Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.115544
Revised: December 2, 2025
Accepted: January 14, 2026
Published online: March 7, 2026
Processing time: 131 Days and 2.7 Hours
Chronic hepatitis B (CHB) infection is a global public health burden, affecting over 250 million persons globally, and is associated with substantial morbidity and mortality due to cirrhosis, hepatic decompensation, and hepatocellular car
Core Tip: In context of a growing global burden of obesity and diabetes mellitus, metabolic dysfunction-associated steatotic liver disease (MASLD) and metabolic syndrome are increasingly observed in patients with chronic hepatitis B (CHB) virus infection. The relationship between hepatic steatosis and/or metabolic syndrome with CHB is complex and remains incompletely defined, although multiple cohort studies reveal an association with liver inflammation, liver fibrosis, hepatitis B virus (HBV)-related liver events such as cirrhosis, liver failure, and liver cancer, as well as biochemical and virologic response to antiviral therapy. Therefore, a dual diagnosis of MASLD and CHB, also termed met-HBV, may have important implications for management. Probable met-HBV can initially be identified on the basis of liver imaging, but a confirmed diagnosis can be established on vibration-controlled transient elastography-based controlled attenuation parameter score or liver biopsy. Patients diagnosed with met-HBV should undergo active management of MASLD and other manifestations of the metabolic syndrome. Future cohort studies and controlled trials are needed to inform evidence-based approaches to management.
- Citation: Bhatti TK, Lim JK. Diagnosis and management of metabolic dysfunction-associated steatohepatitis in patients with chronic hepatitis B infection. World J Gastroenterol 2026; 32(9): 115544
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/115544.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.115544
Chronic hepatitis B (CHB) infection affects an estimated 254 million persons globally and is associated with substantial morbidity and mortality due to liver failure and hepatocellular carcinoma (HCC). Although CHB represents the key etiologic factor in the development of clinical liver events, many co-existing modifiable and non-modifiable risk factors may meaningfully contribute to these events, such as age, gender, hepatitis B virus (HBV) genotype, HBV viral load, hepatitis B e antigen (HBeAg) status, family history of HCC, alcohol, and hepatic steatosis. Of these, hepatic steatosis has been increasingly recognized as a potentially important co-factor in liver disease progression and liver cancer among patients with chronic HBV, particularly in context of the growing epidemic of obesity and metabolic syndrome. Metabolic dysfunction-associated steatotic liver disease (MASLD) and metabolic dysfunction-associated steatohepatitis (MASH), formerly known as non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis, respectively, are characterized by hepatic steatosis in the absence of other liver disease etiologies or heavy alcohol consumption[1,2], with MASH representing a progressive variant of MASLD associated with histologic necroinflammation, liver cell damage, and fibrosis[1-3]. In contrast with NAFLD[3], a diagnosis of exclusion in patients with hepatic steatosis with no excessive alcohol consumption or competing etiologies, the terms metabolic dysfunction-associated fatty liver disease in 2020[4] and MASLD in 2023[3] represented diagnoses of inclusion, the former requiring at least one of overweight, obesity, or diabetes (and at least two additional metabolic risk factors), and the latter requiring at least one of five cardiometabolic risk factors, as well as exclusion of excessive alcohol intake or competing causes of steatosis.
MASH is increasingly recognized as a leading cause of cirrhosis, indication for liver transplantation, and source of significant financial and health burden in the United States and globally[5,6]. Whereas chronic hepatitis C virus infection has been associated with virally-mediated hepatic steatosis[7], HBV is not associated with de novo hepatic steatosis, but increasing data suggest that the co-existence of MASLD in patients with CHB may have important clinical implications for the natural history and clinical course of liver disease[8-11].
The latest meta-analysis revealed that 38% of individuals globally exhibited MASLD (2016-2019), representing a 50% rise during 1990-2006[5], and current models suggest a future increase in the prevalence of MASLD to 55.4% by 2040[12], particularly in geographies with an anticipated rise in cardiometabolic risk factors [obesity and type 2 diabetes mellitus (T2DM)] such as Asia and the Middle East and Northern Africa region[13-19]. A recent report of Global Burden of Disease data 2010-2021 reveal compelling evidence for the rising worldwide age-standardized rates of prevalence and incidence of MASLD, particularly in countries with varying sociodemographic indices such as China, Sudan, and India[20]. Among patients with chronic HBV, available data suggest that the estimated prevalence of hepatic steatosis is 30%-35%[8-10], including a meta-analysis of 98 studies and 484772 patients in whom hepatic steatosis was 34.9%[10].
Although limited data are available regarding the effect of HBV infection on hepatic steatosis[21,22], MASLD appears to be associated with lower levels of HBV viremia. Emerging data suggest that increasing levels of hepatic steatosis, as measured by controlled attenuation parameter (CAP) score on transient elastography, is associated with progressive fibrosis in patients with chronic HBV, as assessed by liver stiffness measurement (LSM)[23-25], although this relationship may be more significant in those with at minimum moderate to severe hepatic steatosis[26]. The strength of this association varies by study population, but ranges from hazard ratio (HR) of 1.96 (1.25-3.06)[23] to 3.26 (1.28-8.24)[24] to 3.60 (1.21-10.75)[25] and is observed both in patients who are treatment-naïve or on antiviral therapy with oral nucleos(t)ide (NUC) analogues[26]. Studies of patients with CHB who have undergone liver biopsy additionally reveal an association between histologically evident hepatic steatosis with significant liver inflammation and risk for advanced fibrosis[8,27,28].
The rising global prevalence of MASLD[5,29] among patients with CHB have heightened interest on its potential effect on HBV-related outcomes[30-34]. Available data have revealed mixed findings with regard to the relationship between hepatic steatosis, HBV infection, and liver outcomes[35-40].
Whereas the presence of CHB appears to be associated with poorer liver outcomes among patients with metabolic dysfunction-associated fatty liver disease[35], a large cohort study of 6786 patients with CHB revealed that fatty liver was not independently associated with cirrhosis or HCC risk among untreated patients, and was associated with a lower risk of cirrhosis [HR = 0.19, 95% confidence interval (CI): 0.12-0.33] and HCC (HR = 0.21, 95%CI: 0.09-0.51) among patients on antiviral therapy. In contrast, a large cohort study of 11502 patients with CHB with median 5.3 year follow-up revealed that whereas hepatic steatosis was not associated with cirrhosis complications, metabolic dysfunction was associated with an increased risk of cirrhosis [adjusted HR (aHR) = 1.82, 95%CI: 1.40-2.37] and cirrhosis complications (aHR = 1.30, 95%CI: 1.03-1.63), and among metabolic diseases, diabetes mellitus was associated with highest risk for cirrhosis complications (aHR = 2.87, 95%CI: 1.34-6.11)[40], signaling that metabolic syndrome rather than hepatic steatosis alone may represent the clinically important modifiable risk factor for liver events. Table 1 summarizes study evidence which characterize the risk of disease progression in patients with CHB and concurrent MASLD.
| Study population | SLD diagnosis | Viral suppression | Disease progression | Outcomes |
| 1202 CHB patients with or without steatosis[57] | VCTE | Lower median serum HBV DNA levels | Severe steatosis associated with increase severe fibrosis in treatment-naïve and treated patients | Fibrosis progression (increase) |
| 330 treatment-naive patients with CHB[58] | VCTE | HBsAg seroclearance | Persistent severe hepatic steatosis independently associated with fibrosis progression (OR = 2.379) | Fibrosis progression (increase) |
| 606 patients with CHB[62] | VCTE | HBsAg seroclearance | Severe steatosis associated with severe fibrosis in treatment-naive (OR = 3.60) and treated (OR: 1.95-2.79) patients | Fibrosis severity (increase) |
| Meta-analysis: 34 studies with 68268 CHB patients[63] | VCTE/biopsy-proven steatosis | HBsAg seroclearance (OR = 2.22) | Steatosis associated with cirrhosis (OR = 1.52) and HCC (OR = 1.59) | Cirrhosis (increase), HCC (increase) |
| 1089 patients with CHB[59] | Biopsy data | HBsAg seroclearance | Steatohepatitis + CHB more advanced fibrosis and shorter time to liver-related outcomes/death | Fibrosis (increase), outcomes worse |
| 197346 patients with CHB (South Korea NHIS)[64] | MASLD criteria | HBsAg seroclearance | Risk of HCC was 1.4-fold higher in CHB + MASLD | HCC (increase) |
| 11502 patients with CHB[65] | MASLD criteria | Lower HBV DNA levels | MASLD patients had higher risk of cirrhosis | Cirrhosis (increase) |
| 10546 treatment-naive CHB patients[66] | MASLD criteria | Fewer HBeAg positivity, lower HBV DNA levels | Steatosis + metabolic dysfunction increase risk of HCC (aHR 1.40 per dysfunction) | HCC (increase) |
The primary cause of death for persons with CHB is HCC[41], and established risk factors among patients receiving oral NUC therapy include cirrhosis, thrombocytopenia at baseline, male sex, and advanced age[42] and individual risk may be predicted using risk scores such as PAGE-B[43]. Among patients with CHB treated with oral antivirals, MASLD has been identified as an independent risk factor for HCC[44], with increasing evidence supporting a mechanistic link with MASLD/MASH and HCC carcinogenesis which may stem from insulin-resistance associated hepatic steatosis, lipotoxicity, endoplastic reticulum stress, inflammasome activation, and stellate cell activation[45-47]. MASLD also appears to serve as a co-factor in HCC development in more than half of cases in the presence of alternative primary liver disorders[48], which may have unique interaction in context of CHB which is strongly associated with HCC independent of cirrhosis due to HBV integration into the host hepatocyte genome, activation of HCC pathways through changes in genomic instability, p53 gene mutations, tumor suppressor genes, and telomerase reverse transcriptase gene activation[49-52] as well as HBV-mediated immune modulation of negative regulators of HCC such as interleukin-10 and transforming growth factor-β[53-56].
Distinct from the effects of severe hepatic steatosis on liver fibrosis and disease-based outcomes[57], MASLD has been associated with an increased likelihood of spontaneous hepatitis B surface antigen (HBsAg) clearance or functional cure[24,58]. These reports are supported by a meta-analysis comprising 34 studies and 68268 CHB patients which confirmed that hepatic steatosis was independently associated with HBsAg seroclearance (odds ratio = 2.22)[58], thus revealing a paradoxical relationship in context of the opposing effects on liver fibrosis, liver events, and mortality[32,58-63]. Furthermore, hepatic steatosis may adversely affect response to antiviral therapy with oral NUC analogues with poorer biochemical [alanine aminotransferase (ALT) normalization] and virologic response (suppression of HBV DNA, pregenomic RNA, quantitative HBsAg, and HBeAg loss/seroconversion)[64-66], as well as poorer improvement in liver stiffness score despite viral decline[67]. Real-world observational studies additionally reveal that diabetes mellitus and metabolic syndrome are also associated with poorer virologic response to oral NUC analogues[68].
Limited data addressing the impact of treatment of MASLD and other manifestations of the metabolic syndrome on HBV-related outcomes are presently available. Multiple studies have confirmed an association between diabetes mellitus and metabolic syndrome with an increased risk of HBV-related liver outcomes such as cirrhosis, HCC, and liver-related death[69-72]. However, prospective data to confirm benefit of treatment of diabetes, metabolic syndrome, or MASLD on HBV outcomes are lacking. One study has reported improvement in steatohepatitis with weight loss intervention in patients with CHB[73].
Future studies examining the impact of weight loss pharmacotherapy [e.g., glucagon-like peptide-1 receptor agonist (GLP1RA)], liver-directed MASH pharmacotherapy (e.g., resmetirom), and other anti-glycemic, lipid lowering, and antihypertensive treatment on HBV-related outcomes are needed. Despite limited evidence, due to the overlapping cardiometabolic risk profile of patients with CHB and hepatic steatosis, and the potential implications on liver outcomes, it is prudent to screen for metabolic syndrome and MASLD in this subpopulation, and consider individualized management of metabolic conditions if identified. Furthermore, this information may influence approach to HBV treatment. As per updated 2024 HBV guidelines of the World Health Organization (WHO)[74], antiviral therapy is recommended for HBsAg positive patients with MASLD or metabolic syndrome, independent of HBV DNA, serum ALT, or liver fibrosis stage.
Screening for concurrent MASLD and metabolic syndrome can be incorporated into standard workflows in the management of patients with CHB, including examination (body mass index, waist circumference, blood pressure), assessment of alcohol intake, laboratory testing (fasting glucose or glycosylated hemoglobin, lipid panel), and liver imaging with ultrasound, computed tomography, or magnetic resonance imaging (MRI), which are routinely performed in context of liver cancer surveillance. The presence of cardiometabolic risk factors should signal increased risk for concurrent MASLD and prompt further investigation.
Radiologic features consistent with hepatic steatosis (e.g., increased echogenicity) may identify patients with “probable met-HBV”. Vibration-controlled transient elastography (VCTE) is commonly used for fibrosis staging in CHB but additionally provides liver fat content assessment with CAP scoring. In a patient with CHB, the presence of at least one cardiometabolic risk factor plus clinically significant hepatic steatosis, based either on histologic grade 1 steatosis or greater (grade 1: 5%-33%, grade 2: 34%-66%, grade 3: > 66%), MRI-proton density fat fraction score ≥ 6.5% (grade 1: 6.5%-17.4%, grade 2: 17.5%-22.0%, grade 3: 22.1% or greater) or CAP score ≥ 238 dB/m (S1: 238-252 dB/m, S2: 253-282 dB/m, S3: 283 dB/m or greater), effectively rules in a diagnosis of “confirmed met-HBV”. Concurrent evidence of hepatic inflammation based on elevated liver transaminases, particularly in context of inactive or NUC-controlled CHB (e.g., HBV DNA < 2000 IU/mL) and absence of other precipitants (e.g., hepatotoxic medications, alcohol), effectively signals concurrent MASLD as the likely etiology.
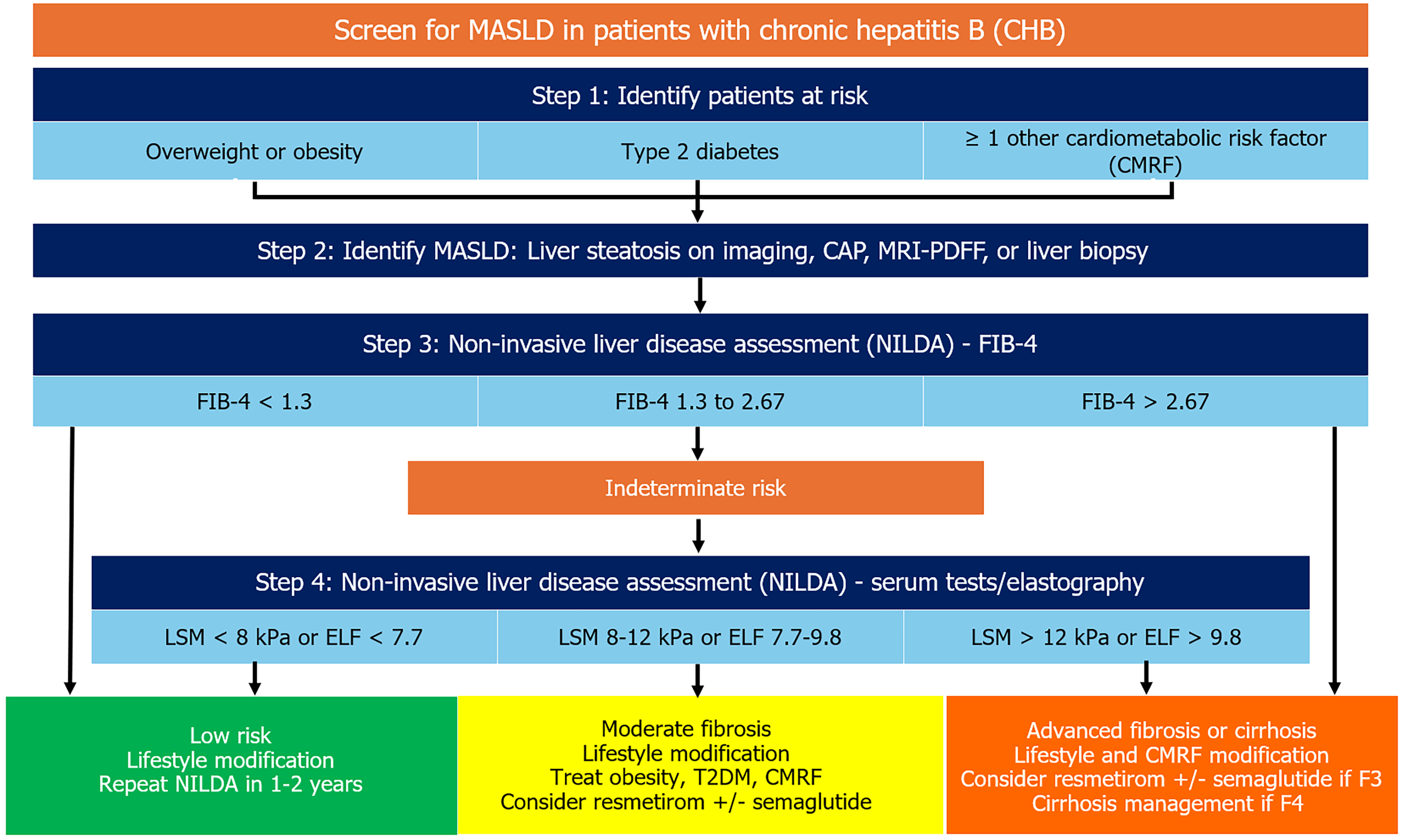
As fibrosis staging is used as a criterion for HBV treatment eligibility in practice guidelines, attribution of key drivers of fibrosis development (concurrent MASLD vs CHB) directly impacts clinical decisions. All patients with CHB and suspected MASLD should undergo initial routine risk stratification with fibrosis-4 (FIB-4), with a score < 1.3 signaling low risk for advanced liver fibrosis. Among individuals with FIB-4 > 1.3, further risk stratification should be pursued with non-invasive tests (NITs) using serum fibrosis assays [e.g., enhanced liver fibrosis (ELF), Fibrotest, Fibrosure, Hepascore] and/or imaging-based elastography (e.g., VCTE, shear wave elastography, magnetic resonance elastography). Dual or sequential NITs combining elastography with serum fibrosis assays (e.g. Fibrotest, ELF) is preferred over a single NIT approach to corroborate fibrosis estimates, recognizing that established cut-offs for F2 fibrosis and cirrhosis were derived from patients with MASLD or CHB alone, and therefore modified cut-offs may be needed for patients with met-HBV[75]. Until additional data are available to inform evidence-based thresholds specific to met-HBV, we suggest the use of the established thresholds of LSM ≥ 8.0 kPa, ELF score ≥ 7.7, or equivalent to rule in F2 fibrosis or greater to identify potential candidates for MASLD pharmacotherapy. Furthermore, liver biopsy should be considered if there is significant discordance in these estimates and/or there remains uncertainty regarding primary drivers of liver injury and fibrosis.
Patients who are determined to have probable or confirmed met-HBV should be counseled regarding the potential implications of this dual liver diagnosis, and advised to pursue a holistic approach to improving liver health, including: (1) Lifestyle modification with diet and exercise as per practice guidelines for MASLD alone; (2) Individualized approach to medical, pharmacologic, endoscopic, or surgical weight loss based on presence of severe obesity and/or overall cardiometabolic risk profile; (3) Restriction of alcohol consumption for patients with elevated liver enzymes or significant fibrosis (F2 or greater), or elimination for patients with cirrhosis; (4) Restriction of potentially hepatotoxic medications or supplements; (5) Targeted treatment of relevant components of the metabolic syndrome (e.g., diabetes mellitus, hypertension, dyslipidemia) to reduce the risk for major adverse cardiovascular events; (6) Individualized approach to liver-directed treatment for MASH and F2-F3 fibrosis with resmetirom and/or semaglutide, particularly in patients with inactive or NUC-controlled CHB and evidence of significant liver inflammation and/or fibrosis; and (7) Individualized consideration for oral NUC therapy of HBV independent of other eligibility criteria, in accordance with 2024 WHO HBV guidelines. The WHO advises that the presence of comorbidities such as T2DM or MASLD should prompt consideration for antiviral therapy, due to the association between T2DM and MASLD on liver fibrosis progression and risk for HCC[76]. In lean non-diabetic patients diagnosed with met-HBV, consideration should be given for screening of other genetic and metabolic liver diseases associated with hepatic steatosis (e.g., Wilson’s disease, alpha-1 anti-trypsin deficiency).
Given limited data specific to this population, the management of patients undergoing liver-directed therapy for MASH in context of met-HBV with the two Food and Drug Administration-approved agents, oral thyroid hormone beta-receptor agonist resmetirom and/or the oral glucagon-like receptor-1 agonist semaglutide, should follow standard American Association for the Study of Liver Diseases guidance for patients with MASH alone, including regular monitoring of liver enzymes every 3-6 months and periodic assessments of CAP score and LSM on VCTE every 6-12 months[77,78]. Although patients with CHB were excluded in the phase 3 trials supporting regulatory approval, resmetirom and semaglutide may considered in the treatment of patients with met-HBV and F2-F3 fibrosis in the absence of independent drug-drug interactions or contraindications. Longitudinal assessment is needed in real-world studies to confirm safety, tolerability, and treatment response, and inform long-term management approach in this population. Among patients who are determined to have evidence of cirrhosis based on clinical markers, NITs, and/or liver biopsy, liver-directed MASLD treatment is not currently recommended, but these patients should be treated with oral NUC therapy and undergo routine cirrhosis management including HCC surveillance twice per year and assessment for clinically significant portal hypertension based on Baveno criteria and/or endoscopic evaluation for gastroesophageal varices. Finally, clinicians should incorporate a multidisciplinary patient-centered approach to management, including coordination of care between primary care, endocrinology, cardiology, and gastrointestinal/hepatology clinicians, and using shared decision making which prioritizes patient preference, and consideration of the impact of met-HBV and its treatment on health-related quality of life, patient-reported outcomes, and adherence to treatment. Figure 1 summarizes a suggested clinical algorithm for initial evaluation and management.
Metabolic syndrome and MASLD are common among patients with CHB, and may have important implications on virologic and other clinical outcomes. The complex interplay between metabolic syndrome, MASLD with or without steatohepatitis, and CHB remain incompletely understood and require further investigation to characterize the bidirectional effects and response to metabolic intervention. Available data are mixed but suggest that MASLD may contribute to an increased risk for liver events in patients with CHB, including cirrhosis, hepatic decompensation, and HCC. Adequately powered prospective cohort studies and controlled trials are needed to support evidence-based recommendations for evaluation and management. These studies should be supported by refined case definitions for met-HBV which account for the presence and severity of MASLD/MASH and other manifestations of the metabolic syndrome, and intersection with alcohol and other sources of hepatic steatosis. Validation studies in patients with met-HBV to derive modified cut-offs for risk stratification using NITs, including serum indices (e.g., FIB-4), serum assays (e.g., ELF), and liver elastography, ideally using liver histology and/or clinical outcome endpoints, would further support decisions regarding treatment initiation and longitudinal assessment of response. In the absence of available evidence, clinicians should consider routine assessment of risk factors for MASLD and metabolic syndrome, limited screening with labs and imaging, VCTE if available, and an individualized approach to metabolic interventions with lifestyle modification and/or pharmacotherapy. Finally, as the presence of HBV is exclusionary for enrollment in therapeutic trials for MASH, real-world observational cohort studies are needed to characterize the effects of GLP1RA, metabolic therapies targeting diabetes, hypertension, and dyslipidemia, and both current and future liver-directed MASH therapies on HBV-specific and overall liver outcomes. Such data will be necessary to inform evidence-based recommendations in future management guidelines.
| 1. | Lindenmeyer CC, McCullough AJ. The Natural History of Nonalcoholic Fatty Liver Disease-An Evolving View. Clin Liver Dis. 2018;22:11-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 229] [Cited by in RCA: 215] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 2. | Pais R, Maurel T. Natural History of NAFLD. J Clin Med. 2021;10:1161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 3. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966-1986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2198] [Cited by in RCA: 2037] [Article Influence: 679.0] [Reference Citation Analysis (8)] |
| 4. | Eslam M, George J. Reply to: correspondence regarding "A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement": Bringing evidence to the NAFLD-MAFLD debate. J Hepatol. 2020;73:1575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 138] [Article Influence: 23.0] [Reference Citation Analysis (3)] |
| 5. | Younossi ZM, Golabi P, Paik JM, Henry A, Van Dongen C, Henry L. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology. 2023;77:1335-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2563] [Cited by in RCA: 2289] [Article Influence: 763.0] [Reference Citation Analysis (7)] |
| 6. | Le MH, Yeo YH, Li X, Li J, Zou B, Wu Y, Ye Q, Huang DQ, Zhao C, Zhang J, Liu C, Chang N, Xing F, Yan S, Wan ZH, Tang NSY, Mayumi M, Liu X, Liu C, Rui F, Yang H, Yang Y, Jin R, Le RHX, Xu Y, Le DM, Barnett S, Stave CD, Cheung R, Zhu Q, Nguyen MH. 2019 Global NAFLD Prevalence: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2022;20:2809-2817.e28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 477] [Article Influence: 119.3] [Reference Citation Analysis (2)] |
| 7. | Adinolfi LE, Rinaldi L, Guerrera B, Restivo L, Marrone A, Giordano M, Zampino R. NAFLD and NASH in HCV Infection: Prevalence and Significance in Hepatic and Extrahepatic Manifestations. Int J Mol Sci. 2016;17:803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 90] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 8. | Machado MV, Oliveira AG, Cortez-Pinto H. Hepatic steatosis in hepatitis B virus infected patients: meta-analysis of risk factors and comparison with hepatitis C infected patients. J Gastroenterol Hepatol. 2011;26:1361-1367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 135] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 9. | Zheng Q, Zou B, Wu Y, Yeo Y, Wu H, Stave CD, Cheung RC, Nguyen MH. Systematic review with meta-analysis: prevalence of hepatic steatosis, fibrosis and associated factors in chronic hepatitis B. Aliment Pharmacol Ther. 2021;54:1100-1109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 75] [Article Influence: 15.0] [Reference Citation Analysis (2)] |
| 10. | Jiang D, Chen C, Liu X, Huang C, Yan D, Zhang X, Zhou Y, Lin Y, Zhou Y, Guan Z, Ding C, Lan L, Zhu C, Wu J, Li L, Yang S. Concurrence and impact of hepatic steatosis on chronic hepatitis B patients: a systematic review and meta-analysis. Ann Transl Med. 2021;9:1718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 39] [Reference Citation Analysis (0)] |
| 11. | Fernandez CJ, Alkhalifah M, Afsar H, Pappachan JM. Metabolic Dysfunction-Associated Fatty Liver Disease and Chronic Viral Hepatitis: The Interlink. Pathogens. 2024;13:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 12. | Le MH, Yeo YH, Zou B, Barnet S, Henry L, Cheung R, Nguyen MH. Forecasted 2040 global prevalence of nonalcoholic fatty liver disease using hierarchical bayesian approach. Clin Mol Hepatol. 2022;28:841-850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 276] [Cited by in RCA: 249] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 13. | NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403:1027-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1583] [Cited by in RCA: 1344] [Article Influence: 672.0] [Reference Citation Analysis (15)] |
| 14. | Younossi ZM, Golabi P, Paik J, Owrangi S, Yilmaz Y, El-Kassas M, Alswat K, Alqahtani SA. Prevalence of metabolic dysfunction-associated steatotic liver disease in the Middle East and North Africa. Liver Int. 2024;44:1061-1070. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 84] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 15. | Le MH, Le DM, Baez TC, Wu Y, Ito T, Lee EY, Lee K, Stave CD, Henry L, Barnett SD, Cheung R, Nguyen MH. Global incidence of non-alcoholic fatty liver disease: A systematic review and meta-analysis of 63 studies and 1,201,807 persons. J Hepatol. 2023;79:287-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 219] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 16. | Zhou F, Zhou J, Wang W, Zhang XJ, Ji YX, Zhang P, She ZG, Zhu L, Cai J, Li H. Unexpected Rapid Increase in the Burden of NAFLD in China From 2008 to 2018: A Systematic Review and Meta-Analysis. Hepatology. 2019;70:1119-1133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 465] [Article Influence: 66.4] [Reference Citation Analysis (3)] |
| 17. | Zhou J, Zhou F, Wang W, Zhang XJ, Ji YX, Zhang P, She ZG, Zhu L, Cai J, Li H. Epidemiological Features of NAFLD From 1999 to 2018 in China. Hepatology. 2020;71:1851-1864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 521] [Article Influence: 86.8] [Reference Citation Analysis (0)] |
| 18. | Estes C, Chan HLY, Chien RN, Chuang WL, Fung J, Goh GB, Hu TH, Huang JF, Jang BK, Jun DW, Kao JH, Lee JW, Lin HC, Razavi-Shearer K, Seto WK, Wong GL, Wong VW, Razavi H. Modelling NAFLD disease burden in four Asian regions-2019-2030. Aliment Pharmacol Ther. 2020;51:801-811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 132] [Article Influence: 22.0] [Reference Citation Analysis (5)] |
| 19. | Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A, Angulo P. The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology. 2005;129:113-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2393] [Cited by in RCA: 2131] [Article Influence: 101.5] [Reference Citation Analysis (3)] |
| 20. | Feng G, Targher G, Byrne CD, Yilmaz Y, Wai-Sun Wong V, Adithya Lesmana CR, Adams LA, Boursier J, Papatheodoridis G, El-Kassas M, Méndez-Sánchez N, Sookoian S, Castera L, Chan WK, Ye F, Treeprasertsuk S, Cortez-Pinto H, Yu HH, Kim W, Romero-Gómez M, Nakajima A, Win KM, Kim SU, Holleboom AG, Sebastiani G, Ocama P, Ryan JD, Lupșor-Platon M, Ghazinyan H, Al-Mahtab M, Hamid S, Perera N, Alswat KA, Pan Q, Long MT, Isakov V, Mi M, Arrese M, Sanyal AJ, Sarin SK, Leite NC, Valenti L, Newsome PN, Hagström H, Petta S, Yki-Järvinen H, Schattenberg JM, Castellanos Fernández MI, Leclercq IA, Aghayeva G, Elzouki AN, Tumi A, Sharara AI, Labidi A, Sanai FM, Matar K, Al-Mattooq M, Akroush MW, Benazzouz M, Debzi N, Alkhatry M, Barakat S, Al-Busafi SA, Rwegasha J, Yang W, Adwoa A, Opio CK, Sotoudeheian M, Wong YJ, George J, Zheng MH. Global burden of metabolic dysfunction-associated steatotic liver disease, 2010 to 2021. JHEP Rep. 2025;7:101271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 72] [Article Influence: 72.0] [Reference Citation Analysis (0)] |
| 21. | Joo EJ, Chang Y, Yeom JS, Ryu S. Hepatitis B virus infection and decreased risk of nonalcoholic fatty liver disease: A cohort study. Hepatology. 2017;65:828-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 129] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 22. | Huang J, Jing M, Wang C, Wang M, You S, Lin S, Zhu Y. The impact of hepatitis B virus infection status on the prevalence of nonalcoholic fatty liver disease: A population-based study. J Med Virol. 2020;92:1191-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Wong SW, Chan WK, Mohamed R. Fatty liver is associated with advanced fibrosis but does not predict adverse outcomes in patients with chronic hepatitis B. J Viral Hepat. 2020;27:1297-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 24. | Mak LY, Hui RW, Fung J, Liu F, Wong DK, Cheung KS, Yuen MF, Seto WK. Diverse effects of hepatic steatosis on fibrosis progression and functional cure in virologically quiescent chronic hepatitis B. J Hepatol. 2020;73:800-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 25. | Seto WK, Hui RWH, Mak LY, Fung J, Cheung KS, Liu KSH, Wong DK, Lai CL, Yuen MF. Association Between Hepatic Steatosis, Measured by Controlled Attenuation Parameter, and Fibrosis Burden in Chronic Hepatitis B. Clin Gastroenterol Hepatol. 2018;16:575-583.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 102] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 26. | Shen F, Mi YQ, Xu L, Liu YG, Wang XY, Pan Q, Zhang RN, Hu XQ, Xu LM, Fan JG. Moderate to severe hepatic steatosis leads to overestimation of liver stiffness measurement in chronic hepatitis B patients without significant fibrosis. Aliment Pharmacol Ther. 2019;50:93-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 27. | Petta S, Cammà C, Di Marco V, Macaluso FS, Maida M, Pizzolanti G, Belmonte B, Cabibi D, Di Stefano R, Ferraro D, Guarnotta C, Venezia G, Craxì A. Hepatic steatosis and insulin resistance are associated with severe fibrosis in patients with chronic hepatitis caused by HBV or HCV infection. Liver Int. 2011;31:507-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 28. | Dai YN, Xu CF, Pan HY, Chen MJ, Yu CH. Fatty liver is associated with significant liver inflammation and increases the burden of advanced fibrosis in chronic HBV infection. BMC Infect Dis. 2023;23:637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Li J, Zou B, Yeo YH, Feng Y, Xie X, Lee DH, Fujii H, Wu Y, Kam LY, Ji F, Li X, Chien N, Wei M, Ogawa E, Zhao C, Wu X, Stave CD, Henry L, Barnett S, Takahashi H, Furusyo N, Eguchi Y, Hsu YC, Lee TY, Ren W, Qin C, Jun DW, Toyoda H, Wong VW, Cheung R, Zhu Q, Nguyen MH. Prevalence, incidence, and outcome of non-alcoholic fatty liver disease in Asia, 1999-2019: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2019;4:389-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 880] [Cited by in RCA: 781] [Article Influence: 111.6] [Reference Citation Analysis (4)] |
| 30. | Hsu YC, Yeh ML, Wong GL, Chen CH, Peng CY, Buti M, Enomoto M, Xie Q, Trinh H, Preda C, Liu L, Cheung KS, Yeo YH, Hoang J, Huang CF, Riveiro-Barciela M, Kozuka R, Istratescu D, Tsai PC, Accarino EV, Lee DH, Wu JL, Huang JF, Dai CY, Cheung R, Chuang WL, Yuen MF, Wong VW, Yu ML, Nguyen MH. Incidences and Determinants of Functional Cure During Entecavir or Tenofovir Disoproxil Fumarate for Chronic Hepatitis B. J Infect Dis. 2021;224:1890-1899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 31. | Huang CF, Jang TY, Jun DW, Ahn SB, An J, Enomoto M, Takahashi H, Ogawa E, Yoon E, Jeong SW, Shim JJ, Jeong JY, Kim SE, Oh H, Kim HS, Cho YK, Kozuka R, Inoue K, Cheung KS, Mak LY, Huang JF, Dai CY, Yuen MF, Nguyen MH, Yu ML. On-treatment gamma-glutamyl transferase predicts the development of hepatocellular carcinoma in chronic hepatitis B patients. Liver Int. 2022;42:59-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 32. | Li J, Yang HI, Yeh ML, Le MH, Le AK, Yeo YH, Dai CY, Barnett S, Zhang JQ, Huang JF, Trinh HN, Wong C, Wong C, Hoang JK, Cheung R, Yu ML, Nguyen MH. Association Between Fatty Liver and Cirrhosis, Hepatocellular Carcinoma, and Hepatitis B Surface Antigen Seroclearance in Chronic Hepatitis B. J Infect Dis. 2021;224:294-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 33. | Huang CF, Liang PC, Tsai PC, Wei YJ, Huang CI, Wang CW, Jang TY, Yeh ML, Hsu PY, Hsieh MY, Lin YH, Dai CY, Chuang WL, Huang JF, Yu ML. The interplay of metabolic dysfunction-associated fatty liver disease and viral hepatitis on liver disease severity: A large community-based study in a viral endemic area. J Gastroenterol Hepatol. 2024;39:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 34. | Lin CL, Kao JH. Perspectives and control of hepatitis B virus infection in Taiwan. J Formos Med Assoc. 2015;114:901-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 35. | Cheng YM, Hsieh TH, Wang CC, Kao JH. Impact of HBV infection on clinical outcomes in patients with metabolic dysfunction-associated fatty liver disease. JHEP Rep. 2023;5:100836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 36. | van der Spek DPC, Katwaroe WK, van Kleef LA, Brakenhoff S, de Man RA, de Knegt RJ, van der Meer AJ, Sonneveld MJ. Time-trends in disease characteristics and comorbidities in patients with chronic hepatitis B in the period 1980-2020. Eur J Intern Med. 2023;107:86-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 37. | Simon TG, Roelstraete B, Khalili H, Hagström H, Ludvigsson JF. Mortality in biopsy-confirmed nonalcoholic fatty liver disease: results from a nationwide cohort. Gut. 2021;70:1375-1382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 558] [Cited by in RCA: 522] [Article Influence: 104.4] [Reference Citation Analysis (0)] |
| 38. | Targher G, Byrne CD, Tilg H. MASLD: a systemic metabolic disorder with cardiovascular and malignant complications. Gut. 2024;73:691-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 396] [Article Influence: 198.0] [Reference Citation Analysis (0)] |
| 39. | Huang SC, Su TH, Tseng TC, Chen CL, Hsu SJ, Liao SH, Hong CM, Liu CH, Lan TY, Yang HC, Liu CJ, Chen PJ, Kao JH. Distinct effects of hepatic steatosis and metabolic dysfunction on the risk of hepatocellular carcinoma in chronic hepatitis B. Hepatol Int. 2023;17:1139-1149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 40. | Huang SC, Su TH, Tseng TC, Liao SH, Hsu SJ, Hong CM, Lan TY, Liu CH, Yang HC, Liu CJ, Kao JH. Pre-Existing and New-Onset Metabolic Dysfunctions Increase Cirrhosis and Its Complication Risks in Chronic Hepatitis B. Am J Gastroenterol. 2025;120:401-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 41. | Chan SL, Wong VW, Qin S, Chan HL. Infection and Cancer: The Case of Hepatitis B. J Clin Oncol. 2016;34:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 134] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 42. | Papatheodoridis GV, Dalekos GN, Yurdaydin C, Buti M, Goulis J, Arends P, Sypsa V, Manolakopoulos S, Mangia G, Gatselis N, Keskın O, Savvidou S, Hansen BE, Papaioannou C, Galanis K, Idilman R, Colombo M, Esteban R, Janssen HL, Lampertico P. Incidence and predictors of hepatocellular carcinoma in Caucasian chronic hepatitis B patients receiving entecavir or tenofovir. J Hepatol. 2015;62:363-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 130] [Article Influence: 11.8] [Reference Citation Analysis (3)] |
| 43. | Voulgaris T, Papatheodoridi M, Lampertico P, Papatheodoridis GV. Clinical utility of hepatocellular carcinoma risk scores in chronic hepatitis B. Liver Int. 2020;40:484-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (2)] |
| 44. | Amano K, Sano T, Ide T, Nakano D, Tsutsumi T, Arinaga-Hino T, Kawaguchi M, Hirai S, Miyajima I, Torimura T, Kawaguchi T. The Effect of MAFLD on Hepatocarcinogenesis in HBeAg-negative Patients with Undetectable HBV-DNA under NA Therapy: A Multicenter Study. Intern Med. 2025;64:1133-1141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 45. | Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018;24:908-922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3652] [Cited by in RCA: 3322] [Article Influence: 415.3] [Reference Citation Analysis (6)] |
| 46. | Hammerich L, Tacke F. Hepatic inflammatory responses in liver fibrosis. Nat Rev Gastroenterol Hepatol. 2023;20:633-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 542] [Article Influence: 180.7] [Reference Citation Analysis (2)] |
| 47. | Ma Y, Wang J, Xiao W, Fan X. A review of MASLD-related hepatocellular carcinoma: progress in pathogenesis, early detection, and therapeutic interventions. Front Med (Lausanne). 2024;11:1410668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 37] [Reference Citation Analysis (0)] |
| 48. | Crane H, Eslick GD, Gofton C, Shaikh A, Cholankeril G, Cheah M, Zhong JH, Svegliati-Baroni G, Vitale A, Kim BK, Ahn SH, Kim MN, Strasser SI, George J. Global prevalence of metabolic dysfunction-associated fatty liver disease-related hepatocellular carcinoma: A systematic review and meta-analysis. Clin Mol Hepatol. 2024;30:436-448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 40] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 49. | Sung WK, Zheng H, Li S, Chen R, Liu X, Li Y, Lee NP, Lee WH, Ariyaratne PN, Tennakoon C, Mulawadi FH, Wong KF, Liu AM, Poon RT, Fan ST, Chan KL, Gong Z, Hu Y, Lin Z, Wang G, Zhang Q, Barber TD, Chou WC, Aggarwal A, Hao K, Zhou W, Zhang C, Hardwick J, Buser C, Xu J, Kan Z, Dai H, Mao M, Reinhard C, Wang J, Luk JM. Genome-wide survey of recurrent HBV integration in hepatocellular carcinoma. Nat Genet. 2012;44:765-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 803] [Cited by in RCA: 746] [Article Influence: 53.3] [Reference Citation Analysis (4)] |
| 50. | Wong DK, Cheng SCY, Mak LL, To EW, Lo RC, Cheung TT, Seto WK, Fung J, Man K, Lai CL, Yuen MF. Among Patients with Undetectable Hepatitis B Surface Antigen and Hepatocellular Carcinoma, a High Proportion Has Integration of HBV DNA into Hepatocyte DNA and No Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:449-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 51. | Mak LY, Wong DK, Pollicino T, Raimondo G, Hollinger FB, Yuen MF. Occult hepatitis B infection and hepatocellular carcinoma: Epidemiology, virology, hepatocarcinogenesis and clinical significance. J Hepatol. 2020;73:952-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 163] [Article Influence: 27.2] [Reference Citation Analysis (1)] |
| 52. | Sze KM, Ho DW, Chiu YT, Tsui YM, Chan LK, Lee JM, Chok KS, Chan AC, Tang CN, Tang VW, Lo IL, Yau DT, Cheung TT, Ng IO. Hepatitis B Virus-Telomerase Reverse Transcriptase Promoter Integration Harnesses Host ELF4, Resulting in Telomerase Reverse Transcriptase Gene Transcription in Hepatocellular Carcinoma. Hepatology. 2021;73:23-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 53. | Li X, Su Y, Hua X, Xie C, Liu J, Huang Y, Zhou L, Zhang M, Li X, Gao Z. Levels of hepatic Th17 cells and regulatory T cells upregulated by hepatic stellate cells in advanced HBV-related liver fibrosis. J Transl Med. 2017;15:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 54. | Nkongolo S, Mahamed D, Kuipery A, Sanchez Vasquez JD, Kim SC, Mehrotra A, Patel A, Hu C, McGilvray I, Feld JJ, Fung S, Chen D, Wallin JJ, Gaggar A, Janssen H, Gehring AJ. Longitudinal liver sampling in patients with chronic hepatitis B starting antiviral therapy reveals hepatotoxic CD8+ T cells. J Clin Invest. 2023;133:e158903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 65] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 55. | Luxenburger H, Neumann-Haefelin C. Liver-resident CD8+ T cells in viral hepatitis: not always good guys. J Clin Invest. 2023;133:e165033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 56. | Cho HJ, Cheong JY. Role of Immune Cells in Patients with Hepatitis B Virus-Related Hepatocellular Carcinoma. Int J Mol Sci. 2021;22:8011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 57. | Hui RWH, Seto WK, Cheung KS, Mak LY, Liu KSH, Fung J, Wong DK, Lai CL, Yuen MF. Inverse relationship between hepatic steatosis and hepatitis B viremia: Results of a large case-control study. J Viral Hepat. 2018;25:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 58. | Mao X, Cheung KS, Peng C, Mak LY, Cheng HM, Fung J, Peleg N, Leung HH, Kumar R, Lee JH, Shlomai A, Yuen MF, Seto WK. Steatosis, HBV-related HCC, cirrhosis, and HBsAg seroclearance: A systematic review and meta-analysis. Hepatology. 2023;77:1735-1745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 79] [Article Influence: 26.3] [Reference Citation Analysis (2)] |
| 59. | Choi HSJ, Brouwer WP, Zanjir WMR, de Man RA, Feld JJ, Hansen BE, Janssen HLA, Patel K. Nonalcoholic Steatohepatitis Is Associated With Liver-Related Outcomes and All-Cause Mortality in Chronic Hepatitis B. Hepatology. 2020;71:539-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 181] [Article Influence: 30.2] [Reference Citation Analysis (1)] |
| 60. | Lin M, Gao B, Peng M, Chen X, Xiao H, Shi M, Zhang X, Zeng F, Chi X. Metabolic dysfunction-associated steatotic liver disease increases hepatocellular carcinoma risk in chronic hepatitis B patients: a retrospective cohort study. Front Physiol. 2024;15:1347459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 61. | van Kleef LA, Choi HSJ, Brouwer WP, Hansen BE, Patel K, de Man RA, Janssen HLA, de Knegt RJ, Sonneveld MJ. Metabolic dysfunction-associated fatty liver disease increases risk of adverse outcomes in patients with chronic hepatitis B. JHEP Rep. 2021;3:100350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 62. | Huang SC, Su TH, Tseng TC, Chen CL, Hsu SJ, Liu CH, Liao SH, Hong CM, Lan TY, Yang HC, Liu CJ, Chen PJ, Kao JH. Metabolic Dysfunction-Associated Steatotic Liver Disease Facilitates Hepatitis B Surface Antigen Seroclearance and Seroconversion. Clin Gastroenterol Hepatol. 2024;22:581-590.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 63. | Yuen MF, Wong DK, Fung J, Ip P, But D, Hung I, Lau K, Yuen JC, Lai CL. HBsAg Seroclearance in chronic hepatitis B in Asian patients: replicative level and risk of hepatocellular carcinoma. Gastroenterology. 2008;135:1192-1199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 336] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 64. | Zhang S, Zhang X, Jin H, Dou Y, Li L, Yuan X, Dong C, Hou M, Nan YM, Shang J. Adverse Effect of Nonalcoholic Fatty Liver Disease on the Therapeutic Response in Patients with Chronic Hepatitis B. J Clin Transl Hepatol. 2023;11:67-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 65. | Kim DS, Jeon MY, Lee HW, Kim BK, Park JY, Kim DY, Ahn SH, Han KH, Kim SU. Influence of hepatic steatosis on the outcomes of patients with chronic hepatitis B treated with entecavir and tenofovir. Clin Mol Hepatol. 2019;25:283-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 66. | Liu SY, Wang D, Liu J, Yang LP, Chen GY. Influence of nonalcoholic fatty liver disease on response to antiviral treatment in patients with chronic hepatitis B: A meta-analysis. World J Hepatol. 2024;16:465-476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 67. | Patmore LA, van Eekhout K, Koc ÖM, de Knegt RJ, Janssen HLA, Brouwer WP, Kramer M, Honkoop P, de Bruijne J, Boland GJ, Postma DF, Blokzijl H, de Man RA, Takkenberg RB, Sonneveld MJ. The Association Between Metabolic Dysfunction-Associated Steatotic Liver Disease and Change in Liver Stiffness in Patients With Chronic Hepatitis B. Liver Int. 2025;45:e70042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 68. | Huang R, Jun DW, Toyoda H, Hsu YC, Trinh H, Nozaki A, Ishikawa T, Watanabe T, Uojima H, Huang DQ, Honda T, Tanaka Y, Vutien P, Marciano S, Abe H, Enomoto M, Atsukawa M, Takahashi H, Tsuji K, Itobayashi E, Takaguchi K, Tsai PC, Dai CY, Huang JF, Huang CF, Yeh ML, Yoon E, Kim SE, Ahn SB, Kim GA, Jung JH, Jeong SW, Oh H, Tseng CH, Ishigami M, Chau A, Hsiao T, Maeda M, Yasuda S, Chuma M, Ito T, Kawashima K, Liu JK, Gadano A, Kozuka R, Itokawa N, Inoue K, Senoh T, Li J, Chuang WL, Cheung R, Wu C, Yu ML, Nguyen MH. Impact of metabolic dysfunction on treatment responses to nucleos(t)ide analogues in chronic hepatitis B: a retrospective multi-center REAL-B cohort study. EClinicalMedicine. 2025;87:103407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 69. | Huang R, Jun DW, Toyoda H, Hsu YC, Trinh H, Nozaki A, Ishikawa T, Watanabe T, Uojima H, Huang DQ, Honda T, Tanaka Y, Vutien P, Marciano S, Abe H, Enomoto M, Atsukawa M, Takahashi H, Tsuji K, Takaguchi K, Tsai PC, Dai CY, Huang JF, Huang CF, Yeh ML, Yoon E, Kim SE, Ahn SB, Kim GA, Jung JH, Jeong SW, Oh H, Tseng CH, Ishigami M, Chau A, Maeda M, Yasuda S, Chuma M, Ito T, Kawashima K, Liu JK, Gadano A, Kozuka R, Itokawa N, Inoue K, Senoh T, Li J, Chuang WL, Cheung R, Wu C, Yu ML, Nguyen MH. Impacts of metabolic syndrome diseases on long-term outcomes of chronic hepatitis B patients treated with nucleos(t)ide analogues. Clin Mol Hepatol. 2025;31:1003-1017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 70. | Thin KN, Tran A, Li J, Lee EY, Yang H, Rui F, Liu C, Stave CD, Cheung RC, Nguyen MH. Increased Risk of Liver-Related Outcomes in Chronic Hepatitis B Patients with Metabolic Syndrome: A Systematic Review and Meta-Analysis. Dig Dis. 2022;40:745-753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 71. | Wang QX, Xue J, Shi MJ, Xie YB, Xiao HM, Li S, Lin M, Chi XL. Association Between Metabolic Dysfunction-Associated Fatty Liver Disease and the Risk of Cirrhosis in Patients with Chronic Hepatitis B-A Retrospective Cohort Study. Diabetes Metab Syndr Obes. 2022;15:2311-2322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 72. | Bockmann JH, Kohsar M, Murray JM, Hamed V, Dandri M, Lüth S, Lohse AW, Schulze-Zur-Wiesch J. High Rates of Liver Cirrhosis and Hepatocellular Carcinoma in Chronic Hepatitis B Patients with Metabolic and Cardiovascular Comorbidities. Microorganisms. 2021;9:968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 73. | Chang XJ, Shi YW, Wang J, Liu HB, Chen Y, Zhu XN, Chen YP, Yu ZJ, Shang QH, Tan L, Li Q, Jiang L, Xiao GM, Chen L, Lu W, Hu XY, Long QH, An LJ, Zou ZY, Wong VW, Yang YP, Fan JG. Influence of weight management on the prognosis of steatohepatitis in chronic hepatitis B patients during antiviral treatment. Hepatobiliary Pancreat Dis Int. 2021;20:416-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 74. | Easterbrook PJ, Luhmann N, Bajis S, Min MS, Newman M, Lesi O, Doherty MC. WHO 2024 hepatitis B guidelines: an opportunity to transform care. Lancet Gastroenterol Hepatol. 2024;9:493-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 65] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 75. | Zhang GL, Zhao QY, Lin CS, Hu ZX, Zhang T, Gao ZL. Transient Elastography and Ultrasonography: Optimal Evaluation of Liver Fibrosis and Cirrhosis in Patients with Chronic Hepatitis B Concurrent with Nonalcoholic Fatty Liver Disease. Biomed Res Int. 2019;2019:3951574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 76. | Guidelines for the prevention, diagnosis, care and treatment for people with chronic hepatitis B infection [Internet]. Geneva: World Health Organization; 2024 . [PubMed] |
| 77. | Chen VL, Morgan TR, Rotman Y, Patton HM, Cusi K, Kanwal F, Kim WR. Erratum: Resmetirom therapy for metabolic dysfunction-associated steatotic liver disease: October 2024 updates to AASLD Practice Guidance. Hepatology. 2025;81:E133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 78. | Bansal MB, Patton H, Morgan TR, Carr RM, Dranoff JA, Allen AM. Semaglutide therapy for metabolic dysfunction-associated steatohepatitis: November 2025 updates to AASLD Practice Guidance. Hepatology. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 79. | Kanwal F, Shubrook JH, Adams LA, Pfotenhauer K, Wai-Sun Wong V, Wright E, Abdelmalek MF, Harrison SA, Loomba R, Mantzoros CS, Bugianesi E, Eckel RH, Kaplan LM, El-Serag HB, Cusi K. Clinical Care Pathway for the Risk Stratification and Management of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2021;161:1657-1669. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 522] [Cited by in RCA: 469] [Article Influence: 93.8] [Reference Citation Analysis (4)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/