Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115912
Revised: November 20, 2025
Accepted: December 23, 2025
Published online: March 15, 2026
Processing time: 135 Days and 6.4 Hours
Primary gastrointestinal lymphomas (PGIL) are rare tumors that can involve the whole gastrointestinal (GI) tract. Although lymphomas can originate from any part of the gastrointestinal tract, the most common sites are the stomach, small intestine, and ileocecal region.
To examine the clinicopathologic and prognostic features of PGIL.
We performed a retrospective single-center analysis of 111 patients diagnosed with gastrointestinal lymphomas (GIL) at Izmir Katip Celebi University Ataturk Training and Research Hospital. Histopathological, clinical, and prognostic para
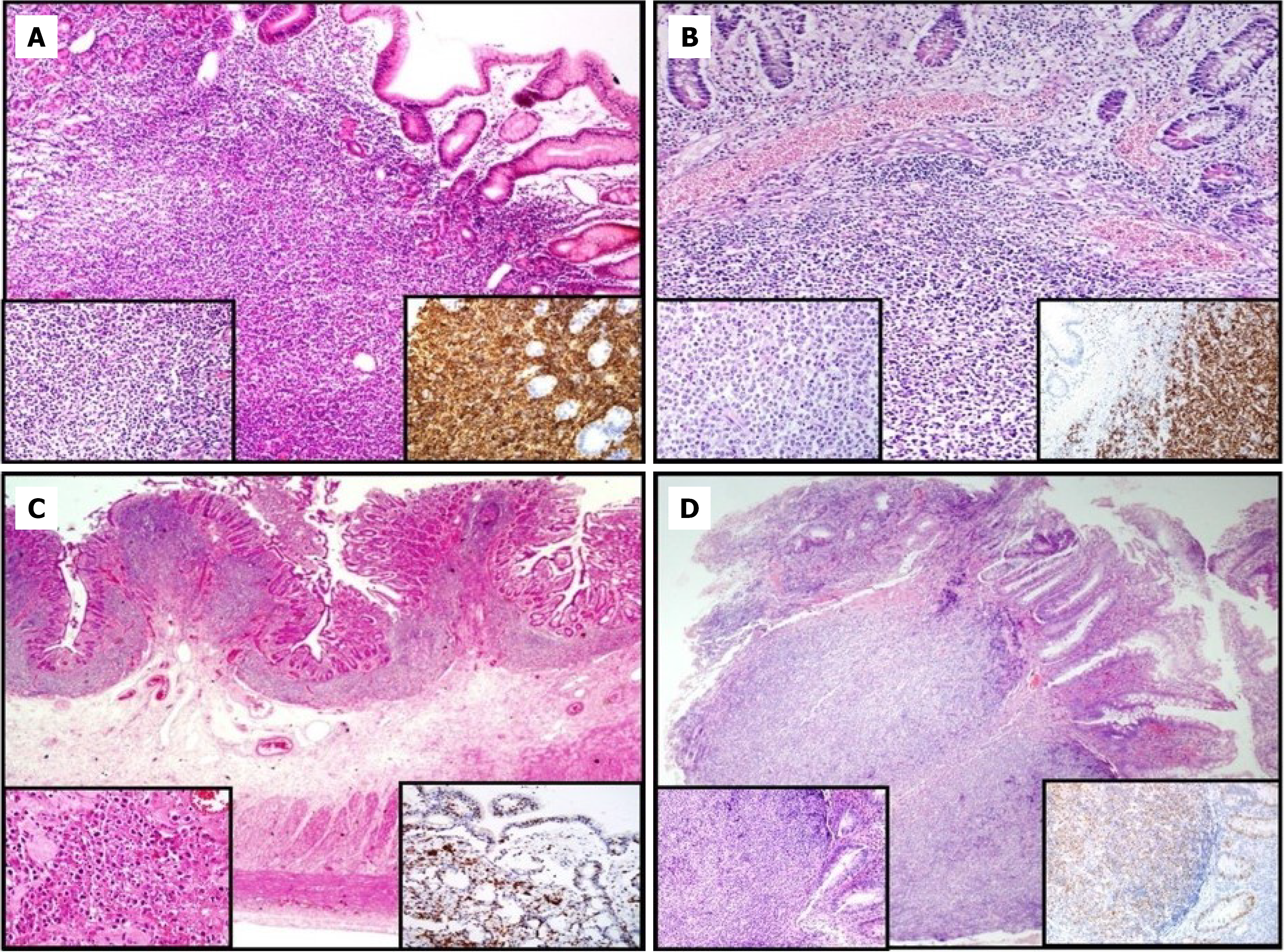
The study included 111 patients diagnosed with GIL. The median age was 66 years (60 males, 51 females). Sites of involvement were stomach (67.5%), small bowel (18%), large bowel/rectum (12.6%), and pancreas (1.8%). Among 62 patients with primary GIL, 49 (79%) had diffuse large B-cell lymphoma (DLBCL), 6 (9.7%) had extranodal mar
In this study, the stomach was the most frequently involved site, DLBCL was the predominant subtype, and poor performance status was associated with poorer survival.
Core Tip: Primary gastrointestinal lymphoma (PGIL) is a rare and heterogeneous disease with diverse clinicopathologic and prognostic features. In this retrospective study of 62 primary cases, the stomach was the most frequently involved site, and diffuse large B-cell lymphoma was the predominant subtype. Poor performance status emerged as an independent predictor of reduced overall survival. This finding contributes to a better understanding of prognostic determinants in PGIL and may guide individualized management strategies in clinical practice.
- Citation: Buyuktalanci DO, Cakir E, Kucukzeybek BB, Altindag SD, Yilmaz AF, Kiper HD, Dilek ON. Analysis of histopathological and clinical prognostic factors in lymphomas involving the gastrointestinal system. World J Gastrointest Oncol 2026; 18(3): 115912
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/115912.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.115912
Primary gastrointestinal lymphomas (PGILs) are the most common type of extranodal malignant lymphomas. They represent 30%-45% of all extranodal lymphomas, 4%-20% of all non-Hodgkin lymphomas, and 1%-4% of all gastro
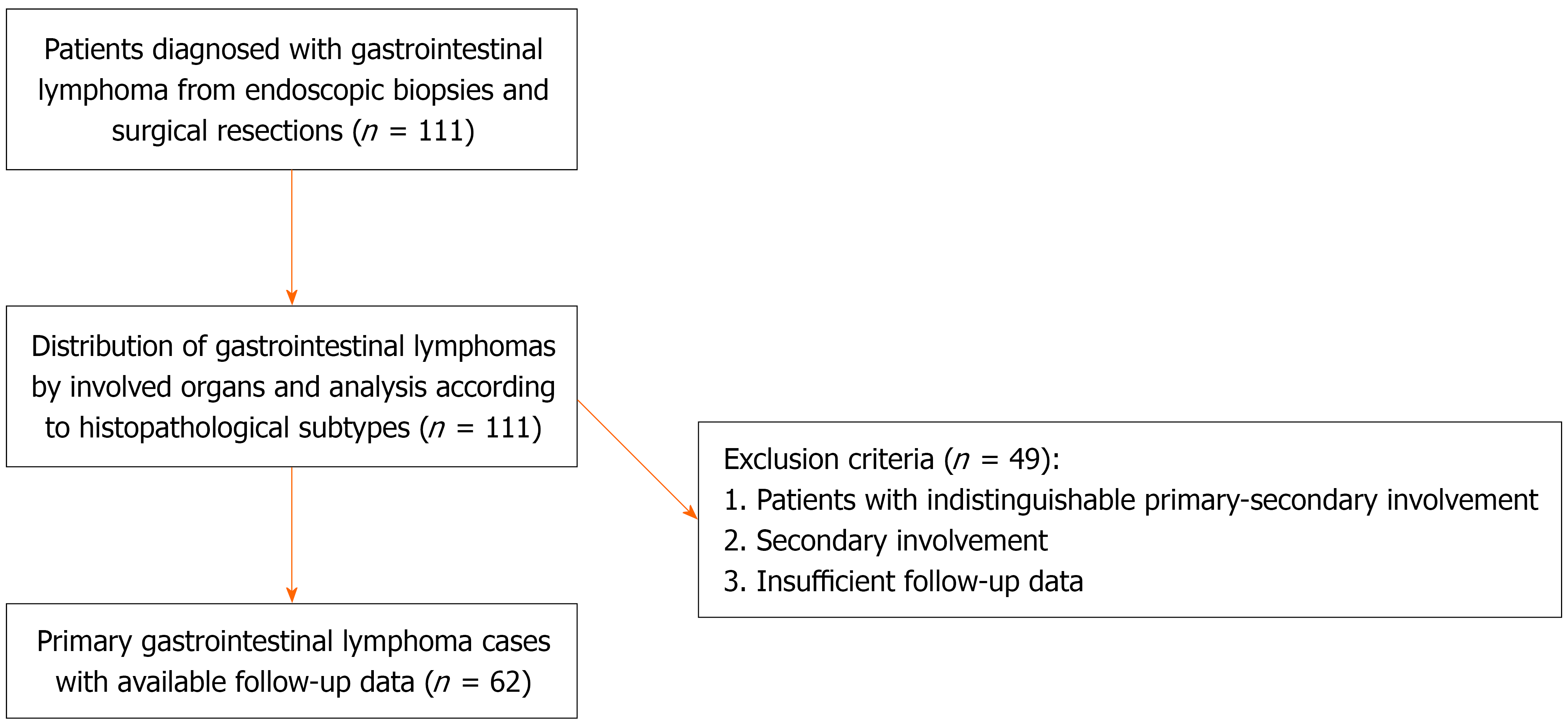
This retrospective study included 111 patients diagnosed with GIL between 2006 to 2018 at Izmir Katip Celebi University Ataturk Training and Research Hospital. Pathological specimens were obtained from endoscopic biopsies and surgical resections. The analysis of organ distribution and histopathological subtypes of gastrointestinal tract lymphomas was performed on a total of 111 cases included in the study. The histopathological diagnosis of GIL was based on the 2008 and 2016 WHO classifications. According to the immunohistochemical results, cases were identified as B-cell or T-cell phenotype, and histopathological subtyping was performed when available. Patients with indistinguishable primary-secondary involvement (n = 21), secondary involvement (n = 21), or insufficient follow-up data (n = 7) were excluded from the study before overall and disease-free survival analyses, based on detailed examination of the radiological findings (Figure 1). Data concerning each patient, including age, gender, anatomic distribution, pathological subtype, Helicobacter pylori (H. pylori) infection, tumor stage, grade, Performance Score (PS), International Prognostic Index (IPI) score, treatment modality, and lactate dehydrogenase (LDH) level, were recorded. The cut-off value for serum LDH was set at ≥ 270 U/L, corresponding to the upper limit of the normal reference range at our institution. This threshold has also been used in previous studies evaluating prognostic factors in lymphoma. Follow-up data, including patient survival, were obtained from the hematology clinic. Clinical stages were evaluated according to the Lugano International Conference classification (I, II1, II2, IIE, or IV), which was recommended as a modified version of the Ann Arbor criteria for PGIL[1,8]. PET-CT was used for staging when available, while contrast-enhanced CT was performed in other cases. Multifocal disease was defined as the presence of two or more non-contiguous gastrointestinal lesions or concurrent involvement of gastrointestinal and other extranodal sites.
Performance status was determined according to the Eastern Cooperative Oncology Group (ECOG) scale[8,9]. Patients were classified into low-risk (IPI score 0-2) and high-risk groups (IPI score ≥ 3) according to the IPI. Patients were classified into an operation group which was defined as those who underwent surgery with postoperative chemotherapy, and a non-operation group. In addition, intestinal and gastric lymphomas were compared with respect to prognostic factors and outcomes.
All statistical analyses were performed using SPSS 25.0 (SPSS, Chicago, IL, United States). Kaplan-Meier curves (log-rank test) were used to analyze event-free survival (EFS) and overall survival (OS). EFS was defined as the time from diagnosis to disease relapse. Multivariate Cox regression analysis was performed to identify independent prognostic factors for OS and EFS. Significant variables (P < 0.05) in univariate analyses were included in the multivariate model. The enter method was used for variable selection. Hazard ratios with 95% confidence intervals were calculated. Other statistical differences were evaluated using the χ2 test. A P value of < 0.05 was considered statistically significant. To reduce potential selection bias between surgical and non-surgical groups, a propensity score for undergoing surgery was calculated using logistic regression based on age, stage, LDH level, ECOG PS, IPI, and histological subtype. Patients were then matched 1:1 using nearest-neighbor matching without replacement, with a caliper width of 0.2 times of the standard deviation of the logit of the propensity score.
Patient characteristics are summarized in Table 1. A total of 111 patients were included in the study, comprising 60 males and 51 females, with a median age of 66 years (range, 21-89 years). The pathological samples were obtained from en
| Histologic type | Stomach (n = 75) | Small bowel | Large bowel rectum | Pancreas (n = 2) | Total patients |
| B-cell lymphoma | 106 (95.4) | ||||
| DLBCL | 65 (86.6) | 13 (65) | 7 (50) | 2 (100) | |
| MALT lymphoma | 6 (8) | 2 (10) | 2 (14.2) | ||
| MCL | 1 (1.3) | 1 (5) | 3 (21.4) | ||
| BL | 1 (1.3) | ||||
| PL | 1 (1.3) | ||||
| Unclassified | 2 (14.2) | ||||
| T-cell lymphoma | 5 (4.6) | ||||
| EATCL | 3 (15) | ||||
| ALCL | 1 (5) | ||||
| Unclassified | 1 (1.3) |
Sixty-two patients remained in the study after excluding patients with indistinguishable primary vs secondary involvement (n = 21), secondary involvement (n = 21), or inadequate follow-up data (n = 7). Among these 62 patients (35 males and 27 females), ages ranged from 21 to 89 years with a median of 60.5 years. Most patients were older than 60 years (n = 34, 54.8%); while 28 patients (45.2%) were younger than 60 years. Of the 62 patients, 60 (96.8%) had a B-cell phenotype, and 2 (3.2%) had a T-cell phenotype. Amongst these patients, 49 cases (79%) were DLBCL, 6 (9.7%) were MALT lymphoma, 2 (3.2%) were MCL, 2 (3.2%) were unclassified B-cell lymphoma, 1 (1.6%) was PL, 1 (1.6%) was EATCL and 1 (1.6%) was unclassified T-cell lymphoma (Figure 2). According to the Lugano staging system, 57 patients (91.9%) had Stage I and II disease, while 5 (8.1%) patients had Stage IV disease. LDH levels were normal in 44 cases (71%), elevated in 18 cases (29%). Regarding treatment, 42 patients (67.7%) received chemotherapy alone, 10 (16.1%) received combined chemoradiotherapy, and 10 (16.1%) underwent surgery followed by chemotherapy. All 62 patients were included in the survival analysis. The median follow-up was 17 (1-119) months, and the median survival time was 63 ± 25.3 months. The 1-year, 3-year, and 5-year OS and EFS rates, estimated by the Kaplan-Meier method, were 67%, 56%, and 47%, and 80%, 58.6%, and 54.4%, respectively. Patients with elevated serum LDH levels had 1-, 3-, and 5-year OS rates of 77.1%, 69.3%, and 62.7%, compared with 44.4%, 27.8%, and 27.8% in those with normal serum LDH levels (< 270 U/L) (P < 0.029). Patients with low IPI scores had significantly better 1-, 3-, and 5-year EFS (92.7%, 57.1%, 27.4%, P = 0.024), but not OS (68.9%, 58.4%, 53.5%, P = 0.095), compared with patients with higher IPI scores (IPI score ≥ 3). In our study, ECOG performance status was divided into two groups: 0–1 and ≥ 2. Among the 62 patients, 41 had a score of 0, 14 had a score of 1, 5 had a score of 2, and 2 had a score of 3, resulting in seven patients (11.3%) classified as having poor performance status (ECOG ≥ 2). Although this subgroup was relatively small, its inclusion still revealed a significant negative impact on OS in both univariate and multivariate analyses, indicating that poor performance status remained a strong prognostic factor despite the limited sample size. The prognosis of patients (OS) with poor PS (ECOG ≥ 2) was statistically worse (P < 0.0001), and patients with poor performance status showed a statistically significant difference in EFS (P = 0.002). Patients who underwent surgical treatment showed a worse OS (P < 0.002) and EFS (P = 0.013) than those who had chemotherapy-based modalities. As a result, in univariate analysis within all prognostic factors, elevated LDH levels (≥ 270 U/L), poor PS (ECOG ≥ 2), and undergoing surgery were associated with reduced survival. Variables that were significant in the univariate analysis -elevated LDH (LDH ≥ 270 U/L), poor PS (ECOG ≥ 2), and surgical treatment- were included in the multivariate analysis, where only poor performance (ECOG ≥ 2) score remained an independent prognostic factor (P = 0.010). Table 2 summarizes the univariate and multivariate analyses of the prognostic factors for OS and EFS. Patients were classified as intestinal (n = 19) or gastric lymphomas (n = 43), and the two groups were compared with respect to age, gender, cell phenotype, Lugano stage, PS, LDH level, and IPI scores. The median age was 59 years (range 21-87) for gastric lymphomas and 66 years (range 24-89) for intestinal lymphomas. Elevated LDH levels were observed in 16 patients (37.2%) in the gastric group and 2 patients (10.5%) in the intestinal group. A high LDH level differed significantly (P < 0.033) between the two groups. Among gastric lymphomas, 5 of 43 patients (11.6%) had a PS ≥ 2, compared with 2 of 19 patients (10.5%) with intestinal lymphomas (P = 1.000). Four patients (9.3%) in the gastric lymphoma group had a high IPI score (IPI score ≥ 3), vs 1 (5.3%) patient in the intestinal lymphoma group (P = 0.666). Of the 43 patients with primary gastric lymphomas, 40 (93%) were in Lugano stage I-II, and 3 (7%) were in stage IV. Among the 19 patients with primary intestinal lymphomas, 17 (89.5%) were stage I-II, and 2 (10.5%) were stage IV (P = 0.638). No significant differences were observed between gastric and intestinal lymphomas in terms of age, gender, cell phenotype, stage, PS, or IPI score.
| Clinical characteristic | Case (n) | OS (%) | EFS (%) | |
| Univariate analysis 1-year/3-year/ 5-year, P value | Multivariate analysis, P value | P value | ||
| Age (years) | 0.529 | - | 0.546 | |
| < 60 | 28 | 64.3/56/56 | ||
| ≥ 60 | 34 | 70.4/49.4/49.4 | ||
| Gender | 0.853 | - | 0.842 | |
| Female | 27 | 54.7/50.2/50.2 | ||
| Male | 35 | 77.1/61.7/54.5 | ||
| Cell Fenotype | 0.900 | - | 0.403 | |
| B-Cell | 60 | 68.1/56.7/52.2 | ||
| T-Cell | 2 | 50/50/50 | ||
| LDH level | 0.029 | 0.085 | 0.090 | |
| < 270 | 44 | 77.1/69.3/62.7 | ||
| ≥ 270 | 18 | 44.4/27.8/27.8 | ||
| Surgical treatment | 0.002 | 0.384 | 0.013 | |
| Surgical treatment (-) | 52 | 74.8/63.7/58.6 | ||
| Surgical treatment (+) | 10 | 30/20/20 | ||
| Stage group | 0.553 | - | 0.909 | |
| I-II | 57 | 68.2/58.1/53.5 | ||
| IV | 5 | 60/40/40 | ||
| Performance score | < 0.0001 | 0.010 | 0.002 | |
| 0-1 | 55 | 74.4/63.9/59 | ||
| ≥ 2 | 7 | 14.3/0/0 | ||
| IPI score | 0.095 | - | 0.024 | |
| 0-2 | 55 | 68.9/58.4/53.5 | ||
| ≥ 3 | 5 | 40/20/20 | ||
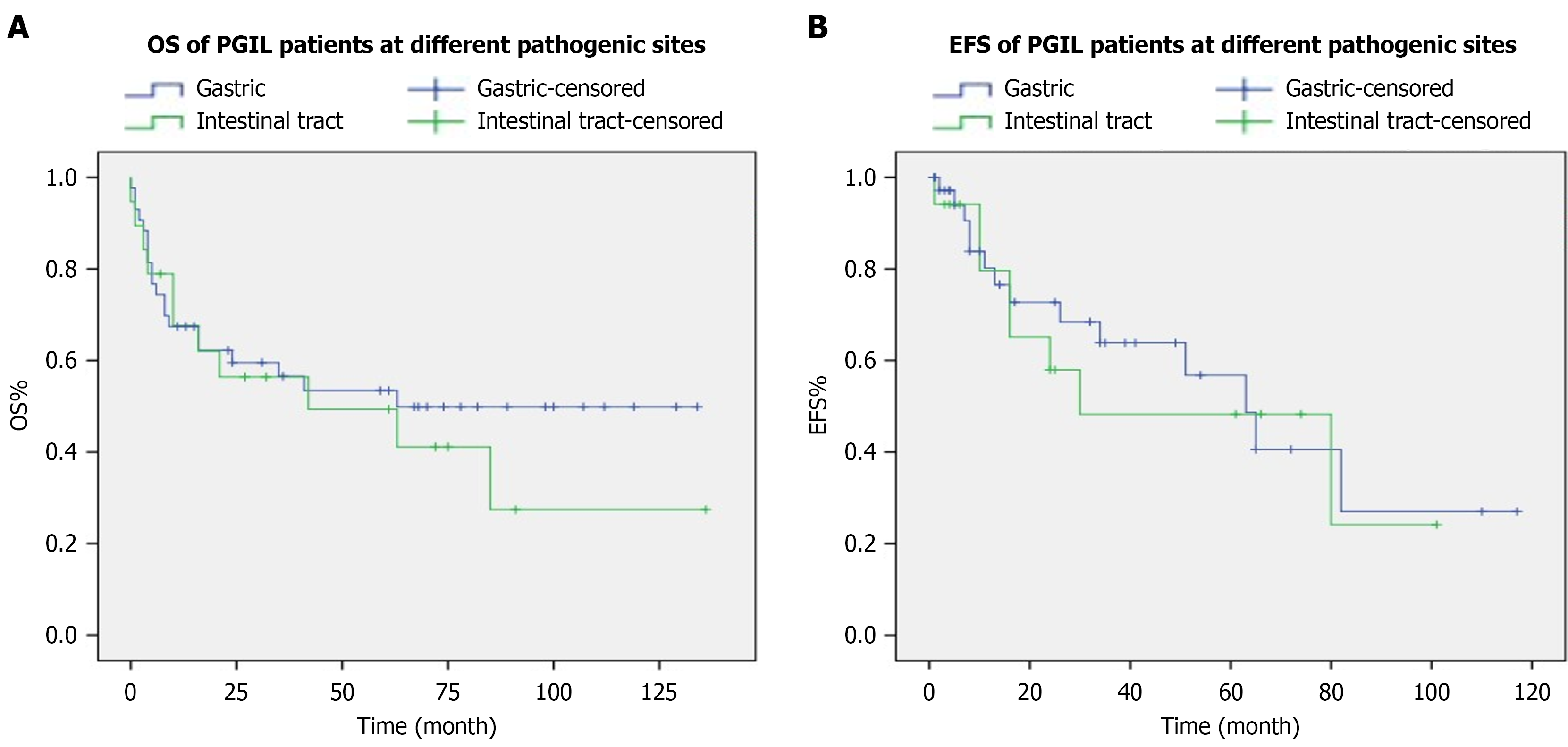
The Kaplan-Meier method was used to analyze survival. Thirty-one patients died; including 20 in the gastric group and 11 in the intestinal group. For intestinal lymphomas, the 1-, 3-, and 5-year the OS rates were 67.7%, 56.4%, and 49.3%, and the corresponding EFS rates were 79.6%, 48.3% and 48.3%. For gastric lymphomas, the 1-, 3-, and 5-year OS rates were 67.4%, 56.6%, and 53.4%, and the EFS rates were 80.2%, 63.9%, and 56.8%. Table 3 summarizes the clinical characteristics and, OS and EFS outcomes for gastric and intestinal lymphomas. There were no statistically significant differences in OS or EFS between the two groups (P = 0.531 and 0.720, respectively) (Figure 3).
| Clinical characteristic | Stomach (n = 43) (69.3%) | Intestinal tract (n = 19) (30.6%) | P value |
| Age (years) | 0.153 | ||
| < 60 | 22 (51.2) | 6 (31.6) | |
| ≥ 60 | 21 (48.8) | 13 (68.4) | |
| Gender | 0.687 | ||
| Female | 18 (41.9) | 9 (47.4) | |
| Male | 25 (58.1) | 10 (52.6) | |
| Histologic type | 0.522 | ||
| B-Cell | 42 (97.7) | 18 (94.7) | |
| T-Cell | 1 (2.3) | 1 (5.3) | |
| LDH level | 0.033 | ||
| < 270 | 27 (62.8) | 17 (89.5) | |
| ≥ 270 | 16 (37.2) | 2 (10.5) | |
| Stage group | 0.638 | ||
| I-II | 40 (93) | 17 (89.5) | |
| IV | 3 (7) | 2 (10.5) | |
| Performance score | 1.000 | ||
| 0-1 | 38 (88.4) | 17 (89.5) | |
| ≥ 2 | 5 (11.6) | 2 (10.5) | |
| IPI score | 0.666 | ||
| 0-2 | 39 (90.7) | 16 (84.2) | |
| ≥ 3 | 4 (9.3) | 1 (5.3) | |
| OS (%) | 0.531 | ||
| 1-year/3-year/5-year | 67.4/56.6/53.4 | 67.7/56.4/49.3 | |
| EFS (%) | 0.720 | ||
| 1-year/3-year/5-year | 80.2/63.9/56.8 | 79.6/48.3/48.3 |
Forty-three primary gastric lymphomas were further evaluated for the presence of H. pylori infection. The microorganism was detected in 21 patients (48%), all of whom underwent eradication therapy. Patients with H. pylori infection had significantly better 1-, 3-, and 5-year OS rates (76%, 47.6%, 28.6%) than those without infection (P < 0.0001). H. pylori infection was detected in four of six MALT lymphoma cases, and all H. pylori-positive patients underwent eradication therapy. However, due to the small sample size, statistical evaluation of the association between H. pylori status or eradication therapy and survival was not feasible.
The gastrointestinal tract is the most common extranodal site of involvement in NHL, whereas PGIL remains relatively rare. PGIL is a heterogeneous disease with respect to patient characteristics, histopathologic subtypes, stage, and treatment outcomes[1,7,10].
PGIL is usually diagnosed between the ages of 50 and 60 years[3]. The median age in the present study was 60.5 years, similar to the findings of Papaxoinis et al[7] (62 years). However, some studies, including those by Wang et al[3] and Wang et al[9], reported median ages of 53 and 58 years, respectively. In our cohort, patients with gastric lymphomas were younger than those with intestinal lymphomas, although the difference was not statistically significant. Regarding gender distribution, our results were consistent with previous studies, showing a higher incidence in males[10-12].
Many studies have shown that the stomach is the most commonly involved site, with a reported frequency ranging from 52.9% to 82%, followed by the intestinal tract. However, in the Pacific region, the small intestine is most commonly affected, followed by the stomach and colon[10,12,13]. In Middle Eastern countries, the frequency of intestinal lymphoma is also high (49%-81%), which is partly attributed to the high prevalence of IPSID in these areas. These differences may also reflect geographic variations in the prevalence of H. pylori infection, celiac disease, or other environmental factors[1,14,15]. In the present study, the stomach was the most common site, with primary gastric lymphomas accounting for 69.3% of cases.
Consistent with previous studies, B-cell lymphomas were more frequent (96.8%) than T-cell lymphomas in our cohort[16,17]. Wang et al[3] reported that B- and T-cell lymphomas accounted for 82.69% and 17.31%, respectively, while Chen et al[18] summarized data from 415 patients, reporting a proportion of 85.8% B-cell lymphomas.
Regarding histological subtypes, DLBCL and MALT lymphomas were the most common forms of PGIL[3,19,20]. DLBCL was the predominant subtype in our study, in line with previous studies[3,12,19]. In this retrospective study, the percentages of DLBCL in primary gastric lymphomas (86%), small bowel lymphomas (70%), and large bowel lymphomas (55.6%) were similar to those reported by Khuroo et al[12] (85%, 50%, and 75%, respectively). However, in the largest series reported by Nakamura et al[1], the most frequent histopathological subtype was MALT lymphoma (44%), followed by DLBCL (22%) and DLBCL plus MALT lymphoma (17%)[1]. Similarly, Ge et al[10] reported a higher proportion of MALT lymphoma than DLBCL in the stomach.
Numerous studies have investigated the prognostic factors associated with PGIL. Previous reports have shown that female gender, low-grade histology, good PS, and surgical resection are associated with improved OS, whereas age > 60 years, advanced stage, poor PS, and elevated LDH levels are linked to worse outcomes[9,14,15,17]. We performed single-factor analysis on 62 patients with PGIL, and on multivariate analysis, we showed that a poor performance status (ECOG ≥ 2) emerged as an independent prognostic factor for inferior OS. Similarly, Wang et al[9] identified high LDH levels, poor PS, advanced stage, an IPI score ≥ 3, conservative treatment, and high-grade histology as predictors of reduced OS.
Papaxoinis et al[7] also reported significantly better OS and EFS in patients diagnosed at earlier stages (I-II) with a 3-year EFS and OS rates of 83% and 87%, respectively, compared with 46% and 60% in patients with advanced-stage disease (P < 0.0001).
The multivariate analysis by Nakamura et al[1] showed that earlier stage, younger age, gastric localization, B-cell phenotype, and absence of B symptoms were independent prognostic factors for improved OS and EFS. In addition, other studies have shown that age over 60 years, elevated LDH levels, advanced stage, and poor PS are associated with poor prognosis[19,21-23]. In our study, a high IPI score was a prognostic factor for EFS but not for OS, consistent with the findings reported by Papaxoinis et al[7].
According to several studies, intestinal lymphomas differ significantly from gastric lymphomas in histopathology, clinical features, management, and prognosis[3,11,18]. Our results indicated a significant difference in LDH levels between intestinal and gastric lymphomas, but no significant differences in age, gender, cell phenotype, stage, PS, or IPI score were observed. Elevated LDH levels were more common in patients with gastric lymphomas than intestinal lymphomas (37.2% vs 10.5%). Wang et al[3] demonstrated statistically significant differences between intestinal and gastric lymphomas with respect to clinical stage, LDH level, and cell phenotype. They also reported that gastric lymphomas had significantly better prognosis than intestinal lymphomas, with 1-, 3-, and 5-year OS rates of 90.20%, 82.40%, and 80.40% for gastric lymphomas vs 90.60%, 79.20%, and 69.80% for intestinal lymphomas, respectively[3]. Similarly, Ge et al[10] found that gastric lymphomas had superior OS compared with intestinal lymphomas (1-year 89 vs 62%; 3-year 84 vs 50%, P = 0.03), and Song et al[24] reported comparable results. However, in the present study, no significant differences in OS or EFS rates were observed between the two groups.
Certain risk factors have been implicated in the pathogenesis of GIL, including H. pylori, human immunodeficiency virus, Campylobacter jejuni, Epstein-Barr virus, hepatitis B virus, human T-cell lymphotropic virus-1 infections, celiac disease, inflammatory bowel disease, and immunosuppression[2]. Infection with H. pylori appears to be a key causal factor in the development of MALT lymphomas[25]. Thus, the treatment of low-grade MALT lymphoma typically includes H. pylori eradication[9]. In the present study, 43 primary gastric lymphomas were evaluated for the presence of H. pylori infection which was detected in 48% of cases, and the presence of H. pylori was significantly associated with better OS (P < 0.0001). In contrast, the large Indian series by George and Lakshmanan[26] reported H. pylori positivity in only 10% of all GIL cases, including both MALT and DLBCL subtypes. This substantial difference may be partly explained by the variation in anatomic distribution between the studies-our cohort consisted predominantly of gastric lymphomas (69.3%), whereas the Indian series also included intestinal and colonic lymphomas where H. pylori-related pathogenesis is less relevant.
While Gao et al[27], who proposed the HLAMA prognostic model for gastrointestinal DLBCL, reported LDH level and age as significant prognostic indicators, LDH level did not remain an independent prognostic factor in our analysis. This model identified LDH level and age as key predictors and demonstrated superior risk stratification compared with the IPI score[27]. Our study, which included a broader spectrum of PGILs with a predominantly gastric distribution, also complements the intestine-restricted series by Chen et al[28]. Although both studies confirmed the adverse impact of elevated LDH and impaired clinical status at least in univariate analyses, Chen et al[28] found that Lugano stage was the only independent predictor of OS/PFS in primary intestinal DLBCL, whereas within our mixed PGIL cohort, ECOG ≥ 2 remained the sole independent prognostic factor for OS.
GI tract lymphoma is a heterogeneous disease, characterized by diverse clinicopathologic, demographic, and prognostic features. Each subtype demonstrates distinct characteristics and exhibits different clinical manifestations, treatment effects, and survival outcomes. The main limitations of this study are its retrospective design and relatively small sample size, from a single center. In this study, the stomach was the most frequent site, and DLBCL was the most common subtype. Poor PS emerged as an independent prognostic factor associated with inferior OS. Larger multicenter cohorts and randomized prospective studies are needed establish more detailed pathogenetic and prognostic information for the management of patients with PGIL. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
The authors would like to express their sincere gratitude to Professor Yuksel Kucukzeybek and Dr. Sidar Bagbudar for their valuable contributions to the statistical analysis of this study. In addition, the authors wish to formally acknowledge Dr. Eylul Gun for her professional assistance with the English language editing of this manuscript.
| 1. | Nakamura S, Matsumoto T, Iida M, Yao T, Tsuneyoshi M. Primary gastrointestinal lymphoma in Japan: a clinicopathologic analysis of 455 patients with special reference to its time trends. Cancer. 2003;97:2462-2473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 204] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 2. | Ghimire P, Wu GY, Zhu L. Primary gastrointestinal lymphoma. World J Gastroenterol. 2011;17:697-707. [PubMed] [DOI] [Full Text] |
| 3. | Wang W, Lin P, Yao H, Jia X, Sun J. Clinical analysis of Primary Gastrointestinal Non-Hodgkin's Lymphoma. Pak J Med Sci. 2017;33:1406-1411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Herrmann R, Panahon AM, Barcos MP, Walsh D, Stutzman L. Gastrointestinal involvement in non-Hodgkin's lymphoma. Cancer. 1980;46:215-222. [PubMed] [DOI] [Full Text] |
| 5. | Harris NL, Jaffe ES, Stein H, Banks PM, Chan JK, Cleary ML, Delsol G, De Wolf-Peeters C, Falini B, Gatter KC. A revised European-American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood. 1994;84:1361-1392. [PubMed] |
| 6. | Jaffe ES, Harris NL, Stein H, Vardiman JW. World Health Organization classification of tumours: Pathology and genetics of tumours of haematopoietic and lymphoid tissues. Lyon: IARC Press, 2001. |
| 7. | Papaxoinis G, Papageorgiou S, Rontogianni D, Kaloutsi V, Fountzilas G, Pavlidis N, Dimopoulos M, Tsatalas C, Xiros N, Economopoulos T. Primary gastrointestinal non-Hodgkin's lymphoma: a clinicopathologic study of 128 cases in Greece. A Hellenic Cooperative Oncology Group study (HeCOG). Leuk Lymphoma. 2006;47:2140-2146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 88] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 8. | Rohatiner A, d'Amore F, Coiffier B, Crowther D, Gospodarowicz M, Isaacson P, Lister TA, Norton A, Salem P, Shipp M. Report on a workshop convened to discuss the pathological and staging classifications of gastrointestinal tract lymphoma. Ann Oncol. 1994;5:397-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 428] [Cited by in RCA: 357] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 9. | Wang YG, Zhao LY, Liu CQ, Pan SC, Chen XL, Liu K, Zhang WH, Yang K, Chen XZ, Zhang B, Chen ZX, Chen JP, Zhou ZG, Hu JK. Clinical characteristics and prognostic factors of primary gastric lymphoma: A retrospective study with 165 cases. Medicine (Baltimore). 2016;95:e4250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 10. | Ge Z, Liu Z, Hu X. Anatomic distribution, clinical features, and survival data of 87 cases primary gastrointestinal lymphoma. World J Surg Oncol. 2016;14:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 11. | Zeggai S, Harir N, Tou A, Medjamia M, Guenaoui K. Gastrointestinal lymphoma in Western Algeria: pattern of distribution and histological subtypes (retrospective study). J Gastrointest Oncol. 2016;7:1011-1016. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Khuroo MS, Khwaja SF, Rather A, Hassan Z, Reshi R, Khuroo NS. Clinicopathological profile of gastrointestinal lymphomas in Kashmir. Indian J Med Paediatr Oncol. 2016;37:251-255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 13. | Soderquist CR, Bhagat G. Gastrointestinal T- and NK-cell lymphomas and indolent lymphoproliferative disorders. Semin Diagn Pathol. 2020;37:11-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 14. | Tarawneh MS. Non-Hodgkin's lymphomas in Jordanians: a histopathological study of 231 cases. Hematol Oncol. 1986;4:91-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Salem P, Anaissie E, Allam C, Geha S, Hashimi L, Ibrahim N, Jabbour J, Habboubi N, Khalyl M. Non-Hodgkin's lymphomas in the Middle East. A study of 417 patients with emphasis on special features. Cancer. 1986;58:1162-1166. [PubMed] [DOI] [Full Text] |
| 16. | Fujishima F, Katsushima H, Fukuhara N, Konosu-Fukaya S, Nakamura Y, Sasano H, Ichinohasama R. Incidence Rate, Subtype Frequency, and Occurrence Site of Malignant Lymphoma in the Gastrointestinal Tract: Population-Based Analysis in Miyagi, Japan. Tohoku J Exp Med. 2018;245:159-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Matysiak-Budnik T, Fabiani B, Hennequin C, Thieblemont C, Malamut G, Cadiot G, Bouché O, Ruskoné-Fourmestraux A. Gastrointestinal lymphomas: French Intergroup clinical practice recommendations for diagnosis, treatment and follow-up (SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO, SFH). Dig Liver Dis. 2018;50:124-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 18. | Chen Y, Chen Y, Chen S, Wu L, Xu L, Lian G, Yang K, Li Y, Zeng L, Huang K. Primary Gastrointestinal Lymphoma: A Retrospective Multicenter Clinical Study of 415 Cases in Chinese Province of Guangdong and a Systematic Review Containing 5075 Chinese Patients. Medicine (Baltimore). 2015;94:e2119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 19. | Koch P, del Valle F, Berdel WE, Willich NA, Reers B, Hiddemann W, Grothaus-Pinke B, Reinartz G, Brockmann J, Temmesfeld A, Schmitz R, Rübe C, Probst A, Jaenke G, Bodenstein H, Junker A, Pott C, Schultze J, Heinecke A, Parwaresch R, Tiemann M; German Multicenter Study Group. Primary gastrointestinal non-Hodgkin's lymphoma: I. Anatomic and histologic distribution, clinical features, and survival data of 371 patients registered in the German Multicenter Study GIT NHL 01/92. J Clin Oncol. 2001;19:3861-3873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 374] [Cited by in RCA: 267] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 20. | Dizengof V, Levi I, Etzion O, Fich A, Blum A, Chertok IRA, Ben-Yakov G, Rouvio O, Greenbaum U. Incidence rates and clinical characteristics of primary gastrointestinal non-Hodgkin lymphoma: a population study. Eur J Gastroenterol Hepatol. 2020;32:569-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 21. | Koch P, del Valle F, Berdel WE, Willich NA, Reers B, Hiddemann W, Grothaus-Pinke B, Reinartz G, Brockmann J, Temmesfeld A, Schmitz R, Rübe C, Probst A, Jaenke G, Bodenstein H, Junker A, Pott C, Schultze J, Heinecke A, Parwaresch R, Tiemann M; German Multicenter Study Group. Primary gastrointestinal non-Hodgkin's lymphoma: II. Combined surgical and conservative or conservative management only in localized gastric lymphoma--results of the prospective German Multicenter Study GIT NHL 01/92. J Clin Oncol. 2001;19:3874-3883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 135] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 22. | Yoon SS, Coit DG, Portlock CS, Karpeh MS. The diminishing role of surgery in the treatment of gastric lymphoma. Ann Surg. 2004;240:28-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 74] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 23. | Ding D, Pei W, Chen W, Zuo Y, Ren S. Analysis of clinical characteristics, diagnosis, treatment and prognosis of 46 patients with primary gastrointestinal non-Hodgkin lymphoma. Mol Clin Oncol. 2014;2:259-264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Song LN, Cen XN, Ou JP, Liang ZY, Qiu ZX, Wang WS, Xu WL, Li Y, Wang MJ, Dong YJ, Yin Y, Sun YH, Liu W, Wang Q, Wang LH, Wang Y, Ren HY. [Clinical and prognostic analysis of 101 cases of primary gastrointestinal non-Hodgkin's lymphoma]. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2013;21:387-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 25. | Olszewska-Szopa M, Wróbel T. Gastrointestinal non-Hodgkin lymphomas. Adv Clin Exp Med. 2019;28:1119-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 26. | George DM, Lakshmanan A. Lymphomas With Primary Gastrointestinal Presentation: A Retrospective Study Covering a Five-Year Period at a Quaternary Care Center in Southern India. Cureus. 2024;16:e75161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Gao F, Wang ZF, Tian L, Dong F, Wang J, Jing HM, Ke XY. A Prognostic Model of Gastrointestinal Diffuse Large B Cell Lymphoma. Med Sci Monit. 2021;27:e929898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | Chen X, Wang J, Liu Y, Lin S, Shen J, Yin Y, Wang Y. Primary intestinal diffuse large B-cell lymphoma: novel insights and clinical perception. Front Oncol. 2024;14:1404298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |