Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115505
Revised: November 18, 2025
Accepted: December 30, 2025
Published online: March 15, 2026
Processing time: 145 Days and 10.1 Hours
Disulfidptosis is a specific form of cell death that occurs in cells with high SL
To investigate whether YWJP promotes gastric cancer cell disulfidptosis via HAase/GLUT1 pathway by reversing cancer-associated fibroblasts (CAFs) se
CAFs were established using transforming growth factor-β1, and senCAFs were induced by H2O2. Senescence proportion was detected via β-galactosidase staining. ELISA and quantitative PCR measured interleukin-6 (IL-6), interleukin-8 (IL-8), hyaluronic acid, HAase, their mRNAs, NADP+/NADPH ratio, and disulfide bond levels. Western blot detected GLUT1 and disulfidptosis-related proteins. Pro
Based on previous research methods, we successfully established the CAFs and senCAFs models. The results showed that YWJP effectively reduced the proportion of senCAFs and simultaneously decreased the secretion of IL-6, IL-8, and HAase. Under HAase culture conditions, YWJP significantly downregulated the expression of GLUT1 and disulfidptosis-related proteins (CYFIP1, Rac1, ARP2, and ARP3), increased the disulfide bond level and the proportion of PI-positive cells in MKN45 cells, and thereby promoted disulfidptosis in MKN45 cells.
YWJP promotes disulfidptosis in MKN45 cells by reversing the proportion of senCAFs and reducing HAase secretion.
Core Tip: This study demonstrates that Yiqi Wenyang Jiedu prescription (YWJP) promotes disulfidptosis in gastric cancer MKN45 cells. The mechanism involves YWJP reversing the senescence of cancer-associated fibroblasts (established by transforming growth factor-β1/H2O2), thereby reducing the secretion of HAase. The decrease in HAase leads to the downregulation of GLUT1 and key disulfidptosis-related proteins (CYFIP1, Rac1, ARP2, ARP3). This process increases intracellular disulfide bond levels and cell death, confirming that YWJP induces disulfidptosis via the HAase/GLUT1 pathway.
- Citation: Kuang ZY, Qin XY, Wang XM, Song XT, Zhu XY, Li J. Yiqi Wenyang Jiedu prescription promotes disulfidptosis of gastric cancer by reversing senescent cancer-associated fibroblasts. World J Gastrointest Oncol 2026; 18(3): 115505
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/115505.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.115505
Gastric cancer ranks as the fifth most common malignant tumor worldwide and the third leading cause of cancer-related deaths[1]. Studies have shown that over 60% of gastric cancer patients experience recurrence and metastasis within two years after surgery[2]. Metastasis is the primary cause of death in patients with advanced gastric cancer, the five-year survival rate for patients with advanced gastric cancer who have developed distant metastases is only 10%[3].
Chemotherapy remains the cornerstone of gastric cancer treatment[4]. Platinum-containing dual-drug regimens are currently the first-line chemotherapy options. However, research has indicated that chemotherapy can induce senescence in the tumor microenvironment[5]. Cancer-associated fibroblasts (CAFs) are one of the major cell types in the tumor microenvironment, chemotherapy can also trigger senescence in CAFs. Senescent CAFs (senCAFs) secrete senescence-associated secretory phenotypes (SASP), which remodel the tumor microenvironment[6]. Currently known SASPs include interleukin-6 (IL-6), interleukin-8 (IL-8), transforming growth factor-β (TGF-β)[7], etc. Nevertheless, in our previous study, by establishing an in vitro senCAFs model, we found that senCAFs not only highly express SASPs such as IL-6 and IL-8 but also strongly express HAase (Supplementary material). HAase is a key component of SASP involved in matrix remodeling, and its main function is to degrade hyaluronic acid (HA) in the extracellular matrix[8]. A study demonstrated that HAase can decompose HA, induce the enrichment of GLUT1 on the cytoplasmic membrane of cancer cells, increase glucose uptake, upregulate glycolysis, and thereby accelerate cancer cell migration[9]. Therefore, the secretion of HAase by senescent CAFs, which promotes the expression of GLUT1 and thus upregulates glycolysis in gastric cancer cells, may be a crucial factor contributing to the high recurrence and metastasis rates of gastric cancer.
Disulfidptosis is a novel form of cell death, which mainly occurs in cancer cells with high SLC7A11 expression (e.g., gastric cancer, liver cancer). Under glucose starvation, the supply of NADPH is insufficient to support the process of reducing cystine to cysteine in cells, leading to disulfide stress, subsequent formation of abnormal disulfide bonds, and ultimately disulfidptosis in tumor cells[10-12]. GLUT1 is a key protein that regulates glucose homeostasis between the intracellular and extracellular environments. High expression of GLUT1 enables cancer cells to evade disulfidptosis; therefore, GLUT1 inhibitors are considered effective agents to promote disulfidptosis in tumor cells, and the development of GLUT1-targeted anti-tumor drugs represents a feasible direction for future research[13].
Yiqi Wenyang Jiedu Prescription (YWJP) is a Chinese herbal formula for gastric cancer treatment that has obtained a Chinese National Invention Patent. Previous explorations have confirmed its favorable clinical efficacy, and it has now entered the phase of double-blind, randomized controlled clinical trials[14,15]. Through drug mass spectrometry analysis and network pharmacology analysis, it was found that YWJP exerts regulatory effects on senescence-related targets (e.g., cell cycle) and matrix-related targets (Supplementary material). Pharmacological studies have shown that the active components of YWJP, such as quercetin, flavonoids, resveratrol, and curcumin, can reverse cellular senescence while inhibiting HAase activity and GLUT1 expression[16,17]. Therefore, the anti-gastric cancer effect of YWJP may be associated with the upregulation of disulfidptosis, which is consistent with the mechanism underlying disulfidptosis.
Based on the above, we hypothesize that YWJP can promote disulfidptosis in gastric cancer cells through the HAase/GLUT1 pathway by interfering with the senescence of CAFs and inhibiting the secretion of HAase.
Human embryonic lung cells (MRC-5 cells, Cat. No. CL-0161) and human gastric cancer cells (MKN45 cells, Cat. No. CL-0292) were purchased from Procell (Wuhan, China). These cells were cultured in DMEM supplemented with 100 mL/L fetal bovine serum (FBS) and maintained in an incubator with 50 mL/L CO2 at 37 °C under saturated humidity.
CAFs and senCAFs were established in our laboratory; Control group (MRC-5): Cells were cultured in DMEM with 100 mL/L FBS for 48 hours; CAFs group: Based on the control group, cells were treated with 10 ng/mL (TGF-β1, Pepro Tech, NJ, United States) in DMEM containing 100 mL/L FBS for 24 hours; senCAFs group: The induction process was initiated similarly to CAFs. After culturing with 10 ng/mL TGF-β1 in DMEM with 100 mL/L FBS for 24 hours, the original me
The culture medium of CAFs and senescent CAFs was discarded. After washing the cells twice with PBS, DMEM basal medium was added for 24 hours of culture. The medium from both groups was collected and filtered to obtain SASP.
Sprague-Dawley rats were acclimated for 3 days under specific pathogen-free conditions: Room temperature 20-26 °C, relative humidity 40%-70%, and a 12 hours light/12 hours dark cycle, with free access to food and water. On the 4th day, rats were weighed and randomly divided into two groups based on body weight; Blank serum (BS) group: Rats were intragastrically administered 2 mL of pure water twice daily for 3 consecutive days; YWJP-containing serum group: Rats were intragastrically administered 2 mL of YWJP twice daily for 3 consecutive days.
The clinical adult dose of YWJP is 108 g/d. The body weight conversion coefficient between rats and a 60 kg adult is 6.3. For a rat with an average body weight of 200 g, the equivalent dose was calculated as follows: 108 g/60 kg × 0.2 kg × 6.3 × 2 = 4.536 g/rat/d. After 6 administrations, rats were anesthetized by intraperitoneal injection of pentobarbital (40 mg/kg), and blood was collected from the abdominal aorta. Blood samples were allowed to stand at room temperature for 3 hours, then centrifuged at 3000 g for 15 minutes to extract the upper serum layer. The serum was filtered, inactivated in a water bath at 56 °C for 30 minutes, aliquoted, and stored at -80 °C.
Cells for immunofluorescence staining were fixed with 40 mL/L tissue/cell fixative (Servicebio, Wuhan, China) at room temperature for 15 min, then permeabilized with 5 mL/L Triton X-100. After blocking in PBS containing 150 mL/L goat serum (Zsbio, Beijing, China) for 1 hour, primary antibodies against fibroblast activation protein (FAP) and α-smooth muscle actin (α-SMA) were diluted to 1:100 with blocking solution, and cells were incubated overnight at 4 °C. Following a series of washes, cells were exposed to fluorescently labeled secondary antibodies (1:500) and incubated for 1 hour in the dark, then washed again. Cells were mounted with DAPI-containing mounting medium and examined under a Zeiss Instruments confocal microscope.
Senescent CAFs were fixed with 40 mL/L paraformaldehyde at room temperature for 5 min and washed 3 times with PBS to remove the fixative. A Cellular Senescence β-Galactosidase Staining Kit (Beyotime, Shanghai, China) was used to prepare the staining solution according to the manufacturer’s instructions. 1 mL of staining solution was added to each well of a 6-well plate, and cells were incubated at 37 °C in the dark for 24 hours. After washing twice with PBS to remove residual dye, the proportion of senescent cells was observed under an optical microscope with 10 × to 20 × objectives.
Cell culture medium from each group was centrifuged at 1000 g at 4 °C for 20 minutes to remove impurities and cell debris, and the supernatant was collected. The following kits were used: Human IL-6 ELISA kit, Human IL-8 ELISA kit, Human HAase ELISA kit, and NADP+/NADPH assay kit (WST-8 method) (Elabscience, Wuhan, China); Human HA ELISA Kit (Boyan Biology, Nanjing, China); protein disulfide bond content assay kit (Servicebio, Wuhan, China). Levels of IL-6, IL-8, HA, HAase, and NADP+/NADPH in SASP, as well as protein disulfide bond content in the supernatant of each group, were measured according to the manufacturers’ instructions.
Human gastric cancer cells (MKN45 cell line) were seeded into culture plates and cultured until reaching 90% confluence. Linear wounds were created using a sterile pipette tip. Cells were then washed to remove debris and cultured in SASP. Changes in wound width were observed under an inverted optical microscope, and images were recorded at 0 hour, 24 hours, and 48 hours after wound creation.
MKN45 cells were adjusted to a concentration of 100 cells/mL, and 2 mL of the cell suspension was added to each well of a 6-well plate. Plates were incubated in a 37 °C incubator with 50 mL/L CO2. When visible cell colonies formed, the medium was discarded. After washing, cells were fixed with 40 mL/L paraformaldehyde at room temperature for 15 minutes, then stained with 1 mL/L crystal violet. Colonies were photographed under an inverted microscope, and colony area or number was analyzed using software such as Image J.
Total protein was extracted from cells using RIPA lysis buffer containing protease and phosphatase inhibitors. Protein concentration was determined using a BCA Protein Quantification Kit (Applygen, Beijing, China). Equal amounts of protein were separated by Tris-Glycine electrophoresis with 200 mL/L methanol, then transferred to a PVDF membrane (Immobilon). The membrane was blocked with protein-free rapid blocking solution at room temperature for 1 hour, then incubated with primary antibodies overnight at 4 °C. After washing, the membrane was incubated with HRP-conjugated goat anti-rabbit secondary antibody at room temperature for 1 hour. Detection was performed using an enhanced chemiluminescence kit, and protein bands were visualized with a molecular imager. The gray values of target proteins and internal reference proteins were quantified using software such as ImageJ to calculate relative expression levels.
Total RNA was extracted using TRIzol reagent (Qiagen, Hilden, Germany). RNA was reverse-transcribed into cDNA using a reverse transcription kit (Qiagen), and cDNA was amplified using SYBR Green PCR Master Mix (Thermo Fisher Scientific, MA, United States). PCR amplification was performed on a Step One Plus Real-Time PCR System (Applied Biosystems, United States). Gene expression levels were normalized using GAPDH as the internal reference gene.
Experimental data were analyzed using SPSS 27.0. All data are presented as mean ± SD. Independent samples t-test was used for statistical analysis of data from two groups. One-way analysis of variance (ANOVA) and multiple comparison tests were used for comparisons among multiple groups. Bar graphs were generated using GraphPad Prism 8.0.2. The P < 0.05 was considered statistically significant.
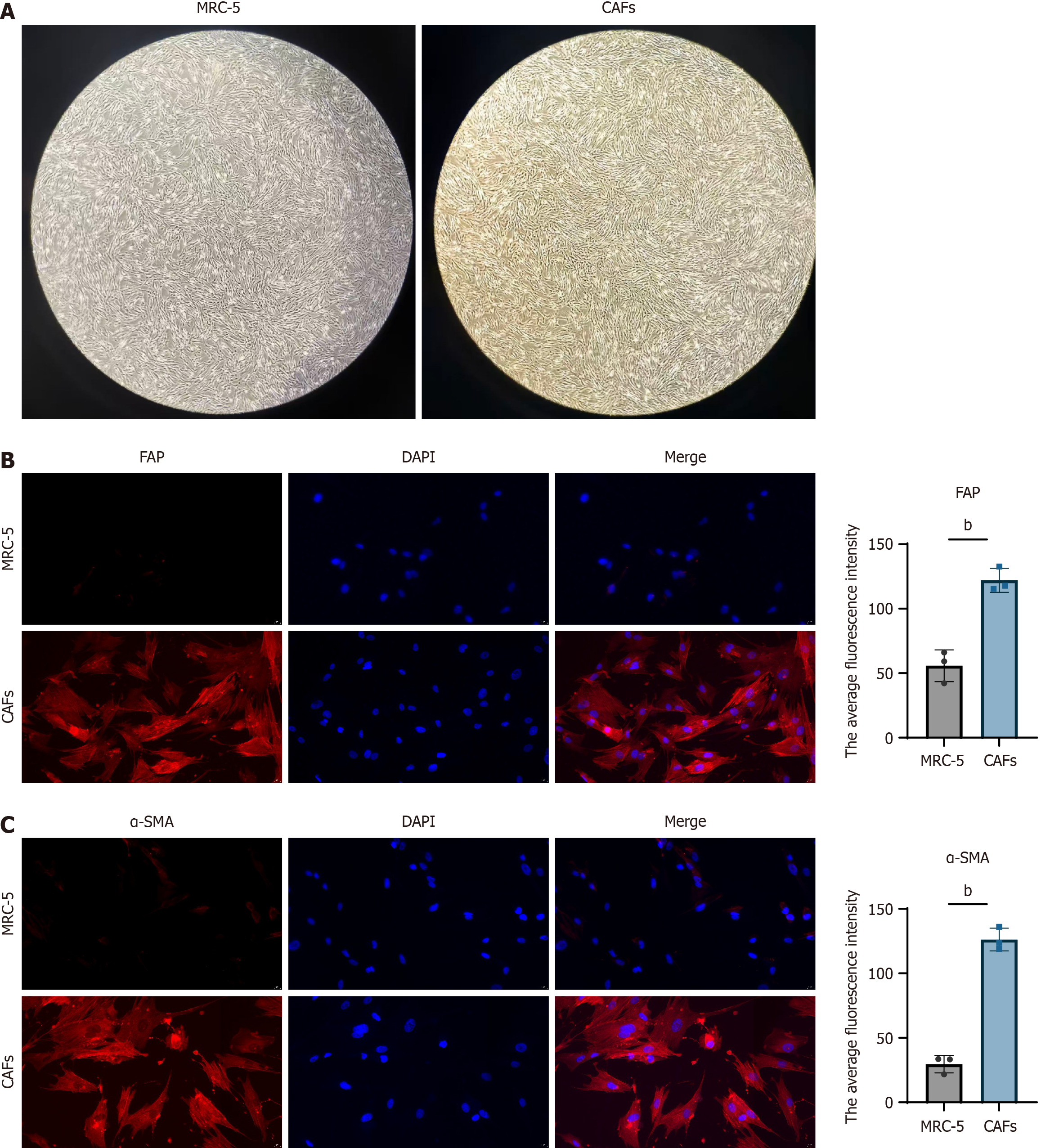
MRC-5 cells grew in an adherent manner and exhibited a spindle-shaped, fibroblast-like morphology. The CAFs model was established by culturing MRC-5 cells in complete medium containing 10 ng/mL TGF-β1 for 24 hours. Results showed that the growth rate of CAFs was faster than that of MRC-5 cells; however, no significant morphological difference was observed between the two groups (Figure 1A). Immunofluorescence analysis revealed that the expression levels of FAP and α-SMA in CAFs were significantly higher than those in MRC-5 cells. This indicates that TGF-β1 induction can cause MRC-5 cells to express CAFs-specific markers, which is consistent with the characteristics of CAFs (Figure 1B and C).
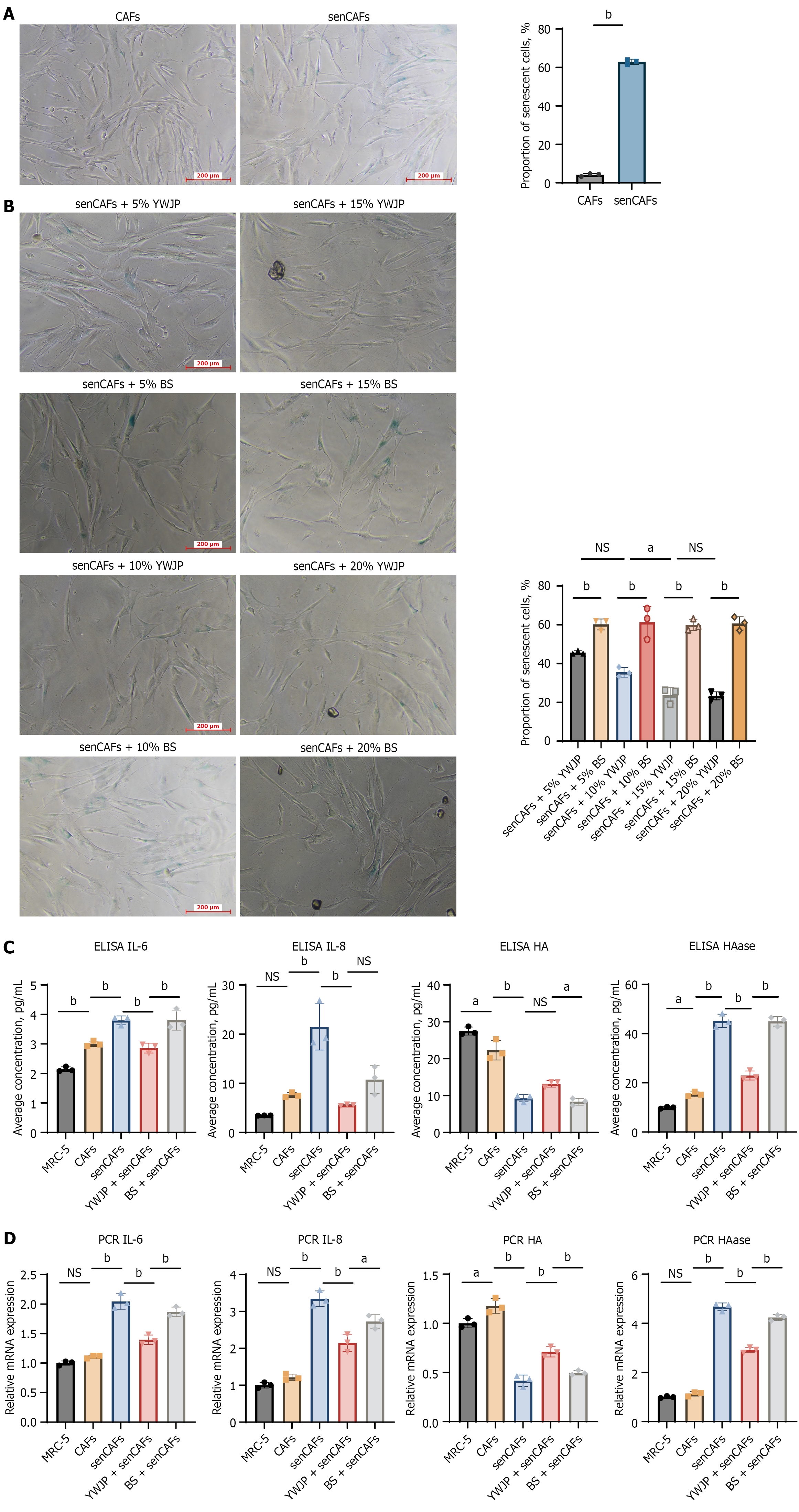
CAFs were treated with 60 μM H2O2 to establish a chemotherapy-induced senCAFs model. β-galactosidase staining showed that compared with the CAFs group, more cells in the CAFs group treated with H2O2 for 24 hours entered a senescent state, with approximately 60% of cells becoming senescent (cells stained blue-green in the figure are senescent cells), which is consistent with the characteristics of senescent CAFs (Figure 2A).
β-galactosidase staining revealed that YWJP-containing serum at 4 concentration gradients (50 mL/L, 100 mL/L, 150 mL/L, and 200 mL/L) all reduced the senescence ratio of CAFs to varying degrees compared with BS of the same proportion. Among them, the 150 mL/L YWJP-containing serum group and 200 mL/L YWJP-containing serum group had the lowest proportion of senescent cells, and there was no statistically significant difference in the proportion of senescent cells between the two groups (Figure 2B). Therefore, 150 mL/L YWJP-containing serum was used in subse
ELISA was used to detect the levels of senescence-associated phenotypes IL-6 and IL-8 in different groups. The results showed that the levels of IL-6 and IL-8 in the senCAFs group were significantly higher than those in the CAFs group; the levels of IL-6 and IL-8 in the YWJP + senCAFs group were significantly lower than those in the senCAFs group; meanwhile, compared with BS, the YWJP-containing serum group had significantly lower levels of IL-6 and IL-8 (Figure 2C). The results of quantitative PCR (qPCR) also confirmed the above conclusions (Figure 2D).
ELISA was used to detect the levels of HA and HAase in different groups. The results showed that HAase levels in the senCAFs group were significantly higher than those in the CAFs group, while HA levels were significantly lower than those in the CAFs group; the YWJP + senCAFs group had significantly lower HAase levels and higher HA content than the senCAFs group; compared with BS, the YWJP-containing serum group had significantly lower HAase levels, while the opposite was true for HA levels (Figure 2C). The results of qPCR also confirmed the above conclusions (Figure 2D). The above results support our previous finding that senCAFs secrete more HAase than CAFs, and YWJP can effectively reduce HAase secretion by senCAFs.
In summary, we confirmed that senCAFs can secrete not only more IL-6 and IL-8 but also HAase related to matrix remodeling compared with CAFs. YWJP can effectively inhibit the expression of β-galactosidase in senCAFs, and simultaneously suppress the secretion of IL-6, IL-8, and HAase by senCAFs, and this effect is not caused by the serum itself.
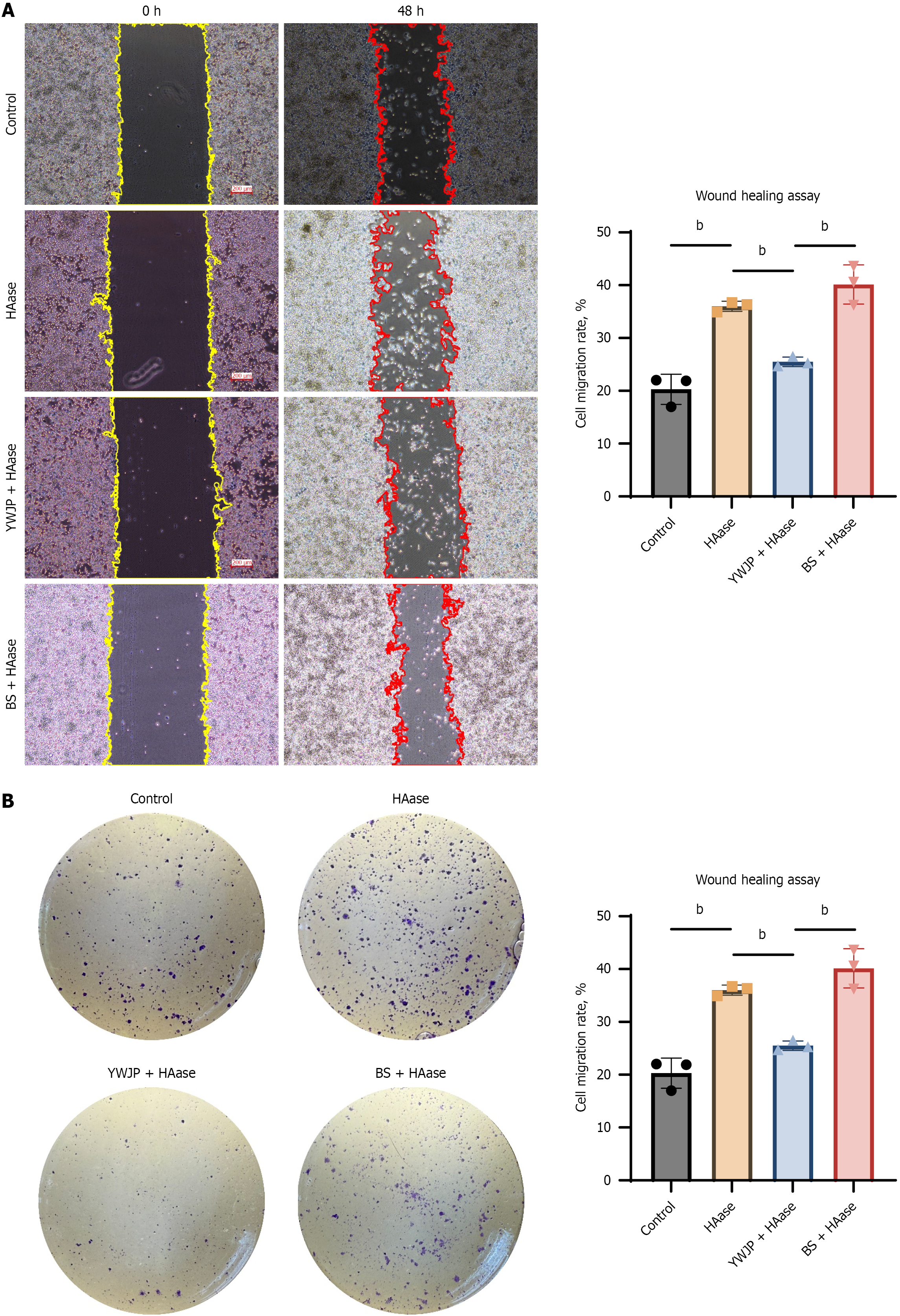
Based on the conclusions in ELISA, MKN45 cells were treated with 45 pg/mL HAase. Results of the wound healing assay showed that the 48 hours migration rate and colony-forming ability of cells in the HAase group were significantly higher than those in the control group, indicating that HAase can promote the migration and proliferation of MKN45 cells (Figure 3). YWJP could effectively reduce the HAase-induced increase in migration rate and colony-forming ability of MKN45 cells, thereby inhibiting the malignant behaviors of MKN45 cells.
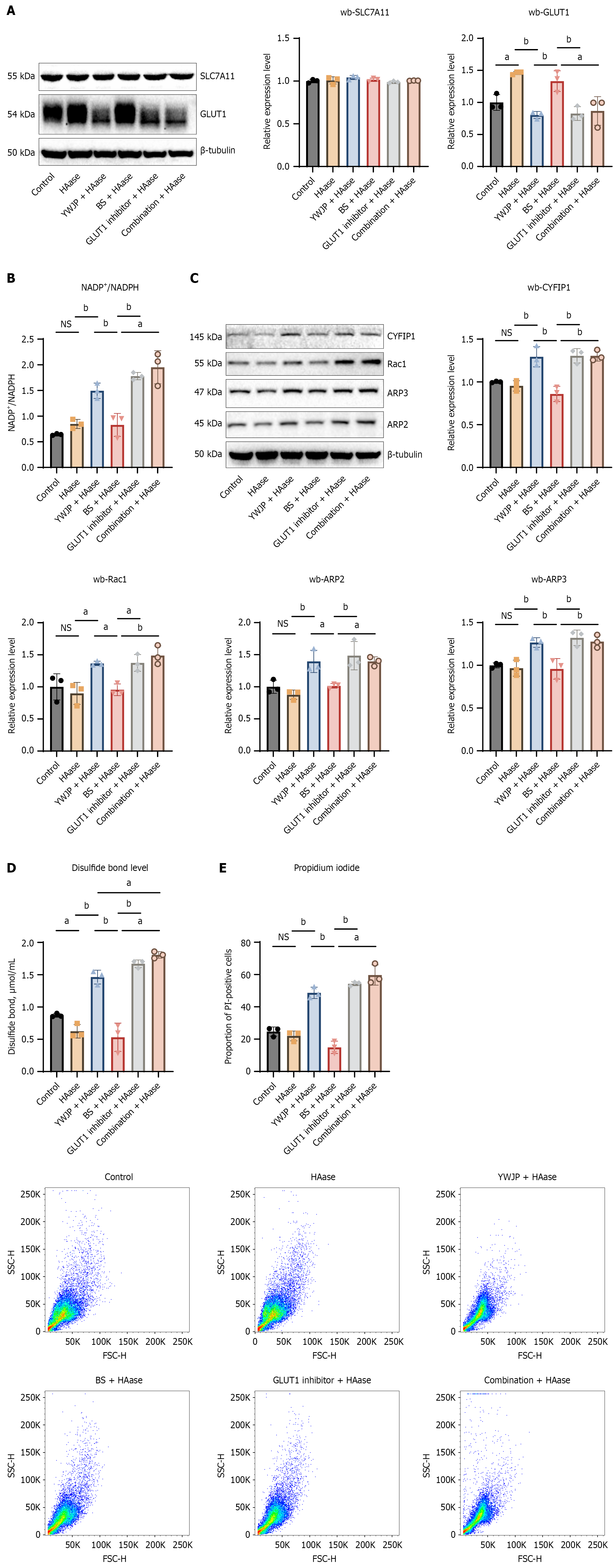
To further explore the mechanism by which YWJP interferes with the malignant behaviors of MKN45 cells under HAase action, combined with the results of previous mass spectrometry analysis, WB was used to detect the expression of disulfidptosis-related key protein SLC7A11 and GLUT1. The results showed that there was no significant difference in the expression level of SLC7A11 among all groups, indicating that YWJP does not act on SLC7A11 but instead exerts its effects upstream of GLUT1. Compared with the Control group, the expression of GLUT1 in the HAase group was significantly upregulated; the expression of GLUT1 in the YWJP + HAase group was significantly lower than that in the HAase group and BS + HAase group, suggesting that YWJP significantly reversed HAase-mediated GLUT1 upregulation; there was no statistically significant difference between the YWJP + HAase group and the GLUT1 Inhibitor + HAase group (Figure 4A).
The NADP+/NADPH ratio was detected by ELISA. The results showed that compared with the HAase group and BS + HAase group, the NADP+/NADPH ratio in the YWJP + HAase group was higher, indicating that YWJP promoted the depletion of NADPH by downregulating the expression of GLUT1. Meanwhile, there was no statistically significant difference in the NADP+/NADPH ratio between the YWJP + HAase group and the GLUT1 Inhibitor + HAase group (Figure 4B).
Further studies were conducted on the expression of proteins related to the Rac1-CYFIP1-ARP2/3 axis downstream of GLUT1. Consistent with the expression pattern of GLUT1, YWJP also significantly reversed the upregulation of Rac1, CYFIP1, ARP2, and ARP3 protein expression. There was no statistically significant difference between the YWJP + HAase group and the GLUT1 Inhibitor + HAase group (Figure 4C).
We further detected the disulfide bond levels in each group using ELISA. The results showed that the disulfide bond level in the YWJP + HAase group was significantly higher than that in the HAase group and BS + HAase group; there was no statistically significant difference between the YWJP + HAase group and the GLUT1 Inhibitor + HAase group (Figure 4D). Similarly, PI staining showed that the proportion of dead cells in the YWJP + HAase group was significantly higher than that in the SASP group and BS + HAase group. There was no statistically significant difference between the YWJP + HAase group and the GLUT1 Inhibitor + HAase group (Figure 4E).
This study aimed to elucidate the novel molecular mechanism underlying the anti-gastric cancer effect of YWJP, with a specific focus on whether it influences the disulfidptosis process of MKN45 cells by regulating the functional state of in the tumor microenvironment. The results demonstrated that YWJP can effectively reverse the senescence of senCAFs, inhibit the secretion of their key effector molecule HAase, and thereby weaken the resistance of MKN45 cells to disulfidptosis through this pathway. This finding links the therapeutic effects of traditional Chinese medicine (TCM) compound prescriptions, the regulation of the tumor microenvironment, and cutting-edge cell death mechanisms, providing a new theoretical basis for gastric cancer treatment strategies.
The core role of the tumor microenvironment in tumor progression has been widely recognized[18-21]; this study further investigated the characteristics of senCAFs and their secreted SASP. Previous studies have demonstrated that CAFs can respond to TGF-β1 regulation, exhibiting a high capacity for HAase production[22,23]. Within SASP, increased TGF-β1 secretion further promotes the secretion of HAase. Currently, there are no specific studies on tumor diseases for this mechanism. This study confirmed that senCAFs not only secrete the inflammatory factors IL-6 and IL-8 but also upregulate HAase secretion. HAase can degrade HA on the cell surface, upregulate the expression of GLUT1, and promote glucose uptake by tumor cells.
Disulfidptosis, a novel form of cancer cell death discovered in 2023, mainly occurs in cancer cells with high SLC7A11 expression[24]. Tumor cells with high SLC7A11 expression have a high demand for NADPH and are strictly dependent on glucose uptake and NADPH supply[25-27]. This dependence makes them susceptible to disulfide stress when glucose uptake is insufficient, leading to the formation of abnormal intracellular disulfide bonds, cytoskeletal contraction, and ultimately disulfidptosis[28,29]. This study found that HAase derived from senCAFs can significantly enhance NADPH supply capacity by upregulating GLUT1 expression in MKN45 cells. This allows MKN45 cells to maintain sufficient NADPH levels to reduce abnormally accumulated disulfide bonds, thereby evading disulfidptosis. Mechanistically, this explains why MKN45 cells with high SLC7A11 expression exhibit higher malignancy in an HAase-rich microenvironment compared to those in regular culture medium.
The core finding of this study is the confirmation that the target of YWJP is not simply MKN45 cells; its active com
The innovation of this study lies in being the first to link the mechanism of action of the traditional TCM compound YWJP in intervening gastric cancer to the cutting-edge concept of disulfidptosis, and clearly defining its effect as the regulation of senCAFs in the tumor microenvironment. However, this study still has limitations. First, as a TCM compound, YWJP has a complex composition; the specific active components and related targets involved in reversing senCAFs require further exploration. Second, the current conclusions are based on in vitro experiments; the effectiveness of YWJP in the complex physiological environment of living animals and whether it exerts synergistic effects such as immunomodulation need to be further verified using in vivo models. Additionally, the conclusions were only drawn using the MKN45 cell line; whether this mechanism is universally applicable to gastric cancer cells with other genetic backgrounds also requires further investigation.
In summary, this study reveals that YWJP promotes disulfidptosis in gastric cancer cells by reversing the proportion of senCAFs, inhibiting their HAase secretion, reducing glucose supply, inducing NADPH depletion and disulfide stress.
| 1. | Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Cooke D, Corvera C, Das P, Enzinger PC, Enzler T, Fanta P, Farjah F, Gerdes H, Gibson MK, Hochwald S, Hofstetter WL, Ilson DH, Keswani RN, Kim S, Kleinberg LR, Klempner SJ, Lacy J, Ly QP, Matkowskyj KA, McNamara M, Mulcahy MF, Outlaw D, Park H, Perry KA, Pimiento J, Poultsides GA, Reznik S, Roses RE, Strong VE, Su S, Wang HL, Wiesner G, Willett CG, Yakoub D, Yoon H, McMillian N, Pluchino LA. Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022;20:167-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1229] [Article Influence: 307.3] [Reference Citation Analysis (5)] |
| 2. | Plotti F, Terranova C, Luvero D, Bartolone M, Messina G, Feole L, Cianci S, Scaletta G, Marchetti C, Di Donato V, Fagotti A, Scambia G, Benedetti Panici P, Angioli R. Diet and Chemotherapy: The Effects of Fasting and Ketogenic Diet on Cancer Treatment. Chemotherapy. 2020;65:77-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 3. | Li Y, Feng A, Zheng S, Chen C, Lyu J. Recent Estimates and Predictions of 5-Year Survival in Patients with Gastric Cancer: A Model-Based Period Analysis. Cancer Control. 2022;29:10732748221099227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 112] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 4. | Xie BW, Zang L, Ma JJ, Sun J, Yang X, Wang ML, Lu AG, Hu WG, Zheng MH. [Safety and Efficacy of Oxaliplatin Combined with Capecitabine or S-Aracabine in Neoadjuvant Chemotherapy for Advanced Gastric Cancer]. Zhonghua Weichang Waike Zazhi. 2021;2:138-144. [RCA] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 5. | Chibaya L, Snyder J, Ruscetti M. Senescence and the tumor-immune landscape: Implications for cancer immunotherapy. Semin Cancer Biol. 2022;86:827-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 107] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 6. | Mavrogonatou E, Pratsinis H, Kletsas D. The role of senescence in cancer development. Semin Cancer Biol. 2020;62:182-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 7. | Prattichizzo F, De Nigris V, La Sala L, Procopio AD, Olivieri F, Ceriello A. "Inflammaging" as a Druggable Target: A Senescence-Associated Secretory Phenotype-Centered View of Type 2 Diabetes. Oxid Med Cell Longev. 2016;2016:1810327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 97] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 8. | Han X, Lei Q, Xie J, Liu H, Li J, Zhang X, Zhang T, Gou X. Potential Regulators of the Senescence-Associated Secretory Phenotype During Senescence and Aging. J Gerontol A Biol Sci Med Sci. 2022;77:2207-2218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 9. | Sullivan WJ, Mullen PJ, Schmid EW, Flores A, Momcilovic M, Sharpley MS, Jelinek D, Whiteley AE, Maxwell MB, Wilde BR, Banerjee U, Coller HA, Shackelford DB, Braas D, Ayer DE, de Aguiar Vallim TQ, Lowry WE, Christofk HR. Extracellular Matrix Remodeling Regulates Glucose Metabolism through TXNIP Destabilization. Cell. 2018;175:117-132.e21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 215] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 10. | Chen J, Ma B, Yang Y, Wang B, Hao J, Zhou X. Disulfidptosis decoded: a journey through cell death mysteries, regulatory networks, disease paradigms and future directions. Biomark Res. 2024;12:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 11. | Li T, Song Y, Wei L, Song X, Duan R. Disulfidptosis: a novel cell death modality induced by actin cytoskeleton collapse and a promising target for cancer therapeutics. Cell Commun Signal. 2024;22:491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 12. | Li X, Xu J, Yan L, Tang S, Zhang Y, Shi M, Liu P. Targeting Disulfidptosis with Potentially Bioactive Natural Products in Metabolic Cancer Therapy. Metabolites. 2024;14:604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 13. | Liu X, Nie L, Zhang Y, Yan Y, Wang C, Colic M, Olszewski K, Horbath A, Chen X, Lei G, Mao C, Wu S, Zhuang L, Poyurovsky MV, James You M, Hart T, Billadeau DD, Chen J, Gan B. Actin cytoskeleton vulnerability to disulfide stress mediates disulfidptosis. Nat Cell Biol. 2023;25:404-414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 940] [Cited by in RCA: 807] [Article Influence: 269.0] [Reference Citation Analysis (0)] |
| 14. | Cao L, Zhu G, Wang X, Kuang Z, Song X, Ma X, Zhu X, Gao R, Li J. Yiqi Wenyang Jiedu prescription for preventing and treating postoperative recurrence and metastasis of gastric cancer: a randomized controlled trial protocol. Front Oncol. 2024;14:1326970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 15. | Cao L, Wang X, Ma X, Zhu G, Li J. Exploration of Constitutional Bias and Associated Factors in Patients with Recurrent and Metastatic Gastric Cancer, and the Therapeutic Effect of Yiqi Wenyang Jiedu Prescription: A one-arm study. Integr Cancer Ther. 2025;24:15347354251331100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H, Giorgadze N, Palmer AK, Ikeno Y, Hubbard GB, Lenburg M, O'Hara SP, LaRusso NF, Miller JD, Roos CM, Verzosa GC, LeBrasseur NK, Wren JD, Farr JN, Khosla S, Stout MB, McGowan SJ, Fuhrmann-Stroissnigg H, Gurkar AU, Zhao J, Colangelo D, Dorronsoro A, Ling YY, Barghouthy AS, Navarro DC, Sano T, Robbins PD, Niedernhofer LJ, Kirkland JL. The Achilles' heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14:644-658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2150] [Cited by in RCA: 1890] [Article Influence: 171.8] [Reference Citation Analysis (1)] |
| 17. | Shriwas P, Chen X, Kinghorn AD, Ren Y. Plant-derived glucose transport inhibitors with potential antitumor activity. Phytother Res. 2020;34:1027-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Mbeunkui F, Johann DJ Jr. Cancer and the tumor microenvironment: a review of an essential relationship. Cancer Chemother Pharmacol. 2009;63:571-582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 393] [Cited by in RCA: 360] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 19. | Ugai T, Yao Q, Ugai S, Ogino S. Advancing precision oncology: Insights into the tumor microenvironment and immunotherapy outcomes. Innovation (Camb). 2024;5:100656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Asadi M, Zafari V, Sadeghi-Mohammadi S, Shanehbandi D, Mert U, Soleimani Z, Caner A, Zarredar H. The role of tumor microenvironment and self-organization in cancer progression: Key insights for therapeutic development. Bioimpacts. 2025;15:30713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 21. | Li Z, Li J, Bai X, Huang X, Wang Q. Tumor microenvironment as a complex milieu driving cancer progression: a mini review. Clin Transl Oncol. 2025;27:1943-1952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 66] [Article Influence: 66.0] [Reference Citation Analysis (1)] |
| 22. | Sapudom J, Müller CD, Nguyen KT, Martin S, Anderegg U, Pompe T. Matrix Remodeling and Hyaluronan Production by Myofibroblasts and Cancer-Associated Fibroblasts in 3D Collagen Matrices. Gels. 2020;6:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 23. | Kabir TD, Leigh RJ, Tasena H, Mellone M, Coletta RD, Parkinson EK, Prime SS, Thomas GJ, Paterson IC, Zhou D, McCall J, Speight PM, Lambert DW. A miR-335/COX-2/PTEN axis regulates the secretory phenotype of senescent cancer-associated fibroblasts. Aging (Albany NY). 2016;8:1608-1635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 67] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 24. | Mao C, Wang M, Zhuang L, Gan B. Metabolic cell death in cancer: ferroptosis, cuproptosis, disulfidptosis, and beyond. Protein Cell. 2024;15:642-660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 135] [Article Influence: 67.5] [Reference Citation Analysis (0)] |
| 25. | Liu X, Olszewski K, Zhang Y, Lim EW, Shi J, Zhang X, Zhang J, Lee H, Koppula P, Lei G, Zhuang L, You MJ, Fang B, Li W, Metallo CM, Poyurovsky MV, Gan B. Cystine transporter regulation of pentose phosphate pathway dependency and disulfide stress exposes a targetable metabolic vulnerability in cancer. Nat Cell Biol. 2020;22:476-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 499] [Cited by in RCA: 439] [Article Influence: 73.2] [Reference Citation Analysis (0)] |
| 26. | Jyotsana N, Ta KT, DelGiorno KE. The Role of Cystine/Glutamate Antiporter SLC7A11/xCT in the Pathophysiology of Cancer. Front Oncol. 2022;12:858462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 188] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 27. | Liu X, Gan B. Glucose starvation induces NADPH collapse and disulfide stress in SLC7A11(high) cancer cells. Oncotarget. 2021;12:1629-1630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 28. | Liu X, Zhang Y, Zhuang L, Olszewski K, Gan B. NADPH debt drives redox bankruptcy: SLC7A11/xCT-mediated cystine uptake as a double-edged sword in cellular redox regulation. Genes Dis. 2021;8:731-745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 29. | Yan Y, Teng H, Hang Q, Kondiparthi L, Lei G, Horbath A, Liu X, Mao C, Wu S, Zhuang L, James You M, Poyurovsky MV, Ma L, Olszewski K, Gan B. SLC7A11 expression level dictates differential responses to oxidative stress in cancer cells. Nat Commun. 2023;14:3673. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 245] [Article Influence: 81.7] [Reference Citation Analysis (0)] |
| 30. | Zoico E, Nori N, Darra E, Tebon M, Rizzatti V, Policastro G, De Caro A, Rossi AP, Fantin F, Zamboni M. Senolytic effects of quercetin in an in vitro model of pre-adipocytes and adipocytes induced senescence. Sci Rep. 2021;11:23237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 31. | Ivan A, Lukinich-Gruia AT, Cristea IM, Pricop MA, Calma CL, Simina AG, Tatu CA, Galuscan A, Păunescu V. Quercetin and Mesenchymal Stem Cell Metabolism: A Comparative Analysis of Young and Senescent States. Molecules. 2024;29:5755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 32. | Onodera Y, Teramura T, Takehara T, Obora K, Mori T, Fukuda K. miR-155 induces ROS generation through downregulation of antioxidation-related genes in mesenchymal stem cells. Aging Cell. 2017;16:1369-1380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 59] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 33. | Yanai H, Shteinberg A, Porat Z, Budovsky A, Braiman A, Zeische R, Fraifeld VE. Cellular senescence-like features of lung fibroblasts derived from idiopathic pulmonary fibrosis patients. Aging (Albany NY). 2015;7:664-672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 149] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 34. | Lim H, Park H, Kim HP. Effects of flavonoids on senescence-associated secretory phenotype formation from bleomycin-induced senescence in BJ fibroblasts. Biochem Pharmacol. 2015;96:337-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 164] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 35. | Sellarés J, Rojas M. Quercetin in Idiopathic Pulmonary Fibrosis: Another Brick in the Senolytic Wall. Am J Respir Cell Mol Biol. 2019;60:3-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 36. | He J, Zhang A, Song Z, Guo S, Chen Y, Liu Z, Zhang J, Xu X, Liu J, Chu L. The resistant effect of SIRT1 in oxidative stress-induced senescence of rat nucleus pulposus cell is regulated by Akt-FoxO1 pathway. Biosci Rep. 2019;39:BSR20190112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 37. | Hwang JW, Yao H, Caito S, Sundar IK, Rahman I. Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic Biol Med. 2013;61:95-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 426] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 38. | Guo Q, Zhang H, Zhang B, Zhang E, Wu Y. Tumor Necrosis Factor-alpha (TNF-α) Enhances miR-155-Mediated Endothelial Senescence by Targeting Sirtuin1 (SIRT1). Med Sci Monit. 2019;25:8820-8835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 39. | Zeng HJ, Ma J, Yang R, Jing Y, Qu LB. Molecular Interactions of Flavonoids to Hyaluronidase: Insights from Spectroscopic and Molecular Modeling Studies. J Fluoresc. 2015;25:941-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 40. | Zambrano A, Molt M, Uribe E, Salas M. Glut 1 in Cancer Cells and the Inhibitory Action of Resveratrol as A Potential Therapeutic Strategy. Int J Mol Sci. 2019;20:3374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 157] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 41. | Jung KH, Lee JH, Thien Quach CH, Paik JY, Oh H, Park JW, Lee EJ, Moon SH, Lee KH. Resveratrol suppresses cancer cell glucose uptake by targeting reactive oxygen species-mediated hypoxia-inducible factor-1α activation. J Nucl Med. 2013;54:2161-2167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 95] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 42. | Thulasiraman P, Foret M, Pandit R, McAlister K. Curcumin Inhibits the PPARδ-p-Akt-GLUT1 Pathway and Ameliorates the Antiproliferative Effects of Doxorubicin in MDA-MB-231 Cells. Asian Pac J Cancer Prev. 2024;25:1035-1043. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 43. | Siddiqui FA, Prakasam G, Chattopadhyay S, Rehman AU, Padder RA, Ansari MA, Irshad R, Mangalhara K, Bamezai RNK, Husain M, Ali SM, Iqbal MA. Curcumin decreases Warburg effect in cancer cells by down-regulating pyruvate kinase M2 via mTOR-HIF1α inhibition. Sci Rep. 2018;8:8323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 109] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 44. | Dai LB, Zhong JT, Shen LF, Zhou SH, Lu ZJ, Bao YY, Fan J. Radiosensitizing effects of curcumin alone or combined with GLUT1 siRNA on laryngeal carcinoma cells through AMPK pathway-induced autophagy. J Cell Mol Med. 2021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |