Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.118422
Revised: January 19, 2026
Accepted: February 26, 2026
Published online: April 16, 2026
Processing time: 102 Days and 16.5 Hours
The increasing incidence and economic burden of pancreatic diseases highlight the urgent need for improved diagnostic tools. While advanced imaging mo
To utilize strain histogram (SH) analysis, a semi-quantitative EUS-elastography method, to establish a reference range of SH values in a cohort of healthy controls and to compare them with a group of patients with confirmed CP.
We consecutively enrolled patients undergoing EUS, applying strict exclusion criteria to select subjects without pancreatic masses or ductal anomalies. SH values were measured three times in each of the three main pancreatic regions (head, body, and tail). In addition, demographic data, smoking habits, and alcohol abuse status were collected. Statistical analysis included de
The study analyzed 143 patients (95 healthy controls, 48 with CP). No significant differences were found in de
SH analysis effectively differentiates pancreatic stiffness between healthy individuals and CP patients, offering potential reference values for early diagnosis. This simple and safe technique aids in the identification of preclinical pathological changes. Future large-scale studies are required for external validation.
Core Tip: This pilot study evaluates endoscopic ultrasound-elastography using strain histogram (SH) analysis to improve the diagnosis of chronic pancreatitis (CP), which remains challenging in its early stages. The study compared 95 healthy controls with 48 CP patients to establish reference stiffness values. Results showed that CP patients had significantly higher pancreatic stiffness, indicated by lower SH values compared to healthy subjects (median 72 vs 116, P < 0.01). In conclusion, SH analysis effectively differentiates healthy tissue from CP, providing a safe tool for identifying preclinical pathological changes.
- Citation: Laurenza C, Zignani N, Elvo B, Pignata L, Frigo F, Di Luna I, Soro S, Dagani D, Drago A, Grassia R. Reference range of pancreatic strain histogram by endoscopic ultrasound elastography: A pilot study. World J Gastrointest Endosc 2026; 18(4): 118422
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/118422.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.118422
The incidence and economic impact of pancreatic diseases are rising rapidly. The estimated global incidence of acute pancreatitis is 34 cases per 100000 people annually, affecting both sexes equally and predominantly occurring in middle-aged or elderly individuals. Recurrent acute pancreatitis may develop in up to 20% of patients following their first episode, with 36% progressing to chronic pancreatitis (CP)[1].
In recent years, imaging techniques for diagnosing pancreatic diseases have improved significantly. Magnetic resonance imaging (MRI) and endoscopic ultrasound (EUS) allow for precise evaluation of the pancreatic parenchyma and the diagnosis of both benign and malignant pathologies with high sensitivity and specificity[2]. For focal pancreatic lesions, primarily pancreatic cancer, EUS-guided fine-needle aspiration/biopsy enables definitive histological diagnosis[3]. Regarding diffuse parenchymal diseases, well-defined clinical and morphological criteria exist for acute and advanced-stage CP, often obviating the need for histological confirmation[4].
CP is a fibro-inflammatory syndrome resulting from recurrent inflammation, which causes extensive replacement of parenchyma with fibrotic tissue. This leads to chronic pain, endocrine and exocrine insufficiency, and a reduced quality and expectancy of life. The morphological features of advanced CP, such as pancreatic atrophy, ductal distortion, strictures, and calcifications, are readily identifiable by EUS, computed tomography and MRI[5,6].
In 2009, a consensus of EUS experts proposed the Rosemont classification to standardize the EUS-based diagnosis of CP[7]. The following year, the Japan Pancreas Society (JPS) introduced the JPS criteria and the concept of early CP[8]. These criteria encompass characteristic imaging and histological findings, recurrent upper abdominal pain, abnormal pancreatic enzyme levels, exocrine dysfunction, and continuous alcohol consumption (80 g/day). The specific EUS findings for early-stage CP include five parenchymal features (lobularity with/without honeycombing, hyperechoic foci without shadowing, stranding, and cysts) and two ductal features (dilated side branches and hyperechoic main pan
In 2019, the JPS revised these diagnostic criteria to further improve the specificity of EUS findings[9]. Early diagnosis and intervention are crucial for prognosis, as early CP is considered a theoretically reversible condition[10]. Conversely, advanced CP causes severe complications. The clinical presentation is dominated by chronic pain – typically a dull, sharp sensation in the upper abdomen radiating to the back, often exacerbated by meals[5]. Exocrine insufficiency leads to bloating, steatorrhea, and diarrhea, requiring enzyme replacement, while endocrine dysfunction may necessitate insulin therapy. Furthermore, patients with CP carry an elevated risk of developing pancreatic cancer[11].
Despite these advances, diagnosing early-stage CP remains challenging due to the limited sensitivity and specificity of traditional methods and the lack of a standardized global definition[12]. Consequently, adjunct non-invasive techniques are needed to better characterize early parenchymal changes. Since fibrosis in CP results in increased tissue stiffness, elastography[13], a qualitative and semi-quantitative method for assessing stiffness, offers a potential solution. EUS-elastography (EUS-EG) is already established in the diagnosis of pancreatic cancer[14] and has recently been applied to evaluate parenchymal fibrosis in CP[15].
Various elastography technologies exist, utilizing distinct mechanisms such as strain elastography, shear wave elastography, transient elastography, and acoustic radiation force impulse imaging. While transient elastography and acoustic radiation force impulse are primarily used for liver assessment, strain and shear wave elastography have gained traction in pancreatic applications[16].
Strain histogram (SH) analysis is a semi-quantitative elastographic technique that calculates the mean value of the hue histogram over several compression cycles, providing a quantitative measure of strain within a selected region of interest (ROI)[17]. By utilizing the B-mode image to detect minor structural deformations caused by compression, strain ela
Few studies have applied SH-EUS-EG to assess the severity of pancreatic fibrosis or to differentiate pancreatic masses, though results have been promising[18]. Notably, however, a reference range for SH values in subjects without identifiable pancreatic disease remains undefined. Therefore, we conducted a pilot study to establish a reference range of SH-EUS-EG values in a cohort of patients with no clinical or radiological evidence of pancreatic disease and compared these values with those of patients with confirmed CP.
In this single-center cross-sectional study, we consecutively enrolled all patients undergoing pancreatic EUS between January and July 2022 at the Gastroenterology and Digestive Endoscopy Unit of ASST Cremona. The exclusion criteria were as follows: (1) Age < 18 years; (2) Presence of a pancreatic mass detected by radiological or EUS examination, or any other pancreatic/extra-pancreatic condition causing compression or stenosis of the pancreatic duct. Inclusion was restricted to cases of pancreatic neuroendocrine tumors showing no evidence of ductal obstruction; (3) Pancreatic in
The cohort was stratified into two groups: (1) Healthy control subjects (patients with other indications for EUS who meet the inclusion criteria, primarily for the evaluation of suspected choledocholithiasis); and (2) Patients with confirmed CP (diagnosed using the Rosemont criteria[7]). The following baseline and clinical parameters were collected: (1) Sex; (2) Age; (3) Body mass index (BMI); (4) Alcohol intake (defined according to the Centers for Disease Control and Prevention[19]); and (5) Smoking status. Additionally, three elastography measurements were obtained for each of the three main pancreatic segments (head, body, and tail).
EUS examinations were performed by two experienced endoscopists (performing > 250 procedures per year) using a linear echoendoscope (Pentax Medical EG 3870UTK and EG38-J10UT) connected to a Hitachi (Arietta v70®) ultrasound platform. Written informed consent was obtained from all patients. Procedures were performed on inpatients or outpatients placed in the left lateral decubitus position under conscious sedation. Examinations followed standard clinical practice.
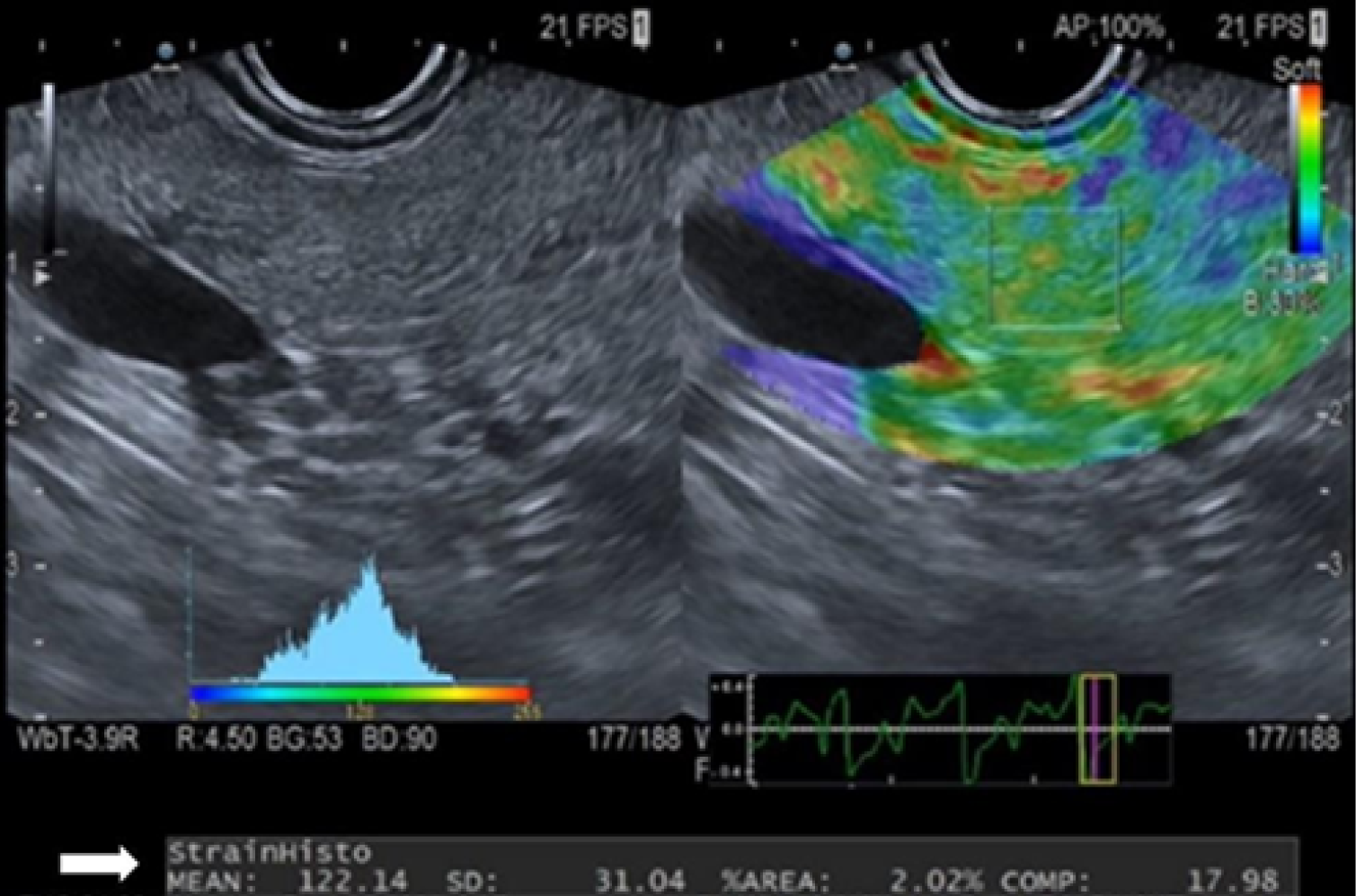
SH analysis was performed after verifying the absence of the aforementioned exclusion criteria. The probe was positioned against the gastric or duodenal wall, applying appropriate pressure to optimize the B-mode image. A frequency of 7.5 MHz was used for the B-mode probe. Measurements were taken exclusively from the pancreatic parenchyma, carefully avoiding the main pancreatic duct and major vascular structures. Elastography and B-mode images were displayed simultaneously in a split-screen format (Figure 1).
The elastography evaluation included a qualitative analysis to detect minor structural deformations (speckle motion) caused by compression within the B-mode image, where the degree of deformation reflects tissue stiffness. Tissue elasticity (on a scale of 0-255) was color-coded and overlaid on the real-time B-mode image. Blue, green/yellow, and red represented hard, intermediate, and soft tissues, respectively. Notably, this scale indicates relative rather than absolute tissue stiffness.
To obtain the SH value, a 1 cm2 square ROI was placed on the pancreatic parenchyma using the qualitative color scale and B-mode image for guidance. Integrated software then automatically generated a histogram, with the X-axis representing tissue elasticity from 0 (hardest) to 255 (softest) and the Y-axis representing the pixel count for each elasticity level within the ROI. The SH value was defined as the mean of all elasticity values within the selected ROI (Figure 1).
To assess vibration energy during analysis, a strain graph displaying waveforms over time was utilized. This graph facilitated the identification of the optimal frame for capturing physiological vibration energy, defined as the point of maximum tissue relaxation during aortic pulsation. To account for potential intra-individual variability, three SH values were obtained from each parenchymal site (head, body, and tail), and the mean value was calculated for analysis.
Descriptive and inferential analyses were conducted to evaluate study variables and assess differences between healthy controls and patients with CP. Continuous variables were compared using the Mann-Whitney test, while categorical variables were assessed using the χ2 test with Yates’ correction.
Subsequently, associations between SH values and other variables were assessed using Spearman’s rank correlation for quantitative variables (age and BMI) and box plots for categorical variables (smoking and alcohol abuse). Finally, a descriptive analysis was performed to identify any sex-based differences within the subgroups.
The sample size was determined based on the working hypotheses and preliminary data, which indicated a mean
A total of 143 patients were enrolled, comprising 95 healthy controls and 48 patients with CP. The study population was predominantly women, with a median age of 65 years and a median BMI of 25 kg/m2, falling within the overweight classification.
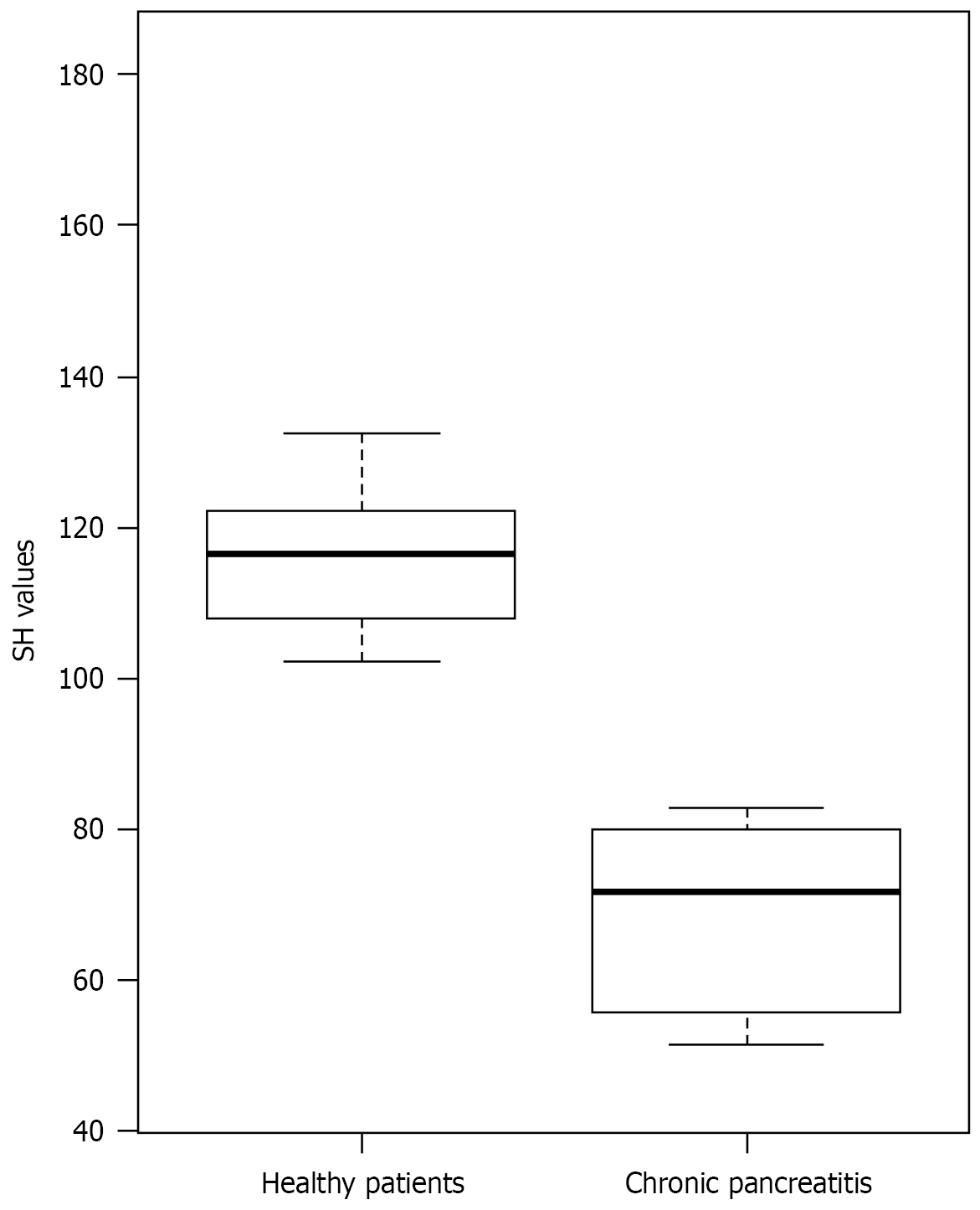
Descriptive and inferential analyses (Table 1) revealed no significant differences between the healthy control and CP subgroups regarding age, sex distribution, or BMI. Conversely, significant clinical and statistical differences were observed in SH values across all pancreatic segments and in the total SH value. Patients with CP exhibited significantly increased pancreatic stiffness, reflected by lower total SH values (median approximately 70) compared to the healthy control group (median approximately 116). These differences are further illustrated in the box plot in Figure 2, which highlights the distribution and main percentiles for each subgroup.
| Baseline | Total (n = 143) | Healthy controls (n = 95) | Chronic pancreatitis (n = 48) | P value |
| Age | 65 (52, 75) | 68 (53, 77) | 62 (51, 72) | |
| Female | 78 (54.5) | 53 (55.8) | 25 (52.1) | |
| Body mass index | 25 (23, 28) | 25 (23, 28) | 26 (22, 28) | |
| Alcohol | 23 (16.1) | 11 (11.6) | 12 (25.0) | 0.07 |
| Smoke | 50 (35.0) | 22 (23.2) | 28 (58.3) | < 0.01 |
| SH head | 108 (78, 122) | 115 (106, 129) | 69 (51, 78) | < 0.01 |
| SH body | 107 (78, 120) | 117 (105, 125) | 71 (56, 79) | < 0.01 |
| SH tail | 104 (80, 120) | 115 (104, 126) | 70 (60, 81) | < 0.01 |
| SH total | 108 (80, 119) | 116 (108, 122) | 72 (56, 80) | < 0.01 |
Finally, a significant difference was observed in the prevalence of alcohol abuse, which was higher in the CP group; similarly, a less pronounced difference was found in the prevalence of smoking within the CP group, with a P value bordering on non-significance.
Subsequently, the association between SH values and other features was evaluated. Specifically, Spearman’s cor
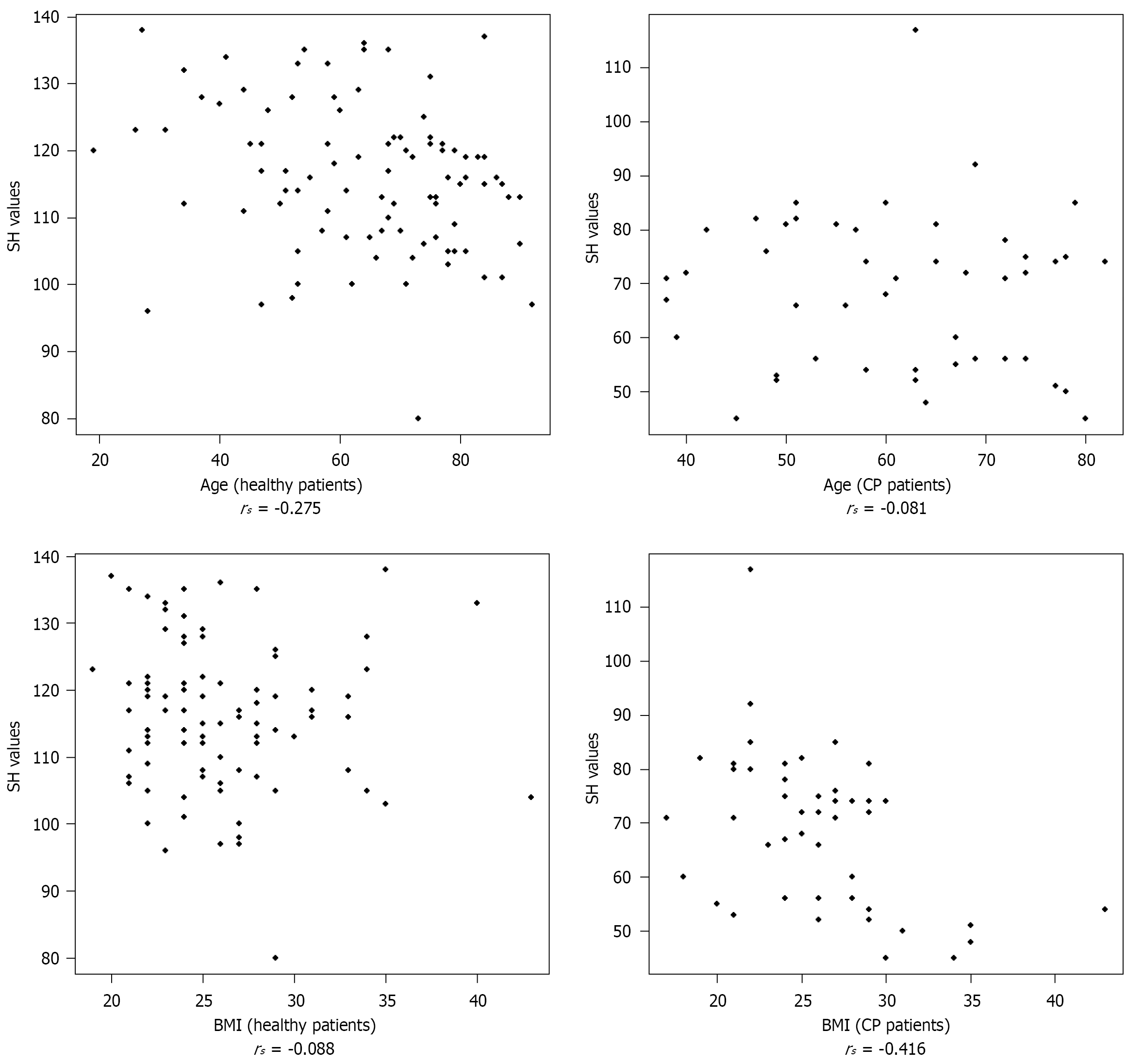
As illustrated in Figure 3, a moderate negative correlation between age and SH values was observed within the healthy control subgroup (rs = -0.275); specifically, as age increases, SH values decrease, reflecting a corresponding increase in pancreatic stiffness. Similarly, BMI was associated with increased pancreatic stiffness, though this correlation reached clinical significance only in the CP group (rs = -0.416), whereas no significant association was found in the control subgroup.
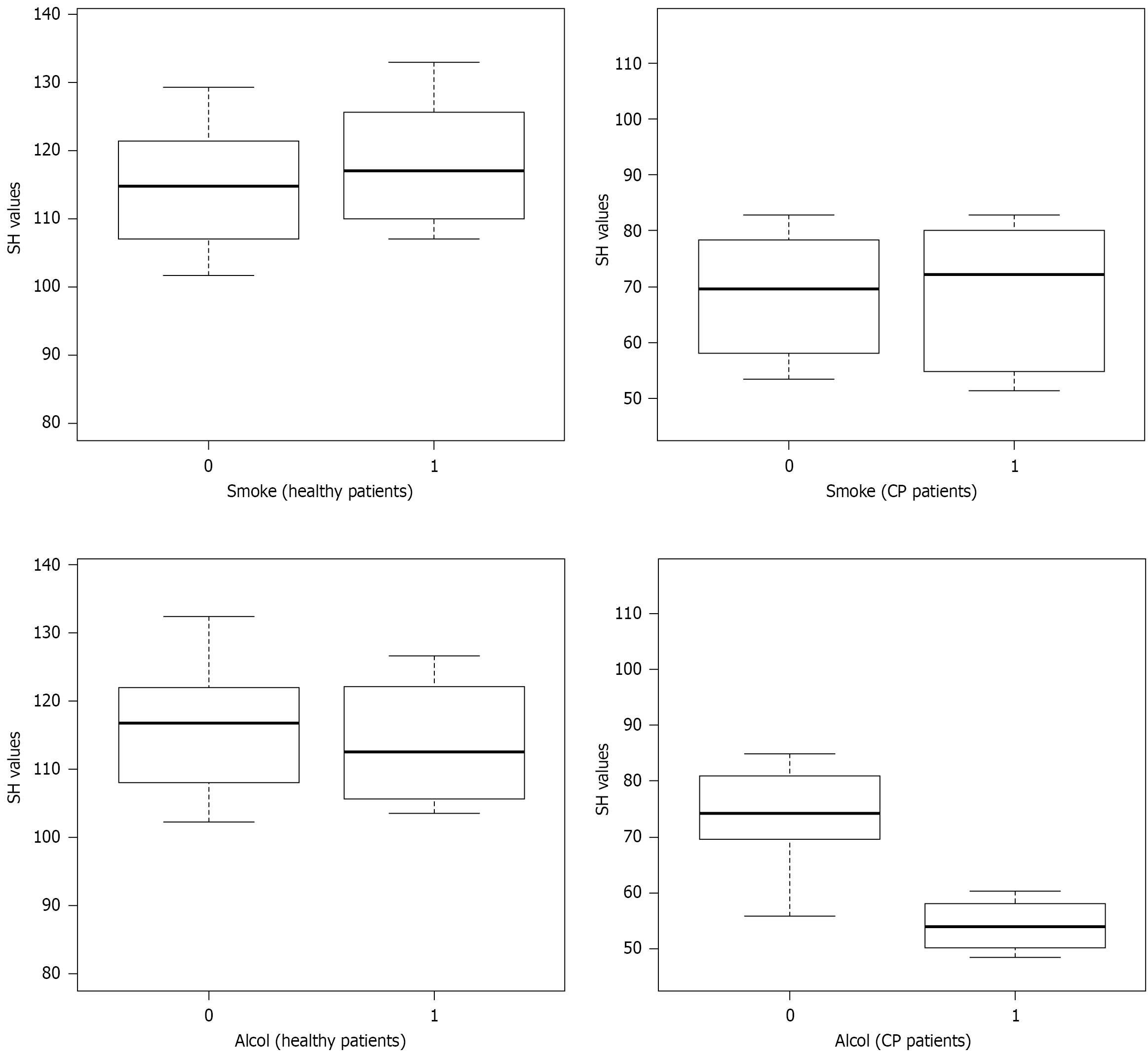
Figure 4 presents boxplots delineating the distribution of categorical variables, such as alcohol abuse and smoking status. Notably, higher SH values were observed among smokers in the control cohort, with no discernible difference in the CP cohort. Conversely, alcohol abuse was associated with markedly lower SH values in the diseased group, while no difference was observed in the healthy group.
Finally, a descriptive analysis based on patient sex was conducted (Tables 2 and 3). Although all subgroups reflected the primary findings of the overall sample (CP patients in both sex-based cohorts exhibited higher pancreatic stiffness and a higher prevalence of smoking and alcohol abuse) no significant differences were found between males and females across any of the analyzed variables.
| Healthy patients | Males (n = 53) | Females (n = 42) |
| Age | 68 (50, 75) | 68 (56, 80) |
| Body mass index | 25 (22, 28) | 26 (24, 28) |
| Alcohol | 6 (11.3) | 5 (11.9) |
| Smoke | 12 (22.6) | 10 (23.8) |
| Strain histogram total | 117 (110, 125) | 114 (107, 120) |
| Chronic pancreatitis patients | Males (n = 25) | Females (n = 23) |
| Age | 60 (52, 71) | 63 (49, 72) |
| Body mass index | 26 (22, 28) | 26 (22, 29) |
| Alcohol | 7 (28) | 5 (21.7) |
| Smoke | 14 (56) | 14 (60.9) |
| Strain histogram total | 72 (55, 81) | 71 (56, 75) |
In this study, SH analysis was used to assess reference ranges for CP and healthy pancreas in patients undergoing EUS, finding a significant difference in pancreatic stiffness between subgroups. Data from both cohorts may suggest a physiological reference range (interquartile range: 108, 122) as well as a pathological range for CP (interquartile range: 56, 80). Furthermore, we presented preliminary findings regarding the association between study variables and SH values; moderate correlations were observed for both BMI and alcohol abuse with increased pancreatic stiffness in CP patients, as well as for age and increased stiffness within the healthy control subgroup. Future large-scale studies are required to validate these predictors and explore additional potential factors.
Over the past 15 years, EUS-EG has been widely applied to assess both focal and diffuse pancreatic pathologies[20], with a particular focus on pancreatic masses[21-24]. These studies have consistently demonstrated that EUS-EG, utilizing various qualitative and quantitative methods, serves as a valid complementary tool to EUS-bronchoscope-guided fine-needle aspiration for the differential diagnosis of pancreatic lesions[12]. Conversely, literature regarding the utility of EUS-EG in early CP remains limited[23].
Domínguez-Muñoz et al[25] proposed a multimodal diagnostic approach for suspected CP, integrating elastography, the endoscopic pancreatic function test (ePFT), and dynamic EUS assessment of ductal compliance following secretin administration. They found that the degree of fibrosis assessed by elastography correlated strongly with ePFT results (r = 0.715, P < 0.0001). Using ePFT as the gold standard, EUS-guided elastography achieved a diagnostic accuracy of 93.4%. However, the complexity of this multimodal methodology may limit its routine clinical applicability. Given that pan
Regarding the specific elastography technique, Yamashita et al[26] compared shear-wave elastography and conventional strain elastography (defined as the strain ratio between the pancreas and the surrounding gastric wall) for diagnosing CP. They concluded that shear-wave elastography was superior to strain ratio analysis, noting that the strain ratio is not an absolute value but is influenced by the positioning of the reference ROI and individual anatomical variability[26].
Nevertheless, strain elastography using histogram analysis has shown promise in diagnosing both pancreatic masses[26,27] and CP[13,18]. Kuwahara et al[18] demonstrated that pancreatic elasticity measured via SH differed significantly according to the Rosemont criteria stage and negatively correlated with the number of EUS features present. Similarly, Itoh et al[13] reported that SH analysis could accurately differentiate histological degrees of fibrosis (normal, mild, marked, severe) in tissue upstream of tumors, with an area under the curve of 0.90. Although neoplasia-induced fibrosis may differ pathophysiologically from non-obstructive CP fibrosis, that study is notable for being the only one to incorporate histological validation.
We also considered the concept of the “aging pancreas”. Radiological studies have shown that pancreatic attenuation decreases with age on computed tomography due to fatty replacement[28], and MRI studies confirm increased adipose tissue starting in the fifth decade[29,30]. However, our multivariate analysis did not identify age as a significant predictor of stiffness in the healthy cohort, suggesting that fibrosis (stiffness), rather than fatty replacement, is the primary driver of SH value reduction in pathology.
Sex-based subgroup analysis revealed no significant differences. This aligns with the relative scarcity of literature on sex-specific pancreatic characteristics, such as the increased hypoechogenicity of the ventral head reported in women[31]. Similarly, data remains limited on the sex-specific pathophysiology of benign conditions like CP[32], as the underlying etiology and sex-dependent mechanisms of pancreatic diseases remain largely unknown[33].
Our study has limitations, primarily the small sample size and the lack of a histological reference standard. However, obtaining histological samples via core needle biopsy or surgery in healthy subjects or early CP patients would be invasive and ethically unjustified. Secondly, inter-observer agreement was not calculated as the data were collected retrospectively from procedures performed according to standard clinical practice, where each operator conducted the examination alone. Furthermore, the images stored in the center’s digital archives did not allow for an adequate calculation of the agreement. Finally, smoking and alcohol consumption were collected as dichotomous variables because quantitative data were unavailable, a limitation inherent to the retrospective design of the study.
Conversely, this study presents several key strengths, including the rigorous selection of the control cohort, the comprehensive collection of clinical variables, and the standardized measurement protocol. Most importantly, our findings have significant clinical applicability. The identified ranges can serve as reference values for diagnosing early CP, particularly when values fall into an indeterminate range (e.g., 80-100). As EUS elastography gains prominence, validating these reference ranges could provide a critical, objective criterion for the early detection of CP.
Alterations in pancreatic elastography can help identify pathological changes in the pancreatic parenchyma at a preclinical stage, thereby supporting the diagnosis of early forms of diffuse pancreatic disease, mainly early CP. The SH requires no patient preparation and is a simple, inexpensive, and safe technique that only slightly increases procedure time. Although our study requires external validation, preferably with larger sample sizes or in prospective studies, all these informations could, in the future, improve disease diagnosis or even reduce the need to collect histological samples.
| 1. | Xiao AY, Tan ML, Wu LM, Asrani VM, Windsor JA, Yadav D, Petrov MS. Global incidence and mortality of pancreatic diseases: a systematic review, meta-analysis, and meta-regression of population-based cohort studies. Lancet Gastroenterol Hepatol. 2016;1:45-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 647] [Cited by in RCA: 547] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 2. | Zhou S, Buxbaum J. Advanced Imaging of the Biliary System and Pancreas. Dig Dis Sci. 2022;67:1599-1612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 3. | Hassan GM, Laporte L, Paquin SC, Menard C, Sahai AV, Mâsse B, Trottier H. Endoscopic Ultrasound Guided Fine Needle Aspiration versus Endoscopic Ultrasound Guided Fine Needle Biopsy for Pancreatic Cancer Diagnosis: A Systematic Review and Meta-Analysis. Diagnostics (Basel). 2022;12:2951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 4. | Shimizu K, Ito T, Irisawa A, Ohtsuka T, Ohara H, Kanno A, Kida M, Sakagami J, Sata N, Takeyama Y, Tahara J, Hirota M, Fujimori N, Masamune A, Mochida S, Enomoto N, Shimosegawa T, Koike K. Evidence-based clinical practice guidelines for chronic pancreatitis 2021. J Gastroenterol. 2022;57:709-724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 58] [Reference Citation Analysis (1)] |
| 5. | Issa Y, Kempeneers MA, van Santvoort HC, Bollen TL, Bipat S, Boermeester MA. Diagnostic performance of imaging modalities in chronic pancreatitis: a systematic review and meta-analysis. Eur Radiol. 2017;27:3820-3844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (2)] |
| 6. | Ren S, Qian L, Daniels MJ, Duan S, Chen R, Wang Z. Evaluation of contrast-enhanced computed tomography for the differential diagnosis of hypovascular pancreatic neuroendocrine tumors from chronic mass-forming pancreatitis. Eur J Radiol. 2020;133:109360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 7. | Catalano MF, Sahai A, Levy M, Romagnuolo J, Wiersema M, Brugge W, Freeman M, Yamao K, Canto M, Hernandez LV. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest Endosc. 2009;69:1251-1261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 353] [Article Influence: 20.8] [Reference Citation Analysis (2)] |
| 8. | Shimosegawa T, Kataoka K, Kamisawa T, Miyakawa H, Ohara H, Ito T, Naruse S, Sata N, Suda K, Hirota M, Takeyama Y, Shiratori K, Hatori T, Otsuki M, Atomi Y, Sugano K, Tanaka M. The revised Japanese clinical diagnostic criteria for chronic pancreatitis. J Gastroenterol. 2010;45:584-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 144] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 9. | Masamune A, Nabeshima T, Kikuta K, Hamada S, Nakano E, Kume K, Kanno A, Sato A, Tachibana Y, Inatomi O, Yamamoto S, Ikeura T, Futagami S, Taguchi M, Hanada K, Shimizu K, Kageoka M, Saito T, Eguchi T, Kubota K, Takenaka M, Mima A, Irisawa A, Ito T, Andoh A, Inui K, Takeyama Y, Yamaue H, Okazaki K, Shimosegawa T. Prospective study of early chronic pancreatitis diagnosed based on the Japanese diagnostic criteria. J Gastroenterol. 2019;54:928-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 10. | Ito T, Ishiguro H, Ohara H, Kamisawa T, Sakagami J, Sata N, Takeyama Y, Hirota M, Miyakawa H, Igarashi H, Lee L, Fujiyama T, Hijioka M, Ueda K, Tachibana Y, Sogame Y, Yasuda H, Kato R, Kataoka K, Shiratori K, Sugiyama M, Okazaki K, Kawa S, Tando Y, Kinoshita Y, Watanabe M, Shimosegawa T. Evidence-based clinical practice guidelines for chronic pancreatitis 2015. J Gastroenterol. 2016;51:85-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 109] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 11. | Bang UC, Benfield T, Hyldstrup L, Bendtsen F, Beck Jensen JE. Mortality, cancer, and comorbidities associated with chronic pancreatitis: a Danish nationwide matched-cohort study. Gastroenterology. 2014;146:989-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 169] [Article Influence: 14.1] [Reference Citation Analysis (2)] |
| 12. | Ge QC, Dietrich CF, Bhutani MS, Zhang BZ, Zhang Y, Wang YD, Zhang JJ, Wu YF, Sun SY, Guo JT. Comprehensive review of diagnostic modalities for early chronic pancreatitis. World J Gastroenterol. 2021;27:4342-4357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 13. | Itoh Y, Itoh A, Kawashima H, Ohno E, Nakamura Y, Hiramatsu T, Sugimoto H, Sumi H, Hayashi D, Kuwahara T, Morishima T, Funasaka K, Nakamura M, Miyahara R, Ohmiya N, Katano Y, Ishigami M, Goto H, Hirooka Y. Quantitative analysis of diagnosing pancreatic fibrosis using EUS-elastography (comparison with surgical specimens). J Gastroenterol. 2014;49:1183-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 14. | Săftoiu A, Vilmann P, Gorunescu F, Janssen J, Hocke M, Larsen M, Iglesias-Garcia J, Arcidiacono P, Will U, Giovannini M, Dietrich C, Havre R, Gheorghe C, McKay C, Gheonea DI, Ciurea T; European EUS Elastography Multicentric Study Group. Accuracy of endoscopic ultrasound elastography used for differential diagnosis of focal pancreatic masses: a multicenter study. Endoscopy. 2011;43:596-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 15. | Yamashita Y, Ashida R, Kitano M. Imaging of Fibrosis in Chronic Pancreatitis. Front Physiol. 2021;12:800516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Hirooka Y, Kuwahara T, Irisawa A, Itokawa F, Uchida H, Sasahira N, Kawada N, Itoh Y, Shiina T. JSUM ultrasound elastography practice guidelines: pancreas. J Med Ultrason (2001). 2015;42:151-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (4)] |
| 17. | Dhar J, Samanta J. The expanding role of endoscopic ultrasound elastography. Clin J Gastroenterol. 2022;15:841-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (2)] |
| 18. | Kuwahara T, Hirooka Y, Kawashima H, Ohno E, Ishikawa T, Kawai M, Suhara H, Takeyama T, Hashizume K, Koya T, Tanaka H, Sakai D, Yamamura T, Furukawa K, Funasaka K, Nakamura M, Miyahara R, Watanabe O, Ishigami M, Hashimoto S, Goto H. Quantitative diagnosis of chronic pancreatitis using EUS elastography. J Gastroenterol. 2017;52:868-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (2)] |
| 19. | CDC Archive. Glossary – Alcohol. 2023. Available from: https://archive.cdc.gov/#/details?url=https://www.cdc.gov/nchs/nhis/alcohol/alcohol_glossary.htm. |
| 20. | Janssen J, Schlörer E, Greiner L. EUS elastography of the pancreas: feasibility and pattern description of the normal pancreas, chronic pancreatitis, and focal pancreatic lesions. Gastrointest Endosc. 2007;65:971-978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 177] [Article Influence: 9.3] [Reference Citation Analysis (2)] |
| 21. | Lu Y, Chen L, Li C, Chen H, Chen J. Diagnostic utility of endoscopic ultrasonography-elastography in the evaluation of solid pancreatic masses: a meta-analysis and systematic review. Med Ultrason. 2017;19:150-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (2)] |
| 22. | Zhang B, Zhu F, Li P, Yu S, Zhao Y, Li M. Endoscopic ultrasound elastography in the diagnosis of pancreatic masses: A meta-analysis. Pancreatology. 2018;18:833-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (3)] |
| 23. | Pei Q, Zou X, Zhang X, Chen M, Guo Y, Luo H. Diagnostic value of EUS elastography in differentiation of benign and malignant solid pancreatic masses: a meta-analysis. Pancreatology. 2012;12:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 24. | Hu DM, Gong TT, Zhu Q. Endoscopic ultrasound elastography for differential diagnosis of pancreatic masses: a meta-analysis. Dig Dis Sci. 2013;58:1125-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 25. | Domínguez-Muñoz JE, Lariño-Noia J, Alvarez-Castro A, Nieto L, Lojo S, Leal S, de la Iglesia-Garcia D, Iglesias-Garcia J. Endoscopic ultrasound-based multimodal evaluation of the pancreas in patients with suspected early chronic pancreatitis. United European Gastroenterol J. 2020;8:790-797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 26. | Yamashita Y, Yamazaki H, Shimokawa T, Kawaji Y, Tamumra T, Hatamaru K, Itonaga M, Ashida R, Kitano M. Shear-wave versus strain elastography in endoscopic ultrasound for the diagnosis of chronic pancreatitis. Pancreatology. 2023;23:35-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 27. | Okasha HH, El-Amin H, Sayed ZE, Maghraby AAE. Role of Endoscopic Ultrasound Elastography Strain Histograms in the Evaluation of Patients with Pancreatic Masses. Turk J Gastroenterol. 2021;32:519-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 28. | Meier JM, Alavi A, Iruvuri S, Alzeair S, Parker R, Houseni M, Hernandez-Pampaloni M, Mong A, Torigian DA. Assessment of age-related changes in abdominal organ structure and function with computed tomography and positron emission tomography. Semin Nucl Med. 2007;37:154-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 76] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 29. | Sato T, Ito K, Tamada T, Sone T, Noda Y, Higaki A, Kanki A, Tanimoto D, Higashi H. Age-related changes in normal adult pancreas: MR imaging evaluation. Eur J Radiol. 2012;81:2093-2098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 30. | Li J, Xie Y, Yuan F, Song B, Tang C. Noninvasive quantification of pancreatic fat in healthy male population using chemical shift magnetic resonance imaging: effect of aging on pancreatic fat content. Pancreas. 2011;40:295-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 31. | Meier J, Lucius C, Möller K, Jenssen C, Zervides C, Gschmack AM, Dong Y, Srivastava D, Dietrich CF. Pancreatic ultrasound: An update of measurements, reference values, and variations of the pancreas. Ultrasound Int Open. 2024;10:a23899085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 32. | Drake M, Dodwad SJ, Davis J, Kao LS, Cao Y, Ko TC. Sex-Related Differences of Acute and Chronic Pancreatitis in Adults. J Clin Med. 2021;10:300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 33. | Wang M, Gorelick F, Bhargava A. Sex Differences in the Exocrine Pancreas and Associated Diseases. Cell Mol Gastroenterol Hepatol. 2021;12:427-441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |