Published online Mar 18, 2026. doi: 10.13105/wjma.v14.i1.112793
Revised: September 26, 2025
Accepted: December 26, 2025
Published online: March 18, 2026
Processing time: 216 Days and 16.2 Hours
Dexmedetomidine (DEX) is a drug that is specifically an agonist for the alpha-2 adrenergic receptor, which has been explored for potential use in the provision of anesthesia during flexible bronchoscopy. Tactical mechanisms enable DEX to be able to provide sedation without marked respiratory depression hence aiding patients with poor respiratory reserve.
To investigate the safety and efficacy of DEX by examining heart rate, blood pre
A meta-analysis following Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines was conducted across PubMed, Scopus, and the Coch
Fourteen randomized controlled trials involving 1920 patients were finalized. Patients using DEX had a higher mean oxygen saturation throughout the procedure (P < 0.0003, I² = 18%) and a decreased mean heart rate (P < 0.00001, I² = 22%). Additionally, DEX significantly increased the incidence of bradycardia (P = 0.00001, I² = 53%). The results show no difference in discomfort experienced by the patients when compared to the control groups.
Results show DEX to have a significant risk of bradycardia and hypotension. The effect on patient satisfaction was indeterminate; larger trials are required to confirm long-term safety and efficacy.
Core Tip: This meta-analysis assessed the effectiveness of dexmedetomidine (DEX), a selective alpha-2 adrenergic receptor agonist, as a sedative agent in flexible bronchoscopy. Compared with other sedative and analgesic regimens, DEX notably reduced the risk of hypoxemia and tachycardia, while mildly improving patient and physician satisfaction. Although extra sedation was administered where necessary, the overall safety profiles were favorable. These findings suggest that DEX is a promising option for patients with compromised respiratory capacity, providing enhanced safety and tolerability during bronchoscopy.
- Citation: Hassni A, Siddiqui AH, Shahid M, Shariq A, Anwar H, Nadeem K, Anjum A, Memon H, Talha A, Khan S, Faisal Y, Naveed M, Moeed A, Surani S. Safety and efficacy of dexmedetomidine for flexible bronchoscopy: A systematic review and meta-analysis. World J Meta-Anal 2026; 14(1): 112793
- URL: https://www.wjgnet.com/2308-3840/full/v14/i1/112793.htm
- DOI: https://dx.doi.org/10.13105/wjma.v14.i1.112793
Bronchoscopy is a crucial technique for the management and detection of numerous respiratory disorders[1]. It enables inspection of the airway’s lumen and mucosa, often utilized for retrieving specimens to biopsy centrally situated tumors, examining lung disorders, and localizing the origin of hemoptysis[2]. Potential complications during this procedure include bleeding, pneumothorax, respiratory infections and adverse reactions to sedation. The main purpose of sedation use is to minimize the patient’s discomfort and anxiety and make things less complicated to accomplish the clinical goals[3,4].
Dexmedetomidine (DEX) and midazolam are two frequently used sedatives, both of which have distinct mechanisms of action and sedation profiles as well as having analgesic properties. In addition to comparing these two drugs, some other agents, including remimazolam, and opioids such as fentanyl, alfentanil, and remifentanil have also been evaluated in earlier studies on sedation during flexible bronchoscopy (FB). These agents were used in several of the randomized controlled trials (RCTs) in our analysis. While DEX and opioids provide both sedation and analgesia[5], benzodiazepines (including midazolam and remimazolam) and propofol primarily act as sedatives without significant analgesic effects[6]. So, the overall efficacy of anesthesia might be impacted by the pharmacological difference between sedative and sedative-analgesic agents, with agents that provide both properties may offer superior patient comfort compared to those with only sedative properties[7].
DEX is a highly selective alpha 2-adrenoceptor with diverse pharmacological effects throughout the body[8]. It is one of the most recently developed and commercialized agents that offers analgesia and conscious sedation without any respiratory depression. By decreasing the sympathetic CNS outflow, DEX poses opioid sparing effects along with organ protection. DEX induces hypnosis by activating alpha 2-adrenergic receptors in the locus ceruleus, lowering the cyclic AMP levels and hyperpolarizing the neurons, thereby inhibiting neuronal activity. It additionally reduces the nociceptor activity and modifies the pain pathway. Reduced release of norepinephrine from the locus ceruleus enhances the release of gamma-aminobutyric acid (GABA) and galanin, which further suppresses the locus ceruleus and tuberomamillary nucleus by lifting the inhibition of the ventrolateral preoptic nucleus. This results in reduced pain signals and leads to a hypnotic state like natural sleep[9].
Midazolam, on the other hand, is a benzodiazepine medication used in the emergency department, commonly for the induction of sedation before surgeries[10]. They are metabolized by cytochrome P450 enzymes and by glucuronide conjugation. The mode of action suggests ionotropic GABA-α mediated neuronal inhibition, which accounts for almost all the effects of midazolam which includes hypnosis, stress alleviation, tranquilization and centrally mediated muscle relaxation.
Propofol is another GABA-α receptor agonist that metabolizes completely into inactive products that are eliminated in the urine largely dependent on liver function[11]. It acts as a versatile agent in clinical anesthesia, exhibiting antipruritic, bronchodilatory, muscle relaxant and anti-epileptic properties[12]. Moreover, remimazolam is an ultra-fast-acting intravenous benzodiazepine used for anesthesia and sedation. It upregulates γ-aminobutyric acid receptors and is broken down by tissue esterases without compromising respiratory rates[13]. Fentanyl is a powerful opioid used during intraoperative and postoperative procedures due to its strong analgesic effects[14] though it is utilized less frequently these days[15]. Some studies indicate that propofol, when combined with opioid agents, reduces the occurrence of propofol-related pain to an extent[16]. Remifentanil is also a potent opioid with a brief half-life of 10 minutes[17]. Although administering alfentanil before propofol increases their synergistic effects and ensures peak alignment, it may also increase the risk of cardiopulmonary depression[18].
Several key differences emerge when comparing the safety profiles and sedative effects of DEX, midazolam and propofol during FB procedures. While these agents are clinically useful, they possess significant limitations. DEX might cause hypotension and bradycardia; benzodiazepines can lead to respiratory depression and amnesia and propofol carries a risk of hypotension. Considering the increasing application in the diagnosis and management of respiratory disorders, choosing the optimal sedative becomes crucial for the procedural effectiveness and safety of patients. In this meta-analysis, we aim to compare these drugs, with a primary focus on the sedative experience, adverse effects and other patient outcomes during bronchoscopy procedures.
This study was conducted in concordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses and the manuscript was prepared and revised according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 Checklist[19]. This meta-analysis has been registered on PROSPERO, No. CRD42024594120.
Literature was searched by two independent authors using the electronic databases PubMed, Scopus and Cochrane Library. The keywords were “dexmedetomidine”, “midazolam”, “remimazolam”, “propofol”, “remifentanil”, “ketamine” and “bronchoscopy”. No filters were used during the literature review. Articles were extracted from the database initialization to September 2025. No language restrictions were applied. A more detailed search strategy is given in Supplementary Table 1.
Screening of the titles and abstracts of the articles via a full-text review was done by two independent authors (Shariq A and Memon H) to ascertain their relevance, followed by a thorough full-text read. Later, discrepancies were resolved by a third person (Hassni A). Selected articles were verified against the predefined inclusion criteria after removing any duplicate studies.
Inclusion criteria: The predefined eligibility criteria for the studies included (1) Study design: Randomized control trials; (2) Population: Adults ( ≥ 18 years) who underwent FB; (3) Intervention: DEX alone or in conjunction with other agents; (4) Comparator: Midazolam, remimazolam, fentanyl, propofol or any other anesthetic drugs, either alone or in combination; and (5) Outcomes: Primary outcomes were mean heart rate, mean oxygen saturation, and the discomfort experienced during the procedure; secondary outcomes included Ramsay Sedation Score (RSS), cough, pain and distress, ease of bronchoscopy, quality of sedation, composite scores, and adverse effects.
Exclusion criteria: The studies were excluded if they were non-randomized control trials, observational studies, conference abstracts, letters, case reports, and studies with incomplete data or irrelevant outcomes or that did not administer DEX were excluded.
Study and baseline data extraction were done by two authors (Hassni A and Memon H) who read the articles and organized information onto an Excel sheet. This was verified by one author (Siddiqui AH) who concluded disagreements. Data extracted was organized according to author name, year of publication, study design, population size, DEX dosage, control drug, outcomes of each study, follow up duration, general patient characteristics (gender, height, age, weight and body mass index), duration of bronchoscopy, bronchoscopy types, route of bronchoscopy, baseline heart rate, baseline mean systolic and diastolic pressure and baseline mean saturation of oxygen in blood. Primary and secondary outcomes data was extracted from relevant trials onto a separate sheet. Data was interconverted from medians to means as required using the Wans method. All the data taken were from previously published articles, so no new patient recruitment or consent was required.
The primary outcomes of interest were mean heart rate and mean oxygen saturation of patients reported by trials at regular intervals of 5 minutes, 10 minutes, at the end of the procedure and 10 minutes after the end of the procedure; and the discomfort experienced by patients during the procedure. Discomfort was reported by trials using a point system of 1 (being the lowest) to 10 (being the greatest) and classified as none, mild, moderate or severe discomfort.
The secondary outcomes included from shortlisted trials were (1) Ramsay Sedation Scale (RSS) score ranging from 1 (least sedation) to 6 (deepest sedation); (2) Cough [reported by studies using the visual analogue scale from 0 (least severe) to 10 (most severe)]; (3) Pain/distress (minimal, mild, or maximal); (4) Ease of bronchoscopy (easy, slightly difficult, or very difficult); (5) Composite scores at trachea and nasopharynx (reported by studies as ideal, acceptable, or unacceptable); (6) Willingness of patients to repeat procedure; (7) Quality of sedation (excellent, good, fair, poor); (8) Need of a rescue dose; and (9) Changes in systolic and diastolic blood pressure (mmHg).
Cochrane risk of bias 2 was used to assess and authenticate the quality of the included RCTs[20] by one author independently (Talha A) verified by another author (Anwar H) in case of doubt. The procedure is based on the risk of the study over the domains of (1) Randomization process; (2) Deviation from the intended intervention; (3) Missing outcome data; (4) Measurement of the outcome; and (5) Selection of the reported results.
Review Manager version 5.4.1 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration 2020) was used to evaluate the studies included. The results were assessed as risk ratio (RR) or mean differences (MD) with a 95%CI. All the outcomes were analyzed via the random effects model to cater to the diversity of articles included. The significance of the results obtained was authenticated via a forest plot. P value lower than 0.05 is taken as significant throughout. Heterogeneity, for each respective outcome was assessed using the Higgins I² statistics. A value of I² = 25%-50% is taken as mild, I² = 50%-75% is moderate and I² > 75% is considered significant heterogeneity. A sensitivity analysis was conducted for all the results that presented significant heterogeneity.
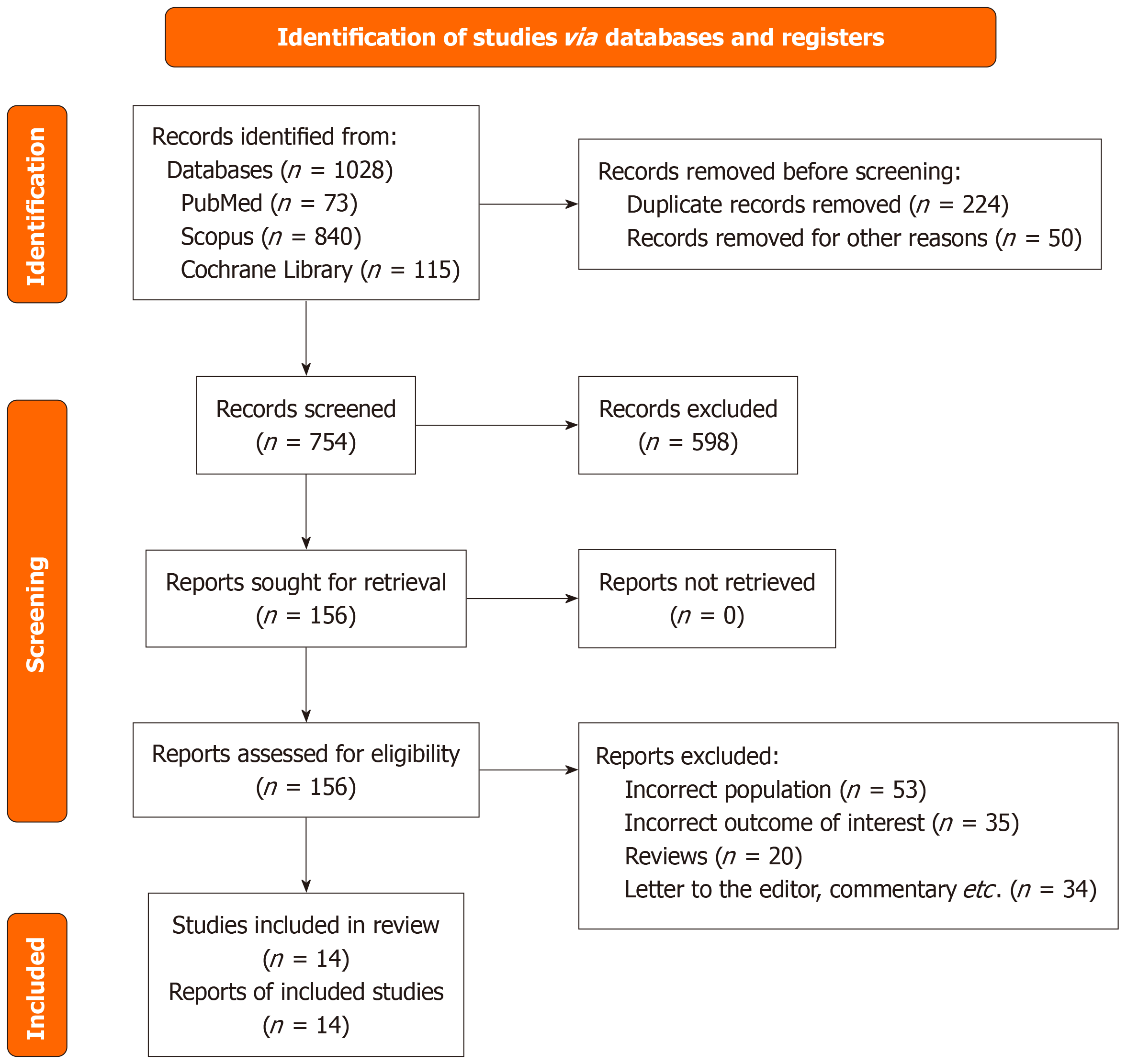
The search initially retrieved 1028 records from databases from which 14 RCTs[21-34] involving 1920 participants undergoing bronchoscopy under the influence of DEX were included. A more detailed overview of the selection process is given in Figure 1. All the studies included are RCTs that administered the intervention drug via intravenous infusion. Among these studies, six compared DEX against midazolam[28-30,32-34]. Two studies[27,31] compared against a combination of midazolam-fentanyl, while two studies had combination of propofol-fentanyl[23,24]. One study compared DEX against propofol[26] and another compared against alfentanil[22]. One study had remifentanil in combination with propofol[25]. Lastly, one study[21] had remimazolam-remifentanil combination as the control.
The characteristics of the included studies are summarized in Table 1 while baseline characteristics are given in Table 2[21-34]. DEX (n = 962) was compared with control interventions (n = 958). The follow-up durations of included studies ranged from 6 months to 41 months. All patients were adults, over the age of 18, with the mean age as 53.04 ± 6.70 years, consisting of 59.4% males and 40.6% females. Patients undergoing bronchoscopy were mostly there for the purpose of inspection/examination, bronchoalveolar lavage or biopsy and the average duration of the procedure across studies was 16.52 ± 6.02 minutes.
| Ref. | Study design | Population | Intervention | Control | Sample size | Males | Study duration | DEX dose | Route of administration | Purpose of bronchoscopy | ||
| Intervention | Control | Intervention | Control | |||||||||
| Magazine et al[27], 2024 | RCT | Patients aged 18-65 years undergoing FB in a tertiary care hospital in South India | DEX (0.75 μg/kg) | Midazolam -fentanyl (0.035 mg/kg and 25 μg) | 31 | 31 | 18 (58) | 20 (64.5) | 41 months | 0.75 µg/kg | Intravenous infusion | To visualize the bronchial tree and perform necessary sampling procedures |
| Zhou et al[21], 2024 | RCT | Patients aged 18-70 undergoing painless flexible fiberoptic bronchoscopy | DEX (4 μg/mL) -remifentanil (20 μg/mL) | Remimazolam (1 mg/mL) -remifentanil (20 μg/mL) | 182 | 182 | 116 (63.7) | 126 (69.2) | 18 months | 4 μg/mL | Intravenous infusion | Evaluation of unexplained cough, hemoptysis, localized wheezing, hoarseness, suspected lung abnormalities, lung disease evaluation, and preoperative assessment |
| Magazine et al[28], 2021 | RCT | Patients (aged 18-65, requiring diagnostic FB) | DEX (0.75 μg/kg) | Midazolam (0.035 mg/kg) | 24 | 24 | 13 (54.2) | 10 (41.7) | 17 months | 0.75 µg/kg | Intravenous infusion | Diagnostic |
| Kim et al[29], 2021 | RCT | Patients undergoing EBUS-TBNA | DEX (0.25-0.75 μg/kg) | Midazolam | 48 | 54 | 33 (68.8) | 39 (72.2) | 14 months | 0.25-0.75 μg/kg | Intravenous infusions | Diagnostic |
| Zhang et al[31], 2021 | RCT | Patients undergoing bronchoscopy | DEX combined with midazolam | Fentanyl combined with midazolam | 222 | 211 | 139 (62.6) | 126 (59.7) | - | - | Intravenous infusion | Diagnostic |
| Magazine et al[30], 2020 | RCT | Patients aged 18-65 requiring diagnostic FB | DEX (0.5 μg/kg) | Midazolam (0.035 mg/kg) | 27 | 27 | 19 (70.4) | 14 (51.9) | 22 months | 0.5 μg/kg | Intravenous infusion | Diagnostic bronchoscopy to evaluate pulmonary pathologies, assess centrally located tumors, and identify the cause of hemoptysis |
| Goneppanavar et al[32], 2015 | RCT | Patients aged 18-70 years undergoing FB | DEX (1 μg/kg) | Midazolam (0.02 mg/kg) | 27 | 27 | 20 (74.1) | 24 (88.9) | - | 1 μg/kg | Intravenous infusion | Evaluation of the airway in the Department of Pulmonary Medicine |
| Liao et al[33], 2012 | RCT | Postoperative adult patients in the intensive care unit who had undergone thoracic surgery and were referred for bronchoscopy | DEX | Midazolam | 99 | 99 | 61 (61.6) | 62 (63.3) | 12 months | - | Intravenous infusion | Postoperative evaluation in patients who had undergone thoracic surgery |
| Kumari et al[34], 2021 | RCT | Patients undergoing EBUS-TBNA | DEX (1 μg/kg) | Midazolam (2 mg/kg) | 99 | 98 | 56 (56.6) | 50 (51) | - | 1 μg/kg | Intravenous infusion | To perform EBUS-TBNA for diagnostic evaluation |
| Riachy et al[22], 2018 | RCT | Patients aged 18-70 undergoing elective FB | DEX (0.5 μg/kg) | Alfentanil (10 μg/kg) | 53 | 55 | 31 (58.5) | 30 (55.6) | 25 months | 0.5 μg/kg | Intravenous infusion | Diagnostic evaluation of pulmonary disease |
| Yuan et al[23], 2016 | RCT | Patients aged 20-75 years with ASA I-III undergoing elective FB | DEX (1 μg/kg) + fentanyl (1 μg/kg) | Propofol (4 μg/mL) + fentanyl (1 μg/kg) | 50 | 50 | 27 (54) | 22 (44) | 6 months | 1 μg/kg | Intravenous infusion | Various procedures (e.g., bronchoalveolar lavage, transbronchial biopsy) |
| Shoukry et al[24], 2016 | RCT | Adult patients (18-70 years old, ASA I-III) undergoing elective fiberoptic bronchoscopy | DEX (1 μg/kg) + fentanyl (1 μg/kg) | Propofol (0.5-1 mg/kg) + fentanyl (1 µg/kg) | 36 | 36 | 23 (63.9) | 25 (69.4) | 9 months | 1 μg/kg | Intravenous infusion | Diagnostic purposes (e.g., lung cancer staging, hemoptysis, interstitial lung disease) |
| Mani et al[26], 2015 | RCT | Patients undergoing FB | DEX (1 μg/kg) | Propofol (1 mg/kg) | 30 | 30 | - | - | - | 1 μg/kg | Intravenous infusion | Diagnostic FB |
| Ryu et al[25], 2012 | RCT | Patients, aged 18-70 years, ASA I-III, undergoing elective diagnostic FB | DEX (0.4-2 μg/kg) + propofol (0.5 mg/kg) | Propofol (0.5 mg/kg) + remifentanil (1-5 μg/kg) | 35 | 35 | 20 (57) | 18 (51) | 7 months | 0.4-2 μg/kg | Intravenous infusion | Diagnostic FB |
| Ref. | Age (year) | Weight (kg) | Body mass index (kg/m²) | Duration of bronchoscopy | Heart rate (bpm) | Mean systolic blood pressure (mmHg) | Mean diastolic blood pressure (mmHg) | Mean oxygen saturation, saturation of oxygen in blood | ||||||||||
| Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | |||
| Magazine et al[27], 2024 | 47.65 ± 13.41 | 50.23 ± 11.97 | 52.94 ± 12.36 | 57.16 ± 10.95 | 20.90 ± 4.86 | 21.83 ± 3.19 | 15.35 ± 11.93 | 12.84 ± 8.11 | 89.87 ± 11.35 | 85.94 ± 16.86 | 133.77 ± 22.38 | 124.68 ± 14.16 | 83.29 ± 8.77 | 76.61 ± 9.31 | 99.45 ± 1.50 | 99.23 ± 1.28 | ||
| Magazine et al[28], 2021 | 48.50 ± 13.138 | 44.42 ± 12.047 | 50.55 ± 10.20 | 50.07 ± 7.81 | 20.25 ± 2.80 | 20.05 ± 3.36 | 13.42 ± 6.801 | 10.54 ± 3.706 | 81.92 ± 18.533 | 84.83 ± 15.670 | 127.42 ± 20.930 | 126.58 ± 20.905 | 74.08 ± 11.088 | 78.92 ± 9.528 | 99.92 ± 0.408 | 100.00 ± 0.000 | ||
| Kim et al[29], 2021 | 63.7 ± 11.3 | 63.9 ± 11.0 | - | - | 23.4 ± 2.9 | 23.7 ± 2.6 | - | - | - | - | - | - | - | - | - | - | ||
| Zhang et al[31], 2021 | 58.03 ± 14.04 | 60.23 ± 11.56 | 63.40 ± 11.60 | 61.76 ± 9.72 | - | - | 24.40 ± 12.37 | 20.29 ± 10.96 | - | - | - | - | - | - | - | - | ||
| Magazine et al[30], 2020 | 47.07 ± 13.40 | 45.00 ± 14.26 | 53.23 ± 8.69 | 50.37 ± 9.84 | 20.61 ± 3.27 | 19.98 ± 3.51 | - | - | 87.85 ± 16.19 | 87.41 ± 16.31 | 131.63 ± 22.56 | 126.67 ± 18.92 | 77.85 ± 9.53 | 77.19 ± 11.49 | 100.00 ± 0 | 99.85 ± 0.53 | ||
| Goneppanavar et al[32], 2015 | 49.70 ± 13.85 | 52.52 ± 15.05 | 53.26 ± 10.10 | 56.07 ± 10.84 | 20.77 ± 3.40 | 20.42 ± 3.46 | 8.93 ± 2.96 | 8.19 ± 2.53 | - | - | - | - | - | - | - | - | ||
| Liao et al[33], 2012 | 58.5 ± 9.1 | 60.1 ± 8.4 | 57.0 ± 8.4 | 58.2 ± 11.3 | - | - | 12.8 ± 2.6 | 13.1 ± 2.6 | - | - | - | - | - | - | - | - | ||
| Kumari et al[34], 2021 | 44 ± 14 | 49 ± 14 | 63 ± 13 | 65 ± 13 | - | - | - | - | - | - | - | - | - | - | - | - | ||
| Zhou et al[21], 2024 | 61 (55-65) | 60 (52-66) | 60 (53-68) | 61 (54-70) | 22.24 ± 2.787 | 22.60 ± 2.742 | 13 (10-18.5) | 13 (10-19) | - | - | - | - | - | - | - | - | ||
| Riachy et al[22], 2018 | 63 | - | - | - | - | 16 | - | - | - | - | - | - | - | - | ||||
| Yuan et al[23], 2016 | 59.76 ± 7.61 | 60.46 ± 69.3 | - | - | 22.20 ± 3.06 | 21.69 ± 3.16 | 20.64 ± 2.08 | 21.14 ± 1.87 | - | - | - | - | - | - | - | - | ||
| Shoukry et al[24], 2016 | 50.3 ± 14.9 | 47.9 ± 15.3 | 77.9 ± 14.2 | 80.3 ± 14.0 | - | - | 21.8 ± 3.9 | 21.5 ± 3.6 | - | - | - | - | - | - | - | - | ||
| Mani et al[26], 2015 | - | - | 164.9 | 61.3 ± 6.15 | - | - | - | - | - | - | - | - | - | - | ||||
| Ryu et al[25], 2012 | 52.9 (27-67) | 52.9 (23-69) | 62.1 ± 13.8 | 60.6 ± 13.8 | - | - | 12.1 ± 2.8 | 12.0 ± 3.4 | - | - | - | - | - | - | - | - | ||
All studies[21-34] were assessed to have the overall low risk of bias. A detailed summary of quality assessment is presented in Supplementary Figure 1.
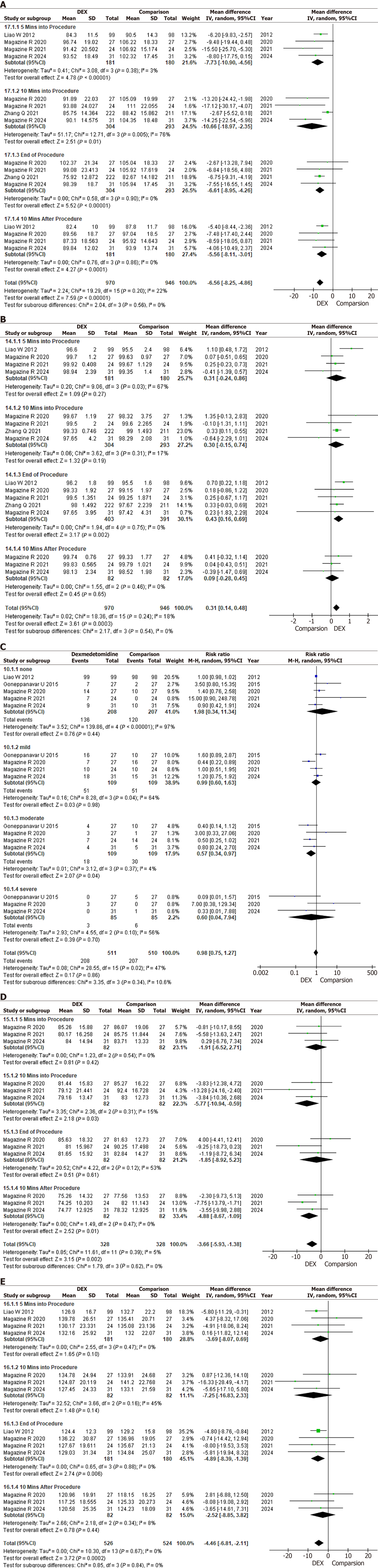
Heart rate (bpm): Five out of fourteen studies recorded the heart rate (bpm) at specific points in the procedure. The results show an overall significant decrease in heart rate in the DEX group vs other control groups (MD = -6.56, 95%CI: -8.25 to -4.86, P < 0.00001, I² = 22%). At all-time points, DEX was associated with a greater likelihood of lowering the heart rate. At 5 minutes into the procedure, DEX significantly lower heart rate (MD = -7.73, 95%CI: -10.90 to -4.56, P < 0.00001, I² = 3%). Similarly, at 10 minutes into the procedure (MD = -10.66, 95%CI: -18.97 to -2.35, P < 0.01, I² = 76%), end of the procedure (MD = -6.61, 95%CI: -8.95 to -4.26, P < 0.00001, I² = 0%), and at 10 minutes after the procedure (MD = -5.56, 95%CI: -8.11 to -3.01, P < 0.0001, I² = 0%) DEX produced significant reduction in the heart rate when compared to the control group (Figure 2A)[27,28,30,31,33]. Due to difference in the control drug used, meta-analysis was performed for subgroup analysis at the mark of end of procedure. DEX vs midazolam (P = 0.26) was not statistically significant. However, DEX vs fentanyl-midazolam (P < 0.00001) was statistically significant (Supplementary Figure 2)[27,28,30,31].
Mean oxygen saturation: Five studies evaluated the mean oxygen saturation at various points (Figure 2B)[27,28,30,31,33]. Overall, the results showed that patients on DEX had a higher mean oxygen saturation throughout the procedure (MD = 0.31, 95%CI: 0.14-0.48, P = 0.0003, I² = 18%). DEX has a significantly higher likelihood of increasing oxygen saturation at the end of the procedure (MD = 0.43, 95%CI: 0.16-0.69, P = 0.002, I² = 0%). Although DEX increased the oxygen saturation at the 5 minutes into the procedure, 10 minutes into the procedure and at the 10-minute mark after the end of the procedure mark, these differences were non-significant (MD = 0.31, 95%CI: -0.24 to 0.86, P = 0.27, I² = 67%), (MD = 0.30, 95%CI: -0.15 to 0.74, P = 0.19, I² = 17%) and (MD = 0.09, 95%CI: -0.28 to 0.45, P = 0.65, I² = 0%), respectively. These results, however, fall within the normal range and are not likely to be clinically meaningful. Since different control drug was used, meta-analysis was performed for subgroup analysis for this outcome at the end of procedure. Here, DEX vs midazolam (P = 0.006) was statistically significant whereas DEX vs fentanyl-midazolam (P = 0.07) was not statistically significant (Supplementary Figure 3)[27,28,30,31,33].
Discomfort during bronchoscopy: Five out of fourteen studies compared the degree of discomfort of bronchoscopy between the groups. The cumulative result reveals no significant decrease in discomfort of DEX when compared to the control groups (RR = 0.98, 95%CI: 0.75-1.27, P = 0.86, I² = 47%). The first category, none (RR = 1.98, 95%CI: 0.34-11.34, P = 0.44, I² = 97%), mild (RR = 0.99, 95%CI: 0.60-1.63, P = 0.98, I² = 64%) and severe (RR = 0.60, 95%CI: 0.04-7.94, P = 0.70 I² = 56%) show no significant effect on the discomfort with use of DEX when compared to control groups. However, a significant improvement in discomfort was observed with the moderate category (RR = 0.57, 95%CI: 0.34-0.97, P = 0.04, I² = 4%). Figure 2C shows the detailed forest plot. Meta-analysis was performed for subgroup analysis for the outcome of moderate discomfort[27,28,30,32,33]. Here, both DEX vs midazolam (P = 0.11) and DEX vs fentanyl-midazolam (P = 0.72) were not statistically significant (Supplementary Figure 4)[27,28,30,32,33].
Mean diastolic blood pressure: Three out of fourteen studies recorded the mean diastolic blood pressure at various time marks. The overall result reveal DEX significantly decreased the mean diastolic blood pressure with the DEX group vs the control groups (MD = -3.66, 95 CI%: -5.93 to -1.38, P = 0.002, I² = 5%) (Figure 2D)[27,28,30]. At the 10 minutes mark into the procedure (MD = -5.77, 95%CI: -10.94 to -0.59, P = 0.003, I² = 15%) as well as the 10 minute-mark after the end of the procedure (MD = -4.88, 95%CI: -8.67 to -1.09, P = 0.01, I² = 0%). DEX was associated with a significant decrease in mean diastolic blood pressure vs the control groups. Conversely, at 5 minutes into the procedure (MD = -1.91, 95%CI: -6.52 to 2.71, P = 0.42, I² = 0%) as well as at the end of the procedure (MD = -1.85, 95%CI: -8.92 to 5.23, P = 0.61, I² = 53%) the blood pressure in both groups was not significantly different.
Mean systolic blood pressure: Four out of fourteen studies evaluated the mean systolic blood pressure at various time marks. The pooled result, including all the time stamps, reveal DEX is significantly associated with a marked decrease in mean systolic blood pressure vs the control groups (MD = -4.69, 95%CI: -6.87 to -2.51, P < 0.0001, I² = 0%). The detailed forest plot is Figure 2E[27,28,30,33]. The 10 minutes mark into the procedure (MD = -6.60, 95%CI: -12.88 to -0.33, P = 0.04,
Composite score at nasopharynx: Three out of fourteen studies record the composite score at the level of the nasopharynx. The pooled data no significant link of DEX with achieving a better score (RR = 1.06, 95%CI: 0.82-1.37, P = 0.65, I² = 61%). The forest plot for this outcome is given in Supplementary Figure 5[27,28,30]. The category of ideal (RR = 1.06, 95%CI: 0.86-1.31, P = 0.60, I² = 73%) and acceptable scores (RR = 1.53, 95%CI: 0.22-10.82, P = 0.67, I² = 50%) show no significant association DEX with attaining ideal and acceptable scores. However, DEX was found to have significant association with reducing unacceptable scores (RR = 0.06, 95%CI: 0.00-0.98, P = 0.05). Heterogeneity was not acceptable since only one study had data for it.
Composite score at trachea: Three out of fourteen studies record the composite score at the level of the trachea. The pooled data reveal that DEX has no significant association with achieving a better score (RR = 0.99, 95%CI: 0.66-1.49, P = 0.97, I² = 66%). Ideal (RR = 1.22, 95%CI: 0.85-1.75, P = 0.28, I² = 72%), acceptable (RR = 0.58, 95%CI: 0.23-1.48, P = 0.26, I² = 41%) and unacceptable scores (RR = 0.93, 95%CI: 0.02-36.63, P = 0.97, I² = 77%) show no significant effect of DEX vs control groups. The forest plot for this is included in Supplementary Figure 6[27,28,30].
Cough: Three out of fourteen studies compare incidence and intensity of cough, as judged by the observer on the VAS scale. Pooled results show DEX has no significant correlation with reducing cough vs other control groups, although there was high heterogeneity among studies (MD = 0.34, 95%CI: -1.04 to 1.73, P = 0.63, I² = 95%). The forest plot is given in Supplementary Figure 7[29,31,34].
Willing to repeat the procedure: Of the fourteen studies, three studies evaluate patient willingness to repeat the procedure. Cumulative results show DEX is not significantly favored by the patients, for repeat use in a future procedure, vs control groups (RR = 1.01, 95%CI: 0.97-1.06, P = 0.59, I² = 65%). This forest plot is given as Supplementary Figure 8[31,33,34].
Need rescue dose: Six studies assessed the need for a rescue dose in the situation when intervention was ineffective. No significant association was found between DEX and the control groups for such need (RR = 0.99, 95%CI: 0.28-3.50, P = 0.98, I² = 91%; Supplementary Figure 9)[21,28,30,32,34].
RSS, during procedure: Four out of fourteen studies record the RSS during the procedure. The pooled results show no significant difference in RSS between DEX and control groups (RR = 1.02, 95%CI: 0.73-1.41, P = 0.93, I² = 61%). The score 1 (RR = 0.64, 95%CI: 0.19-2.11, P = 0.46, I² = 63%), score 2 (RR = 1.17, 95%CI: 0.19-1.60, P = 0.32, I² = 69%) and score 3 or > 3 (RR = 0.93, 95%CI: 0.36-2.42, P = 0.88, I² = 28%) show nominal difference in sedation scores with DEX treatment. A detailed forest plot is available as Supplementary Figure 10[27,28,30,32].
RSS, after procedure: Four out of fourteen studies record the RSS after the procedure had concluded. The pooled results show no significant difference in RSS between DEX and the control groups (RR = 1.04, 95%CI: 0.95-1.14, P = 0.42, I² = 0%). Analysis for score 1 (RR = 1.00, 95%CI: 0.87-1.15, P = 0.99, I² = 0%), score 2 (RR = 0.88, 95%CI: 0.44-1.73, P = 0.71, I² = 0%) and score 3 or > 3 (RR = 1.08, 95%CI: 0.95-1.22, P = 0.24, I² = 0%) reveal DEX was not significantly associated with better sedation scores (Supplementary Figure 11)[27,28,30,32].
Pain, during procedure: Three out of fourteen studies document the intensity of pain during the procedure, as judged by the numerical rating scale. The pooled result shows no significant difference in pain during procedure with use of DEX compared with the control groups (RR = 1.04, 95%CI: 0.73-1.48, P = 0.84, I² = 74%). Minimal pain (0-3) and mild pain (4-6) show no significant association of DEX with lowering the pain experienced during the procedure (RR = 1.28, 95%CI: 0.82-1.99, P = 0.27, I² = 92%) and (RR = 0.60, 95%CI: 0.26-1.40, P = 0.24, I² = 0%), respectively. Only one study documented data for category of maximal pain. Detailed forest plot shown in Supplementary Figure 12[28,30,32].
Pain, 10 minutes after procedure: Four out of fourteen studies recorded a pain rating, 10 minutes after the end of the procedure, as judged by the numerical rating scale. Overall, the pooled results are shown to not be significant (RR = 1.00, 95%CI: 0.96-1.05, P = 0.90, I² = 0%). The category of minimal pain (0-3) (RR = 1.00, 95%CI: 0.96 to 1.05, P = 0.91, I² = 0%) and mild pain (4-6) (RR = 1.38, 95%CI: 0.33 to 5.80, P = 0.66, I² = 0%), show that DEX is not significantly associated with a rating of minimal or mild pain. There was no maximal pain (7-10) category, since there were no events recorded for either the DEX group or the control group, as shown in Supplementary Figure 13[28,30,32].
Distress, during procedure: Four out of fourteen studies recorded distress, during the procedure, between DEX and control groups as judged by the numerical rating scale. The pooled impact shows no significant difference with DEX use (RR = 0.94, 95 CI%: 0.56-1.58, P = 0.83, I² = 75%). Supplementary Figure 14[28,30,32] shows further details. There is no significant association of DEX group with obtaining score of minimal distress (0-3) (RR = 1.59, 95%CI: 0.75-3.35, P = 0.22,
Distress, 10 minutes after procedure: Four out of fourteen studies record the distress, at the 10 minutes-mark after the end of the procedure, between DEX and control groups as judged by the numerical rating scale. The pooled impact shows no significant effect of DEX on reducing distress levels (RR = 1.05, 95 CI%: 0.93-1.19, P = 0.42, I² = 29%). This is illustrated in Supplementary Figure 15[27,28,30,32]. Minimal distress (0-3) (RR = 1.06, 95%CI: 0.92-1.22, P = 0.43, I² = 61%), mild distress (4-6) (RR = 0.71, 95%CI: 0.20-1.58, P = 0.60, I² = 59%) and maximal distress (7-10) (RR = 3.00, 95%CI: 0.32-28.03, P = 0.34, I² = 0%) show no significant association with use of DEX vs control groups.
Ease of bronchoscopy: Four out of fourteen studies compared the ease of bronchoscopy between DEX and control groups. The cumulative impact shows no significant association with DEX (RR = 0.97, 95%CI: 0.68-1.38, P = 0.87, I² = 60%), as seen in Supplementary Figure 16[27,28,30,32]. Easy (RR = 1.36, 95%CI: 1.05-1.77, P = 0.02, I² = 28%) and slightly difficult category (RR = 0.62, 95%CI: 0.41-0.94, P = 0.03, I² = 0%) show DEX has significant association with easier procedures. The very difficult category (RR = 0.80, 95%CI: 0.14-4.59, P = 0.80, I² = 63%) shows no significant association with DEX vs the control groups.
Quality of sedation: Four out of fourteen studies compared the quality of sedation among the groups according to four categories: (1) Excellent; (2) Good; (3) Fair; and (4) Poor. The pooled effect (RR = 1.00, 95%CI: 0.67-1.50, P = 0.99, I² = 60%) show no significant difference between DEX and the control groups. In the excellent category (RR = 2.84, 95%CI: 0.51-15.92, P = 0.23, I² = 43%), good (RR = 1.48, 95%CI: 0.74-2.95, P = 0.26, I² = 65%), fair (RR = 0.68, 95%CI: 0.43-1.08, P = 0.11, I² = 58%) and poor category (RR = 0.73, 95%CI: 0.91-2.75, P = 0.64, I² = 48%), there is no significant association found between DEX and achieving a better score, like excellent, good and fair. The forest plot of this outcome is given in Supplementary Figure 17[27,28,30,32].
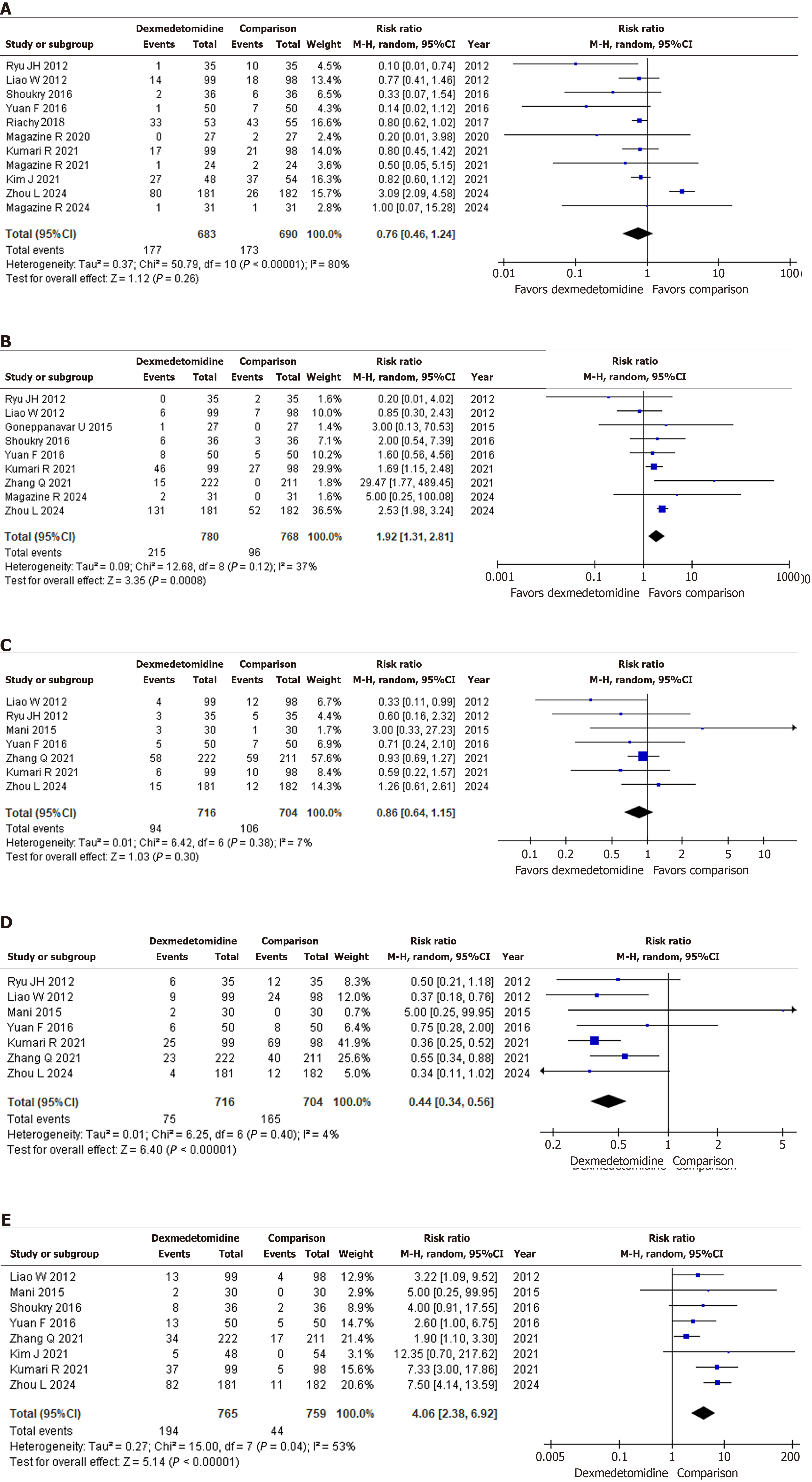
Oxygen de-saturation: A more important measure is the occurrence of desaturation events during bronchoscopy. Eleven out of fourteen studies document oxygen de-saturation (Figure 3A) as an adverse effect[21-25,27,30,33,34]. Although numerically fewer incidences, the analysis shows no significant association of desaturation with the use of DEX vs other control groups desaturation (RR = 0.76, 95%CI: 0.46-1.24, P = 0.26, I² = 80%). While DEX is seen to be linked with a modest increase in mean oxygen saturation, this difference is trivial. The more significant parameter, the desaturation event, was reported as an insignificant outcome.
Hypotension: Nine out of fourteen studies documented hypotension (Figure 3B) as an adverse effect associated with DEX vs the control group[21,23-25,27,31-34]. DEX is found to be significantly associated with the development of hypotension (RR = 1.92, 95%CI: 1.31-2.81, P = 0.0008, I² = 37%).
Hypertension: Seven out of fourteen studies record hypertension (Figure 3C) as an adverse effect[21,23,25,26,31,33,34]. The analysis reveals DEX does not have a significant association with the incidence of hypertension (RR = 0.86, 95%CI: 0.64-1.15, P = 0.30, I² = 7%).
Tachycardia: Seven out of fourteen studies document the incidence of tachycardia (Figure 3D)[21,23,25,26,31,33,34]. DEX is revealed to be significantly associated with fewer incidence of tachycardia vs the control groups (RR = 0.44, 95%CI: 0.34-0.56, P < 0.00001, I² = 4%).
Bradycardia: Eight out of fourteen studies document the incidence of bradycardia (Figure 3E) as an adverse effect[21,23,24,26,29,31,33,34]. DEX is associated with significantly greater incidence of bradycardia vs the control groups (RR = 4.06, 95%CI: 2.38-6.92, P < 0.00001, I² = 53%).
Sensitivity analysis was performed for specific outcomes that had significant heterogeneity. In terms of adverse effects, the outcome of oxygen desaturation had high heterogeneity (P = 0.26, I² = 80%) which, upon odd one out analysis, improved (P = 0.010, I² = 14%). The forest plot is given in Supplementary Figure 18[21-25,27-30,33,34]. Similarly, the heterogeneity in bradycardia also improved from P < 0.00001 (I² = 53%) to P < 0.00001 (I² = 0%) when outlier study was removed which is shown in Supplementary Figure 19[21,23,24,26,29,31,33,34]. Similarly, the subgroup 10 minutes into procedure of the outcome of “heart rate (bpm)” also dropped from P = 0.01 (I² = 76%) to P < 0.00001 (I² = 0%) in Supplementary Figure 20[27,28,30,31,33]. In the outcome of “discomfort during bronchoscopy”, the subgroup none improved from I² = 97% to I² = 52%, mild improved from I² = 64% to I² = 0%, and severe improved from P = 0.70 (I² = 56%) to P = 0.09 (I² = 0%) as seen in Supplementary Figure 21[27,28,30,32,33].
The outcome of “quality of sedation” showed considerable heterogeneity in the subgroups good (P = 0.26, I² = 65%) and fair (P = 0.11, I² = 58%) which improved to P = 0.04 (I² = 44%) and P = 0.001 (I² = 0%) respectively, as seen in Supplementary Figure 22[27,28,30,32]. For the outcome of “RSS, during procedure”, the subgroup score 1 (I² = 63%) and score 2 (P = 0.32, I² = 69%) showed heterogeneity but upon evaluation they improved to I² = 51% and P = 0.003, I² = 0% respectively as in Supplementary Figure 23[27,28,30,32]. In the “ideal” subgroup in both composite score, nasopharynx (P = 0.28, I² = 72%) and “composite score, trachea” (P = 0.60, I² = 73%), removing outliers resulted in a significant reduction in heterogeneity to P = 0.04 (I² = 0%) and P = 0.002 (I² = 0%) respectively, as in Supplementary Figures 24 and 25[27,28,30].
In the outcome assessing “mean oxygen saturation”, improvement was observed in subgroup “5 minutes into procedure” where heterogeneity dropped from I² = 67% to I² = 0% on removing one study (Supplementary Figure 26)[27,28,30,31,33]. Similarly, for “willing to repeat procedure”, improvement from I² = 66% to I² = 0% was seen in Supplementary Figure 27[31,33,34]. Similar effect is observed in the “minimal” category of “pain, during procedure” where it improved from I² = 92% to I² = 0% in Supplementary Figure 28[28,30,32]. Of the minimal (0-3) (I² = 61%) and mild (4-6) (I² = 59%) category of the outcome “distress, 10 minutes, after procedure”, heterogeneity improved to I² = 0% in both outcomes as seen in Supplementary Figure 29[27,28,30,32]. Conversely, in “minimal” category of “distress, during procedure”, the heterogeneity improved only mildly from I² = 86% to I² = 57% as seen in Supplementary Figure 30[28,30,32].
For comparison of “need rescue dose”, the odds improved from I² = 91% to I² = 22% as in Supplementary Figure 31[21,27,28,30,32,34]. For “mean diastolic blood pressure”, the outcome “end of procedure” improved from I² = 53% to I² = 41% (Supplementary Figure 32)[27,28,30]. The heterogeneity in the “very difficult” subgroup of “ease of bronchoscopy” improved marginally from I² = 63% to I² = 30% as in Supplementary Figure 33[27,28,30,32]. Conversely, for assessment of “cough”, removing outliers did not result in a significant reduction in heterogeneity from I² = 95% to I² = 93% as shown in Supplementary Figure 34[29,31,34].
This systematic review and meta-analysis evaluate the safety and efficacy of DEX during bronchoscopy procedures using 14 RCTs[21-34] (n = 1920 patients; DEX, n = 962; control, n = 958). The principal findings of this study report that patients receiving DEX have a statistically significant decrease in mean heart rate and an increase in mean oxygen saturation throughout the procedure. Moreover, DEX was also found to be efficient in lowering the discomfort experienced by the patient during the procedure, due to its effects as an analgesic, however it was statistically not significant. To the best of our knowledge, this is the most recent study analyzing efficacy and safety of DEX, a highly selective alpha 2-adrenoceptor agonist with diverse pharmacodynamic effects[35]. It is commercialized as an agent that offers conscious sedation without much respiratory depression that allows patients to remain cooperative[3,36,37].
In terms of safety, our study analyzed the incidence of abnormal heart rate. The use of DEX was highly associated with bradycardia but significantly less associated with tachycardia. A recently published meta-analysis by Guo et al[38] also reports significant incidence of bradycardia. This effect can be reasonably explained owing to its sympathetic inhibition, peripheral vasoconstriction and inevitable reflex bradycardia[35]. In another trial conducted by Khan et al[39], decrease in heart rate was noted as well, although the patient population did not require treatment for it. In a trial conducted by Zhao et al[40], it is found that co-administration of DEX with Midazolam provided a more stable hemodynamic status than those treated with DEX alone. The hypotensive effects due to the excitation of the alpha 2-adrenergic receptors were noted as early as 1996[41]. This meta-analysis followed the diastolic and systolic blood pressure throughout the process, as a secondary outcome, which always revealed a pattern of significantly lower blood pressure during the procedure. However, either hypertensive or hypotensive effects are believed to be dose dependent[42]. This biphasic effect can prove to be a hindrance for patients with known cardiac problems, arrhythmias, or other vulnerable states. Some studies[22,30] indicate that DEX retains its clinical efficacy at 0.5 μg/kg. On the contrary, Lee et al[43] found the same dose insufficient to maintain RSS at 3, requiring frequent rescue sedation although the patient population was comparable to the former studies. From our included studies, Goneppanavar et al[32] reports one incident of an RSS score 5 sedation in the intervention group being given DEX (1 μg/kg). Here, three patients started the procedure at a sedation level of RSS 4, which might explain the depth of sedation reached as the procedure proceeded. On the other hand, Magazine et al[27] reports one incident of an RSS score 5 sedation in the control group being given a combination of Midazolam-Fentanyl (0.035 mg/kg and 25 μg) which can be explained by the synergistic effect of this combination. These two incidents indicate a need for careful monitoring of different drug interactions causing differences in the depth of sedation.
Additionally, we analyzed the risk of oxygen de-saturation or hypoxemia, defined as oxygen saturation < 90% for more than 30 seconds. Our results demonstrate that DEX was relatively safe to use in this regard with no clinical difference in the risk between both groups. Although numerically fewer events in the DEX group, the results had high heterogeneity. Upon sensitivity analysis, the removal of Zhou et al[21] made the results statistically significant (P = 0.26 vs 0.010). This effect can be explained sufficiently by the action of the comparison drug used i.e., remimazolam, an ultra-short acting benzodiazepine with faster recovery time[44] than midazolam, which was used as comparison by most of the other included studies. The efficacy of remimazolam has been established in fellow meta-analyses[45,46]. Remimazolam is considered as a midazolam hybridized with an ester moiety, which allows its metabolism via esterase[47]. Since its metabolism is not singularly dependent on one organ, i.e., liver, remimazolam is considered reliable due to its predictable recovery.
The study by Guo et al[38] was confined to adverse effects. To present a comprehensive picture of the clinical experience we integrated patient reported outcomes. When assessing the quality of sedation, as experienced by the patients, DEX group reported greater excellent and good scores vs other groups. Similarly, more patients experienced minimal pain and distress during the procedure. These findings, although numerically consistent, were not statistically significant, probably owing to the small population size. A recent trial conducted by Abdelgalil et al[48] also shows that a combination of DEX-propofol produced lower pain scores.
Three studies[27,28,30] used a composite score at the level of nasopharynx and trachea, but these had nominal differences between the groups. This criterion was utilized justifiably by Goneppanavar et al[32], as well as Antony et al[49] to provide a comprehensive understanding of sedation. This parameter represents an average of five categories: Sedation, calmness, respiratory response, physical movement, facial tension which are all judged by the observer. Inclusion of such parameters in future studies may help bring uniformity in the judgment of efficacy of bronchoscopy drugs.
To the best of our knowledge, this is the most recent study amassing the greatest population of people utilizing DEX, making our results impactful. This analysis unveils an alarming link with bradycardia, indicating that the utmost level of care be taken in patients vulnerable to such situations. Our findings also include patient reported outcomes, making our study a true representative of the patient experience. Upon sensitivity analysis for secondary outcome of RSS and the composite score, the results showed significant change but due to the small population group recorded in these outcomes no conclusion can be drawn satisfactorily. For the outcome of quality of discomfort, removal of Magazine et al[27] made the category of good and fair showed significant improvement. This effect may be credited to the use of Fentanyl in combination with midazolam used as the comparison in this study, although, owing to the small population size no reasonable conclusion can be drawn. Future studies can use this data to tailor specific demographics and utilize standardized methods to quantify outcomes such as cough and patient discomfort.
It is beneficial to mention that most studies included in this analysis utilized similar techniques making the pool of the selected studies relatively similar. However, there was notable heterogeneity found in some outcomes. Most of our included studies compared DEX against midazolam, a potent benzodiazepine, or propofol, a short acting GABA agonist. A recent survey among physicians in Turkey indicates that most physicians prefer to use midazolam over propofol if an anesthesiologist is not present[50]. This is likely to explain the rising popularity of midazolam. A recent meta-analysis by Liu et al[51] reports greater association of respiratory depression with midazolam. Both midazolam and propofol are associated with respiratory depression[14,52]. To combat such effects, recent combinations of drugs aim to minimize the dose required[53-55]. DEX is key in avoiding such adverse effects.
Several limitations should be acknowledged. A significant source of heterogeneity among the included studies stems from variations in dosage regimens. While sensitivity analyses were conducted to address this variation, uniform and standardized trials are needed to validate and generalize these findings to broader clinical populations. Most of the secondary outcomes like score at nose, trachea and cough were statistically insignificant. One of the key reasons for this was the limited number of studies along with small patient population that restricted the strength and generalizability of the conclusions. Additionally, due to the absence of predefined guidelines for cough, our study had limited data to analyze which may diminish the validity of our findings. This lack of standardization likely contributed to variability in assessing procedural comfort as well. Few other included studies[23,29,31,34] also documented patient/bronchoscopist sedation satisfaction; however relevant data could not be pooled owing to differences in methods utilized. Future studies would benefit from adopting standardized scales to uniformly quantify patient experiences during sedation. Future studies should concentrate on these secondary results using sufficiently large sample sizes and consistent assessment techniques, as they hold clinical significance for assessing patient comfort and the safety of procedures. Similarly, further research in this regard is bound to provide a better, more consistent picture of the patient’s experience, preferably with a uniform method of documentation.
The results of this meta-analysis show DEX to have a significant risk of bradycardia and hypotension throughout the procedure. Although DEX reduced the distress and pain experienced by patients during the procedure, these effects were not significant after the procedure. Larger clinical trials with more robust methodology are required to confirm the long-term safety and efficacy of DEX.
| 1. | Aravena C, Mehta AC. Rigid versus flexible bronchoscopy in interventional pulmonology: perspective from the master clinicians. Curr Opin Pulm Med. 2025;31:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 2. | Apostolos F, Nikolaos Z, Charalampos M, Kyriakos K, Sotirios F, Gregorios V. Dexmedetomidine-ketamine combination versus fentanyl-midazolam for patient sedation during flexible bronchoscopy: a prospective, single-blind, randomized controlled trial. BMC Pulm Med. 2024;24:301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 3. | Keating GM. Dexmedetomidine: A Review of Its Use for Sedation in the Intensive Care Setting. Drugs. 2015;75:1119-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 288] [Article Influence: 26.2] [Reference Citation Analysis (1)] |
| 4. | Chadha M, Kulshrestha M, Biyani A. Anaesthesia for bronchoscopy. Indian J Anaesth. 2015;59:565-573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 5. | Liu X, Li Y, Kang L, Wang Q. Recent Advances in the Clinical Value and Potential of Dexmedetomidine. J Inflamm Res. 2021;14:7507-7527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (5)] |
| 6. | Dilip Wani M, Dey C, Mistry T. Role of sedation and analgesia in ICU. Anaesth Pain Intensive Care. 2018;22 Suppl 1:S88-S98. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 7. | Hughes CG, McGrane S, Pandharipande PP. Sedation in the intensive care setting. Clin Pharmacol. 2012;4:53-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 8. | Afonso J, Reis F. Dexmedetomidine: current role in anesthesia and intensive care. Rev Bras Anestesiol. 2012;62:118-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 225] [Article Influence: 16.1] [Reference Citation Analysis (1)] |
| 9. | Nordt SP, Clark RF. Midazolam: a review of therapeutic uses and toxicity. J Emerg Med. 1997;15:357-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 198] [Article Influence: 6.8] [Reference Citation Analysis (4)] |
| 10. | Olkkola KT, Ahonen J. Midazolam and other benzodiazepines. Handb Exp Pharmacol. 2008;335-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 285] [Article Influence: 15.8] [Reference Citation Analysis (2)] |
| 11. | Dinis-Oliveira RJ. Metabolic Profiles of Propofol and Fospropofol: Clinical and Forensic Interpretative Aspects. Biomed Res Int. 2018;2018:6852857. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 12. | Lundström S, Twycross R, Mihalyo M, Wilcock A. Propofol. J Pain Symptom Manage. 2010;40:466-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 13. | Hu Q, Liu X, Wen C, Li D, Lei X. Remimazolam: An Updated Review of a New Sedative and Anaesthetic. Drug Des Devel Ther. 2022;16:3957-3974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 143] [Article Influence: 35.8] [Reference Citation Analysis (7)] |
| 14. | Goudra BG, Singh PM, Borle A, Farid N, Harris K. Anesthesia for Advanced Bronchoscopic Procedures: State-of-the-Art Review. Lung. 2015;193:453-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 15. | Peng PW, Sandler AN. A review of the use of fentanyl analgesia in the management of acute pain in adults. Anesthesiology. 1999;90:576-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 270] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 16. | Sridharan K, Sivaramakrishnan G. Comparison of Fentanyl, Remifentanil, Sufentanil and Alfentanil in Combination with Propofol for General Anesthesia: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Curr Clin Pharmacol. 2019;14:116-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (2)] |
| 17. | Hsieh CH, Lin TY, Wang TY, Kuo CH, Lin SM, Kuo HP, Lo YL. The safety and efficacy of alfentanil-based induction in bronchoscopy sedation: A randomized, double-blind, controlled trial. Medicine (Baltimore). 2016;95:e5101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Wang Z, Hu Z, Dai T. The comparison of propofol and midazolam for bronchoscopy: A meta-analysis of randomized controlled studies. Medicine (Baltimore). 2018;97:e12229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int J Surg. 2021;88:105906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7951] [Cited by in RCA: 5389] [Article Influence: 1077.8] [Reference Citation Analysis (5)] |
| 20. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20331] [Article Influence: 2904.4] [Reference Citation Analysis (7)] |
| 21. | Zhou L, Zou J, Li X, Zuo X, Gu M, Sun K, Fan W, Yao Y, Yan M. Efficacy and safety of remimazolam versus dexmedetomidine for patients undergoing flexible fiberoptic bronchoscopy: A randomized, clinical trial. J Clin Anesth. 2024;99:111677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 22. | Riachy M, Khayat G, Ibrahim I, Aoun Z, Dabar G, Bazarbachi T, Khalil N, Habr B. A randomized double-blind controlled trial comparing three sedation regimens during flexible bronchoscopy: Dexmedetomidine, alfentanil and lidocaine. Clin Respir J. 2018;12:1407-1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 23. | Yuan F, Fu H, Yang P, Sun K, Wu S, Lv M, Dong Z, Dong T. Dexmedetomidine-fentanyl versus propofol-fentanyl in flexible bronchoscopy: A randomized study. Exp Ther Med. 2016;12:506-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 24. | Shoukry RA. Safety and efficacy of dexmedetomidine sedation for elective fiberoptic bronchoscopy: A comparative study with propofol. Egypt J Anaesth. 2016;32:483-488. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Ryu JH, Lee SW, Lee JH, Lee EH, Do SH, Kim CS. Randomized double-blind study of remifentanil and dexmedetomidine for flexible bronchoscopy. Br J Anaesth. 2012;108:503-511. [PubMed] [DOI] [Full Text] |
| 26. | Mani B, George SK, Khan MS, Kisku KH. Propofol versus dexmedetomidine as a sole sedative for diagnostic flexible bronchoscopy: A randomized double-blind study. Ain Shams J Anesthesiol. 2015;8:573. [DOI] [Full Text] |
| 27. | Magazine R, Elenjickal VM, Padukone AM, Bhat A, Chogtu B. Comparison Between Dexmedetomidine and Midazolam-Fentanyl Combination in Flexible Bronchoscopy: A Prospective, Randomized, Double-blinded Study. J Bronchology Interv Pulmonol. 2024;31:e0985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Magazine R, Antony T, Chogtu B, Prabhudev AM, Surendra VU, Guddattu V. Clinical usefulness of intermediate-dose dexmedetomidine (0.75 μg/kg) in flexible bronchoscopy - A prospective, randomized, double-blinded study. Indian J Pharmacol. 2021;53:440-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 29. | Kim J, Choi SM, Park YS, Lee CH, Lee SM, Yoo CG, Kim YW, Lee J. Dexmedetomidine versus midazolam for sedation during endobronchial ultrasound-guided transbronchial needle aspiration: A randomised controlled trial. Eur J Anaesthesiol. 2021;38:534-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 30. | Magazine R, Venkatachala SK, Goneppanavar U, Surendra VU, Guddattu V, Chogtu B. Comparison of midazolam and low-dose dexmedetomidine in flexible bronchoscopy: A prospective, randomized, double-blinded study. Indian J Pharmacol. 2020;52:23-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 31. | Zhang Q, Zhou J, He Q, Guan S, Qiu H, Xu X, Xu Q, Dai P, Dong D, Wang X, Zhang S. Dexmedetomidine combined with midazolam infusion guided by bispectral index during bronchoscopy. Clin Respir J. 2021;15:929-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 32. | Goneppanavar U, Magazine R, Periyadka Janardhana B, Krishna Achar S. Intravenous Dexmedetomidine Provides Superior Patient Comfort and Tolerance Compared to Intravenous Midazolam in Patients Undergoing Flexible Bronchoscopy. Pulm Med. 2015;2015:727530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 33. | Liao W, Ma G, Su QG, Fang Y, Gu BC, Zou XM. Dexmedetomidine versus midazolam for conscious sedation in postoperative patients undergoing flexible bronchoscopy: a randomized study. J Int Med Res. 2012;40:1371-1380. [PubMed] [DOI] [Full Text] |
| 34. | Kumari R, Jain K, Agarwal R, Dhooria S, Sehgal IS, Aggarwal AN. Fixed dexmedetomidine infusion versus fixed-dose midazolam bolus as primary sedative for maintaining intra-procedural sedation during endobronchial ultrasound-guided transbronchial needle aspiration: a double blind randomized controlled trial. Expert Rev Respir Med. 2021;15:1597-1604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 35. | Weerink MAS, Struys MMRF, Hannivoort LN, Barends CRM, Absalom AR, Colin P. Clinical Pharmacokinetics and Pharmacodynamics of Dexmedetomidine. Clin Pharmacokinet. 2017;56:893-913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 938] [Cited by in RCA: 842] [Article Influence: 93.6] [Reference Citation Analysis (1)] |
| 36. | Lee S. Dexmedetomidine: present and future directions. Korean J Anesthesiol. 2019;72:323-330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 256] [Article Influence: 36.6] [Reference Citation Analysis (1)] |
| 37. | Wu SH, Lu DV, Hsu CD, Lu IC. The Effectiveness of Low-dose Dexmedetomidine Infusion in Sedative Flexible Bronchoscopy: A Retrospective Analysis. Medicina (Kaunas). 2020;56:193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 38. | Guo Q, An Q, Zhao L, Wu M, Wang Y, Guo Z. Safety and Efficacy of Dexmedetomidine for Bronchoscopy: A Systematic Review and Meta-Analysis. J Clin Med. 2023;12:1607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 39. | Khan A, Sinha R, Kumar KR, Velpandian T, Maitra S, Ray BR. Comparison of plasma concentration and sedative effect of sublingual and intranasal dexmedetomidine in children: A double-blind randomised controlled study. Acta Anaesthesiol Scand. 2025;69:e14583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 40. | Zhao Y, An D, Bi L. Effect of Co-Administration of Midazolam and Dexmedetomidine on Haemodynamics and Stress Response in Elderly Patients with Non-Small Cell Lung Cancer. J Invest Surg. 2025;38:2445587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 41. | MacMillan LB, Hein L, Smith MS, Piascik MT, Limbird LE. Central hypotensive effects of the alpha2a-adrenergic receptor subtype. Science. 1996;273:801-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 339] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 42. | Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93:382-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1056] [Cited by in RCA: 927] [Article Influence: 35.7] [Reference Citation Analysis (2)] |
| 43. | Lee K, Orme R, Williams D, Segal R. Prospective pilot trial of dexmedetomidine sedation for awake diagnostic flexible bronchoscopy. J Bronchology Interv Pulmonol. 2010;17:323-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 44. | Antonik LJ, Goldwater DR, Kilpatrick GJ, Tilbrook GS, Borkett KM. A placebo- and midazolam-controlled phase I single ascending-dose study evaluating the safety, pharmacokinetics, and pharmacodynamics of remimazolam (CNS 7056): Part I. Safety, efficacy, and basic pharmacokinetics. Anesth Analg. 2012;115:274-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 255] [Article Influence: 18.2] [Reference Citation Analysis (5)] |
| 45. | Zhou Y, Zhao C, Tang YX, Liu JT. Efficacy and safety of remimazolam in bronchoscopic sedation: A meta-analysis. World J Clin Cases. 2024;12:1120-1129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (2)] |
| 46. | Zhou YY, Yang ST, Duan KM, Bai ZH, Feng YF, Guo QL, Cheng ZG, Wu H, Shangguan WN, Wu XM, Wang CH, Chai XQ, Xu GH, Liu CM, Zhao GF, Chen C, Gao BA, Li LE, Zhang M, Ouyang W, Wang SY. Efficacy and safety of remimazolam besylate in bronchoscopy for adults: A multicenter, randomized, double-blind, positive-controlled clinical study. Front Pharmacol. 2022;13:1005367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 35] [Reference Citation Analysis (1)] |
| 47. | Sneyd JR. Remimazolam: new beginnings or just a me-too? Anesth Analg. 2012;115:217-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (3)] |
| 48. | Abdelgalil AS, Mohammed AM, Abdelrahman AS, Zayed NOA. Dexmedetomidine-propofol versus ketamine-propofol for sedation of cancer patients undergoing computerized tomography guided bone biopsy: a randomized double-blind trial. Anaesth Pain Intensive Care. 2024;28:809-815. [DOI] [Full Text] |
| 49. | Antony T, Acharya KV, Acharya PR, Kotian H, Kamath S. Clinical usefulness of nebulized dexmedetomidine for conscious sedation in daycare flexible bronchoscopy in Southern India. Indian J Pharmacol. 2024;56:105-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 50. | Akyurt D, Canikli Adigüzel Ş, Güllü Arslan N, Bahadir Altun H, Ültan Özgen G, Kömürcü Ö, Süren M, Tulgar S. Pre-procedure preparation, monitoring, premedication and sedation practices of bronchoscopists in Türkiye. Tuberk Toraks. 2024;72:239-246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 51. | Liu W, Ge X, Gao F, Kan Q, Wang S, Wang Y, He C. Safety and efficacy of dexmedetomidine vs. midazolam in complex gastrointestinal endoscopy: A systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2024;48:102315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 52. | Van de Vel G, Mun S, Zia SUD, Chalasani R, Shukla PS, Malasevskaia I. Impact of Ketamine and Propofol on Cognitive Function in Elderly Patients: A Systematic Review. Cureus. 2025;17:e79091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 53. | Ulutas H, Ucar M, Celik MR, Agar M, Gulcek I. Sedation with Propofol and Propofol-Ketamine (Ketofol) in Flexible Bronchoscopy: A Randomized, Double-Blind, Prospective Study. Niger J Clin Pract. 2023;26:1817-1823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 54. | Nie J, Chen W, Jia Y, Zhang Y, Wang H. Comparison of remifentanil and esketamine in combination with propofol for patient sedation during fiberoptic bronchoscopy. BMC Pulm Med. 2023;23:254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 55. | Matsumoto T, Kaneko A, Fujiki T, Kusakabe Y, Noda A, Tanaka A, Yamamoto N, Tashima M, Tashima N, Ito C, Aihara K, Yamaoka S, Mishima M. Prevalence and characteristics of disinhibition during bronchoscopy with midazolam. Respir Investig. 2022;60:345-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |