Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.115754
Revised: November 18, 2025
Accepted: January 23, 2026
Published online: June 20, 2026
Processing time: 181 Days and 17.6 Hours
Despite recent advancements in healthcare facilities, the early identification and assessment of pneumonia severity remain major challenges that affect clinical outcomes. These challenges emphasise the necessity for reliable biomarkers that can predict disease severity promptly and precisely. Unlike other markers like C-reactive protein or white blood cell count, procalcitonin (PCT) levels specifically increase in bacterial infections, which are a common cause of severe pneumonia in children.
To evaluate the predictive value of serum PCT in assessing pneumonia severity and clinical outcomes in under-five children.
This prospective observational study was conducted in a tertiary care teaching hospital in Eastern India from August 2022 to July 2024. Children aged 2 to 59 months admitted with pneumonia underwent serum PCT estimation on day 1 and day 5 of admission. The pneumonia severity was classified as follows: (1) Very severe (intubation, shock, or death); (2) Severe (intensive care admission without very severe features and/or high-flow nasal canula); (3) Moderate (hos
Two hundred children were included. Majority were < 6 months of age, and male (64%). Children in the viral pneumonia group were significantly younger. The pneumonia severity was as follows: (1) Very severe (18%); (2) Severe (24.5%); and (3) Moderate (57.5%). Eighteen (9%) children died. On day 1, the mean ± SD level of serum PCT level for different pneumonia severity was as follows: (1) Moderate (0.47 ± 0.42); (2) Severe (2.04 ± 1.5); and (3) Very severe (4.29 ± 2.37). The differences across the groups were statistically significant (P < 0.01). By day 5, the pattern of increasing PCT levels with greater disease severity persisted: (1) Moderate (0.26 ± 0.18); (2) Severe (3.14 ± 4.1); and (3) Very severe (9.51 ± 3.62). The differences observed on day 5 were statistically significant (P < 0.01). PCT levels were significantly higher in bacterial pneumonia compared to viral and atypical pneumonia. On day 1, the AUC of PCT in predicting mortality was 0.946 (95%CI: 0.905-0.973), and on day 5, the AUC was 0.948 (95%CI: 0.908-0.975).
The present study found that serum PCT can predict pneumonia severity including mortality in under-five children. The PCT elevation was significantly higher in bacterial pneumonia compared to viral and atypical pne
Core Tip: Severe pneumonia in children is commonly caused by bacteria. Unlike C-reactive protein, procalcitonin (PCT) levels specifically increase in bacterial infections. In this study, we included 200 children with pneumonia and investigated the role of PCT in predicting the severity and mortality due to pneumonia. The serum PCT level paralleled with the severity of pneumonia, and the day 5 level was higher than the day 1 level. PCT elevation was significantly higher in bacterial pneumonia compared to viral and atypical pneumonia. A higher level of PCT on day 5 predicted a longer duration of antibiotics use, prolonged hospitalization, and increased mortality.
- Citation: Kanungo SK, Hembram G, Sahu PK, Das RR. Serum procalcitonin as a predictor of severity of pneumonia in under-five children: A prospective observational study. World J Methodol 2026; 16(2): 115754
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/115754.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.115754
Pediatric community acquired pneumonia (CAP) is a common cause of under-five mortality and hospitalization, particularly in low-income and middle-income countries[1]. World Health Organization (WHO) has classified CAP into 3 categories: (1) No pneumonia; (2) Pneumonia; and (3) Severe/very severe pneumonia[2]. This classification is used to triage children at admission, so that appropriate treatment can be given. It is also important to predict the severity of CAP in children, so that timely referral or treatment can be delivered and mortality can be prevented. One such classification tool was developed from the Etiology of Pneumonia in the Community study[3,4]. In this study, altered mental status, chest indrawing, multilobar or interstitial infiltrates, decreased PaO2/FiO2 ratio, decreased systolic blood pressure, hypothermia, extremes of age, increased heart rate and respiratory rate predicted a more severe course in children with pneumonia. This study also suggested an ordinal outcome scale for childhood pneumonia severity, which is as follows: (1) Mild (discharge); (2) Moderate (hospitalization without severe or very severe features); (3) Severe (intensive care admission without very severe features and/or high-flow nasal cannula); and (4) Very severe (intubation, shock, or death).
Viruses also contribute to a significant proportion pneumonia case to children under-five, and it’s always been a difficult task to differentiate bacterial from viral pneumonia based on clinical or chest X ray findings. To help in such a situation, various biomarkers have been tested, and their use have been increased in last couple of years as point of care tools[5]. Physicians think that biomarkers may allow better identification and thus help in treatment of children with severe/very severe diseases. One such biomarker is procalcitonin (PCT), a peptide precursor of calcitonin[6]. Unlike other markers like C-reactive protein (CRP) or white blood cell (WBC) count, PCT levels specifically increase in bacterial infections, which are a common cause of severe pneumonia in children.
Though studies on PCT as a marker of bacterial infection have been published in children and adults, studies in children have been less conclusive in 7 studies[7-13]. In addition, there is a scarcity of studies in children using PCT to compare the outcomes based on ordinal scales (as mentioned above). The objective of the present study was to evaluate the role of PCT in predicting severity of pneumonia and its outcome severity in children, and to differentiate different etiologies (bacterial, viral, and atypical) of pneumonia.
This prospective observational study was conducted in the Pediatrics department (a 200 bedded hospital dedicated for children), which is attached to a tertiary care teaching institute in Eastern India. The study duration was from August 2022 to July 2024. The study was approved by the Institutional Ethics Committee. Written informed consents were taken from the parents or care givers. The study is reported as per the declaration of Helsinki.
Inclusion criteria included children of 2-59 months of age admitted to the Pediatric inpatient department for management of pneumonia, which was defined as per the WHO revised 2014 definition of pneumonia in children along with radi
After history and clinical examinations, the cases underwent a series of investigations including complete blood count, liver and renal function tests, blood culture, PCT measurement, and imaging (chest X ray). Serum PCT sample was collected on day 1 and day 5 of admission. Serum PCT analysis was done by ARCHITECT® B·R·A·H·M·S PCT™ which is a chemiluminescent microparticle immunoassay for the quantitative determination of PCT in human serum and plasma on the automated ARCHITECT analyzer from Abbott. A level of < 0.5 ng/mL was considered normal. Children were started empirically on injection Ceftriaxone. Any change or upgradation of antibiotics was done based on culture reports. Oxygen, oral or intravenous fluids, and supportive management were provided based on the clinical condition of the patients. Clinical monitoring was done to detect any deterioration.
All the data pertaining to demography, clinical presentation, final diagnosis, investigations, management, and outcome were recorded in the case record forms.
The sample size was estimated based on the expected diagnostic performance of serum procalcitonin (PCT) in predicting severe pneumonia. Previous studies have reported an area under the receiver operating characteristic curve (AUC) for PCT ranging from 0.85 to 0.95 for predicting severe bacterial infections in children[7-13]. Assuming an anticipated AUC of 0.90, a null hypothesis AUC of 0.70, a power of 80%, and a two-sided alpha error of 0.05, the minimum required sample size was calculated to be approximately 180 participants. Considering a potential attrition rate of 10%, the final sample size was increased to 200.
The data were entered into Microsoft excel sheet. The analysis was conducted using MS Excel, SPSS version 22 (IBM SPSS Statistics, NY, United States). Descriptive statistics were used to summarize demographic characteristics, clinical profiles, and serum PCT levels. Pearson or Spearman correlation analysis assessed the relationship between serum PCT levels and clinical severity. Receiver operating characteristic (ROC) curve analysis determined the sensitivity, specificity, and area under curve (AUC) of serum PCT levels in predicting mortality. Multivariate logistic regression analysis (including variables with P < 0.1 on univariate analysis) explored the independent association of serum biomarker levels with pneumonia severity and mortality, adjusting for potential confounders. P < 0.05 was considered as statistically significant.
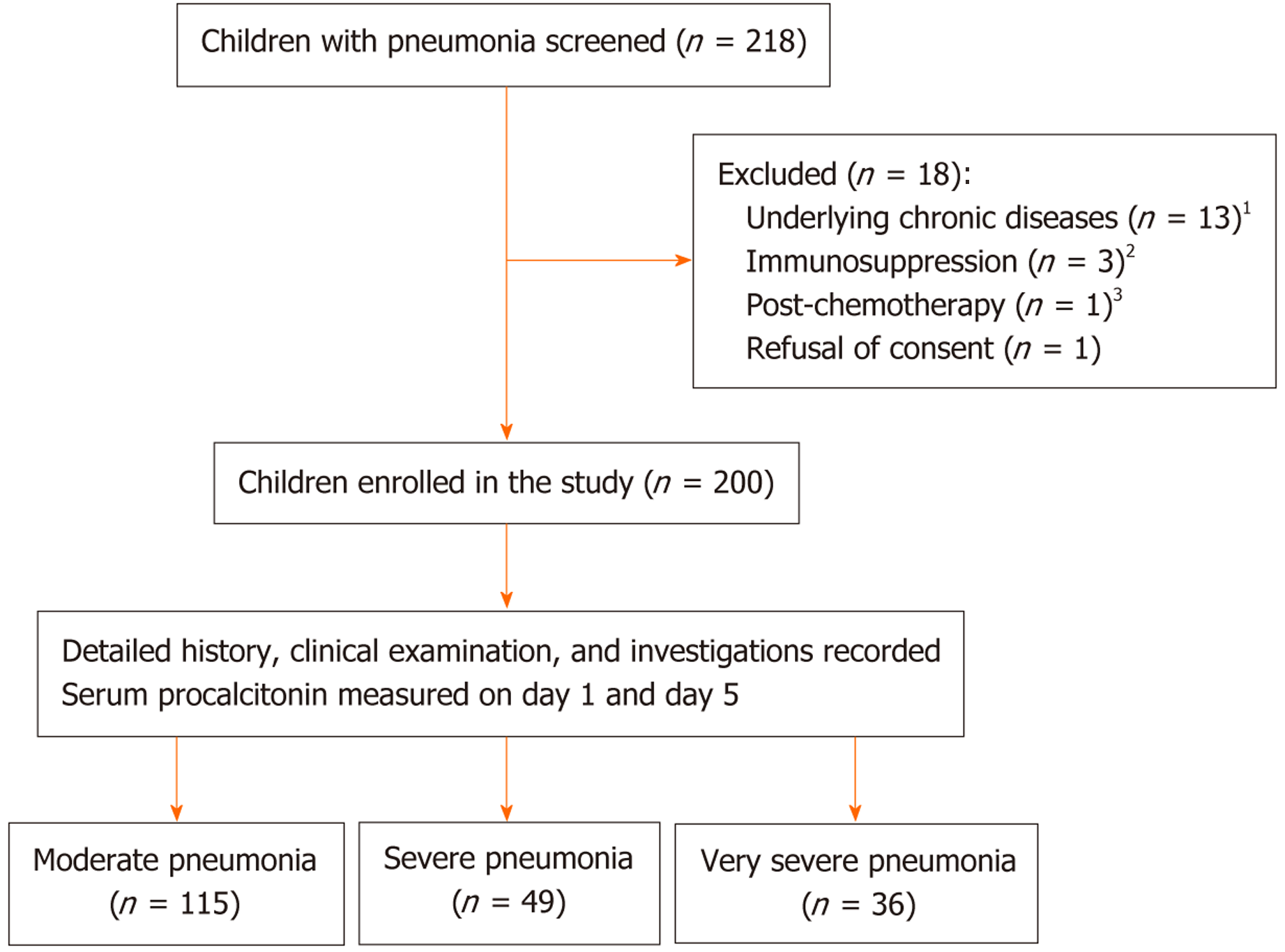
A total of 218 children with pneumonia were screened, and 18 were excluded. The details of the study flow have been mentioned in Figure 1. The baseline characteristics of the included children have been described in Table 1. The mean age was 11.4 months (majority were infants), and males constituted 64%. The mean duration of symptoms prior to hospitalization was 2.5 days. Nineteen (9.5%) children were severely malnourished, and 31 (15.5%) children were not immunized. All children had rapid breathing and chest retractions. Any danger sign as per WHO classification was present in was present in 159 (79.5%) children. Data on prior antibiotics use was available in 109 (54.5%) children. A subgroup analyses on different etiologies (bacterial viral, and atypical) of pneumonia was done and described in Table 1. Children in the viral pneumonia group were significantly younger (most were < 6 months), and those in atypical pneumonia group were significantly older (3-5 years).
| Characteristics | All patients (n = 200) | Bacterial (n = 39) | Viral (n = 15) | Atypical (n = 7) | P value |
| Age (months) | 11.4 ± 8.8 | 28.4 ± 7.2 | 9.7 ± 5.6 | 35.2 ± 6.9 | 0.03 |
| Age sub-groups | |||||
| ≤ 6 months | 96 (48) | 8 (20.5) | 9 (60) | 0 (0) | 0.021 |
| From 6 months to 1 year | 43 (21.5) | 14 (35.9) | 4 (26.7) | 0 (0) | 0.58 |
| > 1-5 year | 61 (31.5) | 17 (43.6) | 2 (13.3) | 7 (100) | 0.042 |
| Male | 128 (64) | 26 (66.7) | 9 (60) | 5 (71.4) | 0.65 |
| Duration of symptoms prior to admission (days) | 2.5 ± 0.8 | 2.3 ± 0.7 | 3.1 ± 0.4 | 3.2 ± 0.1 | 0.92 |
| Not immunized | 31 (15.5) | 7 (17.9) | 3 (20) | 2 (28.6) | 0.34 |
| Severe malnutrition | 19 (9.5) | 8 (20.5) | 2 (13.3) | 2 (28.6) | 0.27 |
| Clinical features | |||||
| Fever | 192 (96) | 37 (94.8) | 14 (93.3) | 6 (85.7) | 0.43 |
| Cough | 176 (88) | 32 (82) | 15 (100) | 6 (85.7) | 0.47 |
| Rapid breathing | 200 (100) | 39 (100) | 15 (100) | 7 (100) | 0.92 |
| Chest retractions | 200 (100) | 39 (100) | 15 (100) | 7 (100) | - |
| Any danger sign1 | 159 (79.5) | 34 (87.2) | 13 (86.7) | 5 (71.4) | - |
| Prior antibiotic use | 109 (54.5) | 24 (61.5) | 8 (53.3) | 4 (57.1) | 0.69 |
These are described in Table 2. Majority of the children had leucocytosis with neutrophilia. Elevated liver enzyme levels were noted in 12 (6%) children, which got resolved in children who survived. ESR and CRP were elevated in all children. A subgroup analysis on different etiologies (bacterial viral, and atypical) of pneumonia did not show significant differences in these laboratory parameters. PCT was measured on day 1 and day 5 of illness and was found to be elevated on both these days with day 5 level being higher compared to day 1 level. PCT levels were significantly higher in bacterial pneumonia compared to viral and atypical pneumonia (Table 3). Chest X ray showed consolidation in 73 (36.5%) children, and it was significantly higher in bacterial pneumonia (Table 3). An organism was isolated from blood culture in 39 (19.5%) children (most common were Staphylococcus aureus and Streptococcus pneumoniae). Mycoplasma serology was positive in 7 (3.5%) children. Nasopharyngeal aspirate PCR for virus isolation could be sent in 56 children, and a virus was isolated in 15 (27.7%) cases (most common was respiratory syncytial virus).
| Characteristics | All patients (n = 200) | Bacterial (n = 39) | Viral (n = 15) | Atypical (n = 7) | P value |
| Complete blood count | |||||
| Total leucocyte count (cumm) | 15417.4 ± 5551.1 | 17417.2 ± 3254.8 | 14226.4 ± 2119.6 | 13112.4 ± 1792.2 | 0.39 |
| Hemoglobin (g/dL) | 10.6 ± 1.8 | 10.9 ± 1.4 | 11.0 ± 1.6 | 11.1 ± 1.2 | 0.76 |
| Neutrophil (%) | 84 ± 6.4 | 84 ± 6.1 | 72 ± 7.3 | 73 ± 7.1 | 0.59 |
| Lymphocyte (%) | 31 ± 5.2 | 28 ± 5.4 | 33 ± 6.1 | 32 ± 4.8 | 0.84 |
| Platelet count (lakhs/cumm) | 2.4 ± 0.9 | 2.4 ± 1.5 | 2.7 ± 1.2 | 2.6 ± 1.2 | 0.91 |
| Liver function tests | |||||
| Total bilirubin (mg/dL) | 0.9 ± 0.2 | 1.1 ± 0.2 | 1.4 ± 0.3 | 0.8 ± 0.1 | 0.65 |
| Total protein (g/L) | 7.7 ± 0.9 | 7.7 ± 0.8 | 7.6 ± 1.1 | 7.6 ± 0.9 | 0.77 |
| Aspartate transaminase (U/L) | 41 ± 8.4 | 36 ± 7.9 | 45 ± 8.1 | 44 ± 7.7 | 0.44 |
| Alanine transaminase (U/L) | 37 ± 6.5 | 36 ± 6.7 | 39 ± 6.1 | 38 ± 6.6 | 0.82 |
| Renal function tests | |||||
| Urea (mg/dL) | 29 ± 8.4 | 29 ± 8.1 | 27 ± 9.1 | 28 ± 8.8 | 0.69 |
| Creatinine (mg/dL) | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.93 |
| Inflammatory markers | |||||
| Erythrocyte sedimentation rate (mm/hour) | 28 ± 12 | 23 ± 11 | 32 ± 8 | 26 ± 10 | 0.46 |
| C-reactive protein (mg/L) | 22 ± 9 | 42 ± 6 | 28 ± 7 | 20 ± 5 | 0.37 |
| Chest X ray findings | |||||
| Heterogenous opacities/infiltrates | 106 (53) | 14 (35.9) | 13 (86.7) | 6 (85.7) | 0.07 |
| Consolidation | 73 (36.5) | 25 (64.1) | 2 (13.3) | 1 (14.3) | P < 0.01 |
| Duration of antibiotics use | |||||
| 5-10 days | 142 (71) | 26 (66.7) | 15 (100) | 7 (100) | - |
| > 10 days | 58 (29) | 13 (33.3) | 0 (0) | 0 (0) | 0.002 |
| Type of respiratory support | |||||
| Nasal cannula/face mask | 200 (100) | 39 (100) | 15 (100) | 7 (100) | - |
| Continuous positive airway pressure | 43 (21.5) | 7 (17.9) | 3 (13.3) | 1 (14.3) | 0.63 |
| Heated humidified high flow nasal cannula | 40 (20) | 15 (38.5) | 6 (40) | 3 (42.9) | 0.38 |
| Mechanical ventilation | 36 (18) | 6 (15.4) | 4 (26.6) | 1 (14.3) | 0.71 |
| Pneumonia severity types | |||||
| Moderate | 115 (57.5) | 19 (48.7) | 9 (60) | 5 (71.4) | 0.33 |
| Severe | 49 (24.5) | 13 (33.3) | 4 (26.7) | 1 (14.3) | 0.27 |
| Very severe | 36 (18) | 7 (18) | 2 (13.3) | 1 (14.3) | 0.48 |
| Outcomes | |||||
| Discharged | 182 (91) | 33 (84.6) | 13 (86.7) | 7 (100) | 0.51 |
| Died | 18 (9) | 6 (15.4) | 2 (13.3) | 0 | 0.42 |
| Severity distribution as per the etiology | Serum procalcitonin levels (ng/mL) | P value | |
| Day 1 | Day 5 | ||
| Moderate | 0.47 ± 0.42 | 0.26 ± 0.18 | 0.09 |
| Bacterial | 2.14 ± 0.77 | 1.92 ± 0.85 | 0.06 |
| Viral | 0.32 ± 0.17 | 0.41 ± 0.19 | 0.29 |
| Atypical | 0.31 ± 0.14 | 0.37 ± 0.15 | 0.47 |
| Severe | 2.04 ± 1.5 | 3.14 ± 4.1 | < 0.01 |
| Bacterial | 7.38 ± 1.01 | 9.56 ± 3.92 | < 0.01 |
| Viral | 0.45 ± 0.16 | 0.49 ± 0.17 | 0.27 |
| Atypical | 0.38 ± 0.15 | 0.41 ± 0.17 | 0.41 |
| Very severe | 4.21 ± 2.37 | 7.46 ± 3.62 | < 0.01 |
| Bacterial | 14.86 ± 2.19 | 19.27 ± 3.54 | < 0.01 |
| Viral | 0.54 ± 0.22 | 0.72 ± 0.18 | 0.25 |
| Atypical | 0.41 ± 0.19 | 0.55 ± 0.16 | 0.34 |
These are described in Table 2. All the children received supportive therapy, oxygen, respiratory support, and antibiotics as per the hospital protocol. Majority of the children (71%) required antibiotics of 5-10 days duration, and rest required for > 10 days. All children requiring antibiotics for > 10 days belonged to the bacterial pneumonia group, and this was statistically significant. Thirty-six (18%) children developed respiratory failure and required mechanical ventilation. On distribution as per the severity of illness, the majority (57.5%) belonged to moderate group followed by severe (24.5%), and very severe (18%). No difference in the severity was found among different etiologies (bacterial, viral and atypical) of pneumonia. Mortality was observed in 18 (9%) children. No difference was found among different etiologies of pne
Correlation of PCT level with CRP levels: (1) A moderate positive correlation (Pearson’s r = 0.444, P < 0.01) indicating that higher CRP levels on day 1 are associated with higher PCT levels on day 1; and (2) A strong positive correlation (Pearson’s r = 0.842, P < 0.001) suggesting that higher CRP levels on day 5 are associated with higher PCT levels on day 5. This correlation was seen in case of bacterial pneumonia, and overall causes of pneumonia.
Correlation of PCT level with duration of hospital stay: The mean ± SD serum PCT level on day 1 in the group treated with antibiotics for > 10 days was 3.48 ± 1.86 ng/mL. In contrast, the group treated for 5-10 days had a significantly lower PCT level of 0.71 ± 1.24 ng/mL (P < 0.01). By day 5, the PCT level in the group treated with > 10 days of antibiotics increased to 7.46 ± 4.92 ng/mL. However, PCT level was still at a lower level in the group treated with 5-10 days of antibiotics with 0.51 ± 1.61 ng/mL (P < 0.01). This correlation was seen in case of bacterial pneumonia, and overall causes of pneumonia.
Comparison of serum PCT level as per the illness severity distribution: This has been described in Table 3. As can be seen, the PCT level significantly correlated with the severity of illness distribution in the included children with the severe and very severe illness showing a statistically significant increase in PCT levels on day 5 compared to day 1. This correlation was seen in case of bacterial pneumonia, and overall causes of pneumonia.
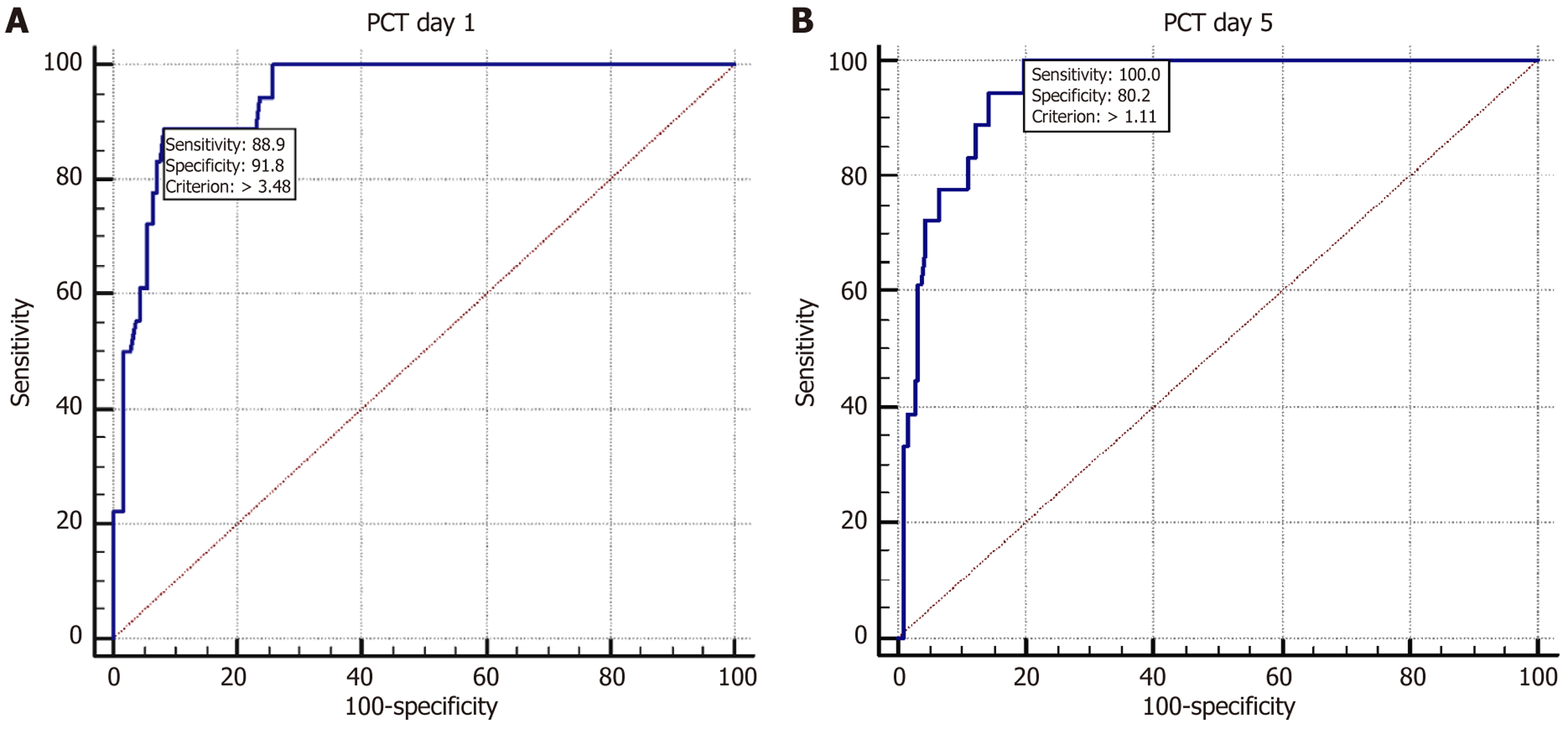
PCT levels on day 1: On multivariate logistic regression analysis, a higher serum PCT level on day 1 was independently associated with increased odds of severe pneumonia and mortality. A cut-off PCT level of > 3.48 on day 1 had a sensitivity of 88.9%, specificity of 91.8% and AUC of 0.946 (95%CI: 0.905-0.973) in predicting subsequent mortality (Figure 2A).
PCT levels on day 5: On multivariate logistic regression analysis, a higher serum PCT level on day 5 was independently associated with increased odds of severe pneumonia and mortality. A cut-off PCT level of > 1.11 on day 5 had a sensitivity of 100%, specificity of 80.2% and AUC of 0.948 (95%CI: 0.908-0.975) in predicting subsequent mortality (Figure 2B).
In the present study, 200 children (majority being < 6 months of age) with pneumonia were included. The serum PCT level paralleled with the severity of pneumonia, and the day 5 level was higher than the day 1 level. A higher day 5 PCT level predicted a longer duration of antibiotics use, prolonged hospitalization, and increased mortality. The latter was significantly seen in case of bacterial pneumonia and overall causes of pneumonia.
The published results on the role of PCT in predicting pneumonia and its outcomes have been variable. In a multicenter prospective from India, serum PCT was measured in 370 patients (median age, 12 months)[13]. The authors found that a raised PCT level was significantly associated with radiographic features of consolidation and complicated pneumonia. In addition, the PCT level was significantly higher in children with severe pneumonia as per the WHO classification. In another prospective study from United States including 477 children, the authors did not find any statistically significant differences in the median total leucocyte count, absolute neutrophil count, CRP, and PCT across pneumonia severity categories[9]. None of these biomarkers could discriminate adequately between severe and non-severe diseases as well as suspected and radiographic pneumonia. However, CRP and PCT were much higher in children with empyema requiring chest drainage and children with pneumonia and septic shock. The authors concluded that CRP and PCT may have some utility in predicting the most severe pneumonia outcomes. In the present study, PCT levels were significantly higher in bacterial pneumonia, which supports the previous study findings emphasizing the fact that a higher initial and serial rising PCT levels favors bacterial over viral and atypical pneumonia diagnosis.
In the present study, majority (57.5%) of the children had moderate followed by severe (24.5%), and very severe (18%) disease. A study from United States included 488 children with pneumonia, of which 30 (6%) were classified as very severe, 106 (22%) as severe, 327 (67%) as moderate, and 25 (5%) as mild disease[10]. The authors correlated serum PCT level with severity of the diseases and found that a higher level of PCT is associated with increased severity of the dis
Serum PCT levels reflect the systemic inflammatory response to bacterial infection. Elevated PCT levels in severe pneumonia likely reflect the extent of pulmonary and systemic inflammation, bacterial load, and cytokine-mediated stimulation of calcitonin gene expression in extrathyroidal tissues. A key strength of PCT lies in its kinetics – rapid elevation within 6-12 hours of bacterial infection onset and a short half-life of 24-36 hours following adequate control of infection. This dynamic profile allows PCT to reflect not only disease severity at presentation but also the trajectory of clinical improvement or deterioration. In our cohort, children with persistently elevated PCT levels on day 5 tended to have more severe disease and adverse outcomes. Conversely, those showing early PCT decline had faster clinical reco
When comparing PCT with CRP, our study found that PCT had a stronger correlation with clinical severity indices. CRP, while widely used, showed greater variability and weaker discrimination between severe and non-severe (mo
Despite these promising findings, it is important to acknowledge certain limitations. First, the study sample size, although adequate for detecting associations, was relatively small for definitive conclusions regarding cutoff values or predictive thresholds. Larger, multicenter studies are necessary to validate the optimal PCT cutoff levels for classifying pneumonia severity in children across different age groups and etiologies. Second, viral infections were identified in some children, and they could have influenced PCT values, potentially leading to variability in results. Third, PCT measurement is relatively costly and not universally available in resource-limited settings, which may restrict its widespread adoption. Cost-effectiveness studies are warranted to evaluate its feasibility in low-and middle-income countries where childhood pneumonia burden remains high. Fourth, potential overlap in PCT levels between moderate bacterial pneumonia and severe viral infections with secondary bacterial colonization. Although PCT is typically low in viral infections, severe viral pneumonias, such as those caused by influenza or respiratory syncytial virus, can elicit modest PCT elevations due to cytokine activation and secondary bacterial infection. Hence, while PCT serves as a helpful adjunct, it should not replace clinical judgment or microbiological confirmation. Rather, it should be interpreted in conjunction with clinical features, radiographic findings, and other laboratory parameters. Fifth, it would have been ideal to include only patients with bacterial infections and exclude those with viral, mycoplasma, and other non-bacterial infections.
Serial PCT monitoring may be more informative than a single measurement at admission. Higher serum PCT levels correlate strongly with pneumonia severity and adverse clinical outcomes in children. PCT, therefore, holds promise as an objective biomarker for early severity prediction, prognosis, and potentially antibiotic stewardship in pediatric pneumonia. Future large-scale prospective studies are warranted to refine threshold values, validate clinical algorithms incorporating PCT, and assess cost-effectiveness in diverse healthcare settings.
| 1. | Marangu D, Zar HJ. Childhood pneumonia in low-and-middle-income countries: An update. Paediatr Respir Rev. 2019;32:3-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 83] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 2. | Revised WHO Classification and Treatment of Pneumonia in Children at Health Facilities: Evidence Summaries. Geneva: World Health Organization; 2014. [PubMed] |
| 3. | Bradley JS, Byington CL, Shah SS, Alverson B, Carter ER, Harrison C, Kaplan SL, Mace SE, McCracken GH Jr, Moore MR, St Peter SD, Stockwell JA, Swanson JT; Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis. 2011;53:e25-e76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1331] [Cited by in RCA: 1168] [Article Influence: 77.9] [Reference Citation Analysis (0)] |
| 4. | Williams DJ, Zhu Y, Grijalva CG, Self WH, Harrell FE Jr, Reed C, Stockmann C, Arnold SR, Ampofo KK, Anderson EJ, Bramley AM, Wunderink RG, McCullers JA, Pavia AT, Jain S, Edwards KM. Predicting Severe Pneumonia Outcomes in Children. Pediatrics. 2016;138:e20161019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 113] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 5. | Thomas J, Pociute A, Kevalas R, Malinauskas M, Jankauskaite L. Blood biomarkers differentiating viral versus bacterial pneumonia aetiology: a literature review. Ital J Pediatr. 2020;46:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 6. | Breimer LH, MacIntyre I, Zaidi M. Peptides from the calcitonin genes: molecular genetics, structure and function. Biochem J. 1988;255:377-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 135] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 7. | Agnello L, Bellia C, Di Gangi M, Lo Sasso B, Calvaruso L, Bivona G, Scazzone C, Dones P, Ciaccio M. Utility of serum procalcitonin and C-reactive protein in severity assessment of community-acquired pneumonia in children. Clin Biochem. 2016;49:47-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 8. | Toikka P, Irjala K, Juvén T, Virkki R, Mertsola J, Leinonen M, Ruuskanen O. Serum procalcitonin, C-reactive protein and interleukin-6 for distinguishing bacterial and viral pneumonia in children. Pediatr Infect Dis J. 2000;19:598-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 166] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 9. | Florin TA, Ambroggio L, Brokamp C, Zhang Y, Rattan M, Crotty E, Belsky MA, Krueger S, Epperson TN 4th, Kachelmeyer A, Ruddy R, Shah SS. Biomarkers and Disease Severity in Children With Community-Acquired Pneumonia. Pediatrics. 2020;145:e20193728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 106] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 10. | Sartori LF, Zhu Y, Grijalva CG, Ampofo K, Gesteland P, Johnson J, McHenry R, Arnold DH, Pavia AT, Edwards KM, Williams DJ. Pneumonia Severity in Children: Utility of Procalcitonin in Risk Stratification. Hosp Pediatr. 2021;11:215-222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 11. | Rezk A, Bakry N, Elfiky S, Metawaa M, Ibrahim A. Diagnostic and prognostic utility of salivary and serum procalcitonin, interleukin-6, and interleukin-10 in pediatric pneumonia: a prospective case-control study. Front Pediatr. 2025;13:1627451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 12. | Omaggio L, Franzetti L, Caiazzo R, Coppola C, Valentino MS, Giacomet V. Utility of C-reactive protein and procalcitonin in community-acquired pneumonia in children: a narrative review. Curr Med Res Opin. 2024;40:2191-2200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 13. | Ratageri VH, Panigatti P, Mukherjee A, Das RR, Goyal JP, Bhat JI, Vyas B, Lodha R, Singhal D, Kumar P, Singh K, Mahapatro S, Charoo BA, Kabra SK, Jat KR. Role of procalcitonin in diagnosis of community acquired pneumonia in Children. BMC Pediatr. 2022;22:217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Baumann P, Baer G, Bonhoeffer J, Fuchs A, Gotta V, Heininger U, Ritz N, Szinnai G, Bonhoeffer J. Procalcitonin for Diagnostics and Treatment Decisions in Pediatric Lower Respiratory Tract Infections. Front Pediatr. 2017;5:183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Li Y, Min L, Zhang X. Usefulness of procalcitonin (PCT), C-reactive protein (CRP), and white blood cell (WBC) levels in the differential diagnosis of acute bacterial, viral, and mycoplasmal respiratory tract infections in children. BMC Pulm Med. 2021;21:386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 16. | Yin H, Mo S. Value of combined detection of serum amyloid A, C-reactive protein and procalcitonin in differential diagnosis of respiratory tract infection in children of China. Ann Med. 2022;54:1732-1737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Khan DA, Rahman A, Khan FA. Is procalcitonin better than C-reactive protein for early diagnosis of bacterial pneumonia in children? J Clin Lab Anal. 2010;24:1-5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |