©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 14, 2026; 32(10): 116152
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.116152
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.116152
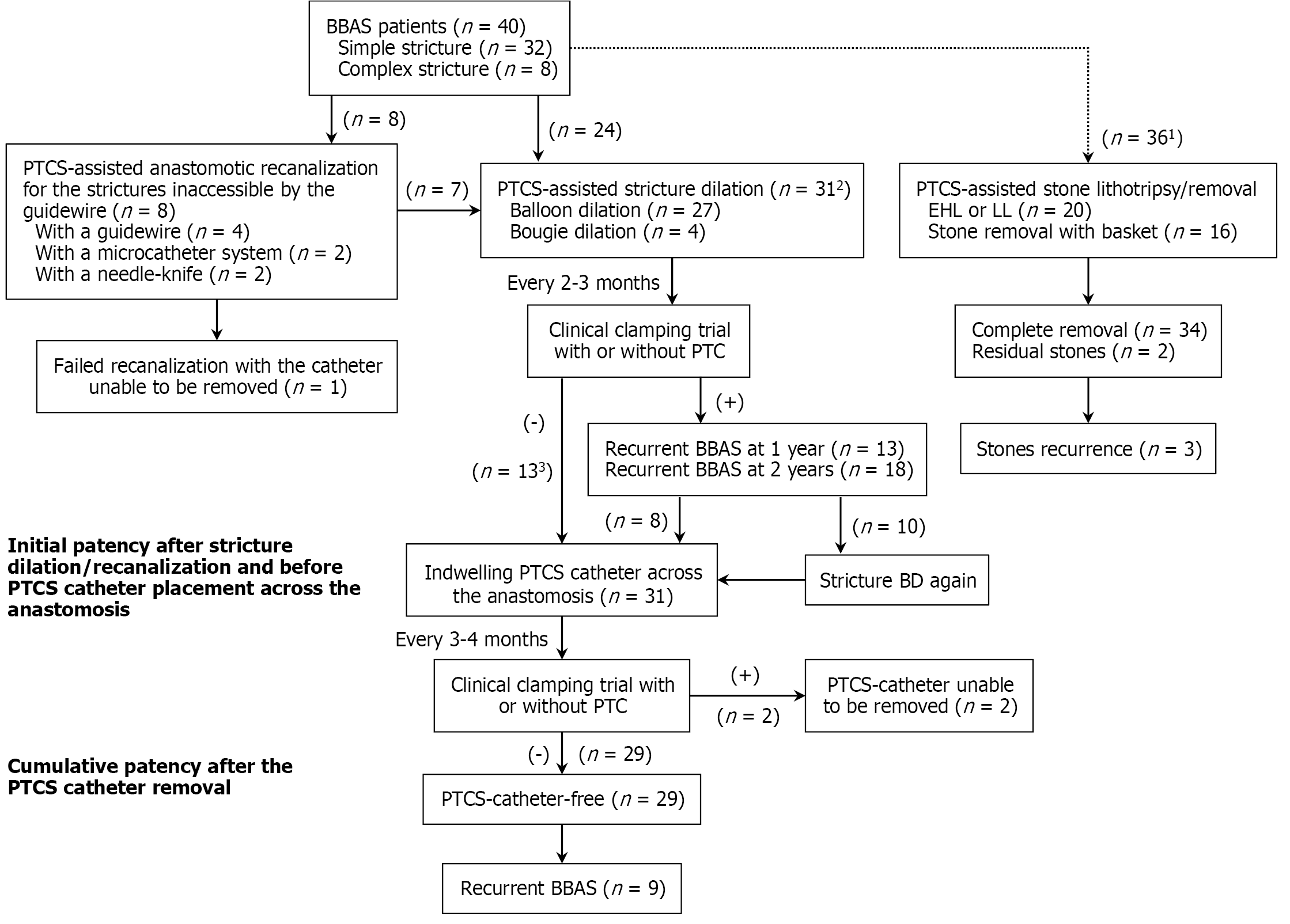
Figure 1 The technical details of percutaneous transhepatic cholangioscopy-assisted procedures for the treatment of benign bilioenteric anastomotic strictures in 40 patients.
1Including 31 patients who underwent percutaneous transhepatic cholangioscopy-assisted stricture dilation (24 with stricture dilation alone and 7 with successful anastomotic recanalization prior to stricture dilation). 2Twenty-eight patients received anastomotic stricture dilation/recanalization and 8 with mild stricture did not receive stricture dilation. 3Placement of an indwelling percutaneous transhepatic cholangioscopy catheter across the anastomosis due to unsuccessful first balloon dilation, residual or potential stone, or bougie dilation alone. PTCS: Percutaneous transhepatic cholangioscopy; BBAS: Benign bilioenteric anastomotic stricture; EHL: Electrohydraulic lithotripsy; LL: Laser lithotripsy; PTC: Percutaneous transhepatic cholangiography; BD: Balloon dilation.
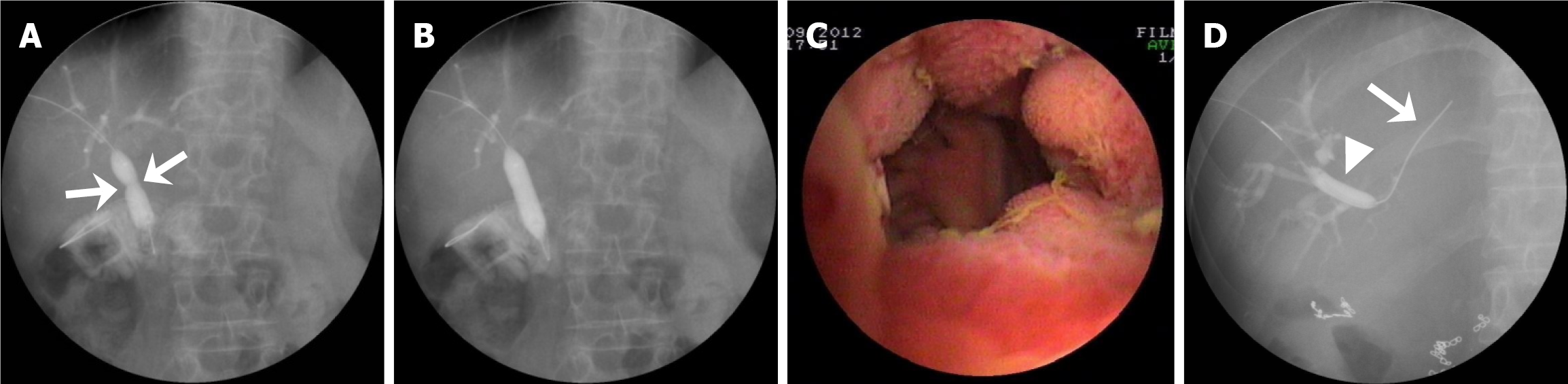
Figure 2 Representative images showing successful percutaneous transhepatic cholangioscopy-assisted balloon dilation for the treatment of simple and complex benign bilioenteric anastomotic strictures.
A-C: Cholangiogram showing that the 10-mm balloon is incompletely inflated in the waist (arrow, A) and the balloon waist disappeared (arrow) when the appropriate pressure had been reached (B), and patent anastomosis 18 months after the dilation (C) in a patient with a simple benign bilioenteric anastomotic stricture (BBAS); D: An 8-mm balloon was inserted under guidance of a guidewire (arrow) to dilate the stricture in the opposite left hepatic duct; the X-ray shows the disappearance of the balloon waist (arrow) in a patient with a complex BBAS.
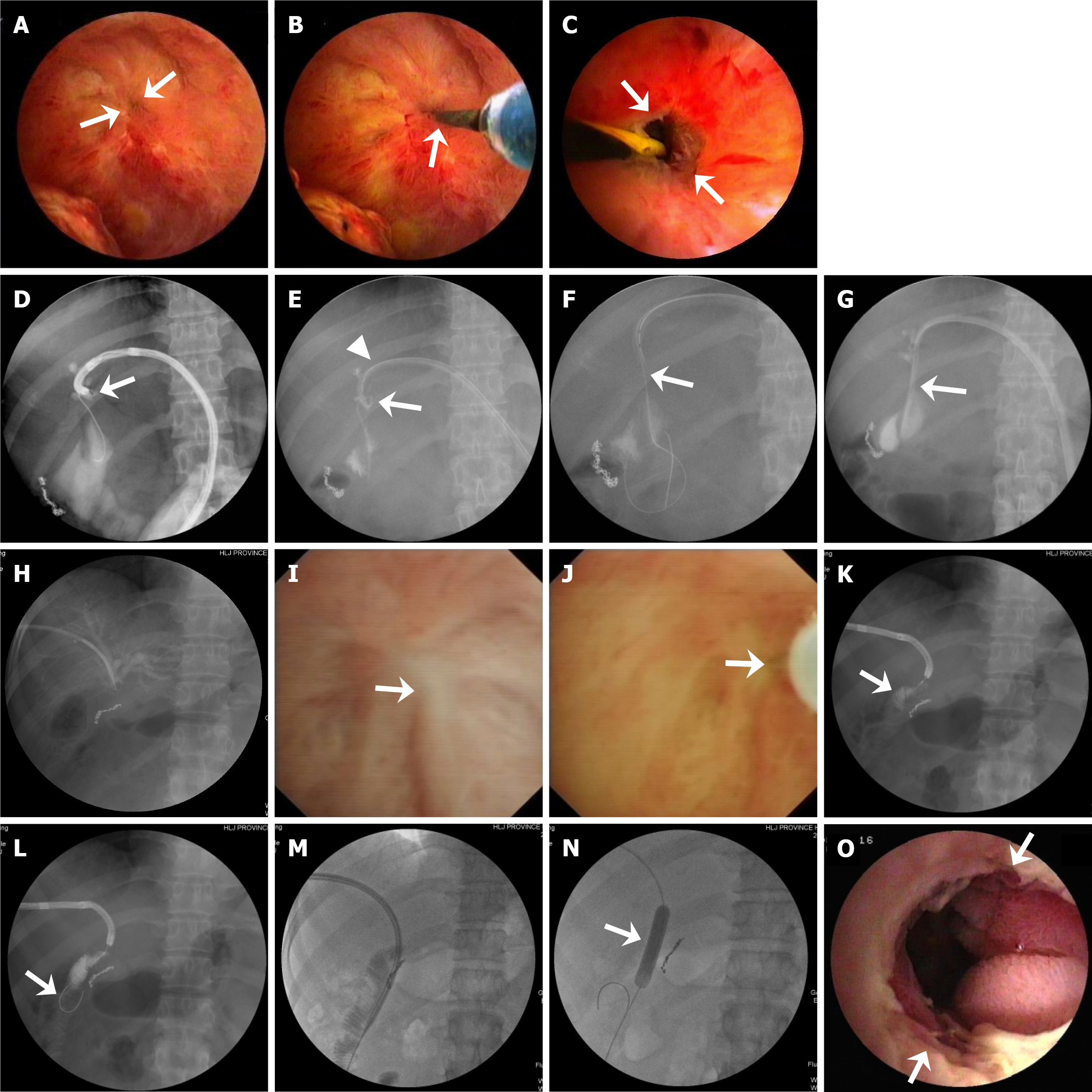
Figure 3 Representative images showing successful percutaneous transhepatic cholangioscopy-assisted anastomotic recanalization in patients with severe benign bilioenteric anastomotic stricture with a guidewire, the microcatheter system, and a needle knife.
A-C: In a patient with a small dimple anastomotic site (arrow) in the center of a fibrous scar (converging mucosal membrane) (A), a zebra guidewire (via an endoscopic retrograde cholangiopancreatography cannula) with a flexible tip was used to tap the mucosa (arrow) in the site of the dimple (B) and after the guidewire passed through the stenotic anastomosis, initial dilation (arrow points to the open anastomosis) for the stricture was performed with a fine bougie (C); D-G: In a patient with a severe anastomotic stricture, a stiff 0.025-inch micro-guidewire, guided under percutaneous transhepatic cholangioscopy (PTCS), was looped through stenotic anastomosis of sharp angulation and looping (arrow in D). Then a 5-Fr microcatheter (≤ 3 Fr at the tip) was looped (arrow) to pass through the stricture and kept in place within the PTCS catheter (arrowhead points to the tip of the PTCS catheter above the stricture) (E). The looping stricture was straightened with a dreamwire (arrow) under fluoroscopy one month later (F), and an 18-Fr PTCS catheter (arrow) was finally placed through the stricture after stepwise bougie dilation (G); H-O: In a patient with complete bilioenteric anastomotic membranous occlusion, percutaneous transhepatic cholangiography shows no passage of the contrast medium through the anastomosis (H). PTCS only shows the converging mucosal membrane (arrow) in the area of scars, but not the anastomosis (I). A 25-gauge injection needle (arrow) was used to puncture the center of the scars under direct visualization (J). Radiography shows the jejunal circular folds (arrow) (K). After a small incision was made with a needle knife at the puncture site, a guidewire was inserted, which was confirmed in the jejunum (arrow) (L). Then, the stenotic anastomosis was dilated with a bougie (M) and a 10-mm balloon with no remaining waist (arrow) (N). PTCS shows enlargement of the anastomosis (arrows) after balloon dilation for nine days (O), indicating successful recanalization and dilation.
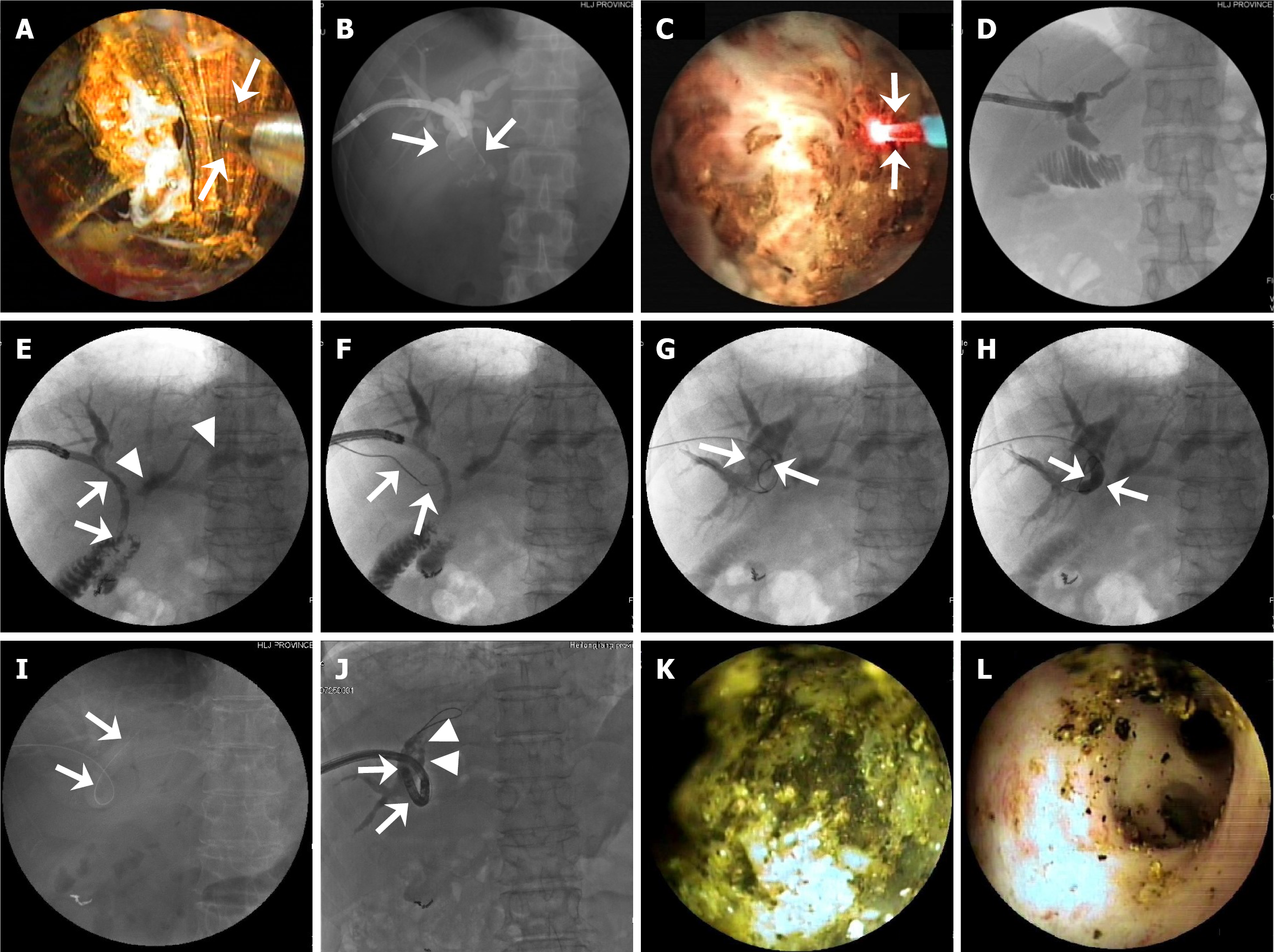
Figure 4 Representative images of successful percutaneous transhepatic cholangioscopy-assisted biliary stone therapy with elec
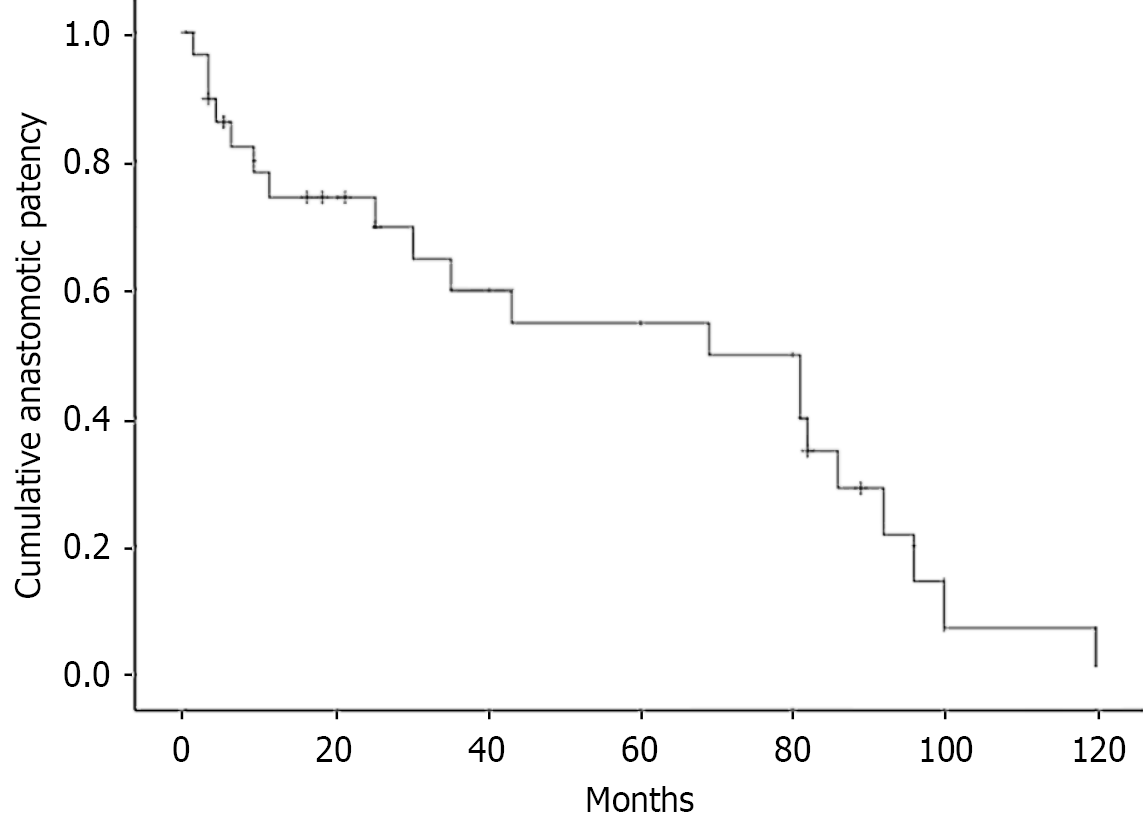
Figure 5 Kaplan-Meier methods.
Results show that in 29 patients with an indwelling percutaneous transhepatic cholangioscopy catheter via the anastomotic stricture that was removed, the cumulative anastomotic patency remained in 74.3%, 69.7%, 59.7%, 49.8%, and 34.8% of patients at 1, 2, 3, 5, and 7 years, respectively. The median patency duration was 69 months, with 95% confidence intervals of 21 to 117 months.
- Citation: Ren X, Wang C, Qu YP, Tang XF, Xia T, Lu YX, Sun XM. Percutaneous transhepatic cholangioscopy-assisted procedures for the management of postoperative benign bilioenteric anastomotic strictures with or without biliary stones. World J Gastroenterol 2026; 32(10): 116152
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/116152.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.116152