Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.115252
Revised: November 19, 2025
Accepted: January 16, 2026
Published online: March 25, 2026
Processing time: 152 Days and 21.7 Hours
Hyponatremia remains the most common electrolyte disorder in clinical practice, with a wide range of causes ranging from endocrine dysregulation to iatrogenic causes. Recent developments in point-of-care ultrasonography, vasopressin physiology, and osmoregulation have transformed diagnostics by emphasizing accuracy above purely clinical judgment. Although traditional management based on fluid restriction and hypertonic solutions remain the cornerstone of treatment, a careful and cautious monitoring is required to avoid osmotic demyelination and overcorrection. The Furst equation, a validated predictor of response to fluid restriction, should be integrated into routine practice, together with objective tools such as point-of-care ultrasonography, to improve the accuracy of volume assessment and guide more precise therapeutic decisions. Likewise, different therapeutic options have been studied for non-responders to fluid restriction, including urea, desmopressin, and selective vasopressin receptor antagonists (vaptans), showing promising results. Ongoing developments in artificial intelligence and laboratory-based decision support tools are expected to improve personalized management and predictive accuracy, potentially reconciling the disparity between evidence and clinical practice.
Core Tip: Hyponatremia, defined as a low concentration of sodium in the blood, is the most frequent electrolyte disorder in clinical practice. Its causes are diverse and often overlap, from hormonal imbalances to medication effects or underlying heart, liver, and kidney conditions. Effective and safe correction depends on accurately identifying whether the body has too much or too little water, rather than simply restricting fluids. Bedside ultrasound (point-of-care ultrasonography) and simple laboratory tools such as the Furst equation allow clinicians to assess fluid status objectively, predict response to fluid restriction, and guide personalized treatment strategies that reduce complications and improve patient outcomes.
- Citation: Martínez-Sánchez FD, Gutierrez-Rosas LE, Barranco-Hernandez LH, Gonzalez-Alvarez G, Bastida-Castro LA, Rocha-Haro A, Barrientos-Cabrera G, Martínez-Cabrera CF, Balderas-Juarez J, Salinas-Ramirez MA, Hernandez-Castillo JL. Hyponatremia: Evolving diagnostics and emerging therapeutics in clinical practice. World J Nephrol 2026; 15(1): 115252
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/115252.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.115252
Hyponatremia, classically defined as a serum sodium concentration below 135 mmol/L, represents the most frequent disturbance of fluid and electrolyte balance encountered in clinical practice[1,2]. Rather than a discrete disease entity, it reflects an underlying pathophysiologic alteration in water homeostasis[2]. Dysnatremias in general are among the most common electrolyte disorders presenting in emergency care, with hyponatremia consistently identified as the leading abnormality in the United States[1,3]. Although data is scarce, approximately 3 million to 6 million patients each year are evaluated for hyponatremia, and sodium derangements are present in 12%-20% of individuals admitted through the emergency department[2,4].
The clinical significance of sodium imbalance lies in its role as the predominant extracellular cation and the principal determinant of plasma osmolality[5]. Disruptions in sodium concentration, therefore, have immediate implications for osmotic equilibrium and ion distribution, with potential consequences for organ function, most notably within the central nervous system[2,5].
Given its high prevalence, diagnostic complexity, and potential for severe clinical consequences, hyponatremia remains a condition of significant concern across multiple disciplines[1]. Recent years have witnessed advances in diagnostic tools and the development of novel therapeutic strategies, yet the optimal management of this disorder continues to be debated. This narrative review aims to explore evolving diagnostic approaches and highlight emerging therapeutic interventions, emphasizing their potential implications in everyday clinical practice.
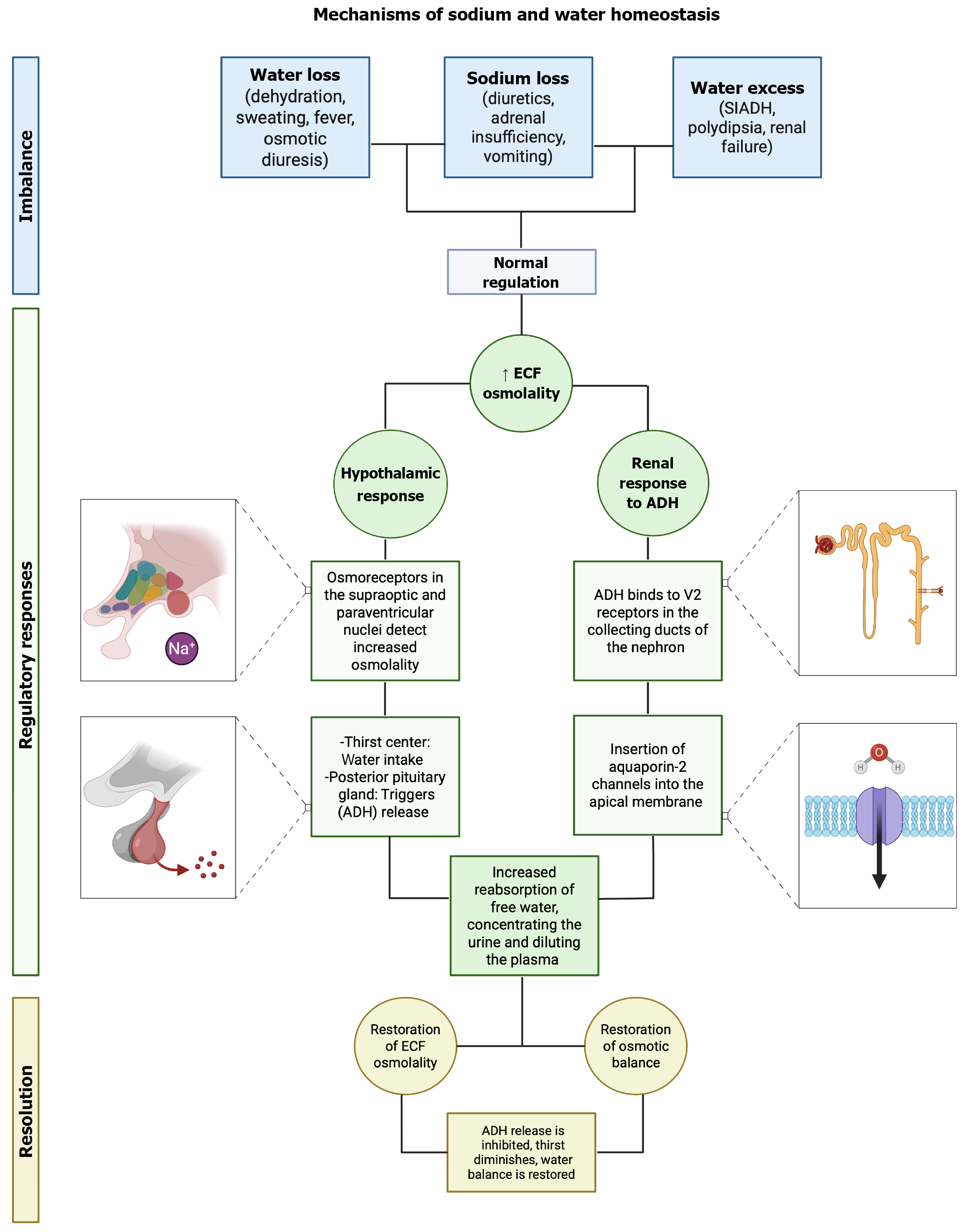
In healthy individuals, water balance is tightly regulated, such that plasma osmolality varies by no more than 1%-2% under normal conditions, provided there is unrestricted access to water[5,6]. The preservation of extracellular fluid (ECF) tonicity is crucial for human physiology and is achieved through a complex process of osmoregulation that integrates thirst, fluid intake, and the secretion and action of arginine vasopressin (AVP)[5,7]. In hyponatremia, alterations in these regulatory mechanisms disrupt water and sodium homeostasis, resulting in clinically significant imbalances (Figure 1).
Under normal circumstances, plasma osmolality is maintained within a narrow range of 275-295 mmol/kg[2,8]. Variations in water balance independent of solute concentration directly influence osmolality. For instance, water loss from the ECF increases osmolality, driving water movement from the intracellular compartment to the extracellular space[5,9]. Even slight elevations in osmolality activate hypothalamic osmoreceptors, stimulating the thirst center and leading to increased water intake[10,11]. Simultaneously, hypothalamic osmoreceptor activation triggers the release of AVP from the posterior pituitary, thereby enhancing renal water reabsorption[11].
Plasma osmolality is sensed by specialized osmoreceptor neurons in the anterior hypothalamus[11-13]. Fenestrations in the blood-brain barrier allow the circumventricular organs, particularly the subfornical organ and the organum vasculosum lamina terminalis, to be directly exposed to plasma[14,15]. These structures project to hypothalamic nuclei such as the supraoptic nucleus and paraventricular nucleus. An increase in osmolality depolarizes these neurons, leading to the synthesis and secretion of AVP, along with its stable surrogate peptide, copeptin[5,16,17].
AVP is a hypothalamic peptide with both antidiuretic and vasopressor properties[5,15]. It mediates its effects through three receptor subtypes, all of which are members of the G protein-coupled receptor family. The V1 receptor, expressed in vascular smooth muscle, activates the Gq/11-phospholipase C pathway, increasing intracellular calcium and producing vasoconstriction, which at higher AVP concentrations translates into a strong vasopressor response[18].
The V2 receptor, localized primarily in the distal nephron and collecting ducts, regulates aquaporin-2 trafficking and thus plays a central role in water reabsorption and urine concentration[5,19]. Once AVP is released into the circulation, it exerts its principal action on the kidney, where binding to V2 receptors on the basolateral membrane of collecting duct cells promotes the insertion of aquaporin-2 water channels into the apical membrane[18]. This increases tubular water permeability, allowing water reabsorption and urine concentration up to approximately 1200 mmol/kg. The osmoregulatory system is highly sensitive, as AVP secretion can change in response to variations in plasma osmolality as small as 1%[12,18].
Finally, the V3 (or V1b) receptor, expressed in anterior pituitary corticotrophs, potentiates adrenocorticotropic hormone release in synergy with corticotropin-releasing hormone, an effect tightly controlled by glucocorticoid feedback[17,18]. Together, these mechanisms maintain plasma osmolality within narrow physiological limits. In hyponatremia, disruption of this finely tuned system, whether due to inappropriate AVP secretion, impaired water excretion, or excessive intake, underpins the pathophysiological basis of the disorder[2,5].
Moreover, understanding AVP’s role has direct therapeutic implications. The development of V2 receptor antagonists, such as tolvaptan and conivaptan, represents a targeted approach to correct water retention and hyponatremia, emphasizing the translation of physiological knowledge into clinical practice[1]. In essence, hyponatremia reflects an excess of water relative to sodium rather than actual sodium depletion. This imbalance results from non-osmotic AVP secretion, impaired renal free-water excretion, or excessive intake, all of which lead to hypotonic plasma. The pathophy
The classification of hyponatremia relies on three main aspects: Volume status, time course, and symptom severity, which collectively guide both diagnosis and treatment decisions[1,2,20]. Volume status remains the cornerstone of etiological classification[2]. In hypovolemic hyponatremia, sodium and water are lost simultaneously, but sodium losses predominate, leading to ECF contraction[5]. Typical extrarenal causes include gastrointestinal fluid losses such as vomiting, diarrhea, or high-output fistulas, as well as excessive sweating or third-spacing in pancreatitis or peritonitis[2,21]. Renal causes include the use of thiazide diuretics, cerebral salt wasting (CSW), mineralocorticoid deficiency, and some forms of tubulointerstitial disease[5]. Clinically, patients often present with orthostatic hypotension, tachycardia, decreased skin turgor, and signs of hypoperfusion[1-4]. A useful diagnostic distinction relies on urinary sodium: Low values suggest extrarenal sodium loss, whereas elevated values indicate renal causes[2,20].
Euvolemic hyponatremia is most frequently due to the syndrome of inappropriate antidiuretic hormone secretion (SIADH; also called syndrome of inappropriate antidiuresis), particularly in hospitalized patients. SIADH can result from pulmonary disorders, central nervous system diseases, malignancies (e.g., small-cell lung carcinoma), or the use of drugs such as selective serotonin reuptake inhibitors, carbamazepine, cyclophosphamide, or vincristine (Table 1)[22,23]. Endocrine disorders such as adrenal insufficiency and hypothyroidism must be excluded, and primary polydipsia remains an additional though less common cause. Importantly, patients with euvolemic hyponatremia usually lack clinical signs of either hypo- or hypervolemia, making laboratory assessment essential[2,5].
| Drug class | Examples | Proposed mechanism | Clinical notes |
| Antidepressants | SSRIs (sertraline, fluoxetine, paroxetine), SNRIs (venlafaxine, duloxetine), tricyclics (amitriptyline), mirtazapine | Enhanced ADH release or potentiation of its renal effect | Most frequent cause in older adults, especially underweight women |
| Antipsychotics | Haloperidol, risperidone, olanzapine, quetiapine | Dopaminergic and serotonergic modulation of ADH | Risk increases with concomitant antidepressants |
| Antiepileptics/mood stabilizers | Carbamazepine, oxcarbazepine, valproate, lamotrigine | Increased ADH release and sensitivity at renal V2 receptor | Carbamazepine is a classic cause of chronic SIADH |
| Chemotherapeutic/oncologic agents | Cyclophosphamide, vincristine, cisplatin, ifosfamide | Direct stimulation of ADH release or renal tubular toxicity | Often transient; risk rises with concomitant nausea or stress |
| Analgesics | Opioids (morphine, tramadol), NSAIDs | Opioids increase ADH release; NSAIDs potentiate its action by reducing prostaglandin inhibition | Particularly relevant postoperatively |
| Diuretics | Thiazides (hydrochlorothiazide, indapamide) | Impaired urinary dilution, sodium loss | Responsible for up to 90% of diuretic-induced hyponatremia |
| Antineoplastic/immunomodulators | Interferon-α, cyclophosphamide, vincristine | Hypothalamic or renal effect on ADH | Usually dose-dependent |
| Other agents | Desmopressin, chlorpropamide, ecstasy (MDMA) | Direct V2 receptor agonism or ADH secretion | Seen in recreational drug users and hospitalized patients receiving DDAVP |
In hypervolemic hyponatremia, there is an increase in both sodium and water content, but with disproportionately greater water retention[1,2]. Although it can occur spontaneously in advanced disease states, it is often exacerbated by iatrogenic factors such as hypotonic fluid administration or aggressive diuretic therapy[2,24]. This imbalance is typically observed in disorders associated with reduced effective arterial blood volume, despite an absolute excess of ECF. Such conditions include advanced heart failure, cirrhosis, and nephrotic syndrome, where decreased perfusion stimulates non-osmotic AVP release, sympathetic activation, and renin-angiotensin-aldosterone system activity. The resulting impaired water excretion aggravates hypotonicity. Clinically, these patients exhibit edema, ascites, and signs of fluid overload[2,20].
The time course of hyponatremia is critical for prognosis and management[2,25]. Acute hyponatremia, defined as a fall in serum sodium within less than 48 hours, carries the highest risk of cerebral edema, seizures, and death due to the lack of sufficient time for osmotic adaptation within neurons[2,4,26]. Chronic hyponatremia (≥ 48 hours or of uncertain duration) tends to present with fewer or subtler symptoms because neurons gradually extrude osmolytes to restore osmotic equilibrium[2,5]. However, this adaptive process renders patients highly vulnerable to osmotic demyelination syndrome (ODS) if serum sodium is corrected too rapidly[1,2,5]. Clinically, the distinction between acute and chronic is often uncertain, and in practice, when the onset is unknown, hyponatremia is assumed to be chronic to minimize treatment-related complications[20,27].
The clinical presentation depends not only on the absolute serum sodium level but also on the rate of decline. Mild-to-moderate hyponatremia often causes nonspecific symptoms such as headache, fatigue, nausea, irritability, dizziness, cognitive impairment, and gait disturbances[1-4,20]. These manifestations may be mistakenly attributed to comorbid conditions, which delays recognition of the electrolyte disorder. Severe or rapidly progressive hyponatremia can precipitate seizures, respiratory depression, coma, or death if untreated[1,2]. Notably, growing evidence highlights that even mild chronic hyponatremia is not benign: Studies have demonstrated associations with increased risk of falls, bone fractures, gait unsteadiness, and impaired attention or memory[28-31].
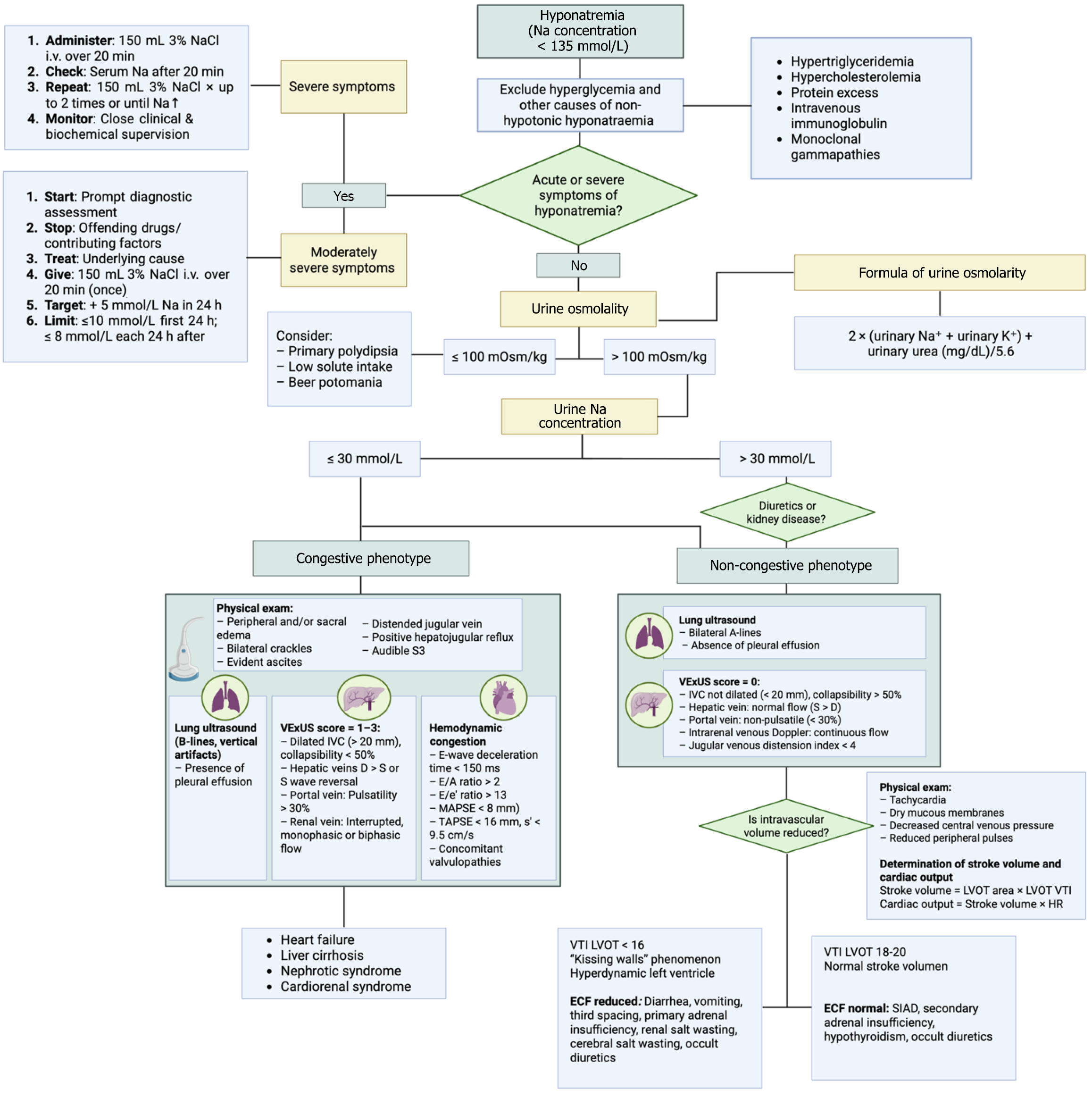
Initial assessment of volume and tonicity status in patients with hyponatremia requires a combination of clinical and laboratory data. Traditionally, urinary osmolality and urinary sodium have been used as key diagnostic tools. In the presence of hypotonic hyponatremia, low urinary osmolality (< 100 mOsm/kg) suggests an excess of free water (as in primary polydipsia), while high values (> 100 mOsm/kg) indicate an appropriate renal response to antidiuretic hormone or its inappropriate secretion, as in SIADH or occult hypovolemia[2,32].
Urinary sodium, on the other hand, has been widely used to discriminate hypovolemic causes (usually < 30 mEq/L) from non-hypovolemic causes (usually > 30 mEq/L)[2,20]. However, its interpretation is affected by the use of diuretics, renal failure, and frequent comorbidities in hospitalized patients[2]. Other classic markers, such as blood urea nitrogen/creatinine ratio or sodium excreted fraction, also have limitations in this context and should not be used in isolation[32].
In patients with hypovolemia, typically with sodium and water losses due to gastrointestinal or renal causes, the reduction in effective arterial volume stimulates AVP secretion, perpetuating hyponatremia[5]. In hypervolemic hy
Given these limitations, new biomarkers have been investigated that more closely reflect the activity of the vasopressin-renal axis. Copeptin, a stable peptide derived from the precursor of vasopressin, has been proposed as an indirect marker of antidiuretic hormone secretion[18,33]. Although its plasma concentration is elevated in states of hypovolemia, SIADH, and acute physiological stress, its diagnostic utility has been questioned due to its wide interindividual variability and overlap between hyponatremia subtypes[2,5,33]. In prospective studies, it has been observed that its isolated value does not allow a precise differentiation between hypovolemia, euvolemia, and hypervolemia; however, some authors have suggested that the urinary copeptin/sodium ratio could play a role in distinguishing between SIADH and hypovolemic causes, although with limited clinical utility[2,33-35]. In fact, the Co-MEDy study[34] demonstrated that copeptin levels < 3.9 pmol/L were highly suggestive of primary polydipsia, while values > 84 pmol/L indicated hypovolemia; however, the overlap in intermediate ranges limited its ability to differentiate among the primary etiologies of profound hyponatremia reliably. Similarly, Fenske et al[33] showed that although copeptin levels were elevated across different hyponatremia subtypes, only the copeptin-to-urinary sodium ratio (> 30 pmol/mmol) provided partial discriminatory power between SIADH and hypovolemic hyponatremia.
An emerging biomarker with more promising results is mid-regional pro-atrial natriuretic peptide (MR-proANP)[36]. This marker, released in response to atrial distension, showed a significant association with volume status in the study by Nigro et al[36], where MR-proANP was independently associated with hypovolemia vs euvolemia (odds ratio: 2.45 per SD increase in multivariable models) and provided diagnostic value beyond traditional urine indices. In contrast, N-terminal pro-brain natriuretic peptide, although widely used in the evaluation of heart failure, did not demonstrate discriminative utility for hyponatremia subtypes in this setting[36]. Complementary evidence comes from the CAPNETZ cohort, in which Krüger et al[37] found that both MR-proANP and C-terminal pro-AVP were significantly elevated in patients with community-acquired pneumonia and dysnatremia, and that their combination with serum sodium achieved the highest accuracy for predicting 28-day mortality. Thus, biomarkers such as MR-proANP could complement conventional clinical and laboratory data, particularly when urinalysis is unavailable or yields inconclusive results. However, further validation is needed before systematic incorporation into diagnostic algorithms.
Accurate assessment of ECV is essential for the differential diagnosis and appropriate treatment of hyponatremia. Traditional tools, such as physical examination, vital signs, and laboratory parameters (e.g., sodium excretion fraction or serum urea nitrogen), have significant limitations, with correct diagnosis rates of less than 50% in some studies[5]. In this context, the use of point-of-care ultrasound (PoCUS) has emerged as a non-invasive, complementary tool that enables a more comprehensive hemodynamic assessment[38].
PoCUS integrates three key components: Lung ultrasound (LUS), venous congestion ultrasound, and focused cardiac ultrasound. This combination enables the estimation of stroke volume, assessment of systemic venous congestion, and detection of pulmonary patterns suggestive of fluid overload or hypovolemia. For example, a pattern with A-lines in LUS, decreased Doppler-measured stroke volume of the left ventricular outflow tract, and a collapsible inferior vena cava suggests hypovolemia[38].
In 2022, Chatterjee and Koratala[39], supported this utility by showing how PoCUS allows the identification of occult hypovolemia in a patient with ambiguous physical findings, since although the examination suggested euvolemia, Doppler showed low stroke volume with a positive response to the passive leg elevation test (increase > 15% in velocity-time integral). This prompted the safe administration of fluids, leading to normalization of both serum sodium and stroke volume, thereby confirming the diagnosis of occult hypovolemia. Such evaluation highlights how PoCUS can guide targeted and safe interventions, resulting in favorable clinical outcomes.
In addition, in 2024, Mazón Ruiz et al[38] suggested incorporating PoCUS early in the diagnostic approach to hyponatremia as a way to improve ECV assessment and to phenotype congestion (non-congestive, congestive, or redistributive), combining LUS, venous congestion ultrasound, and focused cardiac ultrasound to complement clinical and laboratory data. This proposal advances prior guidance, such as the 2014 European Clinical Practice Guideline, which emphasized biochemical and urinary indices while acknowledging the limitations of physical examination in assessing volume status[2]. While innovative, the scheme proposed by Mazón Ruiz et al[38] begins with plasma osmolality as the entry point. In real-world hospitalized or critically ill settings, however, the first abnormality clinicians usually face is the documentation of hyponatremia itself (serum sodium < 135 mmol/L)[1,5]. Using this threshold as the initial step better reflects clinical practice and may streamline subsequent evaluation of tonicity and volume status. Furthermore, incorporating urinary sodium together with PoCUS findings may provide a more precise and pragmatic framework, allowing not only congestion phenotyping but also refinement of the likely etiological diagnoses (Figure 2).
The treatment of acute and symptomatic hyponatremia constitutes a medical emergency[1,2]. Current European guidelines recommend the administration of 3% hypertonic saline as repeated intravenous boluses of 150 mL infused over 20 minutes, which may be repeated up to two additional times depending on the clinical response or until serum sodium has risen by approximately 5 mmol/L[2]. Alternatively, a continuous infusion may be administered at a rate of 0.5-2 mL/kg/hour. Nonetheless, bolus therapy is preferred to achieve a more predictable correction[2,20]. In patients with concomitant hypokalemia, potassium supplementation should be initiated, but clinicians must be aware that potassium repletion itself accelerates the rise in serum sodium by driving intracellular sodium shifts[2,5]. Thus, when potassium chloride is infused (either separately or in combination with hypertonic saline), strict monitoring of serum sodium (every 2 hours) is mandatory to prevent inadvertent overcorrection[2,20]. A sudden increase in free water diuresis further amplifies this risk, with osmotic demyelination as the feared complication[2]. For severe chronic hyponatremia (< 120 mmol/L), although randomized trials are lacking, it has been suggested that slow infusions of hypertonic saline (15-30 mL/hour) may be beneficial even in asymptomatic patients, provided that close surveillance is ensured to avoid overly rapid correction[2,20,40].
Overall, mild to moderate and asymptomatic cases should be addressed with less aggressive strategies. For this, the first fundamental step in evaluating a patient with hyponatremia is to determine whether the condition is real (hypotonic) or spurious[1,2]. Isotonic or hypertonic hyponatremia results from water shifts caused by osmotically active solutes like glucose or lipids and does not indicate a true sodium deficiency (often called pseudohyponatremia; Figure 2). Treatment should target the underlying condition. Hypotonic hyponatremia, on the other hand, is the most frequent and clinically relevant form, involving a relative excess of free water with respect to the body’s sodium content[1,2].
The rate of serum sodium correction is critical to preventing neurologic injury, particularly ODS. Current guidelines recommend aiming for an initial rise of approximately 4-6 mmol/L within the first 6 hours for patients with acute or severely symptomatic hyponatremia, while avoiding increases > 8 mmol/L in any subsequent 24-hour period and > 18 mmol/L within 48 hours[1,2]. Observational data indicate that ODS has occurred most often when serum sodium increased by > 10 mmol/L in the first 24 hours or > 18 mmol/L in 48 hours[1]. Targets should be individualized. In patients at low risk for ODS, many experts accept totals up to 8-10 (occasionally 10-12) mmol/L in 24 hours provided that 48-hour correction remains ≤ 18 mmol/L; conversely, in high-risk patients (chronic and profound hyponatremia, alcoholism, malnutrition, advanced liver disease, or hypokalemia) a more conservative limit of ≤ 6-8 mmol/L per 24 hours (often aiming for 4-6) is advised, with close biochemical monitoring and readiness to halt or re-lower if overcorrection ensues[2,20].
The pathophysiological basis for the strict limits in sodium correction lies in the cerebral adaptation to chronic hypo-osmolality[5]. In hyponatremia of gradual onset, brain cells reduce their content of organic osmolytes (such as taurine, myo-inositol, and creatine) to prevent cerebral edema[2,27]. A rapidly increasing serum sodium level quickly reverses the osmotic gradient, causing cellular dehydration and damage to glial structures[2,5,41]. Current evidence indicates that the initial damage primarily affects astrocytes, which control osmotic balance through aquaporins and connexins; their dysfunction then disrupts the metabolic and osmotic support for oligodendrocytes, leading to myelin rupture and cell death. This process explains the tendency for damage to occur in the pontine and extrapontine areas, resulting in the clinical syndrome of ODS[41].
However, a recent meta-analysis by Ayus et al[42] including more than 11000 patients challenged this paradigm, showing that rapid correction (≥ 8-10 mmol/L per 24 hours) was associated with lower in-hospital and 30-day mortality, as well as shorter hospital stay, without a significant increase in ODS. These findings suggest that excessively slow correction may also be harmful. Nevertheless, as the analysis was based on observational cohort studies with inherent risks of bias and confounding, its results should be interpreted with caution and do not yet justify changes to current guideline recommendations[2,20].
Hypertonic saline 3% remains the cornerstone of therapy for acute symptomatic hyponatremia, and commercially prepared solutions are readily available in many settings[1,2,43]. However, in middle- and low-income countries or in hospitals where ready-to-use 3% saline is not accessible, it can be safely prepared from more concentrated stock solutions (e.g., 17.7% NaCl) by validated dilution methods using 0.9% saline as a diluent, allowing accurate and reproducible preparation for clinical use[44].
The general formula is:
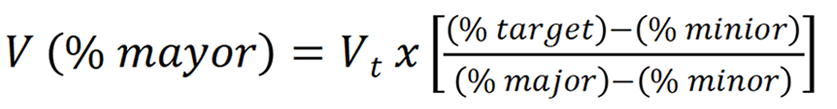
Where:
V(% major): Volume to be used of the most concentrated solution (17.7%).
Vt: Total volume desired.
% target: Desired final concentration (3%).
% minor: Diluent solution concentration (0.9%).
% major: Concentration of the available solution (17.7%).
Example 1: To prepare 1000 mL of 3% sodium chloride (NaCl) using a 17.7% solution:
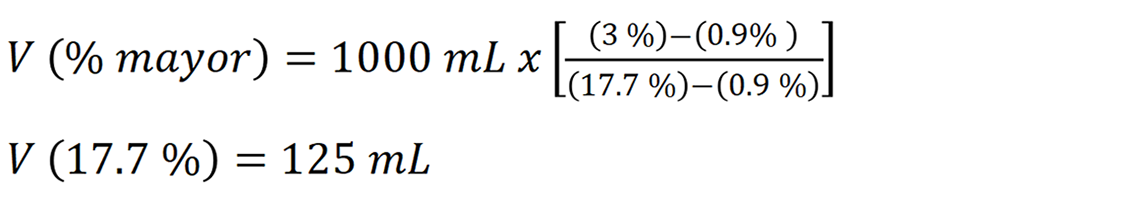
125 mL of 17.7% NaCl should be mixed with 875 mL of 0.9% sodium saline normal. Since commercial presentations of 17.7% NaCl in Mexico typically come in 10 mL ampoules, this equates to 12.5 ampoules. It is important to note that the calculated volume of concentrated solution must be removed from the total volume of the base solution (gauged) to maintain the desired final concentration.
Example 2: To prepare 500 mL of 3% NaCl:
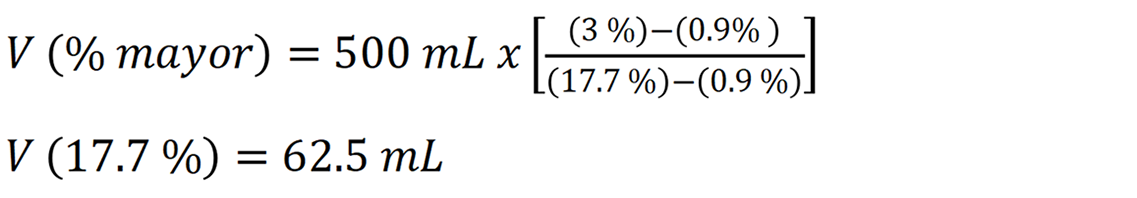
62.5 mL of 17.7% NaCl should be mixed with 437.5 mL of 0.9% sodium saline normal, which corresponds to 6.25 ampoules of 10 mL. This method is practical, reproducible, and clinically safe (when performed under aseptic conditions) for medical practice.
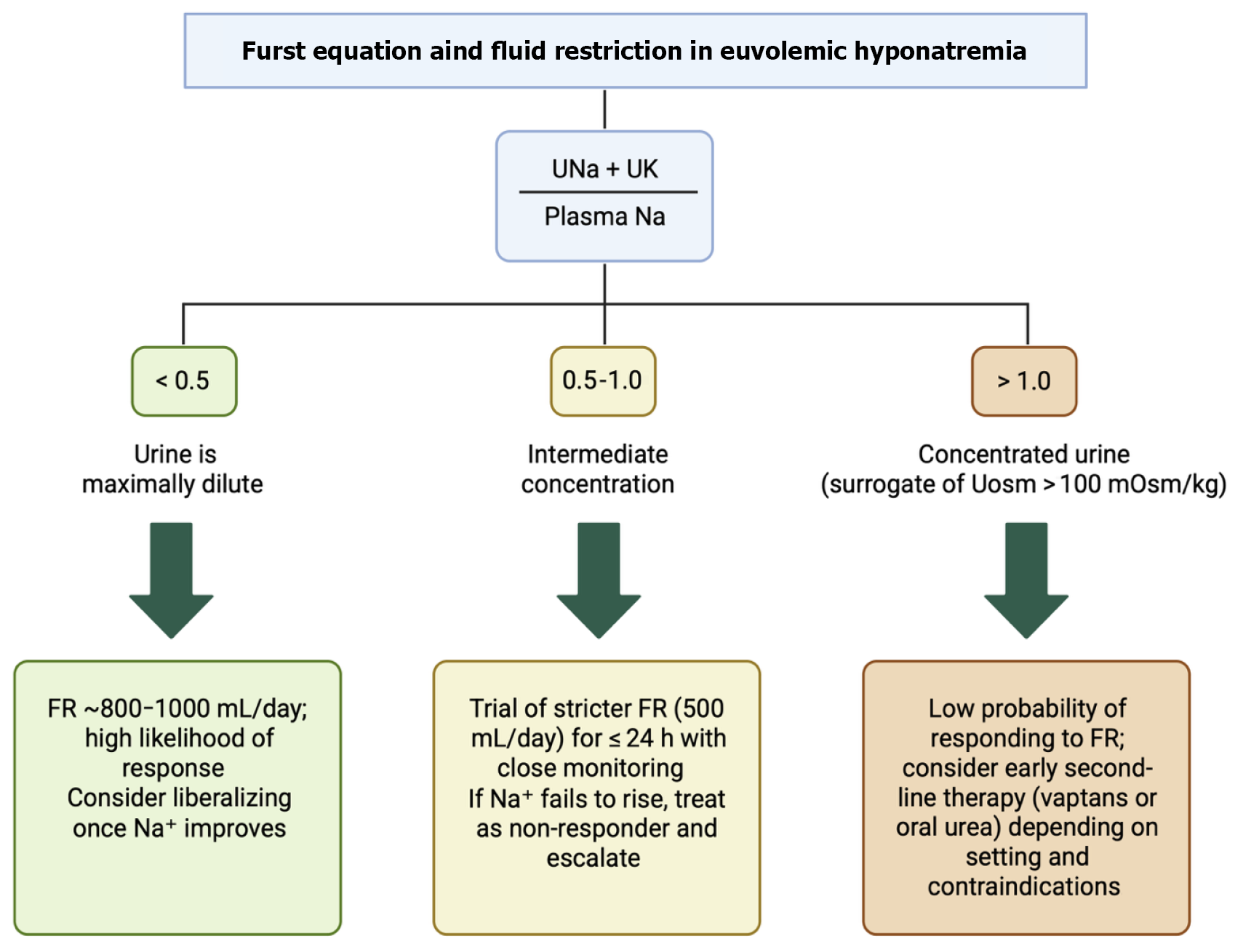
Fluid restriction remains the recommended first-line therapy for chronic euvolemic hyponatremia (commonly seen in SIADH), typically prescribing 800-1000 mL/day, as it is simple, inexpensive, and generally safe[45-47]. Nonetheless, its efficacy is often limited, with up to 40%-50% of patients failing to achieve a meaningful rise in serum sodium[48]. Likewise, data from a randomized controlled trial found that fluid restriction to 1 L/day produced a slight early increase in plasma sodium (median + 3 mmol/L at 3 days) with little additional correction by 30 days[47]. Additionally, observational studies have identified predictors of nonresponse, such as high urine osmolality or elevated urinary sodium, reflecting impaired electrolyte-free water clearance[48].
Unfortunately, in practice, particularly among hospitalized or critically ill patients, it is often challenging to predict who may benefit from fluid restriction based merely on measures. To address this challenge, the Furst equation (also called the Furst index) has been proposed as a simple and practical bedside tool (Figure 3). The Furst equation is calculated as (urinary Na+ + K+)/plasma Na+[45]. Values > 1 indicate concentrated urine and might predict a low likelihood of response to fluid restriction, whereas values < 1 suggest a higher probability of success[45,46]. In a recent study by Chienwichai et al[45], a cutoff of 0.86 optimally identified nonresponders; however, its performance remained modest (AUC: Approximately 0.65). Although studies evaluating the utility and accuracy of the Furst equation in clinical practice are limited, its use needs further validation and opens the door for future research into practical bedside tools for predicting fluid restriction response.
Beyond predictive formulas such as the Furst equation, calculating daily free water excretion provides additional insight into a patient’s ability to eliminate solute-free water and anticipate response to fluid restriction[5]. This parameter can be estimated using the electrolyte-free water clearance formula: Ch2Oe = V × [1 - (Una + Uk)/Pna].
Where V is the urine volume, Una and Uk are urinary sodium and potassium concentrations, and Pna is plasma sodium concentration[1,5]. A daily electrolyte-free water clearance greater than 500-800 mL generally predicts adequate renal water excretion and a favorable response to fluid restriction, whereas lower or negative values indicate impaired clearance and the potential need for adjunctive therapies such as urea or loop diuretics. Although conceptually useful, its application in routine clinical practice remains limited by the need for serial urine studies and laboratory data[1,2,5].
In patients with persistent hyponatremia despite fluid restriction (mainly in SIADH), various pharmacological options have been proposed[1,49,50]. Among these, vasopressin receptor V2 antagonists (vaptans), such as tolvaptan, and the use of urea, an osmotic agent with a more conservative correction profile, stand out[50].
Tolvaptan, licensed in the United States for euvolemic and hypervolemic hyponatremia, has shown efficacy in rapidly increasing serum sodium, but the potential risk of overcorrection and ODS might limit its use[1,51,52]. In a retrospective multicenter study[49], tolvaptan generated an increase of 5.05 mmol/L at 12 hours compared to 1.10 mmol/L with urea (P = 0.001), although the differences at 24 hours and 48 hours were not significant. However, the risk of overcorrection was 43% with tolvaptan compared to 9% with urea. Regarding the doses used, tolvaptan was administered mostly in schedules of 7.5 mg/day to 15 mg/day, while urea was used in average doses of 18.6 g/day, often in combination with other measures such as water restriction or diuretics[49]. However, tolvaptan achieved a faster correction in the short term, hospital duration, normalization rate, and overall safety favored urea.
Even though there has been an increasing recommendation for vaptan use because of their pharmacological effects, they should not be used as first-line therapy because of their cost-benefit profile and the fact that we still don’t know how they will affect people in the long term, especially those with mild to moderate hyponatremia[49-51]. Randomized studies have not shown any improvements in mortality, quality of life, or clinically significant outcomes, and the exorbitant cost makes it even less likely that this will be used regularly[53]. Even at modest dosages (< 15 mg), tolvaptan might represent a significant risk of overcorrection, remarking the need for meticulous monitoring and judicious application[54]. Urea, on the other hand, is a safe, cheap, and widely available alternative that works by osmotic diuresis and slowly raising sodium levels without a high risk of overcorrection/ODS[54,55]. It is usually administered as oral powder dissolved in water or juice (15-30 g/day), allowing a gradual and controlled increase in serum sodium concentration with minimal adverse effects. When given per os or enterally at a dose of 15 g to 30 g per day, urea has been found to be equally effective as vaptans but safer[1,55]. Demeclocycline and lithium are currently mostly not used anymore because they are toxic. Loop diuretics mixed with isotonic saline may only be used in cases of hypervolemia[20,52]. Overall, treatment for refractory hyponatremia should be tailored to each patient[1]. Urea is a practical second-line option once fluid restriction has failed, while vaptans should only be used in particular situations, with risk, expense, and clinical context carefully considered[55].
Another alternative well-known agent for treating severe hyponatremia is desmopressin [1-deamino-8-D-arginine vasopressin (DDAVP)], a synthetic vasopressin analogue that acts on V2 receptors in the renal collecting duct, thereby promoting water reabsorption and suppressing free-water diuresis[56-59]. Its rationale in hyponatremia is counterintuitive, as it deliberately induces antidiuresis. Nevertheless, this strategy is useful to prevent or reverse overly rapid correction of serum sodium, one of the main risk factors for ODS[57].
Different strategies of DDAVP administration have been described. Proactive (given early to tightly control correction), reactive (administered when an unexpectedly brisk rise in sodium or urine output is observed), and rescue (used once correction limits are exceeded)[57,58]. Observational data from large cohorts show that the reactive approach is most common, and while DDAVP effectively slows the rate of correction, it has been associated with longer hospital stay and lower rates of “safe correction” when used late, largely due to overcorrection occurring before its initiation[58]. In contrast, a proactive strategy has been linked with more predictable sodium trajectories, albeit sometimes at the expense of slower normalization[58,59].
Moreover, Sood et al[59] evaluated the combination of hypertonic saline with scheduled DDAVP to achieve controlled correction. In a quality improvement series, this regimen produced steady sodium increases (approximately 5-6 mmol/L at 24 hours) without cases of excessive correction or ODS, highlighting its utility in critically ill or high-risk patients.
Overall, the role of DDAVP in hyponatremia management is not to raise sodium directly, but rather to act as a “brake” that prevents uncontrolled aquaresis once the underlying cause of hyponatremia begins to resolve[60]. Current data suggest that its judicious use, particularly in patients at high risk of ODS (very low baseline sodium, malnutrition, alcoholism, hypokalemia, or liver disease), might improve safety. Nevertheless, the evidence remains observational mainly, and prospective studies are still needed to clarify its optimal timing, dosing, and impact on hard outcomes[58-60]. In clinical practice, DDAVP may be administered to prevent or reverse overly rapid correction of serum sodium levels[60]. Typical regimens include 1-2 μg intravenously or subcutaneously every 6-8 hours, or 10-20 μg intranasally every 8-12 hours, with frequent monitoring of serum sodium and urine output. In cases where overcorrection has already occurred, DDAVP can be combined with intravenous dextrose 5% solution to safely re-lower sodium levels, maintaining correction rates below 8-10 mmol/L per 24 hours. This proactive “DDAVP clamp” strategy has been increasingly adopted to stabilize sodium trajectories and reduce the risk of osmotic demyelination[58-60].
For certain patients with chronic or difficult-to-treat euvolemic hyponatremia, additional osmotic strategies might help support the correction process. For instance, oral salt tablets can be used to increase solute intake and boost free water clearance, especially if fluid restriction alone isn’t enough[1,2]. Similarly, a low-dose oral furosemide combined with oral sodium supplementation can promote the excretion of excess water by lowering medullary osmolarity[2]. Eating a high-protein diet can also raise urea production and promote osmotic diuresis, which can help improve serum sodium levels without needing medication. It’s important to carefully implement these measures under proper medical and biochemical supervision to prevent too rapid correction or volume depletion[1,2].
Evidence supporting the use of fludrocortisone in hyponatremia is limited to specific conditions such as CSW[1,2,61]. In a randomized controlled trial of 42 patients with hyponatremia following subarachnoid hemorrhage, Misra et al[61] demonstrated that fludrocortisone (0.1-0.4 mg daily) significantly shortened the time to normalization of serum sodium compared with standard therapy alone, with minimal adverse effects. These findings suggest that mineralocorticoid supplementation might be partially safe and effective as an adjunct in selected cases of hypovolemic hyponatremia due to renal sodium loss. Although mineralocorticoid therapy has been proposed in various contexts, it is not recommended in SIADH[1,2,22]. European guidelines[2] restrict fludrocortisone to selected cases of CSW, where it may accelerate sodium correction, but evidence remains limited, and it should not be used in euvolemic or hypervolemic hyponatremia. Thus, the management of SIADH should rely on fluid restriction, oral sodium supplementation combined with loop diuretics, or urea and vasopressin receptor antagonists in refractory cases[1,2,22].
The distinction between CSW and SIADH remains controversial and clinically challenging (Table 2)[2,20]. Both entities may present with hyponatremia, high urinary sodium, and elevated urine osmolality, yet differ in ECV status and therapeutic approach[5,20]. Current guidelines acknowledge that reliable clinical or biochemical differentiation is difficult, as assessment of volume status is often inaccurate and markers such as serum uric acid or natriuretic peptides have limited diagnostic specificity[2,5,20]. Persistent hypouricemia and increased fractional excretion of urate after sodium correction may suggest CSW, but these findings are not pathognomonic[62]. European and American guidelines therefore recommend focusing on careful clinical evaluation and response to isotonic saline rather than rigid classification of these entities[2,20].
| Feature | SIADH | Cerebral salt wasting | Reset osmostat |
| Volume status | Euvolemic | Hypovolemic (true salt loss) | Euvolemic |
| Plasma sodium | Low | Low | Mildly low but stable |
| Urine sodium | > 30 mmol/L | > 30 mmol/L | Variable |
| Urine osmolality | > 100 mOsm/kg | > 100 mOsm/kg | Normal dilution capacity maintained |
| Serum uric acid | Low | Low | Normal or low-normal |
| Fractional excretion of uric acid | Elevated during hyponatremia (> 12%), normalizes after correction | Remains elevated after correction | Normal (< 11%) |
| Fractional excretion of sodium | > 0.5%-1% (inappropriately high for euvolemia) | > 1%-2% (due to true renal salt loss) | Normal or slightly increased |
| Fractional excretion of urea | > 55%-60% | > 60%-70% | Normal (< 35%) |
| Response to isotonic saline | No improvement or worsens hyponatremia | Improves serum sodium and volume | No major change |
| ADH secretion pattern | Inappropriate, independent of osmolality | Appropriate (secondary to hypovolemia) | Reset to a lower threshold |
| Volume markers (BUN, hematocrit) | Normal | Elevated (due to hypovolemia) | Normal |
| Treatment approach | Fluid restriction, salt tablets, urea, vaptans | Volume and salt repletion ± fludrocortisone | Usually none; avoid overcorrection |
| Typical setting | Pulmonary disorders, malignancy, CNS disease, drugs | CNS injury, subarachnoid hemorrhage, neurosurgery | Chronic illness, elderly, pregnancy, tuberculosis, carcinoma |
Similarly, reset osmostat represents a mild, stable form of euvolemic hyponatremia in which the hypothalamic threshold for vasopressin release is chronically lowered, resulting in a new steady-state at a reduced plasma sodium concentration[63]. Recent prospective data support its recognition as a distinct clinical entity rather than a subtype of SIADH, characterized by preserved renal water-diluting capacity and stable sodium balance[63]. The water-diluting test remains the gold standard for diagnosis, with ≥ 80% free-water excretion favoring a reset osmostat over SIADH, whereas excretion usually remains < 40%. Fractional uric acid excretion below 11% may serve as a practical surrogate, whereas copeptin has not demonstrated reliable diagnostic accuracy[63]. Recognizing this entity prevents unnecessary fluid restriction and highlights that CSW, SIADH, and reset osmostat likely represent a physiological continuum rather than discrete categories[5,20,63].
Recent advances in artificial intelligence and healthcare information technology are bringing in a new era for the personalized management of hyponatremia[64-66]. Conventional methods depend significantly on clinician interpretation and fixed laboratory thresholds. However, novel models utilize machine learning and decision support systems to dynamically forecast sodium changes and direct customized therapies[65]. A large cohort study in Germany showed that a laboratory-based clinical decision support system indicated that prompt follow-up of sodium measurements (within 12 hours) could markedly enhance correction rates and discharge outcomes. This suggests the potential for automated alerts to improve patient safety and adherence to guideline-based care[66].
Moreover, modern deep learning models have attained enhanced accuracy in forecasting serum sodium variations and recommending personalized therapeutic modifications for critically ill patients[65]. In the future, artificial intelligence-driven prediction, precise monitoring, and customized suggestions could help healthcare providers maintain natremia within a safe range with greater accuracy than ever before. This reflects personalized care, where treatment strength, speed, and tactics are tailored to each patient’s risk and response. At the bedside, this integration can help reduce medical errors, shorten hospital stays, and keep vulnerable groups safer when treatment adjustments are needed.
A serum sodium < 135 mmol/L is only the top of the iceberg. Its approach and management might be challenging in some refractory scenarios and in limited-resource settings. Advances in biomarkers, imaging, and physiologic understanding have refined the diagnostic process, while therapeutic strategies continue to evolve toward safer and more tailored correction. Fluid restriction remains the cornerstone of initial therapy, complemented by tools such as the Furst equation to anticipate response. Importantly, the evaluation of volemia in hyponatremia solely on clinical examination is becoming insufficient in modern times. PoCUS should be a routine extension of the physical exam, providing real-time, objective, and reproducible insights into intravascular volume status. For refractory cases, urea and DDAVP offer safe and practical alternatives, whereas vaptans should be reserved for carefully selected patients under close monitoring. Looking ahead, the integration of artificial intelligence and decision-support systems promises to personalize further and refine the management of hyponatremia, translating evidence-based insights into better patient outcomes at the bedside.
We would like to thank all the medical staff from the internal medicine department at Hospital General Dr. Manuel Gea Gonzalez. Use this review to continue improving residents’ learning.
| 1. | Spasovski G. Hyponatraemia-treatment standard 2024. Nephrol Dial Transplant. 2024;39:1583-1592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 2. | Spasovski G, Vanholder R, Allolio B, Annane D, Ball S, Bichet D, Decaux G, Fenske W, Hoorn EJ, Ichai C, Joannidis M, Soupart A, Zietse R, Haller M, van der Veer S, Van Biesen W, Nagler E; Hyponatraemia Guideline Development Group. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Eur J Endocrinol. 2014;170:G1-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 708] [Cited by in RCA: 541] [Article Influence: 45.1] [Reference Citation Analysis (0)] |
| 3. | Burst V. Etiology and Epidemiology of Hyponatremia. Front Horm Res. 2019;52:24-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 4. | Sterns RH, Rondon-Berrios H, Adrogué HJ, Berl T, Burst V, Cohen DM, Christ-Crain M, Cuesta M, Decaux G, Emmett M, Garrahy A, Gankam-Kengne F, Hix JK, Hoorn EJ, Kamel KS, Madias NE, Peri A, Refardt J, Rosner MH, Sherlock M, Silver SM, Soupart A, Thompson CJ, Verbalis JG; PRONATREOUS Investigators. Treatment Guidelines for Hyponatremia: Stay the Course. Clin J Am Soc Nephrol. 2024;19:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 48] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 5. | Seay NW, Lehrich RW, Greenberg A. Diagnosis and Management of Disorders of Body Tonicity-Hyponatremia and Hypernatremia: Core Curriculum 2020. Am J Kidney Dis. 2020;75:272-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 6. | Robertson GL, Aycinena P, Zerbe RL. Neurogenic disorders of osmoregulation. Am J Med. 1982;72:339-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 186] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 7. | LEAF A, MAMBY AR. The normal antidiuretic mechanism in man and dog; its regulation by extracellular fluid tonicity. J Clin Invest. 1952;31:54-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 27] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 8. | Schrier RW. Molecular mechanisms of clinical concentrating and diluting disorders. Prog Brain Res. 2008;170:539-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Cowen LE, Hodak SP, Verbalis JG. Age-associated abnormalities of water homeostasis. Endocrinol Metab Clin North Am. 2013;42:349-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 102] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 10. | Hochberg Z, Benderly A. Normal osmotic threshold for vasopressin release in the hyponatremia of hypothyroidism. Horm Res. 1983;17:128-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 11. | Bichet DG. Regulation of Thirst and Vasopressin Release. Annu Rev Physiol. 2019;81:359-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Dunn FL, Brennan TJ, Nelson AE, Robertson GL. The role of blood osmolality and volume in regulating vasopressin secretion in the rat. J Clin Invest. 1973;52:3212-3219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 441] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 13. | Knepper MA, Kwon TH, Nielsen S. Molecular physiology of water balance. N Engl J Med. 2015;372:1349-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 190] [Article Influence: 17.3] [Reference Citation Analysis (5)] |
| 14. | Johnson AK, Gross PM. Sensory circumventricular organs and brain homeostatic pathways. FASEB J. 1993;7:678-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 382] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 15. | McKinley MJ, Johnson AK. The physiological regulation of thirst and fluid intake. News Physiol Sci. 2004;19:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 143] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 16. | Kim A, Madara JC, Wu C, Andermann ML, Lowell BB. Neural basis for regulation of vasopressin secretion by anticipated disturbances in osmolality. Elife. 2021;10:e66609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Landgraf R, Ludwig M. Vasopressin release within the supraoptic and paraventricular nuclei of the rat brain: osmotic stimulation via microdialysis. Brain Res. 1991;558:191-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 93] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 18. | Christ-Crain M. Vasopressin and Copeptin in health and disease. Rev Endocr Metab Disord. 2019;20:283-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 19. | Bankir L, Bichet DG, Morgenthaler NG. Vasopressin: physiology, assessment and osmosensation. J Intern Med. 2017;282:284-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 189] [Article Influence: 21.0] [Reference Citation Analysis (3)] |
| 20. | Hoorn EJ, Zietse R. Diagnosis and Treatment of Hyponatremia: Compilation of the Guidelines. J Am Soc Nephrol. 2017;28:1340-1349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 218] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 21. | Mannheimer B, Bergh CF, Falhammar H, Calissendorff J, Skov J, Lindh JD. Association between newly initiated thiazide diuretics and hospitalization due to hyponatremia. Eur J Clin Pharmacol. 2021;77:1049-1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 22. | Adrogué HJ, Madias NE. The Syndrome of Inappropriate Antidiuresis. N Engl J Med. 2023;389:1499-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 23. | Ellison DH, Berl T. Clinical practice. The syndrome of inappropriate antidiuresis. N Engl J Med. 2007;356:2064-2072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 761] [Cited by in RCA: 569] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 24. | Baiu I, Kang M, Weiser TG. Acute severe iatrogenic hyponatremia. Trauma Surg Acute Care Open. 2019;4:e000388. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 25. | Lindner G, Schwarz C, Haidinger M, Ravioli S. Hyponatremia in the emergency department. Am J Emerg Med. 2022;60:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 26. | Sterns RH. Treatment of Severe Hyponatremia. Clin J Am Soc Nephrol. 2018;13:641-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 27. | Rondon-Berrios H. Diagnostic and Therapeutic Strategies to Severe Hyponatremia in the Intensive Care Unit. J Intensive Care Med. 2024;39:1039-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 28. | Renneboog B, Musch W, Vandemergel X, Manto MU, Decaux G. Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits. Am J Med. 2006;119:71.e1-71.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 672] [Cited by in RCA: 563] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 29. | Soiza RL, Cumming K, Clarke JM, Wood KM, Myint PK. Hyponatremia: Special Considerations in Older Patients. J Clin Med. 2014;3:944-958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 30. | Dokmak A, Madias NE. Hyponatremia and In-Hospital Falls and Fractures in Older Adults. J Am Geriatr Soc. 2019;67:1752-1753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 31. | Fratangelo L, Nguyen S, D'Amelio P. Hyponatremia and aging-related diseases: key player or innocent bystander? A systematic review. Syst Rev. 2023;12:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 32. | Fenske W, Maier SK, Blechschmidt A, Allolio B, Störk S. Utility and limitations of the traditional diagnostic approach to hyponatremia: a diagnostic study. Am J Med. 2010;123:652-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 95] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 33. | Fenske W, Störk S, Blechschmidt A, Maier SG, Morgenthaler NG, Allolio B. Copeptin in the differential diagnosis of hyponatremia. J Clin Endocrinol Metab. 2009;94:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 141] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 34. | Nigro N, Winzeler B, Suter-Widmer I, Schuetz P, Arici B, Bally M, Blum CA, Nickel CH, Bingisser R, Bock A, Huber A, Müller B, Christ-Crain M. Evaluation of copeptin and commonly used laboratory parameters for the differential diagnosis of profound hyponatraemia in hospitalized patients: 'The Co-MED Study'. Clin Endocrinol (Oxf). 2017;86:456-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 35. | Christ-Crain M, Morgenthaler NG, Fenske W. Copeptin as a biomarker and a diagnostic tool in the evaluation of patients with polyuria-polydipsia and hyponatremia. Best Pract Res Clin Endocrinol Metab. 2016;30:235-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 36. | Nigro N, Winzeler B, Suter-Widmer I, Schuetz P, Arici B, Bally M, Blum CA, Nickel CH, Bingisser R, Bock A, Rentsch Savoca K, Huber A, Müller B, Christ-Crain M. Mid-regional pro-atrial natriuretic peptide and the assessment of volaemic status and differential diagnosis of profound hyponatraemia. J Intern Med. 2015;278:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 37. | Krüger S, Ewig S, Giersdorf S, Hartmann O, Frechen D, Rohde G, Suttorp N, Welte T; CAPNETZ study group. Dysnatremia, vasopressin, atrial natriuretic peptide and mortality in patients with community-acquired pneumonia: results from the german competence network CAPNETZ. Respir Med. 2014;108:1696-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Mazón Ruiz J, Josue Banegas E, Pérez Canga JL, González-Blas LB, Menéndez García N, Cavada Bustamante A, Serrano Soto M, Sánchez Álvarez E, Alcázar Arroyo R, Romero-González GA. Precision medicine: "Point of Care Ultrasound" (PoCUS) in the diagnostic approach to the patient with hyponatremia. Nefrologia (Engl Ed). 2024;44:159-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 39. | Chatterjee T, Koratala A. Point of care cardiac ultrasound in the management of hyponatremia: an enhancement to physical examination. CEN Case Rep. 2022;11:6-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 40. | Hoorn EJ, Spasovski G. Recent developments in the management of acute and chronic hyponatremia. Curr Opin Nephrol Hypertens. 2019;28:424-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 41. | Ambati R, Kho LK, Prentice D, Thompson A. Osmotic demyelination syndrome: novel risk factors and proposed pathophysiology. Intern Med J. 2023;53:1154-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 42. | Ayus JC, Moritz ML, Fuentes NA, Mejia JR, Alfonso JM, Shin S, Fralick M, Ciapponi A. Correction Rates and Clinical Outcomes in Hospitalized Adults With Severe Hyponatremia: A Systematic Review and Meta-Analysis. JAMA Intern Med. 2025;185:38-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 43. | Baek SH, Jo YH, Ahn S, Medina-Liabres K, Oh YK, Lee JB, Kim S. Risk of Overcorrection in Rapid Intermittent Bolus vs Slow Continuous Infusion Therapies of Hypertonic Saline for Patients With Symptomatic Hyponatremia: The SALSA Randomized Clinical Trial. JAMA Intern Med. 2021;181:81-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 44. | Fandiño PWA. Suero salino hipertónico: un método sencillo, práctico y seguro para su preparación. Rev Cient Soc Esp Enferm Neurol. 2016;43:30-31. [DOI] [Full Text] |
| 45. | Chienwichai K, Jiwakanon S, Chaiviriyawong K, Wattanakul J, Rojnsaengroung W, Manasuth S, Sangkaew S, Chang A, Mongkolrattanakul P. Urine Electrolyte to Predict Treatment Failure of Fluid Restriction in Syndrome of Inappropriate Antidiuresis: A Prospective Multicenter Study. Kidney360. 2025;6:2097-2106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Verbalis JG, Goldsmith SR, Greenberg A, Korzelius C, Schrier RW, Sterns RH, Thompson CJ. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med. 2013;126:S1-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 947] [Cited by in RCA: 736] [Article Influence: 56.6] [Reference Citation Analysis (1)] |
| 47. | Garrahy A, Galloway I, Hannon AM, Dineen R, O'Kelly P, Tormey WP, O'Reilly MW, Williams DJ, Sherlock M, Thompson CJ. Fluid Restriction Therapy for Chronic SIAD; Results of a Prospective Randomized Controlled Trial. J Clin Endocrinol Metab. 2020;105:dgaa619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 48. | Winzeler B, Lengsfeld S, Nigro N, Suter-Widmer I, Schütz P, Arici B, Bally M, Blum C, Bock A, Huber A, Müller B, Christ-Crain M. Predictors of nonresponse to fluid restriction in hyponatraemia due to the syndrome of inappropriate antidiuresis. J Intern Med. 2016;280:609-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 49. | Scalla PA, Palma SM, Dittmar E, Zamora FJ, Trimino E. Comparative Safety and Effectiveness of Urea and Tolvaptan for the Management of Hyponatremia. J Pharm Pract. 2024;37:1252-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 50. | Martin-Grace J, Tomkins M, O'Reilly MW, Thompson CJ, Sherlock M. Approach to the Patient: Hyponatremia and the Syndrome of Inappropriate Antidiuresis (SIAD). J Clin Endocrinol Metab. 2022;107:2362-2376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 51. | Malhotra I, Gopinath S, Janga KC, Greenberg S, Sharma SK, Tarkovsky R. Unpredictable nature of tolvaptan in treatment of hypervolemic hyponatremia: case review on role of vaptans. Case Rep Endocrinol. 2014;2014:807054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 52. | Llewellyn DC, Oštarijaš E, Sahadevan S, Nuamek T, Byrne C, Taylor DR, Vincent RP, Dimitriadis GK, Aylwin SJ. Efficacy and Safety of Low-Dose Tolvaptan (7.5 mg) in the Treatment of Inpatient Hyponatremia: A Retrospective Study. Endocr Pract. 2025;31:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 53. | Gross PA, Wagner A, Decaux G. Vaptans are not the mainstay of treatment in hyponatremia: perhaps not yet. Kidney Int. 2011;80:594-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 54. | Lewellyn D, Nuamek T, Ostarijas E, Logan Ellis H, Drakou EE, Aylwin SJB, Dimitriadis GK. Low-Dose Tolvaptan for the Treatment of Syndrome of Inappropriate Antidiuretic Hormone-Associated Hyponatremia: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Clinical Effectiveness and Safety. Endocr Pract. 2025;31:956-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 55. | Chander S, Kumari R, Lohana AC, Rahaman Z, Parkash O, Shiwlani S, Mohammed YN, Wang HY, Chi H, Tan W, Kumar SK, Sindhu F. Urea to Treat Hyponatremia Due to Syndrome of Inappropriate Antidiuretic Hormone Secretion: A Systematic Review and Meta-Analysis. Am J Kidney Dis. 2025;85:303-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 56. | Pakchotanon K, Kanjanasuphak N, Chuasuwan A, Gojaseni P, Chittinandana A. Safety and efficacy of proactive versus reactive administration of desmopressin in severe symptomatic hyponatremia: a randomized controlled trial. Sci Rep. 2024;14:7487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 57. | Perianayagam A, Sterns RH, Silver SM, Grieff M, Mayo R, Hix J, Kouides R. DDAVP is effective in preventing and reversing inadvertent overcorrection of hyponatremia. Clin J Am Soc Nephrol. 2008;3:331-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 58. | MacMillan TE, Cavalcanti RB. Outcomes in Severe Hyponatremia Treated With and Without Desmopressin. Am J Med. 2018;131:317.e1-317.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 59. | Sood L, Sterns RH, Hix JK, Silver SM, Chen L. Hypertonic saline and desmopressin: a simple strategy for safe correction of severe hyponatremia. Am J Kidney Dis. 2013;61:571-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 60. | Achinger SG, Ayus JC. Use of Desmopressin in Hyponatremia: Foe and Friend. Kidney Med. 2019;1:65-70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 61. | Misra UK, Kalita J, Kumar M. Safety and Efficacy of Fludrocortisone in the Treatment of Cerebral Salt Wasting in Patients With Tuberculous Meningitis: A Randomized Clinical Trial. JAMA Neurol. 2018;75:1383-1391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 62. | Assadi F. Diagnostic Utility of Fractional Excretion of Urate, Urinary Phosphate and Brain Natriuretic Peptide in Distinguishing Cerebral/Renal Salt Wasting From SIADH in Neurologically Ill Children: A Systematic Review. Clin Endocrinol (Oxf). 2026;104:3-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 63. | Kiewiet A, Schuinder R, Doornebal J, Groeneveld P. Chronic Hyponatremia: The Role of Reset Osmostat in Patients with Suspected SIAD. J Clin Med. 2024;13:3538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 64. | Kinoshita T, Oyama S, Hagiwara D, Azuma Y, Arima H. A machine learning approach for predicting treatment response of hyponatremia. Endocr J. 2024;71:345-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 65. | Lu X, Xu H, Dong W, Xin Y, Zhu J, Lin X, Zhuang Y, Che H, Li Q, He K. Dynamic Prediction and Intervention of Serum Sodium in Patients with Stroke Based on Attention Mechanism Model. J Healthc Inform Res. 2025;9:174-190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 66. | Sicker T, Federbusch M, Eckelt F, Isermann B, Fenske W, Fries C, Schmidt M, Kaiser T. Challenge in hyponatremic patients - the potential of a laboratory-based decision support system for hyponatremia to improve patient's safety. Clin Chem Lab Med. 2023;61:1025-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |