©The Author(s) 2020.
World J Gastroenterol. Aug 21, 2020; 26(31): 4589-4606
Published online Aug 21, 2020. doi: 10.3748/wjg.v26.i31.4589
Published online Aug 21, 2020. doi: 10.3748/wjg.v26.i31.4589
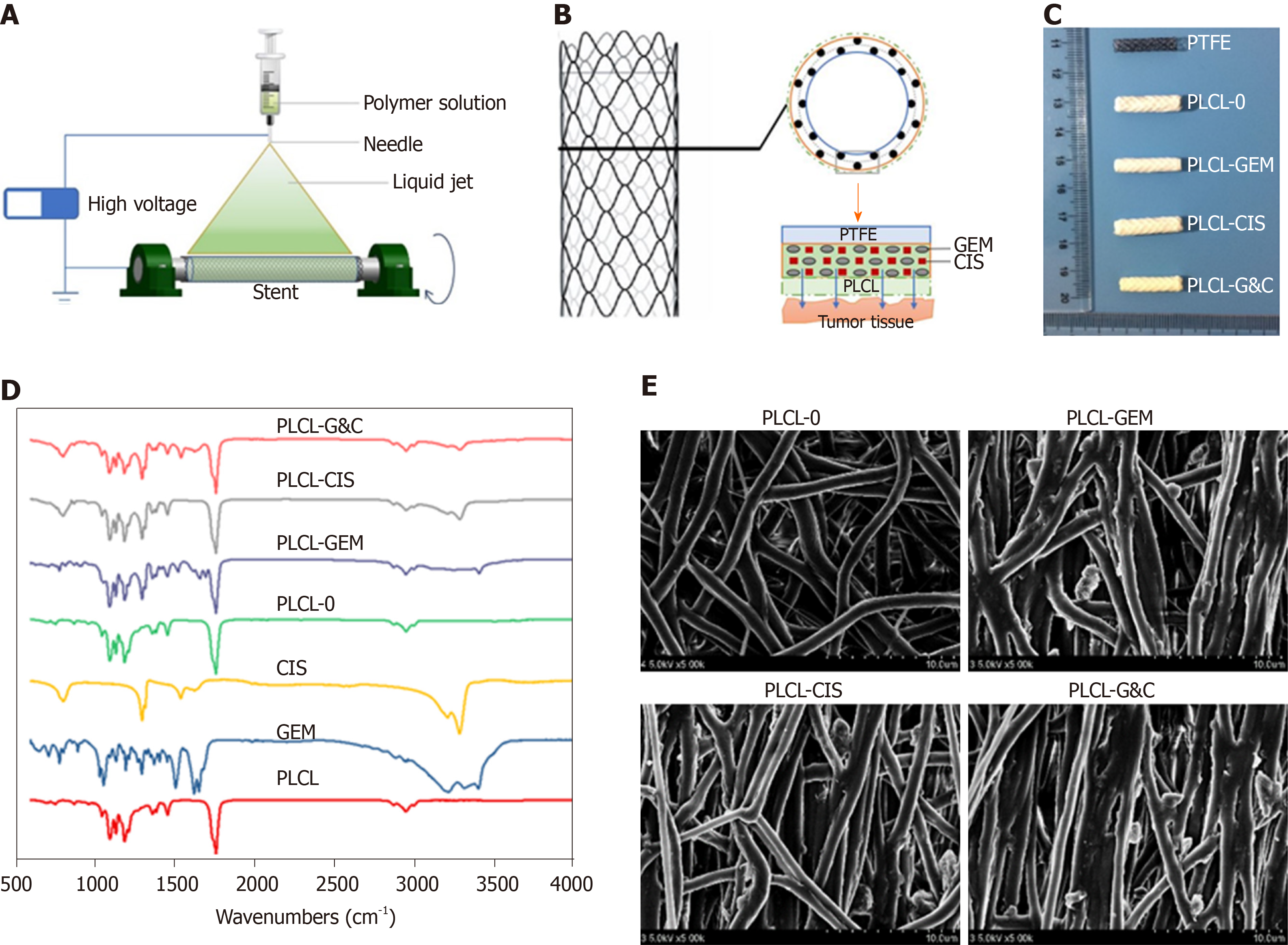
Figure 1 Manufacturing and characteristics of different biliary drug-eluting stents.
A: Schematic illustration of the manufacturing of drug-eluting stents by the mixed electrospinning method; B: Optimized design of the gemcitabine/cisplatin-eluting biliary stent. The entire structure of the coated membranes consists of three layers: the inner polytetrafluoroethylene membrane (as a blocker to avoid drug loss), the middle PLCL layer (as the major drug carrier), and the outer PLCL-unloaded membrane (as a blocker to prevent burst release); C: Image of different types of drug-eluting stents; D: Fourier transform infrared spectra of different prototype drugs and drug-eluting nanofilms; E: Scanning electron microscope images of different drug-eluting nanofilms (magnification ×2000). CIS: Cisplatin; GEM: Gemcitabine; PLCL: Poly-L-lactide-caprolactone; PLCL-0: Non-drug-loaded PLCL nanofilm; PLCL-CIS: PLCL nanofilm loaded with CIS; PLCL-GEM: PLCL nanofilm loaded with GEM; PLCL-GC: PLCL nanofilm loaded with both GEM and CIS; PTFE: Polytetrafluoroethylene.
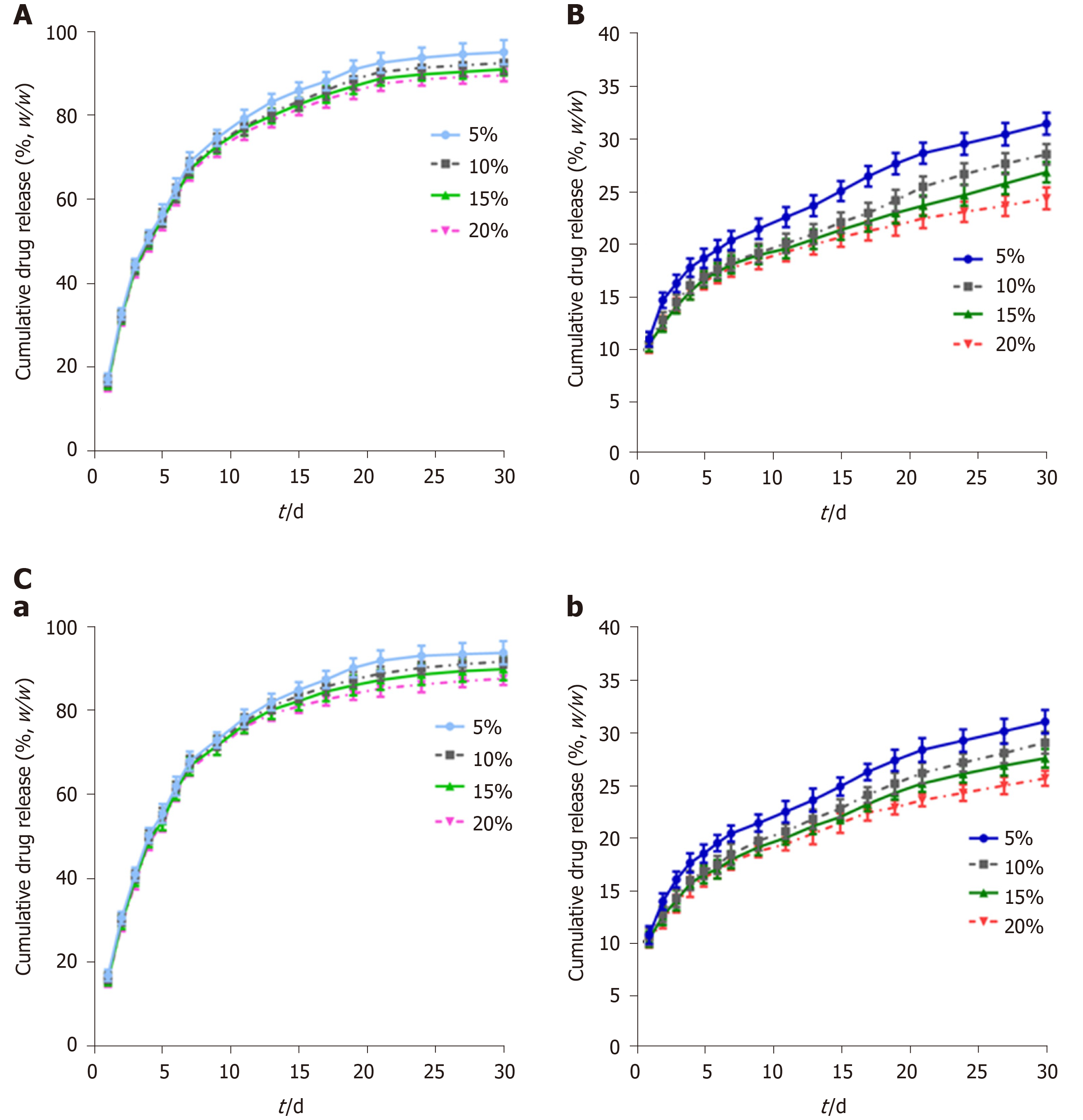
Figure 2 Cumulative drug release of different biliary drug-eluting stents with different drug-loading ratios.
A: Release curves of gemcitabine from PLCL-GEM-eluting stents; B: Release curves of cisplatin from PLCL-CIS-eluting stents; C: Release curves of (a) gemcitabine and (b) cisplatin from PLCL-GEM and CIS-eluting stents. CIS: Cisplatin; GEM: Gemcitabine; PLCL: Poly-L-lactide-caprolactone.
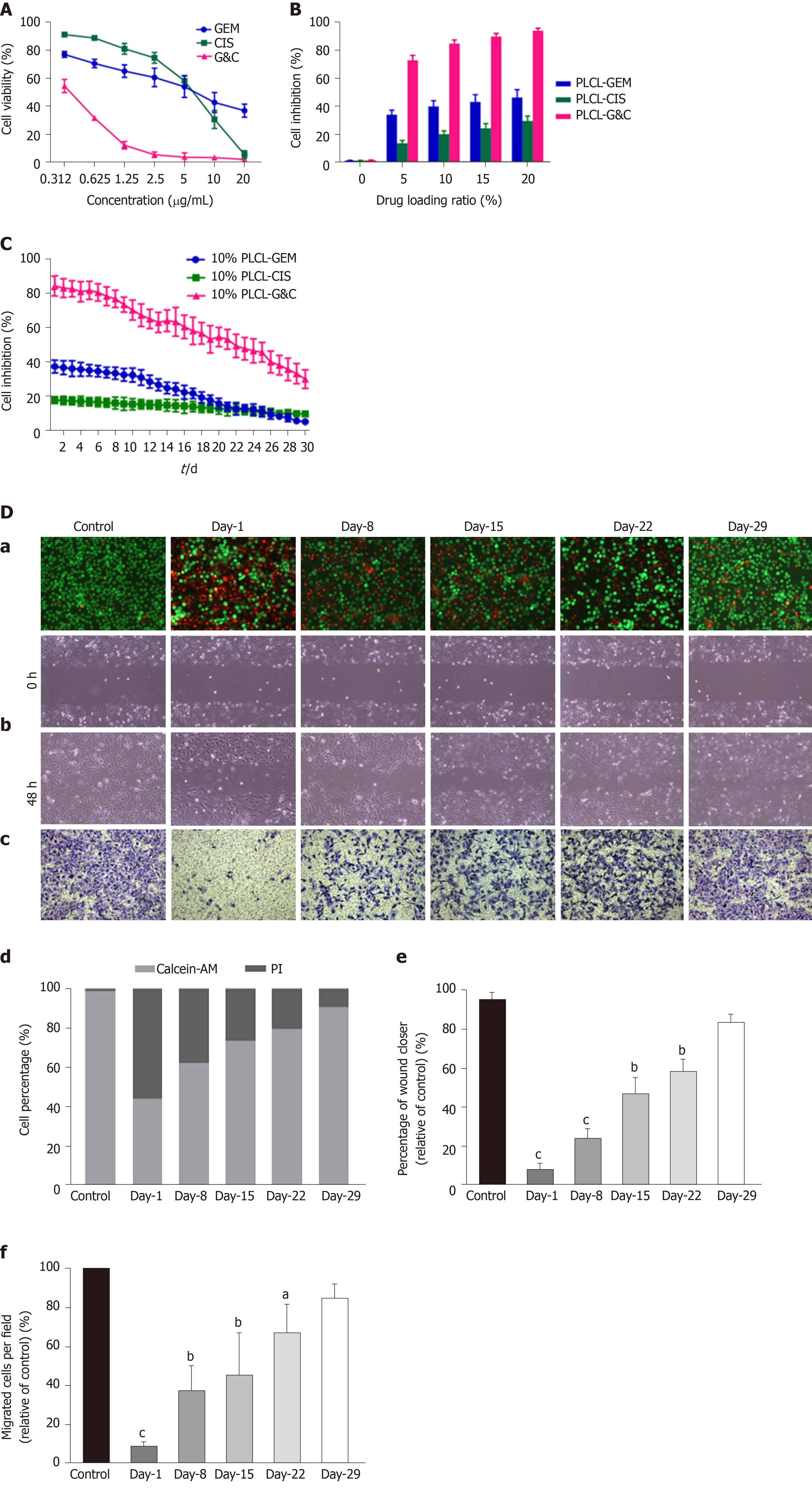
Figure 3 Antitumor activity of different drug-loaded nanofilms against human cholangiocarcinoma cells in vitro.
A: Effects of different concentrations of prototype gemcitabine and cisplatin on the growth of EGI-1 cells; B: Inhibition of EGI-1 cells caused by different drug-loaded nanofilms with different drug-loading ratios for 30 d; C: Inhibition of EGI-1 cells caused by drug-released media from 10% drug-loaded nanofilms every other week for 30 d; D: Effects of 10% drug-loaded nanofilms on the (a) viability (magnification × 100); (b) migration (×100); (c) and invasion (×200) of EGI-1 cells; (d) statistical analysis of the proportion of living and dead cells coincubated with different drug-released media; (e) quantitative analysis of the wound closer area following coincubation with different drug-released media; and (f) quantification of migrated cells coincubated with different drug-released media. aP < 0.05, bP < 0.01, cP < 0.001 vs control (n = 6 mice per group). CIS: Cisplatin; GEM: Gemcitabine; PI: Propidium iodide; PLCL: Poly- L-lactide-caprolactone; PLCL-CIS: PLCL nanofilm loaded with CIS; PLCL-GEM: PLCL nanofilm loaded with GEM; PLCL-GC: PLCL nanofilm loaded with both GEM and CIS.
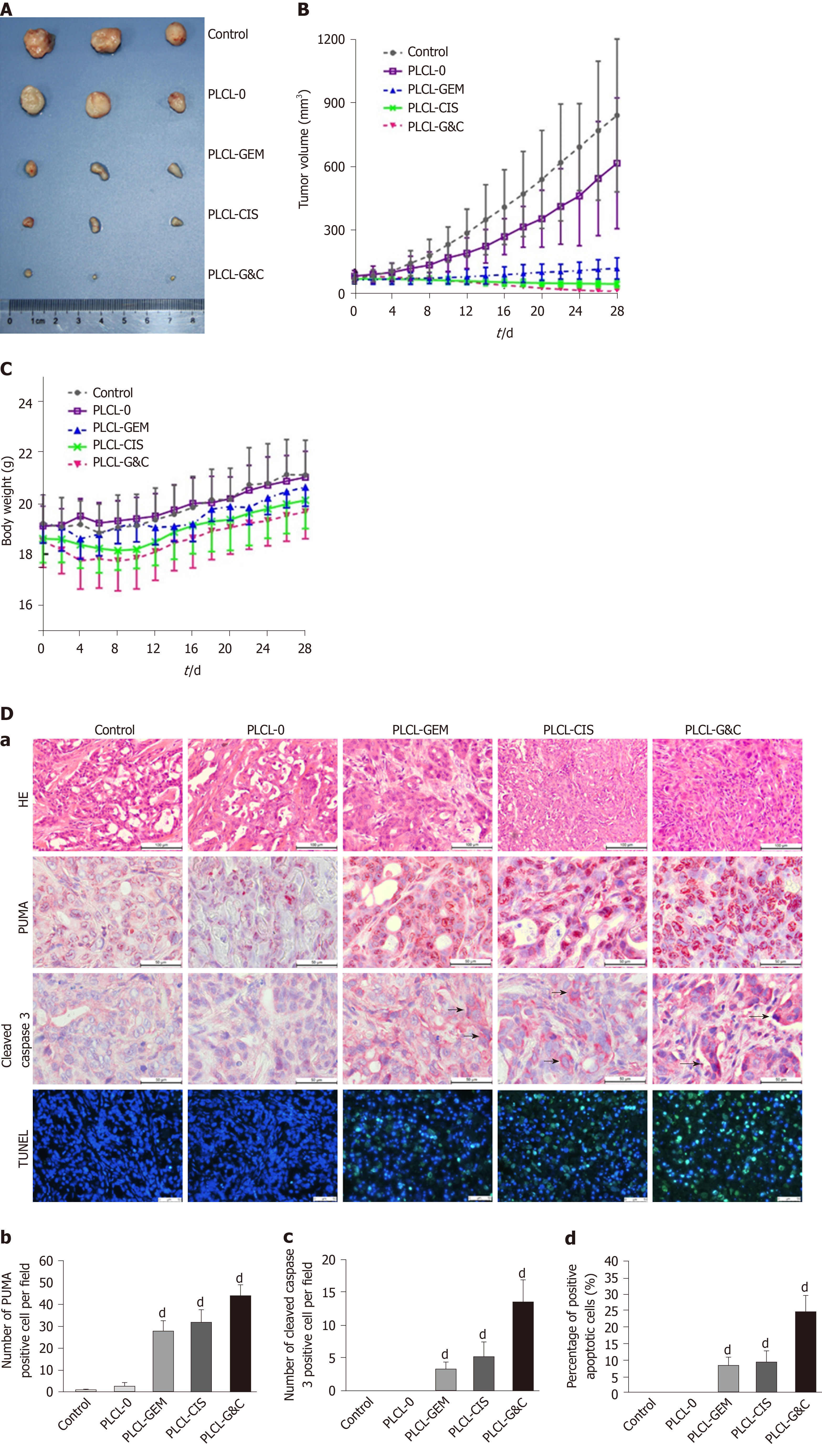
Figure 4 Antitumor activity of drug-loaded nanofilms against subcutaneous tumor xenografts in nude mice in vivo.
A: Image of subcutaneous tumors from nude mice after drug-loaded nanofilm implantation for 4 wk; B: Changes in tumor volume; C: Changes in body weight; and D: (a) HE staining (magnification × 200, scale bar 100 µm), immunohistochemical analysis of p53 upregulated modulator of apoptosis and cleaved caspase-3 (×400, scale bar 50 µm), and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling staining (× 200, scale bar 50 µm) in subcutaneous tumor tissues; (b-d) Statistical analysis results: Quantification of (b) p53 upregulated modulator of apoptosis-positive cell numbers, (c) cleaved caspase-3-positive cell numbers, and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling-positive cell proportion in subcutaneous tumor tissues. dP < 0.001 vs control (n = 6 mice per group). CIS: Cisplatin; GEM: Gemcitabine; HE: Hematoxylin and eosin; PLCL: Poly L-lactide-caprolactone; PLCL-0: Non-drug-loaded PLCL nanofilm; PLCL-CIS: PLCL nanofilm loaded with CIS; PLCL-GEM: PLCL nanofilm loaded with GEM; PLCL-GC: PLCL nanofilm loaded with both GEM and CIS; PUMA: p53 upregulated modulator of apoptosis; TUNEL: Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling.
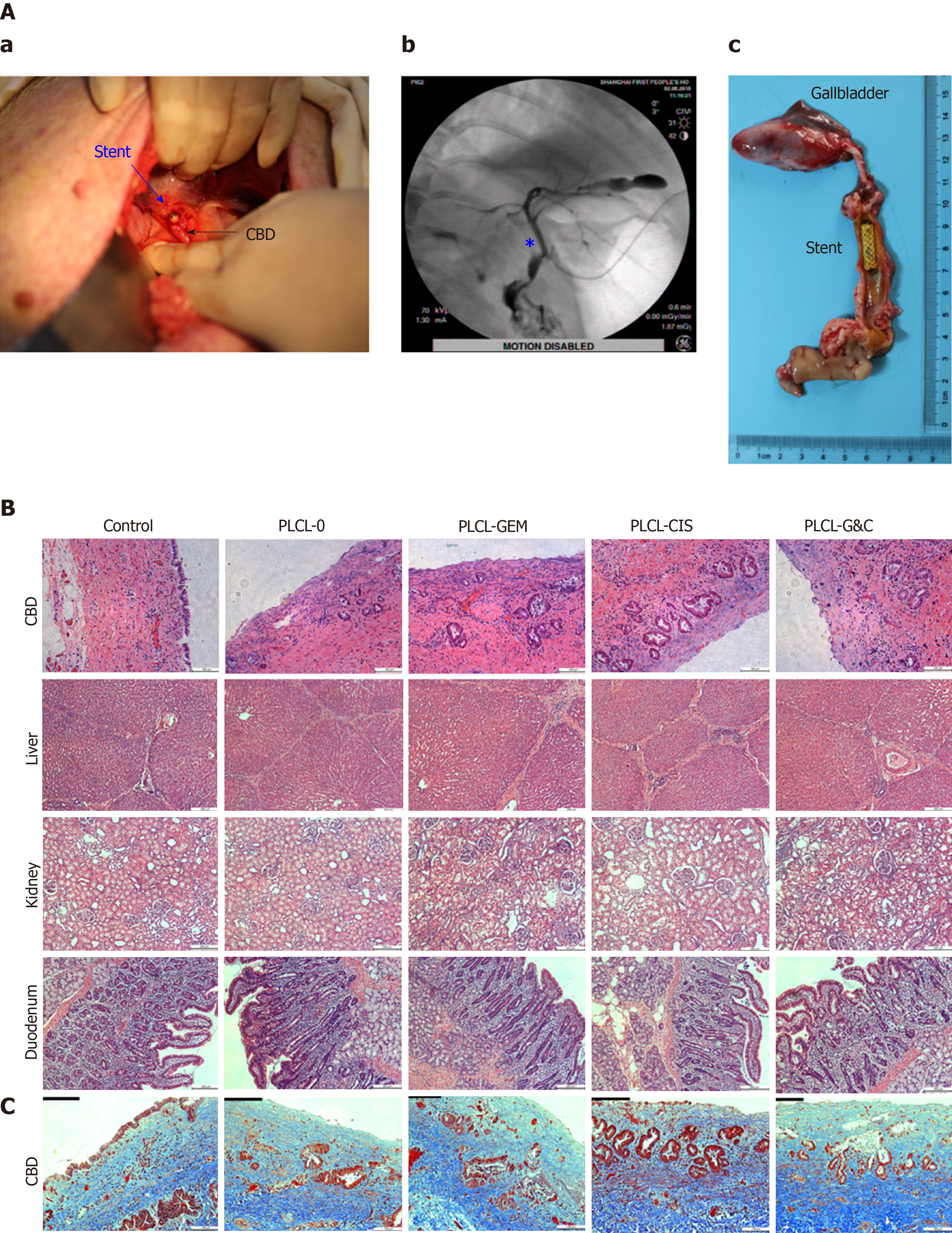
Figure 5 Safety evaluation of different drug-eluting biliary stents in a porcine model.
A: (a) Critical surgical step necessary to place a stent into the porcine CBD; the stent was sutured on the CBD wall carefully when closing the CBD to avoid stent migration; (b) cholangiogram after stent placement; the pentacle marks the location of the stent; (c) macroscopic appearance of the CBD after animals were sacrificed; all the stents were still in the CBD and had not migrated; B: HE staining of the CBD (magnification ×200, scale bar 100 µm), liver, kidney, and duodenum (× 100, scale bar 200 µm) 30 d after placement of different drug-eluting biliary stents; C: Masson trichrome staining of the CBD 30 d after placement of different drug-eluting biliary stents (× 200, scale bar 100 µm). CBD: Common bile duct; CIS: Cisplatin; GEM: Gemcitabine; PLCL: Poly-L-lactide-caprolactone; PLCL-0: Non-drug-loaded PLCL nanofilm; PLCL-CIS: PLCL nanofilm loaded with CIS; PLCL-GEM: PLCL nanofilm loaded with GEM; PLCL-GC: PLCL nanofilm loaded with both GEM and CIS.
- Citation: Xiao JB, Weng JY, Hu YY, Deng GL, Wan XJ. Feasibility and efficacy evaluation of metallic biliary stents eluting gemcitabine and cisplatin for extrahepatic cholangiocarcinoma. World J Gastroenterol 2020; 26(31): 4589-4606
- URL: https://www.wjgnet.com/1007-9327/full/v26/i31/4589.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i31.4589