Published online Nov 26, 2021. doi: 10.4252/wjsc.v13.i11.1747
Peer-review started: February 25, 2021
First decision: May 5, 2021
Revised: May 18, 2021
Accepted: November 15, 2021
Article in press: November 15, 2021
Published online: November 26, 2021
Processing time: 272 Days and 19.4 Hours
Fibrosis is the hyperactivation of fibroblasts that results in excessive accumulation of extracellular matrix, which is involved in numerous pathological changes and diseases. Adipose-derived stem cells (ASCs) are promising seed cells for regenerative medicine due to their bountiful source, low immunogenicity and lack of ethical issues. Their anti-fibrosis, immunomodulation, angiogenesis and other therapeutic effects have made them suitable for treating fibrosis-related diseases. Here, we review the literature on ASCs treating fibrosis, elaborate and discuss their mechanisms of action, changes in disease environment, ways to enhance therapeutic effects, as well as current preclinical and clinical studies, in order to provide a general picture of ASCs treating fibrotic diseases.
Core Tip: Fibrosis is involved in various diseases. Adipose-derived stem cells (ASCs) are promising candidates for regenerative medicine and anti-fibrosis treatment. We herein discuss the mechanisms of action, changes in disease environment, ways to enhance therapeutic effects, as well as current preclinical and clinical studies of ASCs treating fibrotic diseases in order to provide a general picture.
- Citation: Li ZJ, Wang LQ, Li YZ, Wang CY, Huang JZ, Yu NZ, Long X. Application of adipose-derived stem cells in treating fibrosis. World J Stem Cells 2021; 13(11): 1747-1761
- URL: https://www.wjgnet.com/1948-0210/full/v13/i11/1747.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v13.i11.1747
Fibrosis is a common pathological feature in various diseases. It can involve multiple systems and organs, for example, lungs, liver, kidneys, skin, heart and skeletal muscle. Fibrosis is characterized by the hyperactivation of fibroblasts, causing them to transit into contractile myofibroblasts, as well as increased synthesis of extracellular matrix proteins, resulting in excessive deposition of collagen fibers, often in the form of abnormally arranged bundles. Macroscopically, fibrosis leads to the stiffening of tissues, often accompanied by the loss of normal functions[1-4].
Adipose-derived stem cells (ASCs) are a promising candidate for regenerative medicine due to their multifaceted functions, abundance, and lack of ethical problems. Numerous studies have looked into the effectiveness and mechanisms of action of ASCs in treating fibrosis-related diseases, as well as the ways to boost their functions[4-7].
In this review, we summarize and discuss recent studies in the hope of providing a general picture of the present applications of ASCs in fibrotic diseases.
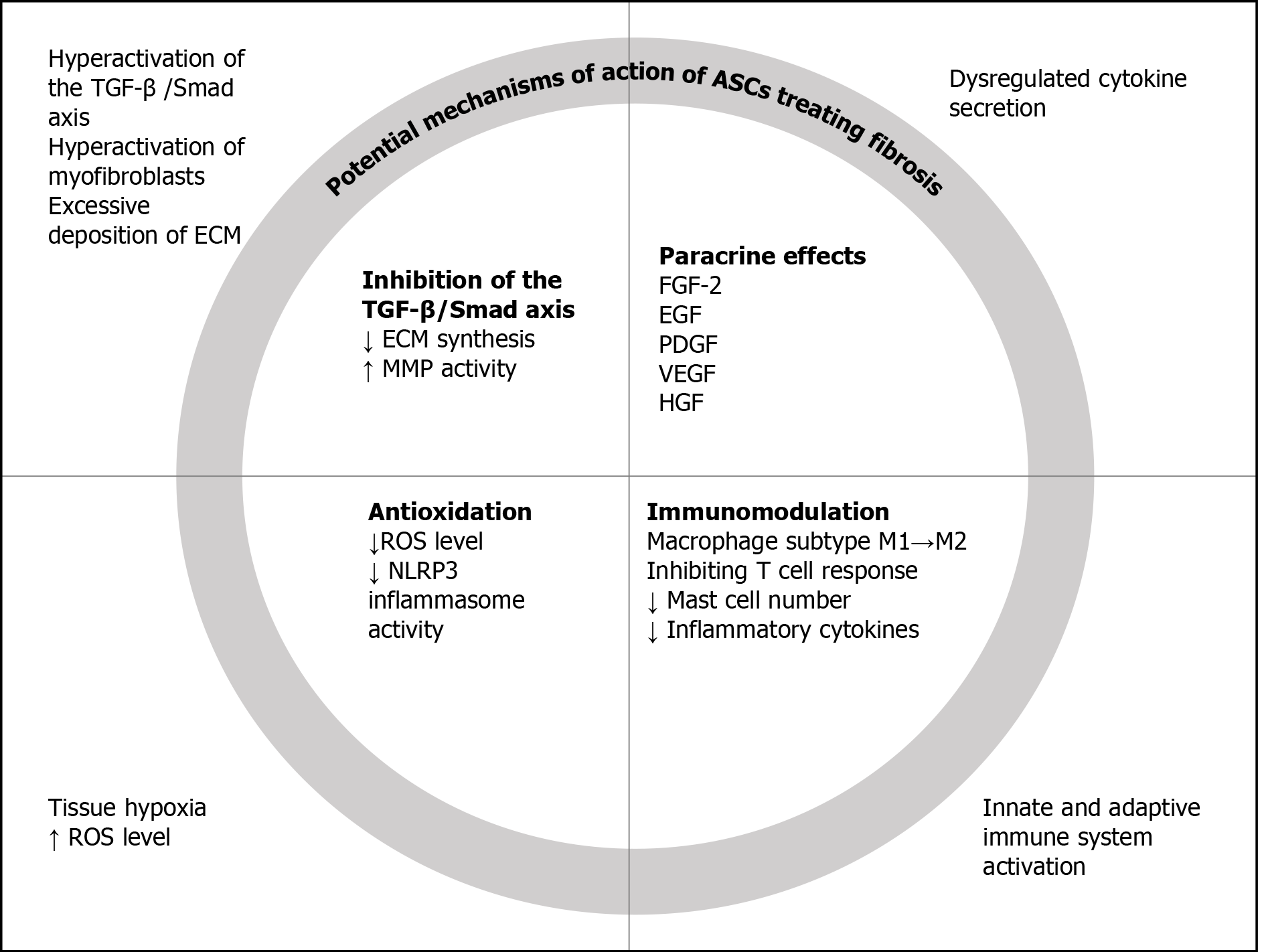
The transforming growth factor-β (TGF-β)/Smad axis is one of the critical players in the wound healing cascade, whose dysregulation leads to pathological fibrosis[8]. In addition to directly stimulating the synthesis of collagen and fibronectin, TGF-β1 also inhibits the extracellular matrix (ECM) decomposing enzyme, matrix metalloproteinases (MMPs), as opposed to the anti-fibrotic tissue inhibitor of metalloproteinases (TIMPs)[9]. By promoting the synthesis while inhibiting the degradation of ECM, the activation of the TGF-β/Smad axis results in excessive accumulation of ECM, which is characteristic of fibrosis. Another important feature of fibrosis is the transition from fibroblast to myofibroblast, which is also induced by TGF-β1[10]. Various studies[10-13] have shown that the decreased expression of TGF-β1 is an important effect of ASCs in the treatment of fibrosis. In fact, it is one of the most widely used indicators of fibrosis treatment.
Dysregulation of cytokine secretion and signaling is present during the fibrotic process[14]. It is well known that adipose-derived mesenchymal stem cells can secrete a large number of soluble factors[15]. The anti-fibrotic function of human adipose tissue is related to the release of fibroblast growth factor-2 (FGF-2), epidermal growth factor (EGF), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), and hepatocyte growth factor (HGF), etc. Those growth factors and cytokines are pivotal ways for ASCs to exert their effects, such as pro-angiogenesis and immu
As a result of tissue damage, fibrosis is associated with hypoxic conditions in the affected area, leading to the production of reactive oxygen species (ROS). Fibrotic tissue is characterized by low capillary density and low oxygen concentration[17]. As a result of chronic hypoxia, ROS have a significant effect on the activation of the TGF-β1/Smad pathway and the accumulation of collagen.
In a study of rat myocardial infarction models, ASCs transplantation significantly decreased ROS level and suppressed the activity of nucleotide-binding oligomerization domain like receptor (NLR) pyrin domain containing 3 (NLRP3) inflammasome, and cardiac fibrosis was markedly ameliorated[18]. At present, many studies have shown that ASCs have powerful antioxidant properties[6,19]. The antioxidative activity of paracrine factors, as well as the improvement in microcirculation due to increased capillary density, leads to a significant reduction in oxidative stress in the surrounding environment[20].
Fibrosis involves the activation of the innate and adaptive immune system[1]. ASCs possess immunomodulatory abilities. Studies have shown that ASCs can reduce inflammation. ASCs play an important role in regulating the function of macrophages, including the conversion from pro-inflammatory M1 subtype to the anti-inflammatory M2 subtype and the recruitment of anti-inflammatory macrophages[21-23]. ASCs also exert powerful immunosuppressive properties by inhibiting T cell response[13]. Mast cells are believed to have a direct effect on stimulating the proliferation of fibroblasts, and the positive results of ASCs scar treatment were associated with a decrease in the number of mast cells in the histological samples of the treatment group[24].
Apart from regulating immune cells, ASCs can also reduce the level of a variety of inflammatory cytokines, including interleukin (IL)-1β, tumor necrosis factor-alpha (TNF-α), and IL-6[13]. The changes in fibrosis and possible mechanisms of action of ASCs are summarized in Figure 1.
ASCs in a distinct disease microenvironment display different characteristics. Taki et al[25] treated ASCs with systemic sclerosis (SSc) lesion tissue fluid, to comprehensively determine the influences of the disease microenvironment on ASCs. Their findings support the idea that ASCs are activated by exposure to the disease microenvironment and can differentiate into fibrotic cells. In addition, subcutaneous ASCs are another potential source of pathogenic myofibroblasts, and multiple factors in the disease microenvironment contribute to the fibrotic transformation (Figure 2).
Adipose tissue in obesity changes into a pro-inflammatory phenotype, fueled by altered ASCs. They exhibit decreased pluripotency, increased secretion of multiple inflammatory factors, including TNF-α, IL-8, IL-6, etc, while the secretion levels of VEGF, FGF, and HGF were reduced. Obese ASCs also synthesize more ROS and recruit more immune cells, continuously promoting the development of inflammation[26]. Over-nutrition also leads to the shortening of telomeres in ASCs, driving them toward cellular senescence, and consequently, fibrosis and functional abnormalities of adipose tissue[27].
The contents of extracellular vesicles (EVs) undergo changes in addition to ASCs themselves. Farahani et al[28] conducted microRNA sequencing on the EVs derived from ASCs of lean and metabolic syndrome (MetS) model pigs, and revealed the changes of 19 microRNAs which were related to mitochondria. Further tests on stenotic kidney injury models uncovered that only MetS EVs failed to improve fibrosis or other indicators. Another study also conducted microRNA sequencing on MetS EVs and compared the differentially expressed microRNAs between lean and MetS individuals, both in pig models and human patients. As a result, there were 57 overlaps in differentially expressed microRNAs between pig models and patients, likely related to MetS-induced changes. In vitro experiments of co-culturing ASCs with renal tubular cells demonstrated that MetS ASCs could induce senescence in tubular cells. MetS ASCs injection was not able to improve renal fibrosis as effectively as lean ASCs[29].
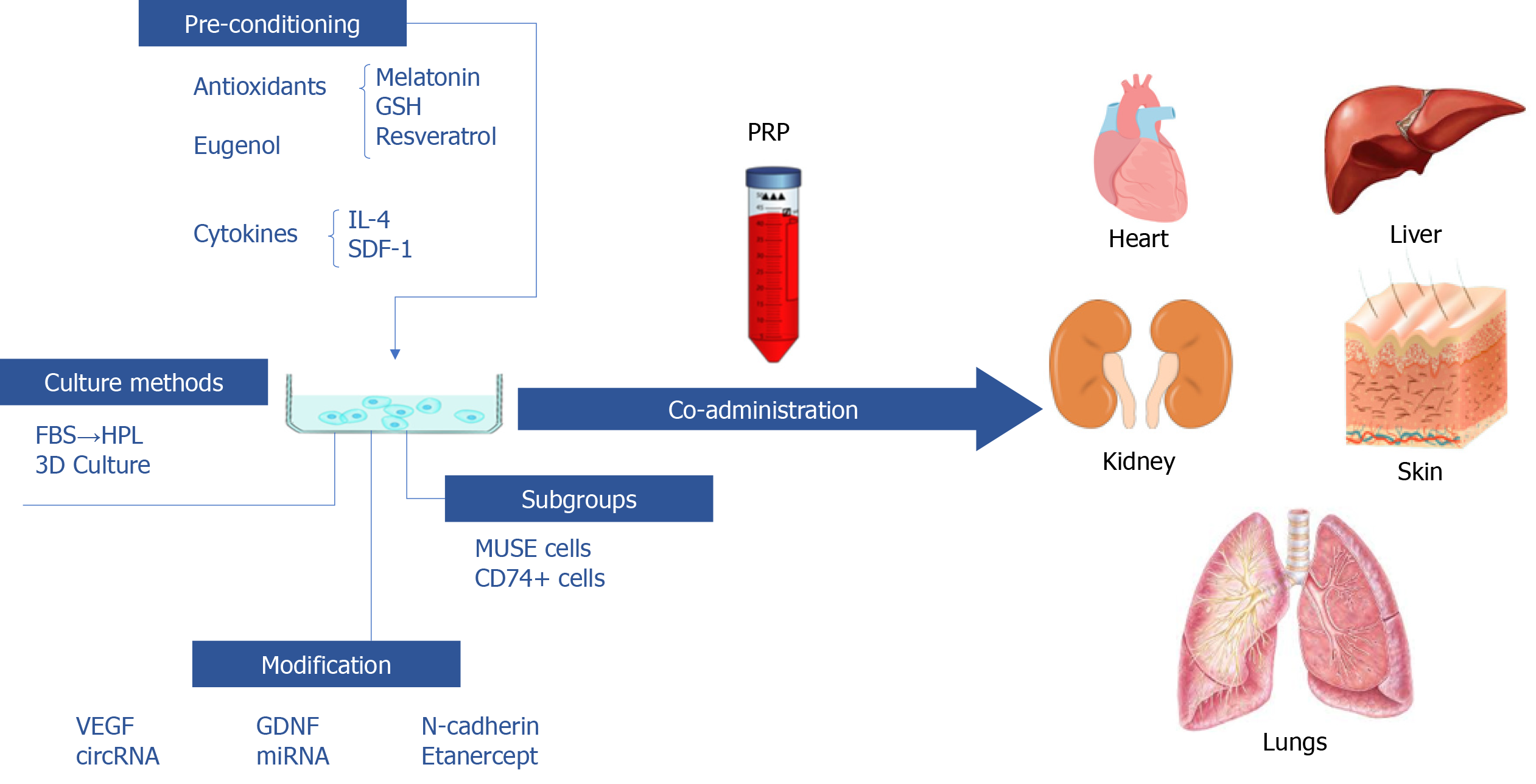
Multiple studies have shown that pre-conditioning with chemicals or protein factors could enhance the therapeutic performance of ASCs. Antioxidants are common sought choices. Liao et al[30] pre-treated ASCs with reduced glutathione (GSH) or melatonin, both of which are endogenous antioxidants. Pre-treatment enhanced the anti-fibrosis and anti-inflammatory effects of transplanted ASCs due to promoted migration and survival. The enhanced survival of ASCs could result from increased expression of Bcl-2 and Cyclin-D1 and reduced expression of Bax, that is, antioxidant pre-treatment promoted cell proliferation and reduced apoptosis. Ex vivo imaging showed that GSH and melatonin pre-treatment promoted ASCs migration into the liver, and allowed them to maintain at a relatively high level. Further investigation revealed that the amelioration of ROS-induced oxidative stress might be the underlying mechanism of action of the antioxidants, and that GSH might be a better choice of pre-conditioning compared to melatonin. Resveratrol is a natural antioxidant extracted from plants which has been proved to benefit health and enhance stem cells therapeutic performance. Chen et al[5] pretreated ASCs with resveratrol (RSVL-ASCs). Compared to untreated ASCs, RSVL-ASCs exhibited a stronger anti-fibrotic effect in diabetes mellitus cardiomyopathy.
Fathy et al[31] discovered that eugenol pre-treatment enhanced ASCs’ self-renewal, proliferation and migration abilities in vitro, as well as their therapeutic effects in rat liver fibrosis models. Compared to untreated ASCs, eugenol treated ASCs (E-ASCs) exhibited better homing ability, further decreased the expression level of inflammatory factors and increased MMPs expression. Histopathological examination revealed similar results.
Zimowska et al[32] studied whether ASCs treated with IL-4 and stromal derived factor-1 (SDF-1), both of which enhance skeletal muscle regeneration, had a better effect in promoting skeletal muscle regeneration. In vitro treatment of IL-4 and SDF-1 significantly enhanced ASCs proliferation and migration.
Pre-conditioning with chemicals and protein factors proved to be effective in improving the functions of ASCs. However, since there could be residual substances in treated cells, it is pivotal to test and determine their safety before application.
Modifying ASCs with various factors could boost their therapeutic effects or expand their mechanisms of action. VEGF is an essential factor in promoting angiogenesis, and is naturally secreted by ASCs. Boosting the concentration of VEGF might result in a boosted therapeutic effect. Yu et al[7] studied the pro-angiogenic, anti-fibrotic and ability to improve fat graft survival rate of VEGF mRNA modified ASCs in vitro and in vivo. Compared to unmanipulated controls, VEGF mRNA modification greatly amplified the therapeutic effects, likely due to the increased secretion of VEGF. Glial cell line-derived neurotrophic factor (GDNF) is a neuroprotective factor that has also been proved to play a critical role in kidney diseases. Li et al[6] transfected GDNF gene into ASCs in order to explore whether the modified cells could perform better in treating renal interstitial fibrosis. It was discovered that the modified ASCs secreted more growth factors, and possessed enhanced abilities in many aspects.
Apart from classical growth factors, N-cadherin is a transmembrane protein that can enhance cell-cell adhesion which has only recently attracted attention in the field of stem cell biology. In a recent study[33], adult mice-ASCs were transfected with adenovirus harboring N-cadherin. N-cadherin overexpression promoted the migration and angiogenic properties, and significantly increased the formation of the N-cadherin/β-catenin complex and the level of active β-catenin in the nucleus, which leads to increased expression levels of MMP-10, MMP-13, and HGF, thus exerting their anti-fibrosis effect.
Another method of exploration is to first compare the differences between disease and normal tissues, identify the genes most likely in play, and adjust ASCs with pertinency. Zhu et al[34] compared the circular RNA (circRNA) expression profiles of normal and fibrotic liver samples from CCl4 induced liver fibrosis mouse models, and discovered that mmu_circ_0000623 was downregulated in fibrotic liver samples. Further exploration revealed that mmu_circ_0000623 interacted with miR-125/ATG4D and modulated autophagy. ASCs were modified with mmu_circ_0000623 and their exosomes were collected. Exosomes from modified ASCs resulted in the best anti-fibrotic effect in vitro and in vivo, by regulating autophagy.
Micro RNAs (miRNAs, or miRs) are small non-coding RNA molecules (about 22 nucleotides) that change gene expression at the post-transcriptional level, leading to changes in protein synthesis. MiR-150 is a representative anti-fibrotic miRNA, which can inhibit the activation of hepatic stellate cells through the inhibition of C-X-C motif chemokine ligand 1 (CXCL1), and is a natural component of ASCs EVs[35]. Paik et al[36] transfected ASCs with miR-150 to explore whether additional miR-150 secretion could boost the anti-fibrotic ability. As expected, transfected ASCs better alleviated liver fibrosis both in vitro and in vivo, due to elevated secretion of anti-fibrotic miRs[36].
TNF-α-driven inflammation plays a key role in the occurrence of liver fibrosis. Han et al[37] designed genetically engineered ASCs that can produce etanercept (an effective TNF-α inhibitor) to play an anti-fibrosis role. They transfected ADSCs with a microcirculatory plasmid containing an insert encoding the etanercept gene to generate synthetic etanercept ADSCs. The results showed that blocking TNF-α-driven inflammation at the appropriate stage of liver fibrosis with the advantage of ADSCs may be an effective strategy to prevent fibrosis.
In summary, genetically manipulated ASCs could be useful in adjusting to different disease conditions and boosting therapeutic effects. However, great care must be taken when considering administrating them into humans. It is crucial to first make sure that the manipulated cells are not tumorigenic or in other ways harmful.
2-dimensional culture remains the primary approach to expand ASCs in vitro. However, various materials used in this process increased the risk of contamination and immune rejection. For example, xenogeneic fetal bovine serum (FBS) might induce immune reactions or transmit bovine diseases. Human platelet lysate (HPL) has been proved to be a promising substitute for FBS. Replacing FBS with HPL in ASCs expansion medium resulted in better proliferative ability without changing cell surface markers. Although TGF-β1-stimulated fibroblasts exhibited improved migration upon HPL-cultured ASCs conditioned medium treatment, there were marked reductions in TGF-β1 and alpha-smooth muscle actin (α-SMA) expression, indicating that the anti-fibrotic ability was likely enhanced by HPL culturing. Further investigation by adding the HGF neutralizing antibody revealed that the anti-fibrotic effect was at least partially related to the increased secretion of HGF[11]. This well-rounded study explored the feasibility of substituting FBS with HPL, and paved the way for safer clinical application of ASCs.
At present, 3-dimensional (3D) cell culture is a frequently-used method to enhance stem cell functions. Transglutaminase cross-linked gelatin (Col-Tgel) is a stiffness-tunable cell culture medium which exhibits excellent performance in cellular adhesion and proliferation and can release entrapped cells. ASCs and Col-Tgel were co-cultured in an in vitro 3D system. Compared with ASCs alone, Col-Tgel embedded ASCs significantly enhanced the long-term retention rate and cardioprotective effect of ASCs in acute myocardial infarction models[2].
Aside from Col-Tgel, various 3D culture methods await exploration. Different target diseases and application methods shall require matching characteristics of the culture medium. ECM mechanical characteristics could affect ASCs phenotype and secretion of cytokines. It has long been known that matrix stiffness is an important influencing factor that affects stem cells lineage specification[38]. Dunham et al[39] discovered that culturing ASCs in a stiff matrix resulted in a more pro-fibrotic phenotype, including increased actin and α-SMA expression and ECM secretion. An adipokine array revealed altered expression levels of multiple adipokines, of which endocan, insulin-like growth factor-binding protein-6 (IGFBP-6), and monocyte chemoattractant protein-1 (MCP-1) were increased in the stiff matrix and might play a part in environment stiffness induced changes of ASCs. In vivo experiments in post-traumatic elbow contracture rat models revealed similar results: ASCs cultured in soft matrix had the best therapeutic effects in increasing elbow range of motion and fibrosis, while ASCs cultured on tissue culture plastic surface made little difference. It was interesting that ASCs transferred from soft matrix into stiff matrix exhibited mechanical memory and maintained the soft matrix-induced phenotypes for a week, which may provide new methods for enhancing the therapeutic effects while avoiding adverse effects of ASCs.
Apart from stiffness, matrix microarchitecture is another factor that influences ASCs characteristics. Seo et al[40] investigated the effects of collagen microarchitecture on ASCs differentiation towards myofibroblasts. ASCs were cultured in collagen fibers that were different in diameter and pore size. Increased contractility, myofibroblast differentiation and pro-angiogenic phenotype were detected in ASCs cultured in thicker fibers and larger pores. The contractile ASCs in turn re-arranged local collagen fibers, therefore increasing local stiffness, and reciprocally, environment stiffness would eventually act on ASCs, forming a circuit of action. Notably, these changes were independent of collagen concentration and bulk stiffness.
There are subgroups within ASCs that possess varied abilities. Multilineage differentiating stress enduring (MUSE) cells are thought to be early-stage MSCs. They can be sorted by severe cellular stress conditions or flow cytometry, using the markers stage-specific embryonic antigen 3 (SSEA-3) (a marker of human ES cells) and endoglin (CD105) (a marker of MSCs). Enhanced pluripotency, paracrine effects, high homing ability and low tumorigenicity have been observed in MUSE cells compared to unsorted ASCs[41].
Borrelli et al[42] identified a subpopulation of ASCs that were positive for CD74. They proved to have enhanced anti-fibrotic abilities both in vitro and in vivo. CD74+ ASCs conditioned medium possessed stronger anti-fibrotic ability, possibly through elevated secretion of HGF, FGF2, TGF-β3 and decreased level of TGF-β1. In radiation-induced fibrosis mouse models, CD74+ ASCs assisted fat graft most significantly, reduced dermal thickness and fibrosis, and resulted in the highest fat survival rate.
As ASCs are inherently heterogenous, subgroups are worth studying in the hope of discovering suitable subgroups for different diseases and the cells that possess maximum healing powers and minimum possibility of inducing adverse effects.
In addition to focusing on ASCs themselves, another way of improving therapeutic effects is the combined usage of ASCs and other substances. Evin et al[3] co-administered ASCs with platelet-rich plasma (PRP) in treating radiation-induced fibrosis. The combination of ASCs and PRP rendered the best results, providing evidence that PRP could augment the therapeutic effects of ASCs.
During muscle repair, fibrosis is a common adverse factor as it stiffens and weakens the healed muscle, depriving it of normal function. In vivo transplantation of ASCs into skeletal muscle injury models resulted in better muscle morphology under the microscope, with more regenerated myofibers and less collagen fiber deposition, through regulating immune responses[32]. It is worth noting that ASCs exhibited better regenerative effects than bone marrow-derived mesenchymal stem cells[43], providing evidence for choosing the most suitable cell type for muscle repair.
Many heart diseases are attributed to cardiac fibrosis and remodeling. Clinically, thrombolytic therapy and primary percutaneous coronary intervention are the most effective treatments for acute myocardial infarction (MI), but due to the limited heart regeneration capacity of adult mammals, irreversible heart failure cannot be prevented. It is expected that stem-cell based regenerative therapy could benefit cardiac fibrosis after infarction. Yan et al[33] aimed to explore whether and how N-cadherin (NCAD) regulates mesenchymal stem cell retention and cardio-protection against ischemic heart failure (IHF). It was found that ASCs transfected with NCAD significantly increased mouse left ventricular ejection fractions and reduced fibrosis. NCAD overexpression can promote ASCs-cardiomyocyte adhesion and migration, and enhance their angiogenesis and cardiomyocyte proliferation abilities. They confirmed for the first time that NCAD overexpression can mediate the expression and production of MMP-10/MMP-13/HGF through β-catenin to enhance the protective effect of ASCs on IHF.
Chen et al[2] delivered the aforementioned Col-Tgel enwrapped ASCs into the heart muscle of acute MI models. The results showed that Col-Tgel provided a suitable microenvironment for the survival, proliferation, and migration of ASCs into the ischemic myocardial tissue, which is essential to exert their regenerative and anti-fibrotic effect. This method can be used clinically to improve the effective rate and reproducibility of cell therapies and heart regeneration research.
EVs deliver genes and proteins to recipient cells and mediate the paracrine activity of their parent cells, which also plays a role in the cardioprotective effect of ASCs. In a pig model with both MetS and renal artery stenosis (RAS), intrarenal injection of EVs derived from ASCs reduced the release of pro-inflammatory cytokines. In MetS+RAS patients, intrarenal injection of EVs reduced myocardial damage, which could be related to the improvement in renal function and systemic inflammation. Local concentrations of inflammatory cytokines such as monocyte chemoattractant protein-1 (MCP-1), TNF-α, and IL-6 were reduced in the stenotic kidney. EVs derived from ASCs improved myocardial fibrosis and remodeling, reduced myocardial hypoxia, improved capillary density and microvascular function, and reduced myocardial cell senescence, thus improving the diastolic function[44].
The efficacy of ASCs has been known to clinicians for some time. Trials have been conducted to explore whether ASCs can safely improve cardiac fibrosis after MI. The results were satisfactory, and were thoroughly elaborated by Li et al[45] and Vazir et al[46].
Although numerous studies support the effectiveness of ASCs in treating cardiac fibrosis, negative results exist where ASCs are not as effective as induced pluripotent stem cell-derived cardiomyocytes[47]. Therefore, it is necessary to make comparisons to understand both the advantages and the disadvantages of ASCs.
Liver fibrosis is an excessive wound healing process that occurs in response to liver injury depending on the underlying cause. Currently, there are no effective treatments for liver fibrosis other than liver transplantation. Due to their advantages over stem cells from other sources, ASCs have received extensive attention as regenerative drugs for the treatment of liver fibrosis[35,48].
Intravenously injected ASCs can migrate into and survive in the fibrotic liver in animal models. Fibrosis was significantly reduced 3 wk after ASCs injection. Immunohistochemistry assay indicated that ASCs had the potential to differentiate into hepatic cells in vivo[48].
However, the inherent limitations of stem cell therapy, such as cell rejection and possibility of tumor formation, hinder the clinical application of ASCs-based therapy. To overcome these problems, extracellular nanovesicles (ENVs) responsible for the treatment of ASCs (A-ENVs) have shown considerable promise as a cell-free treatment of liver diseases. Han et al[49] studied the in vivo and in vitro anti-fibrotic effects of A-ENVs in a thioacetamide-induced liver fibrosis model. A-ENVs significantly down-regulated the expression of fibrogenesis markers, such as MMP-2, collagen-1, and α-SMA. Systemic injection of ENVs can accumulate in fibrotic liver tissue and restore liver function. These results demonstrate the great potential of A-ENVs as a therapeutic method based on ENVs in the treatment of liver fibrosis and possibly other difficult chronic liver diseases.
ASCs could also exert their anti-fibrotic function through an EV component, miR-150-5p and its downstream target, CXCL1, which is related to hepatic stellate cells activation and ensuing fibrosis. Du et al[35] demonstrated that the EVs from ASCs contain miR-150-5p, and EVs treatment downregulated CXCL1 expression in hepatic stellate cells, preventing their activation. In vivo experiments showed reduced fibrosis and inflammation in the EVs treated group that correlated with the inhibition of CXCL1.
With the developing studies on ASCs derivatives, a question naturally arises: are the derivatives as effective as ASCs themselves? Watanabe et al[21] established non-alcoholic steatohepatitis (NASH) models in mice, tested and compared the therapeutic effects of MSCs and their small EVs. Liver fibrosis was significantly reduced after MSCs or sEVs treatment. In terms of relieving fibrosis, the effect of 5.0 μg of sEVs was equal to that of 1 × 106 MSCs, indicating that although ASCs derivatives possess several advantages compared to the direct application of ASCs themselves, a certain amount or concentration would have to be reached for them to have a satisfactory effect.
In order to enhance treatment effectiveness, various modifications were made, including the aforementioned antioxidant and eugenol pre-treatment, modification with various factors including mmu_circ_0000623 and etanercept, all exhibited enhanced abilities in treating liver fibrosis[30,31,34,37].
Attempts have been made in terms of applying ASCs in treating liver fibrosis patients. Huang et al[50] treated liver cirrhosis with GXHPC1, a cell product that contains human ASCs in a phase I clinical trial. Previous trials in animal models proved GXHPC1 to be effective and safe. In their clinical trial, intrahepatic injection of GXHPC1 did not cause any safety problems. The liver function and quality of life of liver cirrhosis patients was improved significantly.
Fibrosis appears in various kidney diseases, and is an indicator of the degree of renal structural damage. Intravenously injected ASCs can migrate into the injured kidney in renal interstitial fibrosis mouse models, promote angiogenesis through the PI3K (phosphatidylinositol-4,5-bisphosphate 3-kinase)/AKT (v-akt murine thymoma viral oncogene homologue) pathway, alleviate oxidative stress and thus significantly reduce renal fibrosis[6]. RAS can lead to kidney ischemia and injury. Kim et al[51] demon
A comparison between ASCs and their derivatives was also conducted in kidney diseases. ASCs and their EVs can both attenuate kidney injury, while focusing on different aspects. Both of them improved kidney fibrosis, hypoxia and cellular apoptosis levels, but the cells were better at increasing capillary density and reducing inflammation, yet their EVs were more efficient in preserving the integrity of kidney cells[52]. The differences represent various components and mechanisms awaiting exploration, which may guide future therapeutic choices.
Apart from kidney fibrosis itself, prolonged peritoneal dialysis (PD) can result in peritoneal fibrosis (PF), a long-term complication jeopardizing peritoneal membrane (PM) function. An intravenous injection of 1 × 106 ASCs proved to not only block the development of PF, but also alleviate the fibrosis and inflammation of already formed PF in rat models of chronic kidney disease (CKD) combined with PF. ASCs treatment significantly downregulated the expression levels of IL-1β, TNF-α, and IL-6, and avoided macrophage and T-cell infiltration into the PM in chlorhexidine gluconate induced PF in CKD rats[13].
Nevertheless, we should recognize the fact that ASCs are not a panacea. Chen et al[19] discussed whether adjunct ASCs could facilitate shockwave therapy in treating atherosclerotic renal artery stenosis (ARAS). Masson trichrome-staining exhibited a similar degree of fibrosis in shockwave therapy alone or shockwave + ASCs; thus, adjunct ASCs did not further improve fibrosis in ARAS. However, adjunct ASCs did have better performance in retaining more capillaries and reducing oxidative stress, which is clearly beneficial to the treatment of kidney diseases.
One of the major differences between normal and fibrotic skin is that the collagen fibers are randomly oriented in normal skin, while arranged in large bundles in fibrotic skin, causing it to thicken and stiffen[53]. Various conditions involve fibrosis of the skin, such as hypertrophic scars, radiation-induced fibrosis, and scleroderma, etc.
Hypertrophic scarring is caused by hyperactivation of fibroblasts and excessive accumulation of extracellular matrix during wound healing. Studies have shown that ASCs are capable of preventing hypertrophic scar occurrence and progression[10,54]. It has also been observed that ASCs themselves are more effective than their conditioned medium, possibly due to insufficient concentration of paracrine factors and lack of cell-cell contact induced regulation which would allow ASCs to react according to their surrounding environment[55].
Since the concentration of paracrine factors in conditioned medium is rather low, lyophilizing is an effective way to condense. Freeze-dried ASCs conditioned medium powder could reduce hypertrophic scar fibroblasts activity in vitro in a dose-dependent manner. In rabbit ear hypertrophic scar models, topical administration of lyophilized ASCs conditioned medium significantly reduced hypertrophic scarring. The effect is most significant when combining the conditioned medium powder with polysaccharide hydrogel, which provided a medium for the sustained release and continuous action of paracrine factors[10].
Other skin fibrotic diseases, for example, radiation-induced fibrosis, is not uncommon among patients receiving radiotherapy. Subcutaneous injection of ASCs alleviated fibrosis, along with other skin complications caused by radiation, and the therapeutic effect was amplified by PRP co-administration[3].
SSc is an autoimmune disease that often involves the skin and lungs. Okamura et al[56] found that ASCs may prove to be a potential therapeutic drug for SSc patients. Administration of ASCs alleviated skin and lung fibrosis of bleomycin-induced scleroderma and sclerodermatous chronic graft-versus-host disease (Scl-cGVHD) model mice. Experimental results showed that ASCs inhibited the infiltration of CD4+ T cells, CD8+ T cells, and macrophages in the dermis of bleomycin model mice and reduced the mRNA levels of collagen and fibrotic cytokines, such as IL-6 and IL-13.
Fat graft is a frequently used method for treating fibrotic skin diseases such as scleroderma. However, the low retention rate had always been a troubling issue. Zhu et al[23] applied ASC-EVs to fat grafts in mice, and measured fat graft survival rate at up to 12 wk post-surgery. Not only did ASC-EVs improve fat retention, they also altered the ratio of M1/M2 macrophages toward an anti-inflammatory state, promoted the browning of white adipose tissue, and reduced fibrosis in fat grafts.
Ogino et al[53] studied the therapeutic effects of ASCs transplantation in secondary lymphedema, where chronic accumulation of tissue fluid often leads to skin fibrosis. Picrosirius red staining revealed that ASCs restored type I collagen orientation and increased type III collagen content, thus relieving dermal fibrosis in lymphedema mice. Promoted lymphangiogenesis was observed, which is pivotal to the alleviation of tissue edema, thereby blocking the development of dermal fibrosis. Since there is a lack of effective treatment methods for lymphatic diseases, ASCs serve as a promising treatment modality due to their multifaceted functions.
In terms of clinical trials, ASCs were used to treat post-acne scars and SSc (Table 1). The hand disability cause by SSc skin lesions is a tricky problem that has significant impact on patients’ quality of life and is difficult to treat. As multiple preclinical studies have demonstrated the anti-fibrosis effect of ASCs, attempts were made to apply ASCs in the treatment of SSc hand disability. Subcutaneous injection of stromal vascular fraction (SVF) greatly improved fibrosis, hand function, quality of life and other aspects[57-59]. ASCs might be the long-expected solution for this debilitating condition.
| Trial number | Ref. | Disease | Study type | Study population | Cell/derivative type | Amount | Method of administration | Outcome measurement | Follow-up period | Results | Complications |
| NCT03060551 | Park et al[58], 2020 | SSc hand disability | Open-label, single center clinical trial | 18 | SVF | 3.61 × 106 each finger on average | Subcutaneous injection | Skin fibrosis, hand edema, hand disability, severity of Raynaud’s phenomenon and hand pain, quality of life, active ulcers, nailfold capillary microscopy | 6 mo | Improved skin fibrosis, edema, quality of life, as well as other aspects | No serious adverse events occurred. Five minor adverse events were reported, including paresthesia in liposuction area, dizziness after lidocaine injection, and transient pallor that soon resolved without sequelae |
| NCT01813279 | Granel et al[57], 2014; Guillaume-Jugnot et al[59], 2015 | SSc hand disability | Open-label, single arm phase I trial | 12 | SVF | 3.76 ± 1.85 × 106 each finger | Subcutaneous injection | Hand disability, fibrosis, vascular manifestations, pain and quality of life | 12 mo | Significant improvements inskin sclerosis, hand function, finger edema and quality of life that lasted for at least a year | No serious adverse events occurred. Four minor adverse events were reported that spontaneously resolved |
| Not applicable | Abou Eitta et al[65], 2019 | Post-acne scars | A single-center, split-face, prospective clinical trial | 10 | SVF | 6 × 106 on average | Subcutaneous injection | The global scoring system, TEWL and skin hydration | 3 mo | A significant improvement in the degree of scar severity, scar area percent, skin hydration, and TEWL | Not mentioned |
| Not applicable | Zhou et al[66], 2016 | Facial atrophic acne scars and skin rejuvenation | A single-center, split-face, prospective clinical trial | 22 | ASC-CM | 3 mL | Topical application on laser treated sites, 3 sessions at one-month intervals | The subjective satisfaction scale, improvement score, biophysical measurements, and skin biopsies | 3 mo | Topical application of ASC-CM can increase the efficacy of FxCR treatment of atrophic acne scars and skin rejuvenation, while simultaneously reduces adverse reactions post laser therapy | No complications reported |
Lung fibrosis is a debilitating condition that can occur in many diseases. Current therapies are insufficient, thus posing the demand for better solutions, such as stem cell therapy[60].
Baer et al[61] transplanted ASCs isolated from luciferase transgenic mice (mASCs) into Atm-deficient mice which mimic the lung injury in human Ataxia-telangiectasia syndrome. Using an in vivo bioluminescence imaging (BLI) system, they found that the intravenously injected ASCs migrated into the injured lungs of mouse models, and were present for up to 9 to 14 d. Since the in vivo disposition of transplanted ASCs is not yet completely clear, BLI might serve as a powerful tool for tracking the whereabouts of cells, providing crucial information regarding the safety of cell therapy.
A comparison between mesenchymal stem cells (MSCs) from different sources was conducted in mouse lung fibrosis models. Among stem cells from adipose tissue (ASCs), Wharton’s jelly (WJ-MSCs), chorionic membrane (CSCs) and chorionic villi (CVCs), ASCs proved to be the most effective and well-rounded in different therapeutic aspects[60]. Another comparison was made in pulmonary hypertension (PAH) models. Mesenchymal stem cells from adipose tissue (ASCs), bone marrow (BMSCs) and umbilical cord blood (UCB-MSCs) were compared. As a result, UCB-MSCs proved to be the most effective in treating PAH, exhibiting the greatest improvement in cardiac function as well as reductions in fibrosis, inflammation, and classic PAH pathways[62]. It is interesting that MSCs from different sources vary so much in their effects. Further research is needed to investigate the mechanisms involved in order to understand and find the best match between diseases and therapeutic cell types.
The functions and mechanisms of ASCs are not singular, but rather intricate. Lim et al[12] found that intravenously injected ASCs migrated into the lungs of murine Scl-GVHD models, but not into the skin. It seems that ASCs exhibited a pro-inflammatory effect in the lungs: CD11b monocyte/macrophages and CD4 T cells were recruited, expression levels of CC chemokine 1 (CCL1) and multiple chemokines were upregulated, and a deterioration in pathological score was observed. Moreover, blocking CCL1 exerted protective effects, relieving inflammation and fibrosis in the lungs. However, despite the possibly detrimental role ASCs play in the lungs, they exhibited anti-inflammatory and anti-fibrotic effects in the skin, which were preserved, or even enhanced, after CCL1-blocking antibody treatment. The fact that ASCs alleviated skin fibrosis while exacerbating lung injuries in Scl-GVHD posed a potential threat to the safety of ASCs therapy. However, CCL1-blocking antibody treatment could avoid detrimental effects while preserving or even enhancing the protective effects which offered a solution. The combination of stem cells and CCL1-blocking antibody provides a new option in exploiting the therapeutic effects of ASCs while avoiding the possible adverse effects.
Not all studies yielded positive healing effects. A study in acute respiratory distress syndrome (ARDS) mouse models revealed that ASCs treatment inhibited the recruitment of neutrophils, reduced short-term lung injury, and alleviated long-term fibrosis. However, the level of inflammatory cytokines did not decrease significantly, the therapeutic effect was minimal and not clinically significant[63].
Despite the possibly unstable effects shown in preclinical studies, a clinical trial of ASCs treating lung fibrosis yielded positive results. Tzouvelekis et al[64] treated idiopathic pulmonary fibrosis with ASCs-SVF in a phase Ib clinical trial, in order to prove the safety of ASCs treatment. There were no serious adverse events, and functional parameters and quality of life indicators did not deteriorate.
ASCs are promising candidates for the treatment of various fibrotic diseases. Multiple methods could be exploited in order to boost the therapeutic effects of ASCs. However, the functions of ASCs are somewhat indeterminate and complicated. The effectiveness and safety issues in ASCs therapy, and the most matching diseases suitable for ASCs therapy remain to be explored.
We would like to thank Xiao K from the Department of Orthopaedic Surgery, West China Hospital for helping with drawing the diagram.
| 1. | Spagnolo P, Distler O, Ryerson CJ, Tzouvelekis A, Lee JS, Bonella F, Bouros D, Hoffmann-Vold AM, Crestani B, Matteson EL. Mechanisms of progressive fibrosis in connective tissue disease (CTD)-associated interstitial lung diseases (ILDs). Ann Rheum Dis. 2021;80:143-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 176] [Article Influence: 35.2] [Reference Citation Analysis (4)] |
| 2. | Chen Y, Li C, Chen J, Li Y, Xie H, Lin C, Fan M, Guo Y, Gao E, Yan W, Tao L. Tailorable Hydrogel Improves Retention and Cardioprotection of Intramyocardial Transplanted Mesenchymal Stem Cells for the Treatment of Acute Myocardial Infarction in Mice. J Am Heart Assoc. 2020;9:e013784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 3. | Evin N, Tosun Z, Aktan TM, Duman S, Harmankaya I, Yavas G. Effects of Adipose-Derived Stem Cells and Platelet-Rich Plasma for Prevention of Alopecia and Other Skin Complications of Radiotherapy. Ann Plast Surg. 2021;86:588-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 4. | Shukla L, Yuan Y, Shayan R, Greening DW, Karnezis T. Fat Therapeutics: The Clinical Capacity of Adipose-Derived Stem Cells and Exosomes for Human Disease and Tissue Regeneration. Front Pharmacol. 2020;11:158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 138] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 5. | Chen TS, Chuang SY, Shen CY, Ho TJ, Chang RL, Yeh YL, Kuo CH, Mahalakshmi B, Kuo WW, Huang CY. Antioxidant Sirt1/Akt axis expression in resveratrol pretreated adipose-derived stem cells increases regenerative capability in a rat model with cardiomyopathy induced by diabetes mellitus. J Cell Physiol. 2021;236:4290-4302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 6. | Li S, Wang Y, Wang Z, Chen L, Zuo B, Liu C, Sun D. Enhanced renoprotective effect of GDNF-modified adipose-derived mesenchymal stem cells on renal interstitial fibrosis. Stem Cell Res Ther. 2021;12:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 7. | Yu F, Witman N, Yan D, Zhang S, Zhou M, Yan Y, Yao Q, Ding F, Yan B, Wang H, Fu W, Lu Y, Fu Y. Human adipose-derived stem cells enriched with VEGF-modified mRNA promote angiogenesis and long-term graft survival in a fat graft transplantation model. Stem Cell Res Ther. 2020;11:490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 8. | Griffin MF, Almadori A, Butler PE. Use of Lipotransfer in Scleroderma. Aesthet Surg J. 2017;37:S33-S37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 9. | Finnson KW, McLean S, Di Guglielmo GM, Philip A. Dynamics of Transforming Growth Factor Beta Signaling in Wound Healing and Scarring. Adv Wound Care (New Rochelle). 2013;2:195-214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 221] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 10. | Zhang C, Wang T, Zhang L, Chen P, Tang S, Chen A, Li M, Peng G, Gao H, Weng H, Zhang H, Li S, Chen J, Chen L, Chen X. Combination of lyophilized adipose-derived stem cell concentrated conditioned medium and polysaccharide hydrogel in the inhibition of hypertrophic scarring. Stem Cell Res Ther. 2021;12:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 11. | Cheng NC, Tu YK, Lee NH, Young TH. Influence of Human Platelet Lysate on Extracellular Matrix Deposition and Cellular Characteristics in Adipose-Derived Stem Cell Sheets. Front Cell Dev Biol. 2020;8:558354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 12. | Lim JY, Ryu DB, Kim TW, Lee SE, Park G, Yoon HK, Min CK. CCL1 blockade alleviates human mesenchymal stem cell (hMSC)-induced pulmonary fibrosis in a murine sclerodermatous graft-versus-host disease (Scl-GVHD) model. Stem Cell Res Ther. 2020;11:254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 13. | Costalonga EC, Fanelli C, Garnica MR, Noronha IL. Adipose-Derived Mesenchymal Stem Cells Modulate Fibrosis and Inflammation in the Peritoneal Fibrosis Model Developed in Uremic Rats. Stem Cells Int. 2020;2020:3768718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Borovikova AA, Ziegler ME, Banyard DA, Wirth GA, Paydar KZ, Evans GRD, Widgerow AD. Adipose-Derived Tissue in the Treatment of Dermal Fibrosis: Antifibrotic Effects of Adipose-Derived Stem Cells. Ann Plast Surg. 2018;80:297-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 15. | Moon KM, Park YH, Lee JS, Chae YB, Kim MM, Kim DS, Kim BW, Nam SW, Lee JH. The effect of secretory factors of adipose-derived stem cells on human keratinocytes. Int J Mol Sci. 2012;13:1239-1257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 16. | Ejaz A, Epperly MW, Hou W, Greenberger JS, Rubin JP. Adipose-Derived Stem Cell Therapy Ameliorates Ionizing Irradiation Fibrosis via Hepatocyte Growth Factor-Mediated Transforming Growth Factor-β Downregulation and Recruitment of Bone Marrow Cells. Stem Cells. 2019;37:791-802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 17. | Lokmic Z, Musyoka J, Hewitson TD, Darby IA. Hypoxia and hypoxia signaling in tissue repair and fibrosis. Int Rev Cell Mol Biol. 2012;296:139-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 153] [Article Influence: 10.9] [Reference Citation Analysis (3)] |
| 18. | Lee TM, Harn HJ, Chiou TW, Chuang MH, Chen CH, Chuang CH, Lin PC, Lin SZ. Host pre-conditioning improves human adipose-derived stem cell transplantation in ageing rats after myocardial infarction: Role of NLRP3 inflammasome. J Cell Mol Med. 2020;24:12272-12284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 19. | Chen XJ, Zhang X, Jiang K, Krier JD, Zhu X, Conley S, Lerman A, Lerman LO. Adjunctive mesenchymal stem/stromal cells augment microvascular function in poststenotic kidneys treated with low-energy shockwave therapy. J Cell Physiol. 2020;235:9806-9818. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 20. | Zhao L, Wei Z, Yang F, Sun Y. [Transforming growth factor-β1 induced cellular proliferation and collagen synthesis was mediated by reactive oxygen species in pulmonary fibroblasts]. Zhonghua Laodong Weisheng Zhiyebing Zazhi. 2015;33:15-19. [PubMed] |
| 21. | Watanabe T, Tsuchiya A, Takeuchi S, Nojiri S, Yoshida T, Ogawa M, Itoh M, Takamura M, Suganami T, Ogawa Y, Terai S. Development of a non-alcoholic steatohepatitis model with rapid accumulation of fibrosis, and its treatment using mesenchymal stem cells and their small extracellular vesicles. Regen Ther. 2020;14:252-261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 22. | Cai C, Kilari S, Zhao C, Simeon ML, Misra A, Li Y, van Wijnen AJ, Mukhopadhyay D, Misra S. Therapeutic Effect of Adipose Derived Mesenchymal Stem Cell Transplantation in Reducing Restenosis in a Murine Angioplasty Model. J Am Soc Nephrol. 2020;31:1781-1795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 23. | Zhu YZ, Zhang J, Hu X, Wang ZH, Wu S, Yi YY. Supplementation with Extracellular Vesicles Derived from Adipose-Derived Stem Cells Increases Fat Graft Survival and Browning in Mice: A Cell-Free Approach to Construct Beige Fat from White Fat Grafting. Plast Reconstr Surg. 2020;145:1183-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 24. | Foley TT, Ehrlich HP. Through gap junction communications, co-cultured mast cells and fibroblasts generate fibroblast activities allied with hypertrophic scarring. Plast Reconstr Surg. 2013;131:1036-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Taki Z, Gostjeva E, Thilly W, Yaseen B, Lopez H, Mirza M, Hassuji Z, Vigneswaran S, Ahmed Abdi B, Hart A, Arumalla N, Thomas G, Denton CP, Suleman Y, Liu H, Venturini C, O'Reilly S, Xu S, Stratton R. Pathogenic Activation of Mesenchymal Stem Cells Is Induced by the Disease Microenvironment in Systemic Sclerosis. Arthritis Rheumatol. 2020;72:1361-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 26. | Ritter A, Kreis NN, Louwen F, Yuan J. Obesity and COVID-19: Molecular Mechanisms Linking Both Pandemics. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 94] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 27. | Gao Z, Daquinag AC, Fussell C, Zhao Z, Dai Y, Rivera A, Snyder BE, Eckel-Mahan KL, Kolonin MG. Age-associated telomere attrition in adipocyte progenitors predisposes to metabolic disease. Nat Metab. 2020;2:1482-1497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 28. | Farahani RA, Zhu XY, Tang H, Jordan KL, Lerman A, Lerman LO, Eirin A. Metabolic Syndrome Alters the Cargo of Mitochondria-Related microRNAs in Swine Mesenchymal Stem Cell-Derived Extracellular Vesicles, Impairing Their Capacity to Repair the Stenotic Kidney. Stem Cells Int. 2020;2020:8845635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (4)] |
| 29. | Li Y, Meng Y, Zhu X, Saadiq IM, Jordan KL, Eirin A, Lerman LO. Metabolic syndrome increases senescence-associated micro-RNAs in extracellular vesicles derived from swine and human mesenchymal stem/stromal cells. Cell Commun Signal. 2020;18:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Liao N, Shi Y, Wang Y, Liao F, Zhao B, Zheng Y, Zeng Y, Liu X, Liu J. Antioxidant preconditioning improves therapeutic outcomes of adipose tissue-derived mesenchymal stem cells through enhancing intrahepatic engraftment efficiency in a mouse liver fibrosis model. Stem Cell Res Ther. 2020;11:237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Fathy M, Okabe M, M Othman E, Saad Eldien HM, Yoshida T. Preconditioning of Adipose-Derived Mesenchymal Stem-Like Cells with Eugenol Potentiates Their Migration and Proliferation In Vitro and Therapeutic Abilities in Rat Hepatic Fibrosis. Molecules. 2020;25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 32. | Zimowska M, Archacka K, Brzoska E, Bem J, Czerwinska AM, Grabowska I, Kasprzycka P, Michalczewska E, Stepaniec I, Soszynska M, Ilach K, Streminska W, Ciemerych MA. IL-4 and SDF-1 Increase Adipose Tissue-Derived Stromal Cell Ability to Improve Rat Skeletal Muscle Regeneration. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 33. | Yan W, Lin C, Guo Y, Chen Y, Du Y, Lau WB, Xia Y, Zhang F, Su R, Gao E, Wang Y, Li C, Liu R, Ma XL, Tao L. N-Cadherin Overexpression Mobilizes the Protective Effects of Mesenchymal Stromal Cells Against Ischemic Heart Injury Through a β-Catenin-Dependent Manner. Circ Res. 2020;126:857-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 34. | Zhu M, Liu X, Li W, Wang L. Exosomes derived from mmu_circ_0000623-modified ADSCs prevent liver fibrosis via activating autophagy. Hum Exp Toxicol. 2020;39:1619-1627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 35. | Du Z, Wu T, Liu L, Luo B, Wei C. Extracellular vesicles-derived miR-150-5p secreted by adipose-derived mesenchymal stem cells inhibits CXCL1 expression to attenuate hepatic fibrosis. J Cell Mol Med. 2021;25:701-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 36. | Paik KY, Kim KH, Park JH, Lee JI, Kim OH, Hong HE, Seo H, Choi HJ, Ahn J, Lee TY, Kim SJ. A novel antifibrotic strategy utilizing conditioned media obtained from miR-150-transfected adipose-derived stem cells: validation of an animal model of liver fibrosis. Exp Mol Med. 2020;52:438-449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 37. | Han JH, Kim OH, Lee SC, Kim KH, Park JH, Lee JI, Lee KH, Hong HE, Seo H, Choi HJ, Ju JH, Kim SJ. A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 38. | Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12260] [Cited by in RCA: 9682] [Article Influence: 484.1] [Reference Citation Analysis (0)] |
| 39. | Dunham C, Havlioglu N, Chamberlain A, Lake S, Meyer G. Adipose stem cells exhibit mechanical memory and reduce fibrotic contracture in a rat elbow injury model. FASEB J. 2020;34:12976-12990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 40. | Seo BR, Chen X, Ling L, Song YH, Shimpi AA, Choi S, Gonzalez J, Sapudom J, Wang K, Andresen Eguiluz RC, Gourdon D, Shenoy VB, Fischbach C. Collagen microarchitecture mechanically controls myofibroblast differentiation. Proc Natl Acad Sci U S A. 2020;117:11387-11398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 148] [Article Influence: 24.7] [Reference Citation Analysis (4)] |
| 41. | Cao J, Yang Z, Xiao R, Pan B. Regenerative potential of pluripotent nontumorgenetic stem cells: Multilineage differentiating stress enduring cells (Muse cells). Regen Ther. 2020;15:92-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 42. | Borrelli MR, Patel RA, Adem S, Diaz Deleon NM, Shen AH, Sokol J, Yen S, Chang EY, Nazerali R, Nguyen D, Momeni A, Wang KC, Longaker MT, Wan DC. The antifibrotic adipose-derived stromal cell: Grafted fat enriched with CD74+ adipose-derived stromal cells reduces chronic radiation-induced skin fibrosis. Stem Cells Transl Med. 2020;9:1401-1413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 43. | Moussa MH, Hamam GG, Abd Elaziz AE, Rahoma MA, Abd El Samad AA, El-Waseef DAA, Hegazy MA. Comparative Study on Bone Marrow-Versus Adipose-Derived Stem Cells on Regeneration and Re-Innervation of Skeletal Muscle Injury in Wistar Rats. Tissue Eng Regen Med. 2020;17:887-900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 44. | Zhang L, Zhu XY, Zhao Y, Eirin A, Liu L, Ferguson CM, Tang H, Lerman A, Lerman LO. Selective intrarenal delivery of mesenchymal stem cell-derived extracellular vesicles attenuates myocardial injury in experimental metabolic renovascular disease. Basic Res Cardiol. 2020;115:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 45. | Li X, Ma T, Sun J, Shen M, Xue X, Chen Y, Zhang Z. Harnessing the secretome of adipose-derived stem cells in the treatment of ischemic heart diseases. Stem Cell Res Ther. 2019;10:196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 46. | Vazir A, Fox K, Westaby J, Evans MJ, Westaby S. Can we remove scar and fibrosis from adult human myocardium? Eur Heart J. 2019;40:960-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 47. | Stępniewski J, Tomczyk M, Andrysiak K, Kraszewska I, Martyniak A, Langrzyk A, Kulik K, Wiśniewska E, Jeż M, Florczyk-Soluch U, Polak K, Podkalicka P, Kachamakova-Trojanowska N, Józkowicz A, Jaźwa-Kusior A, Dulak J. Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes, in Contrast to Adipose Tissue-Derived Stromal Cells, Efficiently Improve Heart Function in Murine Model of Myocardial Infarction. Biomedicines. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 48. | Wu X, Zhang S, Lai J, Lu H, Sun Y, Guan W. Therapeutic Potential of Bama Pig Adipose-Derived Mesenchymal Stem Cells for the Treatment of Carbon Tetrachloride-Induced Liver Fibrosis. Exp Clin Transplant. 2020;18:823-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 49. | Han HS, Lee H, You D, Nguyen VQ, Song DG, Oh BH, Shin S, Choi JS, Kim JD, Pan CH, Jo DG, Cho YW, Choi KY, Park JH. Human adipose stem cell-derived extracellular nanovesicles for treatment of chronic liver fibrosis. J Control Release. 2020;320:328-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 50. | Huang KC, Chuang MH, Lin ZS, Lin YC, Chen CH, Chang CL, Huang PC, Syu WS, Chiou TW, Hong ZH, Tsai YC, Harn HJ, Lin PC, Lin SZ. Transplantation with GXHPC1 for Liver Cirrhosis: Phase 1 Trial. Cell Transplant. 2019;28:100S-111S. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 51. | Kim SR, Zou X, Tang H, Puranik AS, Abumoawad AM, Zhu XY, Hickson LJ, Tchkonia T, Textor SC, Kirkland JL, Lerman LO. Increased cellular senescence in the murine and human stenotic kidney: Effect of mesenchymal stem cells. J Cell Physiol. 2021;236:1332-1344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 52. | Zhao Y, Zhu X, Zhang L, Ferguson CM, Song T, Jiang K, Conley SM, Krier JD, Tang H, Saadiq I, Jordan KL, Lerman A, Lerman LO. Mesenchymal Stem/Stromal Cells and their Extracellular Vesicle Progeny Decrease Injury in Poststenotic Swine Kidney Through Different Mechanisms. Stem Cells Dev. 2020;29:1190-1200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (5)] |
| 53. | Ogino R, Hayashida K, Yamakawa S, Morita E. Adipose-Derived Stem Cells Promote Intussusceptive Lymphangiogenesis by Restricting Dermal Fibrosis in Irradiated Tissue of Mice. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 54. | Foubert P, Zafra D, Liu M, Rajoria R, Gutierrez D, Tenenhaus M, Fraser JK. Autologous adipose-derived regenerative cell therapy modulates development of hypertrophic scarring in a red Duroc porcine model. Stem Cell Res Ther. 2017;8:261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 55. | Zhang Q, Liu LN, Yong Q, Deng JC, Cao WG. Intralesional injection of adipose-derived stem cells reduces hypertrophic scarring in a rabbit ear model. Stem Cell Res Ther. 2015;6:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 106] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 56. | Okamura A, Matsushita T, Komuro A, Kobayashi T, Maeda S, Hamaguchi Y, Takehara K. Adipose-derived stromal/stem cells successfully attenuate the fibrosis of scleroderma mouse models. Int J Rheum Dis. 2020;23:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 57. | Granel B, Daumas A, Jouve E, Harlé JR, Nguyen PS, Chabannon C, Colavolpe N, Reynier JC, Truillet R, Mallet S, Baiada A, Casanova D, Giraudo L, Arnaud L, Veran J, Sabatier F, Magalon G. Safety, tolerability and potential efficacy of injection of autologous adipose-derived stromal vascular fraction in the fingers of patients with systemic sclerosis: an open-label phase I trial. Ann Rheum Dis. 2015;74:2175-2182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 140] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 58. | Park Y, Lee YJ, Koh JH, Lee J, Min HK, Kim MY, Kim KJ, Lee SJ, Rhie JW, Kim WU, Park SH, Moon SH, Kwok SK. Clinical Efficacy and Safety of Injection of Stromal Vascular Fraction Derived from Autologous Adipose Tissues in Systemic Sclerosis Patients with Hand Disability: A Proof-Of-Concept Trial. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 59. | Guillaume-Jugnot P, Daumas A, Magalon J, Jouve E, Nguyen PS, Truillet R, Mallet S, Casanova D, Giraudo L, Veran J, Dignat-George F, Sabatier F, Magalon G, Granel B. Autologous adipose-derived stromal vascular fraction in patients with systemic sclerosis: 12-month follow-up. Rheumatology (Oxford). 2016;55:301-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 60. | Periera-Simon S, Xia X, Catanuto P, Coronado R, Kurtzberg J, Bellio M, Lee YS, Khan A, Smith R, Elliot SJ, Glassberg MK. Anti-fibrotic effects of different sources of MSC in bleomycin-induced lung fibrosis in C57BL6 male mice. Respirology. 2021;26:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 61. | Baer PC, Sann J, Duecker RP, Ullrich E, Geiger H, Bader P, Zielen S, Schubert R. Tracking of Infused Mesenchymal Stem Cells in Injured Pulmonary Tissue in Atm-Deficient Mice. Cells. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 62. | Oh S, Jang AY, Chae S, Choi S, Moon J, Kim M, Spiekerkoetter E, Zamanian RT, Yang PC, Hwang D, Byun K, Chung WJ. Comparative analysis on the anti-inflammatory/immune effect of mesenchymal stem cell therapy for the treatment of pulmonary arterial hypertension. Sci Rep. 2021;11:2012. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 63. | Jung YJ, Park YY, Huh JW, Hong SB. The effect of human adipose-derived stem cells on lipopolysaccharide-induced acute respiratory distress syndrome in mice. Ann Transl Med. 2019;7:674. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 64. | Tzouvelekis A, Paspaliaris V, Koliakos G, Ntolios P, Bouros E, Oikonomou A, Zissimopoulos A, Boussios N, Dardzinski B, Gritzalis D, Antoniadis A, Froudarakis M, Kolios G, Bouros D. A prospective, non-randomized, no placebo-controlled, phase Ib clinical trial to study the safety of the adipose derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis. J Transl Med. 2013;11:171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 200] [Article Influence: 15.4] [Reference Citation Analysis (4)] |
| 65. | Abou Eitta RS, Ismail AA, Abdelmaksoud RA, Ghezlan NA, Mehanna RA. Evaluation of autologous adipose-derived stem cells vs. fractional carbon dioxide laser in the treatment of post acne scars: a split-face study. Int J Dermatol. 2019;58:1212-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 66. | Zhou BR, Zhang T, Bin Jameel AA, Xu Y, Guo SL, Wang Y, Permatasari F, Luo D. The efficacy of conditioned media of adipose-derived stem cells combined with ablative carbon dioxide fractional resurfacing for atrophic acne scars and skin rejuvenation. J Cosmet Laser Ther. 2016;18:138-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Invited article; Externally peer reviewed.
Specialty type: Cell and tissue engineering
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Fraticelli P, Najman SJ S-Editor: Wang LL L-Editor: Webster JR P-Editor: Wang LL