Published online May 14, 2026. doi: 10.3748/wjg.v32.i18.117611
Revised: February 2, 2026
Accepted: March 3, 2026
Published online: May 14, 2026
Processing time: 146 Days and 10.3 Hours
Endoscopy and fecal calprotectin remain the reference standards for assessing in
Core Tip: Leucine-rich α-2-glycoprotein (LRG) is an emerging serum biomarker that showed modest but incremental value for predicting endoscopic activity in Taiwanese patients with ulcerative colitis and Crohn’s disease, particularly when com
- Citation: Guha R, Banerjee A. Letter to the Editor: Leucine-rich α-2-glycoprotein in Taiwanese inflammatory bowel disease: Strengths and limitations of a novel serum marker. World J Gastroenterol 2026; 32(18): 117611
- URL: https://www.wjgnet.com/1007-9327/full/v32/i18/117611.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i18.117611
The prospective study published in World Journal of Gastroenterology by Chen et al[1] on leucine-rich α-2-glycoprotein (LRG) in Taiwanese patients with inflammatory bowel disease provides timely data on an emerging serum biomarker for endoscopic activity. The author has demonstrated LRG as a supportive marker, particularly when combined with C-reactive protein (CRP), hemoglobin, and albumin, but also underscored important limitations compared with fecal calprotectin (FC) and the need for validation beyond a single-center cohort. This Letter briefly highlights these strengths and weaknesses and outlines future research directions to clarify how LRG can be integrated into practical, patient-centered algorithms for noninvasive inflammatory bowel disease (IBD) monitoring.
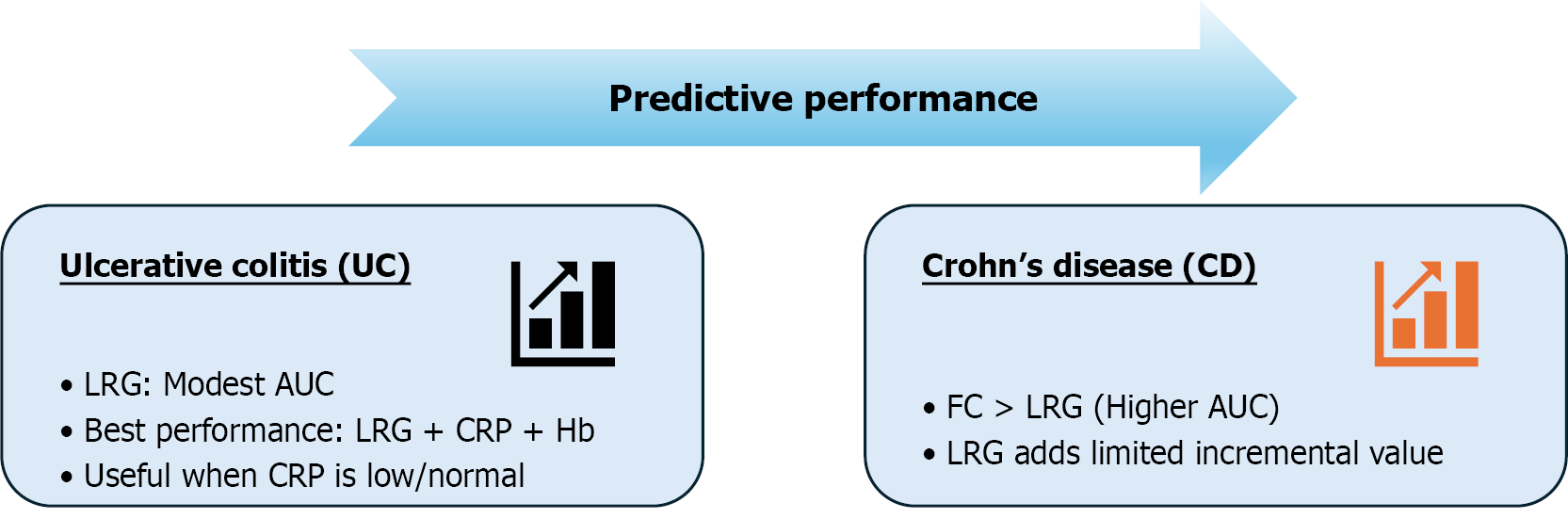
Chen et al[1] reports a prospective, single-center study assessing LRG as a serum biomarker of endoscopic activity in Taiwanese patients with IBD[1] (Figure 1). LRG is a 50-kilodalton glycoprotein released from activated neutrophils and other inflammatory cells and has been implicated as a potential biomarker in immune mediated diseases[2-6]. The investigators enrolled 203 patients [100 with ulcerative colitis (UC) and 103 with Crohn’s disease (CD)] and obtained serum, stool, and clinical data within one month of endoscopy, using the Mayo endoscopic subscore for UC and the Simple En
The main strengths of the study lie in its rigorous methodology and clinically relevant endpoints. Active endoscopic disease is clearly defined, biospecimens are collected within 1 month of endoscopy, and LRG performance is examined separately in UC and CD. In general, all IBD patients showed a significant positive correlation of LRG[8] with FC (r = 0.155, P = 0.028) and CRP (r = 0.565, P = 0.001), which was more striking in UC patients, while in CD patients, LRG was strongly correlated with CRP. In contrast, LRG showed a negative correlation with hemoglobin and albumin. To evaluate diagnostic probability, accuracy, as well as optimal cutoff for each biomarker for predicting active disease, a receiver operating characteristic curve analysis was performed based on the results observed from the endoscopically active vs inactive group, which revealed significant differences in LRG, CRP, and FC levels. Quantitatively, in UC, the area under the curve (AUC) for predicting endoscopic activity is 0.54 for CRP, 0.56 for LRG, and 0.77 for FC, underscoring the superior standalone performance of FC and only modest discrimination by LRG alone. In CD, the corresponding AUCs are 0.69 for CRP, 0.60 for LRG, and 0.72 for FC, with no statistically significant differences between the three biomarkers. Most notably, among UC patients with low or normal CRP, combining CRP, hemoglobin, and LRG increases the AUC from 0.56 to 0.76, approaching the performance of FC (AUC = 0.78) while remaining slightly lower. These data support a nuanced conclusion that LRG adds the most incremental value in UC patients with low or normal CRP, where traditional serum markers underperform, and blood-based assessment is particularly challenging. In these settings, LRG appears best suited as part of a composite serum panel, rather than as a standalone test, with FC remaining the dominant noni
| Biomarker/model | Disease group | AUROC | Cutoff | Sensitivity | Specificity | Key point |
| CRP | UC | 0.475 (0.321-0.629) | 0.045 | 50% | 60% | Weak discrimination compared with endoscopy |
| LRG | UC | 0.654 (0.509-0.799) | 7.15 | 77% | 53% | Modest standalone performance |
| FC | UC | 0.779(0.656-0.903) | 35 | 77% | 73% | Best single biomarker in UC |
| CRP + hemoglobin-LRG | UC, low/normal CRP | 0.76 (0.52-0.82) | NA | 88% | 49% | Improves accuracy, close to FC (0.78) |
| CRP | CD | 0.542 (0.298-0.785) | 0.025 | 80% | 36% | Moderate performance |
| LRG | CD | 0.517 (0.232-0.801) | 7.05 | 80% | 50% | Lower than CRP and FC |
| FC | CD | 0.639 (0.344-0.933) | 37.5 | 80% | 58% | The highest AUC among the three in CD |
Several limitations temper enthusiasm for immediate clinical implementation. First, FC consistently outperforms LRG for predicting endoscopic activity in both UC and CD, with higher AUCs that are in line with prior literature, reinforcing FC’s status as the dominant noninvasive biomarker. Second, the study is conducted at a single tertiary center in Taiwan, and the authors appropriately acknowledge that genetic background, environmental exposures, diet, and healthsystem factors may limit generalizability to other Asian and non-Asian populations. Third, although the paper discusses “optimal” LRG cutoffs, there remains substantial variability compared with prior predominantly Japanese cohorts, highlighting the lack of assay harmonization and the risk of overfitting thresholds to a single cohort. Taken together, these issues suggest that, at present, LRG should be viewed as a supportive adjunct to established markers, rather than a replacement for FC.
Overall, this work provides valuable human data extending LRG research beyond Japanese populations and clarifies where LRG is most likely to be clinically useful. Future research should prioritize multicenter, multiethnic validation with standardized LRG assays, direct head-to-head comparisons of different cutoff strategies, and evaluation of LRG within treat-to-target frameworks that incorporate clinical, endoscopic, and patient-reported outcomes. Such work will be es
Moreover, as noted by the authors, biomarker data collection was done one month after endoscopy, which is essentially a long gap, which potentially could reduce the accuracy of the data due to the short half-life of these acute-phase biomarker proteins. Hence, further future validation of the data at closer time points to endoscopy would be im
In conclusion, the study by Chen et al[1] provides important prospective evidence that serum LRG can serve as a supportive blood-based biomarker of endoscopic activity in Taiwanese patients with IBD, particularly in UC with normal or low CRP levels. Nonetheless, its diagnostic performance remains slower than that of FC, and the single-center design and variable cutoff values limit generalizability across populations and assay platforms. Current data therefore support using LRG as a complementary component within serum-based composite panels, or as an adjunct when FC is un
| 1. | Chen YC, Weng MT, Tsai FP, Chen ZC, Wu HY, Tung CC, Wang CY, Wei SC. Role of leucine-rich α-2-glycoprotein in Taiwanese patients with inflammatory bowel disease as a predictive biomarker for endoscopic activity. World J Gastroenterol. 2026;32:114677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 2. | Serada S, Fujimoto M, Terabe F, Iijima H, Shinzaki S, Matsuzaki S, Ohkawara T, Nezu R, Nakajima S, Kobayashi T, Plevy SE, Takehara T, Naka T. Serum leucine-rich alpha-2 glycoprotein is a disease activity biomarker in ulcerative colitis. Inflamm Bowel Dis. 2012;18:2169-2179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 180] [Article Influence: 12.9] [Reference Citation Analysis (3)] |
| 3. | Mishima T, Fujimoto M, Urushima H, Funajima E, Suzuki Y, Ohkawara T, Murata O, Serada S, Naka T. A Role for Leucine-Rich α2-Glycoprotein in Leukocyte Trafficking and Mucosal Inflammation in Inflammatory Bowel Disease. Inflamm Bowel Dis. 2025;31:1637-1648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | Komatsu M, Sagami S, Hojo A, Karashima R, Maeda M, Yamana Y, Serizawa K, Umeda S, Asonuma K, Nakano M, Hibi T, Matsuda T, Kobayashi T. Leucine-Rich Alpha-2 Glycoprotein Is Associated With Transmural Inflammation Assessed by Intestinal Ultrasound in Patients With Crohn's Disease. Aliment Pharmacol Ther. 2025;61:658-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 5. | Shinzaki S, Matsuoka K, Iijima H, Mizuno S, Serada S, Fujimoto M, Arai N, Koyama N, Morii E, Watanabe M, Hibi T, Kanai T, Takehara T, Naka T. Leucine-rich Alpha-2 Glycoprotein is a Serum Biomarker of Mucosal Healing in Ulcerative Colitis. J Crohns Colitis. 2017;11:84-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 129] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 6. | Shinzaki S, Matsuoka K, Tanaka H, Takeshima F, Kato S, Torisu T, Ohta Y, Watanabe K, Nakamura S, Yoshimura N, Kobayashi T, Shiotani A, Hirai F, Hiraoka S, Watanabe M, Matsuura M, Nishimoto S, Mizuno S, Iijima H, Takehara T, Naka T, Kanai T, Matsumoto T. Leucine-rich alpha-2 glycoprotein is a potential biomarker to monitor disease activity in inflammatory bowel disease receiving adalimumab: PLANET study. J Gastroenterol. 2021;56:560-569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 68] [Article Influence: 13.6] [Reference Citation Analysis (4)] |
| 7. | Mosli MH, Zou G, Garg SK, Feagan SG, MacDonald JK, Chande N, Sandborn WJ, Feagan BG. C-Reactive Protein, Fecal Calprotectin, and Stool Lactoferrin for Detection of Endoscopic Activity in Symptomatic Inflammatory Bowel Disease Patients: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2015;110:802-19; quiz 820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 597] [Cited by in RCA: 530] [Article Influence: 48.2] [Reference Citation Analysis (3)] |
| 8. | Ojaghi Shirmard F, Pourfaraji SM, Saeedian B, Bagheri T, Ismaiel A, Matsumoto S, Babajani N. The usefulness of serum leucine-rich alpha-2 glycoprotein as a novel biomarker in monitoring inflammatory bowel disease: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2025;37:891-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |