Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.114748
Revised: October 1, 2025
Accepted: January 13, 2026
Published online: March 25, 2026
Processing time: 168 Days and 13.6 Hours
Patients initiating hemodialysis face heightened infection risks due to immunodeficiency and frequent healthcare exposure. Vaccinations against hepatitis B virus (HBV), Streptococcus pneumoniae, and influenza are recommended but often underutilized, particularly in resource-constrained settings.
To evaluate the baseline vaccination status and associated factors of patients starting hemodialysis in a tertiary nephrology center in North India, with the goal of identifying any overlooked opportunities for preventive care.
A prospective cross-sectional study was conducted at the Institute of Liver and Biliary Sciences, New Delhi, from June 2023 to December 2024. Adult patients (≥ 18 years) initiating hemodialysis were evaluated for vaccination status (HBV, pn
Among 132 patients (mean age 52.6 years; 72.7% male), 63 (47.7%) completed HBV vaccination, 37 (28.0%) received pneumococcal and influenza vaccines, and 73 (55.3%) were unvaccinated. Prior nephrology consultation significantly predicted vaccine uptake for HBV (72.1% vs 21.9%, P < 0.001), pneumococcal (47.1% vs 7.8%, P < 0.001), and influenza (47.1% vs 7.8%, P < 0.001). Protective anti-HBs titers were observed in 66.7% of vaccinated individuals. Vaccinated patients had higher rates of arteriovenous fistula creation and transplant evaluation.
Vaccination coverage at dialysis initiation remains suboptimal. Early nephrology referral and structured chronic kidney disease (CKD) care pathways are critical to improving vaccine uptake. Integrating adult immunization into routine CKD management may reduce infection-related morbidity and improve outcomes.
Core Tip: This study highlights critical gaps in adult immunization among patients with chronic kidney disease initiating hemodialysis, with suboptimal coverage for hepatitis B virus, pneumococcal, and influenza vaccines. Notably, a high prevalence of tuberculosis (TB) (16.7%) was observed, underscoring the dual infectious burden in this population. Prior nephrology consultation and pre-transplant evaluation were strongly associated with higher vaccine uptake. Integrating structured vaccination and TB screening into pre-dialysis care pathways offers a unique opportunity for preventive nephrology in resource-limited settings.
- Citation: Puri S, Thakur AN, Meshram HS, Bhagat C, Modasia B, Batheja V, Mathur RP. Vaccination gaps in incident hemodialysis patients: An opportunity for preventive nephrology, a prospective, cross-sectional study. World J Nephrol 2026; 15(1): 114748
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/114748.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.114748
Patients with end-stage renal disease (ESRD) undergoing hemodialysis face heightened vulnerability to infections due to both innate immunodeficiency and frequent exposure to healthcare settings. Infectious diseases are a leading cause of morbidity and the second most common cause of mortality in dialysis patients worldwide, trailing only cardiovascular disease[1]. Preventive strategies, particularly vaccination against common pathogens such as hepatitis B virus (HBV), streptococcus pneumoniae, and influenza virus, are essential components of comprehensive dialysis care[2]. Despite clear recommendations by the Kidney Disease: Improving Global Outcomes (KDIGO) and Centers for Disease Control and Prevention, adult vaccination rates in patients initiating dialysis remain alarmingly low[3,4].
The immunological dysfunction in chronic kidney disease (CKD) is multifactorial, involving uremia-induced alterations in innate and adaptive immunity, persistent systemic inflammation, and malnutrition, all of which contribute to suboptimal vaccine responses[5]. Nevertheless, timely vaccination prior to dialysis initiation, particularly during earlier stages of CKD or during predialysis nephrology care, has been shown to enhance seroconversion rates and improve clinical outcomes[6]. Unfortunately, many patients present late to nephrology services, often requiring emergency dialysis initiation with temporary vascular access, missing the optimal immunization window[7].
HBV vaccination is of particular relevance in the dialysis population due to the risk of nosocomial transmission from contaminated equipment or lapses in infection control. Although a four-dose regimen is recommended in CKD patients using higher antigen doses, completion rates are variable, and response rates can be poor in advanced kidney failure[8]. The anti-HBs antibody titer (≥ 10 mIU/mL) is considered the seroprotective threshold, and levels must be periodically monitored post-vaccination[9]. Pneumococcal and influenza vaccinations are similarly recommended, with annual influenza immunization advised for all dialysis patients, yet global adherence is inconsistent[10].
India bears a disproportionate burden of CKD and dialysis initiation in resource-constrained settings, where late referrals and poor awareness of vaccination guidelines may further compound the problem[11]. There is a paucity of real-world data from low- and middle-income countries evaluating vaccination practices at dialysis initiation. While national programs prioritize immunization in pediatric and antenatal populations, adult immunization, particularly in high-risk chronic disease cohorts, remains neglected[12].
This study was conducted to assess the baseline vaccination status among incident hemodialysis patients at a tertiary nephrology center in North India and to identify factors associated with incomplete immunization. The study particularly focused on HBV, pneumococcal, and influenza vaccination coverage, as well as the influence of prior nephrology consultation, nutritional status, and transplant evaluation on vaccine uptake. By delineating gaps in vaccine administration at the point of dialysis initiation, this analysis underscores missed opportunities in preventive nephrology and highlights the need to integrate adult immunization as a core component of CKD care pathways. The findings may inform targeted strategies to improve vaccination rates, reduce infection-related complications, and ultimately enhance outcomes in this vulnerable population.
This cross-sectional study was conducted at the Institute of Liver and Biliary Sciences, New Delhi, from June 2023 to December 2024. A total of 132 adult patients (≥ 18 years) with CKD initiating hemodialysis for the first time were enrolled. Exclusion criteria included patients with acute kidney injury, prior renal replacement therapy, immunosuppressive therapy, or pregnancy. The study was approved by the institutional ethics committee, and written informed consent was obtained from all participants. Baseline demographic data, comorbidities, clinical presentation, vascular access, vaccination status (HBV, pneumococcal, and influenza), and infection history [e.g., tuberculosis (TB), and HBV] were collected using a pre-designed case record form. Vaccination status was ascertained through a combination of patient interview and review of available personal health records or hospital documentation. When available, vaccination cards, discharge summaries, and dialysis unit records were cross-checked to confirm self-reported status. In cases where records were unavailable, information relied on patient recall. Verification from primary care providers was attempted when feasible; however, this was not uniformly possible across all participants. Data entry and management were performed using the REDCap platform. Laboratory investigations, including blood counts, serum creatinine, electrolytes, liver function tests, and serologies, were also recorded. Statistical analyses were conducted using SPSS software version 22.0. Descriptive statistics were used to summarize patient characteristics. Categorical variables were presented as n (%); continuous variables as mean ± SD. Comparisons between vaccinated and unvaccinated groups were made using χ2 or Fisher’s exact test for categorical variables and Student’s t-test for continuous variables. Kaplan-Meier survival analysis and multivariate logistic regression were used where appropriate. A P value < 0.05 was considered statistically significant.
Table 1 shows the baseline demographics of the study population. A total of 132 incident hemodialysis patients were enrolled, comprising 96 men (72.7%) and 36 women (27.3%), with a mean age of 52.6 years (SD = 13.4). Diabetic kidney disease was the most common etiology of ESRD, accounting for 54 (40.9%) patients, followed by hypertensive nephrosclerosis (n = 19; 14.4%), chronic glomerulonephritis (n = 17; 12.9%), obstructive uropathy (n = 12; 9.1%), and unknown causes (n = 30; 22.7%). Comorbidities were prevalent, with hypertension present in 98 (74.2%) patients, diabetes mellitus in 60 (45.5%), and cardiovascular disease in 27 (20.5%). TB was diagnosed in 22 patients (16.7%), with extrapulmonary involvement observed in 12 (54.5%) of these cases.
| Characteristics | n = 132 |
| Age (years) | 52.6 ± 13.4 |
| Sex | |
| Male | 96 (72.7) |
| Female | 36 (27.3) |
| Etiology | |
| Diabetic kidney disease | 54 (40.9) |
| Hypertensive nephrosclerosis | 19 (14.4) |
| Chronic glomerulonephritis | 17 (12.9) |
| Obstructive uropathy | 12 (9.1) |
| Undetermined | 30 (22.7) |
| Comorbidity | |
| Hypertension | 98 (74.2) |
| Diabetes mellitus | 60 (45.5) |
| Cardiovascular disease | 27 (20.5) |
| Tuberculosis | 22 (16.7) |
| Extrapulmonary TB | 12 (54.5 of 22 TB cases) |
Emergency initiation of dialysis was common, seen in 110 patients (83.3%). Temporary vascular access was used in 102 (77.3%) patients, predominantly via internal jugular vein catheterization. Only five patients (3.8%) had a functioning arteriovenous fistula (AVF) at the time of dialysis initiation. Pre-transplant evaluation and nutritional assessment were completed in 35 (26.5%) and 42 (31.8%) patients, respectively.
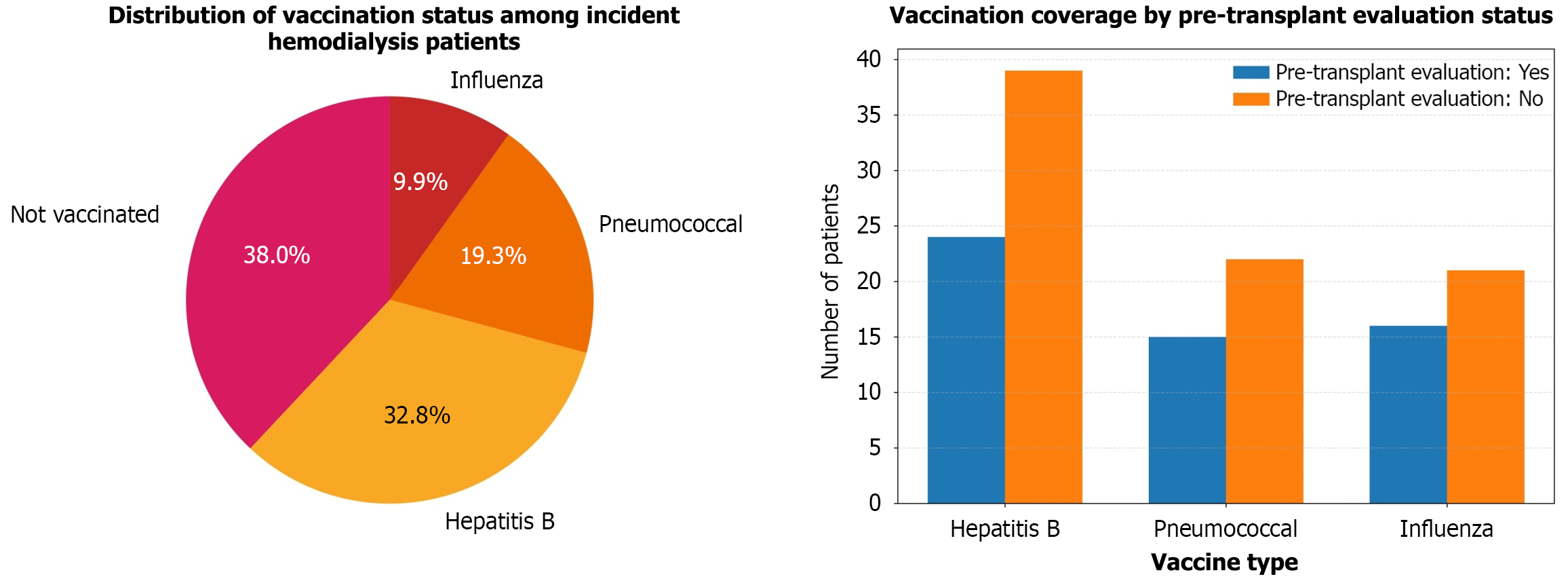
Figure 1 shows the detailed vaccine coverage in the study. Vaccination coverage was suboptimal. Completion of the HBV vaccine series was recorded in 63 patients (47.7%). Pneumococcal and influenza vaccinations were documented in only 37 patients (28.0%). Overall, 73 patients (55.3%) had not received any of the three recommended adult vaccinations. Among vaccinated individuals, a higher prevalence of preformed AVF (7.9% vs 0.0%; P = 0.03) and prior nephrology consultation (52.4% vs 16.4%; P < 0.01) was noted compared to unvaccinated individuals. Infection-related hospitalizations were more frequent in the unvaccinated group (21.9% vs 12.7%), although this difference was not statistically significant (P = 0.08). Hepatitis B surface antigen positivity was identified in eight patients (6.1%), of whom only two had completed the hepatitis B vaccination series. Influenza vaccine uptake was lowest, with only 19 (14.4%) patients vaccinated despite seasonal availability. Stratification by age showed that patients younger than 50 years were more likely to be vaccinated than those aged 50 years or older (56.3% vs 42.9%; P = 0.04). Vaccination status was significantly associated with pre-transplant evaluation. Among those who underwent evaluation, 24 of 35 patients (68.6%) had received HBV vaccination, compared with 39 of 97 (40.2%) in those who had not (P = 0.007). Pneumococcal vaccination rates were also higher among those evaluated for transplant (15 of 35; 42.9%) than in those who were not (22 of 97; 22.7%; P = 0.040). Similarly, influenza vaccination was more common in the pre-transplant group (16 of 35; 45.7%) compared to those without transplant evaluation (21 of 97; 21.6%; P = 0.012).
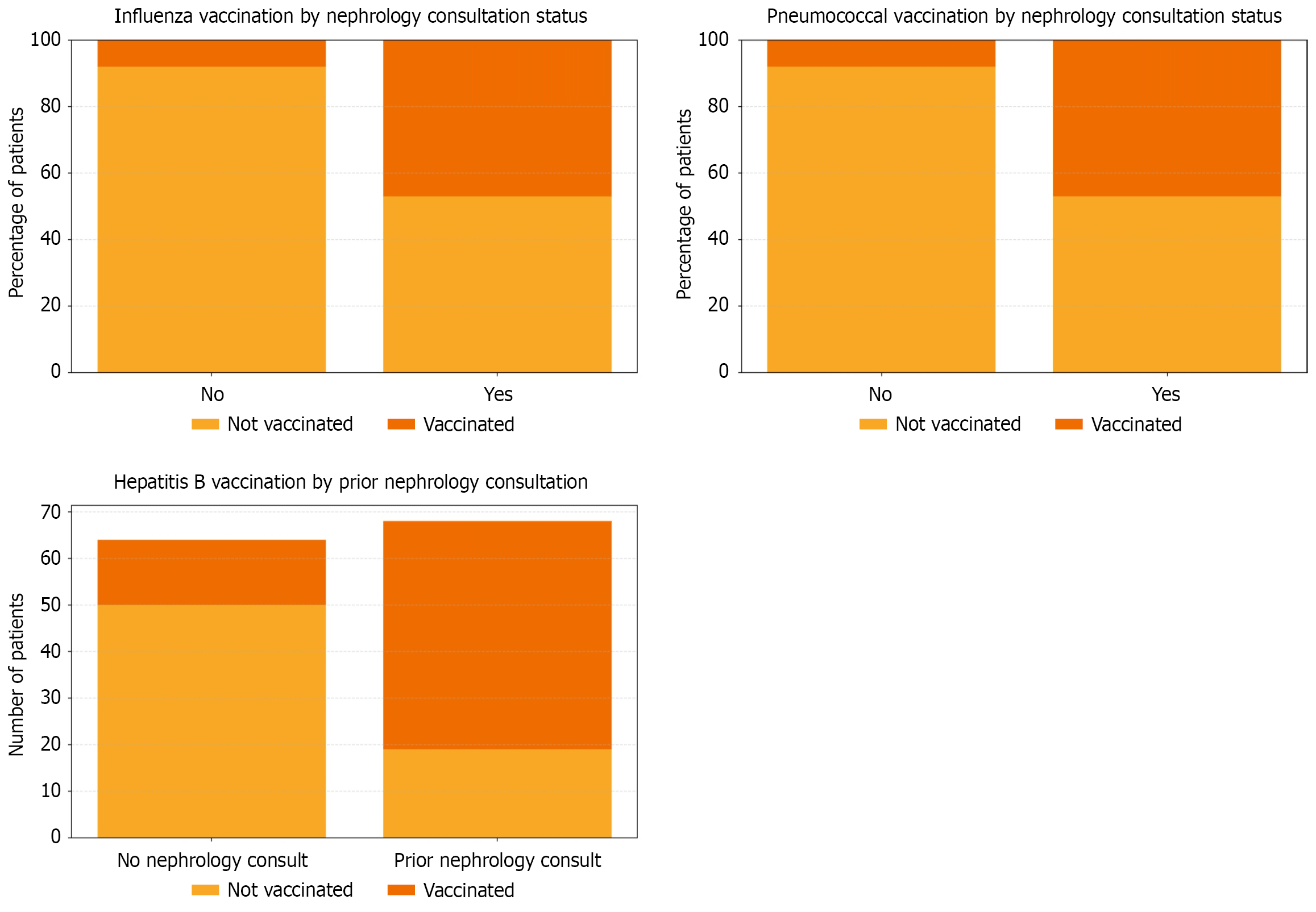
Figure 2 shows the vaccine response with prior nephrology consults. Prior nephrology consultation was significantly associated with completion of the HBV vaccine series (Table 2). Among those with prior nephrology contact, 49 of 68 (72.1%) patients had received the vaccine, compared with 14 of 64 (21.9%) patients without such consultation (P < 0.001; χ2 test). Conversely, 50 of 64 (78.1%) patients without nephrology consultation remained unvaccinated. Similarly, pneumococcal vaccination (Table 3) was more frequent among patients with prior nephrology consultation (32 of 68; 47.1%) than those without (5 of 64; 7.8%) (P < 0.001). Influenza vaccination (Table 4) followed the same trend, with 32 of 68 patients (47.1%) receiving the vaccine in the consultation group vs 5 of 64 (7.8%) in the non-consultation group (P < 0.001).
| HBV vaccination status | False | True | Total | P value1 |
| No | 68 | 1 | 69 | < 0.001 |
| Yes | 21 | 42 | 63 | |
| Total | 89 | 43 | 132 |
| Nephrology consult | No | Yes | Total | P value1 |
| No | 59 | 5 | 64 | < 0.001 |
| Yes | 36 | 32 | 68 | |
| Total | 95 | 37 | 132 |
| Nephrology consult | No | Yes | Total | P value1 |
| No | 59 | 5 | 64 | < 0.001 |
| Yes | 36 | 32 | 68 | |
| Total | 95 | 37 | 132 |
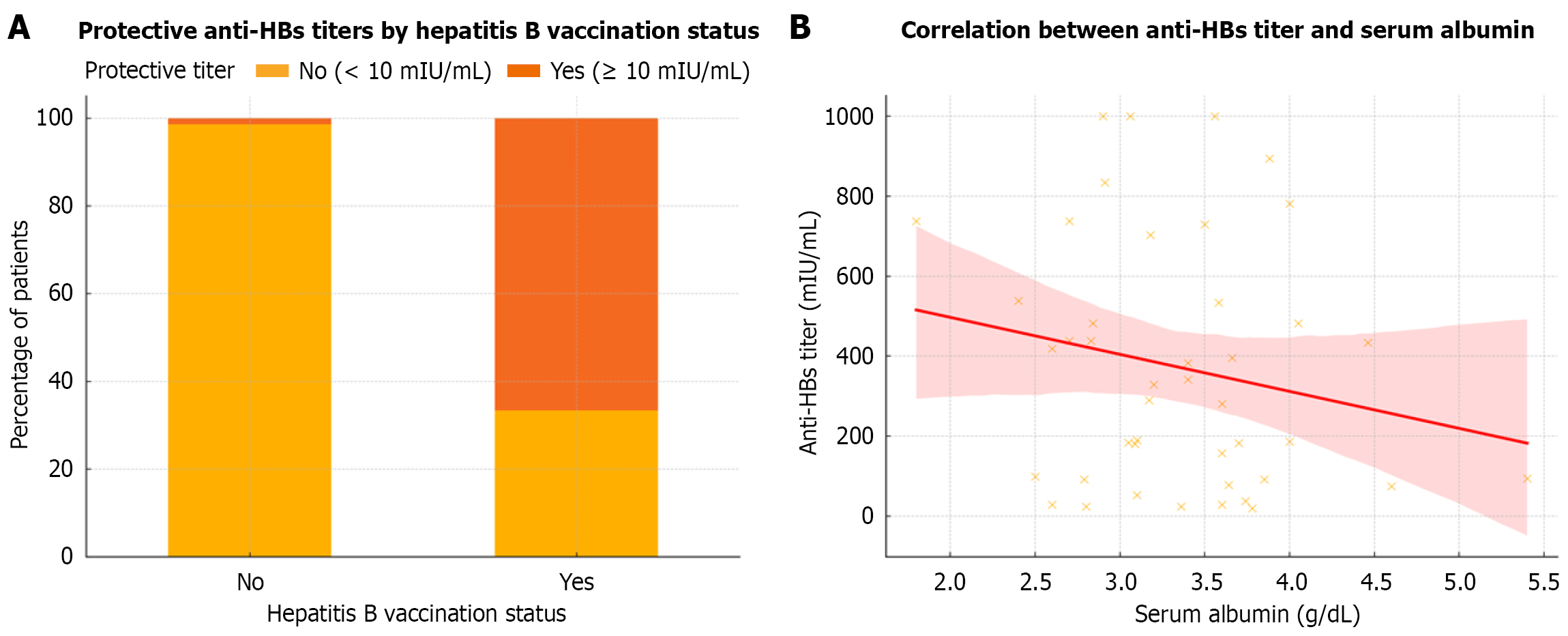
Figure 3 shows the anti-HBs response in the study. Among HBV-vaccinated individuals, 42 of 63 (66.7%) had protective anti-HBs antibody titers (≥ 10 mIU/mL), compared with only one of 69 (1.4%) in the unvaccinated group (P < 0.001), confirming the immunogenicity of the HBV vaccine in this cohort.
No significant correlation was found between anti-HBs titers and serum albumin levels. Spearman’s correlation showed a weak, non-significant negative association (ρ = –0.17; P = 0.28), suggesting that nutritional status, as measured by serum albumin, was not a determinant of vaccine-induced antibody response (Figure 3).
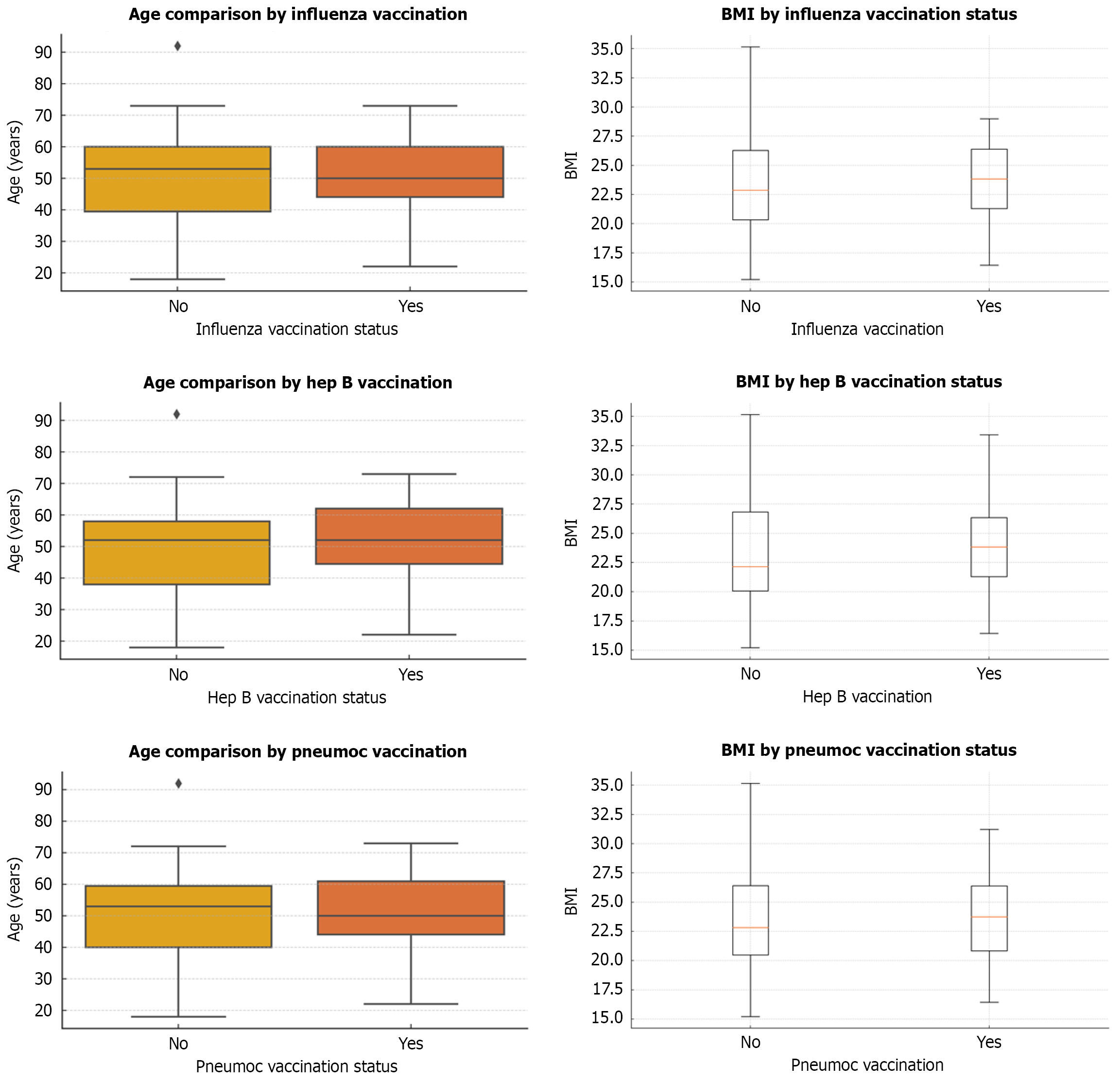
Figure 4 shows a comparison of age and body mass index (BMI) between vaccinated and unvaccinated patients for each vaccine, which both showed no statistically significant differences. For pneumococcal vaccination, mean BMI was 22.04 kg/m2 (SD = 4.77) in vaccinated patients and 21.99 kg/m2 (SD = 4.93) in unvaccinated patients (P = 0.96). For hepatitis B, the vaccinated group had a mean BMI of 22.27 kg/m2 (SD = 4.95) vs 21.69 kg/m2 (SD = 4.83) in the unvaccinated group (P = 0.49). Influenza vaccination showed a mean BMI of 21.83 kg/m2 (SD = 4.87) in vaccinated individuals and 22.13 kg/m2 (SD = 4.92) among those unvaccinated (P = 0.75). Overall, there was no significant association between BMI and vaccination status for any of the three vaccines evaluated.
Patients initiating hemodialysis represent a uniquely vulnerable cohort with high morbidity and mortality due to infections, a consequence of both intrinsic immune dysfunction and the invasive nature of renal replacement therapy. Infections are the second most common cause of death in ESRD, following cardiovascular disease, and a substantial proportion are preventable through vaccination[1]. Our study highlights significant gaps in adult vaccination among incident hemodialysis patients in a North Indian tertiary care center, despite longstanding recommendations advocating timely immunization[2-4].
The observed HBV vaccination rate in our study (47.7%) is suboptimal, especially considering the known risks of nosocomial transmission in hemodialysis units. HBV can spread through contaminated dialysis equipment or lapses in infection control protocols, making immunization critical[5]. Although a higher antigen dose and four-dose schedule are recommended for patients with CKD, the completion and seroconversion rates vary widely[8]. In our cohort, only 66.7% of vaccinated patients achieved protective anti-HBs titers (≥ 10 mIU/mL), emphasizing both the efficacy and importance of timely vaccination before significant uremia-induced immune dysfunction sets in[9]. This finding aligns with the European Renal Best Practice guidelines that recommend early vaccination in predialysis stages to optimize seroconversion[6].
Pneumococcal and influenza vaccine coverage was even lower (28%), despite the established role of these pathogens in respiratory morbidity among dialysis patients. Prior studies have reported variable coverage rates worldwide, with some developed countries achieving > 60% influenza vaccination among dialysis patients[10]. In contrast, our findings mirror those from other resource-limited settings, where adult immunization infrastructure is poorly developed and awareness among healthcare providers remains insufficient[12]. Influenza vaccine uptake was the lowest among the three studied (14.4%), potentially reflecting misconceptions regarding efficacy, safety, or accessibility during seasonal campaigns[7].
A key determinant of vaccine uptake in our study was prior nephrology consultation. Among patients with such contact, 72.1% received HBV vaccination compared to only 21.9% of those without consultation (P < 0.001). Similar trends were noted for pneumococcal and influenza vaccines. These results are consistent with studies demonstrating that early referral to nephrologists facilitates implementation of preventive care practices, including vaccination[7]. Late presentation to nephrology services often results in emergent dialysis initiation with temporary vascular access, bypassing the window for structured pre-dialysis education and immunization[10].
Interestingly, a higher prevalence of AVF at initiation was noted in the vaccinated group, underscoring a broader preparedness among those receiving timely nephrology care. AVF creation requires planning and represents a surrogate marker for proactive CKD management. The correlation between vaccination status and AVF prevalence in our study reaffirms the interdependence of different preventive strategies in CKD care[13,14].
Our study also revealed significantly higher vaccine coverage among patients evaluated for kidney transplantation. This subset likely received structured counseling on immunization as part of pre-transplant protocols, which typically mandate hepatitis B and pneumococcal vaccination. Among those evaluated for transplant, 68.6% received the HBV vaccine compared to 40.2% of those not evaluated (P = 0.007). These findings indicate that transplant readiness workflows may present a parallel opportunity to reinforce vaccine education and delivery[15].
Contrary to expectations, nutritional status as assessed by serum albumin did not significantly correlate with vaccine response, despite literature suggesting that malnutrition may blunt immunogenicity[2]. This could reflect limitations of serum albumin as a sole marker of nutritional status or the modest sample size of our study. Similarly, no significant associations were noted between BMI and vaccine coverage, which may suggest that factors such as healthcare access, awareness, and system-level barriers outweigh individual clinical parameters in influencing vaccination rates.
The high prevalence of infection-related hospitalizations among unvaccinated patients (21.9%) compared to vaccinated counterparts (12.7%), though not statistically significant (P = 0.08), suggests a potential clinical benefit of vaccination in reducing infection burden. While our study was not powered to assess causality, these findings are congruent with data demonstrating reduced hospitalization and mortality with vaccination in dialysis populations[10]. For instance, influenza vaccination has been associated with lower hospitalization rates and all-cause mortality in patients with ESRD[16]. Hence, underutilization of vaccines in this group represents a missed opportunity to reduce morbidity and healthcare costs.
The high prevalence of TB (16.7%) observed in our cohort is a striking finding that warrants closer attention, particularly in the Indian context, where TB remains endemic. CKD itself confers an elevated risk of TB due to the profound immune dysregulation associated with uremia, malnutrition, and repeated healthcare exposure. Both innate and adaptive immune responses are impaired in CKD, leading to defective macrophage function, reduced T-cell responsiveness, and diminished cytokine production, all of which predispose to reactivation of latent TB infection[17]. Several studies have confirmed that the incidence of TB in CKD and ESRD is several-fold higher than in the general population, with extrapulmonary and disseminated presentations being disproportionately represented[18]. This biological vulnerability likely synergizes with the high community prevalence of TB in India, creating a “dual-risk” scenario that amplifies disease burden.
Our findings align with prior Indian studies reporting TB prevalence between 10 and 20% in dialysis populations, underscoring the need for systematic screening and prophylaxis strategies[6,11]. The predominance of extrapulmonary TB in our cohort (54.5% of TB cases) further reflects the atypical disease spectrum commonly seen in immunocompromised states, complicating timely diagnosis and management[19].
Current guidelines, including those from KDIGO and the Indian National Tuberculosis Elimination Program, recommend screening CKD patients for latent TB infection using either the tuberculin skin test (TST) or interferon-gamma release assays, particularly before initiating dialysis or immunosuppression[5]. However, diagnostic limitations-such as poor sensitivity of TST in uremic patients and high background exposure in endemic regions-pose challenges to implementation. Preventive therapy with isoniazid has been advocated in high-risk individuals, though data specific to Indian CKD cohorts remain sparse[1,5].
From a policy standpoint, integrating TB screening into pre-dialysis evaluation could be a valuable addition to CKD care pathways in India. This would involve systematic latent TB infection testing at earlier stages of CKD, chest imaging where indicated, and prompt initiation of anti-TB therapy in confirmed cases. Given the intersection of two major public health challenges of CKD and TB, our study highlights an urgent need for nephrology units to adopt context-specific protocols. Such units can address both infectious and non-communicable disease burdens simultaneously.
The Indian healthcare system has made significant strides in childhood immunization through national programs; however, adult immunization remains largely neglected[20]. Lack of standardized protocols, absence of dedicated immunization clinics for adults, poor record-keeping, and low provider awareness contribute to poor vaccine uptake among high-risk adult populations[21]. In nephrology practice, where patients frequently interface with multiple providers and require coordinated long-term care, integrating vaccine assessment into routine CKD management could yield meaningful benefits. This might include incorporating vaccine status into dialysis initiation checklists, transplant evaluation workflows, and CKD education programs.
Limitations of our study include its single-center design and modest sample size, which may limit generalizability. An important limitation of our study pertains to the method of determining vaccination status. While efforts were made to corroborate patient reports with vaccination cards, hospital records, and dialysis unit documentation, in many instances reliance on patient recall was unavoidable. Patient recall of vaccination history has been shown to be imprecise, with studies demonstrating variable sensitivity and specificity compared to documented records. This introduces the possibility of misclassification bias, potentially leading to under- or overestimation of true vaccine coverage. Given the absence of a national adult immunization registry in India, such challenges are common in clinical research settings. Future studies would benefit from linkage with electronic health records or integration of adult immunization tracking into CKD care pathways to improve data accuracy. Furthermore, we did not assess barriers to vaccination at the patient or provider level, which could have provided actionable insights for quality improvement. However, the study is one of the few from a low- and middle-income country to comprehensively evaluate immunization gaps in incident dialysis patients, with implications for preventive nephrology practice in similar settings.
Our findings underscore a substantial gap in adult vaccination among patients initiating hemodialysis, particularly in hepatitis B, pneumococcal, and influenza vaccines. Timely nephrology consultation, transplant evaluation, and structured CKD care pathways significantly influenced vaccine uptake. Addressing these gaps through integrated, proactive immunization strategies may help reduce infection-related morbidity and mortality in this high-risk population.
| 1. | Dalrymple LS, Go AS. Epidemiology of acute infections among patients with chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:1487-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 189] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 2. | Kato S, Chmielewski M, Honda H, Pecoits-Filho R, Matsuo S, Yuzawa Y, Tranaeus A, Stenvinkel P, Lindholm B. Aspects of immune dysfunction in end-stage renal disease. Clin J Am Soc Nephrol. 2008;3:1526-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 890] [Cited by in RCA: 794] [Article Influence: 44.1] [Reference Citation Analysis (0)] |
| 3. | Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024;105:S117-S314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3017] [Cited by in RCA: 2691] [Article Influence: 1345.5] [Reference Citation Analysis (9)] |
| 4. | Centers for Disease Control and Prevention. Vaccines and Immunizations. [cited 31 December 2025]. Available from: https://www.cdc.gov/vaccines. |
| 5. | Eleftheriadis T, Antoniadi G, Liakopoulos V, Kartsios C, Stefanidis I. Disturbances of acquired immunity in hemodialysis patients. Semin Dial. 2007;20:440-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 249] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 6. | Bharati J, Jha V. Global Dialysis Perspective: India. Kidney360. 2020;1:1143-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 7. | Dhanorkar M, Prasad N, Kushwaha R, Behera M, Bhaduaria D, Yaccha M, Patel M, Kaul A. Impact of Early versus Late Referral to Nephrologists on Outcomes of Chronic Kidney Disease Patients in Northern India. Int J Nephrol. 2022;2022:4768540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 8. | Fabrizi F, Dixit V, Messa P, Martin P. Hepatitis B virus vaccine in chronic kidney disease: improved immunogenicity by adjuvants? A meta-analysis of randomized trials. Vaccine. 2012;30:2295-2300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 9. | Won JH, Zhang Y, Ji B, Logsdon CD, Yule DI. Phenotypic changes in mouse pancreatic stellate cell Ca2+ signaling events following activation in culture and in a disease model of pancreatitis. Mol Biol Cell. 2011;22:421-436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Spetz C, Valkonen JP. Genomic sequence of Wild potato mosaic virus as compared to the genomes of other potyviruses. Arch Virol. 2003;148:373-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 11. | Jha V. Current status of end-stage renal disease care in India and Pakistan. Kidney Int Suppl. 2013;3:157-160. [RCA] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 84] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 12. | Bhattacharyya A, Shahabuddin SM. Adult vaccination in India: A rapid review of current status & implementation challenges. Indian J Med Res. 2024;160:279-292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Kliger AS, Foley RN, Goldfarb DS, Goldstein SL, Johansen K, Singh A, Szczech L. KDOQI US commentary on the 2012 KDIGO Clinical Practice Guideline for Anemia in CKD. Am J Kidney Dis. 2013;62:849-859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 184] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 14. | Allon M, Robbin ML. Increasing arteriovenous fistulas in hemodialysis patients: problems and solutions. Kidney Int. 2002;62:1109-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 585] [Cited by in RCA: 518] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 15. | Kasiske BL, Zeier MG, Chapman JR, Craig JC, Ekberg H, Garvey CA, Green MD, Jha V, Josephson MA, Kiberd BA, Kreis HA, McDonald RA, Newmann JM, Obrador GT, Vincenti FG, Cheung M, Earley A, Raman G, Abariga S, Wagner M, Balk EM; Kidney Disease: Improving Global Outcomes. KDIGO clinical practice guideline for the care of kidney transplant recipients: a summary. Kidney Int. 2010;77:299-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 640] [Cited by in RCA: 520] [Article Influence: 32.5] [Reference Citation Analysis (1)] |
| 16. | McGrath LJ, Kshirsagar AV, Cole SR, Wang L, Weber DJ, Stürmer T, Brookhart MA. Influenza vaccine effectiveness in patients on hemodialysis: an analysis of a natural experiment. Arch Intern Med. 2012;172:548-554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Min J, Kwon SK, Jeong HW, Han JH, Kim YJ, Kang M, Kang G. End-stage Renal Disease and Risk of Active Tuberculosis: a Nationwide Population-Based Cohort Study. J Korean Med Sci. 2018;33:e341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 18. | Subramanian AK, Theodoropoulos NM; Infectious Diseases Community of Practice of the American Society of Transplantation. Mycobacterium tuberculosis infections in solid organ transplantation: Guidelines from the infectious diseases community of practice of the American Society of Transplantation. Clin Transplant. 2019;33:e13513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 100] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 19. | Indian Ministry of Health and Family Welfare. National strategic plan for tuberculosis elimination 2017-2025. [cited 3 March 2025]. Available from: https://ntep.in/node/364/CP-national-strategic-plan-nsp-tb-elimination-2017-25#:~:text=The%20National%20Strategic%20Plan%20(NSP,%2C%20TREAT%2C%20PREVENT%2C%20BUILD. |
| 20. | Lahariya C. A brief history of vaccines and vaccination in India. Indian J Med Res. 2014;139:491-511. [PubMed] |
| 21. | Puliyel JM, Naik P. National immunization policy must tackle vaccine hesitancy and adult vaccination. Indian Pediatr. 2019;56:221-226. |