©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 14, 2026; 32(10): 113771
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.113771
Published online Mar 14, 2026. doi: 10.3748/wjg.v32.i10.113771
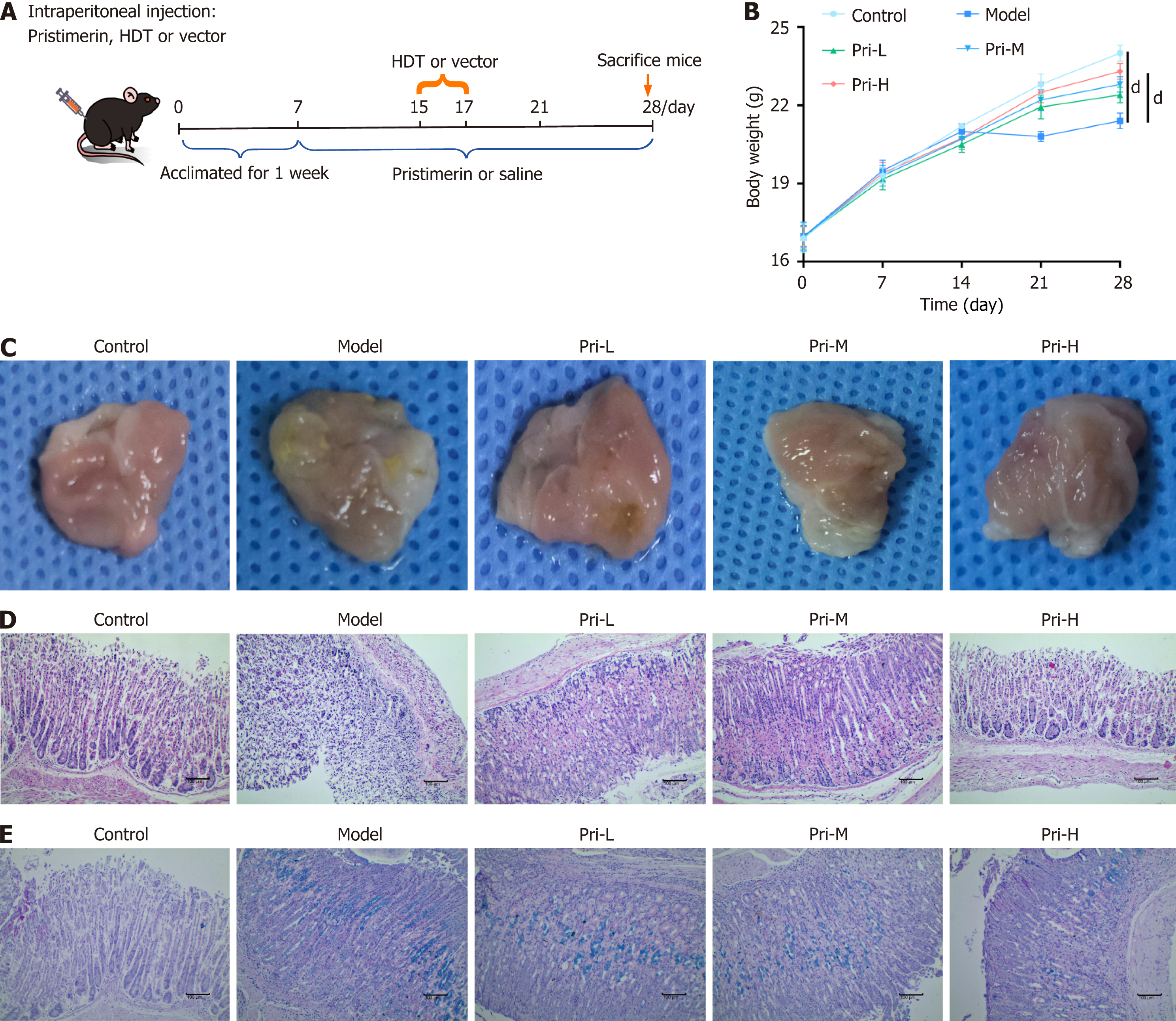
Figure 1 Pristimerin alleviates high-dose tamoxifen-induced gastric mucosal pathological damage in spasmolytic polypeptide-exp
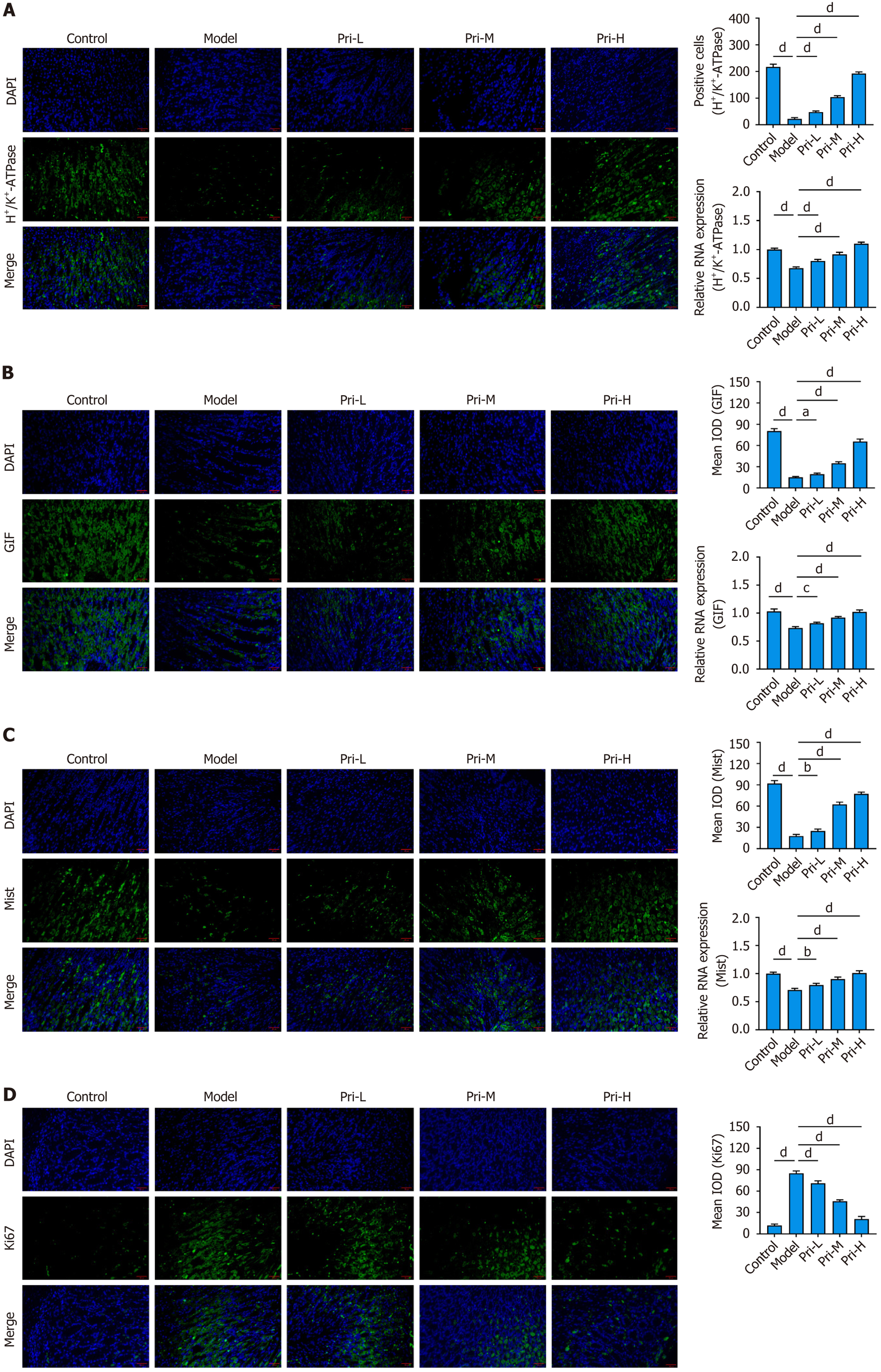
Figure 2 Pristimerin alleviates spasmolytic polypeptide-expressing metaplasia-related oxyntic atrophy in mice.
A-C: Immunofluorescence and reverse transcription quantitative polymerase chain reaction detection of hydrogen-potassium-adenosine-triphosphatase (parietal cells), gastric intrinsic factor and mist (chief cells) expression in gastric mucosa of high-dose tamoxifen-induced spasmolytic polypeptide-expressing metaplasia mice treated with pristimerin. Scale bar, 20 μm; D: Inhibitory effect of pristimerin on gastric mucosal cell proliferation in mice. Scale bar, 20 μm. aP < 0.05; bP < 0.01; cP < 0.001; dP < 0.0001. Values represent the mean ± SD (n = 6). DAPI: 4’,6-diamidino-2-phenylindole; H+/K+ ATPase: Hydrogen-potassium-adenosine-triphosphatase; GIF: Gastric intrinsic factor; Pri-L: Low-dose pristimerin; Pri-M: Medium-dose pristimerin; Pri-H: High-dose pristimerin; IOD: Integrated optical density.
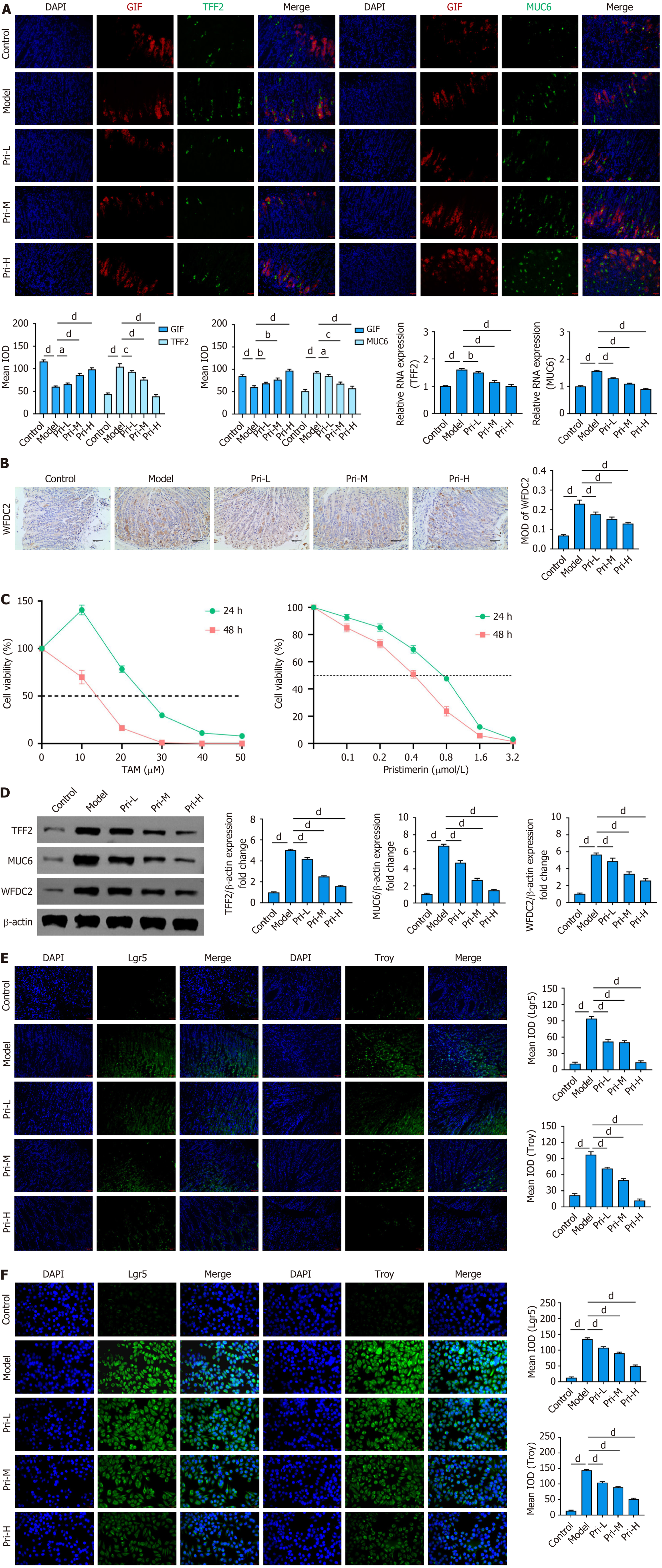
Figure 3 Inhibitory effect of pristimerin on aberrantly expressed spasmolytic polypeptide-expressing metaplasia-specific markers and stem cell markers.
A: Immunofluorescence co-localization analysis of chief cell marker gastric intrinsic factor with spasmolytic polypeptide-expressing metaplasia (SPEM)-specific markers trefoil factor 2 (TFF2)/mucin 6 (MUC6) in mouse gastric tissues, and detection of TFF2/MUC6 expression by reverse transcription qua
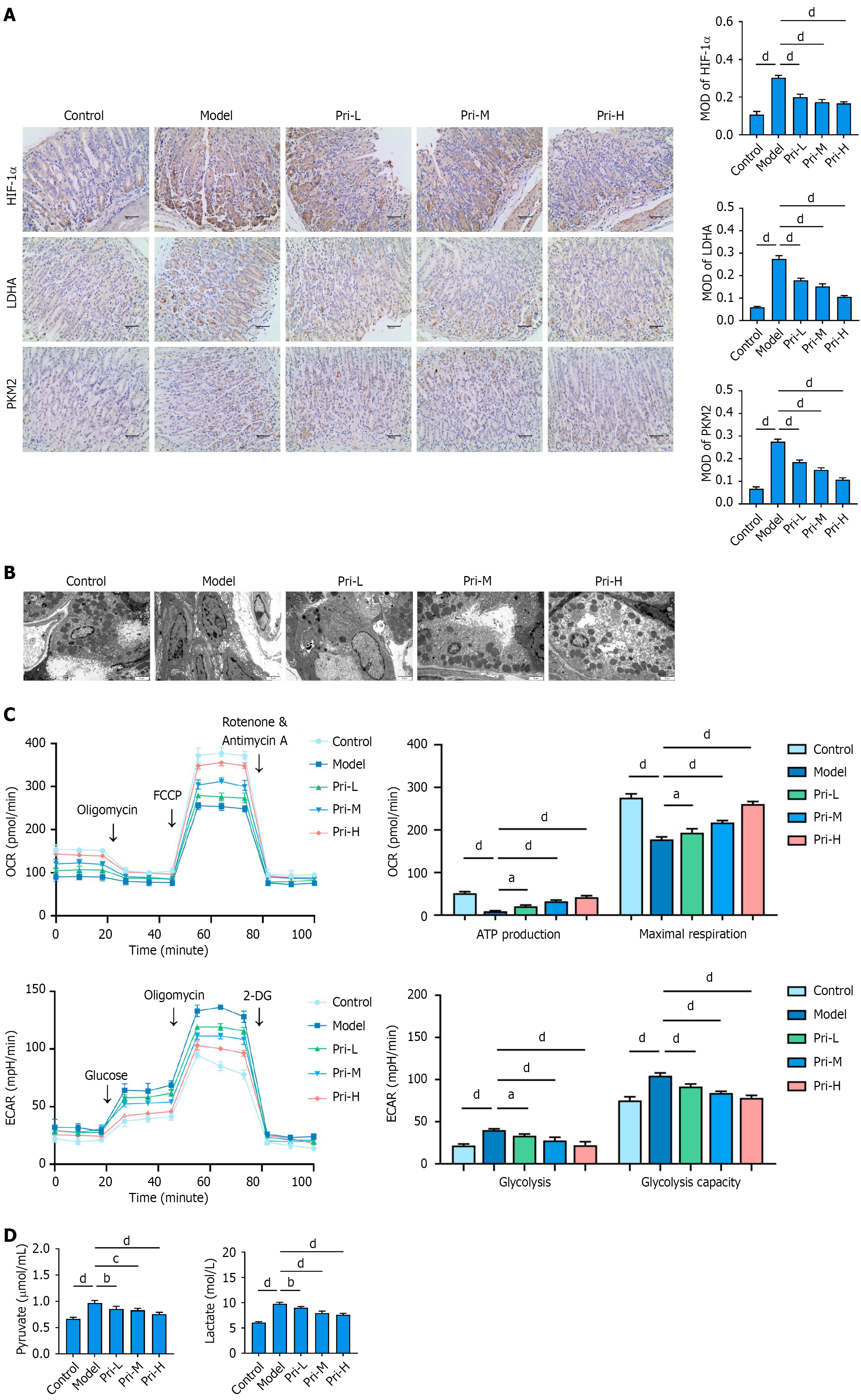
Figure 4 Pristimerin regulates glycolytic reprogramming in gastric mucosal cells.
A: Inhibitory effects of pristimerin on key glycolytic regulators (hypoxia-inducible factor-1α, lactate dehydrogenase A, pyruvate kinase M2) in the gastric mucosa of mice. Scale bar, 100 μm; B: Representative transmission electron microscopy images revealing ultrastructural features in mouse gastric mucosal cells. Scale bar, 2 μm; C: Analysis of cellular energy metabolism by Seahorse XF technology in human gastric epithelial (GES-1) cells following treatment with pristimerin; D: Quantitative analysis of pyruvate and lactate production in GES-1 cells. aP < 0.05. bP < 0.01. cP < 0.001. dP < 0.0001. Values represent the mean ± SD (A: n = 6; C and D: n = 5). HIF-1α: Hypoxia-inducible factor-1α; LDHA: Lactate dehydrogenase A; PKM2: Pyruvate kinase M2; Pri-L: Low-dose pristimerin; Pri-M: Medium-dose pristimerin; Pri-H: High-dose pristimerin; MOD: Mean optical density; FCCP: Trifluoromethoxy carbonylcyanide phenylhydrazone; 2-DG: 2-deoxy-D-glucose; OCR: Oxygen consumption rate; ECAR: Extracellular acidification rate.
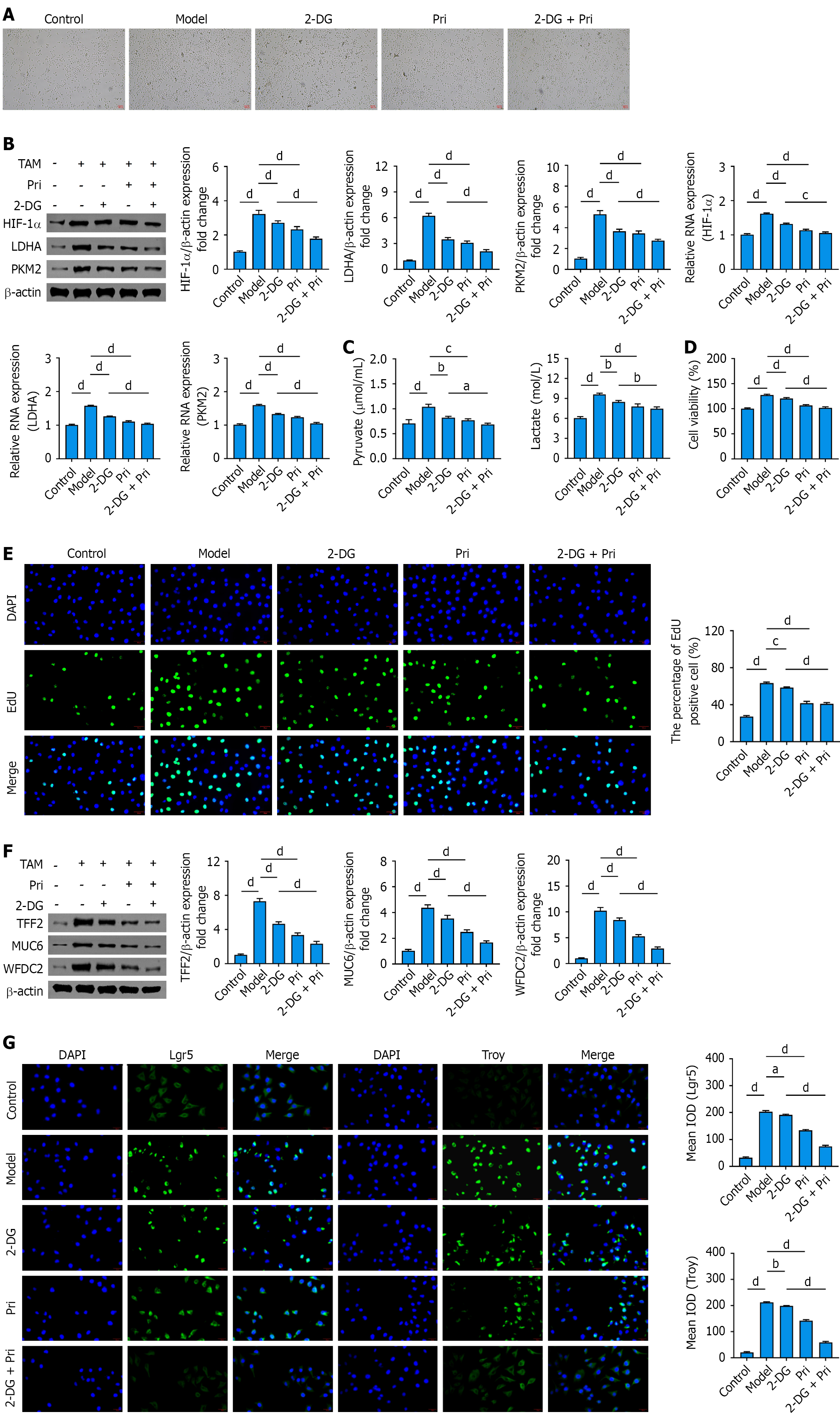
Figure 5 Pristimerin ameliorates tamoxifen-induced spasmolytic polypeptide-expressing metaplasia phenotype in human gastric epithelial cells via suppression of glycolysis.
A: Representative images of tamoxifen-induced human gastric epithelial (GES-1) cells following pristimerin or 2-deoxy-D-glucose (2-DG) intervention. Scale bar, 100 μm; B: Expression of key glycolytic regulators in GES-1 cells following pristimerin or 2-DG intervention; C: Quantitative analysis of pyruvic acid and lactate production in GES-1 cells; D: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay for GES-1 cell viability; E: 5-Ethynyl-2’-deoxyuridine assay for detecting proliferation activity in GES-1 cells. Scale bar, 20 μm; F: Inhibitory effects of pristimerin or 2-DG on spasmolytic polypeptide-expressing metaplasia-specific marker expression in GES-1 cells (Western blot; β-actin-normalized relative expression vs control); G: Expression of stem cell markers in GES-1 cells following pristimerin or 2-DG treatment. Scale bar, 20 μm. aP < 0.05. bP < 0.01. cP < 0.001. dP < 0.0001. Values represent the mean ± SD (n = 5). 2-DG: 2-deoxy-D-glucose; Pri: Pristimerin; HIF-1α: Hypoxia-inducible factor-1α; LDHA: Lactate dehydrogenase A; PKM2: Pyruvate kinase M2; TAM: Tamoxifen; DAPI: 4’,6-diamidino-2-phenylindole; EdU: 5-Ethynyl-2’-deoxyuridine; MUC6: Mucin 6; TFF2: Trefoil factor 2; WFDC2: Whey-acidic-protein four-disulfide core domain protein 2; Lgr5: Leucine-rich repeat-containing G-protein coupled receptor 5; Troy: Tumor necrosis factor receptor superfamily member 19; IOD: Integrated optical density.
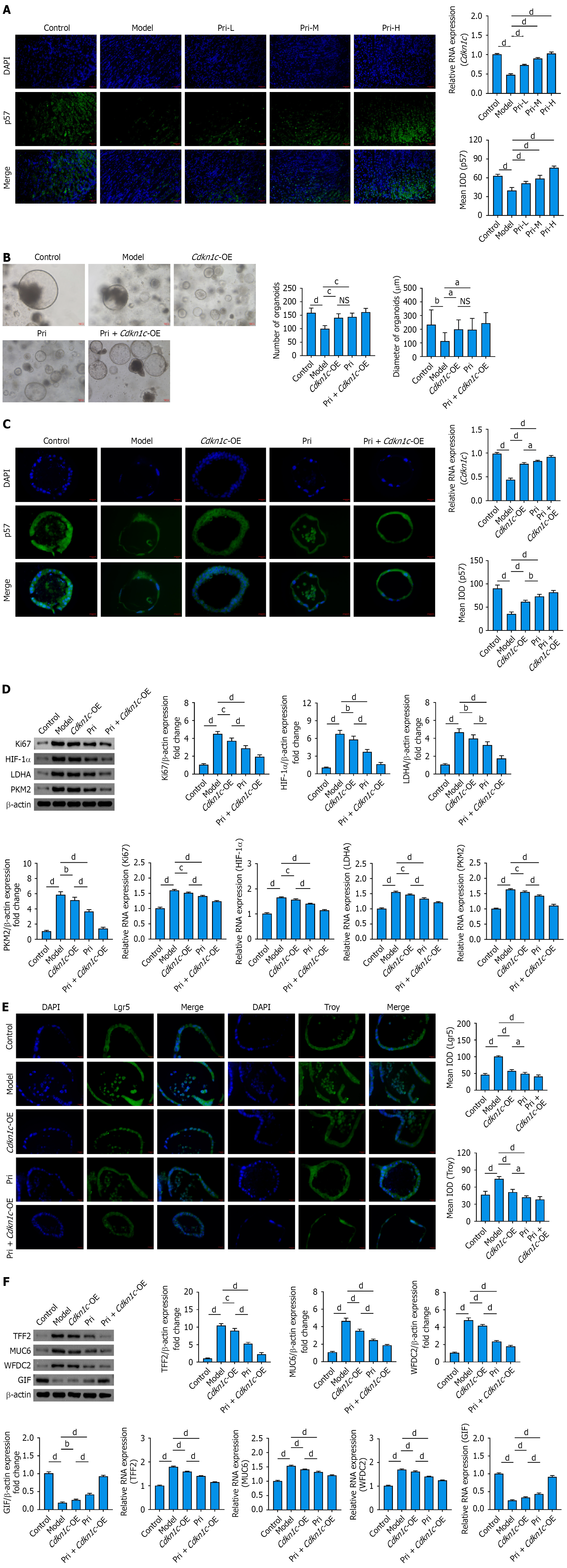
Figure 6 Pristimerin upregulates p57 to modulate glycolytic reprogramming and ameliorate the spasmolytic polypeptide-expressing metaplasia phenotype in gastric organoids.
A: Pristimerin upregulates gastric mucosal p57 in spasmolytic polypeptide-expressing metaplasia (SPEM) mice. Scale bar, 20 μm; B: Representative images of normal gastric organoids, N-methyl-N’-nitro-N-nitrosoguanidine/Helicobacter pylori-induced SPEM gastric organoids, and those following Cdkn1c overexpression or pristimerin intervention, along with quantitative analyses of organoid number and diameter. Scale bar, 100 μm; C: P57 expression in SPEM gastric organoids following Cdkn1c overexpression or pristimerin treatment. Scale bar, 10 μm; D-F: Both Cdkn1c overexpression and pristimerin treatment suppressed cell proliferation [reverse transcription quantitative polymerase chain reaction (RT-qPCR); Western blot, β-actin-normalized relative expression vs control] in SPEM gastric organoids, downregulated key glycolytic regulators (RT-qPCR; Western blot), stem cell markers (immunofluorescence: Scale bar, 10 μm), and SPEM-specific markers (RT-qPCR; Western blot), while promoting gastric intrinsic factor (RT-qPCR; Western blot) expression with pristimerin exhibiting stronger effects. aP < 0.05. bP < 0.01. cP < 0.001. dP < 0.0001. Values represent the mean ± SD (A: n = 6; B-F: n = 5). NS: Not significant; Cdkn1c-OE: Cdkn1c-over
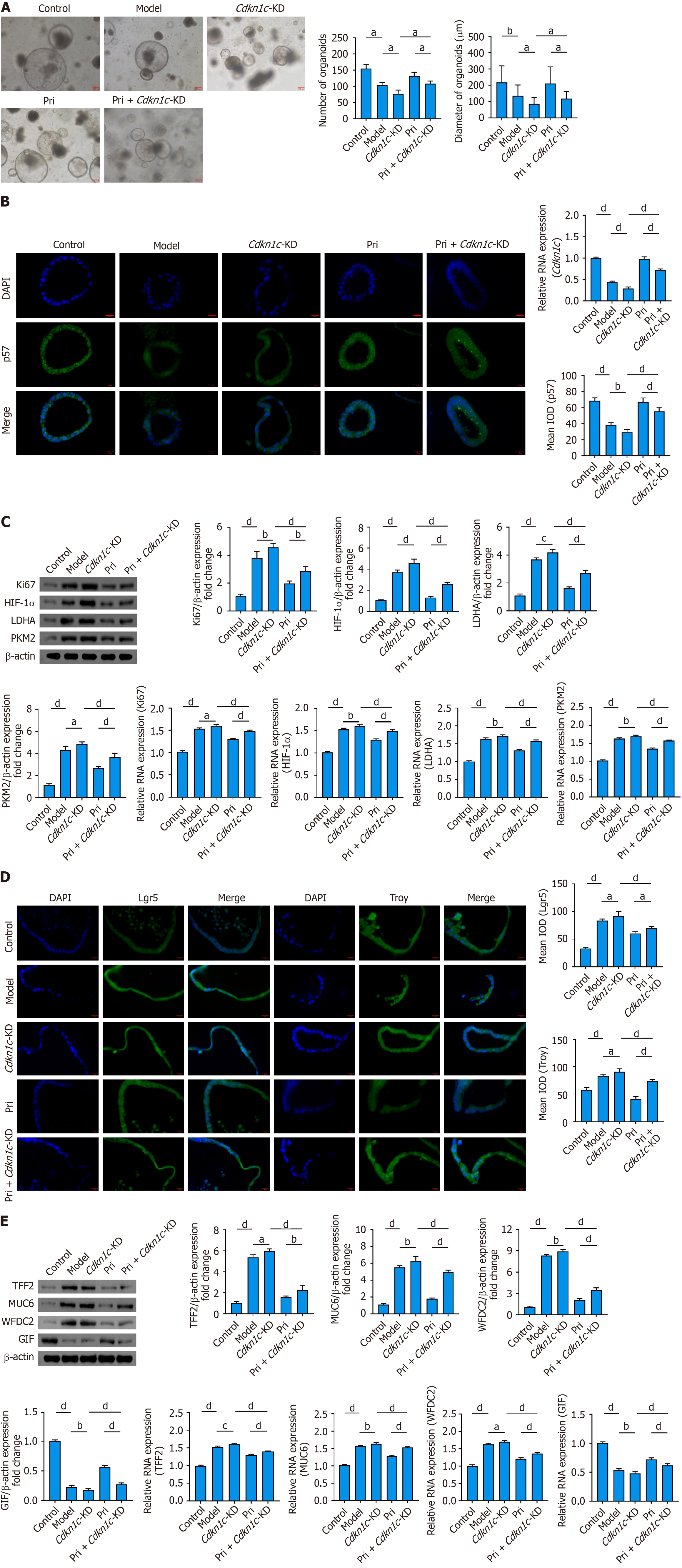
Figure 7 Cdkn1c knockdown attenuates pristimerin’s regulation of glycolytic reprogramming and amelioration of spasmolytic polype
- Citation: Wen JS, Pan ZW, Yao XD, Liu YQ, Zhu YD. Pristimerin ameliorates spasmolytic polypeptide-expressing metaplasia by modulating Cdkn1c (p57)-mediated glycolytic reprogramming. World J Gastroenterol 2026; 32(10): 113771
- URL: https://www.wjgnet.com/1007-9327/full/v32/i10/113771.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i10.113771