Published online Mar 21, 2026. doi: 10.3748/wjg.v32.i11.115488
Revised: November 14, 2025
Accepted: January 12, 2026
Published online: March 21, 2026
Processing time: 149 Days and 22.5 Hours
Pancreatic cancer frequently metastasizes to the liver, and thereby confers high mortality risk.
To investigated the effect of metabolic dysfunction-associated steatotic liver disease (MASLD) on liver metastasis and survival outcomes in patients with pancreatic cancer, as the increasing incidence of MASLD.
We retrospectively analyzed data from 2123 patients who were diagnosed with pancreatic cancer between 2006 and 2021. MASLD was diagnosed based on a hepatic steatosis index (HSI) > 30.
In the study population, which predominantly comprised males, the median age was 66 years (n = 1118, 52.6%). Patients with liver metastasis at baseline (n = 540, 25.4%) had larger tumors (median, 41 mm vs 35 mm) and higher carbohydrate antigen (CA) 19-9 levels (median 975.0 U/mL vs 206.2 U/mL; all P < 0.001). During the follow-up period (median 9.7 months), 502 (23.6%) patients were diagnosed with new liver metastases, and 819 (38.6%) died. MASLD did not significantly correlate with liver metastasis at diagnosis or new liver metastases (all P > 0.05). In contrast, younger age [hazard ratio (HR) = 0.985, 95% confidence interval (CI): 0.974-0.997], increased tumor size (HR = 1.008, 95%CI: 1.001-1.016), and elevated CA19-9 levels (HR = 1.662, 95%CI: 1.279-2.160) were significantly associated with new liver metastases during the follow-up (all P < 0.05). Although MASLD appeared to be as
Thus, HSI-defined MASLD does not directly influence liver metastasis or survival in patients with pancreatic cancer.
Core Tip: Pancreatic cancer frequently spreads to the liver, contributing to its high mortality. With the rising prevalence of metabolic dysfunction-associated steatotic liver disease (MASLD), its potential impact on cancer outcomes remains uncertain. In this large cohort study of 2123 patients with pancreatic cancer, MASLD-defined by a hepatic steatosis index, was not associated with liver metastasis at diagnosis, new liver metastasis during follow-up or overall survival after adjustment for other risk factors. These findings suggest that MASLD does not significantly influence the metastatic pattern or prognosis in patients with pancreatic cancer.
- Citation: Chon HY, Rhee H, Kim J, Leem G, Jo JH, Chung MJ, Park JY, Bang S, Park SW, Kim SU, Lee HS. Impact of metabolic dysfunction-associated steatotic liver disease on liver metastasis and survival in pancreatic cancer. World J Gastroenterol 2026; 32(11): 115488
- URL: https://www.wjgnet.com/1007-9327/full/v32/i11/115488.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i11.115488
Pancreatic cancer remains one of the most challenging malignancies in oncology and is characterized by late diagnosis, aggressive nature, and poor prognosis[1]. Despite advancements in medical science, the 5-year survival rate of pancreatic cancer remains dismally low. A key factor in its lethality is its propensity for early metastasis, particularly to the liver, which significantly worsens patient outcomes[2]. Understanding the factors influencing this metastatic process is crucial for improving prognosis and guiding therapeutic interventions.
Research into the interplay between cancer progression and the associated comorbidities, particularly those affecting the liver, has increasingly generated interest. Metabolic dysfunction-associated steatotic liver disease (MASLD) is a prevalent condition that is characterized by the accumulation of fat in liver cells and is complicated by cardiometabolic factors[3,4]. Although the impact of MASLD on liver function and overall health is well documented, its role in oncology, particularly pancreatic cancer, remains unclear.
Recent studies have shed light on the complex relationship between MASLD and cancer, and thereby suggested its potential influence on tumor progression and metastasis. For example, MASLD is associated with a reduced risk of liver metastasis in colorectal cancer[5,6]. Several mechanisms of liver metastasis in colorectal cancer have been suggested, including increased insulin/insulin growth factor-1 and hepatocyte derived extracellular vesicles which enhance yes-associated protein signaling and immunosuppressive tumor microenvironment[7,8]. However, the applicability of these findings to pancreatic cancer remains unclear. The association between MASLD and pancreatic cancer has been previously reported[9]; however, there is limited research into the impact of MASLD on the development of pancreatic cancer liver metastasis. The unique biology of pancreatic cancer, including its aggressive metastatic behavior, may interact differently with the MASLD-altered hepatic environment[10].
This study aimed to bridge this knowledge gap by investigating the relationship between hepatic steatosis index (HSI)-defined MASLD, liver metastasis, and survival in patients with pancreatic cancer.
This study comprised a retrospective analysis of a cohort of 2123 patients diagnosed with pancreatic cancer between 2006 and 2021 at a large tertiary academic center (Supplementary Figure 1). The primary objective of this study was to investigate the effects of MASLD on liver metastasis and patient survival. The inclusion criterion was a confirmed diagnosis of pancreatic ductal adenocarcinoma. The exclusion criteria were as follows: (1) Age < 19 years (n = 3); (2) Chronic hepatitis B (n = 93); (3) Chronic hepatitis C (n = 2); (4) Alcoholic liver disease (n = 9); (5) Significant alcohol consumption (> 210 g/week for male and > 140 g/week for female; n = 107)[11]; (6) Medication history (e.g., tamoxifen, steroids); (7) Follow-up loss (n = 41); and (8) Insufficient clinical information calculating HSI (n = 2).
The HSI was calculated for each patient using a formula that incorporated the following clinically available parameters: 8 × [aspartate aminotransferase (AST)/alanine aminotransferase (ALT) ratio] + [body mass index (BMI) + 2 if female; if diabetes mellitus]. Liver enzyme and BMI were measured at the time of cancer diagnosis and before treatment. The HSI was developed using Korean 5462 nonalcoholic fatty liver disease and 5262 normal cohort; the HSI could exclude steatosis when it was < 30 (negative likelihood ratio 0.186, sensitivity 93.1%) and could diagnose steatosis with high specificity when > 36 (positive likelihood ratio 6.069, specificity 92.4%)[12]. The HSI has been previously validated as a reliable noninvasive measure of MASLD, which makes it an ideal tool for this retrospective study[13,14]. The HSI cutoff for the diagnosis of MASLD in our study was adopted from original research, wherein both the cutoff values of HSI > 30 and HSI > 36 were evaluated[12]. To maximize the diagnostic sensitivity, we defined MASLD based on an HSI > 30.
To assess hepatic steatosis, we utilized computed tomography (CT) in a subgroup of patients. A total of 500 patients were randomly selected using Excel’s RAND function, and CT scans were available to assess fatty liver in 452 patients. A faculty abdominal radiologist, with more than 10 years of experience in liver imaging who was blinded to the clinical information of the patients, evaluated the CT images of 500 patients. Four regions of interest (ROIs) were manually drawn in the pre-contrast images of each of the four liver sections, and two ROIs, between 300 mm2 and 400 mm2, were randomly drawn in the spleen to carefully avoid the blood vessels, bile ducts, and focal hepatic or splenic lesions. The attenuation values of the liver and spleen ROIs were measured and the average attenuation values for both the liver and spleen were calculated; MASLD was diagnosed if the average attenuation value of the liver was lower than that of the spleen.
Data were meticulously extracted from the electronic medical records. These included demographic details (age and sex), clinical characteristics (BMI, diabetes mellitus, and hypertension), tumor characteristics [i.e., location, size, and carbohydrate antigen (CA) 19-9 levels], and the presence of cirrhosis. The data necessary to calculate the HSI, including liver enzyme levels, were collected.
Statistical analyses were multifaceted. A logistic regression model was used to evaluate the relationship between the HSI and the presence of liver metastasis at the time of diagnosis. The model provided odds ratios (ORs) with 95% confidence intervals (CIs) and P values to assess the significance of the associations. To understand the effect of hepatic steatosis on overall survival and newly developed liver metastases, a Cox proportional hazards model was employed. In the Cox regression model, survival time was measured from the date of diagnosis to death in patients who died, and to the last visit for those alive. Individuals alive at last contact were right-censored. Potential confounders, including demographic and clinical characteristics, were adjusted. The Kaplan-Meier method was used to compare patients with varying HSI levels. A P < 0.05 by two-tailed test was considered statistically significant. All statistical analyses were performed using the SPSS Statistics version 26 (IBM Corp., Armonk, NY, United States).
The study protocol was reviewed and approved by the institutional review board of the healthcare center (No. 4-2024-1444). As this retrospective study involved the analysis of de-identified patient data, the need for informed consent from individual patients was waived. All the procedures were conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
The study population had a median age of 66 [interquartile range (IQR): 58-73] years, with a slight predominance of males (n = 1118, 52.6%; Table 1). This demographic representation is consistent with the general epidemiology of pancreatic cancer, which tends to have a higher incidence in older populations and a slightly higher prevalence in males. The common comorbidities observed included diabetes mellitus (n = 1042, 49.1%) and hypertension (n = 919, 43.3%), aligning with the known risk factors for pancreatic cancer. The median HSI was 33.9 (IQR: 30.6-37.5), which reflected a diverse range of liver health statuses among the participants. The tumor locations were distributed as follows: 42.6% (n = 904) in head, 22.3% (n = 474) in the body, 17.7% (n = 375) in the tail. The median tumor size was 36.0 mm (IQR: 28.0-48.0), and the median CA19-9 level, a tumor marker of pancreatic cancer, was 259.6 U/mL (IQR: 31.4-1647.0).
| Characteristics | Total (n = 2123) | With liver metastasis (n = 540) | Without liver metastasis (n = 1584) | P value |
| Patient | ||||
| Age (years), median IQR | 66 (58-73) | 65 (58.0-72.0) | 66 (58.0-73.0) | 0.057 |
| Male | 1118 (52.6) | 298 (55.2) | 820 (51.8) | 0.174 |
| Body mass index (kg/m2), median IQR | 22.7 (20.8-24.9) | 22.8 (20.7-25.1) | 22.7 (20.8-24.8) | 0.571 |
| Diabetes mellitus | 1042 (49.1) | 245 (45.4) | 797 (50.3) | 0.046 |
| Hypertension | 919 (43.3) | 239 (44.3) | 680 (43.0) | 0.598 |
| Cirrhosis | 4 (0.2) | 1 (0.2) | 3 (0.2) | 0.984 |
| HSI, median IQR | 33.9 (30.6-37.5) | 34.0 (30.7-37.8) | 33.8 (30.6-37.4) | 0.585 |
| HSI > 30 | 1683 (79.3) | 426 (78.9) | 1257 (79.4) | 0.798 |
| HSI > 36 | 729 (34.3) | 192 (35.6) | 537 (33.9) | 0.490 |
| TG (mg/dL), median IQR | 102 (75-138) | 100 (79-143) | 103 (75-136) | 0.929 |
| HDL (mg/dL), median IQR | 42 (32-50) | 40 (32-47) | 42 (32-52) | 0.043 |
| LDL (mg/dL), median IQR | 86 (64-116) | 84 (62-105) | 87 (65-118) | 0.334 |
| Total bilirubin (mg/dL), median IQR | 0.7 (0.5-1.4) | 0.7 (0.5-1.3) | 0.7 (0.5-1.4) | 0.461 |
| Tumor | ||||
| Location | < 0.001 | |||
| Head | 904 (42.6) | 181 (33.5) | 723 (45.7) | |
| Body | 474 (22.3) | 114 (21.1) | 360 (22.8) | |
| Tail | 375 (17.7) | 139 (25.7) | 236 (14.9) | |
| Mixed | 369 (17.4) | 106 (19.7) | 263 (16.6) | |
| Size (mm), median IQR | 36.0 (28.0-48.0) | 41 (32-55) | 35 (26-45) | < 0.001 |
| CA19-9 (U/mL), median IQR | 259.6 (31.4-1647.0) | 975.0 (45.0-6092.3) | 206.2 (30.0-1046.3) | < 0.001 |
Among 2123 patients, 1042 (49.1%) patients were diagnosed with liver metastasis during the study period (n = 540 at their diagnosis and n = 502 after diagnosis). Patients with and without liver metastasis at baseline were compared. Patients with liver metastasis showed a lower prevalence of diabetes mellitus (45.4% vs 50.3%, P = 0.046). In patients with liver metastasis, the tumor size was larger (41 mm vs 35 mm) and the CA19-9 (975.0 U/mL vs 206.2 U/mL) levels were higher (all P < 0.001). After excluding patient with liver metastasis at baseline, a comparison of the patients with and without newly developed liver metastases after baseline is summarized in Supplementary Table 1.
Logistic regression analysis revealed significant associations between several clinical factors and the risk of liver metastasis at baseline (Table 2). Diabetes mellitus emerged as a significant factor; the likelihood of liver metastasis decreased in patients with diabetes mellitus (OR = 0.421, 95%CI: 0.233-0.758, P = 0.004). Tumor size was another critical factor, with larger tumors correlating with an increased risk of liver metastasis (OR = 1.037, 95%CI: 1.021-1.054, P < 0.001). The HSI, our primary measure of hepatic steatosis, did not demonstrate a significant correlation with liver metastasis. This lack of association was consistent even when both thresholds were used (> 30 or > 36). A CA19-9 level > 260 U/mL (OR = 1.747, 95%CI: 1.035-2.948, P = 0.037) was positively correlated with liver metastasis. In subgroup analysis according to tumor location, HSI was not associated with liver metastasis in both of head and body-tail tumors (OR = 1.101, 95%CI: 0.991-1.029, P = 0.299 for head tumor and OR = 1.004, 95%CI: 0.987-1.022, P = 0.646 for body-tail tumor). The investigation of risk factors differentiating patients with new liver metastases from those without is presented in Supplementary Table 2.
| Variables | Univariate | Multivariate | ||||
| OR | 95%CI | P value | OR | 95%CI | P value | |
| Age (years) | 0.991 | 0.982-1.000 | 0.053 | |||
| Male | 1.146 | 0.942-1.394 | 0.174 | |||
| Diabetes mellitus | 0.819 | 0.673-0.996 | 0.046 | 0.421 | 0.233-0.758 | 0.004 |
| Hypertension | 1.054 | 0.866-1.284 | 0.598 | |||
| Cirrhosis | 0.748 | 0.368-1.522 | 0.424 | |||
| Body mass index (kg/m2) | 1.014 | 0.983-1.046 | 0.373 | |||
| Tumor size | 1.026 | 1.018-1.034 | < 0.001 | 1.037 | 1.021-1.054 | < 0.001 |
| HSI | 1.009 | 0.995-1.023 | 0.217 | |||
| HSI > 30 | 0.969 | 0.762-1.232 | 0.798 | |||
| HSI > 36 | 1.075 | 0.876-1.319 | 0.490 | |||
| TG (mg/dL) | 1.000 | 0.998-1.003 | 0.852 | |||
| HDL (mg/dL) | 0.986 | 0.974-0.999 | 0.029 | 0.983 | 0.966-1.001 | 0.059 |
| LDL (mg/dL) | 0.996 | 0.991-1.001 | 0.165 | |||
| CA19-9 > 260 (U/mL) | 1.935 | 1.582-2.367 | < 0.001 | 1.747 | 1.035-2.948 | 0.037 |
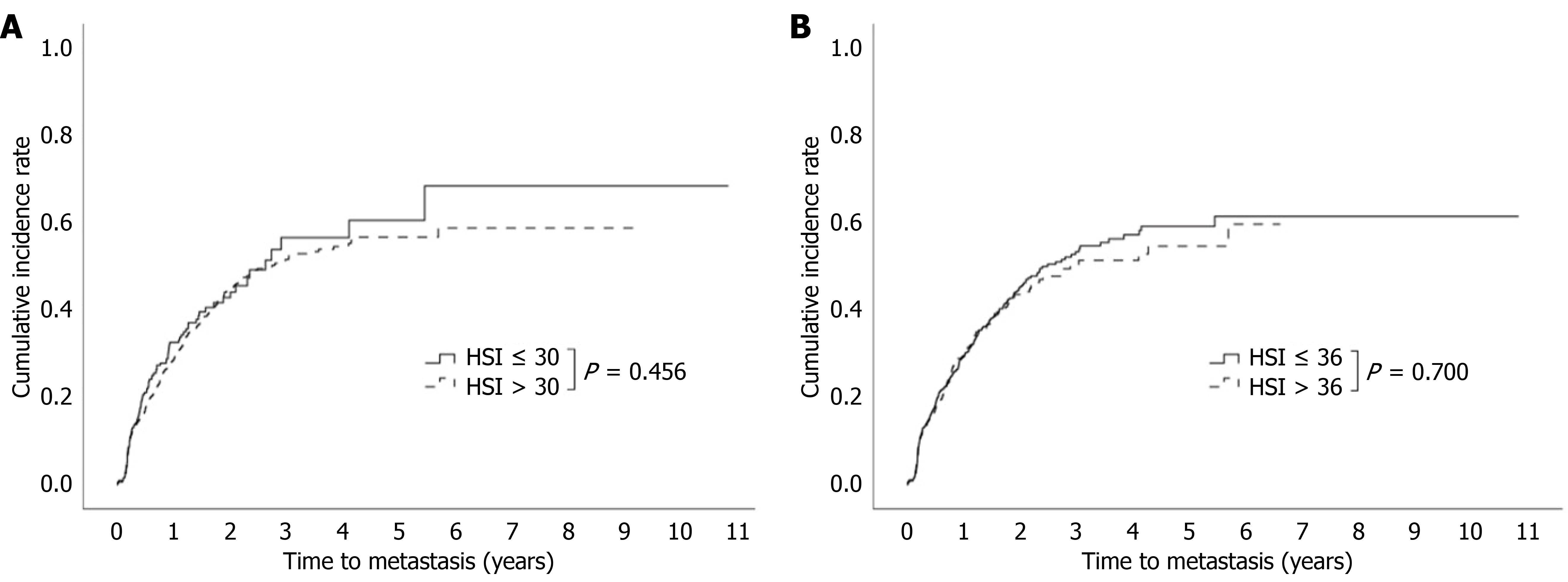
During the follow-up period (9.7 months, IQR: 3.7-18.3), 502 patients experienced newly developed liver metastasis (Table 3). After excluding patients with liver metastasis at baseline, the risk factors for newly developed liver metastasis were investigated using the Cox proportional hazards model. The CA19-9 levels were categorized using their respective median values as cutoff points. In univariate analysis, age [hazard ratio (HR) = 0.987, 95%CI: 0.979-0.995, P = 0.002], tumor size (HR = 1.013, 95%CI: 1.006-1.020, P < 0.001), and CA19-9 > 260 U/mL (HR = 1.662, 95%CI: 1.393-1.984, P < 0.001) correlated with the liver metastasis. Significance was maintained in multivariate analysis. In contrast, the HSI did not constitute a prognostic factor for liver metastasis, regardless of cutoffs (P = 0.457 for HSI > 30 and P = 0.700 for HSI > 36; Figure 1).
| Variables | Univariate | Multivariate | ||||
| HR | 95%CI | P value | HR | 95%CI | P value | |
| Age (years) | 0.987 | 0.979-0.995 | 0.002 | 0.985 | 0.974-0.997 | 0.016 |
| Male | 0.882 | 0.740-1.051 | 0.159 | |||
| Diabetes mellitus | 0.853 | 0.714-1.019 | 0.080 | |||
| Hypertension | 1.080 | 0.906-1.287 | 0.389 | |||
| Cirrhosis | 1.084 | 0.152-7.711 | 0.936 | |||
| Body mass index (kg/m2) | 1.000 | 0.971-1.029 | 0.977 | |||
| Tumor size | 1.013 | 1.006-1.020 | < 0.001 | 1.008 | 1.001-1.016 | 0.031 |
| HSI | 1.004 | 0.990-1.019 | 0.558 | |||
| HSI > 30 | 0.921 | 0.741-1.144 | 0.457 | |||
| HSI > 36 | 0.964 | 0.800-1.161 | 0.700 | |||
| TG (mg/dL) | 1.000 | 0.999-1.002 | 0.733 | |||
| HDL (mg/dL) | 0.994 | 0.985-1.003 | 0.164 | |||
| LDL (mg/dL) | 1.000 | 0.997-1.003 | 0.943 | |||
| CA19-9 > 260 (U/mL) | 1.662 | 1.393-1.984 | < 0.001 | 1.662 | 1.279-2.160 | < 0.001 |
In total, 819 (38.6%) patients died during follow-up. The Cox proportional hazards model identified several key pre
| Variables | Univariate | Multivariate | ||||
| HR | 95%CI | P value | HR | 95%CI | P value | |
| Age (years) | 1.019 | 1.012-1.026 | < 0.001 | 1.014 | 0.997-1.031 | 0.097 |
| Male | 1.079 | 0.941-1.238 | 0.276 | |||
| Diabetes mellitus | 0.652 | 0.567-0.750 | < 0.001 | 0.525 | 0.358-0.769 | 0.001 |
| Hypertension | 1.038 | 0.905-1.191 | 0.593 | |||
| Cirrhosis | 1.497 | 0.374-5.996 | 0.569 | |||
| Body mass index (kg/m2) | 0.980 | 0.958-1.002 | 0.078 | |||
| Tumor size | 1.016 | 1.011-1.021 | < 0.001 | 1.025 | 1.014-1.035 | < 0.001 |
| HSI | 0.997 | 0.985-1.009 | 0.591 | |||
| HSI > 30 | 0.807 | 0.685-0.952 | 0.011 | 1.007 | 0.650-1.561 | 0.975 |
| HSI > 36 | 0.945 | 0.817-1.094 | 0.451 | |||
| TG (mg/dL) | 0.998 | 0.996-1.000 | 0.027 | 0.997 | 0.994-1.000 | 0.026 |
| HDL (mg/dL) | 0.992 | 0.984-1.000 | 0.040 | 0.988 | 0.977-0.998 | 0.023 |
| LDL (mg/dL) | 1.000 | 0.997-1.002 | 0.966 | |||
| CA19-9 > 260 (U/mL) | 1.825 | 1.587-2.099 | < 0.001 | 1.443 | 1.057-1.970 | 0.021 |
In patients with liver metastasis, older age (HR = 1.021, 95%CI: 1.001-1.041, P = 0.043), diabetes mellitus (HR = 0.648, 95%CI: 0.423-0.992, P = 0.046), tumor size (HR = 1.017, 95%CI: 1.005-1.029, P = 0.007), and triglyceride levels (HR = 0.997, 95%CI: 0.994-1.000, P = 0.031) were correlated with the mortality risk (Table 5).
| Variables | Univariate | Multivariate | ||||
| HR | 95%CI | P value | HR | 95%CI | P value | |
| Age (years) | 1.019 | 1.009-1.028 | < 0.001 | 1.021 | 1.001-1.041 | 0.043 |
| Male | 0.975 | 0.816-1.165 | 0.782 | |||
| Diabetes mellitus | 0.682 | 0.569-0.819 | < 0.001 | 0.648 | 0.423-0.992 | 0.046 |
| Hypertension | 0.989 | 0.828-1.182 | 0.904 | |||
| Cirrhosis | 4.304 | 1.071-17.290 | 0.040 | |||
| Body mass index (kg/m2) | 0.993 | 0.964-1.023 | 0.639 | |||
| Tumor size | 1.013 | 1.006-1.020 | < 0.001 | 1.017 | 1.005-1.029 | 0.007 |
| HSI | 1.000 | 0.986-1.016 | 0.953 | |||
| HSI > 30 | 0.820 | 0.661-1.018 | 0.072 | |||
| HSI > 36 | 0.993 | 0.822-1.199 | 0.942 | |||
| TG (mg/dL) | 0.997 | 0.995-1.000 | 0.022 | 0.997 | 0.994-1.000 | 0.031 |
| HDL (mg/dL) | 0.998 | 0.987-1.008 | 0.667 | |||
| LDL (mg/dL) | 0.997 | 0.992-1.002 | 0.199 | |||
| CA19-9 > 260 (U/mL) | 1.690 | 1.407-2.029 | < 0.001 | 1.312 | 0.892-1.928 | 0.168 |
To further investigate the relationship among MASLD, liver metastasis at diagnosis, and newly developed liver metastasis during follow-up, we conducted an additional assessment of the MASLD status using CT imaging. The clinical data of the 452 patients are presented in Supplementary Table 3. Among the 452 patients, 117 (25.9%) were found to have liver metastasis at the time of diagnosis, whereas 97 (21.5%) of those without liver metastasis at baseline developed new liver metastases during follow-up. The analysis revealed that MASLD, as assessed using CT imaging, was not sig
This study showed no significant correlation between hepatic steatosis as measured by HSI and liver metastasis in patients with pancreatic cancer. This finding is particularly intriguing given the established role of the liver in metabolic syndromes and the prevalent assumption that hepatic conditions, such as steatosis, influence metastatic processes. The results of this study suggest a more complex interaction between pancreatic cancer and hepatic steatosis than was previously understood.
Contrary to our findings, several studies have reported a significant association between MASLD and liver metastasis in other types of cancer. In colorectal cancer, clinical studies have reported a protective role of hepatic steatosis against liver metastasis, whereas basic research has described the negative adverse impact of MASLD on liver metastasis[15]. In patients with breast cancer, hepatic metastasis-free survival is longer in patients with MASLD[16]. But, in cases of non-small cell lung cancer, it has been reported that patients with MASLD have a greater risk of liver metastasis[17]. Although several studies have evaluated the relationship between MASLD and liver metastasis, their results have been inconclusive and inconsistent among various types of cancer and research methods[15].
The disparity between our results and those of other studies may be attributable to the distinct pathologies and metastatic mechanisms of pancreatic and other cancers. Pancreatic cancer is known for its aggressive nature and propensity for early metastasis and often involves multiple pathways and factors that may override the influence of hepatic steatosis. Although little research has been conducted on the relationship between MALD and liver metastasis in patients with pancreatic cancer, Javed et al[18] reported an association between liver disease and hepatic recurrence-free survival in a cohort of patients who underwent resection for primary pancreatic cancer. Hepatic recurrence-free survival did not differ between patients without liver disease and those with MASLD or perfusion changes.
Recently, several studies have suggested hypotheses regarding the risk potential of MASLD as a premetastatic niche in cancers other than pancreatic cancer[19,20]. MASLD can provide energy to metastatic tumors via tumor-induced triglyceride lipolysis, and communication between MASLD and primary cancer cells via extracellular vesicles and particles can accelerate hepatic steatosis and primary cancer metastasis. However, there is no basic research regarding the impact of MASLD on liver metastasis in pancreatic cancer, and we believe that the lack of a significant relationship between the HSI and liver metastasis in our study might be related to the aggressive nature of pancreatic cancer. Its ability to modulate distant microenvironments, including the liver, may overwhelm the impact of preexisting liver conditions such as steatosis.
Diabetes is a well-known risk factor for development of pancreatic cancer. In particular, new-onset diabetes could be an alarm sign for underlying pancreatic cancer. Patients with diabetes show worse pathology, larger tumor size, and poor postoperative prognosis[21]. However, in a previous meta-analysis, long-standing diabetes was not associated with decreased survival in patients with advanced pancreatic cancer[22]. In our cohort, diabetes was not associated with liver metastasis but was associated with positive survival outcomes, even in patients with liver metastasis. In our opinion, this phenomenon may be attributable to early diagnosis of pancreatic cancer in patients with diabetes.
Our study confirmed that younger patients had more aggressive tumor features; however, survival was poor in older patients[23]. Short survival of older patients may be correlated with their poor general condition to endure chemotherapy or surgery. Therefore, younger patients are more suitable for the treatment of pancreatic cancer than older patients, even though they have oligometastatic liver lesions.
This retrospective study inherently limited the ability to establish causality. Although practical, the use of HSI as an indirect measure of hepatic steatosis may not capture the complexity of liver pathology as accurately as histological examination. Also, HSI may have limited sensitivity to the impact of MASLD in patients with pancreatic cancer because the HSI was originally derived and validated primarily in non-cancer populations. Specifically, HSI relies on BMI and transaminases, which can be substantially perturbed by cancer cachexia (weight loss/sarcopenia), systemic inflammation, and cholestasis/obstruction. These factors may lower BMI and/or affect ALT/AST, leading to misclassification of steatosis. In addition, our cohort was not limited to surgical cases, pathological variables (lymph node status, lym
The emergence of precision medicine in cancer treatment underscores the need for a detailed understanding of the individual variations in disease progression. Our study contributes to this paradigm by highlighting the fact that hepatic steatosis may not be a one-size-fits-all prognostic factor for pancreatic cancer. Customizing patient management based on a constellation of factors rather than relying on single variables could enhance treatment efficacy and outcomes.
In conclusion, our study revealed that hepatic steatosis, assessed using the HSI, may not have a direct role in liver metastasis in pancreatic cancer. However, its association with patient survival is complex and requires further investigation. This study sheds light on the complex interplay between pancreatic cancer development and systemic conditions, such as hepatic steatosis.
| 1. | Stoffel EM, Brand RE, Goggins M. Pancreatic Cancer: Changing Epidemiology and New Approaches to Risk Assessment, Early Detection, and Prevention. Gastroenterology. 2023;164:752-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 404] [Cited by in RCA: 363] [Article Influence: 121.0] [Reference Citation Analysis (3)] |
| 2. | Takeda T, Sasaki T, Okamoto T, Kasuga A, Matsuyama M, Ozaka M, Inoue Y, Takahashi Y, Saiura A, Sasahira N. Outcomes of pancreatic cancer with liver oligometastasis. J Hepatobiliary Pancreat Sci. 2023;30:229-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 3. | Teng ML, Ng CH, Huang DQ, Chan KE, Tan DJ, Lim WH, Yang JD, Tan E, Muthiah MD. Global incidence and prevalence of nonalcoholic fatty liver disease. Clin Mol Hepatol. 2023;29:S32-S42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 576] [Cited by in RCA: 498] [Article Influence: 166.0] [Reference Citation Analysis (3)] |
| 4. | Ko E, Yoon EL, Jun DW. Risk factors in nonalcoholic fatty liver disease. Clin Mol Hepatol. 2023;29:S79-S85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (1)] |
| 5. | Zhang C, Zhang Y, Dong Y, Zi R, Wang Y, Chen Y, Liu C, Wang J, Wang X, Li J, Liang H, Ou J. Non-alcoholic fatty liver disease promotes liver metastasis of colorectal cancer via fatty acid synthase dependent EGFR palmitoylation. Cell Death Discov. 2024;10:41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (1)] |
| 6. | Mantovani A, Petracca G, Beatrice G, Csermely A, Tilg H, Byrne CD, Targher G. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: a meta-analysis of observational cohort studies. Gut. 2022;71:778-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 254] [Article Influence: 63.5] [Reference Citation Analysis (1)] |
| 7. | Muhidin SO, Magan AA, Osman KA, Syed S, Ahmed MH. The relationship between nonalcoholic fatty liver disease and colorectal cancer: the future challenges and outcomes of the metabolic syndrome. J Obes. 2012;2012:637538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | Wang Z, Kim SY, Tu W, Kim J, Xu A, Yang YM, Matsuda M, Reolizo L, Tsuchiya T, Billet S, Gangi A, Noureddin M, Falk BA, Kim S, Fan W, Tighiouart M, You S, Lewis MS, Pandol SJ, Di Vizio D, Merchant A, Posadas EM, Bhowmick NA, Lu SC, Seki E. Extracellular vesicles in fatty liver promote a metastatic tumor microenvironment. Cell Metab. 2023;35:1209-1226.e13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 137] [Article Influence: 45.7] [Reference Citation Analysis (0)] |
| 9. | Park JH, Hong JY, Han K, Kang W, Park JK. Increased risk of pancreatic cancer in individuals with non-alcoholic fatty liver disease. Sci Rep. 2022;12:10681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 24] [Reference Citation Analysis (1)] |
| 10. | Bojmar L, Zambirinis CP, Hernandez JM, Chakraborty J, Shaashua L, Kim J, Johnson KE, Hanna S, Askan G, Burman J, Ravichandran H, Zheng J, Jolissaint JS, Srouji R, Song Y, Choubey A, Kim HS, Cioffi M, van Beek E, Sigel C, Jessurun J, Velasco Riestra P, Blomstrand H, Jönsson C, Jönsson A, Lauritzen P, Buehring W, Ararso Y, Hernandez D, Vinagolu-Baur JP, Friedman M, Glidden C, Firmenich L, Lieberman G, Mejia DL, Nasar N, Mutvei AP, Paul DM, Bram Y, Costa-Silva B, Basturk O, Boudreau N, Zhang H, Matei IR, Hoshino A, Kelsen D, Sagi I, Scherz A, Scherz-Shouval R, Yarden Y, Oren M, Egeblad M, Lewis JS, Keshari K, Grandgenett PM, Hollingsworth MA, Rajasekhar VK, Healey JH, Björnsson B, Simeone DM, Tuveson DA, Iacobuzio-Donahue CA, Bromberg J, Vincent CT, O'Reilly EM, DeMatteo RP, Balachandran VP, D'Angelica MI, Kingham TP, Allen PJ, Simpson AL, Elemento O, Sandström P, Schwartz RE, Jarnagin WR, Lyden D. Multi-parametric atlas of the pre-metastatic liver for prediction of metastatic outcome in early-stage pancreatic cancer. Nat Med. 2024;30:2170-2180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 44] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 11. | Kang SH, Lee HW, Yoo JJ, Cho Y, Kim SU, Lee TH, Jang BK, Kim SG, Ahn SB, Kim H, Jun DW, Choi JI, Song DS, Kim W, Jeong SW, Kim MY, Koh H, Jeong S, Lee JW, Cho YK; Korean Association for the Study of the Liver (KASL). KASL clinical practice guidelines: Management of nonalcoholic fatty liver disease. Clin Mol Hepatol. 2021;27:363-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 190] [Article Influence: 38.0] [Reference Citation Analysis (1)] |
| 12. | Lee JH, Kim D, Kim HJ, Lee CH, Yang JI, Kim W, Kim YJ, Yoon JH, Cho SH, Sung MW, Lee HS. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010;42:503-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1338] [Cited by in RCA: 1222] [Article Influence: 76.4] [Reference Citation Analysis (3)] |
| 13. | Kim HN, Jeon HJ, Choi HG, Kwon IS, Rou WS, Lee JE, Lee TH, Kim SH, Lee BS, Shin KS, Lee HJ, Eun HS. CT-based Hounsfield unit values reflect the degree of steatohepatitis in patients with low-grade fatty liver disease. BMC Gastroenterol. 2023;23:77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 14. | Chang JW, Lee HW, Kim BK, Park JY, Kim DY, Ahn SH, Han KH, Kim SU. Hepatic Steatosis Index in the Detection of Fatty Liver in Patients with Chronic Hepatitis B Receiving Antiviral Therapy. Gut Liver. 2021;15:117-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (2)] |
| 15. | Lv Y, Patel N, Zhang HJ. The progress of non-alcoholic fatty liver disease as the risk of liver metastasis in colorectal cancer. Expert Rev Gastroenterol Hepatol. 2019;13:1169-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 16. | Wu W, Chen J, Ye W, Li X, Zhang J. Fatty liver decreases the risk of liver metastasis in patients with breast cancer: a two-center cohort study. Breast Cancer Res Treat. 2017;166:289-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 17. | Wu W, Liao H, Ye W, Li X, Zhang J, Bu J. Fatty liver is a risk factor for liver metastasis in Chinese patients with non-small cell lung cancer. PeerJ. 2019;7:e6612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (3)] |
| 18. | Javed A, Baig E, Habib J, C C, Lafaro K, Greer J, Burkhart R, Burns W, Cohen S, Fishman E, Cameron J, He J, Weiss M, Wolfgang C. Impact of liver disease and parenchymal changes on patterns of liver recurrence in patients with resected PDAC: a new insight into the biological behavior of this lethal disease. HPB. 2021;23:S505. [DOI] [Full Text] |
| 19. | Kim SJ, Hyun J. Altered lipid metabolism as a predisposing factor for liver metastasis in MASLD. Mol Cells. 2024;47:100010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 20. | Li Y, Su X, Rohatgi N, Zhang Y, Brestoff JR, Shoghi KI, Xu Y, Semenkovich CF, Harris CA, Peterson LL, Weilbaecher KN, Teitelbaum SL, Zou W. Hepatic lipids promote liver metastasis. JCI Insight. 2020;5:e136215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 21. | Hank T, Sandini M, Qadan M, Weniger M, Ciprani D, Li A, Ferrone CR, Warshaw AL, Lillemoe KD, Fernández-Del Castillo C. Diabetes mellitus is associated with unfavorable pathologic features, increased postoperative mortality, and worse long-term survival in resected pancreatic cancer. Pancreatology. 2020;20:125-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 22. | Jeon CY, Li D, Cleary S, Stolzenberg-Solomon R, Bosetti C, La Vecchia C, Porta M, Toriola AT, Hung RJ, Kurtz RC, Olson SH. The Association of Recently Diagnosed Diabetes and Long-term Diabetes With Survival in Pancreatic Cancer Patients: A Pooled Analysis. Pancreas. 2018;47:314-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 23. | Cao BY, Tong F, Zhang LT, Kang YX, Wu CC, Wang QQ, Yang W, Wang J. Risk factors, prognostic predictors, and nomograms for pancreatic cancer patients with initially diagnosed synchronous liver metastasis. World J Gastrointest Oncol. 2023;15:128-142. [PubMed] [DOI] [Full Text] |