Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.116175
Revised: December 9, 2025
Accepted: February 11, 2026
Published online: April 26, 2026
Processing time: 166 Days and 12.5 Hours
Poly-L-lactic acid (PLLA) has been demonstrated to grow both subcutaneous adipose tissue and the dermis which may occur through stimulation of adipose-derived stem cells (ADSC). These cells maintain the integumentary system by regulating fibroblasts and immune cells, sustaining the extracellular matrix, and combating oxidative stress.
To use multiplex immunofluorescence to characterize macrophage subtypes in
Multiplex immunofluorescence was used to characterize the macrophage sub
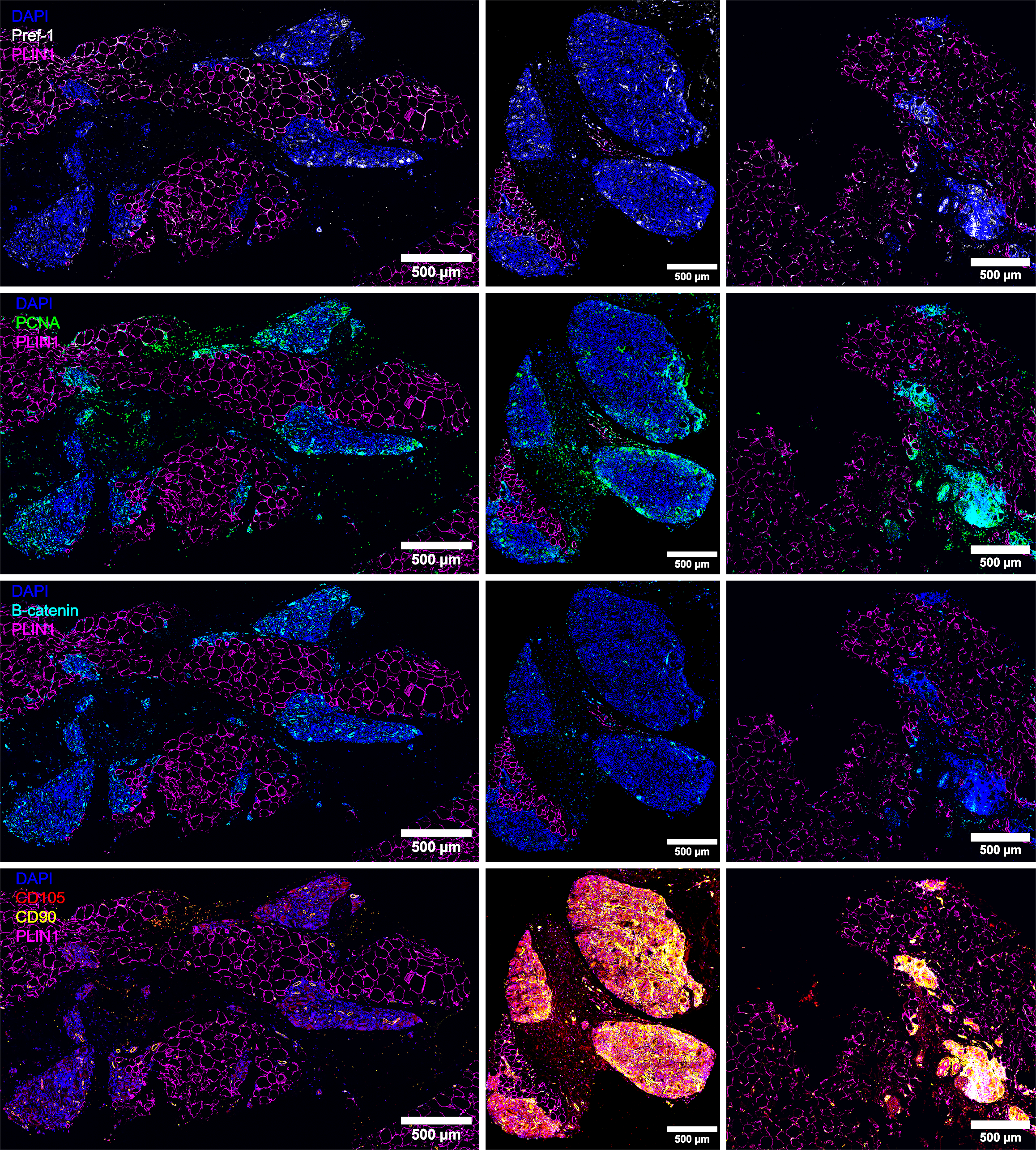
Absolute counts of ADSC and fibroblasts increased markedly in PLLA-treated tissues compared to baseline and controls. PLLA-macrophage-fibroblast encapsulations were high in ADSC with intense CD105, CD90, preadipocyte factor 1, proliferating cell nuclear antigen, β-catenin levels. This contrasts sharply with distant tissue, which was near absent in those signals and instead dominated by perilipin 1 consistent with mature adipocytes. Additionally, M2 macrophages predominated over M1 in these encapsulations, consistent with late-stage wound remodeling rather than infection-associated inflammation.
The study represents the first time native ADSC phenotype cells have been expanded in humans in vivo. PLLA has a well-established three-decade safety record as a minimally invasive skin-rejuvenation and volumization agent. This study suggests PLLA promotes a regenerative environment and may expand progenitor populations in vivo.
Core Tip: This study provides the first in vivo evidence that poly-L-lactic acid stimulates proliferation of cells expressing adipose-derived stem cell phenotype in human subcutaneous tissue. Using multiplex immunofluorescence on archived biopsy samples from a previous randomized clinical trial, poly-L-lactic acid-treated sites demonstrated marked expansion of cells exhibiting adipose-derived stem cell phenotype and fibroblasts in addition to a predominance of M2 macrophages consistent with a remodeling response. Most importantly, this may represent a novel regenerative paradigm using a mini
- Citation: Zubair RA, Zhou XH, Fabi SG. Adipose-derived stem cell stimulation using poly-L-lactic acid injection. World J Stem Cells 2026; 18(4): 116175
- URL: https://www.wjgnet.com/1948-0210/full/v18/i4/116175.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i4.116175
In most organisms, the aging process manifests as a time-dependent loss of physiological integrity, leading to increased susceptibility to chronic diseases and functional impairments. Contemporary research focuses on elucidating the conserved pathways that drive aging, particularly through the 12 hallmarks of aging[1], a framework defining the interconnected biological drivers of aging and potential interventions to slow or reverse them. Among these, stem cell exhaustion is an integrative hallmark characterized by the loss of tissue regenerative capacity, ranging from hema
In aging skin, prolonged inflammation causes collateral cellular damage, and DNA injury and telomere attrition further compromise proliferative capacity[5]. Understanding this progressive loss of intrinsic repair mechanisms has driven interest in transient biocompatible and biodegradable materials that can induce tissue rejuvenation. Injectable hyaluronic acid (HA) and poly-L-lactic acid (PLLA) are at the forefront of this race, demonstrating potential beyond volumetric support. Animal trials and in vitro experiments found that cells expressing the CD44 HA receptor had increased viability, proliferation, and differentiation[6,7]. This led to speculation that HA has the potential to stimulate adipogenesis in addition to serving as scaffolding for growth. Similarly, in a recent double-blinded, split-body trial (SPLASH study), female subjects with hip dell volume deficits were randomized to have one hip dell treated with 150 mg of PLLA while the contralateral hip dell received an equivalent volume of saline[8]. Subjects received three treatments at 1-month intervals and were followed for 9 months. At that time, the dermis and adipose layers increased in thickness by 26.1% and 27%, respectively, while there was no change in saline-treated tissue.
The finding that PLLA treatment results in dermal and hypodermal growth raised the possibility that PLLA may stimulate adipocyte-derived stem cells, positioning it as a modulator of local progenitor niches in aged tissue. When used to treat rat epidermal stem cells, PLLA has been found to enhance viability, migration, and proliferation while delaying differentiation and senescence. The exact mechanisms by which PLLA causes adipose tissue growth in humans remain unknown. One hypothesized mechanism from the SPLASH trial was PLLA upregulation of dikkopf-related protein 1 inhibiting Wnt signaling, leading to a decrease in β-catenin[9]. This allows Peroxisome proliferator-activated receptor gamma (PPARγ) expression so adipocyte differentiation can proceed[9]. It is also possible that PLLA-induced increases in collagen type VI and type IV lead to adipocyte differentiation[10,11].
The primary objective of the following study was to use multiplex immunofluorescence to characterize the macrophage subtypes involved in the PLLA-macrophage-fibroblast encapsulations on previously obtained biopsies (SPLASH
This study utilized subcutaneous adipose tissue obtained from 14 subjects who participated in a previous double-blind, placebo-controlled split-body clinical trial of injectable PLLA (PLLA-SCA. Sculptra® Aesthetic, Galderma Laboratories; Fort Worth, TX, United States) for treating female subjects 31 years old to 60 years old with hip dell volume deficits (SPLASH study)[8]. Subjects were randomized to have one hip dell treated with 150 mg of PLLA-SCA while the contralateral side received an equivalent volume of placebo (bacteriostatic saline). Subjects received three treatments at 1-month intervals and were followed for 9 months. A punch biopsy was performed on the right hip dell at day 0 (baseline) and again at 3 months and 6 months after the final treatment.
In the present study, 28 blocks of adipose tissue, consisting of 14 paired samples of pre- and post-treatment biopsies were analyzed. These samples were provided as formalin-fixed, paraffin-embedded blocks. Samples were shipped in tightly enclosed, well-sealed packages to the immunofluorescence laboratory (Visikol, Inc., Ashland, MA, United States) and stored at room temperature until processing, labeling, and imaging. All materials and respective product information for tissue preparation and labeling are listed below in Table 1.
| Material information | ||
| Glycine | Fisher scientific | 212485 |
| Normal donkey serum | Innovative Research, Inc. | IGDNSER500ML-36672 |
| Goat serum | Gibco | 16210064 |
| TBS (10 ×), pH = 7.4 | Quality Biological | 351-086-491 |
| TritonX-100 | Thermo Scientific | A16046.AP |
| Xylene substitute-3 | StatLab | 7400-1 |
| 100% reagent alcohol | StatLab | 6900-1 |
| 95% reagent alcohol | StatLab | 9500-1 |
| 70% reagent alcohol type 1 water blend | StatLab | 7070-1 |
| Citraconic anhydride | Thermo Scientific Chemicals | L05238.14 |
| PCNA monoclonal antibody (10D10E11) | Thermo Fisher Scientific | 60097-1-IG |
| CD90 monoclonal antibody (2D7D11) | Thermo Fisher Scientific | 66766-1-IG |
| Endoglin/CD105 polyclonal antibody | Thermo Fisher Scientific | 10862-1-AP |
| CD31 monoclonal antibody (2H8) | Invitrogen | MA3105 |
| Adiponectin monoclonal antibody (OTI3G10), TrueMAB™ | Thermo Fisher Scientific | TA804768 |
| DLK1 polyclonal antibody | Thermo Fisher Scientific | 29288-1-AP |
| Β-catenin monoclonal antibody (CAT-5H10) | Invitrogen | 13-8400 |
| PPARγ monoclonal antibody (1F4A2) | Thermo Fisher Scientific | 66936-1-IG |
| Perilipin-1 (D1D8) XP® rabbit mAb #9349 | Cell Signaling Technology | 9349 |
| Anti-CD68 antibody (EPR20545) - goat IgG (Chimeric) | Abcam | ab289671 |
| Anti-CD80 antibody (RM1133) | Abcam | ab315832 |
| CD163 recombinant rabbit monoclonal antibody (JA51-30) | Thermo Fisher Scientific | MA5-32684 |
| DAPI | Invitrogen | D3571 |
| Donkey αGoat 594 | Invitrogen | A11058 |
| Donkey αGoat 647 | Invitrogen | A32849 |
| Donkey αRabbit 647 | Invitrogen | A31573 |
| Goat αMouse IgG1 488 | Invitrogen | A21121 |
| Goat αMouse IgG1 647 | Invitrogen | A21240 |
| Goat αMouse IgG2a 594 | Invitrogen | A21135 |
| Goat αRabbit 555 | Invitrogen | A21428 |
| Goat αRabbit 594 | Invitrogen | A11012 |
| Goat αRabbit 647 | Invitrogen | A21245 |
| Fluoro-Gel (with Tris buffer) | Electron Microscopy Sciences | 17985-10 |
Validation of 13 antibodies [proliferating cell nuclear antigen (PCNA), CD90, CD105, CD31, preadipocyte factor 1 (Pref-1), perilipin 1 (PLIN1), adiponectin, β-catenin, PPAR-γ, CD68, CD80, CD206, CD163] was carried out on the corresponding control tissue (Table 2). All validations were performed using a concentration gradient composed of three antibody dilutions. The dilutions chosen for each antibody were based on the manufacturer’s recommended range. After com
| Antibody | SKU# | Concentration gradient | Optimal concentration | Control tissue |
| PCNA | 60097-1-IG | 1:500, 1:1000, 1:2000 | 1:500 | Human tonsil |
| CD90 | 66766-1-IG | 1:250, 1:500, 1:1000 | 1:250 | Human hippocampus |
| CD105 | 10862-1-AP | 1:1000, 1:2000, 1:4000 | 1:1,000 | Human testis |
| CD31 | MA3105 | 3 μg/mL, 9 μg/mL, 15 μg/mL | 15 μg/mL | Human lung |
| Adiponectin | TA804768 | 1:50, 1:100, 1:200 | 1:50 | Human adipose tissue |
| Pref-1 | 29288-1-AP | 1:100, 1:200, 1:500 | 1:100 | Human placenta |
| β-catenin | 13-8400 | 1:100, 1:500, 1:1000 | 1:100 | Human tonsil |
| PLIN1 | 66936-1-IG | 1:50, 1:100, 1:200 | 1:100 | Human adipose tissue |
| PPARγ | 9349 | 1:250, 1:500, 1:1000 | 1:250 | Human tonsil |
| CD68 | ab289671 | 1:500, 1:1000, 1:2000 | 1:500 | Human lung |
| CD80 | ab315832 | 1:50, 1:100, 1:200 | 1:100 | Human lymph |
| CD163 | MA5-32684 | 1:50, 1:100, 1:200 | 1:50 | Human lung |
The validation slides were baked at 60 °C for 1 hour, dewaxed with xylene substitute, then rehydrated in a stepwise gradient of 100% reagent alcohol (RA) for two rounds (5 minutes each), two rounds of 95% RA (5 minutes each), one round of 70% RA (5 minutes), and one round of 50% RA for 3 minutes. The slides were then placed in three rounds of MilliQ water for 3 minutes each to complete hydration. After rehydration, the validation slides underwent antigen retrieval to expose binding sites using citraconic anhydride for 45 minutes, cooled at room temperature for 30 minutes, followed by two washes with Tris-buffered saline (TBS) and a wash in Tris-buffered saline tween (TBST) on an orbital shaker, 5 minutes each. Following antigen retrieval, the slides were blocked in blocking buffer; either 6% donkey serum or 6% goat serum, depending on the secondary antibody, in TBST with 0.3 M glycine, for 1 hour. This was followed by an overnight incubation at 4 °C with the primary antibody in blocking buffer and the secondary control received blocking buffer only with no antibody added. After the overnight incubation, the slides were washed three times with TBST on an orbital shaker, followed by a 1-hour secondary antibody incubation at room temperature with a dilution of 1:500 for each.
Following the secondary incubation, antibody solution was removed, and slides were incubated with 4’,6-diamidino-2-phenylindole (DAPI) (Invitrogen, D3571, CA, United States), diluted at 1:4000 in TBS for 8 minutes. Following nuclear labeling, slides were washed with two rounds of TBST and one round of TBS on an orbital shaker, 5 minutes each. The slides were then mounted with Fluoro-Gel Mounting Medium with Tris and cover slipped for imaging.
Experimental and control slides were baked at 60 °C for 1 hour, dewaxed with Xylene Substitute, then rehydrated in a stepwise gradient of 100% RA for two rounds (5 minutes each), two rounds of 95% RA (5 minutes each), one round of 70% RA (5 minutes), and one round of 50% RA (3 minutes). The slides were then placed in three rounds of MilliQ water for 3 minutes each to complete hydration. After rehydration, the slides underwent antigen retrieval to expose binding sites using citraconic anhydride for 45 minutes, cooled at room temperature for 30 minutes, followed by two washes with TBS and a wash in TBST on an orbital shaker, 5 minutes each.
Following antigen retrieval, the slides were blocked in blocking buffer; either 6% donkey serum or 6% goat serum, depending on the secondary antibody, in TBST with 0.3 M glycine, for 1 hour. This was followed by an overnight incubation at 4 °C with the primary antibody in blocking buffer and the secondary control received blocking buffer only with no antibody added. Primary antibody labeling of experimental and control samples was labeled with validated concentrations for final tissue labeling (Table 2). After the overnight incubation, the slides were washed three times with TBST on an orbital shaker, followed by a 1-hour secondary antibody incubation at room temperature with a dilution of 1:500 for each. Following the secondary incubation, antibody solution was removed, and slides were incubated with DAPI, diluted at 1:4000 in TBS for 8 minutes. Following nuclear labeling, slides were washed with two rounds of TBST and one round of TBS on an orbital shaker, five minutes each. The slides were then mounted with Fluoro-Gel Mounting Medium with Tris and cover slipped for imaging.
Samples were split into two labeling groups. Primary study samples underwent labeling and stripping for a total of four panels (Table 3). Macrophage analysis samples underwent labeling and stripping for a total of two panels (Table 4). Following imaging, labeled antibodies were stripped using Visikol’s EasyPlex™ Stripping Solution. Antibodies were stripped from the tissue sample via room temperature incubation of samples within the stripping solution on a rocker overnight. After stripping was completed, the samples were washed three times in MilliQ water on a rocker. The next panel was then applied using the method stated above, starting from the blocking step.
| Primary antibody | Vendor and SKU | Primary concentration | Secondary antibody | Vendor and SKU | Secondary concentration |
| Panel 1 | |||||
| PCNA | Thermofisher, 60097-1-IG | 1:500 | Goat αMouse IgG1 488 | Invitrogen, A21121 | 1:500 |
| CD105 | Thermofisher, 10862-1-AP | 1:1000 | Goat αRabbit 555 | Invitrogen, A21428 | 1:500 |
| CD90 | Thermofisher, 66766-1-IG | 1:250 | Goat αMouse IgG2a 594 | Invitrogen, A21135 | 1:500 |
| CD31 | Invitrogen, MA3105 | 15 μg/mL | Donkey αGoat 647 | Invitrogen, A21447 | 1:500 |
| Panel 2 | |||||
| β-catenin | Invitrogen, 13-8400 | 1:100 | Goat αMouse IgG1 488 | Invitrogen, A21121 | 1:500 |
| Adiponectin | Thermofisher, TA804768 | 1:50 | Goat αMouse IgG2b 594 | Invitrogen, A21145 | 1:500 |
| Pref-1 | Thermofisher, 29288-1-AP | 1:100 | Goat αRabbit 647 | Invitrogen, A21245 | 1:500 |
| Panel 3 | |||||
| PLIN1 | Cell Signaling Technology, 9349 | 1:100 | Goat αRabbit 594 | Invitrogen, A11012 | 1:500 |
| Panel 4 | |||||
| PPAR-γ | Thermofisher, 66936-1-IG | 1:250 | Goat αMouse IgG1 647 | Invitrogen, A21240 | 1:500 |
| Primary antibody | Vendor and SKU | Primary concentration | Secondary antibody | Vendor and SKU | Secondary concentration |
| Panel 1 | |||||
| CD68 | Abcam, ab289671 | 1:500 | Donkey αGoat 594 | Invitrogen, A11058 | 1:500 |
| CD80 | Thermofisher, 66406-1-IG | 1:100 | Donkey αRabbit 647 | Invitrogen, A31573 | 1:500 |
| Panel 2 | |||||
| CD163 | Thermofisher, MA5-32684 | 1:50 | Goat αRabbit 647 | Invitrogen, A21245 | 1:500 |
All fluorescently labeled slides were imaged on a Zeiss Axioscan 7 at 20X (0.344 μm/pixel) magnification. The fluorescently labeled slides were imaged with uniform exposure times across all labeled channels for each respective tissue type and controls for group 1 and macrophage analysis samples (Table 5). Primary study, panel 1 channels were DAPI at 350 nm, PCNA at 488 nm, CD105 at 555 nm, CD90 at 594 nm and CD31 at 647 nm; panel 2 channels were DAPI at 350 nm, β-catenin at 488 nm, adiponectin at 594 nm and Pref-1 at 647 nm; panel 3 channels were DAPI at 350 nm and PLIN1 at 594 nm; panel 4 channels were DAPI at 350 nm and PPARγ at 647 nm. Macrophage analysis channels were DAPI at 350 nm, CD68 at 594 nm, CD80 at 647 nm, and CD163 at 647 nm.
After image acquisition, a cell population analysis was performed to ascertain the positivity of the markers within detected cells for each image. The cell population analysis was accomplished by segmenting cells through the DAPI channel then colocalizing the thresholded markers to the detected cells to determine the positivity of each cell. Thresholding for each of the markers was accomplished through an auto-local thresholding algorithm after a background correction was performed to normalize the intensity within the images and outline positive signal. Parameters to guide the algorithm were set by optimizing the thresholding on a subset of representative regions from the images in the sample set for each marker. For identification of the cells, the DAPI marker was thresholded through the aforementioned method and a segmentation algorithm was used to isolate the cells as separate objects within the image. Once complete, positivity was quantified by measuring the colocalization of the thresholded markers within each identified cell, which was slightly expanded to account for the cell body.
The total cell counts and percentage of cells matching each phenotype of interest were measured for each slide. Fibroblast counts and percentages were measured by subtracting the quantity of adipose-derived stem cells (ADSC) (Pref1+/CD105+/CD90+/CD31-) from the total quantity of stromal cells (CD105+/CD90+/CD31-). For each subject, the cell counts and percentage of cells from pre-treatment samples were subtracted from those samples collected at 6 months post-treatment. A similar process was performed for adiponectin, PLIN1, β-catenin, nuclear β-catenin, and PPARγ.
Three subcutaneous tissue samples that had successfully captured PLLA-macrophage-fibroblast encapsulations in the previous trial underwent staining and immunofluorescence imaging as described above. The cell counts of M2 and M1 macrophages were calculated for each of these three samples using CD68 and CD80 colocalization to identify M1 macrophages and by using CD163 to identify M2 macrophages. The ratio of the M1/M2 counts was calculated from the counts.
Statistical analyses were conducted using GraphPad Prism statistical software (Dotmatics, Boston, MA, United States). Each endpoint (individual proteins or cell phenotype panels) had individual change score calculated (Δ = 6-month post-treatment count - baseline count) for each subject. These change scores were then compared between groups using independent-samples t-tests to determine if mean change differs between control and treatment arms. A significance level (α) of 0.05 was used. The false discovery rate was controlled using the Benjamini-Hochberg procedure to adjust P-values across the secondary endpoints.
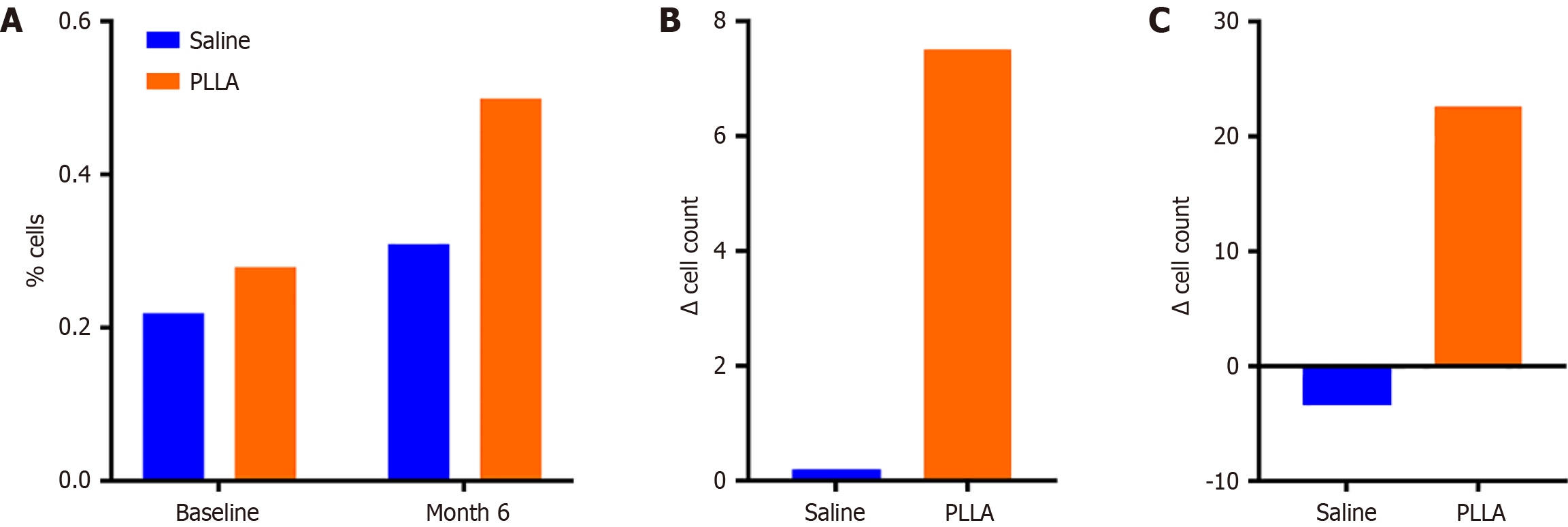
Tissue cellularity increases were greater in PLLA-treated samples than saline-treated samples (saline: 158.5 ± 20.7 to 259.7 ± 61.3; PLLA: 184.0 ± 38.2 nuclei/mm2 to 612.1 ± 111.3 nuclei/mm2).
The increase in ADSCs (identified as Pref-1+/CD105+/CD90+/CD31-) as a percentage of cells was two-fold greater in PLLA- (0.18%, from 0.28% to 0.46%) vs saline-treated tissues (0.9%, from 0.22% to 0.31%) (Figure 1A). Absolute ADSC counts increased markedly in PLLA-treated tissues (7.50 ± 6.81 cells, from 5.5 cells to 13.0 cells) vs saline controls (0.20 ± 0.86 cells, from 4.6 cells to 4.8 cells) (Figure 1B). Similar trends were seen in fibroblasts as a percentage (saline: 0.01% ± 0.19%, from 0.35% to 0.36%; PLLA: 0.56% ± 0.21%, from 0.26% to 0.82%) and absolute counts (saline: -3.4 ± 2.3, from 7 cells to 3.6 cells; PLLA: 22.6 ± 12.4 cells, from 4.5 cells to 27.1 cells) (Figure 1C). The change in proliferating stromal cells (PCNA+/CD105+/CD90+), representing ADSCs and fibroblasts in S-phase, also followed this pattern comparing saline- (-0.18% ± 0.60%, from 2.10% to 1.92%; -5.4 ± 19.1 cells from 38.8 cells to 33.4 cells) and PLLA-treated tissues (0.45% ± 1.16%, from 3.15% to 3.6%; 62.6 ± 52.1 cells, from 52.0 cells to 114.6 cells).
Pref-1 expressing cells increased by 18.62% ± 6.83% in saline-treated tissues as they decreased by 11.80% ± 3.32% in PLLA-treated tissues (P = 0.18). Similar differences were seen with PPARγ and PLIN1. PLIN1+ cells/mm2 decreased in PLLA-treated tissues (-0.32% ± 0.80%) vs saline controls (2.56% ± 1.03%; P = 0.13). PPARγ expressing cells increased by 5.14% ± 3.72% in saline-treated tissues as they decreased by 4.34% in PLLA-treated tissues (P = 0.13). Changes in nuclear β-catenin showed no significant differences between saline- (6.20% ± 4.11%) and PLLA-treated tissues (-0.89% ± 2.29%) in percent positive cells. Similarly, the change in total β-catenin signal per area was non-significant (saline: 1.07% ± 0.93%; PLLA: -0.39% ± 0.51%). Change in adiponectin signal per area changes were also non-significant (saline: 1.16% ± 0.94%; PLLA: -0.22% ± 0.63%).
Histologic sections had notable spatial heterogeneity. Proximity to PLLA particles had a major effect on cell types and protein expression. PLLA-macrophage-fibroblast encapsulations and tissue in close proximity were high in ADSCs with intense CD105, CD90, Pref1, PCNA, and β-catenin levels. This contrasts sharply with distant tissue, which was nearly absent in those signals and instead dominated by PLIN1 consistent with mature adipocytes (Figure 2).
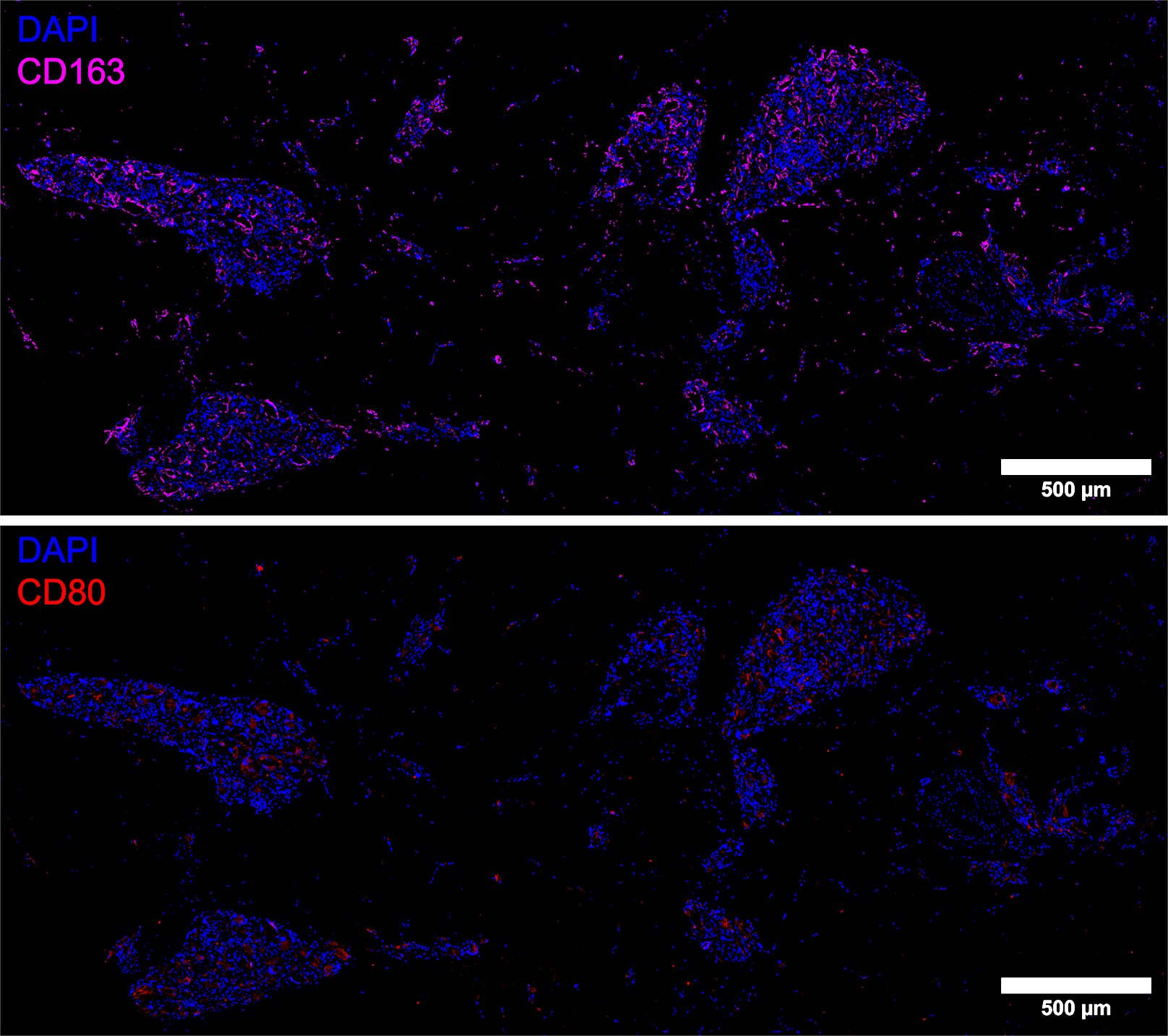
Analysis of PLLA-injected tissues revealed a significantly higher proportion of CD163+ macrophages (8.83% ± 1.48%) compared to CD68+/CD80+ macrophages (1.83% ± 1.02%) (Figure 3). This 4.8:1 ratio of CD163+ (M2) to CD68+/CD80+ (M1) cells indicates a significant deviation from an equal M1/M2 macrophage distribution (P = 0.018).
This study assessed the characterization of PLLA-induced tissue rejuvenation and explored stem cell revitalization within the deep dermal and superficial subcutaneous human tissue layers. Building on the previous SPLASH clinical trial[8], the remodeling microenvironment surrounding the foreign body reactions induced by PLLA were investigated.
This study evaluated proteins that regulate adipocyte differentiation to better understand the mechanisms driving the growth of the subcutaneous adipose layer following injections of PLLA. One such protein, preadipocyte factor 1 (Pref-1, delta-like non-canonical Notch ligand 1) is a regulatory protein that acts as a gatekeeper, maintaining the ADSC and preadipocyte states by blocking differentiation into mature adipocytes[13]. The significant decrease in Pref-1 expression in PLLA-treated tissues suggests the adipogenesis pathway is inhibited in some areas. Consistent with this was a significant decrease in PPARγ, a nuclear receptor necessarily activated before adipocyte differentiation, and a decrease in PLIN1, which is expressed on mature white adipocytes. In the opposing direction, nuclear β-catenin and the overall quantity of β-catenin positive cells decreased. This protein is a downstream part of the Wnt pathway that inhibits adipocyte differentiation when it translocates to the nucleus.
The difference in ADSC quantity in PLLA-treated samples compared to controls was large, approximately doubled as a percentage of cells and 37.5 × greater in absolute counts. We attribute the increase in ADSC (or at least cells exhibiting ADSC-specific markers) to the proliferation of existing ADSCs, as downstream cell types, including fibroblasts and preadipocytes, do not have the ability to dedifferentiate into ADSC. This is supported by the greater increase in adipose precursor cell proliferation (cells expressing PCNA and Pref1) in the PLLA group compared to control (58.4 vs 30.8).
The opposing directions of the proteomic changes observed are disentangled through closer examination of the differing spatial patterns of protein levels (Figure 3). It becomes clear that the tissue response to PLLA depends on the distance from the PLLA particles. Around the PLLA particles is a foreign body reaction, triggered by the greater stiffness of the material compared to the surrounding extracellular matrix[12]. The area of the foreign body response has elevated levels of CD105, CD90, Pref1, PCNA, CD31, and β-catenin in this study as it is populated by M2 macrophages, fibroblasts, ADSC, and preadipocytes. We could also expect this area to have high levels of transforming growth factor-β, interleukin (IL)-10, elastin, collagen type 1, collagen type 3, and matrix metalloproteinases as previous studies have established these are sites of collagen and elastin production[8,14-16]. It is low in PLIN, as there are few adipocytes. In this region, it is hypothesized that adipogenesis is inhibited while the multipotent stem cell state is maintained and proliferation occurs. Over time, PLLA is hydrolyzed into lactic acid and this area shrinks.
Distant from the PLLA-macrophage-fibroblast encapsulations, there is a very different environment. As PLLA particles are hydrolyzed and the foreign body response dissipates, the signals blocking differentiation and promoting proliferation (including β-catenin and Pref-1) have reversed. ADSCs proceed to differentiate into mature adipocytes and the other cell types found in adipose tissue such as fibroblasts and pericytes (vasculature) and we would predict elevated expression of adipogenic factors[14,15]. This results in the bulk increase in adipose volume found in the previous SPLASH hip dell trial[8] as well as many animal studies[17-19]. These areas are high in PLIN1 (Figure 3) and mature adipocytes. Overall, this remote tissue resembles untreated, native tissue.
The observed decrease in adipogenesis markers (Pref-1, PPARγ) contrary to adipose expansion can be reconciled by the temporal and spatial dynamics of the response of adipose tissue to PLLA. Immediately adjacent to PLLA particles, the microenvironment is dominated by macrophage activity, fibroblast recruitment, and proliferation of ADSC phenotype cells, a mileu that favors progenitor expansion rather than adipocyte differentiation. As hydrolysis progresses and macrophage activity decreases, differentiation pathways recover, allowing adipogenesis to occur in regions spatially distant from the encapsulations. Thus, the progressive increase in adipose volume over nine months may result from delayed adipocyte maturation following an initial progenitor expansion phase. Transcriptomic studies support this biphasic model, with later phase findings including upregulated adipocyte-associated genes such as PLIN1, Dickkopf-1, FAB4, LIPE, CIDEC, TRARG1, CMKLR1[14,15]. Cytokines linked to regenerative remodeling including IL-10, IL-13, and transforming growth factor-β have also been found to be upregulated. Follow-up studies separately analyzing PLLA-adjacent and remote tissue at multiple time points should be performed to validate this model.
The 4.8:1 ratio of M2:M1 macrophages supports a model in which the immune response to PLLA is consistent with late-stage wound remodeling rather than infection- or tumor-associated inflammation.
Each subject was treated with the same amount of PLLA; however, their hip dell width and depth varied so there were differences in PLLA spread to maximize aesthetic outcomes. This was a single center study limited to female subjects. A multicenter study of both sexes would have greater population representativeness. Although tissue continues to grow for at least 9 months in response to PLLA, most of the increase in the dermis and adipose tissue had occurred by month 3 post-treatment. The analyzed samples were obtained at 6 months, when the stages of proliferation, differentiation, and bulk increase of adipose tissue had mostly been completed, and only a few regions of PLLA particles and associated reaction remained. Long-term follow-up beyond nine months would still be useful to monitor for delayed adverse events and to confirm the durability of the results.
Each analyzed slide contains only a 4-5 μm thin slice of tissue. PLLA is injected in the superficial part of the adipose layer to maximize its effects on the dermis. The punch biopsies samples of the adipose layer were several millimeters in depth. The histotechnicians that processed the biopsies did not preferentially obtain sections from the superficial part of the biopsy nor did they preferentially choose sections that had PLLA present in them. Thus, the effects of PLLA are likely underestimated.
Measuring changes using the percentage of cells that match a phenotype likely results in an underestimation as the substantial increase in overall cellularity in PLLA-treated samples causes a dilution effect. The lack of statistically significant changes in ADSC, fibroblast, and PCNA+ populations despite large increases further reflects this dilution effect. Alternatively, absolute cell counts can be used. This has its own limitations as total area of tissue on each slide varies. There are other causes for the lack of significance despite large effect sizes. Inter-individual variability was driven primarily by random biopsy sampling, as preferential targeting of PLLA encapsulations was not performed. The small proportion of PLLA encapsulation regions remaining at 6 months post-treatment was variably captured by biopsy. Commensurately, the samples contributing most to variance were those containing PLLA encapsulations. This spatial heterogeneity likely inflated variance and reduced statistical power.
Critically, this study did not have viable cells to perform functional validation to confirm stemness or multipotency of the cells expressing ADSC phenotype on immunofluorescence. Functional validation would include taking fresh tissue, using fluorescence-activated cell sorting to isolate ADSCs, then inducing differentiation into fibroblasts, adipocytes, chondrocytes, and osteoblasts. A functional test of self-renewal capacity is the colony forming unit-fibroblast assay, in which ADSCs plated at low density are cultured and resulting fibroblast colonies are evaluated. No testing of this type was performed due to the aforementioned use of formalin-fixed, paraffin-embedded non-viable tissue.
The use of previously obtained biopsies also resulted in this study being underpowered. A total of 16 subjects had the necessary biopsies taken in the previous trial, and only 14 of these subjects could be reached to consent for the present study. Clinically, PLLA is occasionally injected using a supraperiosteal technique, or less commonly with an intradermal technique. These techniques may result in different tissue responses than the adipose technique reported here. The hip dell was chosen as the treatment area, because it is protected from ultraviolet radiation. Different anatomic areas may respond differently. Threshold segmentation analysis was used for efficiency and objectivity, with manual verification to confirm accuracy. Subsequent investigations should include full inter-observer reproducibility analysis as well as larger sample sizes with subjects of varied gender, age, and ethnicity to improve generalizability.
This study represents the first-time native cells expressing ADSC phenotype have been expanded in humans in vivo, although multipotency and stemness could not be functionally demonstrated with these methods. The subcutaneous adipose tissue, or hypodermis, while often stereotyped as an inert layer merely serving as structural support, play a keystone role in healing and maintaining healthy functioning of the integumentary system. These stem cells regulate fibroblasts and immune cells, combat oxidative stress, and sustain the extracellular matrix[20-24]. Aging and ultraviolet radiation are known to diminish subcutaneous adipose volume as well as ADSC proliferative and differentiation functions[20,25]. Aging fundamentally impairs tissue renewal and repair upon injury, and a general strategy to counter the declining stem cell exhaustion is cellular reprograming[1]. PLLA stands as an exciting therapeutic option to transiently expand local reserves of stem cells to generate any connective tissue cell type necessary to competently respond to future insults, whether it be ultraviolet radiation, chronic disease, surgery, trauma, or aging. While PLLA is sometimes categorized as a dermal filler, in contrast to other implants, it exhibits gradual volume restoration through adipose volumization that recapitulates normal adipose tissue architecture. This also differentiates it from fat grafting, which results in resorption within the first year - a phenomenon believed to be caused by the inability of the recipient area to support the donor cells. Regarding other regenerative therapies, none of those modalities; gene therapy, stem cell grafting, or exosomes, have three decades-long safety record of PLLA. Stem cell transplantation alone is insufficient due to the need to address the age-altered/dysfunctional microenvironment[26]. Reprogramming of somatic cells into stem cells still faces scalability, survival, and immunological barriers. PLLA should be explored as a safe, minimally-invasive, and accessible option to potentially expand ADSC-like progenitors for chronic degeneration and aging-related stem cell exhaustion.
The authors would like to acknowledge Carl S Hornfeldt for editorial assistance.
| 1. | López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186:243-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2880] [Cited by in RCA: 4116] [Article Influence: 1372.0] [Reference Citation Analysis (3)] |
| 2. | Rossi DJ, Seita J, Czechowicz A, Bhattacharya D, Bryder D, Weissman IL. Hematopoietic stem cell quiescence attenuates DNA damage response and permits DNA damage accumulation during aging. Cell Cycle. 2007;6:2371-2376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 122] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 3. | Lin B, Coleman JH, Peterson JN, Zunitch MJ, Jang W, Herrick DB, Schwob JE. Injury Induces Endogenous Reprogramming and Dedifferentiation of Neuronal Progenitors to Multipotency. Cell Stem Cell. 2017;21:761-774.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 4. | Carlson ME, Suetta C, Conboy MJ, Aagaard P, Mackey A, Kjaer M, Conboy I. Molecular aging and rejuvenation of human muscle stem cells. EMBO Mol Med. 2009;1:381-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 178] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 5. | Khalid KA, Nawi AFM, Zulkifli N, Barkat MA, Hadi H. Aging and Wound Healing of the Skin: A Review of Clinical and Pathophysiological Hallmarks. Life (Basel). 2022;12:2142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 69] [Reference Citation Analysis (0)] |
| 6. | Huang SH, Lin YN, Lee SS, Chai CY, Chang HW, Lin TM, Lai CS, Lin SD. New adipose tissue formation by human adipose-derived stem cells with hyaluronic acid gel in immunodeficient mice. Int J Med Sci. 2015;12:154-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 7. | Vassallo V, Di Meo C, Alessio N, La Gatta A, Ferraro GA, Nicoletti GF, Schiraldi C. Highly Concentrated Stabilized Hybrid Complexes of Hyaluronic Acid: Rheological and Biological Assessment of Compatibility with Adipose Tissue and Derived Stromal Cells towards Regenerative Medicine. Int J Mol Sci. 2024;25:2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 8. | Zubair R, Ishii L, Loyal J, Hartman N, Fabi SG. SPLASH: Split-Body Randomized Clinical Trial of Poly- l -Lactic Acid for Adipogenesis and Volumization of the Hip Dell. Dermatol Surg. 2024;50:1155-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 9. | de Winter TJJ, Nusse R. Running Against the Wnt: How Wnt/β-Catenin Suppresses Adipogenesis. Front Cell Dev Biol. 2021;9:627429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 104] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 10. | Moraes DA, Sibov TT, Pavon LF, Alvim PQ, Bonadio RS, Da Silva JR, Pic-Taylor A, Toledo OA, Marti LC, Azevedo RB, Oliveira DM. A reduction in CD90 (THY-1) expression results in increased differentiation of mesenchymal stromal cells. Stem Cell Res Ther. 2016;7:97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 155] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 11. | Bian Y, Deng C, Li W, Lei Z, Li Y, Li X. A Comparative Study on the Biological Characteristics of Human Adipose-Derived Stem Cells from Lipectomy and Liposuction. PLoS One. 2016;11:e0162343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Oh S, Lee JH, Kim HM, Batsukh S, Sung MJ, Lim TH, Lee MH, Son KH, Byun K. Poly-L-Lactic Acid Fillers Improved Dermal Collagen Synthesis by Modulating M2 Macrophage Polarization in Aged Animal Skin. Cells. 2023;12:1320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 63] [Reference Citation Analysis (0)] |
| 13. | Mitterberger MC, Lechner S, Mattesich M, Kaiser A, Probst D, Wenger N, Pierer G, Zwerschke W. DLK1(PREF1) is a negative regulator of adipogenesis in CD105⁺/CD90⁺/CD34⁺/CD31⁻/FABP4⁻ adipose-derived stromal cells from subcutaneous abdominal fat pats of adult women. Stem Cell Res. 2012;9:35-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (3)] |
| 14. | Waibel J, Nguyen TQ, Le JHTD, Qureshi A, Ziegler M, Widgerow A, Meckfessel M. Gene Analysis of Biostimulators: Poly-L-Lactic Acid Triggers Regeneration While Calcium Hydroxylapatite Induces Inflammation Upon Facial Injection. J Drugs Dermatol. 2025;24:34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 15. | Waibel J, Ziegler M, Nguyen TQ, Le JHTD, Qureshi A, Widgerow A, Meckfessel M. Comparative Bulk RNA-Seq Analysis of Poly-l-Lactic Acid Versus Calcium Hydroxylapatite Reveals a Novel, Adipocyte-Mediated Regenerative Mechanism of Action Unique to PLLA. Dermatol Surg. 2024;50:S166-S171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 16. | Huth S, Huth L, Marquardt Y, Jansen M, Lin C, Bartneck M, Baron JM. Molecular Insights Into the Effects of PLLA-SCA on Gene Expression and Collagen Synthesis in Human 3D Skin Models Containing Macrophages. J Drugs Dermatol. 2024;23:285-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 17. | Ogino S, Yamada A, Kambe Y, Nakano T, Lee S, Sakamoto M, Kato Y, Okumura S, Okano J, Yamauchi K, Suzuki Y, Yamaoka T, Morimoto N. Preliminary report of de novo adipogenesis using novel bioabsorbable implants and image evaluation using a porcine model. J Artif Organs. 2022;25:245-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 18. | Ogino S, Yamada A, Nakano T, Lee S, Yamanaka H, Tsuge I, Sowa Y, Sakamoto M, Kyoko F, Kambe Y, Kato Y, Arata J, Yamauchi K, Yamaoka T, Morimoto N. Long term observation of de novo adipogenesis using novel bioabsorbable implants with larger size in a porcine model. Regen Ther. 2023;24:324-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Oh S, Seo SB, Kim G, Batsukh S, Park CH, Son KH, Byun K. Poly-D,L-Lactic Acid Filler Increases Extracellular Matrix by Modulating Macrophages and Adipose-Derived Stem Cells in Aged Animal Skin. Antioxidants (Basel). 2023;12:1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 20. | Gentile P, Garcovich S. Adipose-Derived Mesenchymal Stem Cells (AD-MSCs) against Ultraviolet (UV) Radiation Effects and the Skin Photoaging. Biomedicines. 2021;9:532. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 21. | Chae YB, Lee JS, Park HJ, Park IH, Kim MM, Park YH, Kim DS, Lee JH. Advanced adipose-derived stem cell protein extracts with antioxidant activity modulates matrix metalloproteinases in human dermal fibroblasts. Environ Toxicol Pharmacol. 2012;34:263-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 22. | Guo S, Wang T, Zhang S, Chen P, Cao Z, Lian W, Guo J, Kang Y. Adipose-derived stem cell-conditioned medium protects fibroblasts at different senescent degrees from UVB irradiation damages. Mol Cell Biochem. 2020;463:67-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 23. | Kim J, Park Y, Tae G, Lee KB, Hwang SJ, Kim IS, Noh I, Sun K. Synthesis and characterization of matrix metalloprotease sensitive-low molecular weight hyaluronic acid based hydrogels. J Mater Sci Mater Med. 2008;19:3311-3318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 62] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 24. | Qin F, Huang J, Zhang W, Zhang M, Li Z, Si L, Long X, Wang X. The Paracrine Effect of Adipose-Derived Stem Cells Orchestrates Competition between Different Damaged Dermal Fibroblasts to Repair UVB-Induced Skin Aging. Stem Cells Int. 2020;2020:8878370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Schipper BM, Marra KG, Zhang W, Donnenberg AD, Rubin JP. Regional anatomic and age effects on cell function of human adipose-derived stem cells. Ann Plast Surg. 2008;60:538-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 254] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 26. | Oh J, Lee YD, Wagers AJ. Stem cell aging: mechanisms, regulators and therapeutic opportunities. Nat Med. 2014;20:870-880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 685] [Cited by in RCA: 587] [Article Influence: 48.9] [Reference Citation Analysis (0)] |