Published online May 16, 2026. doi: 10.4253/wjge.v18.i5.119573
Revised: March 11, 2026
Accepted: March 30, 2026
Published online: May 16, 2026
Processing time: 101 Days and 18.2 Hours
Congenital anomalies of the inferior vena cava (IVC) are rare vascular abnor
A 71-year-old female presented with recurrent gastrointestinal bleeding for more than one year. Endoscopic examination revealed large esophagogastric varices. Contrast-enhanced computed tomography demonstrated absence of the hepatic segment of the IVC with azygos continuation. Laboratory testing showed pre
This case highlights the diagnostic complexity of INCPH in patients with congenital IVC anomalies. Long-standing hemodynamic alterations associated with IVC abnormalities may contribute to intrahepatic vascular remodeling and the development of portal hypertension, although a direct causal relationship remains uncertain.
Core Tip: Inferior vena cava anomalies may induce non-cirrhotic portal hypertension by increasing portal venous inflow via abnormal hemodynamics, leading to variceal bleeding. Diagnosis requires exclusion of cirrhosis, supported by patent hepatic veins and liver biopsy showing obliterative portal venopathy. Splenomegaly/pancytopenia are not obligatory. Endoscopic therapy effectively controls bleeding, but long-term monitoring is essential.
- Citation: Li M, Zhang LK, Qu L, Liu JH, Liu JJ, Sun SZ, Zhang D, Hao NB, Li CZ. Absent inferior vena cava combined with idiopathic noncirrhotic portal hypertension resulting esophagogastric variceal bleeding: A case report. World J Gastrointest Endosc 2026; 18(5): 119573
- URL: https://www.wjgnet.com/1948-5190/full/v18/i5/119573.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i5.119573
Idiopathic noncirrhotic portal hypertension (INCPH), currently included within the spectrum of porto-sinusoidal vascular disease (PSVD), is characterized by portal hypertension in the absence of cirrhosis and with preserved hepatic function[1-3]. Patients commonly present with complications of portal hypertension, including splenomegaly, thrombocytopenia, or variceal bleeding, despite relatively normal hepatic biochemical parameters.
The etiology of INCPH is heterogeneous and includes prothrombotic disorders, autoimmune diseases, infections, exposure to medications or toxins, and congenital vascular abnormalities[4-6]. Histologically, INCPH is frequently associated with obliterative portal venopathy, characterized by sclerosis and narrowing of small portal vein branches that lead to altered intrahepatic blood flow[7].
Congenital anomalies of the inferior vena cava (IVC), including interruption or absence of the hepatic segment with azygos continuation, are uncommon vascular malformations with an estimated prevalence of 0.0005%-1% in the general population[8]. Although many individuals remain asymptomatic, certain IVC anomalies may alter venous return and predispose patients to complications such as deep vein thrombosis or abnormal hemodynamic circulation[9].
Several case reports have suggested a potential association between congenital IVC anomalies and portal hypertension[10]. However, the underlying mechanisms remain incompletely understood, and a causal relationship has not been definitively established. Here, we report a rare case of INCPH presenting with esophagogastric variceal bleeding in a patient with congenital absence of the hepatic segment of the IVC. This case highlights the diagnostic challenges and emphasizes the importance of differential diagnosis in patients with portal hypertension without cirrhosis.
A 71-year-old woman was admitted with recurrent melena for more than one year.
One year prior to admission, the patient experienced episodes of melena. Endoscopy performed at another hospital revealed esophagogastric varices, and symptomatic improvement was achieved after hemostatic treatment. In August 2024, the patient developed hematemesis (approximately 250 mL) accompanied by dizziness and recurrent melena. She received conservative treatment at an outside hospital and was subsequently referred to our institution because of persistent gastrointestinal bleeding.
The patient had a several-year history of type 2 diabetes mellitus.
The patient denied a history of chronic liver disease, viral hepatitis, alcohol consumption, or long-term use of hepatotoxic medications. There was no family history of hereditary liver disease or thrombotic disorders.
On admission, vital signs were as follows: Body temperature 36.1 °C, heart rate 62 beats/minute, respiratory rate 18 breaths/minute, and blood pressure 123/65 mmHg. Her height was 158 cm and weight was 55 kg, corresponding to a body mass index of 22.0 kg/m2.
The patient appeared pale and mildly fatigued. No stigmata of chronic liver disease were observed, including spider angiomas or palmar erythema. Abdominal examination revealed a soft, non-tender abdomen without hepatomegaly or splenomegaly on palpation.
Laboratory tests showed mild anemia (hemoglobin 114 g/L) with otherwise normal white blood cell and platelet counts. Liver function tests, including alanine aminotransferase, aspartate aminotransferase, bilirubin, and albumin levels, were within normal limits. Serological tests for viral hepatitis and autoimmune liver disease were negative.
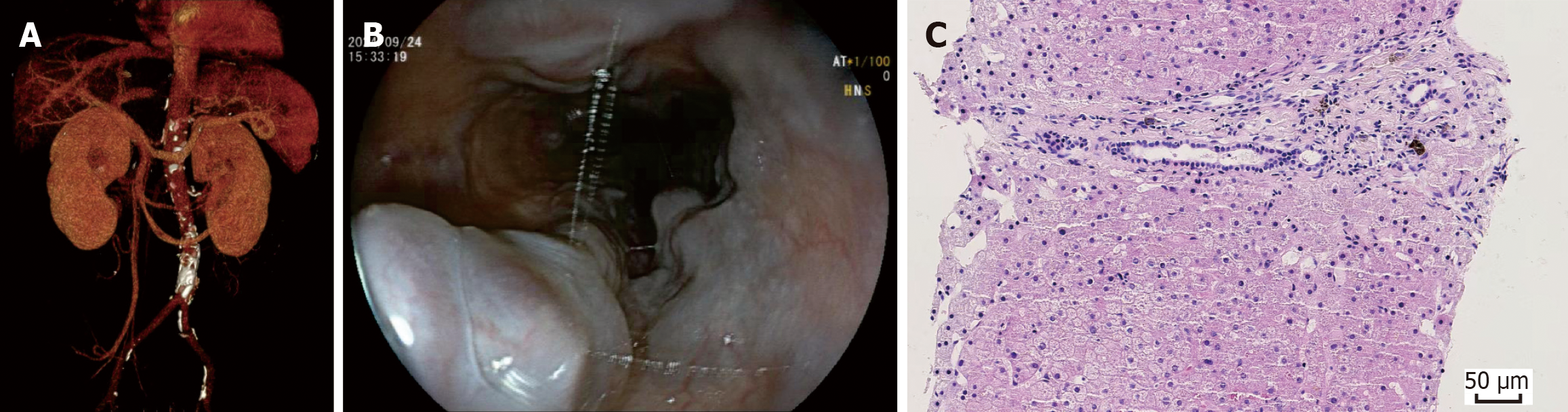
Doppler ultrasound of the portal venous system demonstrated a slightly dilated main portal vein (12 mm in diameter), with a portal vein flow velocity of 24 cm/second within the expected range. No evidence of portal vein thrombosis was detected. Contrast-enhanced computed tomography revealed absence of the hepatic segment of the IVC with drainage of the renal veins into the azygos system. The right and middle hepatic veins were dilated, measuring 11 mm and 9 mm in diameter, respectively (Figure 1A). Esophagogastric varices were also observed on imaging (Figure 1B). Esophagogastroduodenoscopy demonstrated large esophageal varices (grade III) with positive red wale signs and severe gastric fundal varices. Echocardiography showed left atrial enlargement, reduced left ventricular diastolic function, and mild tricuspid regurgitation.
Ultrasound-guided liver biopsy revealed moderate fibrosis in portal areas with narrowing or disappearance of portal vein lumens and multiple thin-walled vascular channels surrounding portal tracts. Focal sinusoidal dilatation was observed without evidence of perisinusoidal fibrosis. These findings were consistent with INCPH associated with obliterative portal venopathy and showed no evidence of hepatic veno-occlusive disease (Figure 1C)[7,11,12].
The case report posits that the long-term hemodynamic alterations caused by the congenital IVC anomaly likely contributed to the development of the portal venopathy and subsequent non-cirrhotic portal hypertension.
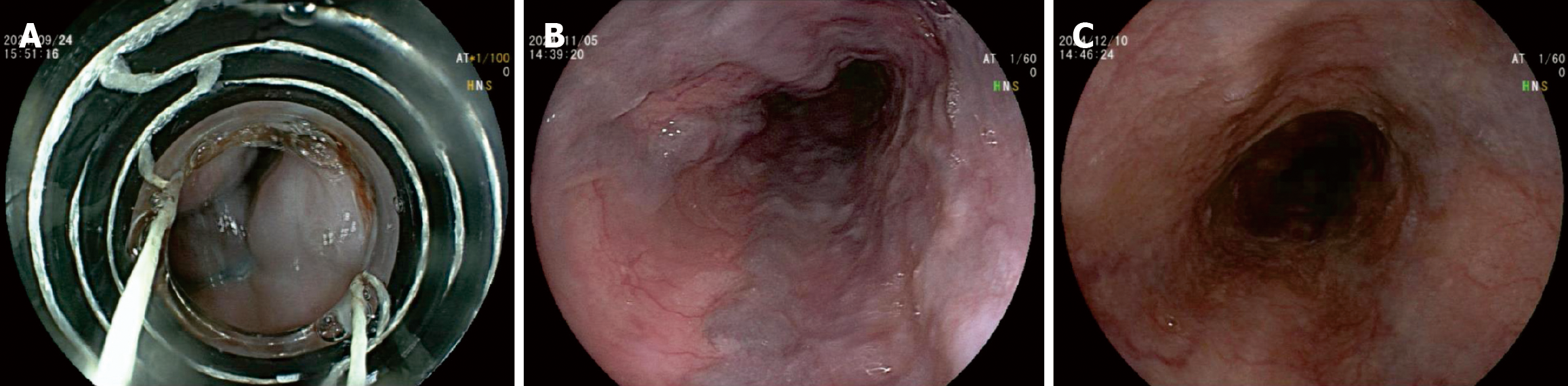
The patient underwent endoscopic variceal ligation. Eleven bands were applied to four large esophageal varices columns located approximately 25 cm from the incisors (Figure 2A). Follow-up endoscopy 1 month later showed improvement in varices, and a second session of variceal ligation was performed (Figure 2B).
No intraoperative complications such as bleeding or perforation occurred. The procedure was performed successfully.
In addition to endoscopic treatment, the patient received supportive therapy including proton pump inhibitors and anemia correction.
Other potential treatment strategies considered included pharmacological therapy (non-selective beta-blockers) and transjugular intrahepatic portosystemic shunt (TIPS). However, TIPS may be technically challenging in patients with IVC anomalies, and conservative pharmacological therapy alone was considered insufficient given the high risk of recurrent bleeding.
The patient recovered uneventfully and was discharged seven days after endoscopic variceal ligation. At two-month follow-up, endoscopy demonstrated marked reduction of esophageal varices (Figure 2C) and complete resolution of gastric fundal varices. The patient remained clinically stable without recurrence of gastrointestinal bleeding.
INCPH, currently classified within the spectrum of PSVD, represents a heterogeneous group of vascular liver disorders characterized by portal hypertension in the absence of cirrhosis[2,4]. Histologically, PSVD is commonly associated with obliterative portal venopathy, nodular regenerative hyperplasia, or incomplete septal fibrosis[7,11].
In the present case, liver biopsy demonstrated narrowing or disappearance of portal vein branches accompanied by periportal fibrosis and sinusoidal dilatation, which are characteristic histopathological features of obliterative portal venopathy. These findings supported the diagnosis of INCPH.
The coexistence of a congenital IVC anomaly and INCPH raises important questions regarding potential patho
Congenital interruption of the hepatic segment of the IVC alters systemic venous return and may result in chronic compensatory collateral circulation through the azygos system. Persistent alterations in venous flow dynamics may influence intrahepatic vascular remodeling and contribute to the development of obliterative portal venopathy, including narrowing of portal venules and formation of collateral circulation[9,10].
Importantly, portal hypertension in PSVD is frequently presinusoidal[11,14]. In such cases, the hepatic venous pressure gradient (HVPG) may remain normal or only mildly elevated despite clinically significant portal hypertension. This phenomenon may explain the relatively normal Doppler-estimated portal hemodynamics observed in the present patient. Previous studies have emphasized that HVPG measurements may underestimate portal pressure in presinusoidal disorders.
Several alternative causes of noncirrhotic portal hypertension were carefully considered[4,5]. Imaging studies showed no evidence of portal vein thrombosis or Budd-Chiari syndrome. Serological testing excluded autoimmune liver disease and chronic viral hepatitis. Furthermore, liver biopsy revealed no histological features of cirrhosis. These findings support the diagnosis of INCPH associated with obliterative portal venopathy.
Although a direct causal relationship between the IVC anomaly and portal hypertension cannot be definitively established, long-standing hemodynamic alterations resulting from abnormal venous anatomy may represent a potential contributing factor. Similar mechanisms have been proposed in other congenital vascular disorders associated with portal hypertension, such as congenital portosystemic shunts and Abernethy malformations[9].
Management of portal hypertension in INCPH primarily focuses on prevention and treatment of complications[5,6,14]. Endoscopic variceal ligation remains the first-line therapy for esophageal varices, while pharmacological therapy with non-selective beta-blockers may reduce portal pressure. In selected cases, transjugular TIPS may be considered[15]. However, anatomical variations of the IVC may increase the technical difficulty of TIPS placement. Therefore, individualized treatment strategies are required for patients with congenital venous anomalies.
This case highlights the diagnostic complexity of portal hypertension occurring in patients with unusual venous anatomy. Recognition of noncirrhotic causes of portal hypertension is particularly important in patients presenting with variceal bleeding but preserved hepatic function.
Several limitations should be acknowledged in the interpretation of this case. First, although extensive clinical, laboratory, imaging, and histopathological evaluations were performed, the differential diagnosis of INCPH cannot be considered completely exhaustive. For example, comprehensive screening for inherited or acquired thrombophilia, including genetic testing for prothrombotic disorders, was not performed; therefore, rare prothrombotic conditions potentially associated with obliterative portal venopathy cannot be completely excluded. Second, while imaging studies showed no evidence of portal vein thrombosis, IVC thrombosis, or Budd-Chiari syndrome, it remains possible that previous transient or subclinical thrombotic events occurred and subsequently resolved before diagnosis. Third, invasive hemodynamic measurements, such as the HVPG, were not performed in this patient. Consequently, although clinical findings and endoscopic evidence supported the presence of portal hypertension, the direct hemodynamic relationship between the IVC anomaly and portal venous circulation could not be quantitatively assessed. Fourth, as this is a single case report, the coexistence of congenital IVC anomaly and INCPH may represent either a potential pathophysiological association or a coincidental finding. Therefore, the findings of this report should be interpreted cautiously.
This case highlights the diagnostic complexity of INCPH in patients with congenital IVC anomalies. Although a direct causal relationship cannot be definitively established, long-term hemodynamic alterations associated with congenital IVC abnormalities may contribute to intrahepatic vascular remodeling and the development of portal hypertension. Careful differential diagnosis and histological evaluation are essential for accurate diagnosis and appropriate management.
| 1. | Hernández-Gea V, Baiges A, Turon F, Garcia-Pagán JC. Idiopathic Portal Hypertension. Hepatology. 2018;68:2413-2423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 2. | Sarin SK, Khanna R. Non-cirrhotic portal hypertension. Clin Liver Dis. 2014;18:451-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 3. | Barisic-Jaman M, Milosevic M, Pastrovic F, Skrtic A, Grgurevic I. Porto-sinusoidal vascular disease: a new definition of an old clinical entity. Clin Exp Hepatol. 2023;9:297-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 4. | Nicoară-Farcău O, Rusu I, Stefănescu H, Tanțău M, Badea RI, Procopeț B. Diagnostic challenges in non-cirrhotic portal hypertension - porto sinusoidal vascular disease. World J Gastroenterol. 2020;26:3000-3011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 5. | Strauss E, Valla D. Non-cirrhotic portal hypertension--concept, diagnosis and clinical management. Clin Res Hepatol Gastroenterol. 2014;38:564-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Kmeid M, Liu X, Ballentine S, Lee H. Idiopathic Non-Cirrhotic Portal Hypertension and Porto-Sinusoidal Vascular Disease: Review of Current Data. Gastroenterology Res. 2021;14:49-65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 7. | Li P, Schiano TD, Thung SN, Ward SC, Fiel MI. Shared Features of Obliterative Portal Venopathy, Normal Liver, and Chronic Liver Disease: A Histologic and Morphometric Analysis. Mod Pathol. 2025;38:100739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 8. | Gerges P, Mian A, Singh G, Aziz M, Guirguis S, Koteish A. Agenesis of the Intrahepatic Inferior Vena Cava: A Case Report and Literature Review. Cureus. 2023;15:e35589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 9. | Chen SJ, Wu MH, Wang JK. Clinical implications of congenital interruption of inferior vena cava. J Formos Med Assoc. 2022;121:1938-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 10. | Fukushima M, Miyaaki H, Sasaki R, Haraguchi M, Miuma S, Ishimaru H, Hidaka M, Okudaira S, Eguchi S, Futakuchi M, Kusano H, Kage M, Nakao K. Inferior Vena Cava Anomalies with Portal Vein System Continuation Presenting as Portal Hypertension with a Long-term Follow-up. Intern Med. 2020;59:2897-2901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | De Gottardi A, Sempoux C, Berzigotti A. Porto-sinusoidal vascular disorder. J Hepatol. 2022;77:1124-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 120] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 12. | Goodman ZD. Grading and staging systems for inflammation and fibrosis in chronic liver diseases. J Hepatol. 2007;47:598-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 743] [Cited by in RCA: 678] [Article Influence: 35.7] [Reference Citation Analysis (1)] |
| 13. | Khot R, Jarmakani HM, Krasinski A, Menias CO, Katz DS, Revzin MV. From congenital variants to critical clues: A radiologic review of inferior vena cava pathologies. J Clin Imaging Sci. 2025;15:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2053] [Article Influence: 513.3] [Reference Citation Analysis (15)] |
| 15. | Lv Y, Li K, He C, Luo B, Zhang B, Liu H, Wang Z, Guo W, Wang Q, Chen H, Bai W, Yuan X, Yu T, Li X, Yuan J, Han N, Zhu Y, Niu J, Xie H, Wang J, Chen L, Yin Z, Fan D, Li Z, Han G. TIPSS for variceal bleeding in patients with idiopathic non-cirrhotic portal hypertension: comparison with patients who have cirrhosis. Aliment Pharmacol Ther. 2019;49:926-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |