Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.117462
Revised: January 28, 2026
Accepted: March 5, 2026
Published online: April 16, 2026
Processing time: 126 Days and 18.1 Hours
Upper gastrointestinal system (UGIS) polyps are detected infrequently during esophagogastroduodenoscopy (EGD). Data on the prevalence, characteristics, and dysplasia risk of these lesions remain limited compared with colorectal polyps.
To determine the detection rate of UGIS polyps in our center and to evaluate their clinical, endoscopic, and histopathological features.
The medical records of 2095 patients who underwent EGD between January 2022 and January 2024 were retrospectively reviewed. A total of 110 patients (5.3%) with endoscopically detected polyps who underwent polypectomy were included. Demographic, clinical, endoscopic and histopathologic features were analyzed.
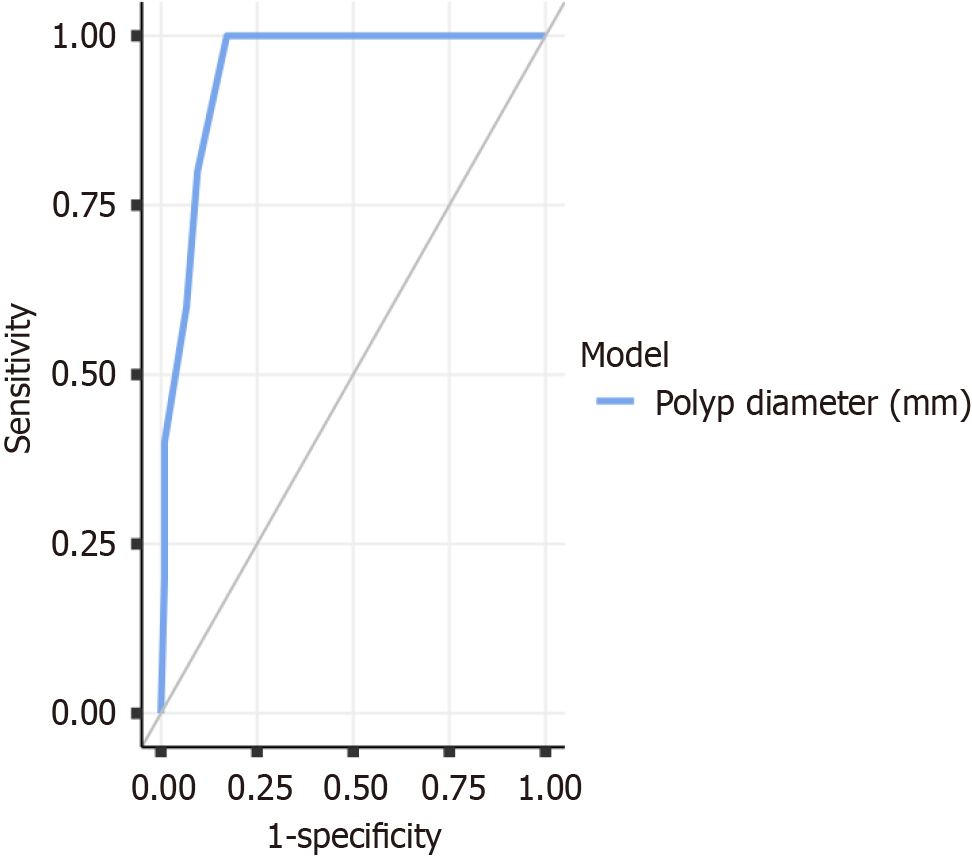
The median age was 65 years (range: 33-86), and 64.5% were female. Hyperplastic polyps were the most common type (70.9%). Dysplasia was identified in 5 patients (4.5%), all of whom had adenomatous or hyperplastic polyps. Dysplastic polyps were significantly larger than non-dysplastic polyps (median 10 mm vs 5 mm, P < 0.001) and were more frequently associated with intestinal metaplasia (P < 0.001) and Helicobacter pylori (H. pylori) infection (P = 0.014). Receiver operating characteristic analysis revealed that polyp size strongly predicted the presence of dysplasia (area under the curve: 0.947; 95%CI: 0.891-1.000; P < 0.001), with an optimal cut-off value of 7.5 mm (sensitivity 100%, specificity 82.9%).
In our cohort, UGIS polyps were detected in approximately 5% of EGDs, and most were benign. Polyp size, H. pylori infection, and intestinal metaplasia were significantly associated with dysplasia. Given the malignant potential of adenomatous polyps and the size-dependent risk of dysplasia, complete endoscopic removal of incidentally detected UGIS polyps is advisable.
Core Tip: Upper gastrointestinal polyps were detected in 5.3% of esophagogastroduodenoscopy, and most were benign. Helicobacter pylori infection, polyp diameter ≥ 7.5 mm, adenomatous histology, and intestinal metaplasia increase the risk of dysplasia. Fundic gland polyps are generally benign, with no dysplasia observed, supporting current literature on their low malignant potential.
- Citation: Yüksel E, Dinçer B. Polypectomy rate and characteristics of polyps in upper gastrointestinal system endoscopy. World J Gastrointest Endosc 2026; 18(4): 117462
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/117462.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.117462
Gastric cancer ranks as the fifth most common malignancy and the fifth leading cause of cancer-related mortality, and despite its declining incidence, it remains an important public health concern[1]. Although patients diagnosed at an early stage have a more favorable prognosis, gastric cancer is often detected at an advanced stage because symptoms usually appear late[2]. Therefore, particularly in East Asian countries where the incidence is higher, endoscopic screening programs have been implemented to increase the rate of early diagnosis[3,4]. Esophagogastroduodenoscopy (EGD) is a commonly used endoscopic method for the screening and diagnosis of gastric cancer[5,6]. Unlike colorectal cancers, gastric cancer rarely arises from polyps, and the detection rate of polyps during EGD is substantially lower than that observed during colonoscopy. Although polyps of the upper gastrointestinal tract are reported to have a lower malignant potential compared with colorectal polyps, the literature on this topic is more limited than that on colorectal polyps[7]. In this study, we aimed to investigate the polypectomy rate during upper gastrointestinal system endoscopy in our center and to evaluate the histopathological characteristics of the identified polyps.
The relevant medical records of 2095 patients who underwent gastroscopy in the surgical endoscopy room from January 1, 2022 to January 1, 2024 were reviewed. Of these, 110 patients (5.3%) in whom a polyp was detected in the upper gastrointestinal tract were included in the final analysis. This study was conducted in accordance with the Declaration of Helsinki and approved by the Non-Interventional Clinical Research Ethics Committee of Ankara Oncology Training and Research Hospital (No. 2025-11/173). Written informed consent was obtained from study participants.
Patients aged > 18 years who underwent EGD due to upper gastrointestinal symptoms or findings and who were found to have a polyp in the upper gastrointestinal tract and underwent polypectomy were included. Exclusion criteria were the absence of polyps on EGD, failure to perform polypectomy, incomplete pathology reports, the presence of upper gastrointestinal malignancy except for incidentally detected lesions during polypectomy, and a history of gastric re
Statistical analyses were carried out using SPSS software (version 22; IBM Corp., Armonk, NY, United States). The dataset was summarized with descriptive metrics such as n (%), and the median with its interquartile range (IQR). Categorical variables were evaluated with Fisher’s exact test, whereas continuous variables were compared using the Mann-Whitney U test due to non-normal distribution. To determine how well polyp diameter discriminated between cases with and without dysplasia, a receiver operating characteristic (ROC) curve was constructed. The area under the curve (AUC) along with the corresponding 95%CI was reported as a measure of diagnostic accuracy. The threshold that best differentiated dysplastic from non-dysplastic lesions was identified using Youden’s index. All statistical tests were two-tailed, and a P-value below 0.05 was considered indicative of statistical significance.
The median age of the 110 patients included in the study was 65 years (range: 33-86), and 71 (64.5%) were female. EGD was performed due to dyspepsia in 49 patients (44.5%) and abdominal pain in 26 patients (23.6%). Of the detected polyps, 38 (34.5%) were located in the gastric corpus, 25 (22.7%) in the gastric fundus, and 24 (21.8%) in the gastric antrum. H. pylori infection was identified in 14 patients (12.7%). Dysplasia was detected in the pathological examination of 5 patients (4.5%). While H. pylori infection was significantly more common in patients with dysplasia (P = 0.014), no significant differences were observed between the groups in terms of other parameters (Table 1).
| Variables | All patients (n = 110) | Dysplasia (-) (n = 105) | Dysplasia (+) (n = 5) | P value |
| Age (years) | 65 (33-86) | 65 (33-86) | 70 (53-81) | 0.5801 |
| Sex | ||||
| Female | 71 (64.5) | 69 (65.7) | 2 (40) | 0.3442 |
| Male | 39 (35.5) | 36 (34.3) | 3 (60) | |
| Indication | ||||
| Dyspepsia | 49 (44.5) | 48 (45.7) | 1 (20) | 0.1072 |
| Abdominal pain | 26 (23.6) | 26 (24.8) | 1 (20) | |
| Reflux symptoms | 13 (11.8) | 11 (10.5) | 1 (20) | |
| Family history of gastric cancer | 10 (9.1) | 10 (9.5) | 0 (0) | |
| Anemia | 9 (8.2) | 8 (7.6) | 1 (20) | |
| Radiologic suspicious findings | 3 (2.7) | 2 (1.9) | 1 (20) | |
| Polyp localisation | ||||
| Esophagus | 2 (1.8) | 2 (1.9) | 0 (0) | 0.5402 |
| Cardia | 17 (15.5) | 17 (16.2) | 0 (0) | |
| Fundus | 25 (22.7) | 25 (23.8) | 0 (0) | |
| Corpus | 38 (34.5) | 35 (33.3) | 3 (60) | |
| Antrum | 24 (21.8) | 22 (21) | 2 (40) | |
| Bulbus | 3 (2.7) | 3 (2.9) | 0 (0) | |
| Second part of the duodenum | 1 (0.9) | 1 (1) | 0 (0) | |
| Helicobacter pylori infection | ||||
| No | 96 (87.3) | 94 (89.5) | 2 (40) | 0.0142 |
| Yes | 14 (12.7) | 11 (10.5) | 3 (60) | |
Review of the pathology reports of the polypectomy specimens showed that the median polyp diameter was 5 mm (IQR: 4-7). Among the 110 polyps, 78 (70.9%) were hyperplastic, 19 (17.3%) were fundic gland polyps, 12 (10.9%) were adenomatous polyps, and 1 (0.9%) was an inflammatory polyp. Intestinal metaplasia was present in 17 patients (15.5%). Patients with dysplasia had significantly larger polyp diameters (P < 0.001) and a higher frequency of intestinal meta
| Variables | All patients (n = 110) | Dysplasia (-) (n = 105) | Dysplasia (+) (n = 5) | P value |
| Polyp diameter (mm) | 5 (4-7) | 5 (4-6) | 10 (9-12) | < 0.0011 |
| Pathological diagnosis | ||||
| Hyperplastic | 78 (70.9) | 77 (73.3) | 1 (20) | 0.0012 |
| Fundic gland | 19 (17.3) | 19 (18.1) | 0 (0) | |
| Adenomatous | 12 (10.9) | 8 (7.6) | 4 (80) | |
| Inflammatory | 1 (0.9) | 1 (1) | 0 (0) | |
| Intestinal metaplasia | ||||
| No | 93 (84.5) | 93 (88.6) | 0 (0) | < 0.0012 |
| Yes | 17 (15.5) | 12 (11.4) | 5 (100) |
When the relationship between H. pylori infection status and polyp types was evaluated, the frequency of H. pylori infection was found to be significantly higher in patients with hyperplastic and adenomatous polyps (Table 3).
| Variables | All patients (n = 110) | Helicobacter pylori (-) (n = 96) | Helicobacter pylori (+) (n = 14) | P value |
| Pathological diagnosis | ||||
| Hyperplastic | 78 (70.9) | 71 (74) | 7 (50) | 0.0031 |
| Fundic gland | 19 (17.3) | 18 (18.8) | 1 (7.1) | |
| Adenomatous | 12 (10.9) | 6 (6.3) | 6 (42.9) | |
| Inflammatory | 1 (0.9) | 1 (1) | 0 (0) |
ROC curve analysis showed that polyp size was associated with the presence of dysplasia, with an AUC of 0.947 (95%CI: 0.891-1.000; P < 0.001). The optimal cut-off value determined by Youden’s index was 7.5 mm. Using this threshold, a polyp diameter ≥ 7.5 mm identified dysplasia with a sensitivity of 100.0% and a specificity of 82.7%. The positive predictive value was 21.7%, and the negative predictive value was 100.0%. Overall diagnostic accuracy was calculated as 83.6% (Figure 1).
In our study, polypectomy rate is only 5.3% of EGDs performed in our center. None of these patients had malignancy arising from a polyp, and dysplasia was identified in only 5 patients. In the literature, H. pylori infection and the resulting intestinal metaplasia and hypergastrinemia are defined as risk factors for gastric cancer[8-10]. However, gastric cancer arising from polyps is reported to be much less common compared with intestinal metaplasia and dysplasia and may also be associated with hereditary syndromes[7,11]. Consistent with this, polyps were detected in only 110 of the 2095 patients who underwent EGD in our study, and dysplasia was found in only 5 polyps. Our findings are consistent with the existing literature.
Hyperplastic polyps are the most common gastric polyps, and studies have reported that they are associated with H. pylori infection and regress following H. pylori eradication. The inflammation induced by H. pylori infection, along with subsequent metaplasia and dysplasia, is thought to contribute to the development of hyperplastic polyps[12]. In a study by Cho et al[13], 201 patients with H. pylori infection and a history of endoscopic polypectomy for hyperplastic polyps were evaluated, and recurrence of hyperplastic polyps was significantly lower in patients who achieved H. pylori eradication. Similarly, a randomized controlled trial by Nam et al[14] demonstrated that H. pylori eradication leads to regression of hyperplastic polyps. In our study, hyperplastic polyps were also most commonly observed in patients with H. pylori infection, and our findings are in line with the current literature.
Fundic gland polyps are one of the commonly observed types of gastric polyps, with an increasing incidence particularly associated with the rising use of proton pump inhibitors, and are considered benign in cases not associated with hereditary syndromes such as familial adenomatous polyposis[15]. Although dysplasia has occasionally been reported in sporadic fundic gland polyps, the literature indicates that their risk of malignant transformation is low[15,16]. In our study, the absence of dysplasia in fundic gland polyps supports the existing literature.
Regarding gastric polyp types, the literature shows that hyperplastic polyps are the most common, followed by fundic gland polyps and adenomatous polyps. Hyperplastic polyps larger than 10 mm are associated with an increased risk of neoplasia. Dysplasia is very rarely observed in sporadic fundic gland polyps, whereas fundic gland polyps in patients with hereditary polyposis syndromes carry a higher risk of dysplasia. Adenomatous polyps account for approximately 3%-26% of all gastric polyps and are reported to be associated with intestinal metaplasia and H. pylori infection; they carry neoplastic potential[17].
The literature indicates that the malignant potential of upper gastrointestinal polyps increases with polyp diameter[18]. However, a definitive polyp diameter cut-off associated with a markedly increased malignancy risk has not been clearly defined. In our study, ROC analysis evaluating the relationship between polyp size and dysplasia risk showed that a diameter of ≥ 7.5 mm was associated with an increased risk of dysplasia.
The retrospective and single-center design of the study and the limited sample size are among its main limitations. Additionally, the absence of malignancy in our series is likely due to the small number of cases, preventing evaluation of the characteristics of malignancies arising from polyps. Furthermore, the assessment of the relationship between polyp size and malignancy risk cannot be generalized given the limited number of large polyps (> 10 mm). Multicenter pro
In our study, the rate of polypectomy during EGD was approximately 5%, similar to the literature, and these polyps were generally benign. However, H. pylori infection, a polyp diameter > 7.5 mm, adenomatous histology, and the presence of intestinal metaplasia were found to increase the risk of dysplasia.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14984] [Article Influence: 7492.0] [Reference Citation Analysis (22)] |
| 2. | Dinçer B, Sağdıç MF, Olgun M, Karayiğit A, Dilek GB, Sarıdemir S, Özaslan C. Subtotal versus total gastrectomy for mid- and distal gastric poorly cohesive carcinoma: a retrospective controlled study. Front Oncol. 2025;15:1717678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Tsubono Y, Hisamichi S. Screening for gastric cancer in Japan. Gastric Cancer. 2000;3:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 90] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | Choi KS, Jun JK, Suh M, Park B, Noh DK, Song SH, Jung KW, Lee HY, Choi IJ, Park EC. Effect of endoscopy screening on stage at gastric cancer diagnosis: results of the National Cancer Screening Programme in Korea. Br J Cancer. 2015;112:608-612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 130] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 5. | Park CH, Kim B, Chung H, Lee H, Park JC, Shin SK, Lee SK, Lee YC. Endoscopic quality indicators for esophagogastroduodenoscopy in gastric cancer screening. Dig Dis Sci. 2015;60:38-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 6. | Yoshimizu S, Yamamoto Y, Horiuchi Y, Omae M, Yoshio T, Ishiyama A, Hirasawa T, Tsuchida T, Fujisaki J. Diagnostic performance of routine esophagogastroduodenoscopy using magnifying endoscope with narrow-band imaging for gastric cancer. Dig Endosc. 2018;30:71-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 7. | Gullo I, Grillo F, Mastracci L, Vanoli A, Carneiro F, Saragoni L, Limarzi F, Ferro J, Parente P, Fassan M. Precancerous lesions of the stomach, gastric cancer and hereditary gastric cancer syndromes. Pathologica. 2020;112:166-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 118] [Article Influence: 19.7] [Reference Citation Analysis (15)] |
| 8. | Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3583] [Cited by in RCA: 3206] [Article Influence: 128.2] [Reference Citation Analysis (4)] |
| 9. | Duan Y, Xu Y, Dou Y, Xu D. Helicobacter pylori and gastric cancer: mechanisms and new perspectives. J Hematol Oncol. 2025;18:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 151] [Article Influence: 151.0] [Reference Citation Analysis (2)] |
| 10. | Du S, Yang Y, Fang S, Guo S, Xu C, Zhang P, Wei W. Gastric Cancer Risk of Intestinal Metaplasia Subtypes: A Systematic Review and Meta-Analysis of Cohort Studies. Clin Transl Gastroenterol. 2021;12:e00402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 11. | Christenson RV, Sood S, Vierkant RA, Schupack D, Boardman L, Grotz TE. Gastric polyposis and risk of gastric cancer in patients with familial adenomatous polyposis. J Gastrointest Surg. 2024;28:1890-1896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 12. | Kővári B, Kim BH, Lauwers GY. The pathology of gastric and duodenal polyps: current concepts. Histopathology. 2021;78:106-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (2)] |
| 13. | Cho YS, Nam SY, Moon HS, Kim TH, Kim SE, Jung JT. Helicobacter pylori eradication reduces risk for recurrence of gastric hyperplastic polyp after endoscopic resection. Korean J Intern Med. 2023;38:167-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (2)] |
| 14. | Nam SY, Lee SW, Jeon SW, Kwon YH, Lee HS. Helicobacter pylori Eradication Regressed Gastric Hyperplastic Polyp: A Randomized Controlled Trial. Dig Dis Sci. 2020;65:3652-3659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 15. | Sano W, Inoue F, Hirata D, Iwatate M, Hattori S, Fujita M, Sano Y. Sporadic fundic gland polyps with dysplasia or carcinoma: Clinical and endoscopic characteristics. World J Gastrointest Oncol. 2021;13:662-672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (6)] |
| 16. | Levy MD, Bhattacharya B. Sporadic Fundic Gland Polyps With Low-Grade Dysplasia: A Large Case Series Evaluating Pathologic and Immunohistochemical Findings and Clinical Behavior. Am J Clin Pathol. 2015;144:592-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 17. | Castro R, Pimentel-Nunes P, Dinis-Ribeiro M. Evaluation and management of gastric epithelial polyps. Best Pract Res Clin Gastroenterol. 2017;31:381-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 18. | Reyes-Placencia D, Cantú-Germano E, Latorre G, Espino A, Fernández-Esparrach G, Moreira L. Gastric Epithelial Polyps: Current Diagnosis, Management, and Endoscopic Frontiers. Cancers (Basel). 2024;16:3771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |