Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 21, 2026; 32(15): 116364
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.116364
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.116364
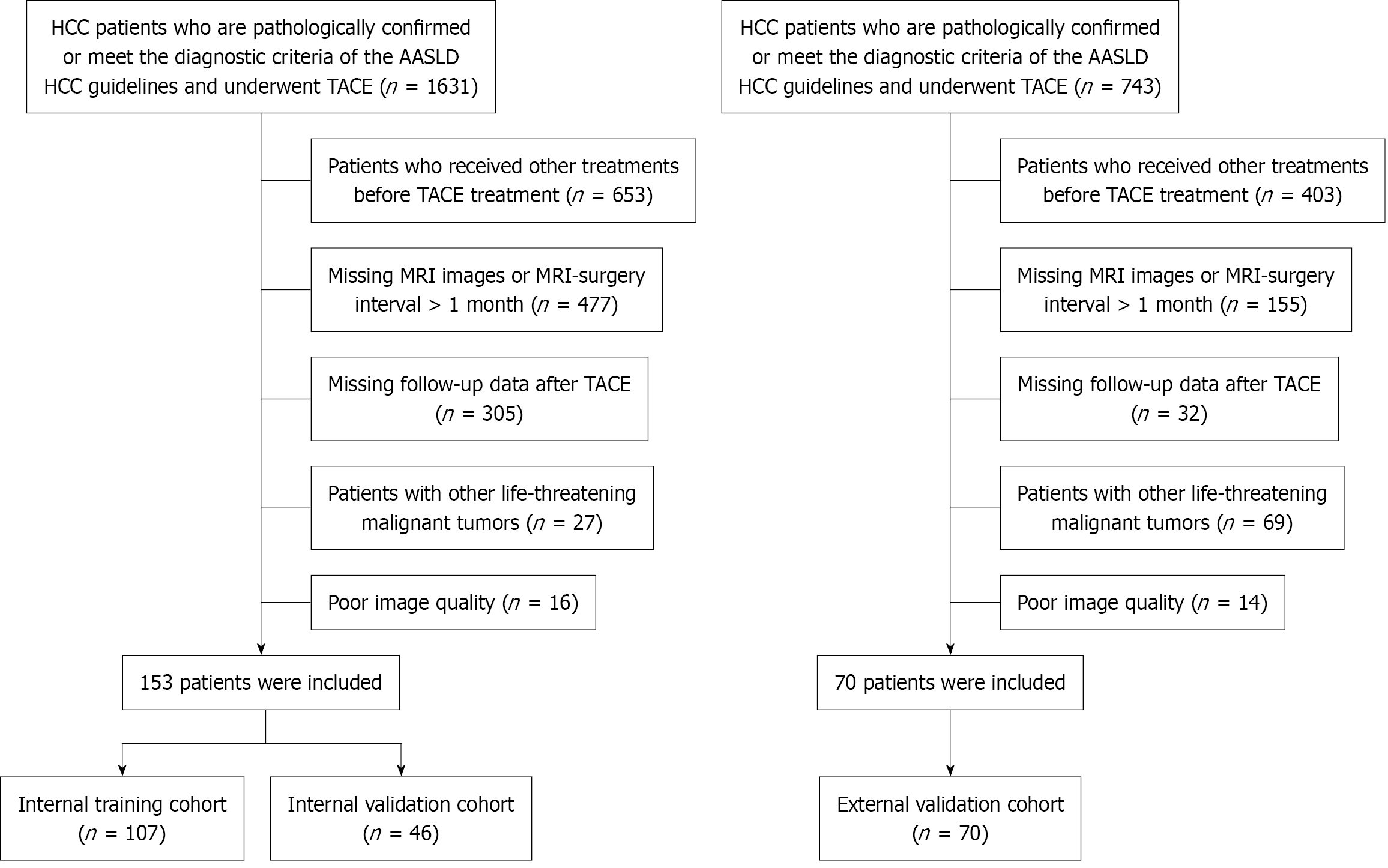
Figure 1 Enrollment and exclusion flowchart of patients with hepatocellular carcinoma.
The left side shows the screening process for institution A, where 1631 initial patients with hepatocellular carcinoma who underwent transcatheter arterial chemoembolization were screened. A total of 153 patients were finally enrolled and divided into an internal training set (n = 107) and an internal validation set (n = 46). The right side shows the screening process for institution B, where 743 initial patients with hepatocellular carcinoma who underwent transcatheter arterial chemoembolization were screened. A total of 70 patients were finally enrolled, serving as the external validation set. HCC: Hepatocellular carcinoma; AASLD: American Association for the Study of Liver Diseases; TACE: Transcatheter arterial chemoembolization; MRI: Magnetic resonance imaging.
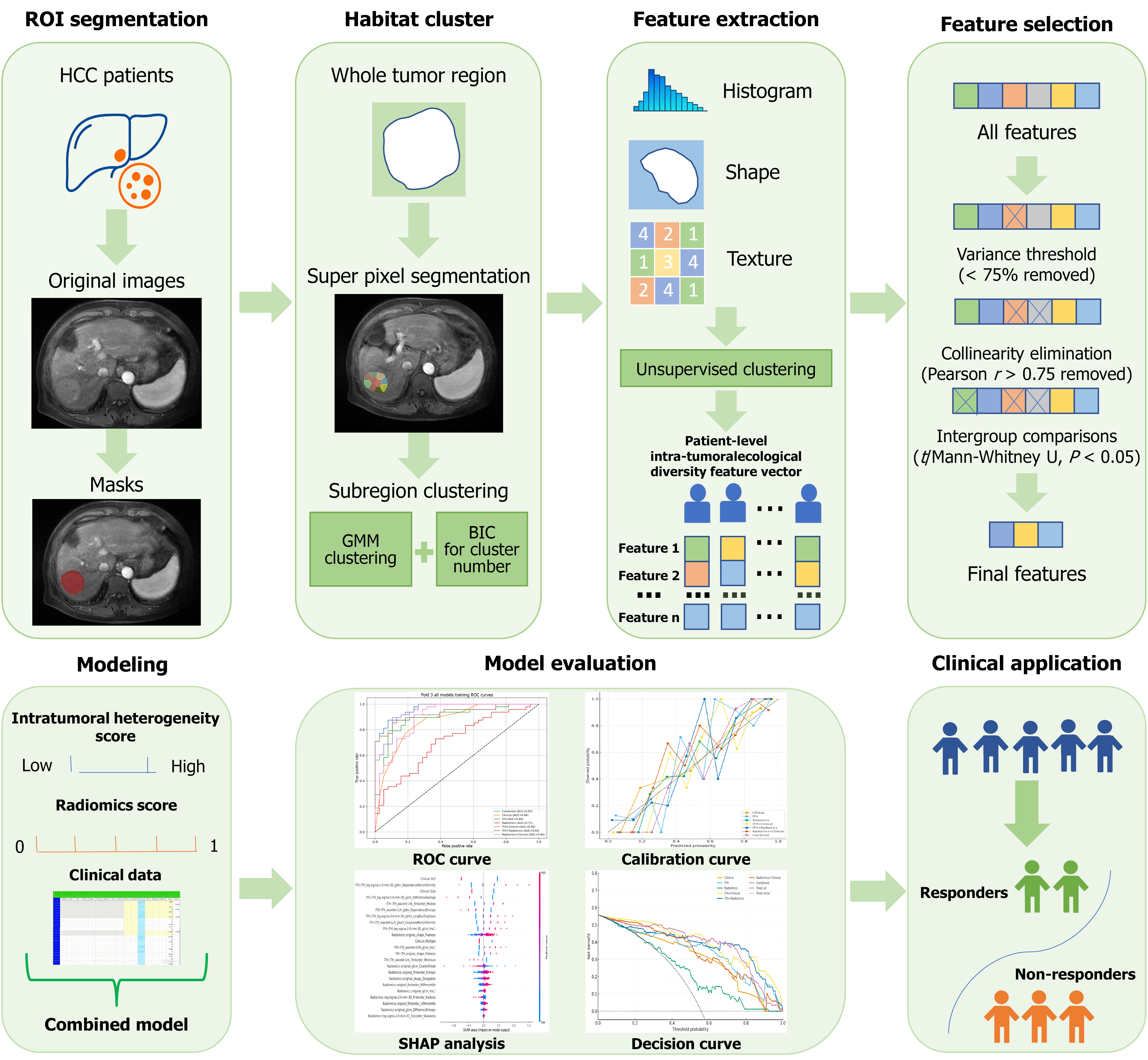
Figure 2 Schematic diagram of the methodological workflow.
The workflow is divided into two parts: The first part is preprocessing and feature acquisition. First, through region of interest segmentation, masks are obtained from the original images of hepatocellular carcinoma patients; then, via habitat clustering, subregion clustering is performed using simple linear iterative clustering superpixels to obtain habitats; subsequently, feature extraction and feature selection are conducted in sequence. The second part is model construction and application: A combined model is built based on the intratumoral heterogeneity score, radiomics score, and clinical data; model evaluation is completed using the receiver operating characteristic curve, calibration curve, SHapley Additive exPlanation analysis, and decision curve; finally, the model is applied in clinical practice to distinguish between responders (treatment responders) and non-responders (non-treatment responders). ROI: Region of interest; HCC: Hepatocellular carcinoma; GMM: Gaussian mixture model; BIC: Bayesian information criterion; ROC: Receiver operating characteristic; SHAP: SHapley Additive exPlanation.
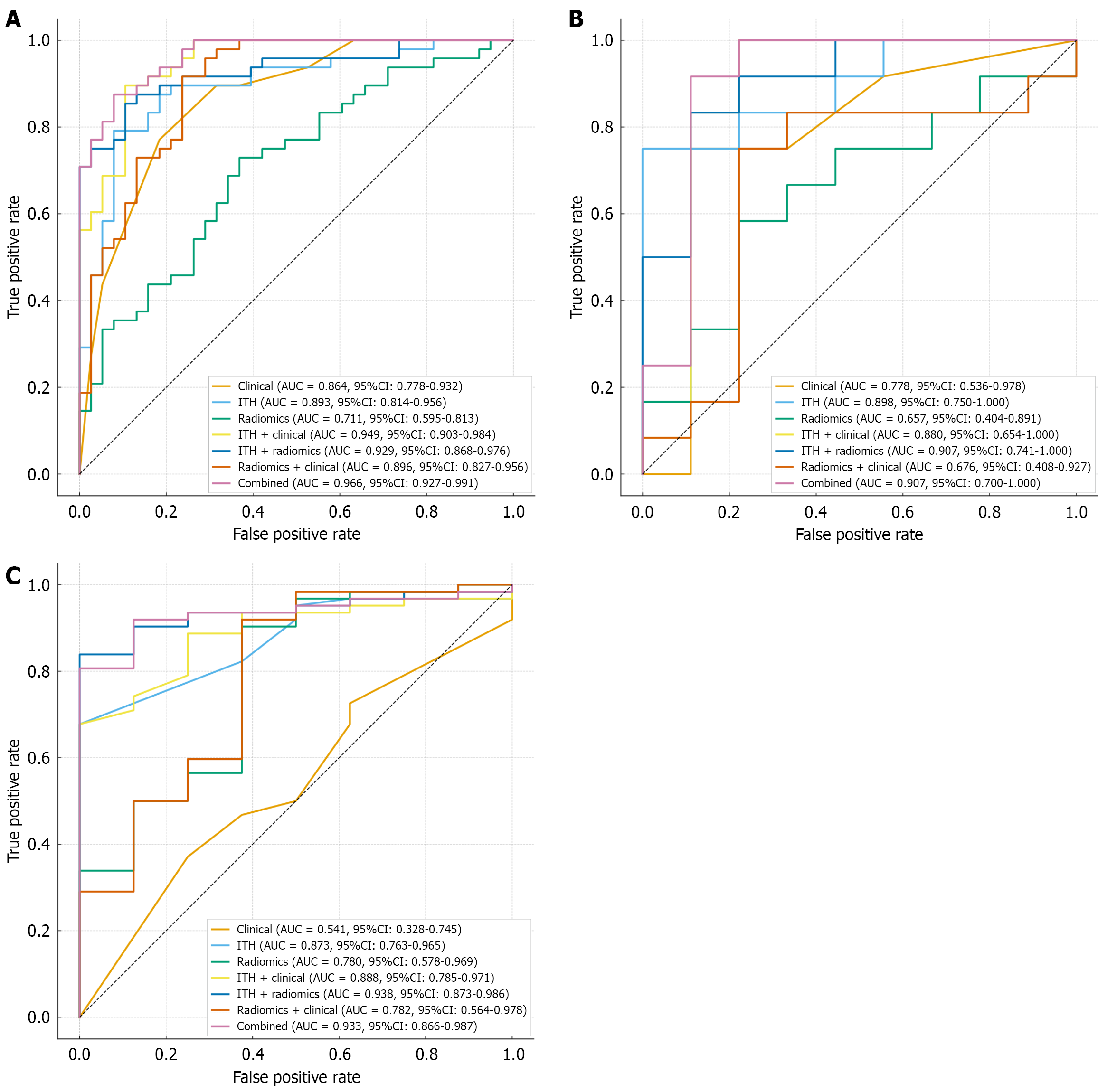
Figure 3 Performance and comparison of the seven models in the internal training set, internal validation set, and external validation set.
This figure presents the receiver operating characteristic curves of the seven models across three cohorts. A: Internal training set; B: Internal validation set; C: External validation set. The results show that the combined model, integrating clinical variables, traditional radiomics features, and the intratumoral heterogeneity index, exhibits the best performance. Its area under the curve values reach 0.97, 0.91, and 0.93 in the internal training set, internal validation set, and external validation set, respectively, which are the highest among all evaluated models. AUC: Area under the curve; CI: Confidence interval; ITH: Intratumoral heterogeneity.
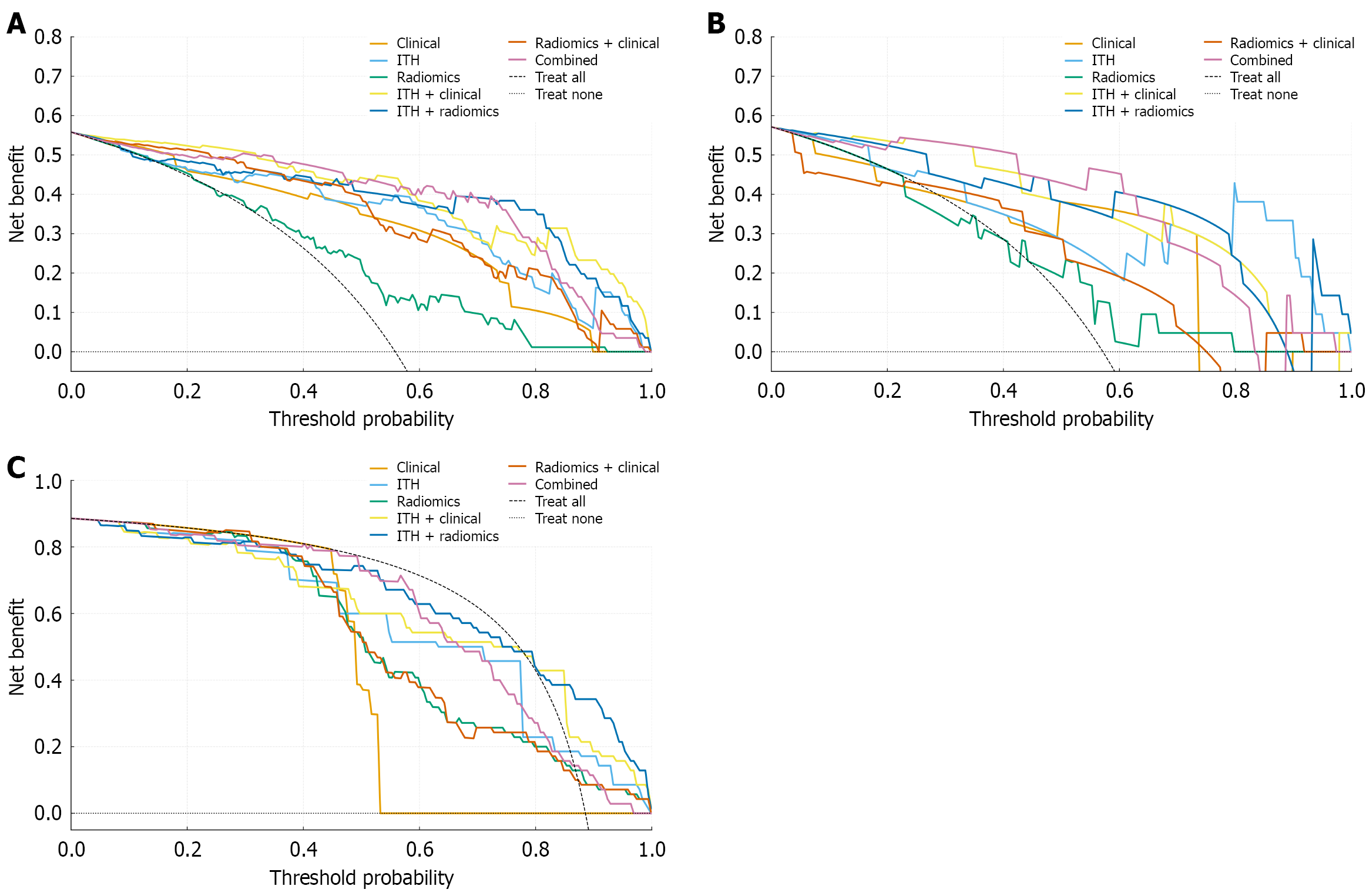
Figure 4 Decision curve analysis of the seven models in the internal training set, internal validation set, and external validation set.
This figure presents the decision curve analysis results of the seven models across three cohorts. A: Internal training set; B: Internal validation set; C: External validation set. As shown in the figure, the combined model, integrating clinical variables, traditional radiomics features, and the intratumoral heterogeneity index, achieves significantly higher net benefits than other single models or pairwise combined models within a wide range of threshold probability intervals in all three panels. This indicates that the combined model has good guiding value for clinical decision-making. ITH: Intratumoral heterogeneity.
- Citation: Lv JB, Liu W, Wei YG, Tang HN, Chen QQ, Hu HJ, Hu JB. Habitat imaging on contrast-enhanced magnetic resonance imaging predicts early response to transarterial chemoembolization in hepatocellular carcinoma. World J Gastroenterol 2026; 32(15): 116364
- URL: https://www.wjgnet.com/1007-9327/full/v32/i15/116364.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i15.116364