Published online Jun 15, 2024. doi: 10.4251/wjgo.v16.i6.2380
Revised: January 19, 2024
Accepted: April 3, 2024
Published online: June 15, 2024
Processing time: 179 Days and 23.1 Hours
Hepatic artery infusion chemotherapy (HAIC) has good clinical efficacy in the treatment of advanced hepatocellular carcinoma (HCC); however, its efficacy varies. This review summarized the ability of various markers to predict the efficacy of HAIC and provided a reference for clinical applications. As of October 25, 2023, 51 articles have been retrieved based on keyword predictions and HAIC. Sixteen eligible articles were selected for inclusion in this study. Comprehensive literature analysis found that methods used to predict the efficacy of HAIC in
Core Tip: Hepatic artery infusion chemotherapy (HAIC) has good clinical efficacy and high safety, and it has become one of the main treatment options for patients with intermediate to advanced hepatocellular carcinoma (HCC). Through predicting the prognosis of HAIC, appropriate patients can be screened for HAIC, and the overall efficacy of HAIC in HCC patients can improve. This review summarized the strategies currently used to predict the efficacy of HAIC in middle and advanced HCC, analyzed each marker's ability to predict HAIC efficacy, and provided a reference for the clinical selection of an appropriate HAIC prediction modality.
- Citation: Wang QF, Li ZW, Zhou HF, Zhu KZ, Wang YJ, Wang YQ, Zhang YW. Predicting the prognosis of hepatic arterial infusion chemotherapy in hepatocellular carcinoma. World J Gastrointest Oncol 2024; 16(6): 2380-2393
- URL: https://www.wjgnet.com/1948-5204/full/v16/i6/2380.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v16.i6.2380
Primary liver cancer is the sixth most common cancer worldwide and the third leading cause of cancer-related deaths. Hepatocellular carcinoma (HCC) accounts for 75%-85% of all primary liver cancer cases[1-4]. With recent advances in the treatment of HCC, patient survival can be improved using effective methods including surgical resection, radiofrequency ablation, microwave ablation, hepatic artery chemoembolization (TACE), hepatic artery infusion chemotherapy (HAIC), multimolecular targeted agents, immunotherapy, and liver transplantation[5,6]. However, due to the nonspecific symptoms of liver cancer, most patients are already in the middle and late stages when diagnosed. Therefore, TACE, HAIC, targeting, and immunization have become the main treatments for middle- and late-stage liver cancers[7]. Studies have shown that HAIC therapy increases local drug concentration in the tumor through selective hepatic artery perfusion, which can significantly improve antitumor treatment and reduce systemic adverse effects[8]. It has been included in guidelines for improving the prognosis of advanced HCC[5,9,10]. In addition, HAIC treatment has also shown good efficacy in biliary tract tumors, and metastatic tumors, such as liver metastases from breast cancer[11-14], liver metastases from gastric cancer, and liver metastases from colorectal cancer[9,15-20].
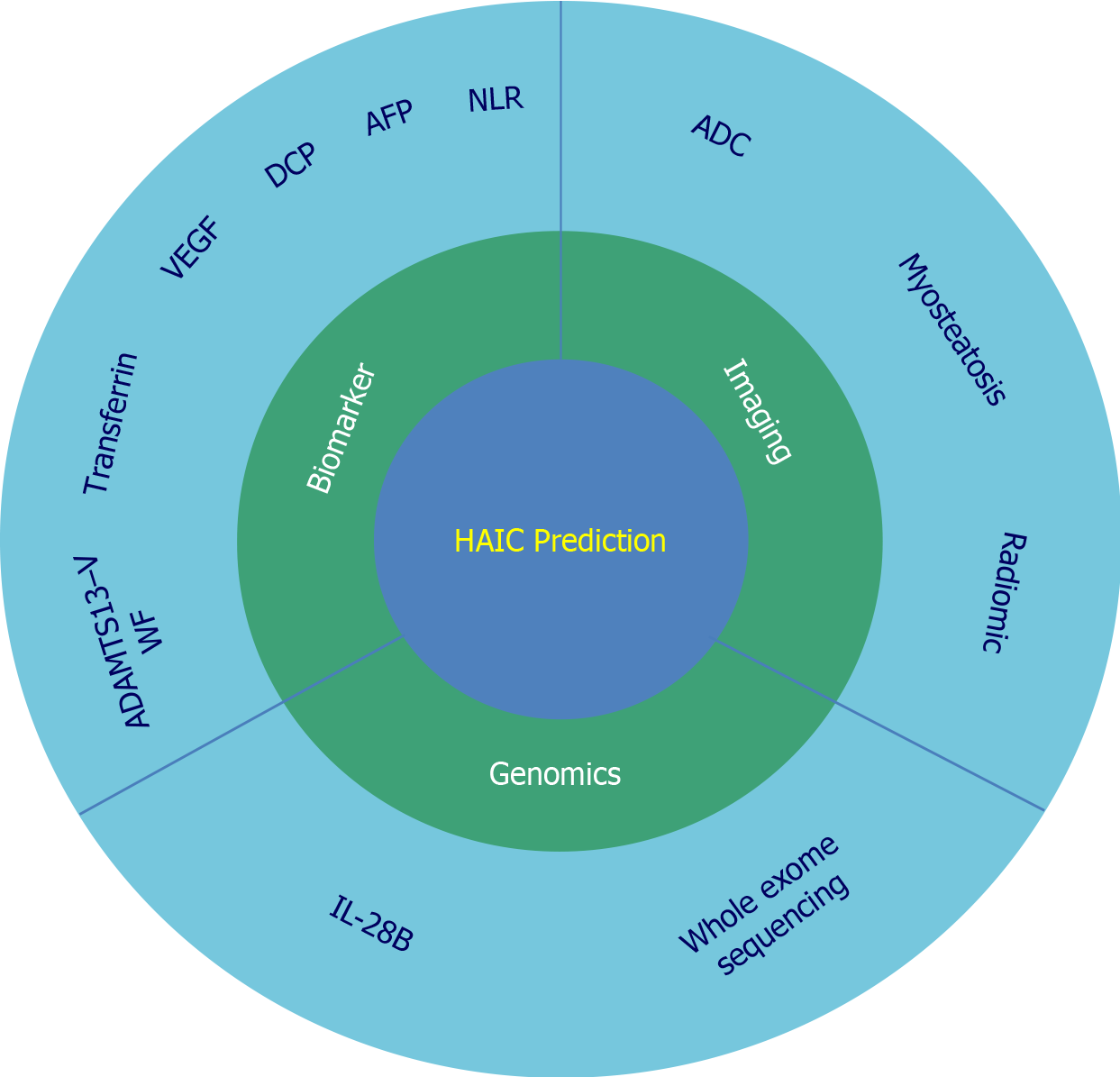
However, owing to the heterogeneity of tumors, the efficacy of HAIC may vary. To improve the efficacy of HAIC treatment, there is an urgent need for the predictive assessment of HAIC efficacy when selecting appropriate patients for treatment. Current prognostic prediction modalities for HAIC treatment include serological indicators, genomic sequencing, and imaging techniques (Figure 1). This review summarized the existing HAIC predictive indicators, provided a reference for the clinical selection of appropriate patients with HCC, and attempted to identify the direction of further development of predictive modalities.
Many biomarkers are commonly used for the early diagnosis, monitoring, and outcome prediction in patients with HCC because of their high sensitivity, specificity, and accessibility[21]. Currently, inflammatory indicators and tumor-specific markers are used to predict the prognosis of HAIC, and we summarized and analyzed the impact of these markers on prognosis (Table 1).
| Biomarker | PFS HR (95%CI) | OS HR (95%CI) | ORR HR (95%CI) | ORR (%) | Ref. |
| NLR | 1.363 (1.008-1.843) | 1.492 (1.106-2.012) | 1.918 (1.092-3.369) | 37.6 | [31] |
| NLR | 1.984 (1.111-3.545) | 24.3 | [39] | ||
| DCP | 2.460 (1.434-4.220) | 3.097 (1.728-5.551) | 24.3 | [39] | |
| AFP DCP | 2.17 (1.23-3.92) | 51.9 | [52] | ||
| 1.9 (1.06-3.42) | 56.4 | [52] | |||
| VEGF | 2.42 (1.33-4.38) | 35.2 | [43] | ||
| Serum transferrin | 1 (0.132-0.603) | 46.9 | [47] | ||
| IL-28B | 1.720 (1.072-2.759) | 2.620(1.124-6.107) | 51.9 | [73] |
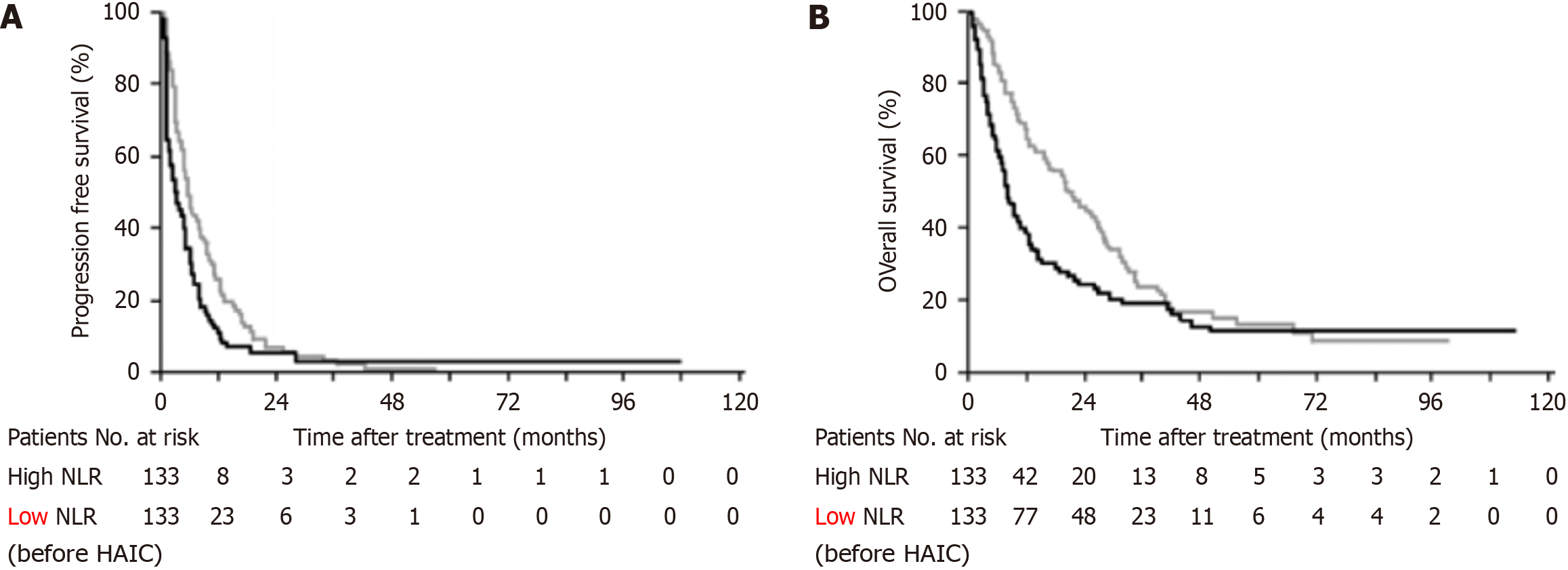
The inflammatory response plays a crucial role in the development and progression of various cancers[22]. Inflammatory factors, such as inflammatory cell levels, active oncogenes, reactive oxygen species, and necrotic substances present in cancerous tissues can promote cancer initiation and progression and affect patient survival[23-25]. Among them, absolute white blood cell count, lymphocyte-to-C-reactive protein ratio, neutrophil to lymphocyte ratio (NLR), platelet-to-lymphocyte ratio, and cytokines are readily available and have been suggested to correlate with the prognosis of patients with various malignancies[26,27]. For example, NLR is now widely recognized as a predictive marker for advanced melanoma, colorectal cancer with liver metastases, and breast cancer[28-30]. Terashima et al[31] analyzed the effect of NLR on the prognosis of HAIC in patients with advanced HCC in the HAIC form of 5-fluorouracil (5-FU) combined with interferon α-2b. The results showed that patients with high NLR have worse objective response rates [27.1 vs 37.6; hazard ratio (HR): 1.918; 95% confidence interval (CI): 1.092-3.369; P = 0.024], progression-free survival (PFS) (3.2 vs 5.6; HR: 1.363; 95%CI: 1.008-1.843; P = 0.044), and overall survival (OS) (8.0 vs 20.7; HR: 1.492; 95%CI: 1.106, 2.012; P < 0.01), which can be used as prognostic predictors for patients with HAIC treatment of advanced HCC (Figure 2). Meanwhile, Tajiri et al[32] also showed that lower NLR suggested better efficacy [odds ratio (OR): 0.49; 95%CI: 0.18-0.96; P = 0.04] and prolonged OS (HR: 3.24; 95%CI: 1.15-9.14; P = 0.03) of HAIC in advanced HCC patients, using HAIC modality of cisplatin combined with 5-FU. Further studies with larger groups of patients are needed to determine the most appropriate NLR threshold and provide better sensitivity and specificity as predictors (Table 1).
Des-γ-carboxy prothrombin (DCP), known as a protein induced by vitamin K absence or antagonist-II, is an abnormal protein produced in HCC. Some studies have shown that DCP-elevated tumors in patients with HCC are more prone to microvascular infiltration, metastasis, and recurrence and can be used as a predictor of HCC prognosis[33,34]. The serum marker alpha-fetoprotein (AFP), the most widely used biomarker for diagnosing HCC, has a diagnostic yield of approximately 46%[35,36]. Whether combined AFP and DCP can improve the prediction effect in HCC diagnosis and prognostic prediction[37]. Yamamoto et al[38] used changes in AFP and DCP levels before and after treatment to evaluate the predictive performance of HAIC (sensitivity, 64% and 79%; specificity, 88% and 64%, respectively). On this basis, the combination of AFP and DCP showed a better predictive ability (sensitivity and specificity of 93% and 60%, respectively). Tsunematsu et al[39] used DCP combined with the NLR to predict the prognosis of cisplatin combined with 5-FU (HAIC) in patients with advanced HCC. It was found that patients with low NLR had significantly longer PFS (PFS: 8.4 vs 2.8; HR: 1.984; 95%CI: 1.111- 3.545; P = 0.021), and early decrease of DCP after HAIC was associated with better prognosis (PFS: 7.2 vs 2.3; HR: 2.460; 95%CI: 1.434-4.220; P = 0.001; OS: 18.5 vs 6.1; HR: 3.097; 95%CI: 1.728-5.551; P < 0.001). Thus, AFP and DCP are effective predictive indicators, and tumor markers, in combination with inflammatory indicators, can improve the prediction of HAIC.
The secretion of VEGF by the tumor cells and the surrounding stroma stimulates endothelial cell proliferation, leading to structural abnormalities under neovascular and hypoxic conditions[40] and significantly correlates with the development, progression, and metastasis of HCC[26,41]. Matsui et al[42] found that a higher serum VEGF level was correlated with a poor prognosis for HAIC. Furthermore, Niizeki et al[43] observed that the serum level of VEGF was an independent predictor of HAIC treatment (cisplatin combined with 5-FU) (OR: 4.77; 95%CI: 1.21-18.90; P = 0.026), and the result indicated that patients with lower serum VEGF levels were more suitable for HAIC treatment.
Serum transferrin levels can predict survival in patients with various liver diseases[44,45]. In addition, it has been shown that serum transferrin, similar to deferoxamine, exerts antitumor effects[46]. Zaitsu et al[47] found that serum transferrin is an independent prognostic predictor of HAIC treatment (low-dose cisplatin combination with 5-FU) (HR: 0.282; 95%CI: 0.132-0.603; P = 0.001) and that patients with serum transferrin ≥190 mg/dL had better survival than those with serum transferrin < 190 mg/dL (median survival time 12.0 vs 4.9; HR: 1; 95%CI: 0.132-0.6030; P = 0.001). These results suggested that serum transferrin can predict the prognosis of HCC patients treated with HAIC and that elevated transferrin in vivo contributes to anti-tumor progression and improves patient survival.
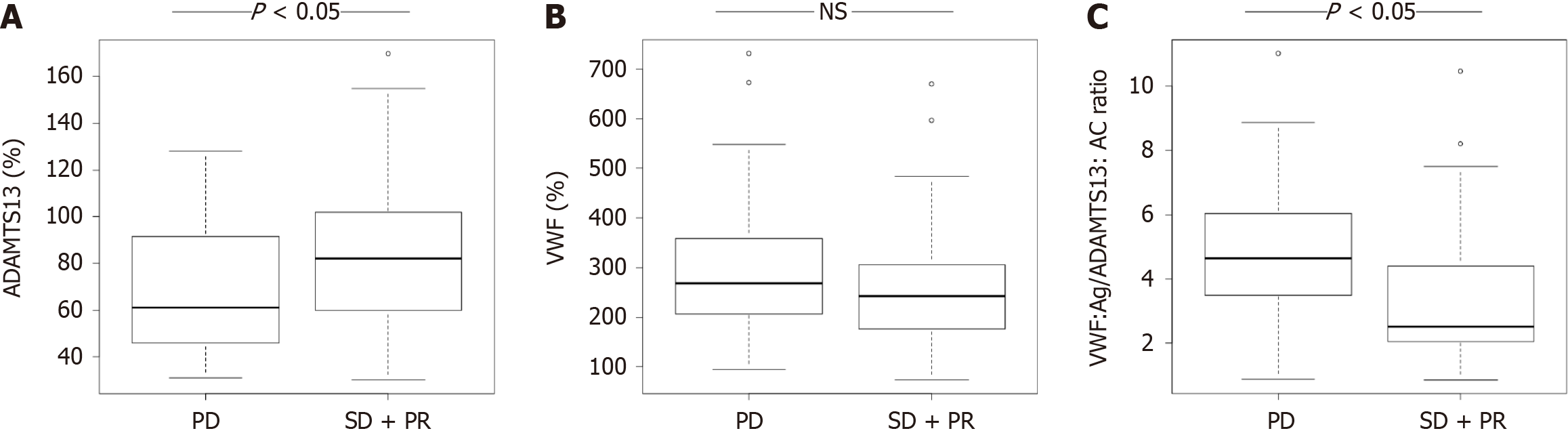
ADAMTS13 is a metalloprotease produced by the hepatic stellate cells, whose main function is to cleave vascular VWF into smaller multimers[48]. ADAMTS13–VWF imbalance is associated with hypercoagulability and cancer progression[48-50]. Takaya et al[51] found that the ratio of VWF to ADAMTS13 was an independent predictor for patients with HCC treated with HAIC (cisplatin combined with 5-FU) (OR: 0.176; 95%CI: 0.0493-0.631; P = 0.00766), with high specificity (87.1%), sensitivity (53.7%), and an AUC of 0.715. The above study used coagulation-related factors to predict the prognosis of HAIC based on the blood coagulation cascade related to cancer progression. Further studies are needed to explore the specific mechanism (Figure 3).
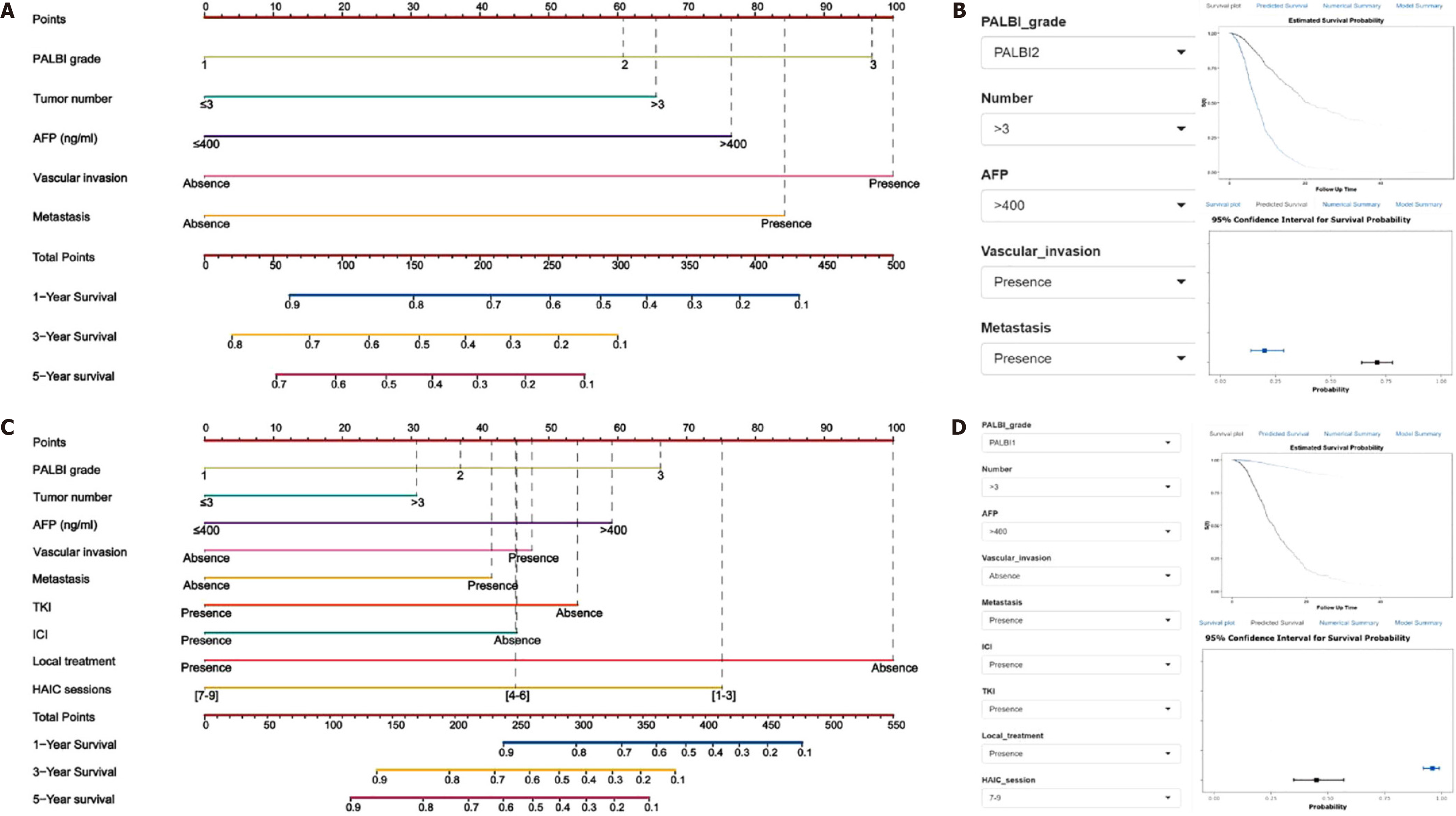
In addition to using a single indicator to predict the efficacy of HAIC, many recent studies have improved predictive efficacy by combining indicators. Saeki et al[52] developed an Assessment for Continuous Treatment score composed of three independent indicators: Child-Pugh score, AFP, and DCP, which ranged from 0 to 3. Patients stratified into two groups according to this score showed significantly different prognoses (≤ 1 vs ≥ 2 points: median survival time, 15.1 vs 8.7 months; P = 0.003). Many HCC staging systems and other prognostic scoring criteria have been developed to predict the prognosis of patients with HCC. Mei et al[53] used six independent predictors, including C-reactive protein, albumin-bilirubin (ALBI) grade, AFP, extrahepatic metastasis, portal vein invasion, and tumor size to establish a nomogram to predict individualized OS in patients with unresectable HCC after HAIC. Compared with the predicted effect of conventional staging systems, such as the Barcelona Clinic Liver Cancer (BCLC), Japan Integrated Staging (JIS) score, American Joint Committee on Cancer (AJCC), and Hong Kong liver cancer (HKLC)[7,54-56], the nomogram showed a better C-index (0.71, Table 2), indicating its superior predictive ability. Furthermore, Wang et al[57] established two nomogram models to assess the survival prognosis of HAIC and HAIC combined therapies in patients with HCC. First, five independent indicators (tumor size, vascular invasion, metastasis, ALBI grade, and AFP level) were identified using multivariate analysis. The C-index value of this model was 0.701 (95%CI: 0.664-0.738) for the external test cohort. The predictive effect improved significantly in the HAIC combined model, including the abovementioned factors and the number of sessions, in combination with immune checkpoint inhibitors, tyrosine kinase inhibitors, and local therapy. The C-index values were 0.816 (95%CI: 0.789-0.843) in the external test cohort (Figure 4). Thus, nomogram models are essential for identifying patients with large HCC who are suitable for treatment with HAIC combination therapy and may potentially benefit personalized decision-making.
| Staging system | Training cohort (n = 308) | Validation cohort (n = 155) | ||||
| C-index | 95%CI | P value1 | C-index | 95%CI | P value1 | |
| The nomogram | 0.710 | 0.674–0.746 | NA | 0.716 | 0.664–0.768 | NA |
| BCLC staging system | 0.562 | 0.523–0.602 | < 0.001 | 0.613 | 0.558–0.668 | 0.005 |
| HKLC staging system | 0.626 | 0.594–0.658 | < 0.001 | 0.659 | 0.616–0.702 | 0.043 |
| CIS | 0.631 | 0.596–0.666 | < 0.001 | 0.658 | 0.610–0.707 | 0.044 |
| AJCC eighth edition | 0.637 | 0.603–0.672 | < 0.001 | 0.634 | 0.588–0.680 | 0.015 |
| JIS score | 0.619 | 0.582–0.655 | < 0.001 | 0.638 | 0.587–0.689 | 0.017 |
| Okuda classification | 0.634 | 0.600–0.668 | < 0.001 | 0.631 | 0.585–0.676 | 0.011 |
Apparent diffusion coefficient (ADC), a magnetic resonance imaging technique, is established based on the diffusion-weighted imaging (DWI) modality, which quantitatively reflects the restriction of water molecule movement[58]. It has been shown that the ADC helps predict HCC with differentiation adverse, proliferative activity, and microvascular infiltration[59-62]. Therefore, the calculation of ADC values using DWI can be used to assess the tumor stage and predict the treatment outcome of liver cancer. Sung et al[63] found that the tumor ADC to liver ADC ratio (OR: 3.217; 95%CI: 1.264-8.187; P = 0.014) was correlated with the response to HAIC treatment for unresectable advanced HCC patients. A ratio < 0.741 suggests a good therapeutic effect of HAIC. In summary, analysis of the changes in the ratio of tumor ADC to liver ADC after HAIC treatment is helpful in predicting the efficacy in patients and can be used as an important predictor of HAIC treatment.
In recent years, preoperative assessment of body composition has received increasing attention. Many studies have shown that the assessment of myosteatosis on computed tomography (CT) can predict the prognosis of patients with HCC. Yi et al[64] established a nomogram by measuring the value of myosteatosis combined with clinical variables (serum red blood cells, hemoglobin, creatinine, and mean CT value of visceral fat) to effectively predict the prognosis of combined anti-PD-1 and HAIC therapy. The AUC of the prediction model was 0.711 (95%CI: 0.75-0.95). However, large-scale multicenter prospective studies with standardized imaging protocols and body composition measurement tools are required to validate these results.
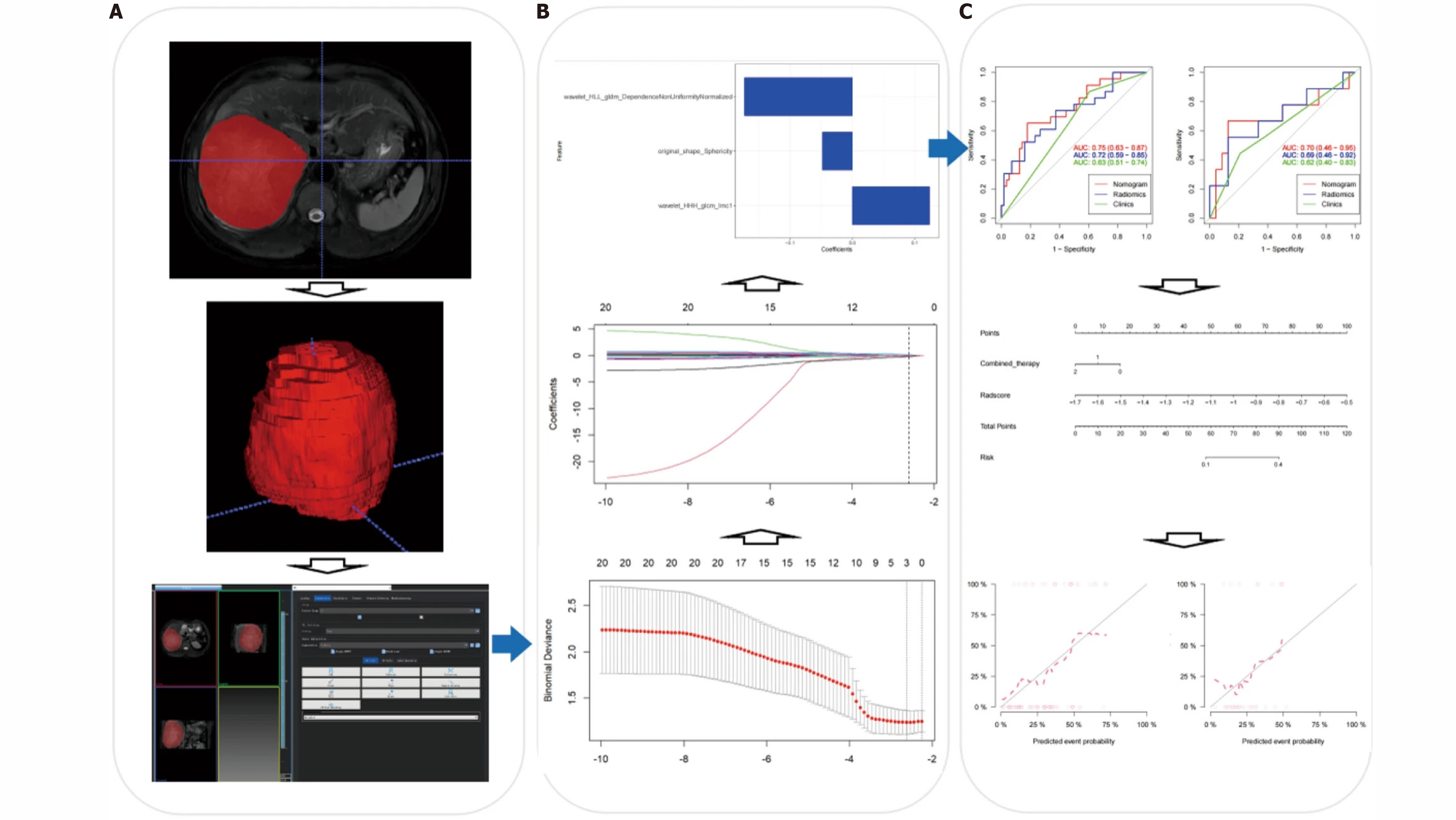
Recently, Zhao et al[65] developed a better prognostic score for predicting HAIC-treated HCC using a nomogram with a combined model of radiomics and albumin-bilirubin scores (Figure 5). The albumin-bilirubin score in this nomogram (AUC 0.69; 95%CI: 0.51-0.88) and the radiomic score (AUC 0.70; 95%CI: 0.58-0.82) were both independent indicators of HAIC treatment response. The result showed that the combined two indicators achieved better performance in the prediction, with AUC scores of 0.79 (95%CI: 0.68-0.90) and 0.75 (95%CI: 0.58-0.92) in the training and validation cohorts, respectively.
HCC exhibits tumor heterogeneity[66]. It has been shown that HCC, which develops on HBV infection, HCV infection, alcohol abuse, and nonalcoholic fatty liver disease, have different molecular mutation profiles[67]. Schulze et al[68] and Gao et al[69] showed that TP53, CTNNB1, and TERT promoters are the most common mutations in HCC, and targeting these molecular mutations can improve patient outcomes. Therefore, the analysis of the mutational characteristics of patients and targeted treatment of genetic targets are beneficial for improving patient prognosis.
The interleukin-28B (IL-28B) gene is a member of the IFN-related cytokine family[70]. Single-nucleotide polymorphisms in IL-28B are associated with the effectiveness of interferon therapy for chronic hepatitis C infection[60,71]. Whether IL-28B genotype affects the prognosis of patients with advanced HCC remains unknown[72]. Terashima et al[73] analyzed whether the IL-28B SNP (rs8099917) was correlated with the therapeutic efficacy and patient prognosis of HAIC in advanced HCC. Genotypes were divided into minor and major genotypes. Patients with minor genotypes had better objective response rates (51.9% vs 29.1%, P = 0.022) and significantly longer median OS (16.9 vs 14.1, P = 0.027) (Table 1). In addition, it has been shown that the SNP polypeptide N -acetylgalactosaminyltransferase14 (GALNT14-rs9679162) genotype "GG" is associated with prolonged overall survival after HAIC treatment using HAIC modality of cisplatin combined with 5-FU in patients with advanced HCC (HR: 0.187; 95%CI: 0.020-1.775; P = 0.019)[74,75].
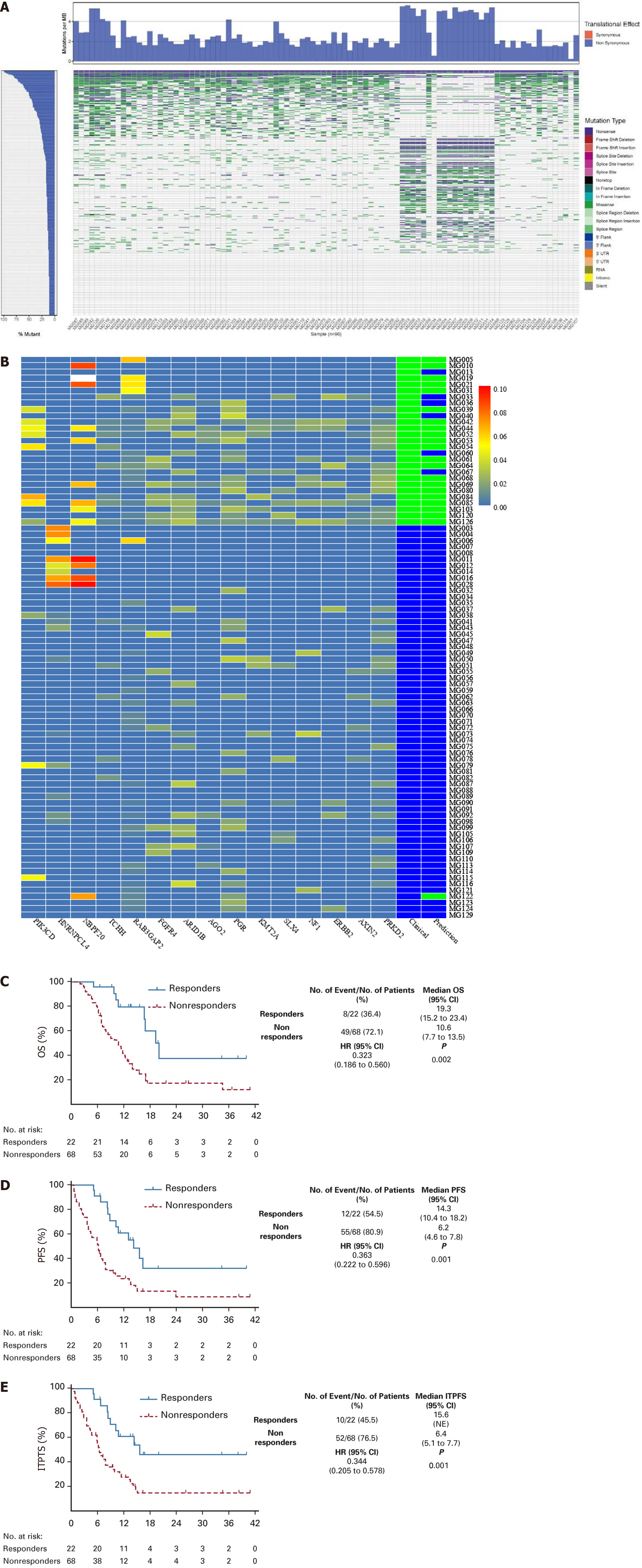
Furthermore, Ning et al[76] developed an exploratory model for predicting the efficacy of FOLFOX HAIC based on genomic sequencing of 96 patients (Figure 6A). A panel of 15 mutant genes (PIK3CD, HNRNPCL4, NBPF20, TCHH, RAB3- GAP2, FGFR4, ARID1B, AGO2, PGR, KMT2A, SLX4, NF1, ERBB2, AXIN2, and PRKD2) was derived from whole-exome sequencing of 90 patients to predict the efficacy of HAIC treatment (Figure 6B). The multivariate model indicated that the mutational panel was independent of clinical characteristics and could differentiate 83% of the patients who responded to HAIC treatment. HAIC responders had longer PFS and OS (PFS 14.3 vs 6.2 months, P = 0.001; OS 19.3 vs 10.6, P = 0.002; Figure 6C-E).
In conclusion, serological indicators, genetic testing, and imaging techniques can predict the prognosis of patients with HCC treated with HAIC and have shown excellent predictive value. Despite the apparent correlation with HAIC efficacy, there are shortcomings of the above markers for predicting the prognosis of HAIC treatment for HCC. First, this was a retrospective study. Second, they were conducted in a single center, and the number of cases in some studies was small, which may lead to potential prognostic bias; therefore, there is a need for validation using large-scale prospective studies of multiple centers. Finally, there is a need to improve prognostic accuracy by combining multiple markers and artificial intelligence to predict the prognosis of patients with HAIC-treated HCC. Although these are promising developments, there is still a need to standardize data for future studies to improve the veracity and accuracy of the results.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69808] [Article Influence: 13961.6] [Reference Citation Analysis (51)] |
| 2. | Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J, Finn RS. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1323] [Reference Citation Analysis (0)] |
| 3. | Singal AG, Lampertico P, Nahon P. Epidemiology and surveillance for hepatocellular carcinoma: New trends. J Hepatol. 2020;72:250-261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 918] [Cited by in RCA: 867] [Article Influence: 144.5] [Reference Citation Analysis (7)] |
| 4. | Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A. Hepatocellular carcinoma. Lancet. 2022;400:1345-1362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1976] [Cited by in RCA: 1736] [Article Influence: 434.0] [Reference Citation Analysis (7)] |
| 5. | Zhou H, Song T. Conversion therapy and maintenance therapy for primary hepatocellular carcinoma. Biosci Trends. 2021;15:155-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 135] [Article Influence: 27.0] [Reference Citation Analysis (6)] |
| 6. | Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301-1314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4664] [Cited by in RCA: 4437] [Article Influence: 554.6] [Reference Citation Analysis (8)] |
| 7. | Reig M, Forner A, Rimola J, Ferrer-Fàbrega J, Burrel M, Garcia-Criado Á, Kelley RK, Galle PR, Mazzaferro V, Salem R, Sangro B, Singal AG, Vogel A, Fuster J, Ayuso C, Bruix J. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol. 2022;76:681-693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3613] [Cited by in RCA: 3336] [Article Influence: 834.0] [Reference Citation Analysis (6)] |
| 8. | Ikeda M, Morizane C, Ueno M, Okusaka T, Ishii H, Furuse J. Chemotherapy for hepatocellular carcinoma: current status and future perspectives. Jpn J Clin Oncol. 2018;48:103-114. [PubMed] [DOI] [Full Text] |
| 9. | Chen LT, Martinelli E, Cheng AL, Pentheroudakis G, Qin S, Bhattacharyya GS, Ikeda M, Lim HY, Ho GF, Choo SP, Ren Z, Malhotra H, Ueno M, Ryoo BY, Kiang TC, Tai D, Vogel A, Cervantes A, Lu SN, Yen CJ, Huang YH, Chen SC, Hsu C, Shen YC, Tabernero J, Yen Y, Hsu CH, Yoshino T, Douillard JY. Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with intermediate and advanced/relapsed hepatocellular carcinoma: a TOS-ESMO initiative endorsed by CSCO, ISMPO, JSMO, KSMO, MOS and SSO. Ann Oncol. 2020;31:334-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (4)] |
| 10. | Li QJ, He MK, Chen HW, Fang WQ, Zhou YM, Xu L, Wei W, Zhang YJ, Guo Y, Guo RP, Chen MS, Shi M. Hepatic Arterial Infusion of Oxaliplatin, Fluorouracil, and Leucovorin Versus Transarterial Chemoembolization for Large Hepatocellular Carcinoma: A Randomized Phase III Trial. J Clin Oncol. 2022;40:150-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 306] [Article Influence: 76.5] [Reference Citation Analysis (7)] |
| 11. | Shirono T, Niizeki T, Iwamoto H, Shimose S, Suzuki H, Kawaguchi T, Kamachi N, Noda Y, Okamura S, Nakano M, Kuromatu R, Koga H, Torimura T. Therapeutic Outcomes and Prognostic Factors of Unresectable Intrahepatic Cholangiocarcinoma: A Data Mining Analysis. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Ishii M, Itano O, Morinaga J, Shirakawa H, Itano S. Potential efficacy of hepatic arterial infusion chemotherapy using gemcitabine, cisplatin, and 5-fluorouracil for intrahepatic cholangiocarcinoma. PLoS One. 2022;17:e0266707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 13. | Gou Q, Wu L, Cui W, Mo Z, Zeng D, Gan L, He J, Mai Q, Shi F, Chen M, Sun Z, Liu Y, Wu J, Chen X, Zhuang W, Xu R, Li W, Cai Q, Zhang J, Li J, Zhou Z. Stent placement combined with intraluminal radiofrequency ablation and hepatic arterial infusion chemotherapy for advanced biliary tract cancers with biliary obstruction: a multicentre, retrospective, controlled study. Eur Radiol. 2021;31:5851-5862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | Tewes M, Peis MW, Bogner S, Theysohn JM, Reinboldt MP, Schuler M, Welt A. Hepatic arterial infusion chemotherapy for extensive liver metastases of breast cancer: efficacy, safety and prognostic parameters. J Cancer Res Clin Oncol. 2017;143:2131-2141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 15. | Datta J, Narayan RR, Kemeny NE, D'Angelica MI. Role of Hepatic Artery Infusion Chemotherapy in Treatment of Initially Unresectable Colorectal Liver Metastases: A Review. JAMA Surg. 2019;154:768-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 112] [Article Influence: 16.0] [Reference Citation Analysis (3)] |
| 16. | Liu P, Zhu X, Li J, Lu M, Leng J, Li Y, Yu J. Retrospective analysis of interventional treatment of hepatic metastasis from gastroenteropancreatic neuroendocrine tumors. Chin J Cancer Res. 2017;29:581-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 17. | Zhang Y, Wang K, Yang T, Cao Y, Liang W, Yang X, Xiao T. Meta-Analysis of Hepatic Arterial Infusion for Liver Metastases From Colorectal Cancer. Front Oncol. 2021;11:628558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Liu P, Zhu H, Zhang X, Feng A, Zhu X, Sun Y. Predicting Survival for Hepatic Arterial Infusion Chemotherapy of Unresectable Colorectal Liver Metastases: Radiomics Analysis of Pretreatment Computed Tomography. J Transl Int Med. 2022;10:56-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 19. | Lim A, Le Sourd S, Senellart H, Luet D, Douane F, Perret C, Bouvier A, Métairie S, Cauchin E, Rougier P, Matysiak-Budnik T, Touchefeu Y. Hepatic Arterial Infusion Chemotherapy for Unresectable Liver Metastases of Colorectal Cancer: A Multicenter Retrospective Study. Clin Colorectal Cancer. 2017;16:308-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Zhang HY, Guo JH, Gao S, Chen H, Wang XD, Zhang PJ, Liu P, Cao G, Xu HF, Zhu LZ, Yang RJ, Li J, Zhu X. Effect of primary tumor side on survival outcomes in metastatic colorectal cancer patients after hepatic arterial infusion chemotherapy. World J Gastrointest Oncol. 2018;10:431-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Casadei-Gardini A, Orsi G, Caputo F, Ercolani G. Developments in predictive biomarkers for hepatocellular carcinoma therapy. Expert Rev Anticancer Ther. 2020;20:63-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (2)] |
| 22. | Diakos CI, Charles KA, McMillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493-e503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1819] [Cited by in RCA: 1750] [Article Influence: 145.8] [Reference Citation Analysis (4)] |
| 23. | Fonseca GWPD, Farkas J, Dora E, von Haehling S, Lainscak M. Cancer Cachexia and Related Metabolic Dysfunction. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 24. | Sangro B, Sarobe P, Hervás-Stubbs S, Melero I. Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2021;18:525-543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1090] [Cited by in RCA: 1002] [Article Influence: 200.4] [Reference Citation Analysis (1)] |
| 25. | Miyata K, Imai Y, Hori S, Nishio M, Loo TM, Okada R, Yang L, Nakadai T, Maruyama R, Fujii R, Ueda K, Jiang L, Zheng H, Toyokuni S, Sakata T, Shirahige K, Kojima R, Nakayama M, Oshima M, Nagayama S, Seimiya H, Hirota T, Saya H, Hara E, Takahashi A. Pericentromeric noncoding RNA changes DNA binding of CTCF and inflammatory gene expression in senescence and cancer. Proc Natl Acad Sci U S A. 2021;118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 26. | Rico Montanari N, Anugwom CM, Boonstra A, Debes JD. The Role of Cytokines in the Different Stages of Hepatocellular Carcinoma. Cancers (Basel). 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 27. | Okugawa Y, Toiyama Y, Yamamoto A, Shigemori T, Ide S, Kitajima T, Fujikawa H, Yasuda H, Hiro J, Yoshiyama S, Yokoe T, Saigusa S, Tanaka K, Shirai Y, Kobayashi M, Ohi M, Araki T, McMillan DC, Miki C, Goel A, Kusunoki M. Lymphocyte-C-reactive Protein Ratio as Promising New Marker for Predicting Surgical and Oncological Outcomes in Colorectal Cancer. Ann Surg. 2020;272:342-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 197] [Article Influence: 32.8] [Reference Citation Analysis (3)] |
| 28. | Capone M, Giannarelli D, Mallardo D, Madonna G, Festino L, Grimaldi AM, Vanella V, Simeone E, Paone M, Palmieri G, Cavalcanti E, Caracò C, Ascierto PA. Baseline neutrophil-to-lymphocyte ratio (NLR) and derived NLR could predict overall survival in patients with advanced melanoma treated with nivolumab. J Immunother Cancer. 2018;6:74. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 401] [Cited by in RCA: 385] [Article Influence: 48.1] [Reference Citation Analysis (4)] |
| 29. | Verter E, Berger Y, Perl G, Peretz I, Tovar A, Morgenstern S, Brenner B, Benchimol D, Kashtan H, Sadot E. Neutrophil-to-Lymphocyte Ratio Predicts Recurrence Pattern in Patients with Resectable Colorectal Liver Metastases. Ann Surg Oncol. 2021;28:4320-4329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (3)] |
| 30. | Grassadonia A, Graziano V, Iezzi L, Vici P, Barba M, Pizzuti L, Cicero G, Krasniqi E, Mazzotta M, Marinelli D, Amodio A, Natoli C, Tinari N. Prognostic Relevance of Neutrophil to Lymphocyte Ratio (NLR) in Luminal Breast Cancer: A Retrospective Analysis in the Neoadjuvant Setting. Cells. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 31. | Terashima T, Yamashita T, Iida N, Nakagawa H, Arai K, Kitamura K, Kagaya T, Sakai Y, Mizukoshi E, Honda M, Kaneko S. Blood neutrophil to lymphocyte ratio as a predictor in patients with advanced hepatocellular carcinoma treated with hepatic arterial infusion chemotherapy. Hepatol Res. 2015;45:949-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 32. | Tajiri K, Kawai K, Minemura M, Yasumura S, Hosokawa A, Kawabe H, Tomizawa G, Sugiyama T. Neutrophil/lymphocyte ratio as a prognostic indicator of hepatic arterial infusion chemotherapy with arterial cisplatin plus continuous 5-fluorouracil. Hepatol Res. 2015;45:755-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Yang Y, Li G, Lu Z, Liu Y, Kong J, Liu J. Progression of Prothrombin Induced by Vitamin K Absence-II in Hepatocellular Carcinoma. Front Oncol. 2021;11:726213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 34. | Liebman HA, Furie BC, Tong MJ, Blanchard RA, Lo KJ, Lee SD, Coleman MS, Furie B. Des-gamma-carboxy (abnormal) prothrombin as a serum marker of primary hepatocellular carcinoma. N Engl J Med. 1984;310:1427-1431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 481] [Cited by in RCA: 418] [Article Influence: 10.0] [Reference Citation Analysis (5)] |
| 35. | Zhang G, Ha SA, Kim HK, Yoo J, Kim S, Lee YS, Hur SY, Kim YW, Kim TE, Park YG, Wang J, Yang Y, Xu Z, Song EY, Huang Z, Jirun P, Zhongtian J, Shishi Q, Zhuqingqing C, Lei G, Kim JW. Combined analysis of AFP and HCCR-1 as an useful serological marker for small hepatocellular carcinoma: a prospective cohort study. Dis Markers. 2012;32:265-271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 36. | Wang T, Zhang KH. New Blood Biomarkers for the Diagnosis of AFP-Negative Hepatocellular Carcinoma. Front Oncol. 2020;10:1316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 126] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 37. | Feng H, Li B, Li Z, Wei Q, Ren L. PIVKA-II serves as a potential biomarker that complements AFP for the diagnosis of hepatocellular carcinoma. BMC Cancer. 2021;21:401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 121] [Article Influence: 24.2] [Reference Citation Analysis (6)] |
| 38. | Yamamoto S, Onishi H, Takaki A, Oyama A, Adachi T, Wada N, Sakata M, Yasunaka T, Shiraha H, Okada H. The Early Decline of α-Fetoprotein and Des-γ-Carboxy Prothrombin Predicts the Response of Hepatic Arterial Infusion Chemotherapy in Hepatocellular Carcinoma Patients. Gastrointest Tumors. 2020;7:83-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 39. | Tsunematsu S, Suda G, Yamasaki K, Kimura M, Takaaki I, Umemura M, Ito J, Sato F, Nakai M, Sho T, Morikawa K, Ogawa K, Kamiyama T, Taketomi A, Sakamoto N. Combination of neutrophil-to-lymphocyte ratio and early des-γ-carboxyprothrombin change ratio as a useful predictor of treatment response for hepatic arterial infusion chemotherapy against advanced hepatocellular carcinoma. Hepatol Res. 2017;47:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Apte RS, Chen DS, Ferrara N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell. 2019;176:1248-1264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2322] [Cited by in RCA: 2058] [Article Influence: 294.0] [Reference Citation Analysis (6)] |
| 41. | Morse MA, Sun W, Kim R, He AR, Abada PB, Mynderse M, Finn RS. The Role of Angiogenesis in Hepatocellular Carcinoma. Clin Cancer Res. 2019;25:912-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 426] [Article Influence: 60.9] [Reference Citation Analysis (0)] |
| 42. | Matsui D, Nagai H, Mukozu T, Ogino YU, Sumino Y. VEGF in patients with advanced hepatocellular carcinoma receiving intra-arterial chemotherapy. Anticancer Res. 2015;35:2205-2210. [PubMed] |
| 43. | Niizeki T, Sumie S, Torimura T, Kurogi J, Kuromatsu R, Iwamoto H, Aino H, Nakano M, Kawaguchi A, Kakuma T, Sata M. Serum vascular endothelial growth factor as a predictor of response and survival in patients with advanced hepatocellular carcinoma undergoing hepatic arterial infusion chemotherapy. J Gastroenterol. 2012;47:686-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 44. | Meier JA, Bokemeyer A, Cordes F, Fuhrmann V, Schmidt H, Hüsing-Kabar A, Kabar I. Serum levels of ferritin and transferrin serve as prognostic factors for mortality and survival in patients with end-stage liver disease: A propensity score-matched cohort study. United European Gastroenterol J. 2020;8:332-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 45. | Viveiros A, Finkenstedt A, Schaefer B, Mandorfer M, Scheiner B, Lehner K, Tobiasch M, Reiberger T, Tilg H, Edlinger M, Zoller H. Transferrin as a predictor of survival in cirrhosis. Liver Transpl. 2018;24:343-351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 46. | Gkouvatsos K, Papanikolaou G, Pantopoulos K. Regulation of iron transport and the role of transferrin. Biochim Biophys Acta. 2012;1820:188-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 362] [Article Influence: 25.9] [Reference Citation Analysis (1)] |
| 47. | Zaitsu J, Yamasaki T, Saeki I, Harima Y, Iwamoto T, Matsumoto T, Urata Y, Hidaka I, Marumoto Y, Ishikawa T, Takami T, Yamamoto N, Kaino S, Uchida K, Terai S, Sakaida I. Serum transferrin as a predictor of prognosis for hepatic arterial infusion chemotherapy in advanced hepatocellular carcinoma. Hepatol Res. 2014;44:481-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 48. | Favaloro EJ, Pasalic L, Henry B, Lippi G. Laboratory testing for ADAMTS13: Utility for TTP diagnosis/exclusion and beyond. Am J Hematol. 2021;96:1049-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 49. | Pépin M, Kleinjan A, Hajage D, Büller HR, Di Nisio M, Kamphuisen PW, Salomon L, Veyradier A, Stepanian A, Mahé I. ADAMTS-13 and von Willebrand factor predict venous thromboembolism in patients with cancer. J Thromb Haemost. 2016;14:306-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 60] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 50. | Guo R, Yang J, Liu X, Wu J, Chen Y. Increased von Willebrand factor over decreased ADAMTS-13 activity is associated with poor prognosis in patients with advanced non-small-cell lung cancer. J Clin Lab Anal. 2018;32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 51. | Takaya H, Namisaki T, Moriya K, Shimozato N, Kaji K, Ogawa H, Ishida K, Tsuji Y, Kaya D, Takagi H, Fujinaga Y, Nishimura N, Sawada Y, Kawaratani H, Akahane T, Matsumoto M, Yoshiji H. Association between ADAMTS13 activity-VWF antigen imbalance and the therapeutic effect of HAIC in patients with hepatocellular carcinoma. World J Gastroenterol. 2020;26:7232-7241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 52. | Saeki I, Yamasaki T, Tanabe N, Iwamoto T, Matsumoto T, Urata Y, Hidaka I, Ishikawa T, Takami T, Yamamoto N, Uchida K, Terai S, Sakaida I. A new therapeutic assessment score for advanced hepatocellular carcinoma patients receiving hepatic arterial infusion chemotherapy. PLoS One. 2015;10:e0126649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 53. | Mei J, Lin WP, Shi F, Wei W, Liang JB, Shi M, Zheng L, Li SH, Guo RP. Prognostic nomogram predicting survival of patients with unresectable hepatocellular carcinoma after hepatic arterial infusion chemotherapy. Eur J Radiol. 2021;142:109890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (3)] |
| 54. | Ishikawa T, Kubota T, Abe S, Watanabe Y, Sugano T, Inoue R, Iwanaga A, Seki K, Honma T, Yoshida T. Hepatic arterial infusion chemotherapy with cisplatin before radical local treatment of early hepatocellular carcinoma (JIS score 0/1) improves survival. Ann Oncol. 2014;25:1379-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 55. | Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more "personalized" approach to cancer staging. CA Cancer J Clin. 2017;67:93-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4716] [Cited by in RCA: 4801] [Article Influence: 533.4] [Reference Citation Analysis (8)] |
| 56. | Yau T, Tang VY, Yao TJ, Fan ST, Lo CM, Poon RT. Development of Hong Kong Liver Cancer staging system with treatment stratification for patients with hepatocellular carcinoma. Gastroenterology. 2014;146:1691-700.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 578] [Cited by in RCA: 548] [Article Influence: 45.7] [Reference Citation Analysis (7)] |
| 57. | Yao W, Wei R, Jia J, Li W, Zuo M, Zhuo S, Shi G, Wu P, An C. Development and validation of prognostic nomograms for large hepatocellular carcinoma after HAIC. Ther Adv Med Oncol. 2023;15:17588359231163845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (8)] |
| 58. | Gong XQ, Tao YY, Wu YK, Liu N, Yu X, Wang R, Zheng J, Huang XH, Li JD, Yang G, Wei XQ, Yang L, Zhang XM. Progress of MRI Radiomics in Hepatocellular Carcinoma. Front Oncol. 2021;11:698373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (6)] |
| 59. | Okamura S, Sumie S, Tonan T, Nakano M, Satani M, Shimose S, Shirono T, Iwamoto H, Aino H, Niizeki T, Tajiri N, Kuromatsu R, Okuda K, Nakashima O, Torimura T. Diffusion-weighted magnetic resonance imaging predicts malignant potential in small hepatocellular carcinoma. Dig Liver Dis. 2016;48:945-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 60. | Roy S, Guha Roy D, Bhushan A, Bharatiya S, Chinnaswamy S. Functional genetic variants of the IFN-λ3 (IL28B) gene and transcription factor interactions on its promoter. Cytokine. 2021;142:155491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 61. | Surov A, Meyer HJ, Wienke A. Correlation between apparent diffusion coefficient (ADC) and cellularity is different in several tumors: a meta-analysis. Oncotarget. 2017;8:59492-59499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 257] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 62. | Surov A, Meyer HJ, Wienke A. Associations between apparent diffusion coefficient (ADC) and KI 67 in different tumors: a meta-analysis. Part 1: ADC(mean). Oncotarget. 2017;8:75434-75444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 117] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 63. | Sung PS, Choi MH, Yang H, Lee SK, Chun HJ, Jang JW, Choi JY, Yoon SK, Choi JI, Lee YJ, Bae SH. Diffusion-Weighted Magnetic Resonance Imaging in Hepatocellular Carcinoma as a Predictor of a Response to Cisplatin-Based Hepatic Arterial Infusion Chemotherapy. Front Oncol. 2020;10:600233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 64. | Yi X, Fu Y, Long Q, Zhao Y, Li S, Zhou C, Lin H, Liu X, Liu C, Chen C, Shi L. Myosteatosis can Predict Unfavorable Outcomes in Advanced Hepatocellular Carcinoma Patients Treated With Hepatic Artery Infusion Chemotherapy and Anti-PD-1 Immunotherapy. Front Oncol. 2022;12:892192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 65. | Zhao Y, Huang F, Liu S, Jian L, Xia X, Lin H, Liu J. Prediction of therapeutic response of unresectable hepatocellular carcinoma to hepatic arterial infusion chemotherapy based on pretherapeutic MRI radiomics and Albumin-Bilirubin score. J Cancer Res Clin Oncol. 2023;149:5181-5192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 66. | Gao Q, Wang XY, Zhou J, Fan J. Multiple carcinogenesis contributes to the heterogeneity of HCC. Nat Rev Gastroenterol Hepatol. 2015;12:13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (3)] |
| 67. | Craig AJ, von Felden J, Garcia-Lezana T, Sarcognato S, Villanueva A. Tumour evolution in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2020;17:139-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 676] [Cited by in RCA: 607] [Article Influence: 101.2] [Reference Citation Analysis (0)] |
| 68. | Schulze K, Imbeaud S, Letouzé E, Alexandrov LB, Calderaro J, Rebouissou S, Couchy G, Meiller C, Shinde J, Soysouvanh F, Calatayud AL, Pinyol R, Pelletier L, Balabaud C, Laurent A, Blanc JF, Mazzaferro V, Calvo F, Villanueva A, Nault JC, Bioulac-Sage P, Stratton MR, Llovet JM, Zucman-Rossi J. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat Genet. 2015;47:505-511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1516] [Cited by in RCA: 1413] [Article Influence: 128.5] [Reference Citation Analysis (5)] |
| 69. | Gao Q, Zhu H, Dong L, Shi W, Chen R, Song Z, Huang C, Li J, Dong X, Zhou Y, Liu Q, Ma L, Wang X, Zhou J, Liu Y, Boja E, Robles AI, Ma W, Wang P, Li Y, Ding L, Wen B, Zhang B, Rodriguez H, Gao D, Zhou H, Fan J. Integrated Proteogenomic Characterization of HBV-Related Hepatocellular Carcinoma. Cell. 2019;179:561-577.e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 780] [Cited by in RCA: 706] [Article Influence: 100.9] [Reference Citation Analysis (3)] |
| 70. | Sheppard P, Kindsvogel W, Xu W, Henderson K, Schlutsmeyer S, Whitmore TE, Kuestner R, Garrigues U, Birks C, Roraback J, Ostrander C, Dong D, Shin J, Presnell S, Fox B, Haldeman B, Cooper E, Taft D, Gilbert T, Grant FJ, Tackett M, Krivan W, McKnight G, Clegg C, Foster D, Klucher KM. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat Immunol. 2003;4:63-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1221] [Cited by in RCA: 1223] [Article Influence: 53.2] [Reference Citation Analysis (0)] |
| 71. | Grzegorzewska AE, Jodłowska E, Mostowska A, Jagodziński P. Effect of interferon λ3 gene polymorphisms, rs8099917 and rs12979860, on response to hepatitis B virus vaccination and hepatitis B or C virus infections among hemodialysis patients. Pol Arch Med Wewn. 2015;125:894-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 72. | Fabris C, Falleti E, Cussigh A, Bitetto D, Fontanini E, Bignulin S, Cmet S, Fornasiere E, Fumolo E, Fangazio S, Cerutti A, Minisini R, Pirisi M, Toniutto P. IL-28B rs12979860 C/T allele distribution in patients with liver cirrhosis: role in the course of chronic viral hepatitis and the development of HCC. J Hepatol. 2011;54:716-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 129] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 73. | Terashima T, Honda M, Toyama T, Shimakami T, Shimizu R, Takatori H, Arai K, Kawaguchi K, Kitamura K, Yamashita T, Sakai Y, Mizukoshi E, Kaneko S. IL-28B variant as a predictor in patients with advanced hepatocellular carcinoma treated with hepatic arterial infusion chemotherapy. J Gastroenterol Hepatol. 2020;35:1813-1820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 74. | Lin CC, Hsu CW, Chen YC, Chang ML, Liang KH, Lai MW, Lin CL, Chien RN, Lin KH, Yeh CT. A GALNT14 rs9679162 genotype-guided therapeutic strategy for advanced hepatocellular carcinoma: systemic or hepatic arterial infusion chemotherapy. Pharmacogenomics J. 2020;20:57-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 75. | Yeh CT, Liang KH, Lin CC, Chang ML, Hsu CL, Hung CF. A single nucleotide polymorphism on the GALNT14 gene as an effective predictor of response to chemotherapy in advanced hepatocellular carcinoma. Int J Cancer. 2014;134:1214-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 76. | Lyu N, Wang X, Li JB, Lai JF, Chen QF, Li SL, Deng HJ, He M, Mu LW, Zhao M. Arterial Chemotherapy of Oxaliplatin Plus Fluorouracil Versus Sorafenib in Advanced Hepatocellular Carcinoma: A Biomolecular Exploratory, Randomized, Phase III Trial (FOHAIC-1). J Clin Oncol. 2022;40:468-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 229] [Article Influence: 57.3] [Reference Citation Analysis (4)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0