Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.114527
Revised: November 14, 2025
Accepted: January 19, 2026
Published online: March 25, 2026
Processing time: 173 Days and 8.5 Hours
Kidney transplantation (KT) improves quality of life and confers a survival ad
Core Tip: Kidney transplantation (KT) is lifesaving as a treatment of kidney failure regardless of race and body habitus. However, long-term data on outcomes in people with obesity who are of Black ethnicity are limited, and this can be a significant deterrent to transplantation in this group. In addition, the available evidence supporting the benefits of KT in the Black population with obesity is controversial. We have reviewed the literature and discuss factors (both modifiable and non-modifiable) affecting access to and outcomes from KT. Issues such as suitable policy changes, better characterisation of Black patients (including genetic studies) and steps to ensure appropriate medication dosing are vital. Emerging medications and new surgical interventions also raise hope in improving access for Black patients.
- Citation: Oluyombo R, Karim M, Mason S, Halawa A. Kidney transplantation in obese Black patients: Strategies to improve access and outcomes. World J Nephrol 2026; 15(1): 114527
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/114527.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.114527
Kidney transplantation (KT) is the preferred modality of renal replacement therapy (RRT) for suitable patients with end-stage renal disease (ESRD). It confers a survival advantage, improves quality of life, and is cheaper than remaining on dialysis. The global increase in rates of obesity is having a significant effect not only on prevalence of ESRD, but also on access to KT and subsequent outcomes. It is estimated that 7% of mildly obese, 28% of moderately obese, and 46% of morbidly obese patients are precluded from KT based on their body weight[1].
Black patients (including Black Africans, Black British, African Americans and African Canadian), have higher rates of obesity together with an increased prevalence of chronic kidney disease (CKD) and accelerated progression to ESRD[2,3]. There is already a disparity in access to KT services among Black patients compared to their White counterparts, and obesity further increases their risk of being excluded from KT[4]. This group, therefore, merits specific attention, particularly since obesity is a modifiable risk factor and so should not automatically preclude Black patients from accessing this lifesaving intervention[5].
The prevalence of obesity is higher among patients with CKD and those starting any form of RRT compared to the general population[6]. Black patients face a double challenge. Compared to White Caucasians, they have significantly higher rates of obesity (and extreme obesity) both within the general population[7,8], and among potential KT recipients[2]. Many transplant centres are reluctant to accept obese Black patients for this important life-improving and cost-saving procedure despite the lack of definitive evidence to support this stance[9]. While there is an increased risk of surgical complications in obese patients, ongoing improvement in surgical techniques and perioperative management has led to better outcomes[10,11]. This review aims to evaluate the existing evidence and highlight challenges and potential interventions that may improve access to KT and subsequent outcomes for obese Black patients.
Obesity is an independent risk factor for the development of CKD and progression to ESRD[12]. Studies from different continents have shown a significantly higher prevalence of obesity among Black patients than Caucasians (45.4% vs 37.4%)[13-15]. In the Coronary Artery Risk Development in Young Adults study, increasing class of obesity was associated with a higher risk of deterioration in kidney function among Black patients than Caucasians[16]. CKD is more prevalent (and, on average, diagnosed seven years earlier) in Black patients than in Caucasians, and albuminuria is 40% higher[17]. High-risk Black patients with hypertension or diabetes, the two main comorbidities associated with obesity, have a higher prevalence of CKD of 35.6% and 32.6% respectively, compared to 15.8% in the general population[18]. In a systematic review from Africa by Adeboye et al[19], obese Black Africans had a higher burden of inflammatory and cardiovascular risk markers and increased vascular resistance than Caucasians. In the United States, African Americans are 1.4 times more likely to be obese, and 40% and 60% more likely to be diagnosed with hypertension and diabetes respectively, than Caucasians[20]. As already mentioned, hypertension and diabetes are major risk factors for CKD, present in more than 70% of patients with ESRD[21]. Rates of hypertension in adults in Africa are amongst the highest globally, with almost half of adults over the age of 25 being affected[22,23]. The prevalence of obesity among urban dwellers in the region is 20%-50%, and individuals with obesity have double the risk of hypertension. In the Research on Obesity and Diabetes among African Migrants study, Agyemang et al[24], found a high prevalence of obesity (up to 21% and 54% in men and women, respectively) and diabetes (up to 15% and 10% in men and women, respectively) among African migrants living in Europe.
In the United Kingdom, the median age for incident dialysis among Black patients is 56 years compared to 66 years in Caucasians. In addition, there is a higher prevalence of ESRD than in Caucasians: 12.3% vs 10.5% in 35-44-year-old. Among patients with ESRD, 43% of Black patients have functioning kidney transplants compared to 59% of Caucasians[25].
Data from the United States have shown that African Americans are four times more likely than Caucasians to be diagnosed with ESRD, and while constituting only 12% of the population, they represent 32% of dialysis patients[26]. The prevalence of ESRD is nearly three and a half times higher than in Caucasians (5854 vs 1703, per million people) and indeed any other ethnic group. Similarly, the incidence of ESRD is higher (834 vs 312, per million people). In terms of waiting time for KT, African Americans had a median of 60 months compared to 41 months in Caucasians. African Americans were also less likely than Caucasians to have received specialist pre-dialysis care for more than 12 months (27.1% vs 33.1%). Furthermore, 34.1% of Caucasians with ESRD underwent KT compared to only 19.5% of African Americans (a ratio of 1.7), and this gap has widened further with a ratio of Caucasians to Black patients of 2.5 in the 2020 report. Black patients are less likely to have pre-emptive KT, and the disparity continues up to 3 years on dialysis[17,26].
Obesity exerts multiple effects throughout the spectrum of kidney disease, from early CKD to ESRD (including dialysis and KT), and this influence is even more pronounced in Black patients. Compared to Caucasians, African Americans with CKD show an increase in mortality from all causes [relative risk (RR): 1.78, 95% confidence interval (CI): 1.14-2.78] and cardiovascular events (RR: 2.11, 95%CI: 1.00-4.47)[27,28]. Outcomes following KT are less clear-cut. While some authors have reported that obesity does not significantly impact results[29,30], others have suggested that it is associated with poorer outcomes, particularly among patients with extreme obesity[31]. Potential perceptions that obese Black patients have improved survival on dialysis and poorer outcomes following KT may lead to their further exclusion from or delay in undergoing transplantation, thus exacerbating the existing disparity. However, this misses the point that, even if there is some narrowing of the risk/benefit ratio, KT still confers better patient outcomes in Black patients than remaining on dialysis and is more cost-effective[32].
Previous studies report that Black adults are less likely to receive both deceased and live donor KT. However, re
To date, most studies on this subject have been conducted in the United States and United Kingdom; however, this racial disparity is global; in Canada, patient race is a strong and independent predictor of access to KT[8], yet in terms of outcomes, graft and patient survival are comparable to Caucasians[38].
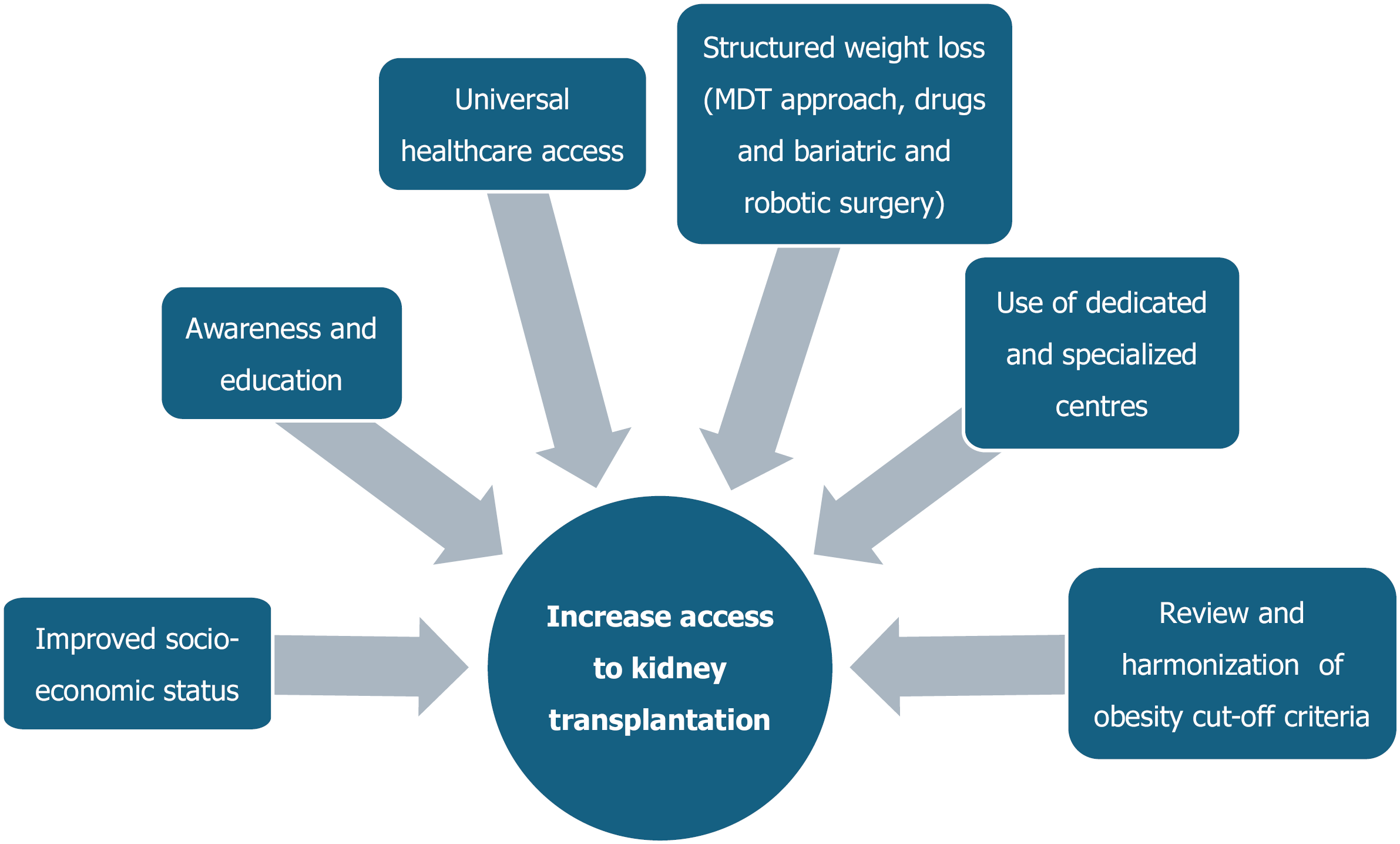
In insurance-based healthcare systems, the lower rate of coverage amongst these disadvantaged patients further impacts decisions by health care practitioners to list them for KT, leading to longer time on dialysis and poorer overall outcomes[39]. Even after successful KT, ongoing follow-up and provision and monitoring of immunosuppressive therapy can be a real challenge in this group, further compromising graft survival and other long-term outcomes. In the United Kingdom, up to 20% of Black patients are classified as socially deprived; despite the universal health care system, this group is less likely to be considered for pre-emptive or live donor transplantation [odds ratios (OR): 0.35 and 0.4, respectively], or to undergo KT (OR: 0.66)[40]. Improvement in socioeconomic status and other factors (Figure 1) will likely improve access to KT among this cohort.
An additional challenge is that Black patients still lag significantly behind in living kidney donation (LKD), which confers even better outcomes than cadaveric KT[41]. There is a clear need for a pragmatic solution to improve access to LKD. The National Black, Asian, Mixed Race and Minority Ethnic Transplant Alliance is heavily involved in raising awareness of organ and stem cell donation, with a view to increasing the number of Black, Asian, Mixed Race and Minority Ethnic donors. Health literacy, LKD transplant knowledge, and culturally sensitive community engagement peer support programmes are potential means of addressing barriers and inequity. Included in the National Black, Asian, Mixed Race and Minority Ethnic Transplant Alliance programme is the GOLD Phone Buddy Scheme, a good example of targeted, tailored peer support for Black patients at the start of their kidney journey to support living donation discussions[42].
In Africa, KT has lagged, with no robust data available. This is compounded by the non-availability of renal registries in most African countries, and the high cost of surgery and anti-rejection medication. The cost of KT for the first six months averages United States $17550[43]. This is out of the patient’s pocket and therefore prohibitive. Almost 60% of kidney grafts are from live donors, with 70% from family members. The bulk of KT in Africa occurs in South Africa, northern Africa, and Nigeria. The level of awareness, knowledge and perception of organ donation has been evaluated among health care workers in Nigeria. Despite good knowledge of organ donation among this group (> 90%), perception of health workers towards organ donation is poor. Only 1 in 10 would donate, while 1 in 3 would volunteer to counsel families of potential donors. Around 40% reported that their religion did not allow them to donate[44]. This implies that to move towards a successful deceased organ donation programme in this population, health care professionals and teams should be targeted for a change of public perception through education, trust building and communication. In Africa, there are legislative, socio-cultural, religious, and administrative challenges confronting cadaveric KT program
Analysis of outcomes following KT is complex, as many variables contribute. However, geographical and racial factors appear to play a significant part. In a report comparing United Kingdom and United States data at one year post KT, the incidence of graft failure in Black patients was twice as high in the United States than in the United Kingdom (hazard ratio: 2.15, 95%CI: 1.91-2.43). Recipient mortality was also 20% lower in the United Kingdom cohort than their United States counterparts[45]. A study examining a cohort of patients in France compared outcomes of Black adults and Caucasians. Encouragingly, there was no difference in patient or graft survival at 5 years[46]. Similarly, a Canadian registry analysis of over 20000 KT patients found that, although Black patients were less likely to be transplanted than Caucasians, there was no difference in graft survival even after correcting for co-morbidities and socioeconomic status. Furthermore, mortality was 50% lower among Black adults than Caucasians[47]. However, it is important to note that the proportions of Black patients in the French and Canadian studies, at 12.8% and 4% respectively, were lower than those reported in most United States studies (sometimes up to 50%)[48]. In contrast, an analysis by Chakkera et al[49] of data from the United States Department of Veterans Affairs revealed that, even in the presence of free access to healthcare, KT graft survival was inferior among Black patients compared to Caucasians (RR: 1.31; 95%CI: 1.26-1.36). The authors could not account for this difference through analysis of comorbidities or socioeconomic factors. However, it should be noted that patients in this study were transplanted before 2000, and clinical practice, particularly regarding the use of immunosuppression, has evolved significantly[50,51].
Obese Black patients face significant challenges in undergoing KT. For surgical procedures generally, Black patients have been demonstrated to have an increased risk of perioperative complications[52]. In KT recipients, obesity is associated with an increase in duration of operation and intensive care unit admission, delayed wound healing and risk of wound infection, delayed graft function, and premature death[31]. However, in a study by Gore et al[53], outcomes of KT in morbidly obese patients were like those in recipients with diabetes. Compared to normal weight Black recipients, in univariate analysis, overall graft and patient survival rates are lower by 38% and 30%, respectively among obese Black patients; however, there is no significant difference in graft failure and patient survival between Black patients and Caucasians in multivariate analysis[54].
Currently, there remains a lack of consensus around the criteria for KT in obese patients in general. This is compounded further by the methods used to define obesity. Although body mass index (BMI) is easy to calculate, its use is not straightforward. The body composition of Black and White adults is different. The distribution of subcutaneous fat and length of bones compared to the trunk are different in the two groups. Black females have higher musculoskeletal mass than White females. Consequently, a methodological error can occur when estimating relative body fat in Black adults if these differences are not taken into account and leading to misclassification of obesity[55].
BMI cannot differentiate between peripheral and visceral adiposity, and it does not account for body composition, thereby overestimating obesity in patients with large muscle mass. As BMI has low sensitivity, the United Kingdom National Institute for Health and Care Excellence (NICE) recommends waist circumference when BMI is less than 35 kg/m2[56]. Interestingly, Kovesdy et al[57] reported lower mortality following KT in people with higher BMI, whereas, after adjusting for BMI, higher waist circumference was associated with a significant increase in mortality. The experience and expertise of the transplanting centre also play an important role in the outcome of KT in obese patients.
Delayed graft function and acute rejection episodes are more common in obese recipients, who also have a higher prevalence of cardiovascular disease, tend to be older, and are on dialysis for longer before KT than non-obese patients. These differences are often even more marked among Black patients (Table 1)[58].
| Modifiable factors | Effect on kidney graft dysfunction | Non-modifiable factors | Effect on kidney graft dysfunction |
| Increasing waiting time | Enhance | APOL1 | Enhance |
| Hypertension | Enhance | MYH9 gene | Enhance |
| Cardiovascular risk factors | Enhance | CYP3A5 | Enhance |
| Induction protocol; alemtuzumab; OKT3; ATG vs IL-2 Ab | Reduce | MDR1 polymorphism | Enhance |
| Poor medication compliance | Enhance | ||
| Persistent proteinuria | Enhance | ||
| New onset diabetes after transplant | Enhance | ||
| Increased leptin: Adiponectin ration | Enhance | ||
| Obesity | Enhance |
Factors related to immunosuppressive regimens also play an important role in graft outcomes, as both obese individuals and Black patients exhibit significant differences in pharmacokinetic and pharmacodynamic characteristics. Drug absorption, distribution, metabolism and excretion can be affected in obese patients, with a significant contribution from racial pharmacogenetic differences. For example, compared to Caucasians, a greater proportion of Black patients (50% vs 10%-30%) express polymorphisms of the cytochrome P450 family 3 subfamily A member 5 enzyme, affecting the metabolism of drugs such as tacrolimus and potentially resulting in increased rates of acute rejection, drug toxicity, or chronic allograft dysfunction[59,60]. Similarly, polymorphism of the multi-drug resistance gene 1 that affects drug absorption, distribution, and metabolism is more common among Black patients than Caucasians (84% vs 22%); this can lead to “steroid-resistant” rejection, in turn resulting in higher intensity immunosuppressive regimens[61]. In Black patients, induction protocols using agents such as alemtuzumab, muromonab (anti-CD3), or anti-thymocyte globulin, when compared to more common regimens using anti-interleukin 2 receptor antibodies (such as basiliximab), are associated with a 32% reduction in the incidence of acute rejection and 9% increase in graft survival, an effect more marked than in Caucasians[62]. In addition to these biological factors, poor concordance with medication regimens is commoner among Black recipients, with one United States study showing rates of 50% compared to 28% in Caucasians[63]. Proteinuria, an independent risk factor for cardiovascular disease and progression of kidney disease, is more common in both obese patients and Black patients. In a cohort of KT recipients with a predominantly obese Black group, Kwan et al[48] showed that proteinuria increased with adiposity, and severity of obesity was associated with an increasing level of proteinuria (OR: 1.48-2.43) and increased risk of graft failure (OR: 1.02-1.25) at 4 years. Obesity likely contributes to this risk by promoting a chronic inflammatory state with resultant metabolic stress that may also reduce graft longevity and increase cardiovascular risk[64].
Other racial and genetic differences may also affect the development and progression of CKD and outcomes following KT. Black patients more commonly express isoforms of the apolipoprotein L1 (APOL1) and myosin heavy chain 9 genes in podocytes, and these are associated with higher rates of hypertensive nephrosclerosis, focal segmental glomerulosclerosis, proteinuria, interstitial fibrosis, human immunodeficiency virus-associated nephropathy, and other renal diseases[65-68]. Such APOL1 variants are present in only around 13% of all United States patients with African ancestry, but in almost 70% of those who develop focal segmental glomerulosclerosis and human immunodeficiency virus-associated nephropathy[69]. There also appear to be racial differences in the pathophysiology of renal disease. For example, while the histopathological features of hypertensive nephropathy in Black patients are mainly glomerulosclerosis and interstitial fibrosis, Caucasians more commonly exhibit intimal and medial thickening[70]. In a United States prospective cohort study by Grams et al[71], the rate of decline in kidney function was 1.5 mL/minute/1.73 m2/year among Caucasians with CKD compared to 2.1 mL/minute/1.73 m2/year and 2.3 mL/minute/1.73 m2/year for African Americans with low and high-risk variants of APOL1, respectively. It is therefore possible that addressing risk factors such as dyslipidaemia, hypertension, and cigarette smoking may confer less benefit on the progression of CKD among Black patients than Caucasians[72]. Post transplant diabetes mellitus (PTDM) is common after KT, with an overall prevalence of around 10%, and the incidence is highest in the first six months (4%-25%)[73,74]. The precise role of PTDM in outcomes following KT is, however, not entirely clear. It is more prevalent among Black recipients than their Caucasian counterparts (OR: 2) and shows a linear increase in prevalence for every 1 kg increase in body weight above 45 kg[75]. Kasiske et al[76] reported a 60% increase in the risk of graft failure in affected patients. Conversely, data from the United States Organ Procurement and Transplantation Network suggest that PTDM alone does not adversely affect graft survival[61]. Regardless, Black recipients have a 1.5-2-fold increased incidence of PTDM compared to Caucasians over and above the effect of obesity[77,78]. Hepatitis C infection, which tends to be more prevalent among Black recipients, has also been suggested to predispose to PTDM[79]. Hypomagnesaemia is common among Black recipients than Caucasians and may promote insulin resistance[80]. Use of steroid-free or steroid-sparing immunosuppressive regimens has been reported to reduce the incidence of PTDM, as have calcineurin inhibitors: For example, prescribing ciclosporin in place of tacrolimus if the immunological risk is low, and avoiding combined use of sirolimus and tacrolimus[51]. Bariatric surgery has been reported to reduce the incidence of PTDM among KT recipients with severe obesity[81].
Application of precision medicine is a viable option to improve transplant outcomes in Black recipients. This combines clinical phenotypes and biological information to formulate suitably refined treatment strategies, rather than a one-size-fits-all approach. For example, a single-centre study in the United Kingdom, where all patients received tacrolimus according to their weights, reported lower median tacrolimus levels among Black patients in the first 4 weeks post KT, leading to a higher incidence of acute rejection compared to other ethnicities[82]. Personalised plans using pharmacogenomic information, monitoring biomarkers, artificial intelligence, and strategies to improve patient concordance may further bolster outcomes. Pre-transplant genotyping of Black patients should be encouraged as it may improve graft and recipients’ survival.
Generally, there is no standardized approach to management strategies and treatment goals in potential KT recipients who are obese. Despite the disproportionately high prevalence of obesity among Black patients, participation in weight loss intervention programmes are lower than that of Caucasians. There is a disparity in the outcomes of behavioural weight intervention programmes, and it is important to review what approaches are most appropriate and effective for Black patients[83]. In managing weight among people with obesity, different approaches, including lifestyle changes, pharmacological and surgical interventions, have been employed.
Structured weight loss programmes for obese potential KT candidates require a multifaceted approach, including dietary and lifestyle changes, psychological interventions, use of pharmacological therapy, and surgery. A personalized, patient-centred programme and a multidisciplinary team (including an experienced renal dietician) are crucial[84]. In one United States study, Black candidates on the KT waiting list were 24% more likely to have lost weight than Caucasians, but improved access to KT following weight loss was only achieved in the latter group[85].
Generally, lifestyle interventions to aid weight loss are extremely challenging, and even more so in Black patients. In a population-based study of about 33000 participants in the United States, middle-aged Black adults were 43% less likely than Caucasians to engage in moderate physical activity. It has been suggested that reduced levels of physical activity and fitness confer as much as a 20% excess risk of CKD in Black patients than Caucasians[86]. Sadly, attempts to modify diet and exercise habits often yield disappointing results, and any weight loss achieved is rarely sustained[87]. Dialysis is associated with fatigue and reduced exercise tolerance, further challenging weight loss. Nevertheless, moderate to vigorous physical activity is recommended to improve health-associated quality of life and exercise tolerance, and to reduce cardiovascular risk in patients with CKD. Regarding dietary interventions, caution is warranted in their use to reduce weight loss in dialysis patients as this group is already subject to significant restrictions (such as potassium and phosphate intake), and further restrictions can increase the risk of malnutrition.
Pharmacological therapy with drugs such as glucagon-like peptide-1 receptor agonists (GLP-RAs) gives a glimmer of hope with obesity management beyond lifestyle modifications (Figure 2)[88]. In a United States-based retrospective study that included KT recipients with type 2 diabetes mellitus and a considerable proportion of Black patients, use of GLP-RAs among dialysis patients was associated with significant weight loss, 23% lower mortality risk, and 66% better chance of being listed for KT[89]. However, in that study, there was a concern about a possible 32% increase in the risk of diabetic retinopathy. In another study, use of GLP-RAs in KT recipients led to a 49% reduction in death-censored graft loss and a 31% reduction in mortality[90]. In the United Kingdom, in addition to reduced calorie intake and regular physical exercise, NICE has now approved both semaglutide and tirzepatide for use in adults with a BMI of at least 35 kg/m2 (reduced by 2.5 kg/m2) in groups with increased risk (including Black Africans) and at least one weight-related health condition. At 6 months, the dose and decision to continue with the medication are to be reviewed if weight reduction is less than 5%. Currently, GLP-RAs are available only as injections, but upcoming oral agents would reduce the burden of repeated injections. In the phase 3 ATTAIN-1 and OASIS-1 trials of oral GLP-RAs, orforglipron and semaglutide reduced average body weight by 12.4% and 15.1% over 72 weeks and 68 weeks, respectively[91,92].
In a United States Renal Data System (USRDS) analysis of 127327 participants, there was 85% higher chance of kidney transplant waitlisting and 21% lower risk of mortality with GLP-RA use[93]. When GLP-RA was compared with bariatric surgery, GLP-RA demonstrated consistently reduced risk of mortality. Those who had surgery were more likely to be Black than were glucagon-like peptide-1 users. There was an initial higher mortality among those who had bariatric surgery, but there was a long-term survival advantage and improved transplant access[94].
In recent years, bariatric surgery has gained an increasingly important role in the management of obesity, with such procedures recommended in patients with a BMI over 40 kg/m2 or who have one or more obesity-related comorbidities (which may improve with weight loss) and a BMI above 35 kg/m2. To expand the uptake and reduce inequalities in access to surgery, NICE in the United Kingdom recommends a 2.5 kg/m2 lower BMI threshold for at-risk ethnic populations, including Black patients. Interestingly, and of benefit to potential KT recipients with obesity, the previous recommendation requiring all non-surgical approaches to have been implemented before considering bariatric surgery has been removed[95].
There are varying opinions on the use of BMI cut-offs for eligibility for KT and indication for bariatric surgery. This may contribute to reduced access to KT by Black patients. While the limits for KT are most commonly around 35 kg/m2 in the United Kingdom, higher levels of 35-45 kg/m2 are often used in the United States and in Canada[96-98]. There is now a more liberal approach, as evidenced by a recent survey of United States transplant programmes where a BMI cutoff of 40 kg/m2 was commonly used for evaluation and listing at more than two-thirds of centres, and over 70% of programmes had weight management units within their centre, with specific integration into transplant program in 1 out of 5 institutions[99].
Bariatric surgical techniques lead to weight reduction by physically restricting the ability to take in food [such as laparoscopic sleeve gastrectomy (LSG), or laparoscopic adjustable gastric bands (LAGB)], by impairing nutrient absorption [for example, by Roux-en-Y gastric bypass (RYGB)], or through a combination of both. Impressive weight loss of up to 70% can be seen in patients undergoing such procedures, but with a reported mortality rate of around 2%[100]. RYGB carries significant risks, including postoperative acute kidney injury, long-term oxalate nephropathy, and nephrolithiasis. In addition, the effects on gut absorption can lead to difficulties in achieving stable levels of immunosuppressant drugs, potentially affecting outcomes following KT[101,102]. A more recent development in bariatric surgery is the use of laparoscopic techniques. These are associated with shorter operating times and lower rates of complications such as post-operative leaks, and their use has further widened the eligibility criteria for bariatric surgery, in turn leading to KT being undertaken in patients who would previously have been excluded[103-105].
In a study by Thomas et al[106] of 31 patients undergoing RYGB before KT (including over 50% Black patients), there were significant improvements in blood pressure and glycaemic control; 90% of patients attained a BMI of < 35 kg/m2 following surgery. There was no reported difference in subsequent overall graft outcome compared with normal-weight recipients. However, surgical complications did occur in 25.8% of patients, and the acute rejection rate was higher than in control KT patients who had not undergone bariatric surgery (42.8% vs 15.7%). In another study of 41 patients (35% Black patients) with morbid obesity undergoing LSG before KT, mean BMI reduction over 16 months after LSG and before KT was 9 kg/m2, with a significant reduction in blood pressure, need for antihypertensive therapy, and insulin requirements. There was a 97% graft survival among the LSG group at 12 months[107]. Lee et al[108] conducted a meta-analysis that included 453 patients undergoing bariatric surgery prior to KT and 201 patients undergoing bariatric surgery after KT (4.6-15 years), respectively. The 1-year graft survival among those who had bariatric surgery ranged from 70% (in the early studies) to 100% most recently. A limitation of this analysis was that the studies included were not randomised controlled trials. A United USRDS registry report of 188 patients (> 40% Black) who underwent bariatric surgery and were then placed on the KT waiting list showed an average reduction in BMI from 40.1 kg/m2 to 35.1 kg/m2 over 16 months following bariatric surgery. Almost 70% of those on the waiting list subsequently underwent KT[109]. There was no mortality up to 3 months post-operatively. Fifteen years on, Sheetz et al[110] analysed updated USRDS data (2006-2015) and reported that, at 5 years, bariatric surgery was associated not only with an increase in the chance of KT by 82%, but also a reduction in all-cause mortality by 49% at 1 year post KT when compared with standard care.
To further reduce waiting times for KT, LSG has been performed concurrently with robot-assisted KT. In one such series of 18 patients, the LSG group had a mean BMI of 46 kg/m2 at the time of transplantation, 36.6 kg/m2 at 3 months and 32 kg/m2 at 12 months. The simultaneous LSG and KT procedure was associated with a longer operative time, but warm ischaemic time and hospital stay were comparable to those in the KT group. Graft function at 12 months showed no statistical difference[111]. However, this data should be interpreted with caution due to the small sample size and the relatively short period of follow-up. Nevertheless, this strategy merits further evaluation.
Despite these potentially positive reports, some cautions have been raised around the role of bariatric surgery in KT. In an analysis of over 430000 patients (including 17.6% Black patients), thirty days after bariatric surgery the Black patient cohort had significantly higher rates of mortality (0.13% vs 0.09%), operative complications (4.14% vs 3.41%), need for further intervention (1.34% vs 1.24%), and hospital readmission (5.03% vs 3.56%) than the overall group[112]. Surgical problems included band displacement and erosion, visceral obstruction, dysfunction of LAGB ports, and infection related to the foreign bodies. LSG has a shorter operative time than RYGB and may be more suitable for patients with significant comorbidities, in whom prolonged anaesthesia and surgery are not ideal[113].
As alluded to above, the effect of bariatric surgery on the absorption of immunosuppressive drugs needs to be considered. Unlike LSG and LAGB, RYGB interferes with gut absorption. It has been shown to affect the pharmacokinetics of tacrolimus, sirolimus, and mycophenolate mofetil, both in terms of the maximum plasma concentration achieved and the time to reach that level[108]. However, if these issues are considered, RYGB can still yield positive outcomes: Choudhury et al[114] found that obese patients with ESRD who had RYGB showed improved survival, gaining 1.3 years of life compared to those undergoing LSG before KT. In a cohort studied by Cohen et al[115], 64 patients (from 1994 to 2017) underwent bariatric surgery before or after KT. Both groups had a reduced risk of graft failure at 5 years compared with obese patients undergoing KT without bariatric surgery, with hazard ratios of 0.31 and 0.85 respectively. BMI reduction was also maintained for at least 5 years after bariatric surgery.
There are other potential risks from bariatric surgery that can particularly affect patients with renal disease. Post-operative acute kidney injury has been described in 2.9% to 10% of cases, so careful attention should be paid to perioperative fluid management as fluid balance and haemodynamic factors may play a role[116,117]. Short bowel syndrome (resulting from issues such as bowel adhesions and mesenteric ischaemia, leading to the need for small bowel resection) can occur in around 4% of patients and may also affect the absorption and monitoring of immunosuppressive drugs[118]. In severe cases, short bowel syndrome, common in RYGB, may necessitate the use of parenteral nutrition or even small bowel transplantation[119]. Finally, bariatric surgical techniques (such as RYGB) that lead to malabsorption can result in the development of hyperoxaluria and consequent oxalate nephropathy, although this does appear to be rare among Black patients[120-122].
Post KT, there is an average weight gain of about 10 kg, exacerbated when steroids are used, and this is worse in recipients with pre-existing obesity. Indiscriminate eating habits and poor physical exercise contribute to this. While it is important to avoid malnutrition and metabolic disorders, it is essential to promote a healthy lifestyle[122].
The global burden of obesity is increasing, contributing to the burden of CKD. Black patients face challenges: Their underlying rates of CKD are higher, they show a faster progression to ESRD, and they tend to have more comorbidities. They suffer longer delays in being listed for KT, and the resultant increased time spent on dialysis further worsens long-term outcomes. The current consensus is that, despite the increased perioperative risks, KT results in better long-term survival than other forms of RRT. Data regarding KT in obese sub-Saharan African patients are particularly limited. There is a need for renal registries, multinational cohort studies, and clinical trials in this population. It is imperative to conduct longitudinal studies on BMI and other relevant anthropometric measurements, adjusted for ethnicity. There is a need for research to establish the mechanistic relationship between obesity and poor outcomes post-KT. Proactive drug prescription, including the use of pharmacogenetics, should help to reduce the risk of under- or over-dosing immunosuppressive medication. The utility of integrated weight management strategies and teams within transplant programmes cannot be over-emphasized. Ethical, cultural, and logistical factors pose major challenges and are daunting the design of randomized controlled trials and establishment of registries. However, it is vital to overcome these obstacles to improve access to optimal treatments and outcomes in these disadvantaged populations.
The authors acknowledge John Losasso of Sir Thomas Browne Library, Norfolk and Norwich University Hospital, Norwich for his tireless assistance in helping with literature search.
| 1. | Segev DL, Simpkins CE, Thompson RE, Locke JE, Warren DS, Montgomery RA. Obesity impacts access to kidney transplantation. J Am Soc Nephrol. 2008;19:349-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 209] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 2. | Laster M, Shen JI, Norris KC. Kidney Disease Among African Americans: A Population Perspective. Am J Kidney Dis. 2018;72:S3-S7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 90] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 3. | Hounkpatin HO, Fraser SDS, Honney R, Dreyer G, Brettle A, Roderick PJ. Ethnic minority disparities in progression and mortality of pre-dialysis chronic kidney disease: a systematic scoping review. BMC Nephrol. 2020;21:217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 68] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 4. | Harding K, Mersha TB, Pham PT, Waterman AD, Webb FA, Vassalotti JA, Nicholas SB. Health Disparities in Kidney Transplantation for African Americans. Am J Nephrol. 2017;46:165-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 5. | Kälble T, Lucan M, Nicita G, Sells R, Burgos Revilla FJ, Wiesel M; European Association of Urology. EAU guidelines on renal transplantation. Eur Urol. 2005;47:156-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 122] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 6. | Lentine KL, Delos Santos R, Axelrod D, Schnitzler MA, Brennan DC, Tuttle-Newhall JE. Obesity and kidney transplant candidates: how big is too big for transplantation? Am J Nephrol. 2012;36:575-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 110] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 7. | National Institute of Diabetes and Digestive and Kidney Diseases. Overweight and obesity statistics. [cited 28 August 2025]. Available from: https://www.niddk.nih.gov/health-information/health-statistics/overweight-obesity. |
| 8. | Government of the United Kingdom. Overweight adults. [cited 28 August 2025]. Available from: https://www.ethnicity-facts-figures.service.gov.uk/health/diet-and-exercise/overweight-adults/latest. |
| 9. | Malek SK, Keys BJ, Kumar S, Milford E, Tullius SG. Racial and ethnic disparities in kidney transplantation. Transpl Int. 2011;24:419-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 10. | Maggard MA, Shugarman LR, Suttorp M, Maglione M, Sugerman HJ, Livingston EH, Nguyen NT, Li Z, Mojica WA, Hilton L, Rhodes S, Morton SC, Shekelle PG. Meta-analysis: surgical treatment of obesity. Ann Intern Med. 2005;142:547-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1135] [Cited by in RCA: 928] [Article Influence: 44.2] [Reference Citation Analysis (0)] |
| 11. | Young MT, Gebhart A, Phelan MJ, Nguyen NT. Use and Outcomes of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Gastric Bypass: Analysis of the American College of Surgeons NSQIP. J Am Coll Surg. 2015;220:880-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 131] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 12. | Vivante A, Golan E, Tzur D, Leiba A, Tirosh A, Skorecki K, Calderon-Margalit R. Body mass index in 1.2 million adolescents and risk for end-stage renal disease. Arch Intern Med. 2012;172:1644-1650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 271] [Cited by in RCA: 232] [Article Influence: 16.6] [Reference Citation Analysis (5)] |
| 13. | Sarathy H, Henriquez G, Abramowitz MK, Kramer H, Rosas SE, Johns T, Kumar J, Skversky A, Kaskel F, Melamed ML. Abdominal Obesity, Race and Chronic Kidney Disease in Young Adults: Results from NHANES 1999-2010. PLoS One. 2016;11:e0153588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (4)] |
| 14. | Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA. 2014;311:806-814. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6575] [Cited by in RCA: 5833] [Article Influence: 486.1] [Reference Citation Analysis (4)] |
| 15. | El-Sayed AM, Scarborough P, Galea S. Ethnic inequalities in obesity among children and adults in the UK: a systematic review of the literature. Obes Rev. 2011;12:e516-e534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 16. | Grubbs V, Lin F, Vittinghoff E, Shlipak MG, Peralta CA, Bansal N, Jacobs DR, Siscovick DS, Lewis CE, Bibbins-Domingo K. Body mass index and early kidney function decline in young adults: a longitudinal analysis of the CARDIA (Coronary Artery Risk Development in Young Adults) study. Am J Kidney Dis. 2014;63:590-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 17. | Ethnicity disparities in patients with kidney failure in England and Wales. [cited 28 August 2025]. Available from: https://www.ukkidney.org/sites/renal.org/files/Ethnicity%20Disparities%202023%20Final.pdf. |
| 18. | Kaze AD, Ilori T, Jaar BG, Echouffo-Tcheugui JB. Burden of chronic kidney disease on the African continent: a systematic review and meta-analysis. BMC Nephrol. 2018;19:125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 132] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 19. | Adeboye B, Bermano G, Rolland C. Obesity and its health impact in Africa: a systematic review. Cardiovasc J Afr. 2012;23:512-521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 20. | United States Department of Health and Human Office of Minority Health. Obesity and Black/African Americans. [cited 28 August 2025]. Available from: https://minorityhealth.hhs.gov/obesity-and-blackafrican-americans. |
| 21. | NCD Risk Factor Collaboration (NCD-RisC) – Africa Working Group. Trends in obesity and diabetes across Africa from 1980 to 2014: an analysis of pooled population-based studies. Int J Epidemiol. 2017;46:1421-1432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 181] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 22. | Njelekela MA, Mpembeni R, Muhihi A, Mligiliche NL, Spiegelman D, Hertzmark E, Liu E, Finkelstein JL, Fawzi WW, Willett WC, Mtabaji J. Gender-related differences in the prevalence of cardiovascular disease risk factors and their correlates in urban Tanzania. BMC Cardiovasc Disord. 2009;9:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 104] [Article Influence: 6.1] [Reference Citation Analysis (4)] |
| 23. | Akpa OM, Made F, Ojo A, Ovbiagele B, Adu D, Motala AA, Mayosi BM, Adebamowo SN, Engel ME, Tayo B, Rotimi C, Salako B, Akinyemi R, Gebregziabher M, Sarfo F, Wahab K, Agongo G, Alberts M, Ali SA, Asiki G, Boua RP, Gómez-Olivé FX, Mashinya F, Micklesfield L, Mohamed SF, Nonterah EA, Norris SA, Sorgho H, Tollman S, Parekh RS, Chishala C, Ekoru K, Waddy SP, Peprah E, Mensah GA, Wiley K, Troyer J, Ramsay M, Owolabi MO; as members of the CVD Working Group of the H3Africa Consortium. Regional Patterns and Association Between Obesity and Hypertension in Africa: Evidence From the H3Africa CHAIR Study. Hypertension. 2020;75:1167-1178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 24. | Agyemang C, Meeks K, Beune E, Owusu-Dabo E, Mockenhaupt FP, Addo J, de Graft Aikins A, Bahendeka S, Danquah I, Schulze MB, Spranger J, Burr T, Agyei-Baffour P, Amoah SK, Galbete C, Henneman P, Klipstein-Grobusch K, Nicolaou M, Adeyemo A, van Straalen J, Smeeth L, Stronks K. Obesity and type 2 diabetes in sub-Saharan Africans - Is the burden in today's Africa similar to African migrants in Europe? The RODAM study. BMC Med. 2016;14:166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 145] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 25. | The United Kingdom Kidney Association. UK Renal Registry 23rd Annual Report. [cited 28 August 2025]. Available from: https://www.ukkidney.org/sites/renal.org/files/23rd_UKRR_ANNUAL_REPORT.pdf. |
| 26. | NIDDK USRDS. 2024 Annual Data Report. [cited 28 August 2025]. Available from: https://usrds-adr.niddk.nih.gov/2024. |
| 27. | Choi AI, Rodriguez RA, Bacchetti P, Bertenthal D, Hernandez GT, O'Hare AM. White/black racial differences in risk of end-stage renal disease and death. Am J Med. 2009;122:672-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 106] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 28. | Yu JW, Gupta G, Kang L, Bandyopadhyay D, Siddiqui MS, Bhati CS, Stravitz RT, Levy M, Reichman TW. Obesity does not significantly impact outcomes following simultaneous liver kidney transplantation: review of the UNOS database - a retrospective study. Transpl Int. 2019;32:206-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 29. | Krishnan N, Higgins R, Short A, Zehnder D, Pitcher D, Hudson A, Raymond NT. Kidney Transplantation Significantly Improves Patient and Graft Survival Irrespective of BMI: A Cohort Study. Am J Transplant. 2015;15:2378-2386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 82] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 30. | Lafranca JA, IJermans JN, Betjes MG, Dor FJ. Body mass index and outcome in renal transplant recipients: a systematic review and meta-analysis. BMC Med. 2015;13:111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 154] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 31. | Gill JS, Lan J, Dong J, Rose C, Hendren E, Johnston O, Gill J. The survival benefit of kidney transplantation in obese patients. Am J Transplant. 2013;13:2083-2090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 164] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 32. | Wong G, Howard K, Chapman JR, Chadban S, Cross N, Tong A, Webster AC, Craig JC. Comparative survival and economic benefits of deceased donor kidney transplantation and dialysis in people with varying ages and co-morbidities. PLoS One. 2012;7:e29591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 153] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 33. | Purnell TS, Luo X, Cooper LA, Massie AB, Kucirka LM, Henderson ML, Gordon EJ, Crews DC, Boulware LE, Segev DL. Association of Race and Ethnicity With Live Donor Kidney Transplantation in the United States From 1995 to 2014. JAMA. 2018;319:49-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 256] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 34. | Buford J, Harding JL, Di M, Laster ML, Drewry K, Pastan SO, Patzer RE. Divergent Trends by Patient Age in Racial Disparities in Kidney Transplant Access. Kidney Int Rep. 2025;10:2766-2777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 35. | Qannus AA, Tanriover B. Beyond the Waitlist: Addressing Structural Racism and Age-Specific Barriers to Equity in Kidney Transplantation. Kidney Int Rep. 2025;10:2542-2544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 36. | Wachterman MW, McCarthy EP, Marcantonio ER, Ersek M. Mistrust, misperceptions, and miscommunication: a qualitative study of preferences about kidney transplantation among African Americans. Transplant Proc. 2015;47:240-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 37. | Feehally J. Ethnicity and renal replacement therapy. Blood Purif. 2010;29:125-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 38. | Yeates KE, Schaubel DE, Cass A, Sequist TD, Ayanian JZ. Access to renal transplantation for minority patients with ESRD in Canada. Am J Kidney Dis. 2004;44:1083-1089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 63] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 39. | Axelrod DA, Dzebisashvili N, Schnitzler MA, Salvalaggio PR, Segev DL, Gentry SE, Tuttle-Newhall J, Lentine KL. The interplay of socioeconomic status, distance to center, and interdonor service area travel on kidney transplant access and outcomes. Clin J Am Soc Nephrol. 2010;5:2276-2288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 200] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 40. | Caskey FJ. Renal replacement therapy: can we separate the effects of social deprivation and ethnicity? Kidney Int Suppl (2011). 2013;3:246-249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 41. | Wu DA, Robb ML, Watson CJE, Forsythe JLR, Tomson CRV, Cairns J, Roderick P, Johnson RJ, Ravanan R, Fogarty D, Bradley C, Gibbons A, Metcalfe W, Draper H, Bradley AJ, Oniscu GC. Barriers to living donor kidney transplantation in the United Kingdom: a national observational study. Nephrol Dial Transplant. 2017;32:890-900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 42. | Gift of Living Donation. Pilot Project Report 2024: Improving Equity of Access to Living Donation Kidney Transplantation for Black Kidney Patients. [cited 11 August 2025]. Available from: https://www.giftoflivingdonation.org.uk/wp-content/uploads/2025/07/GOLD-pilot-report.pdf. |
| 43. | Davis SO, Zubair A, Igbokwe M, Abu M, Chiedozie CA, Sanni Q, Jesuyajolu D. A Scoping Review of Kidney Transplantation in Africa: How Far have We Come? World J Surg. 2023;47:2113-2123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 44. | Oluyombo R, Fawale BM, Busari OA, Ogunmola JO, Olanrewaju TO, Akinleye CA, Ojewola RW, Yusuf M, Obajolowo O, Soje M, Gbadegesin B. Organ Donation Among Tiers of Health Workers: Expanding Resources to Optimize Organ Availability in a Developing Country. Transplant Direct. 2016;2:e52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 45. | Tahir S, Gillott H, Jackson-Spence F, Nath J, Mytton J, Evison F, Sharif A. Do outcomes after kidney transplantation differ for black patients in England versus New York State? A comparative, population-cohort analysis. BMJ Open. 2017;7:e014069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 46. | Pallet N, Thervet E, Alberti C, Emal-Aglaé V, Bedrossian J, Martinez F, Roy C, Legendre C. Kidney transplant in black recipients: are African Europeans different from African Americans? Am J Transplant. 2005;5:2682-2687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 47. | Yeates K, Wiebe N, Gill J, Sima C, Schaubel D, Holland D, Hemmelgarn B, Tonelli M. Similar outcomes among black and white renal allograft recipients. J Am Soc Nephrol. 2009;20:172-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 48. | Kwan JM, Hajjiri Z, Metwally A, Finn PW, Perkins DL. Effect of the Obesity Epidemic on Kidney Transplantation: Obesity Is Independent of Diabetes as a Risk Factor for Adverse Renal Transplant Outcomes. PLoS One. 2016;11:e0165712. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 49. | Chakkera HA, O'Hare AM, Johansen KL, Hynes D, Stroupe K, Colin PM, Chertow GM. Influence of race on kidney transplant outcomes within and outside the Department of Veterans Affairs. J Am Soc Nephrol. 2005;16:269-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 50. | Nicoletto BB, Fonseca NK, Manfro RC, Gonçalves LF, Leitão CB, Souza GC. Effects of obesity on kidney transplantation outcomes: a systematic review and meta-analysis. Transplantation. 2014;98:167-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 145] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 51. | Moreso F, Hernández D. Has the survival of the graft improved after renal transplantation in the era of modern immunosuppression? Nefrologia. 2013;33:14-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 52. | Fiscella K, Franks P, Meldrum S, Barnett S. Racial disparity in surgical complications in New York State. Ann Surg. 2005;242:151-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 53. | Gore JL, Pham PT, Danovitch GM, Wilkinson AH, Rosenthal JT, Lipshutz GS, Singer JS. Obesity and outcome following renal transplantation. Am J Transplant. 2006;6:357-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 258] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 54. | Vranic GM, Ma JZ, Keith DS. The role of minority geographic distribution in waiting time for deceased donor kidney transplantation. Am J Transplant. 2014;14:2526-2534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 55. | Wagner DR, Heyward VH. Measures of body composition in blacks and whites: a comparative review. Am J Clin Nutr. 2000;71:1392-1402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 253] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 56. | Surveillance report 2018 - Obesity: identification, assessment and management (2014) NICE guideline CG189 and BMI: preventing ill health and premature death in black, Asian and other minority ethnic groups (2013) NICE guideline PH46 [Internet]. London: National Institute for Health and Care Excellence (NICE); 2018-May-24 . [PubMed] |
| 57. | Kovesdy CP, Czira ME, Rudas A, Ujszaszi A, Rosivall L, Novak M, Kalantar-Zadeh K, Molnar MZ, Mucsi I. Body mass index, waist circumference and mortality in kidney transplant recipients. Am J Transplant. 2010;10:2644-2651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 58. | Taber DJ, Egede LE, Baliga PK. Outcome disparities between African Americans and Caucasians in contemporary kidney transplant recipients. Am J Surg. 2017;213:666-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 59. | Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J, Watkins PB, Daly A, Wrighton SA, Hall SD, Maurel P, Relling M, Brimer C, Yasuda K, Venkataramanan R, Strom S, Thummel K, Boguski MS, Schuetz E. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet. 2001;27:383-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1583] [Cited by in RCA: 1569] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 60. | Taber DJ, Gebregziabher MG, Srinivas TR, Chavin KD, Baliga PK, Egede LE. African-American race modifies the influence of tacrolimus concentrations on acute rejection and toxicity in kidney transplant recipients. Pharmacotherapy. 2015;35:569-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 61. | Zheng HX, Zeevi A, McCurry K, Schuetz E, Webber S, Ristich J, Zhang J, Iacono A, Dauber J, McDade K, Zaldonis D, Lamba J, Burckart GJ. The impact of pharmacogenomic factors on acute persistent rejection in adult lung transplant patients. Transpl Immunol. 2005;14:37-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 62. | Taber DJ, McGillicuddy JW, Bratton CF, Rohan VS, Nadig S, Dubay D, Baliga PK. Cytolytic Induction Therapy Improves Clinical Outcomes in African-American Kidney Transplant Recipients. Ann Surg. 2017;266:450-456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 63. | Patzer RE, Serper M, Reese PP, Przytula K, Koval R, Ladner DP, Levitsky JM, Abecassis MM, Wolf MS. Medication understanding, non-adherence, and clinical outcomes among adult kidney transplant recipients. Clin Transplant. 2016;30:1294-1305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 64. | Molinero LL, Yin D, Lei YM, Chen L, Wang Y, Chong AS, Alegre ML. High-Fat Diet-Induced Obesity Enhances Allograft Rejection. Transplantation. 2016;100:1015-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 65. | Ekrikpo UE, Mnika K, Effa EE, Ajayi SO, Okwuonu C, Waziri B, Bello A, Dandara C, Kengne AP, Wonkam A, Okpechi I. Association of Genetic Polymorphisms of TGF-β1, HMOX1, and APOL1 With CKD in Nigerian Patients With and Without HIV. Am J Kidney Dis. 2020;76:100-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 66. | Freedman BI, Locke JE, Reeves-Daniel AM, Julian BA. Apolipoprotein L1 Gene Effects on Kidney Transplantation. Semin Nephrol. 2017;37:530-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 67. | Kao WH, Klag MJ, Meoni LA, Reich D, Berthier-Schaad Y, Li M, Coresh J, Patterson N, Tandon A, Powe NR, Fink NE, Sadler JH, Weir MR, Abboud HE, Adler SG, Divers J, Iyengar SK, Freedman BI, Kimmel PL, Knowler WC, Kohn OF, Kramp K, Leehey DJ, Nicholas SB, Pahl MV, Schelling JR, Sedor JR, Thornley-Brown D, Winkler CA, Smith MW, Parekh RS; Family Investigation of Nephropathy and Diabetes Research Group. MYH9 is associated with nondiabetic end-stage renal disease in African Americans. Nat Genet. 2008;40:1185-1192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 534] [Cited by in RCA: 497] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 68. | Parsa A, Kao WH, Xie D, Astor BC, Li M, Hsu CY, Feldman HI, Parekh RS, Kusek JW, Greene TH, Fink JC, Anderson AH, Choi MJ, Wright JT Jr, Lash JP, Freedman BI, Ojo A, Winkler CA, Raj DS, Kopp JB, He J, Jensvold NG, Tao K, Lipkowitz MS, Appel LJ; AASK Study Investigators; CRIC Study Investigators. APOL1 risk variants, race, and progression of chronic kidney disease. N Engl J Med. 2013;369:2183-2196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 709] [Cited by in RCA: 637] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 69. | Kopp JB, Nelson GW, Sampath K, Johnson RC, Genovese G, An P, Friedman D, Briggs W, Dart R, Korbet S, Mokrzycki MH, Kimmel PL, Limou S, Ahuja TS, Berns JS, Fryc J, Simon EE, Smith MC, Trachtman H, Michel DM, Schelling JR, Vlahov D, Pollak M, Winkler CA. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol. 2011;22:2129-2137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 682] [Cited by in RCA: 680] [Article Influence: 45.3] [Reference Citation Analysis (1)] |
| 70. | Hughson MD, Puelles VG, Hoy WE, Douglas-Denton RN, Mott SA, Bertram JF. Hypertension, glomerular hypertrophy and nephrosclerosis: the effect of race. Nephrol Dial Transplant. 2014;29:1399-1409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 71. | Grams ME, Rebholz CM, Chen Y, Rawlings AM, Estrella MM, Selvin E, Appel LJ, Tin A, Coresh J. Race, APOL1 Risk, and eGFR Decline in the General Population. J Am Soc Nephrol. 2016;27:2842-2850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 126] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 72. | Appel LJ, Wright JT Jr, Greene T, Kusek JW, Lewis JB, Wang X, Lipkowitz MS, Norris KC, Bakris GL, Rahman M, Contreras G, Rostand SG, Kopple JD, Gabbai FB, Schulman GI, Gassman JJ, Charleston J, Agodoa LY; African American Study of Kidney Disease and Hypertension Collaborative Research Group. Long-term effects of renin-angiotensin system-blocking therapy and a low blood pressure goal on progression of hypertensive chronic kidney disease in African Americans. Arch Intern Med. 2008;168:832-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 123] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 73. | Karabicak I, Aytug S, Lewis S, Shah S, Sumrani N, Hayat A, Distant DA, Salifu MO. Long-term kidney transplant outcome in obese patients in a predominantly African American population. Clin Transplant. 2011;25:E264-E270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 74. | Davidson J, Wilkinson A, Dantal J, Dotta F, Haller H, Hernández D, Kasiske BL, Kiberd B, Krentz A, Legendre C, Marchetti P, Markell M, van der Woude FJ, Wheeler DC; International Expert Panel. New-onset diabetes after transplantation: 2003 International consensus guidelines. Proceedings of an international expert panel meeting. Barcelona, Spain, 19 February 2003. Transplantation. 2003;75:SS3-S24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 345] [Article Influence: 15.0] [Reference Citation Analysis (6)] |
| 75. | Palepu S, Prasad GV. New-onset diabetes mellitus after kidney transplantation: Current status and future directions. World J Diabetes. 2015;6:445-455. [PubMed] [DOI] [Full Text] |
| 76. | Kasiske BL, Snyder JJ, Gilbertson D, Matas AJ. Diabetes mellitus after kidney transplantation in the United States. Am J Transplant. 2003;3:178-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1046] [Cited by in RCA: 929] [Article Influence: 40.4] [Reference Citation Analysis (3)] |
| 77. | Sulanc E, Lane JT, Puumala SE, Groggel GC, Wrenshall LE, Stevens RB. New-onset diabetes after kidney transplantation: an application of 2003 International Guidelines. Transplantation. 2005;80:945-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 3.7] [Reference Citation Analysis (3)] |
| 78. | Huang E, Bunnapradist S. Pre-Transplant Weight Loss and Survival after Kidney Transplantation. Am J Nephrol. 2015;41:448-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 79. | Pham PT, Pham PM, Pham SV, Pham PA, Pham PC. New onset diabetes after transplantation (NODAT): an overview. Diabetes Metab Syndr Obes. 2011;4:175-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 80. | Fox CH, Mahoney MC, Ramsoomair D, Carter CA. Magnesium deficiency in African-Americans: does it contribute to increased cardiovascular risk factors? J Natl Med Assoc. 2003;95:257-262. [PubMed] |
| 81. | Bellini MI, Paoletti F, Herbert PE. Obesity and bariatric intervention in patients with chronic renal disease. J Int Med Res. 2019;47:2326-2341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 82. | Thomas EC, Randall DW, Khurram MA, Yaqoob MM. Ethnic differences in early blood tacrolimus concentrations after kidney transplantation: a retrospective observational study. BMC Nephrol. 2025;26:436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 83. | Ray S, Idiong CI, Anderson R, Killion K, Antrum C, Puglisi M, Kalinowski J, Cooksey Stowers K, Blackman Carr LT. Examining preferences for weight loss interventions among black men and women with overweight or obesity: A qualitative study. BMC Public Health. 2025;25:2799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 84. | Ku E, Whelan AM, McCulloch CE, Lee B, Niemann CU, Roll GR, Grimes BA, Johansen KL. Weighing the waitlist: Weight changes and access to kidney transplantation among obese candidates. PLoS One. 2020;15:e0242784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 85. | August KJ, Sorkin DH. Racial/ethnic disparities in exercise and dietary behaviors of middle-aged and older adults. J Gen Intern Med. 2011;26:245-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 151] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 86. | Tussing-Humphreys LM, Fitzgibbon ML, Kong A, Odoms-Young A. Weight loss maintenance in African American women: a systematic review of the behavioral lifestyle intervention literature. J Obes. 2013;2013:437369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 87. | MacLaughlin HL, Cook SA, Kariyawasam D, Roseke M, van Niekerk M, Macdougall IC. Nonrandomized trial of weight loss with orlistat, nutrition education, diet, and exercise in obese patients with CKD: 2-year follow-up. Am J Kidney Dis. 2010;55:69-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 88. | Koskinas KC, Van Craenenbroeck EM, Prescott EB. European Society of Cardiology clinical consensus statement on obesity and cardiovascular disease: 10 key points. Eur Heart J. 2025;46:3611-3612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 89. | Orandi BJ, Chen Y, Li Y, Charytan D, Lentine KL, Lee BP, Ali N, DeMarco MP, Weintraub MA, Bae S, Lonze BE, Ren-Fielding CJ, Lofton H, Gujral A, Segev DL, McAdams-DeMarco M. GLP-1 Receptor Agonist Outcomes, Safety, and Body Mass Index Change in a National Cohort of Patients on Dialysis. Clin J Am Soc Nephrol. 2025;20:1100-1110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 90. | Orandi BJ, Chen Y, Li Y, Metoyer GT, Lentine KL, Weintraub M, Bae S, Ali NM, Lonze BE, Ren-Fielding CJ, Lofton H, Gujral A, Segev DL, McAdams-DeMarco M. GLP-1 receptor agonists in kidney transplant recipients with pre-existing diabetes: a retrospective cohort study. Lancet Diabetes Endocrinol. 2025;13:374-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 31] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 91. | Investors. Lilly's oral GLP-1, orforglipron, delivers weight loss of up to an average of 27.3 lbs in first of two pivotal Phase 3 trials in adults with obesity. [cited 25 August 2025]. Available from: https://investor.lilly.com/news-releases/news-release-details/lillys-oral-glp-1-orforglipron-delivers-weight-loss-average-273. |
| 92. | Knop FK, Aroda VR, do Vale RD, Holst-Hansen T, Laursen PN, Rosenstock J, Rubino DM, Garvey WT; OASIS 1 Investigators. Oral semaglutide 50 mg taken once per day in adults with overweight or obesity (OASIS 1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2023;402:705-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 261] [Article Influence: 87.0] [Reference Citation Analysis (0)] |
| 93. | Orandi B, Chen Y, Li Y, Charyton D, Lentine K, Ali N, Bae S, Lonze B, Ren-fielding C, Weintraub M, Segev D, McAdams-DeMarco M. GLP-1 Receptor Agonists: Advancing Kidney Transplant Access and Safety in a National ESKD Cohort. Am J Transplant. 2025;25:S136. [DOI] [Full Text] |
| 94. | Chen Y, Bae S, Li Y, Segev D, Mcadams-Demarco M, Orandi B. Bariatric Surgery vs. GLP-1 Receptor Agonists: Mortality Risk and Access to Kidney Transplantation. Am J Transplant. 2025;25:S417. |
| 95. | Heymsfield SB, Wadden TA. Mechanisms, Pathophysiology, and Management of Obesity. N Engl J Med. 2017;376:254-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1506] [Cited by in RCA: 1271] [Article Influence: 141.2] [Reference Citation Analysis (0)] |
| 96. | Byham-Gray L, Stover J, Wiesen K. A Clinical Guide to Nutrition Care in Kidney Disease. Academy of Nutrition and Dietetics. Chicago: Academy of Nutrition and Dietetics, 2013. |
| 97. | Chan G, Soucisse M. Survey of Canadian Kidney Transplant Specialists on the Management of Morbid Obesity and the Transplant Waiting List. Can J Kidney Health Dis. 2016;3:2054358116675344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 98. | Lambert K, Beer J, Dumont R, Hewitt K, Manley K, Meade A, Salamon K, Campbell K. Weight management strategies for those with chronic kidney disease: A consensus report from the Asia Pacific Society of Nephrology and Australia and New Zealand Society of Nephrology 2016 renal dietitians meeting. Nephrology (Carlton). 2018;23:912-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 99. | Puttarajappa CM, Urbanski M, Fallahzadeh MK, Budhiraja P, Balaraman V, Butsch WS, Morford H, Anand PM, Woodside KJ, Orandi BJ; co-authors. Practices in the management of kidney transplant candidates and recipients with obesity: A survey of United States transplant programs. Am J Transplant. 2025;25:2194-2205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 100. | Yemini R, Nesher E, Winkler J, Carmeli I, Azran C, Ben David M, Mor E, Keidar A. Bariatric surgery in solid organ transplant patients: Long-term follow-up results of outcome, safety, and effect on immunosuppression. Am J Transplant. 2018;18:2772-2780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 101. | Rogers CC, Alloway RR, Alexander JW, Cardi M, Trofe J, Vinks AA. Pharmacokinetics of mycophenolic acid, tacrolimus and sirolimus after gastric bypass surgery in end-stage renal disease and transplant patients: a pilot study. Clin Transplant. 2008;22:281-291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 159] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 102. | Szomstein S, Rojas R, Rosenthal RJ. Outcomes of laparoscopic bariatric surgery after renal transplant. Obes Surg. 2010;20:383-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 103. | Kienzl-Wagner K, Weissenbacher A, Gehwolf P, Wykypiel H, Öfner D, Schneeberger S. Laparoscopic sleeve gastrectomy: gateway to kidney transplantation. Surg Obes Relat Dis. 2017;13:909-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 104. | Oberholzer J, Giulianotti P, Danielson KK, Spaggiari M, Bejarano-Pineda L, Bianco F, Tzvetanov I, Ayloo S, Jeon H, Garcia-Roca R, Thielke J, Tang I, Akkina S, Becker B, Kinzer K, Patel A, Benedetti E. Minimally invasive robotic kidney transplantation for obese patients previously denied access to transplantation. Am J Transplant. 2013;13:721-728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 135] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 105. |
Guggino J, Coumes S, Wion N, Reche F, Arvieux C, Borel AL Effectiveness and Safety of Bariatric Surgery in Patients with End-Stage Chronic Kidney Disease or Kidney Transplant.
|
| 106. | Thomas IA, Gaynor JJ, Joseph T, De La Cruz-Munoz N, Sageshima J, Kupin W, Chen LJ, Ciancio G, Burke GW 3rd, Mattiazzi AD, Roth D, Guerra G. Roux-en-Y gastric bypass is an effective bridge to kidney transplantation: Results from a single center. Clin Transplant. 2018;32:e13232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 107. | Kim Y, Bailey AJ, Morris MC, Kassam AF, Shah SA, Diwan TS. Kidney transplantation after sleeve gastrectomy in the morbidly obese candidate: results of a 2-year experience. Surg Obes Relat Dis. 2020;16:10-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 108. | Lee Y, Raveendran L, Lovrics O, Tian C, Khondker A, Koyle MA, Farcas M, Doumouras AG, Hong D. The role of bariatric surgery on kidney transplantation: A systematic review and meta-analysis. Can Urol Assoc J. 2021;15:E553-E562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 109. | Modanlou KA, Muthyala U, Xiao H, Schnitzler MA, Salvalaggio PR, Brennan DC, Abbott KC, Graff RJ, Lentine KL. Bariatric surgery among kidney transplant candidates and recipients: analysis of the United States renal data system and literature review. Transplantation. 2009;87:1167-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 126] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 110. | Sheetz KH, Gerhardinger L, Dimick JB, Waits SA. Bariatric Surgery and Long-term Survival in Patients With Obesity and End-stage Kidney Disease. JAMA Surg. 2020;155:581-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 111. | Spaggiari M, Di Cocco P, Tulla K, Kaylan KB, Masrur MA, Hassan C, Alvarez JA, Benedetti E, Tzvetanov I. Simultaneous robotic kidney transplantation and bariatric surgery for morbidly obese patients with end-stage renal failure. Am J Transplant. 2021;21:1525-1534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 112. | Mocanu V, Dang JT, Switzer N, Madsen K, Birch DW, Karmali S. Sex and Race Predict Adverse Outcomes Following Bariatric Surgery: an MBSAQIP Analysis. Obes Surg. 2020;30:1093-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 113. | Takata MC, Campos GM, Ciovica R, Rabl C, Rogers SJ, Cello JP, Ascher NL, Posselt AM. Laparoscopic bariatric surgery improves candidacy in morbidly obese patients awaiting transplantation. Surg Obes Relat Dis. 2008;4:159-64; discussion 164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 157] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 114. | Choudhury RA, Hoeltzel G, Prins K, Chow E, Moore HB, Lawson PJ, Yoeli D, Pratap A, Abt PL, Dumon KR, Conzen KD, Nydam TL. Sleeve Gastrectomy Compared with Gastric Bypass for Morbidly Obese Patients with End Stage Renal Disease: a Decision Analysis. J Gastrointest Surg. 2020;24:756-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 115. | Cohen JB, Lim MA, Tewksbury CM, Torres-Landa S, Trofe-Clark J, Abt PL, Williams NN, Dumon KR, Goral S. Bariatric surgery before and after kidney transplantation: long-term weight loss and allograft outcomes. Surg Obes Relat Dis. 2019;15:935-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (3)] |
| 116. | Weingarten TN, Gurrieri C, McCaffrey JM, Ricter SJ, Hilgeman ML, Schroeder DR, Kendrick ML, Greene EL, Sprung J. Acute kidney injury following bariatric surgery. Obes Surg. 2013;23:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 66] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 117. | McBride CL, Petersen A, Sudan D, Thompson J. Short bowel syndrome following bariatric surgical procedures. Am J Surg. 2006;192:828-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 118. | Detwiler RK. Con: Weight loss prior to transplant: no. Nephrol Dial Transplant. 2015;30:1805-1809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 119. | Dumronggittigule W, Marcus EA, Cheng EY, Dubray BJ, Venick RS, Busuttil RW, Farmer DG. Intestinal Failure Following Bariatric Surgery. Transplantation. 2017;101:S33. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 120. | Lumlertgul N, Siribamrungwong M, Jaber BL, Susantitaphong P. Secondary Oxalate Nephropathy: A Systematic Review. Kidney Int Rep. 2018;3:1363-1372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 121. | Camilleri B, Bridson JM, Sharma A, Halawa A. From chronic kidney disease to kidney transplantation: The impact of obesity and its treatment modalities. Transplant Rev (Orlando). 2016;30:203-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 122. | Oniscu GC, Abramowicz D, Bolignano D, Gandolfini I, Hellemans R, Maggiore U, Nistor I, O'Neill S, Sever MS, Koobasi M, Nagler EV. Management of obesity in kidney transplant candidates and recipients: A clinical practice guideline by the DESCARTES Working Group of ERA. Nephrol Dial Transplant. 2021;37:i1-i15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |