INTRODUCTION
Acquired brain injury (BI), including traumatic BI (TBI) and stroke, represents a major global health burden due to its high mortality and long-term morbidity rates[1]. The pathophysiology of BI is a complex process characterized by primary damage caused by the initial insult, followed by a series of secondary injury mechanisms that induce further neuronal death and functional impairment(s)[2]. In clinical practice, treatment strategies have focused on reducing primary brain damage. In recent years, it has been increasingly recognized that systemic complications, especially gastrointestinal (GI) dysfunction, play an important role in the outcomes of patients with BI[3]. Many patients who sustain severe BI present with GI dysfunction, which can manifest as delayed gastric emptying, ileus, stress ulceration, and an increased risk for infection[4]. GI dysfunction not only affects the nutritional status of critically ill patients, but is also an independent predictor of morbidity and mortality.
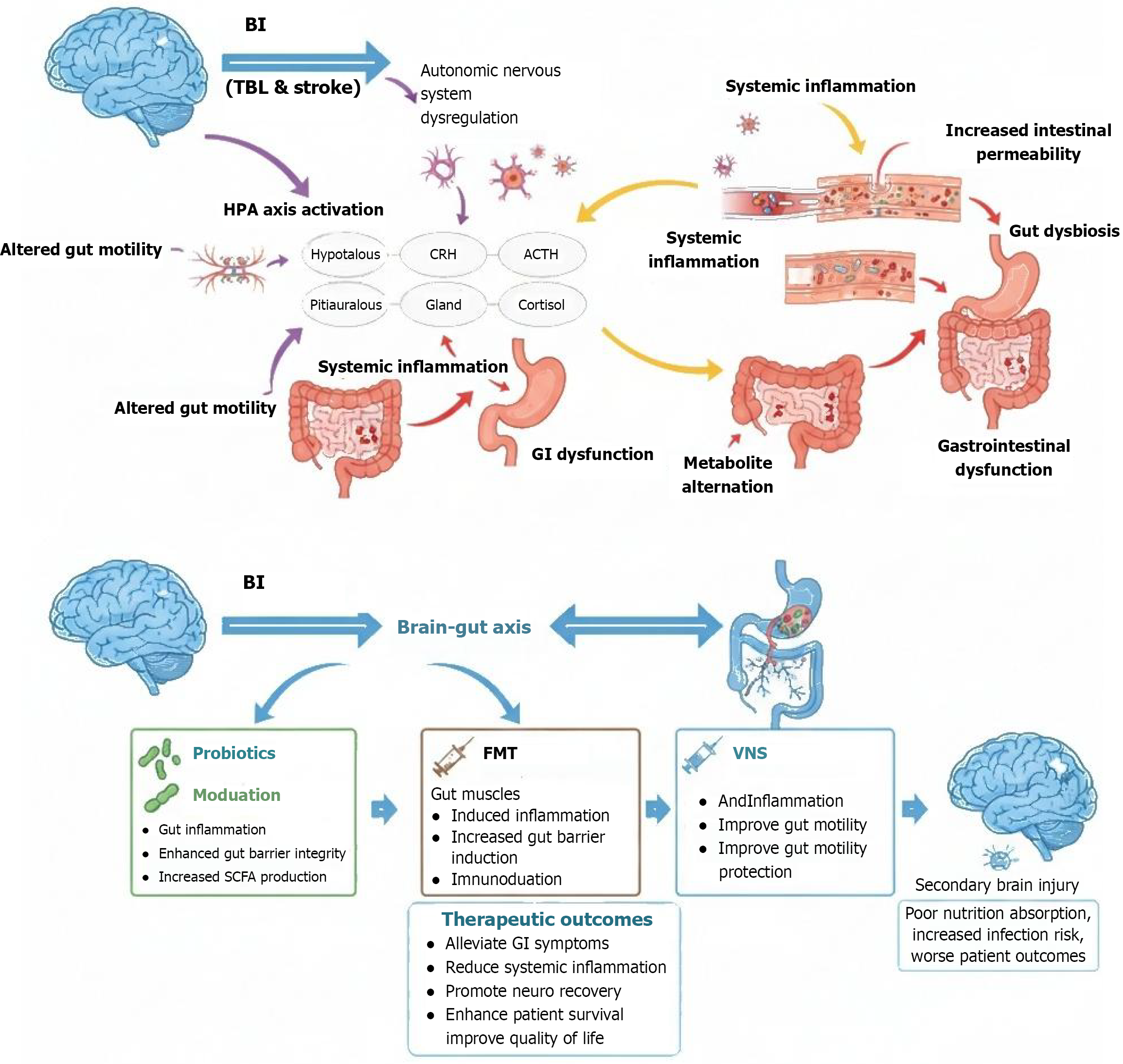
As illustrated in Figure 1, the detailed and complex bidirectional communication network, known as the brain-gut axis, is the key to understanding the association between BI and GI dysfunction[5]. The brain-gut axis involves multiple neural, endocrine, and immune connections between the central nervous system (CNS) and enteric nervous system (ENS)[6]. The core components of this axis include the CNS, autonomic nervous system (ANS) and its sympathetic and parasympathetic branches, ENS, and hypothalamic-pituitary-adrenal (HPA) axis. Bidirectional communication is maintained through various pathways, including the neural, endocrine, and immune signaling pathways. The vagus nerve, a primary component of the parasympathetic nervous system (PNS), serves as a direct and rapid neural conduit that transmits information from the gut microbiota and the intestinal environment to the brain. Conversely, the CNS modulates gut functions, such as motility and secretion via the ANS. Endocrine pathways involve gut-derived hormones and microbial metabolites, such as short-chain fatty acids (SCFAs) and neurotransmitters (e.g., serotonin and gamma-amino butyric acid), which can enter the circulation and influence brain function. This intricate network ensures constant dialogue between the brain and gut, which is vital for homeostasis. However, the delicate balance of the brain-gut axis is profoundly disturbed after acute BI, leading to a vicious cycle in which brain damage causes gut dysfunction, which in turn promotes secondary BI through systemic inflammation and other mechanisms[7].
Figure 1 The brain-gut axis in brain injury: Mechanisms and therapeutic strategies.
BI: Brain injury; TBI: Traumatic brain injury; HPA: Hypothalamic-pituitary-adrenal; CRH: Corticotropin-releasing hormone; ACTH: Adrenocorticotropic hormone; GI: Gastrointestinal; SCFA: Short-chain fatty acid; FMT: Fecal microbiota transplantation; VNS: Vagus nerve stimulation.
The involvement of the gut microbiota in the brain-gut axis has attracted increasing attention in recent years[8]. The gut microbiome represents a collection of microorganisms residing in the GI tract and has been shown to play a key modulatory role in host physiology, including the brain and immune system[9]. After BI, alterations in the composition and diversity of the gut microbiota, such as dysbiosis, have been repeatedly reported in preclinical models and clinical studies[10]. Such dysbiosis is not only a consequence of BI, but also plays an active role in the pathophysiology of BI by promoting intestinal inflammation, disrupting gut barrier function, and affecting neuroinflammation[11].
As such, a better understanding of the mechanisms underlying GI dysfunction after BI and the key role of the brain-gut axis is essential for the development of novel therapeutic strategies. In the present review, we summarize the current knowledge regarding the key pathways involved in BI-induced GI dysfunction, including the ANS, HPA axis, neuroinflammation, and gut microbiota. We also summarize the emerging therapeutic strategies targeting the brain-gut axis for the prevention and treatment of GI complications in patients with BI. Understanding the complex interactions between the brain and gut will provide a better basis for future studies investigating BI and the development of more effective and multitargeted therapies.
ANS DYSFUNCTION AND ITS IMPACT ON GI MOTILITY
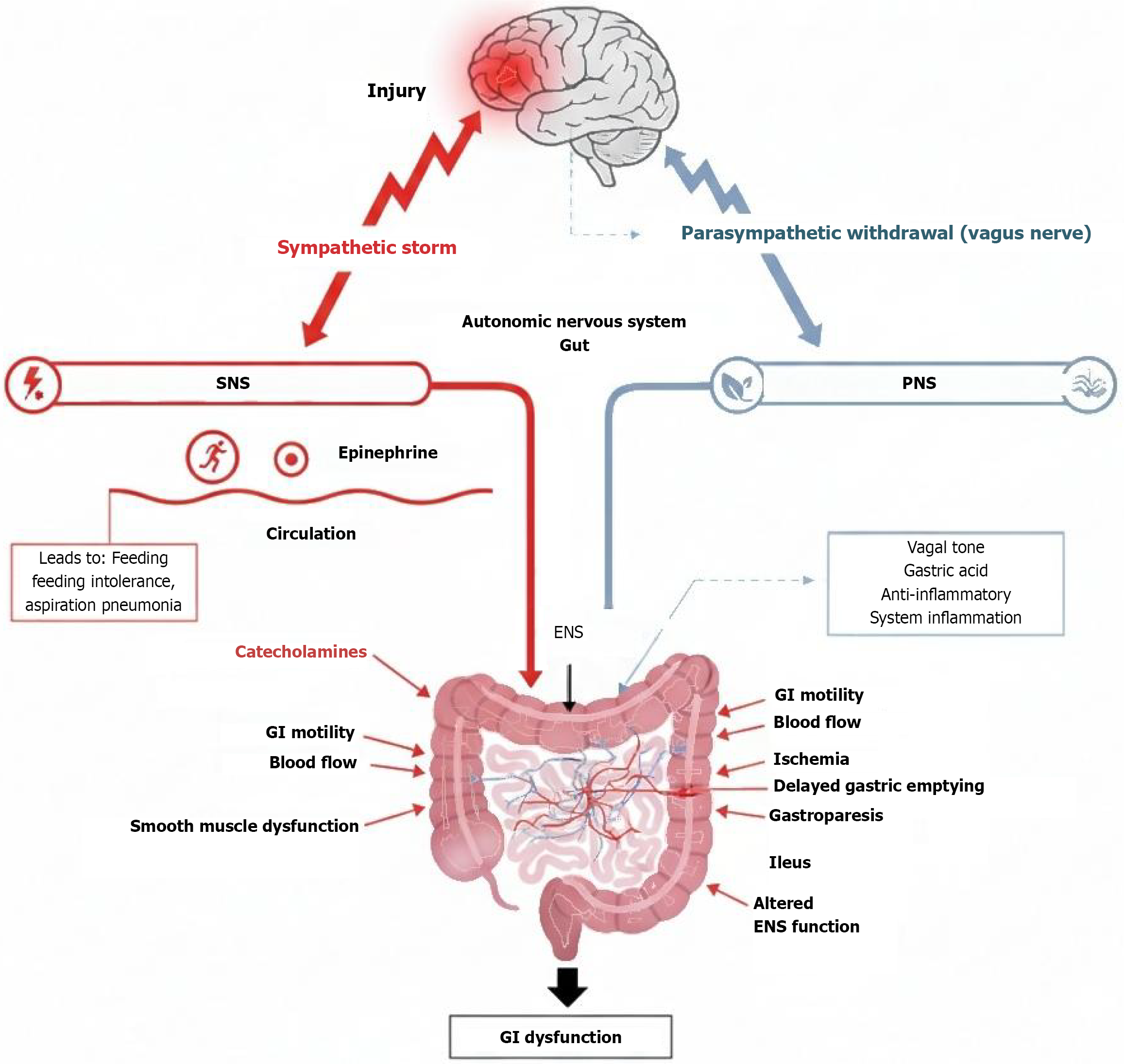
The ANS is an essential component of the brain-gut axis that exerts fine control over virtually all aspects of GI function, including motility, secretion, and blood flow[12]. The ANS is composed of 2 branches, the sympathetic nervous system (SNS) and the PNS, which generally have opposite effects on GI activity[13], as illustrated in Figure 2. After BI, the balance between these 2 branches is usually and profoundly disrupted, leading to dysautonomia, which plays an important role in GI dysfunction[14].
Figure 2 Acute brain injury results in autonomic imbalance and gastrointestinal dysfunction.
SNS: Sympathetic nervous system; PNS: Parasympathetic nervous system; ENS: Enteric nervous system; GI: Gastrointestinal.
An acute BI can cause a high degree of intense and sustained SNS activity (“sympathetic storm”)[15] with copious amounts of norepinephrine and epinephrine released into the circulation. The SNS outflow has inhibitory effects on the GI tract, slowing GI motility by decreasing smooth muscle contraction and blood flow to the gut (possibly by inducing ischemia)[16]. This “sympathetic storm” likely contributes to delayed gastric emptying and the development of gastroparesis and ileus encountered in patients with TBI and stroke, which in turn can lead to feeding intolerance and aspiration pneumonia[17].
In contrast, the PNS (which is mostly mediated by the vagus nerve) has positive effects on GI function. The vagus nerve stimulates gastric acid secretion and promotes gastric motility. It also exerts anti-inflammatory effects in the intestine through the cholinergic anti-inflammatory pathway[18]. After BI, there is generally an overall withdrawal of parasympathetic tone, which further promotes sympathetic dominance and GI dysmotility[19]. Dysautonomia not only slows digestion, but also impairs the ability of the intestine to mount an effective anti-inflammatory response. This may also promote systemic inflammation[20].
The ENS is a myelinated network of neurons within the walls of the gut itself that can function autonomously, and often referred to as the “second brain”. However, the ENS is also modulated by CNS through the ANS[21]. Dysautonomia induced by TBI would, therefore, directly affect the ENS, disrupting the pattern of muscle contraction and relaxation that enables coordinated peristalsis, potentially leading to several different motility disorders, ranging from localized spasms to total paralysis of the gut[22]. Experimental TBI has been shown to induce decreased contractility of intestinal smooth muscles and delayed GI transit, which are likely mediated by alterations in ENS function[23]. In conclusion, dysautonomia appears to play a major role in GI motility disorders after BI. The state of heightened sympathetic activity, coupled with the withdrawal of parasympathetic tone, creates an environment that is highly detrimental to normal GI function and can lead to major clinical complications[24]. Therapies aimed at restoring autonomic balance, such as vagus nerve stimulation (VNS), may prove beneficial in ameliorating these effects[25].
HPA AXIS AND STRESS-INDUCED GI COMPLICATIONS
In addition to the ANS, the HPA axis is another key link in the brain-gut communication network after BI, which is disrupted after TBI or stroke[26]. The HPA axis is the most important neuroendocrine stress response system in the human body. When a major physiological stressor, such as TBI or stroke, is imposed, corticotropin-releasing hormone (CRH) is released from the hypothalamus, which stimulates the pituitary gland to release adrenocorticotropic hormone[27]. Adrenocorticotropic hormone then acts on the adrenal glands, causing them to secrete glucocorticoids (primarily cortisol) in humans. This response is adaptive if activated only temporarily. However, because the HPA axis is chronically activated after severe BI, the continual release of glucocorticoids induced by excessive HPA-axis activation can lead to GI dysfunction[28].
Glucocorticoids have several effects on the GI tract and can lead to dysfunction, directly affecting GI motility, typically causing delayed gastric emptying, which contributes to feeding intolerance[29]. Glucocorticoids exert dual detrimental effects on GI function: They not only disrupt the integrity of the GI mucosal barrier by reducing the secretion of protective mucus and bicarbonate, and decreasing mucosal blood flow - thereby augmenting gut mucosal vulnerability to injury - but also exacerbate this delayed gastric emptying previously described. Preclinical studies in canine models have confirmed that prednisolone, a synthetic glucocorticoid, directly induces gastric mucosal damage and impairs gastric motility, with the prokinetic agent mosapride demonstrating efficacy in alleviating both pathological manifestations[30]. This evidence underscores the interconnected nature of glucocorticoid-induced mucosal barrier disruption and GI dysmotility, two key pathological mechanisms that collectively contribute to the development of GI dysfunction, including stress-related mucosal disease and Cushing’s ulcers, in patients following severe brain injury. This increased susceptibility to injury likely contributes to the development of stress-related mucosal diseases and Cushing’s ulcers, which are shallow or deep gastric or duodenal ulcerations that can result in life-threatening GI bleeding in critically ill patients[31].
CRH peptides, both centrally produced and peripherally synthesized, also play a major role in modulating gut function, and can directly affect GI motility and secretion and increase intestinal permeability[32]. Increased intestinal permeability may be particularly important because it can lead to the translocation of bacteria and endotoxins from the lumen of the gut into the bloodstream, triggering a systemic inflammatory response that exacerbates both primary BI and other forms of organ dysfunction[33].
The HPA axis is closely connected to the immune system[34]. Glucocorticoids are extremely strong immunosuppressants and, although they may help blunt the initial inflammatory response after injury, chronically high levels of glucocorticoids can lead to immunosuppression and increased susceptibility to infections, both of which could involve the gut[35]. Collectively, the effects of glucocorticoids and CRH provide a positive feedback loop, in which stress-induced changes in the gut can contribute to systemic secondary brain dilation and dysregulation of the HPA axis (Figure 2)[36].
In summary, persistent activation of the HPA axis after BI may be an important cause of stress-induced GI complications. High levels of glucocorticoids and CRH after BI may induce abnormal regulation of GI motility, compromise the intestinal mucosal barrier, and modulate the immune response, exerting detrimental effects in affected patients[37]. A better understanding of the role of the HPA axis in BI-induced gut dysfunction is important to develop strategies to prevent these complications, which may encompass pharmacological modulation of the stress response or protection of the gut mucosa against its detrimental effects.
NEUROINFLAMMATION AND THE SYSTEMIC INFLAMMATORY RESPONSE
The brain itself mounts a powerful inflammatory response in response to BI, known as neuroinflammation[38]. This results in the activation of the resident pool of immune cells in the brain, mostly microglia and astrocytes, and the transmigration of immune cells from the surrounding vasculature into the injured brain parenchyma[39]. Initially, this proinflammatory response helps the brain remove cellular debris and promotes repair after injury. However, if this effect is sustained in an exaggerated manner, the neuroinflammatory response can significantly contribute to secondary brain damage[40]. It is important to note that this local inflammation in the brain is not a “standalone” event. It may not only affect the brain itself but also “spread” to the rest of the body, leading to a whole-body inflammatory response syndrome that affects other organs, including the gut[41].
The “spillover” of proinflammatory mediators, such as tumor necrosis factor-alpha, interleukin-1β, interleukin-6, and chemokines, from the injured brain into the cerebrospinal fluid surrounding the brain and subsequently into the circulation is the link between neuroinflammation and systemic inflammation[42]. These inflammatory molecules can reach the GI tract and induce a state of “gut inflammation”[43]. They may impair barrier function in the intestinal wall, alter normal motility of the gut, and modulate the composition of the gut microbiota[44]. Moreover, the inflammatory milieu produced by the injured brain may induce an additional reduction in blood flow to the splanchnic organs as a part of the body’s “stress response”. This in turn may compromise the integrity and function of the gut through ischemia-reperfusion injury[45].
The relationship between neuroinflammation and gut dysfunction is bidirectional[46]. Although brain inflammation may cause gut problems, a dysfunctional gut can worsen brain inflammation[47]. As discussed in the next section, a “leaky gut” may permit the translocation of gut-derived bacterial products, such as lipopolysaccharide, and pro-inflammatory molecules, into the systemic circulation[48]. These inflammatory products may cross the disrupted blood-brain barrier and activate the microglia and astrocyte population in the brain, enhancing neuroinflammation and causing further neuronal damage, even after the initial insult[49].
This concept of a gut-brain inflammatory loop is supported by preclinical and clinical observations[50]. Animal models of TBI and stroke have shown that manipulating the composition of the gut microbiota or attenuating gut barrier dysfunction can reduce the degree of neuroinflammation and improve neurological function[51]. In affected patients, gut dysfunction and systemic inflammation are frequently associated with the degree of BI and poor clinical outcome[52].
In conclusion, the inflammatory response after BI is not strictly a local process in the brain, but rather a systemic process that cannot be dissociated from the gut. The crosstalk between neuroinflammation and the systemic inflammatory response through the brain-gut axis plays an important role in the pathophysiology of secondary injury after brain damage. Breaking this vicious cycle by modulating either central or peripheral inflammation, or the pathways that link them, offers a new opportunity to improve outcomes in patients with brain damage.
GUT BARRIER DYSFUNCTION AND BACTERIAL TRANSLOCATION
The normal gut is protected by a single layer of epithelial cells. With a protective mucosal layer and tight junctions between adjacent cells, the intestinal epithelium acts as a highly selective barrier[53]. This intestinal barrier is important for the absorption of nutrients, but also prevents the translocation of potentially harmful contents from the lumen of the gut into the circulation, such as bacteria, toxins, and large pieces of undigested food[54]. After BI, intestinal barrier function is often compromised. This is known as “leaky gut” or increased intestinal permeability[55].
Several potential mechanisms can lead to disruption of the intestinal barrier after BI. As discussed in the context of autonomic dysfunction, a profound sympathetic surge after BI can lead to splanchnic vasoconstriction and decreased blood flow (i.e., ischemia) to the gut mucosa[56]. Following reperfusion, reactive oxygen species are produced, which induce oxidative stress and injury to epithelial cells[57]. Systemic inflammation mediated by proinflammatory cytokines released from the injured brain also plays a direct role in intestinal barrier disruption[58]. These cytokines can alter the expression and localization of tight junction proteins (occluding, claudins, zonula occludens-1), which are the structures that seal the space between adjacent epithelial cells and control paracellular transport between them[59].
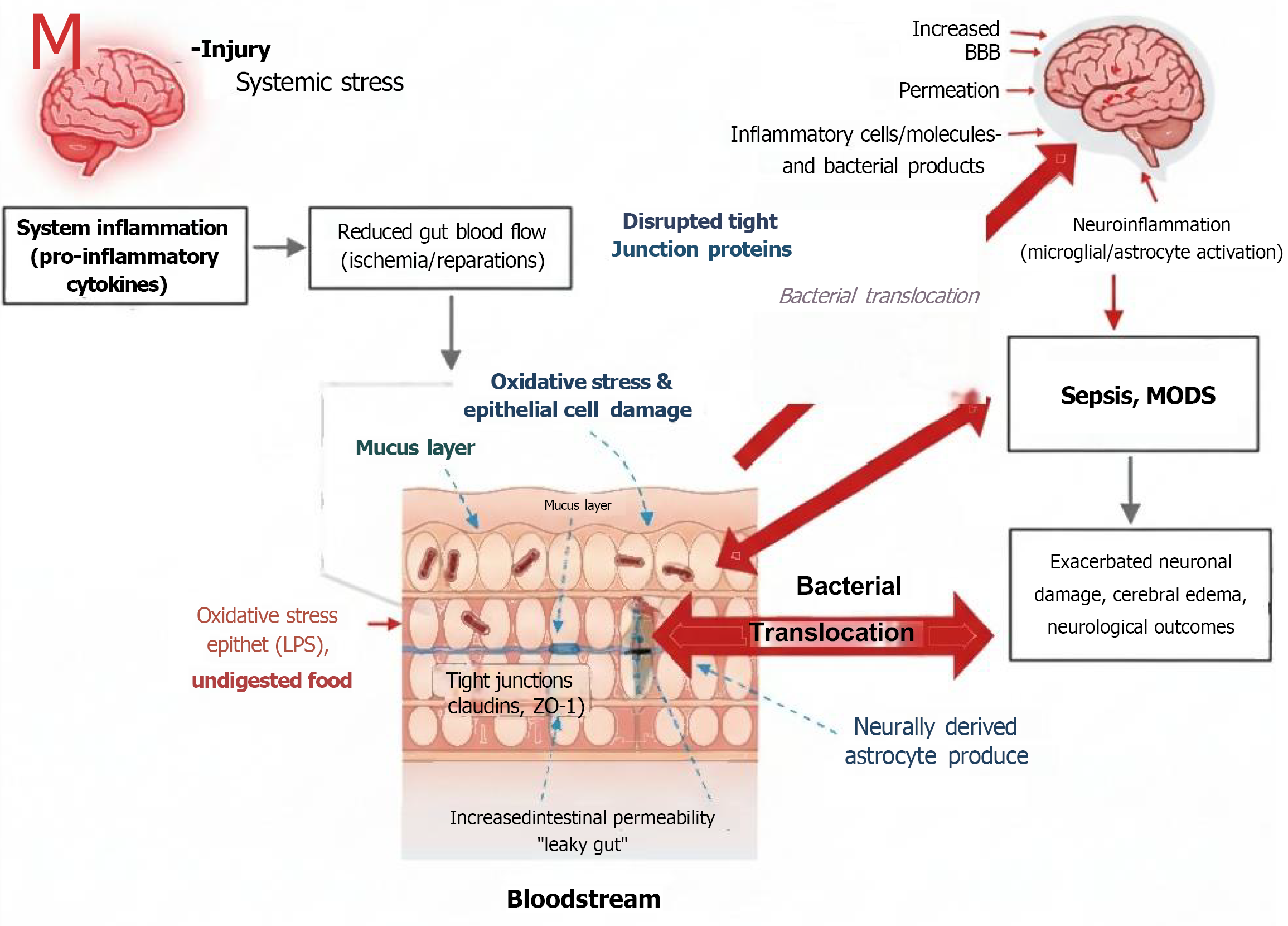
The consequence of this dysfunction of the intestinal barrier is the translocation of contents within the lumen of the gut, particularly bacteria and their products, such as lipopolysaccharide, into the systemic circulation[60]. This process, known as “bacterial translocation”, plays a major role in the drive to systemic inflammation and is strongly associated with the development of sepsis and multiple organ dysfunction syndrome in critically injured patients, including those who experience TBI[61]. Products from bacteria that enter the circulation induce a powerful inflammatory response and a second surge of proinflammatory cytokines[62].
This state of systemic inflammation also affects the brain. A “leaky gut” leads to a “leaky brain”. It has been demonstrated that the inflammatory mediators that increase the permeability of the intestinal barrier also increase the permeability of the blood-brain barrier and can induce the passage of inflammatory cells and molecules, as well as products originating in the gut, into the CNS[63]. These substances can activate microglia and astrocytes and, therefore, potentiate and prolong the inflammatory response in the CNS[64]. This second wave of inflammation can increase neuronal damage, induce cerebral edema, and lead to poorer neurological outcomes (Figure 3)[65].
Figure 3 Gut barrier dysfunction and bacterial translocation following brain injury and their implications for neural and systemic health.
LPS: Lipopolysaccharide; MODS: Multiple organ dysfunction syndrome; ZO-1: Zonula occludens-1.
Gut barrier dysfunction is a key link in the vicious cycle of brain damage, systemic inflammation, and secondary brain damage. Loss of intestinal integrity transforms the gut from a source of nutrition into a source of inflammation, with downstream consequences for the rest of the body. Considering this, protective and repair strategies aimed at intestinal barrier function, such as special enteral nutrition (EN), probiotics, or pharmacological agents targeting the tight junctions, are currently being explored to improve outcomes after BI.
ROLE OF THE GUT MICROBIOTA AND DYSBIOSIS
The human body hosts a large number of microorganisms that collectively comprise the gut microbiota, which play important roles, including digestion, development of the immune system, and brain function[66]. Communication between the gut microbiota and brain is termed the “microbiota-gut-brain axis” and is currently a highly active area of research[67]. A healthy gut microbiota is crucial for maintaining homeostasis. After BI, the delicate ecosystem of the gut is frequently disrupted, resulting in dysbiosis associated with both GI and neurological complications[68].
BI can induce dysbiosis in several ways. The systemic stress response, including activation of the SNS and HPA axes, can have direct effects on the gut environment, including the ability of microbes to grow[69]. Disturbances in gut motility, which the authors have recently reviewed, can lead to the overgrowth of specific pathogenic bacteria[70]. Moreover, factors commonly found in critical care wards, such as antibiotics, opioids, proton-pump inhibitors, and changes in diet and enteral feeding, can affect the composition of the gut microbiota[71].
The effects of dysbiosis are multifold. When the balance between beneficial commensal bacteria of the gut microbiota and pathogenic microbes shifts in favor of the latter, several effects ensue. For example, pathogenic bacteria can release several toxins and other virulence factors that damage the intestinal epithelium and impair gut barrier function[72]. Loss of beneficial bacteria is also a problem[73]. For example, many beneficial bacteria, such as those that produce SCFAs, such as butyrate, can exert several protective effects. SCFAs are an important energy source for colonocytes and have several effects on gut barrier integrity, immune response, and brain function[74]. Therefore, decreased SCFA production after BI can lead to both gut inflammation and neuroinflammation[75].
An altered gut microbiota can also directly affect the immune system. Dysbiosis can result in a proinflammatory state within the gut, with an increase in the production of proinflammatory cytokines and a relative decrease in anti-inflammatory responses[76]. This local inflammatory state can extend into the circulation, contributing to the systemic inflammatory response often observed after BI[77]. Additionally, microbes within the gut can influence the differentiation and function of immune cells, such as T cells, which can then infiltrate the brain and participate in neuroinflammatory responses[78]. An association between gut dysbiosis and neurological outcomes is emerging. In fact, in preclinical studies, stroke-induced dysbiosis has been associated with worse neurological deficits, and interventions to restore a healthy microbiota, such as fecal microbiota transplantation (FMT), have been correlated with improved outcomes[79]. Similarly, in TBI models, dysbiosis has been associated with lesion volume and neurobehavioral deficits[80].
In conclusion, the gut microbiota is an important component of the brain-gut axis, and its disruption after BI is an important pathophysiological mechanism. Dysbiosis promotes gut barrier dysfunction, induces pro-inflammatory states at both the systemic and central levels, and is associated with worse neurological recovery. Thus, the gut microbiota is an attractive target for intervention. The gut microbiota can be modulated with probiotics, prebiotics, and FMT, with the ultimate goal of restoring gut homeostasis and improving outcomes in patients with BI.
NUTRITIONAL ISSUES AND FEEDING INTOLERANCE
A critically injured patient is in a hypermetabolic and hypercatabolic state, and has increased energy and protein requirements to support repair and recovery[81]. Therefore, adequate nutrition is an important component of the critical care management of these patients. Whole-body enteral feeding is preferred over intravenous parenteral nutrition because the enteral route helps maintain gut integrity and modulates the immune response. In addition, outcomes after EN have been better than those after parenteral nutrition[82]. GI dysfunction induced by BI can make the delivery of adequate EN difficult[83].
Feeding intolerance is a common and important issue in patients with TBI and stroke, and has been reported to affect up to one-half of patients in some studies[84]. It is characterized by large residual stomach volumes, abdominal distension, vomiting, and diarrhea. The main cause of feeding intolerance is profound GI dysmotility with delayed gastric emptying and ileus after dysregulation of the ANS and systemic stress response after BI[85].
The inability to tolerate EN has several consequences, including undernutrition and impairment of immune function, delayed wound healing, and increased risk for infection[86]. Malnutrition is an independent predictor of poor outcomes in patients with BI. Feeding intolerance also predisposes patients to aspiration pneumonia secondary to vomiting and regurgitation, which are the major causes of morbidity and mortality in this patient population[87]. The need to frequently interrupt or reduce the rate of EN makes it difficult to achieve patients’ nutritional goals and leads to a state of malnutrition and poor recovery[88].
Feeding intolerance is a common and challenging clinical problem in patients with BI. Approaches to its management include the use of prokinetic agents to enhance gut motility, placement of a feeding tube beyond the stomach into the small intestine (post-pyloric feeding) to bypass the poorly functioning stomach, and the use of special enteral formulas[89]. Peptide-based or elemental formulas, in which nutrients are pre-digested, may be better absorbed when GI function is compromised[90]. However, the rationale for these interventions continues to evolve, and a consistent approach to their use is lacking[91].
In conclusion, although early and adequate nutrition is essential for the recovery of patients with BI, the ability to provide nutrition is limited by feeding intolerance. This intolerance is a result of BI-induced GI dysfunction, in part due to disruption of the brain-gut axis. Understanding the cause of this dysfunction and developing strategies to optimize nutritional delivery are crucial for the care of critically ill patients.
THERAPEUTIC STRATEGIES TARGETING THE BRAIN-GUT AXIS
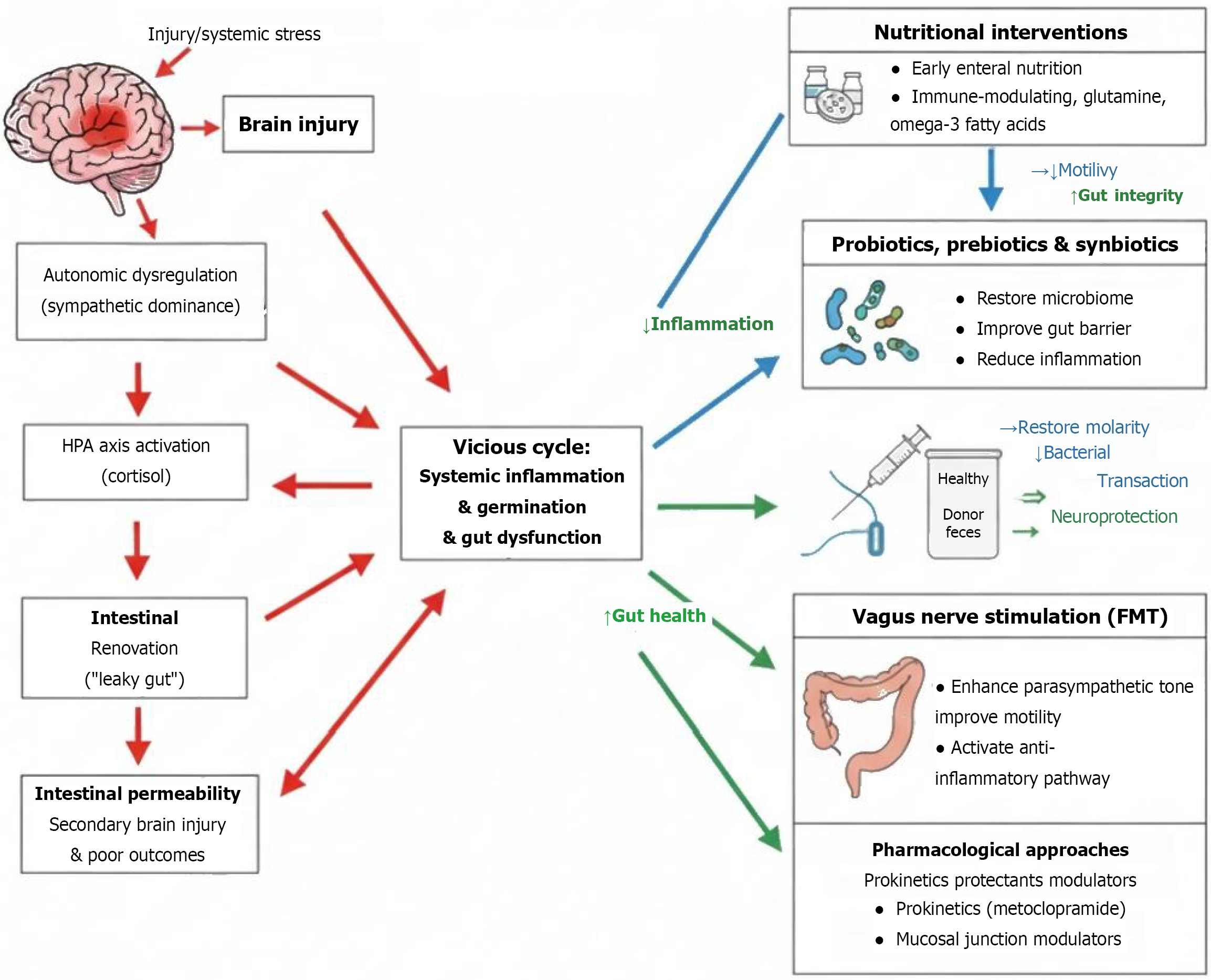
Emerging knowledge regarding the importance of the brain-gut axis in the pathophysiology of BI has created new opportunities for intervention[92]. Strategies targeting the modulation of this axis and GI dysfunction are being explored with the goal of improving both GI and neurological outcomes[93], summarized in Figure 4.
Figure 4 Therapeutic approaches targeting the brain-gut axis following brain injury.
HPA: Hypothalamic-pituitary-adrenal; FMT: Fecal microbiota transplantation.
Nutritional interventions
As previously mentioned, early EN is essential. In addition to providing calories and proteins, special nutritional formulations also play a role[94]. Although early large-scale trials have yielded mixed results, more recent meta-analyses of critically ill patients, including those with TBI, have suggested that immune-modulating diets enriched with components, such as arginine and fish oils, can significantly reduce the incidence of infectious complications and shorten the length of hospital stay, although their impact on mortality remains controversial[95,96].
Probiotics, prebiotics, and synbiotics
Probiotics are live microorganisms that, when administered in a viable form, confer health benefits to the host[97]. Prebiotics are substrates selectively used by beneficial host microorganisms that confer health benefits[98]. Synbiotics are a combination of both, and are believed to restore healthy gut microbiota, offset dysbiosis, improve gut barrier function, reduce inflammation, and modulate the host’s immune response[99]. Several human studies and randomized controlled trials (RCTs) have corroborated these findings. For example, a recent RCT involving patients with severe TBI demonstrated that a seven-strain probiotic formulation not only decreased the incidence of ventilator-associated pneumonia but also significantly reduced systemic inflammatory markers, such as tumor necrosis factor-alpha, and improved feeding tolerance. Larger multicenter trials are required to confirm the effects on long-term neurological outcomes[100]. By restoring beneficial bacteria and their metabolites, such as SCFAs, these interventions may break the cycle of gut-brain inflammation[101].
FMT
FMT consists of transferring fecal matter from a healthy donor to a recipient to rebuild a healthy community of gut microbes[102]. It has been successfully used to treat recurrent Clostridium difficile infection(s) and has been explored for a multitude of other conditions. Although its application in the acute BI setting remains experimental, clinical evidence is slowly emerging. Case series and pilot studies involving stroke patients with severe complications, such as recurrent Clostridium difficile infection or antibiotic-associated diarrhea, have shown that FMT is a feasible rescue therapy. These initial human studies reported the restoration of gut microbial diversity and resolution of GI symptoms and, in some cases, observed improvements in neurological status, providing a crucial foundation for future controlled trials[103]. However, the safety and feasibility of FMT in the acute setting after BI has yet to be investigated[104].
VNS
The PNS plays a critical role in maintaining gut homeostasis and modulating inflammation[105]. Stimulation of the vagus nerve permits electrical stimulation that increases parasympathetic tone, improves gut motility, and activates the cholinergic anti-inflammatory pathway, leading to a reduction in both systemic inflammation and neuroinflammation[106]. The clinical translation of VNS is one of the most advanced areas of brain-gut axis modulation. Landmark clinical trials have demonstrated that invasive VNS combined with rehabilitation significantly improves motor function in survivors of chronic stroke. More recently, clinical studies have shown that noninvasive transcutaneous VNS is safe for patients with acute stroke, effectively modulating autonomic imbalance and reducing systemic levels of proinflammatory cytokines, highlighting its potential as an early intervention[107].
Pharmacological approaches
Various pharmacological agents can be used to target specific aspects of GI dysfunction. Prokinetic drugs, such as metoclopramide and erythromycin, are commonly used to improve gastric emptying and manage feeding intolerance, although their efficacy can vary. Clinical practice guidelines for neurocritical care support a stepwise approach for managing fecal incontinence. Although monotherapy with agents, such as metoclopramide, has yielded variable success, clinical evidence supports the use of combination therapy (e.g., metoclopramide plus erythromycin) in refractory cases. Furthermore, clinical trials are actively investigating novel pharmacological targets, including ghrelin receptor agonists and agents that enhance tight junction integrity (e.g., larazotide) to more effectively restore gut function after BI[108]. However, the effectiveness of these methods varies. Other potential pharmacological strategies include the use of agents that protect the gut mucosa, increase tight junction integrity, and selectively modulate inflammatory responses[109].
BRIDGING BASIC RESEARCH AND CLINICAL TRANSLATION
Bridging the gap between promising basic research and effective clinical translation is a critical challenge. However, the potential for this is significant, particularly in personalized brain-gut axis therapies. Previous preclinical studies have identified specific bacterial strains that produce neuroactive compounds. The next step involves designing robust, large-scale RCTs to validate their efficacy, safety, and optimal dosing in human populations with BI. Furthermore, advanced in vitro models, such as organoids and “gut-on-a-chip” technologies, offer a powerful platform to screen potential therapeutic agents and elucidate their mechanisms of action in a human-like physiological context. This can help “de-risk” and accelerate the transition from laboratory findings to clinical trials, ultimately improving the pipeline for the development of novel treatments for BI-induced GI dysfunction.
In summary, modulating the brain-gut axis represents a novel, multipronged approach for improving the care of patients with BI. Targeting the downstream effects of BI in the gut may reduce systemic inflammation, prevent infections, and mitigate the effects of secondary BI. While many of the strategies discussed above remain in the experimental or early clinical investigation stages, collectively, these findings represent a fundamental shift in the current approach to the management of BI and one that moves beyond the neurocentric view to consider the entire system.
FUTURE DIRECTIONS AND RESEARCH PERSPECTIVES
Despite the exciting findings discussed in the present review, many questions remain regarding the brain-gut axis after BI and there are several areas for future research.
Elucidating causal mechanisms
Despite the strong associations between gut dysfunction, dysbiosis, and poor neurological outcomes, the former causes the latter to be proven[110]. Innovative animal models [e.g., gnotobiotic (germ-free) animal models], as well as more sophisticated multi-omics (genomics, transcriptomics, proteomics, and metabolomics) approaches, are needed to delineate the specific molecular and cellular mechanisms by which the gut microbiota and its metabolites modulate neuroinflammation and brain repair processes[111].
Personalized medicine and biomarkers
The response to BI and subsequent gut decompensation is strikingly heterogeneous. Future efforts should be directed toward identifying biomarkers that can predict which patients will develop the most severe GI dysfunction (i.e., clinical implementation of gut-targeted therapies, biomarkers of gut permeability, specific microbial profiles, and inflammatory markers)[112]. Such biomarkers would enable more personalized care of these patients and would facilitate early targeted interventions for high-risk patients[113]. The role of the gut microbiota in drug metabolism and efficacy (i.e., “pharmacomicrobiomics”) will be important to understand variations in response to neuroprotective and other drugs used to treat the chronic sequelae of BI (e.g., ketogenic diet)[114].
Optimizing therapeutic interventions
Therapies, such as probiotics and VNS, hold promise in animal models; however, their application in acute settings is suboptimal. Large, well-designed, RCTs are needed to determine the optimal timing and dose, as well as the most appropriate probiotic strains/formulations that are safe for use in patients with BI[115]. For VNS, this will include the characterization of the patient population that is most likely to respond to stimulation and the long-term effects of these interventions on cognitive recovery, mood disorders, and other chronic sequelae of BI. In addition, the effects of these interventions on systemic inflammation need to be determined.
Role of diet and nutrition
The effects of specific dietary components on the gut microbiota and brain health are rapidly growing. Identification of the specific role that different types of dietary fibers, polyphenols, and other dietary components play in modulating the gut-brain axis after injury will enable the development of dietary guidelines based on solid evidence beyond providing simple caloric/protein support. The effects of other specialized diets, such as the ketogenic diet, which affect both brain metabolism and the gut microbiota, are exciting areas for future research.
Translational research and clinical implementation
Despite promising findings, significant challenges remain in clinical translation. Host factors, including genetics, baseline microbiome composition, diet, and lifestyle, contribute to substantial individual differences in response to interventions. The timing of an intervention is also a critical, yet poorly understood, factor; administration in the acute phase vs the chronic phase of BI may yield vastly different outcomes. Moreover, the effects of microbial therapies are often highly strain-specific, meaning that the benefits observed with one probiotic strain cannot necessarily be extrapolated to other strains. This necessitates rigorous strain-level characterization and testing. The application of multi-omics technologies, including genomics, transcriptomics, proteomics, and metabolomics, is essential for overcoming these hurdles. By creating a holistic view of host-microbe interactions, multi-omics can help identify novel biomarkers for patient stratification, predict treatment response, and pave the way for truly personalized therapeutic strategies. In summary, the mechanism(s) of the brain-gut axis after BI is a very active and clinically relevant area of investigation. Future basic research efforts focused on providing new mechanistic insights, personalized strategies, and rigorous clinical evaluations of new therapies may significantly improve the standard of care and outcomes in this vulnerable population.
LIMITATIONS OF THIS REVIEW
This study had several limitations. First, as a narrative review, it did not use a systematic search methodology, which may have introduced potential selection bias in the cited literature. Second, the brain-gut axis in BI is rapidly evolving and many of the findings discussed, particularly regarding therapeutic interventions, are derived from preclinical animal models. Translating these results into human patients remains a significant challenge. Finally, due to the breadth of the topic, we could not address all aspects in exhaustive detail, and some nuances of the complex interactions within the brain-gut axis may have been simplified.
CONCLUSION
GI dysfunction is a well-known and highly prevalent post-BI disorder with widespread dysregulation of the brain-gut axis. The subsequent breakdown is believed to form a vicious circle in which the initial CNS trauma leads to systemic inflammation, gut barrier breakdown, and dysregulation of the microbiota (Figure 1). These peripheral insults further induce secondary neuroinflammation and enhance primary brain damage. This model suggests a shift from purely neurocentric concepts toward a more holistic understanding of BI. Modulation of the gut using drugs, specific nutrition, and VNS is a novel approach to attack the gut and induce systemic changes that mitigate complications and secondary BI, which, in turn, improves recovery. Therefore, the brain-gut axis is central to the pathophysiology of BI and represents an important target for novel therapies.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report’s classification
Scientific Quality: Grade B, Grade B
Novelty: Grade C, Grade C
Creativity or Innovation: Grade B, Grade C
Scientific Significance: Grade B, Grade C
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
P-Reviewer: Jeene PM, PhD, Netherlands; Liberman M, PhD, Canada S-Editor: Wang JJ L-Editor: A P-Editor: Wang WB