Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 21, 2026; 32(15): 115995
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.115995
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.115995
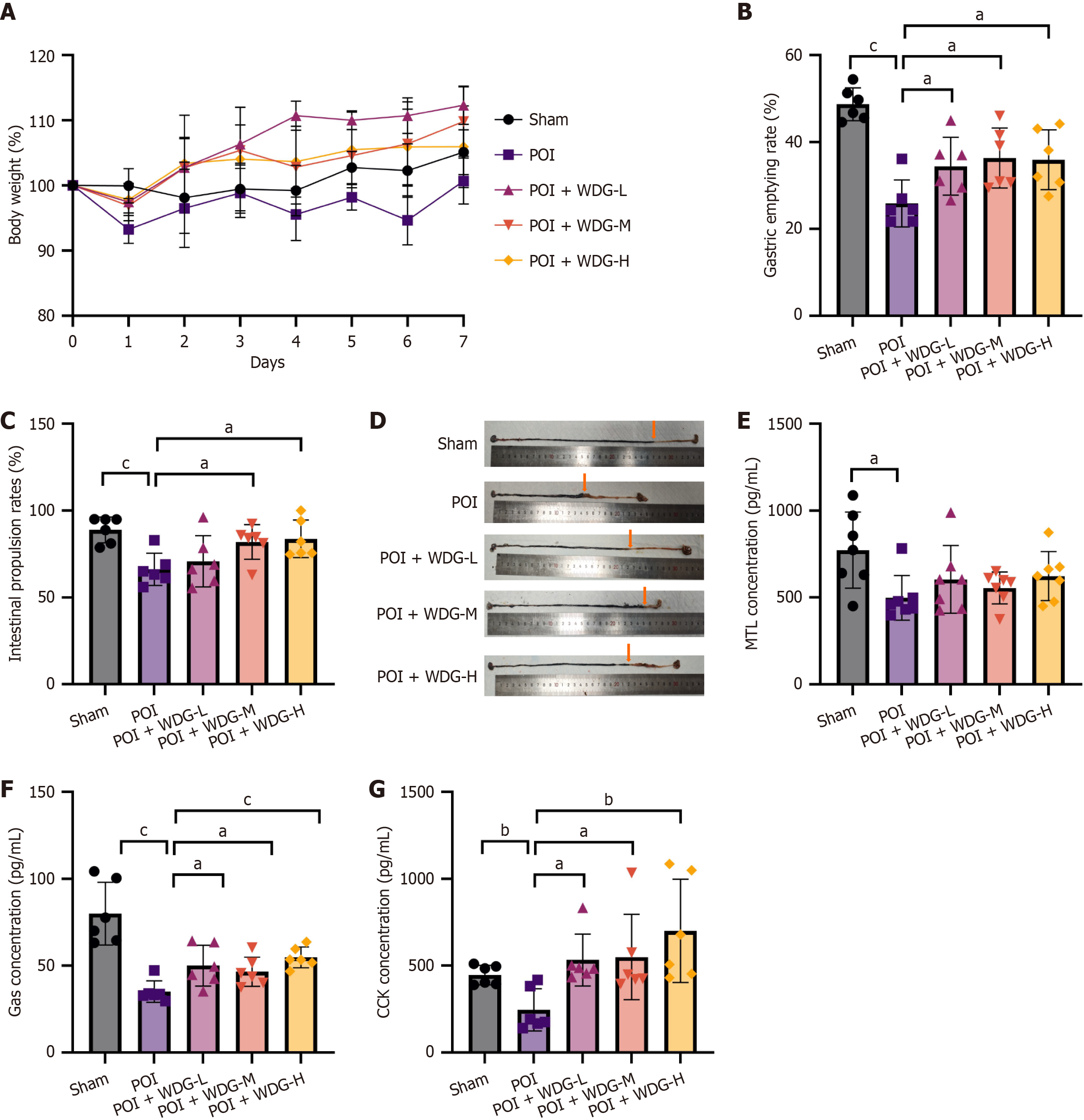
Figure 1 Wuda granules improved gastrointestinal motility in postoperative ileus mice.
A: Body-weight change; B: Gastric emptying rate; C and D: Statistical results and representative images of charcoal powder propulsion distance in the small intestine; E-G: Serum levels of cholecystokinin, gastrin, and motilin. Data are presented as mean ± SD (n = 6). aP < 0.05 vs postoperative ileus, bP < 0.01 vs postoperative ileus, and cP < 0.001 vs postoperative ileus. POI: Post
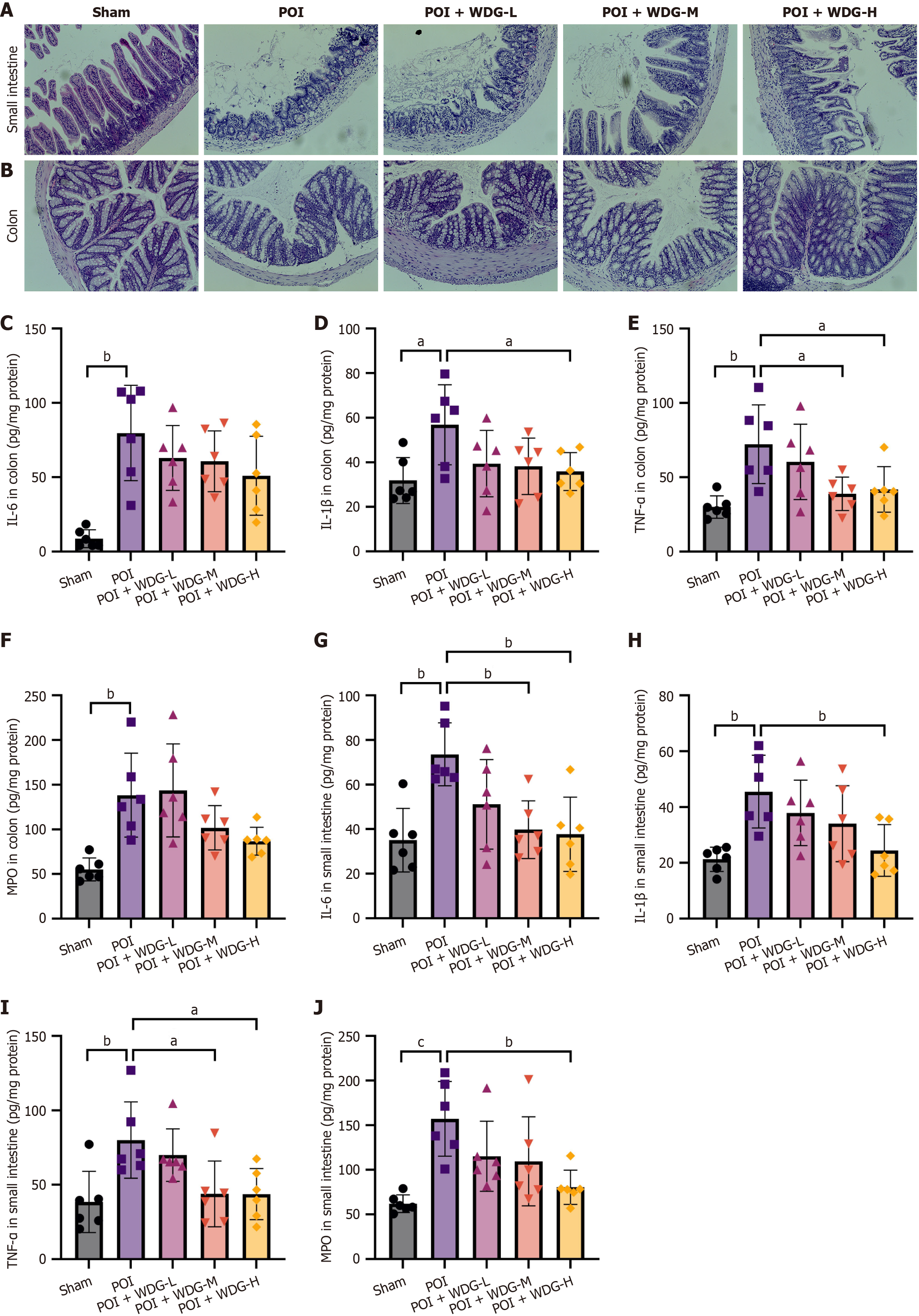
Figure 2 Effects of Wuda granules on histopathological changes and inflammatory cytokine levels in the colon and small intestine of postoperative ileus mice.
A and B: Hematoxylin and eosin-stained images of the small intestine and colon tissues (magnification: 200 ×); C-F: Enzyme-linked immunosorbent assay analysis of interleukin (IL)-6, IL-1β, tumor necrosis factor-α, and myeloperoxidase expression in colon tissue; G-J: Enzyme-linked immunosorbent assay analysis of IL-6, IL-1β, tumor necrosis factor-α, and myeloperoxidase expression in small intestine tissue. aP < 0.05 vs postoperative ileus, bP < 0.01 vs postoperative ileus, and cP < 0.001 vs postoperative ileus. POI: Postoperative ileus; WDG: Wuda granules; IL: Interleukin; TNF-α: Tumor necrosis factor-α; MPO: Myeloperoxidase.
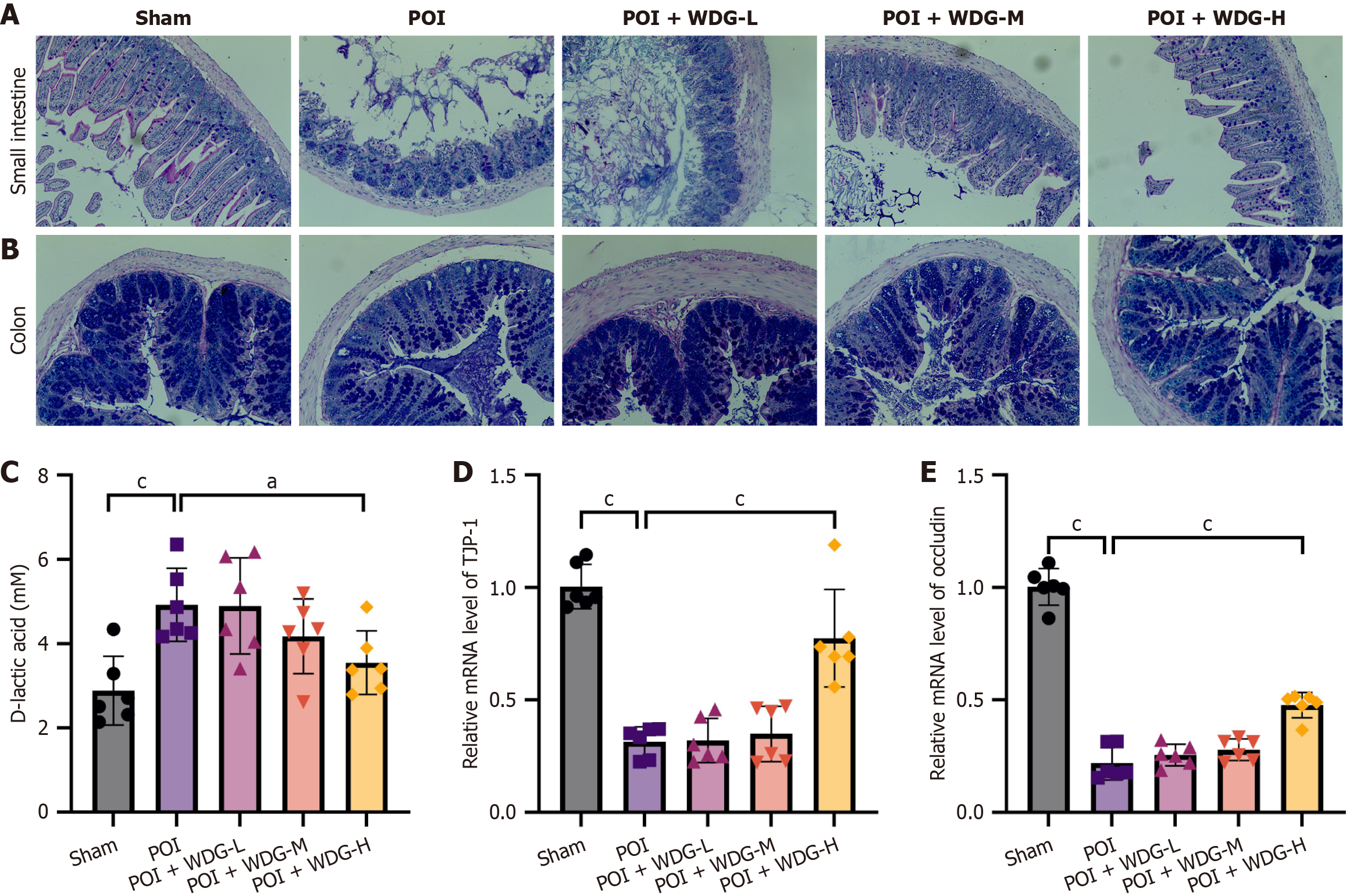
Figure 3 Wuda granules improve intestinal barrier structure and function in postoperative ileus mice.
A and B: Alcian blue-periodic acid Schiff staining of the small intestine and colon (magnification: 200 ×); C: Serum D-lactic acid levels; D and E: Relative mRNA expression of tight-junction protein-1 and occludin in small-intestinal tissue, as determined by reverse transcription-quantitative polymerase chain reaction. Data are presented as mean ± SD (n = 6). aP < 0.05 vs postoperative ileus, and cP < 0.001 vs postoperative ileus. HE: Hematoxylin and eosin; POI: Postoperative ileus; WDG: Wuda granules; TJP-1: Tight-junction protein-1.
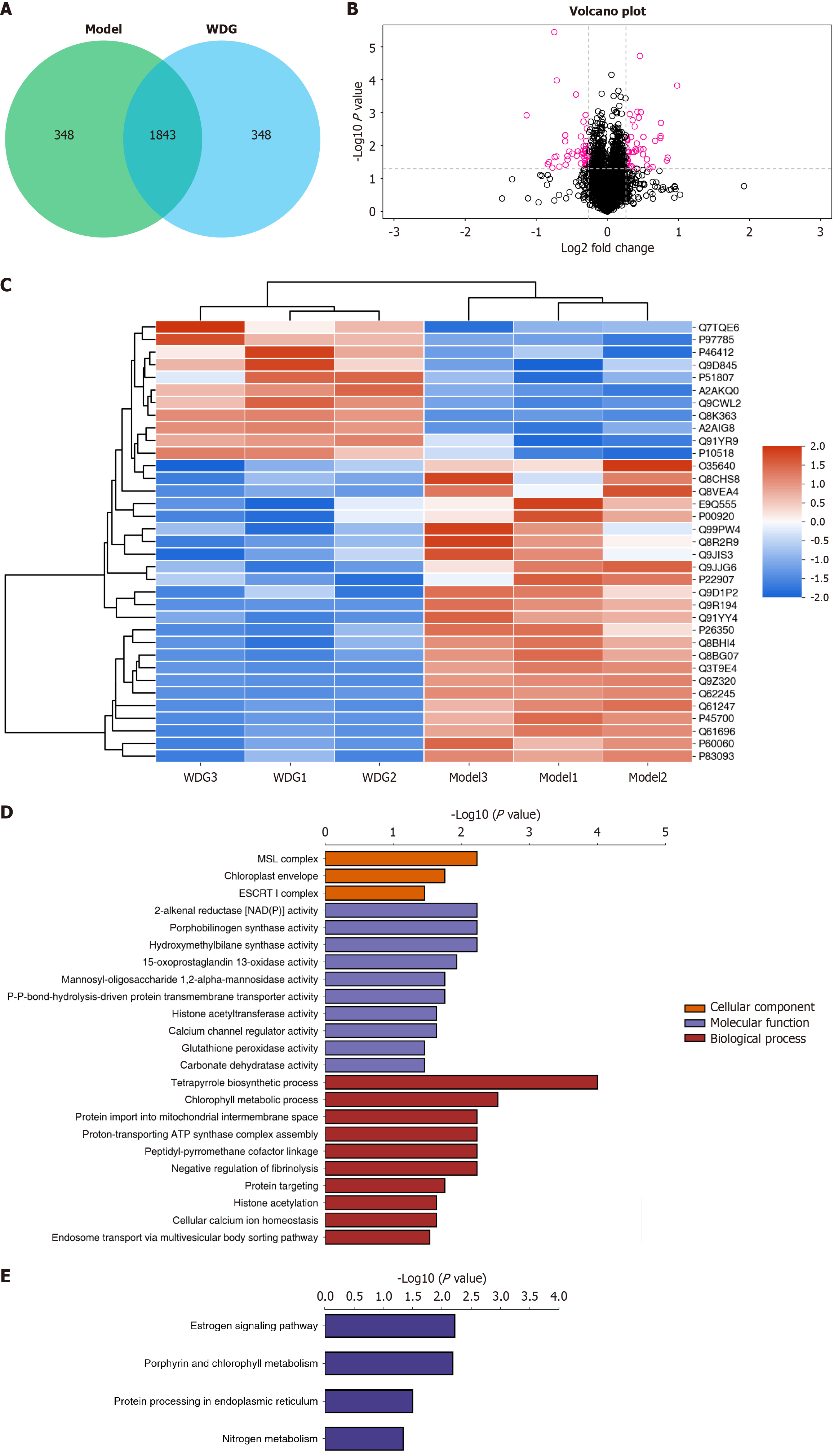
Figure 4 Proteomics analysis of Wuda granules in colon tissues of postoperative ileus mice.
A: Venn diagram showing differentially expressed proteins shared between the postoperative ileus model and Wuda granules-treated groups; B: Volcano plot indicating significantly upregulated and downregulated proteins (red dots) with |log2 fold changes| > 1 and P < 0.05; C: Heatmap illustrating the expression profiles of differentially expressed proteins across samples, with red to blue indicating high to low abundance; D: Gene Ontology enrichment analysis of differential proteins categorized by cellular component, molecular function, and biological process; E: Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis of differentially expressed proteins. WDG: Wuda granules.
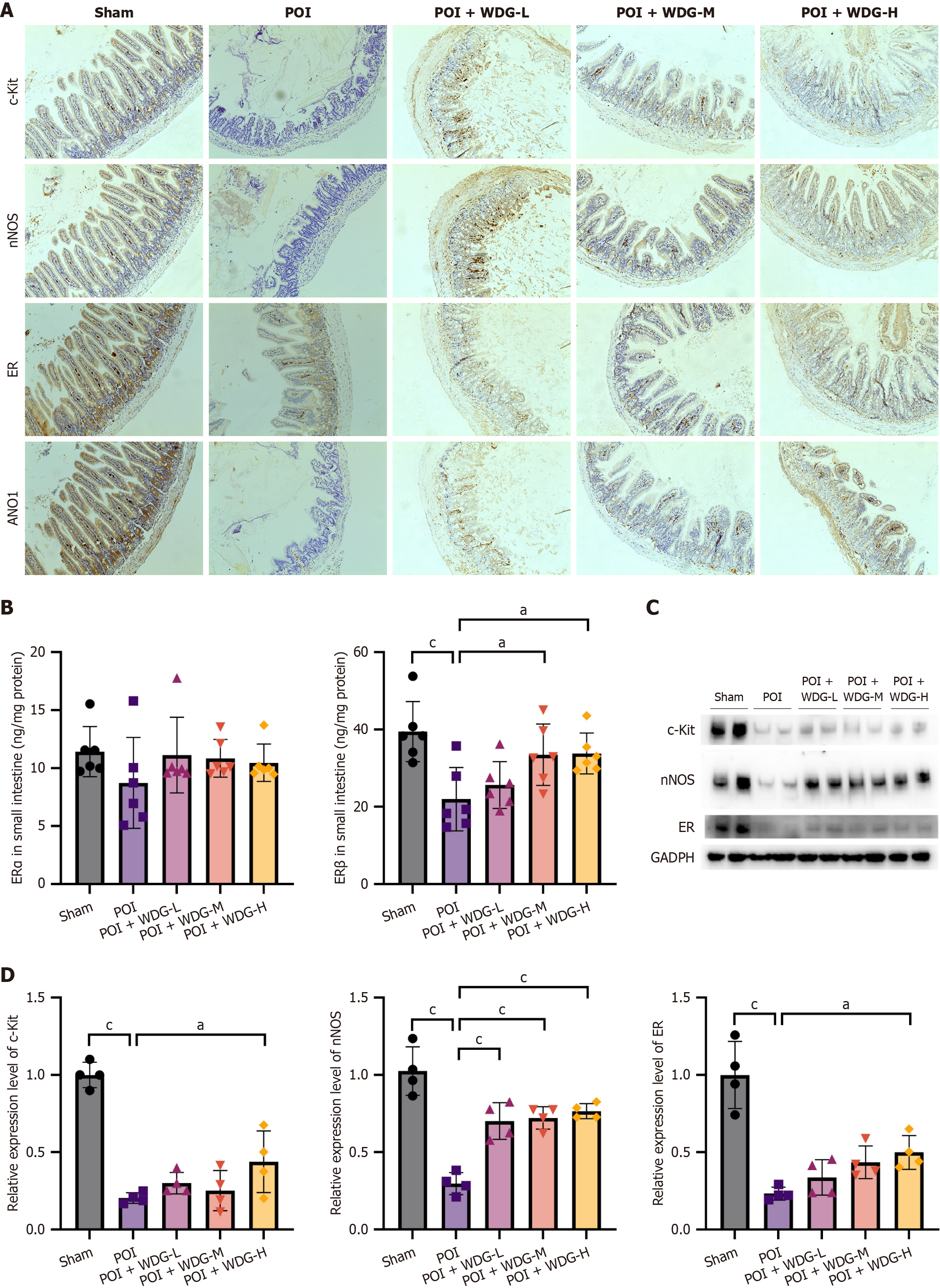
Figure 5 Effects of Wuda granules on the expression of the enteric pacemaker-hormonal-neuronal regulation axis in the small intestine of postoperative ileus mice.
A: Representative immunohistochemical staining of tyrosine-protein kinase kit, neuronal nitric oxide synthase, estrogen receptor (ER), and anoctamin-1 in small-intestinal tissues from different groups (magnification: 200 ×); B: Enzyme-linked immunosorbent assay analysis of ERα and ERβ protein levels in the small intestine (n = 6); C: Western blot analysis of tyrosine-protein kinase kit, neuronal nitric oxide synthase, and ER protein levels in intestinal tissues; D: Quantification of protein expression levels normalized to 3-phosphate dehydrogenase (n = 4). Data are presented as mean ± SD. aP < 0.05 vs postoperative ileus, and cP < 0.001 vs postoperative ileus. POI: Postoperative ileus; WDG: Wuda granules; c-Kit: Tyrosine-protein kinase kit; nNOS: Neuronal nitric oxide synthase; ER: Estrogen receptor; ANO1: Anoctamin-1; GAPDH: 3-phosphate dehydrogenase.
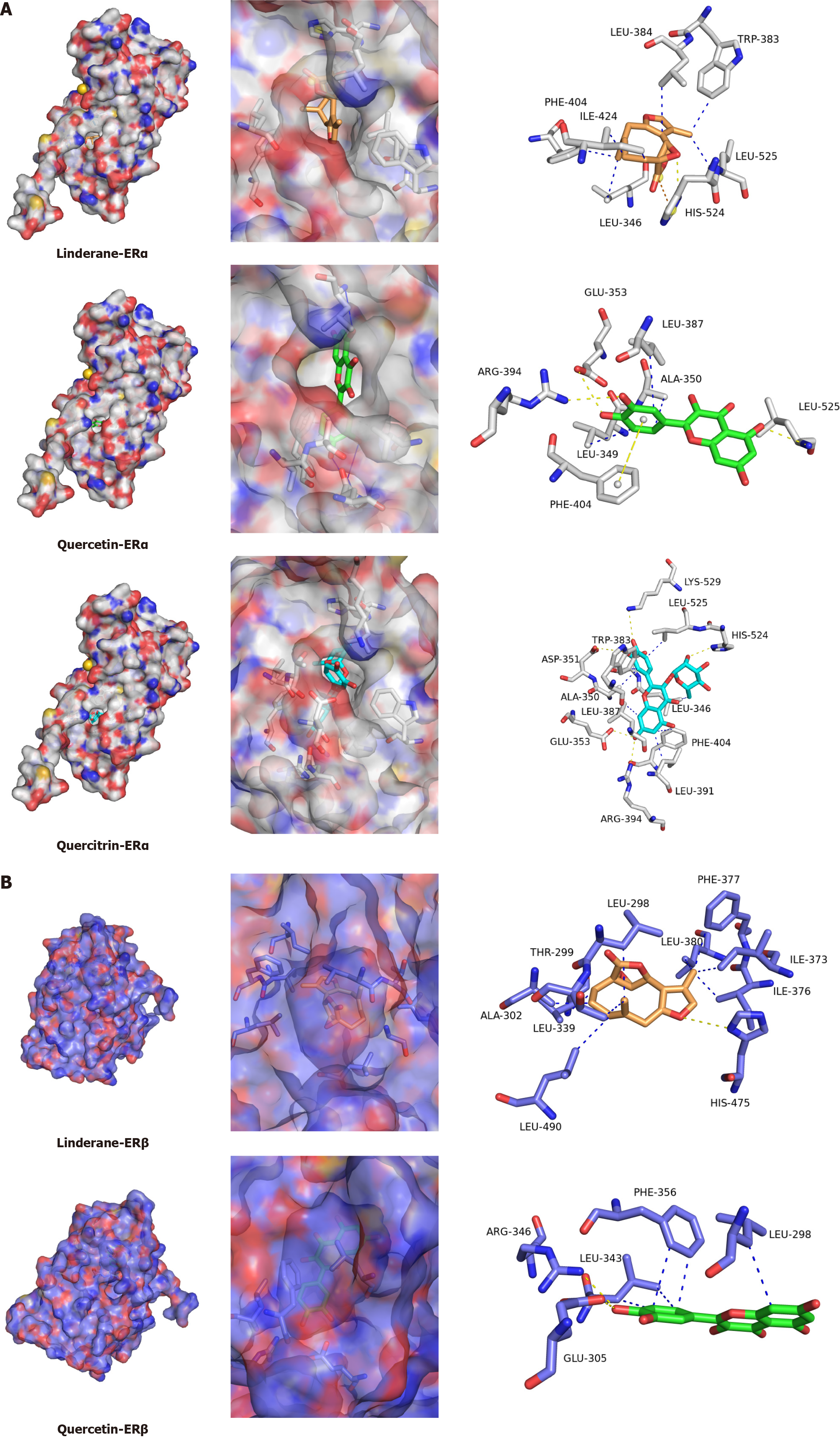
Figure 6 Molecular docking of representative chemical constituents from Wuda granules with estrogen receptors.
A: Binding modes of linderane, quercetin, and quercitrin with estrogen receptor α (PDB ID: 1a52); B: Binding modes of linderane and quercetin with estrogen receptor β (PDB ID: 5toa). Electrostatic surface representations (left), binding pocket interactions (middle), and two-dimensional interaction diagrams (right) are shown. The interaction types are indicated by different colors: Hydrogen bonds, yellow; hydrophobic interactions, blue; and salt bridges, orange. ER: Estrogen receptor.
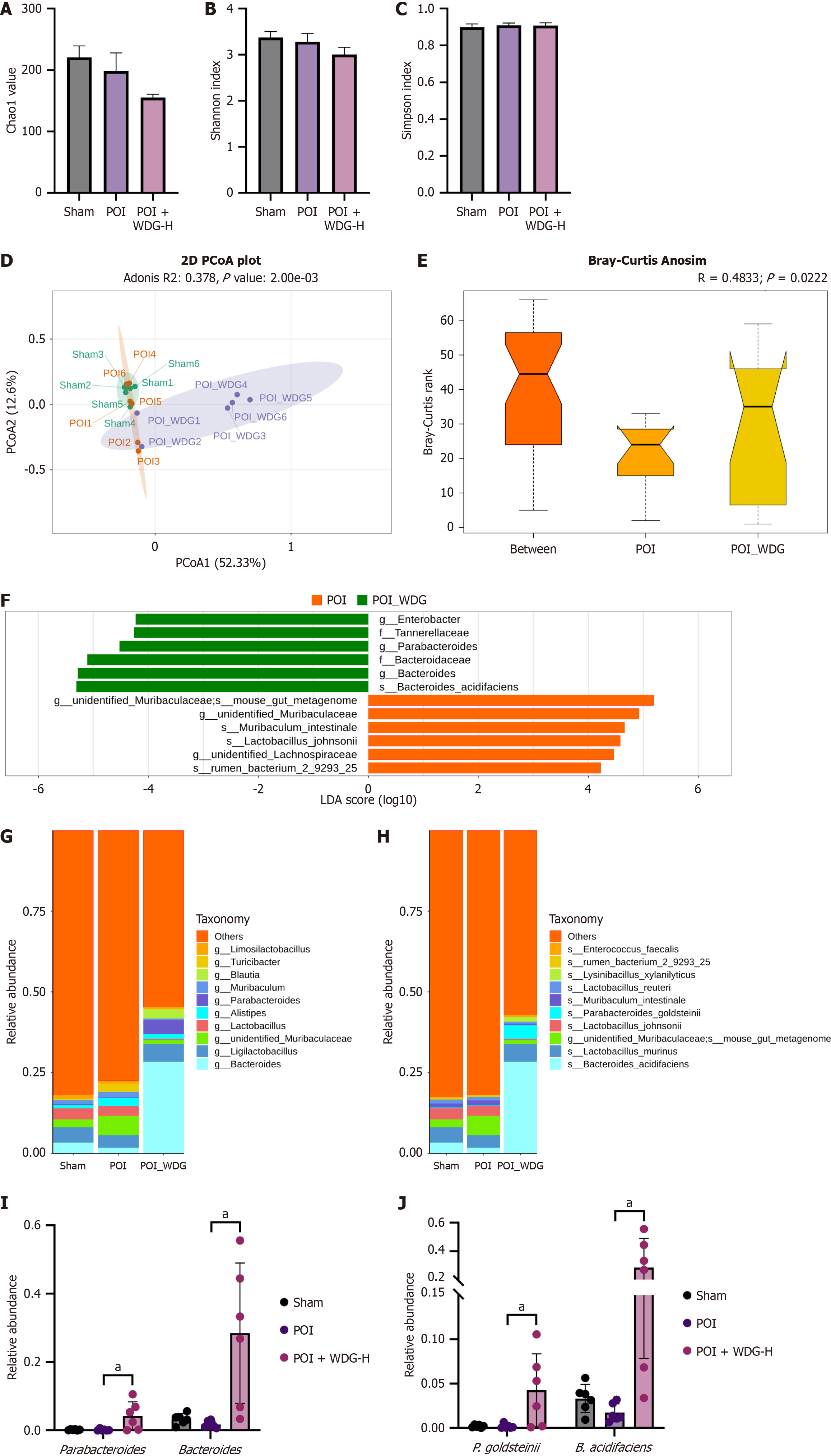
Figure 7 Effects of Wuda granules on the intestinal flora in postoperative ileus mice.
A-C: Α-diversity indices: Chao1, Shannon, and Simpson; D: Principal coordinates analysis plot based on the Bray-Curtis distance showing β-diversity differences among groups; E: Bray-Curtis Anosim test validating community structure differences; F: Linear discriminant analysis revealing significantly different taxa between postoperative ileus and Wuda granules groups (linear discriminant analysis score > 3); G and H: Gut microbiota composition at the genus and species levels; I and J: Relative abundance of key differential genera (Parabacteroides, Bacteroides) and species (Parabacteroides goldsteinii, Bacteroides acidifaciens). aP < 0.05 vs postoperative ileus. POI: Postoperative ileus; WDG: Wuda granules; PCoA: Principal coordinates analysis; P. goldsteinii: Parabacteroides goldsteinii; B. acidifaciens: Bacteroides acidifaciens.
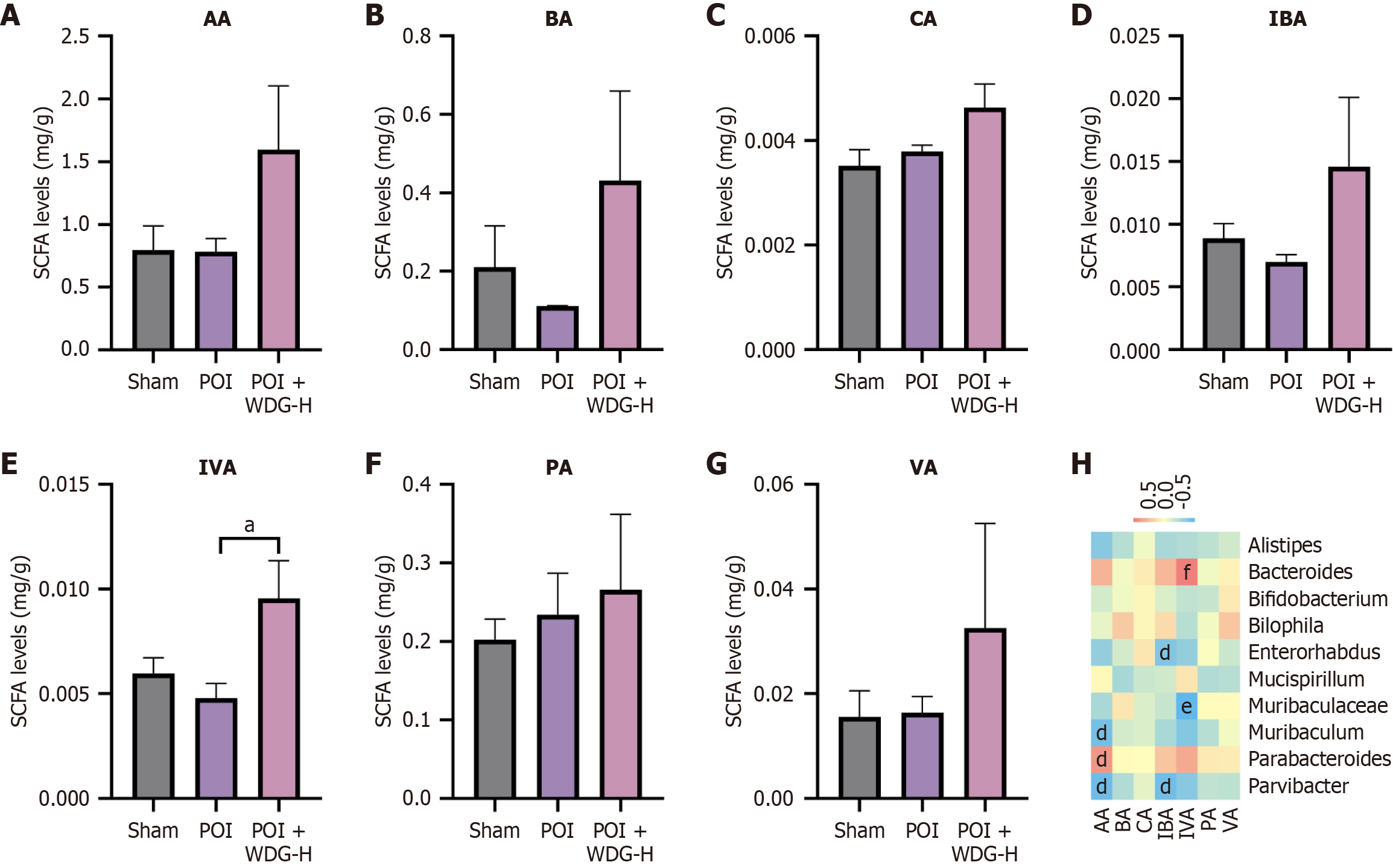
Figure 8 Effects of Wuda granules on fecal short-chain fatty acid levels in postoperative ileus mice.
A-G: Concentrations of various short-chain fatty acids: Acetic acid, butyric acid, caproic acid, isobutyric acid, isovaleric acid, propionic acid and valeric acid; H: Spearman correlation of the top 10 gut microbial genera and short-chain fatty acids. aP < 0.05 vs postoperative ileus; dP < 0.05, eP < 0.01, and fP < 0.001, gut microbial genera vs short-chain fatty acids. AA: Acetic acid; BA: Butyric acid; CA: Caproic acid; IBA: Isobutyric acid; IVA: Isovaleric acid; PA: Propionic acid; VA: Valeric acid; SCFA: Short-chain fatty acid.
- Citation: Wang T, Xu YH, Ou YH, Xiong WJ, Luo LJ, Li J, Peng YH, Chen Y, Zeng HP, Yu Y, Tang HP, Wang F, Yao HL, Wang W. Wuda granules target estrogen receptors and modulate gut microbiota to alleviate postoperative ileus: A multi-omics perspective. World J Gastroenterol 2026; 32(15): 115995
- URL: https://www.wjgnet.com/1007-9327/full/v32/i15/115995.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i15.115995