Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.116415
Revised: December 23, 2025
Accepted: February 3, 2026
Published online: April 14, 2026
Processing time: 138 Days and 22.9 Hours
Extramural vascular invasion (EMVI) represents a crucial high-risk pathological feature in colon cancer, strongly linked to lymph node involvement, distant dissemination, and local recurrence. Advanced analytical techniques such as ma
To develop and validate interpretable DFS prediction models for colon cancer by combining multiple machine-learning algorithms with a nomogram framework based on ctEMVI.
In this retrospective analysis, comprehensive clinical, radiological, and pathological information was collected from 101 patients who underwent curative resection for colon cancer. Based on findings from preoperative contrast-enhanced CT, patients were stratified into ctEMVI-positive and ctEMVI-negative groups. DFS was estimated using Kaplan-Meier methods and compared with log-rank tests. Prognostic variables independently associated with DFS were identified through Cox proportional hazards regression and subsequently integrated into machine-learning-based predictive models and a nomogram framework. The discriminative ability, calibration accuracy, clinical utility, and interpretability of these models were systematically evaluated using receiver operating characteristic curves, calibration plots, decision curve analysis, and SHapley Additive exPlanations.
Among 101 patients, 40 were ctEMVI-positive and 61 ctEMVI-negative. The ctEMVI was significantly associated with T staging determined by CT, lymph node status determined by CT, pathological T stage, pathological nodal status, lymph node ratio, lymphovascular invasion, and perineural invasion (all P < 0.05). During follow-up, 29 patients experienced recurrence or metastasis, including 19 in the ctEMVI-positive group, which showed signi
The ctEMVI was a significant prognostic factor in colon cancer, and CoxBoost and nomogram models accurately predicted DFS after curative resection.
Core Tip: Extramural vascular invasion represents a well-established determinant of prognosis in colon cancer; however, its reliable evaluation before surgery remains clinically challenging. The present study provided evidence that computed tomography-detected extramural vascular invasion (ctEMVI) functioned as an independent predictor of disease-free survival following curative resection. By incorporating ctEMVI with conventional clinicopathological factors, we developed interpretable machine-learning-based prediction models and a nomogram to enhance individualized preoperative risk assessment. Among the evaluated algorithms, CoxBoost demonstrated the strongest predictive capability. Collectively, these results underscored the clinical utility of ctEMVI-driven modeling approaches for refining preoperative risk stratification in patients with colon cancer.
- Citation: Wang MR, Zheng LF, Yang F, Gu XY, Yang JS, Chen FX, Liu JM, He BS. Development and validation of prognostic models for colon cancer incorporating extramural vascular invasion assessed by contrast-enhanced computed tomography. World J Gastroenterol 2026; 32(14): 116415
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/116415.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.116415
According to global cancer statistics released in 2023, colorectal cancer (CRC) ranks as the second most common cause of cancer-related mortality worldwide[1]. Alarmingly, among men under the age of 50, CRC has emerged as the leading contributor to cancer deaths, accounting for nearly 34.3% of all cases in this age group[1]. Surgical resection remains the cornerstone of curative therapy, often complemented by systemic treatments such as immunotherapy, targeted therapy, and chemotherapy[2]. Accurate preoperative tumor assessment is therefore critical for guiding therapeutic decision-making and predicting clinical outcomes[3].
Extramural vascular invasion (EMVI) refers to the extension of malignant cells into venous vessels beyond the muscularis propria of the intestinal wall[4]. Accumulating evidence has identified EMVI as a major high-risk feature in colon cancer, closely associated with lymphatic dissemination, distant metastasis, and local tumor recurrence[5-7]. In rectal cancer, high-resolution magnetic resonance imaging (MRI) is routinely employed for EMVI evaluation[8,9]; however, its utility in colon cancer remains restricted due to physiological bowel motion and respiratory artifacts. Conversely, computed tomography (CT) has become the principal imaging tool for preoperative staging of colon cancer. Recent investigations have demonstrated that CT-detected EMVI (ctEMVI) can be reliably identified prior to surgery and serves as an independent predictor of postoperative recurrence. Moreover, when combined with conventional TNM staging, ctEMVI enables more accurate identification of patients at high risk of disease relapse[10].
In recent years, remarkable progress in the assessment of tumor metastasis and prognosis has been achieved through the application of machine-learning techniques[11]. As an intuitive multivariate analytical framework, a nomogram integrates diverse prognostic variables into a single graphical model, thereby allowing clinicians to quantitatively esti
Building upon these advances, the present study sought to elucidate the prognostic significance of ctEMVI in colon cancer. By integrating multiple machine-learning algorithms with a nomogram-based approach, we aimed to develop and validate predictive models capable of supporting individualized treatment planning and timely clinical decision-making for patients undergoing surgery for colon cancer.
This retrospective study included patients diagnosed with colon cancer who received treatment at the Department of Gastroenterology, Nantong First People’s Hospital, between April 2019 and April 2020. Eligible participants met the following inclusion criteria: (1) Completion of a preoperative contrast-enhanced and non-enhanced abdominal CT scan; (2) Underwent radical resection of CRC confirmed by postoperative histopathology, without severe postoperative compli
Patients were excluded if they (1) Had undergone prior abdominal surgery; (2) Had a concurrent malignancy in another organ; and (3) Presented with preoperative distant metastasis. Demographic and clinical data, including sex, age, carcinoembryonic antigen (CEA), and carbohydrate antigen 19-9 (CA19-9), were collected for all included cases. The study was reviewed and approved by the Ethics Committee of Nantong First People’s Hospital (No. 2025-KT286-03).
All patients fasted for 4-8 hours prior to examination and ingested 2000 mL of isotonic mannitol 1 hour before scanning to achieve adequate bowel distension. CT imaging was performed from the diaphragm to the inferior margin of the pubic symphysis using a Somatom Force scanner (Siemens Healthineers, Germany) with the following acquisition parameters: (1) Tube A, 90 kV and 144 mAs; (2) Tube B, 150 kV and 90 mAs; (3) Collimation, 2 mm × 192 mm × 0.6 mm; (4) Pitch, 1.0; (5) Rotation time, 0.5 seconds; and (6) Linear fusion coefficient, 0.5.
Contrast enhancement was achieved via intravenous administration of iopromide (Ultravist 370, Bayer, Germany) through the antecubital vein at a dose of 1.5 mL/kg and an injection rate of 4 mL/second, followed by a 40 mL saline flush. The arterial phase was triggered when the iodine concentration in the aorta reached 100 Hounsfield units, followed by venous and delayed phases at 40 seconds and 80 seconds, respectively. These sequential acquisitions were used to assess tumor vascularity and depth of invasion.
Contrast-enhanced CT was employed to accurately delineate the location, size, and extent of colon tumors, as well as to assess local invasion and involvement of adjacent structures. The modality also facilitated the detection of regional lymph node involvement and distant metastases, thereby informing clinical management strategies. All images were independently reviewed by two senior radiologists, with discrepancies resolved through consensus discussion.
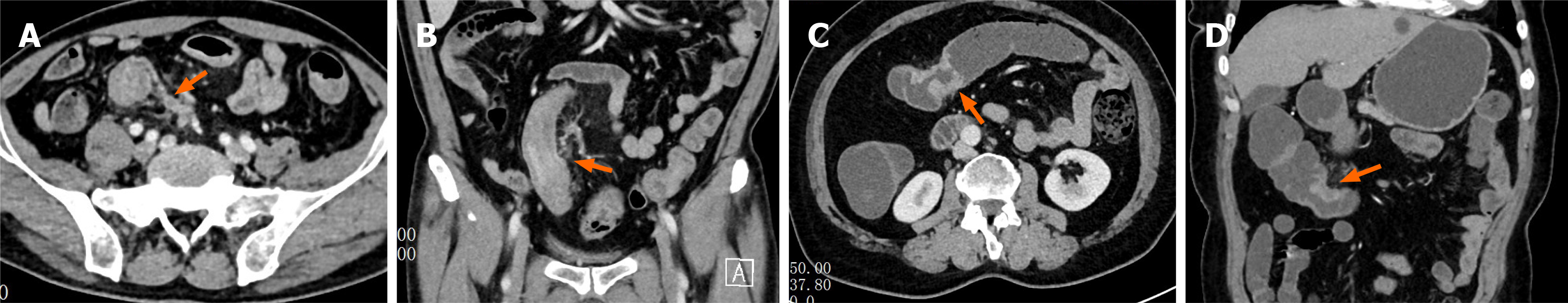
The evaluation of ctEMVI was primarily grounded in the assessment of morphological alterations in mesenteric vessels adjacent to the tumor[12]. Specifically, a lesion was categorized as ctEMVI-positive when at least one of the following radiologic criteria was identified: (1) Irregularity of the vascular wall, characterized by nodular, serrated, or spiculated contours with loss of the normal smooth margin; (2) Abnormal changes in the vascular lumen, including focal or irregular dilatation exceeding the caliber of the corresponding normal vessel at the same anatomical level; and (3) Abnormal vascular density or trajectory, manifested as intraluminal filling defects, rigid or distorted vessel course, circumferential tumor encasement, or abrupt vessel interruption (Figure 1A and B). Tumors lacking all of these imaging features were classified as ctEMVI-negative (Figure 1C and D).
Other tumor indicators were conducted according to the following criteria: (1) Tumor location: The colon was seg
Postoperative follow-up was conducted through outpatient consultations or telephone interviews to document recur
Independent prognostic factors for DFS were identified using multivariate Cox regression analysis. Based on these variables, a nomogram and five machine-learning models were constructed, including XGBoost, random survival forest (RSF), CoxBoost, gradient boosting machine (GBM), and least absolute shrinkage and selection operator (LASSO)-Cox.
Given the modest sample size (n = 101), a predefined training-testing partition was not implemented. Instead, internal validation was conducted using leave-one-out cross-validation (LOOCV), whereby a single patient was sequentially held out as the testing set while the remaining patients served as the training cohort in each iteration. This procedure was repeated across all individuals (n = 101), and model performance indices were subsequently aggregated to obtain averaged estimates.
To enhance predictive accuracy (ACC), hyperparameter optimization for the RSF, XGBoost, GBM, and CoxBoost models was carried out using a grid search strategy. Each candidate hyperparameter configuration was evaluated within the LOOCV framework, with performance quantified by the time-dependent area under the receiver operating characteristic (ROC) curve (AUC). The hyperparameter combination achieving the optimal performance was selected for final model construction. Comprehensive details of the hyperparameter search space and the finalized parameter settings are summarized in Supplementary Table 1.
Among the five machine-learning models, the model demonstrating the highest time-dependent AUC value was selected for direct comparison with the nomogram. Comprehensive model evaluation included ROC analysis, AUC, ACC, precision, F1-score, recall, calibration curves, and decision curve analysis (DCA). SHapley Additive exPlanations (SHAP) were applied to interpret machine-learning model predictions and quantify the relative contribution of each feature to DFS outcomes.
All statistical analyses were performed using SPSS (version 26.0) and R (version 4.4.1). Continuous variables were first assessed for normality. Normally distributed data were presented as mean ± SD, while non-normally distributed variables were expressed as median with interquartile range. Categorical variables are presented as percentage with 95%CI. Between-group comparisons for continuous variables were conducted using independent-sample t-tests, whereas categorical variables were analyzed using the χ2 test.
All dichotomous variables were encoded to reflect increasing risk, with lymphovascular invasion (LVI), perineural invasion (PNI), pathological nodal status (pN), and ctEMVI assigned a value of 1 to indicate presence and 0 to denote absence. Survival analyses were performed using the Kaplan-Meier method, with differences between groups evaluated by log-rank tests. Cox proportional hazards regression was employed to identify independent predictors of DFS. A two-sided P value of < 0.05 was considered statistically significant.
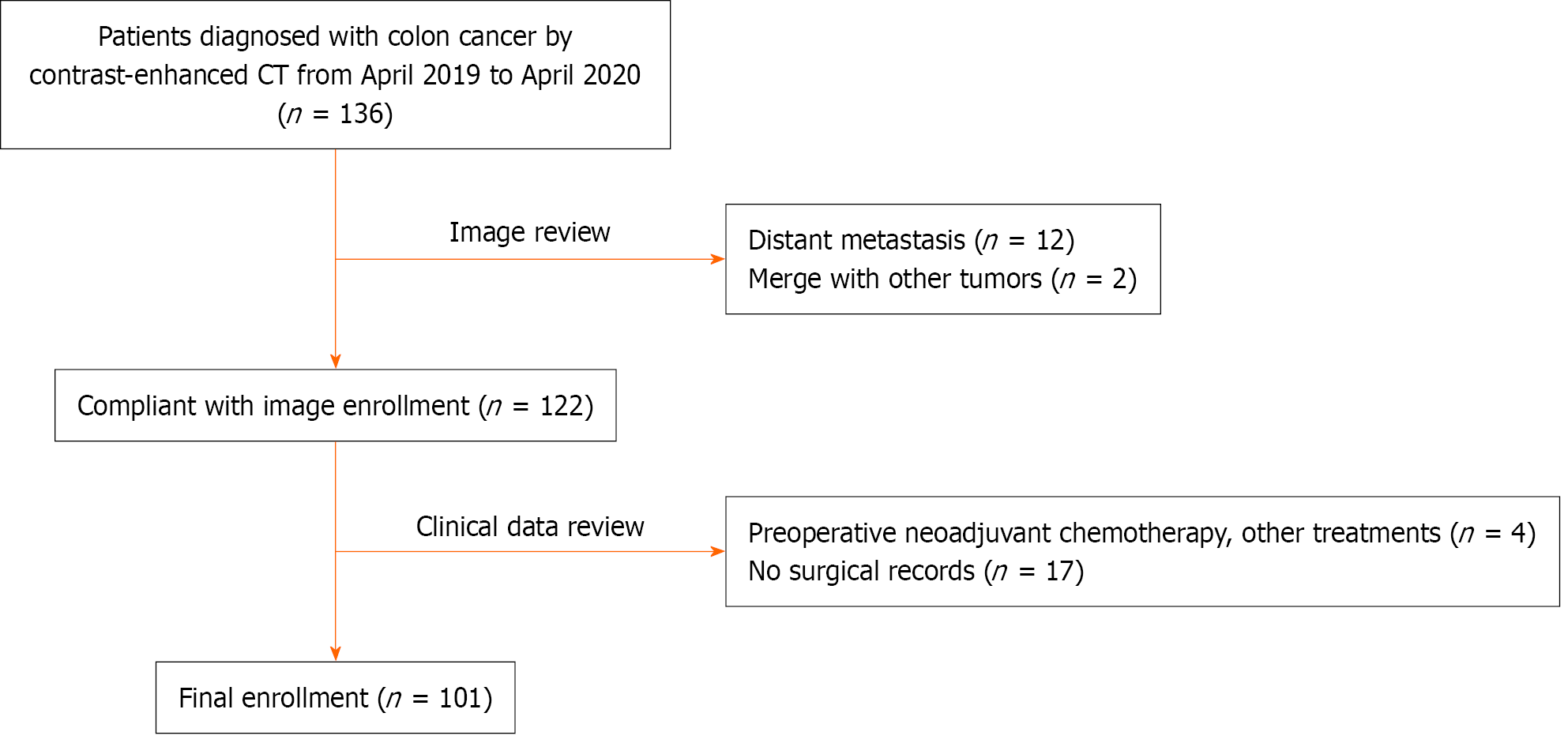
A total of 101 patients (59 men and 42 women) met the predefined inclusion and exclusion criteria and were included in the final analysis (Figure 2). Their ages ranged from 30 years to 93 years, with a mean of 65.87 ± 11.12 years. Of these, 40 patients were classified as ctEMVI-positive and 61 patients as ctEMVI-negative.
The diagnostic assessments by the two senior radiologists demonstrated substantial concordance (kappa = 0.76, P < 0.001), reflecting high interobserver reliability. Correlation analyses revealed that ctEMVI status was significantly associated with multiple clinicopathological parameters, including ctT, ctN, pathological T stage, pathological pN, lymph node ratio, LVI, and PNI (all P < 0.05). In contrast, no significant relationships were observed between ctEMVI and sex, age, CEA, CA19-9, tumor location, tumor size, or histological subtype (P > 0.05; Table 1).
| Factors | ctEMVI negative | ctEMVI positive | t/χ2 | P value | |
| Gender | Male | 35 (59.3) | 24 (40.7) | 0.068 | 0.794 |
| Female | 26 (61.9) | 16 (38.1) | |||
| Age (year) | 66.64 ± 10.95 | 64.70 ± 11.41 | 0.856 | 0.394 | |
| Carcinoembryonic antigen | Normal | 52 (64.2) | 29 (35.8) | 2.471 | 0.116 |
| Increase | 9 (45) | 11 (55) | |||
| Carbohydrate antigen 19-9 | Normal | 52 (61.2) | 33 (38.8) | 0.137 | 0.712 |
| Increase | 9 (56.2) | 7 (43.8) | |||
| Tumor site | Left colon | 33 (63.5) | 19 (36.5) | 0.421 | 0.516 |
| Right colon | 28 (57.1) | 21 (42.9) | |||
| Tumor size | < 5 cm | 48 (60.8) | 31 (39.2) | 0.020 | 0.887 |
| ≥ 5 cm | 13 (59.1) | 9 (40.9) | |||
| T staging determined by CT | T1-2 | 14 (100) | 0 | 10.658 | 0.001 |
| T3-4 | 47 (54) | 40 (46) | |||
| Lymph node status determined by CT | Negative | 39 (83) | 8 (17) | 18.743 | < 0.001 |
| Positive | 22 (40.7) | 32 (59.3) | |||
| Pathological T | T1-2 | 11 (100) | 0 | 8.095 | 0.004 |
| T3-4 | 50 (55.6) | 40 (44.4) | |||
| Pathological nodal status | Negative | 34 (75.6) | 11 (24.4) | 7.798 | 0.005 |
| Positive | 27 (48.2) | 29 (51.8) | |||
| Lymph node ratio | 0.00 (0.00, 0.11) | 0.10 (0.00, 0.18) | -2.903 | 0.004 | |
| Tissue typing | Well differentiated | 53 (64.6) | 29 (35.4) | 3.273 | 0.070 |
| Poorly differentiated | 8 (42.1) | 11 (57.9) | |||
| Lymphovascular invasion | Negative | 2 (8.3) | 22 (91.7) | 35.674 | < 0.001 |
| Positive | 59 (76.6) | 18 (23.4) | |||
| Perineural invasion | Negative | 57 (73.1) | 21 (26.9) | 23.027 | < 0.001 |
| Positive | 4 (17.4) | 19(82.6) |
During follow-up, 26 patients were lost to follow-up. The primary reasons included transfer to other medical institutions (n = 10), nonattendance at scheduled follow-up visits (n = 5), incomplete or unavailable contact information (n = 6), and refusal to participate in telephone-based follow-up (n = 5). For survival analyses, these individuals were handled as censored observations at the time of their last documented follow-up. The median follow-up duration for the entire cohort was 36 months (range, 18-36 months). Among the patients lost to follow-up, 10 belonged to the ctEMVI-positive group and 16 belonged to the ctEMVI-negative group, with no significant difference between groups (P > 0.05). The median DFS was 33 months in the ctEMVI-positive group, whereas the ctEMVI-negative group did not reach the median DFS during the observation period, indicating that more than half of the patients remained event-free at the end of follow-up.
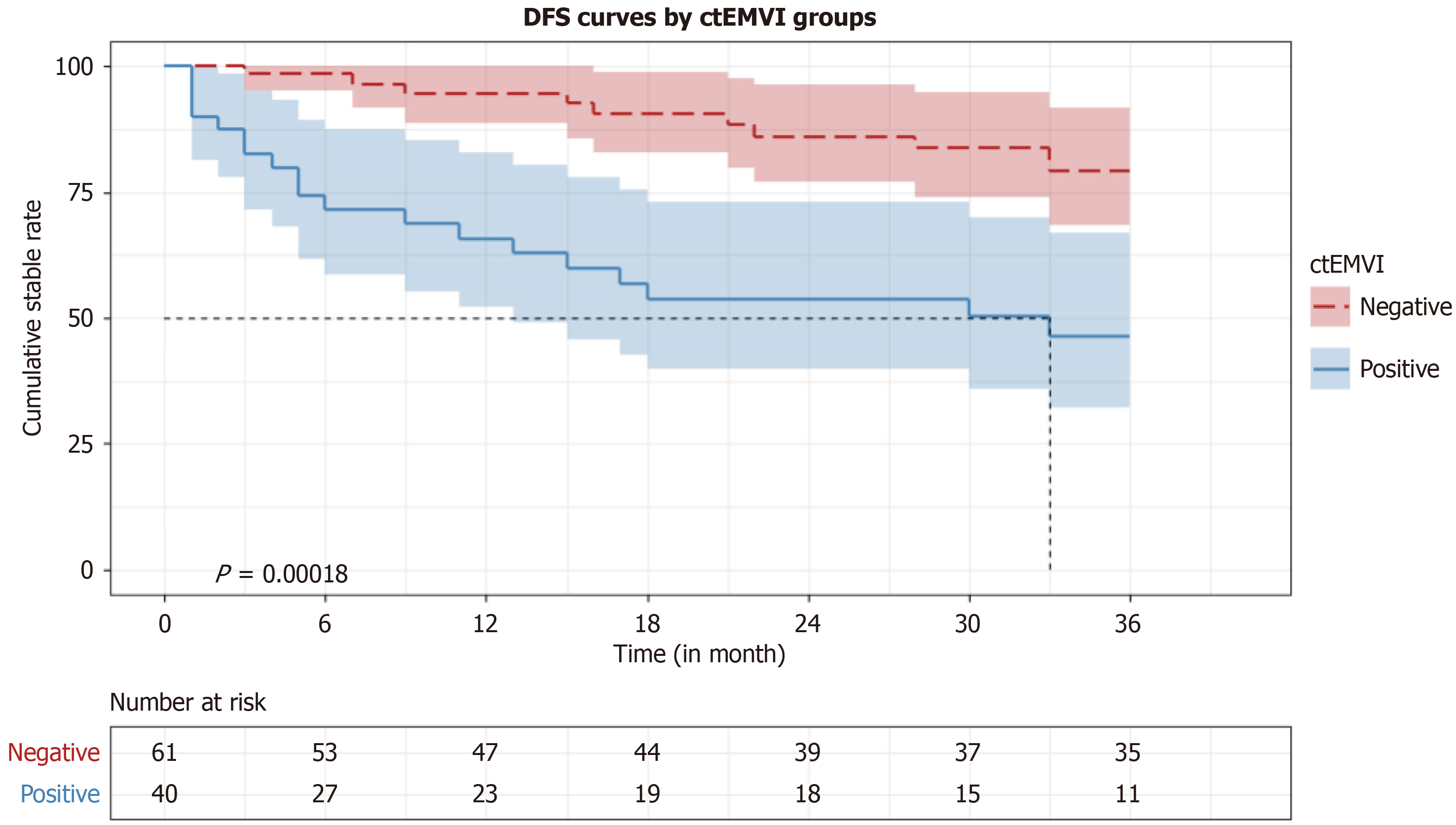
During this period, 72 patients remained free from recurrence, metastasis, or mortality, whereas 29 experienced disease progression, including 19 in the ctEMVI-positive group and 10 in the ctEMVI-negative group. Kaplan-Meier analysis demonstrated that patients with ctEMVI-positive tumors had a markedly shorter DFS compared with those without ctEMVI (χ² = 13.991, P = 0.00018; Figure 3).
Univariate Cox regression identified several variables significantly associated with 3-year DFS, including ctN [hazard ratio (HR): 2.76, 95%CI: 1.81-6.46], ctEMVI (HR: 3.91, 95%CI: 1.81-8.43), pN (HR: 6.36, 95%CI: 2.21-18.30), lymph node ratio (HR: 30.65, 95%CI: 6.70-140.24), tissue typing (HR: 2.64, 95%CI: 1.22-5.70), LVI (HR: 2.65, 95%CI: 1.26-5.56), and PNI (HR: 5.25, 95%CI: 2.49-11.06) (all P < 0.05). No significant associations were observed for sex, age, CEA, CA19-9, tumor location, tumor size, ctT stage, or pathological T stage (P > 0.05).
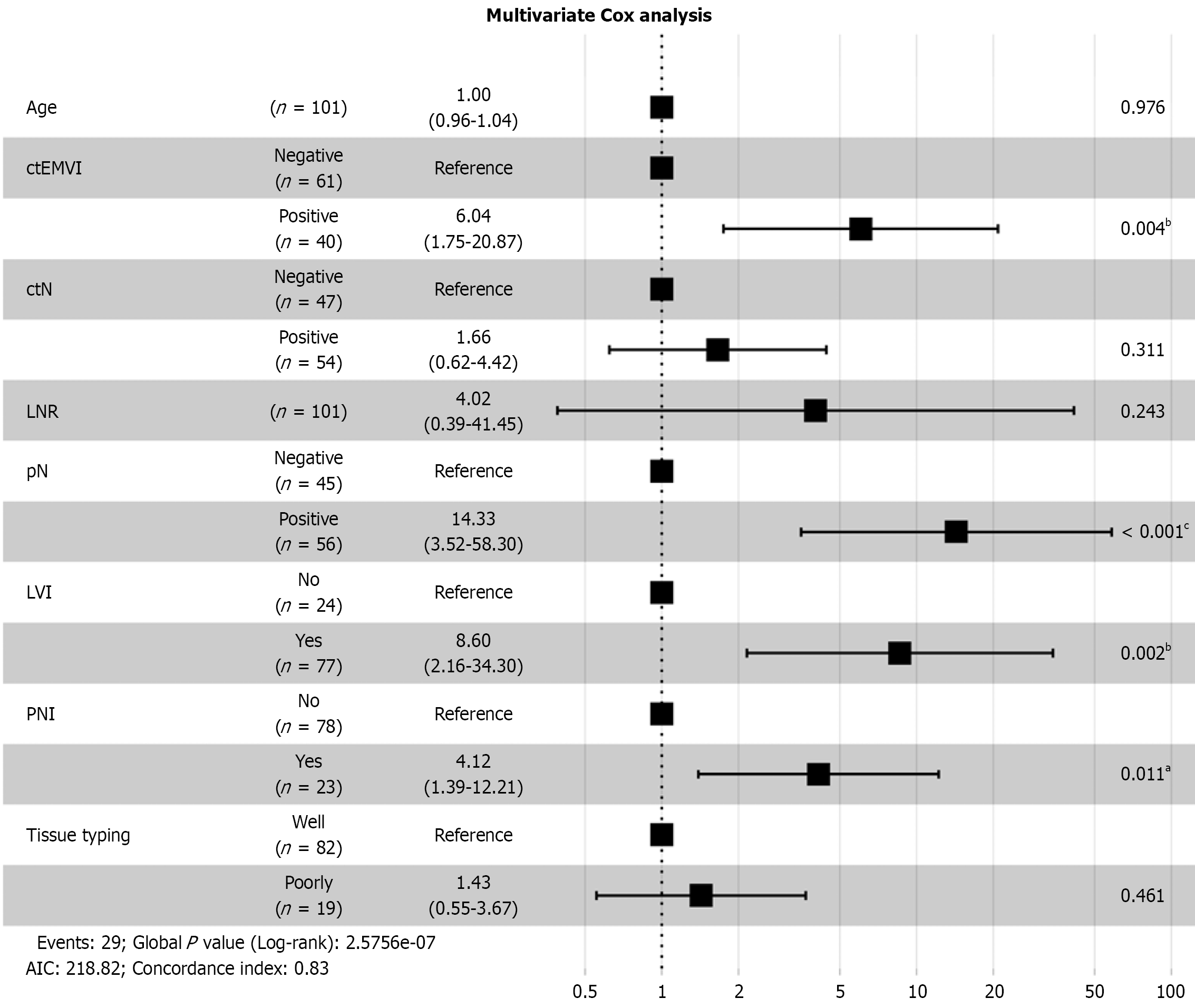
In multivariate analysis, ctEMVI (HR: 6.04, 95%CI: 1.75-20.87), pN (HR: 14.33, 95%CI: 3.52-58.30), LVI (HR: 8.60, 95%CI: 2.16-34.30), and PNI (HR: 4.12, 95%CI: 1.39-12.21) emerged as independent predictors of DFS in patients with colon cancer (P < 0.05; Figure 4).
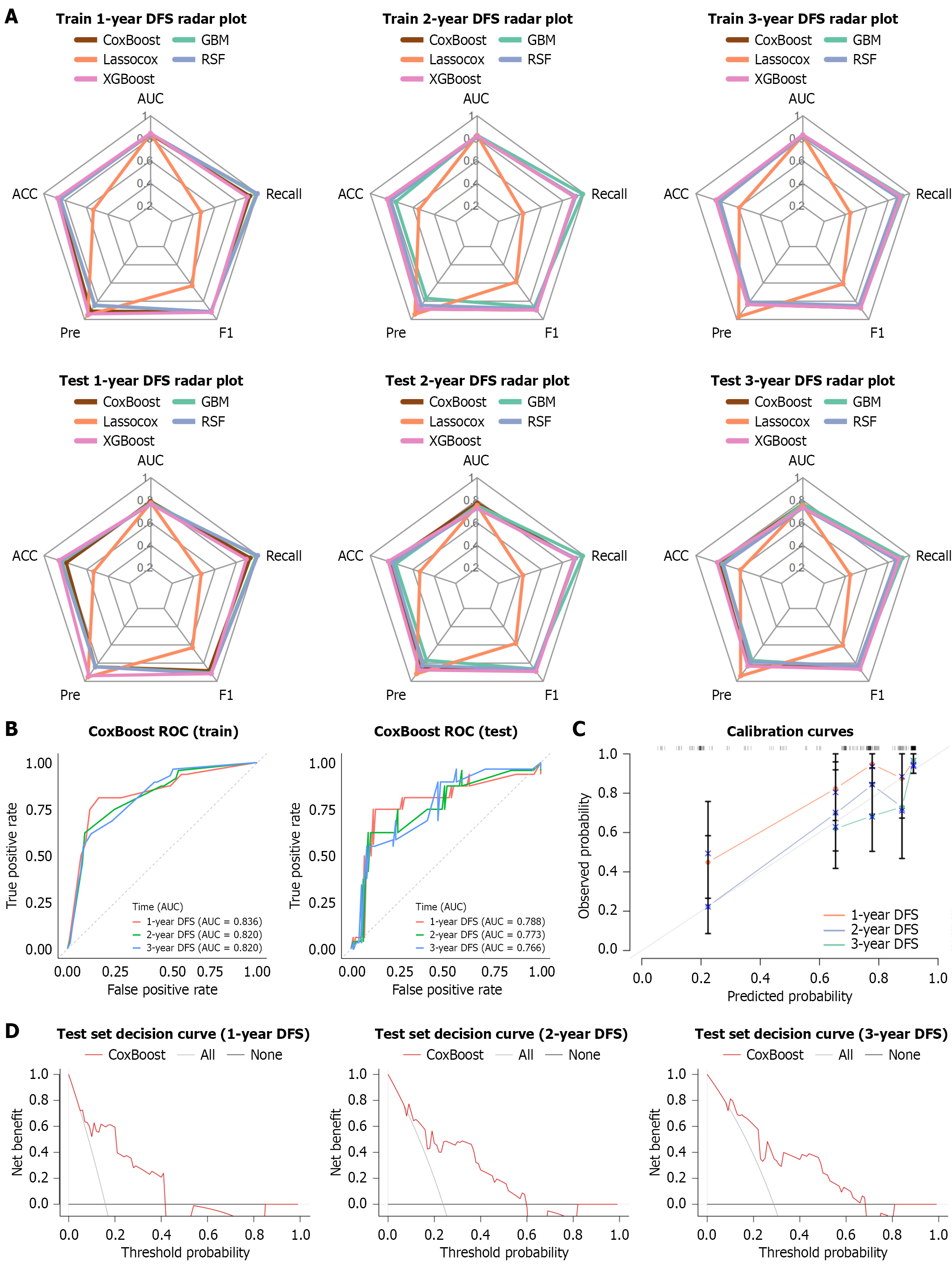
The predictive capabilities of five machine-learning algorithms were systematically compared to identify the optimal model for DFS prediction (Figure 5A). The ROC curve of the selected model was subsequently generated to assess its ACC in forecasting 1-year, 2-year, and 3-year DFS (Figure 5B). Among the evaluated models, the CoxBoost algorithm demonstrated the strongest discriminative performance, yielding AUC values of 0.788 (95%CI: 0.646-0.930), 0.773 (95%CI: 0.659-0.886), and 0.766 (95%CI: 0.662-0.870) for 1-year, 2-year, and 3-year DFS predictions, respectively. These perfor
Calibration curves indicated close alignment between predicted and observed DFS probabilities in both training and test datasets (Figure 5C), confirming the model’s robustness. Furthermore, DCA revealed that the CoxBoost model provided a higher net clinical benefit across a broad range of threshold probabilities for 1-year, 2-year, and 3-year DFS predictions (Figure 5D).
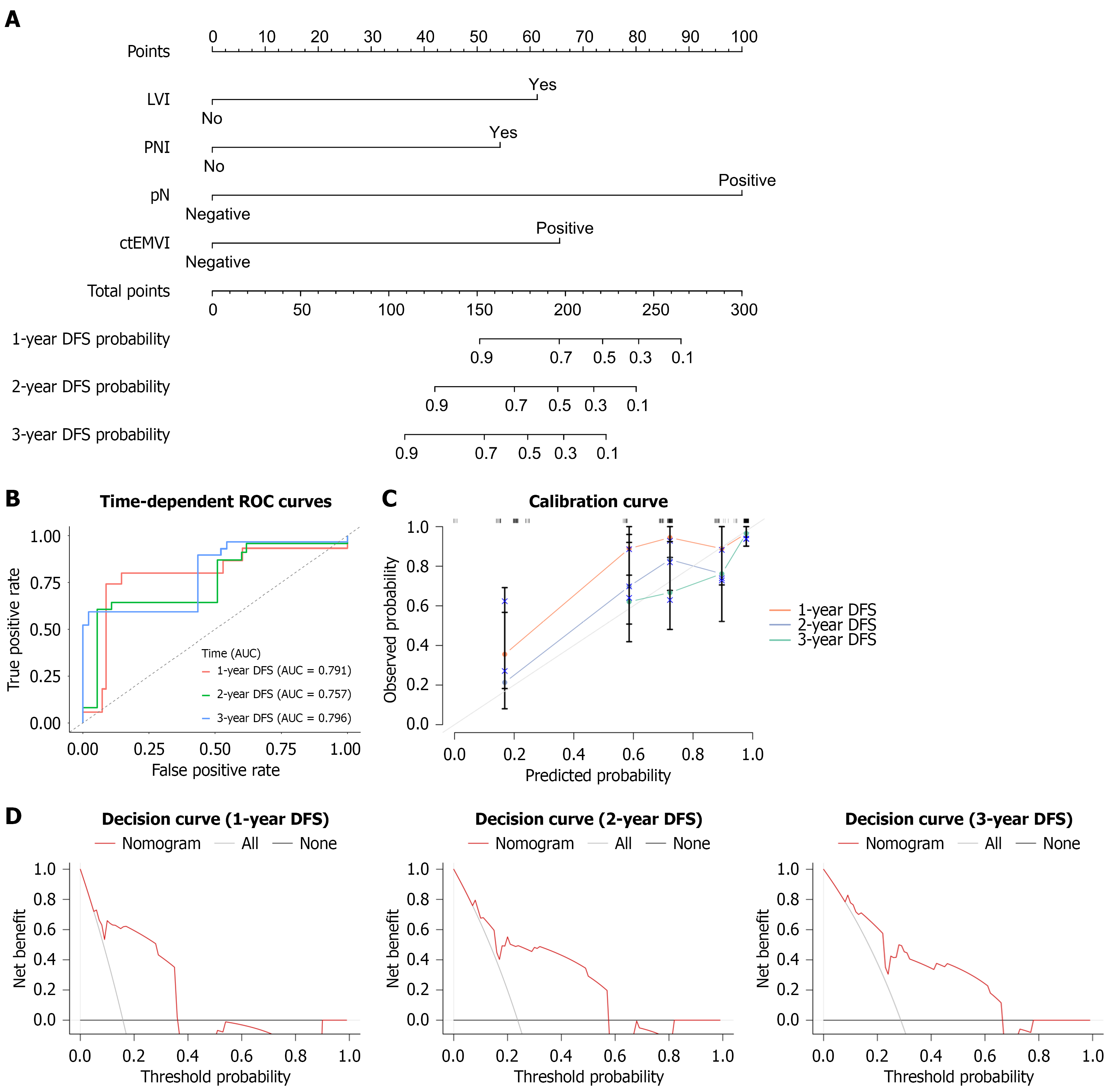
A nomogram was constructed incorporating the four independent prognostic factors identified in multivariate analysis (Figure 6A). Among these variables, pN positivity carried the greatest relative weight, followed by ctEMVI status. Using disease progression (0 = no, 1 = yes) as the dependent outcome and model-predicted probabilities as the independent variable, ROC analysis was performed on the test set. The nomogram achieved AUC values of 0.791 (95%CI: 0.644-0.937), 0.757 (95%CI: 0.630-0.883), and 0.796 (95%CI: 0.686-0.906) for 1-year, 2-year, and 3-year DFS predictions, respectively (Figure 6B). All AUCs exceeded 0.750 and were consistent with the model’s C-index of 0.748, demonstrating favorable discriminative ability.
Calibration plots confirmed strong concordance between predicted and actual DFS outcomes (Figure 6C). DCA further indicated that the nomogram provided consistently higher net benefit than a null model across the entire high-risk threshold range (0-1.0) for 1-year, 2-year, and 3-year DFS (Figure 6D). These results collectively underscored the nomo
Both the CoxBoost model and the nomogram were internally validated using LOOCV. The nomogram demonstrated superior predictive performance for 1-year (AUC = 0.791, 95%CI: 0.644-0.937) and 3-year (AUC = 0.796, 95%CI: 0.686-0.906) DFS following curative resection for colon cancer. In contrast, the CoxBoost model exhibited optimal ACC in predicting 2-year DFS, achieving an AUC of 0.773 (95%CI: 0.659-0.886). These results indicated that while both approa
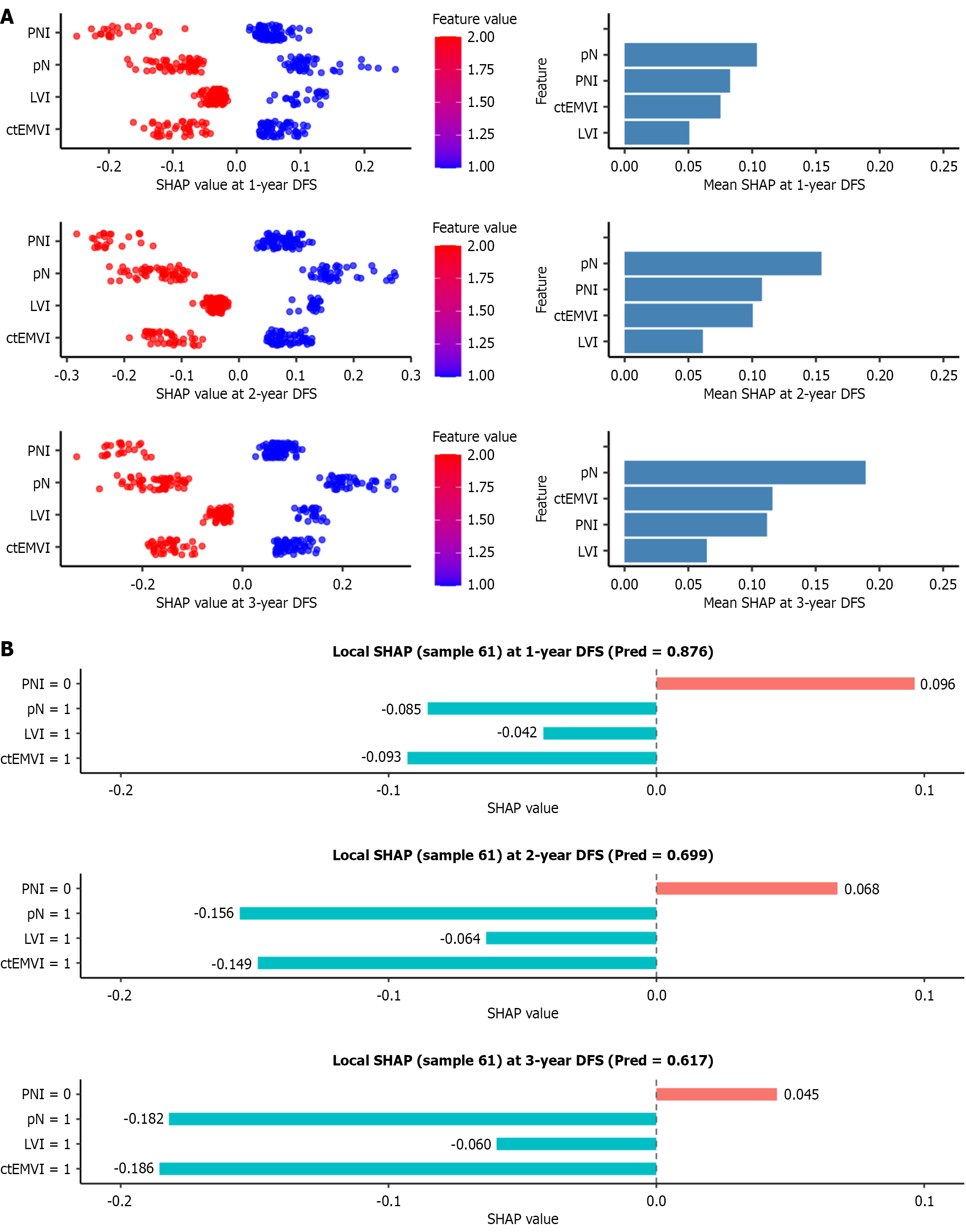
SHAP analysis was employed to elucidate the contribution of individual features to DFS predictions and to enhance model interpretability. In the CoxBoost model, SHAP dependence plots were used to rank feature importance, visually representing the influence of each variable on predicted outcomes (Figure 7A).
The SHAP analysis highlighted that ctEMVI, pN, LVI, and PNI were all key determinants of DFS after curative colon cancer surgery. Among these, pN status consistently exerted the greatest effect on both short-term (1-year) and long-term (3-year) DFS. Although ctEMVI did not rank as the top predictor, its influence was more pronounced in long-term survival outcomes. Additionally, local SHAP analysis was conducted for a randomly selected patient, confirming the interpretability and individualized predictive capacity of the model (Figure 7B).
In the present study, preoperative ctEMVI was identified as a robust prognostic marker in colon cancer. Our findings demonstrated that ctEMVI was strongly associated with advanced clinicopathological features and served as an independent predictor of diminished DFS. Leveraging the results of multivariate regression analysis, we successfully constructed both traditional Cox regression-based nomograms and multiple machine-learning-based predictive models. Among five candidate machine-learning approaches, the CoxBoost model exhibited superior predictive performance, while the Cox-based nomogram also demonstrated reliable discriminative ability upon validation. A direct comparison between the optimal machine-learning model and the nomogram, incorporating time-dependent analysis, allowed for the identification of the most accurate model at different postoperative intervals. Furthermore, SHAP provided quantitative insights into the contribution of individual features to model predictions, enhancing interpretability.
Nomograms have gained widespread acceptance in both biomedical research and routine clinical practice owing to their intuitive interpretability and comparatively robust performance in small-sample contexts, and they are extensively applied for prognostic evaluation in oncology. Despite these advantages, conventional nomogram-based approaches may be inherently limited in their ability to model nonlinear associations and high-order interactions among predictors. In parallel with the rapid evolution of machine-learning methodologies, data-driven models capable of autonomously learning complex patterns from multidimensional data have emerged as increasingly attractive tools for outcome prediction. The incorporation of SHAP facilitates quantitative attribution of individual variables, thereby mitigating the traditional “black-box” nature of machine-learning models and enhancing their interpretability. Nevertheless, whether machine-learning algorithms can reproducibly and consistently surpass traditional nomogram-based models across specific clinical settings remains an area of ongoing debate. Previous studies, such as that by Lei et al[14] have compared nomograms and machine-learning models in predicting overall survival for patients with non-small cell lung cancer. Given the time-dependent nature of DFS and the evolving predictive performance of both nomograms and machine-learning algorithms, incorporating temporal evaluation in our study strengthened the evidence supporting the ACC and reliability of these predictive tools. CRC is characterized by aggressive behavior and a high propensity for distant metastasis, particularly to the liver and lungs, which accounts for the majority of disease-related deaths. Surgical resection remains the only potentially curative treatment modality[15]. Notably, Sargent et al[16] have reported that approximately 80% of colon cancer recurrences occur within the first 3 years post-surgery, underscoring the importance of identifying factors that influence DFS during this critical postoperative window.
EMVI is widely recognized as a high-risk feature in CRC, strongly correlating with elevated rates of metastasis and postoperative recurrence[17-19]. Prior studies have highlighted its prognostic superiority over conventional MRI staging. For instance, Lord et al[20] have reported that EMVI status outperforms MRI in predicting T and N stage, offering enhanced guidance for subsequent therapeutic planning. Similarly, D'Souza et al[21] have emphasized that EMVI detection, alongside tumor deposits and T3 sub-staging, identifies patients at elevated risk for recurrence, providing a rationale for intensified postoperative monitoring. Consistent with these findings, our study confirmed that ctEMVI, pN, LVI, and PNI independently predicted DFS (P < 0.05).
Notably, ctEMVI-positive patients exhibited significantly shorter DFS compared with ctEMVI-negative individuals (P < 0.05). A total of 101 patients were enrolled in the present study, among whom 26 were lost to follow-up, including 10 patients in the ctEMVI-positive group and 16 in the ctEMVI-negative group. The proportion of patients lost to follow-up did not differ significantly between groups (P = 0.89), indicating a balanced distribution. In the survival analyses, these individuals were appropriately handled as censored observations at the time of their last documented follow-up. Although loss to follow-up may compromise data completeness and introduce potential bias, the Cox proportional hazards model is specifically designed to accommodate censored data. Nevertheless, we recognized that the relatively high rate of loss to follow-up constituted an important limitation of this study and might have affected the robustness of the survival estimates. Future prospective investigations with more complete follow-up are therefore required to further corroborate and refine our findings. In a particularly illustrative observation, three patients who had no detectable metastases preoperatively developed liver metastases within 1 month post-surgery; all were classified as ctEMVI-positive prior to surgery. This underscored the potential of ctEMVI as a sensitive marker for occult metastatic risk. For such high-risk patients, even in the absence of preoperative metastases, clinicians might consider supplementary imaging modalities, such as MRI or positron emission tomography/CT, and shorten the postoperative surveillance interval to enable timely intervention.
Beyond conventional prognostic assessment, machine learning offers transformative potential for survival prediction in oncology[22]. CoxBoost, a gradient boosting-based adaptation of the Cox proportional hazards model, combines the statistical rigor of traditional survival analysis with the adaptive learning capacity of modern machine learning. Through iterative optimization, CoxBoost refines predictive models and has demonstrated superior performance in numerous prognostic studies. In the present study, we developed predictive models using XGBoost, RSF, CoxBoost, GBM, and LASSO-Cox algorithms. Within the test set, CoxBoost consistently outperformed its counterparts, achieving AUC values of 0.788 (95%CI: 0.646-0.930) for 1-year DFS, 0.773 (95%CI: 0.659-0.886) for 2-year DFS, and 0.766 (95%CI: 0.662-0.870) for 3-year DFS. These results highlighted CoxBoost as a robust tool for individualized postoperative risk stratification in colon cancer. Taken together, the integration of ctEMVI assessment with machine-learning models provided a powerful framework for precision oncology, enabling clinicians to identify high-risk patients, tailor follow-up schedules, and optimize adjuvant treatment strategies.
Nomograms constructed from clinicopathological parameters have become indispensable tools for predicting disease progression across diverse tumor types and risk groups, reaffirming their central role in oncologic prognostication[23]. By providing a straightforward, multivariable visualization, nomograms enable intuitive, patient-specific risk assessment, allowing clinicians to identify high-risk individuals and implement timely interventions[24]. Several nomograms incorporating EMVI have been developed in CRC to predict postoperative recurrence and survival, thereby guiding decisions regarding intensified adjuvant therapy. For example, Zhao et al[25] have developed a nomogram integrating EMVI grading to predict individualized 3-year and 5-year DFS in rectal cancer patients, while Chen et al[26] have constructed a nomogram for DFS prediction using EMVI and additional prognostic factors, achieving a C-index of 0.688 and AUCs of 0.731, 0.723, and 0.779 for 1-year, 3-year, and 5-year survival, respectively. Consistent with these findings, our study demonstrated that the nomogram achieved AUC values of 0.791 (95%CI: 0.644-0.937), 0.757 (95%CI: 0.630-0.883), and 0.796 (95%CI: 0.686-0.906) for 1-year, 2-year, and 3-year DFS predictions, all exceeding 0.75 and closely aligning with the model’s C-index of 0.748. These results underscored the nomogram’s robust predictive performance and clinical utility in stratifying postoperative risk in colon cancer.
In this study, the nomogram demonstrated robust predictive performance for 1-year and 3-year DFS following curative resection, whereas the CoxBoost model exhibited optimal ACC for 2-year DFS. These findings suggested that integrating machine-learning algorithms with traditional nomograms could harness the strengths of both approaches, potentially enhancing prognostic precision across multiple postoperative intervals. Interpretability remains a critical barrier to the adoption of machine-learning models in clinical practice. By leveraging SHAP, model predictions can be visualized in a manner that quantifies the contribution of individual features to patient outcomes[27]. In this study, SHAP analysis revealed that pN status exerted the greatest influence on DFS predictions. Importantly, local SHAP analyses demon
The American Society of Colon and Rectal Surgeons recommends consideration of adjuvant chemotherapy for patients with stage II colon cancer who exhibit high-risk features, with the aim of avoiding overtreatment in those at low risk[28]. Prior studies have demonstrated that stage II patients with EMVI positivity experience poorer overall survival than EMVI-negative patients with stage III disease, and adjuvant chemotherapy confers significant DFS benefits in both EMVI-positive stage II and stage III colon cancer, underscoring the strong prognostic relevance of EMVI[29]. In parallel, the therapeutic landscape of CRC has evolved substantially, with the availability of precision strategies, including targeted therapies and immunotherapy, for metastatic disease characterized by specific molecular alterations, offering effective and individualized treatment options[30].
In this context, the present study advanced the field by integrating preoperative ctEMVI with established pathological risk factors, performing a direct comparison between conventional Cox-based nomograms and machine-learning models, and leveraging SHAP to validate feature contributions and enhance model interpretability. This integrative framework enables more refined identification of high-risk colon cancer patients than reliance on any single prognostic indicator. Notably, patients with ctEMVI positivity, even those with early pathological stages, should be regarded as high-risk and may warrant multidisciplinary evaluation, individualized treatment planning, and intensified postoperative surveillance. Conversely, ctEMVI-negative patients lacking additional high-risk features generally demonstrated favorable outcomes, suggesting that adjuvant treatment intensity and follow-up strategies could potentially be de-escalated, thereby improving resource utilization without compromising oncologic safety.
Despite these encouraging findings, several limitations merit careful consideration. First, this investigation was conducted as a retrospective, single-center study and was characterized by a relatively high rate of loss to follow-up, which may introduce selection bias and affect the robustness of survival estimates. Although the reasons for loss to follow-up were documented and the distribution between ctEMVI-positive and ctEMVI-negative groups was comparable, incomplete outcome ascertainment may still limit the generalizability of the results. Second, the modest sample size and limited number of observed events might constrain the stability of both multivariable Cox regression and machine-learning-based models. Under such conditions, complex models are inherently more vulnerable to overfitting, and parameter estimates, including hazard ratios, may be unstable. Furthermore, performance metrics such as AUC, ACC, and F1 score may appear overly optimistic and should therefore be interpreted with caution. Accordingly, external validation in larger, independent cohorts is essential to confirm the reproducibility and clinical utility of the proposed models.
In addition, the present study did not explicitly evaluate the impact of adjuvant chemotherapy on survival outcomes. We acknowledge that treatment decisions are influenced by multiple factors, including patient age, comorbidities, and pathological stage, resulting in inherent treatment heterogeneity. To minimize confounding and better isolate the intrinsic prognostic value of ctEMVI, analyses were restricted to patients who did not receive adjuvant therapy during the study period. Although this strategy may introduce selection bias, our findings consistently demonstrate that preoperative ctEMVI remains a strong prognostic marker, supporting its role as an indicator of intrinsic tumor aggressiveness. Future large-scale, prospective studies involving patients treated according to contemporary adjuvant therapy standards are warranted to further validate ctEMVI and the integrated modeling framework for guiding risk stratification, therapeutic decision-making, and postoperative surveillance.
Our findings confirmed that ctEMVI was a valuable preoperative indicator for prognostic assessment in colon cancer. Both the CoxBoost machine-learning model and the clinicopathology-based nomogram, constructed with ctEMVI and associated risk factors, provided independent, interpretable tools for predicting postoperative DFS. By quantitatively stratifying the risk of disease progression, these models enabled personalized clinical decision-making, guiding surveillance intensity and adjuvant therapy selection. Collectively, this integrated approach represented a significant step toward precision oncology, facilitating more accurate and individualized management of patients with colon cancer.
| 1. | Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA Cancer J Clin. 2023;73:233-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2171] [Cited by in RCA: 1974] [Article Influence: 658.0] [Reference Citation Analysis (14)] |
| 2. | Hernandez Dominguez O, Yilmaz S, Steele SR. Stage IV Colorectal Cancer Management and Treatment. J Clin Med. 2023;12:2072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 80] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 3. | Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, Garrido-Laguna I, Grem JL, Gunn A, Hecht JR, Hoffe S, Hubbard J, Hunt S, Johung KL, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Miller ED, Mulcahy MF, Nurkin S, Overman MJ, Parikh A, Patel H, Pedersen K, Saltz L, Schneider C, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Gregory KM, Gurski LA. Colon Cancer, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021;19:329-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1323] [Cited by in RCA: 1121] [Article Influence: 224.2] [Reference Citation Analysis (5)] |
| 4. | Ge Y, Jia Y, Li Y, Dai J, Guan R, Hu S. Synthetic phase-sensitive inversion-recovery vessel for assessing extramural venous invasion in patients with rectal cancer: imaging quality and added value to T2-wighted imaging. Eur Radiol. 2023;33:4148-4157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Guan Z, Zhang XY, Li XT, Sun RJ, Lu QY, Wu AW, Sun YS. Correlation and prognostic value of CT-detected extramural venous invasion and pathological lymph-vascular invasion in colon cancer. Abdom Radiol (NY). 2022;47:1232-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 6. | Mc Entee PD, Shokuhi P, Rogers AC, Mehigan BJ, McCormick PH, Gillham CM, Kennedy MJ, Gallagher DJ, Ryan CE, Muldoon CB, Larkin JO. Extramural venous invasion (EMVI) in colorectal cancer is associated with increased cancer recurrence and cancer-related death. Eur J Surg Oncol. 2022;48:1638-1642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 7. | Cho J, Kim YH, Kim HY, Chang W, Park JH. Extramural venous invasion and depth of extramural invasion on preoperative CT as prognostic imaging biomarkers in patients with locally advanced ascending colon cancer. Abdom Radiol (NY). 2022;47:3679-3687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Tan JJ, Carten RV, Babiker A, Abulafi M, Lord AC, Brown G. Prognostic Importance of MRI-Detected Extramural Venous Invasion in Rectal Cancer: A Literature Review and Systematic Meta-Analysis. Int J Radiat Oncol Biol Phys. 2021;111:385-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 9. | Bates DDB, Homsi ME, Chang KJ, Lalwani N, Horvat N, Sheedy SP. MRI for Rectal Cancer: Staging, mrCRM, EMVI, Lymph Node Staging and Post-Treatment Response. Clin Colorectal Cancer. 2022;21:10-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 10. | Kato T, Tsukamoto S, Miyake M, Kudose Y, Takamizawa Y, Moritani K, Daiko H, Kanemitsu Y. Prognostic impact of extramural venous invasion detected by contrast-enhanced CT colonography in colon cancer. BJS Open. 2024;8:zrad121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Cheng S, Hu G, Zhang S, Lv R, Sun L, Zhang Z, Jin Z, Wu Y, Huang C, Ye L, Feng Y, Chen ZS, Wang Z, Xue H, Yang A. Machine Learning-Based Radiomics in Malignancy Prediction of Pancreatic Cystic Lesions: Evidence from Cyst Fluid Multi-Omics. Adv Sci (Weinh). 2025;12:e2409488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Zhu Z, Mao M, Song A, Gong H, Gu J, Dai Y, Feng F. Study on the diagnostic value of MDCT extramural vascular invasion in preoperative N staging of gastric cancer patients. BMC Med Imaging. 2024;24:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Song W, Chen Z, Zheng Z, Hu J, Chen Y, Deng W, He X, Lan P. Prognostic value of radiologically enlarged lymph nodes in node-negative colon cancer. Colorectal Dis. 2020;22:537-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Lei H, Li X, Ma W, Hong N, Liu C, Zhou W, Zhou H, Gong M, Wang Y, Wang G, Wu Y. Comparison of nomogram and machine-learning methods for predicting the survival of non-small cell lung cancer patients. Cancer Innov. 2022;1:135-145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 15. | Strickler JH, Yoshino T, Graham RP, Siena S, Bekaii-Saab T. Diagnosis and Treatment of ERBB2-Positive Metastatic Colorectal Cancer: A Review. JAMA Oncol. 2022;8:760-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (3)] |
| 16. | Sargent DJ, Wieand HS, Haller DG, Gray R, Benedetti JK, Buyse M, Labianca R, Seitz JF, O'Callaghan CJ, Francini G, Grothey A, O'Connell M, Catalano PJ, Blanke CD, Kerr D, Green E, Wolmark N, Andre T, Goldberg RM, De Gramont A. Disease-free survival versus overall survival as a primary end point for adjuvant colon cancer studies: individual patient data from 20,898 patients on 18 randomized trials. J Clin Oncol. 2005;23:8664-8670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 560] [Cited by in RCA: 506] [Article Influence: 24.1] [Reference Citation Analysis (1)] |
| 17. | Tripathi P, Rao SX, Zeng MS. Clinical value of MRI-detected extramural venous invasion in rectal cancer. J Dig Dis. 2017;18:2-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 18. | Foxtrot Collaborative Group. Feasibility of preoperative chemotherapy for locally advanced, operable colon cancer: the pilot phase of a randomised controlled trial. Lancet Oncol. 2012;13:1152-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 362] [Article Influence: 25.9] [Reference Citation Analysis (1)] |
| 19. | Yang SX, Yao X, Song XH, Cui YC, Ye YJ, Wang Y. Extramural vascular invasion detected by contrast-enhanced multiple-row detectors computed tomography (ceMDCT) as a predictor of synchronous metastases in colon cancer. Oncotarget. 2017;8:94883-94892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 20. | Lord AC, D'Souza N, Shaw A, Rokan Z, Moran B, Abulafi M, Rasheed S, Chandramohan A, Corr A, Chau I, Brown G. MRI-Diagnosed Tumor Deposits and EMVI Status Have Superior Prognostic Accuracy to Current Clinical TNM Staging in Rectal Cancer. Ann Surg. 2022;276:334-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 157] [Article Influence: 39.3] [Reference Citation Analysis (1)] |
| 21. | D'Souza N, Shaw A, Lord A, Balyasnikova S, Abulafi M, Tekkis P, Brown G. Assessment of a Staging System for Sigmoid Colon Cancer Based on Tumor Deposits and Extramural Venous Invasion on Computed Tomography. JAMA Netw Open. 2019;2:e1916987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 22. | Kaur I, Doja MN, Ahmad T. Data mining and machine learning in cancer survival research: An overview and future recommendations. J Biomed Inform. 2022;128:104026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 23. | Wu J, Zhang H, Li L, Hu M, Chen L, Xu B, Song Q. A nomogram for predicting overall survival in patients with low-grade endometrial stromal sarcoma: A population-based analysis. Cancer Commun (Lond). 2020;40:301-312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 466] [Cited by in RCA: 393] [Article Influence: 65.5] [Reference Citation Analysis (1)] |
| 24. | Chen X, He Z, Zhao C, Wu K, Zhu Q, Fu Y, Pan Y, Fan Y, Yang S, Zeng Y, Luo S, Liu L, Du F, Zhou X. Construction and validation of a nomogram based on the log odds of positive lymph nodes to predict the prognosis of T1 gastric cancer. Sci Rep. 2025;15:7788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 25. | Zhao Q, Wan L, Zou S, Zhang C, E T, Yang Y, Ye F, Zhao X, Ouyang H, Zhang H. Prognostic risk factors and survival models for T3 locally advanced rectal cancer: what can we learn from the baseline MRI? Eur Radiol. 2021;31:4739-4750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 26. | Chen F, Zhang S, Fu C, Grimm R, Lu J, Shao C, Shen F, Chen L. Predicting disease-free survival in locally advanced rectal cancer using a prognostic model based on pretreatment b-value threshold map and postoperative pathologic features. Jpn J Radiol. 2025;43:236-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Huang Y, Li N, Han X, Xu S, Zhang G, Zhu X. Radiomics application using non-contrast computed tomography for predicting uric acid kidney stones. BMC Med Imaging. 2025;25:433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Vogel JD, Felder SI, Bhama AR, Hawkins AT, Langenfeld SJ, Shaffer VO, Thorsen AJ, Weiser MR, Chang GJ, Lightner AL, Feingold DL, Paquette IM. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Colon Cancer. Dis Colon Rectum. 2022;65:148-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 252] [Article Influence: 63.0] [Reference Citation Analysis (1)] |
| 29. | Qwaider YZ, Sell NM, Stafford CE, Kunitake H, Ricciardi R, Bordeianou LG, Deshpande V, Goldstone RN, Cauley CE, Berger DL. Adjuvant Chemotherapy Benefits on Patients with Extramural Vascular Invasion in Stages II and III Colon Cancer. J Gastrointest Surg. 2021;25:2019-2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Miller KD, Nogueira L, Devasia T, Mariotto AB, Yabroff KR, Jemal A, Kramer J, Siegel RL. Cancer treatment and survivorship statistics, 2022. CA Cancer J Clin. 2022;72:409-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2024] [Cited by in RCA: 1814] [Article Influence: 453.5] [Reference Citation Analysis (4)] |