Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.113771
Revised: October 31, 2025
Accepted: December 16, 2025
Published online: March 14, 2026
Processing time: 174 Days and 20 Hours
Spasmolytic polypeptide-expressing metaplasia (SPEM) is a gastric precancerous lesion (GPL) with high malignant potential. The ethyl acetate extract of Celastrus orbiculatus Thunb. effectively ameliorates GPL and gastric cancer progression. Meanwhile, the primary active constituent of this plant, pristimerin, also demon
To investigate the therapeutic effects of pristimerin on SPEM and its underlying mechanisms.
Pristimerin was administered to high-dose tamoxifen-induced SPEM mice to assess its effects on pathological progression, glycolytic reprogramming, and Cdkn1c (p57) expression. Human gastric epithelial (GES-1) cells were treated with tamoxifen and then with pristimerin or 2-deoxy-D-glucose to demonstrate that pristimerin ameliorates SPEM by regulating glycolytic reprogramming. Further
Pristimerin effectively ameliorated gastric mucosal damage and oxyntic atrophy induced by high-dose tamoxifen, suppressed the aberrant upregulation of key glycolytic regulators, SPEM-specific markers, and stem cell markers, and upregulated p57 expression. In tamoxifen-induced GES-1 cells, pristimerin exhibited comparable therapeutic effects. Crucially, glycolysis inhibition in GES-1 cells effectively ameliorated tamoxifen-induced SPEM-associated phenotypes. In gastric organoids, Cdkn1c overexpression suppressed glycolytic reprogramming and SPEM pheno
Pristimerin effectively ameliorates gastric mucosal pathological damage and oxyntic atrophy in high-dose tam
Core Tip: This study systematically investigated the therapeutic effects of the natural compound pristimerin on spasmolytic polypeptide-expressing metaplasia (SPEM) using a high-dose tamoxifen-induced mouse model, tamoxifen-treated human gastric epithelial cells, and N-methyl-N’-nitro-N-nitrosoguanidine/Helicobacter pylori-induced gastric organoids. It elucidated the underlying mechanism by which pristimerin ameliorates SPEM through regulating Cdkn1c (p57) to suppress glycolytic reprogramming. These findings not only provide novel mechanistic insights into pristimerin treatment for SPEM but also offer a crucial theoretical basis for targeting p57 in SPEM therapy.
- Citation: Wen JS, Pan ZW, Yao XD, Liu YQ, Zhu YD. Pristimerin ameliorates spasmolytic polypeptide-expressing metaplasia by modulating Cdkn1c (p57)-mediated glycolytic reprogramming. World J Gastroenterol 2026; 32(10): 113771
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/113771.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.113771
Based on 2022 global epidemiology statistics, gastric cancer (GC) was the fifth-leading malignancy worldwide in terms of both incidence and mortality[1]. Due to its insidious early symptoms and lack of effective treatments, GC constitutes a substantial public health burden. In recent years, the research focus has gradually shifted from “GC treatment” to “interception of precancerous lesions”. Among these, spasmolytic polypeptide-expressing metaplasia (SPEM) has garnered widespread attention due to its malignant transformation potential. As a secondary lesion of chronic atrophic gastritis (CAG), SPEM exhibits a higher propensity to progress to malignancy compared to CAG[2]. Indeed, studies dem
As a key candidate tumor suppressor within the cyclin-dependent kinase-interacting protein/kinase inhibitory protein (CIP/KIP) family, p57 (Cdkn1c) inhibits tumorigenesis by regulating critical cancer cell processes such as proliferation, migration, and aerobic glycolysis[5]. Downregulation of this factor in advanced-stage tumors leads to loss of its function, while reactivation of p57 expression restores its tumor-suppressive function[6]. Similarly, evidence indicates that p57 is significantly downregulated in injured gastric chief cells. Insufficient p57 expression directly activates chief cell plasticity, driving the acquisition of malignant phenotypes and resulting in aberrantly enhanced proliferative activity. Conversely, restoring p57 expression maintains the long-term “reserve stem cell (RSC)” state in chief cells[7,8]. It is noteworthy that chief cells, as a known source of SPEM cells, undergo transformation into trefoil factor 2 (TFF2)- and mucin 6 (MUC6)-expressing SPEM cells a hallmark feature of this pathological process[9]. Therefore, p57 may represent a promising the
Metabolic reprogramming is a cancer hallmark in which tumor cells preferentially utilize glycolysis for energy under aerobic conditions[10]. Modulation of glycolytic reprogramming has been demonstrated to ameliorate pathological processes in multiple diseases, especially tumors[11,12]. However, recent studies reveal that in gastric precancerous lesion (GPL), gastric mucosal cells remodel glucose metabolism patterns enhanced glycolysis to sustain aberrant proliferation and malignant progression, whereas regulating glycolytic pathways effectively delays precancerous lesion pro
Celastrus orbiculatus Thunb., a woody vine from the Celastraceae family, has been demonstrated in modern pharmacological studies to significantly inhibit the proliferation, invasion, and metastasis of GC cells through its ethyl acetate extract (COE)[16,17]. Furthermore, COE effectively alleviates inflammatory and metaplastic conditions in GPL by mo
This study explored the therapeutic efficacy of pristimerin in SPEM and its underlying mechanisms. The results demonstrated that pristimerin significantly alleviated gastric mucosal pathological damage and oxyntic atrophy in high-dose tamoxifen (HDT)-induced SPEM mice. More importantly, this study provides the first mechanistic evidence that pristimerin ameliorates SPEM by modulating Cdkn1c (p57)-mediated glycolytic reprogramming. These findings not only provide novel mechanistic insights into pristimerin treatment for SPEM but also establish p57 as a promising therapeutic target for this condition.
Pristimerin (B20098; purity: 99.3%) was obtained from Shanghai Yuanye Biotechnology Co., Ltd. (China). Hematoxylin (G1004; Servicebio, China) and eosin (71014544; Sinopharm, China) were used for histological staining, supplemented with an alcian blue/periodic acid schiff (AB/PAS) kit (60534ES50; Yeasen, China). Antibodies targeting MUC6 (ABIN6263435; Antibodies Online, Germany) and pyruvate kinase M2 (PKM2) (ABIN744773; Antibodies Online, Ger
Following a 7-day acclimation period, thirty female C57BL/6J mice (6-week-old, specific pathogen free, body weight range 16.0-18.0 g) were divided into five weight-matched groups (n = 6 per group): Control, model, and three pristimerin treatment groups. Mice were obtained from Jiangsu GemPharmatech Co., Ltd. and maintained at 24 °C on a 12-hour light/dark cycle, and provided free access to food and water. This study was conducted in accordance with the approved animal ethics protocol. The sample size (n = 6 per group) was determined to balance statistical requirements with the “Reduction” principle of animal ethics, and this protocol was approved by the Institutional Animal Care and Use Com
Mice in the pristimerin-treated (Pri) groups received daily intraperitoneal injections of 0.05 mg/kg/day [low-dose pristimerin (Pri-L)], 0.1 mg/kg/day [medium-dose pristimerin (Pri-M)], or 0.2 mg/kg/day [high-dose pristimerin (Pri-H)] pristimerin for 21 consecutive days, while those in the model and control groups were administered an equivalent volume of saline. Following an 8-day intervention period, the pristimerin and model groups underwent 3-days of HDT administration (250 mg/kg, intraperitoneal injection) dissolved in a vehicle of 10% ethanol and 90% sunflower oil. Control animals received only the vehicle solution. Body weight measurements were systematically performed at 7-day intervals throughout the study period. Upon completion of the treatment regimen, mice were humanely euthanized under terminal anesthesia to harvest gastric tissues.
All procedures were carried out in a specific pathogen free facility. No specific measures, such as randomization of procedural order or counterbalancing of cage positions, were taken to control for these potential confounders. This study implemented full blinding. The personnel responsible for group allocation did not participate in subsequent experimental procedures. By concealing the drug administration process, all researchers remained unaware of the group assignments. The allocation list was unblinded only after the final data analysis was completed.
Gastric corpus specimens were longitudinally incised, immersion-fixed in 10% neutral buffered formalin, and then processed via graded ethanol-xylene dehydration, paraffin embedding, and microtomy (3-4 μm). Dewaxed sections underwent histochemical staining with hematoxylin and eosin (HE) or AB/PAS. Blinded evaluation (Nikon Eclipse E100) assessed mucosal injury, glandular atrophy, and metaplasia. ImageJ was used to analyze histopathological alterations in HE-stained sections (nuclei: Blue/purple; cytoplasm: Pink) and to quantify mucin-positive areas (acidic: Blue; neutral: Magenta) in AB/PAS-stained tissues.
GES-1 cells were routinely propagated in RPMI-1640 supplemented with heat-inactivated fetal bovine serum (FBS) and antibiotic-antimycotic solution (penicillin 100 U/mL, streptomycin 100 μg/mL). To induce SPEM-like phenotypes, cells underwent serum starvation in medium containing 0.1% FBS for 24 hours, followed by incubation with 10 μmol/L TAM in complete medium for an additional 24 hours. For the therapeutic intervention, SPEM cells were exposed to different concentrations of pristimerin or the glycolysis inhibitor 2-deoxy-D-glucose (2-DG) (5 mmol/L) for 24 hours, and controls received equivalent volumes of dimethyl sulfoxide (DMSO).
Murine gastric organoids were constructed as described by Shibata et al[24]. Following microdissection into 1 mm2 fragments, gastric corpus tissues underwent sequential processing: Three ice-cold phosphate-buffered saline (PBS) washes, followed by 3-hour incubation in 10 mmol/L ethylenediaminetetraacetic acid-supplemented PBS under chilled conditions (4 °C). After centrifugation, pellets were mechanically dissociated under microscopy to release gastric glands. The glands were then filtered, centrifuged, and embedded in Matrigel. Organoids were cultured in advanced Dulbecco’s modified eagle’s medium (DMEM)/F12 medium supplemented with B27, N2, N-acetylcysteine, gastrin, epidermal growth factor, R-spondin1, noggin, fibroblast growth factor 10, Wnt3A, and penicillin/streptomycin (37 °C, 5% carbon dioxide, medium refreshed every 48 hours). Organoids > 400 μm were passaged, and third-passage organoids were used. For SPEM induction, organoids were co-cultured with H. pylori (multiplicity of infection: 2 × 105) in 0.02 μg/mL MNNG-containing medium for 24 hours. Following Cdkn1c overexpression/knockdown, gastric organoids were treated with 0.2 μmol/L pristimerin for 24 hours.
The coding sequence of the Cdkn1c gene was cloned into the pcDNA3.1 vector via double digestion with HindIII/EcoRI to construct the overexpression plasmid. SPEM-derived organoids were dissociated into single-cell suspensions by enzymatic digestion and filtration. LipofectamineTM 3000 mediated transfection of Cdkn1c-overexpressing plasmid or empty vector constructs into cells, followed by selection with 500 μg/mL G418 to establish stable cell lines. Transfected cells were re-embedded in Matrigel for three-dimensional (3D) expansion under standard organoid culture conditions.
Negative control small interfering RNA (siRNA) and Cdkn1c-targeting siRNA (sequences listed in Supplementary Table
Following dewaxing, antigen retrieval was performed in citrate buffer. Subsequently, quenching with 3% hydrogen peroxide and blocking with 10% goat serum were performed. Primary antibodies were applied overnight at 4 °C, with su
Following antigen retrieval, tissue sections underwent blocking with 5% FBS prior to overnight exposure to primary antibodies at 4 °C. After three PBS washes, secondary antibody incubation with fluorophore conjugates was performed for 1 hour. Nuclear counterstaining was achieved using DAPI. Fluorescence was visualized using an Olympus BX53 mic
Proteins extracted with radio immunoprecipitation assay buffer (protease/phosphatase inhibitors) were quantified by the bicinchoninic acid assay. Protein electrophoresis utilized sodium dodecyl sulfate-polyacrylamide gel electrophoresis, followed by polyvinylidene difluoride membrane electroblotting. Post-blocking (5% bovine serum albumin), membranes underwent primary antibody incubation, rigorous tris buffered saline tween washes, and HRP-secondary antibody probing. Immunoreactivity was visualized via electrochemiluminescence reagent exposure and signal capture. Target protein expression was determined by ImageJ analysis, normalized to β-actin, and presented as fold changes vs controls.
Complementary DNA synthesis from total RNA employed Moloney murine leukemia virus reverse transcriptase. Polymerase chain reaction amplification was performed with primers listed in Supplementary Table 2 (5-6 replicates/group). Products were electrophoresed on 1% agarose gels and analyzed by GelStain imaging. Relative gene expression was calculated using the 2-ΔΔCT method, normalized to a housekeeping gene, and expressed as fold change relative to the control group.
After treatment with pristimerin or 2-DG, the culture medium in the plates was aspirated, followed by the addition of 20 μL MTT reagent to each well and incubation for 4 hours. Subsequently, 150 μL DMSO was added, and the plates were gently shaken in the dark for 15 minutes. Cell viability was assessed by 490-nm optical density measurements using a microplate reader.
Cells (5 × 10³/well, 24-well plate) were treated with 10 μmol/L EdU for 4 hours, followed by sequential fixation (4% paraformaldehyde solution, 15 minutes) and permeabilization (0.5% Triton X-100 solution, 20 minutes) at room temperature. Subsequently, EdU was labeled using the click reaction mixture under light-protected conditions at room temperature for 30 minutes, and nuclei were counterstained with DAPI (1 μg/mL) for 10 minutes. Final images were acquired using a Nikon Eclipse Ti2 inverted fluorescence microscope.
Following dual fixation (2.5% glutaraldehyde and 2% osmium tetroxide), gastric specimens underwent ethanol gradient dehydration and epoxy resin embedding. Uranyl acetate and lead citrate were applied to counterstain ultrathin sections. Cellular ultrastructure was imaged using a transmission electron microscope (Hitachi HT7800).
Lactate and pyruvate concentrations in the cell culture medium were determined with their respective assay kits, in strict adherence to the manufacturer’s protocols. Briefly, the conditioned medium was collected and centrifuged to remove cellular debris, and the clarified supernatant was taken for detection. For lactate quantification, the enzyme working solution and chromogenic agent were combined with 20 μL of the supernatant. After incubation at 37 °C for 10 minutes, the stop solution was added, and the absorbance was measured at a wavelength of 530 nm. For pyruvate quantification, 0.1 mL of supernatant was subjected to a 10-minute, 37 °C incubation with the chromogenic agent. Subsequently, following the addition of alkaline solution, the reaction mixture was kept at room temperature for 5 minutes before the final absorbance measurement at 505 nm. Standard curves were generated using serial concentrations of standards, and the metabolite concentrations were calculated accordingly.
Mitochondrial respiration was assessed using the Seahorse XF Cell Mito Stress Test Kit. In brief, cells were seeded in XF24 plates at a density of 7 × 104 cells/well and cultured overnight. On the day of assay, cells were equilibrated in XF DMEM assay medium (supplemented with 1 mmol/L pyruvate, 2 mmol/L glutamine, and 10 mmol/L glucose, potential of hydrogen = 7.4) for 60 minutes in a non-carbon dioxide incubator. During the assay, oligomycin (15 μmol), trifluoromethoxy carbonylcyanide phenylhydrazone (2 μmol/L), and rotenone/antimycin A (5 μmol/L) were sequentially injected. Data were normalized to cell number and analyzed using Wave software (Agilent, United States). Glycolytic function was evaluated using the Seahorse XF glycolysis stress test kit. Cells were plated as described above and incubated in XF DMEM medium containing 2 mmol/L glutamine. After equilibration, glucose (100 mmol/L), oligomycin (10 μmol/L), and 2-DG (500 mmol/L) were injected sequentially. All measurements were performed on a Seahorse XFe24 Analyzer (Agilent, United States), and data were processed using the associated glycolysis report generator.
Statistical analysis was conducted on data from a minimum of five independent biological replicates using GraphPad Prism 9.5, with all results expressed as mean ± SD and significance defined as P ≤ 0.05. Following verification of para
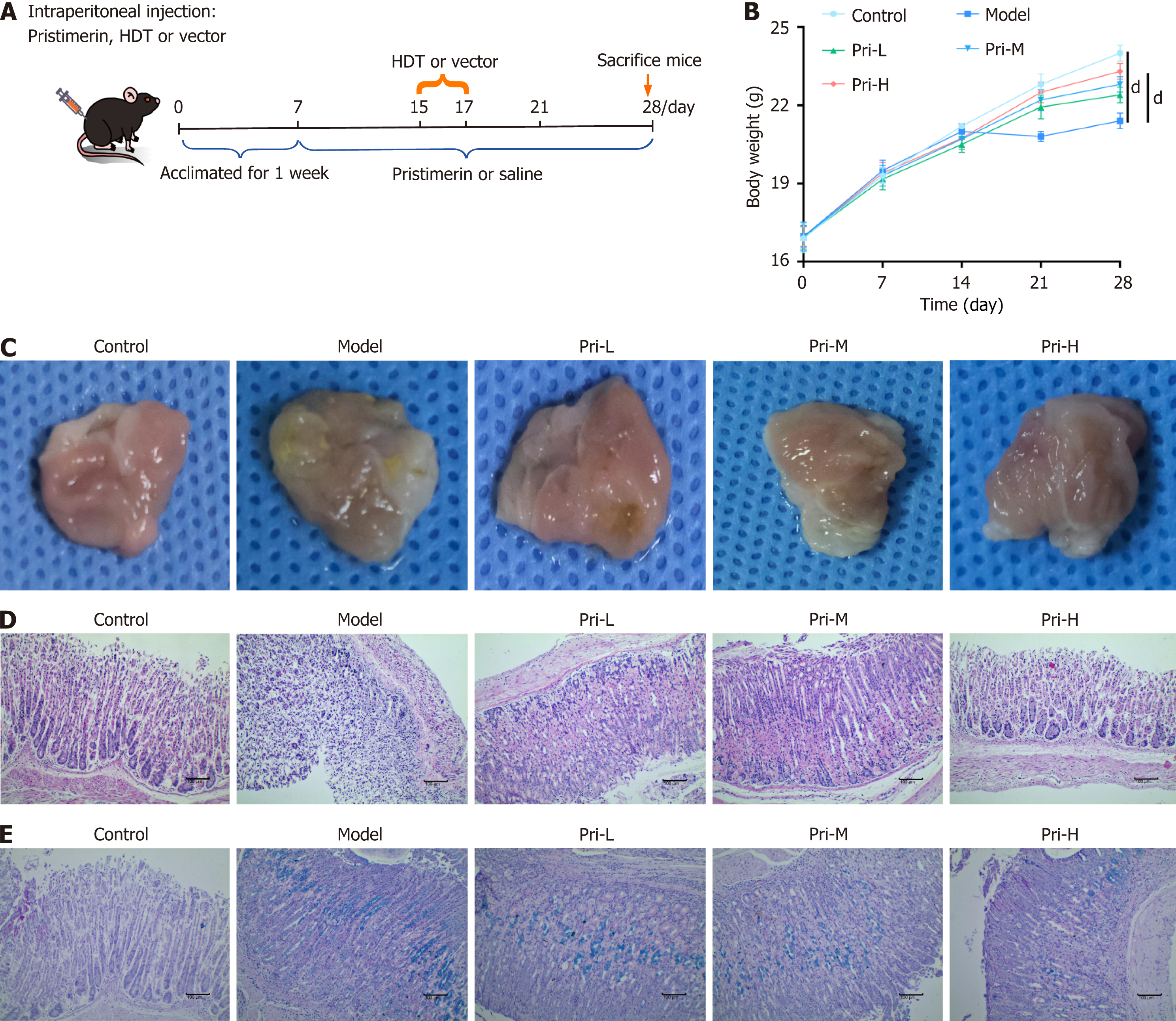
A murine SPEM model was induced by HDT to assess the therapeutic potential of pristimerin[25]. The treatment groups received pristimerin at varying doses via intraperitoneal injection (Figure 1A). During the intervention period, HDT induction significantly attenuated body weight gain in mice, whereas pristimerin treatment effectively maintained growth trajectory (Figure 1B). Macroscopic evaluation revealed that gastric mucosa in the model group exhibited pallor/duskiness, increased firmness, rugae flattening or disappearance, accompanied by focal nodular/ulcer-like lesions. Conversely, pristimerin substantially ameliorated these pathological phenotypes (Figure 1C). Histopathological analysis of the gastric mucosa in model mice revealed severely disrupted glandular architecture, a disorganized cellular arr
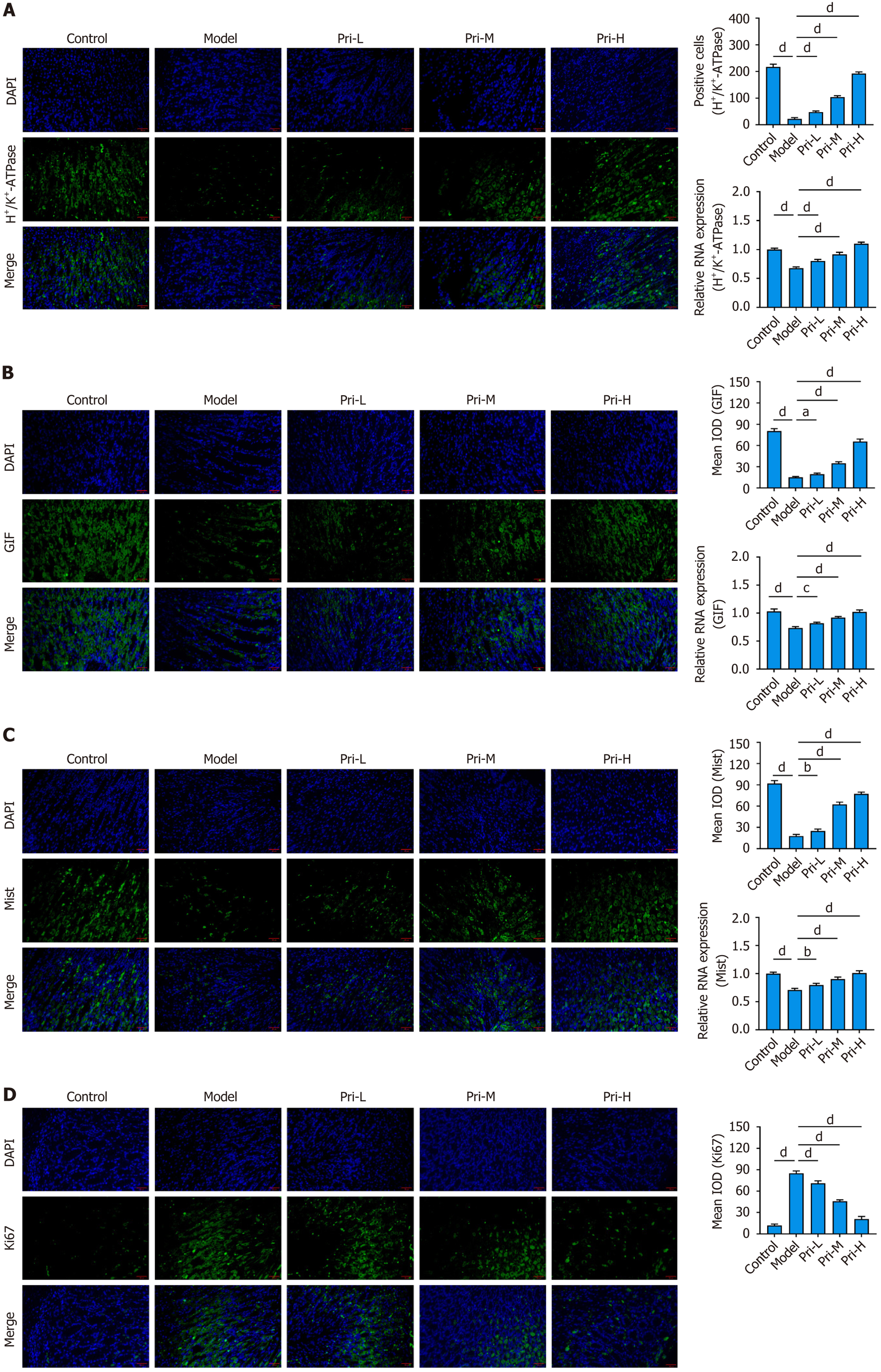
Oxyntic atrophy, a landmark pathological alteration in SPEM, is characterized by progressive depletion and functional impairment of chief cells (enzyme-producing) and parietal cells (acid-secreting)[26]. To elucidate the impact of pri
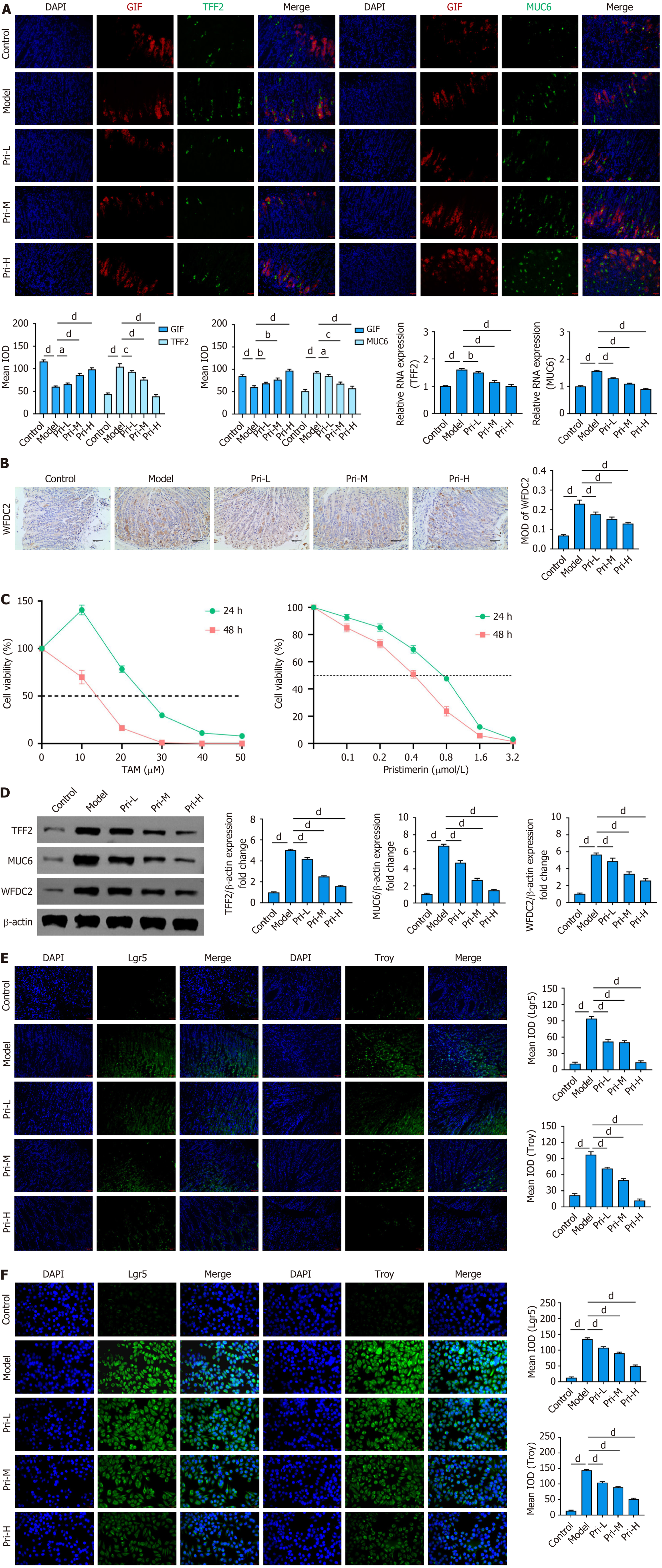
SPEM is a metaplastic lesion arising during gastric mucosal carcinogenesis, characterized by chief cell trans differentiation into TFF2/MUC6-aberrant metaplastic cells localized at gastric corpus gland bases[9,27]. WFDC2 is another imp
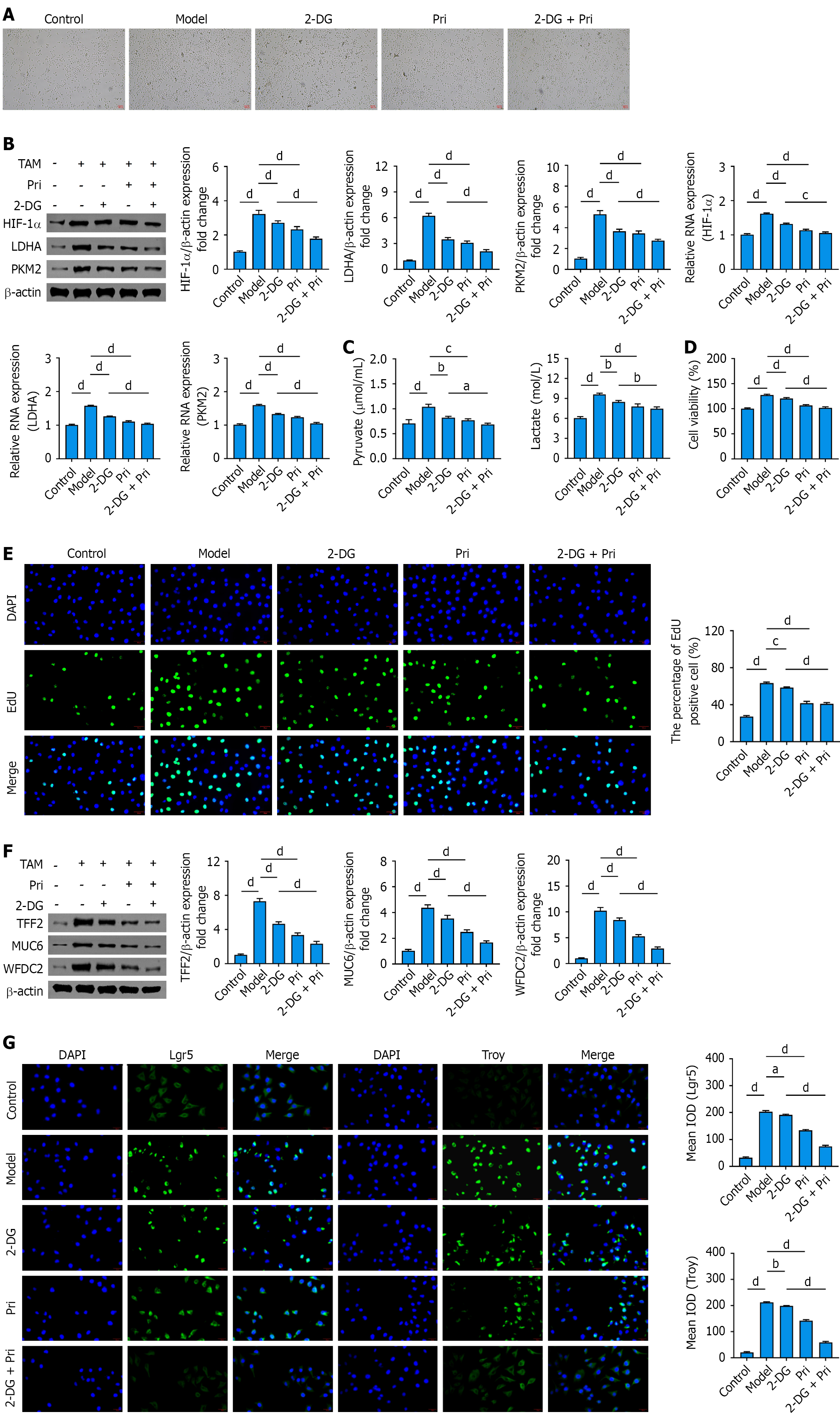
To validate this mechanism in vitro, GES-1 cells were treated with varying concentrations of TAM, and the half-maximal inhibitory concentration (IC50) was determined. The results showed IC50 values of 25.90 μmol/L and 12.57 μmol/L after 24 hours and 48 hours of treatment, respectively. Notably, treatment with 10 μmol/L TAM for 24 hours significantly enhanced cell viability (Figure 3C), a phenotype consistent with the aberrant proliferative characteristics of SPEM[26]. The significantly upregulated expression of SPEM characteristic marker genes (TFF2, MUC6, and WFDC2; Supplementary Figure 2A) further confirmed the reliability of the model. In addition, cytotoxicity assessment of pristimerin revealed IC50 values of 0.65 μmol/L and 0.38 μmol/L after 24 hours and 48 hours of treatment in GES-1 cells, respectively. Following 24 hours of intervention, cell viability remained above 60% when the concentration was below 0.4 μmol/L (Figure 3C). Therefore, 0.1 μmol/L (Pri-L), 0.2 μmol/L (Pri-M), and 0.4 μmol/L (Pri-H) were selected as the low-, medium-, and high-intervention concentrations for subsequent experiments. Western blotting revealed significantly elevated expression of TFF2, MUC6, and WFDC2 in TAM-induced GES-1 cells; pristimerin treatment suppressed this effect. In vitro experiments further validated pristimerin’s regulatory capacity on these markers (Figure 3D).
Lgr5 and Troy, recognized as stem cell markers, are broadly expressed in the gastric pylorus and fundus, predominantly localized to the basal and isthmus regions of the gastric glands. Studies have demonstrated that aberrant overexpression of these two markers in gastric body glands strongly correlates with gastric mucosal malignant progression[29]. Therefore, we further analyzed the expression profiles of Lgr5 and Troy. Significant upregulation of both markers was observed in gastric body tissues of SPEM mouse models. Notably, pristimerin treatment significantly suppressed their expression (Figure 3E). Consistent results were obtained in vitro (Figure 3F). Collectively, these findings establish pri
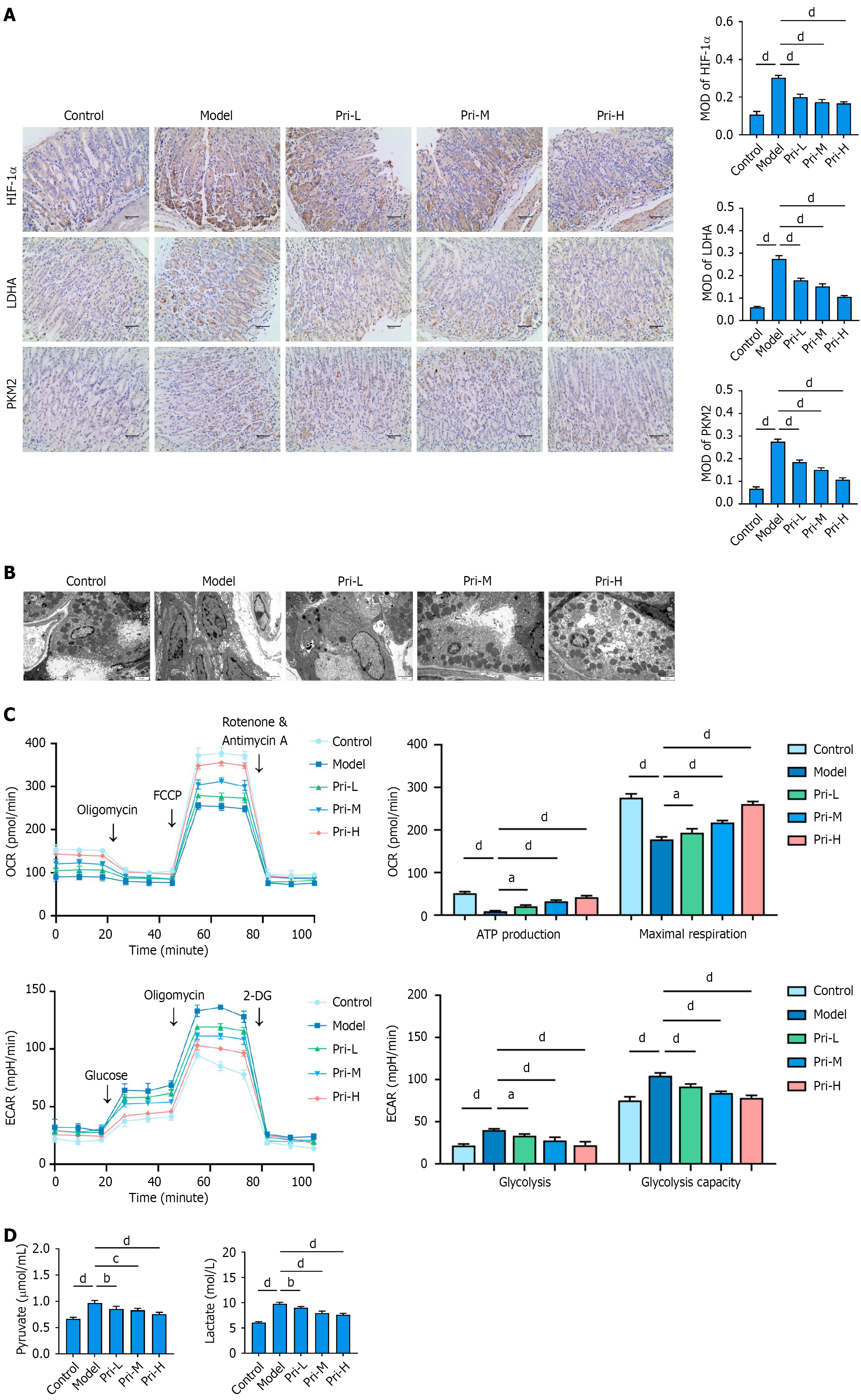
Glycolytic reprogramming is the key metabolic basis promoting the malignant transformation of gastric mucosal cells and disease progression. Analysis of key glycolytic regulators (HIF-1α, LDHA, and PKM2) demonstrated a notable elevation of their expression in the HDT-induced mice. Pristimerin treatment suppressed the expression of these factors (Figure 4A). Ultrastructural observation revealed that gastric mucosal tissue cells in SPEM model mice exhibited dilated endoplasmic reticulum, swollen mitochondria, accompanied by a significant decrease in the number of secretory granules and autophagic vesicles. In contrast, the Pri group demonstrated a denser endoplasmic reticulum structure, well-pre
In vitro, treatment of TAM-induced GES-1 cells with varying concentrations of pristimerin, followed by assessment using the Seahorse XF Metabolic Analyzer, revealed that TAM induction significantly enhanced the glycolytic rate and glycolytic capacity of GES-1 cells, while markedly reducing both the basal oxygen consumption rate (OCR) and maximal OCR. Pristimerin treatment rescued this metabolic imbalance (Figure 4C). Metabolite analysis further demonstrated that pristimerin effectively suppressed the TAM-induced elevation in pyruvate and lactate levels (Figure 4D). Collectively, these results confirm that gastric mucosal cells in SPEM lesions exhibit a metabolic disorder characterized by aberrantly enhanced glycolysis and suppressed oxidative phosphorylation. Pristimerin restores metabolic homeostasis by rep
To further investigate the impact of glycolytic reprogramming on SPEM and its role in pristimerin-mediated amelioration of SPEM pathology, we treated TAM-induced GES-1 cells with medium-concentration pristimerin or 2-DG (Figure 5A). The results revealed that 24-hour treatment with 10 μmol/L TAM induced abnormally increased cell density, dis
Further investigation revealed that 2-DG and pristimerin treatment differentially suppressed the TAM-induced pathological enhancement of cell viability and proliferative activity in GES-1 cells (Figure 5D and E). More importantly, both agents significantly suppressed the aberrant upregulation of SPEM-specific markers (TFF2, MUC6, WFDC2) and stem cell markers (Lgr5, Troy). Furthermore, the combined treatment of pristimerin and 2-DG exerted a markedly str
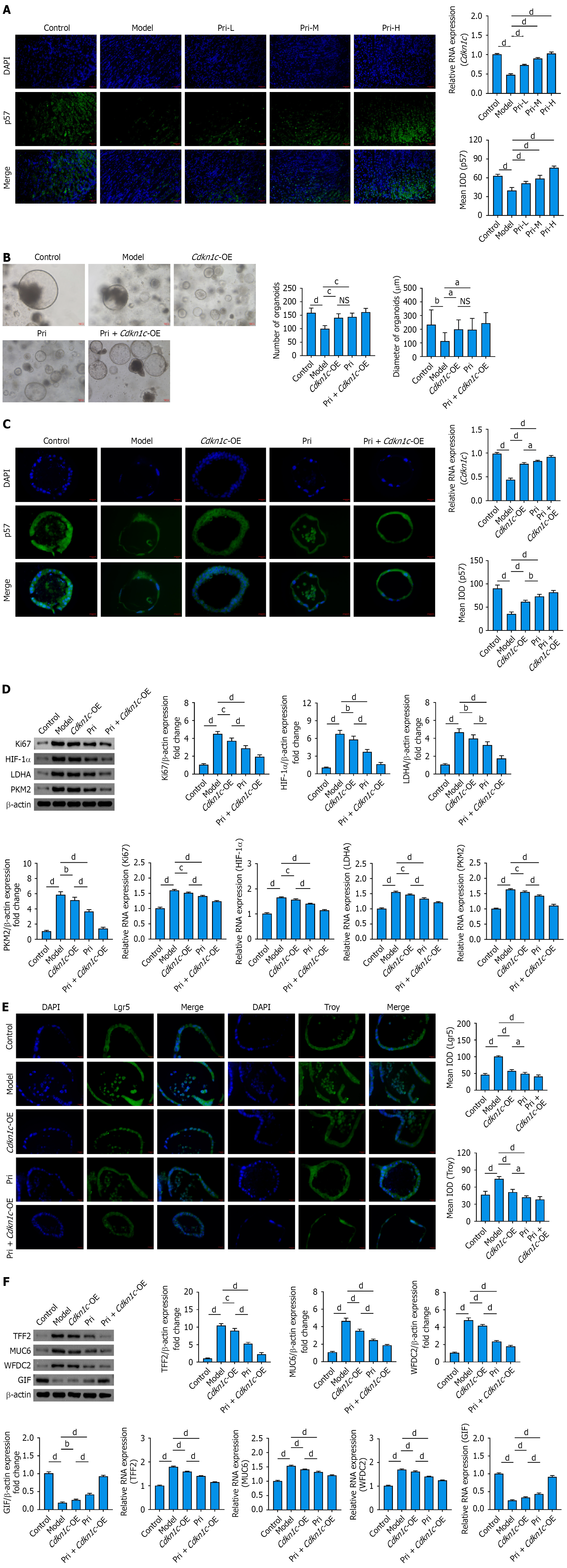
To investigate pristimerin’s regulation of p57, we assessed its expression in mouse gastric tissues via immunofluorescence. HDT-induced SPEM mice exhibited dramatically reduced p57 expression, while pristimerin treatment sig
Post-intervention analysis revealed that MNNG/H. pylori treatment significantly reduced both the number and diameter of organoids, whereas both Cdkn1c overexpression and pristimerin intervention effectively ameliorated this phenotype (Figure 6B). Immunofluorescence confirmed that both plasmid transfection and pristimerin treatment upregulated p57 expression in gastric organoids (Figure 6C) while concurrently suppressing Ki67, key glycolytic regulators (HIF-1α/PKM2/LDHA; Figure 6D), stem cell markers (Lgr5/Troy; Figure 6E), and SPEM-specific markers (TFF2/MUC6/WFDC2; Figure 6F). These results demonstrate that downregulated p57 expression in SPEM lesions is closely associated with aberrant glycolytic reprogramming and activation of malignant phenotypes, and that restoration of p57 expression effectively suppresses this pathological mechanism, while pristimerin inhibits glycolytic pathways and SPEM phenotypes by upregulating p57.
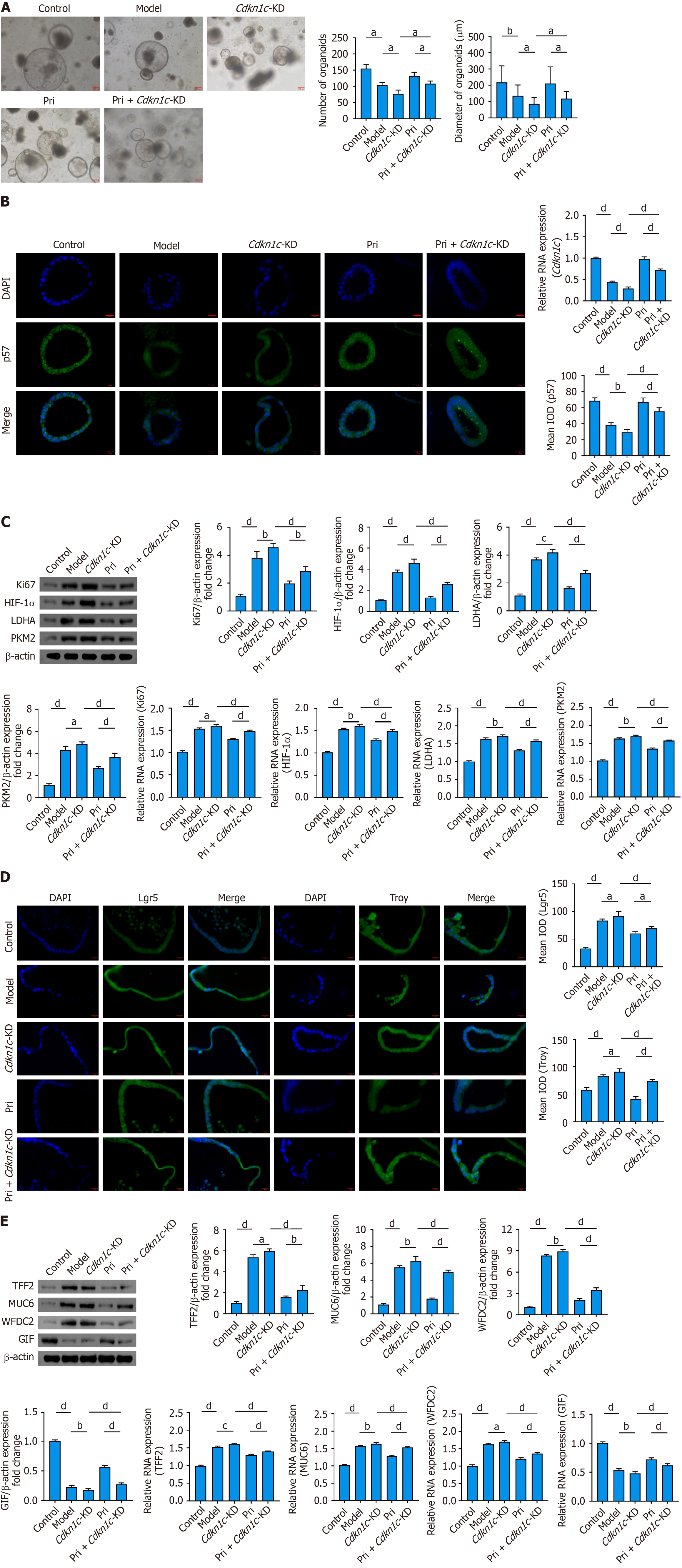
To validate the regulatory mechanism by which pristimerin targets p57, we constructed a p57-knockdown model
According to the Correa cascade hypothesis, the normal gastric mucosa must undergo a series of pathological stages to develop into GC, primarily including CAG, intestinal metaplasia (IM), and dysplasia. Among these stages, the transition into metaplasia represents the pivotal point at which the gastric mucosa acquires malignant transformation potential[30]. Recent studies have found that SPEM is not only an important precursor lesion of IM, but also a key link in the malignant progression of gastric mucosa, and its persistent existence will significantly increase the risk of mucosal carcinogenesis[31,32]. Furthermore, clinicopathological analysis further shows that SPEM cells are found in 90% of resected GC tissues and exhibit high microsatellite instability and genome-wide genetic instability[33]. This further demonstrates the central role of SPEM in the development process of GC. Therefore, early intervention targeting SPEM is of great significance for blocking the malignant progression of gastric mucosa.
Recent studies on Traditional Chinese medicine compound formulas in SPEM treatment mechanisms have achieved preliminary progress. For instance, Weiwei decoction can improve SPEM in both H. pylori-infected mice and Atp4a gene knockout mice by inhibiting intestinal-type gastric adenocarcinoma-associated transcripts, restoring the expression of Atp4a and pepsinogen II, and reducing M2 macrophage infiltration[34]. Similarly, Zuojin capsule alleviates TAM-induced SPEM by modulating cyclins and the phosphatidylinositol 3-kinase/protein kinase B signaling axis[26]. It is noteworthy that, compared to complex herbal preparations, their bioactive compounds generally exhibit superior cell membrane per
In this study, utilizing an HDT-induced SPEM mouse model, we demonstrated that pristimerin significantly alleviates gastric mucosal pathological damage, including improvement of oxyntic atrophy and suppression of metaplastic lesions. This robustly confirms pristimerin’s therapeutic potential in reversing SPEM. At the molecular level, pristimerin effectively suppressed SPEM characteristics in gastric mucosal cells by downregulating specific markers TFF2 and MUC6. WFDC2 a small secretory protein overexpressed in numerous cancers and correlated with adverse prognosis[36,37] was recently shown to promote SPEM progression via interleukin-33 upregulation[27]. Our findings reveal that pristimerin significantly downregulates WFDC2 in gastric tissues of SPEM mice. Furthermore, we observed aberrantly elevated expression of the stem cell markers Lgr5 and Troy in the gastric corpus of SPEM mice. As reported, Lgr5+/Troy+ stem cells serve as critical origins of GC cells, where their dysregulated overexpression drives tumorigenesis, progression, and metastasis[29,38,39]. Consequently, the pathological upregulation of Lgr5 and Troy in SPEM mice may suggest that these tissues have already acquired features predisposing them to malignant transformation. Importantly, pristimerin tre
Mechanistically, our data demonstrate that the ameliorative effect of pristimerin on SPEM is mediated through its modulation of glycolytic reprogramming. Glycolytic reprogramming is an adaptive change in energy metabolism for cells coping with pathological microenvironments, by regulating gene expression, enzyme activity, and signaling pathways to satisfy demands for proliferation, stress response, or differentiation[40]. This phenomenon is extensively documented in tumors, where its functional scope extends beyond rapid energy provision. Studies demonstrate that tumor cells competitively deplete nutrients to impair immunometabolic fitness, thereby suppressing antitumor immunity[41,42]. Critical rate-limiting enzymes (e.g., HIF-1α, PKM2, LDHA) and metabolites (e.g., lactate) within this metabolic cascade directly drive malignant progression[43-46]. Recent studies have shown that gastric mucosal cells in GPL enhance glycolysis to adapt to the chronic inflammatory and hypoxic microenvironment, whereas inhibition of glycolysis reverses GPL pro
Further investigation identified Cdkn1c/p57, a member of the CIP/KIP family, as the key molecular target through which pristimerin regulates glycolytic reprogramming and ameliorates SPEM phenotypes. Previous studies have est
Finally, it is imperative to conscientiously acknowledge the limitations of this study. First, as a preclinical investigation, the findings are entirely derived from animal, cell, and organoid models, and lack human pharmacokinetic, bioavailability, and toxicity data, which poses challenges for clinical translation. Second, the experimental sample size was relatively small, and the in vivo studies did not include mechanistically well-defined positive control groups. Third, real-time metabolic analysis of glycolytic reprogramming was lacking. Fourth, the upstream regulatory mechanisms of pri
In summary, this study provides the first demonstration of pristimerin’s significant ameliorative effect on SPEM and elucidates its core mechanism in ameliorating disease progression through regulation of Cdkn1c (p57)-mediated glycolytic reprogramming (Supplementary Figure 4). This finding not only establishes p57 as a critical target for pristimerin-mediated amelioration of SPEM, but also provides an important theoretical foundation for further exploration of the molecular mechanisms in gastric mucosal lesions and the development of therapeutic strategies targeting the glycolytic pathway.
The authors sincerely thank the laboratory colleagues for their excellent technical assistance.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14951] [Article Influence: 7475.5] [Reference Citation Analysis (22)] |
| 2. | Li ML, Hong XX, Zhang WJ, Liang YZ, Cai TT, Xu YF, Pan HF, Kang JY, Guo SJ, Li HW. Helicobacter pylori plays a key role in gastric adenocarcinoma induced by spasmolytic polypeptide-expressing metaplasia. World J Clin Cases. 2023;11:3714-3724. [PubMed] [DOI] [Full Text] |
| 3. | Srivastava S, Huang KK, Rebbani K, Das K, Fazreen Z, Yeoh KG, Tan P, Teh M. An LCM-based genomic analysis of SPEM, Gastric Cancer and Pyloric Gland Adenoma in an Asian cohort. Mod Pathol. 2020;33:2075-2086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 4. | Zhang G, Zhang X, Pan W, Chen X, Wan L, Liu C, Yong Y, Zhao Y, Sang S, Zhang L, Yao S, Guo Y, Wang M, Wang X, Peng G, Yan X, Wang Y, Zhang M. Dissecting the Spatial and Single-Cell Transcriptomic Architecture of Cancer Stem Cell Niche Driving Tumor Progression in Gastric Cancer. Adv Sci (Weinh). 2025;12:e2413019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 5. | Zhou X, Gao W, Hua H, Ji Z. LncRNA-BLACAT1 Facilitates Proliferation, Migration and Aerobic Glycolysis of Pancreatic Cancer Cells by Repressing CDKN1C via EZH2-Induced H3K27me3. Front Oncol. 2020;10:539805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (4)] |
| 6. | Kavanagh E, Joseph B. The hallmarks of CDKN1C (p57, KIP2) in cancer. Biochim Biophys Acta. 2011;1816:50-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 60] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 7. | Lee JH, Kim S, Han S, Min J, Caldwell B, Bamford AD, Rocha ASB, Park J, Lee S, Wu SS, Lee H, Fink J, Pilat-Carotta S, Kim J, Josserand M, Szep-Bakonyi R, An Y, Ju YS, Philpott A, Simons BD, Stange DE, Choi E, Koo BK, Kim JK. p57(Kip2) imposes the reserve stem cell state of gastric chief cells. Cell Stem Cell. 2022;29:826-839.e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 8. | Radyk MD, Burclaff J, Willet SG, Mills JC. Metaplastic Cells in the Stomach Arise, Independently of Stem Cells, via Dedifferentiation or Transdifferentiation of Chief Cells. Gastroenterology. 2018;154:839-843.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 9. | Teal E, Dua-Awereh M, Hirshorn ST, Zavros Y. Role of metaplasia during gastric regeneration. Am J Physiol Cell Physiol. 2020;319:C947-C954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 10. | Vargas N, Rondeau M, Fortune T, Luk T, Brandenburg LO. Tumor Acidity as a Result of the Warburg Effect in Cancer Glucose Metabolism and Its Role in Cancer Progression-A Review. Mil Med. 2025;190:e1844-e1850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 11. | Dong XM, Chen L, Xu YX, Wu P, Xie T, Liu ZQ. Exploring metabolic reprogramming in esophageal cancer: the role of key enzymes in glucose, amino acid, and nucleotide pathways and targeted therapies. Cancer Gene Ther. 2025;32:165-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 12. | Worral Wilfred Raj AS, Manoharan R. NUAKs promote mTOR/c-Myc-induced glucose and glutamine reprogramming for cell growth and metastasis in breast cancer cells. Biochim Biophys Acta Mol Basis Dis. 2025;1871:167508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 13. | Liu S, Ji H, Zhang T, Huang J, Yin X, Zhang J, Wang P, Wang F, Tang X. Modified Zuojin pill alleviates gastric precancerous lesions by inhibiting glycolysis through the HIF-1α pathway. Phytomedicine. 2025;136:156255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 14. | Zhang C, Cai T, Zeng X, Cai D, Chen Y, Huang X, Gan H, Zhuo J, Zhao Z, Pan H, Li S. Astragaloside IV reverses MNNG-induced precancerous lesions of gastric carcinoma in rats: Regulation on glycolysis through miRNA-34a/LDHA pathway. Phytother Res. 2018;32:1364-1372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (3)] |
| 15. | Zhang JX, Bao SC, Chen J, Chen T, Wei HL, Zhou XY, Li JT, Yan SG. Xiaojianzhong decoction prevents gastric precancerous lesions in rats by inhibiting autophagy and glycolysis in gastric mucosal cells. World J Gastrointest Oncol. 2023;15:464-489. [PubMed] [DOI] [Full Text] |
| 16. | Wang H, Tao L, Ni T, Gu H, Jin F, Dai X, Feng J, Ding Y, Xiao W, Guo S, Hisamitsu T, Qian Y, Liu Y. Anticancer efficacy of the ethyl acetate extract from the traditional Chinese medicine herb Celastrus orbiculatus against human gastric cancer. J Ethnopharmacol. 2017;205:147-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 17. | Wang H, Luo Y, Ou S, Ni T, Chu Z, Feng X, Dai X, Zhang X, Liu Y. Celastrus orbiculatus Thunb. extract inhibits EMT and metastasis of gastric cancer by regulating actin cytoskeleton remodeling. J Ethnopharmacol. 2023;301:115737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Zhu FY, Zhu YD, Peng Q, Zhang XZ, Li P. [Expression and clinical significance of glycolysis-related genes HIF-1α, FOXO4 and LDHA in gastric cancer]. Linchuang Zhongliuxue Zazhi. 2023;28:798-805. [DOI] [Full Text] |
| 19. | Zhang XZ, Zhu FY, Wen JS, Liu YQ, Zhu YD. [Mechanism of Reversal of Precancerous Lesions of Gastric Cancer by Celastrus orbiculatus Extract via Regulating Lgr5/Wnt/β-catenin Signaling Pathway]. Zhongguo Shiyan Fangjixue Zazhi. 2024;30:70-77. [DOI] [Full Text] |
| 20. | Zhu YD, Hu L, Li P, Zhang M, Liu YQ. Effects of Celastrus orbiculatus on Epithelial Mesenchymal Transition in Gastric Mucosal Epithelial Cells by Inhibiting Lgr5 Expression from Rats with Gastric Precancerous Lesions. Am J Chin Med. 2018;46:1129-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 21. | Wang Y, Feng W, Wang X, Li X, Mou Y, Wang X, Zhang Y. The multifaceted mechanisms of pristimerin in the treatment of tumors state-of-the-art. Biomed Pharmacother. 2022;154:113575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 22. | Tang Y, Chen J, Li J, Zheng Y, Zhong X, Huang S, Chen B, Peng B, Zou X, Chen X. Pristimerin synergistically sensitizes conditionally reprogrammed patient derived-primary hepatocellular carcinoma cells to sorafenib through endoplasmic reticulum stress and ROS generation by modulating Akt/FoxO1/p27(kip1) signaling pathway. Phytomedicine. 2021;86:153563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 23. | Wu H, Li L, Ai Z, Yin J, Chen L. Pristimerin induces apoptosis of oral squamous cell carcinoma cells via G(1) phase arrest and MAPK/Erk1/2 and Akt signaling inhibition. Oncol Lett. 2019;17:3017-3025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Shibata W, Sue S, Tsumura S, Ishii Y, Sato T, Kameta E, Sugimori M, Yamada H, Kaneko H, Sasaki T, Ishii T, Tamura T, Kondo M, Maeda S. Helicobacter-induced gastric inflammation alters the properties of gastric tissue stem/progenitor cells. BMC Gastroenterol. 2017;17:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (4)] |
| 25. | Huh WJ, Khurana SS, Geahlen JH, Kohli K, Waller RA, Mills JC. Tamoxifen induces rapid, reversible atrophy, and metaplasia in mouse stomach. Gastroenterology. 2012;142:21-24.e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 206] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 26. | Xiong M, Chen X, Wang H, Tang X, Wang Q, Li X, Ma H, Ye X. Combining transcriptomics and network pharmacology to reveal the mechanism of Zuojin capsule improving spasmolytic polypeptide-expressing metaplasia. J Ethnopharmacol. 2024;318:117075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 27. | Ye Q, Zhu Y, Ma Y, Wang Z, Xu G. Emerging role of spasmolytic polypeptide-expressing metaplasia in gastric cancer. J Gastrointest Oncol. 2024;15:2673-2683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 28. | Jeong H, Lee B, Kim KH, Cho SY, Cho Y, Park J, Lee Y, Oh Y, Hwang BR, Jang AR, Park JH, Park JH, Jeong SH, Lee D, Lee YC, Lim KM, Goldenring JR, Nam KT. WFDC2 Promotes Spasmolytic Polypeptide-Expressing Metaplasia Through the Up-Regulation of IL33 in Response to Injury. Gastroenterology. 2021;161:953-967.e15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 29. | Wuputra K, Ku CC, Pan JB, Liu CJ, Liu YC, Saito S, Kato K, Lin YC, Kuo KK, Chan TF, Chong IW, Lin CS, Wu DC, Yokoyama KK. Stem Cell Biomarkers and Tumorigenesis in Gastric Cancer. J Pers Med. 2022;12:929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 30. | Liao W, Wang J, Li Y. Natural products based on Correa's cascade for the treatment of gastric cancer trilogy: Current status and future perspective. J Pharm Anal. 2025;15:101075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 31. | Lee SH, Jang B, Min J, Contreras-Panta EW, Presentation KS, Delgado AG, Piazuelo MB, Choi E, Goldenring JR. Up-regulation of Aquaporin 5 Defines Spasmolytic Polypeptide-Expressing Metaplasia and Progression to Incomplete Intestinal Metaplasia. Cell Mol Gastroenterol Hepatol. 2022;13:199-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 32. | Chong Y, Yu D, Lu Z, Nie F. Role and research progress of spasmolytic polypeptide‑expressing metaplasia in gastric cancer (Review). Int J Oncol. 2024;64:33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 33. | Chen J, Zhu C, Wang C, Hu C, Czajkowsky DM, Guo Y, Liu B, Shao Z. Evidence for heightened genetic instability in precancerous spasmolytic polypeptide expressing gastric glands. J Med Genet. 2020;57:385-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 34. | Hong X, Li H, Lin Y, Luo L, Xu W, Kang J, Li J, Huang B, Xu Y, Pan H, Guo S. Efficacy and potential therapeutic mechanism of Weiwei decoction on Spasmolytic polypeptide-expressing metaplasia in Helicobacter pylori-infected and Atp4a-knockout mice. J Ethnopharmacol. 2024;319:117062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 35. | Zhang X, Qiu H, Li C, Cai P, Qi F. The positive role of traditional Chinese medicine as an adjunctive therapy for cancer. Biosci Trends. 2021;15:283-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 283] [Article Influence: 56.6] [Reference Citation Analysis (3)] |
| 36. | Li Y, Yu C, Li H, Feng Y, Fan P, Chen X. Relationship between human epididymal protein 4 and depth of tumor invasion, postoperative recurrence, and metastasis of epithelial epithelial ovarian cancer. Asia Pac J Clin Oncol. 2024;20:472-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 37. | Ohkuma R, Yada E, Ishikawa S, Komura D, Kubota Y, Hamada K, Horiike A, Ishiguro T, Hirasawa Y, Ariizumi H, Shida M, Watanabe M, Onoue R, Ando K, Tsurutani J, Yoshimura K, Sasada T, Aoki T, Murakami M, Norose T, Ohike N, Takimoto M, Kobayashi S, Tsunoda T, Wada S. High levels of human epididymis protein 4 mRNA and protein expression are associated with chemoresistance and a poor prognosis in pancreatic cancer. Int J Oncol. 2021;58:57-69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 38. | Fischer AS, Sigal M. The Role of Wnt and R-spondin in the Stomach During Health and Disease. Biomedicines. 2019;7:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 39. | Wilhelm F, Böger C, Krüger S, Behrens HM, Röcken C. Troy is expressed in human stomach mucosa and a novel putative prognostic marker of intestinal type gastric cancer. Oncotarget. 2017;8:50557-50569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 40. | Xu X, Peng Q, Jiang X, Tan S, Yang Y, Yang W, Han Y, Chen Y, Oyang L, Lin J, Xia L, Peng M, Wu N, Tang Y, Li J, Liao Q, Zhou Y. Metabolic reprogramming and epigenetic modifications in cancer: from the impacts and mechanisms to the treatment potential. Exp Mol Med. 2023;55:1357-1370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 176] [Article Influence: 58.7] [Reference Citation Analysis (0)] |
| 41. | Guerra L, Bonetti L, Brenner D. Metabolic Modulation of Immunity: A New Concept in Cancer Immunotherapy. Cell Rep. 2020;32:107848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 125] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 42. | Shang Z, Ma Z, Wu E, Chen X, Tuo B, Li T, Liu X. Effect of metabolic reprogramming on the immune microenvironment in gastric cancer. Biomed Pharmacother. 2024;170:116030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 43. | Feng Y, Xiong Y, Qiao T, Li X, Jia L, Han Y. Lactate dehydrogenase A: A key player in carcinogenesis and potential target in cancer therapy. Cancer Med. 2018;7:6124-6136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 565] [Cited by in RCA: 512] [Article Influence: 64.0] [Reference Citation Analysis (1)] |
| 44. | Wang JX, Choi SYC, Niu X, Kang N, Xue H, Killam J, Wang Y. Lactic Acid and an Acidic Tumor Microenvironment suppress Anticancer Immunity. Int J Mol Sci. 2020;21:8363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 321] [Article Influence: 53.5] [Reference Citation Analysis (2)] |
| 45. | Wu T, Yang Y, Zhang B, Zhang ZS, Zhou S, Jia GZ, Liu SQ, He XL, He JX, Wang N. EDDM3A drives gastric cancer progression by promoting HIF-1α-dependent aerobic glycolysis. Oncogenesis. 2022;11:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 46. | Zhu S, Guo Y, Zhang X, Liu H, Yin M, Chen X, Peng C. Pyruvate kinase M2 (PKM2) in cancer and cancer therapeutics. Cancer Lett. 2021;503:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 194] [Article Influence: 38.8] [Reference Citation Analysis (0)] |
| 47. | Chelakkot C, Chelakkot VS, Shin Y, Song K. Modulating Glycolysis to Improve Cancer Therapy. Int J Mol Sci. 2023;24:2606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 260] [Article Influence: 86.7] [Reference Citation Analysis (0)] |
| 48. | Qiu Z, Li Y, Zeng B, Guan X, Li H. Downregulated CDKN1C/p57(kip2) drives tumorigenesis and associates with poor overall survival in breast cancer. Biochem Biophys Res Commun. 2018;497:187-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 49. | Sun C, Ma P, Wang Y, Liu W, Chen Q, Pan Y, Zhao C, Qian Y, Liu J, Li W, Shu Y. KLF15 Inhibits Cell Proliferation in Gastric Cancer Cells via Up-Regulating CDKN1A/p21 and CDKN1C/p57 Expression. Dig Dis Sci. 2017;62:1518-1526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 50. | Zhao F, Yang Z, Gu X, Feng L, Xu M, Zhang X. miR-92b-3p Regulates Cell Cycle and Apoptosis by Targeting CDKN1C, Thereby Affecting the Sensitivity of Colorectal Cancer Cells to Chemotherapeutic Drugs. Cancers (Basel). 2021;13:3323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 51. | Yang C, Yan Z, Sun Z, Hu F, Xu W. FOXO3 Inhibits the Cisplatin Resistance and Progression of Melanoma Cells by Promoting CDKN1C Transcription. Appl Biochem Biotechnol. 2024;196:7834-7848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/