Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.114793
Revised: November 25, 2025
Accepted: February 10, 2026
Published online: April 27, 2026
Processing time: 205 Days and 13.4 Hours
Hepatology and gastroenterology encompass a wide spectrum of acute and chronic conditions, many of which require urgent decision-making, rendering the concept of time-sensitive intervention crucial. Certain diagnostic and therapeutic actions must be undertaken within specific time windows, as even short delays may result in irreversible organ damage, systemic decompensation, or death. These interventions are not only lifesaving but also directly influence long-term prognosis, transplant candidacy, and quality of life. Time-sensitive interventions are relevant across diverse clinical contexts as described in the following ex
Core Tip: Practitioners in hepato-gastroenterology should respect the clock ticking during the management (diagnosis and treatment) of different liver and gastrointestinal diseases. The concept of time-sensitive management is not clearly defined in current practice guidelines. Clinicians, therefore, need a consolidated one-file resource - such as this review - which gathers the available evidence on interventions where timing is critical. Delays in these diagnostic or therapeutic steps can lead to substantial adverse outcomes, including irreversible organ injury, systemic decompensation, prolonged hospitalization, long-term disability, reduced transplant eligibility, diminished quality of life, and even mortality.
- Citation: Said EM, Soliman HH, Gabal HMR, Emara MH, Fouad Y, Elzahaby A, Anees M, Zaky S, Sakr MA. Time sensitive managements in hepato-gastroenterology. World J Hepatol 2026; 18(4): 114793
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/114793.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.114793
Patient-related factors such as age, immune status, and comorbidities play a major role in determining morbidity and mortality in liver and gastrointestinal (GI) diseases. Equally important are disease characteristics and their clinical management, both of which fundamentally influence the prognosis. Clinical management frequently requires critical diagnostic and therapeutic interventions that must be performed within strict time limits, whether the patients are hospitalized or not. Delays in implementing these measures can result in irreversible damage, making them true “time-sensitive interventions” where the clock is always ticking. Failure to make the right decision at the perfect time could ultimately jeopardize the patient’s health. Hence, there is a need for shedding light on the most serious time-sensitive interventions and decisions in diseases affecting the liver, biliary system, and GI tract, providing clinicians with clear guidance for prompt and effective action to reduce harmful delays and achieve quality in health care.
In hepato-gastroenterology, several diagnostic procedures are time sensitive, including diagnostic paracentesis for suspected spontaneous bacterial peritonitis (SBP), endoscopy for acute GI bleeding, and serum amylase measurement in suspected acute pancreatitis, among others. Likewise, therapeutic interventions demand strict timing, such as post-exposure administration of hepatitis B immunoglobulin (HBIG) in non-immune individuals, prompt parenteral antibiotics for SBP, intravenous (IV) albumin infusion for hepatorenal syndrome (HRS) prophylaxis in SBP, vasopressor therapy for active variceal bleeding, and urgent endoscopic retrograde cholangio-pancreatography (ERCP) for acute gallstone pancreatitis. Despite the critical importance of these interventions, the available literature remains fragmented, with no single source summarizing the evidence. This review, therefore, aims to consolidate the data on time-sensitive diagnostic and therapeutic interventions in hepato-gastroenterology, highlight the importance of each, and emphasize the optimal time frame to guide clinicians in improving patient outcomes.
This is a narrative review aiming to synthesize current evidence, guideline recommendations, and expert perspectives on clinical scenarios in hepatology and gastroenterology where timely diagnostic or therapeutic interventions influence patient outcomes. A narrative approach was chosen over a systematic to allow broad coverage of heterogeneous topics and integration of multiple types of evidence.
The objectives of this review were to: Identify hepato-gastroenterological conditions in which the timing of diagnosis or treatment is critical, and outline the precise time-sensitive diagnostic steps and therapeutic strategies for each. Summarize the available evidence supporting the impact of timely intervention on morbidity, mortality, hospital stay, long-term prognosis, and transplant candidacy. Highlight knowledge gaps, controversies, and priorities for future research.
A comprehensive literature search up to August 2025 was conducted in the following electronic databases: PubMed/MEDLINE, Scopus, Web of Science, Google Scholar for grey literature, key citations, and guidelines. Searches used combinations of keywords and MeSH terms related to timing and urgency of care, including: “time-sensitive”, “timing”, “urgent management”, “delayed diagnosis”, “early intervention”, “hepatology”, “gastroenterology”, “spontaneous bacterial peritonitis”, “mesenteric ischemia”, “acute severe ulcerative colitis”, “acute liver failure”, “DILI”, “acetaminophen toxicity”, “GI bleeding”, “endoscopy timing”, “mesenteric venous thrombosis”, “HBV immunoglobulin”, “post-exposure prophylaxis”, “screening endoscopy”, “endoscopic surveillance”. The focus was on articles published in the last 20 years, although older landmark studies and guidelines were included when foundational.
Inclusion criteria: Studies on adult populations, addressing the timing of diagnostic or therapeutic interventions in hepatology or gastroenterology. Clinical trials, cohort and case-control studies, case series, meta-analyses, narrative reviews, and international guidelines.
Exclusion criteria: Editorials, commentaries, or expert opinions without supporting evidence. Non-English articles, unless providing unique or critical data.
Titles and abstracts were initially screened to identify relevant studies. Full-text articles were assessed for eligibility based on the inclusion criteria. Additional articles were identified through citation tracing and review of references from key guidelines (snowballing technique).
For each identified condition [e.g., drug-induced liver injury, mesenteric ischemia, acute severe ulcerative colitis (UC), acute liver failure (ALF), GI bleeding], the following data elements were extracted: (1) Identification of the time-sensitive intervention; (2) Description of procedure, define the name, dose, route of administration, and possible adverse events of the used medications; (3) Strength and nature of evidence supporting the recommended timing; (4) Reported optimal timing window, when specified; (5) Clinical and prognostic consequences of delayed intervention; (6) Areas of uncertainty or debate; and (7) Data extraction was performed manually by the reviewer to allow integration of heterogeneous evidence.
A qualitative synthesis approach was used. Findings were organized thematically into: (1) Time-sensitive diagnostic interventions; (2) Time-sensitive therapeutic interventions; and (3) Cross-cutting concepts such as rapid pathophysiologic deterioration, “golden hours”, and the impact of system delays. Evidence across topics was compared to identify common principles, such as early recognition, expedited diagnostic pathways, and the impact of timing on organ salvage and survival.
Because this is a narrative review, no formal systematic risk-of-bias tools were applied. Instead, a qualitative appraisal considered: (1) Study design and hierarchy of evidence; (2) Sample size and consistency of results; (3) Alignment with international guidelines; and (4) Clinical relevance and applicability to the contemporary practice.
This review did not involve human participants or identifiable data and therefore did not require ethical approval.
ALF is a rare, life-threatening, but potentially reversible condition characterized by the rapid loss of hepatic function in patients without preexisting liver disease[1]. While encephalopathy and coagulopathy are the hallmark features, ex
One of the major pathophysiologic derangements in ALF is cerebral edema, and it is a major risk of mortality risk among this subgroup of patients. Cerebral edema is reported among 80% of patients with grade IV hepatic encephalopathy and can lead to brain herniation and death[1-3]. The underlying mechanism of cerebral edema is related to the acute elevation in blood ammonia level following the loss of detoxification function of the failing liver[1-3]. Ammonia leads to astrocyte swelling and dysfunction, and hence cerebral edema. There is a direct relation between the degree of rise in arterial ammonia level and the risk of cerebral edema. The acute nature of the condition impairs the body’s ability to create osmolar buffers to compensate for the sharp rises and floods of toxins, including ammonia, a mechanism which is not totally impaired among patients with chronic liver failure, where the brain had enough time to create these buffers, and hence the likelihood of having severe cerebral edema is low[4-7].
The overarching management goal for ALF patients is to prevent death from cerebral edema and multiple organ failure, thereby allowing sufficient time for spontaneous liver regeneration or ELT. Although ammonia is only one of several endogenous toxins implicated in cerebral edema, it serves as a useful surrogate marker, and severe hyperammonemia (> 150 μmol/L) is strongly associated with neurological injury[8]. Management requires prompt evaluation for reversible causes and early transfer to a transplant center.
While the relationship between hyperammonemia and neurological complications varies among patients, the onset of high-grade hepatic encephalopathy requiring intubation is considered an appropriate threshold for initiating continuous renal replacement therapy (CRRT)[8]. Blood purification is a central neuroprotective strategy, with evidence showing that CRRT can effectively reduce or prevent severe hyperammonemia[8]. For this reason, CRRT is now regarded as the standard of care for neuroprotection in ALF, even before conventional criteria for acute kidney injury are met[9]. In contrast, intermittent hemodialysis is not recommended due to hemodynamic instability and the risk of exacerbating intracranial hypertension (ICP)[10]. Plasma exchange may be performed in parallel with CRRT via separate vascular access or sequentially, depending on patient status and institutional expertise[11].
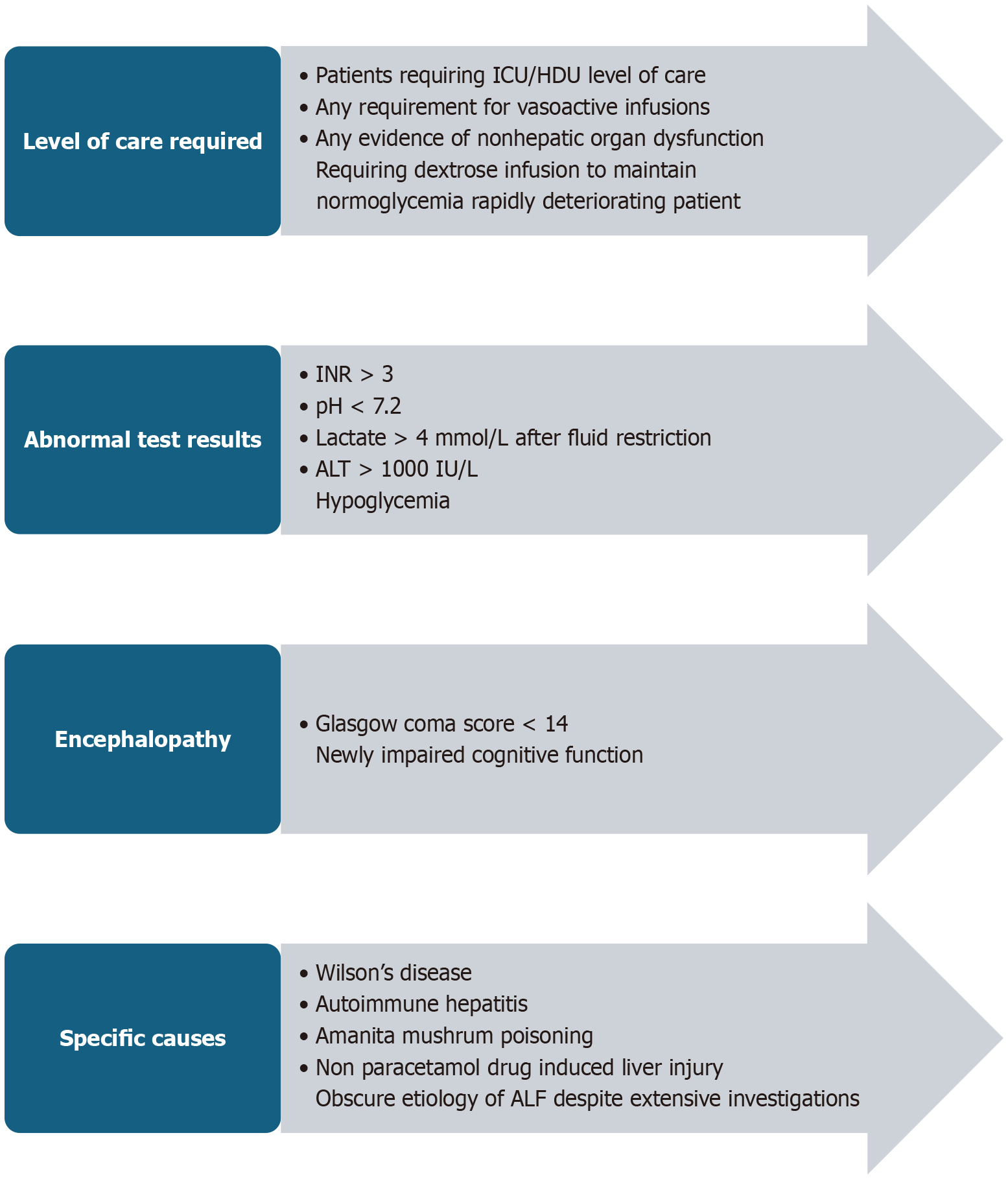
The rapid initiation of multimodal neuroprotective measures of hemo(dia)filtration, mild hyperventilation, and mild hypothermia (referred to collectively as “quadruple-H therapy”) are key management priority to prevent death from cerebral oedema in ALF[12]. For patients with severe hyperammonemia or refractory shock, high or standard volume plasma exchange may provide additional benefits through removal of inflammatory mediators and other endogenous toxins. Daily treatments of plasma exchange have been prevent improve transplant-free survival[13]. Prompt treatment of ALF raises the transplant-free survival to 55%-60%. According to Reuben et al[14], almost all interventions recommended for ALF can be provided at nontransplant centers, but patients can deteriorate rapidly, and ELT can be lifesaving for those who are eligible. The decision of ELT necessitates combining the established criteria and local multidisciplinary expertise (Figure 1)[14].
Airway and breathing: Intubate if grade ≥ III encephalopathy. Prevent aspiration, enable sedation, and control ICP.
Circulation: Aggressive IV fluids; use norepinephrine as first-line vasopressor if hypotensive. Avoid fluid overload (risk of cerebral edema).
Monitoring: Admit to the intensive care unit (ICU) with invasive arterial blood pressure and hourly neurological checks. Early referral to a transplant center.
Diagnostic workup: The work should explore the history with a focus on exposure to drugs and herbal remedies, risk of exposure to viral hepatitis, autoimmune and metabolic disorders, among others. Pattern of liver enzyme elevation and aspartate aminotransferase/alanine transaminase ratio with levels ≥ 3000 IU/L may point towards viral hepatitis, acetaminophen overdose, or liver ischemia. A comprehensive laboratory workup should be sent that evaluates the differential diagnosis and prepares for the prospect of a liver transplant evaluation. These include complete liver and kidney functions, blood gases, ammonia level, viral and immune markers, metabolic profile, and serum drug level, among others. Evaluation of the prognosis by frequent assessment and calculation of different scores, e.g., the model of end-stage liver disease (MELD) imaging, is very crucial for planning liver transplantation. Liver biopsy is indicated in very limited situations when the diagnosis is unclear and should be done through a transjugular approach.
N-acetylcysteine: (1) Indication: All paracetamol (acetaminophen) ALF. Potential benefit in all non-paracetamol ALF as well. Although using N-acetylcysteine (NAC) in non-acetaminophen ALF is recommended by some evidence[4,5], other authors and international guidelines[6,7] remain cautious due to limited randomized controlled trials data. Therefore, NAC may be considered in selected cases of non-acetaminophen ALF given potential survival benefits, although high-quality evidence is limited; (2) Timing: Start immediately, do not wait for toxicology confirmation; and (3) Protocol: IV NAC 150 mg/kg over 1 hour → 50 mg/kg over 4 hours → 100 mg/kg over 16 hours (repeat as guided by response).
Treat the precipitating cause: (1) Infections: Start broad-spectrum antibiotics empirically (high infection risk in ALF); (2) Herpes simplex virus hepatitis: Start IV acyclovir if suspected; (3) Autoimmune hepatitis: Corticosteroids in selected cases; and (4) Mushroom ingestion: Silibinin or penicillin G if available.
Head elevation 30° and maintain venous drainage. Avoid hyponatremia; keep serum Na 145-150 mmol/L.
Osmotherapy: Mannitol 0.5-1 g/kg IV bolus if ICP rises. Hypertonic saline if refractory.
Sedation/paralysis: Propofol preferred; avoid benzodiazepines.
Ventilation: Mild hyperventilation only as a bridge (not long-term therapy).
ICP monitoring: Consider in advanced encephalopathy if listed for transplant.
Maintain mean arterial pressure ≥ 65 mmHg (norepinephrine ± vasopressin).
Glucose control: IV dextrose infusion; avoid hypoglycemia. Correct metabolic acidosis only if pH < 7.1 with instability.
Renal support: CRRT preferred over intermittent dialysis (better hemodynamic stability + ammonia clearance).
Do not correct international normalized ratio (INR) routinely unless: (1) Active bleeding; (2) Before invasive procedures; (3) FFP, platelets, cryoprecipitate only if clinically bleeding; and (4) Avoid prophylactic correction (can mask severity and interfere with transplant assessment).
Empiric antibiotics ± antifungals recommended in: (1) Advanced encephalopathy; (2) Invasive line use; and (3) Transplant candidates. Perform daily cultures and maintain strict aseptic technique.
Refer early to the transplant center. Apply King’s College Criteria[15].
Paracetamol-induced ALF: (1) The pH < 7.3 (irrespective of encephalopathy); or (2) INR > 6.5 + Cr > 300 μmol/L + grade III-IV encephalopathy.
Non-paracetamol ALF: INR > 6.5 or any 3 of: (1) Age < 10 or age > 40; (2) Etiology: Non-A, non-B, halothane, idiosyncratic drugs; (3) Jaundice > 7 days before encephalopathy; (4) INR > 3.5; and (5) Bilirubin > 300 μmol/L.
Although the King’s College Criteria is widely used. Yet recently, a multimodal approach incorporating King’s College Criteria, MELD scores, ammonia levels, and clinical trajectory has been proposed in determining candidacy for ELT. Current prognostic models have good accuracy to identify patients who will die without liver transplantation, but are not able to identify those in whom transplantation is futile. New prognostic markers to select patients for transplantation are still in the pipeline[5].
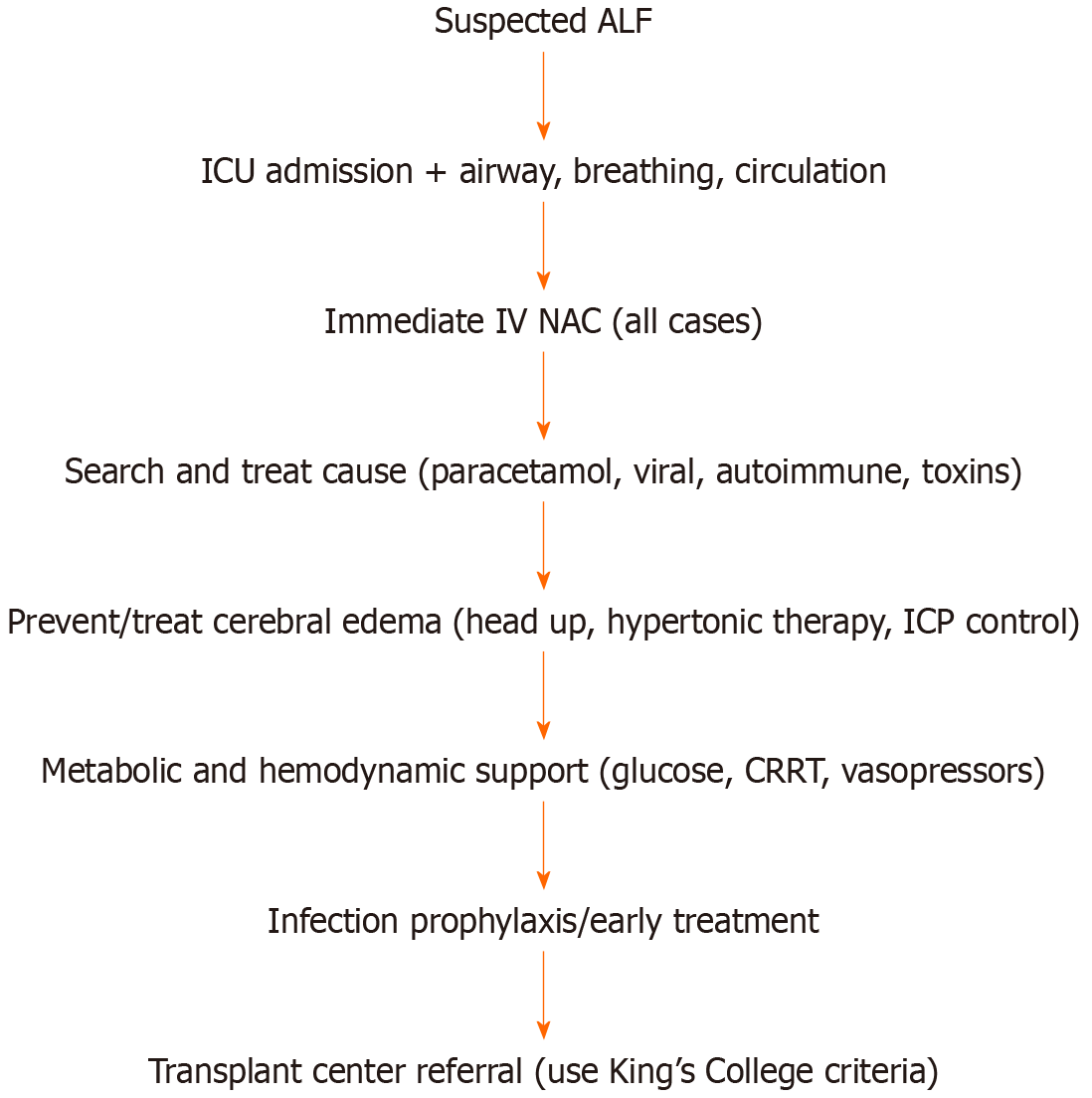
The causes of ALF are diverse, and determining the etiology is essential for guiding targeted therapy. A timely and accurate diagnosis is therefore critical to enable early treatment, maximize the chances of transplant-free survival, or ensure prompt ELT when necessary[16] as shown in Figure 2.
The most common drug-induced liver injuries (DILI) that require urgent recognition[17] include.
Although acetaminophen is generally considered safe (up to 4 g/day), its overdose induces both fatal and nonfatal hepatic necrosis[18]. Acetaminophen is the leading cause of ALF in the United States, responsible for about 20% of liver transplants[1,17]. Besides, patients with alcohol use disorders, patients at extremes of age, underweight, and those on concurrent interacting medications such as carbamazepine, phenytoin, or isoniazid (INH) are vulnerable to suffer hepatic toxicity with repeated therapeutic or mildly excessive dosing[19].
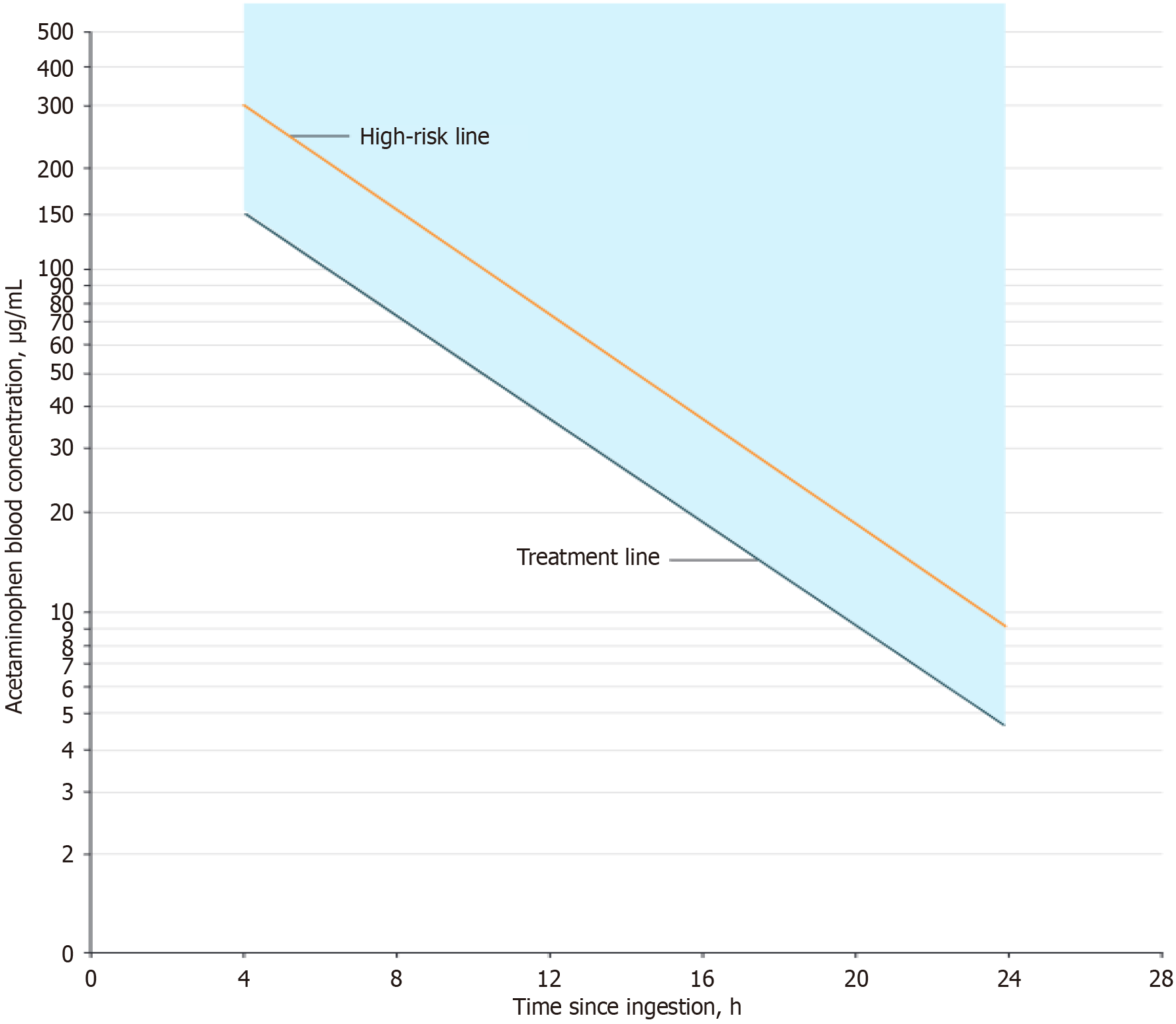
Time-sensitive management: Management of paracetamol overdose involves gastric decontamination and prompt antidote administration. A Cochrane review showed that activated charcoal and gastric lavage can reduce paracetamol absorption if given within 1-2 hours of ingestion. NAC is the preferred antidote, as it reduces liver injury and has fewer side effects compared to alternatives like dimercaprol and cysteamine[20]. NAC is the gold-standard antidote for acetaminophen toxicity. It acts as a glutathione precursor, detoxifying N-acetyl-p-benzoquinoneimine, enhancing hepatic perfusion, scavenging free radicals, and improving mitochondrial energy production. NAC provides the greatest benefit when given within 8 hours of ingestion, but may still offer benefit even if delayed, as shown in a single late-reversal case[21]. NAC should be administered whenever acetaminophen levels are potentially toxic on the Rumack-Matthew nomogram (Figure 3)[22]. The original Rumack-Matthew nomogram has a reference line that begins at 200 μg/mL at 4 hours post-ingestion[23], then is reduced to 150 μg/mL to enhance patient safety[24]. with high-risk line (to identify patients at elevated risk of hepatic injury) at 300 μg/mL at 4 hours[25]. Hence, NAC should be started if acetaminophen levels (measured 4-24 hours post-ingestion) fall on or above the treatment line. Higher doses are often used when levels exceed the high-risk line, but there is no standardized evidence-based adjustment protocol.
INH (isonicotinyl hydrazide/isonicotinic acid hydrazine) is the second most common cause of DILI. It is presented as mild liver injury or symptomatic hepatitis (with 10% mortality)[26]. Risk increases with age, alcoholism, previous INH intolerance, concurrent liver disease, or cytochrome P450-inducing drugs[27]. Typically, INH hepatitis presented within three months of initiating therapy, though it was reported up to 14 months after[28]. Management requires early recognition and prompt discontinuation of INH if bilirubin ≥ 3 mg/dL or transaminases > 5 × ULN, as delayed withdrawal worsens outcomes[27]. Most patients (about 80%) may tolerate cautious reintroduction, though close monitoring is essential[29]. Severe cases may require transplantation[30]. NAC may be considered in selected cases of non-acetaminophen ALF given potential survival benefits[31], although high-quality evidence is limited.
Time-sensitive aspects: (1) Monitoring: Frequent ALT checks, especially in the first 3 months; (2) Immediate discontinuation: Crucial to prevent progression to liver failure; and (3) Reintroduction: Only with close monitoring if clinically mandated (Table 1).
| Drug/class | Mechanism | Time sensitivity | Key intervention |
| Acetaminophen | Direct toxin (NAPQI) | < 8-12 hours for NAC efficacy | NAC, early levels, transplant if needed |
| Isoniazid | Hepatocellular (immune) | Days-weeks; need early stop | Monitor LFTs, stop the drug promptly |
| Halothane | Immune hepatitis | Re-exposure fatal | Avoid re-exposure, early stop |
| Amiodarone (IV) | Direct toxic | Hours-days | Discontinue if ALT/AST rise |
| Valproic acid | Mitochondrial dysfunction | Hours-days | Stop the drug, consider L-carnitine |
| Methotrexate (acute) | Overdose, accumulation | Hours (folinic acid critical) | Leucovorin rescue |
| DRESS-related drugs | Immune-mediated | Days; worsening if delayed stop | Stop all suspect drugs urgently |
| Herbal toxins | Idiosyncratic or direct | Hours-days | Stop supplement, list in history |
Prolonged use of amiodarone may induce asymptomatic transaminase elevation or, less commonly, steatohepatitis, cirrhosis, and liver failure. The patient should be followed every 6 months and consider dose reduction or drug substitution if aminotransferase elevation exceeds twice the upper normal limit[32]. Occasionally, IV amiodarone may induce fatal hepatocellular injury. Usually manifests within 24 hours of administration but is reported later[33]. The mechanism of IV amiodarone DILI is not certain. Possible explanations include toxicity from the infusion solvent polysorbate 80[34] or dose-related hepatotoxicity, as hyperacute hepatitis has also been observed with high oral doses[35]. Management requires prompt discontinuation of IV amiodarone and avoidance of reintroduction, which is usually followed by gradual improvement in liver function[33]. If continued therapy is essential, it could be given orally with a reduced dosage (< 200 mg/day) while maintaining vigilant monitoring of liver function[36].
Time-sensitive decisions: So, during the first 24 hours of IV amiodarone, close monitoring for hepatic toxicity is essential, as injury may be rapidly fatal. In contrast, with long-term oral therapy, liver enzymes can be monitored every 6 months since drug-induced steatohepatitis develops more slowly.
Halothane is no longer commercially available in North America, Europe, and many other countries due to its adverse effects (particularly the possibility of halothane hepatitis). However, due to its low cost, halothane is still used for both induction and maintenance of general anesthesia in resource-challenged countries, particularly in children[37]. Its liver toxicity has two patterns: A mild, self-limited postoperative hepatitis occurring in 20%-30% of patients, with spontaneous resolution in two weeks[38], and a rarer, unpredictable halothane hepatitis that develops within 2 days to 3 weeks, often presenting as acute hepatitis and sometimes progressing to fatal liver failure. Eosinophilia occurs in about 40% of cases, suggesting an immunoallergic mechanism[39]. The incidence is about 1 in 6000-15000 after a single exposure, but rises to 1 in 1000 with repeated exposures, particularly if given within 28 days[40]. Risk is highest in patients over 40 years, women, obese individuals, and those with pre-existing liver disease or genetic susceptibility. NAC may be used in management[37].
Time-sensitive actions: Monitoring of the patient postoperatively is important for early diagnosis. Despite the drug discontinuation occurring already before diagnosis it is still time sensitive for prevention of re-exposure. NAC may be used.
In long-term low-dose methotrexate (MTX) therapy, early transient transaminase elevations are common, while prolonged use may lead to hepatic steatosis, fibrosis, cirrhosis, and even hepatocellular carcinoma[41]. High-dose MTX (≥ 500 mg/m2) is associated with transient acute hepatitis in 60%-80% of patients[42]. Monitoring liver enzymes is therefore time-sensitive, with dose reduction or substitution needed based on hepatic pathology[41]. Although no standardized guidelines exist for hepatic dose adjustment, a 25% reduction is advised in patients with third-space fluid (e.g., ascites), and discontinuation is recommended if bilirubin exceeds 3-5 mg/dL or if transaminase elevation persists for two cycles[43]. Folinic acid (leucovorin) rescue, administered 24 hours after high-dose MTX, is essential to preserve antimitotic efficacy while protecting bone marrow, GI mucosa, and liver folate stores[42].
Time-sensitive actions: Both cumulative dose and acute high dose can induce hepatic injury; folinic acid should start within 24 hours of MTX. Monitor liver and renal function closely.
Valproic acid (VPA) commonly causes hyperammonemia (> 80 mcg/dL), often asymptomatic, though it may progress to valproate-related hyperammonemic encephalopathy, which can occur even with normal liver biochemical tests and therapeutic serum concentrations[44]. Onset may be immediate with loading or insidious with chronic therapy. Acute or chronic VPA use can induce reversible, dose-related hepatocellular injury with mild aminotransferase elevations[45,46]. Rarely, idiosyncratic reactions resembling Reye syndrome led to fulminant hepatic failure, often within the first six months and without preceding enzyme elevation - necessitating close monitoring, especially in infants and those on multiple antiseizure drugs[46]. VPA depletes carnitine, contributing to hepatic manifestations[47]. L-carnitine supplementation (100 mg/kg IV, max 6 g, then 50 mg/kg IV q8h, max 3 g) is recommended in toxicity[48]. Diagnosis of acute poisoning relies on elevated serum VPA, while chronic toxicity may present as hepatocellular injury, hyperammonemia without injury, or valproate-related hyperammonemic encephalopathy. Delayed discontinuation in symptomatic patients can be fatal[49].
Time-sensitive actions: Patients ingesting > 200 mg/kg or with serum levels > 180 mcg/mL require observation, even if asymptomatic[49]. Observation lasts 6 hours for immediate-release and 12 hours for extended-release forms, with serial levels every 2-4 hours to detect delayed peaks[50]. Activated charcoal (1 g/kg, max 50 g) is effective if given within 1 hour[43]. In severe poisoning, intermittent hemodialysis is the preferred extracorporeal removal method[51].
Drug reaction with eosinophilia and systemic symptoms is a rare but life-threatening hypersensitivity reaction occurring 2-8 weeks after exposure to drugs such as allopurinol, sulfonamides, phenytoin, vancomycin, minocycline, and carbamazepine[52]. It is characterized by rash, fever, lymphadenopathy, hematologic abnormalities, and multi-organ involvement, with liver affection in 90% of cases[53]. Most patients recover within weeks to months after drug withdrawal, though mortality ranges between 2% and 10%. Importantly, Drug reaction with eosinophilia and systemic symptoms can trigger long-term immune hyperactivation, leading to polysensitization and cross-reactivity with unrelated drugs, hence additional medications (especially antibiotics) should be avoided during the acute phase[54].
Time-sensitive actions: Management is highly time-sensitive, requiring early identification and immediate withdrawal of the culprit drug to prevent progression of immune activation and multi-organ failure.
High-risk supplements are defined as supplements that have the highest likelihood of containing listed ingredients or adulterants with unknown or inadequate safety data[55]. Diagnosis of supplement-induced liver injury relies on clinical suspicion, typically supported by the absence of prior illness, onset of symptoms or biochemical abnormalities after supplement use, and improvement after discontinuation. The mainstay of therapy for herbal hepatotoxicity is withdrawal of the offending toxin and supportive care[56]. Some cases may progress rapidly to ALF requiring transplantation. The diagnosis is often delayed due to underreporting, as patients may not consider supplements to be medications. Timely recognition and discontinuation are therefore critical to prevent progression or preserve the opportunity for liver transplantation.
There are many scenarios during the course of hepatitis B virus (HBV) infection that necessitate timely intervention to halt the development of florid HBV infection[57].
Pregnant ladies with high viremia (> 200000 IU/mL) should start antiviral therapy by the 2nd trimester to reduce transmission risk[58]. Antiviral agents with established safety profiles in pregnancy, e.g., tenofovir, are preferred[57-59]. Infants of HBV surface-protein (HBsAg)-positive mothers should receive HBV vaccine within 12 hours of birth and HBIG at the same time, up to 7 days. Infants should complete the hepatitis B series and adhere to the minimum intervals between doses[59]. To assess perinatal transmission and confirm immunity, post-vaccination serology test combining HBsAg and hepatitis B surface antibody should be performed at 9-12 months to confirm protection and detect late infections. Post-vaccination serology test at 9-12 months is time sensitive, as earlier testing may detect passive HBIG antibodies and miss late HBV infection.
Hepatitis B is of particular concern following occupational exposures. It remains infectious on surfaces for ≥ 7 days and can be transmitted without visible blood. Health care personnel (HCP) with significant exposure should be urgently evaluated to allow for post exposure prophylaxis (PEP). The type of PEP depends on HBV immune status of HCP and the HBsAg status of the source patient as shown in Table 2[59]. Fully vaccinated responders (anti-HBs > 10 mIU/mL) are considered immune; no PEP required regardless of source status[59]. Nonimmune, unvaccinated, or incompletely vaccinated (anti-HBs < 10 IU/L) HCP requires baseline anti-the core protein (HBc) and follow-up testing after 6 months with anti-HBc and HBsAg[57,59]. For risky exposures, immediate PEP with both hepatitis B vaccine and HBIG has to be given at the same time. HBIG should be given within hours for a maximum of 7 days of exposure (0.06 mL/kg IM, protection lasts 3-6 months). If the patient is documented vaccination non-responder, PEP should be limited to two doses of HBIG one month apart[58,59].
| HCP vaccination status | HCP immune testing (HBsAb titre) | Source status (HBsAg) testing | Postexposure prophylaxis (PEP) | PEP protection testing (HBsAb titre) | |
| Vaccination | HBIG | ||||
| Documented responder after CVCS | Not indicated | Not indicated | Not indicated | Not indicated | Not indicated |
| Documented non-responder after 2 CVCS | Anti-HBs < 10 IU/L | Negative | Not indicated | Not indicated | Not indicated |
| Positive/unknown | Not indicated | Of 2 doses one month apart | Not indicated | ||
| Response not tested after CVCS | Anti-HBs ≥ 10 IU/L | Not indicated | Not indicated | Not indicated | Not indicated |
| Anti-HBs < 10 IU/L | Negative | Re-vaccinate | Not indicated | Yes | |
| Positive/unknown | Start CVCS | One dose ASAP up to 7 days | Yes | ||
| Unvaccinated/incomplete VCS | Anti-HBs ≥ 10 IU/L (post infection recovery) | Not indicated | Not indicated | Not indicated | Not indicated |
| Anti-HBs < 10 IU/L | Negative | Start CVCS | Not indicated | Yes | |
| Positive/unknown | Start CVCS | One dose ASAP up to 7 days | Yes | ||
All patients with chronic HBV infection should receive nucleoside analog therapy pre-transplantation and continue post-transplantation to reduce the risk of reinfection of the transplant. The decision to add HBIG with antivirals to prevent graft infection varies by center, as its added benefit remains uncertain[60]. For high-risk patients, HBIG has been given as a 10000 IU IV bolus dose/day perioperatively, at transplantation, and postoperatively for 7 days[61] or longer if the patient remains HBsAg positive and has high HBV DNA levels post-transplant. This fixed-dose schedule routinely results in anti-HBc > 500 IU/L for a few months post-transplant. Nucleoside analogs should not be discontinued, regardless of the anti-HBs response[61]. In patients with high HBV DNA, we use dual antivirals (entecavir and tenofovir) to rapidly decrease viral load (thus decreasing the risk of recurrence). One of the antivirals (tenofovir) is stopped at 3 months or 6 months after LT when HBsAg and HBV DNA are undetectable[57,62].
Although the role of HBV immunization in preventing HBV reinfection after liver transplantation is unclear. Although it is reasonable to administer HBV vaccination to patients on low-dose immune suppression, nucleoside analogs should not be discontinued, regardless of the anti-HBs response. Active immunization using hepatitis B vaccine for im
The most common sequel of decompensation in liver cirrhosis is ascites; nearly 5% to 10% of patients with compensated cirrhosis per year develop ascites. Approximately 10% of inpatients with ascites will develop SBP. Early recognition and treatment can reduce mortality from > 90% to approximately 20%[65].
Prompt paracentesis facilitates timely diagnostic and therapeutic interventions, which help improve outcomes and reduce resource utilization. Many studies recommended prioritizing diagnostic paracentesis within the first two days of admission[66], even preferably within 12 hours of admission to prevent the development of HRS and reduce 30-day mortality, particularly in patients found to have SBP[67]. If ascitic fluid protein content is low (typically < 1.5 g/dL) without evidence of SBP, antibiotics including daily oral fluoroquinolones (ciprofloxacin or norfloxacin) are re
If the ascitic fluid neutrophil count > 250/mm3, which is diagnostic of SBP, immediate empirical antibiotic therapy should be prescribed considering the context of SBP (community-acquired or health care associated), severity of infection, and local bacterial resistance profile. Cefotaxime has been widely studied, but the choice of antibiotic should be guided by local resistance patterns and protocol. A second diagnostic paracentesis at 48 hours from the start of treatment is recommended to check the efficacy of antibiotic therapy, especially in patients who have an apparently inadequate response or where secondary bacterial peritonitis is suspected[65]. Combining prompt initiation of antibiotics with IV human albumin can prevent the development of HRS. Human albumin is given in high doses on days 1 and 3. Mortality associated with the development of HRS can be as high as > 40%[68]. Once a diagnosis of HRS is established, immediate treatment should be initiated, aiming to reverse this fatal complication. Treatment should include a vasoconstrictor therapy combined with albumin infusion. When available, terlipressin is the recommended vasopressor because of its high efficacy. Other treatment options include a combination of midodrine and octreotide. In the context of ICU admission or lack of response, norepinephrine infusion is currently recommended given its comparable efficacy to terlipressin[68,69]. Hepatic encephalopathy is another common complication of hepatic decompensation and carries a high morbidity and short-term survival. However, no current evidence-based studies define a precise time to initiate specific treatment, although logically early initiation of treatment, workup to define any precipitating factor, and ICU level of care is associated with improved outcomes[68,69].
Delayed paracentesis in cirrhotic ascites is associated with longer hospital stays, subsequently higher costs, and increased complications, including HRS, variceal bleeding, and sepsis[65,70]. In an observational study, each hour of delay in diagnostic paracentesis after admission was associated with a 3.3% increase in in-hospital mortality after adjusting for MELD score[71]. The same study showed that delays in paracentesis beyond 12 hours of admission accounted for a 2.7-fold increase in in-hospital mortality. These findings support the prioritization of early (within 12 hours) diagnostic paracentesis in all cirrhotic ascitic patients admitted[71]. An interesting retrospective cohort study of 126 patients with cirrhosis and SBP-associated septic shock demonstrated that those who survived were more likely to have been administered red antibiotics earlier [median (range) = 1.8 (1.1-5.2) vs 9.5 (3.9-14.3) hours; P < 0.001]. They also demonstrated that each hour of delay in appropriate antimicrobial therapy was associated with a 1.86 times increased hospital mortality[72].
Upper GI bleeding (UGIB) is a medical emergency that needs to be treated quickly in order to lower morbidity and death rates. Although endoscopy and medication have advanced, mortality is still high (5%-14% in high-risk individuals).
Time is very important in decision-making and intervention. The first 15 minutes to 60 minutes of immediate stabilization for patients who are unstable (shock, hazard ratio > 100, SBP < 90), administration of IV access using two large-bore cannulas and crystalloids is done to prevent over-resuscitation in cases of variceal haemorrhage. The fluid resuscitation should be judicious with careful monitoring to avoid precipitation of rebleeding. This is followed by a blood transfusion, which should be a large-scale procedure in the event of continuous bleeding. We endorse the most recent guidelines with a restrictive policy of red blood cell transfusion with a threshold for transfusion at a hemoglobin level of 7 g/dL for patients with UGIB. Hypotensive patients may be transfused at higher hemoglobin levels given equilibration that occurs with fluid resuscitation, and a threshold of 8 g/dL is reasonable in patients with pre-existing cardiovascular disease[73,74]. In case of variceal suspicion (cirrhosis, stigmata), IV ceftriaxone (prevents bacterial infections) plus vasoactive medications (octreotide/terlipressin) are administered. Endoscopic timing is critical and is generally recommended in the first 12 hours (emergent) in cases of hematemesis and shock, and in the first 24 hours (urgent) in cases of stable patients with melena or hematochesia. Delayed endoscopy (after 24 hours) is associated with increased mortality. Endoscopy is beneficial in both diagnosis and treatment. Esophageal band ligation and cyanoacrylate injection of gastric varices are done through endoscopy. Endoscopic hemostatic therapy with bipolar electrocoagulation, heater probe, or injection of absolute ethanol is done for patients with UGIB due to ulcers (strong recommendation, moderate-quality evidence). Endoscopic hemostatic therapy with clips, argon plasma coagulation, or soft monopolar electrocoagulation is also a therapeutic intervention for patients with UGIB due to ulcers. Post-endoscopic early management includes IV proton pump inhibitors twice daily for 72 hours in high-risk ulcers and not in variceal bleeding[73,74]. Trans-jugular intra-hepatic - porto-systemic shunt will be of great value for variceal bleeding if done in the first 72 hours for high-risk patients (Child-Pugh B/C)[75-77].
Early intervention in UGIB - including rapid resuscitation, timely endoscopy (< 24 hours), and pharmacologic therapy - has been shown to improve clinical outcomes. However, health care systems must balance efficacy, resource utilization, and cost. Early endoscopy (< 24 hours) reduces rebleeding rate by 30%-50% and shortens hospital stay by 1-2 days. High-risk patients are cost-effective for urgent endoscopy (prevents costly complications like shock, surgery). While for low-risk patients, outpatient management (avoiding hospitalization) is cost-saving. Early trans-jugular intra-hepatic - porto-systemic shunt is cost-effective in advanced cirrhosis[78].
Lower GI bleeding accounts for approximately 20% of acute GI hemorrhages, with diverticulosis being the most common cause.
Rapid risk stratification, hemodynamic stabilization, and timely intervention are critical to reducing morbidity and mortality. Initial assessment and risk stratification: Hemodynamic instability (tachycardia, hypotension) necessitates immediate resuscitation with IV fluids and blood transfusion (target hemoglobin ≥ 7 g/dL, or hemoglobin ≥ 8 g/dL in cardiovascular disease).
Colonoscopy: Optimal timing is within 24 hours (vs elective) for improved diagnostic yield (22%-45% vs 12%) and therapy (odds ratio = 2.7 for hemostasis).
Preparation: Rapid 4-6 L polyethylene glycol or oral sulfate solutions enable early colonoscopy[79,80]. Endoscopic therapy (clips, coagulation, epinephrine) is first-line for stigmata of bleeding (e.g., diverticula, angioectasia).
Radiologic rescue: Computed tomography (CT) angiography (CTA) localizes bleeding (> 0.3-0.5 mL/minute) before transcatheter arterial embolization (success rate 65%-90%). Surgery (< 10% of cases) is reserved for refractory bleeding, with segmental resection preferred[73,74]. Early, risk-stratified management of acute lower GI bleeding significantly reduces mortality and health care costs by avoiding delays in critical interventions. Early colonoscopy (< 24 hours) decreases 30-day mortality (odds ratio = 0.48, 95% confidence interval: 0.25-0.92) by enabling timely hemostasis. Reduces rebleeding rates (9% vs 23% with delayed scope).
CTA-guided embolization: Mortality < 5% when performed within 6 hours vs 15%-20% with delayed intervention[79-82].
Risk-stratified discharge (e.g., Oakland Score): Safely avoids unnecessary hospitalization.
Early colonoscopy vs routine admission: A lot of money is saved by reducing ICU stays and rebleeding.
CTA-first approach for unstable patients: More cost-effective than empiric surgery.
Thromboembolic disease is the third most common acute cardiovascular disease, after cardiac ischemic syndromes and stroke[83]. Acute mesenteric venous thrombosis (MVT) causes 5% to 15% of all cases of mesenteric ischemia and accounts for 1 in 5000 to 15000 inpatient admissions and 1 in 1000 emergency department visits. The incidence of MVT is estimated to be 2.7 per 100000 persons[83]. CT has increased the number of cases diagnosed during evaluation for other reasons, and this imaging modality identifies approximately 90% of mesenteric venous thromboses. Although mortality remains between 19% and 23%, there has been a trend toward earlier diagnosis and improved outcomes. MVT is slightly more common in men and most frequently presents in persons between the ages of 40 and 60[83,84].
Patients diagnosed with MVT require close monitoring, and their outcomes depend on timely communication between the interprofessional team and the ability to rapidly adapt to clinical circumstances and changes. MVT should be diagnosed early using a combination of clinical manifestations and CTA. The CTA should be introduced earlier instead of CT as the imaging modality of choice for the diagnosis of MVT. In addition, magnetic resonance imaging and Doppler ultrasonography are reserved for specific indications (e.g., magnetic resonance imaging for patients with contraindications to contrast). Patients with acute MVT usually present within 24-72 hours of symptoms. The predominant symptom is midabdominal pain. Patients with acute mesenteric venous thrombi often experience pain out of proportion to physical findings. Fever and signs of peritonitis suggest progression of ischemia to infarction[83,85].
Once diagnosed, treatment must be introduced as early as possible, and any delay beyond 24 hours should be avoided. Treatment goals are to prevent bowel infarction, mitigate clotextension, and minimize recurrence risk. Initial treatment depends on clinical presentation, the acuity of the thrombosis, and the underly in etiology[83,85].
Non-operative therapy: Stable patients may be treated nonoperatively with bowel rest, electrolyte replacement, and anticoagulation. Unfractionated IV heparin is initiated when diagnosed early without the development of peritonitis. Low-molecular-weight heparin may be administered subcutaneously in patients with milder symptoms. Switch to direct oral anticoagulants or vitamin K antagonist is indicated when symptoms decline, indicating clinical improvement, and when imaging shows stable disease[86]. Direct oral anticoagulants are increasingly preferred due to the ease of use and the fewer monitoring requirements. Anticoagulation is given for six months in the presence of an unidentifiable transient risk factor, whereas patients with underlying thrombophilia or idiopathic MVT may be considered for lifelong anticoagulation[87] because recurrence of MVT may be fatal[88]. Nasogastric decompression is often necessary, and total parenteral nutrition may be indicated. Febrile patients may benefit from broad-spectrum antibiotics to treat thrombophlebitis or enteric bacterial translocation[87].
Endovascular therapy: Significant clot burden and clinical deterioration warrant invasive intervention. Historically, the only option for urgent intervention was open surgery. Interventional radiology can offer precise therapy such as catheter-directed thrombolysis, thrombectomy, and focal administration of pharmacologic agents, including papaverine for vasodilation, or tissue-type plasminogen activator or streptokinase for clot lysis. These interventional radiology measures often result in symptomatic improvement and lower bowel resection rates, even with persistent radiographic thrombus[83].
Surgery: Although the availability of CT scanners has improved, exploration remains the leading method for diagnosing MVT worldwide. Explorative laparotomy is preferred over laparoscopy because of the better exposure and the ability to assess bowel viability. Surgery is indicated if a patient becomes septic from ischemic bowel or a perforated viscus. The area of thrombosis is identified, and the affected bowel segment is resected. The goal of surgery is bowel conservation while respecting necrotic segments, and the average length of resection is 50 cm to 60 cm. Intraoperative Doppler ultrasonography may be used for timely diagnosis to assess the mesenteric blood supply. The surgeon may return in 24 hours for a second look before completing the anastomosis if there are concerns for ongoing ischemia[89].
Although the overall mortality has decreased due to earlier diagnosis and treatment, MVT remains a life-threatening condition, and a delay in diagnosis directly correlates with worse outcomes. The most common causes of death in patients with MVT are sepsis, pulmonary emboli, and the sequelae from recurrent thrombi. Patient age, comorbidities, time to diagnosis, and successful revascularization impact outcomes[89]. Acute MVT has a 30-day mortality of up to 32%. Time-related decisions reduce mortality to less than 10%; the mortality rate approaches 60% when diagnosis is delayed[89]. If treatment is delayed longer than 24 hours from symptom onset, mortality ranges between 80% and 100%[83,89,90].
Delay in management of inflammatory bowel disease (IBD), like UC and Crohn’s disease (CD), could lead to complications like acute severe UC, toxic megacolon, bowel perforation, severe bleeding, sepsis, or irreversible damage requiring surgery.
In most UC patients, the disease has a mild to moderate course, but nearly a quarter of them require hospitalization due to severe flare with a 20% risk of colectomy on first hospitalization, increasing to 40% with subsequent admissions[91]. In Western countries, acute severe UC (ASUC) is considered a life-threatening condition leading to a 1% mortality rate[92]. It is defined according to the modified Truelove and Witts criteria by the passage of bloody stools ≥ 6 times/day associated with at least one marker of systemic toxicity such as: Fever ≥ 37.8 °C, pulse ≥ 90 beats/minute, hemoglobin < 10.5 g/dL, erythrocytes sedimentation rate (ESR) > 30 mm/hour or C-reactive protein (CRP) > 30 mg/dL[93].
Early intervention in ASUC within the first 24-72 hours is critical to avoid complications, including colectomy or death. The standard of care of this IBD emergency includes a multidisciplinary approach of early initiation of IV steroids and timely escalation of treatment to either medical rescue therapy or surgery[94].
Initial management (day 1-3): Hospital admission for intensive therapy and close monitoring[95].
Baseline laboratory investigations: Including blood tests (complete blood count urea, creatinine, electrolytes, CRP/erythrocytes sedimentation rate, and liver profile) and stool tests [stool analysis, culture, and Clostridioides difficile (CDI) assay].
Imaging studies: Including pelvi-abdominal ultrasound, abdominal X-ray (or CT if suspected abdominal complications).
Early flexible sigmoidoscopy: It is recommended to be done ideally within 24 hours of admission, without preparation, by an experienced endoscopist with minimal inflation to assess the disease severity, rule out CMV infection, and guide treatment escalation for shorter hospital stay and better clinical outcome[91].
Pre-biologic therapy screening: Hepatitis B, hepatitis C, human immunodeficiency and varicella zoster viruses, tuberculosis screening (chest X-ray and Quantiferon test), thiopurine methyl transferase, and lipid profile.
Thromboembolism prophylaxis: Low molecular weight heparin and compression stockings.
First-line therapy: Early IV corticosteroid - equivalent to 0.8-1 mg/kg of methylprednisolone over 24 hours or 100 mg hydrocortisone qid for 5-7 days.
Precautions: Avoid drugs - non-steroid anti-inflammatory drugs, opioids, anti-diarrhoeal and anticholinergic. Also, avoid routine use of antimicrobial agents unless infection is suspected.
Nutrition care: Correct dehydration, enteral nutrition, and correct any electrolyte imbalance.
Anticipated steroid failure: The Oxford cohort developed and validated the Admission Model for Intensification of Therapy in Acute Severe Colitis index, Ulcerative Colitis Endoscopic Index of Severity, CRP, and albumin levels at admission for predicting response to IV steroids. Patients admitted with the highest Admission Model for Intensification of Therapy in Acute Severe Colitis index, corresponding to those having CRP ≥ 100 mg/L, Ulcerative Colitis Endoscopic Index of Severity ≥ 7, and albumin ≤ 25 g/L, will fail IV steroids in 100% of cases[96,97] (Table 3).
| Item | Score |
| CRP ≥ 100 mg/L | 1 |
| Albumin ≤ 25 gm/L | 1 |
| UCEIS ≥ 4 | 1 |
| UCEIS ≥ 7 | 2 |
| ADMIT-ASC score | Rate of steroid response |
| 0 | 100% |
| 1 | 69.6% |
| 2 | 40.7% |
| 3 | 17.5% |
| 4 | 0% |
Prediction of response to corticosteroid therapy (on day 3 of corticosteroid therapy): The last update of European Crohn’s and Colitis Organisation guidelines recommended assessing the number of bowel movements with CRP levels 3 days after IV steroids to predict steroid failure[98]. Patients with a stool frequency of 8/day, or a stool frequency of 3/day plus a CRP concentration > 45 mg/dL have an 85% chance of needing colectomy during that admission[91]. At this stage, a decision must be made regarding the need for rescue medical therapy or surgery. Overall, initiation of second-line medical treatment or salvage colectomy should be initiated in the absence of response to IV steroids beyond three to a maximum of five days after admission[98].
Rescue therapy: Both infliximab (5 mg/kg IV) and ciclosporin (2 mg/kg IV) drugs are equally effective in ASUC if the decision is to use rescue medical therapy at day 3, and either of them can be used, but infliximab can be easier to administer from a practical perspective. Patients must be closely monitored after rescue therapy has been initiated. Surgery is indicated by day 7 if there is an inadequate response to rescue medical treatment. Emergency indications for surgery in ASUC are toxic megacolon, life-threatening haemorrhage, and perforation[91]. The gold standard operation in the emergent setting of acute severe colitis (ASC) is subtotal colectomy with end ileostomy[99].
It is a severe variant of ASC. It is more commonly seen in UC patients than in CD patients, with an incidence of 8% to 10% vs 2.3%, respectively. The diagnostic criteria for toxic megacolon (TM) include clinical findings of toxic colitis at least three of the following: Fever > 38.6 °C, 101.5 °F, tachycardia > 120 beats/minute, leukocytosis > 10.5 × 103/μL, anemia), sign of end organ damage (at least one of the following: Hypotension, hypovolemia, altered mental status, electrolyte disorders) and radiographic findings of colonic distention greater than 6 cm[100].
Time-sensitive decision: Medical management with steroids may prevent the need for urgent surgery in up to 50% of TM patients; however, these patients frequently undergo colectomy later for recurrent episodes of ASC or TM. Indications that medical management has failed are persistent fevers after 48 hours to 72 hours, persistent hemodynamic instability, increasing transfusion requirements, or increased colonic dilation on repeat imaging[101]. Infliximab and tacrolimus have shown some efficacy as salvage agents for acute prevention of colectomy; however, delaying surgery in TM may lead to perforation with its associated mortality rate of 27% to 57%. The suggested surgical intervention is a subtotal colectomy and end ileostomy when medical management fails[101].
Delay in surgical intervention is associated with a mortality rate up to 57%[101].
The incidence of colonic perforation for IBD patients is highest in UC, especially in the setting of TM[102]. Diagnosis is a mixture of clinical assessment and radiographic evidence. Diffuse peritonitis may be present on physical examination or absent secondary to immunosuppression. Abdominal X-rays or CT scans can diagnose free intra-abdominal perforation with pneumoperitoneum seen in both modalities, and free fluid specifically seen in the CT scan[99].
Time-sensitive decision: Immediate exploratory laparotomy with control of sepsis[102].
CDI is a common cause of infectious diarrhea. It is associated with significant morbidity and mortality and carries a high risk of recurrence. Early diagnosis and initiation of empirical antibiotic treatment for suspected CDI can reduce mortality and improve clinical outcomes.
The diagnosis and treatment of CDI (Table 4) should be a speedy and accurate process. The key is to confirm CDI diagnosis within 1 day and start empiric treatment when awaiting results to reduce hospitalization time. Multi-step testing using nucleic acid amplification test (polymerase chain reaction) plus toxin assay boosts specificity, minimizing overtreatment, especially in immunocompromised patients who often are colonized rather than infected[103]. Rapid initiation of therapy (within 24 hours of sample collection) shortens hospital stay (approximately 6 days vs 8 days), though it hasn’t clearly improved mortality or recurrence in adults[104].
| Patient phenotype (stage of illness) | Recommended action | Time frame |
| Symptomatic (suspected) | Multi-step diagnostics + start empiric therapy (fidaxomicin or vancomycin) | Within 24 hours |
| Severe/fulminant | High-dose oral vancomycin + IV metronidazole; consider rectal vancomycin | Immediately |
| First recurrence | Switch to fidaxomicin | As early as possible after recurrence |
| ≥ 2 recurrences | Add fecal microbiota transplantation promptly | As early as possible |
| Fulminant refractory to medical therapy | Surgical consultation (or ileostomy lavage) | Within hours/days of deterioration |
First-line: Fidaxomicin (if available) is preferred over vancomycin due to lower recurrence; oral vancomycin remains an alternative. Metronidazole - only for oral use in mild cases when other agents are unavailable[105].
Severe or fulminant CDI (e.g., hypotension, shock, ileus or megacolon): High-dose oral/nasogastric tube vancomycin (500 mg q6h) + IV metronidazole; add rectal vancomycin if ileus is present[105].
Recurrent CDI: Fidaxomicin (standard or extended-pulse) is preferable over conventional vancomycin[105]. Bezlo
Immunocompromised patients: No special first-line antibiotics; follow fidaxomicin/vancomycin regimens. FMT is recommended with caution; it is discouraged in patients with severe immunocompromised conditions but accepted in mild-to-moderate situations.
Subtotal colectomy + end ileostomy is standard for fulminant CDI refractory to medical care - carries approximately 41% 30-day mortality[108].
Sequential FMT: In severe cases that do not respond within 48 hours, sequential FMT may also be attempted[107].
Rapid diagnosis and early initiation of optimal therapy – especially fidaxomicin first-line, rigid protocols for severe cases, timely use of FMT in recurrences, and early surgical involvement in fulminant disease - are critical to improving outcomes and reducing mortality in Clostridium difficile colitis. Delays at any stage of management lead to longer hospitalization, which increases costs and risk of in-hospital complications. Higher recurrence and late mortality, especially in high-risk groups (e.g., cancer, elderly) - up to 40% mortality at 1 year; metronidazole use associated with delayed death[109]. Greater likelihood of needing surgery and its associated high mortality if progression to the fulminant stage is not prevented.
Foreign body (FB) ingestion is a common cause for emergency department presentation. It comprises a true FB (non-food) ingestion and food bolus impaction. In adults, FB ingestion is more common in patients with psychiatric disorders, the elderly, prisoners, drug dealers (body packing), and alcohol intoxication, where food bolus impaction (including Steakhouse syndrome), animal bones, toothpicks, dentures, and fish bones are the most frequent. True FB ingestion is mostly encountered in pediatric populations where coins, buttons, plastic items, batteries, and bones are commonly encountered[110-112].
Initial evaluation for risk stratification is very crucial and can be achieved by combining careful history taking with clinical examination and investigations if needed. Precise history (type of FB, time of onset, previous GI tract surgery, caustic ingestions, congenital gut malformations) is essential, and physical examination is also mandatory. Most patients are asymptomatic. Symptoms arise when the FB is stuck in the esophagus or when a complication occurs (obstruction and perforation). Hypersialorrhoea and inability to manage secretions (drooling of saliva) suggest complete esophageal obstruction, warranting emergent endoscopic intervention. Cervical crepitus, neck swelling, or pneumomediastinum are suggestive of esophageal perforation. Choking, stridor, wheezing, or dyspnoea can be seen as a result of aspiration or tracheal compression by the FB[113,114].
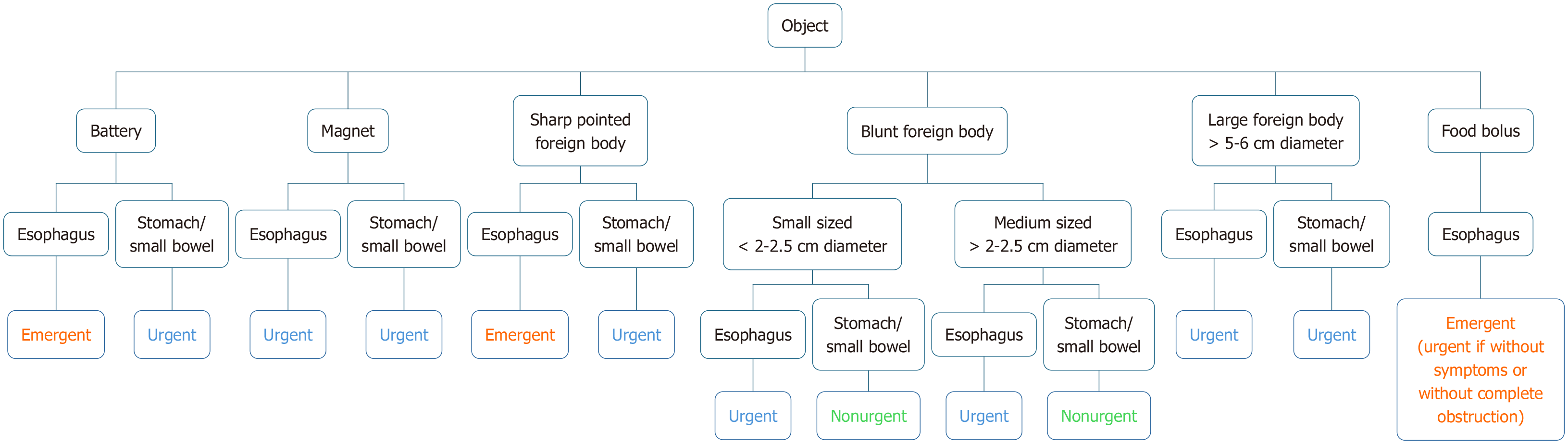
Judgment of the risks of aspiration, obstruction, or perforation determines the timing of endoscopy. Accordingly, timing of the therapeutic esophagogastroduodenoscopy (EGD) will be either emergent (preferably within 2 hours, but at least within 6 hours), urgent (within 24 hours), or nonurgent (within 72 hours). Emergent intervention is recommended for FB ingestion causing complete esophageal obstruction, and for sharp-pointed objects and batteries in the esophagus. Liquefaction necrosis and perforation can occur rapidly when a disk battery is lodged in the esophagus, leading to severe and potentially fatal complications. Urgent (within 24 hours) therapeutic EGD is recommended for other esophageal foreign bodies without complete obstruction and for foreign bodies in the stomach, such as sharp-pointed objects, magnets, batteries, and large/Long (> 5-6 cm) objects. Nonurgent (within 72 hours) therapeutic EGD is suggested for medium-sized (> 2-2.5 cm and < 5 cm diameter) blunt foreign bodies in the stomach (Figure 4). Clinical observation without the need for endoscopic removal is recommended for the management of asymptomatic patients with ingestion of blunt and small (< 2-2.5 cm diameter) objects (except batteries and magnets).
Underlying esophageal conditions, including eosinophilic esophagitis, motility disorders, stenosis, and diverticula, are frequent[115]. The majority of ingested foreign bodies pass through the alimentary tract without complication. Endoscopic retrieval is recommended following ingestions with high risk of complications, or ingestion of foreign bodies that are unlikely to traverse the GI tract, and approximately 1% of patients require surgery due to complications[113].
Corrosive ingestion is a medical emergency that is especially prevalent in developing countries. Airway assessment and prompt management are priorities in emergency settings. It is usually accidental in children, while in adults, it might be related to suicide. Acids cause coagulation necrosis, and alkalies cause liquefaction necrosis[116,117]. Morbidity and mortality rates of corrosive injuries are high. Extensive burns can cause the fragile esophageal wall to become perforated, while post-corrosive esophageal strictures cause patients to suffer and are difficult to treat[118,119].
Tissue injuries after corrosive ingestion go through three phases. Phase 1 is characterized by cell necrosis and thrombosis, 48-72 hours after the event. Next, in phase 2, there is mucosal sloughing with ulceration of the esophageal wall plus fibroblast colonization and granulation. This phase continues for 14 days from phase 1, and the esophagus is friable during this phase. Finally, in phase 3, the healing process starts in the third week and usually continues for 3-6 months[119].
Upon arrival at the emergency department, patient stabilization is the most important target for this stage. The airway is the 1st to be examined, especially for signs of aspiration or laryngeal injury. Physical examination and history taking should be done for details of the corrosive substance, the volume, timing before admission, pre-hospital treatment, and cause of ingestion. The patient should be given nil per os and adequate resuscitation. Nasogastric tube intubation, gastric lavage, administration of emetic drugs, and neutralizing agents are not recommended because reflux of these agents into the esophagus could result in further damage. IV broad-spectrum antibiotics may benefit a patient with high-grade esophageal injuries. The investigation by chest and abdominal radiography should be evaluated[120].
EGD is contraindicated when there are suspected perforations, oral cavity necrosis, and airway injury with com
CT can assist in the prognosis after ingestion, but it is still inconclusive. CT also provides extraesophageal information regarding anatomies such as the mediastinum, lung, and pleural cavity, which endoscopies do not. CT scans can safely provide details about esophageal transmural necrosis, consisting of esophageal wall blurring, peri-esophageal fat stranding, and no enhancement of the esophageal wall after administration of IV contrast. The combination of CT and endoscopy is especially useful for examining patients with Zargar 3b injuries. Esophageal perforation looks in the CT as edematous wall thickening with periesophageal soft tissue infiltration plus blurring of tissue interface or localized fluid collection around the esophagus or the descending aorta[122,123].
Phase 1: Within 24-48 hours, in the emergency department, the target is stabilization and severity evaluation (after exclusion of esophageal perforation, where surgical intervention should be considered) with EGD performance (unless contraindicated) and a CT scan.
Phase 2: Within 3-14 days, the target is supportive care in the hospital.
Phase 3: Continues until 3-6 months, the target is to prevent, evaluate, and treat post- corrosive esophageal stricture (Table 5).
| Zargar classification | Findings |
| Grade 0 | Normal finding on endoscopic examination |
| Grade 1 | Edema and hyperemia of the mucosa |
| Grade 2a | Friability, blisters, exudates, hemorrhages, whitish membrane, erosions, and superficial ulceration |
| Grade 2b | Grade 2a + deep discrete or circumferential ulceration |
| Grade 3a | Small scattered areas of multiple ulceration and areas of necrosis with brown-black or grayish discoloration |
| Grade 3b | Extensive necrosis |
Screening is the process of examining a subgroup of patients or population at high risk to develop a certain pathology before it occurs, when they have no symptoms, while endoscopic screening refers to gut/bowel examination by the endoscopy to detect pre-malignant lesions or early stages of malignancy, aiming to prevent the development of advanced malignancy and hence apply eradicative treatment. Time-sensitive endoscopic screening refers to endoscopic screening in a timely manner for each individual patient/population that would early pick the suspicious lesions and hence prevent the development of frank malignancy. The term time sensitive entails exposure of the individual/patient to hazard if the screening procedure is not performed at the proposed time point. Endoscopic surveillance refers to the frequent endoscopic examination, at certain time points, of the high-risk patients who already have a pre-malignant lesion, aiming to evaluate the efficacy of the treatment applied, completeness of treatment, and diagnosis of early recurrence. There are many pathological conditions in the bowel that necessitate endoscopic screening at certain time points. Although these are not acute conditions and hence the time factor is not so sensitive as other acute conditions, e.g., UGIB, yet screening/surveillance in a timely fashion is associated with improved patient outcomes.
Endoscopic screening of the upper digestive tract is crucial for detecting many premalignant lesions, and surveillance of high-risk patients who already have potentially cancerous pathologies. In fact, many sporadic and inherited pathologic lesions affecting the esophagus, stomach, upper small bowel, and biliary system should be focused on by the endoscopic screening and surveillance as described below.
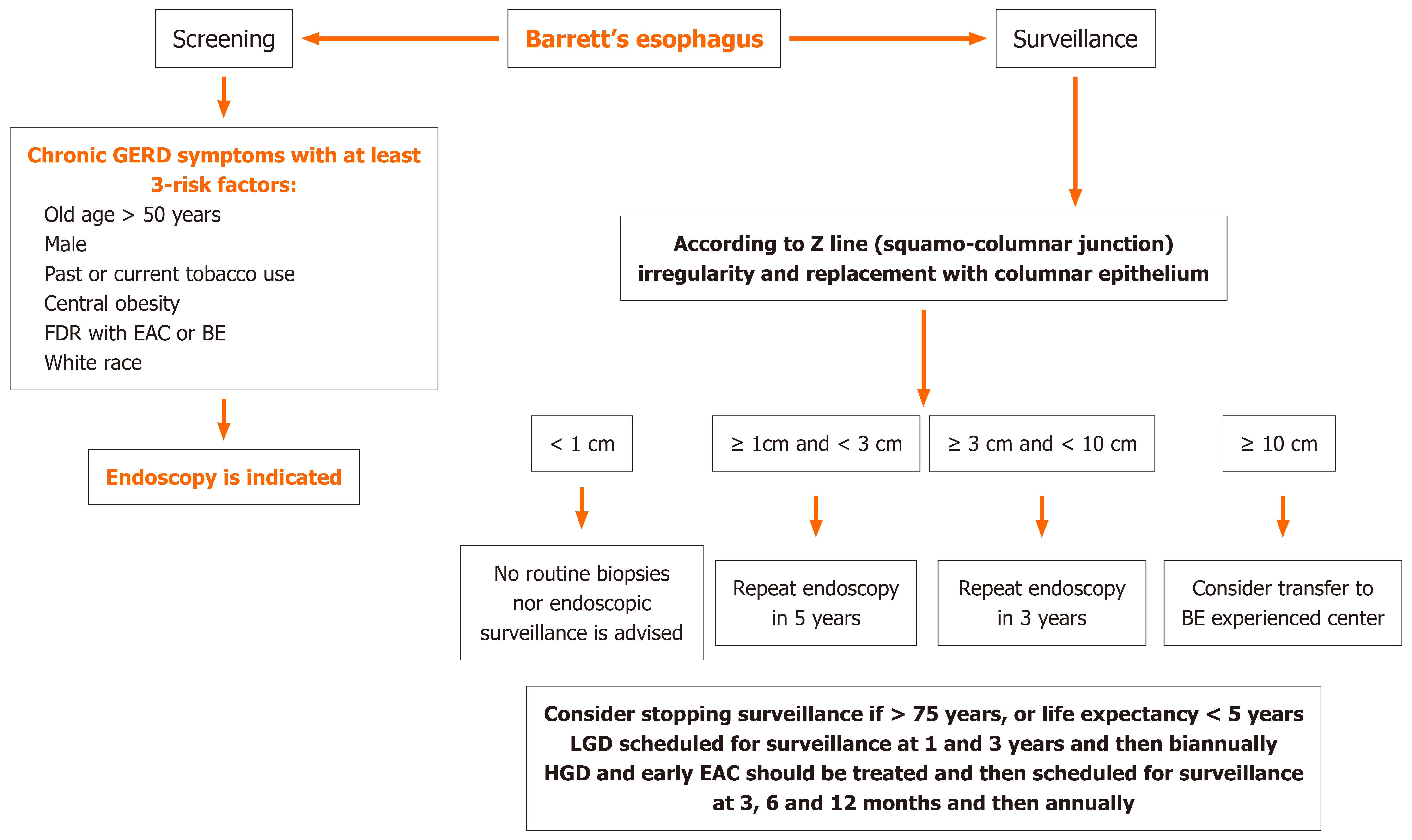
Barrett’s esophagus (BE) is diagnosed by endoscopic illustration of columnar mucosa that extends ≥ 1 cm above the gastroesophageal junction, and biopsies demonstrate specialized intestinal metaplasia (IM), containing goblet cells. It is the only known precursor to esophageal adenocarcinoma (EAC), which has a poor 5-year survival rate of only 20%, because of the tendency toward early metastasis as the tumor is located in close proximity to lymphatic drainage in the submucosa, and the late diagnosis because the symptoms (dysphagia and weight loss) develop late when the cancer has reached an advanced stage[124]. There are variable recurrence rates of metaplasia after eradicative therapies for BE, hence[125], the screening/surveillance process for BE to early recognize and treat the advanced histologic changes is justified.
Timing: Is illustrated in Figure 5, following the American College of Gastroenterology guidelines[126]. A single-screener endoscopy is recommended to establish the diagnosis of BE in individuals with chronic reflux symptoms and 3-risk factors, which include male sex, age > 50 years, White race, current or prior tobacco use, central obesity, and first-degree relative(s) with BE or EAC. Currently, a repeat endoscopic screening in patients who have undergone an initial negative screening examination by endoscopy is not recommended. Once a BE diagnosis is established, American College of Gastroenterology recommends an endoscopic surveillance program guided by both the length/height of the BE mucosa and the degree of dysplasia (based on the biopsies obtained during the screening endoscopy). Non-dysplastic BE segments of (≥ 3 cm) are surveyed on a 3-year interval, and shorter segments (< 3 cm) are surveyed on a 5-year interval. Patients with BE associated with low-grade dysplasia are preferably treated with endoscopic eradicative therapies (EET) to halt its progression to high-grade dysplasia (HGD) or early cancer; otherwise be scheduled for endoscopic surveillance at 6 months and 12 months and thereafter annually. Patients with BE-associated HGD or early stages of EAC should be treated with EET that includes resection of all visible lesions, followed by eradication of any remaining BE mucosa, aiming to achieve complete eradication. Once complete eradication of IM is achieved, patients should be scheduled for endoscopic surveillance based on baseline dysplastic state. In case of baseline low-grade dysplasia, endoscopic surveillance should be scheduled at 1 year and 3 years and thereafter biannually, whereas if baseline HGD/early EAC, endoscopic surveillance should be scheduled at 3 months, 6 months, and 12 months and then annually. Consider cessation of endoscopic surveillance when a patient is no longer a candidate for EET (Figure 5). There are no major differences between American and European GI endoscopic societies regarding the timing intervals in endoscopic screening/surveillance for BE[126,127].
Hazard/benefit: The principal rationale for performing BE screening is that, if the screening examination yields a diagnosis of BE, patients can be placed in an appropriate endoscopic surveillance program to allow timely detection of BE dysplasia or early-stages EAC amenable to endoscopic treatment, preventing EAC or enabling effective treatment of its early stages. However, non-endoscopic screening tools had been introduced as an acceptable alternative to endoscopic screening for BE, including swallowable, non-endoscopic capsule devices combined with biomarkers.
There are many polyposis syndromes[126,128,129] that mainly affect the lower GI tract but can affect the upper GI tract as well and predispose to malignancy. These should be exposed to upper endoscopic screening in conjunction with the colonoscopy, as shown in Table 6.
| Clinical situation | Ref. | Recommended age/time for screening colonoscopy | Recommended age/time for screening gastroscopy |
| Lynch syndrome | Ladigan-Badura et al[128], 2021 | Every 1-2 years beginning between ages 20 years and 25 years or 2-5 years before earliest CRC diagnosis in the family, whichever is earlier | Considered especially for those with family history of gastric cancer and those of Asian ancestry; every 3-5 years beginning between the ages 30 years and 35 years |
| FAP | Weiss et al[129], 2021 | Yearly starting at age 10 and continuing until colectomy; post-colectomy rectal or ileal pouch colonoscopy every 1-2 years; for attenuated FAP, yearly screenings should begin by age 20 | Upper endoscopy to examine the stomach and duodenum at 20 years to 25 years; screening may start earlier if a patient undergoes colectomy before this age; the endoscopy should adequately visualize the ampulla of Vater (use of the side viewing endoscope may be advised) |
| Peutz-Jeghers syndrome | Weiss et al[129], 2021 | Once the diagnosis is confirmed, regular surveillance and imaging are essential due to the increased malignancy risk; upper gastrointestinal endoscopy, video capsule endoscopy, and colonoscopy are recommended between ages 8 and 10 to screen for gastric, duodenal, and small bowel polyps; if polyps are detected on baseline screening, endoscopic evaluation should be repeated every 2 years to 3 years; if polyps are absent, screening should resume every 2 years to 3 years beginning at age 18 | |
| MUTYH-associated polyposis | Weiss et al[129], 2021 | Colonoscopy with polypectomy every one to two years beginning at age 25-30 years; prophylactic colectomy when the polyps became unmanageable | Consider upper endoscopy (including side viewing duodenoscopy) exam at 30-35 years examination, which should evaluate the ampulla of Vater; repeat every three months to four years based on initial findings (number, size, and type of polyps found) |
| Juvenile polyposis syndrome | Shaheen et al[126], 2022 | About 1-3 year interval range, start with symptoms or latest at 18-20 years | |
| Hyperplastic polyposis | Every 1-2 years, prophylactic colectomy is performed when the polyps became unmanageable | ||
| Inflammatory bowel disease | See later | ||
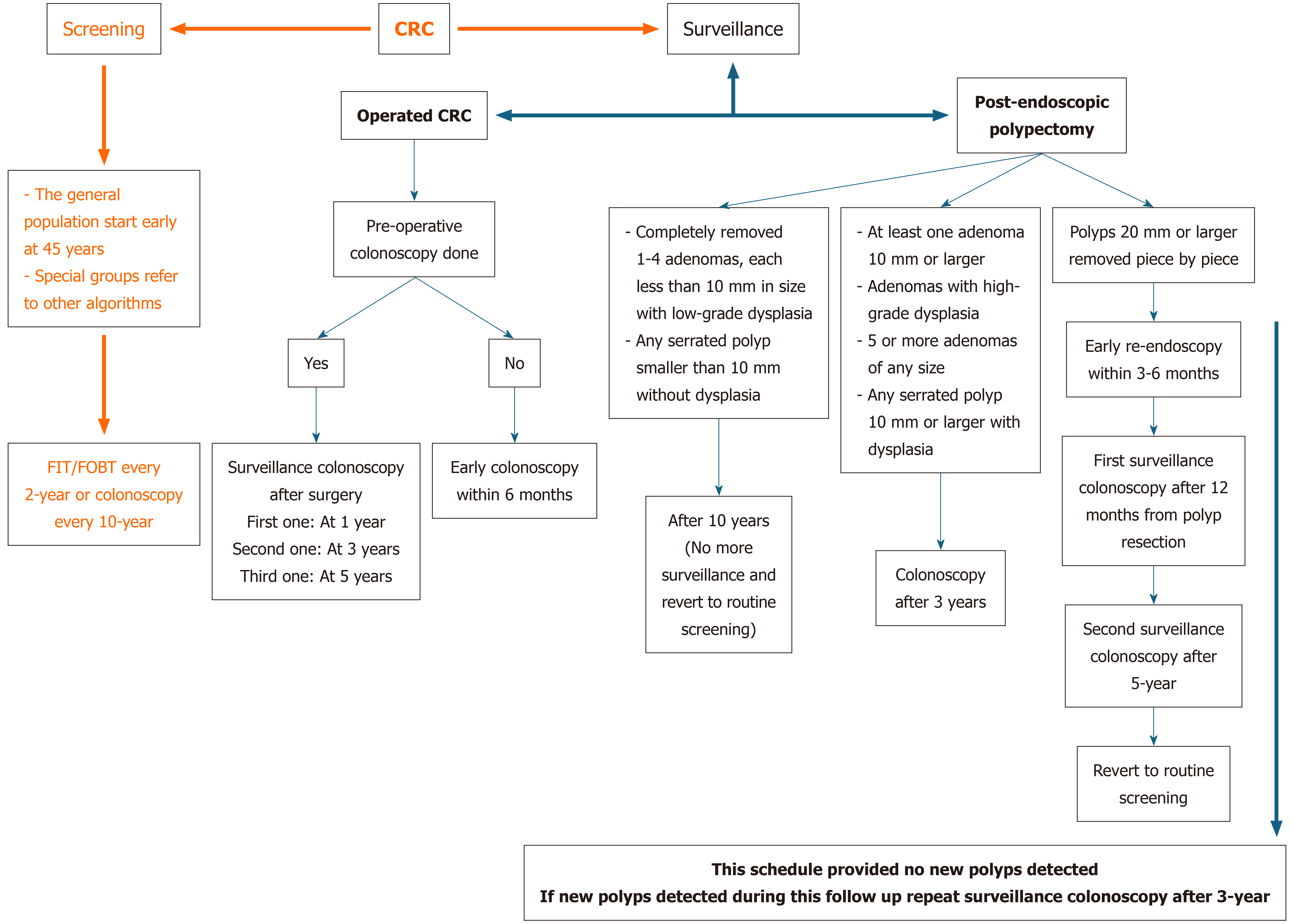
| Post-endoscopic polypectomy | Depend on the size, morphology and histology of the polyp (Figure 2) | ||
| Post-surgical resection of CRC | In case of operated obstructive CRC where preoperative colonoscopy was not done, a colonoscopy should be carried out within 3-6 months after surgery; operated cases with pre-operative colonoscopy done should have colonoscopy at 1-year and 3-years from surgery, and once examination is unremarkable, revert to 5-year interval; if polyps are detected during colonoscopy the polyp surveillance intervals protocol should be followed | ||
Gastric cancer (GC) remains a global health challenge, with over a million new cases in 2020. Despite the secular decline, an ageing population is expected to sustain the absolute number of new cases. The decline is uneven, with a high incidence present in several regions. Recent studies also suggest an increased incidence among young adults (below 50 years). GC is a stage-dependent disease with poor survival related to the late detection of advanced symptomatic disease in the majority of patients. The success of population-based endoscopic screening programs in East Asian countries with high GC incidence in improving outcomes by detecting early, curable cancers, as well as the increasing dissemination of endoscopic imaging and resection techniques, has motivated interest in early detection and curative endoscopic resection. Yet the evidence supporting a role for endoscopic screening and surveillance in GC compared to BE and colorectal cancer (CRC) is still evolving, and this calls for a more nuanced approach towards the adoption of screening and surveillance strategies[130].
Timing: In high-risk populations, mainly in East Asia, it is currently practiced to screen people aged 40 years and older in Korea and those ≥ 50 years in Japan every 2 years by upper endoscopy. While in the low-risk population, surveillance of pre-malignant lesions, including atrophic gastritis and gastric IM, may be feasible, aiming to detect dysplasia and early gastric cancer stages can facilitate curative resection. The decision for surveillance should be guided by the additional risk factors such as incomplete gastric IM, extensive gastric IM, advanced Operative Link for Gastritis Intestinal metaplasia Assessment staging, family history of GC, and some other factors such as ethnic risk. European Society of Gastrointestinal Endoscopy[131] and American Gastroenterological Association[132] recommendations are consistent in not recom
Hazard/benefit: In one meta-analysis of observational studies from the Far East, endoscopic screening was associated with a 40% reduction in GC mortality but not GC incidence. This outcome is achieved with highly intensive screening protocols. In contrast, less frequent endoscopic screening (approximately every 4.5 years) does not appear to be effective in reducing GC mortality, as the intervals are too long for the detection of early-stage GC. Gastric IM is an independent risk factor for GC with a pooled incidence rate of progression to GC of 12.4 GC cases per 10000 patient-years. The risk of malignant progression in atrophic gastritis is estimated to range from 0.1% to 0.3% per year, but it may be higher with more severe and extensive atrophic gastritis (higher the Operative Link on Gastritis Assessment and Operative Link on Gastric Intestinal Metaplasia Assessment stages) and concomitant gastric IM[130].
This is a rare malignancy; however, certain risk factors, e.g., familial adenomatous polyposis, should be screened every 2 years starting at the age of 20 (Table 6).
Endoscopic screening of the lower digestive tract is crucial for detecting many premalignant lesions, and surveillance of high-risk patients who already have potentially cancerous pathologies. In fact, many sporadic and inherited pathologic lesions affecting the colon should be focused on by the endoscopic screening and surveillance as described below.
CRC is a preventable disease because many known risk factors can be identified. Hence, screening at-risk as well as average-risk individuals would pick up pre-malignant lesions and early stages of CRC and would reduce and/or prevent advanced CRC. The risk factors include advanced age, certain dietary habits, obesity, long-standing IBD, family history of CRC, familial polyposis, Lynch syndrome, some other hereditary syndromes, and ethnic groups. All individuals with a first-degree relative (defined as a parent, sibling, or child) who were diagnosed with CRC, particularly before the age of 50 years, should be considered at increased risk for CRC. All individuals without a personal history of CRC, IBD, hereditary CRC syndromes, other CRC predisposing conditions, or a family history of CRC should be considered as average risk for CRC[134]. Screening is associated with 52% and 62% reduction in the incidence of CRC and CRC-related mortality, respectively. Colonoscopy is considered the gold standard screening tool for CRC risk patients. It is currently recommended to screen the average risk population at the age of 45 instead of 50 years, because the predictive value of initiating screening at this age promotes 25 more life-years gained per 1000 individuals screened[135].
Timing: The recommended age/time of screening/surveillance differs from one situation to another and is summarized in Table 6. The decision to continue screening/surveillance beyond the age of 75-80 years should be individualized[136]. CRC and post-endoscopic polypectomy surveillance are summarized in Figure 6[137].
The most serious, devastating long-term complication among patients with IBD is CRC. Studies showed a colitis-associated CRC rate of 5% and 40% after 10 years and 25 years of disease duration, respectively[138]. One huge meta-analysis recruiting 116 studies with 54478 UC patients found an overall colitis-associated CRC prevalence of 3.7% among UC patients. The cumulative risk of colitis-associated CRC in this study was 2% after 10 years, 8% at 20 years, and 18% at 30 years[139]. IBD-CRC accounts for up to 15% of annual deaths in patients with IBD[140].
Timing: Initial screening colonoscopy for dysplasia among patients with IBD should be performed at 8-10 years after disease diagnosis in all people with colonic IBD, and immediately on diagnosis of primary sclerosing cholangitis (PSC). Consequently, staging biopsies should be taken from different colonic segments to assess histologic disease activity and extent and to help guide future surveillance intervals. After a negative screening colonoscopy, surveillance colonoscopy should be performed every 1-5years based on risk factors for CRC, considering current and prior burden of colonic inflammation, family history of CRC, PSC, history of colorectal dysplasia, and frequency and quality of prior surveillance examinations. For most patients in remission, a 2-year or 3-year interval is appropriate unless additional high-risk factors are present, in which case a 1-year follow up should be performed[141] (Table 7).
| Visible dysplastic lesions | ||
| Lesion | Treatment | Colonoscopy surveillance intervals |
| Small < 2 cm + resectable + no histologic features of invasive CRC | Endoscopic resection + continuous surveillance | Every 3-6 months in HGD or incomplete resection; 12 months: More than 1 cm and LGD; 24 months if < 1 cm, pedunculated, LGD |
| Large ≥ 2 cm, complex lesions, incomplete resection, after several attempts, local recurrence | Refer to a highly experienced center for resection vs surgery | Every 3-6 months if resected |
| Non-visible dysplastic lesions (detected by non-targeted biopsy) or incompletely delineated lesions on target biopsy should be examined by SDC | ||
| Persistent HGD or multifocal invisible dysplasia | Surgery | |
| Persistent unifocal low-grade invisible dysplasia | Intensive surveillance with SDC | Every 3-6 months in HGD or multifocal dysplasia; 6-12 months: If LGD continues, surveillance till 2 consecutive negative high-quality SDC colonoscopies |
| No dysplasia at index colonoscopy; timing for the next colonoscopy depends on many factors for CRC risk | ||
| 1-year | 2-3 years | 5-year |
| Moderate or severe inflammation at index colonoscopy; family Hx of CRC in FDR < 50 years; PSC; dense pseudo-polyposis; history of invisible dysplasia < 5 years | Mild inflammation at index colonoscopy; strong family Hx of CRC but no FDR < 50 years; features of prior severe colitis (moderate pseudo-polyposis + extensive scarring); history of invisible dysplasia > 5 years | Continuous disease remission since the last colonoscopy with mucosal healing on the current examination, plus either: ≥ 2 consecutive exams without dysplasia; minimal historical colitis extent (ulcerative proctitis or < 1/3 in Crohn’s disease) |
The process of colonoscopy screening/surveillance should adhere to best practice to optimize dysplasia detection, including control of inflammation, use of high-definition endoscopes, bowel preparation, careful washing and in-section of all colorectal mucosa, and targeted sampling of any suspicious mucosal irregularities. The term targeted biopsies refers to sampling all mucosal lesions suspicious for dysplasia or mucosal areas inexplicably different from the surrounding mucosa. Endoscopic resection is preferred to biopsies when lesions are clearly demarcated without stigmata of invasive cancer or submucosal fibrosis. Mucosal biopsies surrounding a resected lesion are not required unless there are concerns about resection completeness. Adequate delineation of the mucosal lesion can be achieved using spray-dye chromoendoscopy (SDC) or virtual chromoendoscopy (VC) through the high-definition scopes.
The term nontargeted biopsies refers to random sampling of the colonic mucosa when no clear suspicious lesions are seen. Extensive nontargeted biopsies (roughly 4 adequately spaced biopsies every 10 cm) should be taken from flat colorectal mucosa in areas previously affected by colitis when white light endoscopy is used without SDC or VC. Additional biopsies should be taken from areas of prior dysplasia or poor mucosal visibility. Nontargeted biopsies are not routinely required if SDC or VC is performed using a high-definition endoscope, but should be considered if there is a history of dysplasia or PSC.
All clearly delineated dysplastic-appearing lesions without stigmata of invasive cancer or significant submucosal fibrosis should be considered for endoscopic resection while referral to surgery should be considered for clear indications. After endoscopic resection of a visible dysplastic lesion, the next surveillance examination should occur at 3-6 months for the highest-risk lesions and no more than 24 months after for the lowest risk, with the latter group including simple sub-centimeter sessile or pedunculated low-grade adenomas[141] (Table 7). Pouch surveillance should be performed at least annually in those at high risk for developing colorectal dysplasia (prior CRC or dysplasia, PSC), as well as in those with persistent moderate to severe pouchitis and/or pre-pouch ileitis (to assess for treatment response). On the contrary, surveillance intervals for lower-risk patients should be individualized.
Hazard/benefit: Ameta-analysis, conducted in 2018, highlighted the importance of appropriate surveillance[132]. Bye et al[14] assessed the effectiveness of endoscopic surveillance in decreasing IBD-CRC-related mortality and found that the cancer detection rate was significantly higher in the non-surveillance group (3.2%) than in the surveillance group (1.8%). Moreover, CRC-associated death was significantly lower in the surveillance group (8.5%, 15/176) than in the non-surveillance group (22.3%, 79/354). In addition, the early-stage CRC detection rate was significantly higher in the surveillance group (15.5%) than in the non-surveillance group (7.7%). Patients with IBD-related CRCs exhibit a poorer prognosis than those with sporadic CRCs, owing to their aggressive histological characteristics and lower curative resection rate, and hence need to be addressed early[142].
Hepato-pancreatico-biliary emergencies often arise from cholelithiasis (acute cholecystitis, pancreatitis, and cholangitis) and non-traumatic injuries (common bile duct or duodenal)[143]. With overlapping presentations and diverse therapeutic options, timely decision-making is essential to reduce morbidity and mortality.
Acute cholangitis is a bacterial infection of the biliary system, usually due to obstruction of the biliary or hepatic duct. Diagnosis relies on clinical features, labs, and imaging. Classically, patients present clinically with high fever persisting for more than 24 hours, abdominal pain, and jaundice (Charcot’s triad), laboratory studies suggestive of infection and hyperbilirubinemia, and abnormal imaging studies suggestive of biliary obstruction. Timely treatment is critical, as delays increase mortality. Severity is graded as follows[144].
Grade I (mild): Patients have no organ dysfunction and respond to the initial antibiotic treatment.
Grade II (moderate): Patients have no organ dysfunction, but do not respond to the initial antibiotic therapy and have any 2 of these 5 conditions: (1) Leukocytosis or leukopenia; (2) Fever (≥ 39 °C); (3) Elderly (age > 75 years); (4) Hyperbilirubinemia (bilirubin ≥ 5 mg/dL); and (5) Hypoalbuminemia (< 0.7 × lower limit of normal).
Grade III (severe): Patients do not respond to initial medical treatment and have at least one of the following organ/system dysfunction: (1) Cardiovascular system: Hypotension requiring dopamine infusion ≥ 5 μg/kg per minute, or any dose of norepinephrine; (2) Nervous system: Disturbance of consciousness; (3) Respiratory system: PaO2/FiO2 ratio < 300; (4) Renal system: Oliguria, serum creatinine > 2 mg/dL; (5) Hepatic system: INR > 1.5; and (6) Hematological system: Platelet count < 100000/mm3.
The impact of ERCP is related to disease severity. In patients with severe sepsis or septic shock, prompt ERCP is crucial for source control. The Tokyo 2018 guidelines recommend performing ERCP as soon as possible, ideally within 24-48 hours after stabilization in cases of severe cholangitis[145]. The 2021 American Society for Gastrointestinal Endoscopy guidelines recommend ERCP over percutaneous drainage, with biliary decompression ideally performed within 48 hours. They also advise combining sphincterotomy and stone removal with drainage, when possible, unless the patient is too unstable for more extensive endoscopic intervention[146].
Timing: ERCP is indicated within the first 48 hours for biliary drainage in all patients with moderate or severe cholangitis, and in mild cases that fail conservative management. Elective laparoscopic cholecystectomy is especially indicated in choledocholithiasis. Guidelines[145,146] recommend interval laparoscopic cholecystectomy during the same hospitalization or shortly after recovery once the patient is fit for surgery.
Acute pancreatitis is an inflammatory disorder that may lead to local injury, systemic inflammation, and organ failure, with an overall mortality of about 5% (higher in severe cases). It commonly presents with abdominal pain, nausea, and vomiting. Gallstones and alcohol account for approximately 80% of cases, while less common causes include drugs, pancreatic tumors, and hypertriglyceridemia.
Timely decisions: The updated guidelines[147,148] recommend the following: (1) Fluid resuscitation: Start immediately at presentation, using a goal-directed approach (avoid both under-and over-hydration); (2) Feeding: Begin early enteral feeding (within 24 hours) as tolerated; (3) Cholecystectomy (for acute biliary pancreatitis): Perform once the patient is stabilized (same admission), to prevent recurrence; and (4) ERCP is reserved for those with cholangitis or persistent biliary obstruction; otherwise, conservative management suffices. Antibiotics for predicted severe necrotizing AP only, and avoid prophylactic use.
Acute cholecystitis is gallbladder inflammation, mostly due to gallstones (90%-95% of cases). Acalculous cholecystitis causes < 10% of cases, and usually occurs in elderly, postoperative, or critically ill patients. In calculous cholecystitis, cystic duct obstruction by a gallstone leads to pain and inflammation, which, if not timely treated, may progress to necrosis, gangrene, or perforation[149]. Its severity is graded as follows[150].
Grade III (severe): Is associated with dysfunction of any one of the following organs/systems: (1) Cardiovascular dysfunction: Hypotension requiring treatment with dopamine ≥ 5 μg/kg per minute, or any dose of norepinephrine; (2) Neurological dysfunction: Decreased level of consciousness; (3) Respiratory dysfunction: PaO2/FiO2 ratio < 300; (4) Renal dysfunction: Oliguria, creatinine > 2.0 mg/dL; (5) Hepatic dysfunction: Prothrombin time-INR > 1.5; and (6) Hematological dysfunction: Platelet count < 100000/mm3.
Grade II (moderate): Is associated with any one of the following conditions: (1) Elevated white blood cell count (> 18000/mm3); (2) Palpable tender mass in the right upper abdominal quadrant; (3) Duration of complaints > 72 hours; and (4) Marked local inflammation (gangrenous cholecystitis, pericholecystic abscess, hepatic abscess, biliary peritonitis, emphysematous cholecystitis).
Grade I (mild): Does not meet the criteria of grade II nor grade III.
According to the Tokyo guidelines 2018[145], management algorithms are based on the degree of gallbladder inflammation and disease severity. Laparoscopic cholecystectomy is recommended for grade I and grade II cholecystitis, and may also be performed in grade III cases if there are no negative predictive factors, the patient has good performance status, organ dysfunction is favorable, and the procedure is undertaken in an advanced center with intensive care support and expertise in advanced laparoscopy.
Time-sensitive decision: So, in terms of time-sensitive intervention, early cholecystectomy (within the first week) should be performed to reduce cost, hospital stay, and possibility of recurrence in all mild and moderate cases, but delayed in severe cases till improvement of the patient’s general condition.
Hazard/benefit: Meta-analyses comparing early and delayed surgery found no differences in morbidity and mortality, or in the percentage of bile duct injury with either open or laparoscopic techniques. However, the length of hospital stays, and cost/benefits of early cholecystectomy (performed within 7-10 days of symptom onset)[151] are better than those of delayed surgery (antibiotic treatment and surgery after 5-6 weeks)[152,153].
Among the diagnostic interventions are the diagnostic paracentesis in ascitic patients at presentation to decide chemoprophylaxis vs diagnosis of SBP, and ideally should be performed within 12 hours of admission. Once SBP is diagnosed, a second diagnostic paracentesis is recommended at 48 hours from the first one to evaluate the treatment efficacy and decide the need to change the treatment. Another time-sensitive diagnostic intervention is the diagnostic upper endoscopy for patients with acute UGIB and impacted ingested foreign bodies in the upper GI tract. In the first clinical scenario delay in doing endoscopy beyond 12-24 hours is associated with negative outcomes, while in the second scenario, endoscopy should be done within a few hours. There are many other conditions whose diagnosis should be achieved within a few hours, e.g., MVT, acute cholangitis, and acute pancreatitis, based on a combination of many diagnostic modalities. Early diagnosis and prompt initiation of treatment in these conditions are associated with good outcomes.
There are numerous therapeutic interventions that should be carried out in a timely manner to avoid the drawbacks for the patients as well as the healthcare systems. ALF is a medical emergency that should be treated in a fully equipped center, and the early referral for ELT should not be overlooked. Many clinical scenarios necessitate the administration of HBV immunoglobulin with or without HBV vaccine to prevent HBV infection. Many drugs are hepatotoxic, and any offending drug should be discontinued. Immediate interventions, e.g., administration of N-acetyl cysteine in ace
There is growing recognition that timely diagnostic and therapeutic decisions in hepatology and gastroenterology significantly influence morbidity, mortality, and long-term outcomes. However, several important gaps in evidence persist. First, most recommendations on optimal timing - for example, paracentesis in suspected SBP, CTA for mesenteric ischemia, or endoscopy in GI bleeding - are based on observational studies or expert opinion rather than high-quality prospective or randomized trials. Precise, evidence-based thresholds defining what constitutes “timely” vs “delayed” care remain poorly established, and guideline recommendations vary widely. Uncertainty regarding the timing and benefit of therapies such as corticosteroids or NAC in non–acetaminophen DILI necessitates more research in this topic.
System-level contributors to delays are recognized, yet interventional studies testing pathways to reduce delays are scarce. Moreover, few studies link timing to long-term outcomes such as transplant candidacy, quality of life, and cost-effectiveness. Key research priorities may include: Prospective multicenter studies to define evidence-based timing windows for major conditions; standardized definitions and reporting metrics for timing; pragmatic and adaptive trials testing timing of therapies such as NAC or early anticoagulation; implementation studies evaluating fast-track pathways; development of predictive tools or electronic triggers to identify patients needing urgent intervention; and studies focusing on long-term outcomes, and cost-effectiveness. Translational research exploring biomarkers that define biological “windows of reversibility” is also needed. Together, these priorities can transform timing recommendations from expert-driven to evidence-driven standards of care.
In the field of hepatology and gastroenterology there are many diagnostic as well as therapeutic interventions that should be carried out within a certain time frame to achieve the best outcomes and any delay in performing these interventions at the defined time is associated with harm to the patient that is not limited to loss of the chance of cure, development of complications, prolonged hospital stay, lost probability of transplant candidacy, but also mortality. These interventions are gathered under the title “time sensitive interventions” to emphasize the importance of time as the o’clock in knocking behind the clinician, however the evidence-based summaries for these interventions are not collected in one single handout and that is why we tried to summarize the evidence of these interventions in this file to help the practitioners to take their decisions in a timely fashion that is consequently reflected as improved outcomes.
The authors would thank all colleagues who expressed their support during the preparation of this review.
| 1. | Shingina A, Mukhtar N, Wakim-Fleming J, Alqahtani S, Wong RJ, Limketkai BN, Larson AM, Grant L. Acute Liver Failure Guidelines. Am J Gastroenterol. 2023;118:1128-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 121] [Article Influence: 40.3] [Reference Citation Analysis (2)] |
| 2. | European Association for the Study of the Liver. Clinical practice guidelines panel, Wendon, J; Panel members, Cordoba J, Dhawan A, Larsen FS, Manns M, Samuel D, Simpson KJ, Yaron I; EASL Governing Board representative, Bernardi M. EASL Clinical Practical Guidelines on the management of acute (fulminant) liver failure. J Hepatol. 2017;66:1047-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 816] [Cited by in RCA: 696] [Article Influence: 77.3] [Reference Citation Analysis (4)] |
| 3. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on acute-on-chronic liver failure. J Hepatol. 2023;79:461-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 252] [Article Influence: 84.0] [Reference Citation Analysis (1)] |
| 4. | Amjad W, Thuluvath P, Mansoor M, Dutta A, Ali F, Qureshi W. N-acetylcysteine in non-acetaminophen-induced acute liver failure: a systematic review and meta-analysis of prospective studies. Prz Gastroenterol. 2022;17:9-16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 5. | Fernández J, Bassegoda O, Toapanta D, Bernal W. Acute liver failure: A practical update. JHEP Rep. 2024;6:101131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 6. | Orban C, Agapie M, Bratu A, Jafal M, Duțu M, Popescu M. No Significant Beneficial Effects of Intravenous N-Acetylcysteine on Patient Outcome in Non-Paracetamol Acute Liver Failure: A Meta-Analysis of Randomized Controlled Trials. Biomedicines. 2024;12:1462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Karvellas CJ, Bajaj JS, Kamath PS, Napolitano L, O'Leary JG, Solà E, Subramanian R, Wong F, Asrani SK. AASLD Practice Guidance on Acute-on-chronic liver failure and the management of critically ill patients with cirrhosis. Hepatology. 2024;79:1463-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 88] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 8. | Warrillow S, Fisher C, Bellomo R. Correction and Control of Hyperammonemia in Acute Liver Failure: The Impact of Continuous Renal Replacement Timing, Intensity, and Duration. Crit Care Med. 2020;48:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (2)] |
| 9. | Dong V, Robinson AM, Dionne JC, Cardoso FS, Rewa OG, Karvellas CJ. Continuous renal replacement therapy and survival in acute liver failure: A systematic review and meta-analysis. J Crit Care. 2024;81:154513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 10. | Davenport A, Will EJ, Davison AM. Early changes in intracranial pressure during haemofiltration treatment in patients with grade 4 hepatic encephalopathy and acute oliguric renal failure. Nephrol Dial Transplant. 1990;5:192-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 55] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Veeramachaneni H, Subramanian RM. Combined High-Dose Continuous Renal Replacement Therapy and Plasma Exchange in the Management of Severe Multiorgan System Dysfunction Associated With Acute Liver Failure. Hepatology. 2021;74:1124-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Warrillow SJ, Bellomo R. Preventing cerebral oedema in acute liver failure: the case for quadruple-H therapy. Anaesth Intensive Care. 2014;42:78-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Beran A, Mohamed MFH, Shaear M, Nayfeh T, Mhanna M, Srour O, Nawras M, Mentrose JA, Assaly R, Kubal CA, Ghabril MS, Hernaez R, Patidar KR. Plasma exchange for acute and acute-on-chronic liver failure: A systematic review and meta-analysis. Liver Transpl. 2024;30:127-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 14. | Reuben A, Tillman H, Fontana RJ, Davern T, McGuire B, Stravitz RT, Durkalski V, Larson AM, Liou I, Fix O, Schilsky M, McCashland T, Hay JE, Murray N, Shaikh OS, Ganger D, Zaman A, Han SB, Chung RT, Smith A, Brown R, Crippin J, Harrison ME, Koch D, Munoz S, Reddy KR, Rossaro L, Satyanarayana R, Hassanein T, Hanje AJ, Olson J, Subramanian R, Karvellas C, Hameed B, Sherker AH, Robuck P, Lee WM. Outcomes in Adults With Acute Liver Failure Between 1998 and 2013: An Observational Cohort Study. Ann Intern Med. 2016;164:724-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 317] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 15. | O'Grady JG, Alexander GJ, Hayllar KM, Williams R. Early indicators of prognosis in fulminant hepatic failure. Gastroenterology. 1989;97:439-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1699] [Cited by in RCA: 1303] [Article Influence: 35.2] [Reference Citation Analysis (5)] |
| 16. | Nilsen O, Fisher C, Warrillow S. Update on the management of acute liver failure. Curr Opin Crit Care. 2025;31:219-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Fontana RJ, Liou I, Reuben A, Suzuki A, Fiel MI, Lee W, Navarro V. AASLD practice guidance on drug, herbal, and dietary supplement-induced liver injury. Hepatology. 2023;77:1036-1065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 157] [Article Influence: 52.3] [Reference Citation Analysis (5)] |
| 18. | Davidson DG, Eastham WN. Acute liver necrosis following overdose of paracetamol. Br Med J. 1966;2:497-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 355] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 19. | Kholili U, Haryo Nugroho Y, Sugihartono T, A. Nusi I, Boedi Setiawan P. Liver injury associated with Acetaminophen: A Review. Res J Pharm Technol. 2023. [DOI] [Full Text] |
| 20. | Chiew AL, Gluud C, Brok J, Buckley NA. Interventions for paracetamol (acetaminophen) overdose. Cochrane Database Syst Rev. 2018;2:CD003328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 58] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 21. | Alizadeh N, Yaryari AM, Behnoush AH, Raoufinejad K, Behnoush B. Late N-acetylcysteine for successful recovery of acetaminophen-related acute liver failure: A case report. Clin Case Rep. 2023;11:e7946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 22. | Chomchai S, Mekavuthikul P, Phuditshinnapatra J, Chomchai C. Sensitivity of dose-estimations for acute acetaminophen overdose in predicting hepatotoxicity risk using the Rumack-Matthew Nomogram. Pharmacol Res Perspect. 2022;10:e00920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 23. | Rumack BH, Matthew H. Acetaminophen poisoning and toxicity. Pediatrics. 1975;55:871-876. [PubMed] |
| 24. | Rumack BH, Peterson RC, Koch GG, Amara IA. Acetaminophen overdose. 662 cases with evaluation of oral acetylcysteine treatment. Arch Intern Med. 1981;141:380-385. [PubMed] [DOI] [Full Text] |
| 25. | Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N Engl J Med. 1988;319:1557-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 782] [Cited by in RCA: 738] [Article Influence: 19.4] [Reference Citation Analysis (4)] |
| 26. | Lei S, Gu R, Ma X. Clinical perspectives of isoniazid-induced liver injury. Liver Res. 2021;5:45-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 27. | Hassan HM, Guo HL, Yousef BA, Luyong Z, Zhenzhou J. Hepatotoxicity mechanisms of isoniazid: A mini-review. J Appl Toxicol. 2015;35:1427-1432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 28. | Hayashi PH, Fontana RJ, Chalasani NP, Stolz AA, Talwalkar JA, Navarro VJ, Lee WM, Davern TJ, Kleiner DE, Gu J, Hoofnagle JH; US Drug-Induced Liver Injury Network Investigators. Under-reporting and Poor Adherence to Monitoring Guidelines for Severe Cases of Isoniazid Hepatotoxicity. Clin Gastroenterol Hepatol. 2015;13:1676-82.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 29. | Saukkonen J. Challenges in reintroducing tuberculosis medications after hepatotoxicity. Clin Infect Dis. 2010;50:840-842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Centers for Disease Control and Prevention (CDC). Severe isoniazid-associated liver injuries among persons being treated for latent tuberculosis infection - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:224-229. [PubMed] |
| 31. | Jiang SX, Hussaini T, Yoshida EM. N-acetylcysteine for non-acetaminophen induced acute liver failure: A review. Saudi J Gastroenterol. 2022;28:85-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 32. | Frenkel A, Smadar Shneyour R, Shiloh A, Morad M, Shimoni-Rachmilev O, Dreiher J. Adherence to monitoring of patients treated with amiodarone: a nationwide study. Front Med (Lausanne). 2024;11:1408799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 33. | Rätz Bravo AE, Drewe J, Schlienger RG, Krähenbühl S, Pargger H, Ummenhofer W. Hepatotoxicity during rapid intravenous loading with amiodarone: Description of three cases and review of the literature. Crit Care Med. 2005;33:128-34; discussion 245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 58] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 34. | Mohamed M, Al-Hillan A, Flores M, Kaunzinger C, Mushtaq A, Asif A, Hossain M. Concomitant Acute Hepatic Failure and Renal Failure Induced by Intravenous Amiodarone: A Case Report and Literature Review. Gastroenterology Res. 2020;13:40-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Waldhauser KM, Török M, Ha HR, Thomet U, Konrad D, Brecht K, Follath F, Krähenbühl S. Hepatocellular toxicity and pharmacological effect of amiodarone and amiodarone derivatives. J Pharmacol Exp Ther. 2006;319:1413-1423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 90] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 36. | Lahbabi M, Aqodad N, Ibrahimi A, Lahlou M, Aqodad H. Acute hepatitis secondary to parenteral amiodarone does not preclude subsequent oral therapy. World J Hepatol. 2012;4:196-198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Black GW. Clinical features of halothane and enflurane. Acta Anaesthesiol Belg. 1980;31:207-214. [PubMed] |
| 38. | Safari S, Motavaf M, Seyed Siamdoust SA, Alavian SM. Hepatotoxicity of halogenated inhalational anesthetics. Iran Red Crescent Med J. 2014;16:e20153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 39. | Neuberger J. Halothane hepatitis. Eur J Gastroenterol Hepatol. 1998;10:631-633. [PubMed] |
| 40. | Habibollahi P, Mahboobi N, Esmaeili S, Safari S, Dabbagh A, Alavian SM. Halothane-induced hepatitis: A forgotten issue in developing countries: Halothane-induced hepatitis. Hepat Mon. 2011;11:3-6. [PubMed] |
| 41. | Schmidt S, Messner CJ, Gaiser C, Hämmerli C, Suter-Dick L. Methotrexate-Induced Liver Injury Is Associated with Oxidative Stress, Impaired Mitochondrial Respiration, and Endoplasmic Reticulum Stress In Vitro. Int J Mol Sci. 2022;23:15116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 47] [Reference Citation Analysis (0)] |
| 42. | Estève MA, Devictor-Pierre B, Galy G, André N, Coze C, Lacarelle B, Bernard JL, Monjanel-Mouterde S. Severe acute toxicity associated with high-dose methotrexate (MTX) therapy: use of therapeutic drug monitoring and test-dose to guide carboxypeptidase G2 rescue and MTX continuation. Eur J Clin Pharmacol. 2007;63:39-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | Floyd J, Mirza I, Sachs B, Perry MC. Hepatotoxicity of chemotherapy. Semin Oncol. 2006;33:50-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 143] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 44. | Dealberto MJ. Valproate-induced hyperammonaemic encephalopathy: review of 14 cases in the psychiatric setting. Int Clin Psychopharmacol. 2007;22:330-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 45. | Dupuis RE, Lichtman SN, Pollack GM. Acute valproic acid overdose. Clinical course and pharmacokinetic disposition of valproic acid and metabolites. Drug Saf. 1990;5:65-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 36] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Bryant AE 3rd, Dreifuss FE. Valproic acid hepatic fatalities. III. U.S. experience since 1986. Neurology. 1996;46:465-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 200] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 47. | Matsumoto J, Ogawa H, Maeyama R, Okudaira K, Shinka T, Kuhara T, Matsumoto I. Successful treatment by direct hemoperfusion of coma possibly resulting from mitochondrial dysfunction in acute valproate intoxication. Epilepsia. 1997;38:950-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 21] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 48. | Perrott J, Murphy NG, Zed PJ. L-carnitine for acute valproic acid overdose: a systematic review of published cases. Ann Pharmacother. 2010;44:1287-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 49. | Allen RJ. Valproic acid toxicity. Am J Dis Child. 1985;139:648-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 50. | Ingels M, Beauchamp J, Clark RF, Williams SR. Delayed valproic acid toxicity: a retrospective case series. Ann Emerg Med. 2002;39:616-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 51. | Farrar HC, Herold DA, Reed MD. Acute valproic acid intoxication: enhanced drug clearance with oral-activated charcoal. Crit Care Med. 1993;21:299-301. [PubMed] |
| 52. | Sharifzadeh S, Mohammadpour AH, Tavanaee A, Elyasi S. Antibacterial antibiotic-induced drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome: a literature review. Eur J Clin Pharmacol. 2021;77:275-289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 53. | Husain Z, Reddy BY, Schwartz RA. DRESS syndrome: Part I. Clinical perspectives. J Am Acad Dermatol. 2013;68:693.e1-14; quiz 706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 273] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 54. | Santiago LG, Morgado FJ, Baptista MS, Gonçalo M. Hypersensitivity to antibiotics in drug reaction with eosinophilia and systemic symptoms (DRESS) from other culprits. Contact Dermatitis. 2020;82:290-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Ronis MJJ, Pedersen KB, Watt J. Adverse Effects of Nutraceuticals and Dietary Supplements. Annu Rev Pharmacol Toxicol. 2018;58:583-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 187] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 56. | Woo SM, Davis WD, Aggarwal S, Clinton JW, Kiparizoska S, Lewis JH. Herbal and dietary supplement induced liver injury: Highlights from the recent literature. World J Hepatol. 2021;13:1019-1041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 57. | Wong GL, Lemoine M. The 2024 updated WHO guidelines for the prevention and management of chronic hepatitis B: Main changes and potential implications for the next major liver society clinical practice guidelines. J Hepatol. 2025;82:918-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 46.0] [Reference Citation Analysis (2)] |
| 58. | Guidelines for the prevention, diagnosis, care and treatment for people with chronic hepatitis B infection [Internet]. Geneva: World Health Organization; 2024 Mar- . [PubMed] |
| 59. | Weng MK, Doshani M, Khan MA, Frey S, Ault K, Moore KL, Hall EW, Morgan RL, Campos-Outcalt D, Wester C, Nelson NP. Universal Hepatitis B Vaccination in Adults Aged 19-59 Years: Updated Recommendations of the Advisory Committee on Immunization Practices - United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:477-483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 139] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 60. | Fox AN, Terrault NA. The option of HBIG-free prophylaxis against recurrent HBV. J Hepatol. 2012;56:1189-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (3)] |
| 61. | Duvoux C, Belli LS, Fung J, Angelico M, Buti M, Coilly A, Cortesi P, Durand F, Féray C, Fondevila C, Lebray P, Martini S, Nevens F, Polak WG, Rizzetto M, Volpes R, Zoulim F, Samuel D, Berenguer M. 2020 position statement and recommendations of the European Liver and Intestine Transplantation Association (ELITA): management of hepatitis B virus-related infection before and after liver transplantation. Aliment Pharmacol Ther. 2021;54:583-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 62. | Kumar N, Choudhary NS. Managing HBV and HCV Infection Pre- and Post-liver Transplant. J Clin Exp Hepatol. 2024;14:101287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (3)] |
| 63. | Wang SH, Loh PY, Lin TL, Lin LM, Li WF, Lin YH, Lin CC, Chen CL. Active immunization for prevention of De novo hepatitis B virus infection after adult living donor liver transplantation with a hepatitis B core antigen-positive graft. Liver Transpl. 2017;23:1266-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (2)] |
| 64. | Rosenau J, Hooman N, Hadem J, Rifai K, Bahr MJ, Philipp G, Tillmann HL, Klempnauer J, Strassburg CP, Manns MP. Failure of hepatitis B vaccination with conventional HBsAg vaccine in patients with continuous HBIG prophylaxis after liver transplantation. Liver Transpl. 2007;13:367-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 55] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 65. | Aithal GP, Palaniyappan N, China L, Härmälä S, Macken L, Ryan JM, Wilkes EA, Moore K, Leithead JA, Hayes PC, O'Brien AJ, Verma S. Guidelines on the management of ascites in cirrhosis. Gut. 2021;70:9-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 328] [Cited by in RCA: 288] [Article Influence: 57.6] [Reference Citation Analysis (1)] |
| 66. | Beran A, Mohamed MFH, Vargas A, Aboursheid T, Aziz M, Hernaez R, Patidar KR, Nephew LD, Desai AP, Orman E, Chalasani N, Ghabril MS. Early Diagnostic Paracentesis Improves Outcomes of Hospitalized Patients With Cirrhosis and Ascites: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2024;119:2259-2266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 67. | Tocia C, Dumitru A, Alexandrescu L, Popescu R, Dumitru E. Timing of paracentesis and outcomes in hospitalized patients with decompensated cirrhosis. World J Hepatol. 2020;12:1267-1275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 68. | Sherman Z, Soltani A, Steel P, Jesudian A. Time-Sensitive Interventions in Hospitalized Patients With Cirrhosis. Clin Liver Dis (Hoboken). 2020;15:36-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 69. | European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53:397-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1309] [Cited by in RCA: 1141] [Article Influence: 71.3] [Reference Citation Analysis (6)] |
| 70. | Abusneineh B, Garg S, Janbain M, Davitkov P, Falck-ytter Y. Su1705 Paracentesis Within 12 Hours of Admission Improves Outcomes in Patients With Cirrhosis. Gastroenterology. 2013;144:S-1001. [DOI] [Full Text] |
| 71. | Kim JJ, Tsukamoto MM, Mathur AK, Ghomri YM, Hou LA, Sheibani S, Runyon BA. Delayed paracentesis is associated with increased in-hospital mortality in patients with spontaneous bacterial peritonitis. Am J Gastroenterol. 2014;109:1436-1442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 142] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 72. | Karvellas CJ, Abraldes JG, Arabi YM, Kumar A; Cooperative Antimicrobial Therapy of Septic Shock (CATSS) Database Research Group. Appropriate and timely antimicrobial therapy in cirrhotic patients with spontaneous bacterial peritonitis-associated septic shock: a retrospective cohort study. Aliment Pharmacol Ther. 2015;41:747-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 83] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 73. | Laine L, Barkun AN, Saltzman JR, Martel M, Leontiadis GI. ACG Clinical Guideline: Upper Gastrointestinal and Ulcer Bleeding. Am J Gastroenterol. 2021;116:899-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 392] [Article Influence: 78.4] [Reference Citation Analysis (2)] |
| 74. | Gralnek IM, Stanley AJ, Morris AJ, Camus M, Lau J, Lanas A, Laursen SB, Radaelli F, Papanikolaou IS, Cúrdia Gonçalves T, Dinis-Ribeiro M, Awadie H, Braun G, de Groot N, Udd M, Sanchez-Yague A, Neeman Z, van Hooft JE. Endoscopic diagnosis and management of nonvariceal upper gastrointestinal hemorrhage (NVUGIH): European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2021. Endoscopy. 2021;53:300-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 351] [Article Influence: 70.2] [Reference Citation Analysis (8)] |
| 75. | Lau JYW, Yu Y, Tang RSY, Chan HCH, Yip HC, Chan SM, Luk SWY, Wong SH, Lau LHS, Lui RN, Chan TT, Mak JWY, Chan FKL, Sung JJY. Timing of Endoscopy for Acute Upper Gastrointestinal Bleeding. N Engl J Med. 2020;382:1299-1308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 223] [Article Influence: 37.2] [Reference Citation Analysis (4)] |
| 76. | Tejedor-Tejada J, Hermida B, Camblor C, Emara M, Youssef Mohamed S, Perez-Pariente JM. Timing of endoscopy in high-risk patients with acute upper gastrointestinal bleeding: Results of a multicenter international cohort study. Gastroenterol Hepatol. 2025;48:502455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 77. | Barkun AN, Almadi M, Kuipers EJ, Laine L, Sung J, Tse F, Leontiadis GI, Abraham NS, Calvet X, Chan FKL, Douketis J, Enns R, Gralnek IM, Jairath V, Jensen D, Lau J, Lip GYH, Loffroy R, Maluf-Filho F, Meltzer AC, Reddy N, Saltzman JR, Marshall JK, Bardou M. Management of Nonvariceal Upper Gastrointestinal Bleeding: Guideline Recommendations From the International Consensus Group. Ann Intern Med. 2019;171:805-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 391] [Article Influence: 55.9] [Reference Citation Analysis (9)] |
| 78. | Strate LL, Gralnek IM. ACG Clinical Guideline: Management of Patients With Acute Lower Gastrointestinal Bleeding. Am J Gastroenterol. 2016;111:459-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 401] [Cited by in RCA: 318] [Article Influence: 31.8] [Reference Citation Analysis (4)] |
| 79. | Niikura R, Nagata N, Yamada A, Honda T, Hasatani K, Ishii N, Shiratori Y, Doyama H, Nishida T, Sumiyoshi T, Fujita T, Kiyotoki S, Yada T, Yamamoto K, Shinozaki T, Takata M, Mikami T, Mabe K, Hara K, Fujishiro M, Koike K. Efficacy and Safety of Early vs Elective Colonoscopy for Acute Lower Gastrointestinal Bleeding. Gastroenterology. 2020;158:168-175.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (3)] |
| 80. | ASGE Standards of Practice Committee; Pasha SF, Shergill A, Acosta RD, Chandrasekhara V, Chathadi KV, Early D, Evans JA, Fisher D, Fonkalsrud L, Hwang JH, Khashab MA, Lightdale JR, Muthusamy VR, Saltzman JR, Cash BD. The role of endoscopy in the patient with lower GI bleeding. Gastrointest Endosc. 2014;79:875-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 150] [Article Influence: 12.5] [Reference Citation Analysis (3)] |
| 81. | Nagata N, Ishii N, Manabe N, Tomizawa K, Urita Y, Funabiki T, Fujimori S, Kaise M. Guidelines for Colonic Diverticular Bleeding and Colonic Diverticulitis: Japan Gastroenterological Association. Digestion. 2019;99 Suppl 1:1-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 141] [Article Influence: 20.1] [Reference Citation Analysis (4)] |
| 82. | García-Blázquez V, Vicente-Bártulos A, Olavarria-Delgado A, Plana MN, van der Winden D, Zamora J; EBM-Connect Collaboration. Accuracy of CT angiography in the diagnosis of acute gastrointestinal bleeding: systematic review and meta-analysis. Eur Radiol. 2013;23:1181-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 121] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 83. | Singal AK, Kamath PS, Tefferi A. Mesenteric venous thrombosis. Mayo Clin Proc. 2013;88:285-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 129] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 84. | Sakr M, Barakat E, Abdelhakam S, Dabbous H, Yousuf S, Shaker M, Eldorry A. Epidemiological aspects of Budd-Chiari in Egyptian patients: a single-center study. World J Gastroenterol. 2011;17:4704-4710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 25] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 85. | Hmoud B, Singal AK, Kamath PS. Mesenteric venous thrombosis. J Clin Exp Hepatol. 2014;4:257-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 83] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 86. | Salim S, Ekberg O, Elf J, Zarrouk M, Gottsäter A, Acosta S. Evaluation of direct oral anticoagulants and vitamin K antagonists in mesenteric venous thrombosis. Phlebology. 2019;34:171-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 87. | Björck M, Koelemay M, Acosta S, Bastos Goncalves F, Kölbel T, Kolkman JJ, Lees T, Lefevre JH, Menyhei G, Oderich G; Esvs Guidelines Committee; Kolh P, de Borst GJ, Chakfe N, Debus S, Hinchliffe R, Kakkos S, Koncar I, Sanddal Lindholt J, Vega de Ceniga M, Vermassen F, Verzini F, Document Reviewers, Geelkerken B, Gloviczki P, Huber T, Naylor R. Editor's Choice - Management of the Diseases of Mesenteric Arteries and Veins: Clinical Practice Guidelines of the European Society of Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg. 2017;53:460-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 569] [Cited by in RCA: 474] [Article Influence: 52.7] [Reference Citation Analysis (1)] |
| 88. | Brandt LJ, Boley SJ. AGA technical review on intestinal ischemia. American Gastrointestinal Association. Gastroenterology. 2000;118:954-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 448] [Cited by in RCA: 328] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 89. | Russell CE, Wadhera RK, Piazza G. Mesenteric venous thrombosis. Circulation. 2015;131:1599-1603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 90. | Schoots IG, Koffeman GI, Legemate DA, Levi M, van Gulik TM. Systematic review of survival after acute mesenteric ischaemia according to disease aetiology. Br J Surg. 2004;91:17-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 293] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 91. | Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, Hayee B, Lomer MCE, Parkes GC, Selinger C, Barrett KJ, Davies RJ, Bennett C, Gittens S, Dunlop MG, Faiz O, Fraser A, Garrick V, Johnston PD, Parkes M, Sanderson J, Terry H; IBD guidelines eDelphi consensus group, Gaya DR, Iqbal TH, Taylor SA, Smith M, Brookes M, Hansen R, Hawthorne AB. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1-s106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1995] [Cited by in RCA: 1775] [Article Influence: 253.6] [Reference Citation Analysis (8)] |
| 92. | Dong C, Metzger M, Holsbø E, Perduca V, Carbonnel F. Systematic review with meta-analysis: mortality in acute severe ulcerative colitis. Aliment Pharmacol Ther. 2020;51:8-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 93. | Harbord M, Eliakim R, Bettenworth D, Karmiris K, Katsanos K, Kopylov U, Kucharzik T, Molnár T, Raine T, Sebastian S, de Sousa HT, Dignass A, Carbonnel F; European Crohn’s and Colitis Organisation [ECCO]. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 2: Current Management. J Crohns Colitis. 2017;11:769-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1029] [Cited by in RCA: 919] [Article Influence: 102.1] [Reference Citation Analysis (3)] |
| 94. | Kuriakose Kuzhiyanjal AJ, Limdi JK. Management of acute severe ulcerative colitis-an update for generalist and specialist clinicians. Br Med Bull. 2024;151:3-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 96. | Adams A, Gupta V, Mohsen W, Chapman TP, Subhaharan D, Kakkadasam Ramaswamy P, Kumar S, Kedia S, McGregor CG, Ambrose T, George BD, Palmer R, Brain O, Walsh A, Ahuja V, Travis SPL, Satsangi J. Early management of acute severe UC in the biologics era: development and international validation of a prognostic clinical index to predict steroid response. Gut. 2023;72:433-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 97. | Gisbert JP, Chaparro M. Common Mistakes in Managing Patients with Inflammatory Bowel Disease. J Clin Med. 2024;13:4795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 98. | Spinelli A, Bonovas S, Burisch J, Kucharzik T, Adamina M, Annese V, Bachmann O, Bettenworth D, Chaparro M, Czuber-Dochan W, Eder P, Ellul P, Fidalgo C, Fiorino G, Gionchetti P, Gisbert JP, Gordon H, Hedin C, Holubar S, Iacucci M, Karmiris K, Katsanos K, Kopylov U, Lakatos PL, Lytras T, Lyutakov I, Noor N, Pellino G, Piovani D, Savarino E, Selvaggi F, Verstockt B, Doherty G, Raine T, Panis Y. ECCO Guidelines on Therapeutics in Ulcerative Colitis: Surgical Treatment. J Crohns Colitis. 2022;16:179-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 211] [Article Influence: 52.8] [Reference Citation Analysis (15)] |
| 99. | Holubar SD, Lightner AL, Poylin V, Vogel JD, Gaertner W, Davis B, Davis KG, Mahadevan U, Shah SA, Kane SV, Steele SR, Paquette IM, Feingold DL; Prepared on behalf of the Clinical Practice Guidelines Committee of the American Society of Colon and Rectal Surgeons. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Surgical Management of Ulcerative Colitis. Dis Colon Rectum. 2021;64:783-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (2)] |
| 100. | Desai J, Elnaggar M, Hanfy AA, Doshi R. Toxic Megacolon: Background, Pathophysiology, Management Challenges and Solutions. Clin Exp Gastroenterol. 2020;13:203-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (3)] |
| 101. | Pantel H, Reddy VB. Management of Colonic Emergencies. Surg Clin North Am. 2023;103:1133-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (2)] |
| 102. | Van Eaton J, Hatch QM. Surgical Emergencies in Inflammatory Bowel Disease. Surg Clin North Am. 2024;104:685-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 103. | Darkow A, Boreyko J, Patel M. Clostridioides difficile Infection: A Review of Emerging Practices for Infection Treatment and Prevention of Recurrence. J Transl Gastroenterol. 2024;2:200-213. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 104. | Drozdinsky G, Vronsky D, Atamna A, Ben-Zvi H, Bishara J, Eliakim-Raz N. Early treatment for Clostridioides difficile infection: retrospective cohort study. Intern Emerg Med. 2025;20:189-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 105. | Johnson S, Lavergne V, Skinner AM, Gonzales-Luna AJ, Garey KW, Kelly CP, Wilcox MH. Clinical Practice Guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused Update Guidelines on Management of Clostridioides difficile Infection in Adults. Clin Infect Dis. 2021;73:e1029-e1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 417] [Article Influence: 83.4] [Reference Citation Analysis (0)] |
| 106. | Vinterberg JE, Oddsdottir J, Nye M, Pinton P. Management of Recurrent Clostridioides difficile Infection (rCDI): A Systematic Literature Review to Assess the Feasibility of Indirect Treatment Comparison (ITC). Infect Dis Ther. 2025;14:327-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 107. | Abreu Y Abreu AT, Velarde-Ruiz Velasco JA, Zavala-Solares MR, Remes-Troche JM, Carmona-Sánchez RI, Aldana-Ledesma JM, Camacho-Ortiz A, Contreras-Omaña R, Díaz-Seoane R, Elizondo-Vázquez CT, Garza-González E, Grajales-Figueroa G, Gómez-Escudero O, Jacobo-Karam JS, Morales-Arámbula M, Olivares-Guzmán LO, Sifuentes-Osornio J, Siu-Moguel AG, Soto-Solís R, Valdovinos-García LR, Valdovinos-Díaz MA, Vázquez-Elizondo G, Lazo-de la Vega Jasso SA. Consensus on the prevention, diagnosis, and treatment of Clostridium difficile infection. Rev Gastroenterol Mex (Engl Ed). 2019;84:204-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 108. | Bhangu A, Nepogodiev D, Gupta A, Torrance A, Singh P; West Midlands Research Collaborative. Systematic review and meta-analysis of outcomes following emergency surgery for Clostridium difficile colitis. Br J Surg. 2012;99:1501-1513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 107] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 109. | Calbo E, Hernández S, Sopena N, Castellá L, Lopez-Contreras J, Cuquet J, López-Sánchez M, Pérez R, Gudiol C, Coloma A, Marimón M, Espinach J, Andres M, Martos P, Carvalho Rodrigues G, Limón E. Late poor outcomes of Clostridioides difficile infections in oncological patients: A multicentre cohort study. J Infect Prev. 2023;24:182-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 110. | Tambakis G, Schildkraut T, Delaney I, Gilmore R, Loebenstein M, Taylor A, Holt B, Tsoi EH, Cameron G, Demediuk B, Miller A, Connell W, Wright E, Thompson A, Holmes J. Management of foreign body ingestion in adults: Time to STOP and rethink endoscopy. Endosc Int Open. 2023;11:E1161-E1167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 111. | Becq A, Camus M, Dray X. Foreign body ingestion: dos and don'ts. Frontline Gastroenterol. 2021;12:664-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (10)] |
| 112. | Demiroren K. Management of Gastrointestinal Foreign Bodies with Brief Review of the Guidelines. Pediatr Gastroenterol Hepatol Nutr. 2023;26:1-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 113. | Birk M, Bauerfeind P, Deprez PH, Häfner M, Hartmann D, Hassan C, Hucl T, Lesur G, Aabakken L, Meining A. Removal of foreign bodies in the upper gastrointestinal tract in adults: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2016;48:489-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 553] [Cited by in RCA: 457] [Article Influence: 45.7] [Reference Citation Analysis (3)] |
| 114. | ASGE Standards of Practice Committee; Ikenberry SO, Jue TL, Anderson MA, Appalaneni V, Banerjee S, Ben-Menachem T, Decker GA, Fanelli RD, Fisher LR, Fukami N, Harrison ME, Jain R, Khan KM, Krinsky ML, Maple JT, Sharaf R, Strohmeyer L, Dominitz JA. Management of ingested foreign bodies and food impactions. Gastrointest Endosc. 2011;73:1085-1091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 680] [Cited by in RCA: 540] [Article Influence: 36.0] [Reference Citation Analysis (6)] |
| 115. | Mosca S, Manes G, Martino R, Amitrano L, Bottino V, Bove A, Camera A, De Nucci C, Di Costanzo G, Guardascione M, Lampasi F, Picascia S, Picciotto FP, Riccio E, Rocco VP, Uomo G, Balzano A. Endoscopic management of foreign bodies in the upper gastrointestinal tract: report on a series of 414 adult patients. Endoscopy. 2001;33:692-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 119] [Article Influence: 4.8] [Reference Citation Analysis (3)] |
| 116. | Hoffman RS, Burns MM, Gosselin S. Ingestion of Caustic Substances. N Engl J Med. 2020;382:1739-1748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 117. | Struck MF, Beilicke A, Hoffmeister A, Gockel I, Gries A, Wrigge H, Bernhard M. Acute emergency care and airway management of caustic ingestion in adults: single center observational study. Scand J Trauma Resusc Emerg Med. 2016;24:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 118. | Zargar SA, Kochhar R, Nagi B, Mehta S, Mehta SK. Ingestion of corrosive acids. Spectrum of injury to upper gastrointestinal tract and natural history. Gastroenterology. 1989;97:702-707. [PubMed] |
| 119. | Bird JH, Kumar S, Paul C, Ramsden JD. Controversies in the management of caustic ingestion injury: an evidence-based review. Clin Otolaryngol. 2017;42:701-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 120. | Chirica M, Kelly MD, Siboni S, Aiolfi A, Riva CG, Asti E, Ferrari D, Leppäniemi A, Ten Broek RPG, Brichon PY, Kluger Y, Fraga GP, Frey G, Andreollo NA, Coccolini F, Frattini C, Moore EE, Chiara O, Di Saverio S, Sartelli M, Weber D, Ansaloni L, Biffl W, Corte H, Wani I, Baiocchi G, Cattan P, Catena F, Bonavina L. Esophageal emergencies: WSES guidelines. World J Emerg Surg. 2019;14:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 258] [Cited by in RCA: 179] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 121. | Zargar SA, Kochhar R, Mehta S, Mehta SK. The role of fiberoptic endoscopy in the management of corrosive ingestion and modified endoscopic classification of burns. Gastrointest Endosc. 1991;37:165-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 286] [Article Influence: 8.2] [Reference Citation Analysis (2)] |
| 122. | Agarwal A, Srivastava DN, Madhusudhan KS. Corrosive injury of the upper gastrointestinal tract: the evolving role of a radiologist. Br J Radiol. 2020;93:20200528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 123. | Ryu HH, Jeung KW, Lee BK, Uhm JH, Park YH, Shin MH, Kim HL, Heo T, Min YI. Caustic injury: can CT grading system enable prediction of esophageal stricture? Clin Toxicol (Phila). 2010;48:137-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 124. | Vantanasiri K, Kamboj AK, Kisiel JB, Iyer PG. Advances in Screening for Barrett Esophagus and Esophageal Adenocarcinoma. Mayo Clin Proc. 2024;99:459-473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 125. | Krishnamoorthi R, Singh S, Ragunathan K, A Katzka D, K Wang K, G Iyer P. Risk of recurrence of Barrett's esophagus after successful endoscopic therapy. Gastrointest Endosc. 2016;83:1090-1106.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 95] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 126. | Shaheen NJ, Falk GW, Iyer PG, Souza RF, Yadlapati RH, Sauer BG, Wani S. Diagnosis and Management of Barrett's Esophagus: An Updated ACG Guideline. Am J Gastroenterol. 2022;117:559-587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 424] [Cited by in RCA: 357] [Article Influence: 89.3] [Reference Citation Analysis (1)] |
| 127. | Weusten BLAM, Bisschops R, Dinis-Ribeiro M, di Pietro M, Pech O, Spaander MCW, Baldaque-Silva F, Barret M, Coron E, Fernández-Esparrach G, Fitzgerald RC, Jansen M, Jovani M, Marques-de-Sa I, Rattan A, Tan WK, Verheij EPD, Zellenrath PA, Triantafyllou K, Pouw RE. Diagnosis and management of Barrett esophagus: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2023;55:1124-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 119] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 128. | Ladigan-Badura S, Vangala DB, Engel C, Bucksch K, Hueneburg R, Perne C, Nattermann J, Steinke-Lange V, Rahner N, Schackert HK, Weitz J, Kloor M, Kuhlkamp J, Nguyen HP, Moeslein G, Strassburg C, Morak M, Holinski-Feder E, Buettner R, Aretz S, Loeffler M, Schmiegel W, Pox C, Schulmann K; German Consortium for Familial Intestinal Cancer. Value of upper gastrointestinal endoscopy for gastric cancer surveillance in patients with Lynch syndrome. Int J Cancer. 2021;148:106-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 129. | Weiss JM, Gupta S, Burke CA, Axell L, Chen LM, Chung DC, Clayback KM, Dallas S, Felder S, Gbolahan O, Giardiello FM, Grady W, Hall MJ, Hampel H, Hodan R, Idos G, Kanth P, Katona B, Lamps L, Llor X, Lynch PM, Markowitz AJ, Pirzadeh-Miller S, Samadder NJ, Shibata D, Swanson BJ, Szymaniak BM, Wiesner GL, Wolf A, Yurgelun MB, Zakhour M, Darlow SD, Dwyer MA, Campbell M. NCCN Guidelines® Insights: Genetic/Familial High-Risk Assessment: Colorectal, Version 1.2021. J Natl Compr Canc Netw. 2021;19:1122-1132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 58] [Reference Citation Analysis (1)] |
| 130. | Namasivayam V. Endoscopic screening and surveillance for gastric cancer: challenges and opportunities. Fac Rev. 2023;12:17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 131. | Pimentel-Nunes P, Libânio D, Marcos-Pinto R, Areia M, Leja M, Esposito G, Garrido M, Kikuste I, Megraud F, Matysiak-Budnik T, Annibale B, Dumonceau JM, Barros R, Fléjou JF, Carneiro F, van Hooft JE, Kuipers EJ, Dinis-Ribeiro M. Management of epithelial precancerous conditions and lesions in the stomach (MAPS II): European Society of Gastrointestinal Endoscopy (ESGE), European Helicobacter and Microbiota Study Group (EHMSG), European Society of Pathology (ESP), and Sociedade Portuguesa de Endoscopia Digestiva (SPED) guideline update 2019. Endoscopy. 2019;51:365-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 747] [Article Influence: 106.7] [Reference Citation Analysis (5)] |
| 132. | Gupta S, Li D, El Serag HB, Davitkov P, Altayar O, Sultan S, Falck-Ytter Y, Mustafa RA. AGA Clinical Practice Guidelines on Management of Gastric Intestinal Metaplasia. Gastroenterology. 2020;158:693-702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 251] [Cited by in RCA: 229] [Article Influence: 38.2] [Reference Citation Analysis (5)] |
| 133. | Joo DC, Kim GH. Optimal Surveillance of Metachronous Gastric Lesion after Endoscopic Resection of Early Gastric Cancer. Gut Liver. 2024;18:781-788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 134. | Issaka RB, Chan AT, Gupta S. AGA Clinical Practice Update on Risk Stratification for Colorectal Cancer Screening and Post-Polypectomy Surveillance: Expert Review. Gastroenterology. 2023;165:1280-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 135. | Barauskaite E, Raciunas A, Vaicekauskas R. Endoscopic Screening and Surveillance of Gastrointestinal Cancer. Cureus. 2025;17:e79274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 136. | Zhang J, Chen G, Li Z, Zhang P, Li X, Gan D, Cao X, Du H, Zhang J, Zhang L, Ye Y. Colonoscopic screening is associated with reduced Colorectal Cancer incidence and mortality: a systematic review and meta-analysis. J Cancer. 2020;11:5953-5970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 137. | Kahi CJ, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, Lieberman D, Levin TR, Robertson DJ, Rex DK; United States Multi-Society Task Force on Colorectal Cancer. Colonoscopy Surveillance After Colorectal Cancer Resection: Recommendations of the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2016;150:758-768.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 153] [Article Influence: 15.3] [Reference Citation Analysis (3)] |
| 138. | Abu-Freha N, Cohen B, Gordon M, Weissmann S, Kestenbaum EH, Vosko S, Abu-Tailakh M, Ben-Shoshan L, Cohen DL, Shirin H. Colorectal cancer among inflammatory bowel disease patients: risk factors and prevalence compared to the general population. Front Med (Lausanne). 2023;10:1225616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 139. | Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526-535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2395] [Cited by in RCA: 2133] [Article Influence: 85.3] [Reference Citation Analysis (5)] |
| 140. | Sato Y, Tsujinaka S, Miura T, Kitamura Y, Suzuki H, Shibata C. Inflammatory Bowel Disease and Colorectal Cancer: Epidemiology, Etiology, Surveillance, and Management. Cancers (Basel). 2023;15:4154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 74] [Article Influence: 24.7] [Reference Citation Analysis (1)] |
| 141. | Murthy SK, Feuerstein JD, Nguyen GC, Velayos FS. AGA Clinical Practice Update on Endoscopic Surveillance and Management of Colorectal Dysplasia in Inflammatory Bowel Diseases: Expert Review. Gastroenterology. 2021;161:1043-1051.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 199] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 142. | Bye WA, Ma C, Nguyen TM, Parker CE, Jairath V, East JE. Strategies for Detecting Colorectal Cancer in Patients with Inflammatory Bowel Disease: A Cochrane Systematic Review and Meta-Analysis. Am J Gastroenterol. 2018;113:1801-1809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 79] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 143. | Miura F, Okamoto K, Takada T, Strasberg SM, Asbun HJ, Pitt HA, Gomi H, Solomkin JS, Schlossberg D, Han HS, Kim MH, Hwang TL, Chen MF, Huang WS, Kiriyama S, Itoi T, Garden OJ, Liau KH, Horiguchi A, Liu KH, Su CH, Gouma DJ, Belli G, Dervenis C, Jagannath P, Chan ACW, Lau WY, Endo I, Suzuki K, Yoon YS, de Santibañes E, Giménez ME, Jonas E, Singh H, Honda G, Asai K, Mori Y, Wada K, Higuchi R, Watanabe M, Rikiyama T, Sata N, Kano N, Umezawa A, Mukai S, Tokumura H, Hata J, Kozaka K, Iwashita Y, Hibi T, Yokoe M, Kimura T, Kitano S, Inomata M, Hirata K, Sumiyama Y, Inui K, Yamamoto M. Tokyo Guidelines 2018: initial management of acute biliary infection and flowchart for acute cholangitis. J Hepatobiliary Pancreat Sci. 2018;25:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 366] [Cited by in RCA: 321] [Article Influence: 40.1] [Reference Citation Analysis (2)] |
| 144. | Wada K, Takada T, Kawarada Y, Nimura Y, Miura F, Yoshida M, Mayumi T, Strasberg S, Pitt HA, Gadacz TR, Büchler MW, Belghiti J, de Santibanes E, Gouma DJ, Neuhaus H, Dervenis C, Fan ST, Chen MF, Ker CG, Bornman PC, Hilvano SC, Kim SW, Liau KH, Kim MH. Diagnostic criteria and severity assessment of acute cholangitis: Tokyo Guidelines. J Hepatobiliary Pancreat Surg. 2007;14:52-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 191] [Article Influence: 10.1] [Reference Citation Analysis (4)] |
| 145. | Gomi H, Solomkin JS, Schlossberg D, Okamoto K, Takada T, Strasberg SM, Ukai T, Endo I, Iwashita Y, Hibi T, Pitt HA, Matsunaga N, Takamori Y, Umezawa A, Asai K, Suzuki K, Han HS, Hwang TL, Mori Y, Yoon YS, Huang WS, Belli G, Dervenis C, Yokoe M, Kiriyama S, Itoi T, Jagannath P, Garden OJ, Miura F, de Santibañes E, Shikata S, Noguchi Y, Wada K, Honda G, Supe AN, Yoshida M, Mayumi T, Gouma DJ, Deziel DJ, Liau KH, Chen MF, Liu KH, Su CH, Chan ACW, Yoon DS, Choi IS, Jonas E, Chen XP, Fan ST, Ker CG, Giménez ME, Kitano S, Inomata M, Mukai S, Higuchi R, Hirata K, Inui K, Sumiyama Y, Yamamoto M. Tokyo Guidelines 2018: antimicrobial therapy for acute cholangitis and cholecystitis. J Hepatobiliary Pancreat Sci. 2018;25:3-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 295] [Article Influence: 36.9] [Reference Citation Analysis (3)] |
| 146. | Buxbaum JL, Buitrago C, Lee A, Elmunzer BJ, Riaz A, Ceppa EP, Al-Haddad M, Amateau SK, Calderwood AH, Fishman DS, Fujii-Lau LL, Jamil LH, Jue TL, Kwon RS, Law JK, Lee JK, Naveed M, Pawa S, Sawhney MS, Schilperoort H, Storm AC, Thosani NC, Qumseya BJ, Wani S. ASGE guideline on the management of cholangitis. Gastrointest Endosc. 2021;94:207-221.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 147. | Crockett SD, Wani S, Gardner TB, Falck-Ytter Y, Barkun AN; American Gastroenterological Association Institute Clinical Guidelines Committee. American Gastroenterological Association Institute Guideline on Initial Management of Acute Pancreatitis. Gastroenterology. 2018;154:1096-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 757] [Cited by in RCA: 618] [Article Influence: 77.3] [Reference Citation Analysis (3)] |
| 148. | Nve E, Badia JM, Amillo-Zaragüeta M, Juvany M, Mourelo-Fariña M, Jorba R. Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines. J Clin Med. 2023;12:4711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 149. | Vohra RS, Spreadborough P, Johnstone M, Marriott P, Bhangu A, Alderson D, Morton DG, Griffiths EA; West Midlands Research Collaborative. Protocol for a multicentre, prospective, population-based cohort study of variation in practice of cholecystectomy and surgical outcomes (The CholeS study). BMJ Open. 2015;5:e006399. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 150. | Yokoe M, Takada T, Strasberg SM, Solomkin JS, Mayumi T, Gomi H, Pitt HA, Garden OJ, Kiriyama S, Hata J, Gabata T, Yoshida M, Miura F, Okamoto K, Tsuyuguchi T, Itoi T, Yamashita Y, Dervenis C, Chan AC, Lau WY, Supe AN, Belli G, Hilvano SC, Liau KH, Kim MH, Kim SW, Ker CG; Tokyo Guidelines Revision Committee. TG13 diagnostic criteria and severity grading of acute cholecystitis (with videos). J Hepatobiliary Pancreat Sci. 2013;20:35-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 322] [Cited by in RCA: 274] [Article Influence: 21.1] [Reference Citation Analysis (1)] |
| 151. | Khashab MA, Tariq A, Tariq U, Kim K, Ponor L, Lennon AM, Canto MI, Gurakar A, Yu Q, Dunbar K, Hutfless S, Kalloo AN, Singh VK. Delayed and unsuccessful endoscopic retrograde cholangiopancreatography are associated with worse outcomes in patients with acute cholangitis. Clin Gastroenterol Hepatol. 2012;10:1157-1161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 152. | Zhou MW, Gu XD, Xiang JB, Chen ZY. Comparison of clinical safety and outcomes of early versus delayed laparoscopic cholecystectomy for acute cholecystitis: a meta-analysis. ScientificWorldJournal. 2014;2014:274516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 153. | Gallagher TK, Kelly ME, Hoti E. Meta-analysis of the cost-effectiveness of early versus delayed cholecystectomy for acute cholecystitis. BJS Open. 2019;3:146-152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |