Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.117271
Revised: December 20, 2025
Accepted: February 4, 2026
Published online: April 26, 2026
Processing time: 138 Days and 13.2 Hours
Knee osteoarthritis (OA) represents a progressive degenerative joint disorder characterized by articular cartilage degradation, chronic synovial inflammation, and pathological subchondral bone remodeling. Despite its pre
Core Tip: Mesenchymal stromal cell-derived secretomes, rich in exosomes, cytokines, and growth factors, offer a cell-free therapeutic strategy for knee osteoarthritis by modulating inflammation, promoting chondroprotection, and restoring subchondral bone homeostasis. Their paracrine-driven mechanisms target multiple pathological pathways simultaneously, surpassing symptomatic treatments. However, clinical translation demands rigorous standardization, optimized delivery systems, and clear regulatory frameworks to unlock their full regenerative potential in osteoarthritis management.
- Citation: Muruganandam A, Jeyaraman N, Sukumaran AM, Ramasubramanian S, Devanand V, Nallakumarasamy A, Muthu S, Jeyaraman M. Future perspectives on mesenchymal stromal cell-derived secretomes in knee osteoarthritis. World J Stem Cells 2026; 18(4): 117271
- URL: https://www.wjgnet.com/1948-0210/full/v18/i4/117271.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i4.117271
Knee osteoarthritis (OA) constitutes one of the most prevalent and debilitating musculoskeletal disorders worldwide, affecting approximately 365 million individuals globally and representing a leading cause of disability in aging populations[1,2]. The confluence of demographic aging, rising obesity rates, and increasing metabolic syndrome prevalence has amplified the disease burden substantially over recent decades[3,4]. OA imposes considerable socioeconomic costs through direct healthcare expenditures, lost productivity, and diminished quality of life, yet therapeutic options remain remarkably limited[5,6].
Current management strategies are largely confined to symptomatic palliation. Non-steroidal anti-inflammatory drugs, though widely prescribed, provide only temporary pain relief while carrying significant cardiovascular and gastroin
The fundamental limitation of existing therapies lies in their failure to address the core pathophysiological processes driving OA progression[13,14]. These include the inexorable degradation of articular cartilage through imbalanced matrix metalloproteinase activity, aberrant subchondral bone remodeling with increased sclerosis and osteophyte formation, and chronic low-grade synovial inflammation perpetuating a catabolic joint environment[15-17]. This pathophysiological cascade creates an urgent and unmet need for disease-modifying OA drugs capable of interrupting disease progression, promoting tissue regeneration, and preserving native joint architecture[18,19].
The emergence of regenerative medicine approaches, particularly mesenchymal stromal cell (MSC)-based therapies, initially generated considerable enthusiasm within the orthopedic community[20,21]. Early preclinical studies and pilot clinical trials demonstrated promising results through MSCs’ potent anti-inflammatory capabilities and tissue repair potential[20,22]. However, the path toward widespread clinical adoption of whole-cell therapies encountered significant obstacles[23]. These challenges encompass inherent product variability arising from donor heterogeneity and culture conditions, limited cell viability and engraftment following transplantation, logistical complexities in manufacturing and distribution, regulatory hurdles, and theoretical concerns regarding immunogenicity and malignant transformation potential[23-26].
Building on the recognition of paracrine-mediated MSC effects described above[21,27], attention has shifted toward the MSC-derived secretome, a complex repertoire of bioactive components that includes extracellular vesicles (EVs), growth factors, cytokines, and immunomodulatory proteins[27,28]. The secretome represents a sophisticated, cell-free delivery system capable of simultaneously targeting multiple pathological pathways without the complications inherent to cellular transplantation[29,30]. The term ‘secretome’ encompasses both EVs (particularly exosomes) and soluble factors (growth factors, cytokines), though many therapeutic products consist of EV-enriched or small EV (sEV) fractions specifically isolated for clinical use.
The utilization of MSC-derived secretomes as therapeutic agents offers substantial practical advantages[29]. These include enhanced product stability, simplified standardization protocols, streamlined storage and distribution logistics, elimination of cell viability concerns, and potentially reduced immunogenic risk[30,31]. While numerous isolated studies have confirmed chondroprotective and immunomodulatory properties in various OA models, a comprehensive synthesis bridging fundamental molecular mechanisms with clinical translation potential remains necessary[21,32]. The comparative injectable regenerative therapies for knee OA are summarized in Table 1.
| Parameter | MSC secretomes | Whole-cell MSCs | PRP | Hyaluronic acid |
| Mechanism | Paracrine signaling via EVs, growth factors, cytokines | Paracrine + potential differentiation | Growth factor delivery, platelet-derived factors | Viscosupplementation, mild anti-inflammatory |
| Standardization | Moderate challenge; biochemical characterization possible | High challenge; cellular variability | High variability; preparation method dependent | Well-established; pharmaceutical-grade products |
| Storage/stability | Freeze/Lyophilize possible; moderate stability | Requires cryopreservation; viability concerns | Fresh preparation typically required | Excellent; room temperature stable |
| Regulatory pathway | Evolving; biological therapeutic | Complex; cellular product regulations | Variable; may be considered device or biologic | Established; device or drug depending on claims |
| Manufacturing scalability | Good potential with optimization | Limited; donor and passage constraints | Limited; autologous preparation | Excellent; large-scale synthesis feasible |
| Immunogenicity risk | Minimal; cell-free | Low to moderate depending on allogeneic vs autologous | Minimal; typically autologous | Minimal; well-tolerated |
| Chondroprotective mechanism | Direct: Matrix synthesis, MMP inhibition | Direct and indirect through secretome | Indirect: Growth factor stimulation | Minimal direct chondroprotection |
| Anti-inflammatory potency | High; multiple mechanisms | High; primarily via secretome | Moderate; transient | Mild |
| Subchondral bone effects | Demonstrated in preclinical models | Demonstrated in preclinical models | Limited evidence | Minimal |
| Clinical evidence level | Phase I/II; early data | Phase II/III; mixed results | Multiple RCTs; modest benefit | Extensive RCTs; modest, transient benefit |
| Cost considerations | Moderate; dependent on manufacturing scale | High; labor-intensive production | Low to moderate | Low; established manufacturing |
| Practical advantages | Off-the-shelf potential, consistent dosing | Potentially more potent for severe disease | Point-of-care preparation, familiar to clinicians | Established reimbursement, physician familiarity |
| Practical limitations | Novel product; limited clinical data | Viability concerns, regulatory complexity | High preparation variability | Limited efficacy; short duration |
This narrative review aims to consolidate the expanding literature on MSC-derived secretomes in knee OA[33]. We systematically examine the secretome’s molecular composition, elucidate detailed mechanisms through which it modifies the OA microenvironment, critically analyze existing preclinical and clinical evidence, address formidable challenges related to product standardization and delivery optimization, and delineate future perspectives necessary to translate this promising cell-free strategy into robust clinical therapy[21,33,34]. Beyond summarizing existing evidence, this review advances the field by integrating mechanistic insights into EV-associated and soluble MSC secretome components with a critical evaluation of the manufacturing-to-clinic continuum, encompassing good manufacturing practice (GMP)-compliant production, potency assay development, and regulatory considerations. By situating these analyses within the broader regenerative therapy landscape and outlining a pragmatic translational roadmap with defined milestones, the manuscript directly links biological understanding to the practical requirements of clinical implementation in OA.
This narrative review synthesizes the expanding literature through a comprehensive search of PubMed, Scopus, and Web of Science databases (January 2015 to November 2024) using key terms including ‘mesenchymal stromal cells’, ‘secretome’, ‘extracellular vesicles’, ‘exosomes’, and ‘osteoarthritis’. While we acknowledge that a systematic review with PRISMA methodology would provide additional rigor in evidence grading, our narrative approach allows broader conceptual integration of mechanistic, preclinical, and translational data across this rapidly evolving field. This narrative review emphasizes conceptual connections and practical relevance, rather than formal evidence grading used in systematic reviews or meta-analyses.
Terminology in this field requires clarification: ‘secretome’ refers to the complete collection of factors secreted by MSCs, encompassing both membrane-bound EVs (including exosomes, microvesicles, and apoptotic bodies) and soluble proteins, growth factors, and cytokines. Many therapeutic products are EV-enriched or sEV fractions obtained through differential centrifugation or filtration. Throughout this review, we use ‘secretome’ when discussing the complete mixture, ‘EVs’ or ‘exosomes’ when referring to vesicular components specifically, and note the product composition when describing individual studies. Most mechanistic and clinical data discussed derive from EV-enriched preparations, though soluble factors contribute importantly to therapeutic effects, particularly in whole-conditioned medium applications. MSC-derived secretome comprises a heterogeneous mixture of bioactive components that collectively orchestrate therapeutic effects in OA[35]. Its complexity underlies the multifaceted mechanisms through which secretomes modulate joint pathology, spanning EVs, soluble growth factors, and immunomodulatory molecules[36].
EVs are arguably the most critical component of the MSC secretome, serving as nature’s evolved system for intercellular communication[37]. EVs include exosomes (30-150 nm, endosomal origin), microvesicles (100-1000 nm, plasma membrane budding), and apoptotic bodies (> 1000 nm). Among these, exosomes warrant particular attention due to their abundance, stability, and cargo specificity[38].
Exosomes originate from multivesicular bodies and are enriched with molecular cargo. Their membranes contain characteristic proteins (CD9, CD63, CD81, tumor susceptibility gene 101) that facilitate targeting and uptake[39]. Encapsulated biomolecules - microRNAs (miRNAs), messenger RNAs, proteins, lipids, and metabolites - are protected from degradation and delivered to recipient cells such as chondrocytes, synoviocytes, and macrophages[39]. This transfer alters cellular function via post-transcriptional regulation, protein delivery, and metabolic modulation. Xu et al[40] highlighted exosome-mediated communication as a sophisticated system coordinating multicellular responses to pathological stimuli.
Exosomal miRNAs are particularly relevant in OA. These approximately 22 nucleotide regulators bind target mRNAs to repress translation or induce degradation. MSC-derived exosomes deliver miRNAs with chondroprotective effects such as miR-140-5p promotes chondrocyte proliferation and matrix synthesis while suppressing catabolism[41]; miR-146a attenuates nuclear factor kappa B (NF-κB)-driven inflammation[42]; and miR-21 enhances survival and reduces apoptosis in stressed chondrocytes[43]. Exosomal proteins also contribute significantly. Cheng et al[44] identified over 1000 proteins involved in extracellular matrix organization, immune regulation, and metabolism. This molecular diversity enables simultaneous engagement of multiple therapeutic targets, a capability unmatched by single-molecule drugs.
Beyond vesicles, MSC-derived secretome contains soluble factors that directly regulate tissue homeostasis and repair. These act through receptor-mediated signaling to promote anabolic processes and suppress catabolism.
Transforming growth factor-beta: A key chondroprotective factor. Through SMAD2/3 signaling, transforming growth factor-beta (TGF-β) induces cartilage-specific genes [SRY-related HMG-box gene 9, collagen type II alpha 1 chain (COL2A1), aggrecan]. Ying et al[45] showed MSC-derived TGF-β1 enhances chondrocyte differentiation while preventing hypertrophy, maintaining stable cartilage phenotype.
Insulin-like growth factor-1: Potent anabolic mediator stimulating proliferation, proteoglycan synthesis, and survival via phosphatidylinositol 3-kinase/protein kinase B and mitogen-activated protein kinases/extracellular regulated protein kinases pathways. Montaseri et al[46] demonstrated insulin-like growth factor-1 (IGF-1) counteracts interleukin (IL)-1β-induced catabolism, preserving matrix synthesis and preventing apoptosis.
Bone morphogenetic proteins: Particularly bone morphogenetic protein (BMP)-2 and BMP-7, which stimulate chondrocyte differentiation, matrix synthesis, and subchondral bone remodeling. Kang et al[47] emphasized BMPs’ dual role, as aberrant activity may drive osteophyte formation, necessitating precise regulation.
Vascular endothelial growth factor: Plays a nuanced role. While angiogenesis supports subchondral bone integrity and synovial perfusion, excessive vascular endothelial growth factor (VEGF) promotes pathological vascular invasion into cartilage. Palamà et al[48] and Műzes and Sipos[31] highlighted MSC-derived secretome modulation of VEGF as a mechanism to normalize aberrant angiogenesis in OA.
The immunomodulatory capacity of MSC-derived secretomes is central to their therapeutic potential. Chronic low-grade inflammation drives OA progression, and secretome-derived molecules actively resolve this state[21].
IL-10: A potent anti-inflammatory cytokine. Through signal transducer and activator of transcription 3 activation, IL-10 suppresses NF-κB-driven pro-inflammatory gene expression. Hutchins et al[49] showed MSC-derived IL-10 reduces synovial inflammation, shifting the cytokine milieu toward anti-inflammatory dominance.
Prostaglandin E2: Exerts immunosuppressive effects via EP2/EP4 receptor signaling, reducing pro-inflammatory cytokines and promoting regulatory T cell differentiation. Jin et al[50] noted dose-dependent effects, with physiological levels suppressing inflammation but excessive concentrations potentially enhancing certain pathways.
Tumor necrosis factor-stimulated gene-6: A multifunctional glycoprotein with strong anti-inflammatory properties. Tumor necrosis factor (TNF)-stimulated gene-6 (TSG-6) inhibits neutrophil infiltration, reduces cytokine production, and stabilizes extracellular matrix via hyaluronan interactions. Choi et al[51] demonstrated that TSG-6 knockdown diminishes MSC-derived secretome efficacy in arthritis models. Exosomes also contribute to immunomodulation. miRNAs such as miR-146a and miR-223 suppress inflammatory signaling in immune cells, while exosomal proteins including TGF-β and hepatocyte growth factor directly engage macrophages and T cells to promote immunosuppression[52].
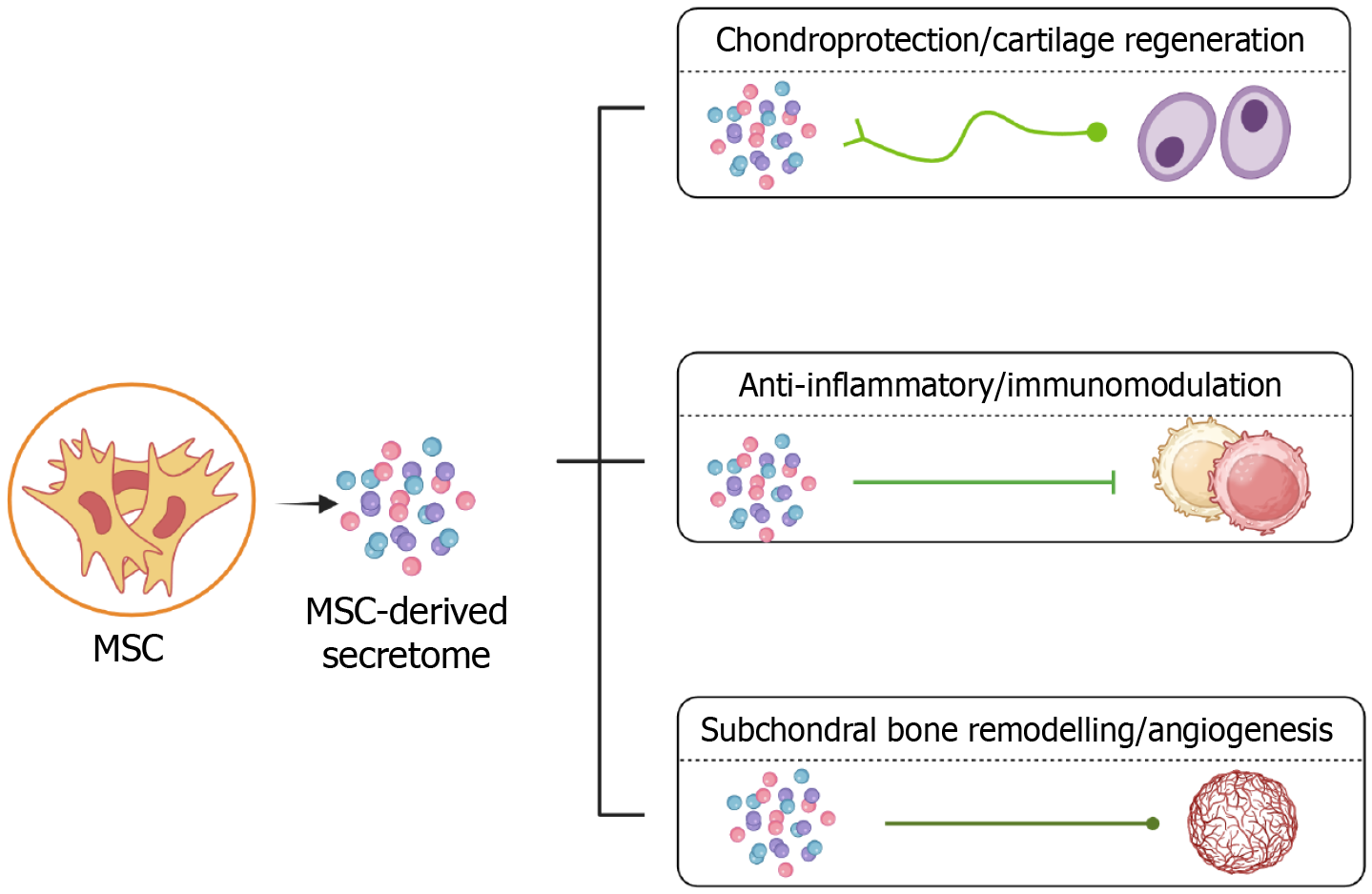
The therapeutic potential of MSC-derived secretomes stems from their capacity to simultaneously target multiple interconnected pathological processes driving OA progression (Figure 1). This section examines in detail the mechanisms through which secretomes modulate chondrocyte function, inflammatory pathways, and subchondral bone homeostasis[53].
Articular cartilage degradation is the hallmark of knee OA, driven by imbalance between anabolic synthesis and catabolic breakdown. The MSC secretome restores this equilibrium through complementary mechanisms[52].
Stimulation of chondrocyte anabolism: Secretome-derived growth factors promote chondrocyte proliferation and extracellular matrix synthesis. Dalle Carbonare et al[54] showed upregulation of SRY-related HMG-box gene 9, the master regulator of chondrogenesis, which drives COL2A1 and aggrecan expression. Guo et al[55] confirmed enhanced COL2A1 transcription and translation, increasing type II collagen deposition. Wen et al[56] demonstrated increased aggrecan synthesis with reduced ADAMTS-5-mediated degradation, preserving cartilage’s compressive resilience. Mechanistically, TGF-β activates SMAD2/3 signaling, while IGF-1 engages phosphatidylinositol 3-kinase/protein kinase B and mitogen-activated protein kinases/extracellular regulated protein kinases cascades. Hui et al[57] highlighted synergistic effects of combined TGF-β and IGF-1, yielding superior chondroprotection.
Suppression of catabolic enzyme activity: MSC-derived secretomes directly suppress matrix-degrading enzymes. Matrix metalloproteinase 13 (MMP-13), the principal collagenase in OA, is reduced via NF-κB and AP-1 inhibition, mediated by exosomal miRNAs and soluble anti-inflammatory factors[58]. ADAMTS-5 downregulation occurs through transcriptional repression and miRNA targeting, preserving aggrecan[59]. Lu et al[60] identified broad suppression of catabolic enzymes (MMP-1, MMP-3, cathepsins), indicating global reprogramming of chondrocyte gene expression toward anabolism.
miRNA-mediated gene regulation: Exosomal miRNAs fine-tune chondrocyte gene expression. Li et al[61] identified dysregulated miRNAs in OA cartilage, with MSC exosomes delivering corrective signals. miR-140-5p promotes proliferation and matrix synthesis while suppressing ADAMTS-5 and inflammatory genes[62]. miR-146a reduces IL-1β-induced inflammation by targeting TNF receptor-associated factor 6 and IL-1 receptor-associated kinase 1[63]. miR-21 protects against apoptosis via phosphatase and tensin homolog/protein kinase B modulation, though its effects can be context-dependent[64]. Collectively, these miRNAs reprogram chondrocytes toward anabolic and anti-inflammatory states.
Chronic synovial inflammation drives OA progression and pain. The MSC secretome exerts potent anti-inflammatory actions.
Cytokine modulation and NF-κB suppression: Secretome treatment reduces TNF-α, IL-1β, and pathological IL-6[65]. Mechanisms include receptor antagonism, enhanced IL-1Ra production, and direct signaling suppression. NF-κB inhibition is central: TSG-6 and exosomal miRNAs block nuclear translocation and DNA binding dampening inflammatory gene expression[51,66].
Macrophage polarization and immune modulation: MSC-derived secretomes reprogram macrophages from pro-inflammatory M1 to anti-inflammatory M2 phenotypes. Cho et al[67] showed reduced M1 markers (inducible nitric oxide synthase, TNF-α, IL-1β) and increased M2 markers (arginase-1, CD206, IL-10). Mechanisms include prostaglandin E2-mediated EP2/EP4 signaling, exosomal miR-146a/miR-223 transfer, and soluble IL-10/TGF-β signaling[68,69]. M2 macrophages not only suppress inflammation but also promote repair via debris clearance and growth factor secretion[69].
Resolution of synovial inflammation: Khatab et al[70] demonstrated reduced synovial thickening, immune infiltration, and normalized fluid composition after secretome treatment, correlating with pain reduction. Mancuso et al[21] documented cytokine profile shifts and increased anti-inflammatory mediators, indicating transformation of the joint microenvironment into a repair-permissive state.
OA involves the entire osteochondral unit, including pathological subchondral bone remodeling with sclerosis and osteophyte formation.
Regulation of bone cell activity: Secretome components normalize osteoblast and osteoclast activity. Zhang et al[71] showed enhanced osteoblast differentiation and mineralization via BMP/Wnt signaling, balanced by osteoclast regulation. Cho et al[72] demonstrated reduced osteoclastogenesis through altered receptor activator of NF-кB ligand/osteoprotegerin ratios.
Modulation of pathological angiogenesis: Aberrant vascularization contributes to remodeling and pain. Li et al[73] described hypervascularization and impaired perfusion in OA bone. MSC-derived secretome VEGF supports balanced angiogenesis, improving perfusion without cartilage invasion[28]. Sears and Ghosh[74] emphasized the secretome’s ability to normalize angiogenesis through combined pro-angiogenic and regulatory factors.
Inhibition of osteophyte formation: Osteophytes, driven by aberrant BMP signaling, impair mobility and cause pain. Giannasi et al[75] showed secretome treatment reduced osteophyte size and BMP-2 expression. Chou et al[76] identified TSG-6 as a key mediator inhibiting inflammatory signals that initiate osteophyte development. Together, these effects highlight the secretome’s ability to address structural joint changes beyond cartilage preservation.
Preclinical studies of MSC-derived secretome therapy in knee OA span in vitro systems, small animal models, and large animal experiments. Collectively, they support therapeutic potential, but critical appraisal highlights both strengths and limitations that shape clinical translation.
Cell culture systems remain the main entry point for dissecting MSC-derived secretome mechanisms in OA. Across multiple studies, MSC-derived EVs and broader secretome fractions consistently enhance anabolic markers such as COL2A1 and aggrecan, while suppressing catabolic mediators including MMP-13, ADAMTS-5, IL-1β, and TNF-α in primary human OA chondrocytes and synoviocytes[33,77-79]. These effects are particularly clear in experiments that use patient-derived cartilage or synovium rather than immortalised lines, which better preserve OA-specific transcriptional and epigenetic changes[33,79]. Three-dimensional pellet or hydrogel systems help maintain chondrocyte phenotype and allow evaluation of matrix deposition, but introduce variability in diffusion, nutrient gradients, and EV penetration, complicating dose standardisation and between-laboratory comparability[77,80].
Mechanistically, EV cargo appears central rather than soluble proteins alone. Exosomes enriched in specific miRNAs reproducibly modulate canonical OA pathways: MiR-140-5p-overexpressing synovial MSC-EVs enhance cartilage matrix synthesis and reduce MMP-13 and ADAMTS-5 in IL-1β-challenged chondrocytes and prevent OA development in rat models; miR-92a-3p-rich EVs promote chondrogenesis and inhibit WNT5A-driven catabolic signaling; hypoxia-enhanced miR-216a-5p or miR-122-5p in MSC-EVs improves chondrocyte proliferation, survival, and autophagy through JAK2/signal transducer and activator of transcription 3 and DUSP2-regulated pathways[81-86]. These data support a model in which specific miRNA-target pairs, rather than bulk trophic factors, drive much of the observed chondroprotection.
Co-culture and organoid approaches partially address the oversimplification of single-cell-type monolayers. Tri- or multi-cell co-cultures including chondrocytes, synovial fibroblasts, and macrophage-lineage cells show that MSC-EVs simultaneously dampen inflammatory cytokine release, shift macrophage phenotype toward an M2-like profile, and preserve cartilage matrix, effects that are substantially weaker in isolated chondrocyte cultures[33,78,87]. However, even sophisticated in vitro models cannot recapitulate joint biomechanics, subchondral bone crosstalk, or adaptive immune contributions. Dose-response curves are another limitation: Secretome concentrations needed to normalise catabolic marker expression often exceed by one to two orders of magnitude the joint levels that are realistically achievable in vivo with a single intra-articular injection, even if one assumes slow clearance[33,77,87,88].
Rodent OA models, whether surgically induced (e.g., destabilization of medial meniscus, anterior cruciate ligament transection) or chemically induced (e.g., monoiodoacetate), consistently show that MSC-EVs or broader secretomes reduce cartilage degeneration scores, preserve proteoglycan content, and attenuate synovial inflammation[33,77,78,84,86,87]. These outcomes are supported by meta-analyses indicating significant improvements in histological OA scores and pain-related behaviours compared with vehicle controls[86,89,90]. Reduction of CGRP-positive nerve fibres in sub
These models are well suited to mechanistic dissection. Genetic manipulation of donor MSCs or recipient mice has linked specific EV components to functional outcomes, for example miR-140-5p or miR-92a-3p in exosomes, and has clarified that macrophage reprogramming is a major contributor to reduced synovitis and pain[81,82,87,90,91]. Yet several features limit translational relevance. Disease in rodents progresses rapidly over weeks, which magnifies the apparent benefit of early intervention and does not capture the decades-long, multifactorial course of human OA[78,87,88]. Many protocols use preventive or very early treatment immediately after joint insult, whereas patients typically present with established cartilage loss and chronic synovitis. Follow-up rarely exceeds 8-12 weeks, so durability of structural and symptomatic benefit remains largely unknown[33,77,84,86]. Histological scoring at a single endpoint remains the dominant outcome, with relatively few studies combining serial imaging, behavioural measures, and molecular readouts in the same animals.
Large animals such as rabbits, dogs, sheep, and minipigs offer joint size, cartilage thickness, and loading patterns that more closely approximate the human knee. Secretome and EV studies in these species have demonstrated improved cartilage integrity, reduced osteophyte formation, and better biomechanical properties after intra-articular administration, sometimes in combination with hyaluronic acid (HA) or hydrogels[77,78,88,91,92]. For example, in rabbit and minipig models of post-traumatic OA, exosome-enriched preparations from bone marrow or umbilical cord MSCs have led to thicker, more organised cartilage repair tissue with higher Safranin O staining intensity and improved indentation modulus compared with vehicle or HA alone[33,77,92].
Despite this anatomical relevance, the evidence base in large animals is thin and heterogeneous. Sample sizes are often underpowered, blinding of histological and biomechanical assessment is inconsistently reported, and outcome measures vary widely between laboratories. Most models rely on surgical injury rather than spontaneous age-related OA, and follow-up rarely extends beyond 3-6 months[77,78,88,91,92]. These constraints mean that large-animal data are more useful for optimisation of delivery strategies and surgical techniques than for definitive efficacy estimates. They do, however, provide a realistic testbed for clinically relevant injection volumes, imaging protocols, and repeated-dose schedules.
Head-to-head comparisons of MSC-derived secretomes with standard intra-articular therapies are comparatively scarce. In rodent and rabbit models, EV-based preparations have generally outperformed HA, platelet-rich plasma, or corticosteroids in terms of histological cartilage preservation and pain-related behaviours[33,77,78,86,87,89]. However, many of these experiments use aggressive induction of OA, high doses of regenerative products, and relatively short follow-up, which tend to accentuate differences in favour of more potent anabolic or anti-inflammatory agents.
Comparisons between whole-cell MSC therapy and acellular secretomes are particularly informative. Several preclinical studies using paired designs have found broadly similar structural and functional outcomes when total EV dose is scaled to the secretory output of the corresponding cell number, with secretomes showing reduced risk of ectopic tissue formation and greater batch-to-batch uniformity[33,87,88,92,93]. Yet true dosing equivalence is difficult to define. EV preparations are quantified using protein mass, particle number, or specific marker content, while cell therapies are dosed by viable cell counts. The relationship between these metrics and intra-articular exposure at the tissue level is not linear, so apparent “equivalence” probably masks meaningful differences in pharmacokinetics and tissue targeting.
The translation of MSC-derived secretomes from preclinical promise to clinical reality requires addressing multiple interconnected challenges spanning manufacturing, regulatory approval, and clinical validation. The following sections examine the current state of clinical evidence and the principal barriers to widespread therapeutic adoption.
The clinical data set for MSC-derived secretomes in knee OA remains small but is growing. Phase I/II trials have primarily evaluated safety of intra-articular EV or secretome injections derived from placental, umbilical cord, or bone-marrow MSCs[93-95]. A recent triple-blind, randomised, placebo-controlled trial of placental MSC-derived EVs in bilateral knee OA (grade 2-3) used a within-patient design, with one knee receiving EVs and the contralateral knee saline. A single 5-mL injection containing a clinically standardised EV dose was safe over 12 months, with no serious adverse events and only transient local reactions, but did not significantly improve Western Ontario and McMaster Universities Arthritis Index pain or function vs placebo[94]. These neutral efficacy findings are important, because they temper expectations derived from aggressive rodent models.
In parallel, first-in-human work with clinical-grade umbilical cord MSC sEVs has shown that large-scale, GMP-compliant manufacturing with reproducible miRNA and protein profiles is feasible and that intra-articular administration appears well tolerated in early cohorts of OA patients[95]. Although these early studies are underpowered for efficacy, modest improvements in pain scores and function have been reported in some open-label series, but the absence of rigorous controls and the known magnitude of placebo responses after intra-arterial injection make these signals difficult to interpret.
Several aspects of trial design remain unresolved. Dose optimisation is particularly complex. EV products can be quantified by total protein, particle number, or copy number of specific bioactive miRNAs, and the clinically relevant parameter is likely a composite of these features[93,96-98]. Translation from preclinical dosing is hindered by scale differences between rodent and human joints and by differences in clearance kinetics. Most planned or ongoing trials use one or two injections separated by weeks to months, whereas preclinical protocols often use multiple closely spaced doses.
Outcome measures present further difficulties. Structural endpoints such as magnetic resonance imaging cartilage thickness or T2 mapping change slowly and require large sample sizes and long follow-up to detect disease-modifying effects. Patient-reported outcomes such as Western Ontario and McMaster Universities Arthritis Index or visual analog scale scores are sensitive but prone to large placebo responses that can exceed the expected incremental benefit of a single biologic injection[94]. Biomarkers including serum or synovial cartilage oligomeric matrix protein, C-telopeptide of type II collagen, or neoepitope fragments of collagen may offer earlier signals, yet none is validated as a surrogate endpoint for regulatory decisions in OA. Heterogeneity of patient populations, with diverse disease phenotypes under the umbrella of “knee OA”, further dilutes treatment effects.
For regulators, MSC-derived secretomes occupy a grey zone between biologics and advanced therapy medicinal products. In most jurisdictions, EVs isolated from MSCs with more than minimal manipulation are regulated as biologic drugs, while whole MSCs are classified as cell-based advanced therapy medicinal products; classification can shift depending on processing steps and intended mechanism of action[95-97,99]. This ambiguity affects requirements for preclinical safety, potency assays, and lot-release criteria. On the positive side, the acellular, non-replicative nature of EV preparations simplifies viral safety testing, sterility assurance, and stability studies compared with living cell products, which may ultimately make large-scale, standardised manufacturing more tractable once regulatory expectations are fully defined.
Secretome composition is highly sensitive to the characteristics of the parental MSC population. Tissue origin, donor age, comorbidities, and passage number all influence EV cargo and soluble factor profiles[33,88,93,96,98-100]. Bone marrow MSCs tend to generate EVs enriched in chondrogenic and osteogenic factors, whereas adipose-derived MSCs show stronger immunomodulatory signatures; umbilical cord MSCs, sourced from young donors, often display greater proliferative capacity and produce EVs with distinct miRNA spectra that may favour anti-inflammatory and anti-senescent effects[33,78,80,84,92]. MSCs from older or comorbid donors generally show reduced secretory potency, which constrains autologous approaches and favours allogeneic master cell banks[33,93,98-100]. Passage number is equally important: Late-passage MSCs display senescence-associated transcriptomic and secretory changes, with EVs that may lose chondroprotective capacity or become pro-fibrotic, which argues for tight control of expansion protocols[96,98,100].
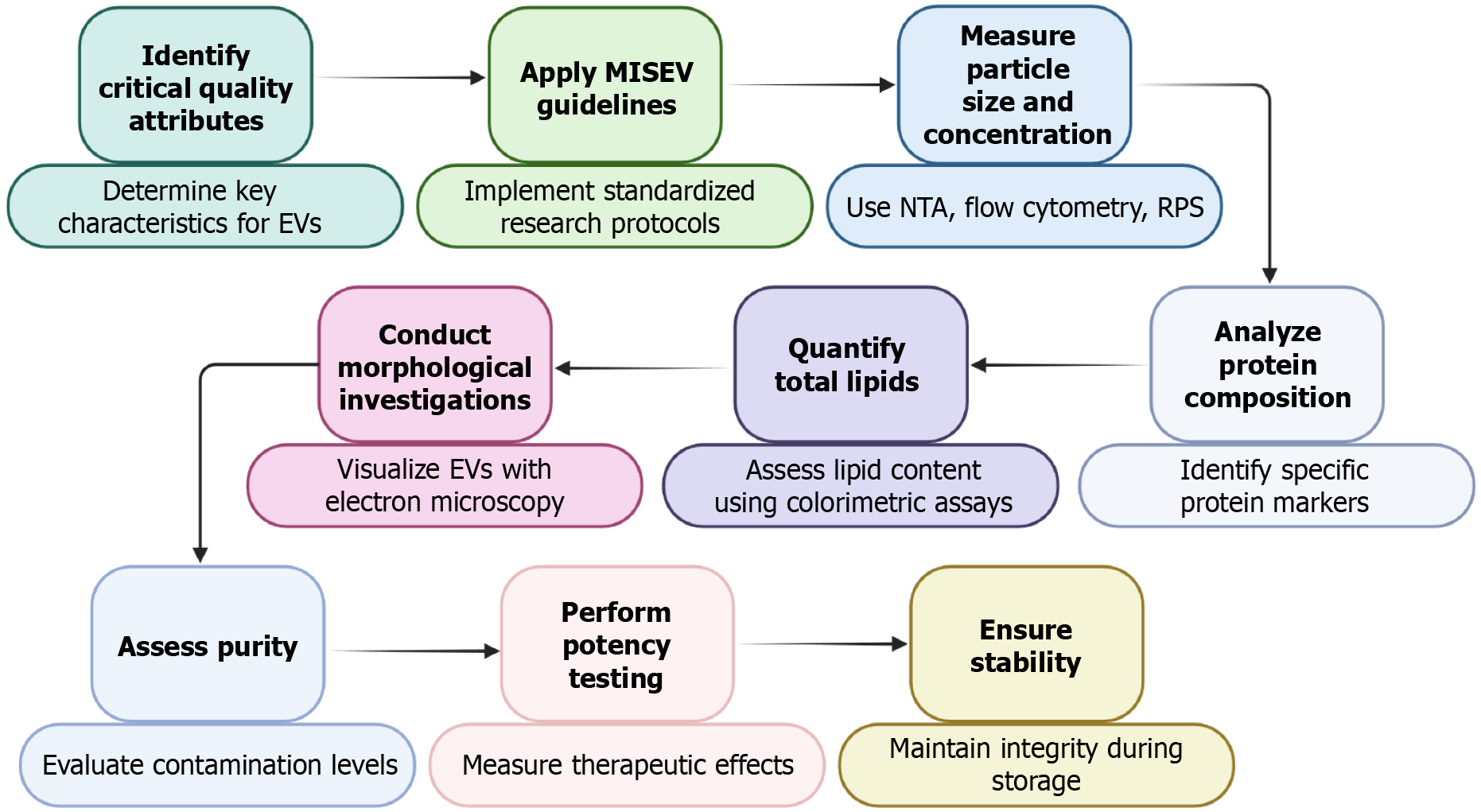
Collection conditions profoundly shape the final product. Serum-free or xeno-free media avoid contaminating bovine EVs but can stress MSCs and alter secretome composition; supplementation with human platelet lysate introduces competing vesicles and growth factors that complicate downstream characterisation[33,87,98]. Duration of conditioning, typically 24-72 hours, trades off yield against accumulation of cell debris, apoptotic bodies, and metabolic waste. Isolation methods including ultracentrifugation, size-exclusion chromatography, tangential-flow filtration, polymer precipitation, and immunoaffinity capture differ in scalability, purity, and preservation of vesicle integrity[95-98]. Ultracentrifugation remains widely used in academic studies but is poorly suited to GMP scale; precipitation methods yield abundant but impure products, while chromatography and filtration approaches offer better scalability at the cost of more complex validation. Minimal Information for Studies of Extracellular Vesicles 2018 and the more recent Minimal Information for Studies of Extracellular Vesicles 2023 update define minimal requirements for EV characterisation, including particle and protein quantification, marker profiling, and basic functional assays[95,96]. However, these guidelines were not written specifically for therapeutic manufacturing, so they need adaptation for lot-release testing and regulatory submissions.
Secretomes contain thousands of proteins, lipids, and nucleic acids, which challenges conventional single-analyte quality control (Figure 2). Basic batch-release criteria generally include sterility, endotoxin, total protein, and EV particle size and concentration, but these metrics do not adequately capture biological potency[93,96-98]. Functional potency assays are increasingly advocated. Examples include in vitro readouts of chondrocyte proliferation and matrix synthesis under inflammatory stress, suppression of macrophage TNF-α or IL-6 secretion and promotion of M2 markers, or reduction of catabolic gene expression in OA synoviocytes[33,87,97,98]. Gimona et al[97] emphasise the need for assays that reflect the proposed mechanism of action and that correlate with in vivo outcomes, while remaining robust enough for routine GMP use. At present, no universally accepted potency assay exists for MSC-EV products in OA, which complicates cross-trial comparison and may explain some variability in clinical responses.
Stability is a practical constraint for multi-centre trials and eventual commercialisation. Fresh or short-term refrigerated preparations show the highest activity, but cold-chain distribution at 4 °C is logistically demanding. Storage at -80 °C or in vapour phase nitrogen maintains EV integrity for months, yet repeated freeze-thaw cycles can cause aggregation and loss of function[93,96-98]. Lyophilisation with appropriate excipients has emerged as a promising strategy to enable room-temperature storage, but formulations must be carefully optimised to prevent damage to lipid membranes and labile miRNAs[93,96]. Different components of the secretome exhibit distinct stability profiles; proteins are generally more resilient than small RNAs, which suggests that degradation may selectively erode some mechanistic pathways over time.
Intra-articular injection is the logical route for knee OA, but pharmacokinetics within the joint are unfavourable. Classical tracer studies show that albumin-sized molecules clear from the joint within 1-13 hours, and native HA clears within roughly a day, although stabilised or crosslinked HA formulations can achieve half-lives of several days to weeks[101,102]. EVs are larger than most soluble cytokines and may persist somewhat longer, yet the synovial capillary bed and lymphatics still clear particles in the 50-200 nm range relatively quickly[101,103,104]. This rapid turnover aligns with clinical observations that many intra-articular biologics produce only transient symptom relief after a single injection.
Nanoparticle studies provide a useful analogy. Cationic nanoparticles can increase residence time from hours to around one week in animal knees, showing that electrostatic interactions and size can be exploited to retard clearance, but even these systems eventually drain via lymphatic pathways[103,104]. Secretome-based therapies that are delivered as a simple bolus injection probably suffer from similar rapid washout, particularly for the soluble fraction.
Therapeutic benefit requires not only residence in the joint but also effective uptake by relevant cell types. EV internalisation by chondrocytes is limited by the dense, negatively charged cartilage matrix and by reduced diffusion in late-stage OA where fibrillation and calcification alter tissue architecture[77,78,87]. In vitro, chondrocytes take up EVs through clathrin- and caveolin-mediated endocytosis, macropinocytosis, and possibly direct membrane fusion, whereas in vivo, a substantial proportion of intra-articular EVs are captured by synovial macrophages and lining cells, which can be beneficial for immunomodulation but reduces the amount reaching deep cartilage[78,87]. Engineering EV surfaces with cartilage-binding peptides or antibodies has improved chondrocyte targeting and cartilage penetration in preclinical studies, but these approaches add manufacturing complexity and may introduce new immunogenic epitopes[105,106].
To counteract rapid clearance and limited penetration, multiple groups have combined secretomes or EVs with advanced delivery systems. Hydrogels based on HA, chitosan, PEG, or composite materials can entrap EVs and provide sustained release while physically filling cartilage defects and supporting tissue repair[107-111]. Thermosensitive or injectable adhesive hydrogels loaded with EVs have improved cartilage regeneration and reduced OA progression in small-animal models, with better retention of fluorescently labelled EVs in the joint space over weeks compared with free EV injections[112-114]. Scaffold-based systems are being explored for focal cartilage defects, where local high-density delivery of EVs or secretomes can be maintained within engineered matrices[113-115].
Nanoparticle platforms, including PLGA and lipid-based carriers, offer another route to provide controlled release and protect EV cargo from enzymatic degradation, though scaling, regulatory scrutiny, and long-term safety remain considerable hurdles[103,104,113]. Secretome-HA co-formulations and other combinations with established viscosupplements may provide incremental benefit by combining mechanical lubrication, extended residence time, and biological activity, but robust comparative data are lacking.
Preconditioning MSCs before secretome collection is one of the most active areas of optimisation. Hypoxic culture (typically 1%-5% O2) increases EV yield and enriches pro-regenerative miRNAs such as miR-216a-5p and miR-122-5p, leading to improved chondrocyte proliferation, migration, matrix synthesis, and autophagy in vitro and enhanced cartilage repair in OA models[83,85,88,92,93]. Inflammatory priming with IL-1β, TNF-α, or interferon gamma can upregulate anti-inflammatory mediators such as TSG-6, prostaglandin E2, indoleamine 2,3-dioxygenase, and specific miRNAs, strengthening immunomodulatory effects but sometimes reducing chondrogenic potential[87,93,99,100]. Three-dimensional spheroid culture reshapes MSC phenotype and enhances secretion of angiogenic and chondrogenic factors, often producing EVs with stronger cartilage-regenerative effects than those from two-dimensional cultures[87,88,99]. The challenge will be to balance enhanced potency with manufacturing complexity. Each preconditioning strategy effectively defines a new product from a regulatory standpoint, and combining multiple stimuli (e.g., hypoxia plus inflammatory priming plus three-dimensional culture) may yield synergistic effects at the cost of substantially more demanding process validation.
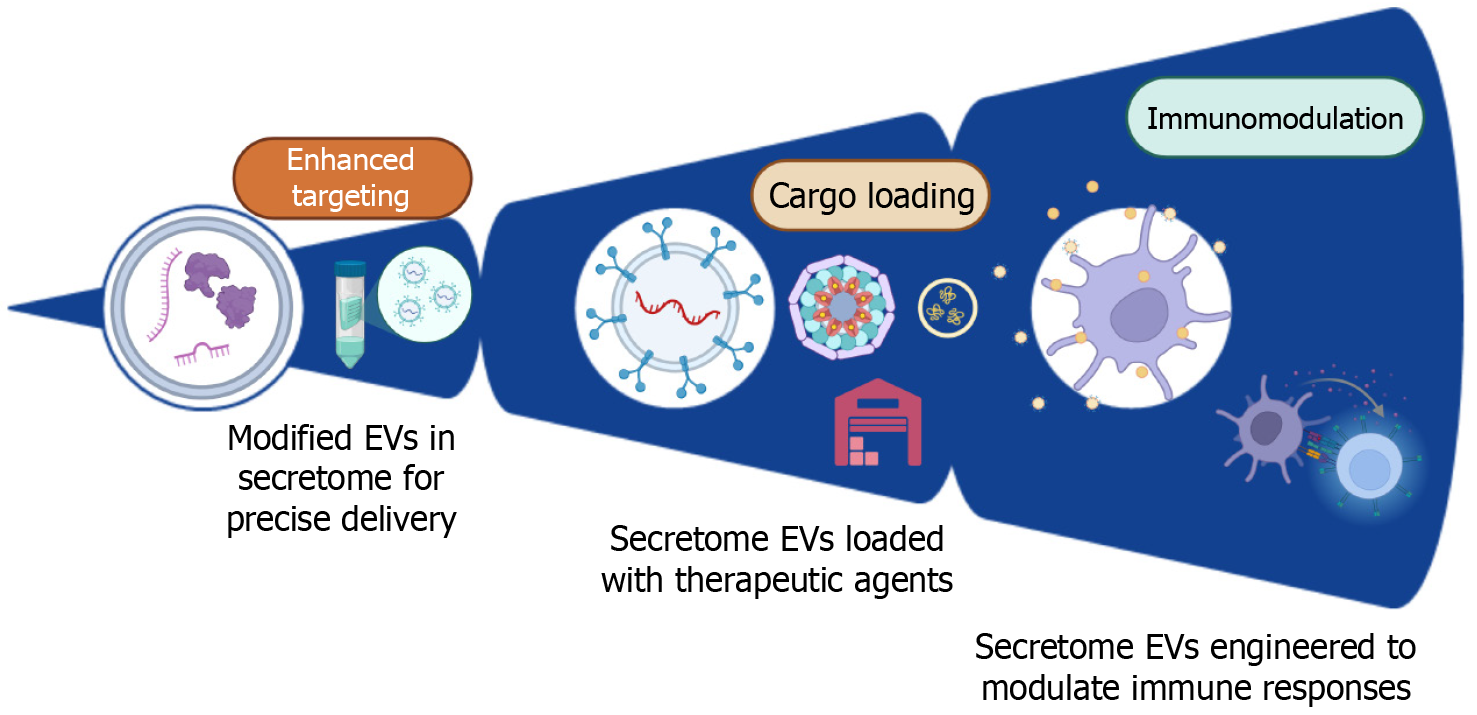
Genetic modification of MSCs enables targeted enrichment of secretome cargo (Figure 3). Overexpression of specific growth factors (such as TGF-β3, BMP-7, or IGF-1), anti-inflammatory cytokines, or therapeutic miRNAs (miR-140-5p, miR-92a-3p, miR-155-5p, among others) in MSCs leads to EVs with amplified chondroprotective properties and more robust disease modification in animal OA models[81,82,87,97]. Viral vectors remain the most efficient tool for stable engineering but raise concerns about insertional mutagenesis in donor cells and theoretical risks of genomic alterations in recipient tissues, even though EVs themselves do not replicate. CRISPR-based editing offers more precise modulation of MSC transcriptional programmes and EV cargo, but applications to clinical-grade MSCs and their secretomes are still largely preclinical.
Hydrogel-based sustained delivery systems: Injectable thermosensitive hydrogels (e.g., chitosan-glycerophosphate or pluronic F-127 formulations) loaded with EVs can provide 2-4 weeks sustained release profiles while maintaining EV integrity. Optimization requires balancing gel degradation kinetics with EV release rates and demonstrating preserved bioactivity post-encapsulation through standardized chondroprotection assays.
Artificial intelligence-guided secretome optimization: Machine learning algorithms trained on multi-omics secretome datasets (proteomics, lipidomics, miRNA profiles) paired with functional outcomes can identify minimal effective cargo signatures. This approach could streamline manufacturing by focusing on defined critical quality attributes rather than whole-secretome complexity, enabling rational design of next-generation products.
Bioengineering cargo enrichment: Beyond genetic modification, biophysical preconditioning protocols combining specific oxygen tension (2%-5% O2), mechanical stimulation mimicking joint loading, and inflammatory priming with defined cytokine cocktails can systematically tune EV cargo. Standardized preconditioning platforms with real-time monitoring would enable reproducible potency enhancement while maintaining GMP compliance.
Post-collection engineering bypasses modification of the parental cells. EVs can be loaded with small molecules, siRNAs, or proteins via electroporation, sonication, or membrane permeabilisation, creating hybrid therapeutics that combine endogenous targeting properties of EVs with defined pharmacologic agents[115,116]. Surface modification with cartilage-homing peptides, antibodies, or aptamers can enhance retention within cartilage and uptake by chondrocytes while reducing non-specific clearance by synovial macrophages[115,116]. These engineered EVs blur the line between biological and nanomedicine products and will likely face some of the most demanding regulatory scrutiny, but they may also offer the most substantial gains in efficacy per dose.
Given the multifactorial nature of OA, it is unlikely that MSC-derived secretome monotherapy will deliver durable disease modification in advanced disease. Rational combinations are therefore an attractive direction. These include pairing EVs with biomaterial scaffolds for focal cartilage repair, combining intra-articular secretomes with HA or other viscosupplements to extend residence time, and integrating biologic injections with structured physiotherapy and weight-management programmes to optimise joint mechanics[112-115]. An important question for future trials will be whether secretomes are best positioned as stand-alone injectables, as adjuncts to arthroscopic or osteotomy procedures, or as part of staged regimens in early OA to delay the need for joint replacement.
The advancement of MSC-derived secretome therapy in knee OA requires coordinated efforts across manufacturing, clinical validation, and translational science. The critical challenges in MSC-derived secretome translation for clinical usage is tabulated in Table 2.
| Challenge category | Specific issues | Current status | Proposed solutions | Timeline to resolution |
| Product standardization | ||||
| Source MSC variability | Donor age, tissue source, comorbidities affect secretome | Widely recognized; limited consensus | Establish standardized donor selection criteria; focus on allogeneic young, healthy donors; develop potency-based lot selection | 2-3 years |
| Isolation method heterogeneity | Multiple protocols yield different products | MISEV guidelines provide research framework | Develop GMP-compliant isolation protocols; comparative studies of methods vs clinical outcomes | 3-5 years |
| Batch-to-batch consistency | Manufacturing variability affects reproducibility | Early-stage optimization in progress | Implement process analytical technology; establish critical quality attributes | 3-5 years |
| Quality control | ||||
| Potency assay development | Lack of validated functional assays | Multiple candidate assays under evaluation | Validate multi-parameter potency testing; correlate with clinical outcomes | 4-6 years |
| Characterization complexity | Thousands of components; incomplete understanding of active elements | Improving with advanced analytics | Employ systems biology approaches; identify critical therapeutic components | 5-7 years |
| Stability testing | Degradation kinetics poorly defined | Limited systematic data | Conduct comprehensive stability studies across formulations; develop real-time potency monitoring | 2-4 years |
| Delivery optimization | ||||
| Rapid intra-articular clearance | Limits sustained therapeutic effect | Well-documented in preclinical models | Develop sustained-release formulations; explore biomaterial carriers; optimize injection timing | 3-5 years |
| Limited cartilage penetration | Chondrocyte targeting inefficient | Recognized challenge; solutions in early development | Engineer targeting moieties; optimize EV size for matrix penetration | 4-6 years |
| Dosing regimen uncertainty | Single vs multiple injections; optimal intervals unknown | Clinical trials using variable regimens | Conduct systematic dose-finding and dose-timing studies | 5-7 years |
| Clinical validation | ||||
| Lack of phase III data | Efficacy vs placebo unproven | Phase I/II ongoing; phase III planning | Execute large, adequately powered RCTs with appropriate controls | 5-8 years |
| Patient selection optimization | Ideal disease stage and phenotype undefined | Exploratory analyses in early trials | Develop predictive biomarkers; conduct stratified analyses | 4-6 years |
| Outcome measure sensitivity | Traditional outcomes require large N and long duration | Alternative outcomes under evaluation | Validate imaging and biochemical biomarkers; incorporate patient-reported outcomes | 3-5 years |
| Regulatory challenges | ||||
| Classification ambiguity | Unclear regulatory pathway | Ongoing agency discussions | Establish precedent through lead product approvals; develop guidance documents | 3-5 years |
| Manufacturing requirements | GMP standards incompletely defined for secretomes | Evolving with agency feedback | Develop industry consensus standards; leverage existing biologic manufacturing frameworks | 3-5 years |
| Economic challenges | ||||
| Reimbursement pathway | Unclear payer coverage for novel biologic | No established codes or reimbursement | Demonstrate cost-effectiveness vs current care; establish value-based pricing | 5-10 years |
| Manufacturing economics | Production costs potentially prohibitive | Economies of scale not yet achieved | Optimize yields; develop scalable processes; explore biosimilar pathways post-approval | 5-7 years |
Immediate research priorities (1-3 years): Consensus protocols for MSC culture, secretome collection, and EV isolation are urgently needed, with collaboration among International Society for Extracellular Vesicles, International Society for Cell and Gene Therapy, and regulators. Parallel efforts must validate candidate potency assays against clinical outcomes, enabling their adoption as quality control tools. Early clinical trials should integrate bioanalytical assessments to link in vitro assays with therapeutic efficacy. Preclinical studies in large animal models with longitudinal follow-up are essential to optimize delivery formulations, comparing hydrogels, nanoparticles, and HA combinations.
Medium-term goals (3-5 years): Definitive phase III trials must be adequately powered, placebo-controlled, and of sufficient duration (12-24 months) to assess durability. Standardized outcome measures and patient stratification will be critical. Identification of predictive biomarkers - imaging features, synovial fluid cytokines, or genetic variants - will enable precision medicine and improve trial efficiency. Concurrently, engineering next-generation secretomes with enhanced potency, improved targeting, or integration with complementary therapeutics should progress alongside validation of first-generation products.
Long-term vision (5-10 years): The ultimate goal is personalized secretome medicine, tailoring products to disease stage, phenotype, or patient-specific autologous sources. Proven efficacy in established OA could extend applications to prevention in high-risk groups, such as post-traumatic or obesity-related OA, shifting therapy toward prophylaxis. Finally, secretomes will likely integrate into multimodal treatment algorithms, complementing physical therapy, weight management, and biomechanical optimization to achieve comprehensive disease modification.
MSC-derived secretomes represent a paradigm shift in regenerative medicine for knee OA, offering cell-free therapies that address multiple pathological processes simultaneously. Through exosomal miRNAs, growth factor signaling, and immunomodulation, secretomes provide chondroprotective, anti-inflammatory, and bone-modulating effects, enabling comprehensive disease modification. Translating this promise requires standardized manufacturing, validated potency assays, and optimized delivery systems. Rigorous phase III trials are essential to confirm efficacy and safety. If phase III trials demonstrate efficacy, MSC-derived secretomes may potentially achieve clinical adoption within 5-10 years, offering promise for disease modification beyond symptomatic relief.
| 1. | Yang G, Wang J, Liu Y, Lu H, He L, Ma C, Zhao Z. Burden of Knee Osteoarthritis in 204 Countries and Territories, 1990-2019: Results From the Global Burden of Disease Study 2019. Arthritis Care Res (Hoboken). 2023;75:2489-2500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 83] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 2. | Cui A, Li H, Wang D, Zhong J, Chen Y, Lu H. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine. 2020;29-30:100587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1098] [Cited by in RCA: 910] [Article Influence: 151.7] [Reference Citation Analysis (0)] |
| 3. | Mocanu V, Timofte DV, Zară-Dănceanu CM, Labusca L. Obesity, Metabolic Syndrome, and Osteoarthritis Require Integrative Understanding and Management. Biomedicines. 2024;12:1262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 4. | Wang H, Cheng Y, Shao D, Chen J, Sang Y, Gui T, Luo S, Li J, Chen C, Ye Y, Yang Y, Li Y, Zha Z. Metabolic Syndrome Increases the Risk for Knee Osteoarthritis: A Meta-Analysis. Evid Based Complement Alternat Med. 2016;2016:7242478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 5. | Puig-Junoy J, Ruiz Zamora A. Socio-economic costs of osteoarthritis: a systematic review of cost-of-illness studies. Semin Arthritis Rheum. 2015;44:531-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 6. | Jiang P, Hu K, Jin L, Luo Z. A brief review of current treatment options for osteoarthritis including disease-modifying osteoarthritis drugs (DMOADs) and novel therapeutics. Ann Med Surg (Lond). 2024;86:4042-4048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 7. | Richard MJ, Driban JB, McAlindon TE. Pharmaceutical treatment of osteoarthritis. Osteoarthritis Cartilage. 2023;31:458-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 199] [Article Influence: 66.3] [Reference Citation Analysis (0)] |
| 8. | Ayhan E, Kesmezacar H, Akgun I. Intraarticular injections (corticosteroid, hyaluronic acid, platelet rich plasma) for the knee osteoarthritis. World J Orthop. 2014;5:351-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 226] [Cited by in RCA: 287] [Article Influence: 23.9] [Reference Citation Analysis (12)] |
| 9. | Wheeler SG. Steroid Knee Injections for Arthritis Are No Better than Placebo in a Randomized Controlled Trial. J Gen Intern Med. 2020;35:3137-3139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Liu H, Qin L, Liu Y, Meng X, Li C, He M. Knee osteoarthritis rehabilitation: an integrated framework of exercise, nutrition, biomechanics, and physical therapist guidance-a narrative review. Eur J Med Res. 2025;30:826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 11. | Ferket BS, Feldman Z, Zhou J, Oei EH, Bierma-Zeinstra SM, Mazumdar M. Impact of total knee replacement practice: cost effectiveness analysis of data from the Osteoarthritis Initiative. BMJ. 2017;356:j1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 119] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 12. | Bhandari M, Smith J, Miller LE, Block JE. Clinical and economic burden of revision knee arthroplasty. Clin Med Insights Arthritis Musculoskelet Disord. 2012;5:89-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 164] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 13. | Li S, Cao P, Chen T, Ding C. Latest insights in disease-modifying osteoarthritis drugs development. Ther Adv Musculoskelet Dis. 2023;15:1759720X231169839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 14. | Rodriguez-Merchan EC. The Current Role of Disease-modifying Osteoarthritis Drugs. Arch Bone Jt Surg. 2023;11:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 15. | Rose BJ, Kooyman DL. A Tale of Two Joints: The Role of Matrix Metalloproteases in Cartilage Biology. Dis Markers. 2016;2016:4895050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 138] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 16. | Donell S. Subchondral bone remodelling in osteoarthritis. EFORT Open Rev. 2019;4:221-229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 147] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 17. | Terkawi MA, Ebata T, Yokota S, Takahashi D, Endo T, Matsumae G, Shimizu T, Kadoya K, Iwasaki N. Low-Grade Inflammation in the Pathogenesis of Osteoarthritis: Cellular and Molecular Mechanisms and Strategies for Future Therapeutic Intervention. Biomedicines. 2022;10:1109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 68] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 18. | Jenei-Lanzl Z, Maurer S, Brenner RE, Zaucke F, Fuchs M, Riegger J. Emerging concepts and challenges in the development of disease-modifying osteoarthritis drugs - a more refined perspective. Arch Pharm Res. 2025;48:467-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 19. | Huang Z, Ding C, Li T, Yu SP. Current status and future prospects for disease modification in osteoarthritis. Rheumatology (Oxford). 2018;57:iv108-iv123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 20. | Zhu C, Wu W, Qu X. Mesenchymal stem cells in osteoarthritis therapy: a review. Am J Transl Res. 2021;13:448-461. [PubMed] |
| 21. | Mancuso P, Raman S, Glynn A, Barry F, Murphy JM. Mesenchymal Stem Cell Therapy for Osteoarthritis: The Critical Role of the Cell Secretome. Front Bioeng Biotechnol. 2019;7:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 164] [Article Influence: 23.4] [Reference Citation Analysis (1)] |
| 22. | Chahal J, Gómez-Aristizábal A, Shestopaloff K, Bhatt S, Chaboureau A, Fazio A, Chisholm J, Weston A, Chiovitti J, Keating A, Kapoor M, Ogilvie-Harris DJ, Syed KA, Gandhi R, Mahomed NN, Marshall KW, Sussman MS, Naraghi AM, Viswanathan S. Bone Marrow Mesenchymal Stromal Cell Treatment in Patients with Osteoarthritis Results in Overall Improvement in Pain and Symptoms and Reduces Synovial Inflammation. Stem Cells Transl Med. 2019;8:746-757. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 215] [Cited by in RCA: 182] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 23. | Lukomska B, Stanaszek L, Zuba-Surma E, Legosz P, Sarzynska S, Drela K. Challenges and Controversies in Human Mesenchymal Stem Cell Therapy. Stem Cells Int. 2019;2019:9628536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 433] [Cited by in RCA: 389] [Article Influence: 55.6] [Reference Citation Analysis (1)] |
| 24. | Ouzin M, Kogler G. Mesenchymal Stromal Cells: Heterogeneity and Therapeutical Applications. Cells. 2023;12:2039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (3)] |
| 25. | Musiał-Wysocka A, Kot M, Majka M. The Pros and Cons of Mesenchymal Stem Cell-Based Therapies. Cell Transplant. 2019;28:801-812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 464] [Cited by in RCA: 398] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 26. | Sanz-Nogués C, O'Brien T. Current good manufacturing practice considerations for mesenchymal stromal cells as therapeutic agents. Biomater Biosyst. 2021;2:100018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 27. | Alvites R, Branquinho M, Sousa AC, Lopes B, Sousa P, Maurício AC. Mesenchymal Stem/Stromal Cells and Their Paracrine Activity-Immunomodulation Mechanisms and How to Influence the Therapeutic Potential. Pharmaceutics. 2022;14:381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 108] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 28. | Trigo CM, Rodrigues JS, Camões SP, Solá S, Miranda JP. Mesenchymal stem cell secretome for regenerative medicine: Where do we stand? J Adv Res. 2025;70:103-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 106] [Article Influence: 106.0] [Reference Citation Analysis (1)] |
| 29. | Vizoso FJ, Eiro N, Cid S, Schneider J, Perez-Fernandez R. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18:1852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1124] [Cited by in RCA: 1000] [Article Influence: 111.1] [Reference Citation Analysis (0)] |
| 30. | González-González A, García-Sánchez D, Dotta M, Rodríguez-Rey JC, Pérez-Campo FM. Mesenchymal stem cells secretome: The cornerstone of cell-free regenerative medicine. World J Stem Cells. 2020;12:1529-1552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 139] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 31. | Műzes G, Sipos F. Mesenchymal Stem Cell-Derived Secretome: A Potential Therapeutic Option for Autoimmune and Immune-Mediated Inflammatory Diseases. Cells. 2022;11:2300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 128] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 32. | Gwam C, Mohammed N, Ma X. Stem cell secretome, regeneration, and clinical translation: a narrative review. Ann Transl Med. 2021;9:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 33. | Nguyen TH, Duong CM, Nguyen XH, Than UTT. Mesenchymal Stem Cell-Derived Extracellular Vesicles for Osteoarthritis Treatment: Extracellular Matrix Protection, Chondrocyte and Osteocyte Physiology, Pain and Inflammation Management. Cells. 2021;10:2887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 34. | Chouaib B, Haack-Sørensen M, Chaubron F, Cuisinier F, Collart-Dutilleul PY. Towards the Standardization of Mesenchymal Stem Cell Secretome-Derived Product Manufacturing for Tissue Regeneration. Int J Mol Sci. 2023;24:12594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 35. | Harrell CR, Fellabaum C, Jovicic N, Djonov V, Arsenijevic N, Volarevic V. Molecular Mechanisms Responsible for Therapeutic Potential of Mesenchymal Stem Cell-Derived Secretome. Cells. 2019;8:467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 415] [Cited by in RCA: 358] [Article Influence: 51.1] [Reference Citation Analysis (5)] |
| 36. | Munoz-Perez E, Gonzalez-Pujana A, Igartua M, Santos-Vizcaino E, Hernandez RM. Mesenchymal Stromal Cell Secretome for the Treatment of Immune-Mediated Inflammatory Diseases: Latest Trends in Isolation, Content Optimization and Delivery Avenues. Pharmaceutics. 2021;13:1802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 37. | Bazzoni R, Takam Kamga P, Tanasi I, Krampera M. Extracellular Vesicle-Dependent Communication Between Mesenchymal Stromal Cells and Immune Effector Cells. Front Cell Dev Biol. 2020;8:596079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 38. | Fernández-Pérez AG, Herrera-González A, López-Naranjo EJ, Martínez-Álvarez IA, Uribe-Rodríguez D, Ramírez-Arreola DE, Sánchez-Peña MJ, Navarro-Partida J. Extracellular Vesicles from Different Mesenchymal Stem Cell Types Exhibit Distinctive Surface Protein Profiling and Molecular Characteristics: A Comparative Analysis. Int J Mol Sci. 2025;26:3393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 39. | Liu M, Wen Z, Zhang T, Zhang L, Liu X, Wang M. The role of exosomal molecular cargo in exosome biogenesis and disease diagnosis. Front Immunol. 2024;15:1417758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 40. | Xu M, Ji J, Jin D, Wu Y, Wu T, Lin R, Zhu S, Jiang F, Ji Y, Bao B, Li M, Xu W, Xiao M. The biogenesis and secretion of exosomes and multivesicular bodies (MVBs): Intercellular shuttles and implications in human diseases. Genes Dis. 2023;10:1894-1907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 180] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 41. | Lao TD, Le TAH. Data Integration Reveals the Potential Biomarkers of Circulating MicroRNAs in Osteoarthritis. Diagnostics (Basel). 2021;11:412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 42. | Liao Z, Zheng R, Shao G. Mechanisms and application strategies of miRNA146a regulating inflammation and fibrosis at molecular and cellular levels (Review). Int J Mol Med. 2023;51:7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 43] [Reference Citation Analysis (0)] |
| 43. | Zhang Y, Jia J, Yang S, Liu X, Ye S, Tian H. MicroRNA-21 controls the development of osteoarthritis by targeting GDF-5 in chondrocytes. Exp Mol Med. 2014;46:e79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 44. | Cheng J, Ji D, Yin Y, Wang S, Song K, Pan Q, Zhang Q, Yang L. Proteomic profiling of serum small extracellular vesicles reveals immune signatures of children with pneumonia. Transl Pediatr. 2022;11:891-908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 45. | Ying J, Wang P, Zhang S, Xu T, Zhang L, Dong R, Xu S, Tong P, Wu C, Jin H. Transforming growth factor-beta1 promotes articular cartilage repair through canonical Smad and Hippo pathways in bone mesenchymal stem cells. Life Sci. 2018;192:84-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 46. | Montaseri A, Busch F, Mobasheri A, Buhrmann C, Aldinger C, Rad JS, Shakibaei M. IGF-1 and PDGF-bb suppress IL-1β-induced cartilage degradation through down-regulation of NF-κB signaling: involvement of Src/PI-3K/AKT pathway. PLoS One. 2011;6:e28663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 168] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 47. | Kang Q, Song WX, Luo Q, Tang N, Luo J, Luo X, Chen J, Bi Y, He BC, Park JK, Jiang W, Tang Y, Huang J, Su Y, Zhu GH, He Y, Yin H, Hu Z, Wang Y, Chen L, Zuo GW, Pan X, Shen J, Vokes T, Reid RR, Haydon RC, Luu HH, He TC. A comprehensive analysis of the dual roles of BMPs in regulating adipogenic and osteogenic differentiation of mesenchymal progenitor cells. Stem Cells Dev. 2009;18:545-559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 317] [Cited by in RCA: 321] [Article Influence: 18.9] [Reference Citation Analysis (2)] |
| 48. | Palamà MEF, Shaw GM, Carluccio S, Reverberi D, Sercia L, Persano L, Pisignano D, Cortese K, Barry FP, Murphy JM, Gentili C. The Secretome Derived From Mesenchymal Stromal Cells Cultured in a Xeno-Free Medium Promotes Human Cartilage Recovery in vitro. Front Bioeng Biotechnol. 2020;8:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 49. | Hutchins AP, Diez D, Miranda-Saavedra D. The IL-10/STAT3-mediated anti-inflammatory response: recent developments and future challenges. Brief Funct Genomics. 2013;12:489-498. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 339] [Article Influence: 26.1] [Reference Citation Analysis (3)] |
| 50. | Jin K, Qian C, Lin J, Liu B. Cyclooxygenase-2-Prostaglandin E2 pathway: A key player in tumor-associated immune cells. Front Oncol. 2023;13:1099811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 122] [Reference Citation Analysis (0)] |
| 51. | Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-κB signaling in resident macrophages. Blood. 2011;118:330-338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 558] [Cited by in RCA: 525] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 52. | Zheng Q, Zhang S, Guo WZ, Li XK. The Unique Immunomodulatory Properties of MSC-Derived Exosomes in Organ Transplantation. Front Immunol. 2021;12:659621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 53. | Liu X, Wei Q, Lu L, Cui S, Ma K, Zhang W, Ma F, Li H, Fu X, Zhang C. Immunomodulatory potential of mesenchymal stem cell-derived extracellular vesicles: Targeting immune cells. Front Immunol. 2023;14:1094685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 54. | Dalle Carbonare L, Bertacco J, Minoia A, Cominacini M, Bhandary L, Elia R, Gambaro G, Mottes M, Valenti MT. Modulation of miR-204 Expression during Chondrogenesis. Int J Mol Sci. 2022;23:2130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 55. | Guo L, Xiang W, Pan Z, Gu H, Jiang X. Post-translational modifications of collagen and its related diseases in metabolic pathways. Acta Pharm Sin B. 2025;15:1773-1795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 56. | Wen C, Xu L, Xu X, Wang D, Liang Y, Duan L. Insulin-like growth factor-1 in articular cartilage repair for osteoarthritis treatment. Arthritis Res Ther. 2021;23:277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 57. | Hui W, Cawston T, Rowan AD. Transforming growth factor beta 1 and insulin-like growth factor 1 block collagen degradation induced by oncostatin M in combination with tumour necrosis factor alpha from bovine cartilage. Ann Rheum Dis. 2003;62:172-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 58. | Miao C, Zhou W, Wang X, Fang J. The Research Progress of Exosomes in Osteoarthritis, With Particular Emphasis on the Mediating Roles of miRNAs and lncRNAs. Front Pharmacol. 2021;12:685623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 59. | Jiang L, Lin J, Zhao S, Wu J, Jin Y, Yu L, Wu N, Wu Z, Wang Y, Lin M. ADAMTS5 in Osteoarthritis: Biological Functions, Regulatory Network, and Potential Targeting Therapies. Front Mol Biosci. 2021;8:703110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 84] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 60. | Lu X, Lin J, Jin J, Qian W, Weng X. Hsa-miR-15a exerts protective effects against osteoarthritis by targeting aggrecanase-2 (ADAMTS5) in human chondrocytes. Int J Mol Med. 2016;37:509-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 61. | Li HZ, Xu XH, Lin N, Wang DW, Lin YM, Su ZZ, Lu HD. Overexpression of miR-10a-5p facilitates the progression of osteoarthritis. Aging (Albany NY). 2020;12:5948-5976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 62. | Duan L, Liang Y, Xu X, Xiao Y, Wang D. Recent progress on the role of miR-140 in cartilage matrix remodelling and its implications for osteoarthritis treatment. Arthritis Res Ther. 2020;22:194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 76] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 63. | Wu C, Wei Y, Kolmas J, Yang K. MicroRNA-146a-3p as an alternative to anti-IL-6 monoclonal antibody therapy: Intra-articular liposome delivery for osteoarthritis treatment. Med Hypotheses. 2025;200:111666. [DOI] [Full Text] |
| 64. | Chen B, Huang SG, Ju L, Li M, Nie FF, Zhang Y, Zhang YH, Chen X, Gao F. Effect of microRNA-21 on the proliferation of human degenerated nucleus pulposus by targeting programmed cell death 4. Braz J Med Biol Res. 2016;49:e5020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 65. | Klaymook S, Tirawanchai N, Wichitwiengrat S, Chuaynarong P, Thongbopit S, Chareancholvanich K, Phermthai T. MSC secretome from amniotic fluid halts IL-1β and TNF-α inflammation via the ERK/MAPK pathway, promoting cartilage regeneration in OA in vitro. J Stem Cells Regen Med. 2024;20:3-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 66. | Gupta SC, Sundaram C, Reuter S, Aggarwal BB. Inhibiting NF-κB activation by small molecules as a therapeutic strategy. Biochim Biophys Acta. 2010;1799:775-787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 656] [Cited by in RCA: 619] [Article Influence: 38.7] [Reference Citation Analysis (0)] |
| 67. | Cho DI, Kim MR, Jeong HY, Jeong HC, Jeong MH, Yoon SH, Kim YS, Ahn Y. Mesenchymal stem cells reciprocally regulate the M1/M2 balance in mouse bone marrow-derived macrophages. Exp Mol Med. 2014;46:e70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 443] [Cited by in RCA: 419] [Article Influence: 34.9] [Reference Citation Analysis (1)] |
| 68. | Wang W, Liang M, Wang L, Bei W, Rong X, Xu J, Guo J. Role of prostaglandin E2 in macrophage polarization: Insights into atherosclerosis. Biochem Pharmacol. 2023;207:115357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 29] [Reference Citation Analysis (0)] |
| 69. | Song Y, Dou H, Li X, Zhao X, Li Y, Liu D, Ji J, Liu F, Ding L, Ni Y, Hou Y. Exosomal miR-146a Contributes to the Enhanced Therapeutic Efficacy of Interleukin-1β-Primed Mesenchymal Stem Cells Against Sepsis. Stem Cells. 2017;35:1208-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 397] [Article Influence: 44.1] [Reference Citation Analysis (4)] |
| 70. | Khatab S, van Osch GJ, Kops N, Bastiaansen-Jenniskens YM, Bos PK, Verhaar JA, Bernsen MR, van Buul GM. Mesenchymal stem cell secretome reduces pain and prevents cartilage damage in a murine osteoarthritis model. Eur Cell Mater. 2018;36:218-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 71. | Zhang H, Wang J, Deng F, Huang E, Yan Z, Wang Z, Deng Y, Zhang Q, Zhang Z, Ye J, Qiao M, Li R, Wang J, Wei Q, Zhou G, Luu HH, Haydon RC, He TC, Deng F. Canonical Wnt signaling acts synergistically on BMP9-induced osteo/odontoblastic differentiation of stem cells of dental apical papilla (SCAPs). Biomaterials. 2015;39:145-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 136] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 72. | Cho KA, Park M, Kim YH, Ryu KH, Woo SY. Mesenchymal stem cells inhibit RANK-RANKL interactions between osteoclasts and Th17 cells via osteoprotegerin activity. Oncotarget. 2017;8:83419-83431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 73. | Li XL, Zhao YQ, Miao L, An YX, Wu F, Han JY, Han JY, Tay FR, Mu Z, Jiao Y, Wang J. Strategies for promoting neurovascularization in bone regeneration. Mil Med Res. 2025;12:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 74. | Sears V, Ghosh G. Harnessing mesenchymal stem cell secretome: Effect of extracellular matrices on proangiogenic signaling. Biotechnol Bioeng. 2020;117:1159-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 75. | Giannasi C, Della Morte E, Cadelano F, Valenza A, Casati S, Dei Cas M, Niada S, Brini AT. Boosting the therapeutic potential of cell secretome against osteoarthritis: Comparison of cytokine-based priming strategies. Biomed Pharmacother. 2024;170:115970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 76. | Chou C, Wisniewski H, Band P, Huebner J, Stabler T, Lattermann C, Kraus V. TSG-6 Activity reflects severity of inflammation in knee osteoarthritis and acute joint injury. Osteoarthr Cartil. 2016;24:S81-S82. [DOI] [Full Text] |
| 77. | Li JJ, Hosseini-Beheshti E, Grau GE, Zreiqat H, Little CB. Stem Cell-Derived Extracellular Vesicles for Treating Joint Injury and Osteoarthritis. Nanomaterials (Basel). 2019;9:261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 78. | Ni Z, Zhou S, Li S, Kuang L, Chen H, Luo X, Ouyang J, He M, Du X, Chen L. Exosomes: roles and therapeutic potential in osteoarthritis. Bone Res. 2020;8:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 219] [Article Influence: 36.5] [Reference Citation Analysis (0)] |
| 79. | Sun C, Teng F, Xia Y. Extracellular vesicles in osteoarthritis: mechanisms, therapeutic potential, and diagnostic applications. Front Immunol. 2025;16:1595095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 80. | Wu CW, Huang YH, Shao PL, Chang LH, Lu CC, Chen CH, Fu YC, Ho ML, Chang JK, Wu SC. Umbilical Cord Mesenchymal Stem Cell-Derived Extracellular Vesicles Enhance Chondrocyte Function by Reducing Oxidative Stress in Chondrocytes. Int J Mol Sci. 2025;26:7683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 81. | Tao SC, Yuan T, Zhang YL, Yin WJ, Guo SC, Zhang CQ. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics. 2017;7:180-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 629] [Cited by in RCA: 591] [Article Influence: 65.7] [Reference Citation Analysis (4)] |
| 82. | Mao G, Zhang Z, Hu S, Zhang Z, Chang Z, Huang Z, Liao W, Kang Y. Exosomes derived from miR-92a-3p-overexpressing human mesenchymal stem cells enhance chondrogenesis and suppress cartilage degradation via targeting WNT5A. Stem Cell Res Ther. 2018;9:247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 369] [Article Influence: 46.1] [Reference Citation Analysis (1)] |
| 83. | Rong Y, Zhang J, Jiang D, Ji C, Liu W, Wang J, Ge X, Tang P, Yu S, Cui W, Cai W. Hypoxic pretreatment of small extracellular vesicles mediates cartilage repair in osteoarthritis by delivering miR-216a-5p. Acta Biomater. 2021;122:325-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (3)] |
| 84. | Zhou H, Shen X, Yan C, Xiong W, Ma Z, Tan Z, Wang J, Li Y, Liu J, Duan A, Liu F. Extracellular vesicles derived from human umbilical cord mesenchymal stem cells alleviate osteoarthritis of the knee in mice model by interacting with METTL3 to reduce m6A of NLRP3 in macrophage. Stem Cell Res Ther. 2022;13:322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 122] [Article Influence: 30.5] [Reference Citation Analysis (4)] |
| 85. | Zhang H, Yang Y, Wu Y, Denslin V, Koh YWJ, Liu L, Zhuo W, Lam WMR, Yu Y, Hui JHP, Yang Z. Unveiling the potential of MSC extracellular vesicles: MiR-122-5p enhancing chondrocyte regeneration in osteoarthritis via autophagy mechanism. Stem Cell Res Ther. 2025;16:289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 86. | Kong Y, Wang Y, Yang Y, Hou Y, Yu J, Liu M, Xie S, Song Y. Intra-articular injection of exosomes derived from different stem cells in animal models of osteoarthritis: a systematic review and meta- analysis. J Orthop Surg Res. 2024;19:834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 87. | Zhuang Y, Jiang S, Yuan C, Lin K. The potential therapeutic role of extracellular vesicles in osteoarthritis. Front Bioeng Biotechnol. 2022;10:1022368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 88. | De Luna A, Otahal A, Nehrer S. Mesenchymal Stromal Cell-Derived Extracellular Vesicles - Silver Linings for Cartilage Regeneration? Front Cell Dev Biol. 2020;8:593386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 89. | Wang Z, Hu Z, Niu L, Xu Y, Qi Y. Mesenchymal stem cell-derived exosomes for the treatment of knee osteoarthritis: a systematic review and meta-analysis based on rat model. Front Pharmacol. 2025;16:1588841. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 90. | Su J, Qi Y, Niu L, Wang Y, Wei B, Ma B, Kong K, Zhai Z, Xu Y. The role of synovial mesenchymal stem cell-derived exosomes in cartilage repair: a systematic review. Front Pharmacol. 2025;16:1617874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 91. | Vonk LA, van Dooremalen SFJ, Liv N, Klumperman J, Coffer PJ, Saris DBF, Lorenowicz MJ. Mesenchymal Stromal/stem Cell-derived Extracellular Vesicles Promote Human Cartilage Regeneration In Vitro. Theranostics. 2018;8:906-920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 303] [Cited by in RCA: 293] [Article Influence: 36.6] [Reference Citation Analysis (4)] |
| 92. | Yang H, Yang H, Wang Q, Ji H, Qian T, Qiao Y, Shi J, Cong M. Mesenchymal stem cells and their extracellular vesicles: new therapies for cartilage repair. Front Bioeng Biotechnol. 2025;13:1591400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 93. | Figueroa-Valdés AI, Luz-Crawford P, Herrera-Luna Y, Georges-Calderón N, García C, Tobar HE, Araya MJ, Matas J, Donoso-Meneses D, de la Fuente C, Cuenca J, Parra E, Lillo F, Varela C, Cádiz MI, Vernal R, Ortloff A, Nardocci G, Castañeda V, Adasme-Vidal C, Kunze-Küllmer M, Hidalgo Y, Espinoza F, Khoury M, Alcayaga-Miranda F. Clinical-grade extracellular vesicles derived from umbilical cord mesenchymal stromal cells: preclinical development and first-in-human intra-articular validation as therapeutics for knee osteoarthritis. J Nanobiotechnology. 2025;23:13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 94. | Bolandnazar NS, Raeissadat SA, Haghighatkhah H, Rayegani SM, Oshnari RS, Keshel SH, Zahraei M, Aalipour K, Babaee M, Zamani A, Rad ZB, Soleimani M, Sefat F. Safety and efficacy of placental mesenchymal stromal cells-derived extracellular vesicles in knee osteoarthritis: a randomized, triple-blind, placebo-controlled clinical trial. BMC Musculoskelet Disord. 2024;25:856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 95. | Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F, Atkin-Smith GK, Ayre DC, Bach JM, Bachurski D, Baharvand H, Balaj L, Baldacchino S, Bauer NN, Baxter AA, Bebawy M, Beckham C, Bedina Zavec A, Benmoussa A, Berardi AC, Bergese P, Bielska E, Blenkiron C, Bobis-Wozowicz S, Boilard E, Boireau W, Bongiovanni A, Borràs FE, Bosch S, Boulanger CM, Breakefield X, Breglio AM, Brennan MÁ, Brigstock DR, Brisson A, Broekman ML, Bromberg JF, Bryl-Górecka P, Buch S, Buck AH, Burger D, Busatto S, Buschmann D, Bussolati B, Buzás EI, Byrd JB, Camussi G, Carter DR, Caruso S, Chamley LW, Chang YT, Chen C, Chen S, Cheng L, Chin AR, Clayton A, Clerici SP, Cocks A, Cocucci E, Coffey RJ, Cordeiro-da-Silva A, Couch Y, Coumans FA, Coyle B, Crescitelli R, Criado MF, D'Souza-Schorey C, Das S, Datta Chaudhuri A, de Candia P, De Santana EF, De Wever O, Del Portillo HA, Demaret T, Deville S, Devitt A, Dhondt B, Di Vizio D, Dieterich LC, Dolo V, Dominguez Rubio AP, Dominici M, Dourado MR, Driedonks TA, Duarte FV, Duncan HM, Eichenberger RM, Ekström K, El Andaloussi S, Elie-Caille C, Erdbrügger U, Falcón-Pérez JM, Fatima F, Fish JE, Flores-Bellver M, Försönits A, Frelet-Barrand A, Fricke F, Fuhrmann G, Gabrielsson S, Gámez-Valero A, Gardiner C, Gärtner K, Gaudin R, Gho YS, Giebel B, Gilbert C, Gimona M, Giusti I, Goberdhan DC, Görgens A, Gorski SM, Greening DW, Gross JC, Gualerzi A, Gupta GN, Gustafson D, Handberg A, Haraszti RA, Harrison P, Hegyesi H, Hendrix A, Hill AF, Hochberg FH, Hoffmann KF, Holder B, Holthofer H, Hosseinkhani B, Hu G, Huang Y, Huber V, Hunt S, Ibrahim AG, Ikezu T, Inal JM, Isin M, Ivanova A, Jackson HK, Jacobsen S, Jay SM, Jayachandran M, Jenster G, Jiang L, Johnson SM, Jones JC, Jong A, Jovanovic-Talisman T, Jung S, Kalluri R, Kano SI, Kaur S, Kawamura Y, Keller ET, Khamari D, Khomyakova E, Khvorova A, Kierulf P, Kim KP, Kislinger T, Klingeborn M, Klinke DJ 2nd, Kornek M, Kosanović MM, Kovács ÁF, Krämer-Albers EM, Krasemann S, Krause M, Kurochkin IV, Kusuma GD, Kuypers S, Laitinen S, Langevin SM, Languino LR, Lannigan J, Lässer C, Laurent LC, Lavieu G, Lázaro-Ibáñez E, Le Lay S, Lee MS, Lee YXF, Lemos DS, Lenassi M, Leszczynska A, Li IT, Liao K, Libregts SF, Ligeti E, Lim R, Lim SK, Linē A, Linnemannstöns K, Llorente A, Lombard CA, Lorenowicz MJ, Lörincz ÁM, Lötvall J, Lovett J, Lowry MC, Loyer X, Lu Q, Lukomska B, Lunavat TR, Maas SL, Malhi H, Marcilla A, Mariani J, Mariscal J, Martens-Uzunova ES, Martin-Jaular L, Martinez MC, Martins VR, Mathieu M, Mathivanan S, Maugeri M, McGinnis LK, McVey MJ, Meckes DG Jr, Meehan KL, Mertens I, Minciacchi VR, Möller A, Møller Jørgensen M, Morales-Kastresana A, Morhayim J, Mullier F, Muraca M, Musante L, Mussack V, Muth DC, Myburgh KH, Najrana T, Nawaz M, Nazarenko I, Nejsum P, Neri C, Neri T, Nieuwland R, Nimrichter L, Nolan JP, Nolte-'t Hoen EN, Noren Hooten N, O'Driscoll L, O'Grady T, O'Loghlen A, Ochiya T, Olivier M, Ortiz A, Ortiz LA, Osteikoetxea X, Østergaard O, Ostrowski M, Park J, Pegtel DM, Peinado H, Perut F, Pfaffl MW, Phinney DG, Pieters BC, Pink RC, Pisetsky DS, Pogge von Strandmann E, Polakovicova I, Poon IK, Powell BH, Prada I, Pulliam L, Quesenberry P, Radeghieri A, Raffai RL, Raimondo S, Rak J, Ramirez MI, Raposo G, Rayyan MS, Regev-Rudzki N, Ricklefs FL, Robbins PD, Roberts DD, Rodrigues SC, Rohde E, Rome S, Rouschop KM, Rughetti A, Russell AE, Saá P, Sahoo S, Salas-Huenuleo E, Sánchez C, Saugstad JA, Saul MJ, Schiffelers RM, Schneider R, Schøyen TH, Scott A, Shahaj E, Sharma S, Shatnyeva O, Shekari F, Shelke GV, Shetty AK, Shiba K, Siljander PR, Silva AM, Skowronek A, Snyder OL 2nd, Soares RP, Sódar BW, Soekmadji C, Sotillo J, Stahl PD, Stoorvogel W, Stott SL, Strasser EF, Swift S, Tahara H, Tewari M, Timms K, Tiwari S, Tixeira R, Tkach M, Toh WS, Tomasini R, Torrecilhas AC, Tosar JP, Toxavidis V, Urbanelli L, Vader P, van Balkom BW, van der Grein SG, Van Deun J, van Herwijnen MJ, Van Keuren-Jensen K, van Niel G, van Royen ME, van Wijnen AJ, Vasconcelos MH, Vechetti IJ Jr, Veit TD, Vella LJ, Velot É, Verweij FJ, Vestad B, Viñas JL, Visnovitz T, Vukman KV, Wahlgren J, Watson DC, Wauben MH, Weaver A, Webber JP, Weber V, Wehman AM, Weiss DJ, Welsh JA, Wendt S, Wheelock AM, Wiener Z, Witte L, Wolfram J, Xagorari A, Xander P, Xu J, Yan X, Yáñez-Mó M, Yin H, Yuana Y, Zappulli V, Zarubova J, Žėkas V, Zhang JY, Zhao Z, Zheng L, Zheutlin AR, Zickler AM, Zimmermann P, Zivkovic AM, Zocco D, Zuba-Surma EK. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9138] [Cited by in RCA: 8549] [Article Influence: 1068.6] [Reference Citation Analysis (17)] |
| 96. | Zhang Y, Lan M, Chen Y. Minimal Information for Studies of Extracellular Vesicles (MISEV): Ten-Year Evolution (2014-2023). Pharmaceutics. 2024;16:1394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 51] [Reference Citation Analysis (1)] |
| 97. | Gimona M, Brizzi MF, Choo ABH, Dominici M, Davidson SM, Grillari J, Hermann DM, Hill AF, de Kleijn D, Lai RC, Lai CP, Lim R, Monguió-Tortajada M, Muraca M, Ochiya T, Ortiz LA, Toh WS, Yi YW, Witwer KW, Giebel B, Lim SK. Critical considerations for the development of potency tests for therapeutic applications of mesenchymal stromal cell-derived small extracellular vesicles. Cytotherapy. 2021;23:373-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 193] [Article Influence: 38.6] [Reference Citation Analysis (3)] |
| 98. | Ragni E, Papait A, Taiana MM, De Luca P, Grieco G, Vertua E, Romele P, Colombo C, Silini AR, Parolini O, de Girolamo L. Cell culture expansion media choice affects secretory, protective and immuno-modulatory features of adipose mesenchymal stromal cell-derived secretomes for orthopaedic applications. Regen Ther. 2025;28:481-497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 99. | Patel JC, Shukla M, Shukla M. From bench to bedside: translating mesenchymal stem cell therapies through preclinical and clinical evidence. Front Bioeng Biotechnol. 2025;13:1639439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 29.0] [Reference Citation Analysis (1)] |
| 100. | Cherian DS, Bhuvan T, Meagher L, Heng TSP. Biological Considerations in Scaling Up Therapeutic Cell Manufacturing. Front Pharmacol. 2020;11:654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 101. | Jones IA, Togashi R, Wilson ML, Heckmann N, Vangsness CT Jr. Intra-articular treatment options for knee osteoarthritis. Nat Rev Rheumatol. 2019;15:77-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 392] [Article Influence: 56.0] [Reference Citation Analysis (12)] |
| 102. | Fonsi M, El Amrani AI, Gervais F, Vincent P. Intra-Articular Hyaluronic Acid and Chondroitin Sulfate: Pharmacokinetic Investigation in Osteoarthritic Rat Models. Curr Ther Res Clin Exp. 2020;92:100573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 103. | Morgen M, Tung D, Boras B, Miller W, Malfait AM, Tortorella M. Nanoparticles for improved local retention after intra-articular injection into the knee joint. Pharm Res. 2013;30:257-268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 104. | Gambaro FM, Ummarino A, Torres Andón F, Ronzoni F, Di Matteo B, Kon E. Drug Delivery Systems for the Treatment of Knee Osteoarthritis: A Systematic Review of In Vivo Studies. Int J Mol Sci. 2021;22:9137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 105. | Li L, Wang F, Zhu D, Hu S, Cheng K, Li Z. Engineering exosomes and exosome-like nanovesicles for improving tissue targeting and retention. Fundam Res. 2025;5:851-867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 44] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 106. | Zhang H, Yan W, Wang J, Xie S, Tao WA, Lee CW, Zhang X, Zhang G, Liu Y, Wei D, Hu J, Liu H, Liu F, Nie Y, Chen X, Xu H, Xia J, Wang S. Surface functionalization of exosomes for chondrocyte-targeted siRNA delivery and cartilage regeneration. J Control Release. 2024;369:493-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 57] [Reference Citation Analysis (0)] |
| 107. | Li X, Xu M, Geng Z, Liu Y. Functional hydrogels for the repair and regeneration of tissue defects. Front Bioeng Biotechnol. 2023;11:1190171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 108. | Zhu S, Li Y, He Z, Ji L, Zhang W, Tong Y, Luo J, Yu D, Zhang Q, Bi Q. Advanced injectable hydrogels for cartilage tissue engineering. Front Bioeng Biotechnol. 2022;10:954501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 109. | Wang M, Deng Z, Guo Y, Xu P. Designing functional hyaluronic acid-based hydrogels for cartilage tissue engineering. Mater Today Bio. 2022;17:100495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 47] [Reference Citation Analysis (0)] |
| 110. | Hui Y, Zheng X, Zheng Z, Wu C, Hao Y, Zhou B. Progress in the Application of Multifunctional Composite Hydrogels in Promoting Tissue Repair. ACS Omega. 2024;9:47964-47975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 111. | Tang G, Tan Z, Zeng W, Wang X, Shi C, Liu Y, He H, Chen R, Ye X. Recent Advances of Chitosan-Based Injectable Hydrogels for Bone and Dental Tissue Regeneration. Front Bioeng Biotechnol. 2020;8:587658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 110] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 112. | Sang X, Zhao X, Yan L, Jin X, Wang X, Wang J, Yin Z, Zhang Y, Meng Z. Thermosensitive Hydrogel Loaded with Primary Chondrocyte-Derived Exosomes Promotes Cartilage Repair by Regulating Macrophage Polarization in Osteoarthritis. Tissue Eng Regen Med. 2022;19:629-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 113. | Zhang FX, Liu P, Ding W, Meng QB, Su DH, Zhang QC, Lian RX, Yu BQ, Zhao MD, Dong J, Li YL, Jiang LB. Injectable Mussel-Inspired highly adhesive hydrogel with exosomes for endogenous cell recruitment and cartilage defect regeneration. Biomaterials. 2021;278:121169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 221] [Article Influence: 44.2] [Reference Citation Analysis (0)] |
| 114. | Wang S, Qiu Y, Qu L, Wang Q, Zhou Q. Hydrogels for Treatment of Different Degrees of Osteoarthritis. Front Bioeng Biotechnol. 2022;10:858656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 115. | Singhmar R, Lamba JK, Panwar A, Hong CA. Exploring exosomes in osteoarthritis: biogenesis, functional roles, and recent hydrogel-based delivery strategies. Biomater Sci. 2025;13:6525-6544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 116. | Ehab S, Gaser OA, Oyouni AAA, Kameli N, Alzahrani F, Abdal Dayem A. Engineered Extracellular Vesicles in Arthritic Diseases: Therapeutic Applications & Challenges. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2025;17:e70031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |