Published online May 16, 2026. doi: 10.4253/wjge.v18.i5.119587
Revised: April 4, 2026
Accepted: April 21, 2026
Published online: May 16, 2026
Processing time: 101 Days and 4.2 Hours
Biliary complications are the Achilles heel of the living donor liver transplantation (LDLT) and compromise the graft survival and the overall survival. Anastomotic biliary strictures (ABS) are the most common and challenging complication within this category. While endoscopic retrograde cholangiopancreatography (ERCP) serves as the standard first-line treatment modality to navigate the strictures, intraductal cholangioscopy, percutaneous transhepatic cholangiography (PTC), and surgical approaches are reserved for the technically challenging cases where standard ERCP remains unsuccessful.
To investigate the prevalence of ABS across various biliary reconstructions in LDLT recipients and analyze the outcomes of endoscopic interventions.
This retrospective cohort study was conducted between March 2019 and De
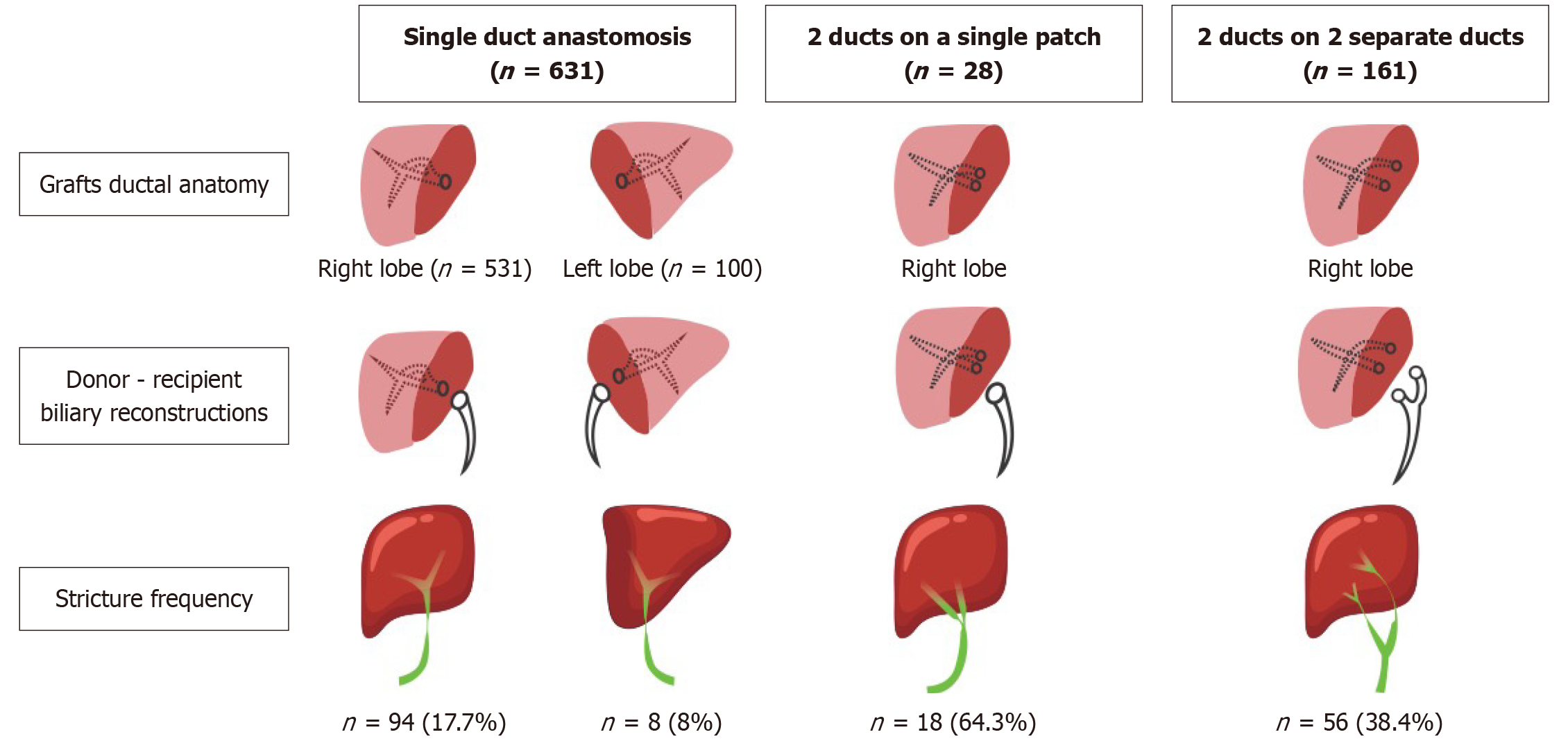
Frequency of anastomotic biliary stricture was 16.2% (102 of 631), in single duct-duct anastomosis, 34.8% (56 of 161) in two duct-duct anastomosis, and 64.3% (18 of 28) where 2 ducts were reconstructed on a single patch. The dual duct reconstruction exhibited a higher risk for ABS development vs single duct anastomosis (odds ratio = 3.34, 95%CI: 2.33-4.78). A total of 792 ERCPs were performed in 176 patients. Only 114 (65%) cases completed the therapeutic course and were included in the treatment outcome analysis. Stricture resolution with endotherapy alone was achieved in 90.2%. PTC improved the overall stricture resolution rates to 99.1%. Stricture recurred among 15 (13.3%) patients. Procedure-related complications included cholangitis (2.4%), pancreatitis (2.3%), and bleeding in (1.52%); no perforation or death occurred.
In the LDLT setting, dual duct biliary reconstruction is strongly associated with ABS. ERCP is a highly effective, safe, and preferred modality for managing post-LDLT ABS.
Core Tip: Anastomotic biliary strictures (ABS) remain the most common and challenging biliary complication after living donor liver transplantation (LDLT). Dual duct biliary reconstruction in the LDLT setting is associated with a significantly high risk of stricture incidence and recurrence. Endoscopic retrograde cholangiopancreatography complemented with intraductal cholangioscopy (IDC) has emerged as a highly effective and safe treatment modality in managing complex biliary strictures. Knowledge and understanding of biliary reconstruction are critical in defining treatment strategies to achieve optimal outcomes. percutaneous transhepatic cholangiography is the preferred modality when endotherapy remains unsuccessful despite using IDC. Surgery serves as a rescue strategy when percutaneous and endoscopic approaches remain unsuccessful to address the ABS.
- Citation: Aujla UI, Syed IA, Malik AK, Khan MMZ, Rafi K, Dar FS. Endoscopic management of post-living donor liver transplant anastomotic biliary strictures: A quaternary care transplant center experience. World J Gastrointest Endosc 2026; 18(5): 119587
- URL: https://www.wjgnet.com/1948-5190/full/v18/i5/119587.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i5.119587
Liver transplant is an effective curative approach for selected patients with end-stage liver disease and acute liver failure, and has shown excellent survival rates. Deceased donor liver transplant (DDLT) has been the gold standard and preferred modality for several decades. However, owing to the continued shortage of deceased donor organs and the growing number of patients on the waiting list, living donor liver transplant (LDLT) has become an excellent alternative option with comparable survival rates[1]. Although LDLT is a more complex and technically challenging procedure compared to DDLT, it still provides excellent graft and survival outcomes. A recent meta-analysis revealed better 1-, 3- and 5-year survival outcomes in LDLT as compared to DDLT[2].
Although there have been significant advances in surgical methods, immunosuppressive medications, and post-operative management, which have improved the outcomes of LDLT recipients to a great extent, surgical complications continue to pose a challenge. Literature review shows that LDLT recipients experience higher surgical complications as compared to DDLT, with a higher frequency of biliary complications[3]. A meta-analysis demonstrated that the risk of developing biliary complications is higher for LDLT as compared to DDLT, with an odds ratio of 1.96 (95%CI: 1.56-2.47)[4]. Biliary strictures, bile leaks, biloma, biliary casts, and bile duct stones represent the spectrum of biliary complications. With reported rates generally ranging from 13% to 36%, the overall incidence of biliary strictures is higher after LDLT than DDLT[5].
The majority of anastomotic biliary strictures (ABS) occur within the first year of liver transplant surgery, with a mean time of diagnosis within 5-8 weeks. The ABS occurring within the first 3 months of transplant is due to surgical and technical issues, whereas the late ABS, which occur after 3 months, are due to localized ischemia and fibrotic healing processes[5,6]. Several risk factors are linked to the development of ABS and include advanced donor and recipient age, hepatic artery thrombosis, use of multiple biliary anastomoses, and technical issues such as tension at the anastomosis or excessive electrocautery use for biliary ductal bleeding control. Additional contributing factors are prolonged cold and warm ischemia times, small bile duct caliber of the graft, preceding bile leakage, and suboptimal surgical technique[6,7].
ABS can significantly impact long-term patient outcomes, diminish quality of life & lead to graft loss or even mortality. The spectrum of clinical presentation is diverse and includes cholangitis, fever, jaundice, pruritus, anorexia, stomach discomfort, and a cholestatic pattern of liver enzymes[6]. Historically, surgical repair or re-transplantation were the mainstay of managing these complications, carrying considerable risk of morbidity and mortality. However, with the advancements in endotherapy and interventional radiology, there has been a revolutionary change in the management of post-transplant biliary strictures[8]. Endoscopic retrograde cholangiopancreatography (ERCP) has emerged as the preferred first-line modality for the management of ABS in post-LDLT patients, providing a less invasive and highly effective therapeutic modality[9]. Moreover, the advent of intraductal cholangioscopy (IDC) has significantly enhanced the therapeutic landscape of endoscopic management of ABS by providing direct visualization and therapeutic inter
This study seeks to assess the occurrence of ABS among the recipients of LDLT, examine the success of ERCP in resolving these strictures, and highlight the evolving importance of IDC as a vital modality for managing complex strictures. This study strives to disseminate valuable insights derived from a leading liver transplant center in Southeast Asia.
This was a retrospective cohort study with prospectively controlled database conducted at Pakistan Kidney and Liver Institute and Research Center, a quaternary care transplant center. Medical records of the enrolled patients were reviewed from March 2019 to December 2024, in accordance with the STROBE recommendations for observational studies.
The inclusion and exclusion criteria were determined a priori.
Inclusion criteria: All patients who developed ABS following LDLT during the study period and completed treatment interventions were enrolled in the study.
Exclusion criteria: (1) Non-ABS; (2) Patients who could not complete the therapeutic course or had ongoing treatment interventions were not included in the outcome analysis; and (3) Patients who died or were lost to follow-up before completing the treatment cascade were excluded from outcome analysis
The non-probability consecutive sampling method was used, in which all patients who fulfilled the criteria of eligibility were selected throughout the study period. A total of 176 patients were selected based on the completeness and availability of medical records. A total of 114 patients completed the therapeutic course and were included in the outcome analysis.
Clinical, laboratory, endoscopy, and radiological data were collected retrospectively from hospital electronic medical records, using a standardized data extraction form.
Demographic variables: (1) Age; (2) Sex; and (3) Comorbidities.
Clinical variables: (1) Indications for liver transplant; (2) Type of graft; (3) Type of biliary anastomosis; (4) Time from liver transplant to first ERCP time; and (5) Time to stricture recurrence after initial resolution.
Intervention-related variables: (1) Type of procedure or treatments provided (ERCP/IDC/PTC/surgical revision); (2) Number of ERCPs; (3) ERCP-related complications; and (4) Prophylactic pancreatic duct stenting.
Outcome variables: (1) ABS incidence across different types of biliary reconstructions; (2) ABS resolution and recurrence rates; and (3) Frequency of ERCP-related complications.
Data extraction was performed by two trained investigators, and a third independent reviewer cross-verified the data to minimize extraction errors.
All study variables were predefined before data collection.
Primary objective: To determine the safety and efficacy of ERCP in managing ABS after LDLT.
Secondary objective: To compare the ABS rates across various biliary ductal reconstruction techniques performed in the setting of LDLT.
Primary outcomes including: (1) The ABS resolution rates after scheduled ERCP-guided treatment sessions for 12 months; (2) The ABS stricture recurrence rates and time to recurrence after initial resolution; and (3) Post-ERCP procedure-related complications.
Secondary outcomes including Incidence of ABS across different types of biliary reconstructions during the follow-up period after LDLT.
Operational definitions were applied consistently. Clinically, ABS was suspected based on the presence of cholestatic liver enzymes, either detected in an asymptomatic LDLT recipient during routine evaluation or often accompanied by symptoms like jaundice, fever, and pruritus. Ultrasound served as the initial imaging modality to diagnose the presence of an ABS, while the definitive confirmation was obtained through cross-sectional imaging such as magnetic resonance cholangiopancreatography or triphasic computed tomography scans. The hallmark radiological feature of ABS included a subtle narrowing or an abrupt cutoff at the site of biliary anastomosis with resultant upstream intrahepatic biliary ductal dilatation.
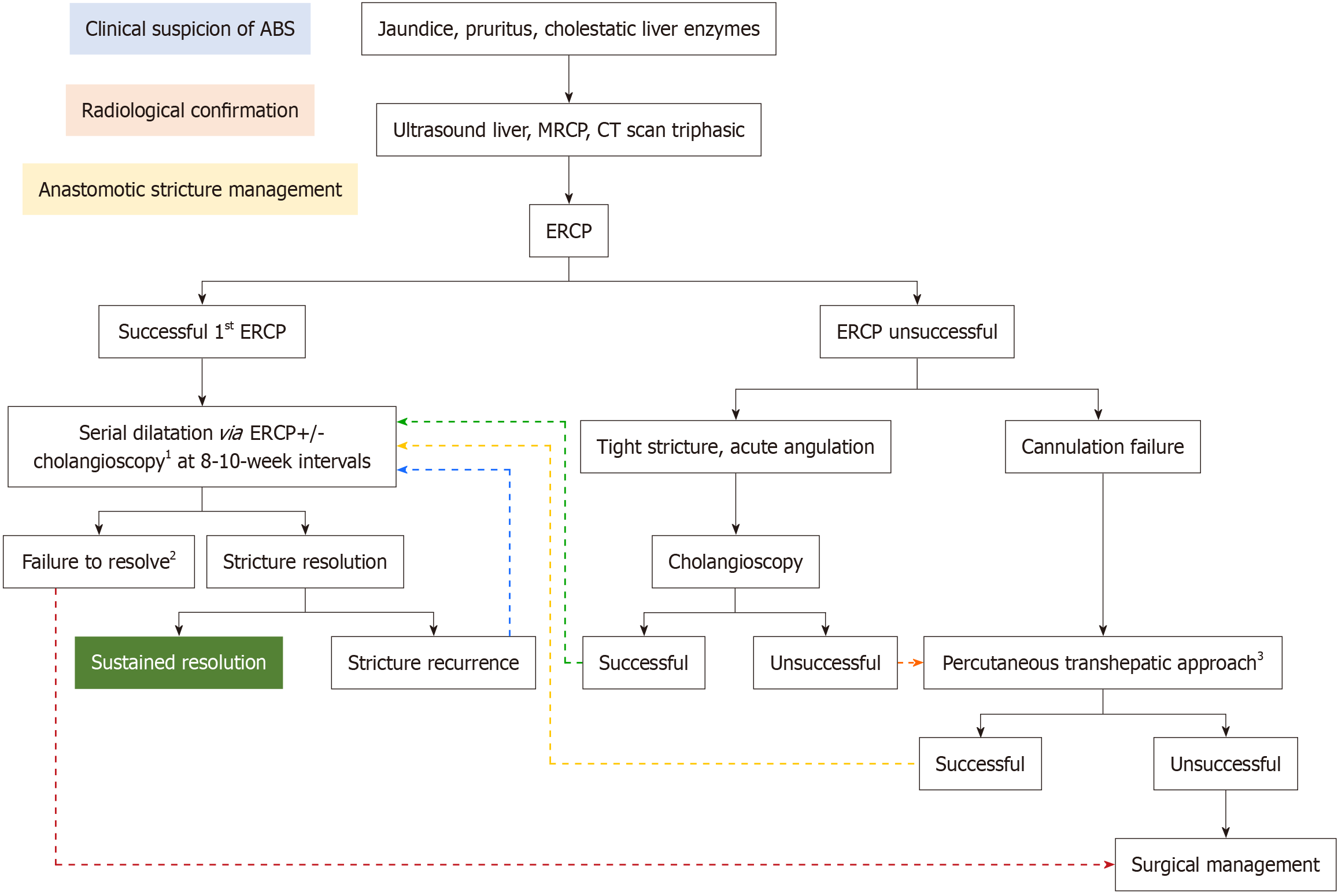
ERCP was the first-line treatment modality for the management of ABS using an Olympus 190 duodenoscope (Olympus Corporation). A cholangiogram mapped the stricture, which was dilated with biliary dilatation balloons or Soehendra dilators, and then stented (7-12 French plastic biliary stents). Figure 1 shows various dilators utilized to dilate the ABS. All patients received pre & post procedural antibiotics as per institutional guidelines and were subsequently scheduled for stent exchanges every 8-12 weeks. Technical success was defined as successful biliary cannulation and traversal of the ABS by placement of the guidewire across the stricture, followed by dilatation of the ABS and deployment of the biliary stent(s). Clinical success was defined as significant improvement/normalization of cholestatic liver enzymes and resolution of symptoms. ERCP failure was defined as the inability to cannulate the bile duct, traverse the stricture, or deploy the stent across the stricture. Among patients where ERCP remained unsuccessful despite successful biliary cannulation, utilization of IDC for direct visualization and assessment of ABS was the preferred option to facilitate the stricture management during the same session. IDC not only served as an adjunct tool, allowing traversal of complex strictures, permitting access and drainage of sectoral biliary branches, but also remained instrumental in providing direct confirmation of stricture resolution. PTC was reserved for cases with biliary cannulation failure, inability to traverse the stricture, and incomplete sectoral duct drainage despite utilization of IDC. Surgical revision was undertaken as a final step, applied only when both initial endotherapy and PTC were unsuccessful or when strictures remained unresolved following at least 12 months of repeated ERCPs ± PTC as an adjunct when indicated. Figure 2 illustrates the management algorithm of ABS at our center.
Stricture resolution was confirmed by ERCP with an occlusion choalngiogram demonstrating disappearance of the stricture waist, spontaneous drainage of contrast from the proximal biliary tree, and absence of resistance to trawl a balloon catheter of appropriate size. IDC was not routinely performed to document stricture resolution. It was reserved for selected cases only where ERCP findings were equivocal to demonstrate complete stricture resolution, persistent cholestatic pattern of liver enzymes despite apparent resolution of ABS on cholangiogram, suspicion of early stricture recurrence, or among cases with complex biliary anatomy and high risk of recurrence.
Post-ERCP complications assessed included bleeding, cholangitis, perforation, and pancreatitis, as well as procedure-related mortality. These complications were diagnosed according to ESGE 2020 guidelines for post ERCP adverse events[13]. Post-ERCP pancreatitis (PEP) was defined as the development of abdominal pain accompanied by a serum amylase or lipase level exceeding three times the upper limit of normal at 24 hours post ERCP, requiring hospitalization. Significant bleeding was considered present if there was clinical evidence, such as hematemesis or melena, or a hemoglobin drop greater than 2 g/dL within 7-10 days following ERCP. Perforation was diagnosed based on radiological confirmation or direct visualization during the ERCP procedure. Cholangitis was defined as the presence of clinical signs of infection, such as fever > 38 °C for more than 24 hours and cholestasis following ERCP. High-risk patients for PEP were defined as patients in whom the double wire technique was used for biliary cannulation and those with contrast opacification of the pancreatic duct.
Selection bias was minimized by including all consecutive eligible patients. Information bias was reduced through standardized data collection procedures. Missing or incomplete data were handled by complete-case analysis, with exclusion of patients who had incomplete follow-up, died before completing therapy, or were still undergoing treatment at the time of analysis. To minimize the observer variability, interventions were performed by the experienced operators.
Data was analyzed using IBM SPSS version 27. Descriptive statistics were used to find out various n (%). Quantitative variables were assessed for normality and expressed as mean ± SD, or median [interquartile range (IQR)], depending on distribution. Nominal data were analyzed using the t-test, where 2 groups were involved, and for the comparison of more than 2 groups, the ANOVA test was used. Categorical data was compared using the χ2 test or Fisher's exact test. Variables that yielded a P < 0.05 were considered statistically significant.
Ethical approval was obtained from the Institutional Review Board of Pakistan Kidney and Liver Institute and Research Center (Approval No. 00672025). Due to the retrospective nature of the research and utilization of anonymized data, the requirement for informed consent was waived.
A total of 820 patients underwent LDLT between March 2019 and December 2024. Single-duct to duct biliary recon
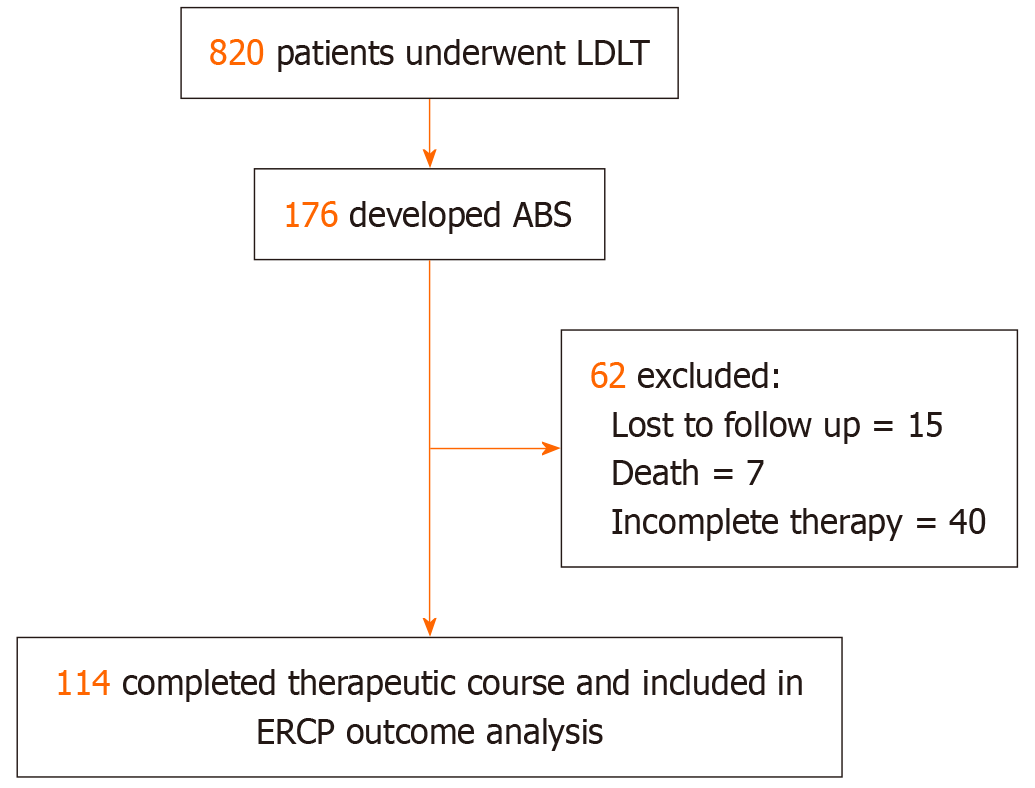
Out of 820 LDLT recipients, 176 (21.5%) patients developed ABS and were included in the study and subsequently analyzed to extract the relevant data. The mean age was 51.5 ± 11 years (range 14-71 years). The majority (74%, n = 130) were males. Diabetes mellitus (27%) and hypertension (19%) were the most prevalent comorbidities observed in the cohort. The median time to ABS development was 5 months (IQR 2.5-8.3 months). Table 1 summarizes the clinical characteristics of the study cohort.
| Parameter | n (%) |
| Demographics | |
| Total | 176 |
| Male | 130 (74) |
| Female | 46 (26) |
| Male/female | 2.82/1 |
| Age distribution (year) | |
| 14-18 | 2 (1) |
| 18-50 | 67 (38) |
| > 50 | 107 (61) |
| Co-morbid conditions | |
| Diabetes mellitus | 47 (27) |
| Hypertension | 33 (19) |
| Ischemic heart disease | 4 (2) |
| Chronic obstructive pulmonary disease | 4 (2) |
| Indication for liver transplant | |
| Hepatitis C virus | 106 (60.23) |
| Cryptogenic cirrhosis | 21 (11.93) |
| Hepatitis B virus | 17 (9.66) |
| Metabolic dysfunction associated steatohepatitis | 8 (4.55) |
| Budd-Chiari syndrome | 7 (3.98) |
| Hepatitis B & delta virus coinfection | 5 (2.84) |
| Wilson’s disease | 3 (1.7) |
| Alcoholic liver disease | 3 (1.7) |
| Primary biliary cholangitis | 2 (1.1) |
| Hepatitis B & C virus coinfection | 2 (1.1) |
| Primary hyperoxaluria | 2 (1.1) |
| Hepatocellular carcinoma | 37 (38.1) |
| Type of graft with anastomotic biliary stricture | |
| Right lobe | 168 (95.45) |
| Left lobe | 8 (4.55) |
| Type of biliary reconstructions among 820 recipients | |
| Single duct-duct anastomosis | 631 (77) |
| Right lobe | 531 (84) |
| Left lobe | 100 (16) |
| Two separate duct – duct anastomosis | 161 (20) |
| Two ducts on a single patch | 28 (3) |
Of the 176 patients included in the study, 15 were lost to follow-up, and 7 died during the course of observation. At the time of data analysis, 40 patients were still undergoing endotherapy sessions. Hence, these 62 patients were excluded from the outcome analysis due to the incomplete therapeutic course. Figure 4 shows the participant flow chart. Among the remaining 114 patients, initial stricture resolution was achieved in 102 (90.2%) cases with endothrapy alone, and the success rate was 99.1% when PTC was utilized to manage ABS among cases of ERCP failure. One patient did not achieve stricture resolution despite maximal endo-therapy sessions and was referred for redo surgery (hepaticojejunostomy).
After initial resolution, 15 patients (13.27%) experienced stricture recurrence. Among the recurrent strictures, 8 reci
| Stricture status | Total, | No. of ERCP (mean ± SD) | Duration between liver transplant to 1st ERCP (months) | Anastomosis, n (%) | ||
| Single | 2 ducts | 2 ducts on a single patch | ||||
| Patients completing the therapeutic course | 114 | 5.18 ± 2.01 | 5.89 ± 5.58 (95%CI: 4.87-6.91) | 67 (58.8) | 36 (31.6) | 11 (9.6) |
| Stricture resolution with endo therapy alone | 102 (90.2) | 5.04 ± 2.04 | 6 ± 5.77 (95%CI: 4.88-7.12) | 58 (86.6) | 33 (91.7) | 11 (100) |
| Percutaneous transhepatic cholangioscopy referrals (cannulation failure/inability to traverse the stricture via ERCP) | 11 (9.7) | 6.27 ± 1.35 | 5.10 ± 3.65 (95%CI: 2.94-7.26) | 8 (13.4) | 3 (9.3) | - |
| Redo surgery | 1 (0.9) | 7.0 | 3.27 | 1 (100) | - | - |
| Combined percutaneous transhepatic cholangioscopy & endotherapy success rates | 113 (99.1) | 5.16 ± 2.02 | 5.92 ± 5.59 (95%CI: 4.89-6.95) | 66 (98.5) | 36 (100) | 11 (100) |
| Stricture recurrence rates | 15 (13.27) | 7.5 ± 1.9 | 5.93 ± 4.57 (95%CI: 3.62-8.24) | 6 (9.1) | 7 (19.4) | 2 (18.2) |
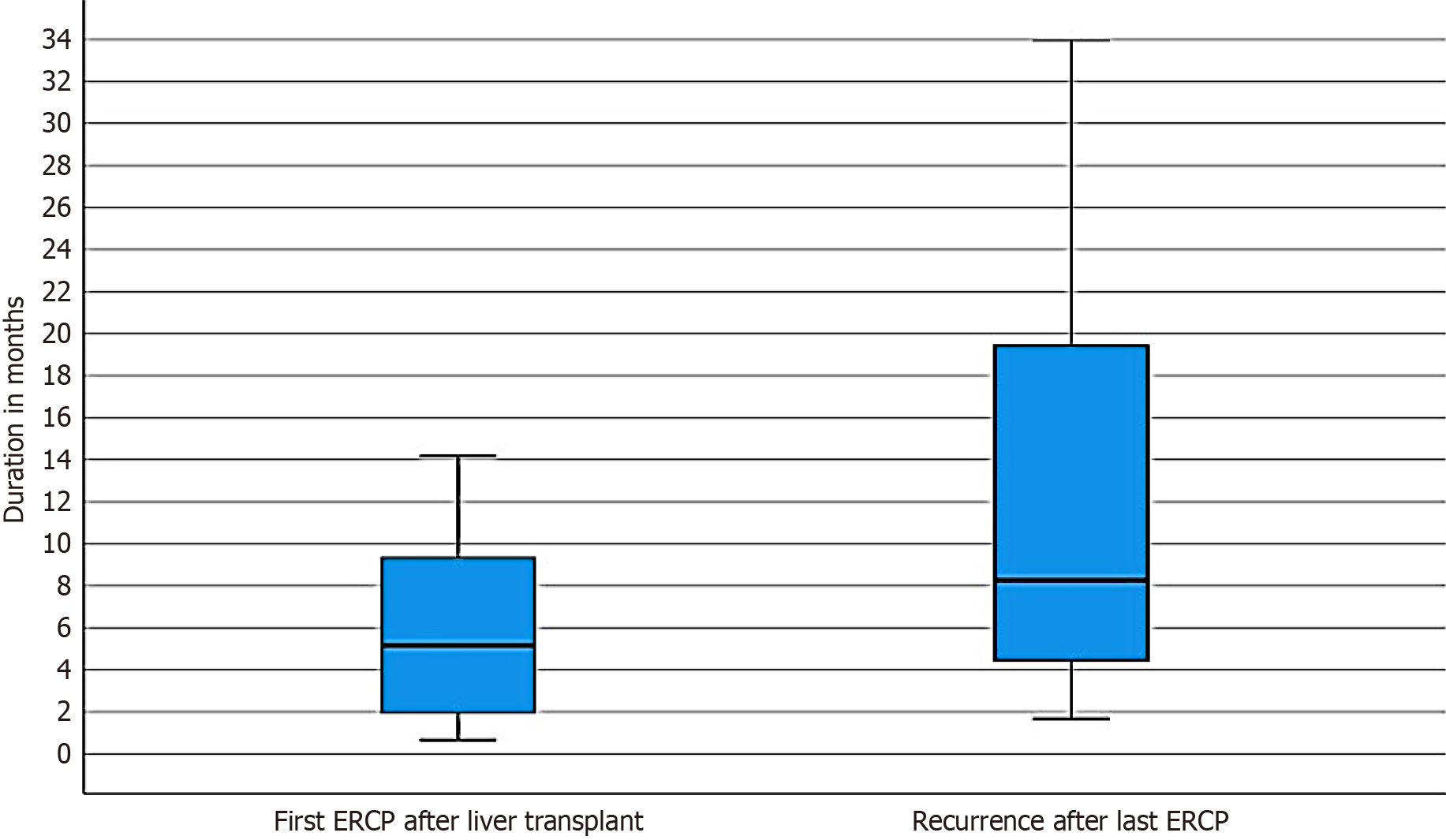
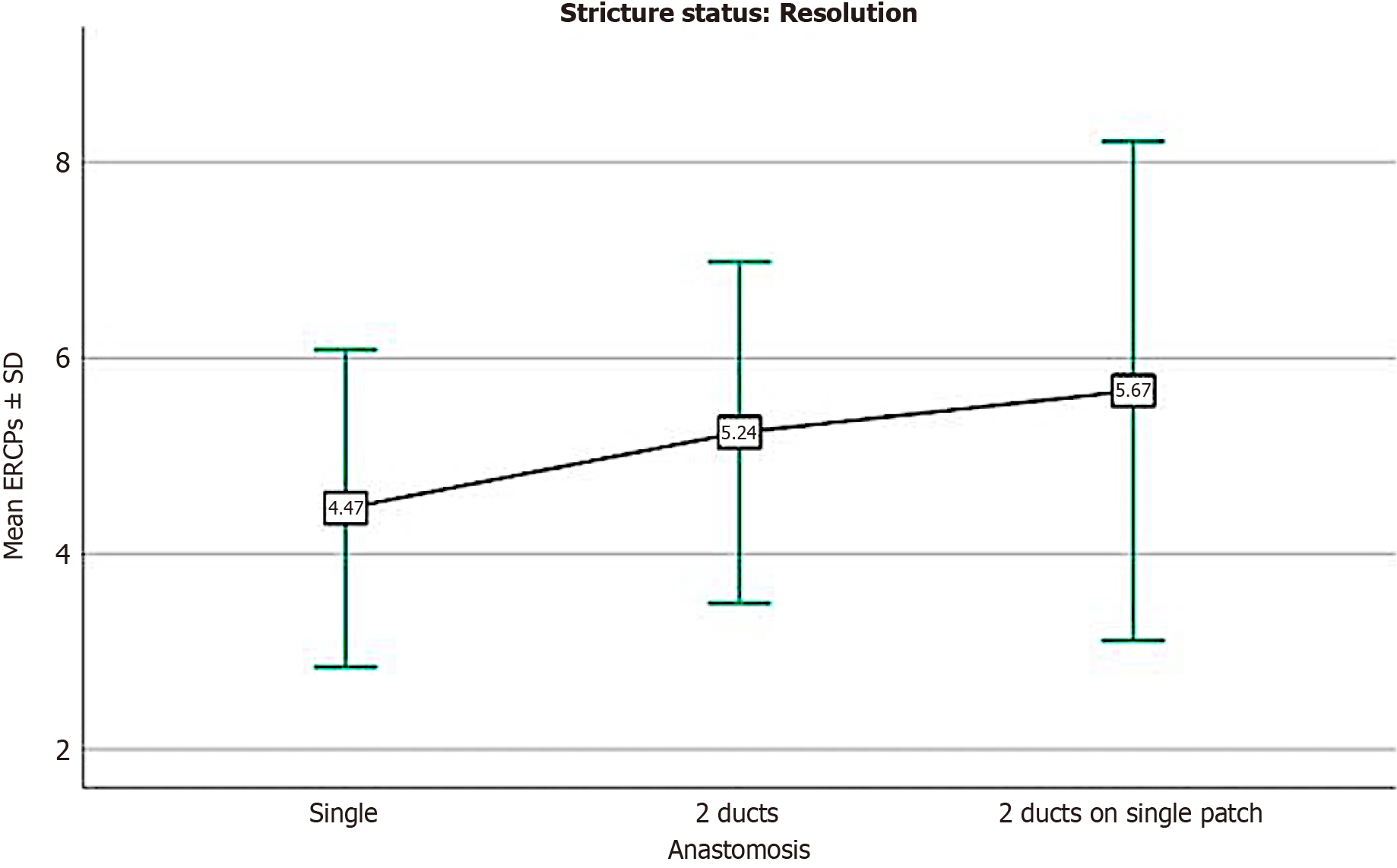
The median time to the stricture formation after liver transplantation was 5 months (IQR 2-9 months), with the majority occurring within the first year. In contrast, the median time to stricture recurrence after the last ERCP was 8 months (IQR 4-19 months), with a range extending up to 34 months. This indicates that recurrence commonly occurred within the first year, but late recurrences were also observed. Figure 5 illustrates the timelines from liver transplantation to the first ERCP and also the time to stricture recurrence after initial resolution. The mean number of ERCPs (stent exchanges) required for stricture resolution varied notably based on the type of anastomosis (with single duct anastomosis requiring fewer ERCPs on average compared to two duct configuration), demonstrating a statistically significant association (P < 0.001). Figure 6 shows the impact of the type of biliary anastomosis on the number of ERCPs required for stricture resolution.
The IDC played a crucial role in the management of ABS. IDC was carried out in 18 patients, accounting for 15.8% of the total cohort of 114 patients who completed the therapeutic course. The main indications included facilitating traversal of acutely angulated/extremely tight strictures in 7 (6%) patients, enabling drainage of sectoral branches in 5 patients (4.4%), and performing ductal assessment to optimize endotherapy in 6 patients (5.3%). Of the 7 cases in which ERCP failed to traverse the stricture, IDC achieved success in 6 cases (86%).
A wide range of procedure-related complications was observed in the study cohort of 176 patients undergoing a total of 792 ERCP procedures. Cholangitis was the most frequent complication and developed in 19 (2.40%) procedures, followed by PEP in 18 (2.27%). ERCP-related bleeding occurred in 12 (1.52%) cases; notably, 6 (50%) of these episodes were self-limiting and required no active intervention. Hemostasis was achieved in 4 cases (33.4%) using electrocautery ± balloon tamponade. The remaining 2 cases (16.6%) required combination therapies, including adrenaline injection with hemo-clip placement and adrenaline injection combined with electrocautery. Importantly, no cases of perforation or procedure-related mortality were observed in our cohort. Prophylactic pancreatic duct stenting was performed in 51 (29%) patients during the first ERCP procedure and successfully prevented PEP in 78% (40 of 51) of high-risk cases for PEP.
ABS are still considered one of the most difficult complications encountered after LDLT, with higher incidence rates as compared to DDLT. This high incidence rate is likely due to multifactorial reasons, including the technical difficulty, particularly in cases involving multiple or small-calibre bile ducts. The ABS incidence of 21.5% in our study is in keeping with the literature[5]. The time interval from liver transplantation to the development of ABS is highly variable. In our study, ABS developed from 12 days to as late as 5 years after LDLT, with a median of 5 months (IQR 2.5-8.3 months), which is similar to the 5.9 months described by Shin and Joh[10].
Over the years, there has been a definite shift noticed in the management of biliary complications post-transplant. The use of PTC as the primary therapeutic tool for managing biliary complications has gradually decreased, whereas the use of ERCP as the first line of treatment modality has steadily increased. Surgical revision has been a less common practice, especially reserved for difficult and refractory strictures. The overall success rate for stricture resolution with endo
The number of ERCPs needed to treat the stricture depends on the number and nature of the ductal anastomosis performed during the liver transplant. In our cohort, patients with a single-duct anastomosis required fewer ERCP sessions for stricture resolution (mean 4.47) than those with double-duct configurations, whether anastomosed separately (mean 5.24) or on a single patch (mean 5.67), confirming prior observations that multiple anastomoses not only increase the risk but also enhance the technical challenge in the endoscopic management[5,9].
ABS recurrence does occur after initial resolution and varies according to the follow-up period. Shin and Joh[10] reported ABS recurrence rates of 13%-44%. Another study showed that stricture recurrence rates after multiple plastic stent placement via ERCP were 3%-20%[6]. The recurrence rate of ABS after an initial resolution was 13.27% in our study, which is well within the rates reported in the literature. Although the time to recurrence was variable, as shown in Figure 5, most of these recurrences were successfully managed by repeated ERCPs, thus decreasing the need for surgical interventions. In our cohort, only one patient required surgical intervention (hepaticojejunostomy), thus confirming the safety and efficacy of endoscopic management in these patients.
Cholangitis (2.40%) and pancreatitis (2.27%) were the two most common post-ERCP complications noted in our study. The incidence of pancreatitis and cholangitis varies from 2.7% to 5.7% and from 1.02% to 4.5%, respectively[17,18]. There were no cases of perforation or ERCP-related mortality in our study. Prophylactic pancreatic duct stenting in the high-risk group for PEP showed favorable results, with 78% of patients showing no PEP, and 22% showing mild PEP, with no cases of moderate to severe PEP; thus, confirming the selective use of pancreatic duct stenting in patients identified as high risk, as per current clinical practice[19-21]. Although the technical success rate of ERCP was high, a proportion of patients were still referred for PTC because of additional challenges, such as inability to cross the stricture or cannulation of sectoral ducts, apart from failed biliary cannulation. These findings underscore the significance of PTC as a preferred rescue modality in cases where ERCP remains unsuccessful.
The strengths of this study include its relatively large sample size, focus on a well-defined single-center cohort, and utilization of both conventional ERCP and advanced cholangioscopic tools. However, its retrospective nature and relatively short follow-up period are potential limitations. Additionally, 55 patients (15 lost to follow-up, 40 still undergoing therapy) had incomplete follow-up, and 7 died before completion of therapy. Exclusion of these patients from outcome analysis could have potentially impacted the study findings and must be kept in consideration. However, a sensitivity analysis may be considered appropriate to assess the magnitude of any resulting bias.
Our findings support current international trends favoring endoscopic management of post-LDLT ABS, and suggest that graft anatomy and stricture characteristics should guide management strategy. Selective use of IDC in difficult cases may enhance technical and clinical success and the avoidance of undesired PTC and surgical referrals. Prospective, multicenter studies are warranted to further define optimal stenting duration, recurrence predictors, and preventive strategies, including refinements in anastomotic techniques during LDLT. As previously noted, our data demonstrated that on average, 4-5 ERCP sessions are required for stricture resolution, depending on the type of biliary anastomosis.
At our center, we primarily used plastic biliary stents for managing ABS. Nonetheless, fully covered self-expandable metallic stents are gaining traction in numerous transplant facilities because of their proven effectiveness, which includes requiring fewer ERCP sessions and a reduced overall treatment time. Even with these benefits, one major drawback of metallic stents is the potential for migration and technical complexity in double-duct anastomosis[22]. Consequently, upcoming studies need to focus on creating novel strategies guiding decisions about choice and feasibility of metal stents in view of the smaller number of ERCPs required for stricture resolution, along with cost effectiveness and less procedure related adverse events than in the multiple plastic stents strategy.
ABS continues to be a frequent challenge after LDLT with associated morbidity and mortality. However, they can be successfully managed through ERCP, known for its excellent technical and clinical outcomes and well-established safety profile. The need for endotherapy and stricture recurrence rates tend to rise in cases of complicated biliary anatomies, especially when multiple duct reconstructions are performed. Adding IDC and selective PTC for challenging cases can greatly improve outcomes in complicated biliary strictures. The evidence reiterates the significance of endoscopic therapy as the mainstay of treatment and the need for standardized approaches to improve long-term success.
Arsalan Saleem Chughtai performed the data analysis, and Saira Imtiaz reviewed the data analysis.
| 1. | Sequeira LM, Ozturk NB, Sierra L, Gurakar M, Toruner MD, Zheng M, Simsek C, Gurakar A, Kim AK. Hepatocellular Carcinoma and the Role of Liver Transplantation: An Update and Review. J Clin Transl Hepatol. 2025;13:327-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 2. | Cavalcante LN, Queiroz RMT, Paz CLDSL, Lyra AC. Better living donor liver transplantation patient survival compared to deceased donor - a systematic review and meta-analysis. Arq Gastroenterol. 2022;59:129-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 3. | Li Z, Rammohan A, Gunasekaran V, Hong S, Chih-Yi Chen I, Kim J, Hervera Marquez KA, Hsu SC, Kirimker EO, Akamatsu N, Shaked O, Finotti M, Yeow M, Genedy L, Dutkowski P, Nadalin S, Boehnert MU, Polak WG, Bonney GK, Mathur A, Samstein B, Emond JC, Testa G, Olthoff KM, Rosen CB, Heimbach JK, Taner T, Wong TC, Lo CM, Hasegawa K, Balci D, Cattral M, Sapisochin G, Selzner N, Jeng LB, Broering D, Joh JW, Chen CL, Suh KS, Rela M, Clavien PA. Biliary complications after adult-to-adult living-donor liver transplantation: An international multicenter study of 3633 cases. Am J Transplant. 2024;24:1233-1246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 4. | Giri S, Sahu SK, Mohapatra V, Chaudhary M, Panigrahi M, Nath P, Mallick B, Praharaj DL. Comparison of Biliary Complications Between Living and Deceased Donor Liver Transplantations: A Systematic Review and Meta-analysis. Cureus. 2024;16:e69019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Boeva I, Karagyozov PI, Tishkov I. Post-liver transplant biliary complications: Current knowledge and therapeutic advances. World J Hepatol. 2021;13:66-79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (1)] |
| 6. | Bofill A, Cárdenas A. A practical approach to the endoscopic management of biliary strictures after liver transplantation. Ann Hepatol. 2024;29:101186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 7. | Hassouneh R, Beran A, Rosenheck M, Sosio J, Olchawa N, Kubal C, Ghabril M, Gromski MA. Risk factors for biliary strictures and leaks after living-donor liver transplantation: a systematic review and meta-analysis. J Gastrointest Surg. 2024;28:1870-1882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 8. | Lee HW, Shah NH, Lee SK. An Update on Endoscopic Management of Post-Liver Transplant Biliary Complications. Clin Endosc. 2017;50:451-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 9. | Rao HB, Prakash A, Sudhindran S, Venu RP. Biliary strictures complicating living donor liver transplantation: Problems, novel insights and solutions. World J Gastroenterol. 2018;24:2061-2072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 42] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (5)] |
| 10. | Shin M, Joh JW. Advances in endoscopic management of biliary complications after living donor liver transplantation: Comprehensive review of the literature. World J Gastroenterol. 2016;22:6173-6191. [PubMed] [DOI] [Full Text] |
| 11. | Yasen A, Feng J, Dai TX, Zhu CH, Liang RB, Liao ZH, Li K, Cai YH, Wang GY. Management of anastomotic biliary stricture through utilizing percutaneous transhepatic cholangioscopy. Clin Radiol. 2024;79:e868-e877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 12. | Cortez AR, Morris MC, Brown NG, Winer LK, Safdar K, Poreddy S, Shah SA, Quillin RC 3rd. Is Surgery Necessary? Endoscopic Management of Post-transplant Biliary Complications in the Modern Era. J Gastrointest Surg. 2020;24:1639-1647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 13. | Dumonceau JM, Kapral C, Aabakken L, Papanikolaou IS, Tringali A, Vanbiervliet G, Beyna T, Dinis-Ribeiro M, Hritz I, Mariani A, Paspatis G, Radaelli F, Lakhtakia S, Veitch AM, van Hooft JE. ERCP-related adverse events: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2020;52:127-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 630] [Article Influence: 105.0] [Reference Citation Analysis (5)] |
| 14. | Cho IR, Lee SH, Kang J, Kim J, Lee TS, Lee MH, Lee MW, Choi JH, Paik WH, Ryu JK, Kim YT, Hong SK, Choi Y, Yi NJ, Lee KW, Suh KS. Digital single-operator cholangioscopy for difficult anastomotic biliary strictures in living donor liver transplant recipients after failure of standard ERCP: SPYPASS-2 study (with videos). Gastrointest Endosc. 2025;101:979-987.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 15. | Sharma ZD, Karunakaran M, Bansal RK, Gandhi A, Singh MK, Soin AS, Puri R, Sud R. Cholangioscopic classification of post-living donor liver transplantation biliary strictures can predict their natural history and response to therapy. ANZ J Surg. 2023;93:911-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Woo YS, Lee JK, Noh DH, Park JK, Lee KH, Lee KT. SpyGlass cholangioscopy-assisted guidewire placement for post-LDLT biliary strictures: a case series. Surg Endosc. 2016;30:3897-3903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (3)] |
| 17. | Ambrus RB, Svendsen LB, Hillingsø JG, Hansen ML, Achiam MP. Post-endoscopic retrograde cholangiopancreaticography complications in liver transplanted patients, a single-center experience. Scand J Surg. 2015;104:86-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Hüsing A, Cicinnati VR, Maschmeier M, Schmidt HH, Wolters HH, Beckebaum S, Kabar I. Complications after endoscopic sphincterotomy in liver transplant recipients: A retrospective single-centre study. Arab J Gastroenterol. 2015;16:46-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Kaimakliotis PI, Kochman ML. Prophylactic pancreatic stent placement to prevent pancreatitis after ERCP. Lancet. 2024;403:411-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Dubravcsik Z, Hritz I, Keczer B, Novák P, Lovász BD, Madácsy L. Network meta-analysis of prophylactic pancreatic stents and non-steroidal anti-inflammatory drugs in the prevention of moderate-to-severe post-ERCP pancreatitis. Pancreatology. 2021;21:704-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 21. | Buxbaum JL, Freeman M, Amateau SK, Chalhoub JM, Coelho-Prabhu N, Desai M, Elhanafi SE, Forbes N, Fujii-Lau LL, Kohli DR, Kwon RS, Machicado JD, Marya NB, Pawa S, Ruan WH, Sheth SG, Thiruvengadam NR, Thosani NC, Qumseya BJ; (ASGE Standards of Practice Committee Chair). American Society for Gastrointestinal Endoscopy guideline on post-ERCP pancreatitis prevention strategies: summary and recommendations. Gastrointest Endosc. 2023;97:153-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 127] [Article Influence: 42.3] [Reference Citation Analysis (1)] |
| 22. | Baraldo S, Meine GC, Santo PAE, Barbosa EC, Nau AL, Keane MG, Chavan R, Kuo YT, Ligresti D. Fully covered self-expandable metal stents versus multiple plastic stents for the treatment of post-orthotopic liver transplant anastomotic biliary strictures: An updated systematic review and meta-analysis of randomized controlled trials. Surg Endosc. 2025;39:721-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |