, Lee HW, Kang SY, Jeong GS, Choi JH, Ahn MS, Sheen SS. Trastuzumab-based palliative chemotherapy for HER2-positive gastric cancer: a single-center real-world data. BMC Cancer. 2021;21:325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Al-Batran SE, Moorahrend E, Maintz C, Goetze TO, Hempel D, Thuss-Patience P, Gaillard VE, Hegewisch-Becker S. Clinical Practice Observation of Trastuzumab in Patients with Human Epidermal Growth Receptor 2-Positive Metastatic Adenocarcinoma of the Stomach or Gastroesophageal Junction. Oncologist. 2020;25:e1181-e1187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 17. | Ding Y, Yuan Y, Weng S. KEYNOTE-811: Potential to revise first-line therapy for metastatic human epidermal growth factor receptor 2-positive gastric cancer. MedComm (2020). 2024;5:e545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 18. | Meric-Bernstam F, Anoka C, Dobrowolska A, Chaudhry A, Rowbottom J, Gustavson M, Puvvada S. 1869TiP A phase II, multicenter, open-label study evaluating trastuzumab deruxtecan (T-DXd) for the treatment of select HER2-expressing solid tumors (DESTINY-PanTumor02). Ann Oncol. 2021;32:S1253-S1254. [RCA] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Weidanz J. Targeting cancer with bispecific antibodies. Science. 2021;371:996-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Zhang J, Liu R, Gao S, Li W, Chen Y, Meng Y, Liu C, Jin W, Wu J, Wang Y, Hao Y, Yi S, Qing Y, Ge J, Hu X. Phase I study of A166, an antibody‒drug conjugate in advanced HER2-expressing solid tumours. NPJ Breast Cancer. 2023;9:28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 47] [Reference Citation Analysis (0)] |
| 21. | Shin SH, Park YH, Park SS, Ju EJ, Park J, Ko EJ, Bae DJ, Kim SY, Chung CW, Song HY, Jang SJ, Jeong SY, Song SY, Choi EK. An Elaborate New Linker System Significantly Enhances the Efficacy of an HER2-Antibody-Drug Conjugate against Refractory HER2-Positive Cancers. Adv Sci (Weinh). 2021;8:e2102414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Friedlaender A, Subbiah V, Russo A, Banna GL, Malapelle U, Rolfo C, Addeo A. EGFR and HER2 exon 20 insertions in solid tumours: from biology to treatment. Nat Rev Clin Oncol. 2022;19:51-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 145] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 23. | Sareyeldin RM, Gupta I, Al-Hashimi I, Al-Thawadi HA, Al Farsi HF, Vranic S, Al Moustafa AE. Gene Expression and miRNAs Profiling: Function and Regulation in Human Epidermal Growth Factor Receptor 2 (HER2)-Positive Breast Cancer. Cancers (Basel). 2019;11:646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 24. | Dumitru M, Mihăilă M, Hotnog C, Panait M, Vasilescu M, Hortopan M, Braşoveanu L. 80P HER2 expression in primary bladder tumors and paired regional lymph nodes: Relationship to cell cycle profile. Ann Oncol. 2021;32:S1369-S1370. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Jensen SG, Thomas PE, Christensen IJ, Balslev E, Hansen A, Høgdall E. Evaluation of analytical accuracy of HER2 status in patients with breast cancer: Comparison of HER2 GPA with HER2 IHC and HER2 FISH. APMIS. 2020;128:573-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Kashiwada T, Saeki H, Uenosono Y, Makiyama A, Iwatsuki M, Narita Y, Satake H, Matsuda Y, Sonoda H, Oki E, Maehara Y. Multicenter observational study on re-evaluation of HER2 status in patients with HER2-positive advanced or recurrent gastric cancer refractory to trastuzumab. J Clin Oncol. 2018;36:4038. [DOI] [Full Text] |
| 27. | Wada R, Hirabayashi K, Ohike N, Morii E. New guidelines for HER2 pathological diagnostics in gastric cancer. Pathol Int. 2016;66:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 28. | Phan DAT, Nguyen VT, Hua TNH, Ngo QD, Doan TPT, Nguyen ST, Thai AT, Nguyen VT. HER2 Status and Its Heterogeneity in Gastric Carcinoma of Vietnamese Patient. J Pathol Transl Med. 2017;51:396-402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Valtorta E, Martino C, Sartore-Bianchi A, Penaullt-Llorca F, Viale G, Risio M, Rugge M, Grigioni W, Bencardino K, Lonardi S, Zagonel V, Leone F, Noe J, Ciardiello F, Pinto C, Labianca R, Mosconi S, Graiff C, Aprile G, Frau B, Garufi C, Loupakis F, Racca P, Tonini G, Lauricella C, Veronese S, Truini M, Siena S, Marsoni S, Gambacorta M. Assessment of a HER2 scoring system for colorectal cancer: results from a validation study. Mod Pathol. 2015;28:1481-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 241] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 30. | Subasinghe D, Acott N, Kumarasinghe MP. A survival guide to HER2 testing in gastric/gastroesophageal junction carcinoma. Gastrointest Endosc. 2019;90:44-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Boyraz B, Ly A. Discerning subsets of breast cancer with very low and absent HER2 protein expression. Hum Pathol. 2022;127:50-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Alrhmoun S, Sennikov S. The Role of Tumor-Associated Antigen HER2/neu in Tumor Development and the Different Approaches for Using It in Treatment: Many Choices and Future Directions. Cancers (Basel). 2022;14:6173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 33. | Goutsouliak K, Veeraraghavan J, Sethunath V, De Angelis C, Osborne CK, Rimawi MF, Schiff R. Towards personalized treatment for early stage HER2-positive breast cancer. Nat Rev Clin Oncol. 2020;17:233-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 208] [Article Influence: 34.7] [Reference Citation Analysis (1)] |
| 34. | Esposito A, Viale G, Criscitiello C, Curigliano G. A clinical perspective on escalating or de-escalating adjuvant therapy in HER2+ breast cancer. Expert Rev Clin Pharmacol. 2019;12:9-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 35. | Pernas S, Tolaney SM. HER2-positive breast cancer: new therapeutic frontiers and overcoming resistance. Ther Adv Med Oncol. 2019;11:1758835919833519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 261] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 36. | Itagaki T, Kajitani K, Ohara M. MO58-6 Evaluation of cardiotoxicity by anti-HER2 therapy. Ann Oncol. 2023;34:S1433-S1434. [DOI] [Full Text] |
| 37. | Wasselin J, Vivier-Chicoteau J, Pozet A, Toullec C, Racine Doat S, Smolenschi C, Turpin A, De Rauglaudre B, Sefrioui D, Bouche O, Zaanan A, Kroemer M, Pellat A, Bignon Bretagne A, Vitellius C, Lavole J, Goujon G, Touchefeu Y, Tougeron D, Hautefeuille V. 1454P PARAFIRIGAST: Paclitaxel/ramucirumab versus FOLFIRI as second line chemotherapy of metastatic gastric cancer: An AGEO multicenter comparative study with a propensity score analysis. Ann Oncol. 2020;31:S912-S913. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 38. | Chow LWC, Lie EF, Toi M. Advances in EGFR/HER2-directed clinical research on breast cancer. Adv Cancer Res. 2020;147:375-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 39. | Wang J, Xu B. Targeted therapeutic options and future perspectives for HER2-positive breast cancer. Signal Transduct Target Ther. 2019;4:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 268] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 40. | Swain SM, Shastry M, Hamilton E. Targeting HER2-positive breast cancer: advances and future directions. Nat Rev Drug Discov. 2023;22:101-126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 938] [Cited by in RCA: 773] [Article Influence: 257.7] [Reference Citation Analysis (4)] |
| 41. | Ferrando-Díez A, Felip E, Pous A, Bergamino Sirven M, Margelí M. Targeted Therapeutic Options and Future Perspectives for HER2-Positive Breast Cancer. Cancers (Basel). 2022;14:3305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 42. | Seliger B. Combinatorial Approaches With Checkpoint Inhibitors to Enhance Anti-tumor Immunity. Front Immunol. 2019;10:999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 43. | Kitano S. SY14-2 Strategies for cancer immunotherapy based on breast cancer microenvironment. Ann Oncol. 2022;33:S438. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 44. | Hurwitz J, Haggstrom LR, Lim E. Antibody-Drug Conjugates: Ushering in a New Era of Cancer Therapy. Pharmaceutics. 2023;15:2017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 45. | Pegram MD, Miles D, Tsui CK, Zong Y. HER2-Overexpressing/Amplified Breast Cancer as a Testing Ground for Antibody-Drug Conjugate Drug Development in Solid Tumors. Clin Cancer Res. 2020;26:775-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 46. | Shitara K, Shen L, Lee J, Hoff PM, Smyth EC, Barrios D, Kobayashi K, Okuda Y, Kamio T, Janjigian YY. An open-label, randomized, multicenter, phase 3 study of trastuzumab deruxtecan (T-DXd) + chemotherapy (chemo) ± pembrolizumab (pembro) versus chemo + trastuzumab ± pembro in first-line metastatic HER2+ gastric or gastroesophageal junction (GEJ) cancer: DESTINY-Gastric05. J Clin Oncol. 2025;44:TPS468. [DOI] [Full Text] |
| 47. | Tabernero J, Shen L, Elimova E, Ku G, Liu T, Shitara K, Lin X, Boyken L, Li H, Grim J, Ajani J. HERIZON-GEA-01: Zanidatamab + chemo ± tislelizumab for 1L treatment of HER2-positive gastroesophageal adenocarcinoma. Future Oncol. 2022;18:3255-3266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 48. | Wang D, Lin J, Yang X, Long J, Bai Y, Yang X, Mao Y, Sang X, Seery S, Zhao H. Combination regimens with PD-1/PD-L1 immune checkpoint inhibitors for gastrointestinal malignancies. J Hematol Oncol. 2019;12:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 49. | Cheng AL, Hsu C, Chan SL, Choo SP, Kudo M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J Hepatol. 2020;72:307-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 415] [Article Influence: 69.2] [Reference Citation Analysis (4)] |
| 50. | Liu Y, Li C, Lu Y, Liu C, Yang W. Tumor microenvironment-mediated immune tolerance in development and treatment of gastric cancer. Front Immunol. 2022;13:1016817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 129] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 51. | Yamashita K, Iwatsuki M, Yasuda-Yoshihara N, Morinaga T, Nakao Y, Harada K, Eto K, Kurashige J, Hiyoshi Y, Ishimoto T, Nagai Y, Iwagami S, Baba Y, Miyamoto Y, Yoshida N, Ajani JA, Baba H. Trastuzumab upregulates programmed death ligand-1 expression through interaction with NK cells in gastric cancer. Br J Cancer. 2021;124:595-603. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 52. | Liefaard MC, van der Voort A, van Seijen M, Thijssen B, Sanders J, Vonk S, Mittempergher L, Bhaskaran R, de Munck L, van Leeuwen-Stok AE, Salgado R, Horlings HM, Lips EH, Sonke GS. Tumor-infiltrating lymphocytes in HER2-positive breast cancer treated with neoadjuvant chemotherapy and dual HER2-blockade. NPJ Breast Cancer. 2024;10:29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 53. | Lee SC, Shimasaki N, Lim JSJ, Wong A, Yadav K, Yong WP, Tan LK, Koh LP, Poon MLM, Tan SH, Ow SGW, Bharwani L, Yap YS, Foo MZQ, Coustan-Smith E, Sundar R, Tan HL, Chong WQ, Kumarakulasinghe NB, Lieow JLM, Koe PJX, Goh BC, Campana D. Phase I Trial of Expanded, Activated Autologous NK-cell Infusions with Trastuzumab in Patients with HER2-positive Cancers. Clin Cancer Res. 2020;26:4494-4502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 54. | Mandó P, Rivero SG, Rizzo MM, Pinkasz M, Levy EM. Targeting ADCC: A different approach to HER2 breast cancer in the immunotherapy era. Breast. 2021;60:15-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 55. | Li L, Li Y, Yang CH, Radford DC, Wang J, Janát-Amsbury M, Kopeček J, Yang J. Inhibition of Immunosuppressive Tumors by Polymer-Assisted Inductions of Immunogenic Cell Death and Multivalent PD-L1 Crosslinking. Adv Funct Mater. 2020;30:1908961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 56. | Zhu S, Zhang T, Zheng L, Liu H, Song W, Liu D, Li Z, Pan CX. Combination strategies to maximize the benefits of cancer immunotherapy. J Hematol Oncol. 2021;14:156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 468] [Article Influence: 93.6] [Reference Citation Analysis (4)] |
| 57. | Yu J, He X, Wang Z, Wang Y, Liu S, Xie Z, Li X, Huang Y. Correction: Combining PD-L1 inhibitors with immunogenic cell death triggered by chemo-photothermal therapy via a thermosensitive liposome system to stimulate tumor-specific immunological response. Nanoscale. 2021;13:13907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 58. | Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K. Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer. 2022;21:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1172] [Cited by in RCA: 1048] [Article Influence: 262.0] [Reference Citation Analysis (4)] |
| 59. | Oaknin A, Lee JY, Makker V, Oh DY, Banerjee S, González-Martín A, Jung KH, Ługowska I, Manso L, Manzano A, Melichar B, Siena S, Stroyakovskiy D, Fielding A, Puvvada S, Smith A, Meric-Bernstam F. Summary of Research: Efficacy of Trastuzumab Deruxtecan in HER2-Expressing Solid Tumors by Enrollment HER2 IHC Status: Post Hoc Analysis of DESTINY-PanTumor02. Adv Ther. 2025;42:2015-2018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 60. | Aoki M, Iwasa S, Boku N. Trastuzumab deruxtecan for the treatment of HER2-positive advanced gastric cancer: a clinical perspective. Gastric Cancer. 2021;24:567-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 61. | Chen Z, Yuan J, Xu Y, Zhang C, Li Z, Gong J, Li Y, Shen L, Gao J. From AVATAR Mice to Patients: RC48-ADC Exerted Promising Efficacy in Advanced Gastric Cancer With HER2 Expression. Front Pharmacol. 2021;12:757994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 62. | Janjigian YY, Oh D, Rha SY, Lee KW, Steeghs N, Chao Y, Di Bartolomeo M, Díez Garcia M, Haj Mohammad N, Stein A, Mcadoo W, Winter M, Croydon L, Lee J. Dose-escalation and dose-expansion study of trastuzumab deruxtecan (T-DXd) monotherapy and combinations in patients (pts) with advanced/metastatic HER2+ gastric cancer (GC)/gastroesophageal junction adenocarcinoma (GEJA): DESTINY-Gastric03. J Clin Oncol. 2022;40:295. [DOI] [Full Text] |
| 63. | Janjigian YY, Van Laarhoven HW, Rha SY, Kozlov V, Oh D, Gravina A, Rapatoni L, Shoji H, Hofheinz RD, Chen L, Ford H, Chenard-Poirier M, de Giorgio-Miller V, Chang Y, Anadu O, Lee J. Updated results from the trastuzumab deruxtecan (T-DXd) 5.4 mg/kg triplet combination of DESTINY-Gastric03 (DG-03): First-line (1L) T-DXd with fluoropyrimidine (FP) and pembrolizumab in advanced/metastatic HER2-positive (HER2+) esophageal adenocarcinoma, gastric cancer (GC), or gastroesophageal junction adenocarcinoma (GEJA). J Clin Oncol. 2025;43:448. [DOI] [Full Text] |
| 64. | Shitara K, Gumus M, Pietrantonio F, Lonardi S, De La Fouchardiere C, Coutzac C, Dekervel J, Hochhauser D, Shen L, Mansoor W, Liu B, Fornaro L, Ryu M, Lee J, Souza F, Jukofsky L, Zhao Y, Kamio T, Zaanan A, Van Cutsem E. Trastuzumab deruxtecan (T-DXd) vs ramucirumab (RAM) + paclitaxel (PTX) in second-line treatment of patients (pts) with human epidermal growth factor receptor 2-positive (HER2+) unresectable/metastatic gastric cancer (GC) or gastroesophageal junction adenocarcinoma (GEJA): Primary analysis of the randomized, phase 3 DESTINY-Gastric04 study. J Clin Oncol. 2025;43:LBA4002. [DOI] [Full Text] |
| 65. | Shitara K, Van Cutsem E, Gümüş M, Lonardi S, de la Fouchardière C, Coutzac C, Dekervel J, Hochhauser D, Shen L, Mansoor W, Liu B, Fornaro L, Ryu MH, Lee J, Faustino C, Metges JP, Tabernero J, Franke F, Janjigian YY, Souza F, Jukofsky L, Zhao Y, Kamio T, Zaanan A, Pietrantonio F; DESTINY-Gastric04 Trial Investigators. Trastuzumab Deruxtecan or Ramucirumab plus Paclitaxel in Gastric Cancer. N Engl J Med. 2025;393:336-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 57] [Article Influence: 57.0] [Reference Citation Analysis (0)] |
| 66. | Yamaguchi K, Bang Y, Iwasa S, Sugimoto N, Ryu M, Sakai D, Chung H, Kawakami H, Yabusaki H, Lee J, Saito K, Kawaguchi Y, Kamio T, Kojima A, Sugihara M, Shitara K. 1422MO Trastuzumab deruxtecan (T-DXd; DS-8201) in patients with HER2-low, advanced gastric or gastroesophageal junction (GEJ) adenocarcinoma: Results of the exploratory cohorts in the phase II, multicenter, open-label DESTINY-Gastric01 study. Ann Oncol. 2020;31:S899-S900. [RCA] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 67. | Peng Z, Chen P, Lu J, Wan Y, Zheng Y, Ye F, Yang J, Liu Y, Pan H, Sun M, Fan Q, Yuan Y, Chen K, Sun Z, Tian H, Xia Y, Shen L. Trastuzumab deruxtecan in patients from China with previously treated human epidermal growth factor receptor 2-positive locally advanced/metastatic gastric or gastroesophageal junction adenocarcinoma (DESTINY-Gastric06): results from a single-arm, multicenter, phase 2 trial. EClinicalMedicine. 2025;87:103404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 68. | Díaz-Rodríguez E, Gandullo-Sánchez L, Ocaña A, Pandiella A. Novel ADCs and Strategies to Overcome Resistance to Anti-HER2 ADCs. Cancers (Basel). 2021;14:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 69. | Xu Y, Wang Y, Gong J, Zhang X, Peng Z, Sheng X, Mao C, Fan Q, Bai Y, Ba Y, Jiang D, Yang F, Qi C, Li J, Wang X, Zhou J, Lu M, Cao Y, Yuan J, Liu D, Wang Z, Fang J, Shen L. Phase I study of the recombinant humanized anti-HER2 monoclonal antibody-MMAE conjugate RC48-ADC in patients with HER2-positive advanced solid tumors. Gastric Cancer. 2021;24:913-925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 122] [Article Influence: 24.4] [Reference Citation Analysis (1)] |
| 70. | Peng Z, Liu T, Wei J, Wang A, He Y, Yang L, Zhang X, Fan N, Luo S, Li Z, Gu K, Lu J, Xu J, Fan Q, Xu R, Zhang L, Li E, Sun Y, Yu G, Bai C, Liu Y, Zeng J, Ying J, Liang X, Xu N, Gao C, Shu Y, Ma D, Dai G, Li S, Deng T, Cui Y, Fang J, Ba Y, Shen L. Efficacy and safety of a novel anti-HER2 therapeutic antibody RC48 in patients with HER2-overexpressing, locally advanced or metastatic gastric or gastroesophageal junction cancer: a single-arm phase II study. Cancer Commun (Lond). 2021;41:1173-1182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 160] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 71. | Barfield RM, Kim YC, Chuprakov S, Zhang F, Bauzon M, Ogunkoya AO, Yeo D, Hickle C, Pegram MD, Rabuka D, Drake PM. A Novel HER2-targeted Antibody-drug Conjugate Offers the Possibility of Clinical Dosing at Trastuzumab-equivalent Exposure Levels. Mol Cancer Ther. 2020;19:1866-1874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 72. | You J, Zhang J, Wang J, Jin M. Cysteine-Based Coupling: Challenges and Solutions. Bioconjug Chem. 2021;32:1525-1534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 73. | Hu X, Zhang J, Liu R, Gao S, Qing Y, Yi S, Yuan J, Chen H, Fan B, Zheng H, Wang J. Phase I study of A166 in patients with HER2-expressing locally advanced or metastatic solid tumors. J Clin Oncol. 2021;39:1024. [DOI] [Full Text] |
| 74. | Liu Y, Lian W, Zhao X, Qi W, Xu J, Xiao L, Qing Y, Xue T, Wang J. A first in-human study of A166 in patients with locally advanced/metastatic solid tumors which are HER2-positive or HER2-amplified who did not respond or stopped responding to approved therapies. J Clin Oncol. 2020;38:1049. [DOI] [Full Text] |
| 75. | Hu X, Zhang J, Liu R, Gao S, Wu J, Wang Y, Hao Y, Ge J, Qing Y, Yi S, Yang Q, Rao H, Yuan J. Updated results and biomarker analyses from the phase I trial of A166 in patients with HER2-expressing locally advanced or metastatic solid tumors. J Clin Oncol. 2022;40:1037. [DOI] [Full Text] |
| 76. | Zhang Y, Fan W, Su F, Zhang X, Du Y, Li W, Gao Y, Hu W, Zhao J. Discussion on the mechanism of HER2 resistance in esophagogastric junction and gastric cancer in the era of immunotherapy. Hum Vaccin Immunother. 2025;21:2459458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (4)] |
| 77. | Shen L, Peng Z, Li C, Lu L, Wang X, Zhang J, Zhang Y, Deng T, Sun M, Cai D, Jin Y, Zong H, Ye F, Xu H, Zhang M, Liu J, Feng D, Fang J. Disitamab vedotin (DV) plus toripalimab (Tor) and chemotherapy (C)/trastuzumab (Tra) as first-line (1L) treatment of patients (pts) with HER2-expressing locally advanced or metastatic (la/m) gastric cancer. J Clin Oncol. 2025;43. [DOI] [Full Text] |
| 78. | Hinner MJ, Aiba RSB, Jaquin TJ, Berger S, Dürr MC, Schlosser C, Allersdorfer A, Wiedenmann A, Matschiner G, Schüler J, Moebius U, Rothe C, Matis L, Olwill SA. Tumor-Localized Costimulatory T-Cell Engagement by the 4-1BB/HER2 Bispecific Antibody-Anticalin Fusion PRS-343. Clin Cancer Res. 2019;25:5878-5889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 79. | Lin W, Zhang Y, Yang Y, Lin B, Zhu M, Xu J, Chen Y, Wu W, Chen B, Chen X, Liu J, Wang H, Teng F, Yu X, Wang H, Lu J, Zhou Q, Teng L. Anti-PD-1/Her2 Bispecific Antibody IBI315 Enhances the Treatment Effect of Her2-Positive Gastric Cancer through Gasdermin B-Cleavage Induced Pyroptosis. Adv Sci (Weinh). 2023;10:e2303908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 80. | Ji D, Zhang J, Shen W, Du Y, Xu J, Yang J, Luo X, Kong P, Yang F, Hu X. Preliminary safety, efficacy and pharmacokinetics (PK) results of KN026, a HER2 bispecific antibody in patients (pts) with HER2-positive metastatic breast cancer. J Clin Oncol. 2020;38:1041. [DOI] [Full Text] |
| 81. | Mohammadi M, Jeddi-Tehrani M, Golsaz-Shirazi F, Arjmand M, Bahadori T, Judaki MA, Shiravi F, Zare HA, Haghighat FN, Mobini M, Amiri MM, Shokri F. A Novel Anti-HER2 Bispecific Antibody With Potent Tumor Inhibitory Effects In Vitro and In Vivo. Front Immunol. 2020;11:600883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 82. | Sun W, Wang X, Wang D, Lu L, Lin H, Zhang Z, Jia Y, Nie X, Liu T, Fu W. CD40×HER2 bispecific antibody overcomes the CCL2-induced trastuzumab resistance in HER2-positive gastric cancer. J Immunother Cancer. 2022;10:e005063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 83. | Zhang B, Shi J, Shi X, Xu X, Gao L, Li S, Liu M, Gao M, Jin S, Zhou J, Fan D, Wang F, Ji Z, Bian Z, Song Y, Tian W, Zheng Y, Xu L, Li W. Development and evaluation of a human CD47/HER2 bispecific antibody for Trastuzumab-resistant breast cancer immunotherapy. Drug Resist Updat. 2024;74:101068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 84. | Lum LG, Al-Kadhimi Z, Deol A, Kondadasula V, Schalk D, Tomashewski E, Steele P, Fields K, Giroux M, Liu Q, Flaherty L, Simon M, Thakur A. Phase II clinical trial using anti-CD3 × anti-HER2 bispecific antibody armed activated T cells (HER2 BATs) consolidation therapy for HER2 negative (0-2+) metastatic breast cancer. J Immunother Cancer. 2021;9:e002194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 85. | Budi HS, Ahmad FN, Achmad H, Ansari MJ, Mikhailova MV, Suksatan W, Chupradit S, Shomali N, Marofi F. Human epidermal growth factor receptor 2 (HER2)-specific chimeric antigen receptor (CAR) for tumor immunotherapy; recent progress. Stem Cell Res Ther. 2022;13:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 86. | Xu J, Meng Q, Sun H, Zhang X, Yun J, Li B, Wu S, Li X, Yang H, Zhu H, Aschner M, Relucenti M, Familiari G, Chen R. HER2-specific chimeric antigen receptor-T cells for targeted therapy of metastatic colorectal cancer. Cell Death Dis. 2021;12:1109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 87. | Song Y, Tong C, Wang Y, Gao Y, Dai H, Guo Y, Zhao X, Wang Y, Wang Z, Han W, Chen L. Effective and persistent antitumor activity of HER2-directed CAR-T cells against gastric cancer cells in vitro and xenotransplanted tumors in vivo. Protein Cell. 2018;9:867-878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 88. | Qi C, Gong J, Li J, Liu D, Qin Y, Ge S, Zhang M, Peng Z, Zhou J, Cao Y, Zhang X, Lu Z, Lu M, Yuan J, Wang Z, Wang Y, Peng X, Gao H, Liu Z, Wang H, Yuan D, Xiao J, Ma H, Wang W, Li Z, Shen L. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial interim results. Nat Med. 2022;28:1189-1198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 519] [Cited by in RCA: 477] [Article Influence: 119.3] [Reference Citation Analysis (5)] |
| 89. | Simon AG, Lyu SI, Laible M, Wöll S, Türeci Ö, Şahin U, Alakus H, Fahrig L, Zander T, Buettner R, Bruns CJ, Schroeder W, Gebauer F, Quaas A. The tight junction protein claudin 6 is a potential target for patient-individualized treatment in esophageal and gastric adenocarcinoma and is associated with poor prognosis. J Transl Med. 2023;21:552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 90. | Guzman G, Reed MR, Bielamowicz K, Koss B, Rodriguez A. CAR-T Therapies in Solid Tumors: Opportunities and Challenges. Curr Oncol Rep. 2023;25:479-489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 137] [Reference Citation Analysis (0)] |
| 91. | Mu J, Edwards J, Zaritskaya L, Swers J, Gupta A, Lafleur D, Hilbert D, Richman L. Selective targeting of HER2-overexpressing solid tumors with a next-generation CAR-T cell therapy. J Clin Oncol. 2020;38:3041. [DOI] [Full Text] |
| 92. | Wickstroem K, Karlsson J, Ellingsen C, Cruciani V, Kristian A, Hagemann UB, Bjerke RM, Ryan OB, Linden L, Mumberg D, Brands M, Cuthbertson A. Synergistic Effect of a HER2 Targeted Thorium-227 Conjugate in Combination with Olaparib in a BRCA2 Deficient Xenograft Model. Pharmaceuticals (Basel). 2019;12:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 93. | Karlsson J. Abstract ND07: HER2-TTC: A Targeted thorium conjugate to treat HER2 expressing cancers with potent alpha radiation. Cancer Res. 2021;81:ND07. [RCA] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 94. | Garg P, Malhotra J, Kulkarni P, Horne D, Salgia R, Singhal SS. Emerging Therapeutic Strategies to Overcome Drug Resistance in Cancer Cells. Cancers (Basel). 2024;16:2478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 157] [Reference Citation Analysis (0)] |
| 95. | Anderson PM, Subbiah V, Trucco MM. Current and future targeted alpha particle therapies for osteosarcoma: Radium-223, actinium-225, and thorium-227. Front Med (Lausanne). 2022;9:1030094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 96. | Schiff R, Veeraraghavan J, De Angelis C, Osborne C, Rimawi M. Abstract SP139: HER2 targeted therapy: Determinants of response and mechanisms of resistance. Cancer Res. 2021;81:SP139. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 97. | Chen B, Liao N, Zhang G, Wang Y, Chen X, Guo L, Cao L, Mok H, Ren C, Li K, Jia M, Li C, Wen L, Lin J, Wei G, Hou T, Lizaso A, Liu J, Balch CM. Distinct mutational landscape between HR+ and HR- HER2+ early-stage breast cancer patients. J Clin Oncol. 2019;37:543. [DOI] [Full Text] |
| 98. | O'keefe K, Elliott A, Livasy C, Steiner M, Kang I, Hoon DSB, Korn WM, Walker P, Radovich M, Pohlmann PR, Swain SM, Tan AR, Heeke AL. HER2 alterations and prognostic implications in all subtypes of breast cancer. J Clin Oncol. 2022;40:1041. [DOI] [Full Text] |
| 99. | Jebbink M, de Langen AJ, Boelens MC, Monkhorst K, Smit EF. The force of HER2 - A druggable target in NSCLC? Cancer Treat Rev. 2020;86:101996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 100. | Jiao XD, Liu K, Wu Y, Zhou XC, Qin BD, Ling Y, Liu J, He X, Du H, Xiang J, Zang YS. HER2 Splice Site Mutation c.1899-1G>A as the Potential Acquired Resistance to Trastuzumab in a Patient with HER2-Positive Gastric Adenocarcinoma. Oncologist. 2021;26:717-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 101. | Marchiò C, Annaratone L, Marques A, Casorzo L, Berrino E, Sapino A. Evolving concepts in HER2 evaluation in breast cancer: Heterogeneity, HER2-low carcinomas and beyond. Semin Cancer Biol. 2021;72:123-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 259] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 102. | Sperinde J, Huang W, Vehtari A, Chenna A, Kellokumpu-Lehtinen PL, Winslow J, Bono P, Lie YS, Petropoulos CJ, Weidler J, Joensuu H. p95HER2 Methionine 611 Carboxy-Terminal Fragment Is Predictive of Trastuzumab Adjuvant Treatment Benefit in the FinHer Trial. Clin Cancer Res. 2018;24:3046-3052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 103. | Goh CW, Zhang L, Chi W, Xiu B, Chen J, Yang W, Ao C, Tang J, Xue J, Chi Y, Wu J. Abstract P5-02-53: P95HER2 Expression in HER2-Positive Breast Cancer. Cancer Res. 2023;83:P5-02. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 104. | Janiszewska M, Stein S, Metzger Filho O, Eng J, Kingston NL, Harper NW, Rye IH, Alečković M, Trinh A, Murphy KC, Marangoni E, Cristea S, Oakes B, Winer EP, Krop IE, Russnes HG, Spellman PT, Bucher E, Hu Z, Chin K, Gray JW, Michor F, Polyak K. The impact of tumor epithelial and microenvironmental heterogeneity on treatment responses in HER2+ breast cancer. JCI Insight. 2021;6:e147617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 105. | Smith AE, Chandarlapaty S. Abstract 1910: Resistance to anti-HER2 therapy in HER2+ breast cancer is mediated by genomic alterations that switch pathway dependence from PI3K/AKT to RAS/MAPK. Cancer Res. 2020;80:1910. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 106. | Yu L, Wei J, Liu P. Attacking the PI3K/Akt/mTOR signaling pathway for targeted therapeutic treatment in human cancer. Semin Cancer Biol. 2022;85:69-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 530] [Article Influence: 132.5] [Reference Citation Analysis (3)] |
| 107. | Garay JP, Smith R, Devlin K, Hollern DP, Liby T, Liu M, Boddapati S, Watson SS, Esch A, Zheng T, Thompson W, Babcock D, Kwon S, Chin K, Heiser L, Gray JW, Korkola JE. Sensitivity to targeted therapy differs between HER2-amplified breast cancer cells harboring kinase and helical domain mutations in PIK3CA. Breast Cancer Res. 2021;23:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 108. | Garay J, Korkola J, Gray J. Abstract P3-03-04: Sensitivity to lapatinib differs between HER2-amplified breast cancer cells harboring kinase and helical domain mutations in PIK3CA and relies on production of PIP3. Cancer Res. 2017;77:P3-03. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 109. | Smith AE, Ferraro E, Safonov A, Morales CB, Lahuerta EJA, Li Q, Kulick A, Ross D, Solit DB, de Stanchina E, Reis-Filho J, Rosen N, Arribas J, Razavi P, Chandarlapaty S. HER2 + breast cancers evade anti-HER2 therapy via a switch in driver pathway. Nat Commun. 2021;12:6667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 110. | Wang Q, Wu S, Gu Y, Liang H, He F, Wang X, He D, Wu K. RASAL2 regulates the cell cycle and cyclin D1 expression through PI3K/AKT signalling in prostate tumorigenesis. Cell Death Discov. 2022;8:275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 111. | Bassi C, Fortin J, Snow BE, Wakeham A, Ho J, Haight J, You-Ten A, Cianci E, Buckler L, Gorrini C, Stambolic V, Mak TW. The PTEN and ATM axis controls the G1/S cell cycle checkpoint and tumorigenesis in HER2-positive breast cancer. Cell Death Differ. 2021;28:3036-3051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 112. | Szymczyk J, Sluzalska KD, Materla I, Opalinski L, Otlewski J, Zakrzewska M. FGF/FGFR-Dependent Molecular Mechanisms Underlying Anti-Cancer Drug Resistance. Cancers (Basel). 2021;13:5796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 113. | Wang X, Jiang W, Du Y, Zhu D, Zhang J, Fang C, Yan F, Chen ZS. Targeting feedback activation of signaling transduction pathways to overcome drug resistance in cancer. Drug Resist Updat. 2022;65:100884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (0)] |
| 114. | Recondo G, Che J, Jänne PA, Awad MM. Targeting MET Dysregulation in Cancer. Cancer Discov. 2020;10:922-934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 179] [Article Influence: 29.8] [Reference Citation Analysis (4)] |
| 115. | Mami-Chouaib F, Terry S, Buart S, Gausdal G, Angelsen A, Lorens J, Chouaib S. Role of the receptor tyrosine kinase AXL in regulating antitumor response and relationship with hypoxic stress. J Immunol 2020; 204: 242. 50;. [DOI] [Full Text] |
| 116. | Koirala N, Dey N, Aske J, De P. Targeting Cell Cycle Progression in HER2+ Breast Cancer: An Emerging Treatment Opportunity. Int J Mol Sci. 2022;23:6547. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 117. | Mahdi H, Hasipek M, Guan Y, Grabowski D, Al-sudani H, Parker Y, Boyd A, Rose P, Lindner D, Jha B. Combined therapy with HER2 and CDK4/6 inhibitors in HER2+ uterine and ovarian carcinomas: Synergistic effect with combined therapy. Gynecol Oncol. 2019;154:87-88. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 118. | Shagisultanova E, Crump LS, Borakove M, Hall JK, Rasti AR, Harrison BA, Kabos P, Lyons TR, Borges VF. Triple Targeting of Breast Tumors Driven by Hormonal Receptors and HER2. Mol Cancer Ther. 2022;21:48-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 119. | Clark AS, Makhlin I, DeMichele A. Setting the Pick: Can PI3K Inhibitors Circumvent CDK4/6 Inhibitor Resistance? Clin Cancer Res. 2021;27:371-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 120. | Suenaga M, Stintzing S, Cao S, Zhang W, Yang D, Ning Y, Okazaki S, Berger MD, Miyamoto Y, Schirripa M, Soni S, Barzi A, Heinemann V, Lenz HJ. Role of CCL5 and CCR5 gene polymorphisms in epidermal growth factor receptor signalling blockade in metastatic colorectal cancer: analysis of the FIRE-3 trial. Eur J Cancer. 2019;107:100-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 121. | Abuelreich S, Leon N, Medina D, Sahni M, Krishnan K, Bush J. Evaluating the Metabolite Profile of HER2‐positive Breast Cancer Cells. FASEB J. 2020;34:1. [DOI] [Full Text] |
| 122. | Wu X, Yang H, Yu X, Qin JJ. Drug-resistant HER2-positive breast cancer: Molecular mechanisms and overcoming strategies. Front Pharmacol. 2022;13:1012552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 44] [Reference Citation Analysis (0)] |
| 123. | Ughetto S, Migliore C, Pietrantonio F, Apicella M, Petrelli A, D'Errico L, Durando S, Moya-Rull D, Bellomo SE, Rizzolio S, Capelôa T, Ribisi S, Degiuli M, Reddavid R, Rapa I, Fumagalli U, De Pascale S, Ribero D, Baronchelli C, Sgroi G, Rausa E, Baiocchi GL, Molfino S, Manenti S, Bencivenga M, Sacco M, Castelli C, Siena S, Sartore-Bianchi A, Tosi F, Morano F, Raimondi A, Prisciandaro M, Gloghini A, Marsoni S, Sottile A, Sarotto I, Sapino A, Marchiò C, Cassoni P, Guarrera S, Corso S, Giordano S. Personalized therapeutic strategies in HER2-driven gastric cancer. Gastric Cancer. 2021;24:897-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 124. | Ning G, Zhu Q, Kang W, Lee H, Maher L, Suh YS, Michaud M, Silva M, Kwon JY, Zhang C, Lee C. A novel treatment strategy for lapatinib resistance in a subset of HER2-amplified gastric cancer. BMC Cancer. 2021;21:923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 125. | van Hasselt JGC, Iyengar R. Systems Pharmacology: Defining the Interactions of Drug Combinations. Annu Rev Pharmacol Toxicol. 2019;59:21-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 126. | Chan TG, O'Neill E, Habjan C, Cornelissen B. Combination Strategies to Improve Targeted Radionuclide Therapy. J Nucl Med. 2020;61:1544-1552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 127. | Finn L, Lunski M, Gadde S, Alberti M, Markabawi D, Finn L. Strategizing the Treatment Approach to Acute Myeloid Leukemia. Clin Oncol Res. 2021. [DOI] [Full Text] |
| 128. | Manzari MT, Shamay Y, Kiguchi H, Rosen N, Scaltriti M, Heller DA. Targeted drug delivery strategies for precision medicines. Nat Rev Mater. 2021;6:351-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 659] [Article Influence: 131.8] [Reference Citation Analysis (4)] |
| 129. | Ocaña A, Amir E, Pandiella A. HER2 heterogeneity and resistance to anti-HER2 antibody-drug conjugates. Breast Cancer Res. 2020;22:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 130. | Mahalingaiah PK, Ciurlionis R, Durbin KR, Yeager RL, Philip BK, Bawa B, Mantena SR, Enright BP, Liguori MJ, Van Vleet TR. Potential mechanisms of target-independent uptake and toxicity of antibody-drug conjugates. Pharmacol Ther. 2019;200:110-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 209] [Article Influence: 29.9] [Reference Citation Analysis (1)] |
| 131. | Chen YF, Xu YY, Shao ZM, Yu KD. Resistance to antibody-drug conjugates in breast cancer: mechanisms and solutions. Cancer Commun (Lond). 2023;43:297-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 110] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 132. | Liu-Kreyche P, Shen H, Marino AM, Iyer RA, Humphreys WG, Lai Y. Lysosomal P-gp-MDR1 Confers Drug Resistance of Brentuximab Vedotin and Its Cytotoxic Payload Monomethyl Auristatin E in Tumor Cells. Front Pharmacol. 2019;10:749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 133. | Ceci C, Lacal PM, Graziani G. Antibody-drug conjugates: Resurgent anticancer agents with multi-targeted therapeutic potential. Pharmacol Ther. 2022;236:108106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 134. | McLemore LE, Albarracin CT, Gruschkus SK, Bassett RL Jr, Wu Y, Dhamne S, Yim I, Lin K, Bedrosian I, Sneige N, Chen H. HER2 testing in breast cancers: comparison of assays and interpretation using ASCO/CAP 2013 and 2018 guidelines. Breast Cancer Res Treat. 2021;187:95-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 135. | Taylor VJ, Barnes PJ, Godwin SC, Bethune GC. Assessment of HER2 using the 2018 ASCO/CAP guideline update for invasive breast cancer: a critical look at cases classified as HER2 2+ by immunohistochemistry. Virchows Arch. 2021;479:23-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 136. | Klc TR, Wu S, Wilhite AM, Jones NL, Powell MA, Olawaiye A, Girda E, Brown J, Puechl A, Ali-Fehmi R, Winer IS, Herzog TJ, Korn WM, Erickson BK. HER2 in Uterine Serous Carcinoma: Testing platforms and implications for targeted therapy. Gynecol Oncol. 2022;167:289-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 137. | Ciesielski M, Lewandowska M, Szajewski M, Walczak J, Pęksa R, Zieliński J, Lenckowski R, Supeł M, Kruszewski W. Impact of chromosome 17 polysomy on HER2 receptor overexpression and patients survival in gastric adenocarcinoma. Is HER2/CEP17 ratio an appropriate method of FISH assessment? Europe J Surg Oncol. 2019;45:e60. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 138. | Vermij L, Singh N, Leon-Castillo A, Horeweg N, Oosting J, Carlson J, Smit V, Gilks B, Bosse T. Performance of a HER2 testing algorithm specific for p53-abnormal endometrial cancer. Histopathology. 2021;79:533-543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 139. | Ho HY, Chung KK, Kan CM, Wong SC. Liquid Biopsy in the Clinical Management of Cancers. Int J Mol Sci. 2024;25:8594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 140. | Koessler T, Addeo A, Nouspikel T. Implementing circulating tumor DNA analysis in a clinical laboratory: A user manual. Adv Clin Chem. 2019;89:131-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 141. | Rossi G, Ignatiadis M. Promises and Pitfalls of Using Liquid Biopsy for Precision Medicine. Cancer Res. 2019;79:2798-2804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 142. | Li Q, Lv M, Lv L, Cao N, Zhao A, Chen J, Tang X, Luo R, Yu S, Zhou Y, Cui Y, Guo W, Liu T. Identifying HER2 from serum-derived exosomes in advanced gastric cancer as a promising biomarker for assessing tissue HER2 status and predicting the efficacy of trastuzumab-based therapy. Cancer Med. 2023;12:4110-4124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 143. | Massihnia D, Pizzutilo EG, Amatu A, Tosi F, Ghezzi S, Bencardino K, Di Masi P, Righetti E, Patelli G, Scaglione F, Vanzulli A, Siena S, Sartore-Bianchi A. Liquid biopsy for rectal cancer: A systematic review. Cancer Treat Rev. 2019;79:101893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 144. | Yuan Q, Deng D, Pan C, Ren J, Wei T, Wu Z, Zhang B, Li S, Yin P, Shang D. Integration of transcriptomics, proteomics, and metabolomics data to reveal HER2-associated metabolic heterogeneity in gastric cancer with response to immunotherapy and neoadjuvant chemotherapy. Front Immunol. 2022;13:951137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 145. | Pfeifer B, Schimek MG. A hierarchical clustering and data fusion approach for disease subtype discovery. J Biomed Inform. 2021;113:103636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 146. | Kerr DJ, Yang L. Personalising cancer medicine with prognostic markers. EBioMedicine. 2021;72:103577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 147. | Bueno Muiño C, Martín M, Del Monte-Millán M, García-Saénz JÁ, López-Tarruella S. HER2+ Breast Cancer Escalation and De-Escalation Trial Design: Potential Role of Intrinsic Subtyping. Cancers (Basel). 2022;14:512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |