Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.114946
Revised: December 11, 2025
Accepted: January 8, 2026
Published online: March 14, 2026
Processing time: 151 Days and 13.6 Hours
Cystic fibrosis liver disease (CFLD) is a significant comorbidity in individuals with cystic fibrosis (CF), marked by biliary fibrosis and progressive cholestasis. The advent of CF transmembrane conductance regulator (CFTR) modulators has revolutionized care for lung disease, but their impact on liver-specific disease and outcomes remain unclear. Additionally, the risk of comorbid cholestatic liver in
To provide a clinical framework for differentiating CFLD and drug induced liver injury (DILI), including from CFTR modulators.
A comprehensive literature review was conducted using PubMed, EMBASE, and Cochrane Library databases through March 2025. Studies evaluating pathogene
CFLD typically presents with insidious progression, portal hypertension, and biliary cirrhosis, whereas CFTR modulator-induced DILI often manifests acutely with jaundice, elevated liver enzymes, and a temporal association with therapy initiation. Key differentiators include biochemical patterns, imaging findings, response to drug withdrawal, and, when necessary, liver histology. Management strategies range from dose modification and supportive care in DILI to ursodeoxycholic acid, nutritional optimization, and portal hypertension management in CFLD.
Early recognition and differentiation between DILI and underlying CFLD are essential for optimizing therapy, pre
Core Tip: Cystic fibrosis (CF) liver disease is a major comorbidity in CF, complicated by progressive cholestasis and over
- Citation: Lee J, Yekula A, Wexler A, Zhuang W, Elangovan A, Rosario J, Burger P, Ramaraju G, Addissie B, Lim N, Narkewicz MR, Twohig P. Liver injury in cystic fibrosis: How to distinguish drug-induced liver injury from cystic fibrosis liver disease? World J Gastroenterol 2026; 32(10): 114946
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/114946.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.114946
Cystic fibrosis (CF) is a multisystem genetic disorder characterized by dysfunction of the CF transmembrane conductance regulator (CFTR) protein. While pulmonary complications dominate the clinical course, liver involvement, termed as CF hepatobiliary involvement (CFHBI) and advanced CF liver disease (aCFLD) are leading cause of non-pulmonary morbidity and mortality in CF patients[1]. CFHBI is the currently preferred term for hepatobiliary involvement in CF. This can range from liver enzyme abnormalities with or without imaging changes to portal hypertensions with or without cirrhosis[2]. While the pathophysiology of CFHBI is unclear, two theories predominate: Dysfunction of the CFTR protein resulting in defective chloride transport across epithelial cells, leading to cholestasis in the liver. This leads to inflammation, chronic infections, and organ damage[3]. Alternatively defective CFTR leads to alterations in intestinal permeability combined with an altered microbiome leads to hepatic inflammation due to gut liver axis abnormalities with the same end point[4].
The introduction of CFTR modulators has transformed CF care. These drugs include ivacaftor, lumacaftor/ivacaftor, tezacaftor/ivacaftor, elexacaftor/tezacaftor/ivacaftor (ETI) and the most recent addition vanzacaftor/tezacaftor/deuterated ivacaftor (VTD). Ivacaftor and deuterated ivacafotor, are part of the potentiator class that help open the CFTR channel and increase the flux of chloride and bicarbonate across the apical cell surface. Modulators such as lumacaftor, elexacaftor, tezacaftor and vanzacaftor are part of the corrector class that help normalize the folding of the defective CFTR protein and its movement to the cell surface[5,6].
Though these drugs have enhanced treatment options, they are known to cause elevations in serum hepatic enzyme levels compared to placebo. Hepatotoxicity associated with these agents has raised new challenges in distinguishing drug induced liver injury (DILI) from underlying CFHBI, when either hepatocellular or cholestatic liver test abnormalities emerge[3]. This systematic review aims to evaluate current evidence on pathogenesis, clinical features, diagnostics, and management to aid in distinguishing CF liver disease (CFLD) progression from DILI from CFTR modulators.
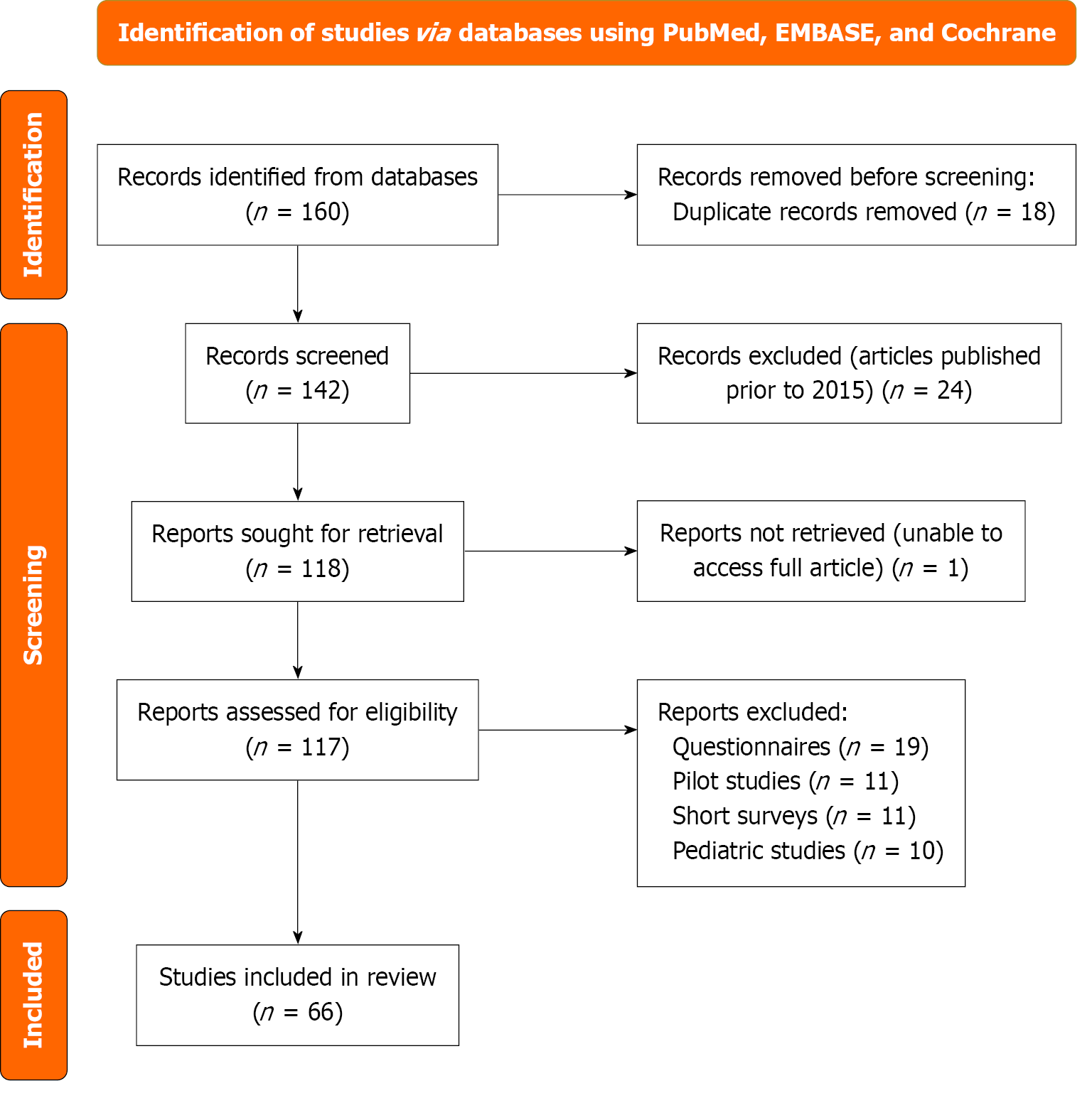
A comprehensive literature search was conducted to identify relevant studies evaluating CFHBI (often referred to as CFLD in older literature) and DILI from CFTR modulators. The search included articles from 2015 up to August 2025 across PubMed, EMBASE, and the Cochrane Library. The search strategy combined Medical Subject Headings and keywords related to CFLD, DILI, and CFTR modulators. Relevant key words such as “cystic fibrosis liver disease” or “CFTR modulators” or “drug induced liver injury” were used to find relevant sources. Additionally, the reference lists of included studies and relevant reviews were manually screened to identify any additional eligible studies. Studies were included based on the following criteria: Adult patients (≥ 18 years) with CFLD and/or CFTR modulator uses. We excluded literature related to non-human subjects, pediatric populations and articles not published in English. The authors independently screened titles and abstracts for eligibility, and articles were then retrieved for further evaluation of meeting eligibility criteria. Discrepancies were resolved through discussion by the authors to reach consensus (Figure 1).
A total of 59 articles were reviewed by the authors for inclusion in this systematic review. Once all the data from our literature search was collected, it was synthesized and organized into the following sections of the manuscript: Pathophysiology of liver injury in CF, DILI from CFTR modulators, differentiating CFLD and DILI in CF patients with abnormal liver function tests (LFTs), biochemical and imaging workup, diagnostic criteria and algorithms, management strategies, future directions and research needs. Furthermore, given the nature of this systematic review, the results and discussion sections were grouped together as both raw data from sources and our interpretation or summary of those results are outlined under each individual section of this paper.
CFLD: aCFLD may arise from inspissation of biliary secretions, ductal plugging, and periductal inflammation, leading to non-cirrhotic portal hypertension and ultimately multilobular cirrhosis. There is also a form of aCFLD that is associated with portal vasculopathy nodular regenerative hyperplasia leading to noncirrhotic portal hypertension whose pathophy
More recently, a phenotypic approach to CFHBI has been advocated[2]. This system uses liver enzyme abnormalities, imaging abnormalities, liver stiffness, presence or absence of portal hypertension with or without portal hypertension, and liver biopsy findings to classify hepatobiliary involvement. Pertinent to DILI, the prevalence of abnormal liver enzymes in CF is actually quite high. In a large study in children 95% of children had at least one abnormal liver enzyme by 18 years of age and 30% had persistently abnormal liver enzymes, typically < 3 times the upper limit of normal (ULN)[10]. The frequency of abnormal liver enzymes in persons with CF in clinical trials who were in the placebo arms of clinical drug trials demonstrated a prevalence of 2-4/100 person months[11]. This means that any evaluation for DILI is complicated by a relatively frequent rate of abnormal liver enzymes in CF that may be uncovered with the more frequent monitoring for DILI.
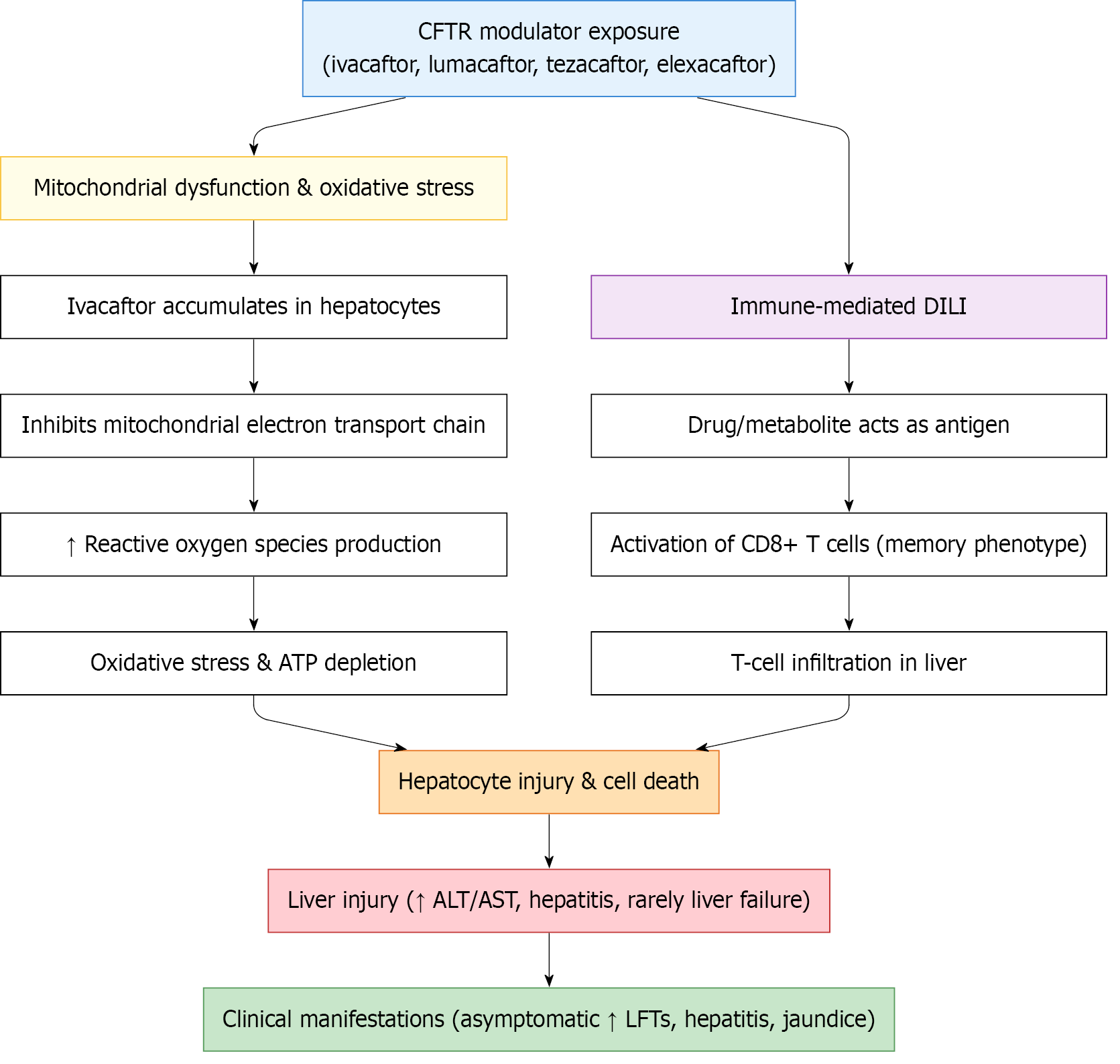
aCFLD typically manifests during childhood or adolescence but may be subclinical for years. Early signs of disease include mild transaminase elevations or hepatosplenomegaly, while advanced disease features cirrhosis and other sequelae of cirrhosis, like portal hypertension or variceal development[12]. Liver disease is an independent risk factor for mortality and represents one of the top 5 leading cause of death in CF, contributing to an overall 10-year cumulative mortality for cirrhotic CFLD in the CFF registry was 40%[13]. The development of cirrhosis, which occurs in about 7 percent of patients, is thought to be influenced by non-CFTR genetic variation and environmental factors[7]. There is conflicting evidence on the correlation of CFLD and sex, with some reports stating that portal hypertension specifically affects more males than females[7]. Contrarily, however, other reports suggest that there is no difference between sexes in the development of liver disease[14]. Genetic modifiers like SERPINA1 variants, particularly the Z allele, have been linked to a higher risk of liver damage in patients with CF. Carriers of these variants may show earlier or more aggressive progression of CFLD, highlighting the role of genetic modifiers in disease severity[15]. Proposed physiologic mechanisms of injury are outlined in Figure 2.
DILI from CFTR modulators: CFTR modulators have transformed CF care in recent years. However, concerns have been raised in the literature about their potential for DILI[16,17]. Ivacaftor, tezacaftor, and elexacaftor are primarily metabo
Clinical features and biomarkers: CFHBI spans a wide spectrum, from mild biochemical abnormalities and steatosis to hepatosplenomegaly, multilobular cirrhosis, and complications of portal hypertension including ascites and variceal bleeding[21]. Hepatomegaly is the most common physical examination finding in aCFLD, with splenomegaly also observed; however, the sensitivity and specificity of physical examination for detecting aCFLD are limited[22]. Many patients remain asymptomatic until advanced disease becomes clinically detectable, at which point features such as splenomegaly, ascites, and peripheral stigmata of chronic liver disease may appear[21].
Abnormalities in liver biochemistry are common in CFHBI, 53%-93% of patients demonstrating elevated aspartate aminotransferase (AST) or alanine aminotransferase levels in early adulthood and one-third showing elevated gamma-glutamyl transpeptidase (GGT) levels[10,23]. However, LFTs alone lack specificity for diagnosing or predicting a risk for development of aCFLD, as the values may be intermittently normal, even in advanced disease[21]. aCFLD usually manifests as a cholestatic or mixed biochemical pattern with elevated alkaline phosphatase (ALP) and GGT levels, whereas DILI is more likely to cause abrupt hepatocellular or mixed injury[24,25]. GGT is the most sensitive indicator, with high mean GGT levels > 35 U/L often associated with presence or risk for aCFLD with odds ratio of 39[26,27]. CFTR modulator DILI often presents acutely within weeks to months of therapy initiation, with disproportionate transaminase elevations or a mixed pattern[28]. Across clinical trials and observational studies, aminotransaminase elevations exceeding three times the ULN are observed in approximately 5%-11% of patients, with a smaller subset experiencing increases greater than five times the ULN[29-32]. Less marked increases are common; in one cohort, 75% had increases of at least 25% above baseline[32].
Biochemical and imaging workup: In CFHBI, ultrasonography may reveal heterogeneous echogenicity, nodular contour, or splenomegaly, and elastography demonstrates elevated liver stiffness (> 5.9 kPa for hepatobiliary involvement; > 8-9 kPa for advanced fibrosis)[33]. Magnetic resonance imaging with elastography offers high diagnostic accuracy and can demonstrate the patchy focal fibrosis characteristic of CFLD[34]. Signs of portal hypertension, such as varices and splenomegaly, further support the diagnosis of aCFLD. In contrast, imaging in DILI is nonspecific. Hepatomegaly, steatosis, or biliary dilation may be seen, but increased stiffness on elastography typically reflects transient inflammation and edema rather than established fibrosis[35].
Histopathology: Liver biopsy, although limited by sampling errors in CFHBI due to its patchy distribution, remains valuable in indeterminate cases[36]. A minority classified as possible or probable, emphasizing the utility of Roussel Uclaf Causality Assessment Method (RUCAM) in guiding management and minimizing unwarranted treatment discontinuation[37]. In adults, ALP may decline when elevated at baseline, GGT generally remains stable, and bilirubin may rise modestly, with approximately 9% developing new values above ULN and often isolated, likely reflecting OATP1B1/1B3 inhibition rather than intrinsic hepatotoxicity[37]. In children, ETI has been associated with reductions in AST among those with hepatic involvement, while GGT may increase slightly but remains within the normal range[38].
CFTR modulators carry strong liver safety precautions and require extra care in patients with hepatic impairment. For ETI (Trikafta), the United States Food and Drug Administration (FDA) label includes a boxed warning for serious and potentially fatal DILI, with cases of liver failure leading to transplantation and death. Use is not recommended in Child-Pugh B, and if being used, then reduced dose with close monitoring is advised. These drugs should not used in Child-Pugh C[39,40]. Patients with aCFLD may have a higher likelihood of severe outcomes if DILI occurs, and post-marketing/FDA Adverse Event Reporting System signals and label updates specifically highlight fatalities in patients with underlying hepatic impairment[41]. While case reports and small series document ETI use in aCFLD, any initiation should be individualized with heightened vigilance along with baseline and periodic LFT monitoring.
Diagnostic criteria and algorithms: Because unnecessary interruption of CFTR modulators can jeopardize clinical benefit, standardized approaches to causality assessment are recommended. The updated RUCAM provides a quantitative framework incorporating timing of exposure, biochemical profile, and competing diagnoses, with higher scores corre
| CFLD | DILI | |
| Clinical features | Spectrum from mild biochemical changes to hepatosplenomegaly, cirrhosis, and portal hypertension (ascites, variceal bleeding). Hepatomegaly is most common; splenomegaly also observed. Often asymptomatic until advanced stages (jaundice, pruritus, ascites) | Typically presents acutely after starting CFTR modulators. Jaundice and pruritus are common, especially in cholestatic or mixed injury patterns |
| Biochemical patterns | Elevated AST/ALT in 53%-93% of patients; GGT elevated in approximately 1/3. Cholestatic or mixed pattern with elevated ALP and GGT. LFTs may be intermittently normal | Hepatocellular or mixed injury more common. ALT/AST elevations > 3 times ULN in 5%-11%, > 5 times ULN in a subset. Often within weeks to months of drug initiation |
| Imaging findings | Ultrasound: Heterogeneous echogenicity, nodular contour, splenomegaly. Elastography: Elevated liver stiffness (> 5.9-9 kPa). MRI: Patchy focal fibrosis, high diagnostic accuracy, signs of portal hypertension | Imaging findings are nonspecific: Hepatomegaly, steatosis, biliary dilation. Elastography: Stiffness usually reflects inflammation/edema, not fibrosis |
| Histopathology | Focal biliary fibrosis, bile duct proliferation, bridging fibrosis, and multi-lobular cirrhosis. Nodular regenerative hyperplasia with portal hypertension | Acute injury with lobular disarray, hepatocellular necrosis, cholestasis, eosinophilic infiltrates. Severe: Extensive necrosis, ductular reactions, fibrosis |
| Diagnostic criteria | RUCAM used to assess causality. Biochemistry and imaging support diagnosis; biopsy helpful in uncertain cases. LFTs alone are not sufficient | RUCAM scores help determine likelihood of DILI. Useful in avoiding unnecessary CFTR therapy discontinuation |
| Response to CFTR modulators | ALP may decline, GGT stable, bilirubin may modestly increase (adults). AST may decrease in children; GGT may slightly rise but remains normal. Underlying CFLD does not increase DILI risk | Liver enzyme changes often reflect transporter inhibition (OATP1B1/1B3), not intrinsic toxicity. Most elevations are mild or transient |
In adults, ALP may decline when elevated at baseline, GGT generally remains stable, and bilirubin may rise modestly, with approximately 9% developing new values above ULN and often isolated, likely reflecting OATP1B1/1B3 inhibition rather than intrinsic hepatotoxicity[30]. In children, ETI has been associated with reductions in AST among those with hepatic involvement, while GGT may increase slightly but remains within the normal range[39]. Of note, CFLD itself does not appear to increase the risk of ETI related liver enzyme changes, indicating that underlying CFLD should not preclude therapy[29].
The main goals of management are to prevent progression of liver injury, optimize CF-related therapies, and maintain candidacy for advanced therapies (e.g., lung or liver transplantation).
CFLD: Ursodeoxycholic acid (UDCA) was the most used pharmacologic agent in the management of CFLD. It was thought to help protect hepatocytes from bile acid-induced injury by increasing bile flow by reducing bile viscosity and enhancing choleresis. Earlier studies suggested that long-term UDCA use stabilized or slow progression of CFLD, although recent randomized controlled trials have shown mixed efficacy and no improvement in disease progression to portal hypertension/aCFLD[12,22,41,42]. Updated guidelines recommend against the routine use of UDCA to prevent advanced liver disease in all people with CF.
Providers should not routinely prescribe UDCA to all CF patients with mild liver involvement with the sole purpose of preventing disease progression. Use of UCDA should be individualized, such as for patients who have obstruction of bile flow, or when cholestatic features are prominent. The patient’s liver disease, symptoms, risk factors, and assessment of benefits vs risks should be evaluated when deciding on treatment with UDCA.
Nutritional optimization is also central to aCFLD management, as malabsorption in these patients is exacerbated by both pancreatic insufficiency and impaired bile flow. Supplementation of fat-soluble vitamins (A, D, E, and K) is recommended. Vitamin K deficiency is particularly common in aCFLD, necessitating either high-dose oral or parenteral replacement. In addition to vitamin supplementation, optimizing caloric intake with high-energy diets and pancreatic enzyme replacement further supports growth and metabolic function[43,44].
As aCFLD progresses, portal hypertension is a frequent complication, leading to splenomegaly, hypersplenism, and esophageal varices. Thus, screening for varices and hepatocellular carcinoma is indicated in advanced disease[45]. Management strategies are extrapolated largely from non-CF populations, as robust CF-specific trials are lacking. Non-selective beta-blockers such as propranolol may be used to reduce variceal bleeding risk in patients at risk of decompen
Liver transplantation is the definitive therapy for patients with decompensated cirrhosis, progressive portal hyper
For DILI from CFTR modulators: For suspected CFTR modulator-associated DILI, the decision to discontinue therapy hinges on the injury severity, the trajectory of laboratory values, and the clinical benefit of continuing the modulator. Most programs pause therapy when the alanine aminotransferase/AST exceed 5-8 times the ULN or bilirubin ≥ 3 times the ULN, thresholds commonly used to flag clinically significant liver injury which are reflected in product labeling for ETI (Trikafta) and VTD[16,50]. Isolated low level bilirubin elevations should not lead to therapy pause. Patients with laboratory elevations below these values have been shown to be able to continue therapy[32]. However, pharmacovigilance data from the FDA Adverse Event Reporting System demonstrate a disproportionate association of ETI with DILI, supporting a low threshold for interruption and hepatology consultation[20]. Mechanistically, idiosyncratic DILI path
If liver tests return to baseline and an alternative cause is not identified, cautious rechallenge can be considered given the pulmonary and nutritional benefits of CFTR modulators among these patients. DILI data suggests rechallenging does not inherently increase fatal outcomes when done selectively with strict criteria and close monitoring[27,51]. In practice, this is often done in a stepwise fashion, starting with a reduced dose accompanied by frequent LFT monitoring and a low threshold to stop if labs rebound[52]. Although much literature in CF rechallenge therapy focuses on non-hepatic adverse events (e.g., rash) successfully managed with graded re-exposure, the same approach is used for hepatic DILI[52,53].
When a positive rechallenge or recurrent LFT injury confirms ETI-related DILI, switching to a different modulator (e.g., tezacaftor/ivacaftor or ivacaftor monotherapy or VTD) may be appropriate because cross-hepatotoxicity is not universal[30]. The current standards-of-care emphasize genotype-specific alternatives and individualized risk-benefit discussions[22]. Case reports of hepatitis/necrosis with ETI underscore the importance of ETI discontinuation in severe cases, with the option for a subsequent trial of an alternative modulator with intensified monitoring[16,22].
There is no proven hepatoprotective antidote for CFTR modulator-associated DILI. Current innovation focuses on better risk detection (e.g., pharmacovigilance analytics showing ETI safety signals, exploratory biomarkers), safer re-initiation frameworks (stepwise dosing, potential therapeutic drug monitoring to individualize exposure), and mecha
CFTR modulators and the gut microbiota: CFTR modulators, especially ETI, partially reverse gut dysbiosis in CF by reducing pro-inflammatory bacteria, such as Escherichia coli, Shigella, and Staphylococcus) and increasing beneficial taxa, such as Blautia and Romboutsia. These changes usually result after about 6 months and often correlate with lower fecal calprotectin levels and intestinal inflammation[54-56]. However, persistent dysbiosis remains common due to ongoing antibiotics, pancreatic insufficiency, and dietary factors, so microbiome benefits are modest, delayed, and variable across patients[57-60]. Longer follow-up and integrated large-scale studies are needed to clarify durability and clinical impact[54].
Over the past decade, CFTR modulators have slowly become ubiquitous along with dramatic advances in CTFR science that have changed the therapeutic landscape. There is still an unmet need for comprehensive, prospective, longitudinal studies to better understand the outcomes, especially of liver disease in CF and drug related adverse events given current scarce data. While recent real world and registry data such as the French national CF cohorts suggest that CFTR modu
There is a growing recognition of genetic and environmental risk modifiers in CF. For example, a recent case report identified heterozygous alpha1-antitrypsin deficiency (SERPINA1 Z allele) as possibly predisposing to severe DILI in a child started on ETI[15,64]. Incorporating genotyping assessment of prior liver disease, nutritional status, microbiome and bile acid metabolism could help stratify risk, guide dosage or monitoring schedules. Studies like the PREDICT-CF are studying the variations in ppFEV1 and improvement on lumacaftor/ivacaftor. Development of patient models using patient cells gives access to understanding individual transcriptomic and proteomic background and its impact on drug pharmacology, which also help with personalized therapies[65,66].
Genetic predisposition contributes to the heterogeneity of CFLD. Although the disease is largely confined to severe genotypes, no correlation has been established with specific mutations. To date, SERPINA1, encoding α-1 antitrypsin, is the only modifier gene identified[67]. The SERPINA1 Z allele increases the risk of portal hypertension and advanced liver disease, possibly through endoplasmic reticulum stress and cholangiocyte injury[34,68]. Its identification as the first genetic marker of severe CFLD highlights the potential for early risk stratification and intervention. However, because the Z allele occurs in only approximately 2.7% of patients with CF and only accounted for 3% of aCFLD subjects, which suggests additional modifier genes involvement[68,69].
Overall, the development and research on these liver-targeted therapies offers promising opportunities in the management of CF. A recent preclinical study in CF rabbit models showed that the sodium-glucose co-transporter 1/2 inhibitor sotagliflozin improves liver disease markers, bile acid profiles, and reduces fibrosis and endoplasmic reticulum stress[70]. Ongoing studies have also explored the use of prenatal CFTR modulators which showed interesting the
While this study provides a comprehensive synthesis of current literature on distinguishing CFLD from DILI in the context of CFTR modulator therapy, several limitations should be acknowledged. First, much of the available evidence is derived from retrospective studies, case series, and registry data, which are subject to selection bias, incomplete data capture, and lack of standardized diagnostic criteria for both CFLD and DILI. The heterogeneity in study populations, definitions of liver injury, and outcome measures further limits the generalizability of findings. Many studies included small sample sizes and short follow-up periods, restricting the ability to assess long-term outcomes and rare adverse events. Additionally, pediatric populations and non-English language studies were excluded, potentially omitting relevant data and limiting applicability to all CF patients. The evolving landscape of CFTR modulator use means that real-world experience and longitudinal data remain limited, particularly regarding the natural history of liver disease in the era of highly effective modulator therapy. Finally, the study relies on published literature up to March 2025, and emerging data or unpublished studies may not be reflected. These limitations highlight the need for prospective, mul
The advent of CFTR modulators has significantly altered the disease trajectory for many patients with CF, yet persistent challenges in managing liver manifestations necessitate continued research. Specifically, further investigation is war
| 1. | Flass T, Narkewicz MR. Cirrhosis and other liver disease in cystic fibrosis. J Cyst Fibros. 2013;12:116-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 141] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 2. | Bodewes FAJA, Freeman AJ, Weymann A, Debray D, Scheers I, Verkade HJ, Narkewicz MR. Towards a Standardized Classification of the Hepatobiliary Manifestations in Cystic Fibrosis (CFHBI): A Joint ESPGHAN/NASPGHAN Position Paper. J Pediatr Gastroenterol Nutr. 2024;78:153-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 3. | Moiceanu ES, Leucuța DC, Nițescu VG, Lescaie A, Iacobescu M, Stan IV, Moșescu SE, Vivisenco IC, Dumitrașcu DL. Impact of CFTR Modulator Therapies on Liver Function in Cystic Fibrosis Patients: A Systematic Review of Hepatic Biomarkers. J Gastrointestin Liver Dis. 2024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Flass T, Tong S, Frank DN, Wagner BD, Robertson CE, Kotter CV, Sokol RJ, Zemanick E, Accurso F, Hoffenberg EJ, Narkewicz MR. Intestinal lesions are associated with altered intestinal microbiome and are more frequent in children and young adults with cystic fibrosis and cirrhosis. PLoS One. 2015;10:e0116967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 5. | Taylor-Cousar JL, Robinson PD, Shteinberg M, Downey DG. CFTR modulator therapy: transforming the landscape of clinical care in cystic fibrosis. Lancet. 2023;402:1171-1184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 118] [Article Influence: 39.3] [Reference Citation Analysis (0)] |
| 6. | Veit G, Vaccarin C, Lukacs GL. Elexacaftor co-potentiates the activity of F508del and gating mutants of CFTR. J Cyst Fibros. 2021;20:895-898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 7. | Stonebraker JR, Ooi CY, Pace RG, Corvol H, Knowles MR, Durie PR, Ling SC. Features of Severe Liver Disease With Portal Hypertension in Patients With Cystic Fibrosis. Clin Gastroenterol Hepatol. 2016;14:1207-1215.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 96] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 8. | Fiorotto R, Strazzabosco M. Pathophysiology of Cystic Fibrosis Liver Disease: A Channelopathy Leading to Alterations in Innate Immunity and in Microbiota. Cell Mol Gastroenterol Hepatol. 2019;8:197-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 9. | Betapudi B, Aleem A, Kothadia JP. Cystic Fibrosis and Liver Disease. 2023 Jan 9. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 10. | Woodruff SA, Sontag MK, Accurso FJ, Sokol RJ, Narkewicz MR. Prevalence of elevated liver enzymes in children with cystic fibrosis diagnosed by newborn screen. J Cyst Fibros. 2017;16:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 11. | Mayer-Hamblett N, Kloster M, Ramsey BW, Narkewicz MR, Saiman L, Goss CH. Incidence and clinical significance of elevated liver function tests in cystic fibrosis clinical trials. Contemp Clin Trials. 2013;34:232-238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Toledano MB, Mukherjee SK, Howell J, Westaby D, Khan SA, Bilton D, Simmonds NJ. The emerging burden of liver disease in cystic fibrosis patients: A UK nationwide study. PLoS One. 2019;14:e0212779. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 13. | Leung DH, Ye W, Schwarzenberg SJ, Freeman AJ, Palermo JJ, Weymann A, Alonso EM, Karnsakul WW, Murray KF, Stoll JM, Huang S, Karmazyn B, Masand P, Magee JC, Alazraki AL, Towbin AJ, Nicholas JL, Green N, Otto RK, Siegel MJ, Ling SC, Navarro OM, Harned RK, Narkewicz MR, Molleston JP; CFLD Research Network. Long-term follow-up and liver outcomes in children with cystic fibrosis and nodular liver on ultrasound in a multi-center study. J Cyst Fibros. 2023;22:248-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 14. | Wilschanski M, Rivlin J, Cohen S, Augarten A, Blau H, Aviram M, Bentur L, Springer C, Vila Y, Branski D, Kerem B, Kerem E. Clinical and genetic risk factors for cystic fibrosis-related liver disease. Pediatrics. 1999;103:52-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 82] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Boëlle PY, Debray D, Guillot L, Corvol H; French CF Modifier Gene Study Investigators. SERPINA1 Z allele is associated with cystic fibrosis liver disease. Genet Med. 2019;21:2151-2155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 16. | Lowry S, Mogayzel PJ, Oshima K, Karnsakul W. Drug-induced liver injury from elexacaftor/ivacaftor/tezacaftor. J Cyst Fibros. 2022;21:e99-e101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 17. | Stylemans D, François S, Vincken S, Verbanck S, Vanderhelst E. A case of self-limited drug induced liver injury under treatment with elexacaftor/tezacaftor/ivacaftor: When it is worth taking the risk. J Cyst Fibros. 2021;20:712-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Hong E, Almond LM, Chung PS, Rao AP, Beringer PM. Physiologically-Based Pharmacokinetic-Led Guidance for Patients With Cystic Fibrosis Taking Elexacaftor-Tezacaftor-Ivacaftor With Nirmatrelvir-Ritonavir for the Treatment of COVID-19. Clin Pharmacol Ther. 2022;111:1324-1333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 19. | Apel K, Pütz K, Tolkach Y, Canbay A, Drebber U. [Drug-induced liver injury-significance of pathology]. Pathologe. 2020;41:457-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 20. | Shi A, Nguyen H, Kuo CB, Beringer PM. Drug-induced liver injury associated with elexacaftor/tezacaftor/ivacaftor: A pharmacovigilance analysis of the FDA adverse event reporting system (FAERS). J Cyst Fibros. 2024;23:566-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 21. | Debray D, Kelly D, Houwen R, Strandvik B, Colombo C. Best practice guidance for the diagnosis and management of cystic fibrosis-associated liver disease. J Cyst Fibros. 2011;10 Suppl 2:S29-S36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 293] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 22. | Sellers ZM, Assis DN, Paranjape SM, Sathe M, Bodewes F, Bowen M, Cipolli M, Debray D, Green N, Hughan KS, Hunt WR, Leey J, Ling SC, Morelli G, Peckham D, Pettit RS, Philbrick A, Stoll J, Vavrina K, Allen S, Goodwin T, Hempstead SE, Narkewicz MR. Cystic fibrosis screening, evaluation, and management of hepatobiliary disease consensus recommendations. Hepatology. 2024;79:1220-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 59] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 23. | Sakiani S, Kleiner DE, Heller T, Koh C. Hepatic Manifestations of Cystic Fibrosis. Clin Liver Dis. 2019;23:263-277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 24. | Fontana RJ, Liou I, Reuben A, Suzuki A, Fiel MI, Lee W, Navarro V. AASLD practice guidance on drug, herbal, and dietary supplement-induced liver injury. Hepatology. 2023;77:1036-1065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 156] [Article Influence: 52.0] [Reference Citation Analysis (5)] |
| 25. | Cho EJ, Jeong SM, Chung GE, Yoo JJ, Cho Y, Lee KN, Shin DW, Kim YJ, Yoon JH, Han K, Yu SJ. Gamma-glutamyl transferase and risk of all-cause and disease-specific mortality: a nationwide cohort study. Sci Rep. 2023;13:1751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 26. | Bodewes FA, van der Doef HP, Houwen RH, Verkade HJ. Increase of Serum γ-Glutamyltransferase Associated With Development of Cirrhotic Cystic Fibrosis Liver Disease. J Pediatr Gastroenterol Nutr. 2015;61:113-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 27. | Højte C, Olsen MF, Faurholt-Jepsen D, Jørgensen MH, Pressler T, Leo-Hansen C, Bryrup T, Jeppesen M, Olesen HV, Petersen J, Katzenstein TL, Nielsen BU, Skov M; TransformCF Study Group. Severe hepatotoxicity is uncommon following the introduction of Elexacaftor/Tezacaftor/Ivacaftor: A real-world two-years follow-up study of the Danish cystic fibrosis cohort. J Cyst Fibros. 2025;24:998-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 28. | Regard L, Martin C, Burnet E, Da Silva J, Burgel PR. CFTR Modulators in People with Cystic Fibrosis: Real-World Evidence in France. Cells. 2022;11:1769. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 29. | Heijerman HGM, McKone EF, Downey DG, Van Braeckel E, Rowe SM, Tullis E, Mall MA, Welter JJ, Ramsey BW, McKee CM, Marigowda G, Moskowitz SM, Waltz D, Sosnay PR, Simard C, Ahluwalia N, Xuan F, Zhang Y, Taylor-Cousar JL, McCoy KS; VX17-445-103 Trial Group. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: a double-blind, randomised, phase 3 trial. Lancet. 2019;394:1940-1948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1220] [Cited by in RCA: 1082] [Article Influence: 154.6] [Reference Citation Analysis (0)] |
| 30. | Middleton PG, Mall MA, Dřevínek P, Lands LC, McKone EF, Polineni D, Ramsey BW, Taylor-Cousar JL, Tullis E, Vermeulen F, Marigowda G, McKee CM, Moskowitz SM, Nair N, Savage J, Simard C, Tian S, Waltz D, Xuan F, Rowe SM, Jain R; VX17-445-102 Study Group. Elexacaftor-Tezacaftor-Ivacaftor for Cystic Fibrosis with a Single Phe508del Allele. N Engl J Med. 2019;381:1809-1819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1922] [Cited by in RCA: 1685] [Article Influence: 240.7] [Reference Citation Analysis (0)] |
| 31. | Tewkesbury DH, Athwal V, Bright-Thomas RJ, Jones AM, Barry PJ. Longitudinal effects of elexacaftor/tezacaftor/ivacaftor on liver tests at a large single adult cystic fibrosis centre. J Cyst Fibros. 2023;22:256-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 32. | Wood M, Babowicz F, Kennedy AG, Antell M, Gilhooly E, Tompkins BJ, Reddy SS. Incidence of transaminitis in adults with cystic fibrosis taking elexacaftor/tezacaftor/ivacaftor. J Am Pharm Assoc (2003). 2023;63:920-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 33. | Piñar-Gutiérrez A, Quintana-Gallego E, Remón-Ruiz PJ, Pizarro Á, González-Navarro I, Jiménez-Sánchez A, García-Rey S, Roque-Cuéllar MDC, Gato S, Domínguez I, Castell FJ, Romero-Gómez M, García-Luna PP. Non-invasive evaluation of steatosis and fibrosis in the liver in adults patients living with cystic fibrosis. J Cyst Fibros. 2025;24:581-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 34. | Dana J, Debray D, Beaufrère A, Hillaire S, Fabre M, Reinhold C, Baumert TF, Berteloot L, Vilgrain V. Cystic fibrosis-related liver disease: Clinical presentations, diagnostic and monitoring approaches in the era of CFTR modulator therapies. J Hepatol. 2022;76:420-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 75] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 35. | Sagir A, Erhardt A, Schmitt M, Häussinger D. Transient elastography is unreliable for detection of cirrhosis in patients with acute liver damage. Hepatology. 2008;47:592-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 382] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 36. | Lewindon PJ, Shepherd RW, Walsh MJ, Greer RM, Williamson R, Pereira TN, Frawley K, Bell SC, Smith JL, Ramm GA. Importance of hepatic fibrosis in cystic fibrosis and the predictive value of liver biopsy. Hepatology. 2011;53:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 83] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 37. | Tewkesbury D, Jones AM, Bright-Thomas R, Cratchley A, Hanley KP, Wyatt J, Athwal V, Barry PJ. Aetiology of Significant Liver Test Abnormalities in a Single-Centre Cohort of People with Cystic Fibrosis Exposed to Elexacaftor/Tezacaftor/Ivacaftor, Utilizing the Updated RUCAM. Drugs. 2023;83:1699-1707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 38. | Diemer S, Elidottir H, Eklund EA, Påhlman LI, Hansen C. The effect of elexacaftor-tezacaftor-ivacaftor on liver stiffness in children with cystic fibrosis. J Pediatr Gastroenterol Nutr. 2025;81:74-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Anton-Păduraru DT, Azoicăi AN, Trofin F, Murgu AM, Mîndru DE, Bocec AS, Halițchi COI, Zota GR, Păduraru D, Nastase EV. Diagnosis, Management, and Prognosis of Cystic Fibrosis-Related Liver Disease in Children. Diagnostics (Basel). 2024;14:538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 40. | Johnson E, Riddles T, Smith D, Henderson D, Masel P, Reid DW, Moore V, Evans IES. Disproportionate Rates of Liver Injury in People With Cystic Fibrosis on Elexacaftor/Tezacaftor/Ivacaftor in Queensland, Australia. Gastro Hep Adv. 2025;4:100641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 41. | Shi A, Cornwell C, Yang K, Beringer PM. Quantitative Systems Toxicology Predicts Ivacaftor-Induced Oxidative Stress Contributes to CFTR Modulator Hepatotoxicity. Clin Pharmacol Ther. 2026;119:208-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 42. | Colombo C, Alicandro G, Oliver M, Lewindon PJ, Ramm GA, Ooi CY, Alghisi F, Kashirskaya N, Kondratyeva E, Corti F, Padoan R, Asherova I, Evans H, de Monestrol I, Strandvik B, Lindblad A; CF UDCA study group. Ursodeoxycholic acid and liver disease associated with cystic fibrosis: A multicenter cohort study. J Cyst Fibros. 2022;21:220-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 43. | Connor AA, Huang HJ, Mobley CM, Graviss EA, Nguyen DT, Goodarzi A, Saharia A, Yau S, Hobeika MJ, Suarez EE, Moaddab M, Brombosz EW, Moore LW, Yi SG, Gaber AO, Ghobrial RM. Progress in Combined Liver-lung Transplantation at a Single Center. Transplant Direct. 2023;9:e1482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 44. | Krzyżanowska P, Drzymała-Czyż S, Pogorzelski A, Duś-Żuchowska M, Skorupa W, Bober L, Sapiejka E, Oralewska B, Rohovyk N, Moczko J, Nowak J, Wenska-Chyży E, Rachel M, Lisowska A, Walkowiak J. Vitamin K status in cystic fibrosis patients with liver cirrhosis. Dig Liver Dis. 2017;49:672-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 45. | Rowland M, Bourke B. Liver disease in cystic fibrosis. Curr Opin Pulm Med. 2011;17:461-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 46. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2053] [Article Influence: 513.3] [Reference Citation Analysis (15)] |
| 47. | Tanner S, Schulman AR. Endoscopic Management of Portal Hypertension and Varices. Tech Vasc Interv Radiol. 2025;28:101053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 48. | Albaiz FA, Semenchuk J, Ma X, Lightfoot D, Gonska T, Tullis E, Stephenson AL. Clinical outcomes after liver transplant in people with cystic fibrosis: A systematic review and meta-analysis. J Cyst Fibros. 2024;23:1112-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 49. | Sherrard LJ, Einarsson GG, Johnston E, O'Neill K, McIlreavey L, McGrath SJ, Gilpin DF, Downey DG, Reid A, McElvaney NG, Boucher RC, Muhlebach MS, Elborn JS, Tunney MM. Assessment of stability and fluctuations of cultured lower airway bacterial communities in people with cystic fibrosis. J Cyst Fibros. 2019;18:808-816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 50. | Mosedale M, Watkins PB. Drug-induced liver injury: Advances in mechanistic understanding that will inform risk management. Clin Pharmacol Ther. 2017;101:469-480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 158] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 51. | Pinazo-Bandera JM, Niu H, Alvarez-Alvarez I, Medina-Cáliz I, Del Campo-Herrera E, Ortega-Alonso A, Robles-Díaz M, Hernández N, Paraná R, Nunes V, Girala M, Bessone F, Lucena MI, Andrade RJ, García Cortés M. Rechallenge in idiosyncratic drug-induced liver injury: An analysis of cases in two large prospective registries according to existing definitions. Pharmacol Res. 2024;203:107183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 52. | Hunt CM, Papay JI, Stanulovic V, Regev A. Drug rechallenge following drug-induced liver injury. Hepatology. 2017;66:646-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (3)] |
| 53. | Diseroad ER, Mogayzel PJ Jr, Pan A. Rechallenge of Elexacaftor/Tezacaftor/Ivacaftor After Skin Rash in Two Pediatric Patients. J Pediatr Pharmacol Ther. 2022;27:463-466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 54. | Knoll RL, Brauny MM, Robert E, Cloos L, Waser L, Hilbert K, Ulmer N, Hillen B, Birkner T, Bartolomaeus TUP, Nitsche O, Jarquín-Díaz VH, Lynch S, Gehring S, Maier L, Poplawska K, Forslund-Startceva SK. CFTR modulator therapy drives microbiome restructuring through improved host physiology in cystic fibrosis: the IMMProveCF phase IV trial. Nat Commun. 2025;16:10111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 55. | Gutiérrez-Díaz I, Gutiérrez-Martinez JR, García-Romero R, Vicente-Santamaría S, De La Mano-Hernández A, Torcuato-Rubio E, Garriga-Garcia M, Martin-Fernández C, Baston-Paz N, Simon-Bernaldo de Quiros C, Díaz-Martín JJ, Delgado-Palacios S, González-Jiménez D. Changes in intestinal microbiota in pediatric cystic fibrosis patients after 6 months of elexacaftor-tezacaftor-ivacaftor therapy. J Pediatr Gastroenterol Nutr. 2025;81:1451-1459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 56. | Duong JT, Hayden HS, Verster AJ, Pope CE, Miller C, Kelsi Penewit, Salipante SJ, Rowe SM, Solomon GM, Nichols D, Kelly A, Schwarzenberg SJ, Freedman SD, Hoffman LR. Fecal microbiota changes in people with cystic fibrosis after 6 months of elexacaftor/tezacaftor/ivacaftor: Findings from the promise study. J Cyst Fibros. 2025;24:792-800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 57. | Marsh R, Santos CD, Yule A, Dellschaft NS, Hoad CL, Ng C, Major G, Smyth AR, Rivett D, van der Gast C. Impact of extended Elexacaftor/Tezacaftor/Ivacaftor therapy on the gut microbiome in cystic fibrosis. J Cyst Fibros. 2024;23:967-976. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 58. | Pope CE, Vo AT, Hayden HS, Weiss EJ, Durfey S, McNamara S, Ratjen A, Grogan B, Carter S, Nay L, Parsek MR, Singh PK, McKone EF, Aitken ML, Rosenfeld MR, Hoffman LR. Changes in fecal microbiota with CFTR modulator therapy: A pilot study. J Cyst Fibros. 2021;20:742-746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 59. | Kristensen M, Prevaes SMPJ, Kalkman G, Tramper-Stranders GA, Hasrat R, de Winter-de Groot KM, Janssens HM, Tiddens HA, van Westreenen M, Sanders EAM, Arets B, Keijser B, van der Ent CK, Bogaert D. Development of the gut microbiota in early life: The impact of cystic fibrosis and antibiotic treatment. J Cyst Fibros. 2020;19:553-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 60. | Wainwright CE. A New Era for Cystic Fibrosis and Cystic Fibrosis Transmembrane Conductance Regulator Modulator Trials in Infants. Am J Respir Crit Care Med. 2022;206:1193-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 61. | Mouliade C, Parlati L, Tzedakis S, Collier M, Bouam S, Vallet-Pichard A, D'Halluin-Venier V, Kanaan R, Pol S, Sogni P, Burgel PR, Mallet V; Demosthenes Research Group. Cystic fibrosis liver disease progression in the era of elexacaftor-tezacaftor-ivacaftor. JHEP Rep. 2025;7:101512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 62. | Vongsavath T, Tun KM, Mukhopadhyay D, Lo CH, Singh A, Amin R, Twohig P, Saab S, Manne V. Effect of Cystic Fibrosis Transmembrane Conductance Regulator Modulators on Liver Enzymes Among Patients With Cystic Fibrosis: A Systematic Review and Meta-Analysis. Gastro Hep Adv. 2025;4:100752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 63. | Auvray M, Laborde N, Mittaine M, Barreau F, Labouret G, Roditis L, Flumian C, Arrouy A, Mas E. Evolution of hepatobiliary involvement in cystic fibrosis children on CFTR modulators. J Cyst Fibros. 2025;24:1006-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 64. | Kinuani R, Ezri J, Kernen Y, Rochat I, Blanchon S. Case Report: When cystic fibrosis, elexacaftor/tezacaftor/ivacaftor therapy, and alpha1 antitrypsin deficiency get together. Front Pediatr. 2024;12:1378744. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 65. | Bardin E, Pastor A, Semeraro M, Golec A, Hayes K, Chevalier B, Berhal F, Prestat G, Hinzpeter A, Gravier-Pelletier C, Pranke I, Sermet-Gaudelus I. Modulators of CFTR. Updates on clinical development and future directions. Eur J Med Chem. 2021;213:113195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 66. | Bouazza N, Urien S, Foissac F, Choupeaux L, Lui G, Froelicher Bournaud L, Rouillon S, Zheng Y, Bardin E, Stremler N, Bessaci K, Bihouee T, Coirier-Duet E, Marguet C, Deneuville E, Laurans M, Reix P, Gerardin M, Mittaine M, Epaud R, Thumerelle C, Weiss L, Berthaud R, Semeraro M, Treluyer JM, Benaboud S, Sermet-Gaudelus I. Lumacaftor/Ivacaftor Population Pharmacokinetics in Pediatric Patients with Cystic Fibrosis: A First Step Toward Personalized Therapy. Clin Pharmacokinet. 2024;63:333-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 67. | Boëlle PY, Debray D, Guillot L, Clement A, Corvol H; French CF Modifier Gene Study Investigators. Cystic Fibrosis Liver Disease: Outcomes and Risk Factors in a Large Cohort of French Patients. Hepatology. 2019;69:1648-1656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 68. | Bartlett JR, Friedman KJ, Ling SC, Pace RG, Bell SC, Bourke B, Castaldo G, Castellani C, Cipolli M, Colombo C, Colombo JL, Debray D, Fernandez A, Lacaille F, Macek M Jr, Rowland M, Salvatore F, Taylor CJ, Wainwright C, Wilschanski M, Zemková D, Hannah WB, Phillips MJ, Corey M, Zielenski J, Dorfman R, Wang Y, Zou F, Silverman LM, Drumm ML, Wright FA, Lange EM, Durie PR, Knowles MR; Gene Modifier Study Group. Genetic modifiers of liver disease in cystic fibrosis. JAMA. 2009;302:1076-1083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 209] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 69. | Debray D, Corvol H, Housset C. Modifier genes in cystic fibrosis-related liver disease. Curr Opin Gastroenterol. 2019;35:88-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 70. | Liang X, Hou X, Bouhamdan M, Sun Y, Song Z, Rajagopalan C, Jiang H, Wei HG, Song J, Yang D, Guo Y, Zhang Y, Mou H, Zhang J, Chen YE, Sun F, Jin JP, Zhang K, Xu J. Sotagliflozin attenuates liver-associated disorders in cystic fibrosis rabbits. JCI Insight. 2024;9:e165826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 71. | Gómez-Montes E, Salcedo Lobato E, Galindo Izquierdo A, García Alcázar D, Villalain González C, Moral-Pumarega MT, Bustos Lozano G, Luna-Paredes C. Prenatal Cystic Fibrosis Transmembrane Conductance Regulator Modulator Therapy: A Promising Way to Change the Impact of Cystic Fibrosis. Fetal Diagn Ther. 2023;50:136-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 72. | Pastor-Vivero MD, Costa I Colomer J, Martín de Vicente C, Vicente-Santamaria S, García Romero R, González Jiménez D, Luna Paredes C. Avances en el tratamiento de la fibrosis quística: los moduladores de la CFTR. An Pediatr. 2025;102:503857. [DOI] [Full Text] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/