©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 14, 2026; 32(10): 115371
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.115371
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.115371
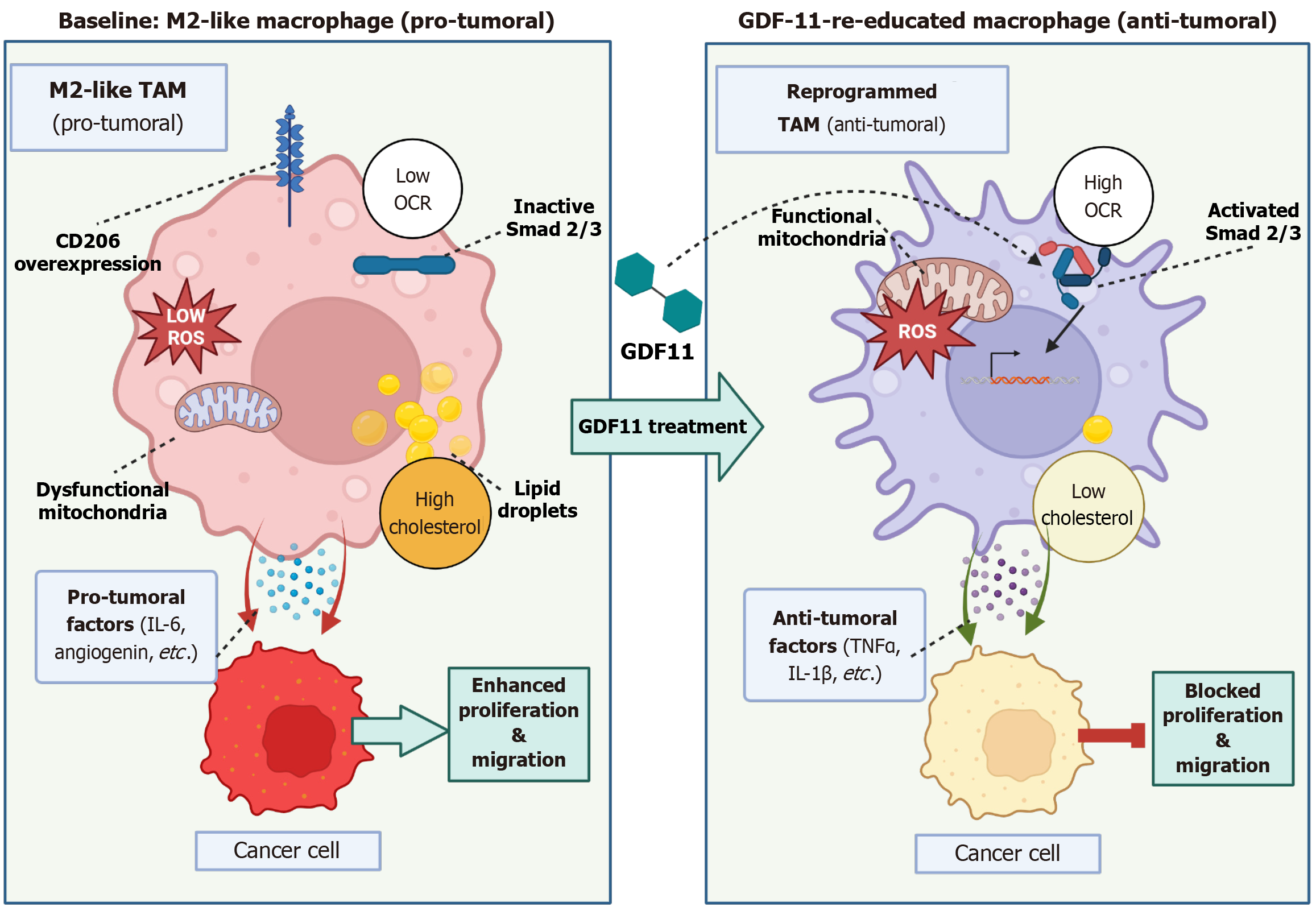
Figure 1 Growth differentiation factor 11-mediated immunometabolic reprogramming drives macrophage re-education and suppresses tumor aggressiveness.
This figure is a mechanistic diagram showing how growth differentiation factor 11 (GDF11) treatment flips pro-tumoral macrophages into an anti-tumoral state, based on findings primarily in the hepatocellular carcinoma context. The baseline (left) shows an M2-like tumor-associated macrophage (pro-tumoral) characterized by small, dysfunctional mitochondria (low oxygen consumption rate), high cholesterol (lipid droplets), and high cluster of differentiation (CD) 206. It secretes pro-tumoral factors [interleukin (IL)-6, angiogenin] that promote cancer cell proliferation and migration. GDF11 treatment (center) leads to the GDF11-re-educated macrophage (anti-tumoral) on the right. Signaling through Smad2/3 triggers immunometabolic reprogramming, resulting in large, functional mitochondria (high oxygen consumption rate), reduced cholesterol, loss of CD206, and high reactive oxygen species production. This re-educated macrophage secretes anti-tumoral factors (tumor necrosis factor-alpha, IL-1beta), which effectively inhibit cancer cell proliferation and migration. TAM: Tumor-associated macrophage; CD: Cluster of differentiation; GDF11: Growth differentiation factor 11; OCR: Oxygen consumption rate; ROS: Reactive oxygen species; IL: Interleukin; TNF: Tumor necrosis factor.
- Citation: Mohammadi S, Darweesh M, Al-Harrasi A. Growth differentiation factor 11 reprograms M2-like macrophages: Targeting immunometabolism for cancer therapy. World J Gastroenterol 2026; 32(10): 115371
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/115371.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.115371