Copyright: ©Author(s) 2026.
World J Methodol. Jun 20, 2026; 16(2): 117099
Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.117099
Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.117099
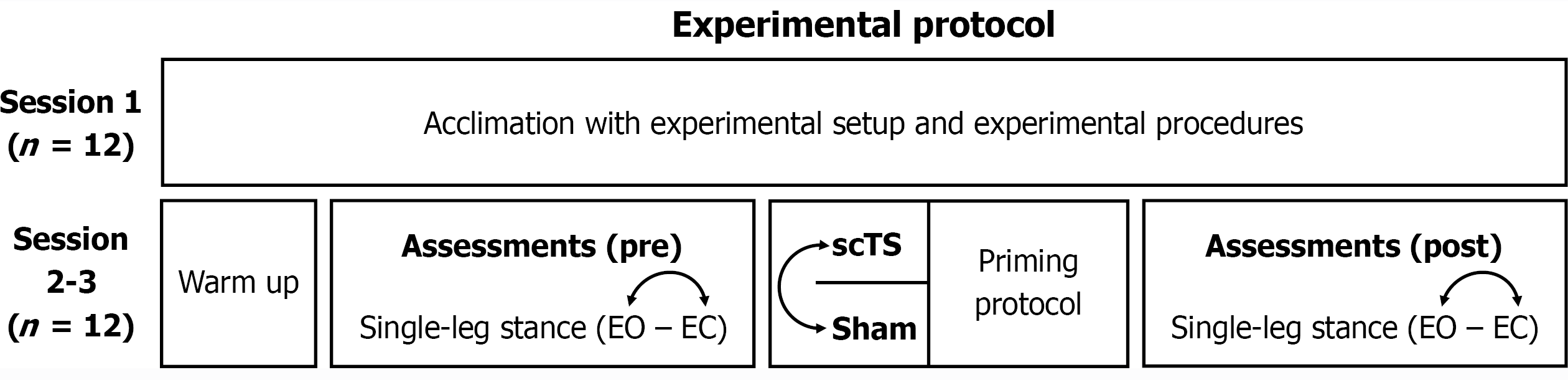
Figure 1 Overview of the experimental protocol.
All subjects enrolled (n = 12) completed the experimental protocol. In sessions 2-3, testing of spinal cord transcutaneous stimulation or sham stimulation priming, as well as the assessments of single-leg stance with eyes open and eyes closed conditions, was proposed in a randomized order. scTS: Spinal cord transcutaneous stimulation; Sham: Sham stimulation; EO: Eyes open; EC: Eyes closed.
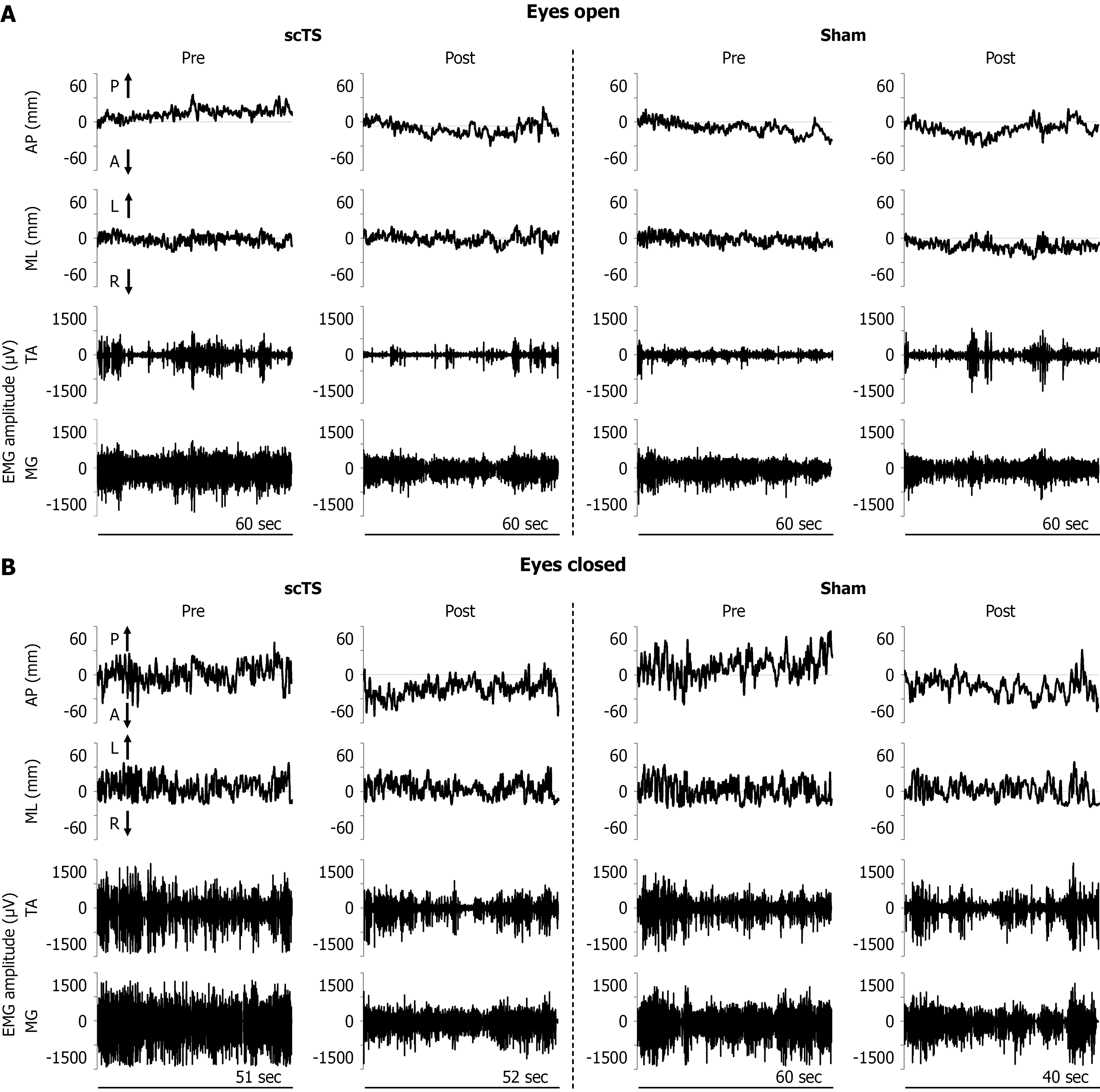
Figure 2 Exemplary time course of center of pressure displacement and electromyographic activity.
A: Time course of the center of pressure displacements in the anterior-posterior and medio-lateral directions, as well as electromyographic activity of tibialis anterior and medial gastrocnemius, during a representative single-leg stance performed before and after the priming protocol with spinal cord transcutaneous stimulation or sham stimulation with eyes open; B: Time course of the center of pressure displacements in the anterior-posterior and medio-lateral directions, as well as electromyographic activity of tibialis anterior and medial gastrocnemius, during a representative single-leg stance performed before and after the priming protocol with spinal cord transcutaneous stimulation or sham stimulation with eyes closed. scTS: Spinal cord transcutaneous stimulation; Sham: Sham stimulation; EMG: Electromyographic; AP: Anterior-posterior; ML: Medio-lateral; TA: Tibialis anterior; MG: Medial gastrocnemius; P: Posterior; A: Anterior; L: Left; R: Right.
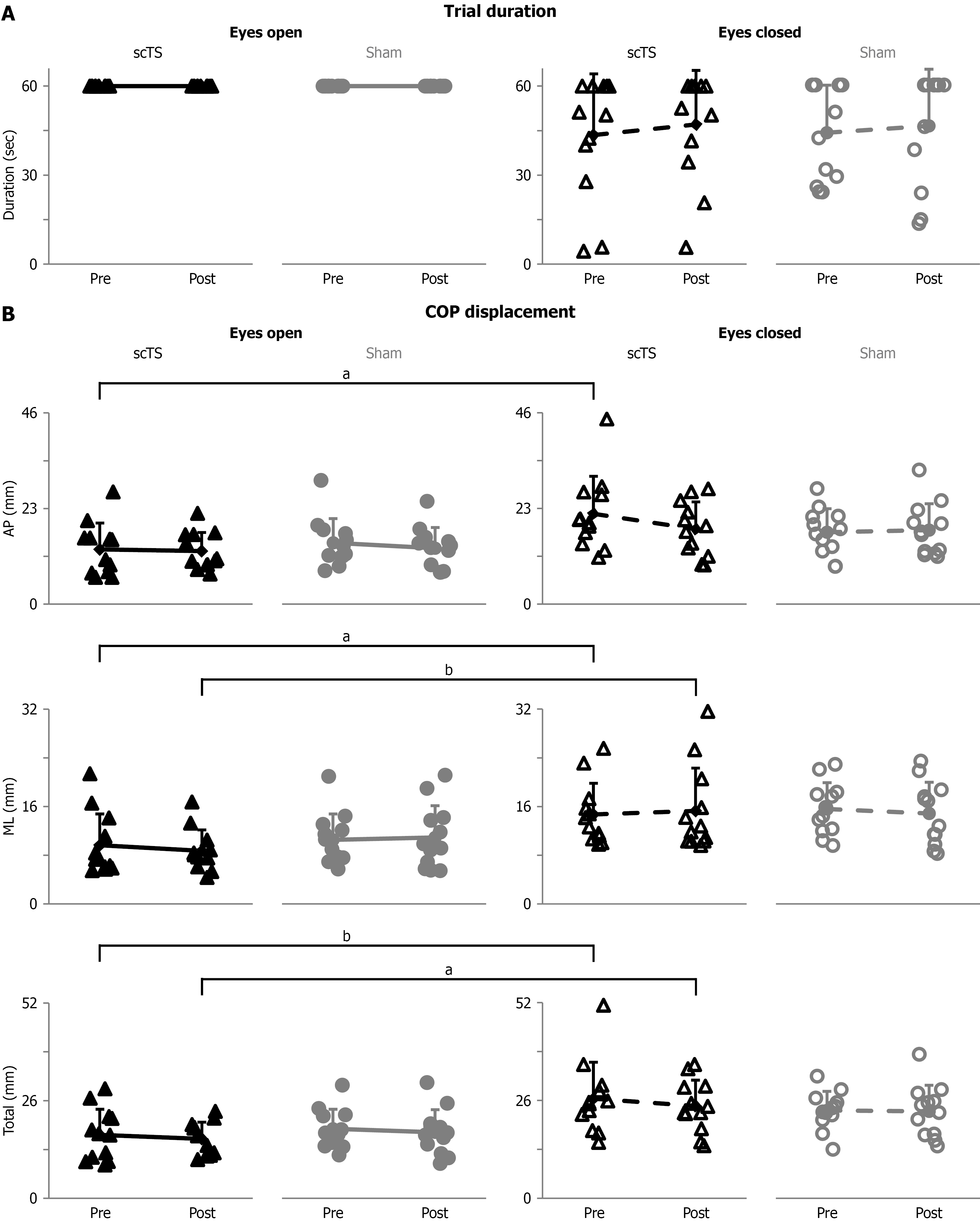
Figure 3 Group data of trial duration and center of pressure displacement.
A: Trial duration (seconds) for single-leg stance attempts with eyes open and eyes closed, before and after the priming protocol with spinal cord transcutaneous stimulation (scTS) or sham stimulation (sham; P < 0.05); B: Center of pressure displacement in the anterior-posterior direction (P < 0.01), medio-lateral direction (P < 0.001), and composite total displacement (quantified as root mean square; P < 0.001) for single-leg stance attempts with eyes open and eyes closed, before and after the priming protocol with spinal cord transcutaneous stimulation or sham. Results are described as individual data points (solid black triangles: ScTS, eyes open; solid grey circles: Sham, eyes open; empty black triangles: ScTS, eyes closed; empty grey circles: Sham, eyes closed) as well as mean and standard deviation. Significant difference by Bonferroni post hoc test: aP < 0.05; bP < 0.01. scTS: Spinal cord transcutaneous stimulation; Sham: Sham stimulation; AP: Anterior-posterior; ML: Medio-lateral; COP: Center of pressure.
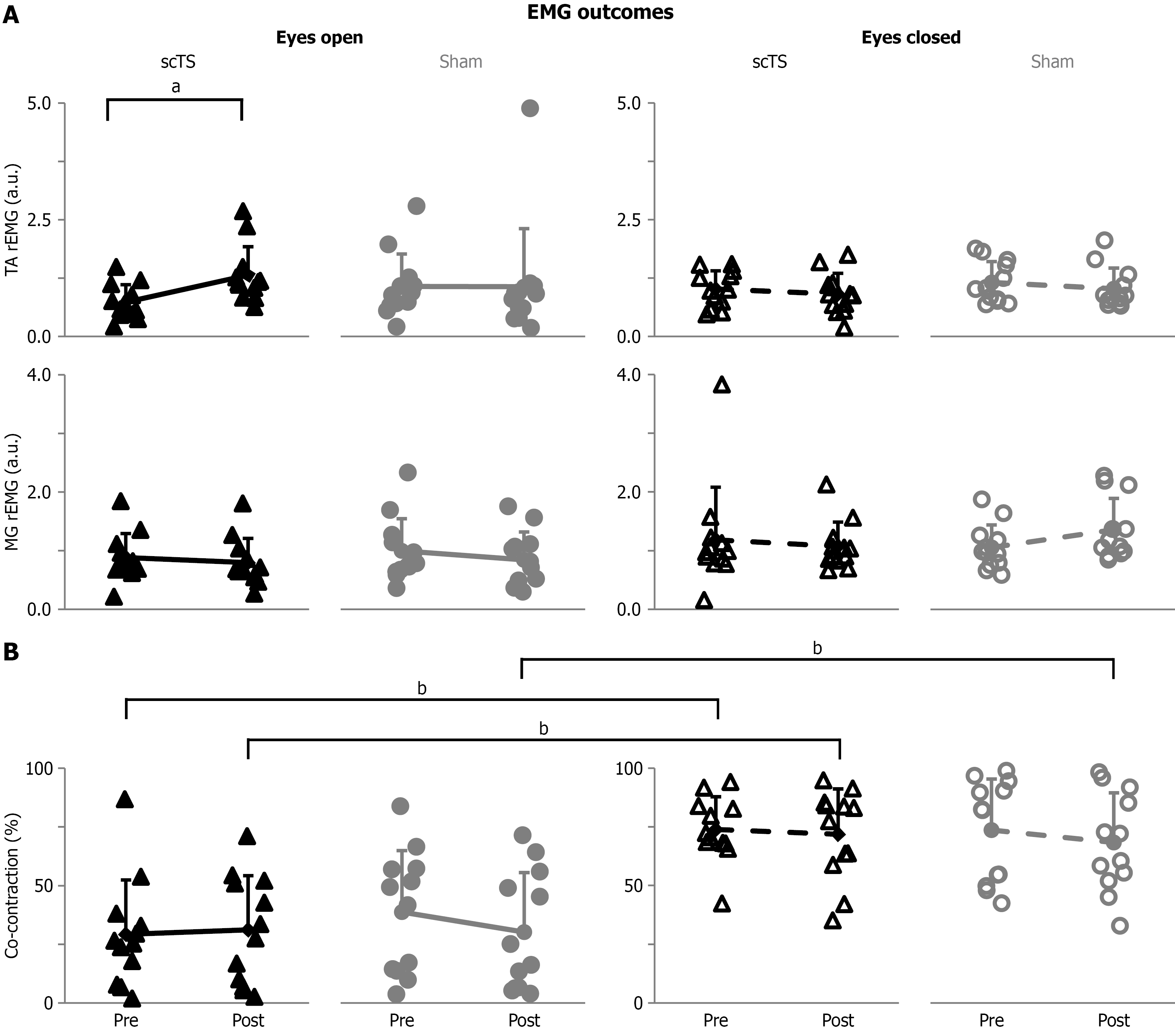
Figure 4 Group data of electromyographic activity.
A: Ratio of electromyography (EMG) amplitude assessed at the end of each trial by the EMG amplitude detected at the start of the trial for the tibialis anterior (P < 0.05) and medial gastrocnemius (P < 0.01). Ratio of EMG are expressed in arbitrary units; B: Single-leg stance duration in which tibialis anterior and medial gastrocnemius muscles were active at the same time above their respective 10% maximal voluntary contraction (co-contraction; P < 0.001). Both metrics are reported for the single-leg stance attempts with eyes open and eyes closed, before and after the priming protocol with spinal cord transcutaneous (scTS) or sham stimulation (sham). Results are described as individual data points (solid black triangles: ScTS, eyes open; solid grey circles: Sham, eyes open; empty black triangles: ScTS, eyes closed; empty grey circles: Sham, eyes closed) as well as mean and standard deviation. Significant difference by Bonferroni post hoc test: aP < 0.05; bP < 0.01. EMG: Electromyographic; scTS: Spinal cord transcutaneous stimulation; Sham: Sham stimulation; TA: Tibialis anterior; MG: Medial gastrocnemius; a.u.: Arbitrary unit; rEMG: Ratio of electromyography.
- Citation: Zaccaron S, D’Alleva M, Mari L, Stafuzza J, Lazzer S, Rejc E. Effects of spinal cord transcutaneous stimulation priming on single-leg balance control. World J Methodol 2026; 16(2): 117099
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/117099.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.117099