Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.114861
Revised: December 8, 2025
Accepted: January 8, 2026
Published online: April 27, 2026
Processing time: 203 Days and 3.6 Hours
Alpha-fetoprotein (AFP), which is re-expressed during hepatocyte regeneration, may serve as a surrogate marker of hepatic recovery in liver failure. We read with great interest the study by Guo et al. This study suggests that dynamic changes in serum AFP possesses potential prognostic value in patients with liver failure undergoing artificial liver support. Despite the limitations of a small sample size and lack of mechanistic validation, the findings nonetheless suggest that AFP holds potential for risk stratification and future individualized treatment. Future studies should emphasize multicenter prospective validation, integration with multidimensional biomarkers, and mechanistic investigation of AFP-related signaling pathways. In this letter, we summarize the established roles of AFP-associated pathways in liver regeneration. Further elucidation of AFP signaling may contribute to more effective strategies for precision artificial liver therapy and improved prognostic assessment.
Core Tip: Guo et al highlighted that serum alpha-fetoprotein (AFP) kinetics may serve as an important prognostic indicator for patients with liver failure and help stratify risk in those receiving artificial liver support therapy. Despite limitations such as a small sample size and a lack of mechanistic validation, dynamic changes in AFP remain a promising complement to traditional prognostic scores. Future research should prioritize multicenter prospective validation and integrate AFP kinetics with multidimensional biomarkers and existing prognostic models, while also exploring regeneration-related signaling such as Wnt/β-catenin, STAT3, and immune pathways.
- Citation: Niu XX, Yang XJ. Letter to the Editor: Serum alpha-fetoprotein dynamics in liver failure - prognostic implications and clinical insights. World J Hepatol 2026; 18(4): 114861
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/114861.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.114861
We read with great interest the recent article by Guo et al[1], which investigated the prognostic value of serum alpha-fetoprotein (AFP) dynamics in patients with liver failure undergoing artificial liver support therapy. Traditionally, AFP has been applied for static assessment in hepatocellular carcinoma (HCC) and certain liver diseases; this study, however, adopted a sequential monitoring approach and highlighted the clinical significance of its dynamic changes. Through retrospective analysis of 194 patients receiving artificial liver support, the authors stratified patients by baseline AFP levels and incorporated serial AFP measurements with multivariate regression analysis. The authors reported that patients with elevated baseline AFP levels and sustained increases after treatment exhibited faster liver function recovery, fewer complications, reduced treatment cycles, and higher 3-month survival rates. The study concluded that AFP kinetics may serve as an effective adjunct indicator for evaluating liver regenerative capacity and predicting prognosis in liver failure. Together, these findings suggest an expanded role for AFP beyond a static diagnostic biomarker, potentially offering additional opportunities for early risk stratification and more individualized management, while calling for further multicenter prospective validation before broader clinical integration.
Globally, liver failure remains a major clinical challenge, with acute-on-chronic liver failure (ACLF) accounting for a substantial proportion and associated with extremely high mortality rates[2,3]. Although artificial liver support systems (such as plasma exchange) have advanced, their therapeutic efficacy varies considerably among patients[4], likely due to differences in hepatic regenerative capacity. Currently, widely used prognostic assessment models (Table 1) include the Child-Turcotte-Pugh (CTP) score, the model for end-stage liver disease (MELD), the Sequential Organ Failure Assessment (SOFA) score, and the Chinese group on the study of severe hepatitis B-ACLF (COSSH-ACLF) score[5,6]. While informative, these models do not adequately reflect the liver’s regenerative potential[7].
| Prognostic tool | Main inputs | Strengths | Limitations | Reflects regenerative potential |
| CTP score | Hepatic encephalopathy, ascites, total bilirubin, albumin, prothrombin time | Simple | Subjective, grading stenosis and lack of corresponding weights for each indicator | Low |
| MELD score | Bilirubin, INR, creatinine | Widely validated; useful for transplant allocation; objective indicators; reliable indicators; easy to obtain | Some clinical factors are not taken into account | Low |
| SOFA | Six organ systems: Respiration; coagulation; liver; cardiovascular; renal, CNS | Simple; continuous monitoring; widely used in ICU | Primarily reflects organ injury, not liver regeneration | Low |
| COSSH-ACLF score | Bilirubin, INR, creatinine, neutrophil count, organ failures | Developed from HBV-ACLF cohort; good regional fit | Limited generalizability; retrospective | Low-moderate |
| CLIF-C ACLF score | Bilirubin, creatinine, INR, organ failures | Tailored for ACLF; good short-term | Requires multiple parameters; complex | Low-moderate |
| AARC score | Bilirubin, INR, lactate, creatinine, encephalopathy | Useful in Asian cohorts; emphasizes metabolic stress | Limited global validation; static | Low-moderate |
| AFP dynamics | Baseline AFP + serial change | Inexpensive; widely available; reflects regeneration | Confounded by etiology, necrosis, malignancy; not yet standardized | High |
Against this background, serum AFP, which is re-expressed during hepatocyte regeneration, has emerged as a potential indicator for assessing hepatic regenerative capacity. However, its clinical application remains challenging. AFP expression is affected by multiple factors, including underlying etiology, extent of hepatocyte necrosis, and concomitant malignancies[8]. Moreover, most current evidence-including the study by Guo et al[1] is derived from single-center retrospective cohorts, limiting the generalizability of the findings. The mechanistic basis for AFP elevation during liver regeneration also remains unclear. It is not yet determined whether AFP functions solely as a surrogate marker of regenerative capacity or as a direct regulatory factor in the regenerative process.
Therefore, in-depth exploration of AFP-related signaling pathways-particularly Wnt/β-catenin and STAT3-and their association with hepatic regeneration is essential for establishing more effective prognostic assessment models and optimizing management and treatment strategies for liver failure.
ACLF is an acute deterioration syndrome that develops on the basis of chronic liver disease or cirrhosis, with short-term mortality rates reaching 50%-90%[9,10]. It is frequently accompanied by multiple organ dysfunction and progresses rapidly. Liver regeneration is an important determinant of survival following acute liver injury. Therefore, early identification of high-risk patients and accurate prognostic assessment are essential for formulating individualized treatment strategies, optimizing interventions, and improving patient outcomes.
Current prognostic assessment models (Table 1), including the CTP score, MELD, SOFA, and COSSH-ACLF score[5,6], primarily focus on organ function and static indicators, limiting their ability to directly capture the liver’s intrinsic regenerative potential. Against this backdrop, Guo et al[1] systematically evaluated AFP kinetics, suggesting its potential to partially address the limitations of existing models. As a low-cost, readily available, and dynamically measurable biomarker, serum AFP shows promise as a valuable supplement to conventional prognostic systems, with the potential to refine risk stratification in ACLF patients.
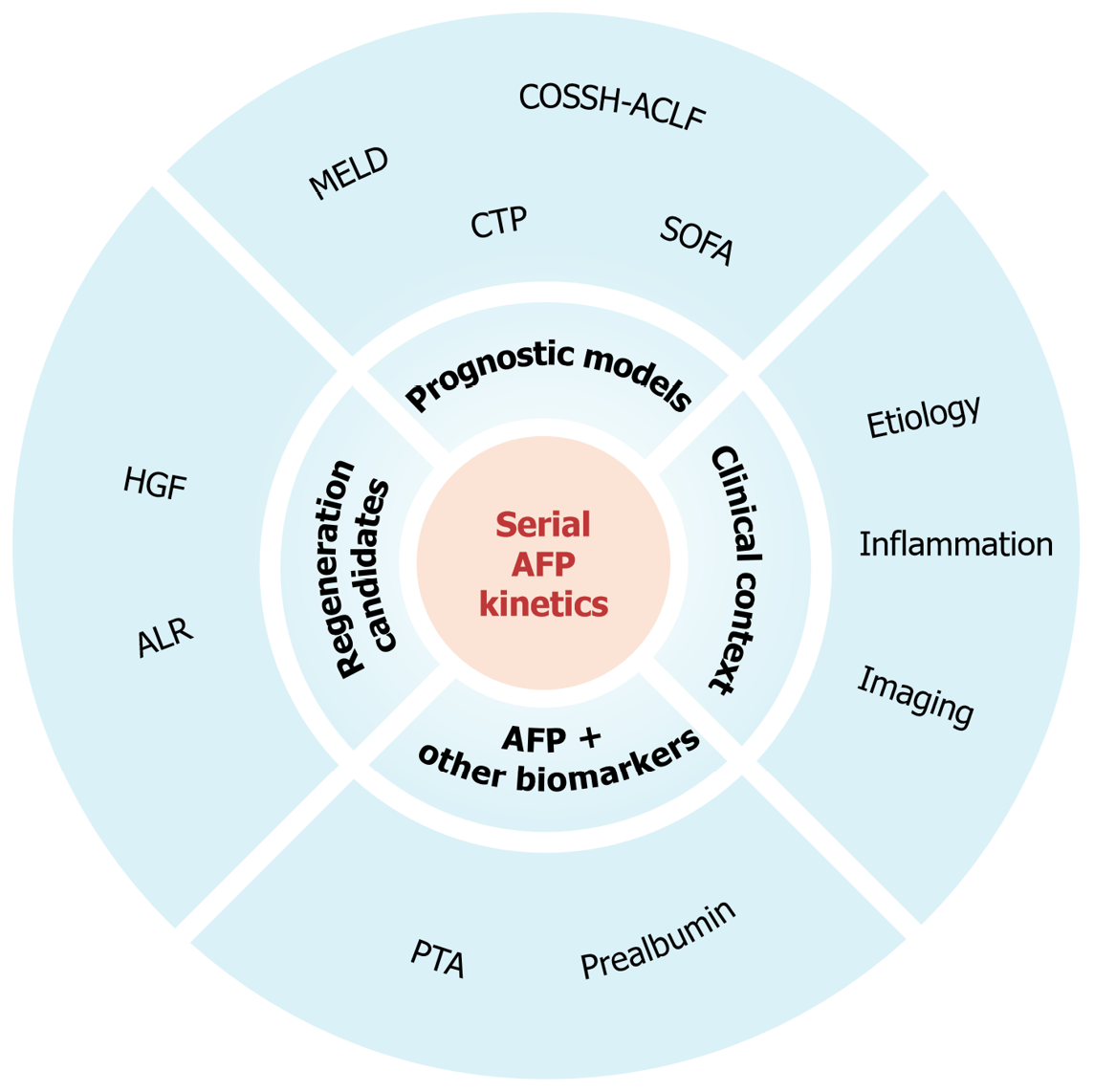
One potential approach is to integrate serial AFP monitoring with existing prognostic assessment models including MELD-based scoring systems, alongside regeneration-related candidates such as hepatocyte growth factor and augmenter of liver regeneration and relevant dynamic clinical indicators[11], while accounting for key clinical context such as etiology, baseline liver status and systemic inflammation. This may be viewed as an operationally feasible complementary integration of serial AFP monitoring with the routine laboratory biomarkers underpinning MELD and related scoring systems such as bilirubin, international normalized ratio, creatinine and serum sodium. Beyond score-based assessment, combined evaluation of AFP with indicators such as prothrombin activity and prealbumin can enhance the accuracy of short-term ACLF prognosis evaluation[12], thus offering a practical example of a potential “AFP + other biomarkers” combination strategy (Figure 1). Importantly, Guo et al[1] explicitly conducted serial AFP assessments during plasma exchange-based artificial liver support therapy, supporting the feasibility of longitudinal AFP monitoring.
Most currently reported comprehensive assessment tools[13,14] incorporating AFP still rely on a single baseline measurement, such as the ALSS-specific artificial liver support system-prognosis model (APM) score[15]. However, the APM score that includes AFP has shown superior short-term prognostic performance compared with MELD and MELD-Na in plasma exchange-treated hepatitis B virus-related acute-on-chronic liver failure patients, supporting the incremental value of AFP beyond traditional scoring systems. From a sequential monitoring perspective, evidence from acute liver failure also provides a kinetics-based rationale, in that early AFP elevation within the first three days as reflected by the AFP ratio correlates with spontaneous survival[16]. In summary, these findings suggest that formal integration of standardized serial AFP monitoring metrics into existing prognostic assessment models represents a direction worthy of investigation in future multicenter prospective validation studies. Nevertheless, a deeper un
In our opinion, the unique value of AFP dynamics lies in linking clinical risk stratification with underlying regenerative mechanisms, which has been underappreciated in previous studies. AFP has traditionally been considered a fetal protein, characterized by high expression during embryonic development and progressive silencing after birth. However, AFP is re-expressed in adults during liver injury and regeneration[11]. For a long time, AFP has primarily been recognized as a tumor marker, particularly as a serum biomarker for HCC. Increasing evidence, however, indicates that AFP not only reflects hepatocyte dedifferentiation and regenerative potential but may also actively participate in regulating regeneration-associated signaling pathways and mediating cell-cell interactions, thereby contributing to liver injury repair[11]. Therefore, in clinical practice, it is necessary to distinguish the range of AFP elevation across different disease contexts.
When interpreting serum AFP, clinicians should consider its absolute level, duration, and trajectory. Compared with non-malignant liver injury, persistent, marked, and progressively rising AFP levels-often reaching the hundreds to even thousands of ng/mL in routine practice-are more suggestive of hepatocarcinogenesis[17,18]. By contrast, in non-malignant conditions such as acute viral hepatitis, drug-induced liver injury, and ACLF, AFP elevations are typically mild to moderate and transient, and may parallel biochemical improvement. Moreover, AFP may also be elevated in physiological or extrahepatic settings including pregnancy and certain germ cell tumours, as well as in rare benign conditions, underscoring the need for etiological and clinical contextualization when interpreting AFP. Accordingly, when evaluating AFP kinetics in patients with liver failure, clinicians should take into account the underlying etiology, baseline cirrhosis, the balance between hepatocyte necrosis and regeneration, viral activity, systemic inflammation, and concomitant malignancy, and interpret AFP dynamics alongside imaging and other relevant biomarkers. This letter focuses on regeneration-related AFP dynamics and their potential to complement conventional prognostic systems, rather than on tumor-associated AFP elevations.
Importantly, AFP elevations linked to dedifferentiation and regeneration are typically more modest than those associated with hepatocarcinogenesis, underscoring the need to interpret AFP levels within the appropriate clinical context.
Research indicates that AFP is involved in multiple signaling pathways and may promote hepatocyte proliferation by activating intracellular signaling cascades. Previous studies have suggested that AFP is associated with the Wnt/β-catenin pathway, which plays a critical role in liver development and regeneration[19,20]. Specifically, in HCC models, AFP can upregulate CMTM6 expression, thereby stabilizing β-catenin by inhibiting its ubiquitination and promoting HCC cell proliferation[21]. Whether this axis operates similarly in non-malignant liver failure remains unclear. Nevertheless, these findings raise the possibility that elevated AFP levels may not only indirectly reflect pathway activity but may also directly accelerate post-injury hepatocyte expansion and repair.
In addition, AFP may interact with STAT3 signaling, a central regulator of liver regeneration and cell survival[11]. STAT3 activation promotes hepatocyte proliferation, exerts anti-apoptotic and antioxidant effects, and enhances regenerative capacity. In a diclofenac-induced liver and kidney injury model, AFP elevation was accompanied by oxidative stress, increased inflammatory cytokines (tumour necrosis factor alpha, interleukin-6), and STAT3 activation, suggesting that AFP may reflect tissue injury associated with inflammation and immune dysregulation[22]. Further research showed that co-administration of honey flower extract mitigates these effects by restoring antioxidant defenses, suppressing STAT3-mediated inflammatory signaling, and improving tissue integrity[22]. This pattern suggests that pairing AFP kinetics with STAT3-related inflammatory and redox markers may improve future risk stratification by helping to distinguish predominantly reversible inflammatory injury from compromised regenerative capacity.
Notably, AFP may also influence liver regeneration through immune regulation. Emerging evidence suggests that AFP may participate in immunoregulatory processes during liver injury and regeneration. During acute injury-induced regeneration, a subset of hepatocytes transiently upregulates fetal genes such as AFP to reprogram and promote regeneration. This process involves dynamic recruitment of immune cells, particularly myeloid cells, and matrix remodeling, suggesting that AFP functions not only as a regenerative marker but may also shape the immune microenvironment to support liver repair[23]. In liver failure, characterized by pronounced immune injury and systemic inflammation, the immunoregulatory effects of AFP may directly influence the balance between injury and repair, fostering an immune milieu conducive to regeneration. However, the specific cell types and signaling mechanisms underlying these effects remain to be elucidated.
Guo et al’s study[1] suggests that AFP offers a complementary dimension for assessing hepatic regenerative capacity, functioning both as a dynamic biomarker and potentially participating in regeneration-related signaling pathways, thereby addressing a limitation of traditional scoring systems in evaluating “regenerative potential”. Although preliminary mechanistic evidence exists, several critical questions remain unresolved. For example, does elevated AFP directly drive regeneration, or does it serve merely as a surrogate indicator for other mechanisms, such as progenitor cell activation or cytokine-mediated repair? In addition, AFP expression patterns and functional roles may vary across different etiologies, including viral hepatitis, alcoholic liver disease, and autoimmune liver injury. Addressing these questions will require comprehensive studies that integrate clinical observations with mechanistic investigations.
Future research should aim to systematically validate the relationship between AFP dynamics and hepatic regenerative capacity across diverse etiologies and clinical settings. Combining in vivo and in vitro liver injury models with omics-based approaches may clarify the AFP-associated signaling networks that underlie liver repair. If AFP is confirmed as an active regulator rather than merely a passive biomarker, it could emerge as a candidate therapeutic target to enhance regeneration. Clinically, integrating serial AFP monitoring into established prognostic frameworks (e.g., MELD) or developing composite models alongside multidimensional biomarkers may further refine risk stratification and help inform more individualized artificial liver strategies. Such an approach would leverage AFP both for prognostic evaluation and as a foundation for targeted intervention strategies. These steps may help lay the groundwork for a more precision-oriented approach to artificial liver support, pending standardized assays and multicenter prospective validation.
Guo et al[1] demonstrated that AFP kinetics provide a valuable adjunct for prognostic assessment in liver failure. AFP currently functions more as a preliminary adjunct biomarker, and robust multicenter validation is required before it can be incorporated into routine decision-making. Future studies should further investigate its involvement in Wnt/β-catenin, STAT3, and immune signaling pathways. These insights may ultimately advance AFP from an “adjunct biomarker” to a “mechanistic regulator”, enabling more comprehensive and dynamic evaluation of regenerative capacity. Such progress could support the development of precision artificial liver therapy and personalized intervention strategies.
| 1. | Guo WB, Wang LY, Guo XJ, Yang J, Li W, Shen FY, Li YT, Yang JH, Tai WL. Prognostic value of serum alpha-fetoprotein kinetics in liver failure on artificial liver support. World J Gastroenterol. 2025;31:111914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 2. | Aggarwal A, Biswas S, Arora U, Vaishnav M, Shenoy A, Swaroop S, Agarwal A, Elhence A, Kumar R, Goel A, Shalimar. Definitions, Etiologies, and Outcomes of Acute on Chronic Liver Failure: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2024;22:2199-2210.e25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 3. | Mezzano G, Juanola A, Cardenas A, Mezey E, Hamilton JP, Pose E, Graupera I, Ginès P, Solà E, Hernaez R. Global burden of disease: acute-on-chronic liver failure, a systematic review and meta-analysis. Gut. 2022;71:148-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 174] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 4. | Beran A, Mohamed MFH, Shaear M, Nayfeh T, Mhanna M, Srour O, Nawras M, Mentrose JA, Assaly R, Kubal CA, Ghabril MS, Hernaez R, Patidar KR. Plasma exchange for acute and acute-on-chronic liver failure: A systematic review and meta-analysis. Liver Transpl. 2024;30:127-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 5. | Cai X, Chong Y, Gan W, Li X. Progress on clinical prognosis assessment in liver failure. Liver Res. 2023;7:101-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Goyes D, Trivedi HD, Curry MP. Prognostic Models in Acute-on-Chronic Liver Failure. Clin Liver Dis. 2023;27:681-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 7. | Luo J, Li J, Li P, Liang X, Hassan HM, Moreau R, Li J. Acute-on-chronic liver failure: far to go-a review. Crit Care. 2023;27:259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 8. | Yeo YH, Lee YT, Tseng HR, Zhu Y, You S, Agopian VG, Yang JD. Alpha-fetoprotein: Past, present, and future. Hepatol Commun. 2024;8:e0422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 62] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 9. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on acute-on-chronic liver failure. J Hepatol. 2023;79:461-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 252] [Article Influence: 84.0] [Reference Citation Analysis (1)] |
| 10. | Zaccherini G, Weiss E, Moreau R. Acute-on-chronic liver failure: Definitions, pathophysiology and principles of treatment. JHEP Rep. 2021;3:100176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 127] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 11. | Liu Q, Wang S, Fu J, Chen Y, Xu J, Wei W, Song H, Zhao X, Wang H. Liver regeneration after injury: Mechanisms, cellular interactions and therapeutic innovations. Clin Transl Med. 2024;14:e1812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 12. | Duan RX, Liu L, Wang Y, Wu WM. Prognostic value of combined detection of alpha-fetoprotein, plasma prothrombin activity, and serum prealbumin in acute-on-chronic liver failure. World J Gastrointest Surg. 2025;17:99531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 13. | Peng H, Zhang Q, Luo L, Lei S, Xiong T, Long L, Xiong Y, Zhang L, Zheng J, Luo X. A prognostic model of acute-on-chronic liver failure based on sarcopenia. Hepatol Int. 2022;16:964-972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 14. | Li C, Hu H, Bai C, Xu H, Liu L, Tang S. Alpha-fetoprotein and APRI as predictive markers for patients with Type C hepatitis B-related acute-on-chronic liver failure: a retrospective study. BMC Gastroenterol. 2024;24:191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 15. | Xie Z, Violetta L, Chen E, Huang K, Wu D, Xu X, Ouyang X, Zhao Y, Li L. A prognostic model for hepatitis B acute-on-chronic liver failure patients treated using a plasma exchange-centered liver support system. J Clin Apher. 2020;35:94-103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Qi R, Wang X, Kuang Z, Shang X, Lin F, Chang D, Mu J. Alpha-fetoprotein and carbohydrate antigen 19-9 as prognostic biomarkers in acute liver failure: A retrospective study. J Int Med Res. 2025;53:3000605251332808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Turshudzhyan A, Wu GY. Persistently Rising Alpha-fetoprotein in the Diagnosis of Hepatocellular Carcinoma: A Review. J Clin Transl Hepatol. 2022;10:159-163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 18. | Hanif H, Ali MJ, Susheela AT, Khan IW, Luna-Cuadros MA, Khan MM, Lau DT. Update on the applications and limitations of alpha-fetoprotein for hepatocellular carcinoma. World J Gastroenterol. 2022;28:216-229. [PubMed] [DOI] [Full Text] |
| 19. | Russell JO, Monga SP. Wnt/β-Catenin Signaling in Liver Development, Homeostasis, and Pathobiology. Annu Rev Pathol. 2018;13:351-378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 389] [Article Influence: 48.6] [Reference Citation Analysis (0)] |
| 20. | Huang R, Zhang X, Gracia-Sancho J, Xie WF. Liver regeneration: Cellular origin and molecular mechanisms. Liver Int. 2022;42:1486-1495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 21. | Liu Q, Wang J, Guo Z, Zhang H, Zhou Y, Wang P, Li T, Lu W, Liu F, Han W. CMTM6 promotes hepatocellular carcinoma progression through stabilizing β-catenin. Cancer Lett. 2024;583:216585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 22. | Salama AR, Ashoura NR, Esmail KA, Tohamy HG, Shukry M, Alotaibi BS, Aboushouk AA. Melissa officinalis Attenuates Diclofenac-Induced Hepatorenal Toxicity via AMPK/SIRT1 and STAT3/Nrf2 Pathways. J Appl Toxicol. 2025;45:1649-1663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 23. | Ben-Moshe S, Veg T, Manco R, Dan S, Papinutti D, Lifshitz A, Kolodziejczyk AA, Bahar Halpern K, Elinav E, Itzkovitz S. The spatiotemporal program of zonal liver regeneration following acute injury. Cell Stem Cell. 2022;29:973-989.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 150] [Article Influence: 37.5] [Reference Citation Analysis (0)] |