Copyright: ©Author(s) 2026.
World J Gastroenterol. May 7, 2026; 32(17): 117823
Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.117823
Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.117823
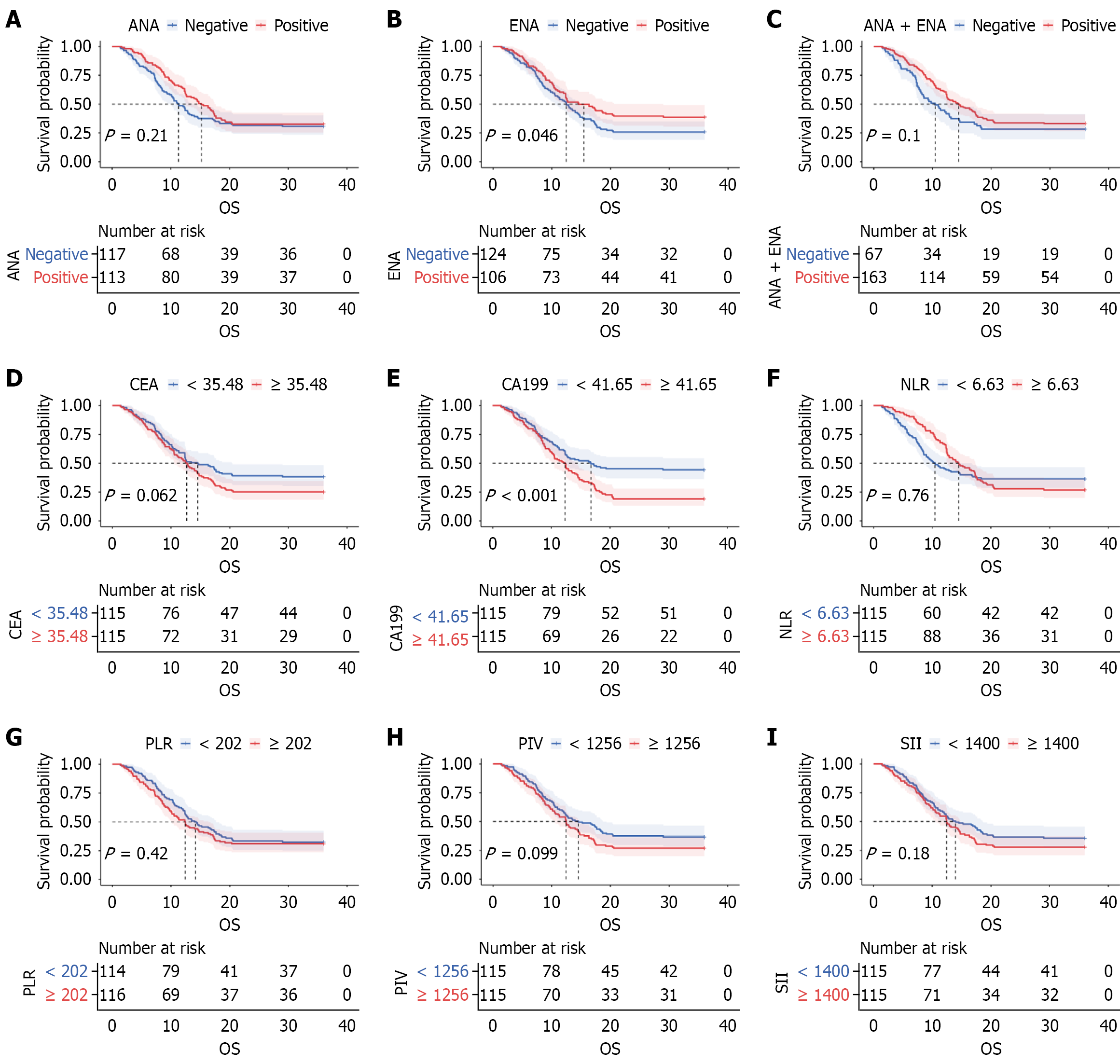
Figure 1 Kaplan-Meier analysis of overall survival according to autoantibody status and laboratory parameters.
A-H: Kaplan-Meier curves depict overall survival probabilities for patients stratified by antinuclear antibody (ANA) status (A), extractable nuclear antigen (ENA) status (B), combined ANA or ENA status (C), carcinoembryonic antigen levels using a cutoff of 35.48 (D), carbohydrate antigen 199 levels using a cutoff of 41.65 (E), neutrophil-to-lymphocyte ratio using a cutoff of 6.63 (F), platelet-to-lymphocyte ratio using a cutoff of 202 (G), pan-immune-inflammation value using a cutoff of 1256 (H); I: Systemic immune-inflammation index using a cutoff of 1400. ANA: Antinuclear antibody; ENA: Extractable nuclear antigen; OS: Overall survival; CEA: Carcinoembryonic antigen; CA199: Carbohydrate antigen 199; NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; PIV: Pan-immune-inflammation value; SII: Systemic immune-inflammation index.
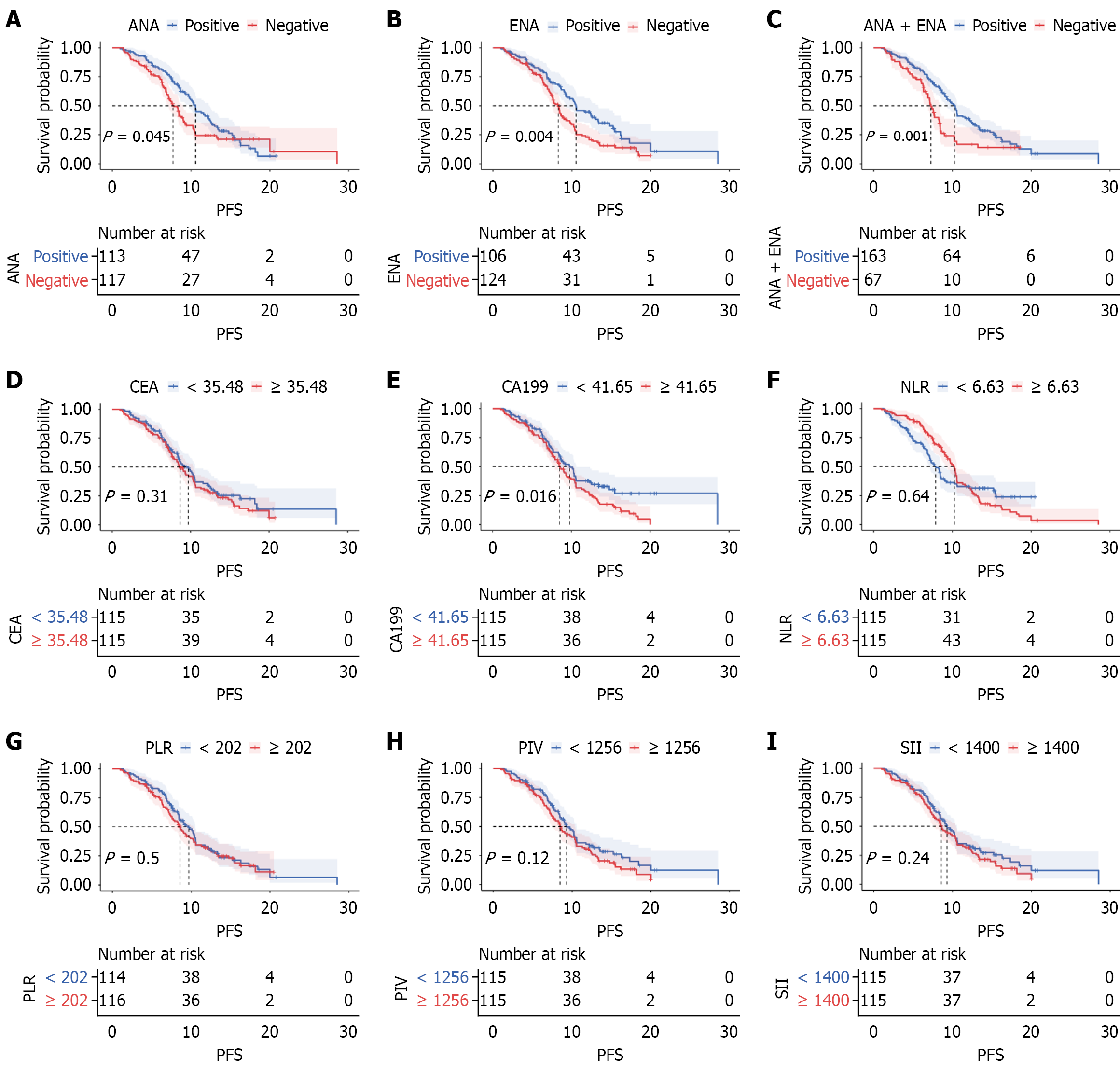
Figure 2 Kaplan-Meier analysis of progression-free survival based on autoantibody profiles.
A-C: Progression-free survival curves are shown for patient groups categorized by antinuclear antibody (ANA) status (A), extractable nuclear antigen (ENA) status (B), and the combined positive autoantibody criterion (ANA or ENA) (C); D: Carcinoembryonic antigen levels using a cutoff of 35.48; E: Carbohydrate antigen 199 levels using a cutoff of 41.65; F: Neutrophil-to-lymphocyte ratio using a cutoff of 6.63; G: Platelet-to-lymphocyte ratio using a cutoff of 202; H: Pan-immune-inflammation value using a cutoff of 1256; I: Systemic immune-inflammation index using a cutoff of 1400. PFS: Progression-free survival; ANA: Antinuclear antibody; ENA: Extractable nuclear antigen; CEA: Car
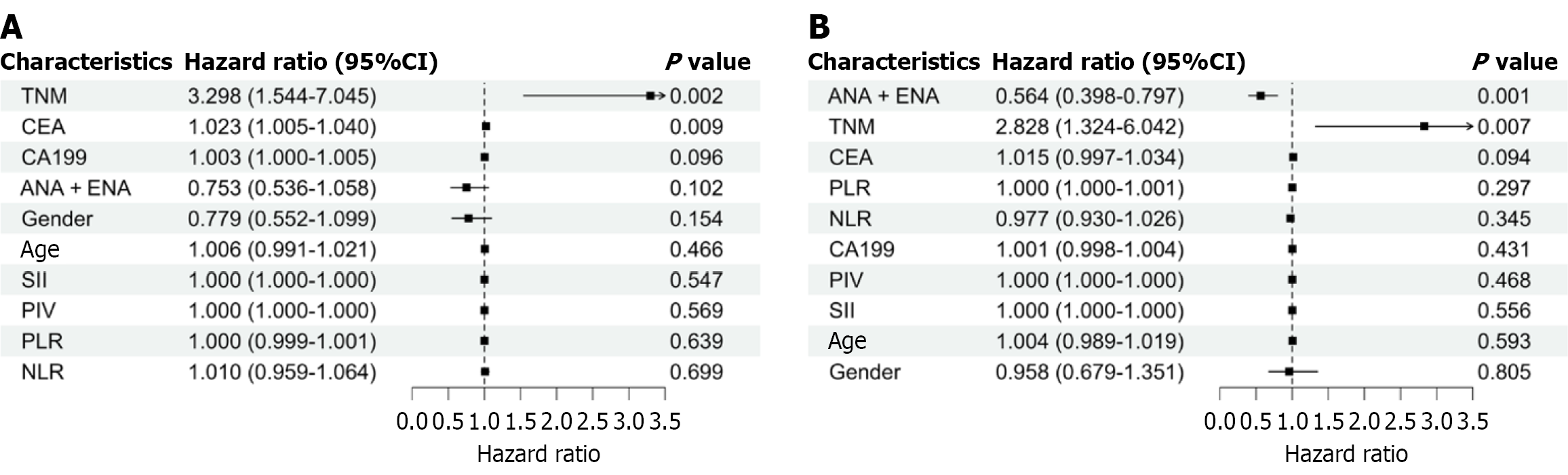
Figure 3 Univariate Cox regression analysis for overall survival and progression-free survival.
A: Forest plot of univariate Cox regression analysis for overall survival; B: Forest plot of univariate Cox regression analysis for progression-free survival. TNM: Tumor node metastasis; CI: Confidence interval; ANA: Antinuclear antibody; ENA: Extractable nuclear antigen; CEA: Carcinoembryonic antigen; CA199: Carbohydrate antigen 199; NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; PIV: Pan-immune-inflammation value; SII: Systemic immune-inflammation index.
Figure 4 Multivariate Cox regression analysis for overall survival and progression-free survival.
A: Forest plot of multivariate Cox regression analysis for overall survival; B: Forest plot of multivariate Cox regression analysis for progression-free survival. CI: Confidence interval; TNM: Tumor node metastasis; CEA: Carcinoembryonic antigen; ANA: Antinuclear antibody; ENA: Extractable nuclear antigen.
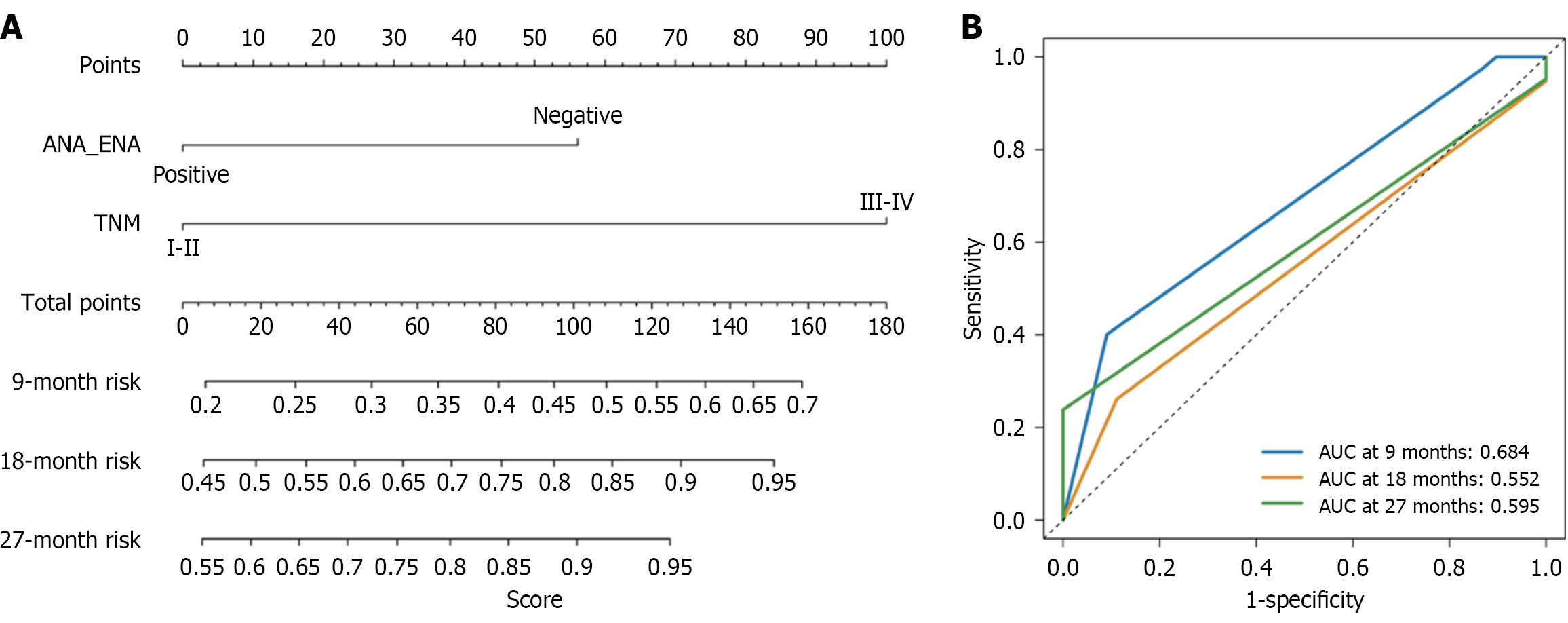
Figure 5 Multivariate Cox regression model for progression-free survival.
A: Nomogram of multivariate Cox regression analysis for progression-free survival; B: Receiver operating characteristic analysis of multivariate Cox regression analysis for progression-free survival with different time. ANA: Antinuclear antibody; ENA: Extractable nuclear antigen; TNM: Tumor node metastasis; AUC: Area under the curve.
- Citation: Zheng PM, Ouyang LB, Wang R, Jing KY, Gao HJ, Zhu CK. Clinical significance of autoantibody profiling and systemic inflammation in predicting outcomes of gastric cancer patients undergoing immunotherapy. World J Gastroenterol 2026; 32(17): 117823
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/117823.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.117823