Copyright: ©Author(s) 2026.
World J Hepatol. Mar 27, 2026; 18(3): 115221
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115221
Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.115221
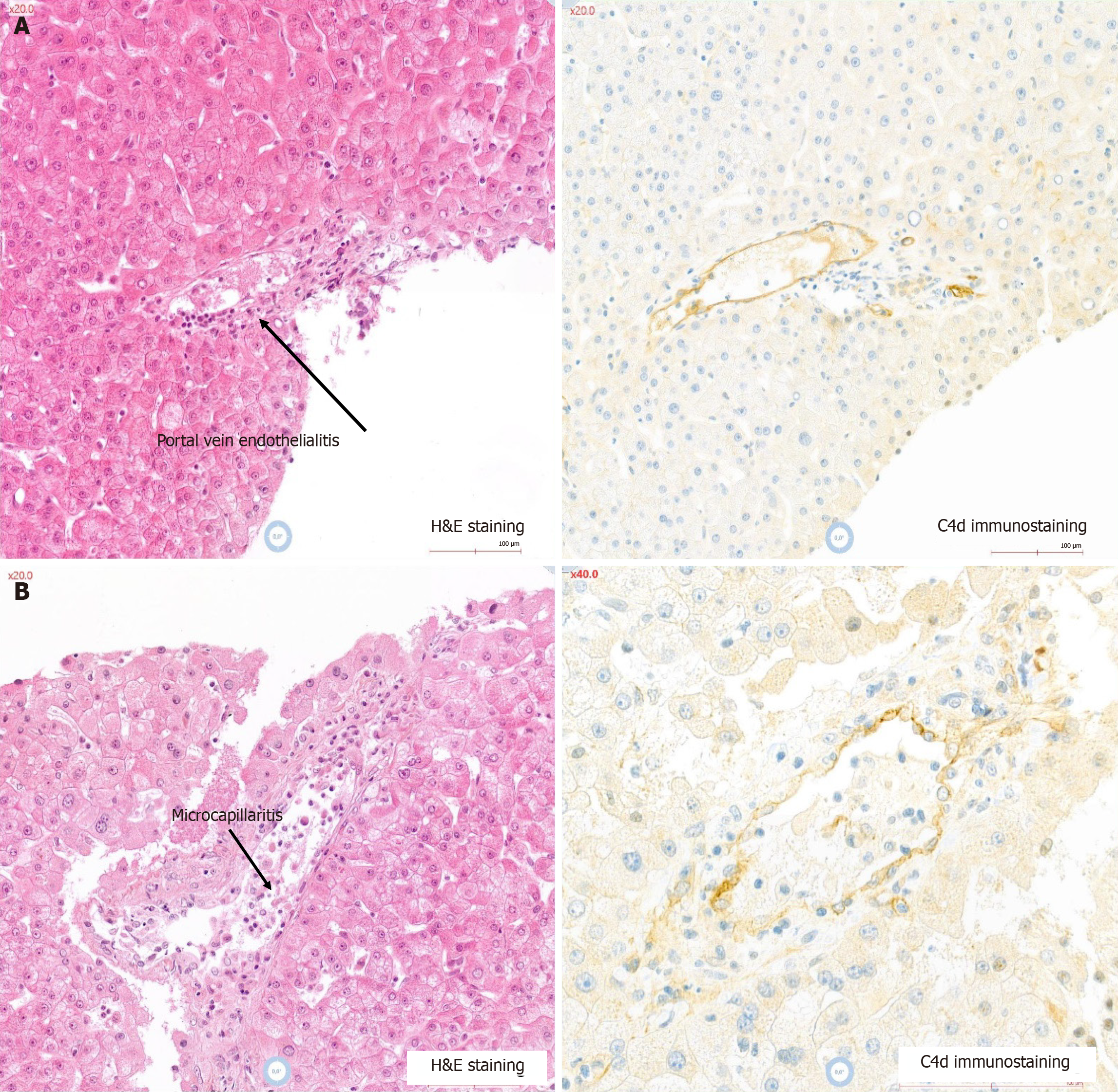
Figure 1 Liver biopsy of a patient with lesions compatible with combined T-cell mediated rejection and acute antibody-mediated rejection.
A: Portal tract: Mild portal infiltrate, endothelialitis and C4d positivity; B: Centrilobular vein: Endothelialitis and microcapillaritis, and C4d positivity. C4d: Complement component 4d.
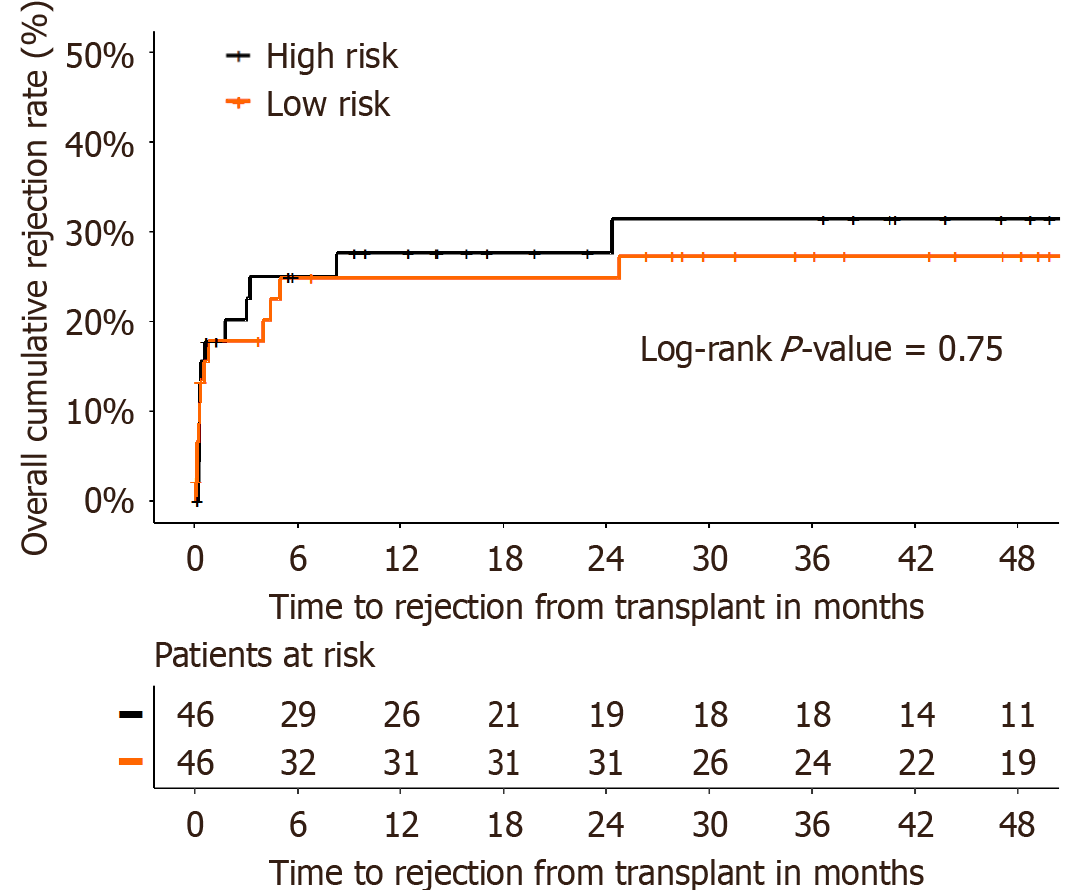
Figure 2 Overall cumulative time to biopsy-proven acute rejection after liver transplantation according to the immunological status.
Kaplan-Meier analysis of cumulative time to biopsy-proven acute rejection after liver transplantation during the study period, comparing the high-risk and low-risk groups (log rank P = 0.75).
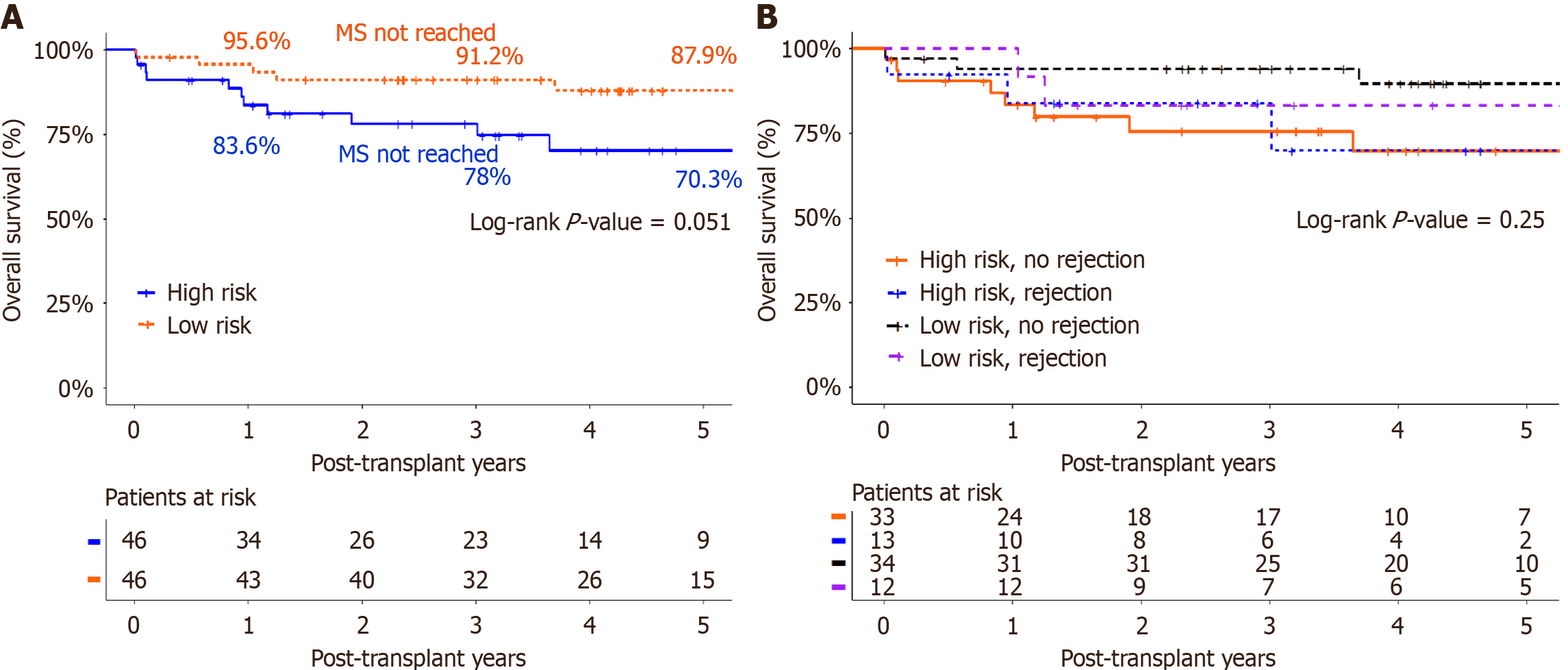
Figure 3 Kaplan-Meier analysis of patient survival.
A: Patient survival after liver transplantation according to immunological status. Kaplan-Meier analysis of patient survival after liver transplantation during the study period, comparing the high-risk and low-risk groups (log rank P = 0.051); B: Patient survival after liver transplantation in both groups according to the occurrence of rejection. Kaplan-Meier analysis of patient survival after liver transplantation showing comparison between the high-risk with rejection, high-risk without rejection, low-risk with rejection and low-risk without rejection groups (log rank P = 0.25). MS: Median survival.
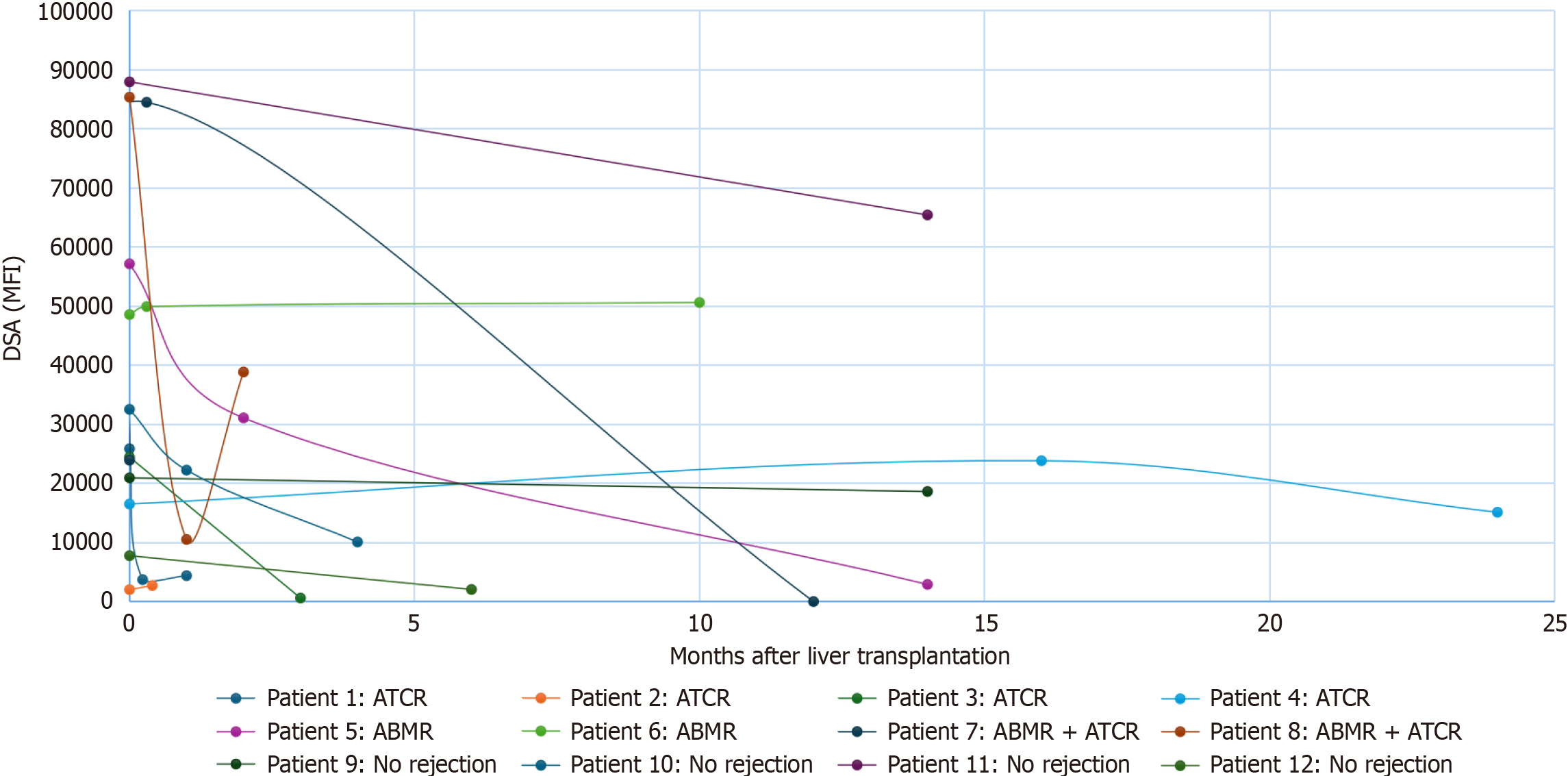
Figure 4 Donor-specific antibodies monitoring following liver transplantation in 12 patients (4 patients with acute antibody-mediated rejection, 4 with T-cell mediated rejection and 4 without rejection).
ABMR: Antibody-mediated rejection; ATCR: Acute T-cell rejection; DSA: Donor specific antibodies; MFI: Mean fluorescence intens.
- Citation: EL-Domiaty N, Coilly A, Sebagh M, Taupin JL, Ibrahim W, Sacleux SC, Ichai P, Duhaut L, Pittau G, Ciacio O, Salloum C, Sa-Cunha A, Azoulay D, Feray C, Cherqui D, Shiha G, Samuel D, Saliba F. Induction with anti-T-lymphocyte globulins and human immunoglobulins: A strategy for hyperimmunized liver transplant patients. World J Hepatol 2026; 18(3): 115221
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/115221.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.115221