Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. Mar 15, 2026; 18(3): 115679
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115679
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115679
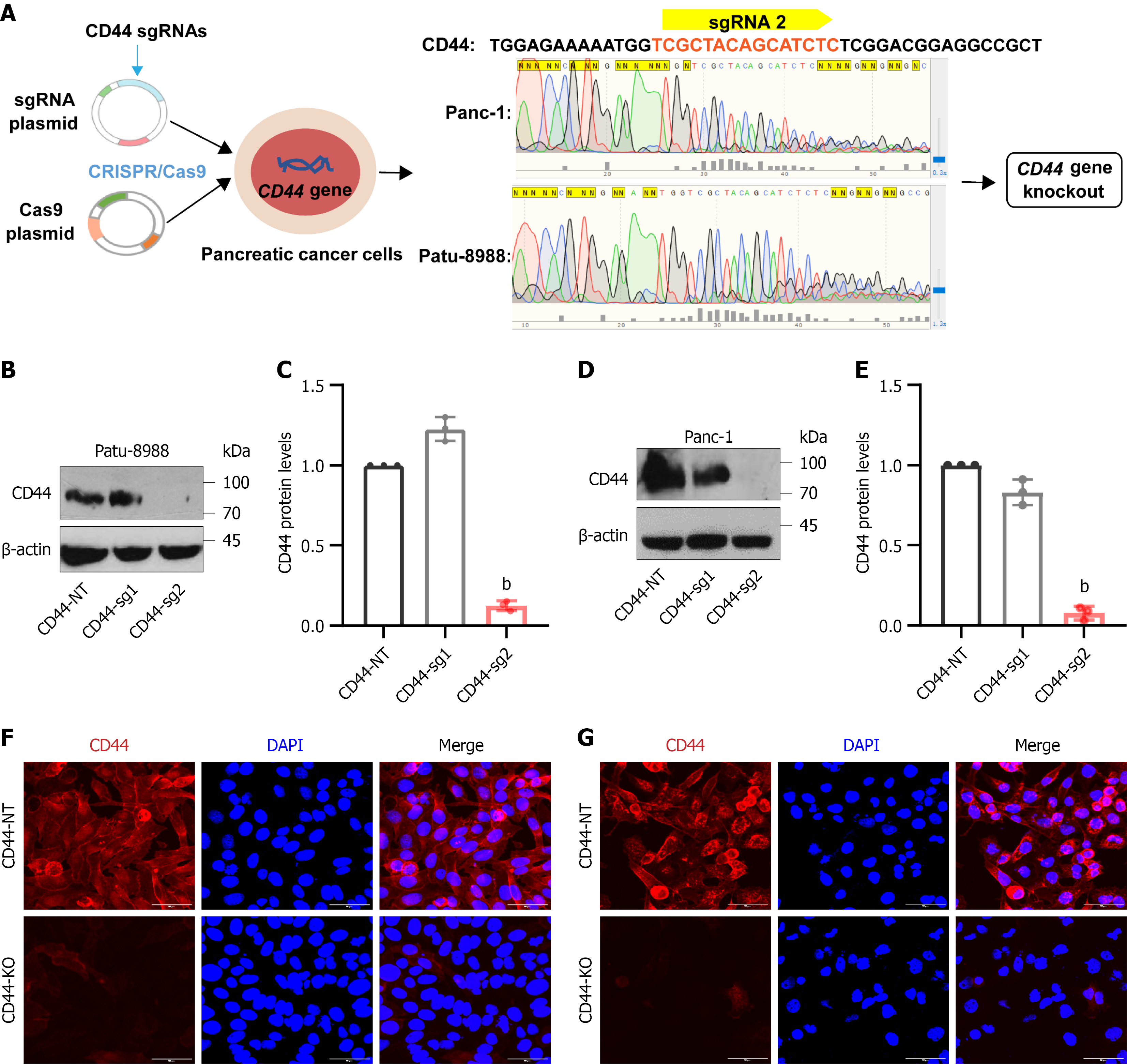
Figure 1 Establishment and verification of CD44 knockout pancreatic cancer cell lines.
Pancreatic cancer Patu-8988 and Panc-1 cells were stably transfected with Cas9-cDNA-vector. A: CD44 single-chain guide RNAs (sgRNAs) were cloned into a lentivirus vector, and the infectious sgRNAs-lentiviruses were prepared as described in the methods section. Cas9-positive pancreatic cancer Patu-8988 and Panc-1 cells were infected with CD44-sgRNA1 lentivirus, CD44-sgRNA2 lentivirus, or control sgRNA-lentivirus (CD44-NT). Further, Sanger sequencing confirmed the efficacy of CRISPR/Cas9 gene editing; B-E: CD44 knockout was verified by western blotting; F and G: Immunofluorescence staining, and confocal microscope analysis (600 ×), red: CD44, blue: DAPI, Merge: Both CD44 and DAPI. Ctrl: CD44-expressing cells, CD44-sgRNA1: CD44 gene single-chain guide RNA1, CD44-sgRNA2: CD44 gene single-chain guide RNA2. Notably, CD44 was almost completely knocked out by CD44-sgRNA2 in pancreatic cancer cells; accordingly, CD44-sgRNA2-mediated CD44 knockout pancreatic cells were named CD44-KO. Data in Figure 1B-E are shown as the mean ± SD of three independent replicates. bP < 0.01 vs CD44-NT in an unpaired t test.
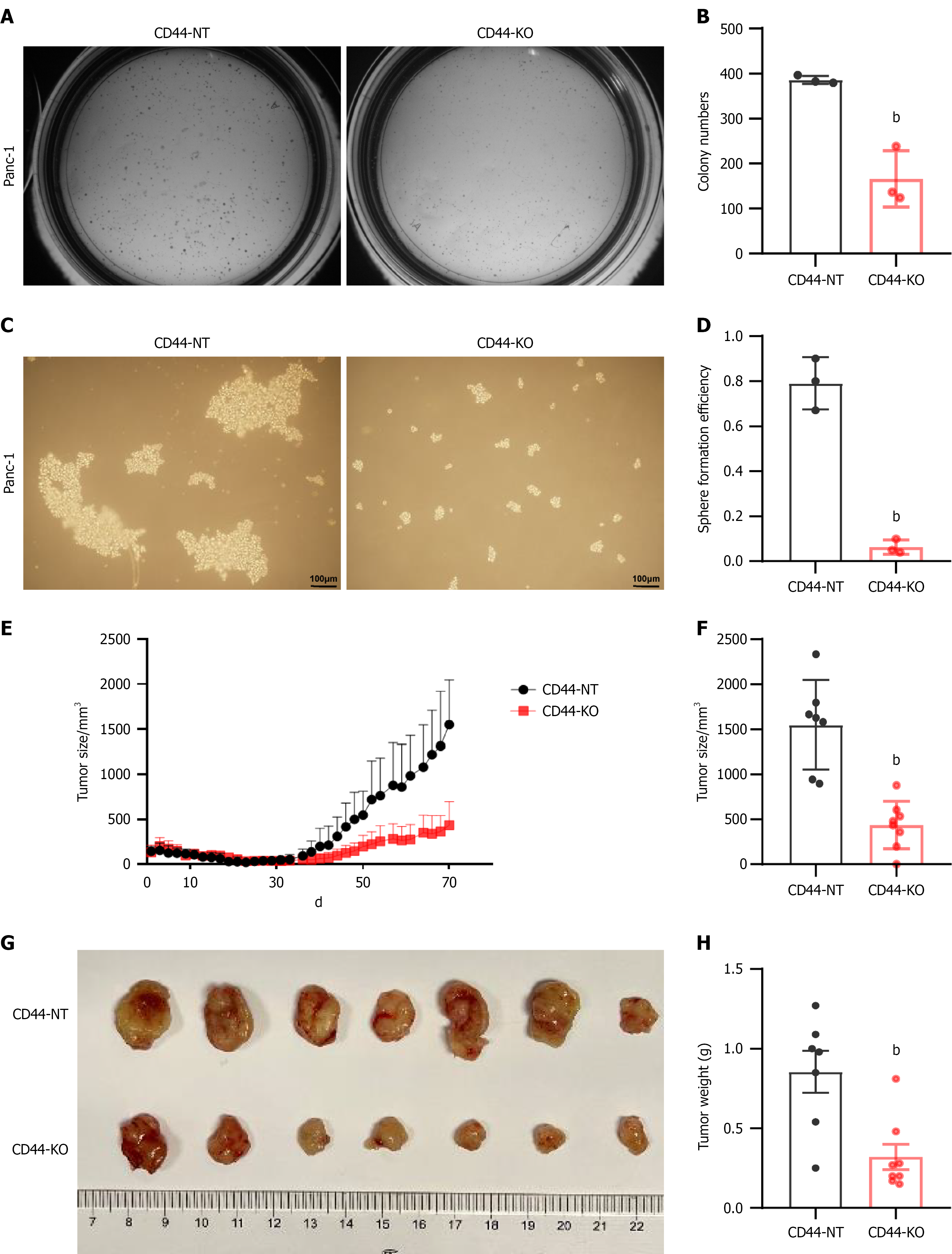
Figure 2 CD44 knockout effectively inhibits pancreatic cancer cell tumorigenesis and reduces cancer cell stemness.
A and B: The tumorigenic capability of CD44-KO and control CD44-expressing (Ctrl) pancreatic cancer Panc-1 cells was measured by a colony formation assay; C and D: The stemness of CD44-KO and Ctrl Panc-1 cells was assessed by a cell spherical formation assay; E and F: Additionally, the effect of CD44 gene knockout on tumor growth in vivo was examined in xenograft mice as described in the methods section. The tumor volumes in the CD44-KO and Ctrl groups of xenograft mice were measured every other day; G: The tumors in the CD44-KO and Ctrl groups were collected, photographed; H: Weighed, and statistically analyzed. Data in Figure 2A-D are shown as the mean ± SD of three independent replicates. bP < 0.01 vs CD44-NT in an unpaired t test.
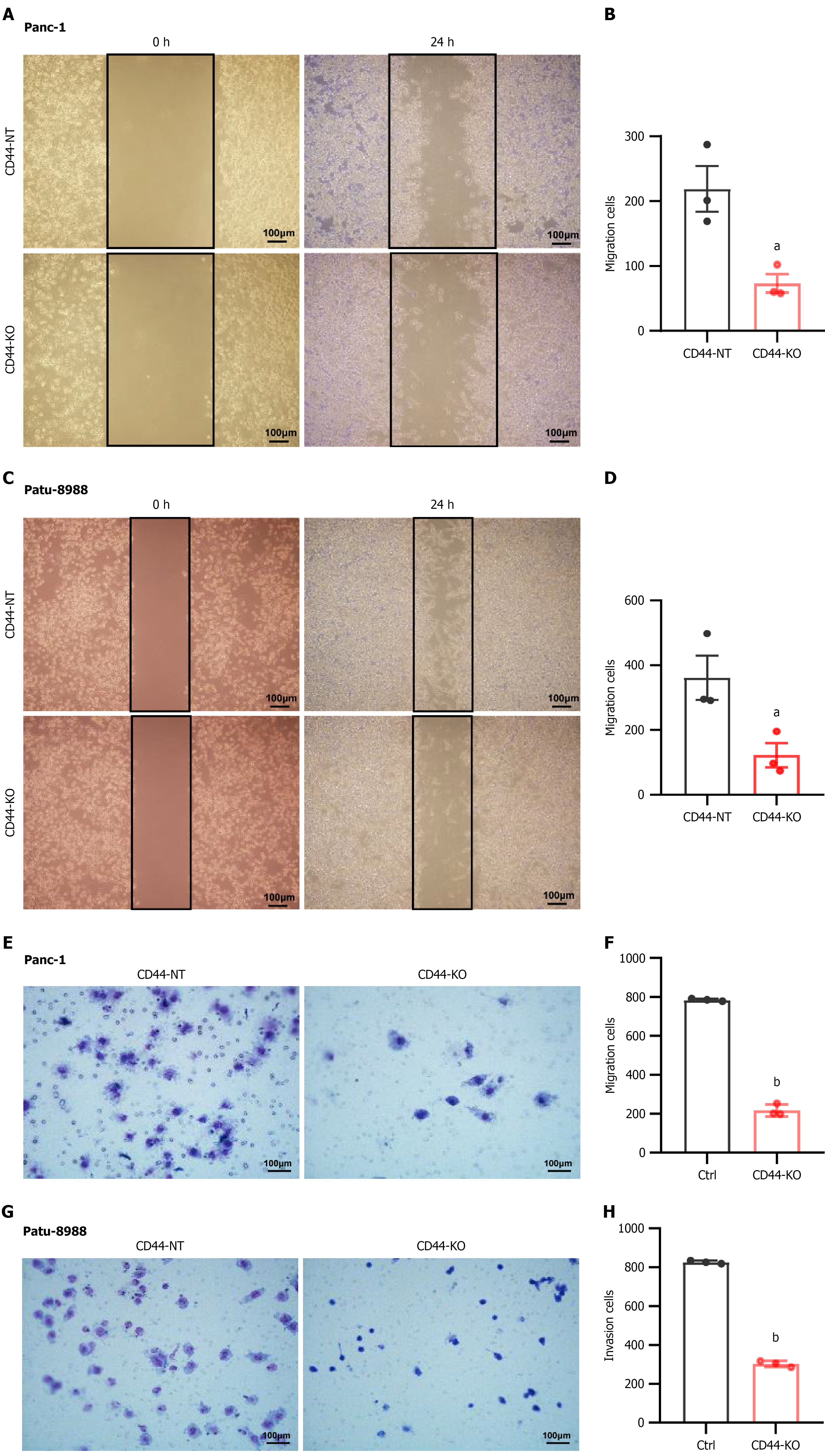
Figure 3 CD44 knockout inhibits pancreatic cancer cell migration and invasion.
Pancreatic cancer Patu-8988 and Panc-1 cells were cultured in six-well plates for a wound-healing assay. A-D: The cell monolayer was photographed at 0 hour and 24 hours of the assay, respectively, and the number of migrated cells was counted and statistically analyzed. In the cell invasion assay, Patu-8988 and Panc-1 cells were cultured in Transwell chambers pre-coated with 100 mL/L Matrigel and incubated for 24 hours; E-H: The invaded cells were stained by Giemsa staining and photographed under an orthostatic microscope (200 ×), and the number of invaded cells was counted and statistically analyzed. Data are shown as the mean ± SD of three independent replicates. aP < 0.05, bP < 0.01 vs CD44-NT in an unpaired t test.
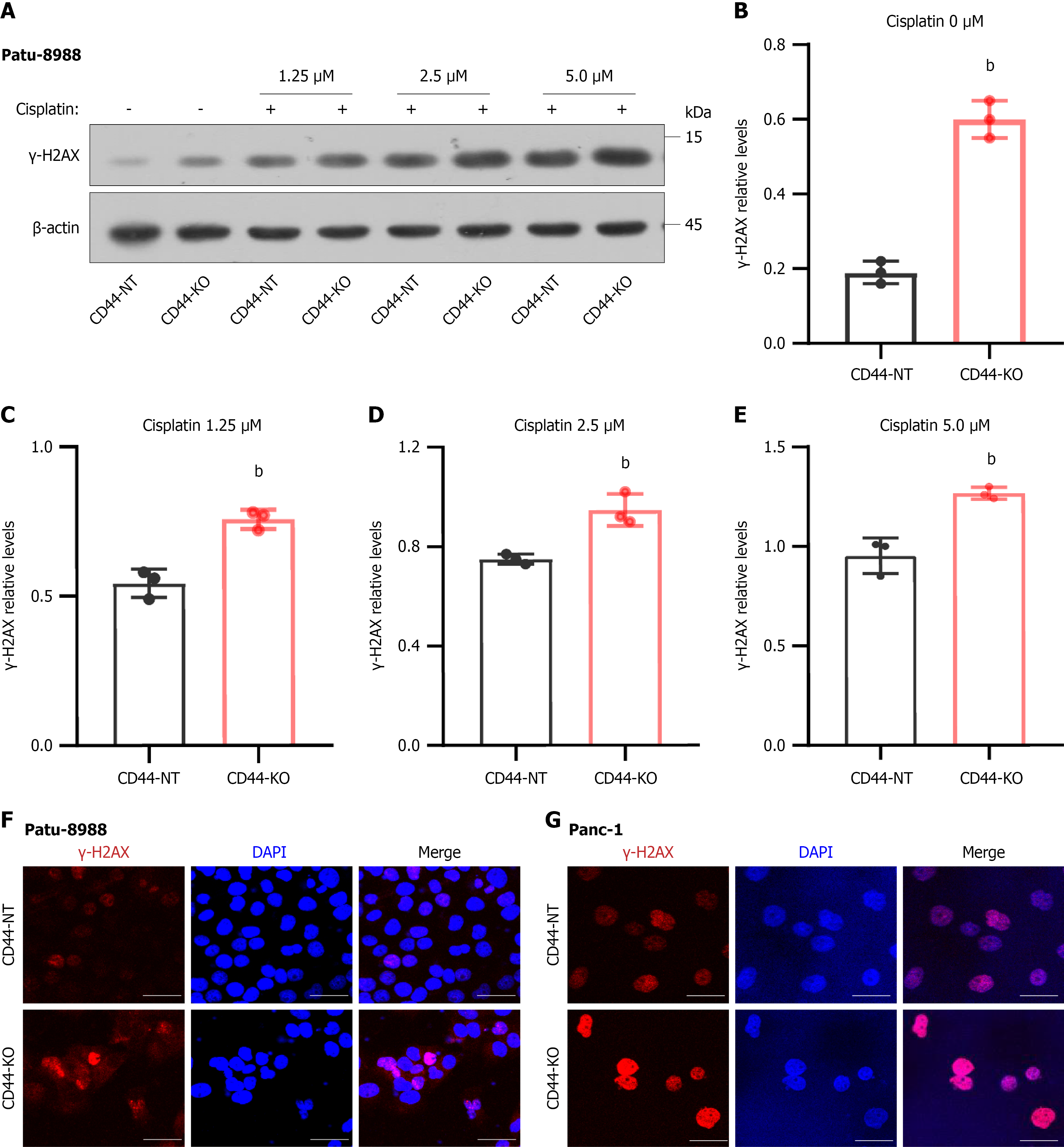
Figure 4 CD44 knockout increases DNA damage and enhances cisplatin-induced DNA cleavage in pancreatic cancer cells.
A: DNA damage in pancreatic cells was assessed by the measurement of the protein level of γ-H2AX, a widely used DNA breakage marker, with western blotting. CD44-KO and CD44-expressing Panc-1 cells (Ctrl) were incubated with the anti-cancer drug cisplatin at concentrations of 0-5 μM; B-E: CD44-KO induced an increase in the γ-H2AX level, even in the absence of cisplatin, and further enhanced DNA damage in the presence of cisplatin; F and G: CD44-KO and CD44-expressing Panc-1 cells were first treated with either 0 μM or 5 μM cisplatin for 48 hours, then stained with γ-H2AX-specific antibody and FITC-secondary antibody, and imaged with a confocal microscope (800 ×); red: γ-H2AX, blue: DAPI, Merge: γ-H2AX and DAPI. Data in Figure 4A-E are shown as the mean ± SD of three independent replicates. bP < 0.01 vs CD44-NT in an unpaired t test.
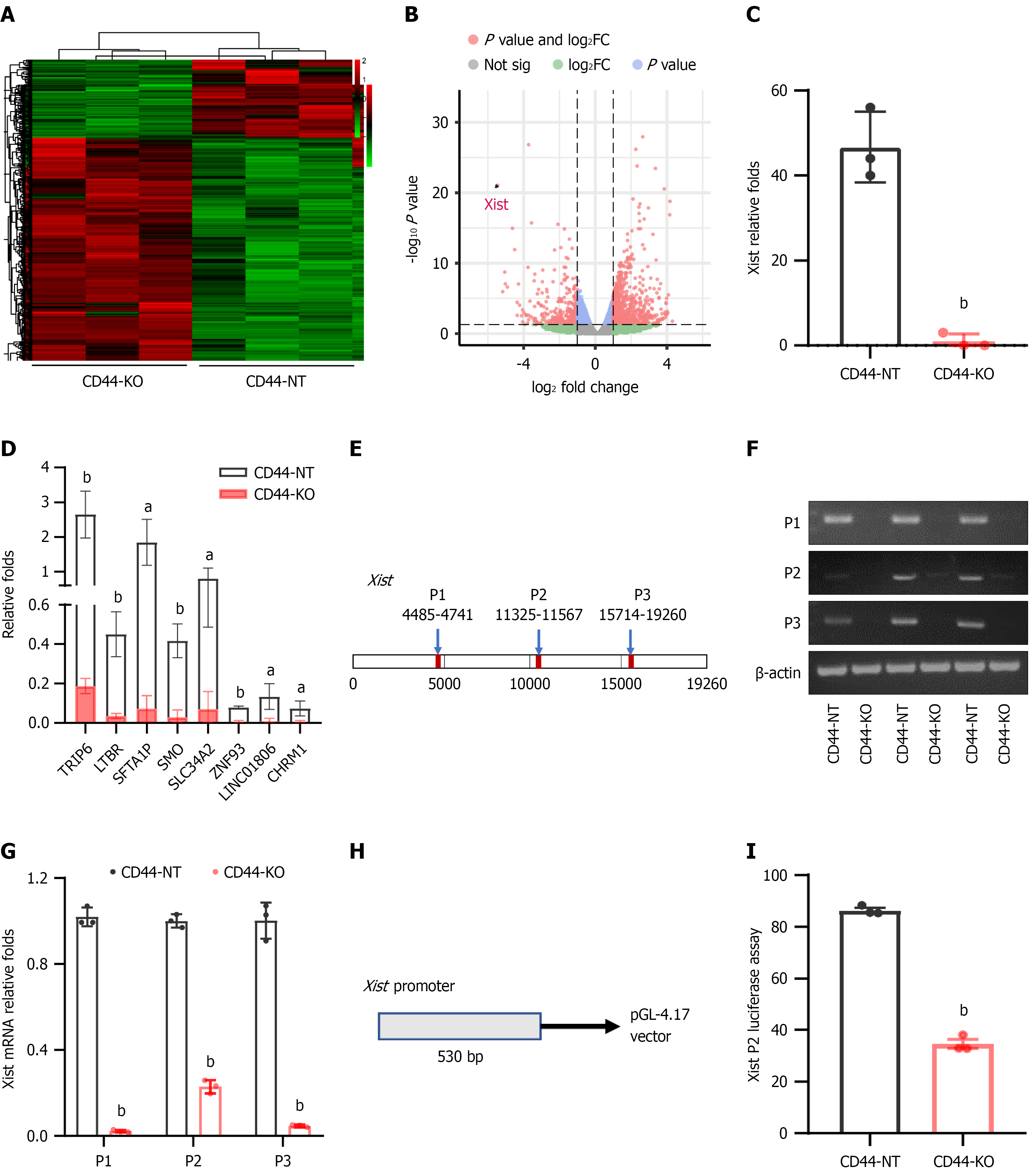
Figure 5 CD44 knockout markedly reduces the expression of oncogenic X-inactive-specific transcript and various other tumorigenic genes in pancreatic cancer cells.
A and B: The gene expression profiles in three independent sets of CD44-KO and control CD44-expressing Panc-1 cells were analyzed by RNA sequencing (RNA-Seq), and a volcano map of the gene expression profiles is depicted in Figure 5B, red: Up-regulated genes; green: Down-regulated genes; black: Gene expression levels did not significantly change; C: RNA-Seq showed that CD44 knockout markedly reduced the expression levels of oncogenic X-inactive-specific transcript (Xist); D: The expression levels of various other tumorigenic genes in CD44-KO Panc-1 cells were also significantly diminished compared to those in control cells; E: To further confirm CD44 knockout-mediated Xist expression, three Xist primers (P1, P2, and P3) were designed at the indicated position of the long non-coding RNA Xist (19260 bp); F and G: Xist expression levels in CD44-KO and control CD44-expressing Panc-1 cells were detected by real-time PCR and real-time quantitative PCR, respectively; H: To study the effect of CD44 on Xist gene transcription, a promoter region DNA fragment (530 bp) of the Xist gene was cloned into the pGL-4.17-luciferase vector, and the construct was used to transfect CD44-KO and CD44-expressing Panc-1 cells, respectively; I: Luciferase activity in these cells was determined and statistically analyzed. Data are shown as the mean ± SD of three independent replicates. aP < 0.05, bP < 0.01 vs CD44-NT in an unpaired t test.
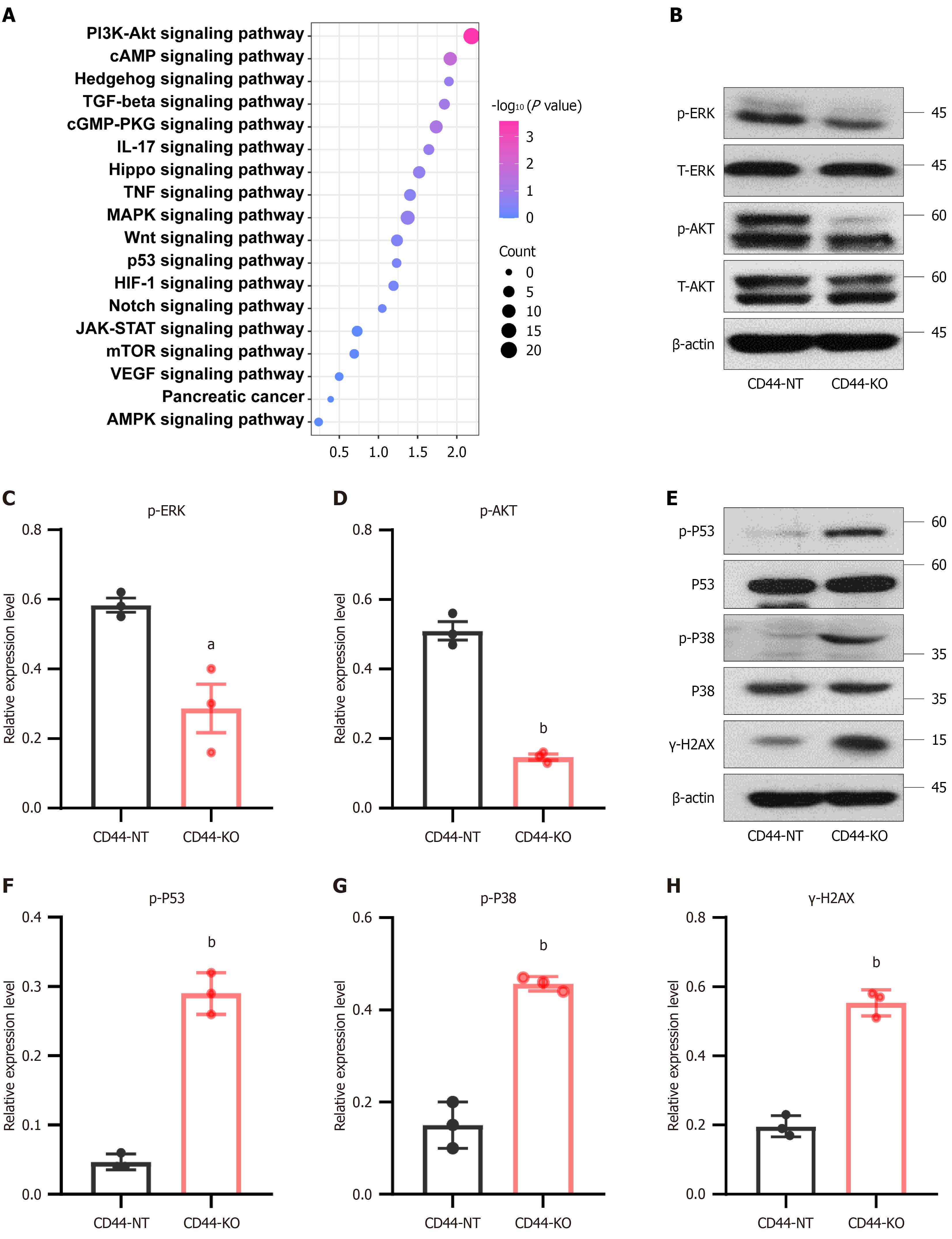
Figure 6 CD44 knockout inhibits the tumorigenic ERK and AKT signaling pathways and enhances the tumor-suppressive p38-p53 sig
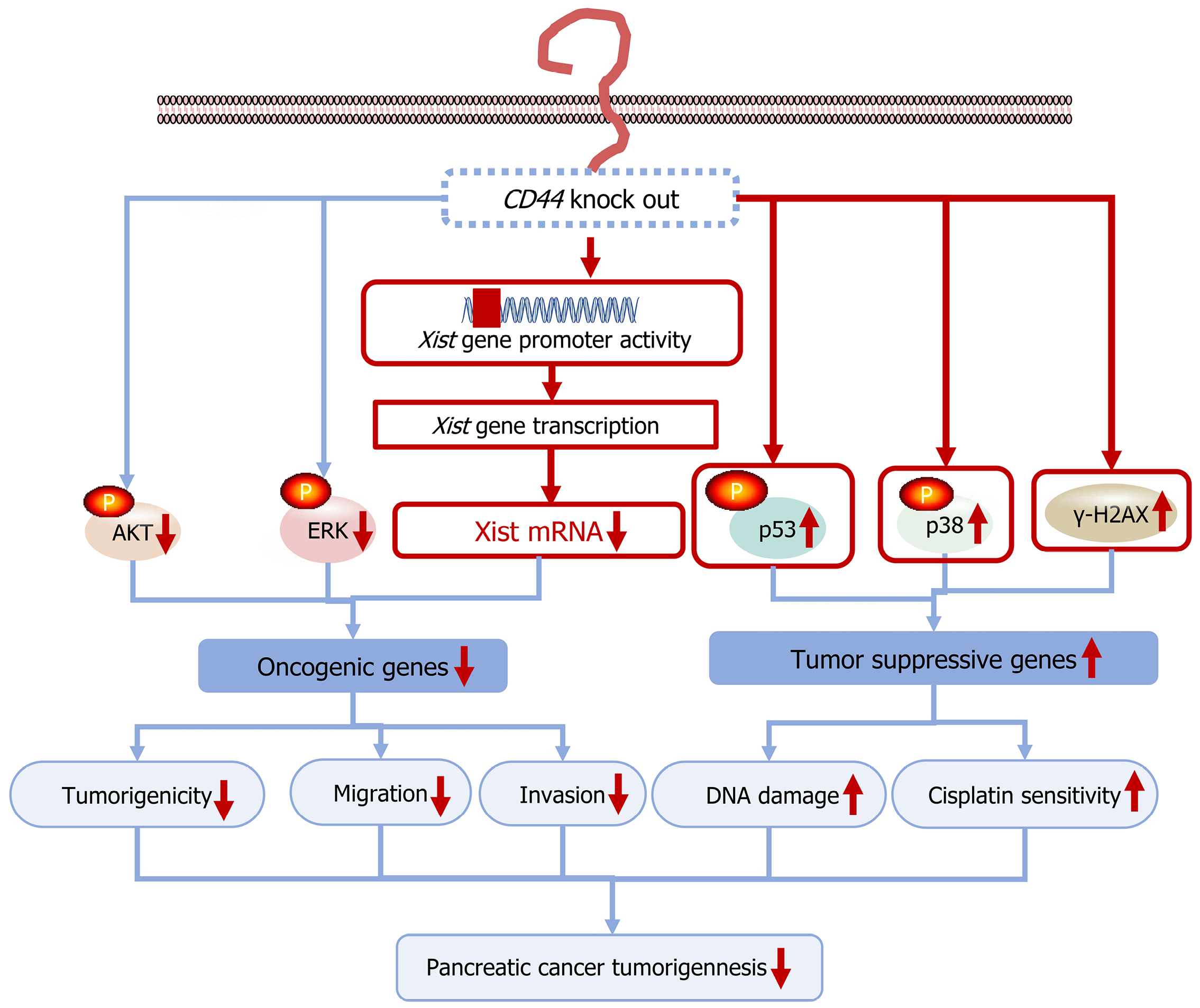
Figure 7 Graphical abstract demonstrating how CD44 knockout inhibits pancreatic cancer cell tumorigenesis and is a new option for tar
- Citation: Liu YX, Zheng NN, Wang XX, Zhou QS, Meng M. Oncogenic CD44 is essential for pancreatic cancer tumorigenesis: A novel targeted therapeutic strategy. World J Gastrointest Oncol 2026; 18(3): 115679
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/115679.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.115679