Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.118485
Revised: January 29, 2026
Accepted: March 13, 2026
Published online: April 16, 2026
Processing time: 99 Days and 22 Hours
Immunoglobulin G4-related sclerosing cholangitis (IgG4-SC) is a rare auto
An 88-year-old male presented with jaundice, epigastric pain, and biochemical evidence of cholestasis. Laboratory tests revealed markedly elevated serum IgG4 (2460 mg/dL), and imaging studies demonstrated bile duct wall thickening with multiple strictures. Histopathological examination showed minimal fibrosis accompanied by chronic inflammatory infiltration. After excluding malignancy, a diagnosis of isolated IgG4-SC was established. Steroid therapy with prednisone (40 mg/day) led to significant improvement in symptoms and biochemical mar
Isolated IgG4-SC mimics primary sclerosing cholangitis or cholangiocarcinoma, making early, accurate differentiation crucial to guide therapy and avoid unne
Core Tip: Immunoglobulin G4-related sclerosing cholangitis (IgG4-SC) is a relatively rare autoimmune disease that typically presents with bile duct stenosis and cholestasis. It is frequently associated with autoimmune pancreatitis; however, a small subset of patients develop IgG4-SC without any extra-biliary involvement from IgG4-related disease. This form, known as isolated IgG4-SC, is particularly challenging to diagnose. This report describes a case of isolated IgG4-SC and suggests that clinicians begin considering this condition in patients who present with unexplained biliary strictures and cholestasis, especially when serum IgG4 levels are elevated.
- Citation: Xia Y, Zhu JD, Su T, Shi L, Zhao SL, Xu HW, Xu CQ. Immunoglobulin G4-related sclerosing cholangitis: A case report. World J Gastrointest Endosc 2026; 18(4): 118485
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/118485.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.118485
Immunoglobulin G4-related sclerosing cholangitis (IgG4-SC) is an autoimmune disorder of unknown pathogenesis characterized by elevated serum IgG4 levels, dense infiltration of IgG4-positive plasma cells and lymphocytes, fibrosis of the bile duct wall, and obliterative phlebitis. It is considered the biliary manifestation of IgG4-related disease (RD) and often presents with biliary strictures and cholestasis, and frequent coexistence with other IgG4-RDs, particularly autoimmune pancreatitis (AIP)[1-3]. Tanaka et al[4] conducted the first epidemiological study of IgG4-SC in Japan in 2019, reporting a prevalence of 2.18 per 100000 individuals. The condition predominantly affects older males, with a mean onset age of 67 years and a male-to-female ratio of approximately 4:1. Notably, 83.7% of patients had concomitant type 1 AIP. To date, no epidemiological data on IgG4-SC have been reported in China.
Differentiating IgG4-SC from primary sclerosing cholangitis (PSC), cholangiocarcinoma, pancreatic cancer, and other hepatobiliary conditions is essential, as treatment strategies and prognoses vary significantly. Glucocorticoids are currently the first-line therapy for IgG4-SC, with most patients achieving complete resolution of biliary strictures and normalization of liver function following treatment. The presence of coexisting AIP often enables earlier recognition of IgG4-SC; however, diagnosing isolated IgG4-SC in the absence of AIP remains particularly challenging[1-3]. Currently, two major diagnostic frameworks are used: The Histology, Imaging, Serology, Other organ involvement, Response to steroid therapy (HISORT) criteria developed in the United States and the Japanese Clinical Diagnostic Criteria 2020[1,5]. Early and accurate diagnosis - integrating clinical presentation, serological testing, imaging findings, and histopathological evidence - is critical for optimizing patient outcomes. This article reports a case of IgG4-SC presenting primarily with jaundice and abdominal pain, highlighting the diagnostic process and clinical management of this condition.
An 88-year-old male patient presented with generalized jaundice, scleral icterus, and epigastric pain for one week.
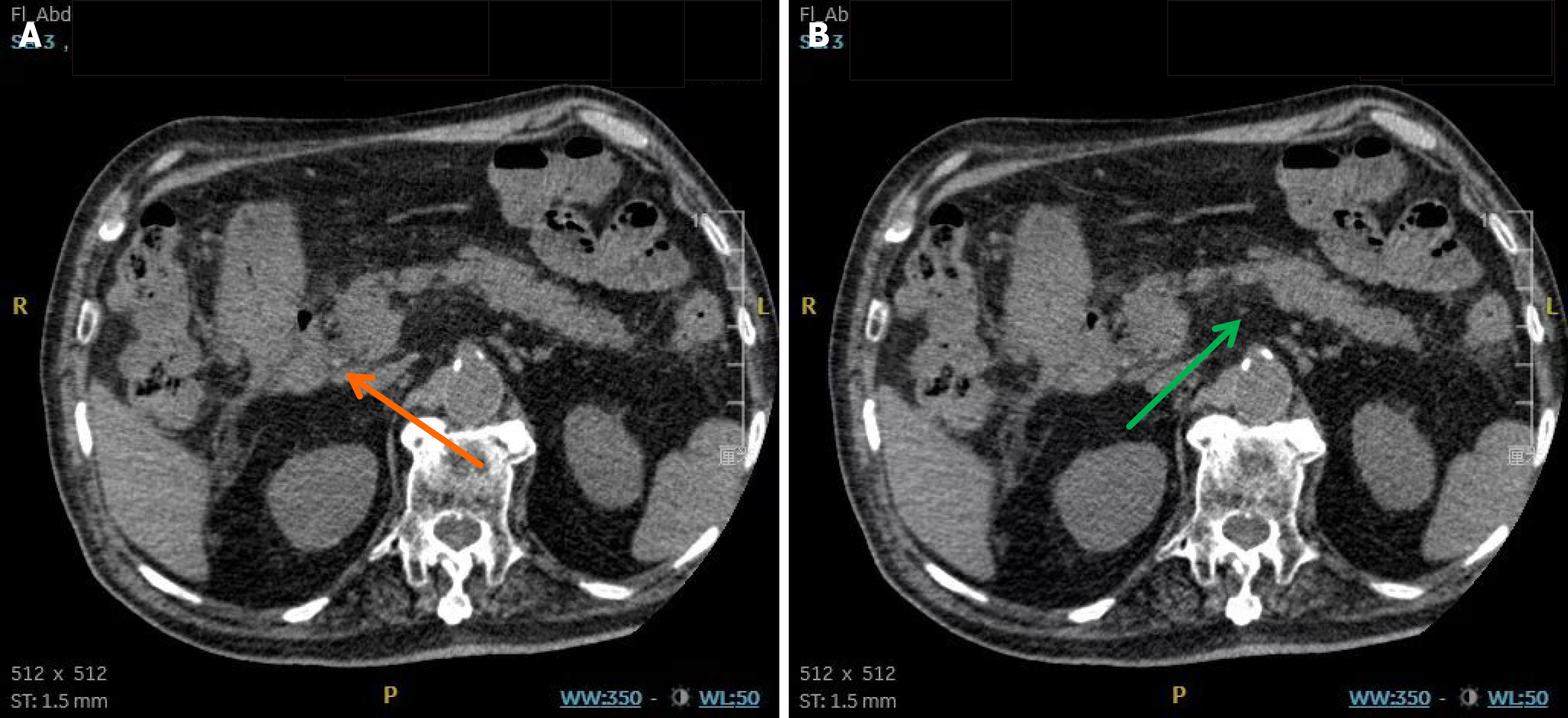
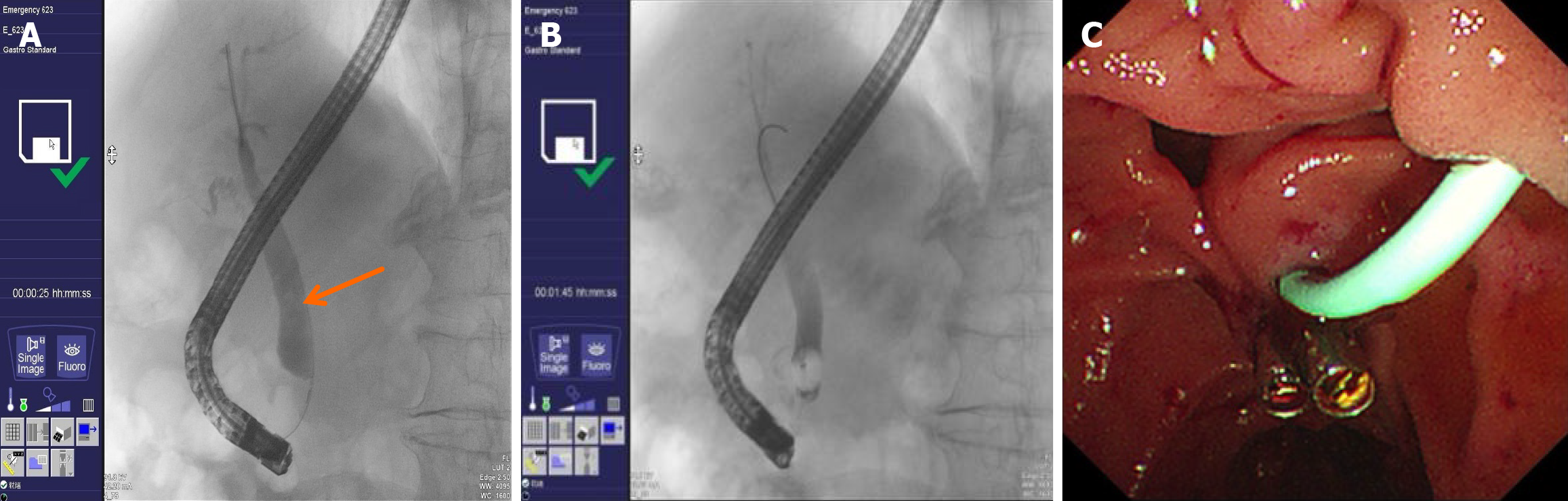
Three months before the current admission (June 29, 2024), the patient experienced recurrent abdominal pain, bloating, and vomiting. Laboratory testing (Table 1) revealed elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and γ-glutamyl transpeptidase (GGT) levels, with bilirubin levels within normal range. Abdominal computed tomography (CT) indicated gallbladder stones, common bile duct stones, and hazy peripancreatic fat stranding, raising concern for possible pancreatitis (Figure 1). Based on these findings, biliary pancreatitis was suspected. Then the patient underwent endoscopic retrograde cholangiopancreatography (ERCP) with endoscopic sphincterotomy, balloon stone extraction, and endoscopic retrograde biliary drainage on July 1, 2024, resulting in resolution of symptoms (Figure 2). No large stones were found during this ERCP, only sludge-like stones, suggesting that larger stones had passed spontaneously. The patient recovered well postoperatively and was discharged. The patient was discharged without timely follow-up and was readmitted on September 17, 2024, due to abdominal pain and persistent jaundice for one week. Repeated laboratory studies demonstrated worsening liver enzyme elevations and hyperbilirubinemia compared with previous levels. Abdominal magnetic resonance imaging (MRI) revealed heterogeneous signal intensity in the distal common bile duct with intra- and extrahepatic biliary dilation, along with multiple gallbladder stones.
| Variables | June 27, 2024 | September 18, 2024 | September 20, 2024 | September 23, 2024 | October 16, 2024 | October 18, 2024 | November 14, 2024 | July 8, 2025 |
| Pancreatic enzymes | ||||||||
| LPS (U/L) | 49 | 53 | ||||||
| P-AMY (U/L) | 74 | 55 | ||||||
| Hepatic enzymes | ||||||||
| AST (U/L) | 71 | 127 | 114 | 88 | 69 | 54 | 34 | 21.7 |
| ALT (U/L) | 84 | 77 | 62 | 44 | 50 | 43 | 36 | 12.7 |
| GGT (U/L) | 494 | 754 | 734 | 507 | 109 | 92 | 91 | 58.8 |
| ALP (U/L) | 507 | 719 | 640 | 460 | 256 | 223 | 97 | 95.9 |
| ALB (g/L) | 29 | 24.8 | 27.7 | 28.8 | 32.9 | 29.5 | 29 | 40.3 |
| TBIL (μmol/L) | 11 | 169 | 216 | 186 | 67 | 53 | 20.4 | 14.90 |
| DBIL (μmol/L) | 4.6 | 100 | 133 | 110 | 32 | 25 | 6.2 | 4.4 |
| IBIL (μmol/L ) | 7.37 | 69.5 | 83 | 76.5 | 35.3 | 28.2 | 14.3 | 10.5 |
| Other indicators | ||||||||
| IgG4 (mg/L) | 24600 | 19000 | 14500 | |||||
| CEA (ng/mL) | 1.13 | 1.05 | ||||||
| CA199 (IU/mL) | 753 | 29.3 | ||||||
The patient’s medical history was significant for cerebral infarction and coronary heart disease.
There was no notable family history of hepatobiliary or autoimmune diseases, and the patient’s personal history was unremarkable.
The patient exhibited generalized jaundice involving both the skin and sclera, accompanied by pruritus. The abdomen was flat and soft, with tenderness localized to the epigastric region but without rebound tenderness.
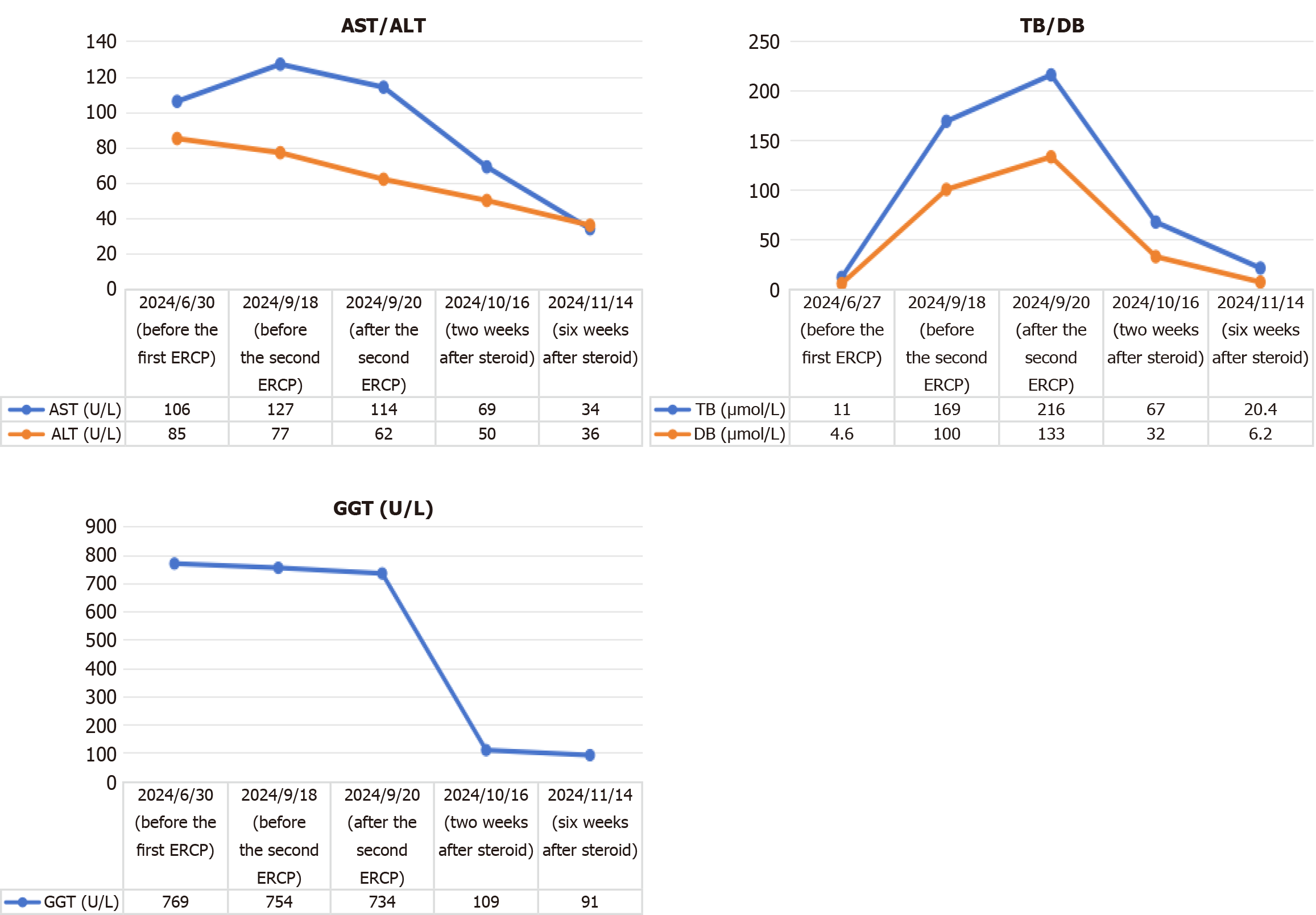
Blood tests performed on September 18, 2024, showed marked abnormalities: Total bilirubin (TBIL) 169.77 µmol/L; direct bilirubin 100.27 µmol/L; ALP 719 U/L; GGT 754 U/L; AST 127 U/L; and ALT 77 U/L. All values were elevated relative to normal reference ranges.
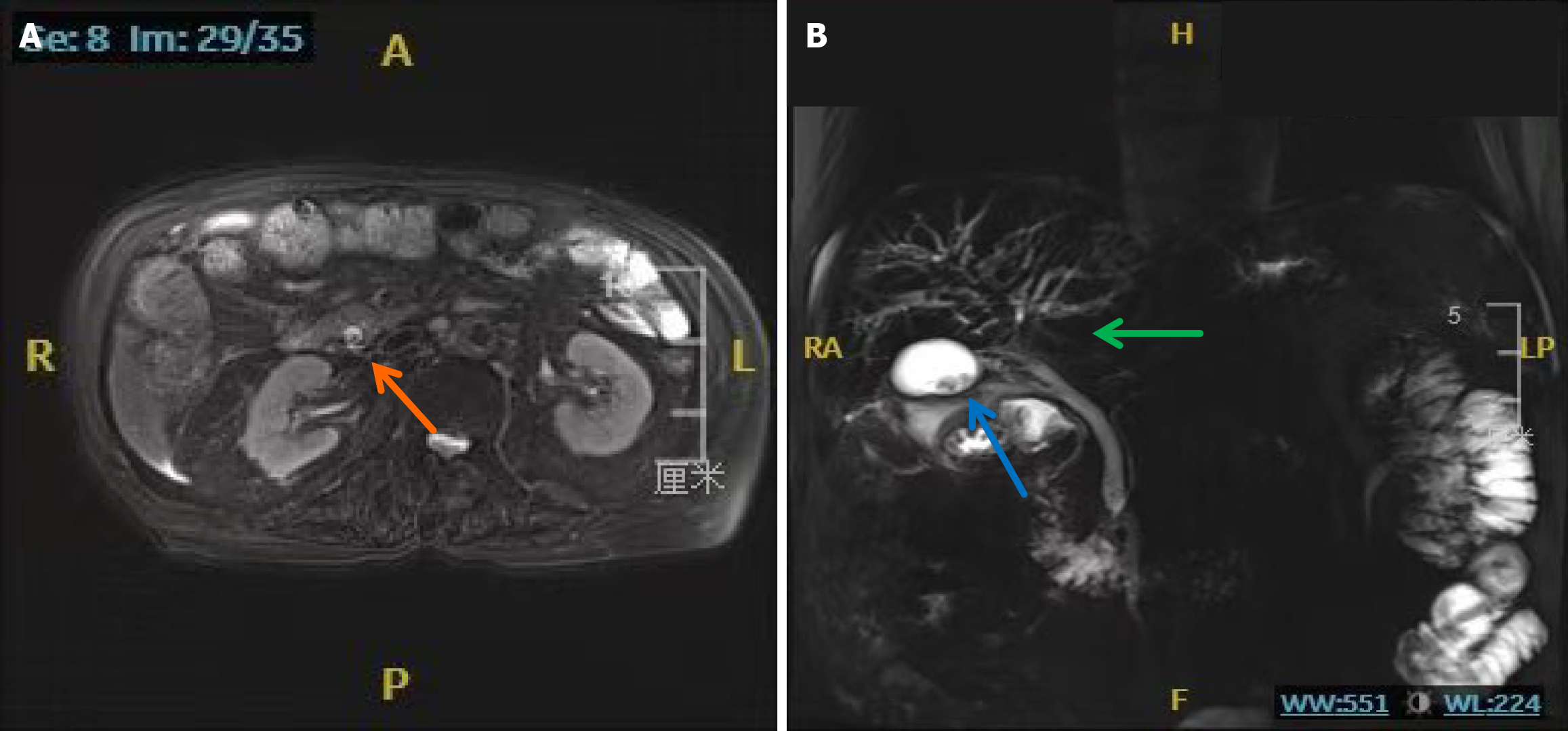
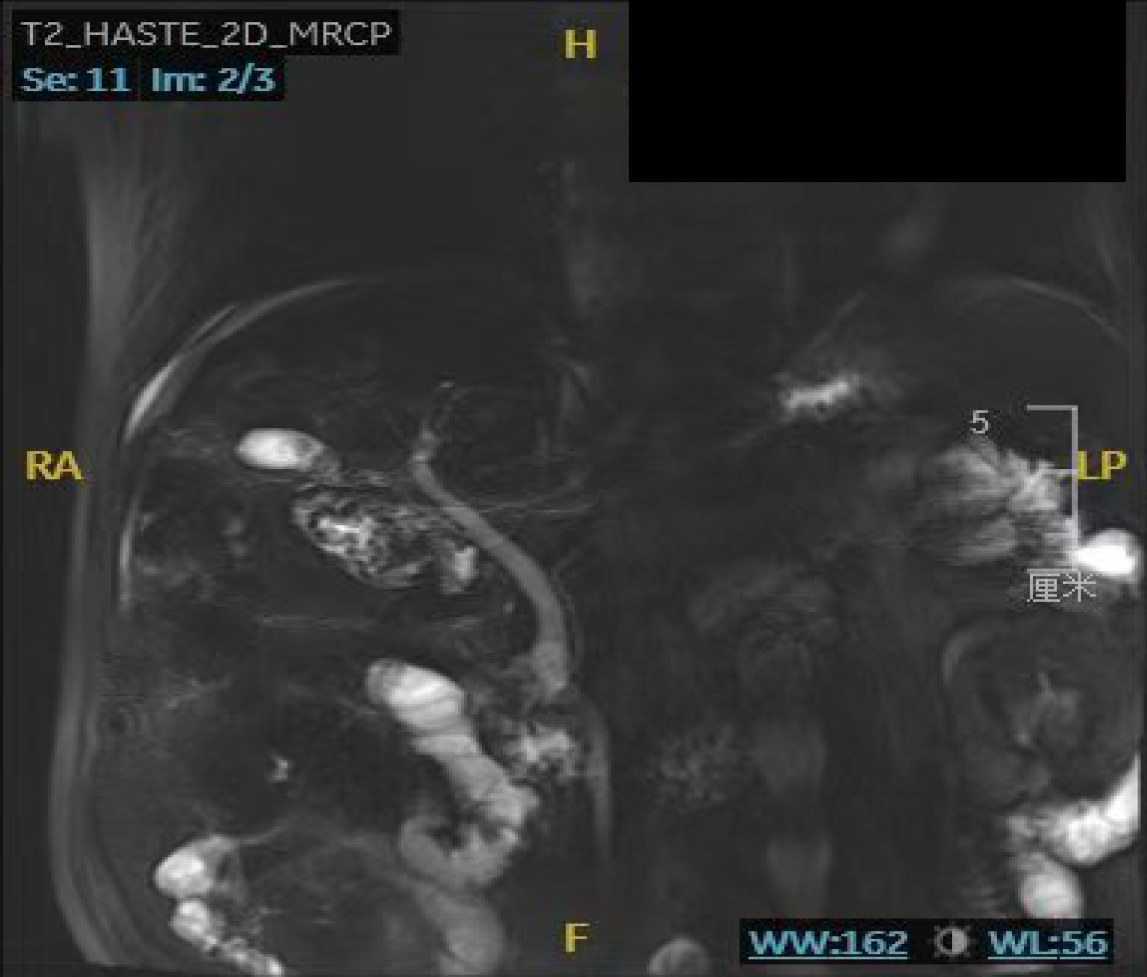
All MRI data were acquired using a MAGNETOM Skyra 3.0T MR scanner (Siemens Healthcare, Erlangen, Germany). Axial fat-suppressed T2-weighted images were obtained with a motion-corrected BLADE (PROPELLER) sequence employing continuous table motion (MBH). Key sequence parameters were as follows: Repetition time = 2400 milliseconds; echo time = 78 milliseconds; flip angle = 89; slice thickness = 5.5 mm. In addition, two-dimensional magnetic resonance cholangiopancreatography (T2-HASTE-2D-MRCP) was performed for a comprehensive evaluation of the biliary and pancreatic ducts. Upper abdominal MRI with magnetic resonance cholangiopancreatography (MRI/MRCP) demonstrated gallbladder stones, common bile duct stones, mild intrahepatic and extrahepatic bile duct dilation, thickening of the cystic gall duct wall with luminal narrowing; features of cholecystitis; abnormal enhancement in the left lobe of the liver, suggestive of abnormal perfusion; and absence of contrast agent excretion in the hepatobiliary phase, indicating possible liver damage (Figure 3).
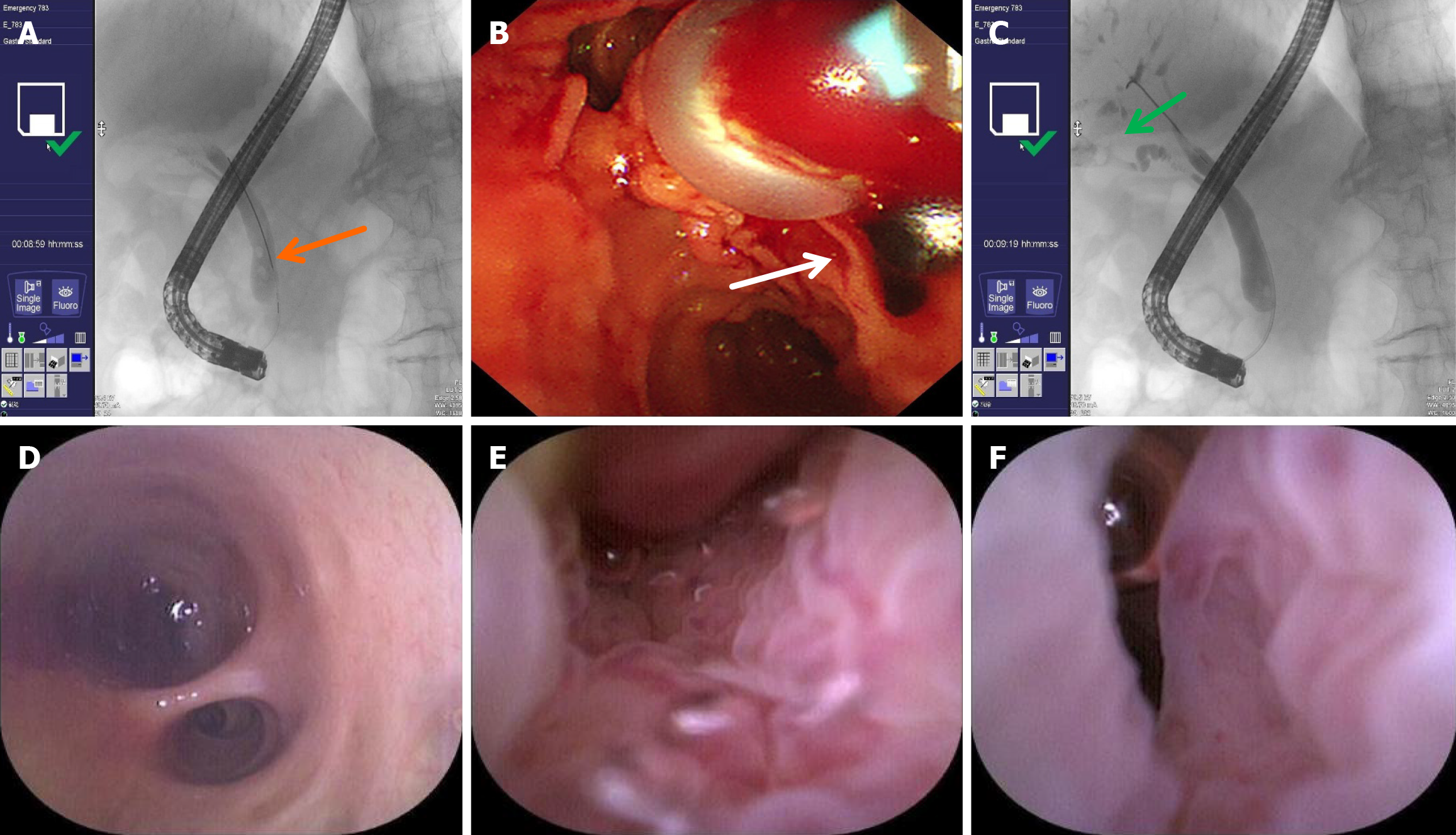
Based on the integrated clinical presentation, laboratory data, and imaging findings, IgG4-SC was highly suspected. For definitive diagnosis, the patient underwent ERCP with biliary balloon dilation, balloon stone extraction, cholangioscopy-guided biopsy, brush cytology, and endoscopic retrograde biliary drainage on September 19, 2024 (Figure 4). Intraoperative cholangiography and endoscopic inspection revealed segmental strictures within the intrahepatic bile ducts and a narrowing that extended from the common hepatic duct to the hepatic bifurcation, with dilation of the intra- and extrahepatic bile ducts. Brush cytology was obtained from the hilar ducts, and a plastic biliary stent (7 Fr, 12 cm) was placed. Postoperatively, the patient was managed with proton-pump inhibitor 40 mg twice daily, octreotide 0.6 mg daily via continuous infusion, cefoperazone sodium and sulbactam sodium 3 g every 12 hours, and intravenous fluids. On the fourth day after ERCP, the patient developed a fever of 38 °C. Due to concerns that drainage via the biliary plastic stent was insufficient, the patient underwent percutaneous transhepatic cholangial drainage.
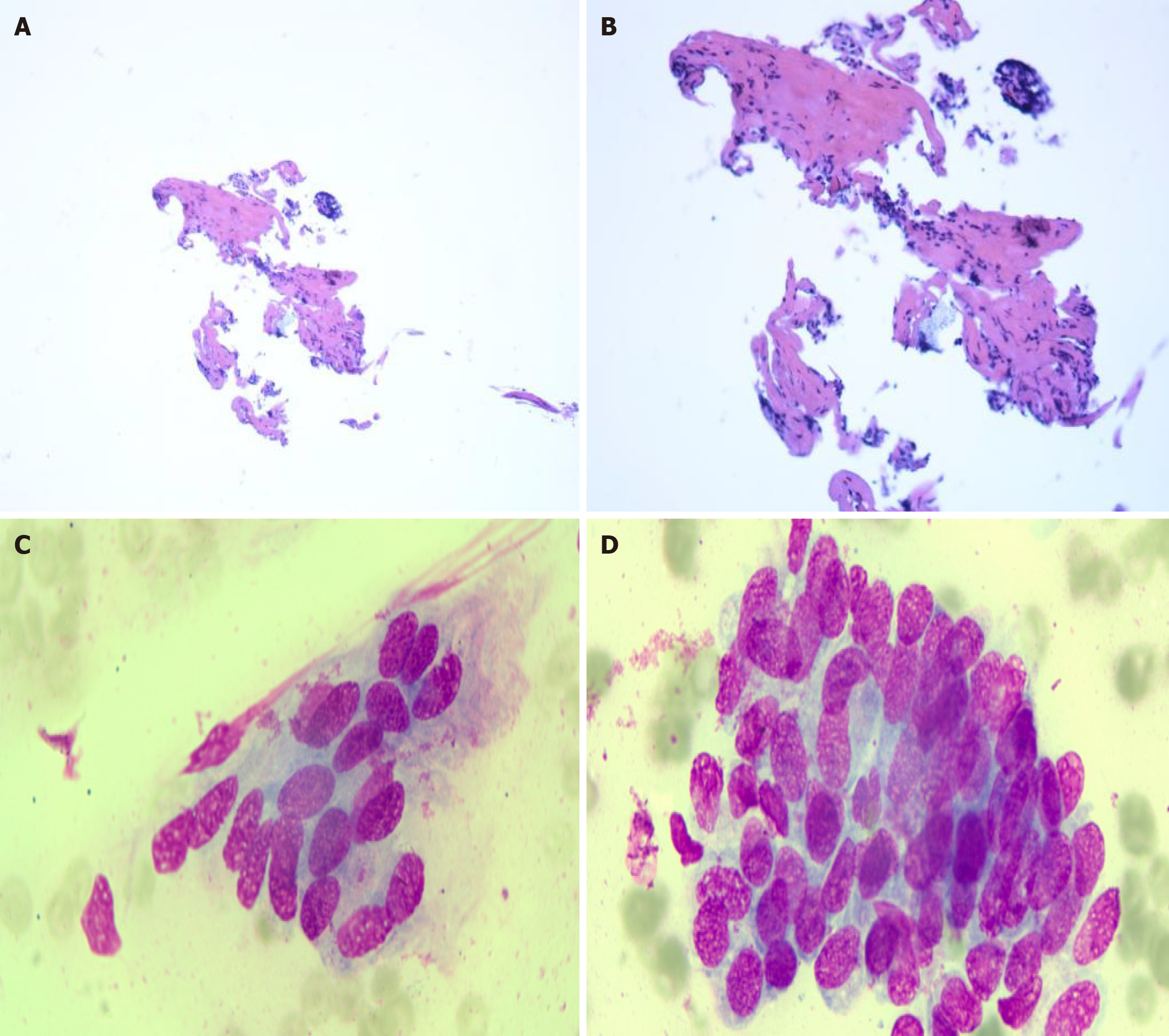
Relevant laboratory values at that time included white blood cells: 10.40 × 109/L (neutrophils, 88.4%), lipase: 53 U/L, amylase: 99 U/L, pancreatic amylase: 55 U/L, AST: 114 U/L, ALT: 62 U/L, GGT: 734 U/L, ALP: 640 U/L, TBIL: 216 μmol/L, direct bilirubin: 133 μmol/L, IBIL: 83 μmol/L, carbohydrate antigen 19-9: 753 IU/mL, IgE: 237 IU/mL, IgG: 36.8 g/L, and IgG4: 24600 mg/L. Autoimmune liver antibodies were negative. Histopathological examination of the bile duct from the common hepatic duct to the hepatic bifurcation showed minimal fibrosis with chronic inflammatory cell infiltration (Figure 5A and B). Due to the small size of the biopsy specimen, the proportion of IgG4-positive plasma cells could not be determined, and thus, this diagnostic criterion could not be applied. Cytological analysis of brushing showed epithelial cells with nuclear enlargement, increased nuclear-to-cytoplasmic ratio, coarse chromatin with hyperchromasia, and mild atypia (Figure 5C and D).
After pathological exclusion of cholangiocarcinoma, the patient was diagnosed with IgG4-SC based on the clinical picture and investigative findings. Diagnosis was supported by the following criteria: (1) Clinical manifestations: Scleral icterus, pruritus, and epigastric pain; (2) Serology: Markedly elevated serum IgG - greater than four times the upper limit of normal; (3) Imaging: Concentric bile duct wall thickening with segmental luminal occlusion involving the cystic duct, the bilateral hepatic ducts at the porta hepatis, the common hepatic duct, and the proximal common bile duct; and (4) Histopathology: Minimal fibrosis with chronic inflammatory cell infiltration extending from the common bile duct to the hepatic bifurcation.
The patient received oral glucocorticoid therapy with prednisone 40 mg once daily for two weeks, followed by a gradual taper of 5 mg every two weeks until a maintenance dose of 5 mg daily was reached.
The patient was admitted on October 15, 2024, for re-evaluation, showing improvement in various parameters, and the percutaneous transhepatic cholangial drainage tube was removed. On November 14, 2024, the patient was readmitted for endoscopic removal of the biliary stent. Following steroid therapy and stent removal, scleral icterus resolved, and the patient reported symptomatic improvement. Laboratory showed marked improvement in cholestatic and hepatocellular enzymes (Figure 6), and follow-up imaging demonstrated regression of biliary wall thickening and reduction of ductal dilation, consistent with disease remission (Figure 7). During the follow-up in July 2025, the patient was unable to recheck IgG4 due to limited conditions; however, liver function tests showed that ALT, AST, GGT, and TBIL were all within normal ranges.
IgG4-SC is the major hepatobiliary manifestation of IgG4-RD, an immune-mediated, systemic fibroinflammatory disorder. The pathogenesis of IgG4-RD is multifactorial, implicating genetic susceptibility, environmental factors, autoantigens, and dysregulated T-cell subsets[3]. IgG4-RD can involve multiple organ systems; the pancreas and salivary glands are most commonly affected, while the bile ducts, thyroid, aorta, retroperitoneum, orbits, lacrimal glands, meninges, lungs, and kidneys are less frequently involved[6,7]. Isolated IgG4-SC (without concomitant AIP) represents only 15%-20% of IgG4-SC cases[1] and often closely mimics cholangiocarcinoma, resulting in frequent misdiagnosis and unnecessary surgical resections[6].
Accurate differentiation of IgG4-SC from PSC, cholangiocarcinoma, and pancreatic cancer is essential because management strategies and prognoses differ markedly. Two diagnostic frameworks are widely used: (1) The Histology, Imaging, Serology, Other organ involvement, Response to steroid therapy (HISORT) criteria, originally developed by Chari et al[8] for AIP, encompass five domains - histology, imaging, serology, other-organ involvement, and steroid responsiveness - and have been adapted for IgG4-SC[8,9]; and (2) The revised Japanese Clinical Diagnostic Criteria (2020), which update the 2012 guidelines, define six diagnostic elements: Intrahepatic and extrahepatic bile duct stenosis, bile duct wall thickening, serological abnormalities, characteristic pathology, involvement of other organs, and steroid responsiveness[1].
Elevated serum IgG4 levels are an important diagnostic clue for IgG4-SC, occurring in approximately 90% of patients with IgG4-SC[10-12]. However, mild IgG4 elevation is not specific: Up to 30% of patients with cholangiocarcinoma may also show modest increases[13]. A serum IgG4 ≥ 135 mg/dL is commonly used as a serological cutoff in diagnostic criteria, but this threshold poorly discriminates IgG4-SC from PSC. Raising the cutoff to > 4 × upper limit of normal markedly improves specificity and positive predictive value (approaching 100%) but lowers sensitivity (approximately 42%)[10,12]. Alternative markers - such as an IgG4/IgG ratio > 0.10 or an IgG4/IgG1 ratio > 0.24 - have been proposed to enhance discrimination between IgG4-SC and other causes of biliary strictures[12,14].
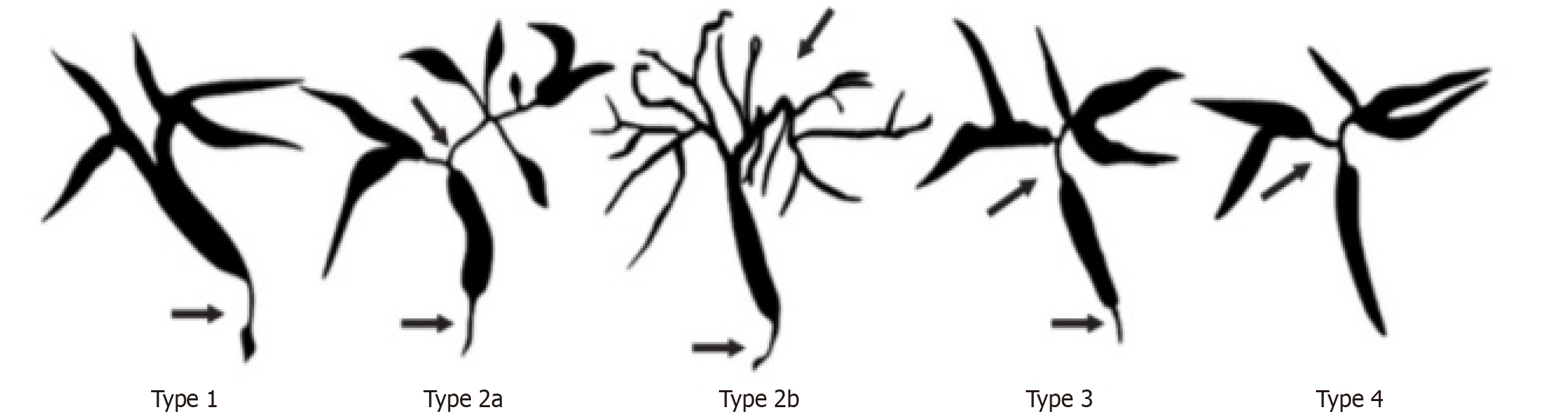
The 2020 Japanese clinical diagnostic criteria first stratify patients by the presence or absence of AIP and then apply cholangiographic classification to aid differential diagnosis. Cholangiographic subtypes are (Figure 8)[9]: (1) Type 1 - strictures confined to the intrapancreatic bile duct; (2) Type 2 - diffuse intra- and extrahepatic strictures (2a: Intrahepatic strictures with prestenotic dilation; 2b: Intrahepatic strictures without prestenotic dilation and with reduced arbo
In this case, the patient presented with obstructive jaundice. Imaging revealed hilar bile duct strictures with wall thickening, and the tumor marker carbohydrate antigen 19-9 was markedly elevated (753 IU/mL), findings that strongly suggested cholangiocarcinoma. However, a markedly increased serum IgG4 level, together with pathological exclusion of malignancy, supported a preliminary diagnosis of type 4 (isolated, hilar) IgG4-SC, based on characteristic hepatobiliary imaging and serology. The subsequent resolution of jaundice and biochemical improvements after corticosteroid therapy corroborated the diagnosis. This case illustrates a clear diagnostic paradigm for rare isolated IgG4-SC through a triad of assessment: Elevated serum IgG4, characteristic imaging findings, and a positive response to corticosteroid therapy. Furthermore, the observation of an extremely high baseline serum IgG4 level followed by rapid clinical improvement upon steroid initiation in this case suggests that IgG4 may serve as a potential biomarker for predicting responsiveness to first-line corticosteroid therapy in IgG4-SC.
The histopathological hallmark of IgG4-SC is dense lymphoplasmacytic infiltration rich in IgG4-positive plasma cells within the bile duct wall, often accompanied by storiform fibrosis and obliterative phlebitis[1,2,4]. However, IgG4-positive cell infiltration is not pathognomonic, since it can also be seen in PSC or cholangiocarcinoma. Crucially, these characteristic changes typically lie deep to the intact epithelial surface, so superficial bile duct biopsies frequently show only nonspecific chronic inflammation because they fail to sample the deeper tissue. Consequently, bile duct biopsy has limited sensitivity for diagnosing IgG4-SC and is most useful to exclude malignancy[16].
Glucocorticoids are the first-line treatment for IgG4-SC. Induction with prednisone at approximately 0.6 mg/kg/day produces a partial response in the vast majority (97%) of patients and complete resolution of biliary strictures and liver-test abnormalities in roughly two-thirds of cases by three months[3,17]. Adjuvant immunomodulators (e.g., azathioprine, methotrexate, mycophenolate mofetil, or cyclophosphamide) can be used as steroid-sparing agents to maintain remission and reduce relapse risk[18]. Rituximab (anti-CD20) is the preferred biologic for steroid-refractory disease, contraindication, or relapse[19]. Emerging data on anti-CD19 (inebilizumab) suggest efficacy in reducing flares, but safety concerns (notably infections and cytopenias) require further evaluation[20]. Typical tapering begins after an induction period (for example, decreasing prednisone by 5 mg every two weeks until reaching 20 mg/day, then by 2.5 mg every four weeks), followed by long-term low-dose maintenance (commonly approximately 5 mg/day for two to three years)[21]. Relapse during tapering occurs in a substantial proportion of patients (reported 31%-57%), and there is evidence suggesting an increased malignancy risk[12]. Japanese nationwide data indicate significantly increased malignancy risk (including lymphoma, gastric, and colorectal carcinomas) among patients with IgG4-RD. Because of the high relapse rate and potential oncologic risks, prolonged - often lifelong - clinical and oncologic surveillance is generally recommended even after sustained remission[22,23].
IgG4-SC presents a major diagnostic challenge because it can closely mimic biliary malignancy, especially when definitive deep-tissue histopathology is unavailable. This case highlights the importance of including IgG4-SC in the differential diagnosis of hilar strictures suspicious for cholangiocarcinoma and of routinely measuring serum IgG4 in such patients. When IgG4-SC is confirmed, prompt initiation of glucocorticoid therapy is essential, together with careful malignancy screening and long-term clinical and oncologic surveillance.
| 1. | Nakazawa T, Kamisawa T, Okazaki K, Kawa S, Tazuma S, Nishino T, Inoue D, Naitoh I, Watanabe T, Notohara K, Kubota K, Ohara H, Tanaka A, Takikawa H, Masamune A, Unno M. Clinical diagnostic criteria for IgG4-related sclerosing cholangitis 2020: (Revision of the clinical diagnostic criteria for IgG4-related sclerosing cholangitis 2012). J Hepatobiliary Pancreat Sci. 2021;28:235-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 2. | Tanaka A. Immunoglobulin G4-related sclerosing cholangitis. J Dig Dis. 2019;20:357-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 3. | Kersten R, Trampert DC, Herta T, Hubers LM, Maillette de Buy Wenniger LJ, Verheij J, van de Graaf SFJ, Beuers U. IgG4-related cholangitis - a mimicker of fibrosing and malignant cholangiopathies. J Hepatol. 2023;79:1502-1523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 4. | Tanaka A, Mori M, Kubota K, Naitoh I, Nakazawa T, Takikawa H, Unno M, Kamisawa T, Kawa S, Okazaki K. Epidemiological features of immunoglobulin G4-related sclerosing cholangitis in Japan. J Hepatobiliary Pancreat Sci. 2020;27:598-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 5. | Ellett TR, Pippas A, Rios H, Jabbar SB. The Deadly Masquerade: Unveiling the Fatal Facade of the Great Imitator. Cureus. 2025;17:e86819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 6. | Kamisawa T, Zen Y, Pillai S, Stone JH. IgG4-related disease. Lancet. 2015;385:1460-1471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1058] [Cited by in RCA: 890] [Article Influence: 80.9] [Reference Citation Analysis (1)] |
| 7. | Wallace ZS, Deshpande V, Mattoo H, Mahajan VS, Kulikova M, Pillai S, Stone JH. IgG4-Related Disease: Clinical and Laboratory Features in One Hundred Twenty-Five Patients. Arthritis Rheumatol. 2015;67:2466-2475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 552] [Cited by in RCA: 490] [Article Influence: 44.5] [Reference Citation Analysis (1)] |
| 8. | Chari ST, Smyrk TC, Levy MJ, Topazian MD, Takahashi N, Zhang L, Clain JE, Pearson RK, Petersen BT, Vege SS, Farnell MB. Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin Gastroenterol Hepatol. 2006;4:1010-6; quiz 934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 774] [Cited by in RCA: 650] [Article Influence: 32.5] [Reference Citation Analysis (6)] |
| 9. | Naitoh I, Nakazawa T. Classification and Diagnostic Criteria for IgG4-Related Sclerosing Cholangitis. Gut Liver. 2022;16:28-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 10. | Ohara H, Nakazawa T, Kawa S, Kamisawa T, Shimosegawa T, Uchida K, Hirano K, Nishino T, Hamano H, Kanno A, Notohara K, Hasebe O, Muraki T, Ishida E, Naitoh I, Okazaki K. Establishment of a serum IgG4 cut-off value for the differential diagnosis of IgG4-related sclerosing cholangitis: a Japanese cohort. J Gastroenterol Hepatol. 2013;28:1247-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 11. | Boonstra K, Culver EL, de Buy Wenniger LM, van Heerde MJ, van Erpecum KJ, Poen AC, van Nieuwkerk KM, Spanier BW, Witteman BJ, Tuynman HA, van Geloven N, van Buuren H, Chapman RW, Barnes E, Beuers U, Ponsioen CY. Serum immunoglobulin G4 and immunoglobulin G1 for distinguishing immunoglobulin G4-associated cholangitis from primary sclerosing cholangitis. Hepatology. 2014;59:1954-1963. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 12. | Ghazale A, Chari ST, Zhang L, Smyrk TC, Takahashi N, Levy MJ, Topazian MD, Clain JE, Pearson RK, Petersen BT, Vege SS, Lindor K, Farnell MB. Immunoglobulin G4-associated cholangitis: clinical profile and response to therapy. Gastroenterology. 2008;134:706-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 753] [Cited by in RCA: 593] [Article Influence: 32.9] [Reference Citation Analysis (4)] |
| 13. | Inoue D, Yoshida K, Yoneda N, Ozaki K, Matsubara T, Nagai K, Okumura K, Toshima F, Toyama J, Minami T, Matsui O, Gabata T, Zen Y. IgG4-related disease: dataset of 235 consecutive patients. Medicine (Baltimore). 2015;94:e680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 337] [Article Influence: 30.6] [Reference Citation Analysis (1)] |
| 14. | Nakazawa T, Ohara H, Sano H, Ando T, Joh T. Schematic classification of sclerosing cholangitis with autoimmune pancreatitis by cholangiography. Pancreas. 2006;32:229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 130] [Article Influence: 6.5] [Reference Citation Analysis (2)] |
| 15. | Furukawa M, Ishii Y, Tatsukawa Y, Nakamura S, Ikemoto J, Miyamoto S, Nakamura K, Yamashita Y, Iijima N, Okuda Y, Nomura R, Arihiro K, Hanada K, Oka S. Endoscopic Retrograde Cholangiopancreatography-Related Procedures for the Differential Diagnosis of Isolated Immunoglobin G4-Related Sclerosing Cholangitis and Perihilar Cholangiocarcinoma. Diagnostics (Basel). 2024;14:1621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 16. | Deshpande V, Zen Y, Chan JK, Yi EE, Sato Y, Yoshino T, Klöppel G, Heathcote JG, Khosroshahi A, Ferry JA, Aalberse RC, Bloch DB, Brugge WR, Bateman AC, Carruthers MN, Chari ST, Cheuk W, Cornell LD, Fernandez-Del Castillo C, Forcione DG, Hamilos DL, Kamisawa T, Kasashima S, Kawa S, Kawano M, Lauwers GY, Masaki Y, Nakanuma Y, Notohara K, Okazaki K, Ryu JK, Saeki T, Sahani DV, Smyrk TC, Stone JR, Takahira M, Webster GJ, Yamamoto M, Zamboni G, Umehara H, Stone JH. Consensus statement on the pathology of IgG4-related disease. Mod Pathol. 2012;25:1181-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2174] [Cited by in RCA: 1798] [Article Influence: 128.4] [Reference Citation Analysis (2)] |
| 17. | Löhr JM, Vujasinovic M, Rosendahl J, Stone JH, Beuers U. IgG4-related diseases of the digestive tract. Nat Rev Gastroenterol Hepatol. 2022;19:185-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (2)] |
| 18. | Omar D, Chen Y, Cong Y, Dong L. Glucocorticoids and steroid sparing medications monotherapies or in combination for IgG4-RD: a systematic review and network meta-analysis. Rheumatology (Oxford). 2020;59:718-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 19. | Carruthers MN, Topazian MD, Khosroshahi A, Witzig TE, Wallace ZS, Hart PA, Deshpande V, Smyrk TC, Chari S, Stone JH. Rituximab for IgG4-related disease: a prospective, open-label trial. Ann Rheum Dis. 2015;74:1171-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 504] [Article Influence: 45.8] [Reference Citation Analysis (2)] |
| 20. | Stone JH, Khosroshahi A, Zhang W, Della Torre E, Okazaki K, Tanaka Y, Löhr JM, Schleinitz N, Dong L, Umehara H, Lanzillotta M, Wallace ZS, Ebbo M, Webster GJ, Martinez Valle F, Nayar MK, Perugino CA, Rebours V, Dong X, Wu Y, Li Q, Rampal N, Cimbora D, Culver EL; MITIGATE Trial Investigators. Inebilizumab for Treatment of IgG4-Related Disease. N Engl J Med. 2025;392:1168-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 88] [Article Influence: 88.0] [Reference Citation Analysis (1)] |
| 21. | Kamisawa T, Nakazawa T, Tazuma S, Zen Y, Tanaka A, Ohara H, Muraki T, Inui K, Inoue D, Nishino T, Naitoh I, Itoi T, Notohara K, Kanno A, Kubota K, Hirano K, Isayama H, Shimizu K, Tsuyuguchi T, Shimosegawa T, Kawa S, Chiba T, Okazaki K, Takikawa H, Kimura W, Unno M, Yoshida M. Clinical practice guidelines for IgG4-related sclerosing cholangitis. J Hepatobiliary Pancreat Sci. 2019;26:9-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 119] [Article Influence: 17.0] [Reference Citation Analysis (2)] |
| 22. | Sumimoto K, Uchida K, Ikeura T, Hirano K, Yamamoto M, Takahashi H, Nishino T, Mizushima I, Kawano M, Kamisawa T, Saeki T, Maguchi H, Ushijima T, Shiokawa M, Seno H, Goto H, Nakamura S, Okazaki K; Research Committee for an Intractable Disease of IgG4-related disease. Nationwide epidemiological survey of immunoglobulin G4-related disease with malignancy in Japan. J Gastroenterol Hepatol. 2022;37:1022-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 23. | Orozco-Gálvez O, Fernández-Codina A, Lanzillotta M, Ebbo M, Schleinitz N, Culver EL, Rebours V, D'Cruz DP, Della-Torre E, Martínez-Valle F. Development of an algorithm for IgG4-related disease management. Autoimmun Rev. 2023;22:103273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 30] [Reference Citation Analysis (1)] |