Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.116152
Revised: December 18, 2025
Accepted: January 13, 2026
Published online: March 14, 2026
Processing time: 118 Days and 5.9 Hours
Postoperative benign bilioenteric anastomotic stricture (BBAS) is a serious compli
To determine the efficacy and safety of percutaneous transhepatic cholangioscopy (PTCS)-assisted procedures, including stricture dilation and recanalization for BBAS and biliary stone therapy for concomitant stones.
Patients with BBAS who received PTCS-assisted procedures were included. Primary outcomes were anastomotic patency (including initial anastomotic patency after stricture dilation and cumulative anastomotic patency after PTCS catheter removal) and treatment success (i.e., negative clinical clamping trial and removable indwelling PTCS catheter). Secondary outcomes were technical success for the three PTCS-assisted procedures, duration of the indwelling PTCS catheter, and adverse events.
Forty patients were included; 32 underwent PTCS-assisted stricture dilation/recanalization and 36 underwent biliary stone therapy. The initial patency rates were 58.1% and 41.9% at 1 and 2 years, respectively, and the cumulative patency rates were 74.3%, 59.7, 49.8%, and 34.8% at 1, 3, 5, and 7 years, respectively. The treatment success rate was 90.6% (29/32) for PTCS-assisted stricture procedures. The technical success rates were 100% (31/31), 87.5% (7/8), and 94.5% (34/36) for PTCS-assisted stricture dilation, recanalization, and biliary stone therapy, respectively. The median duration of the indwelling PTCS catheter in the 29 patients with treatment success was 6 (range: 2-109) months. Only one patient experienced acute bleeding, which was manageable.
PTCS-assisted procedures are efficacious and safe for the management of BBAS and concomitant biliary stones. Placement of an indwelling PTCS catheter across the anastomosis after stricture dilation improves the anastomotic patency.
Core Tip: Forty patients with benign bilioenteric anastomotic stricture received percutaneous transhepatic cholangioscopy (PTCS)-assisted procedures, including stricture dilation/recanalization and stone therapy. The cumulative anastomotic patency due to a PTCS catheter across the anastomosis placed 6 months ago is superior to initial anastomotic patency (balloon dilation alone), i.e., 74.3% and 69.7% vs 58.1% and 41.9% at 1 and 2 years, respectively, with a treatment success rate of 90.6% (29/32). Technical success for stone therapy, including 20 patients with electrohydraulic or laser lithotripsy, was achieved in 34 (94.4%) patients. PTCS-assisted procedures are efficacious, especially for the treatment of intrahepatic stones. PTCS catheter placement across the anastomosis after stricture dilation can improve the anastomotic patency.
- Citation: Ren X, Wang C, Qu YP, Tang XF, Xia T, Lu YX, Sun XM. Percutaneous transhepatic cholangioscopy-assisted procedures for the management of postoperative benign bilioenteric anastomotic strictures with or without biliary stones. World J Gastroenterol 2026; 32(10): 116152
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/116152.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.116152
Postoperative benign bilioenteric anastomotic stricture (BBAS) is a serious complication of biliary surgeries, with an incidence of 2.6%-40%[1-3], mostly due to biliary injuries following bilioenteric anastomosis through laparoscopic cholecystectomy[4-6]. Clinically, BBAS is suspected in patients who have undergone bilioenteric anastomosis and present with cholangitis, laboratory evidence of cholestasis, and frequent bile duct dilation or biliary stones; diagnosis is confirmed through imaging studies. Although percutaneous transhepatic cholangioscopy (PTCS) is mainly a therapeutic procedure, it can be used as a useful diagnostic tool with imaging to further distinguish between benign and malignant conditions, especially in patients with an altered anatomy[7,8]. Without appropriate treatment, BBAS can lead to recurrent cholangitis, choledocholithiasis, secondary biliary cirrhosis, or even liver failure and death[2,9]. In addition, bilioenteric reconstructive surgical treatment for BBAS is associated with a high rate (20%-25%) of recurrent BBAS and a high mortality rate (2%-13%)[5]. Therefore, optimal therapeutic approaches are required for BBAS and recurrent BBAS.
Endoscopic retrograde cholangiopancreatography (ERCP) is currently the gold standard for the diagnosis and treatment of biliary disease[10]; however, it is a challenging procedure for BBAS due to the surgically altered anatomy[3,6,11,12]. Balloon dilation (BD) under ERCP assisted by balloon enteroscopy[13] or guided by endoscopic ultrasound may be considered for BBAS. However, the procedures require not only a skilled endoscopist, but also specific tools that are not always readily available, and procedure-related adverse events (AEs) are common (up to 20%)[10]. Therefore, percutaneous approaches, usually guided under percutaneous transhepatic cholangiography (PTC), are often used as the initial therapy for BBAS[3,4,6,14-16]. The currently used percutaneous treatment procedures for BBAS include the place
Moreover, recurrent BBAS occurs after anastomotic BD, especially in patients with extensive fibrosis and scarring at the anastomotic site[5]. It is believed that the placement of a large-size draining catheter is more important than BD for the treatment of BBAS and the prevention of recurrent BBAS[6]. However, there is no consensus on the optimal diameter of the catheter or the optimal indwelling duration[3,4,6,15,19,20].
We applied PTCS to treat patients with various biliary tract diseases including BBAS in 1998[21]. Since then, approximately 150 patients with BBAS were diagnosed. Whereas PTBD-guided BD therapy or balloon-assisted enteroscopy was performed in patients with simple BBAS with no or only small (< 1 cm) and single stones in the extrahepatic biliary duct, PTCS-assisted procedures with a large-size indwelling catheter were applied in patients with complex or severe strictures or those concomitant with biliary stones, especially, multiple or intrahepatic duct (IHD) stones, in addition to PTBD. The aim of the present retrospective study was to determine the efficacy and safety of PTCS-assisted procedures including stricture dilation and recanalization for BBAS and biliary stone therapy for concomitant stones.
Adult (≥ aged 18 years) hospitalized patients who were diagnosed with BBAS after Roux-en-Y bilioenterostomy, underwent PTCS-assisted procedures, and were regularly followed up between August 1998 and December 2023 were included. Specific inclusion criteria were as follows: (1) Patients with symptomatic BBAS after Roux-en-Y bilioenterostomy and concomitant multiple stones in intra- and extrahepatic biliary ducts or large biliary stones (> 1.5 cm); (2) Patients with unsuccessful PTBD-guided anastomotic recanalization or stone removal due to almost or completely closed anastomosis or stones > 1 cm; and (3) Patients with unsuccessful treatment using balloon-assisted enteroscopy (i.e., unable to reach the anastomosis, dilate the anastomosis, or remove the stones). BBAS was diagnosed by clinical manifestations, laboratory and imaging examination such as abdominal ultrasound, computed tomography (CT), or magnetic resonance cholangiopancreatography, or PTBD. Patients without intra- and extrahepatic biliary duct dilation, those with a single stone ≤ 1 cm or < 3 small stones, those with severe coagulation dysfunction that could not be corrected, or those with ascites were excluded. The study protocol was approved by the Ethics Committee of Heilongjiang Provincial Hospital.
Demographic and clinical data and laboratory and PTC and PTCS findings were collected. BBAS was graded as mild, moderate, or severe based on PTCS and PTC, during which a contrast medium was used to indicate the patency of the anastomosis. A mild stricture was defined as a stricture with mild proximal bile duct dilation of the anastomosis, but the cholangioscope was able to pass through the stricture. A severe stricture was defined when the anastomotic stricture was focal tight or completely closed, with a very slow (incomplete drainage within 5 minutes) or unable passage of the contrast medium to the anastomosis. Then, a moderate stricture was defined when the severity of the anastomotic stricture was between the mild and severe grades described above. BBAS was also classified as simple, defined as a BBAS without an additional stricture in the hilar biliary duct or in the IHD, or complex, defined as a BBAS with one or more additional strictures in the above-mentioned locations.
PTBD and percutaneous sinus tract dilation with bougie as well as the placement of an indwelling PTCS catheter (18-Fr or 20-Fr, Sumitomo, Tokyo, Japan) were performed as previously described[22]. Then, PTCS was carried out 2 weeks after PTBD by using a percutaneous cholangioscope with a 2-mm working channel (ED-270F, Fujifilm, Tokyo, Japan).
Three major PTCS-assisted procedures, including stricture dilation, recanalization, and biliary stone fragmentation/removal, were applied; biopsy was performed under PTCS when necessary.
PTCS-assisted stricture dilation was performed with a balloon or a bougie for an anastomotic stricture with a slow or unable passage of the contrast medium. Briefly, for a simple anastomotic stricture, a balloon dilator (CRE, Boston Scientific, United States, or QBD, Cook Medical, United States) was used over a 0.035-inch guidewire to dilate the stric
PTCS-assisted recanalization was performed for some cases with severe strictures for which the guidewire could not pass through the anastomosis. It was performed with a 0.035-inch guidewire, a 5-Fr microcatheter (≤ 3 Fr at the tip) and micro-guidewire (0.018-inch or 0.025-inch) system, a 4 mm 25-gauge injection needle (NM-201 L-0425, Olympus, Japan), or a needle knife, depending on the features of the stricture (Supplementary material).
PTCS-assisted stone fragmentation/removal procedures were performed for concomitant biliary stones. Small or fragmented stones were removed using a basket or pushed through the anastomosis by a cholangioscope. For impacted bile duct stones, or stones unremovable via the sinus tract or anastomosis (e.g., > 1 cm stones), PTCS-assisted electrohydraulic lithotripsy (PTCS-EHL) (Lithotron, EL-27, Walz Electronic Inc. Rohrdorf, Germany) or PTCS-assisted laser lithotripsy (PTCS-LL) (Laser U100 Plus, W.O.M, Berlin, Germany) was applied. In patients with the stricture near the stone that interfered with the entry of the choledoscope and stone removal, PTCS-guided BD was performed prior to lithotripsy or stone removal.
After the first successful attempt for BD and complete stone clearance, the indwelling PTCS catheter was left above the anastomosis or in the IHD. However, if the first BD attempt failed, the indwelling PTCS catheter needed to be placed across the anastomosis. All patients with anastomotic patency were re-evaluated with a clinical clamping trial or PTC[6] at approximately 2-3 months after the stricture dilation. During the catheter clamping trial, an 18-20-Fr PTCS catheter was placed above the anastomosis, and then the PTCS catheter was clamped for 2 weeks. If the clamping trial was positive, or PTC showed incomplete drainage of the contrast medium, anastomotic dilation was repeated[6] or the indwelling PTCS catheter was placed via the anastomosis.
The primary outcomes were anastomotic patency and treatment success. Anastomotic patency was defined as no signs or symptoms of cholangitis and no cholestasis according to the liver function test, or PTC confirmed the anastomotic patency (i.e., the proper flow of the contrast medium through the anastomosis with complete drainage within 5 minutes). In the present study, anastomotic patency was represented by the initial and cumulative anastomotic patency rates. The initial anastomotic patency rates were defined as the proportions of patients who achieved anastomotic patency at 1 year and 2 years after successful anastomotic BD alone, respectively. The cumulative anastomotic patency rates were defined as the proportions of patients who achieved anastomotic patency at 1, 2, 3, 5, and 7 years after indwelling PTCS catheter placement across the anastomosis and its successful removal. Treatment success was defined when the clinical clamping trial which was performed 3-4 months after the indwelling PTCS catheter across the anastomosis was negative, and the indwelling PTCS catheter was able to be removed.
Secondary outcomes included technical success for PTCS-assisted stricture dilation and PTCS-assisted recanalization prior to stricture dilation, initial and overall technical success for PTCS-assisted stricture procedures, technical success for PTCS-assisted biliary stone therapy, duration of an indwelling PTCS catheter, and PTCS-assisted procedure-related AEs. Technical success for stricture dilation was defined when the balloon waist disappeared during the inflation[5,6] or the anastomosis was dilated to up to 18-20 Fr with a bougie. Technical success for recanalization was defined as the success of a recanalization technique (i.e., the PTCS-assisted guidewire or catheter successfully passed through the narrowed or occluded anastomosis and entered the jejunum) before anastomotic stricture dilation was performed. The initial technical success for PTCS-assisted stricture procedures was defined as technical success for PTCS-assisted stricture dilation without a prior recanalization technique. Overall technical success for PTCS-assisted stricture procedures was defined as the technical success of any of two PTCS-assisted stricture procedures including dilation and recanalization.
Technical success for biliary stone therapy was defined when all biliary stones were completely removed as confirmed by imaging, including abdominal ultrasound, CT, PTC, and PTCS. The duration of the indwelling PTCS catheter was defined as the time interval between the PTCS catheter placement across the anastomosis and the catheter removal. The PTCS catheter was removed when the clinical clamping trial was negative (i.e., there were no signs or symptoms of cholangitis and no cholestasis according to the liver function test at 2 weeks after clamping the catheter), and PTC confirmed the anastomotic patency (i.e., the proper flow of the contrast medium through the anastomosis with complete drainage within 5 minutes)[6,15]. However, for patients with failure of the first BD session for the anastomosis, bougie dilation alone, residual or potential stones, and recurrence of BBAS, the duration of catheter indwelling was prolonged for at least 3 months, even with confirmed anastomotic patency. If the clamping trial was positive, a repeated anastomotic dilation was performed, and the catheter via the stricture was kept for at least 3 months until a negative result was obtained in the clamping trial.
PTCS-assisted procedure-related AEs during the three stages (namely, PTBD, sinus tract dilation, and the PTCS-assisted procedure)[23] were recorded, with the latter including anastomotic stricture dilation, recanalization, and stone therapy-related complications. The AEs according to the Society of Interventional Radiology Clinical Practice Guidelines[24] were classified as minor (mild) AEs (defined as complications with no therapy, nominal therapy, or no consequence) and major AEs (defined as complications that required therapy, hospitalization < 48 hours, or those with an unplanned increase in the level of care, hospitalization > 48 hours, permanent adverse sequelae, or death). Specifically, for PTCS-assisted procedures, the related minor (mild) AEs included cholangitis, bacteremia, right pleural effusion, catheter migration; and major (serious) AEs included severe hemobilia, defined as clinically significant bleeding requiring trans
All patients were followed up for clinical evaluation by hospital visits every 2-3 months within the first year, and then once a year by telephone, or anytime when fever, jaundice, or abdominal pain occurred, suggesting the recurrence of a stone or recurrent BBAS; recurrent BBAS was diagnosed according to the criteria for BBAS[6].
All data were analyzed in statistics analysis system (version 9.4, Cary, NC, United States). Continuous data were pre
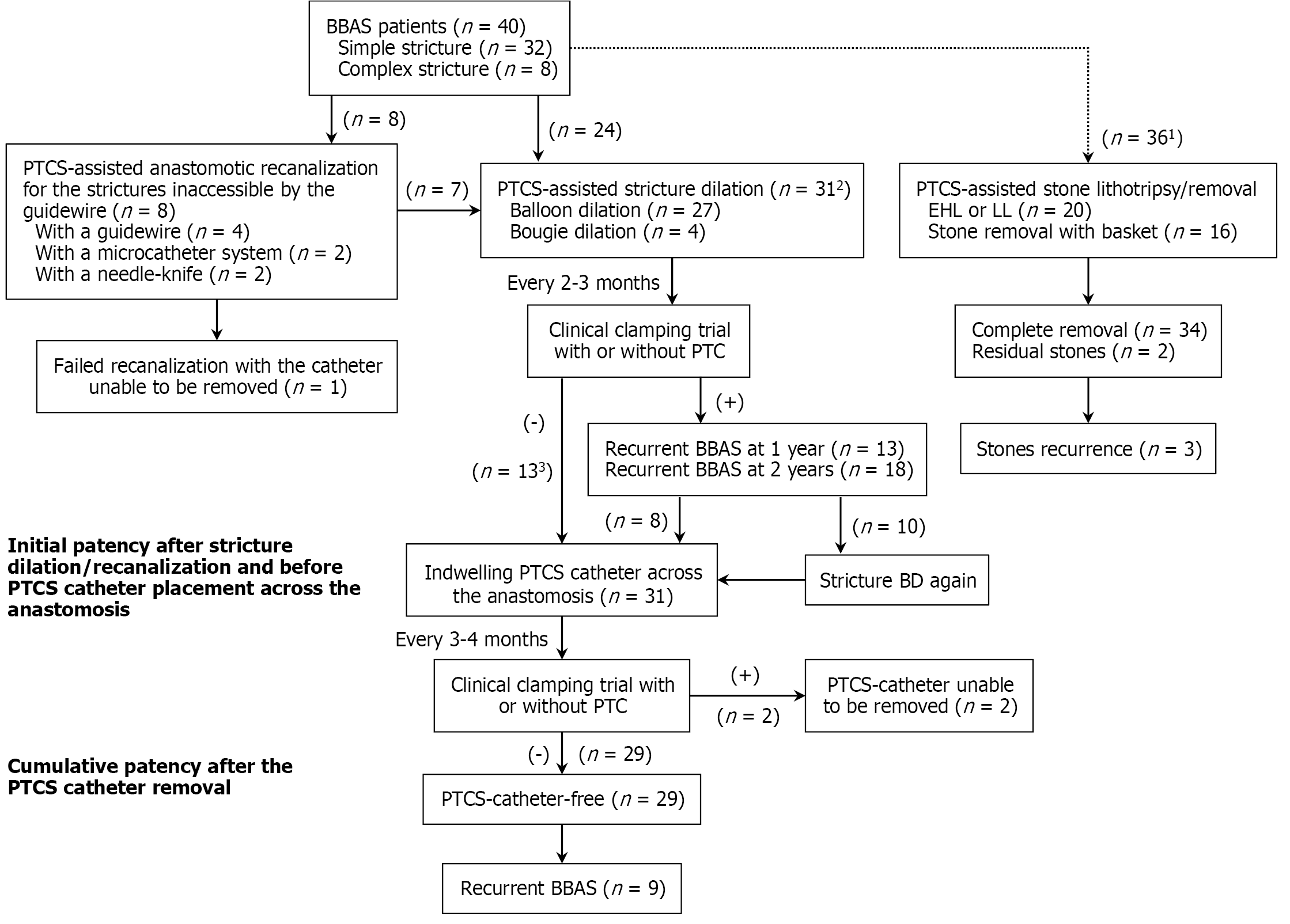
During the present study, 148 patients after Roux-en-Y bilioenterostomy were diagnosed with BBAS with or without biliary stones. Of these patients, 141 met the inclusion criteria, but 101 of them were excluded due a simple anastomotic stricture without concomitant biliary stones (n = 31), only a concomitant single stone (< 1 cm) or fewer than 3 small stones (n = 46), the absence of biliary dilation (n = 15), or no intervention received (n = 9). Finally, 40 eligible patients with BBAS were included in the present study (Figure 1). Their baseline demographic and clinical characteristics, underlying diseases for Roux-en-Y bilioenterostomy, and laboratory findings are listed in Table 1.
| Characteristics | Value |
| Age (years), median (range) | 53 (25-72) |
| Sex | |
| Male | 16 (40.0) |
| Female | 24 (60.0) |
| Clinical presentation | |
| Repeated cholangitis | 36 (90.0) |
| Obstructive jaundice | 4 (10.0) |
| Underlying causes of Roux-en-Y bilioenterostomy | |
| LC with intraoperative bile duct injury | 14 (35.0) |
| Choledochal cyst | 8 (20.0) |
| Hepatolithiasis | 7 (17.5) |
| Pancreatic cystic or solid tumor | 5 (12.5) |
| Ampulloma of the papilla of Vater | 2 (5.0) |
| Others1 | 4 (10.0) |
| Time interval from bilioenteric anastomosis to BBAS treatment (months), median (range) | 66 (2-254) |
| Abnormal liver function test results (n = 40) | |
| γ-glutamyl transferase (IU/L) (upper normal limit 50 IU/L) | 38 (95.0) |
| Alkaline phosphatase (IU/L) (upper normal limit 112 IU/L) | 34 (85.0) |
| Total bilirubin (mmol/L) (upper normal limit 17.1 mmol/L) | 19 (47.5) |
| Tumor marker level increased (n = 36) | |
| CA19-9 (U/L) (normal limit 37 U/L) | 14 (38.9) |
| CEA (ng/mL) (upper normal limit 5 ng/mL) | 2 (5.6) |
The features of the anastomotic strictures, including location, grade, severity, length, and complexity, are shown in Table 2. Simple and complex strictures were present in 32 (80.0%) and 8 (20.0%) patients, respectively, and mild, moderate, and severe anastomotic strictures were present in 10 (25.0%), 11 (27.5%), and 19 (47.5%) patients, respectively. Concomitant biliary stones were observed in 36 (90.0%) patients. The size, number, location, and composition of the stones in these patients are listed in Supplementary Table 1.
| Characteristics | n (%) |
| Location of BBAS1 | |
| Common bile duct | 4 (10.0) |
| Common hepatic duct | 19 (47.5) |
| Hilar bifurcation | 15 (37.5) |
| RHD and LHD | 2 (5.0) |
| Severity of BBAS | |
| Mild | 10 (25.0) |
| Moderate | 11 (27.5) |
| Severe2 | 19 (47.5) |
| Length of BBAS (mm) | |
| ≤ 5 | 20 (76.9) |
| 6-9 | 6 (23.1) |
| ≥ 10 | 0 (0.0) |
| Complexity of BBAS | |
| Simple | 32 (80.0) |
| Complex3 | 8 (20.0) |
| Strictures opposite of PTBD | 4 (10.0) |
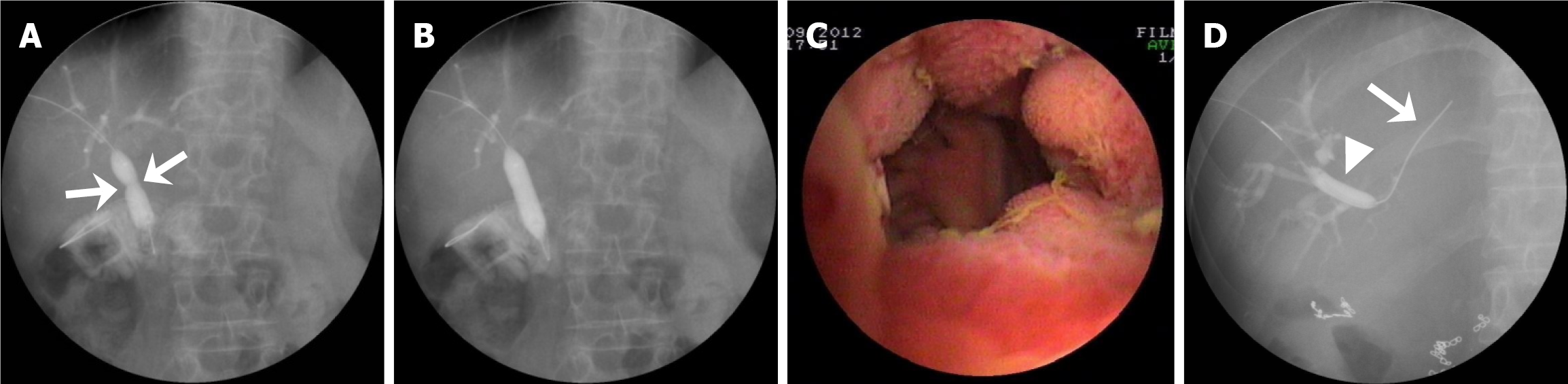
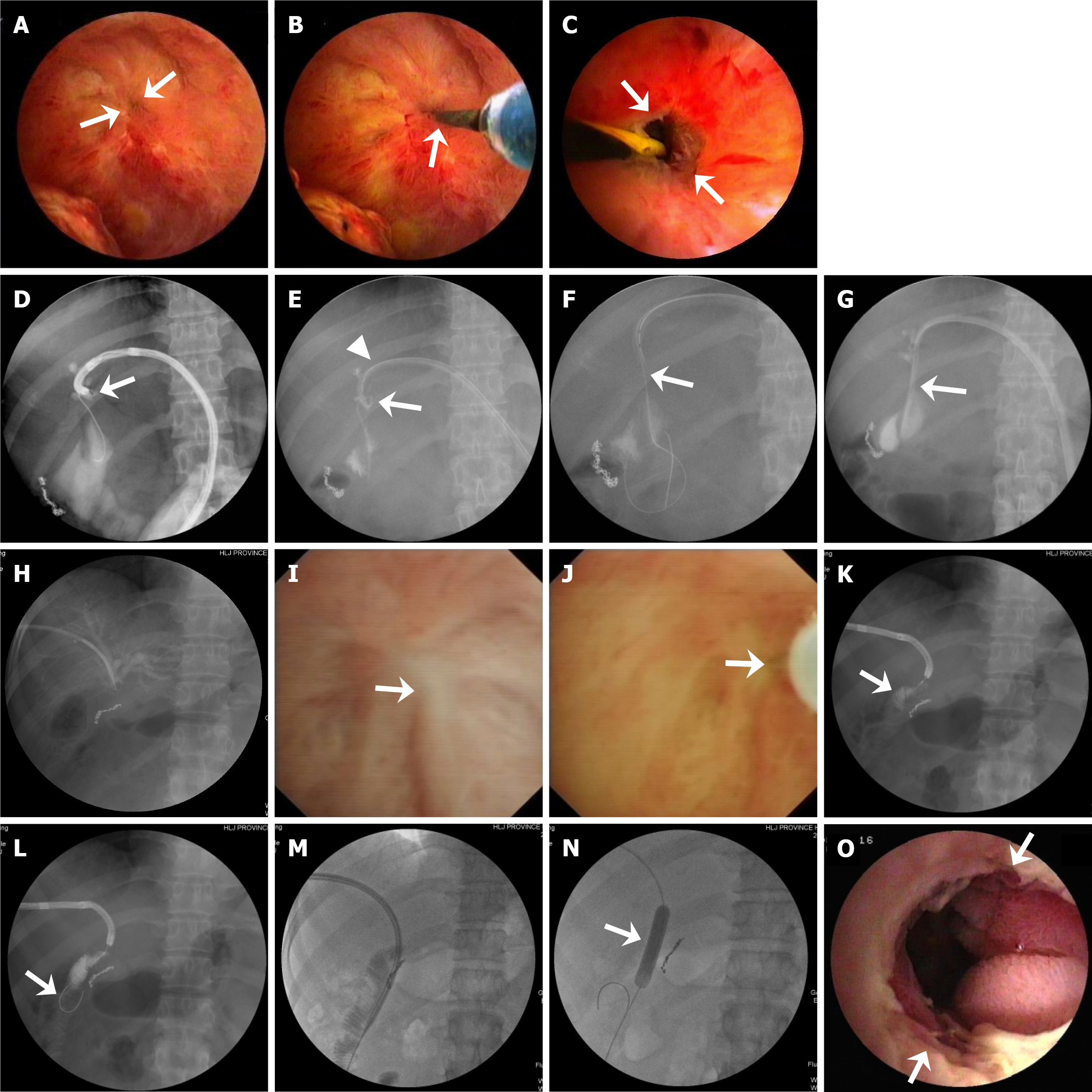
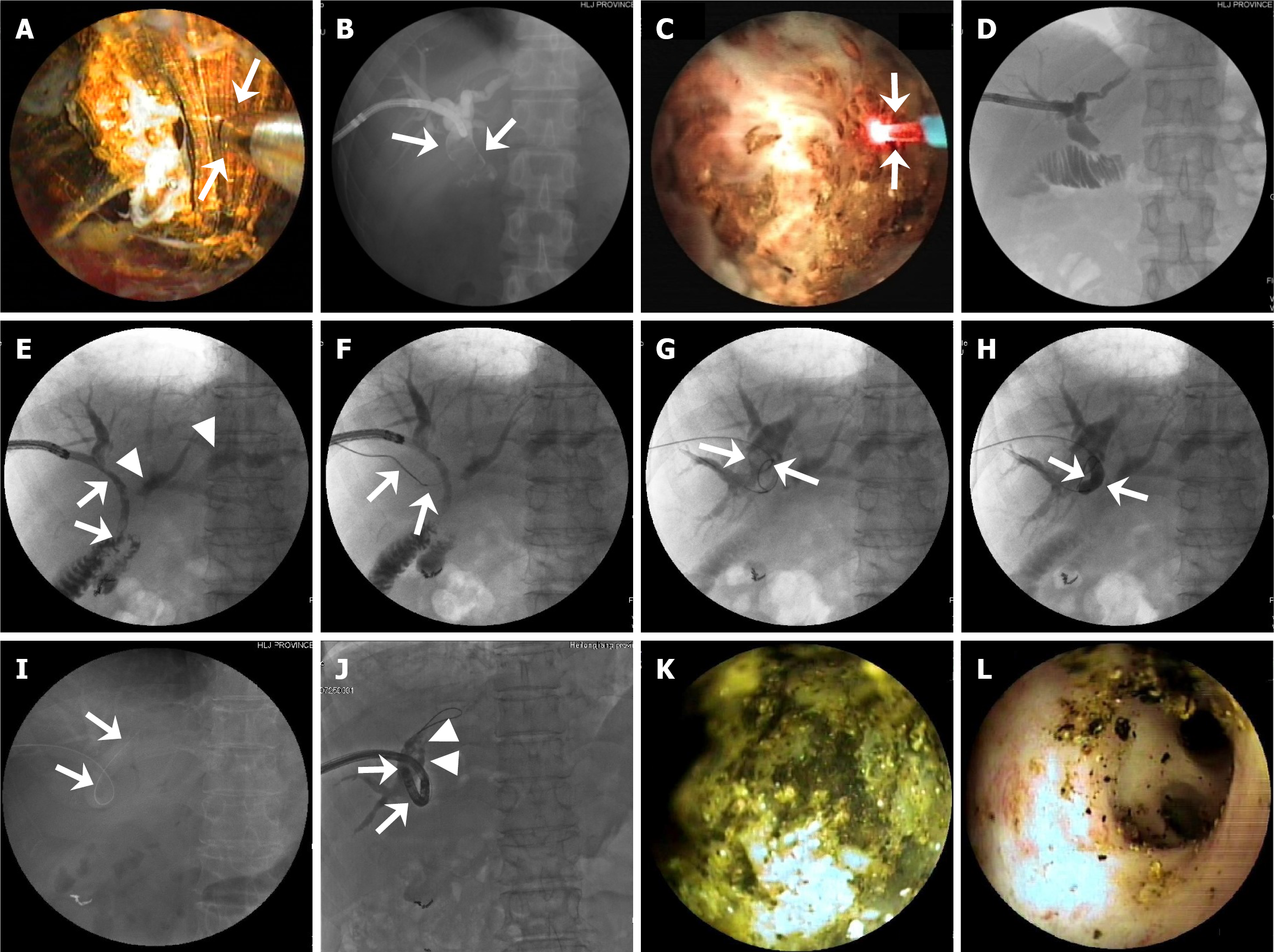
PTCS-assisted stricture dilation, anastomotic recanalization, and biliary stone therapy were performed in 31, 8, and 36 patients, respectively (Figure 1). Specifically, of the 40 patients, 31 patients (including 2 with mild, 11 with moderate, and 18 with severe anastomotic strictures) received BD (n = 27) (Figure 2A-C) or bougie dilation (n = 4). Nine patients did not receive stricture dilation due to complete anastomotic occlusion with failed anastomotic recanalization (n = 1) or mild strictures (n = 8). The eight patients with complex strictures achieved successful BD through PTCS-assisted guidewire passage beyond the stricture (Figure 2D). Among the 19 patients with a severe stricture, 11 patients received stricture dilation without pre-recanalization, and 8 patients received PTCS-assisted recanalization with a guidewire (n = 4, Figure 3A-C), microcatheter and microguidewire system (n = 2, Figure 3D-G), or a needle knife (n = 2, Figure 3H-O). The recanalization procedure was successful in seven (87.5%) of these patients who subsequently received stricture dilation by a balloon dilator (Supplementary Figure 1A and B) or a plastic bougie (Figure 3D-G). Of the 36 patients with biliary stones (9 and 27 with extrahepatic stones and intrahepatic stones, respectively), 20 and 16 patients received EHL/LL and stone removal with basket, respectively (Figure 4A-D).
Eventually, negative clinical clamping trial was achieved in 29 (93.5%) of the 31 patients, and the indwelling PTCS catheters were removed in these patients with the median indwelling duration of 6 months (range: 2-109 months). The catheter was left in the remaining two patients due to a positive clamping trial and repeated recurrence of the stricture and stones.
All 40 patients were discharged from the hospital, and the median follow-up duration was 46 months (range: 2-122 months). Of the 29 catheter-free patients, 5 were lost to follow-up within 1 year, whereas one patient with unsuccessful recanalization and two other patients with a positive clamping trial were followed up beyond 1 year (Figure 1). Of the 32 patients with stricture dilation, 27 (84.4%) were available at the latest follow-up visit. Recurrent BBAS developed in 13 (n = 31, 41.9%) and 18 (n = 31, 58.1%) patients, respectively, within 1 and 2 years after stricture dilation (Table 3). Anasto
| Outcomes | Value |
| Primary outcomes | |
| Anastomotic patency rate | |
| Initial patency rate after stricture dilation (n = 31) | |
| 1 year (3-11 months) | 18 (58.1) |
| 2 years (3-21 months) | 13 (41.9) |
| Cumulative patency rate after catheter removal (n = 29) | |
| 1 year (%) | (74.3) |
| 2 years (%) | (69.7) |
| 3 years (%) | (59.7) |
| 5 years (%) | (49.8) |
| 7 years (%) | (34.8) |
| 8 years (%) | (21.8) |
| 9 years (%) | (14.5) |
| Treatment success (n = 32) | 29 (90.6) |
| Secondary outcomes | |
| Technical success for PTCS-assisted stricture dilation (n = 31) | 31 (100.0) |
| Simple stricture for mean 1.2 BD sessions (n = 19) | 19 (100.0) |
| Complex stricture for mean 1.4 BD sessions (n = 8) | 8 (100.0) |
| Simple strictures for bougie dilation (n = 4) | 4 (100.0) |
| Initial technical success without a prior recanalization technique (n = 32) | 24 (75.0) |
| Technical success of PTCS-assisted recanalization (n = 8) | 7 (87.5) |
| With guidewire (n = 4) | 4 (100.0) |
| With microcatheter system (n = 2) | 2 (100.0) |
| With needle knife (n = 2) | 1 (50.0) |
| Overall technical success with or without a recanalization technique (n = 32) | 31 (96.9) |
| Technical success of PTCS-assisted lithotripsy/stone removal (n = 36) | 34 (94.4) |
| Stones recurred (n = 28) | 3 (10.7) |
| Successful removal of indwelling PTCS catheter (n = 32) | 291 (90.6) |
| Duration of indwelling PTCS catheter, median months (range), (n = 29) | 6 (2-109) |
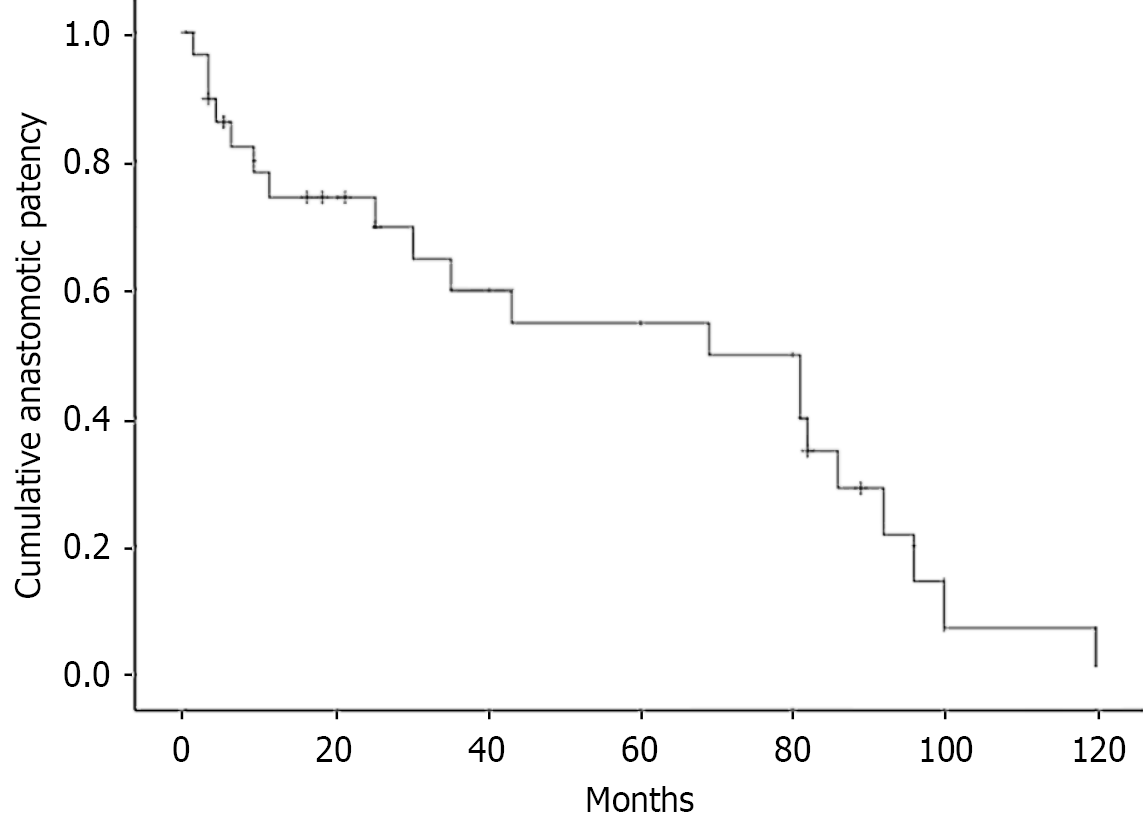
Initial anastomotic patency rate was achieved in 58.1% (n = 18) and 41.9% (n = 13) at 1 and 2 years, respectively, of the 31 patients who received anastomotic stricture dilation (Table 3). Of the 29 catheter-free patients, the cumulative anastomotic patency rates were 74.3%, 69.7%, 59.7%, 49.8%, and 34.8% at 1, 2, 3, 5, and 7 years, respectively (Figure 5), and the median patency duration was 69 months [95% confidence interval (CI): 21-117 months]. The treatment success rate was 90.6% (29/32) (95%CI: 80.5%-100.0%) (Table 3).
Technical success for PTCS-assisted stricture dilation was achieved in all 31 (100%) patients, including 7 patients with and 24 patients without a prior recanalization. The first BD session failed in 3 patients with simple strictures and in 3 patients with complex strictures in the opposite-side bile duct of PTBD; all these patients achieved success with a second BD session.
Technical success for PTCS-assisted recanalization was achieved in 87.5% (7/8) of the patients with severe and completely closed anastomotic strictures (Table 3 and Figure 3). Notably, the initial success and overall technical success rates for PTCS-assisted anastomotic stricture procedures were 75.0% (24/32) and 96.9% (31/32), respectively (Table 3).
PTCS-assisted biliary stone fragmentation/removal was successful in 34 (94.4%) of the 36 patients with biliary stones (Figure 4 and Table 3). Particularly, technical success was achieved in 25 (92.6%) of the 27 patients with intrahepatic stones, including 11 (40.7%) and 16 (59.3%) with monolateral and bilateral stones, respectively (Supplementary Table 1); the successful PTCS-assisted stone removal procedures in a case with intrahepatic stones including a large stone (1.4 cm × 1.9 cm) in the right posterior upper branch duct are illustrated in Figure 4E-L.
Overall, procedure-related AEs developed in 25 patients, including 11 (27.5%), 7 (17.5%), and 7 (21.2%, 4 related to stricture dilation and 3 to stone therapy) patients, during or after PTBD, percutaneous sinus dilation, and PTCS-assisted procedures, respectively (Table 4). The AEs were cholangitis (n = 11), bacteremia (n = 7), right pleural effusion (n = 3), hemobilia (n = 2) (Supplementary Figure 1C), and catheter migration (n = 2); all were minor (mild) except for acute hemobilia which occurred from the sinus tract during percutaneous sinus tract dilation and considered as a major (serious) AE (Table 4). Catheter migration occurred in two cases two weeks after PTBD. The catheter was successfully reinserted under ultrathin cholangioscopy on the same day in one case, and with a repeated PTBD procedure one week later in the other (Table 4). In one patient, blood flowed out from the sinus tract during preparation for the second sinus dilation after the catheter was removed, likely due to a biliovenous fistula caused by blood vessel injury, which was stopped by inserting a catheter larger than the outer caliber of the original catheter over the guidewire (Table 4). Additionally, catheter migration, cholangitis, and bile extravasation occurred, respectively, in 2, 5 and 3 of the 29 patients with treatment success during a median 6 months (range: 2-109 months) of indwelling PTCS catheter. Thus, all 25 AEs, including 24 mild and 1 serious, during the procedures were successfully managed through conservative symptomatic treatment without serious consequences.
| PTBD (n = 40) | Percutaneous sinus tract dilation (n = 40) | PTCS-assisted anastomotic stricture dilation (n = 31)1 | PTCS-assisted anastomotic recanalization (n = 8) | PTCS-assisted stone therapy (n = 36)2 | |
| Cholangitis | 5 (12.5) | 2 (5.0) | 2 (6.5) | 0 (0.0) | 2 (5.6) |
| Bacteremia | 4 (10.0) | 2 (5.0) | 1 (3.3) | 0 (0.0) | 0 (0.0) |
| Right pleural effusion | 2 (5.0) | 1 (2.5) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Hemobilia | 0 (0.0) | 1 (2.5)3 | 1 (3.3) | 0 (0.0) | 0 (0.0) |
| Catheter migration | 0 (0.0) | 1 (2.5)4 | 0 (0.0) | 0 (0.0) | 1 (2.8)4 |
| Overall | 11 (27.5) | 7 (17.5) | 4 (12.9) | 0 (0.0) | 3 (8.3) |
Two patients died of liver and renal failure, respectively, at 51 and 22 months of the follow-up period; both were not related to the PTCS-assisted procedures (Figure 1).
In the present study, PTCS-assisted procedures achieved initial anastomotic patency rates of 58.1% and 41.9% at 1 and 2 years, respectively, and cumulative anastomotic patency rates of 74.3%, 59.7, 49.8%, and 34.8% at 1, 3, 5, and 7 years, respectively, with a treatment success rate of 90.6% for the management of postoperative BBAS. Technical success rates for PTCS-assisted dilation and recanalization were 100% and 87.5%, respectively. The initial and overall technical success rates for the PTCS-assisted procedures were 75.0%, and 96.9%, respectively. The duration of the indwelling PTCS catheter in the catheter-free patients was 6 months (2-109 months). The success rate of PTCS-assisted biliary stone therapy was 94.4%.
In the present study, the initial anastomotic patency rates at 1 (58.1%) and 2 years (41.9%) were not high, likely due to the temporary effect of stricture dilation. However, the cumulative anastomotic patency rates at 1, 3, 5, and 7 years were 74.3%, 59.7%, 49.8%, and 34.8%, respectively. In addition, the Kaplan-Meier analysis showed the median patency duration was 69 months, with a wide range of 95%CIs (range: 21-117 months) in the catheter-free patients. These findings indicate that the placement of an indwelling PTCS catheter increases the efficacy of PTCS-assisted procedures for BBAS. Indeed, the 3-year (59.7%) cumulative anastomotic patency rate is greater than that (49%) using a short double balloon-assisted enteroscopy (DBE) alone[11]. Eventually, indwelling PTCS catheters were successfully removed from 93.5% of patients with a negative clinical clamping trial, with a duration of the indwelling PTCS catheter of 6 months (range: 2-109 months). The relatively long duration of the indwelling PTCS catheter was due to several reasons, including failure of the first BD session for the anastomosis, bougie dilation alone, residual or potential stones, and recurrent BBAS (Figure 1); the indwelling duration was prolonged for at least 3 months even if the clamping trial was negative to improve the ana
We used PTCS-assisted stricture dilation in 31 patients, 27 with BD and 4 with bougie dilation. The technical success rates for the first BD session were 84.2% (16/19) and 62.5% (5/8) for simple and complete strictures, respectively, which are higher than that (52.6%) reported by Kim et al[5] for the first BD session. Three patients with simple strictures failed the first BD session but achieved technical success with a second BD session at 1-2 weeks after placement of the indwelling 18-Fr PTCS catheter across the stricture. Also, three patients with complex strictures located in the hilar biliary duct or IHD on the opposite side of the PTBD failed the first BD session, but technical success was achieved through a PTCS-guided guidewire reaching the stricture site on the opposite side. Our experience is that when BD is difficult, placement of an indwelling PTCS catheter via the stricture or a guidewire under PTCS guidance for BD is required.
It has been recommended that a microcatheter, thin bougie, or special dilation device be applied when a balloon dilator cannot pass through a severe stricture[7,26,27]. In the present study, PTCS-assisted recanalization was successful with a guidewire in four patients whose anastomosis was as small as a pinhole, a dimple, or even hard to locate; a microcatheter system in two patients with strictures of sharp angulation and/or a looping configuration; or a needle knife in one of two patients with scars converging the mucous membrane at the stricture with an invisible anastomotic orifice. Since 2014, we have used the microcatheter system with a 5-Fr microcatheter (made ≤ 3 Fr at the tip) and a stiff 0.025-inch guidewire in patients with PTCS therapy. We assumed that the microcatheter would reduce the resistance of the catheter to pass through the stricture due to its small caliber and the very small space between the lumen of the catheter tip and the guidewire, allowing it to pass through the stricture. We designed a microcatheter (75 cm in length) that was used to dilate the stricture and also guide the indwelling PTCS catheter to connect with the bile bag and maintain PTCS access. Our experience is that when the microcatheter is left in the stenotic place for 1-2 weeks, the stricture becomes relaxed, further enabling the thin bougie to pass through and eventually dilate the stricture. Additionally, our technique for PTCS-assisted recanalization with a needle knife is different from the previously reported by Lim et al[18]. Since fistulotomy with a needle knife is a blind technique and associated with a risk of biliary perforation or bleeding, we punctured a 25-gauge injection needle into the center of the scar under direct visualization (Figure 3J) and injected the contrast medium in two patients. After the jejunal circular fold was visualized by radiography under fluoroscopy, a needle-knife pa
At present, BBAS is mainly treated with BD of anastomotic stenosis, through enteroscopy-assisted ERCP or PTBD[4,6,7], and bile duct stones are mainly cleared by PTCS[3]. However, there are few reports on the treatment of BBAS concomitant with bile duct stones[11]. It is difficult to clear intrahepatic stones with ERCP, especially those in the B6, B7, and B3 segments of the bile duct. In a recent European multicenter study[8], a thin (10.8 F) and short (65-cm) device (i.e., SpyGlass Discover) was used for PTCS to diagnose and treat biliary diseases. SpyGlass-guided EHL with a median of one session achieved a clinical success rate of 96.6% for the treatment of bile duct stones in 29 patients, of whom only 14.7% had intrahepatic stones, which were more difficult to clear[8]. However, this study did not explore the treatment of anastomotic strictures. Recently, a Japanese study compared DBE with PTBD for the treatment of hepatolithiasis after hepaticojejunostomy[28]. It was reported that of the 84 patients eligible for DBE, the hepaticojejunostomy site was reached in 42 (87.5%) patients. The complete stone removal rate of 85.7% (36/42) was achieved with DBE in patients with a median stone diameter of 8 mm (interquartile range: 6-10 mm), with 29 (69.0%) patients being treated with mechanical lithotripsy, whereas the rate was 90.2% (37/41) with PTBD in those with a stone diameter of 7 mm (interquartile range: 7-10 mm), with 31 (75.6%) patients being treated with PTCS-assisted EHL[28]. These results revealed that the success rate for stone therapy is not very high with DBE and PTBD, even for small stones (all ≤ 10 mm). In the present study, we observed that PTCS-assisted stone therapy achieved technical success (i.e., complete stone removal) in 94.4% of cases. Notably, 50.0% and 22.2% of the stones were between 10 mm and 20 mm and > 20 mm, respectively, and the technical success rate was 88.9% for these large stones. Moreover, 75.0% of the stones were located in the IHD, and technical success was achieved in 92.6% of these cases. Our findings indicate that PTCS-assisted stone therapy has advantages, especially for large stones by PTCS-EHL or PTCS-LL lithotripsy, or multiple intrahepatic stones, since the cholangioscope has accessibility to any location of biliary tree dilation. In the present study, one patient with a complex stricture had bilateral intrahepatic stones, including a large stone in the right posterior upper branch duct. Because of the presence of variations in the right anterior and right posterior sectoral ducts (segments 6 and 7)[29] in this patient, PTCS initially failed to observe the stenotic bile duct opening, and cholangiography also failed to show the right posterior branch duct and stones. However, both abdominal ultrasound and CT showed a large stone in the duct (segment 7). Under the guidance of PTCS, a guidewire entered the right posterior branch bile duct, and cholangiography with an ERCP cannula clearly showed the large stone in the right posterior upper branch duct. PTCS-assisted stricture dilation procedures (i.e., BD and the placement of an indwelling PTCS catheter in the branch) were performed, and the stricture was relaxed one week later. Then, a cholangioscope was sharply looped and inserted into the right posterior upper branch duct, showing that the blocky stones had been ground into multiple pieces by the indwelling PTCS catheter. Finally, the stones were successfully removed by further using a basket mechanical lithotripsy and washed out of the duct (Figure 4E-L).
In the present study, mild AEs such as cholangitis, bacteremia, and hemobilia were observed. In one patient with severe hemobilia, acute bleeding from the sinus tract developed, likely due to a biliovenous fistula caused by blood vessel injury, which was quickly stopped with appropriate measures described earlier. Previously, Schumacher et al[20] reported a similar case of bleeding that was stopped through catheter compression during PTCS for BBAS. Therefore, PTCS-assisted procedures for BBAS are relatively safe. Nevertheless, we would recommend that blood vessels be avoided during ultrasound-guided bile duct puncture, which is the key to preventing blood vessel injury and bleeding caused by sinus dilation, and that timely and appropriate management be implemented in case of acute bleeding, which can be rapidly stopped without requirement for blood transfusion and/or angiographic intervention, and serious consequences.
This study has some limitations. First, it was a retrospective and single-center study. Second, the number of studied patients was relatively small, despite 25 years of experience. Third, the relatively long median duration (6 months) of the indwelling PTCS catheter may cause inconvenience to patients, although it does improve the anastomotic patency. Therefore, a well-designed prospective study with more patients is required to confirm our findings and to accumulate more experience in PTCS-assisted procedures for the management of BBAS.
In conclusion, PTCS-assisted procedures, including stricture dilation, recanalization, and biliary stone therapy, are efficacious and safe for the management of BBAS and concomitant biliary stones. Placement of an indwelling PTCS catheter across the anastomosis after stricture dilation improves the anastomotic patency.
We would like to express our appreciation to Professor Ogoshi K (Niigata Cancer Center Hospital, Niigata, Japan), who passed away on July 27, 2021, for his constant instructions and support during the development of our PTCS technique over the years. He will be missed forever. We would also like to thank Professor Yamakawa T (Teikyo University Hospital at Mizonokuchi, Kanagawa, Japan) for his dedicated guidance on the PTCS technique.
| 1. | Dimou FM, Adhikari D, Mehta HB, Olino K, Riall TS, Brown KM. Incidence of hepaticojejunostomy stricture after hepaticojejunostomy. Surgery. 2016;160:691-698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 2. | Yamauchi H, Kida M, Miyata E, Okuwaki K, Iwai T, Minato N, Tadehara M, Watanabe M, Imaizumi H, Koizumi W. Endoscopic Balloon Dilation for Benign Bilioenteric Stricture: Outcomes and Factors Affecting Recurrence. Dig Dis Sci. 2019;64:3557-3567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 3. | Bonnel DH, Fingerhut AL. Percutaneous transhepatic balloon dilatation of benign bilioenteric strictures: long-term results in 110 patients. Am J Surg. 2012;203:675-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Lee AY, Gregorius J, Kerlan RK Jr, Gordon RL, Fidelman N. Percutaneous transhepatic balloon dilation of biliary-enteric anastomotic strictures after surgical repair of iatrogenic bile duct injuries. PLoS One. 2012;7:e46478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 5. | Kim JH, Lee SK, Kim MH, Song MH, Park DH, Kim SY, Lee SS, Seo DW, Bae JS, Kim HJ, Han J, Sung KB, Min YI. Percutaneous transhepatic cholangioscopic treatment of patients with benign bilio-enteric anastomotic strictures. Gastrointest Endosc. 2003;58:733-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 44] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | Hai R, Kuban J. Percutaneous intervention for bilioenteric anastomotic strictures: Current strategies and future directions. Int J Gastrointest Interv. 2017;6:70-77. [DOI] [Full Text] |
| 7. | Fang A, Kim IK, Ukeh I, Etezadi V, Kim HS. Percutaneous Management of Benign Biliary Strictures. Semin Intervent Radiol. 2021;38:291-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 8. | Pérez-Cuadrado-Robles E, Phillpotts S, Bronswijk M, Conrad CC, Binda C, Monino L, Basiliya K, Hollenbach M, Papaefthymiou A, Alric H, Quénéhervé L, Di Gaeta A, Pioche M, Khani A, Lorenzo D, Moreels TG, Rahmi G, Boeken T, Fabbri C, Prat F, Laleman W, Cellier C, Van der Merwe S, Webster G, Ellrichmann M. PERcutaneous transhepatic CHOLangioscopy using a new single-operator short cholangioscope (PERCHOL): European feasibility study. Dig Endosc. 2024;36:719-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 9. | Oh HC. Percutaneous Transhepatic Cholangioscopy in Bilioenteric Anastomosis Stricture. Clin Endosc. 2016;49:530-532. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 10. | Chon HK, Choi KH, Seo SH, Kim TH. Efficacy and Safety of Percutaneous Transhepatic Cholangioscopy with the Spyglass DS Direct Visualization System in Patients with Surgically Altered Anatomy: A Pilot Study. Gut Liver. 2022;16:111-117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 11. | Mizukawa S, Tsutsumi K, Kato H, Muro S, Akimoto Y, Uchida D, Matsumoto K, Tomoda T, Horiguchi S, Okada H. Endoscopic balloon dilatation for benign hepaticojejunostomy anastomotic stricture using short double-balloon enteroscopy in patients with a prior Whipple's procedure: a retrospective study. BMC Gastroenterol. 2018;18:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 12. | Krutsri C, Kida M, Yamauchi H, Iwai T, Imaizumi H, Koizumi W. Current status of endoscopic retrograde cholangiopancreatography in patients with surgically altered anatomy. World J Gastroenterol. 2019;25:3313-3333. [PubMed] [DOI] [Full Text] |
| 13. | Tsutsumi K, Kato H, Yabe S, Mizukawa S, Seki H, Akimoto Y, Uchida D, Matsumoto K, Tomoda T, Yamamoto N, Horiguchi S, Kawamoto H, Okada H. A comparative evaluation of treatment methods for bile duct stones after hepaticojejunostomy between percutaneous transhepatic cholangioscopy and peroral, short double-balloon enteroscopy. Therap Adv Gastroenterol. 2017;10:54-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 14. | Yun G, Yoon CJ, Seong NJ. Percutaneous treatment of benign bilioenteric anastomotic strictures: temporary covered stent placement versus balloon dilatation. Eur Radiol. 2019;29:2690-2697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 15. | Weber A, Rosca B, Neu B, Rösch T, Frimberger E, Born P, Schmid RM, Prinz C. Long-term follow-up of percutaneous transhepatic biliary drainage (PTBD) in patients with benign bilioenterostomy stricture. Endoscopy. 2009;41:323-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Akinci D, Unal E, Ciftci TT, Kyendyebai S, Abbasoglu O, Akhan O. Endobiliary Radiofrequency Ablation in the Percutaneous Management of Refractory Benign Bilioenteric Anastomosis Strictures. AJR Am J Roentgenol. 2019;212:W83-W91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Yang DH, Lee SK, Moon SH, Park DH, Lee SS, Seo DW, Kim MH. Percutaneous transhepatic cholangioscopic intervention in the management of complete membranous occlusion of bilioenteric anastomosis: report of two cases. Gut Liver. 2009;3:352-355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Lim JU, Joo KR, Cha JM, Shin HP, Lee JI, Park JJ, Jeon JW. Needle-knife fistulotomy with percutaneous transhepatic cholangioscopy for managing complete bilioenteric anastomosis occlusion. Surg Laparosc Endosc Percutan Tech. 2014;24:e10-e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Lee MJ, Mueller PR, Saini S, Hahn PF, Dawson SL. Percutaneous dilatation of benign biliary strictures: single-session therapy with general anesthesia. AJR Am J Roentgenol. 1991;157:1263-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 20. | Schumacher B, Othman T, Jansen M, Preiss C, Neuhaus H. Long-term follow-up of percutaneous transhepatic therapy (PTT) in patients with definite benign anastomotic strictures after hepaticojejunostomy. Endoscopy. 2001;33:409-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 62] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 21. | Ren X, Tang XF, Si LJ, Zhu CL, Zhang GL, Sun XZ. [The percutaneous cholangioscopy therapy in treating intra and extra hepatic calculi]. Zhonghua Xiaohuaneijing Zazhi. 2004;21:13-16. [DOI] [Full Text] |
| 22. | Ren X, Qu YP, Zhu CL, Xu XH, Jiang H, Lu YX, Xue HP. Percutaneous transhepatic cholangioscopy-assisted biliary polypectomy for local palliative treatment of intraductal papillary neoplasm of the bile duct. World J Gastrointest Oncol. 2024;16:1821-1832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 23. | Oh HC, Lee SK, Lee TY, Kwon S, Lee SS, Seo DW, Kim MH. Analysis of percutaneous transhepatic cholangioscopy-related complications and the risk factors for those complications. Endoscopy. 2007;39:731-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 108] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 24. | Sacks D, McClenny TE, Cardella JF, Lewis CA. Society of Interventional Radiology clinical practice guidelines. J Vasc Interv Radiol. 2003;14:S199-S202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1406] [Cited by in RCA: 1344] [Article Influence: 58.4] [Reference Citation Analysis (0)] |
| 25. | Glas L, Courbière M, Ficarelli S, Milot L, Mennesson N, Pilleul F. Long-term outcome of percutaneous transhepatic therapy for benign bilioenteric anastomotic strictures. J Vasc Interv Radiol. 2008;19:1336-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Inoue T, Ibusuki M, Kitano R, Kobayashi Y, Ishii N, Ohashi T, Nakade Y, Sumida Y, Ito K, Nakao H, Yoneda M. Severe Biliary Stricture Dilation Using the Soehendra Stent Retriever with a Short-Type Balloon Enteroscope in Patients with Surgically Altered Anatomies. J Gastrointest Surg. 2019;23:953-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Miyata E, Yamauchi H, Kida M, Okuwaki K, Miyazawa S, Iwai T, Koizumi W. Successful endoscopic dilation of severe bilioenteric strictures with a wire-guided diathermic dilator and short-type single-balloon enteroscope. Endoscopy. 2015;47 Suppl 1 UCTN:E94-E95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Nakamura K, Ishii Y, Tatsukawa Y, Nakamura S, Ikemoto J, Miyamoto S, Furukawa M, Iijima N, Hanada K, Oka S. Comparative study of therapeutic outcomes in patients who developed hepatolithiasis after hepaticojejunostomy: balloon-assisted enteroscopic approach versus percutaneous transhepatic approach. Surg Endosc. 2025;39:1160-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 29. | Toouli J, Bhandari M. Anatomy and Physiology of the Biliary Tree and Gallbladder. In: Clavien PA, Baillie J. Diseases of the Gallbladder and Bile Ducts: Diagnosis and Treatment. Wiley-Blackwell, 2006: 1-20. |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/