Published online May 16, 2026. doi: 10.4253/wjge.v18.i5.119952
Revised: April 7, 2026
Accepted: April 28, 2026
Published online: May 16, 2026
Processing time: 89 Days and 7.7 Hours
Aortoduodenal fistula (ADF) managed with duodenal stenting has never before been reported in the literature.
A 75-year-old Chinese female with metastatic ureteric carcinoma presented with massive gastrointestinal bleeding secondary to ADF. Initial haemorrhage was managed with endovascular aortic repair and endoscopic haemostasis. However, she refused definitive open surgery. Her duodenal fistula was managed with partially covered stenting of the duodenum, which was complicated with duode
This case report demonstrates that combined endoscopic and endovascular mana
Core Tip: Aorto-duodenal fistula is a rare cause of gastrointestinal bleeding and most commonly managed with open surgery. This case reports the first ever use of minimally invasive treatment of aortoduodenal fistula with endovascular repair of aorta and endoscopic stenting of duodenum. The duodenal stent was complicated with migration, which can be prevented with clipping of the duodenal stent to the mucosa.
- Citation: Chan ST, Lo STW, Cheung VWS, Chu CCW, Pang SYC. Duodenal and aortic stenting for aortoduodenal fistula secondary to malignancy: A case report. World J Gastrointest Endosc 2026; 18(5): 119952
- URL: https://www.wjgnet.com/1948-5190/full/v18/i5/119952.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i5.119952
Aortoduodenal fistula (ADF) is traditionally managed with open surgery, although endovascular aortic repair (EVAR) has been advocated even by International Societies[1]. However, management of the duodenal defect with endoscopic treatment has never before been reported.
A 75-year-old Chinese female complained of abdominal pain, dizziness and coffee-ground vomiting.
She was found unconscious by pedestrians and transported to the Department of Accident and Emergency, Pamela Youde Nethersole Eastern Hospital in April 2025. Patient complained of diffuse abdominal pain and dizziness, followed by one episode of coffee-ground vomiting. Shortly after admission, she developed cardiac arrest with pulseless electrical activity. Cardiopulmonary resuscitation was initiated and she had return of spontaneous circulation after 2 minutes.
She had a known history of right ureteral transitional cell carcinoma with aortocaval lymph node metastasis since November 2023. The patient received multiple lines of chemotherapy with slow progression of para-aortic nodal and pulmonary metastases. The latest computed tomography (CT) scan, performed two months prior to this admission, already demonstrated interval enlargement of the para-aortic lymph node closely abutting, or possibly invading, the third part of the duodenum.
Her abdomen was soft and nontender, and no pulsatile mass was felt.
Emergency oesophagogastroduodenoscopy (OGD) revealed active bleeding from a large ulcer at the third part of the duodenum. Endoscopic haemostasis using adrenaline injection and hemospray was unsuccessful.
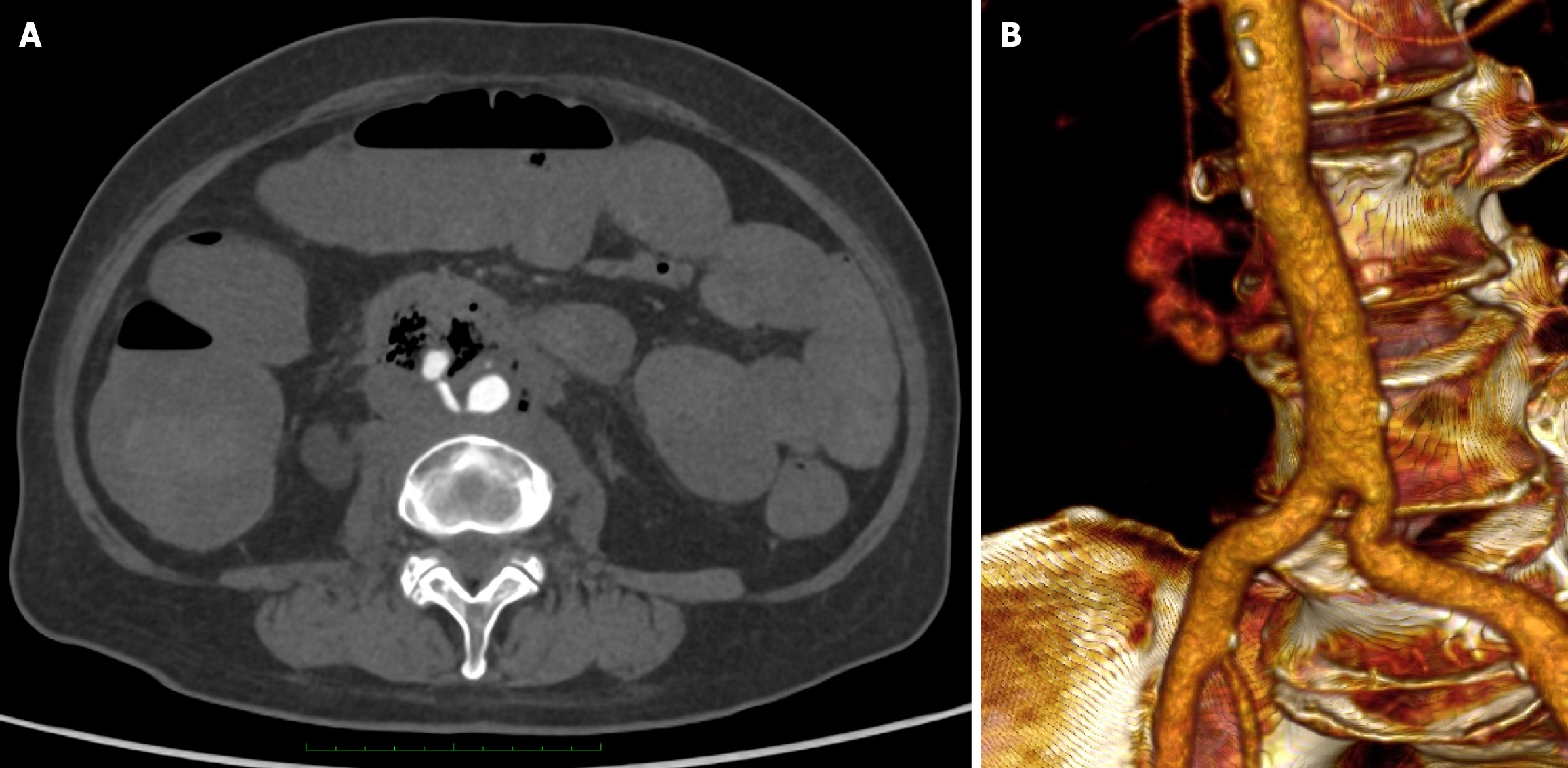
Given the patient’s poor premorbid status and suspected duodenal involvement by metastatic lymph node, laparotomy was not pursued. A CT angiogram (Figure 1) was performed to evaluate for embolization, which showed extravasation of contrast from infrarenal aorta into the third part of duodenum, diagnostic of ADF with haemorrhage. There were multiple gas locules within previously known para-aortic metastatic nodal conglomerates, which were encasing the aorta. There was no aortic aneurysm.
A multidisciplinary discussion involving the patient’s family, intensivist and oncologist concluded that, despite her slowly progressive terminal malignancy, the patient had remained ambulatory with satisfactory quality of life prior to admission, and her oncological survival potentially exceed six months. The family opted for EVAR to control active bleeding, knowing that it is a temporizing measure with high risk of infection without definite open repair, for which the patient was likely unfit.
The final diagnosis was ADF.
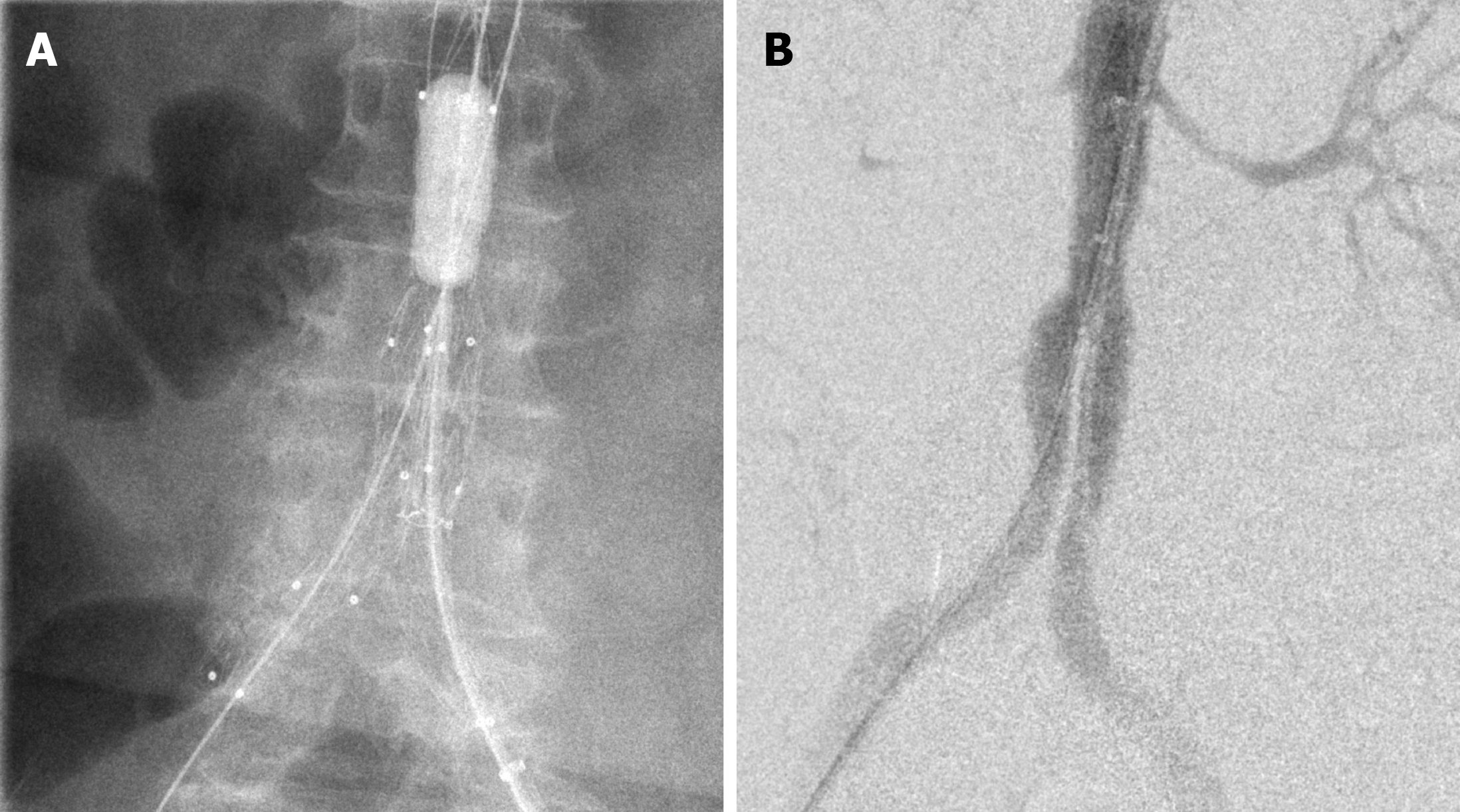
Emergency EVAR was performed for haemostasis despite her abdominal aorta measuring only 12-14 mm, below the minimum requirement. The aorto-enteric fistula was covered with the smallest bifurcated stent-graft and bilateral iliac extensions. Completion angiography showed proper graft placement without extravasation or endoleak (Figure 2).
The patient recovered well following EVAR, thus definitive open repair for ADF was offered. However, upon regai
She was initially managed by keeping strict nil per oral with intravenous proton pump inhibitor and antibiotics. Ente
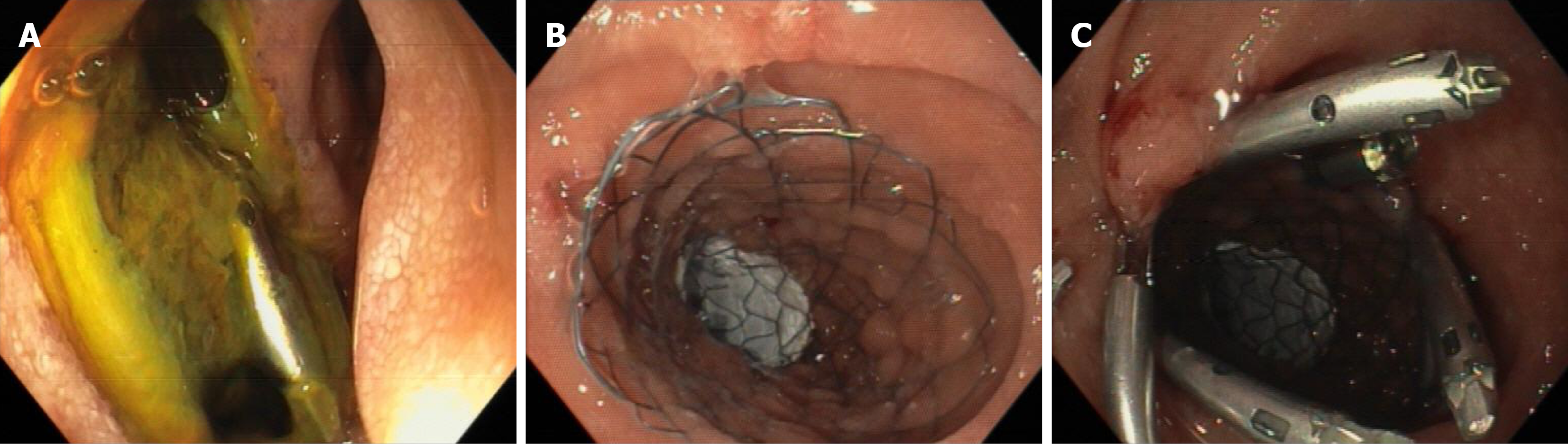
Other nonsurgical methods of pallative treatment were explored and duodenal stenting was offered (Figure 3). OGD was performed on post EVAR day 17 to deploy two partially covered, self-expanding metal stents – one across the pylorus and another across the duodenal defect, overlapping by 2 cm. The patient made a good recovery and could tolerate oral diet after the procedure. She was discharged with a prolonged course of antibiotics.
Two weeks later, the patient was readmitted for a follow up CT scan, which incidentally found that both the duodenal stents had migrated. OGD guided removal of stents was done and subsequently a single partially covered, self-expandable stent was placed from the pylorus to the 4th part of the duodenum. The stent was anchored to the duodenum with multiple endoscopic clips. She could tolerate oral diet after the procedure.
The patient eventually developed cauda equina syndrome secondary to infective spondylitis and sepsis as evidenced by blood culture growing polymicrobial bacteremia, despite being on long-term antibiotics. The patient was eventually managed with palliative care, and she passed a month later.
This is a rare case of ADF caused due to metastatic cancer in para-aortic lymph nodes, invading into the duodenum and aorta. Saers and Scheltinga[2] showed that the most common cause of ADF is aortic aneurysm, while less than 10% of ADF are caused by cancer related etiologies. Aorto-enteric fistula after abdominal aortic aneurysm repair (open or endovascular) is considered secondary. Aorto-enteric fistulas are primary if there was no preceeding aortic intervention[3].
The classical triad of ADF include gastrointestinal (GI) bleeding (as much as 94% of patients affected), pain and pulsating mass on abdominal palpation. Yet this triad is only present in around 11% of patients[4]. Moreover, the differential diagnosis of ADF is often overlooked as it has a very low prevalence (around 0.07% for primary ADF and 1.6% after open abdominal aortic aneurysm repair)[5]. These make the diagnosis of ADF difficult and a high index of suspicion is required.
Workup of ADF includes contrast-enhanced CT angiography and OGD. CT angiography has a low sensitivity (50%), and a high specificity (100%) for diagnosing ADF[6], which makes it highly reliable in positive results, but also a negative result does not necessarily rule out ADF. OGD is an important investigation for upper GI bleeding, which is the most common presentation of ADF. However, the endoscopist must be aware of the possibility of ADF, as the pathology may only be detected if the scope is put down to the 3rd or even the 4th part of the duodenum[7]. It is reported that OGD can only detect 25%-50% of ADF[8].
The surgical management of ADF requires management of the aorta and the management of the duodenum. The management of the aorta has traditionally been with open surgery. In an infected surgical field, such as in a patient with ADF, after wide debridement, the options of aortic repair include using spliced autologous femoral vein graft[9], antibiotic-impregnated prosthetic graft[10], or aortic transection and extra-anatomical bypass (e.g. axillo-femoral bypass)[11], which is the historical golden standard[12]. Yet, emergency ultra-major surgery for ADF carries a mortality rate around 30%-40%[13].
Another approach is EVAR, which has revolutionized the management of aortic aneurysm and is also utilised in ADF as a bridge to definitive surgery[1]. The ESVS guidelines on Vascular Graft and Endograft Infections 2020[14] recommend initial temporary treatment with an endograft in the emergency setting of active bleeding of aorto-enteric fistula (class IIb). EVAR has also been used in the reported patient to achieve hemostasis and to manage the aortic defect.
The surgical management of the duodenum in ADF has also mainly been with open surgery[15]. However, advances in endoscopy allowing for closure of full thickness defects of the GI tract have been reported[16].
The reported patient refused definitive treatment of ADF with open surgery, so endoscopic options were being explored. The endoscopic treatment options of duodenal fistula included primary closure of the defect (e.g. with through-the-scope or over-the-scope endoclips) as Berner-Hansen et al[17] suggested that endoscopic clips or stents could be used to manage aorto-enteric fistula before open surgery. However, clipping was not feasible for this patient because of large defect size and friable ulcer edges. Instead, partially-covered duodenal stenting was used, aiming to exclude the duodenal fistula defect. Fully covered duodenal stent has the disadvantage of causing biliary obstruction and higher chance of migration compared to partially covered stent. The treatment of duodenal stenting may facilitate oral feeding and a better quality of life[18].
Whilst being minimally invasive, endoscopic stenting of the duodenum for ADF has several limitations. First, stents are prone to migration (9.1% risk of migration for partially covered, self-expandable metal stent used in GI pathologies)[19]. Measures to prevent migration such as clipping of the stent to anchor it to the duodenum are recommended[20]. Another is that even though defect coverage could reduce chance of recurrence and sepsis[21], they do not control existing infection, and in fact, infected stent (e.g. EVAR stentgraft) is an anticipated complication - hence the need for lifelong antibiotics. IV antibiotics and percutaneous drainage may control the sepsis temporarily, but inadequate source control means that the infection may worsen and spread[22] – as evidenced by this reported patient with infective spondylitis due to posterior local invasion by the retroperitoneal collection, due to being in a prolonged supine position.
The reported outcome for patients after EVAR is limited. Kehagias et al[21] reported apparently 41.6% long term survival for post EVAR aorto-enteric fistula patients. In their series, EVAR was definitive therapy for 8 out of 12 patients. Antoniou et al[3] in their systematic review showed that infection occurred in 44% of patients after EVAR for aorto-enteric fistula after a mean follow up of 13 months.
We hope that this first reported case of duodenal stenting for ADF would help others to consider it as a treatment option, as well as being aware of the risk of stent migration. Our opinion is that endoscopic management of ADF may be feasible for some patients, and could be considered, especially in patients who are not candidates for open surgery.
To our knowledge, this is the first reported case of duodenal stenting used after EVAR in ADF secondary to cancer metastasis. This is a rare disease with high mortality, therefore high clinical suspicion is needed to correctly diagnose and treat this condition. Duodenal stenting may facilitate oral feeding but carries a risk of stent migration and uncontrolled sepsis[23]. It may be feasible in selected patients with limited life expectancy in centres with expertise.
| 1. | Chakfé N, Diener H, Lejay A, Assadian O, Berard X, Caillon J, Fourneau I, Glaudemans AWJM, Koncar I, Lindholt J, Melissano G, Saleem BR, Senneville E, Slart RHJA, Szeberin Z, Venermo M, Vermassen F, Wyss TR; Esvs Guidelines Committee; de Borst GJ, Bastos Gonçalves F, Kakkos SK, Kolh P, Tulamo R, Vega de Ceniga M, Document Reviewers, von Allmen RS, van den Berg JC, Debus ES, Koelemay MJW, Linares-Palomino JP, Moneta GL, Ricco JB, Wanhainen A. Editor's Choice - European Society for Vascular Surgery (ESVS) 2020 Clinical Practice Guidelines on the Management of Vascular Graft and Endograft Infections. Eur J Vasc Endovasc Surg. 2020;59:339-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 478] [Cited by in RCA: 389] [Article Influence: 64.8] [Reference Citation Analysis (0)] |
| 2. | Saers SJ, Scheltinga MR. Primary aortoenteric fistula. Br J Surg. 2005;92:143-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 189] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 3. | Antoniou GA, Koutsias S, Antoniou SA, Georgiakakis A, Lazarides MK, Giannoukas AD. Outcome after endovascular stent graft repair of aortoenteric fistula: A systematic review. J Vasc Surg. 2009;49:782-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 104] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 4. | Akabane K, Tajima Y, Toyama S, Tamate Y, Watanabe T, Uchida T. Case Report: Primary aortoduodenal fistula caused by a 30-mm inflammatory abdominal aortic aneurysm. Front Surg. 2025;12:1582823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Šumskienė J, Šveikauskaitė E, Kondrackienė J, Kupčinskas L. Aorto-duodenal fistula: a rare but serious complication of gastrointestinal hemorrhage. A case report. Acta Med Litu. 2016;23:165-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Hughes FM, Kavanagh D, Barry M, Owens A, MacErlaine DP, Malone DE. Aortoenteric fistula: a diagnostic dilemma. Abdom Imaging. 2007;32:398-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Alzobydi AH, Guraya SS. Primary aortoduodenal fistula: a case report. World J Gastroenterol. 2013;19:415-417. [PubMed] [DOI] [Full Text] |
| 8. | Lin TC, Tsai CL, Chang YT, Hu SY. Primary aortoduodenal fistula associated with abdominal aortic aneurysm with presentation of gastrointestinal bleeding: a case report. BMC Cardiovasc Disord. 2018;18:113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 9. | Pallister ZS, Chung J. Femoral Vein Reconstruction for Aortic Infections. Vasc Specialist Int. 2021;37:4-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Nasim A, Hayes P, London N, Barrie WW, Bell PR, Naylor AR. Vascular surgical society of great britain and ireland: In situ replacement of infected aortic grafts with rifampicin-bonded prostheses. Br J Surg. 1999;86:695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 11. | Bacourt F, Koskas F. Axillobifemoral bypass and aortic exclusion for vascular septic lesions: a multicenter retrospective study of 98 cases. French University Association for Research in Surgery. Ann Vasc Surg. 1992;6:119-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 64] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Kakkos SK, Bicknell CD, Tsolakis IA, Bergqvist D; Hellenic Co-operative Group on Aortic Surgery. Editor's Choice - Management of Secondary Aorto-enteric and Other Abdominal Arterio-enteric Fistulas: A Review and Pooled Data Analysis. Eur J Vasc Endovasc Surg. 2016;52:770-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 13. | Bartley A, Scali ST, Patterson S, Rosenthal MD, Croft C, Arnaoutakis DJ, Cooper MA, Upchurch GR Jr, Back MR, Huber TS. Improved perioperative mortality after secondary aortoenteric fistula repair and lessons learned from a 20-year experience. J Vasc Surg. 2022;75:287-295.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 14. | Baril DT, Carroccio A, Ellozy SH, Palchik E, Sachdev U, Jacobs TS, Marin ML. Evolving strategies for the treatment of aortoenteric fistulas. J Vasc Surg. 2006;44:250-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 88] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 15. | Jung JE, Oh S, Kwon IG. Experiences of Surgical Approach for Aortoduodenal Fistula Repairs in Abdominal Aortic Aneurysm Patients. Yonsei Med J. 2025;66:37-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Papaefthymiou A, Norton B, Telese A, Murray C, Murino A, Johnson G, Tsiopoulos F, Simons-Linares R, Seghal V, Haidry R. Endoscopic suturing and clipping devices for defects in the GI tract. Best Pract Res Clin Gastroenterol. 2024;70:101915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Berner-Hansen V, Olsen AA, Brandstrup B. Endoscopic treatment of primary aorto-enteric fistulas: A case report and review of literature. World J Gastrointest Endosc. 2021;13:189-197. [PubMed] [DOI] [Full Text] |
| 18. | Jung K, Ahn JY, Jung HY, Cho CJ, Na HK, Jung KW, Lee JH, Kim do H, Choi KD, Song HJ, Lee GH, Kim JH. Outcomes of endoscopically inserted self-expandable metal stents in malignancy according to the type of stent and the site of obstruction. Surg Endosc. 2016;30:4001-4010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Jena A, Chandnani S, Jain S, Sharma V, Rathi P. Efficacy of endoscopic over-the-scope clip fixation for preventing migration of self-expandable metal stents: a systematic review and meta-analysis. Surg Endosc. 2023;37:3410-3418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 20. | Kakkos SK, Papadoulas S, Tsolakis IA. Endovascular management of arterioenteric fistulas: a systemic review and meta-analysis of the literature. J Endovasc Ther. 2011;18:66-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Kehagias D, Mulita F, Panagiotopoulos I, Lampropoulos C, Markopoulos G, Verras GI, Kehagias I. Primary aortoenteric fistula: is endovascular repair the prime option? A review of the literature. Kardiochir Torakochirurgia Pol. 2022;19:220-225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Tellor B, Skrupky LP, Symons W, High E, Micek ST, Mazuski JE. Inadequate Source Control and Inappropriate Antibiotics are Key Determinants of Mortality in Patients with Intra-Abdominal Sepsis and Associated Bacteremia. Surg Infect (Larchmt). 2015;16:785-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 23. | Masuda S, Ichita C, Koizumi K. Practical guide to duodenal stenting for gastric outlet obstruction: Clinical outcomes, selection criteria, placement techniques, and management strategies. World J Gastrointest Endosc. 2025;17:107189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (3)] |