INTRODUCTION
Anastomosis of the digestive tract is commonly performed in general surgery, with primary methods including manual suturing, laparoscopic instrument-sutured anastomosis, and fully stapled anastomosis[1,2]. However, regardless of the anastomotic method employed, they all carry the risk of digestive tract fistulas due to severe anastomotic infection, insufficient blood supply to the tissue wall, or excessive anastomotic tension[3-5]. These fistulas pose significant therapeutic challenges and life-threatening consequences in clinical practice.
Esophageal pleural fistula (EPF), a particularly severe subtype, predominantly occurs following proximal gastrectomy, involving the thoracic cavity. Its additional etiologies mainly include esophageal tumor perforation, iatrogenic thoracic surgical injuries, and radiation therapy complications[6,7]. The incidence of EPFs post-lobectomy is approximately 0.2% to 1.0%[8], while gastric interposition surgeries exhibit incidence rates ranging from 1.9% to 11.3%[9-11].
Clinically, EPFs typically manifest with pyothorax-related symptoms, including dysphagia, chest pain, fever, dyspnea, pleural effusion, and malnutrition[12-15]. Current therapeutic strategies include abscess drainage, delayed surgical repair after infection control, and stent placement[16,17]. Nevertheless, treatment outcomes are poor with mortality rates exceeding 31%[8,18]. The high mortality underscores the urgent need for innovative management approaches.
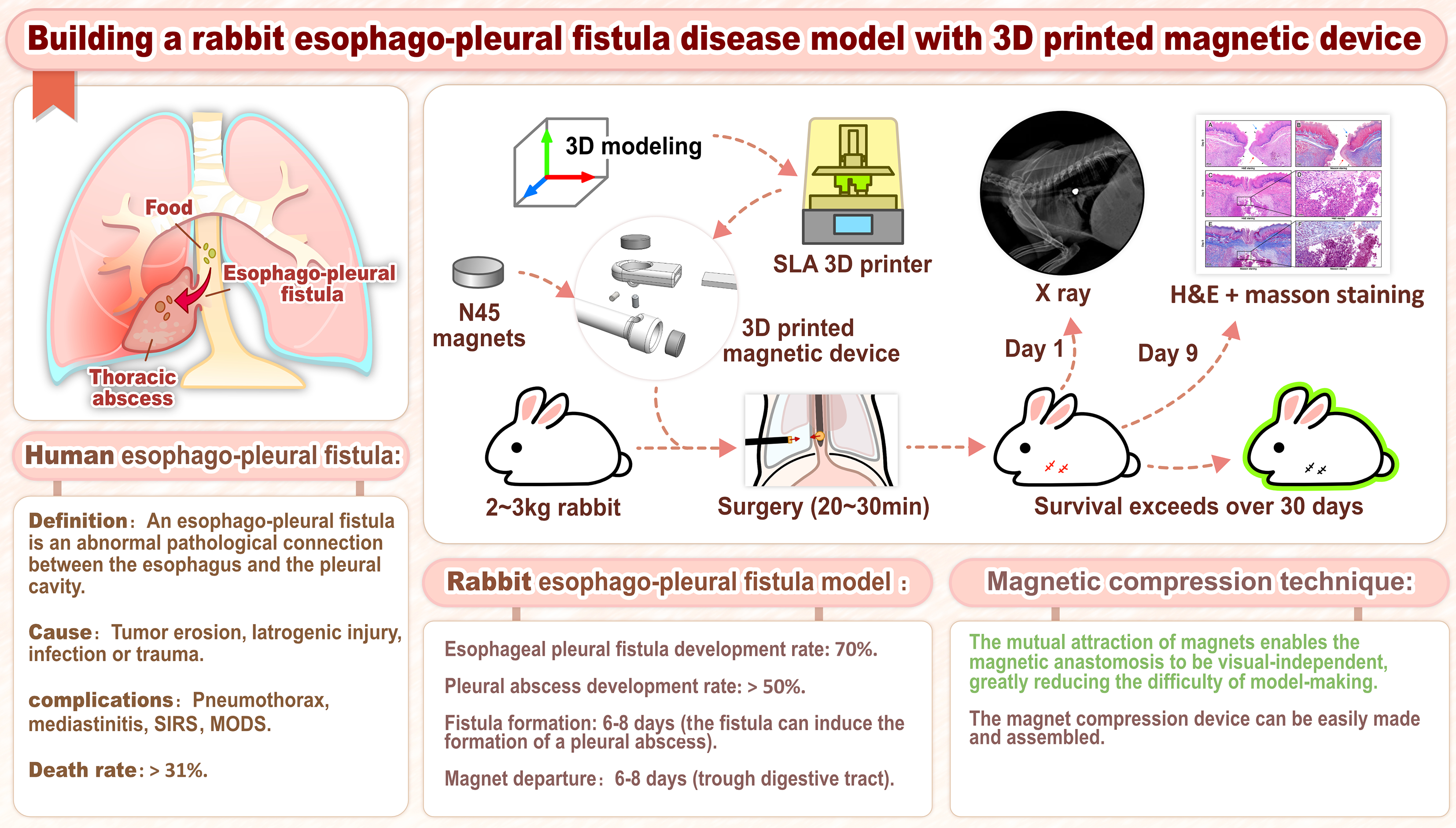
To date, no established animal models of EPFs in any species have been reported. In this pioneering study, we developed a novel magnetic compression technology (MCT)-based rabbit EPF model and described the production method for the modeling (Figure 1). This study provides crucial preclinical evidence for evaluating various sealing materials (e.g., bioadhesives, tissue sealants), surgical interventions, and other therapeutic modalities.
Figure 1 Graphical abstract of the study.
SIRS: Systemic inflammatory response syndrome; MODS: Multiple organ dysfunction syndrome; H&E: Hematoxylin & eosin; SLA: Sterolithography apparatus.
MATERIALS AND METHODS
Ethical approval
The animal use protocol was reviewed and approved by the Institutional Animal Care and Use Committee of the Second Xiangya Hospital, Central South University, China (Approval No. 2021491).
Thirty New Zealand White rabbits weighing 2-3 kg were included in this study. Due to its exploratory nature, 10 rabbits were allocated to preliminary experiments and 20 to formal experiments. At the study endpoint, all rabbits were euthanized via intravenous injection of sodium pentobarbital (100 mg/kg) for anatomical analysis and tissue collection.
Magnet selection
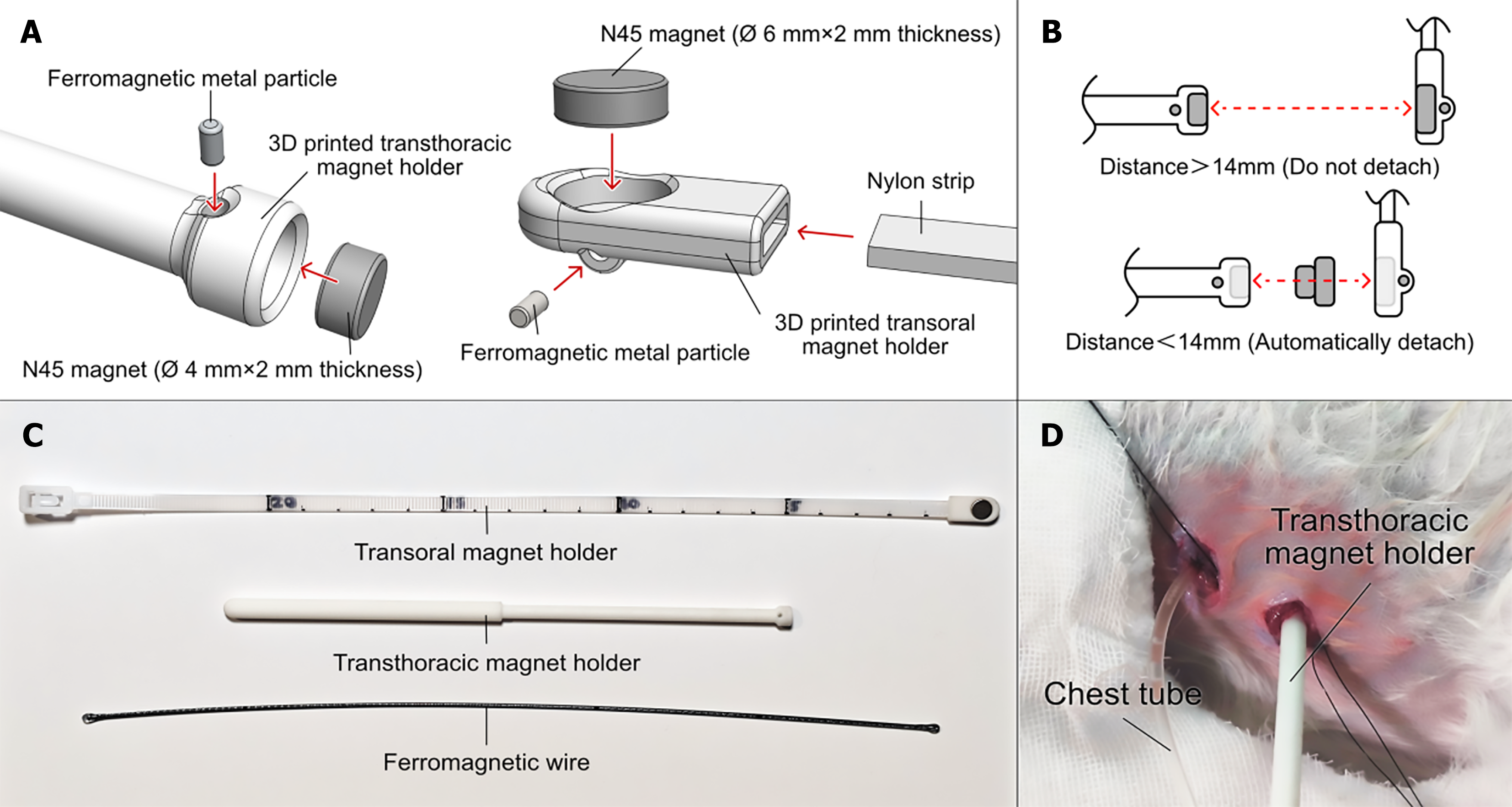
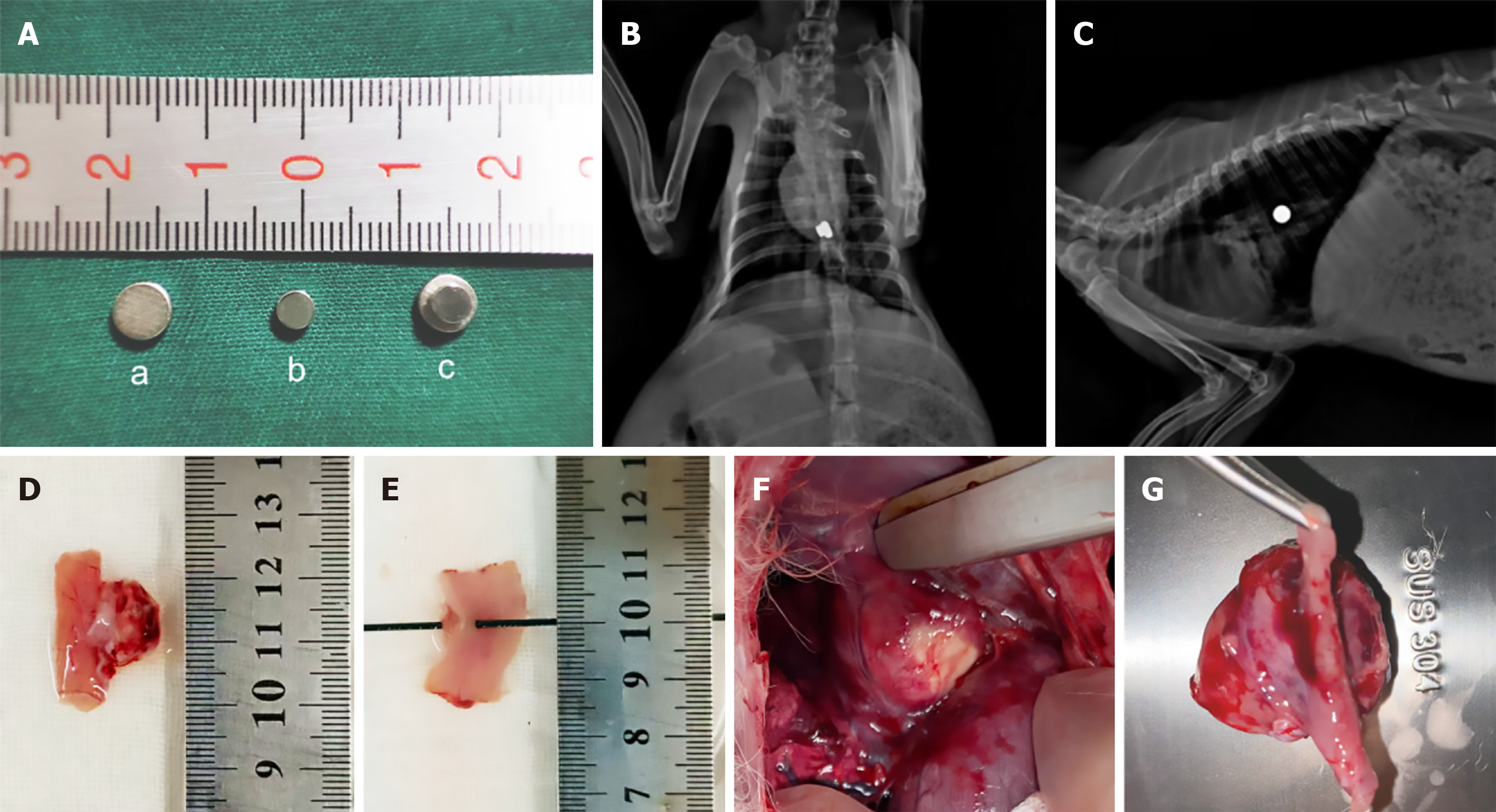
Transoral magnet: N45-grade neodymium-iron-boron (NdFeB) magnet (Ø 6 mm × 2 mm thickness); Transthoracic magnet: N45-grade NdFeB magnet (Ø 4 mm × 2 mm thickness) (Figure 2A); both magnets were coated with an anti-erosion coating. On direct contact (d = 0 mm), the magnets generate a force of 3.64 N. When the magnets align within the rabbit, they generate a compressive force of approximately 3.30 N, resulting in a localized tissue pressure of 262.7 kPa.
Figure 2 Magnetic compression technology-based modeling tool for esophageal pleural fistula induction in rabbits.
A: Schematic assembly of the modeling tool; B: Schematic of the magnetic release mechanism; C: Picture of the modeling tool; D: Pre-alignment setup: Transthoracic magnet holder insertion (right side, parallel to xiphoid process on the anterior axillary line) and a chest tube should be in place.
3D modeling and 3D printing
The transoral and transthoracic magnet holders were modeled in Shapr3D and fabricated via stereolithography using ABS-like photopolymer resin. Post-printing, both holders underwent secondary ultraviolet curing to eliminate residual monomers. Each holder encapsulates a ferromagnetic metal particle within its structure. This design generates an adequate magnetic retention force to counteract gravity, ensuring stable positioning of the magnets. Upon proximity (d < 14 mm) of the two magnet holders, the inter-magnet attractive force exceeds the retaining magnetic force, triggering controlled magnet detachment and release (Figure 2).
Study procedures
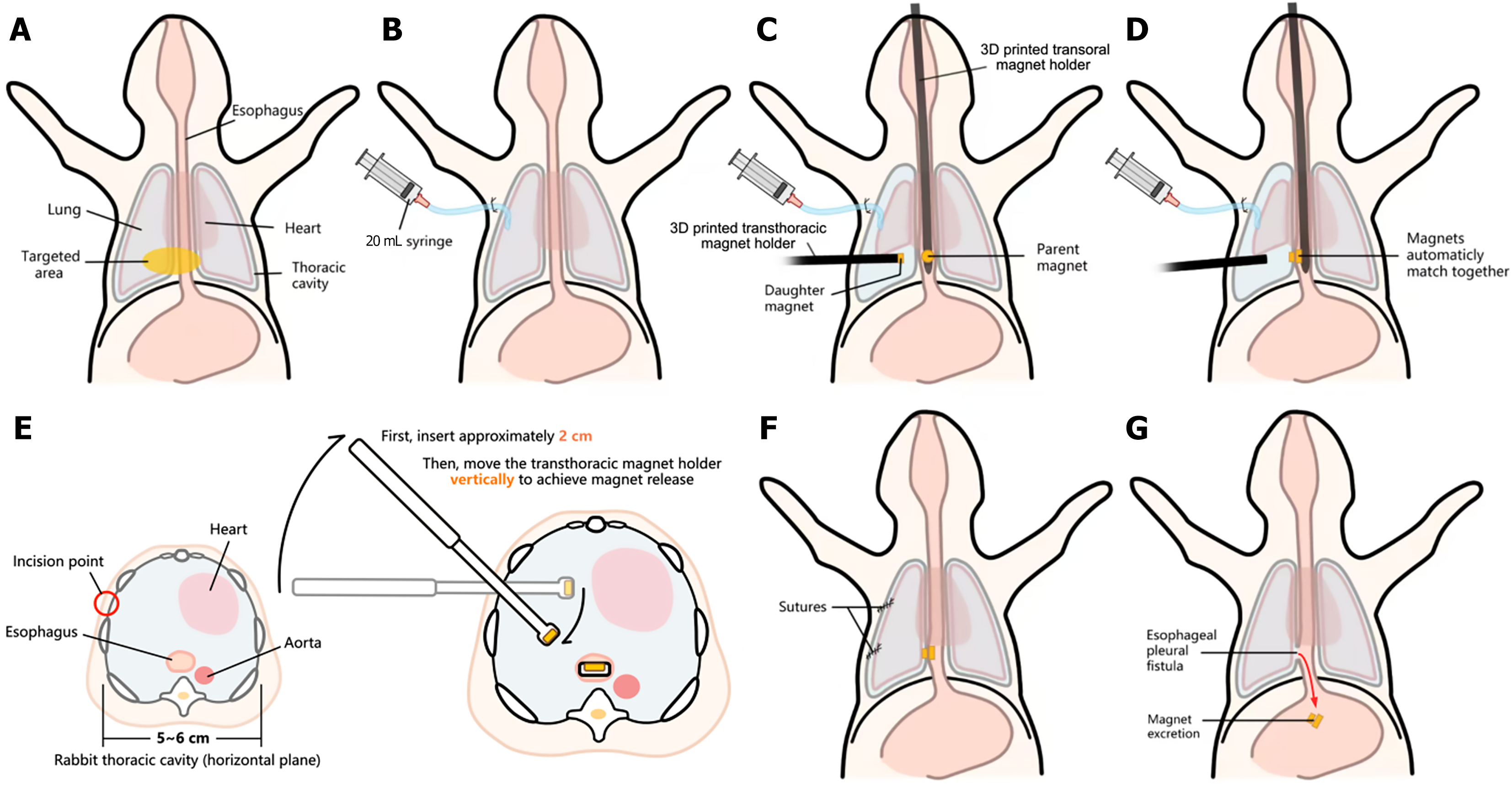
Following successful anesthesia, New Zealand White rabbits (3 kg) were positioned supinely with the right thoracic wall prepped surgically through hair removal, povidone-iodine/alcohol sterilization, and sterile fenestrated draping. Anatomical landmarks were established at the mid-axillary-xiphoid intersection for magnetic device insertion and 1 cm superior in the adjacent intercostal space for drainage access (Figure 3A). Local anesthesia (lidocaine) was then performed at both sites, followed by 1 cm incisions through the skin, subcutaneous layers, and intercostal muscles. Hemorrhage was controlled using 6-0 sutures and preemptive purse-string sutures were placed around both sites to facilitate subsequent thoracic closure. A curved forceps-guided catheter (syringe-connected, side-ported) was inserted 2 cm into the thorax and secured (Figures 2D and 3B). Magnet deployment was then performed: The transthoracic magnet holder was inserted post-pneumothorax induction (15-20 mL air injection, collapsing the right lung) (Figures 2D and 3C). The transoral magnet holder was placed directly above the rabbit to estimate the insertion length (most likely to be 15-17 cm in 3 kg rabbits). The position of the transthoracic magnet holder was continuously adjusted until a brief traction was felt (magnet release) (Figure 3C-E). The reserved suture was ligated to seal the thoracic cavity. The ferromagnetic wire was inserted into the rabbit’s esophagus to investigate the correct positioning and alignment of the magnets. The plastic catheter was removed after extracting all air from the thoracic cavity, and layered muscle/skin repair was performed (Figure 3F). Postoperatively, levofloxacin was administered, and the rabbits received liquid diet maintenance for 48 hours. The magnet is expected to detach spontaneously and be excreted via the digestive tract within 6-8 days after the procedure (Figure 3G).
Figure 3 Schematic diagram of the esophageal pleural fistula modeling process.
A: The primary operative site (marked yellow); B: Insertion of a 20 mL syringe-connected catheter into the thoracic cavity; C: After injecting 20 mL of air to create space for operation, the transthoracic magnet holder and the transoral magnet holder were inserted; D: Magnetic force pulls the magnets together, compressing the tissue in between; E: A cross-sectional view at the xiphoid level of the thorax; F: Surgical closure after removal of both magnet holders; G: Magnets naturally fall off within 6-8 days postoperatively.
RESULTS
A group of 20 New Zealand White rabbits (body weight: 2-3 kg) underwent EPF modeling surgery. Among them, pleural abscesses formed in 14 rabbits, with two cases exhibiting abscess encapsulation that prevented normal magnet expulsion. The remaining six rabbits failed to develop pleural abscesses. In the EPF disease tolerance test, all eight rabbits survived until postoperative day (POD) 30, followed by euthanasia and tissue harvesting.
Statistical analysis
Key metrics included: Modeling surgery duration (26.60 ± 3.99 minutes), anesthesia duration (7.95 ± 1.20 minutes), magnet excretion time (7.06 ± 0.70 days), abscess volume on day 9 (5.0 ± 1.0 cm3) (n = 8), and abscess volume on day 30 (71.4 ± 31.7 cm3) (n = 5).
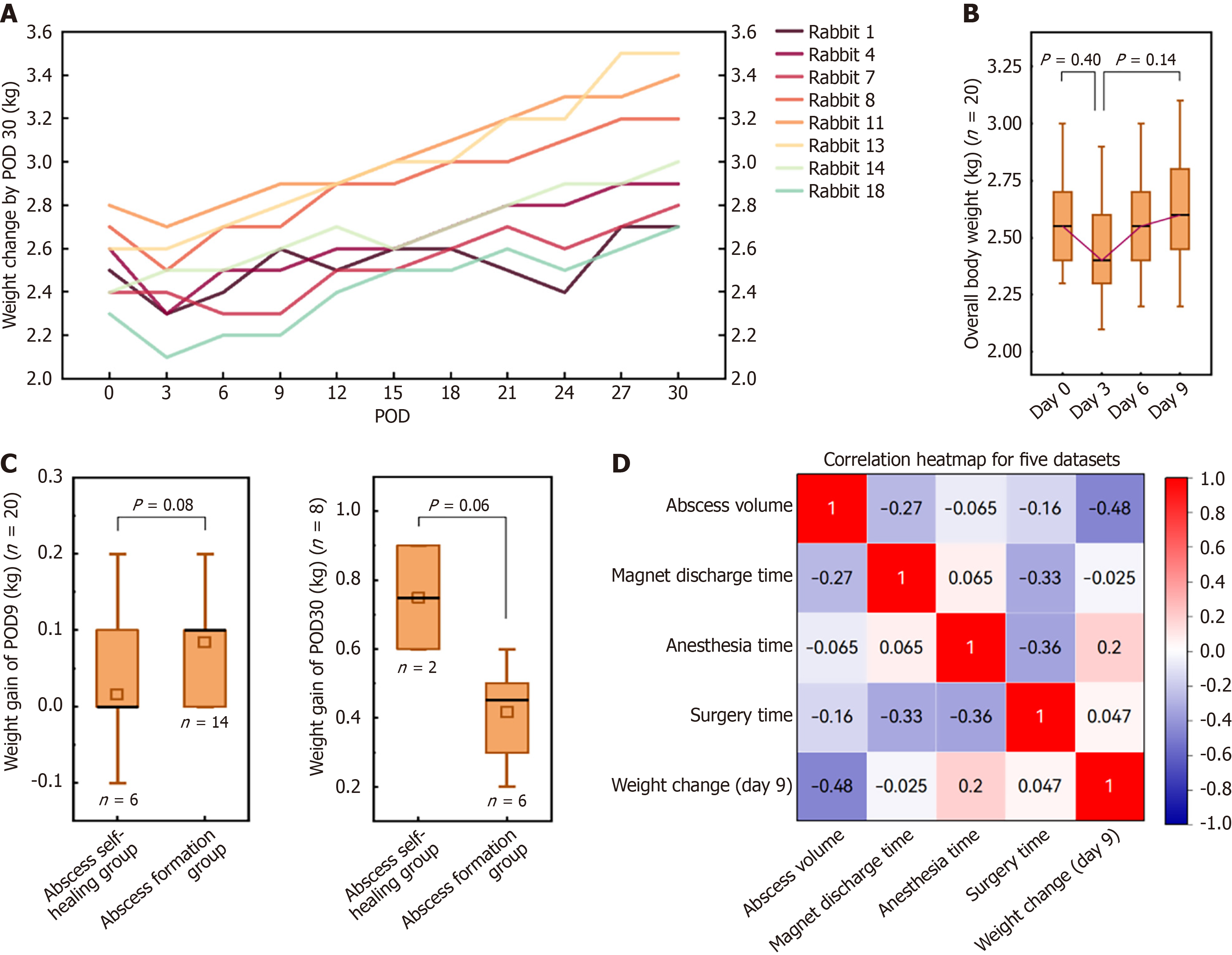
All eight rabbits subjected to surgical tolerance testing exhibited weight gain (0.5 ± 0.2 kg) (n = 20) postoperatively (Figure 4A). Monitoring of 20 rabbits over the first 9 PODs revealed an initial weight loss, which generally recovered to or exceeded baseline levels by POD 6. However, these weight change trends were subtle, and no statistically significant differences were detected (all P values > 0.05) (Figure 4B).
Figure 4 Results of perioperative weight changes and correlation analysis in esophageal pleural fistula modeling.
A: Body weight changes in 8 rabbits during the 30-day perioperative period; B: Body weight changes in 20 rabbits over the 9-day perioperative period; C: Changes in rabbit body weight after surgery (the body weight of postoperative day 9 minus the body weight before surgery, and the body weight of postoperative day 30 minus the body weight before surgery), the “□” represents the mean value; D: Correlation analysis among five datasets. POD: Postoperative day.
As abscess formation indicates successful modeling, rabbits were categorized into 2 groups: Abscess formation group and abscess self-healing group. Analysis of weight gain in 20 rabbits over 9 days and 8 rabbits over 30 days showed apparent differences on box plots, but were not statistically significant (P value > 0.05), due to sample size (Figure 4C).
Furthermore, the correlation between abscess volume and several variables (magnet discharge time, anesthesia time, surgery time, and weight change over the first 9 PODs) was weak and mostly statistically insignificant (Figure 4D).
Imaging findings (X-ray)
On POD 1, anteroposterior and lateral radiographs confirmed appropriate magnet pairing at the sub-xiphoid region, inferior to the cardiac border, with no displacement or detachment observed. The bilateral costophrenic angles remained sharp, and no pleural effusion or other pathological manifestations were detected (Figure 5A-C).
Figure 5 Magnets and gross observations.
A: Picture of the magnets, transoral magnet (Ø 6 mm × 2 mm thickness) (a), transthoracic magnet (Ø 4 mm × 2 mm thickness) (b), magnetically coupled magnets (c); B and C: X-ray day 1 post-operation; D: Gross specimen of the esophagus and the esophageal pleural fistula (EPF) abscess (day 9); E: Gross specimen of EPF (day 9) (with the EPF abscess cleared away and a metal wire passing through the fistula); F: Gross specimen of the EPF abscess in vivo (day 30); G: Gross specimen of the EPF abscess in vitro (day 30).
Gross and histological observations
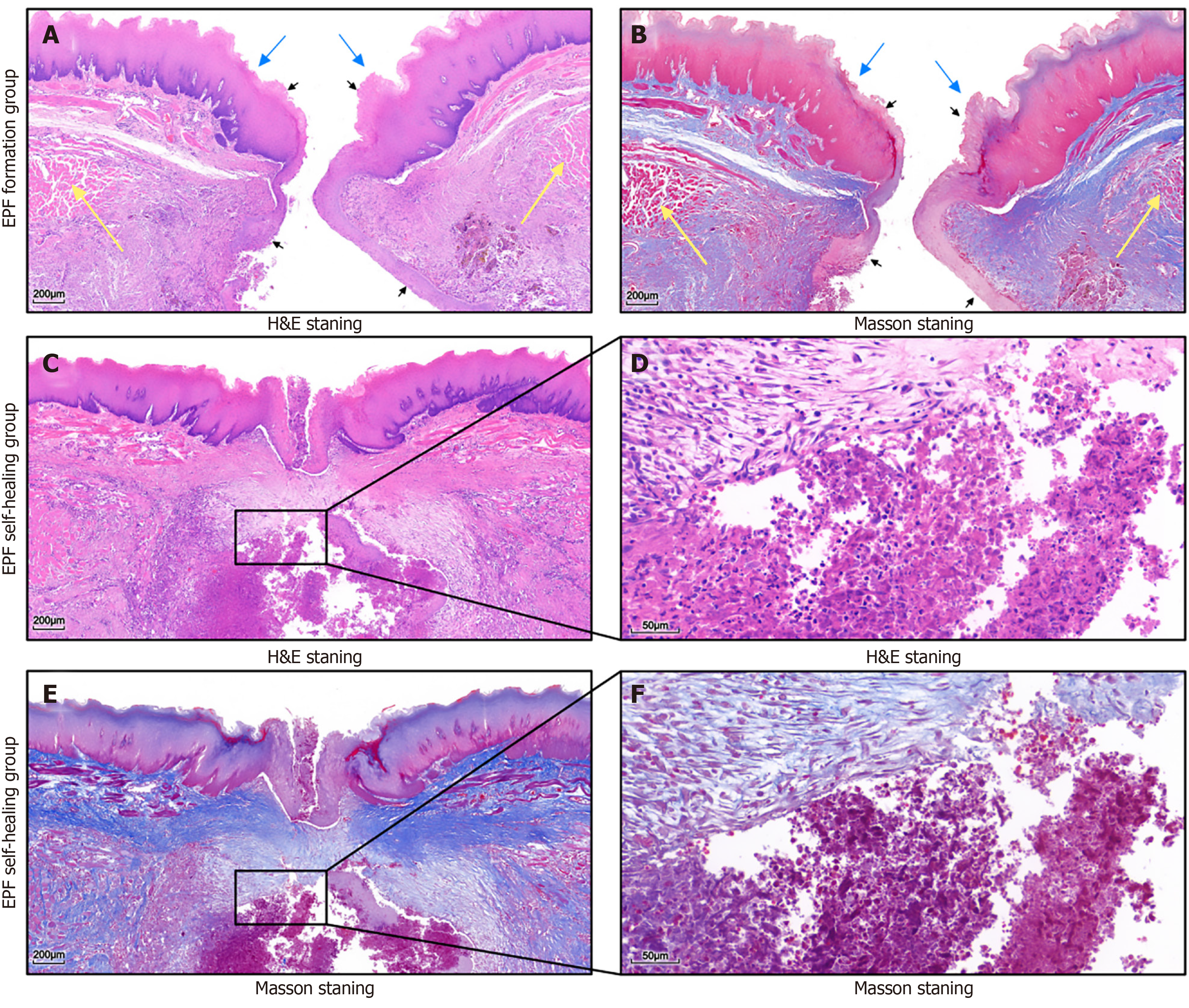
Gross examination of the specimen from POD 9 revealed a pleural abscess adhering to the esophagus. Following resection, EPF formation was observed (Figure 5D-G). Tissue samples were collected from both EPF formation sites and spontaneously healed EPF regions for histological analysis using hematoxylin-eosin staining and Masson’s trichrome staining (Figure 6).
Figure 6 Histological observations.
A and B: Histological analysis shows disruption of the esophageal mucosa (blue arrow) and muscular layer (yellow arrow), with esophageal squamous epithelium extending towards the thoracic cavity (black arrow), representing the formation of esophageal pleural fistula (EPF); C-F: The EPF self-healing group shows that mucosal restitution is the primary reparative event; the abscess cavity contains abundant necrotic debris and infiltrating inflammatory cells, surrounded by newly synthesized fibrous tissue that initiates the encapsulation of the abscess. H&E: Hematoxylin & eosin; EPF: Esophageal pleural fistula.
In the EPF formation group, histopathological examination showed disruption of the esophageal mucosa and muscular layer, with esophageal squamous epithelium extending along the ruptured margins into the pleural cavity (Figure 6A and B).
In the EPF self-healing group, re-epithelialization across the fistula site and submucosal fibrosis were observed, accompanied by early-stage abscess formation adjacent to the esophagus, characterized by peripheral chronic inflammatory infiltration (eosinophils, lymphocytes, plasma cells) and central coagulative necrosis with karyorrhexis (Figure 6C-F).
DISCUSSION
In this study, we discovered the feasibility of creating an EPF model in rabbits using MCT via oral and right thoracic cavity approaches. The rabbits generally tolerated the procedure well. None of the models developed postoperative complications such as hemothorax, pneumothorax, or vascular injuries. All the magnets were positioned correctly. The positioning of the magnet is primarily determined by the transthoracic magnet holder. Therefore, as long as the movement range of the magnet holder is controlled within the level of the xiphoid process, positional misalignment is unlikely to occur (Figure 3E). After the magnets had detached, most rabbits can form a relatively stable fistula.
The development of a rabbit EPF model in this study represents the first report of a successful animal model for this condition, for which no prior models exist in any species. Although studies for esophageal-pleural fistula animal models are currently lacking, the MCT used in this study has been extensively investigated. The historical application of MCT in animal studies dates to the 1980s, commencing with Obora et al’s magnetic ring for vascular anastomosis in dogs and rats[19]. Key developments followed, including intestinal anastomosis in pigs and use in canine liver transplantation (2013)[20,21], small intestinal anastomosis in rats (2018)[22], and the establishment of a canine tracheoesophageal fistula model (2019)[23]. In clinical practice, MCT has seen diverse applications, most notably in treating biliary obstruction since 2003[24-27], as well as in managing esophageal strictures, diverticula, and other intestinal procedures[28-31].
EPFs are fatal, often secondary to trauma, tumors, esophageal diverticulum perforation, post-lobectomy complications, and post-radiation therapy complications[32-36]. Due to the non-specific clinical presentations of EPF (e.g., fever, dyspnea, or chest pain), its diagnosis depends critically on imaging findings[32,37]. With the emergence of some new imaging techniques, we may achieve earlier and more accurate diagnosis and treatment[38-41]. Conservative treatments include pleural effusion drainage, local irrigation, tube feeding, gastrostomy or jejunostomy, followed by surgical treatment, including repair or direct reconstruction of the esophagus. Due to the poor prognosis of EPFs, constructing corresponding animal models to verify new treatment methods is particularly important.
At the beginning of this experiment, our team considered the option of operating under fluoroscopy or with a thoracoscope for better vision. Given the concerns over fluoroscopic radiation exposure, as well as the fact that conventional thoracoscopic instruments are too large for the relatively limited thoracic volume of rabbits and require a complex surgical setup for one-lung ventilation, we did not adopt these two approaches for modeling. We first attempted to perform the magnet alignment operation under direct vision through a large incision in the rabbit’s chest. However, this method caused considerable trauma, resulting in hypoxia. The surgical operation time window was narrow, making it difficult to complete the operation with one attempt. The postoperative recovery and survival conditions were also poor, and complications such as hemothorax, pneumothorax, and pleural effusion were likely to occur. Therefore, we developed the MCT tool set. The method of automatic magnet alignment can greatly reduce the difficulty of modeling and allows position control of the magnet alignment. The rabbits demonstrated good tolerance to the injection of 20 mL of gas into the thoracic cavity, which was performed to create operational space for the magnetic anastomosis, and the surgical window was relatively wide (> 10 minutes). Even if the rabbits showed symptoms of hypoxia, their breathing conditions could quickly improve after aspirating the excessive gas in the thoracic cavity. Since the transoral magnet holder would bend, the insertion depth of the guidewire would exceed the distance from the rabbit’s upper incisors to the xiphoid process by about 1 cm. The transoral magnet holder was assembled from a 6 mm wide × 1 mm thick nylon cable tie, a 3D-printed transoral magnet holder, and a magnetic metal particle. A nylon strip below 6 mm wide × 1 mm thick would not provide enough strength and width suitable for surgical operations. The transthoracic magnet holder was assembled from a 3D-printed rod and a magnetic metal particle (Figure 2A-C). During magnet release, a brief pulling sensation should be felt. Due to the thin tissue layer between the esophagus and the thoracic cavity, the magnetic compression force achieved upon magnet apposition is relatively strong, thereby accidental displacement during the procedure is not likely to occur. However, this also implies that once the magnets are aligned, repositioning the compression site is generally not recommended, as it is most likely to cause esophageal rupture. Post-operation, the position of the magnet can be confirmed by simply inserting the ferromagnetic wire into the rabbit’s esophagus or X ray imaging. Since the force generated by the magnet was relatively small, the ferromagnetic wire would not shift the position of the magnet while transmitting enough force for the operator to feel.
Applying MCT to construct EPFs has great advantages at the anatomical level. By utilizing the characteristic of mutual attraction between magnets, the process of directly visualizing and searching for the esophagus during surgery is avoided, the surgical time is shortened, and the overall trauma is reduced. Thus, the majority of rabbits can tolerate the surgical process and survive for a relatively long period (> 30 days) after surgery, providing a relatively stable animal model for the treatment and research of EPFs.
The EPF model establishment success rate in this study was approximately 70%. Most of the models developed pleural abscesses as intended. In some cases, magnets failed to detach normally, potentially due to excessive compression of adjacent tissues on the thoracic side - including partial lung lobes. The potential factors contributing to spontaneous healing in certain cases may include: (1) The use of an undersized magnet: Creating a fistula too small, prone to spontaneous closure; (2) Adhesion formation induced by local inflammation: Encapsulating the magnet, thereby preventing the dissemination of esophageal contents into the pleural cavity; (3) Lack of physical support at the fistula site to maintain luminal patency; and (4) Small quantity or low virulence of bacterial formation at the fistula site. Additionally, varying postoperative dietary regimens may influence the microenvironment around the fistula, thereby potentially affecting the overall modeling success rate.
Although postoperative magnet detachment time could be roughly assessed through X-ray imaging, examination of excretions, or using the ferromagnetic wire, comprehensively, the use of a ferromagnetic wire seemed to be the most accurate and efficient method.
In the successfully established rabbit EPF models from this study, pathological examination revealed that squamous epithelial cells on the esophageal side exhibited a tendency to migrate inward along the fistula tract. This cellular extension may impede direct apposition and healing of the submucosal tissues across the fistula, thereby permitting the leakage of esophageal contents into the thoracic cavity (i.e., the abscess cavity). This mechanism might also contribute to the recalcitrant nature of human EPF, explaining in part its poor healing outcomes.
There are limitations in this study. As noted previously, the small thoracic size of rabbits may limit the applicability of thoracoscopic modeling techniques. Consequently, the rabbit EPF model may not be suitable for evaluating certain surgical interventions designed for larger anatomies. Using larger animal models, such as swine or canines, would be more appropriate for validating surgical treatments. Moreover, the success rate of the current modeling protocol requires improvement. While pleural abscesses formed in most rabbits, their sizes exhibited substantial variability, indicating insufficient modeling stability. A potential solution for future studies would be to use larger magnets to create more consistent and sizable fistulas.