Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. May 15, 2026; 18(5): 116882
Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116882
Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116882
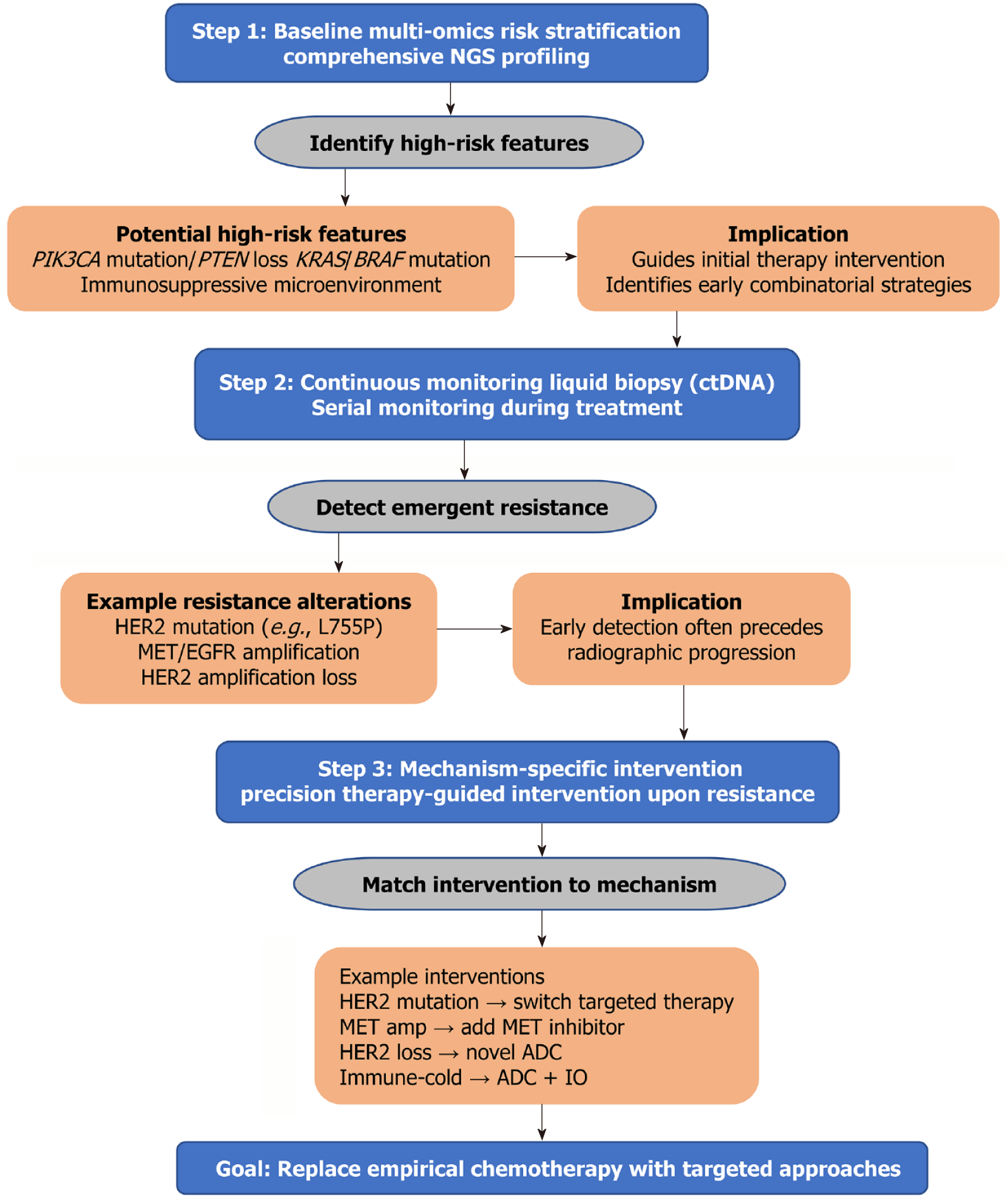
Figure 1 A dynamic, biomarker-informed decision model for overcoming resistance in human epidermal growth factor receptor 2-positive gastric cancer.
This flowchart outlines a proposed dynamic decision-model for overcoming therapy resistance. The model shifts the paradigm from a reactive, standardized sequence to a proactive, adaptive process of precision medicine. It operates through three core, iterative steps: (1) Initial multi-omics stratification: A comprehensive molecular profile at diagnosis (e.g., via next-generation sequencing) identifies high-risk features (e.g., PIK3CA mutations, PTEN loss) to guide first-line therapy intensity; (2) Longitudinal liquid biopsy surveillance: Periodic circulating tumor DNA analysis enables the early detection of emerging resistant clones and their mechanisms, often prior to radiographic progression; and (3) Mechanism-directed therapeutic rechallenge: Upon resistance detection, therapy is adaptively switched to a matched, mechanism-specific regimen (e.g., a tyrosine kinase inhibitor for an acquired human epidermal growth factor receptor 2 mutation), bypassing empirical chemotherapy. The model emphasizes continuous adaptation and highly individualized treatment. HER2: Human epidermal growth factor receptor 2; PI3K: Phosphatidylinositol 3-kinase; AKT: Protein kinase B; mTOR: Mammalian target of rapamycin; MET: Mesenchymal-epithelial transition factor; ER: Estrogen receptor; FGFR: Fibroblast growth factor receptor; EMT: Epithelial-mesenchymal transition; CDK: Cyclin-dependent kinase; CAV-1: Caveolin-1; ECM: Extracellular matrix; CD: Cluster of differentiation; TIL: Tumor-infiltrating lymphocyte; CTL: Cytotoxic T lymphocyte; TCR: T cell receptor; MHC: Major histocompatibility complex; PD-1: Programmed cell death 1; PD-L1: Programmed cell death ligand 1; PH: Potential of hydrogen.
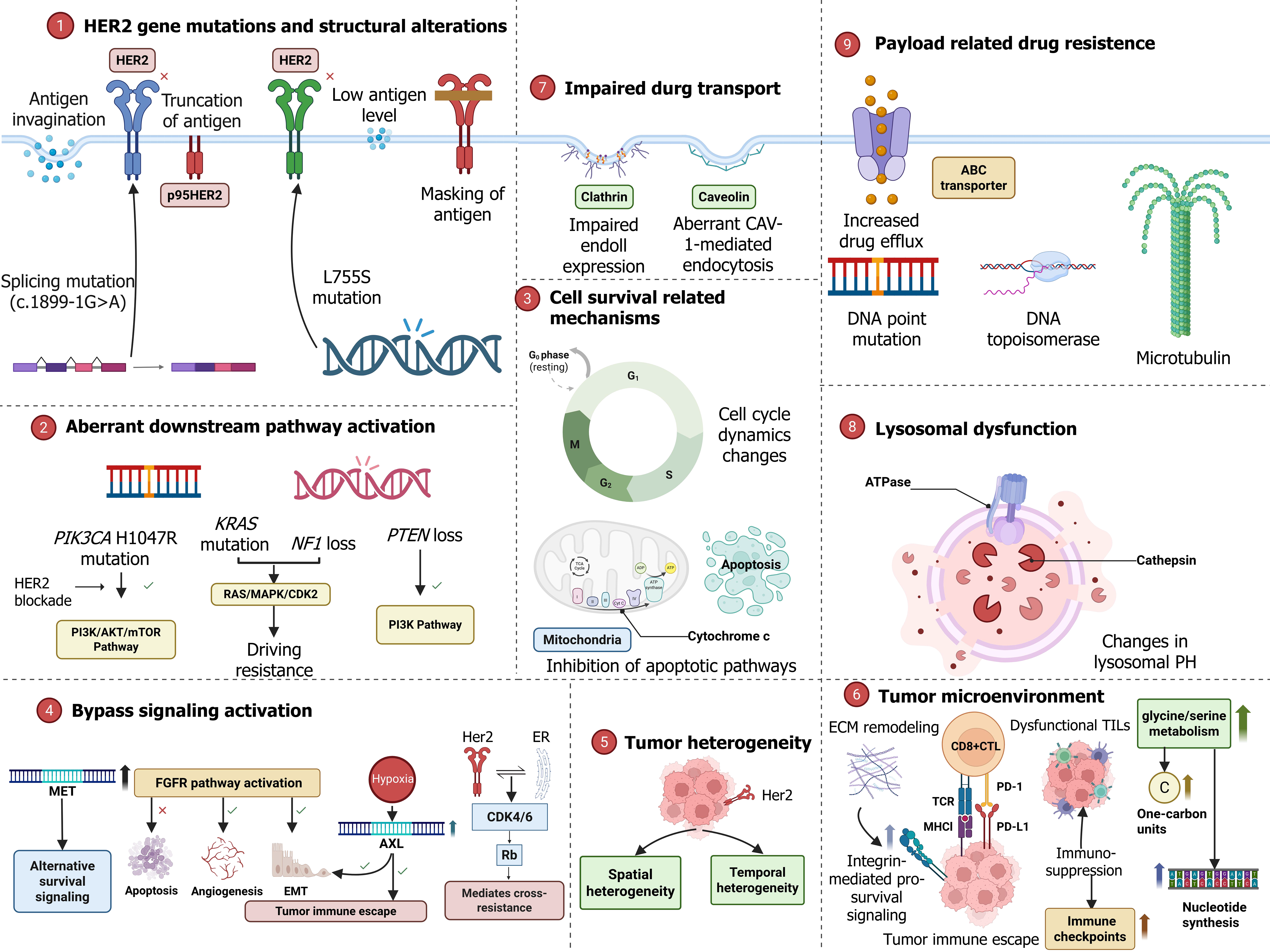
Figure 2 Resistance mechanisms to targeted and antibody-drug conjugate therapy in human epidermal growth factor receptor 2-positive gastric cancer a visual summary of the diverse mechanisms underlying treatment resistance.
(1) Human epidermal growth factor receptor 2 gene mutations and structural alterations; (2) Impaired drug transport; (3) Lysosomal dysfunction; (4) Payload-related drug resistance; (5) Cell survival related mechanisms; (6) Tumor microenvironment remodeling; (7) Bypass signaling activation; (8) Aberrant downstream pathway activation; and (9) Tumor heterogeneity. HER2: Human epidermal growth factor receptor 2; IHC: Immunohistochemistry; ISH: In situ hybridization; ctDNA: Circulating tumor DNA; CTC: Circulating tumor cell; IO: Immunotherapy; T-DXd: Trastuzumab deruxtecan; RC48: Disitamab vedotin; ADC: Antibody-drug conjugate; CAR-T: Chimeric antigen receptor T-cell im
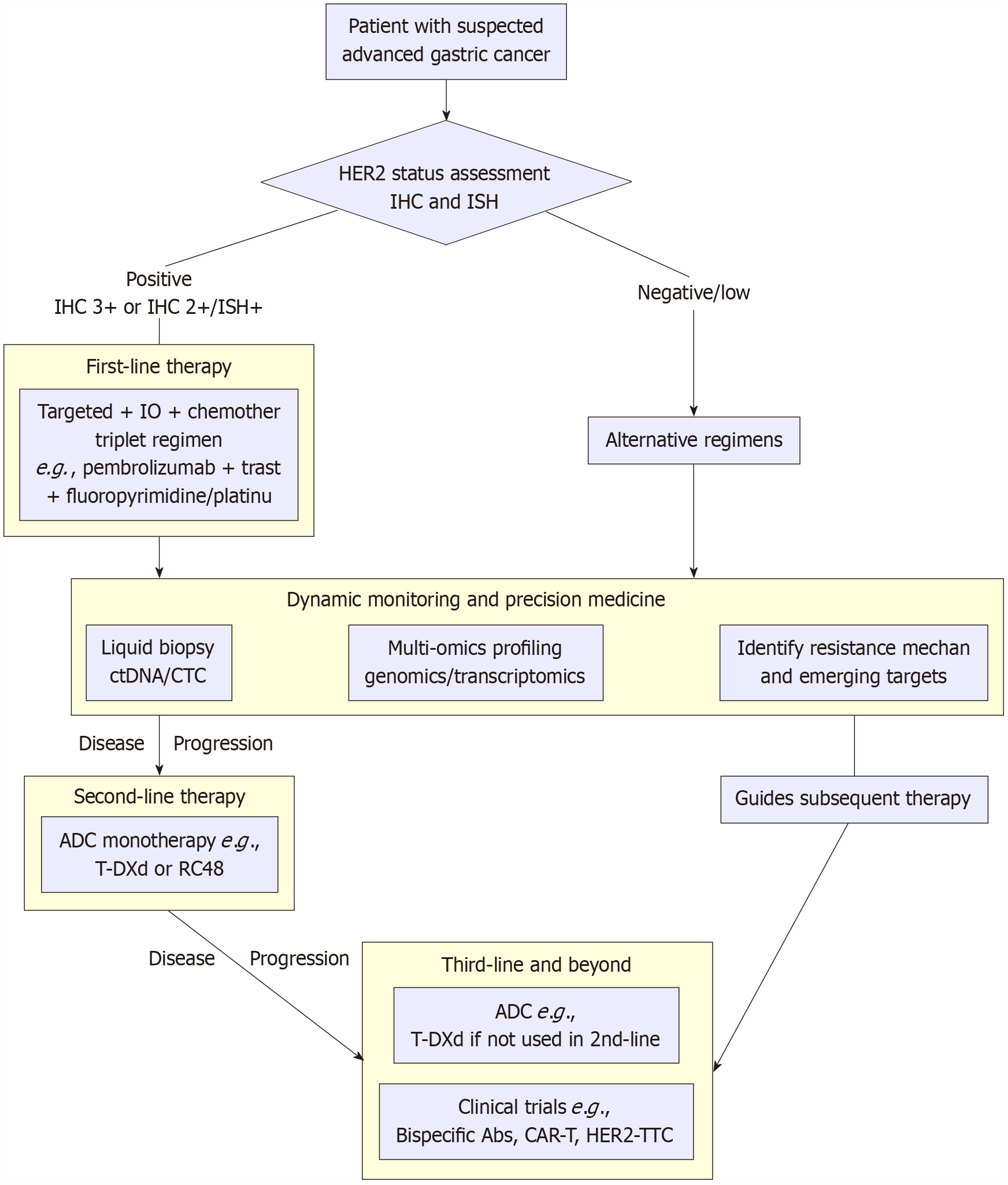
Figure 3 A precision diagnostic and therapeutic roadmap for human epidermal growth factor receptor 2-positive gastric cancer.
This flowchart delineates the integrated clinical decision-making pathway, from initial diagnosis and molecular subtyping through the sequential selection of therapies (first- to later-line) and ongoing dynamic monitoring. NGS: Next-generation sequencing; ctDNA: Circulating tumor DNA; HER2: Human epidermal growth factor receptor 2; MET: Mesenchymal-epithelial transition factor; EGFR: Epidermal growth factor receptor; IO: Immunotherapy; ADC: Antibody-drug conjugate.
- Citation: Xu JJ, Ni CX, Wang P, Qin LD, Xu JJ. Advancing human epidermal growth factor receptor 2-positive gastric cancer therapy: Toward targeted immunotherapy and antibody-drug conjugates. World J Gastrointest Oncol 2026; 18(5): 116882
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/116882.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.116882