Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.118038
Revised: January 8, 2026
Accepted: February 5, 2026
Published online: April 27, 2026
Processing time: 120 Days and 15.3 Hours
Portal hypertension (PHT) in children is associated with a high risk of esophag
To systematically evaluate and meta-analyse diagnostic accuracy of blood-based non-invasive tests for predicting EV and CSV in pediatric PHT.
We conducted a systematic review and diagnostic accuracy meta-analysis following Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Test Accuracy Studies guidelines. PubMed, EMBASE, and Scopus were searched from inception to October 2025. Studies evaluating aminotransferase-to-platelet ratio index (APRI), platelet count, clinical prediction rule (CPR), varices prediction rule (VPR), or King’s variceal prediction score (K-VaPS) against UGIE in children (≤ 18 years) with PHT were included. Hierarchical summary receiver operating characteristic models were used to pool sensitivity, specificity, diagnostic odds ratios, and area under the curve (AUC). Subgroup, sensitivity, and publication bias analyses were performed.
Twenty-four studies were included [APRI-17 studies (n = 1355), platelet count-11 studies (n = 1040), CPR-12 studies (n = 944), VPR-7 studies (n = 655), K-VaPS-5 studies (n = 315)]. APRI demonstrated pooled sensitivities and specificities of 0.77 and 0.71 for EV and 0.72 and 0.61 for CSV, with corresponding AUCs of 0.795 and 0.776, respectively. Platelet count showed pooled sensitivities and specificities of 0.69 and 0.77 for EV and 0.66 and 0.72 for CSV, with AUCs of 0.834 and 0.714, respectively. Among prediction scores, VPR showed highest accuracy for CSV prediction (AUC = 0.864), followed by K-VaPS (0.727) and CPR (0.717). Substantial heterogeneity was observed across most of the analyses. Diagnostic accuracy was consistently higher in biliary atresia–exclusive cohorts and Asian populations. The certainty of evidence ranged from moderate to very low.
Blood-based non-invasive tests demonstrate moderate accuracy for predicting varices in pediatric PHT and may serve as adjunctive triage tools; however, standardized pediatric thresholds and prospective validation are urgently needed.
Core Tip: Blood-based non-invasive tests offer a pragmatic approach for risk stratification of varices in children with portal hypertension. This meta-analysis reveals that commonly used indices, such as aminotransferase-to-platelet ratio index, platelet count, demonstrate moderate diagnostic accuracy and may help identify children at lower risk who could safely defer immediate endoscopic surveillance. Their advantages include wide availability, low cost, and ease of repeat testing, making them particularly relevant in resource-limited settings. However, substantial heterogeneity, variability in applied cut-off thresholds, and low-to-moderate certainty of evidence limit their reliability as standalone tools. Prospective pediatric studies with standardised and aetiology-specific cut-offs are essential before implementing routine guidelines.
- Citation: Panda K, Dash DP, Pati GK, Maria A, Gautam V. Diagnostic accuracy of blood-based non-invasive tests for prediction of varices in pediatric portal-hypertension: A systematic review and meta-analysis. World J Hepatol 2026; 18(4): 118038
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/118038.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.118038
Portal hypertension (PHT) is the central pathophysiological consequence of a heterogeneous spectrum of chronic liver diseases and portal venous system disorders arising from increased intrahepatic vascular resistance and altered splanchnic hemodynamics[1,2]. One of the most serious complications of PHT is the development of esophageal/gastric varices (EV), which can lead to life-threatening upper gastrointestinal bleeding, especially in patients harbouring high-risk or clinically significant varices (CSV)[3,4]. Upper gastrointestinal endoscopy (UGIE) is the established reference standard for detecting and stratifying EV/CSV; however, being an invasive procedure, it often requires general anaesthesia or deep sedation in children and carries procedural risks[5-8]. The need for repeated surveillance in a chronic disease context also poses a significant burden on families and healthcare systems, particularly in resource-constrained settings. Furthermore, only a subset of children with PHT harbour CSV that warrants treatment with endoscopic band ligation/beta-blocker prophylaxis. Accurate identification of this high-risk subgroup is therefore critical to enable an effective triage strategy, minimising unnecessary endoscopic procedures while ensuring timely prophylaxis in those most likely to benefit[9,10]. This highlights the clinical imperative for reliable non-invasive alternatives capable of identifying and stratifying variceal risk.
Although non-invasive imaging modalities, such as liver stiffness measurement and splenic stiffness measurement, have shown promise in predicting EV/CSV, their widespread use in pediatric practice is limited by higher costs, restricted availability, the need for pediatric-specific probes, and dependence on technical expertise. In contrast, blood-based non-invasive tests are inexpensive, universally available, and easily repeatable, making them particularly well-suited for large-scale risk stratification and triage in children with PHT.
Over the past two decades, several blood-based markers and composite scores-such as platelet count, platelet count to spleen diameter ratio, aspartate aminotransferase-to-platelet ratio index (APRI), fibrosis-4 index (FIB-4), etc. have been identified, and their variceal predictive ability has been rigorously investigated. Multiple meta-analyses in the adult population have already evaluated the diagnostic performance of these indices with variable results[11-13]. However, children differ fundamentally from adults in terms of disease etiology, the pathophysiology of PHT, and haematological reference ranges, which limits the applicability of adult-derived evidence to pediatric practice. To date, only one meta-analysis has evaluated the diagnostic accuracy of platelet count for variceal prediction in pediatric PHT; however, it included only four studies available at the time[12]. Moreover, several composite scores, such as the clinical prediction rule (CPR), varices prediction rule (VPR), and King’s variceal prediction score (K-VaPS), which combine blood tests such as platelet count, serum albumin, with splenic z score assessed through ultrasonography, have been devised and used exclusively in pediatric cohorts[14].
The pediatric literature evaluating these blood-based non-invasive predictors remains fragmented and inconsistent, with varying diagnostic accuracy and cut-off values. Consequently, there is currently no consensus regarding the clinical utility of these tests for guiding endoscopic surveillance in children with PHT[14]. The present study aims to address this critical knowledge gap by systematically evaluating and quantitatively summarizing the diagnostic performance of these blood-based non-invasive tests for variceal prediction in pediatric PHT. By synthesising available evidence and exploring sources of heterogeneity, this study aims to inform evidence-based triage strategies, optimise surveillance practices, and identify priorities for future research in pediatric hepatology.
This study was designed and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Test Accuracy Studies (PRISMA-DTA) guidelines[15]. The review protocol was developed a priori and registered with the International Prospective Register of Systematic Reviews, registration No. CRD42025
A comprehensive literature search was conducted across the electronic databases PubMed/MEDLINE, EMBASE, and Scopus, from their inception through October 2025, with an English language filter applied. The search strategy combined controlled vocabulary (MeSH/Emtree terms) and free-text keywords related to PHT, EV, children, and blood-based non-invasive tests. Search terms for PHT included “portal hypertension”, “chronic liver disease”, and “cirrhosis”. Terms related to the target condition comprised “esophageal varices”, “oesophageal varices”, “gastric varices”, and “gastroesophageal varices”. Pediatric populations were identified using terms such as “child”, “infant”, “adolescent”, “pediatric”, and “paediatric”. Blood-based non-invasive tests were captured using keywords and phrases including “platelet count”, “thrombocytopenia”, “platelet count to spleen size”, “platelet count to spleen diameter ratio”, “platelet count to spleen z-score”, “aspartate aminotransferase-to-platelet ratio index”, and “Fibrosis-4 index”. In addition, pediatric-specific blood-based composite indices such as the “clinical prediction rule”, “varices prediction rule”, and “King’s variceal prediction score”, were explicitly included.
A preliminary literature review indicated heterogeneity in the assessment of splenic size for the composite index “platelet count-to-spleen size ratio”, with studies variably using splenic diameter, spleen size z-score, or estimated splenic size. Additionally, only a single pediatric study has evaluated the accuracy of FIB-4[16]. Due to this heterogeneity and limited data availability, the present analysis was restricted to evaluating the pooled accuracy of APRI, platelet count and pediatric-specific prediction scores, including the CPR, VPR, and K-VaPS for variceal prediction.
Inclusion criteria: Prospective or retrospective cross-sectional and cohort studies were included if they met the following predefined criteria based on the Population/Intervention/Comparison/Outcome framework.
Population (P): Studies involving pediatric patients (≤ 18 years) with PHT of any etiology, including both cirrhotic causes like biliary atresia, metabolic liver disease, Wilson disease, autoimmune hepatitis, etc. and non-cirrhotic causes, such as extrahepatic portal vein obstruction.
Index tests (I): Studies evaluating the diagnostic accuracy of one or more of the following five blood-based non-invasive tests/composite indices (APRI, platelet count, CPR, VPR, K-VaPS) for the prediction of EV/CSV.
Reference standard (C): Studies using UGIE as the reference standard for EV/CSV detection. We accepted the definition of EV/CSV as used in the respective studies for this meta-analysis.
Outcome (O): Studies having a 2 × 2 contingency table or providing adequate data on diagnostic accuracy to calculate true positive (TP), false negative (FN), false positive (FP), and true negative (TN).
Exclusion criteria: Studies were excluded if they involved adult or mixed (adult + pediatric) populations or lacked sufficient diagnostic accuracy data to construct 2 × 2 contingency tables. Additional exclusions included non-English language studies, case reports, case series with < 10 patients, review articles, editorials, conference abstracts without full-text data, and animal studies.
After removing duplicates, two reviewers (Dash DP and Pati GK) independently scrutinized the titles and abstracts of all retrieved records for potential eligibility. Full-text articles of potentially relevant studies were then assessed against the predefined inclusion and exclusion criteria to determine their eligibility for inclusion in the meta-analysis. The reference lists of included studies and relevant review articles were manually screened to identify additional eligible studies. Any disagreements were resolved through discussion or, when necessary, by consultation with a third reviewer (Panda K).
Data were extracted independently by two reviewers (Maria A and Gautam V) using a standardized data extraction proforma. Extracted variables included study characteristics (author, year, country, study design and duration, sample size), participant characteristics (age, gender, etiology of PHT, prevalence of EV/CSV), details of the index test(s) (name of the test/composite index, cut-off values, whether cut-offs were pre-defined, duration between index test and UGIE, blinding of the investigator to reference standard results), reference standard details (UGIE findings regarding presence or absence of EV/CSV, blinding of endoscopist to index test results), outcome data [2 × 2 contingency tables (TP, FN, FP, TN) for each index test]. When a study reported diagnostic accuracy for an index test at multiple cut-off values, we extracted accuracy estimates corresponding to the cut-off most commonly used across the included studies to ensure consistency in pooled analyses.
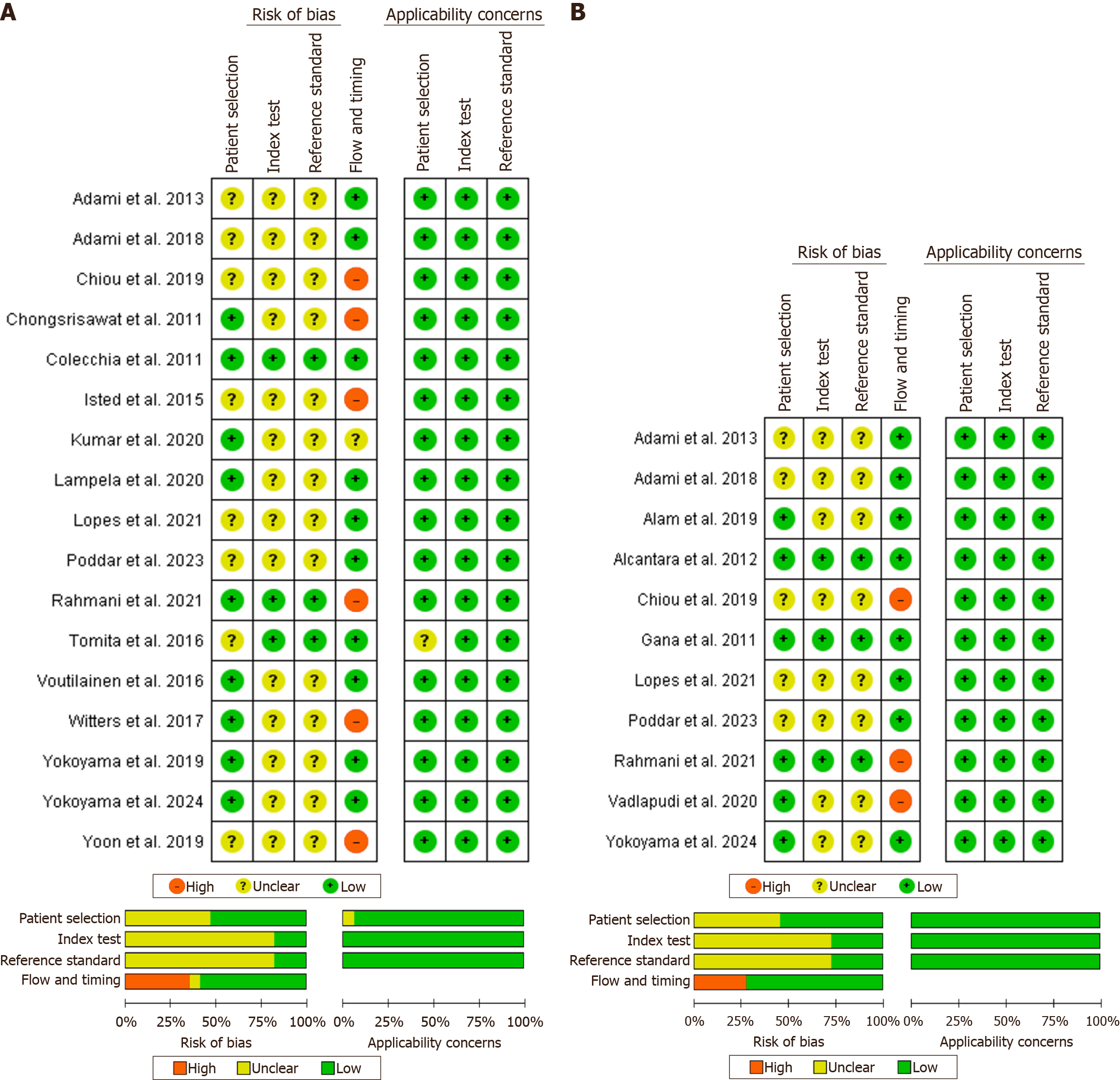
The methodological quality of the included studies was assessed independently by two reviewers (Panda K and Pati GK) using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool[17]. Risk of bias was evaluated across four domains: Patient selection, index test, reference standard, and flow and timing. Applicability concerns were also assessed for the first three domains. Any discrepancies were resolved by consensus.
The primary outcome of this meta-analysis is to determine the diagnostic accuracy of five blood-based non-invasive tests (APRI, platelet count, CPR, VPR, K-VaPS) for the prediction of EV and CSV in pediatric patients with PHT, using UGIE as the reference standard.
In view of the variability in cut-off thresholds across studies for blood-based non-invasive tests, a hierarchical summary receiver operating characteristic (HSROC) model was used to obtain pooled estimates of sensitivity, specificity, positive and negative likelihood ratios, and diagnostic odds ratios (DOR), along with their 95%CI. Pooled estimates were generated only when three or more studies were available for a given index test for a particular target condition (EV/CSV). HSROC curves were constructed with corresponding 95% confidence and prediction regions, and the area under the HSROC curve (AUC) was calculated to summarise overall diagnostic performance. An AUC > 0.9 was considered to indicate high diagnostic accuracy, values between 0.7 and 0.9 were suggestive of moderate accuracy, whereas values between 0.5 and 0.7 were indicative of low accuracy.
Heterogeneity was assessed qualitatively via visual inspection of forest plots and quantitatively using Cochrane Q test and the Higgins I2 statistic, with P < 0.05 or I2 > 60% indicating substantial heterogeneity. To explore potential sources of heterogeneity, subgroup analyses were performed using relevant study-level covariates, where data permitted. In addition, the clinical applicability of blood-based non-invasive tests for predicting EV/CSV was evaluated using Fagan nomograms, which integrate likelihood ratios with pre-test probabilities to estimate positive/negative post-test probabilities. Sensitivity analyses were performed by excluding studies at high risk of bias. Publication bias was assessed using Deeks’ funnel plot asymmetry test, with a P < 0.10 considered significant[18]. The overall certainty of evidence for each index test was assessed using a Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach adapted for diagnostic test accuracy studies, considering risk of bias, inconsistency, indirectness, imprecision, and publication bias[19,20]. All statistical analyses were conducted using MetaDTA version 1.5.2[21-23], Meta-Disc version 1.4, and Review Manager version 5.4.
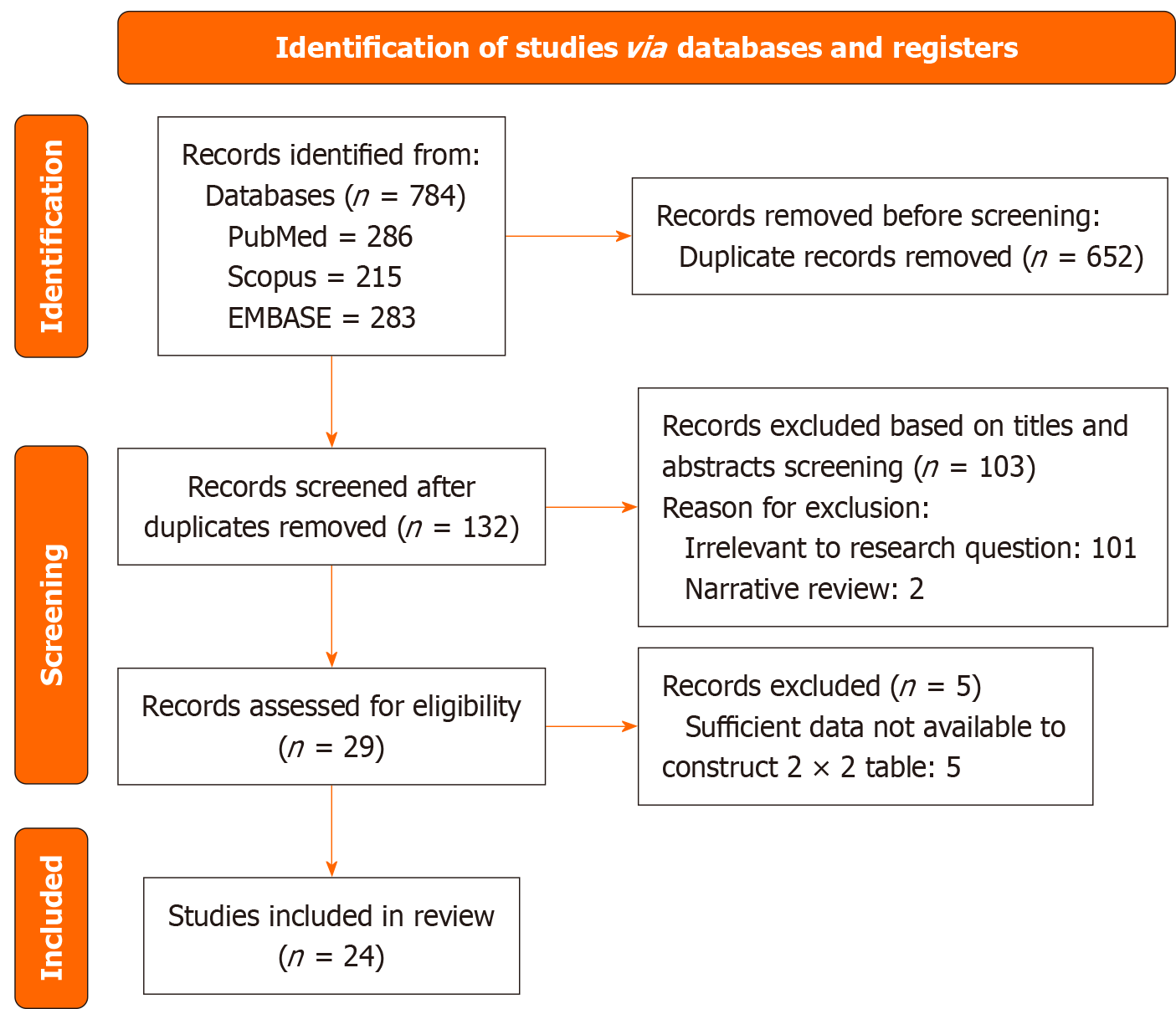
The systematic database search yielded 784 records (PubMed: 286; Scopus: 215; EMBASE: 283). After removing duplicates, 132 records were screened based on titles and abstracts. Of these, 103 studies were excluded for reasons including lack of relevance to the study question (n = 101) and narrative reviews (n = 2). Full-text assessment was performed for 29 articles, of which five were excluded owing to insufficient data to construct diagnostic 2 × 2 contingency tables[24-28]. Ultimately, 24 studies fulfilled the inclusion criteria and were included in the meta-analysis[16,29-52]. The study selection process is depicted in the PRISMA flow diagram (Figure 1).
Table 1 summarises the demographic characteristics and diagnostic accuracy parameters of the included studies separately for APRI and platelet count, and Table 2 summarises those for CPR, VPR, and K-VaPS.
| Index test | Ref. | Study design/period | Sample size/gender | Age | Etiology of portal hypertension | Prevalence of varices | Interval between index test and reference standard | Any grade oesophageal/gastric varices | Clinically significant varices | ||||||||||
| Cut-off value | TP | FP | FN | TN | AUROC | Cut-off value | TP | FP | FN | TN | AUROC | ||||||||
| APRI | Colecchia et al[29], Italy, 2011 | Prospective single-center (1990-2009) | 31. Male = 13 (43) | 12 ± 15.5 years | Cirrhotic. Biliary atresia-post KPE | 48.3% | 1 day | > 0.96 | 13 | 3 | 2 | 13 | 0.88 | ||||||
| Chongsrisawat et al[30], Thailand, 2011 | Prospective single-center | 73. Gender = NA | 9.11 ± 5.64 years | Cirrhotic. Biliary atresia-post KPE | 53.4% | NA | > 1.92 | 33 | 6 | 6 | 28 | 0.87 | |||||||
| Adami et al[34], Brazil, 2013 | Retrospective single-center (2000-2011) | 103. Male = 47 (45.7) | 8.9 ± 4.7 years | Cirrhotic + non-cirrhotic | 68.9% | Up to 3 months | > 1.4 | 45 | 11 | 26 | 21 | ||||||||
| Isted et al[35], United Kingdom, 2015 | Retrospective single-center (2004-2012) | 195. Male = 83 (42.5) | 55 (11-216) days | Cirrhotic. Biliary atresia post-KPE | 21.5% | NA | ≥ 1.64 | 29 | 57 | 13 | 96 | 0.72 | |||||||
| Voutilainen et al[36], Finland, 2016 | Prospective single-center (2012-2015) | 64. Male = 34 (53.1) | 5.2 (2.1-13.9) years | Cirrhotic. Biliary atresia-post KPE (34.3%). IFALD (42.1%). Cholestatic liver disease (9.3%). Miscellaneous (14.3%) | 21.8% | 1 day | > 0.6 | 14 | 17 | 0 | 33 | 0.832 | |||||||
| Tomita et al[37], Japan, 2016 | Prospective single-center (2011-2014) | 16. Gender = NA | 6.3 (0.7-33.5) years | Cirrhotic. Biliary atresia-post KPE | 18.7% | Up to 3 months | > 4.87 | 2 | 0 | 1 | 13 | 0.72 | |||||||
| Witters et al[38], United Kingdom, 2017 | Prospective single-center (2005-2014) | 124. Male = 66 (53.2) | 8.81 ± 5.60 years | Cirrhotic + non-cirrhotic | 63.7% | Up to 6 months | > 1.603 | 48 | 20 | 31 | 25 | 0.61 | |||||||
| Adami et al[39], Brazil, 2018 | Retrospective single-center (2000-2011) | 98. Male = 46 (46.9) | 9.1 ± 4.7 years | Cirrhotic. Autoimmune hepatitis (21.4%). Biliary atresia (26.5%). Sclerosing cholangitis (6.1%). Cystic fibrosis (6.1%). Idiopathic biliary cirrhosis (12.2%). Miscellaneous (27.5%) | 32.6% | Up to 3 months | > 1.4 | 18 | 35 | 14 | 31 | ||||||||
| Yoon et al[41], South Korea, 2019 | Retrospective single-center (2015-2016) | 22. Male = 10 (45.4) | 10 (4-18) years | Cirrhotic. Biliary atresia-post KPE | 27.2% | Up to 6 months | > 0.65 | 6 | 4 | 0 | 12 | 0.844 | |||||||
| Chiou et al[42], Singapore, 2019 | Retrospective single-center (1989-2016) | 72. Male = 35 (48.6) | NA | Cirrhotic. Biliary atresia-post KPE | 22.2% | 6 months | ≥ 3 | 13 | 13 | 3 | 43 | 0.828 | |||||||
| Yokoyama et al[43], Japan, 2019 | Prospective single-center (2018-2019) | 34. Male = 11 (32.2) | 5.2 ± 5.92 years | Cirrhotic. Biliary atresia-post KPE | 41.1% | 1-2 days | ≥ 1.63 | 10 | 3 | 4 | 17 | 0.750 | |||||||
| Lampela et al[44], Finland, 2020 | Prospective single-center (1989-2017) | 45. Gender = NA | 1.6 (0.7-14) years | Cirrhotic. Biliary atresia-post KPE | 46.6% | None. Both done on same day | ≥ 1.01 | 19 | 12 | 2 | 12 | 0.64 | |||||||
| Kumar et al[46], India, 2020 | Prospective single-center (2018-2019) | 59. Gender = NA | NA | Cirrhotic. Wilson disease (25%). Autoimmune hepatitis (16%). Biliary atresia (11%). Cholestatic liver disease (8%). Miscellaneous (40%) | 54.2% | Up to 1 week | ≥ 1.5 | 26 | 7 | 6 | 20 | 0.81 | |||||||
| Lopes et al[48], Brazil, 2021 | Retrospective single-center (2004-2018) | 158. Male = 70 (44.3) | 5.38 (2.08-11.52) years | Cirrhotic. Biliary atresia (35.4%). Autoimmune hepatitis (27.8%). Sclerosing cholangitis (14.5%). Alpha1-antitrypsin deficiency (8.2%). Cryptogenic cirrhosis (6.3%). Miscellaneous (7.5%) | 18.9% | Up to 3 months | ≥ 1.4 | 26 | 63 | 4 | 65 | 0.744 | |||||||
| Rahmani et al[49], Iran, 2021 | Prospective single-center | 98. Male = 66 (67.3) | 9.48± 4.9 years | Cirrhotic | 43.8% | NA | ≥ 0.45 | 33 | 18 | 10 | 37 | 0.799 | |||||||
| Poddar et al[16], India, 2023 | Retrospective single-center (2010-2022) | 110. Male = 75 (68.1) | 12 (8-24) months | Cirrhotic. Biliary atresia | 31.8% | None | ≥ 2 | 29 | 28 | 6 | 47 | 0.720 | |||||||
| Yokoyama et al[50], Japan, 2024 | Prospective single-center (2018-2022) | 53. Male = 17 | 4 (0-18) years | Cirrhotic. Biliary atresia-post KPE | 52.8% | Up to 1 month | ≥ 1.476 | 22 | 5 | 6 | 20 | 0.826 | |||||||
| Platelet count | Gana et al[32], Multicentric, 2011 | Prospective multi-center (2007-2010) | 108. Male = 53 (48) | 11 ± 5.4 years | Cirrhotic + non-cirrhotic | 68.5% | Up to 3 weeks | < 1.15 lakh/mm3 | 60 | 10 | 14 | 24 | 0.790 | ||||||
| Alcantara et al[33], Brazil, 2012 | Prospective. Single-center (2007-2011) | 35. Gender = NA | Median: 11 years | Cirrhotic. Autoimmune hepatitis (54.2%). Biliary atresia (28.5%). Miscellaneous (17.1%) | 48.5% | Up to 3 months | < 1.19 lakh/mm3 | 13 | 3 | 4 | 15 | 0.830 | |||||||
| Adami et al[34], Brazil, 2013 | Retrospective single-center (2000-2011) | 103. Male = 47 (45.7) | 8.9 ± 4.7 years | Cirrhotic + non-cirrhotic | 68.9% | Up to 3 months | < 1.15 lakh/mm3 | 48 | 6 | 23 | 26 | 0.820 | |||||||
| Adami et al[39], Brazil, 2018 | Retrospective single-center (2000-2011) | 98. Male = 46 (46.9) | 9.1 ± 4.7 years | Cirrhotic | 32.6% | Up to 3 months | < 115 × 109/L | 21 | 30 | 11 | 36 | 0.67 | |||||||
| Chiou et al[42], Singapore, 2019 | Retrospective single-center (1989-2016) | 72. Male = 35 (48.6) | NA | Cirrhotic. Biliary atresia post-KPE | 22.2% | 6 months | < 100 × 109/L | 12 | 11 | 4 | 45 | 0.800 | |||||||
| Alam et al[45], Bangladesh, 2019 | Prospective single-center (2016-2018) | 84. Male = 44 (52.4) | 9.7 ± 3.2 years | Cirrhotic. Wilson disease (65.4%). Hepatitis B/C (7.1%). Autoimmune hepatitis (7.1%). Biliary atresia (5.9%). Miscellaneous (14.2%) | 71.4%-EV. 55%-CSV | None | < 1 lakh/mm3 | 26 | 1 | 34 | 23 | 0.740 | < 100 × 109/L | 17 | 9 | 16 | 18 | 0.640 | |
| Vadlapudi et al[47], India, 2020 | Prospective single-center (2017-2018) | 55. Male = 33 (60) | 9.37 ± 3.12 years | Non-cirrhotic Extrahepatic portal vein thrombosis | 87.2% | NA | < 117 × 109/L | 37 | 3 | 11 | 4 | 0.568 | |||||||
| Lopes et al[48], Brazil, 2021 | Retrospective single-center (2004-2018) | 158. Male = 70 (44.3) | 5.38 (2.08-11.52) years | Cirrhotic | 18.9% | Up to 3 months | < 100 × 109/L | 13 | 21 | 17 | 107 | 0.709 | |||||||
| Rahmani et al[49], Iran, 2021 | Prospective single-center | 98. Male = 66 (67.3) | 9.48 ± 4.9 years | Cirrhotic | 43.8% | NA | < 1.1 lakh/mm3 | 36 | 18 | 7 | 37 | 0.833 | |||||||
| Poddar et al[16], India, 2023 | Retrospective single-center (2010-2022) | 110. Male = 75 (68.1) | 12 (8–24) months | Cirrhotic. Biliary atresia | 31.8% | None | < 150 × 109/L | 26 | 25 | 9 | 50 | 0.690 | |||||||
| Yokoyama et al[50], Japan, 2024 | Prospective single-center (2018-2022) | 53. Male = 17 | 4 (0-18) years | Cirrhotic. Biliary atresia post-KPE | 52.8% | Up to 1 month | < 141 × 109/L | 20 | 10 | 8 | 15 | 0.721 | |||||||
| Index test | Ref. | Study design/period | Sample size/gender | Age | Prevalence of varices | Etiology of portal hypertension | Interval between index test and reference standard | Any grade oesophageal/gastric varices | Clinically significant varices | ||||||||||
| Cut-off value | TP | FP | FN | TN | AUROC | Cut-off value | TP | FP | FN | TN | AUROC | ||||||||
| Clinical prediction rule | Gana et al[31], Canada, 2010 | Retrospective single-center (2000-2007) | 51. Male= 25 (49) | Mean (range): 11 (0.2-17) years | 33.3% | Cirrhotic + non-cirrhotic | Up to 6 months | ≤ 116 | 14 | 4 | 3 | 30 | 0.93 | ||||||
| Gana et al[32], Multicentric, 2011 | Prospective multi-center (2007-2010) | 108. Male = 53 (48) | 11 ± 5.4 years | 68.5% | Cirrhotic + non-cirrhotic | Up to 3 weeks | ≤ 116 | 60 | 9 | 14 | 25 | 0.80 | |||||||
| Adami et al[34], Brazil, 2013 | Retrospective single-center (2000-2011) | 103. Male = 47 (45.7) | 8.9 ± 4.7 years | 68.9% | Cirrhotic + non-cirrhotic | Up to 3 months | ≤ 114 | 53 | 7 | 18 | 25 | 0.77 | |||||||
| Isted et al[35], United Kingdom, 2015 | Retrospective single-center (2004-2012) | 195. Male = 83 (42.5) | 55 (11-216) days | 21.5% | Cirrhotic. Biliary atresia post-KPE | NA | ≤ 114 | 29 | 55 | 13 | 98 | 0.74 | |||||||
| Tomita et al[37], Japan, 2016 | Prospective single-center (2011-2014) | 16 | Median (range): 6.3 (0.7-33.5) years | 18.7% | Cirrhotic. Biliary atresia-post KPE | Up to 3 months | ≤ 106 | 2 | 2 | 1 | 11 | 0.76 | |||||||
| Witters et al[38], United Kingdom, 2017 | Prospective single-center (2005-2014) | 124. Male = 66 (53.2) | 8.81 ± 5.60 years | 63.7% | Cirrhotic + non-cirrhotic | Up to 6 months | ≤ 114 | 60 | 18 | 19 | 27 | 0.73 | |||||||
| Adami et al[39], Brazil, 2018 | Retrospective single-center (2000-2011) | 98. Male= 46 (46.9) | 9.1 ± 4.7 years | 32.6% | Cirrhotic | Up to 3 months | ≤ 114 | 26 | 31 | 6 | 35 | 0.65 | |||||||
| Sutton et al[40], United Kingdom, 2018 | Prospective single-center (2015-2016) | 67. Male= 32 (47.7) | 8.5 (0.3-18) years | 56.7% | Cirrhotic + non-cirrhotic | None | ≤ 106.3 | 17 | 10 | 21 | 19 | 0.54 | |||||||
| Yokoyama et al[43], Japan, 2019 | Prospective single-center (2018-2019) | 34. Male= 11 (32.2) | 5.2 ± 5.92 years | 41.1% | Cirrhotic. Biliary atresia-post KPE | 1-2 days | ≤ 120.3 | 12 | 7 | 2 | 13 | 0.782 | |||||||
| Lampela et al[44], Finland, 2020 | Prospective single-center (1989-2017) | 45. Gender = NA | 1.6 (0.7-14) years | 46.6% | Cirrhotic. Biliary atresia-post KPE | None. Both done on same day | ≤ 105 | 11 | 4 | 10 | 20 | 0.69 | |||||||
| Diéguez Hernández-Vaquero et al[51], Spain, 2024 | Retrospective single-center | 45 UGIE (in 18 patients). Male = 11 (61.1)1 | NA | 80% | Non-cirrhotic. Extrahepatic portal vein thrombosis | Up to 6 months | ≤ 115 | 8 | 0 | 28 | 9 | 0.815 | |||||||
| Jezequel et al[52], France, 2025 | Retrospective single-center (2020-2024) | 58. Male = 28 (48.3) | 9.20 (4.0-14.3) years | 53.4% | Cirrhotic + non-cirrhotic. Biliary atresia (32.8%). Autoimmune hepatitis (19%). Portal vein thrombosis (13.8%). Wilson disease (5.2%). Miscellaneous (29.2%) | NA | ≤ 116.4 | 23 | 6 | 8 | 21 | 0.71 | |||||||
| Varice prediction rule | Isted et al[35], United Kingdom, 2015 | Retrospective single-center (2004-2012) | 195. Male = 83 (42.5) | 55 (11-216) days | 21.5% | Cirrhotic. Biliary atresia post-KPE | NA | ≤ 7.2 | 36 | 44 | 6 | 109 | 0.75 | ||||||
| Tomita et al[37], Japan, 2016 | Prospective single-center (2011-2014) | 16. Gender = NA | Median (range): 6.3 (0.7-33.5) years | 18.7% | Cirrhotic. Biliary atresia post-KPE | Up to 3 months | ≤ 7.16 | 3 | 3 | 0 | 10 | 0.85 | |||||||
| Witters et al[38], United Kingdom, 2017 | Prospective single-center (2005-2014) | 124. Male = 66 (53.2) | 8.81 ± 5.60 years | 63.7% | Cirrhotic. Biliary atresia post-KPE | Up to 6 months | ≤ 3.63 | 63 | 18 | 16 | 27 | 0.72 | |||||||
| Chiou et al[42], Singapore, 2019 | Retrospective single-center (1989-2016) | 72. Male = 35 (48.6) | NA | 22.2% | Cirrhotic. Biliary atresia post-KPE | 6 months | ≤ 3 | 13 | 8 | 3 | 48 | 0.833 | |||||||
| Lampela et al[44], Finland, 2020 | Prospective single-center (1989-2017) | 45. Gender = NA | 1.6 (0.7-14) years | 46.6% | Cirrhotic. Biliary atresia post-KPE | None. Both done on same day | ≤ 2.76 | 16 | 10 | 5 | 14 | 0.720 | |||||||
| Lopes et al[48], Brazil, 2021 | Retrospective single-center (2004-2018) | 158. Male = 70 (44.3) | 5.38 (2.08-11.52) years | 18.9% | Cirrhotic | Up to 3 months | ≤ 7.2 | 25 | 66 | 5 | 62 | 0.752 | |||||||
| Diéguez Hernández-Vaquero et al[51], Spain, 2024 | Retrospective single-center | 45 UGI. (in 18 patients). Male = 11 (61.1) | NA | 80% | Non-cirrhotic. Extrahepatic portal vein thrombosis | Up to 6 months | ≤ 7.3 | 24 | 0 | 12 | 9 | 0.858 | |||||||
| King’s variceal prediction score (K-VaPS) | Witters et al[38], United Kingdom, 2017 | Prospective single-center (2005-2014) | 124. Male = 66 (53.2) | 8.81 ± 5.60 years | 63.7% | Cirrhotic + non-cirrhotic | Up to 6 months | ≤ 76 | 57 | 12 | 22 | 33 | 0.77 | ||||||
| Sutton et al[40], United Kingdom, 2018 | Prospective single-center (2015-2016) | 67. Male = 32 (47.7) | Median (range): 8.5 (0.3-18) years | 56.7% | Cirrhotic + non-cirrhotic | None | ≤ 75.2 | 15 | 11 | 23 | 18 | 0.48 | |||||||
| Yokoyama et al[43], Japan, 2019 | Prospective single-center (2018-2019) | 34. Male = 11 (32.2) | 5.2 ± 5.92 years | 41.1% | Cirrhotic. Biliary atresia-post KPE | 1-2 days | ≤ 96.1 | 10 | 5 | 4 | 15 | 0.775 | |||||||
| Lampela et al[44], Finland, 2020 | Prospective single-center (1989-2017) | 45. Gender = NA | 1.6 (0.7-14) years | 46.6% | Cirrhotic. Biliary atresia-post KPE | None. Both done on same day | ≤ 68 | 14 | 8 | 7 | 16 | 0.670 | |||||||
| Diéguez Hernández-Vaquero et al[51], Spain, 2024 | Retrospective single-center | 45 UGIE (in 18 patients). Male = 11 (61.1)1 | NA | 80% | Non-cirrhotic. Extrahepatic portal vein thrombosis | Up to 6 months | ≤ 76 | 2 | 0 | 34 | 9 | 0.753 | |||||||
APRI: Seventeen studies, including 1355 pediatric patients, evaluated the diagnostic accuracy of APRI for variceal prediction and were published between 2011 and 2022[16,29,30,34-39,41-44,46,48-50]. Ten of them were conducted pro
Using the QUADAS-2 tool (Figure 2A), 6 studies were identified as having a high risk of bias across the flow and timing domains, as the interval between UGIE and APRI estimation was either not reported or more than three months[30,35,38,41-42,49]. Eight studies had unclear risk of bias in patient selection domain due to retrospective design in 7 studies[16,34,35,39,41,42,48] and non-random sampling in one study[37]. For the index test and reference standard domain, also, the majority of the included studies were labelled as having unclear risk of bias due to the absence of reporting on the blinding of investigators. One study had applicability concerns in the patient selection domain due to poorly defined inclusion criteria[37].
Platelet count: Eleven studies (6-prospective, 5-retrospective), including 968 children with PHT, were identified[16,32-34,39,42,45,47-50]. Of these, four (n = 428) and six studies (n = 612) predicted diagnostic accuracy for EV[32-34,49] and CSV[16,39,42,47,48,50], respectively, while one study evaluated accuracy for both EV and CSV[45]. Six studies were from Asian countries, four from Brazil, and the rest was a multicentre study. Cut-off values of platelet count for EV and CSV prediction ranged from 100 × 109/L to 119 × 109/L and 100 × 109/L to 150 × 109/L respectively and all were derived post-hoc. The prevalence of EV and CSV in the included studies ranged from 48.3% to 71.4% and 18.9% to 87.2%, respectively (Table 1). Three studies have a high risk of bias across the flow and timing domain, as the time interval between the index test and the reference standard was more than 6 months in one study[42] and was not reported in the other two studies[47,49] (Figure 2B).
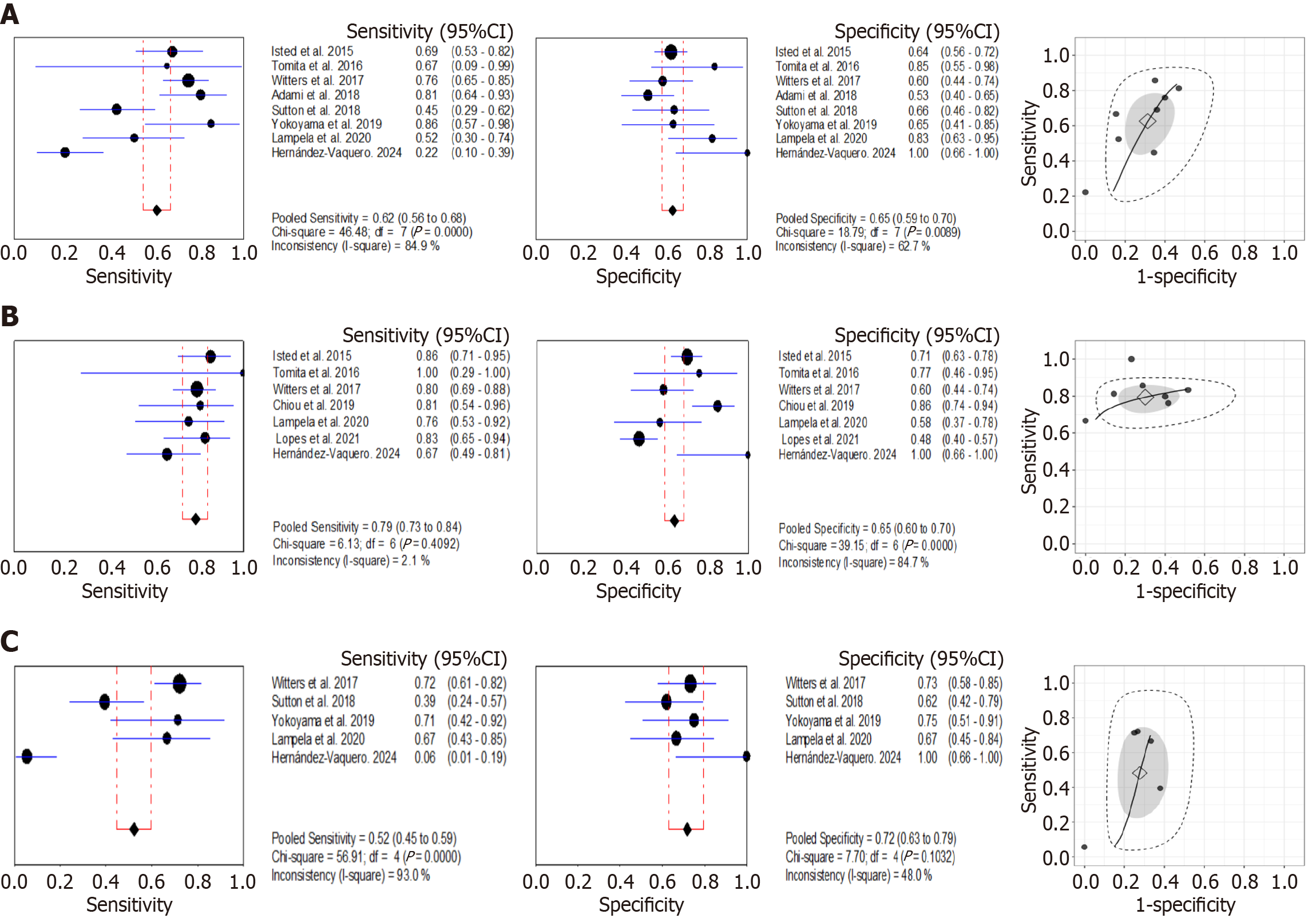
CPR: Of the total 12 included studies, 4 studies (n = 320) evaluated the diagnostic accuracy of CPR for EV prediction[31,32,34,52]. The remaining 8 studies (n = 624) assessed accuracy for CSV prediction with cut-off values ranging from 105 to 120.3[35,37-40,43,44,51] (Table 2). Five studies have a high risk of bias across the flow and timing domain[32,35,38,51,52] (Supplementary Figure 1A).
VPR: Data from seven studies (n = 655) were pooled to assess the diagnostic accuracy of VPR for CSV prediction, with cut-offs ranging from 2.76 to 7.3[35,37,38,42,44,48,51] (Table 2). Four among the seven studies have a high risk of bias across the flow and timing domain[35,38,42,51] (Supplementary Figure 1B).
K-VaPS: Five studies (n = 315) estimated the diagnostic accuracy for CSV prediction with cut-offs ranging from 68 to 96.1[38,40,43,44,51] (Table 2). Two studies have a high risk of bias across the flow and timing domain[38,51] (Supplementary Figure 1C).
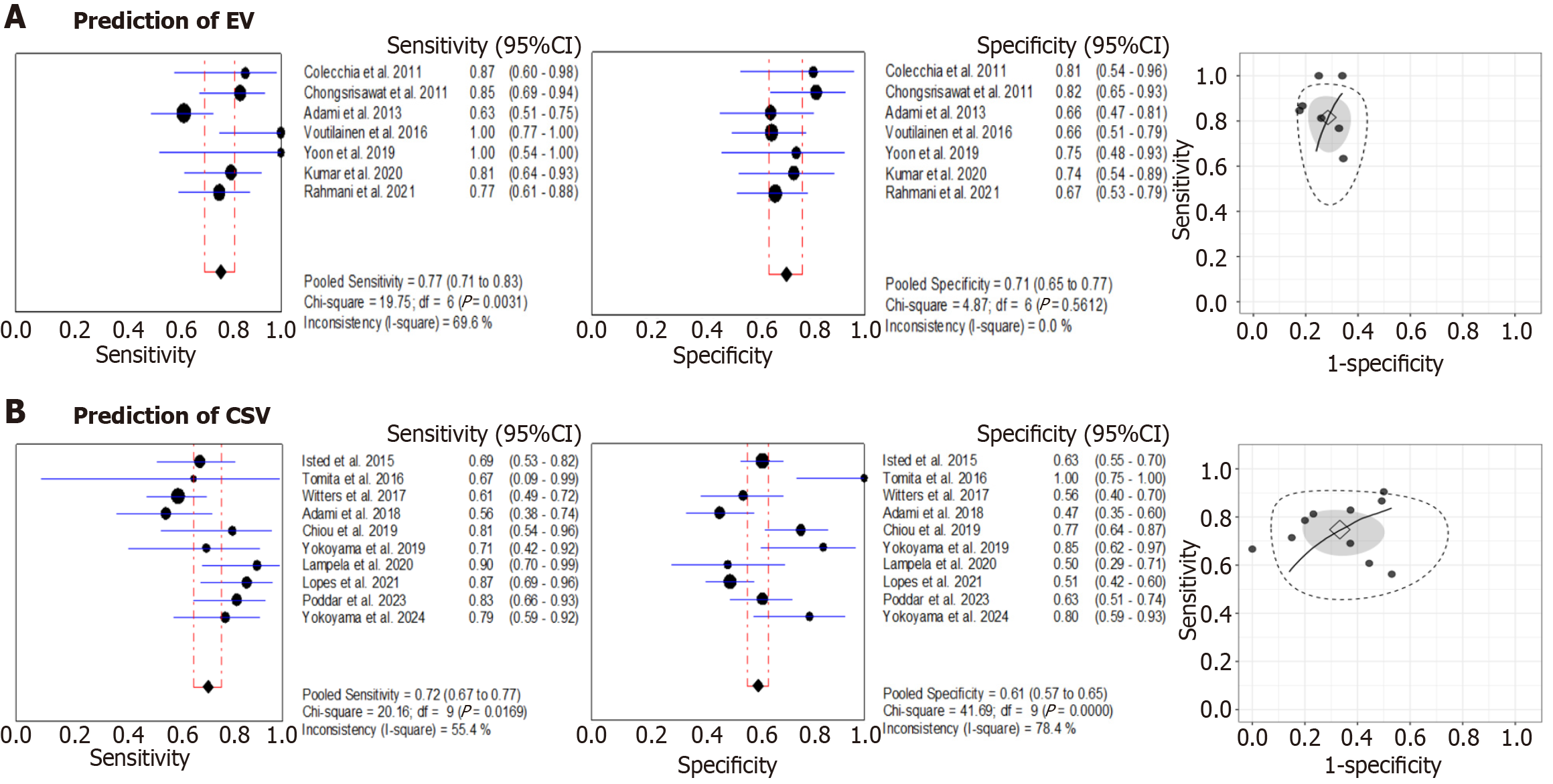
APRI: For the prediction of EV, APRI demonstrated a pooled sensitivity of 0.77 (95%CI: 0.71-0.83) and a pooled specificity of 0.71 (95%CI: 0.65-0.77), with a pooled DOR of 11.15 (95%CI: 4.75-33.09) and an AUC of 0.795. Between-study heterogeneity was substantial for sensitivity (I2 = 69.6%), whereas no heterogeneity was observed for specificity (I2 = 0%) in EV prediction Figure 3A. At a pre-test probability of 50%, a positive APRI result increased the post-test probability of EV to approximately 73%, while a negative result reduced it to 24% (Table 3). For the prediction of CSV, the pooled sensitivity was 0.72 (95%CI: 0.67-0.77) and the pooled specificity was 0.61 (95%CI: 0.57-0.65), yielding a pooled DOR of 5.91 (95%CI: 2.91-18.62) and an AUC of 0.776. Moderate to substantial heterogeneity was observed for both sensitivity (I2 = 55.4%) and specificity (I2 = 78.4%) (Figure 3B). At a pre-test probability of 50%, a negative APRI reduced the post-test probability of CSV to approximately 32%, whereas a positive test increased it to 65% (Table 3).
| Index test | Target condition | No. of study cohorts | Pooled sensitivity (95%CI) | Pooled specificity (95%CI) | Diagnostic odds ratio (95%CI) | PLR (95%CI) | NLR (95%CI) | AUC | Pre-test probability (%) | Post-test probability (+) (%) | Post-test probability (-) (%) |
| APRI | EV | 7 | 0.77 (0.71-0.83) | 0.71 (0.65-0.77) | 11.15 (4.75-33.09) | 2.85 (2.06-4.07) | 0.26 (0.11-0.46) | 0.795 | 25 | 47 | 10 |
| 50 | 73 | 24 | |||||||||
| 75 | 89 | 49 | |||||||||
| CSV | 10 | 0.72 (0.67-0.77) | 0.61 (0.57-0.65) | 5.91 (2.91-18.62) | 2.23 (1.57-4.94) | 0.38 (0.24-0.55) | 0.776 | 25 | 38 | 13 | |
| 50 | 65 | 32 | |||||||||
| 75 | 85 | 58 | |||||||||
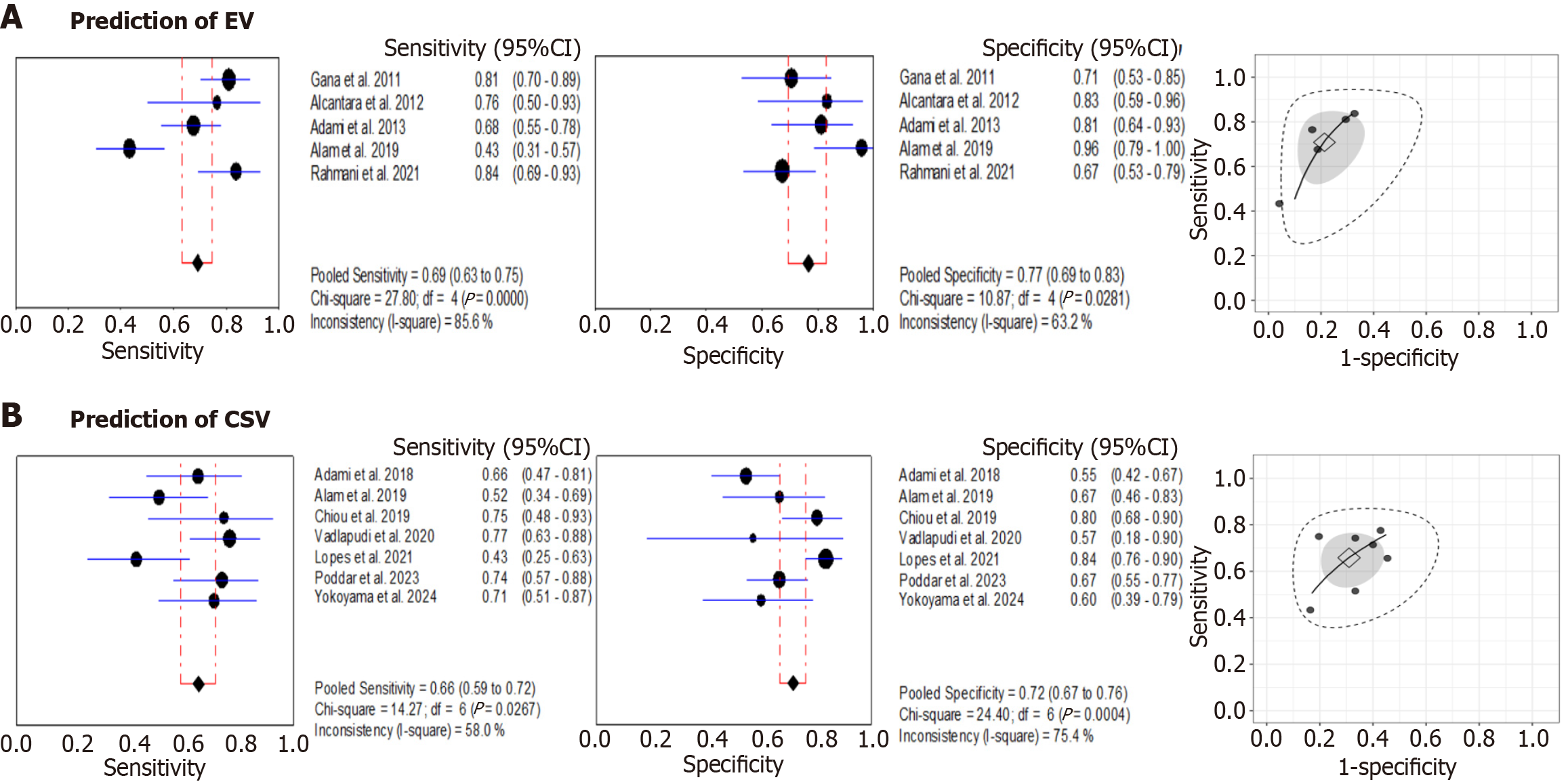
| Platelet count | EV | 5 | 0.69 (0.63-0.75) | 0.77 (0.69-0.83) | 9.23 (3.40-25.67) | 3.30 (1.94-6.34) | 0.36 (0.19-0.62) | 0.834 | 25 | 50 | 12 |
| 50 | 75 | 29 | |||||||||
| 75 | 90 | 55 | |||||||||
| CSV | 7 | 0.66 (0.59-0.72) | 0.72 (0.67-0.76) | 4.25 (1.95-8.56) | 2.1 (1.38-3.14) | 0.5 (0.33-0.71) | 0.714 | 25 | 61 | 5 | |
| 50 | 83 | 15 | |||||||||
| 75 | 93 | 34 | |||||||||
| CPR | EV | 4 | 0.78 (0.71-0.83) | 0.80 (0.71-0.86) | 12.91 (5.72-28.50) | 3.68 (2.14-6.17) | 0.29 (0.19-0.43) | 0.848 | 25 | 70 | 6 |
| 50 | 87 | 17 | |||||||||
| 75 | 95 | 38 | |||||||||
| CSV | 8 | 0.62 (0.56-0.68) | 0.65 (0.59-0.70) | 3.76 (1.74-8.40) | 2.0 (1.35-3.26) | 0.54 (0.32-0.79) | 0.717 | 25 | 37 | 16 | |
| 50 | 64 | 37 | |||||||||
| 75 | 84 | 64 | |||||||||
| VPR | CSV | 7 | 0.79 (0.73-0.84) | 0.65 (0.60-0.70) | 9.19 (4.06-21.7) | 2.64 (1.73-5.11) | 0.29 (0.19-0.44) | 0.864 | 25 | 43 | 10 |
| 50 | 70 | 24 | |||||||||
| 75 | 87 | 49 | |||||||||
| K-VaPS | CSV | 5 | 0.52 (0.45-0.59) | 0.72 (0.63-0.79) | 2.43 (0.72-8.17) | 1.72 (0.78-3.16) | 0.72 (0.36-1.08) | 0.727 | 25 | 38 | 18 |
| 50 | 65 | 40 | |||||||||
| 75 | 85 | 67 |
Platelet count: For the prediction of EV, platelet count showed a pooled sensitivity of 0.69 (95%CI: 0.63-0.75) and a pooled specificity of 0.77 (95%CI: 0.69-0.83) with substantial heterogeneity (I2: 85.6% for sensitivity and 63.2% for specificity). Pooled DOR was 9.23 (95%CI: 3.40-25.67) with an AUC of 0.834 (Figure 4A). At a pre-test probability of 50%, a positive platelet count result increased the probability of EV to approximately 75%, while a negative result decreased it to 29%. When evaluated for CSV, platelet count yielded a pooled sensitivity of 0.66 (95%CI: 0.59-0.72) and a pooled specificity of 0.72 (95%CI: 0.67-0.76), with a pooled DOR of 4.25 (95%CI: 1.95-8.56) and an AUC of 0.714. Moderate to substantial heterogeneity was again evident, with I2 values of 58.0% for sensitivity and 75.4% for specificity (Figure 4B). At a pre-test probability of 50%, a negative platelet count reduced the likelihood of CSV to approximately 15%, whereas a positive result increased it to 83% (Table 3).
CPR: For the prediction of EV, CPR demonstrated a pooled sensitivity of 0.78 (95%CI: 0.71-0.83), a pooled specificity of 0.80 (95%CI: 0.71-0.86) and a pooled DOR of 12.91 (95%CI: 5.72-28.50) with an AUC of 0.848 (Supplementary Figure 2). The pooled accuracy parameters for CSV detection were as follows: Sensitivity: 0.62 (95%CI: 0.56–0.68); specificity: 0.65 (95%CI: 0.59-0.70), DOR: 3.76 (95%CI: 1.74-8.40) with an AUC of 0.717. Significant heterogeneity was observed in sensitivity (I2 = 84.9%) and specificity (I2 = 62.7%) for CSV prediction (Figure 5A, Table 3).
VPR: It showed a pooled sensitivity of 0.79 (95%CI: 0.73-0.84), I2 = 2.1% and a pooled specificity of 0.65 (95%CI: 0.60-0.70), I2 = 84.7%, with a pooled DOR of 9.19 (95%CI: 4.06-21.7) and an AUC of 0.864 for CSV prediction (Figure 5B, Table 3).
K-VaPS: It demonstrated a pooled sensitivity of 0.52 (95%CI: 0.45-0.59), I2 = 93% and a pooled specificity of 0.72 (95%CI: 0.63-0.79), I2 = 48% and a pooled DOR of 2.43 (95%CI: 0.72-8.17) with an AUC of 0.727 (Figure 5C, Table 3).
Meta-analyses performed after excluding studies at high risk of bias did not reveal any substantial changes in the pooled diagnostic accuracy estimates of the above five index tests for EV or CSV prediction (Supplementary Table 1). Given that APRI has been validated primarily as a non-invasive surrogate marker for variceal prediction in cirrhotic adults, a sensitivity analysis was undertaken, excluding the two studies that enrolled mixed (cirrhotic and non-cirrhotic) population of pediatric patients. The pooled diagnostic accuracy parameters remained largely unchanged, indicating the robustness of the overall findings.
To explore potential sources of heterogeneity, subgroup analyses were performed based on study design, sample size, geographic region, etiology of PHT, prevalence of EV/CSV, cut-off thresholds, and timing between index test and UGIE.
For both EV and CSV prediction, APRI has better accuracy in the Asian population (vs non-Asian population), study cohorts with only biliary atresia children (vs mixed etiology), prospective studies, sample size < 50 (vs > 50) and in studies with an interval between UGIE and index test < 1 week (vs > 1 week). Studies using APRI cut-off values < 1 demonstrated higher accuracy for the detection of EV, whereas for CSV prediction, studies with cut-off values > 2 showed superior performance (Supplementary Table 2). For platelet count in EV prediction, studies with platelet count cut-off values < 100 × 109/L showed higher accuracy compared to those using > 100 × 109/L (Supplementary Table 3). Likewise, CPR, VPR, and K-VaPS showed better accuracy in cohorts with Asian population, with exclusively biliary atresia children and with CSV prevalence < 50% (Supplementary Table 4).
Deek’s funnel plot asymmetry test indicated no significant evidence of publication bias for any of the five index tests for prediction of either EV or CSV, as shown in Supplementary Figures 3 and 4.
Using the GRADE approach, the overall certainty of evidence for the diagnostic accuracy of the five index tests ranged from “moderate” to “very low” for the target condition (EV/CSV prediction), with downgrading driven primarily by the risk of bias and inconsistency, as summarized in Table 4.
| Diagnostic test for the target condition | No. of studies | Study design | Accuracy parameter | Risk of bias | Indirectness | Inconsistency | Imprecision | Publication bias | Overall certainty of evidence |
| APRI for EV | 7 studies (n = 450) | Cross-sectional | Sensitivity | Serious1 | Not serious | Serious2 | Not serious | None | Low |
| Specificity | Serious1 | Not serious | Not serious | Not serious | None | Moderate | |||
| APRI for CSV | 10 studies (n = 892) | Cross-sectional | Sensitivity | Serious1 | Not serious | Not serious | Serious3 | None | Low |
| Specificity | Serious1 | Not serious | Serious2 | Not serious | None | Low | |||
| Platelet count for EV | 5 studies (n = 368) | Cross-sectional | Sensitivity | Serious1 | Not serious | Serious2 | Not serious | None | Low |
| Specificity | Serious1 | Not serious | Serious2 | Not serious | None | Low | |||
| Platelet count for CSV | 7 studies (n = 612) | Cross-sectional | Sensitivity | Serious1 | Not serious | Not serious | Not serious | None | Moderate |
| Specificity | Serious1 | Not serious | Serious2 | Serious3 | None | Very low | |||
| CPR for EV | 4 studies (n = 320) | Cross-sectional | Sensitivity | Serious1 | Not serious | Not serious | Not serious | None | Moderate |
| Specificity | Serious1 | Not serious | Not serious | Not serious | None | Moderate | |||
| CPR for CSV | 8 studies (n = 624) | Cross-sectional | Sensitivity | Serious1 | Not serious | Serious2 | Serious3 | None | Very low |
| Specificity | Serious1 | Not serious | Serious2 | Not serious | None | Low | |||
| VPR for CSV | 7 studies (n = 655) | Cross-sectional | Sensitivity | Serious1 | Not serious | Not serious | Serious3 | None | Low |
| Specificity | Serious1 | Not serious | Serious2 | Not serious | None | Low | |||
| K-VaPS for CSV | 5 studies (n = 315) | Cross-sectional | Sensitivity | Serious1 | Not serious | Serious2 | Not serious | None | Low |
| Specificity | Serious1 | Not serious | Not serious | Not serious | None | Moderate |
This comprehensive systematic review and meta-analysis assessed the diagnostic accuracy of five blood-based non-invasive tests for predicting EV and CSV in children with PHT. Our findings indicate that commonly available tests/indices, including APRI, platelet count, and pediatric-specific prediction scores (CPR, VPR, and K-VaPS), demonstrate moderate diagnostic accuracy, with substantial heterogeneity across studies. Collectively, these results suggest that blood-based tests are at best suited as triage or rule-out tools rather than replacements for endoscopic screening.
Both APRI and platelet count exhibited relatively balanced sensitivity and specificity for EV prediction, with slightly lower performance for CSV, ranging from 60% to 80%. Of the various pediatric-specific prediction scores, VPR demonstrated the strongest performance for CSV prediction (AUC = 0.864), followed by K-VaPS (AUC = 0.727) and CPR (AUC = 0.717). Meta-analyses in adult populations have documented comparable pooled diagnostic accuracy estimates for APRI and platelet count[11,12].
Clinical utility was further supported by Fagan nomogram analysis for both APRI and platelet count, demonstrating their ability to meaningfully modify post-test probability. At a pre-test probability of 50%, a positive result for APRI increased the likelihood of EV to approximately 73%, while a negative result reduced it to 24%. For CSV, the corresponding post-test probabilities were 65% and 32%, respectively. Similarly, for platelet count, a positive test increased the probability of EV to 75%, whereas a negative test lowered it to 29%; for CSV, corresponding post-test probabilities were 83% and 15%. Among the prediction scores, VPR has the best post-test probabilities compared to CPR and K-VaPS. However, as with other non-invasive modalities, false-negative results remain a concern. Up to 24%-30% of children may be misclassified as not having EV after a negative APRI or platelet count result, and approximately 15%-32% may be misclassified as not having CSV, thereby missing UGIE and appropriate prophylaxis or follow-up. These findings underscore that while APRI and platelet count are useful triage tools to reduce unnecessary UGIE, they should not be used in isolation to definitively exclude EV/CSV.
Substantial between-study heterogeneity was noted across almost all analyses. Subgroup analyses indicated that heterogeneity in the diagnostic performance of blood-based tests was variably influenced by disease etiology, geographic region, study design, sample size, applied cut-off thresholds, and the interval between index testing and endoscopic assessment. However, one consistent pattern across all five blood-based tests/indices was the superior diagnostic performance observed in cohorts restricted to biliary atresia and in studies conducted in Asian populations. This is likely attributable to the early onset of cirrhosis in biliary atresia, with variceal development closely paralleling worsening hepatic fibrosis, portal pressure, and hypersplenism, resulting in a stronger and more predictable relationship between PHT severity and blood-based parameters. In contrast, cohorts with mixed etiologies include children with non-cirrhotic PHT, such as extrahepatic portal vein obstruction, in whom portal pressure and variceal formation may occur despite preserved hepatic synthetic function and near-normal biochemical profiles. Additionally, most of the Asian studies included in this meta-analysis consisted of biliary atresia-only cohorts, which further explains the superior performance of these index tests in this ethnic group.
In this context, APRI was originally developed as a surrogate marker of hepatic fibrosis and cirrhosis and has been extensively studied and validated in adult cirrhotic populations for assessing PHT and variceal risk; however, its biological applicability in non-cirrhotic PHT is inherently less robust. Nevertheless, pediatric PHT has a distinct pathophysiology, with a higher prevalence of non-cirrhotic PHT than the adult population, and both cirrhotic and non-cirrhotic children usually undergo similar endoscopic surveillance. Of the 17 studies evaluating APRI in this meta-analysis, two included mixed cohorts comprising both cirrhotic and non-cirrhotic patients. Sensitivity analyses excluding these studies did not materially alter the pooled diagnostic accuracy estimates, thereby confirming the robustness of the overall findings. However, this should not be interpreted as evidence of uniform applicability of APRI across all etiologies. As discussed earlier, etiology-based subgroup analyses consistently demonstrated superior diagnostic accuracy in biliary atresia–exclusive (cirrhotic) cohorts compared with mixed-etiology populations. Collectively, these findings indicate that APRI performs best in cirrhotic pediatric PHT and should be interpreted with caution in non-cirrhotic cases.
Variation in the cut-off thresholds used for APRI and platelet count emerged as another important contributor to between-study heterogeneity. Divergent patterns were observed, with studies applying lower APRI cut-offs (below 1) demonstrating better diagnostic performance for EV prediction, whereas studies using higher APRI thresholds (above 2) showed improved accuracy for CSV prediction. These findings do not imply that lower APRI values are intrinsically associated with higher EV risk. Rather, this observation reflects a diagnostic threshold effect inherent to ROC-derived cut-offs wherein lower APRI cut-offs increase sensitivity at the expense of specificity. Hence, their utility lies primarily in triage rather than definitive diagnosis. Similarly, studies employing lower platelet count cut-offs (below 100 × 109/L) exhibited superior performance for EV detection, while no consistent difference in diagnostic accuracy was observed for CSV prediction with regard to platelet cut-off. These seemingly paradoxical findings are likely influenced by the interplay of other covariates, including underlying etiology, study design, sample size, and baseline disease severity. These results also highlight the absence of standardised etiology-specific pediatric cut-offs and the pressing need for harmonised methodologies.
The certainty of evidence, as assessed using the GRADE framework, ranged from “moderate” to “very low” for the sensitivity and specificity of these five index tests, with downgrading primarily attributed to risk of bias and inconsistency. For all five index tests, one or more included studies were judged to be at high risk of bias in the flow and timing domain, mainly due to a lack of reporting of time interval or a prolonged interval (> 3 months) between the index test and UGIE. Furthermore, most included studies exhibited an unclear risk of bias in the patient selection domain, attributable to retrospective study designs, poorly defined inclusion criteria, or non-consecutive patient recruitment. A similar unclear risk of bias was also noted in the index test and reference standard domains, largely due to the absence of blinding of the respective investigators and the post hoc derivation of diagnostic cut-offs, which may have compromised internal validity. Despite these methodological limitations, sensitivity analyses excluding studies at high risk of bias did not materially alter the pooled diagnostic accuracy estimates, supporting the overall robustness of the findings of this meta-analysis.
There is only a single published study (a 2017 Cochrane review) that analysed the pooled accuracy of four pediatric studies using platelets as a non-invasive predictor of varices in children[12]. Hence, our study is the first comprehensive meta-analysis of diagnostic accuracy focused exclusively on blood-based non-invasive tests for predicting EV and CSV in children with PHT. The review followed PRISMA-DTA methodology, applied a rigorous hierarchical modelling ap
However, this study has several limitations, which should be acknowledged. First, most included studies were single-center, with relatively small sample sizes, and many employed retrospective designs, increasing the risk of selection bias. Cut-off values in all included studies were derived post hoc, which limits generalizability and precludes the identification of optimal pediatric thresholds. Second, although we performed subgroup analyses to explore potential sources of heterogeneity, it is possible that additional unmeasured or unreported factors-such as variations in age, gender, disease se
Despite these methodological limitations, the findings of this meta-analysis have important implications for the clinical management and variceal surveillance of children with PHT, for whom a non-invasive, risk-based triage approach is desirable given the invasiveness of pediatric UGIE. Although liver and splenic stiffness measurements have shown promise for variceal prediction, their routine pediatric use is constrained by cost, need for specialized equipment and pediatric probes, operator dependency, and limited accessibility, particularly outside tertiary centers. On the other hand, blood-based non-invasive tests, especially APRI and platelet counts, are inexpensive, widely available, easily repeatable, and independent of technical expertise, making them well-suited for longitudinal monitoring and large-scale triage. However, as stated earlier, their moderate diagnostic accuracy, coupled with between-study heterogeneity and the lack of standardized pediatric-specific cut-offs, suggests that these tests should be used as adjuncts rather than standalone tools when guiding endoscopic decision-making. The Baveno VII consensus recommends initiating endoscopic surveillance in adults with chronic liver disease when the platelet count falls below 150 × 109/L[12]. However, corresponding platelet count thresholds for the pediatric population have not been established. Notably, most studies included in this meta-analysis employed lower platelet cut-off values, typically ranging from 100 × 109/L to 120 × 109/L, for the prediction of both EV and CSV, highlighting the need for pediatric-specific threshold validation. Lastly, all three pediatric prediction scores require radiological assessment, typically ultrasonography, to determine splenic size or z-scores, which ne
The integration of blood-based tests with elastography-rather than reliance on a single modality-may represent the most effective strategy for risk stratification in clinical practice. From a practical standpoint, clinicians may consider using blood-based non-invasive tests as an initial filter, followed by targeted use of liver stiffness measurement/splenic stiffness measurement and UGIE to reduce unnecessary endoscopic procedures, optimise resource utilisation, and improve patient and family experience, without compromising safety.
Blood-based non-invasive tests, including APRI, platelet count, and pediatric-specific prediction scores (CPR, VPR, and K-VaPS), demonstrate moderate diagnostic accuracy for predicting EV and CSV in children with PHT. Although these tests cannot replace UGIE, they may serve as useful adjunctive tools to support risk stratification and guide surveillance strategies. A stepwise, risk-based approach integrating blood-based tests with elastography, where available, may represent the most practical strategy for pediatric variceal surveillance. Future prospective, adequately powered multicenter pediatric studies are required to establish etiology-specific and age-appropriate cut-off thresholds, validate combined diagnostic algorithms, and inform evidence-based guidelines for variceal surveillance in pediatric PHT.
| 1. | Grammatikopoulos T, McKiernan PJ, Dhawan A. Portal hypertension and its management in children. Arch Dis Child. 2018;103:186-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 2. | Chapin CA, Bass LM. Cirrhosis and Portal Hypertension in the Pediatric Population. Clin Liver Dis. 2018;22:735-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 3. | Costaguta A, Alvarez F. Etiology and management of hemorrhagic complications of portal hypertension in children. Int J Hepatol. 2012;2012:879163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Ackermann O, Bernard O, Franchi-Abella S, Almes M, Tanase C, Jacquemin E, Duché M. The Natural History of Gastroesophageal Varices in Children With Portal Hypertension. Gastroenterology. 2026;170:188-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Thakkar K, El-Serag HB, Mattek N, Gilger MA. Complications of pediatric EGD: a 4-year experience in PEDS-CORI. Gastrointest Endosc. 2007;65:213-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 131] [Article Influence: 6.9] [Reference Citation Analysis (3)] |
| 6. | Tringali A, Balassone V, De Angelis P, Landi R. Complications in pediatric endoscopy. Best Pract Res Clin Gastroenterol. 2016;30:825-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (3)] |
| 7. | Attard TM, Grima AM, Thomson M. Pediatric Endoscopic Procedure Complications. Curr Gastroenterol Rep. 2018;20:48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Hathagoda W, Rajindrajith S, Devanarayana NM. Upper gastrointestinal endoscopy in pediatric gastroenterology: Diagnostic and therapeutic applications. World J Gastrointest Endosc. 2025;17:111386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Duché M, Ducot B, Ackermann O, Guérin F, Jacquemin E, Bernard O. Portal hypertension in children: High-risk varices, primary prophylaxis and consequences of bleeding. J Hepatol. 2017;66:320-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 77] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 10. | Alatas FS, Monica E, Ongko L, Kadim M. Endoscopic Primary Prophylaxis to Prevent Bleeding in Children with Esophageal Varices: A Systematic Review and Meta-Analysis. Pediatr Gastroenterol Hepatol Nutr. 2023;26:231-238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 11. | Deng H, Qi X, Guo X. Diagnostic Accuracy of APRI, AAR, FIB-4, FI, King, Lok, Forns, and FibroIndex Scores in Predicting the Presence of Esophageal Varices in Liver Cirrhosis: A Systematic Review and Meta-Analysis. Medicine (Baltimore). 2015;94:e1795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 90] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 12. | Colli A, Gana JC, Yap J, Adams-Webber T, Rashkovan N, Ling SC, Casazza G. Platelet count, spleen length, and platelet count-to-spleen length ratio for the diagnosis of oesophageal varices in people with chronic liver disease or portal vein thrombosis. Cochrane Database Syst Rev. 2017;4:CD008759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 13. | Chen R, Deng H, Ding X, Xie C, Wang W, Shen Q. Platelet Count to Spleen Diameter Ratio for the Diagnosis of Gastroesophageal Varices in Liver Cirrhosis: A Systematic Review and Meta-Analysis. Gastroenterol Res Pract. 2017;2017:7407506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Sutton H, Dhawan A, Grammatikopoulos T. Non-invasive Markers of Portal Hypertension: Appraisal of Adult Experience and Potential Utilisation in Children. J Pediatr Gastroenterol Nutr. 2018;66:559-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | McInnes MDF, Moher D, Thombs BD, McGrath TA, Bossuyt PM; and the PRISMA-DTA Group, Clifford T, Cohen JF, Deeks JJ, Gatsonis C, Hooft L, Hunt HA, Hyde CJ, Korevaar DA, Leeflang MMG, Macaskill P, Reitsma JB, Rodin R, Rutjes AWS, Salameh JP, Stevens A, Takwoingi Y, Tonelli M, Weeks L, Whiting P, Willis BH. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA. 2018;319:388-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2615] [Cited by in RCA: 2380] [Article Influence: 297.5] [Reference Citation Analysis (3)] |
| 16. | Poddar U, Samanta A, Sarma MS, Kumar B, Lal R, Srivastava A, Upadhyaya VD, Yachha SK, Mandelia A. How to suspect the presence of high-risk esophageal varices and when to start endoscopic surveillance in children with biliary atresia? J Gastroenterol Hepatol. 2023;38:1610-1617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM; QUADAS-2 Group. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155:529-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11012] [Cited by in RCA: 10558] [Article Influence: 703.9] [Reference Citation Analysis (5)] |
| 18. | Deeks JJ, Macaskill P, Irwig L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol. 2005;58:882-893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2470] [Cited by in RCA: 2329] [Article Influence: 110.9] [Reference Citation Analysis (3)] |
| 19. | Schünemann HJ, Mustafa RA, Brozek J, Steingart KR, Leeflang M, Murad MH, Bossuyt P, Glasziou P, Jaeschke R, Lange S, Meerpohl J, Langendam M, Hultcrantz M, Vist GE, Akl EA, Helfand M, Santesso N, Hooft L, Scholten R, Rosen M, Rutjes A, Crowther M, Muti P, Raatz H, Ansari MT, Williams J, Kunz R, Harris J, Rodriguez IA, Kohli M, Guyatt GH; GRADE Working Group. GRADE guidelines: 21 part 1. Study design, risk of bias, and indirectness in rating the certainty across a body of evidence for test accuracy. J Clin Epidemiol. 2020;122:129-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 262] [Article Influence: 43.7] [Reference Citation Analysis (0)] |
| 20. | Schünemann HJ, Mustafa RA, Brozek J, Steingart KR, Leeflang M, Murad MH, Bossuyt P, Glasziou P, Jaeschke R, Lange S, Meerpohl J, Langendam M, Hultcrantz M, Vist GE, Akl EA, Helfand M, Santesso N, Hooft L, Scholten R, Rosen M, Rutjes A, Crowther M, Muti P, Raatz H, Ansari MT, Williams J, Kunz R, Harris J, Rodriguez IA, Kohli M, Guyatt GH; GRADE Working Group. GRADE guidelines: 21 part 2. Test accuracy: inconsistency, imprecision, publication bias, and other domains for rating the certainty of evidence and presenting it in evidence profiles and summary of findings tables. J Clin Epidemiol. 2020;122:142-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 271] [Article Influence: 45.2] [Reference Citation Analysis (1)] |
| 21. | Freeman SC, Kerby CR, Patel A, Cooper NJ, Quinn T, Sutton AJ. Development of an interactive web-based tool to conduct and interrogate meta-analysis of diagnostic test accuracy studies: MetaDTA. BMC Med Res Methodol. 2019;19:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 210] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 22. | Patel A, Cooper N, Freeman S, Sutton A. Graphical enhancements to summary receiver operating characteristic plots to facilitate the analysis and reporting of meta-analysis of diagnostic test accuracy data. Res Synth Methods. 2021;12:34-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 138] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 23. | Cerullo E, Sutton AJ, Jones HE, Wu O, Quinn TJ, Cooper NJ. MetaBayesDTA: codeless Bayesian meta-analysis of test accuracy, with or without a gold standard. BMC Med Res Methodol. 2023;23:127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 24. | Fagundes ED, Ferreira AR, Roquete ML, Penna FJ, Goulart EM, Figueiredo Filho PP, Bittencourt PF, Carvalho SD, Albuquerque W. Clinical and laboratory predictors of esophageal varices in children and adolescents with portal hypertension syndrome. J Pediatr Gastroenterol Nutr. 2008;46:178-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Hasan MI, Rukunuzzaman M, Nurullah M, Sultana F. Clinical and Laboratory Predictors of Esophageal Varices in Children with Chronic Liver Disease. Mymensingh Med J. 2017;26:341-350. [PubMed] |
| 26. | Sintusek P, Siriporn N, Punpanich D, Chongsrisawat V, Poovorawan Y. Spleen and Liver Stiffness to Detect Esophageal Varices in Children with Biliary Atresia. J Pediatr Gastroenterol Nutr. 2019;69:411-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 27. | Kang Y, Park S, Kim S, Han SJ, Koh H. Validating the BAVENO VI criteria to identify low risk biliary atresia patients without endoscopy for esophageal varix. Clin Res Hepatol Gastroenterol. 2021;45:101437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 28. | Upadhyay P, Khanna R, Sood V, Lal BB, Patidar Y, Alam S. Splenic Stiffness Is the Best Predictor of Clinically Significant Varices in Children With Portal Hypertension. J Pediatr Gastroenterol Nutr. 2023;76:364-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 29. | Colecchia A, Di Biase AR, Scaioli E, Predieri B, Iughetti L, Reggiani ML, Montrone L, Ceccarelli PL, Vestito A, Viola L, Paolucci P, Festi D. Non-invasive methods can predict oesophageal varices in patients with biliary atresia after a Kasai procedure. Dig Liver Dis. 2011;43:659-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 30. | Chongsrisawat V, Vejapipat P, Siripon N, Poovorawan Y. Transient elastography for predicting esophageal/gastric varices in children with biliary atresia. BMC Gastroenterol. 2011;11:41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 31. | Gana JC, Turner D, Roberts EA, Ling SC. Derivation of a clinical prediction rule for the noninvasive diagnosis of varices in children. J Pediatr Gastroenterol Nutr. 2010;50:188-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Gana JC, Turner D, Mieli-Vergani G, Davenport M, Miloh T, Avitzur Y, Yap J, Morinville V, Brill H, Ling SC. A clinical prediction rule and platelet count predict esophageal varices in children. Gastroenterology. 2011;141:2009-2016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 33. | Alcantara RV, Yamada RM, De Tommaso AM, Bellomo-Brandão MA, Hessel G. Non-invasive predictors of esophageous varices in children and adolescents with chronic liver disease or extrahepatic portal venous obstruction. J Pediatr (Rio J). 2012;88:341-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 34. | Adami MR, Ferreira CT, Kieling CO, Hirakata V, Vieira SM. Noninvasive methods for prediction of esophageal varices in pediatric patients with portal hypertension. World J Gastroenterol. 2013;19:2053-2059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 32] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Isted A, Grammatikopoulos T, Davenport M. Prediction of esophageal varices in biliary atresia: Derivation of the "varices prediction rule", a novel noninvasive predictor. J Pediatr Surg. 2015;50:1734-1738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 36. | Voutilainen S, Kivisaari R, Lohi J, Jalanko H, Pakarinen MP. A Prospective Comparison of Noninvasive Methods in the Assessment of Liver Fibrosis and Esophageal Varices in Pediatric Chronic Liver Diseases. J Clin Gastroenterol. 2016;50:658-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 37. | Tomita H, Ohkuma K, Masugi Y, Hosoe N, Hoshino K, Fuchimoto Y, Fujino A, Shimizu T, Kato M, Fujimura T, Ishihama H, Takahashi N, Tanami Y, Ebinuma H, Saito H, Sakamoto M, Nakano M, Kuroda T. Diagnosing native liver fibrosis and esophageal varices using liver and spleen stiffness measurements in biliary atresia: a pilot study. Pediatr Radiol. 2016;46:1409-1417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 38. | Witters P, Hughes D, Karthikeyan P, Ramakrishna S, Davenport M, Dhawan A, Grammatikopoulos T. King's Variceal Prediction Score: A Novel Noninvasive Marker of Portal Hypertension in Pediatric Chronic Liver Disease. J Pediatr Gastroenterol Nutr. 2017;64:518-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 39. | Adami MR, Kieling CO, Schwengber FP, Hirakata VN, Vieira SMG. Noninvasive Methods of Predicting Large Esophageal Varices in Children With Intrahepatic Portal Hypertension. J Pediatr Gastroenterol Nutr. 2018;66:442-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 40. | Sutton H, Fitzpatrick E, Davenport M, Burford C, Alexander E, Dhawan A, Grammatikopoulos T. Transient Elastography Measurements of Spleen Stiffness as a Predictor of Clinically Significant Varices in Children. J Pediatr Gastroenterol Nutr. 2018;67:446-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 41. | Yoon H, Shin HJ, Kim MJ, Han SJ, Koh H, Kim S, Lee MJ. Predicting gastroesophageal varices through spleen magnetic resonance elastography in pediatric liver fibrosis. World J Gastroenterol. 2019;25:367-377. [PubMed] [DOI] [Full Text] |
| 42. | Chiou FK, Ong C, Low Y, Chiang LW, Phua KB. Non-Invasive Predictors for the First Variceal Hemorrhage in Children With Biliary Atresia After Kasai Portoenterostomy. J Clin Exp Hepatol. 2019;9:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 43. | Yokoyama S, Ishigami M, Honda T, Kuzuya T, Ishizu Y, Ito T, Hirooka Y, Tanaka Y, Tainaka T, Shirota C, Chiba K, Uchida H, Fujishiro M. Spleen stiffness by 2-D shear wave elastography is the most accurate predictor of high-risk esophagogastric varices in children with biliary atresia. Hepatol Res. 2019;49:1162-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 44. | Lampela H, Hukkinen M, Kosola S, Jahnukainen T, Pakarinen MP. Poor performance of noninvasive predictors of esophageal varices during primary prophylaxis surveillance in biliary atresia. J Pediatr Surg. 2020;55:2662-2667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Alam R, Karim ASMB, Rukunuzzaman M, Yasmin A, Hossen K, Benzamin M. Non-endoscopic predictors of esophageal varices in children with chronic liver disease and their utility in resource-constrained countries. Indian J Gastroenterol. 2019;38:310-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 46. | Kumar A, Lal SB, Bhatia A, Das A. Role of noninvasive tools for prediction of clinically evident portal hypertension in children. Eur J Gastroenterol Hepatol. 2020;32:931-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (3)] |
| 47. | Vadlapudi SS, Jagadisan B, Ananthkrishnan R, Narayanaswamy S. Splenic stiffness and platelet count to predict varices needing treatment in pediatric extrahepatic portal vein obstruction. Indian J Gastroenterol. 2020;39:576-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 48. | Lopes JRB, Ferreira AR, Liu PMF, Queiroz TCN, Fagundes EDT, Pimenta JR, Neto JAF, Carvalho SD, Borges MEM, Colin LGF. Non-invasive Predictors of Esophageal Varices With a High Risk of Bleeding in Pediatric Cirrhotic Patients. J Pediatr Gastroenterol Nutr. 2021;72:802-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 49. | Rahmani P, Farahmand F, Heidari G, Sayarifard A. Noninvasive markers for esophageal varices in children with cirrhosis. Clin Exp Pediatr. 2021;64:31-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Yokoyama S, Ishizu Y, Honda T, Imai N, Ito T, Yamamoto K, Muto H, Shirota C, Tainaka T, Sumida W, Makita S, Takada S, Nakagawa Y, Maeda T, Nakamura M, Ishigami M, Uchida H, Kawashima H. A new criterion including the aspartate aminotransferase-to-platelet ratio index and liver and spleen stiffness to rule out varices needing treatment in children with biliary atresia: Modification of the Baveno VII criteria. Hepatol Res. 2024;54:347-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 51. | Diéguez Hernández-Vaquero I, Domènech Tàrrega A, Costa-Roig A, Couselo Jerez M, Vila Carbó JJ. Use of non-invasive scales for detecting esophageal varices in paediatric patients with portal vein thrombosis. Gastroenterol Hepatol. 2024;47:433-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 52. | Jezequel M, El Fayoumi M, Aumar M, Tran L, Saingier C, Behal H, Antoine M, Gottrand F. Splenic stiffness does not predict esophageal varices in children with portal hypertension. J Pediatr Gastroenterol Nutr. 2026;82:156-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 53. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2061] [Article Influence: 515.3] [Reference Citation Analysis (15)] |