Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.116386
Revised: January 6, 2026
Accepted: February 13, 2026
Published online: May 7, 2026
Processing time: 164 Days and 2.5 Hours
Qiweizhigan granule (QWZG) is employed in clinical settings for the treatment of metabolic dysfunction-associated steatohepatitis (MASH). However, the precise biological mechanisms underlying its therapeutic effects are not yet fully elucidated.
To assess the efficacy and the mechanism of QWZG against MASH.
Animal models were established, including normal group, a choline-deficient, L-amino acid-defined high-fat diet (CDAHFD) group, and low/medium/high-dose QWZG groups, as well as a rosiglitazone group. Through com
QWZG significantly ameliorated liver pathology by reducing steatosis, inflammation, and fibrosis. RNA se
QWZG ameliorated the progression of MASH by modulating ferroptosis through the LGALS3/TRAF6/GPX4 axis.
Core Tip: This study reveals that galectin 3 plays a critical role in the progression of metabolic dysfunction-associated steatohepatitis by regulating ferroptosis through the tumor necrosis factor receptor-associated factor 6/glutathione peroxidase 4 axis. The traditional Chinese medicine Qiweizhigan granule significantly alleviates the progression of metabolic dysfun
- Citation: Yang YN, Sun YH, Zhu MZ, Wang YR, Li M, Wang K, Ma J, Zhang L, Hu D, Zhou WJ, Ji G, Dang YQ. Qiweizhigan granule ameliorates metabolic dysfunction-associated steatohepatitis by modulating the galectin 3/tumor necrosis factor receptor-associated factor 6-mediated ferroptosis. World J Gastroenterol 2026; 32(17): 116386
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/116386.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.116386
Metabolic dysfunction-associated steatohepatitis (MASH), a progressive subtype of metabolic dysfunction-associated steatotic liver disease (MASLD), manifests through hepatic lipid overload, sustained inflammatory responses, cellular damage and programmed cell death, eventually leading in later phases to fibrotic scarring, including cirrhosis, hepatocellular carcinoma, and end-stage hepatic insufficiency[1,2]. Around one-fifth of individuals diagnosed with MASLD advance to MASH, and more than 40% of this subgroup subsequently develop hepatic fibrosis[3]. MASH ranks as the second leading cause for liver transplantation worldwide, and represents a growing challenge in global healthcare systems[4]. There is an urgent need to identify effective therapies for the treatment of MASH.
Currently, the main intervention strategy for MASH involves lifestyle modifications, including dietary caloric restric
Galectin 3 (LGALS3) predominantly exerts pro-inflammatory effects and is implicated in a range of chronic disorders associated with persistent inflammatory responses[15]. It modulates toll-like receptor 4 and promotes the expression of tumor necrosis factor receptor-associated factor 6 (TRAF6), thereby inducing inflammatory responses[16]. In addition, elevated levels of LGALS3 are associated with MASH and contribute to liver fibrosis[17]. LGALS3 initiates and amplifies acute inflammatory responses by recruiting macrophages to sites of injury and sustains chronic inflammatory states through the activation of pro-inflammatory pathways, ultimately leading to fibrosis. Numerous inhibitors targeting LGALS3 are currently under development. Key compounds, including belapectin and olitigaltin, have demonstrated efficacy in alleviating liver and pulmonary fibrosis, supporting LGALS3 as a therapeutic target for MASH[18].
Ferroptosis represents a type of programmed cell death driven by iron, characterized by its strong links to oxidative damage, peroxidation of lipids, and the disintegration of the cell membrane[19]. It relies on the accumulation of intracellular iron and the subsequent iron-driven generation of reactive oxygen species. Excess intracellular ferrous iron (Fe2+) generates a substantial amount of hydroxyl radicals via the Fenton reaction. Studies have demonstrated that ferroptosis could promote the progression of MASH[20,21]. In addition, LGALS3 exacerbates inflammatory responses while simultaneously promoting ferroptosis by modulating iron metabolism pathways. A study has demonstrated that inhibiting LGALS3 significantly reduces abnormal iron accumulation, thereby effectively suppressing ferroptosis.
Qiweizhigan granule (QWZG) has been applied in clinical practice for the treatment of MASH. It is composed of seven herbal components: (1) Siegesbeckia pubescens Makino (aerial part; Xixiancao); (2) Sedum sarmentosum Bunge (herb; Chuipencao); (3) Ostrea gigas Thunberg (shell; Muli); (4) Crataegus pinnatifida Bge. var. major N.E.Br. (fruit; Shanzha); (5) Sargassum pallidum (Turner) C. Agardh (seaweed; Haizao); (6) Polygonum cuspidatum Siebold and Zucc. (root and stem; Huzhang); and (7) Schisandra chinensis (Turcz.) Baill. (fruit; Wuweizi). However, the mechanisms underlying efficacy of QWZG in treating MASH remain unclear, which need to be illuminated. This study aimed to evaluate the therapeutic effect and underlying mechanism of QWZG on MASH. The central hypothesis proposed is that QWZG alleviates MASH by inhibiting LGALS3 and thereby modulating downstream ferroptosis. In this study, MASH mice were induced by using a choline-deficient, L-amino acid-defined high-fat diet (CDAHFD), followed by intervention with QWZG. Transcriptomic profiling via RNA sequencing was employed to analyze alterations in mRNA expression.
The TCM formula QWZG comprises seven medicinal materials in a ratio of 3:3:3:1:2:1:1, specifically: (1) Siegesbeckia pubescens Makino (aerial part; Xixiancao); (2) Sedum sarmentosum Bunge (herb; Chuipencao); (3) Ostrea gigas Thunberg (shell; Muli); (4) Crataegus pinnatifida Bge. var. major N.E.Br. (fruit; Shanzha); (5) Sargassum pallidum (Turner) C. Agardh (seaweed; Haizao); (6) Polygonum cuspidatum Siebold and Zucc. (root and stem; Huzhang); and (7) Schisandra chinensis (Turcz.) Baill. (fruit; Wuweizi), which were obtained in Longhua Hospital, Shanghai University of Traditional Chinese Medicine. These botanical materials were combined and immersed in water, followed by two cycles of reflux extraction using an eight fold volume of aqueous solvent per cycle. Each extraction process lasted 1.5 hours. The combined liquid extracts were subsequently filtered, and the filtrate was concentrated using reduced-pressure evaporation at 70 ± 5 °C to obtain a condensed extract with a relative density ranging between 1.25 and 1.30 (measured at 60 ± 5 °C). A dry powdered form of QWZG was subsequently prepared, yielding a final product accounting for 16% of the initial raw material mass.
The components of QWZG were analyzed using ultra-performance liquid chroma-tography coupled with quadrupole time-of-flight mass spectrometry. The liquid chromatography system employed was a Waters Acquity UPLC H-Class system. Chromatographic separation was performed on a Waters ACQUITY UPLC® HSS T3 column (2.1 mm × 100 mm, 1.8 μm) maintained at 30 °C. The injection volume was 3 μL. The detection wavelength was set at 190-400 nm. The mobile phase consisted of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B). Mass spectrometric detection was carried out using an AB Sciex Triple TOF® 4600 high-resolution mass spectrometer with simultaneous data acquisition in positive and negative ion modes. Data were acquired in high-resolution mode, and compound identification was performed by matching with literature references and NP-HRMS Database (Nature Standard, Shanghai).
Approximately 0.5 g of the test sample was accurately weighed and placed in a 50 mL screw-capped conical flask. Total 20 mL of 80% methanol was added, and the mix-ture was sonicated (300 W, 40 kHz) for 30 minutes. After cooling to room temperature, the weight was adjusted, and the mixture was shaken well. The solution was then centrifuged at 12000 rpm for 5 minutes, and the supernatant was collected for analysis.
A total of 48 male C57BL/6J mice, aged six weeks, were sourced from Jihui Laboratory Animal Care Co., Ltd., located in Shanghai, China. The animals were randomly assigned to six experimental groups. Eight mice in the normal group received a standard diet over a two-week period, while the remaining forty mice were maintained CDAHFD (formulated with 60% kcal fat, non-trans fats, 0.1% methionine, no added choline and l-amino acid-defined, supplied by Changzhou SYSE Biotechnology Co., Ltd., China) for the same duration. Following this period, the CDAHFD-fed mice were further randomized into five subgroups: (1) The CDAHFD group (n = 8); (2) Low-dose QWZG (QWZG-L) (n = 8); (3) Medium-dose QWZG (QWZG-M) (n = 8); (4) High-dose QWZG (QWZG-H) (n = 8) groups; and (5) The positive control group receiving rosiglitazone (RSG) (n = 8). RSG, a peroxisome proliferator-activated receptor gamma agonist, has been shown in studies to effectively alleviate insulin resistance, thereby mitigating MASLD[22]. Ultimately, this study established a total of six experimental groups: (1) Normal; (2) CDAHFD; (3) QWZG-L; (4) QWZG-M; (5) QWZG-H; and (6) RSG. The normal group and CDAHFD group were administered sterile purified water once daily by oral gavage for four weeks. QWZG-treated groups were administered QWZG suspension at doses of 2 g raw herb/kg/day (QWZG-L), 4 g raw herb/kg/day (QWZG-M, clinically equivalent dose), and 8 g raw herb/kg/day (QWZG-H) through oral gavage for four weeks. The positive control group received RSG at 1 mg/kg/day. Following a four-week treatment period, the animals were subjected to a 12-hour fasting interval, euthanized via isoflurane anesthesia, and subsequent samples of plasma and hepatic tissue were harvested.
Serum concentrations of alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), total bilirubin (TBIL), triglycerides (TG), and total cholesterol (TC) were quantified with an automated biochemistry analyzer (Toshiba, Japan) and matching reagent kits following the supplier’s guidelines. Additionally, liver tissue homogenates prepared at 4 °C were subjected to analysis for TG and TC content using the same instrument. The biochemical kits were purchased from Shanghai Huachen Bio-reagent Co., Ltd. (Shanghai, China).
Liver tissue specimens from the left lateral lobe were fixed in 10% neutral buffered formalin, paraffin-embedded, and sectioned at 4 µm thickness. Sections were stained with hematoxylin and eosin to evaluate hepatic steatosis and Masson’s trichrome for fibrosis assessment (kits from Zhuhai Beisuo Cell Science and Technology Co., Ltd., Zhuhai, Guangdong Province, China). For lipid visualization, fresh tissues were embedded in OCT compound (Sakura Finetek, Tokyo, Japan), cryosectioned at 8 μm, and stained with Oil Red O (Sigma-Aldrich, St. Louis, MO, United States). Hepatic stellate cell activation was quantified through α-smooth muscle actin (α-SMA) immunostaining (1:200 dilution, 19245, Cell Signaling Technology) and western blotting (1:1000 dilution) following established protocols. NAFLD activity score (NAS) and fibrosis stage were estimated according to Kleiner’s criteria[23].
RNA sequencing analysis was conducted by Shanghai Applied Protein Technology Co., Ltd. (Shanghai, China) in compliance with established protocols. Total RNA was isolated from hepatic tissue samples with TRIzol® Reagent (Magen, China). Following quality assessment, eligible RNA was utilized to construct paired-end sequencing libraries with the ABclonal mRNA-seq Lib Prep Kit (ABclonal, China), according to the guidelines. Library integrity and quality were verified using an Agilent Bioanalyzer 4150 system. Ultimately, raw sequencing data were generated using an Illumina Novaseq 6000 platform.
The initial raw sequencing data were processed using custom Perl scripts developed in-house. HISAT2 software was used to obtain mapped reads. FeatureCounts was used to count the reads numbers mapped to each gene. Subsequently, gene expression levels were quantified in FPKM by incorporating transcript length and the number of reads aligned to each gene. Differential gene expression analysis was carried out with the DESeq2 package in R to identify significantly dysregulated genes. The differentially expressed genes (DEGs) were visualized on a volcano plot. Venn diagram was performed to identify overlapping DEGs between pairwise groups (CDAHFD vs normal and QWZG-H vs CDAHFD). Using Cluster 3.0 software, hierarchical clustering analysis was conducted to illustrate the expression profiles of DEGs modulated by QWZG treatment. For functional enrichment analysis, DAVID 6.8 was used to obtain enriched Gene Ontology terms and Kyoto Encyclopedia of Genes and Genomes pathways.
Hepatocytes (HPs) were isolated by a two-step in situ collagenase perfusion method: Cannulation of the portal vein was performed, followed by sequential circulation perfusion with EGTA buffer and type IV collagenase. After digestion, the cells were filtered and purified by centrifugation. Kupffer cells (KCs) and hepatic stellate cells (HSCs) were isolated via perfusion of the liver with pronase and collagenase. Non-parenchymal cells were collected by low-speed centrifugation and further separated by Percoll gradient centrifugation (25% and 50%). KCs were enriched in the intermediate layer, while stellate cells settled in the bottom layer. All procedures were conducted under aseptic and low-temperature conditions.
The protein concentration was quantified using a BCA assay kit (BN27109, Biorigin, Beijing). Following denaturation, equal amounts of protein from each sample were separated via electrophoresis and transferred onto polyvinylidene difluoride membranes. The membranes were subsequently blocked with 5% skim milk at room temperature for one hour. Primary antibodies, including LGALS3 (1:1000, 89572S, Cell Signaling Technology), glutathione peroxidase 4 (GPX4) (1:10000, ET1706-45, Huabio), TRAF6 (1:10000, A23385, ABclonal), NOD-like receptor family pyrin domain containing 3 (NLRP3) (1:1000, A24294, ABclonal), Achaete-scute family bHLH transcription factor 4 (1:3000, A20414, ABclonal), solute carrier family 7 member 11 (1:500, A25302, ABclonal), α-SMA (1:1000, 19245, Cell Signaling Technology), β-ACTIN (1:20000, ET1602-67, Huabio) were subjected to overnight incubation at a temperature of 4 °C. Following membrane washing, incubation with secondary antibodies was carried out. Acquired images were subsequently evaluated for grayscale intensity utilizing ImageJ software.
RNA was isolated from samples, reverse-transcribed into cDNA, and real-time quantitative PCR was carried out to determine relative expression levels. β-ACTIN served as the internal control for normalization. Corresponding primer sequences are provided in Table 1.
| Gene | Forward primer (5’->3’) | Reverse primer (5’->3’) |
| LGALS3 | AGACAGCTTTTCGCTTAACGA | GGGTAGGCACTAGGAGGAGC |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
| IL-6 | CCAAGAGGTGAGTGCTTCCC | CTGTTGTTCAGACTCTCTCCCT |
| TNF-α | CGGGCAGGTCTACTTTGGAG | ACCCTGAGCCATAATCCCCT |
| NLRP3 | CTCAACAGTCGCTACACGCAG | CGTCCTCGGGCTCAAACA |
| TRAF6 | TAACCTTGCTTTGCGTCCG | TCCGAATGGTCCGTTTGAG |
| ACSL4 | CTCACCATTATATTGCTGCCTGT | TCTCTTTGCCATAGCGTTTTTCT |
| GPX4 | GCCTGGATAAGTACAGGGGTT | CATGCAGATCGACTAGCTGAG |
| SLC7A11 | GGCACCGTCATCGGATCAG | CTCCACAGGCAGACCAGAAAA |
| Tfrc | GTTTCTGCCAGCCCCTTATTAT | GCAAGGAAAGGATATGCAGCA |
| β-Actin | GAGACCTTCAACACCCCAGC | ATGTCACGCACGATTTCCC |
The levels of inflammatory biomarkers were measured with commercial enzyme-linked immunosorbent assay (ELISA) kits. The specific procedure was as follows: After collection, the supernatant was centrifuged at 3000 rpm for 10 minutes. The detected indicators include interleukin (IL)-6 (HJ182, Shanghai Epizyme Biomedical Technology Co., Ltd, China), tumor necrosis factor-α (TNF-α) (HJ207, Shanghai Epizyme Biomedical Technology Co., Ltd, China), and LGALS3 (JL20279, Shanghai Jianglai Biotechnology, Shanghai, China).
Tissue sections were treated with a hydrochloric acid and potassium ferrocyanide mixture. After rinsing, endogenous peroxidase was blocked. Sections were then incubated with DAB to enhance chromogenic signals, followed by hematoxy
The malondialdehyde content in mouse liver was determined using the thiobarbituric acid method according to the manufacturer’s instructions (Nanjing Jiancheng, A003-1) after homogenization of liver tissues.
Superoxide dismutase (SOD) activity in liver was measured according to the manufacturer’s instructions (Nanjing Jiancheng, A001-3) following tissue homogenization.
The total glutathione (GSH) content in liver was determined according to the manufacturer’s instructions (Elabscience, E-BC-K097-M) with supernatant obtained from liver homogenates.
Liver tissue sections were immunostained with primary antibodies against LGALS3 (89572, Cell Signaling Technology), NLRP3 (A24294, Abclonal), and F4/80 (GB113373, Servicebio) at 4 °C overnight. Following this, a secondary antibody incubation was carried out at room temperature for one hour. Finally, imaging of the stained specimens was performed.
Paraffin-embedded mouse liver tissue sections were first dewaxed and rehydrated. Antigen retrieval was then performed, after which endogenous peroxidase activity was quenched using hydrogen peroxide. Non-specific binding sites were subsequently blocked with goat serum. The α-SMA (19245, Cell Signaling Technology), TRAF6 antibody (A23385, Abclonal) was applied and incubated overnight at 4 °C. Following washes with phosphate-buffered saline, an HRP-conjugated secondary antibody was applied and allowed to incubate at ambient temperature. Finally, the tissue samples were dehydrated, cleared, mounted using resinous medium, and observed under a microscope.
Liver tissue samples were homogenized on ice, followed by centrifugation to collect the supernatant. The levels of ferrous iron (Fe2+) and total iron in the tissues were determined colorimetrically in strict accordance with the instructions of the Fe2+ assay kit (Elabscience, E-BC-K773-M) and total iron assay kit (Elabscience, E-BC-K772-M), respectively. Additionally, RAW264.7 cells were collected, lysed, and centrifuged to obtain the supernatant. The intracellular levels of ferrous iron and total iron were then measured according to the recommended procedures of the Fe2+ assay kit (Elabscience, E-BC-K881-M) and total iron assay kit (Elabscience, E-BC-K880-M), respectively.
RAW264.7 cells were grown in dulbecco’s modified eagle medium containing 10% fetal bovine serum, 100 U/mL penicillin/streptomycin. The cells were incubated at 37 °C under a humidified atmosphere of 5% CO2. Following the recommended procedures, Lipofectamine LTX was used to transiently transfect both LGALS3 overexpression and silencing plasmids (Genomeditech, China), along with their respective control plasmids. The LGALS3 recombinant protein was purchased from MCE (HY-P77684) and used to treat RAW264.7 cells at a final concentration of 1000 ng/mL for 24 hours.
Cells were first centrifuged to obtain sufficient pellets, followed by fixation with electron microscopy fixative for four hours. Then, the samples were rinsed with buffer solution. The cells were pre-embedded in molten agarose. Subsequent post-fixation was performed with osmium tetroxide for two hours, followed by dehydration and acetone treatment. The specimens subsequently underwent stepwise infiltration and embedding using a graded concentration series, followed by polymerization at 60 °C for 48 hours to produce hardened resin blocks. Ultrathin sections of 60 nm were cut, collected, and doubly stained. Ultimately, the specimens were examined and visualized via transmission electron microscopy.
Data are expressed as mean ± SEM. Statistical evaluations were conducted using GraphPad Prism version 9.0. Comparisons between groups were carried out by one-way analysis of variance or independent two-sample t-test. A P value of less than or equal to 0.05 was considered statistically significant.
Using ultra-performance liquid chromatography-mass spectrometry, the chemical composition of the QWZG was analyzed. As shown in Supplementary Figure 1 and Supplementary Table 1, a total of 101 characteristic peaks were detected, including the characteristic components of Chuipencao and Shanzha (chlorogenic acid, catechin, quercetin 3,4’-diglucoside, Kaempferol), the characteristic components of Xixiancao (sarmenoside and its isomers), the characteristic components of Huzhang (polydatin, emodin-8-O-glucoside, emodin, physcion-8-O-glucoside sulfate isomer), and the characteristic components of Wuweizi (schisandrin A, schisandrin B), etc.
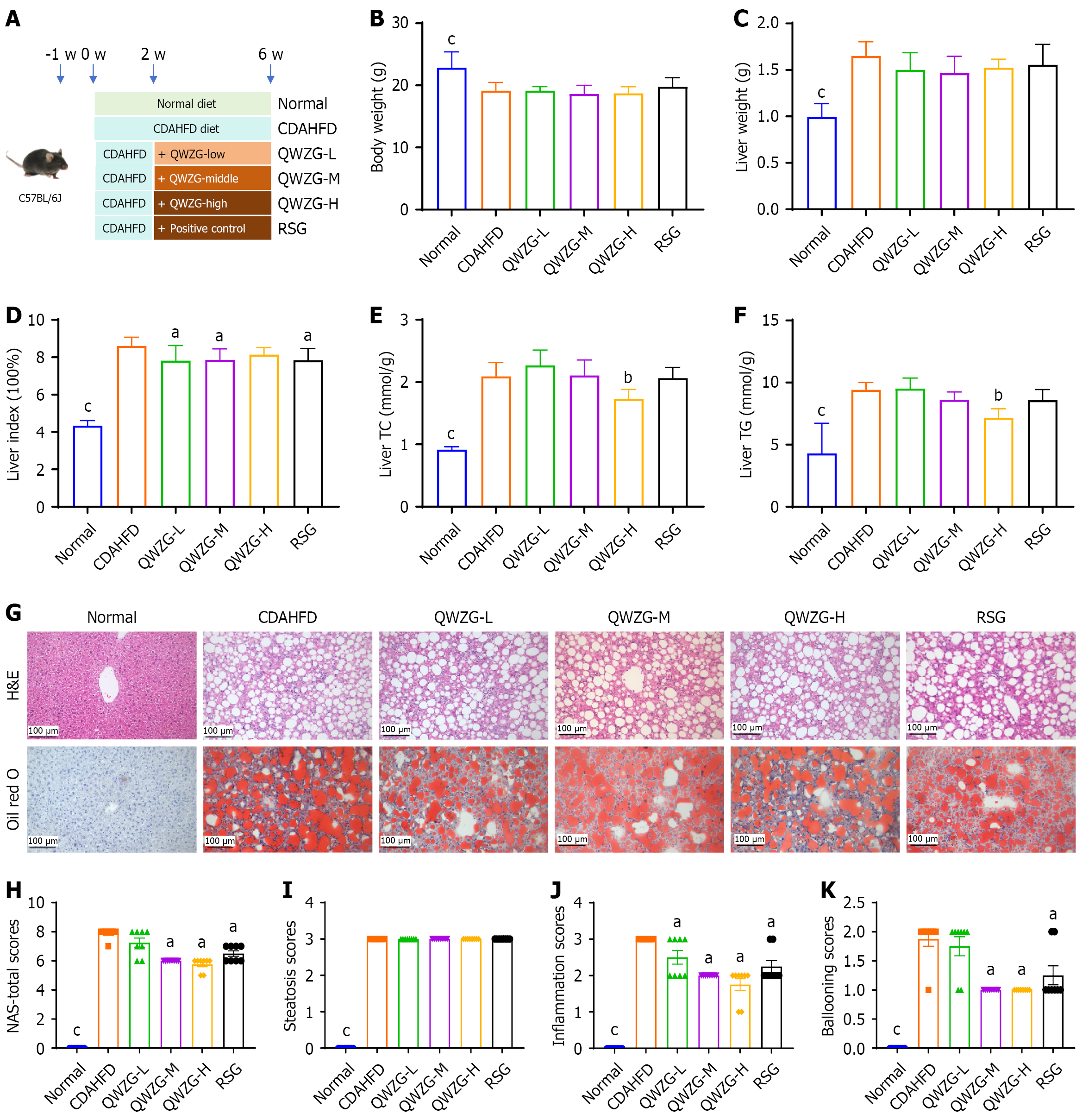
A CDAHFD diet-induced MASH mouse model was used to conduct interventions with QWZG-L, QWZG-M, QWZG-H, as well as the positive control drug RSG (Figure 1A). Compared to the normal group, the CDAHFD group showed significant changes in both body weight (Figure 1B) and liver weight (Figure 1C). However, no significant differences in body or liver weight were observed after intervention with QWZG or RSG. The liver index was significantly higher in the CDAHFD group than that in the normal group, but interventions with QWZG-L, QWZG-M, and RSG resulted in a significant reduction (Figure 1D). Compared to the normal group, the CDAHFD group had markedly elevated levels of liver TC (Figure 1E) and TG (Figure 1F). Interventions with QWZG-H demonstrated significant decreases in liver TG and TC levels. Compared to RSG, the high-dose QWZG exhibited better efficacy in reducing TG and TC levels. Additionally, hematoxylin and eosin and Oil Red O staining on liver tissues were performed (Figure 1G). The results indicated an absence of noticeable lipid droplets in the normal group, whereas abundant red lipid droplet accumulation was observed in the CDAHFD group. All QWZG dosage groups ameliorated hepatic lipid accumulation in mice, with the QWZG-H group exhibiting the most pronounced effect. Compared to the RSG group, QWZG-H demonstrated significantly superior improvement. The CDAHFD group exhibited significantly higher scores for steatosis, inflammation, and ballooning compared to the normal group (Figure 1H-K). The administration of QWZG at all dosage levels significantly alleviated inflammation, with the QWZG-H showing the most substantial reduction. Moreover, QWZG-H demonstrated a more pronounced anti-inflammatory effect compared to the RSG group. In terms of ballooning degeneration, significant improvements were observed in the QWZG-M, QWZG-H, and RSG groups. These results suggest that QWZG, particularly at high doses, can improve MASH.
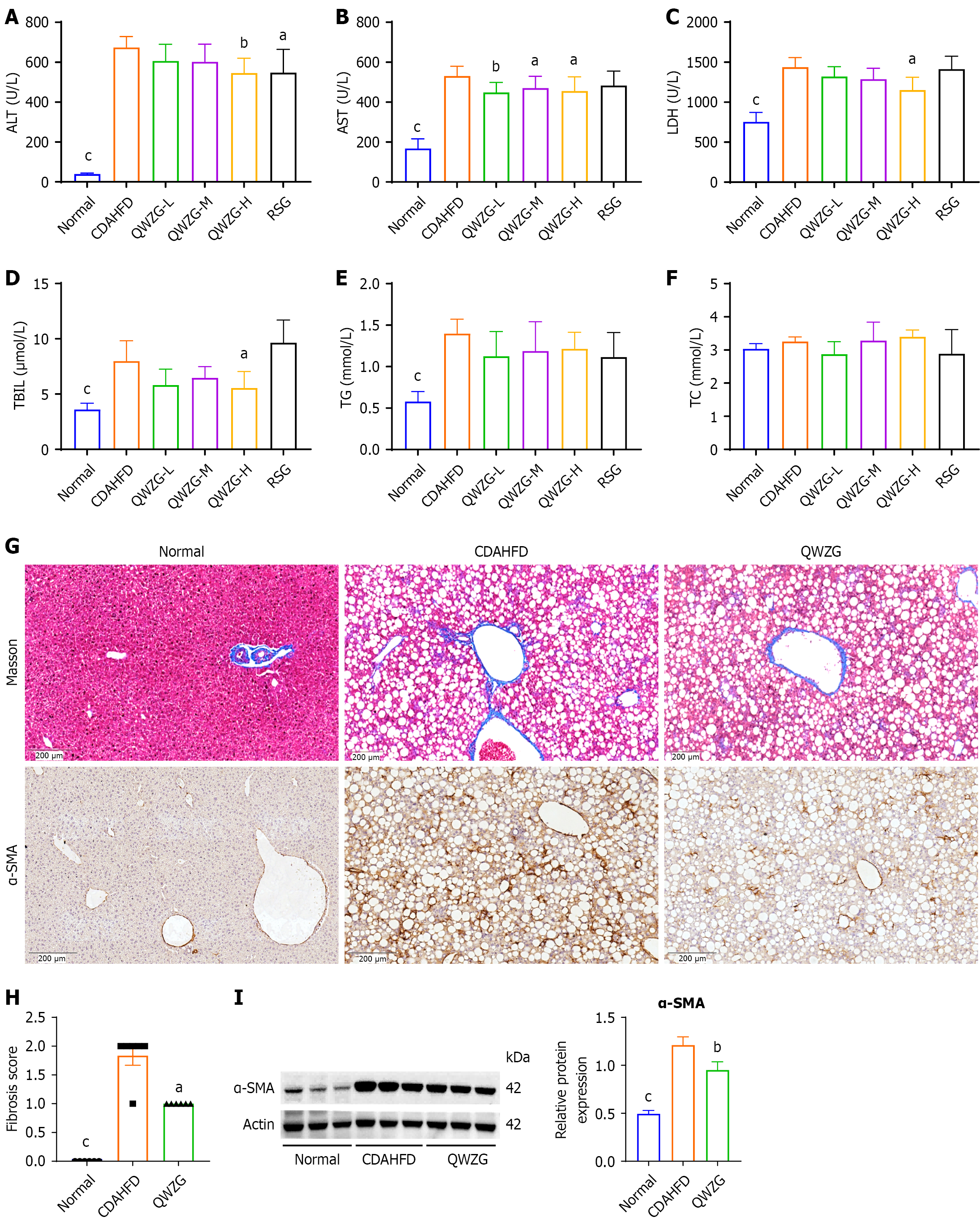
To further assess the phenotypic alterations in mice, relevant biochemical indicators were evaluated. The results demonstrated that the CDAHFD significantly upregulated serum levels of ALT, AST, LDH, TBIL, and TG. The QWZG-H group showed a significant reduction in ALT levels. Although RSG also ameliorated ALT levels in CDAHFD-fed mice, its effect was less pronounced compared to that of the QWZG-H group (Figure 2A). All dosage levels of QWZG effectively decreased AST levels in CDAHFD-induced MASH mice (Figure 2B). Furthermore, only the QWZG-H group demon
To observe the change of fibrosis among normal, CDAHFD, and QWZG groups, Masson’s staining and immunohistochemistry analysis of α-SMA were performed. Results revealed that fibrosis of liver tissues was more serious in the CDAHFD group compared to the normal group, and QWZG restored the fibrosis in CDAHFD-induced mice (Figure 2G-I). These data suggest that QWZG may exert therapeutic effects against fibrosis in MASH mice.
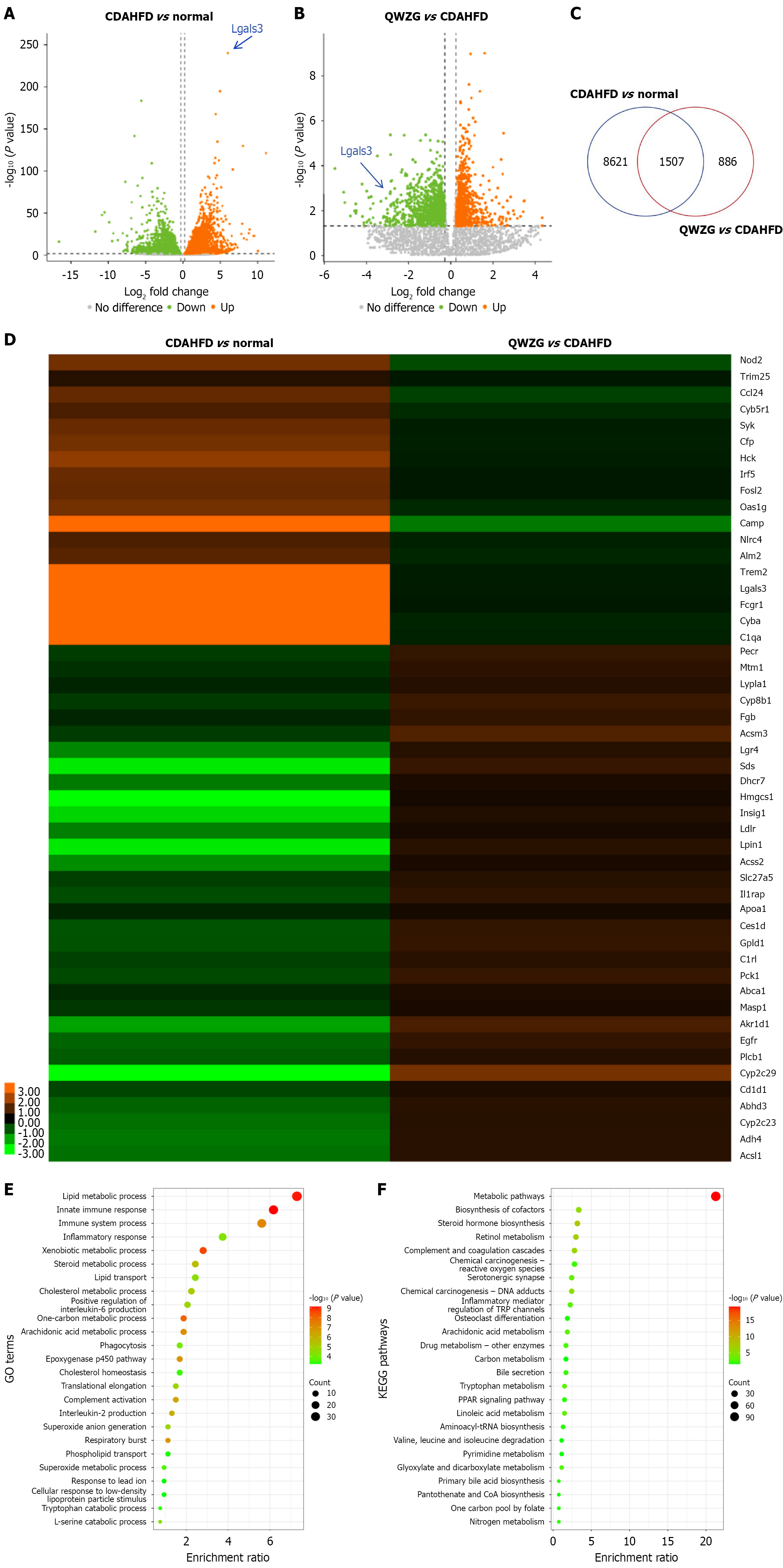
To uncover the underlying mechanisms of QWZG in treating MASH, RNA-sequencing was performed to profile the change of mRNA expression in liver tissues. Using the threshold of fold change of more than 1.2 and P value of less than 0.05, a total of 10128 DEGs were identified between the CDAHFD group and the normal group, comprising 5323 upregulated and 4805 downregulated genes (Figure 3A). In addition, 2393 DEGs (1250 up-regulated and 1143 down-regulated DEGs) in the QWZG group compared to the CDAHFD group were identified (Figure 3B, Supplementary Tables 2 and 3). Using Venn diagram (Figure 3C), we obtained 1507 overlapped DEGs between pairwise groups. The expression of 562 overlapped DEGs was reversed by QWZG. The expression patterns of 50 representative QWZG reversed DEGs were presented in Figure 3D and Supplementary Table 4. All 562 DEGs were significantly enriched in 154 Gene Ontology terms, including lipid metabolic process and inflammatory response, and 30 Kyoto Encyclopedia of Genes and Genomes pathways, including metabolic pathways and inflammatory mediator regulation of TRP channels (Figure 3E and F, and Supplementary Tables 5 and 6). Among these, LGALS3 was identified as one of the most significantly DEGs. These observations indicate that LGALS3 could serve as a key target of QWZG in managing MASH.
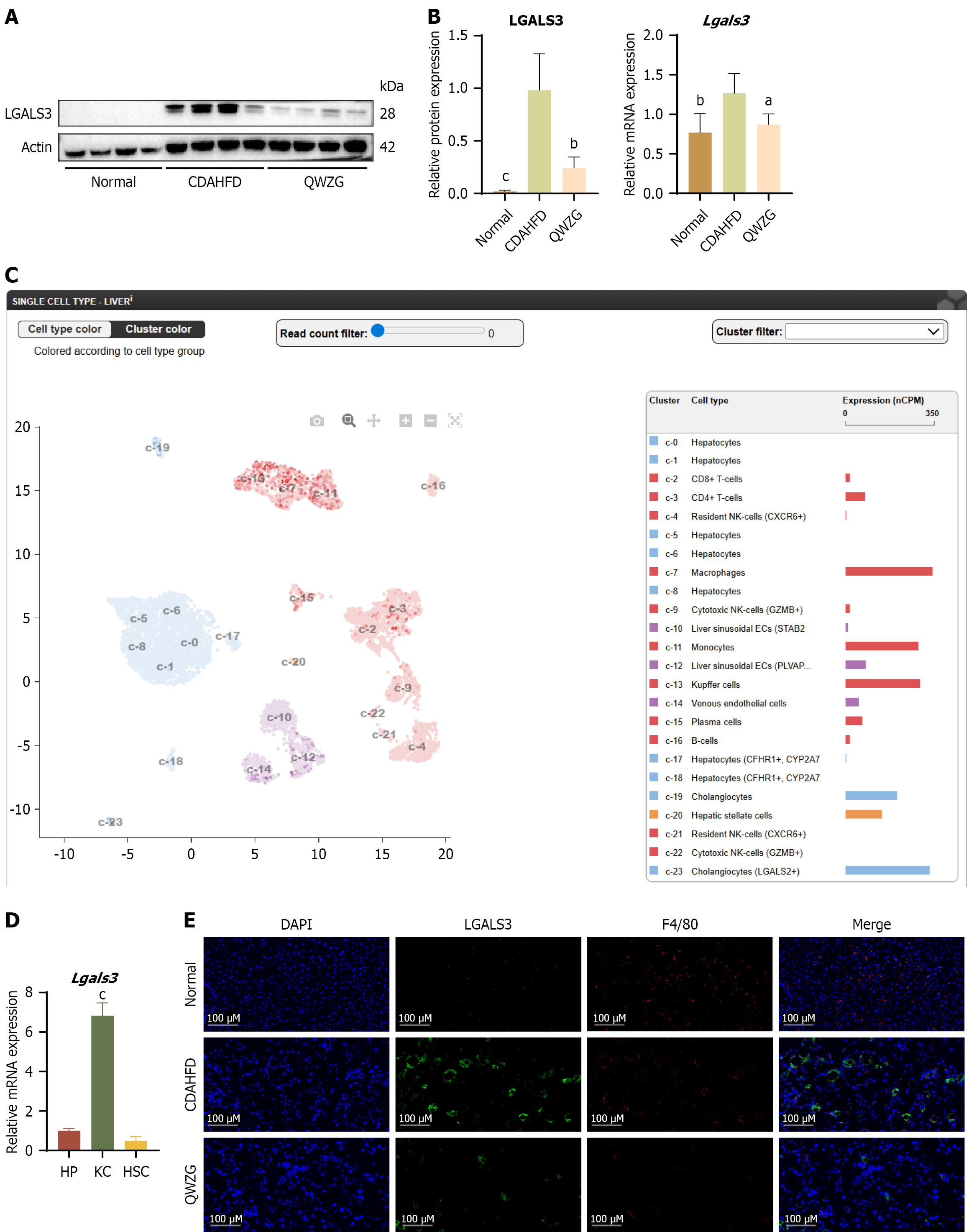
To further validate the regulatory effect of QWZG on LGALS3 expression, level of LGALS3 were verified. Results revealed a significant upregulation of LGALS3 mRNA and protein levels in the liver of the CDAHFD group compared to the normal group. In contrast, QWZG intervention markedly downregulated LGALS3 protein expression (Figure 4A and B). To identify the primary cell type responsible for LGALS3 expression in the liver, a bioinformatic analysis of The Human Protein Atlas database was conducted. The database revealed that in human liver tissue, LGALS3 protein is predominantly enriched in KCs, while exhibiting relatively low expression levels in HPs and HSCs (Figure 4C). To further corroborate these results, KCs, HPs, and HSCs were isolated from livers of mice and subsequently cultured in vitro. Results indicated that mRNA levels of LGALS3 were markedly elevated in KCs relative to the other two hepatic cells, aligning closely with prior database predictions (Figure 4D). Furthermore, the result of immunofluorescence showed clear co-localization of LGALS3 with the macrophage marker F4/80 in liver tissue (Figure 4E). Based on these results, LGALS3 primarily functions within KCs in the liver.
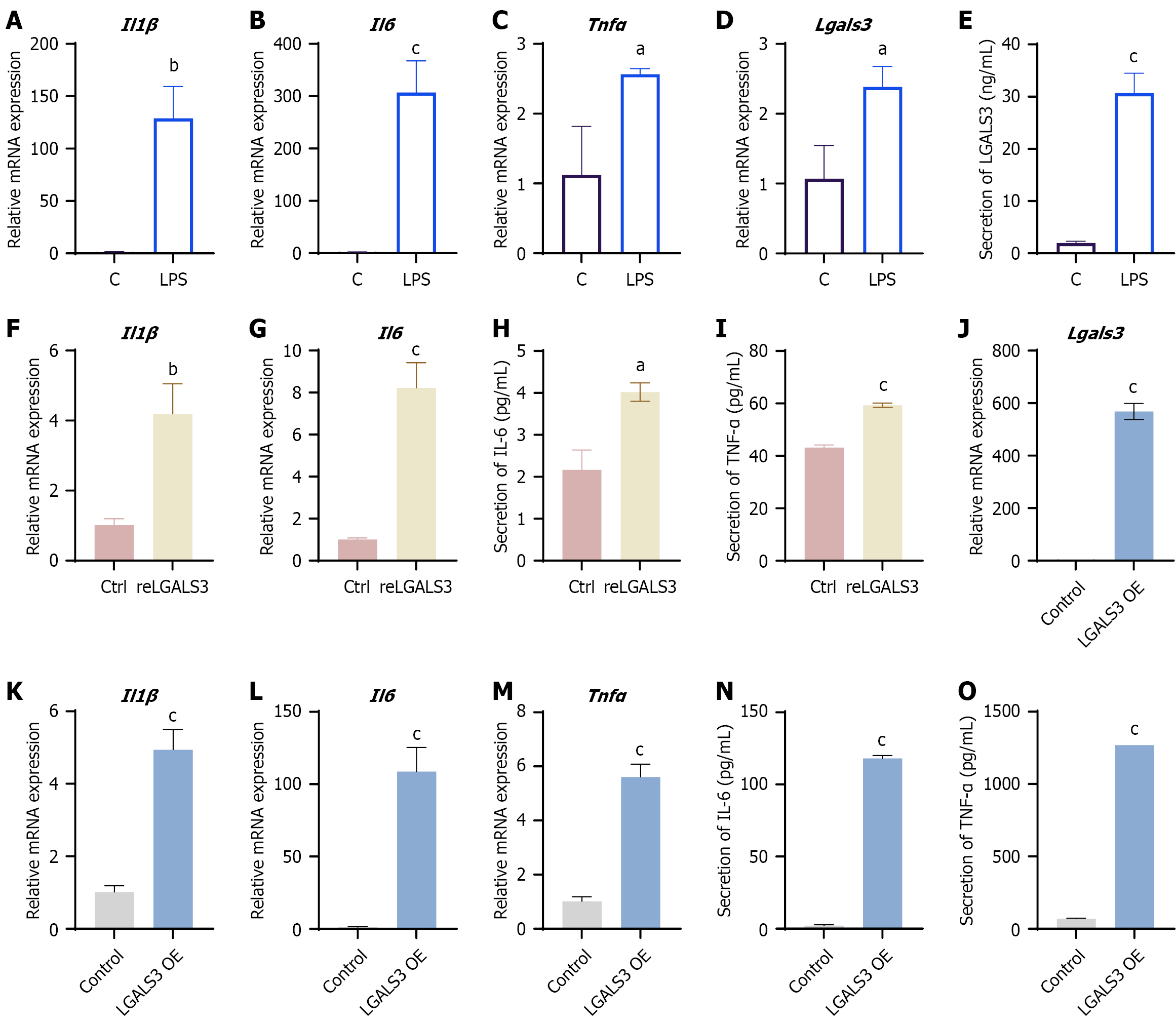
To explore the functional involvement of LGALS3 in inflammation modulation within macrophages, an inflammatory environment was generated through lipopolysaccharide (LPS) (100 ng/mL) stimulation of RAW264.7 cells for 24 hours. Analysis indicated that relative to the control group, cells treated with LPS showed markedly elevated mRNA expression of the pro-inflammatory mediators TNF-α, IL-6, and IL-1β (Figure 5A-C). Additionally, LGALS3 level was found to be substantially increased in the LPS-stimulated RAW264.7 cells (Figure 5D). This study also examined its secretory properties in macrophages. The results showed that following 24 hours of LPS stimulation, the secretion level of LGALS3 was significantly increased compared to the control group (Figure 5E). To further investigate the pro-inflammatory role of LGALS3, RAW264.7 cells were treated with recombinant LGALS3 protein for 24 hours. The results indicated that intervention with LGALS3 recombinant protein significantly elevated the mRNA levels of IL-1β and IL-6 in RAW264.7 cells (Figure 5F and G). ELISA assays also revealed that after LGALS3 recombinant protein intervention, the secretion levels of IL-6 and TNF-α in RAW264.7 cells were markedly increased (Figure 5H and I). A RAW264.7 cell line with overexpressing LGALS3 was successfully generated (Figure 5J). Subsequent real-time quantitative PCR analysis revealed that overexpression of LGALS3 led to a marked upregulation in the mRNA expression levels of inflammatory cytokines (IL-1β, IL-6 and TNF-α) relative to the control group (Figure 5K-M). ELISA results further demonstrated that the LGALS3 overexpression showed significantly elevated secretion levels of IL-6 and TNF-α (Figure 5N and O). Therefore, LGALS3 exerts a pro-inflammatory effect.
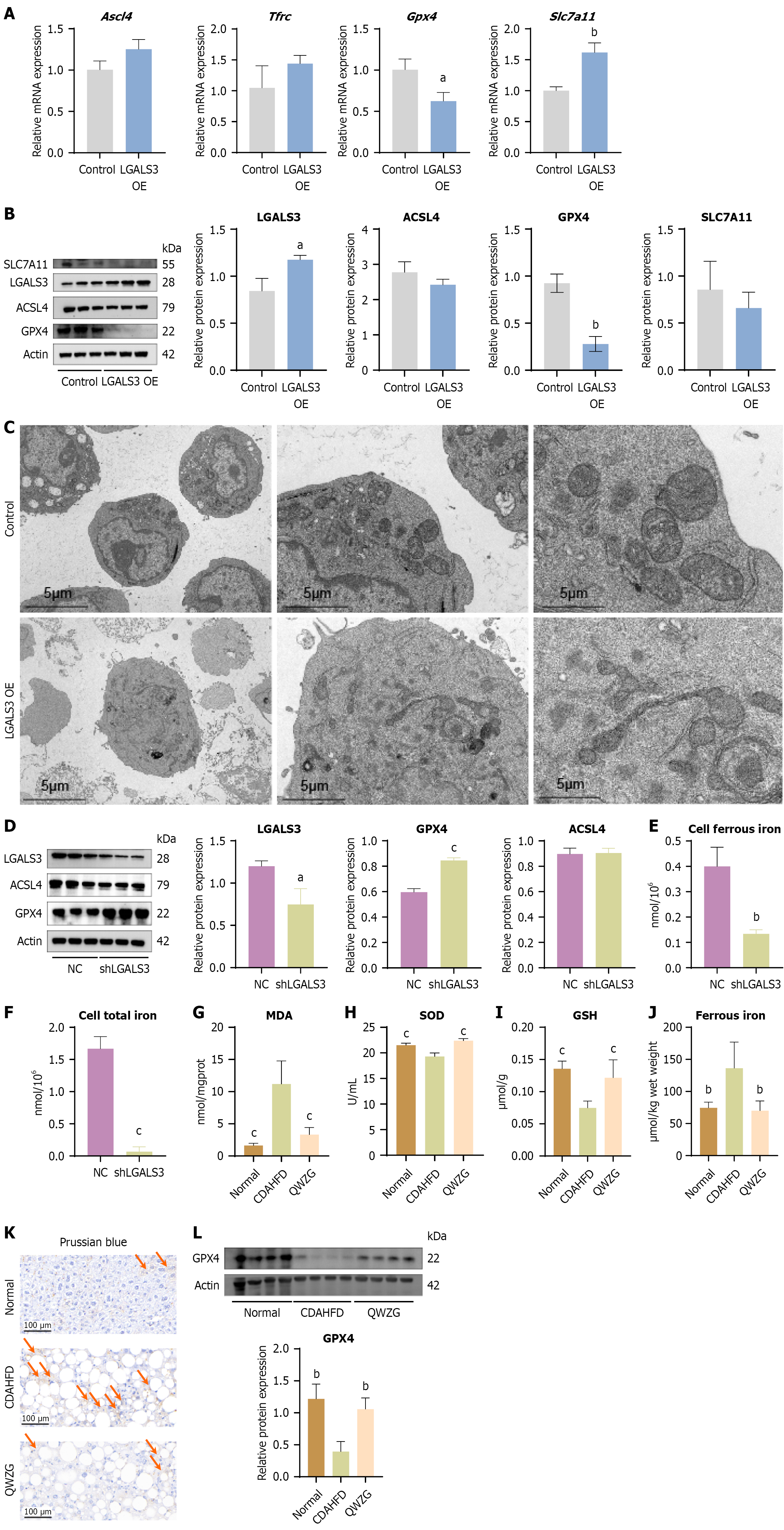
Current findings reveal that LGALS3 exacerbates inflammatory pathways while simultaneously driving ferroptosis via dysregulation of iron metabolic processes. To further investigate the role of LGALS3 in ferroptosis, this study examined key genes involved in the ferroptosis, including Achaete-scute family bHLH transcription factor 4, TFRC, GPX4, solute carrier family 7 member 11. The results indicated that LGALS3 overexpression significantly downregulated the mRNA level of GPX4 (Figure 6A). A consistent conclusion was reached at the protein level (Figure 6B). Furthermore, trans
Studies have demonstrated that LGALS3 could regulate the inflammation by mediating TRAF6 expression[24-26]. Studies have also revealed that TRAF6 regulates the expression of NLRP3, which is closely associated with macrophage-mediated inflammatory responses[27,28]. Furthermore, current research also indicates that TRAF6 is closely associated with GPX4. TRAF6 mediates the ubiquitination of GPX4 and enhances its recognition by p62, resulting in subsequent degradation[29].
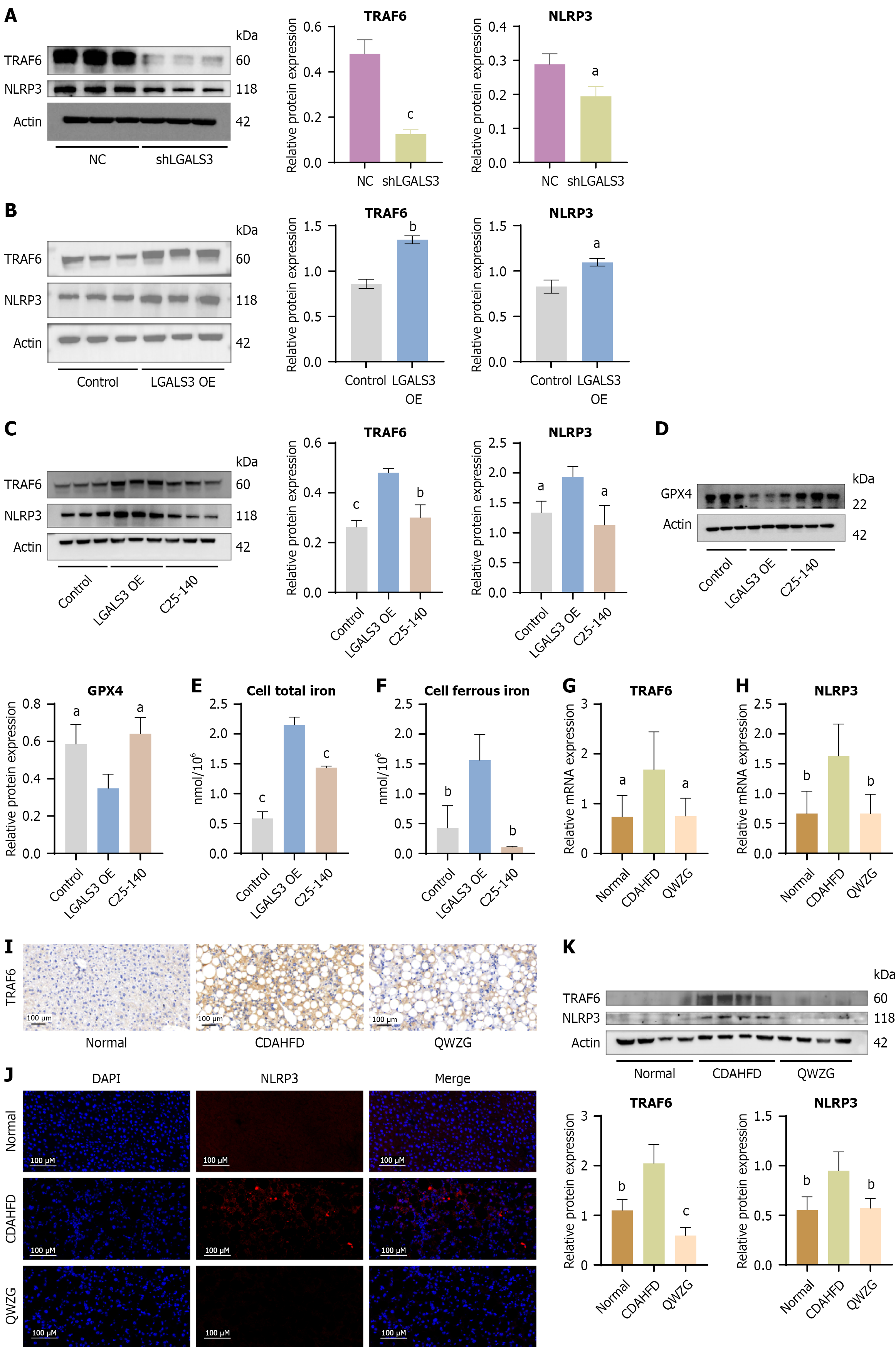
This research further explored the molecular mechanism, which LGALS3 modulates the development of MASH. LGALS3 knockdown indicated a substantial reduction in TRAF6 and NLRP3 protein levels (Figure 7A). Conversely, LGALS3 overexpression significantly upregulated the levels of TRAF6 and NLRP3 (Figure 7B). Then TRAF6 inhibitor C25-140 (HY-120934, MCE, 20 μM) was applied in RAW264.7 cells overexpressing LGALS3 for 24 hours. The results demonstrated that inhibition of TRAF6 effectively reversed the upregulation of NLRP3 and the downregulation of GPX4induced by LGALS3 overexpression (Figure 7C and D). Measurements of ferrous iron (Fe2+) and total iron levels revealed that LGALS3 overexpression significantly increased their content in RAW264.7 cells. Conversely, inhibition of TRAF6 markedly reduced their levels (Figure 7E and F). Finally, the results indicated that QWZG suppressed the elevated mRNA expression of TRAF6 and NLRP3 induced by CDAHFD in the livers of mice with MASH (Figure 7G and H). Their protein levels were consistently reduced after QWZG treatment (Figure 7I-K). Therefore, these findings suggest that QWZG ameliorates MASH by modulating the LGALS3/TRAF6/GPX4 axis.
MASH is a multifactorial disorder with a pathogenesis that remains incompletely elucidated. The clinical landscape of MASH is characterized by substantial patients, yet therapeutic options remain severely limited, creating an urgent for effective pharmacotherapies. TCM formulations have broad application value in the treatment of MASH. This study identified QWZG ameliorated MASH by modulating the LGALS3/TRAF6/GPX4 axis. This work represented the first exploration into the mechanism underlying the efficacy of QWZG.
In this study, our results showed that the QWZG significantly ameliorated hepatic steatosis, inflammation, and fibrosis in mice with MASH. Transcriptomic analysis identified LGALS3 as a key gene. LGALS3 expression was significantly upregulated in MASH mice, then was effectively reversed by QWZG administration. Current research indicated that LGALS3 was closely associated with MASH[18,30,31]. The level of LGALS3 was markedly increased in patients with MASH compared to hepatic steatosis[31]. LGALS3 inhibition could decrease accumulation of hepatic lipoxidation products, inhibit inflammation and fibrosis, to protect from diet-induced MASH[32]. In addition, LGALS3 could be associated with liver fibrosis, and promote the progression of liver fibrosis in MASLD[30]. LGALS3-binding protein (LGALS3BP) was significantly increased in the patients with MASH, whereas deletion of LGALS3BP in HPs inhibits transforming growth factor-beta 1 and ameliorates CCl4-induced fibrosis. Furthermore, depletion of LGALS3BP delays hepatocellular carcinogenesis by downregulating the transforming growth factor-beta 1[33]. A study has demonstrated that LGALS3 inhibitor selvigaltin could reduce liver inflammation and fibrosis in rabbit with MASH[34]. Isoquercitrin could inhibit the level of LGALS3, to regulate insulin resistance and lipid metabolism, finally ameliorating MASH[35]. In this study, our results confirmed that intervention with recombinant LGALS3 protein promoted pro-inflammatory response in RAW264.7 cells. In addition, LGALS3 overexpression significantly promoted inflammatory response. These results validate the important function of LGALS3 in modulating inflammatory response.
Newly recognized modes of cell death, including ferroptosis, cuproptosis and pyroptosis, have attracted growing interest and play important roles in MASH[36]. Neutrophil cytosolic factor 1 could promote iron overload of KCs, to aggravate MASH progression[37]. The novel iron chelator FerroTerminator1 could effectively reverse liver injury and inflammation in multiple MASH models[38], demonstrating that targeting ferroptosis for the treatment of MASH is an effective strategy. Furthermore, emerging evidence suggests that LGALS3 mediates ferroptosis[39]. In this study, we established LGALS3-knockdown and -overexpressing RAW264.7 cell lines, and results indicated that LGALS3 overexpression exacerbated mitochondrial damage, and reduced the level of GPX4, whereas LGALS3 knockdown reduced the levels of total iron and ferrous iron, and increased the level of GPX4, indicating that LGALS3 could regulate ferroptosis. Furthermore, QWZG intervention also reduced the level of ferrous iron (Fe2+), and markedly upregulated GPX4 level. Therefore, our results have confirmed the association between LGALS3 and ferroptosis in MASH, and QWZG may improve MASH by inhibiting LGALS3-mediated ferroptosis. Furthermore, studies have shown that LGALS3 could interact with TRAF6 to promote inflammation[25,26], while TRAF6 could activate macrophage by regulating NLRP3[27,28]. In addition, studies have demonstrated that TRAF6 facilitates GPX4 degradation and induces ferroptosis[40]. These results demonstrated that LGALS3 may regulate ferroptosis by mediating TRAF6-NLRP3 axis. In the study, our results indicated that LGALS3 knockdown significantly reduced the levels of TRAF6 and NLRP3, whereas LGALS3 overexpression significantly promoted their levels. However, TRAF6 inhibitor reversed the effect of LGALS3 on ferroptosis. Finally, our results also showed that QWZG significantly reduced the levels of TRAF6 and NLRP3. Therefore, this study innovatively investigated the pathogenesis of MASH from the perspective of ferroptosis and elucidated the mechanistic basis of QWZG. The research identified LGALS3 as a key regulatory factor that markedly promoted ferroptosis and exacerbated hepatic inflammatory responses. This work was the first to reveal the role of the LGALS3/TRAF6/GPX4 axis in MASH pathogenesis, providing novel evidence that QWZG alleviated MASH. These findings also offer new theoretical support and potential therapeutic targets for the prevention and treatment of MASH with TCM.
In summary, this study confirmed that QWZG could alleviate the progression of MASH by inhibiting LGALS3. Mechanistically, LGALS3 induced macrophage inflammation via the TRAF6/NLRP3 axis-mediated ferroptosis. Therefore, QWZG ameliorated the progression of MASH by modulating the LGALS3/TRAF6/GPX4 axis. These findings suggest that LGALS3 may serve as a potential therapeutic target for MASH, and QWZG was an effective compound for treating MASH.
| 1. | Raza S, Rajak S, Upadhyay A, Tewari A, Anthony Sinha R. Current treatment paradigms and emerging therapies for NAFLD/NASH. Front Biosci (Landmark Ed). 2021;26:206-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 273] [Cited by in RCA: 235] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 2. | Noureddin M. MASH clinical trials and drugs pipeline: An impending tsunami. Hepatology. 2025;82:1325-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 3. | Xu X, Poulsen KL, Wu L, Liu S, Miyata T, Song Q, Wei Q, Zhao C, Lin C, Yang J. Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH). Signal Transduct Target Ther. 2022;7:287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 292] [Cited by in RCA: 275] [Article Influence: 68.8] [Reference Citation Analysis (4)] |
| 4. | Younossi Z, Anstee QM, Marietti M, Hardy T, Henry L, Eslam M, George J, Bugianesi E. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2018;15:11-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4585] [Cited by in RCA: 4100] [Article Influence: 512.5] [Reference Citation Analysis (5)] |
| 5. | Sumida Y, Yoneda M. Current and future pharmacological therapies for NAFLD/NASH. J Gastroenterol. 2018;53:362-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 579] [Cited by in RCA: 539] [Article Influence: 67.4] [Reference Citation Analysis (2)] |
| 6. | Zhang C, Yang M. Current Options and Future Directions for NAFLD and NASH Treatment. Int J Mol Sci. 2021;22:7571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (1)] |
| 7. | Ledford H. First US drug approved for a liver disease surging around the world. Nature. 2024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 8. | Petta S, Kim K, Targher G, Romeo S, Sookoian S, Zheng MH, Aghemo A, Valenti L. Focus on Semaglutide 2.4 mg/week for the Treatment of Metabolic Dysfunction-Associated Steatohepatitis. Liver Int. 2025;45:e70407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 9. | Dang Y, Xu J, Yang Y, Li C, Zhang Q, Zhou W, Zhang L, Ji G. Ling-gui-zhu-gan decoction alleviates hepatic steatosis through SOCS2 modification by N6-methyladenosine. Biomed Pharmacother. 2020;127:109976. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Ye Z, Zhao Y, Cui Y, Xu B, Wang F, Zhao D, Dong G, Wang Z, Wu R. Ling-gui-zhu-gan promotes adipocytes browning via targeting the miR-27b/PRDM16 pathway in 3T3-L1 cells. Front Pharmacol. 2024;15:1386794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Li Y, Ye Z, Zhao Y, Xu B, Xue W, Wang Z, An R, Wang F, Wu R. Ling-gui-zhu-gan granules reduces obesity and ameliorates metabolic disorders by inducing white adipose tissue browning in obese mice. Front Physiol. 2024;15:1427722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Lan T, Xu T, Fu Y, Jiang S, Liang X, Yu Z, Pan L, Rong X, Guo J. Fufang Zhenzhu Tiaozhi Capsule Prevents Intestinal Inflammation and Barrier Disruption in Mice With Non-Alcoholic Steatohepatitis. Front Endocrinol (Lausanne). 2022;13:864703. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Wu J, Xian S, Zhang S, Yang Y, Pan J, Zhou W, Hu D, Ji G, Dang Y. Gan-Jiang-Ling-Zhu decoction improves steatohepatitis induced by choline-deficient-high-fat-diet through the METTL14/N6-methyladenosine-mediated Ugt2a3 expression. J Ethnopharmacol. 2025;339:119153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 14. | Cao Y, Shu X, Li M, Yu S, Li C, Ji G, Zhang L. Jiangzhi granule attenuates non-alcoholic steatohepatitis through modulating bile acid in mice fed high-fat vitamin D deficiency diet. Biomed Pharmacother. 2022;149:112825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Anila MM, Rogowski P, Różycki B. Scrutinising the Conformational Ensemble of the Intrinsically Mixed-Folded Protein Galectin-3. Molecules. 2024;29:2768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Li H, Cao Z, Wang L, Li J, Cheng X, Tang Y, Xing M, Yao P. Chronic high-fat diet induces galectin-3 and TLR4 to activate NLRP3 inflammasome in NASH. J Nutr Biochem. 2023;112:109217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 17. | An L, Chang G, Zhang L, Wang P, Gao W, Li X. Pectin: Health-promoting properties as a natural galectin-3 inhibitor. Glycoconj J. 2024;41:93-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 18. | Bouffette S, Botez I, De Ceuninck F. Targeting galectin-3 in inflammatory and fibrotic diseases. Trends Pharmacol Sci. 2023;44:519-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 114] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 19. | Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6378] [Cited by in RCA: 5757] [Article Influence: 1151.4] [Reference Citation Analysis (9)] |
| 20. | Warasnhe K, Sano A, Guo Q, Satthawiwat N, Dai X, Classon PA, Meroueh C, Kim Lee HS, Valenzuela-Pérez L, Victorelli S, Bamidele AO, Hirsova P, Povero D, Ibrahim SH. Glycogen Synthase Kinase 3 β Hepatocyte Deletion Attenuates Ferroptosis and Metabolic Dysfunction-associated Steatohepatitis in Mice. Cell Mol Gastroenterol Hepatol. 2026;20:101633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 21. | Liu Y, Zhang M, Zhu C, Zhang Y, Huang Y, Xia S, Liao J. FATP5 deficiency alleviates MASH via remodeling hepatic lipid composition to suppress ferroptosis. Free Radic Biol Med. 2025;240:170-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Qiu YY, Zhang J, Zeng FY, Zhu YZ. Roles of the peroxisome proliferator-activated receptors (PPARs) in the pathogenesis of nonalcoholic fatty liver disease (NAFLD). Pharmacol Res. 2023;192:106786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 185] [Article Influence: 61.7] [Reference Citation Analysis (0)] |
| 23. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A, Yeh M, McCullough AJ, Sanyal AJ; Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9231] [Cited by in RCA: 8614] [Article Influence: 410.2] [Reference Citation Analysis (7)] |
| 24. | Ghanta P, Hessel E, Arias-Alvarado A, Aghayev M, Ilchenko S, Kasumov T, Oyewumi MO. Lung cancer exosomal Gal3BP promotes osteoclastogenesis with potential connotation in osteolytic metastasis. Sci Rep. 2024;14:27201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 25. | Xu G, Xia Z, Deng F, Liu L, Wang Q, Yu Y, Wang F, Zhu C, Liu W, Cheng Z, Zhu Y, Zhou L, Zhang Y, Lu M, Liu S. Inducible LGALS3BP/90K activates antiviral innate immune responses by targeting TRAF6 and TRAF3 complex. PLoS Pathog. 2019;15:e1008002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 62] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 26. | Simon D, Derer A, Andes FT, Lezuo P, Bozec A, Schett G, Herrmann M, Harre U. Galectin-3 as a novel regulator of osteoblast-osteoclast interaction and bone homeostasis. Bone. 2017;105:35-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 27. | Cai H, Li H, Xiao X, Wang S, Liu R, Qin Y, Zhou Y, Yao C. TRAF6 promotes abdominal aortic aneurysm development by activating macrophage pyroptosis via the NLRP3/Caspase1/GSDMD pathway. FASEB J. 2025;39:e70318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 28. | Li Y, Yang Y, Xia D, Fang Y, Tang C, Yu J, Geng D, Wang Z, Xiao L. Nrf2/Nlrp3 signaling in aging BMSCs: Traf6 intervention as a novel approach to osteoporosis treatment. Redox Biol. 2025;86:103804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Gong R, Wan X, Jiang S, Guan Y, Li Y, Jiang T, Chen Z, Zhong C, He L, Xiang Z, Yang J, Xu B, Yang J, Cheng Y. GPX4-AUTAC induces ferroptosis in breast cancer by promoting the selective autophagic degradation of GPX4 mediated by TRAF6-p62. Cell Death Differ. 2025;32:2022-2037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 30. | Sotoudeheian M. Galectin-3 and Severity of Liver Fibrosis in Metabolic Dysfunction-Associated Fatty Liver Disease. Protein Pept Lett. 2024;31:290-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 31. | Azevedo Foinquinos G, Azevedo Acioli ME, Santana Cavalcanti AH, Barbosa Junior WL, Lima RE, Juca NT, de Azevedo Foinquinos RC, Rocha da Cruz C, Fernandez Pereira FM, de Carvalho SR, de Mendonça Belmont TF, Vasconcelos LRS, Beltrão Pereira LMM. Influence of LGALS3 and PNPLA3 genes in non-alcoholic steatohepatitis (NASH) in patients undergone bariatric surgery. Obes Res Clin Pract. 2020;14:326-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 32. | Iacobini C, Menini S, Ricci C, Blasetti Fantauzzi C, Scipioni A, Salvi L, Cordone S, Delucchi F, Serino M, Federici M, Pricci F, Pugliese G. Galectin-3 ablation protects mice from diet-induced NASH: a major scavenging role for galectin-3 in liver. J Hepatol. 2011;54:975-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (2)] |
| 33. | Kim DH, Sung M, Park MS, Sun EG, Yoon S, Yoo KH, Radhakrishnan K, Jung SY, Bae WK, Cho SH, Chung IJ. Galectin 3-binding protein (LGALS3BP) depletion attenuates hepatic fibrosis by reducing transforming growth factor-β1 (TGF-β1) availability and inhibits hepatocarcinogenesis. Cancer Commun (Lond). 2024;44:1106-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 34. | Comeglio P, Guarnieri G, Filippi S, Cellai I, Acciai G, Holyer I, Zetterberg F, Leffler H, Kahl-Knutson B, Sarchielli E, Morelli A, Maggi M, Slack RJ, Vignozzi L. The galectin-3 inhibitor selvigaltin reduces liver inflammation and fibrosis in a high fat diet rabbit model of metabolic-associated steatohepatitis. Front Pharmacol. 2024;15:1430109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 35. | Jin HL, Feng XY, Feng SL, Dai L, Zhu WT, Yuan ZW. Isoquercitrin attenuates the progression of non-alcoholic steatohepatitis in mice by modulating galectin-3-mediated insulin resistance and lipid metabolism. Phytomedicine. 2024;123:155188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 36. | Yang Y, Wu J, Wang L, Ji G, Dang Y. Copper homeostasis and cuproptosis in health and disease. MedComm (2020). 2024;5:e724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 55] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 37. | Zhang J, Wang Y, Fan M, Guan Y, Zhang W, Huang F, Zhang Z, Li X, Yuan B, Liu W, Geng M, Li X, Xu J, Jiang C, Zhao W, Ye F, Zhu W, Meng L, Lu S, Holmdahl R. Reactive oxygen species regulation by NCF1 governs ferroptosis susceptibility of Kupffer cells to MASH. Cell Metab. 2024;36:1745-1763.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 51] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 38. | Tao L, Yang X, Ge C, Zhang P, He W, Xu X, Li X, Chen W, Yu Y, Zhang H, Chen SD, Pan XY, Su Y, Xu C, Yu Y, Zheng MH, Min J, Wang F. Integrative clinical and preclinical studies identify FerroTerminator1 as a potent therapeutic drug for MASH. Cell Metab. 2024;36:2190-2206.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 66] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 39. | Chen L, Ye Z, Yang S, Xie J, Li H, Zhou X, Cheng F. RNA-Binding Protein Lgals3 , Ferroptosis, and Acute Kidney Injury. J Am Soc Nephrol. 2025;36:1249-1263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 40. | Shi Y, Yang X, Jiang H, Wu S, Hong Y, Su W, Wang X. Alpinia officinarum Hance extract relieved sepsis-induced myocardial ferroptosis and inflammation by inhibiting lncRNA MIAT/TRAF6/NF-κB axis. Allergol Immunopathol (Madr). 2024;52:21-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |